Open Access Article
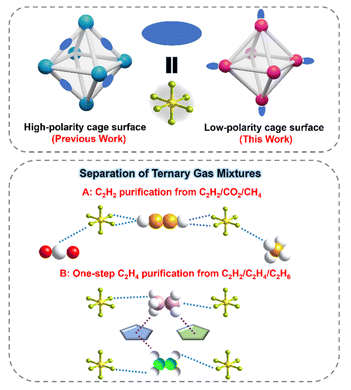
Open Access ArticleLinker desymmetrisation directs low-polar cages in an anion-pillared MOF for acetylene and ethylene purification from ternary mixtures
Jun-Jie Wua,
Peng-Dan Zhang*ac,
Xue-Qian Wu*a,
Shuai-Hao Huanga,
Wen-Wen Donga,
Ya-Pan Wu a and
Dong-Sheng Li
a and
Dong-Sheng Li *ab
*ab
aCollege of Materials and Chemical Engineering, China Three Gorges University, Yichang 443002, P. R. China. E-mail: dana929@163.com; wuxueqiansnail@163.com; lidongsheng1@126.com
bHubei Three Gorges Laboratory, Yichang 443007, Hubei, P. R. China
cCollege of Hydraulic & Environmental Engineering, China Three Gorges University, Yichang 443002, P. R. China
First published on 10th April 2026
Abstract
Anion-pillared metal–organic frameworks (APMOFs) constitute a promising class of porous adsorbents, yet precise organization of anionic groups to maximize their strong electronegative character while circumventing highly polar pore environments and high adsorption enthalpies remains a substantial challenge. Here, we report a novel APMOF, CTGU-45, featuring an atypical low-polar cage surface distinct from reported APMOFs and then explore its C2H2 and C2H4 purification from ternary C2H2/CO2/CH4 and C2H2/C2H4/C2H6 mixtures. Theoretical calculation results revealed the mechanisms of simultaneous recognition of C2H2 and C2H6 molecules from C2 hydrocarbons: the SiF62− pillars provide strong binding sites for C2H2 via C–H⋯F hydrogen bonds, while the TIB-derived low-polar surface offers an optimal environment for C2H6 adsorption through synergistic C–H⋯π and C–H⋯N interactions. Dynamic breakthrough experiments further confirm the efficient separation of C2H2/CO2, C2H2/CO2/CH4, and notably one-step production of polymer-grade C2H4 (≥99.95%, 9.57 L kg−1) from ternary C2H2/C2H4/C2H6 mixtures with low regeneration energy. Crucially, CTGU-45 can be synthesized on a 500 mL scale with retained performance, underscoring its practical feasibility and laying a solid foundation for future industrial applications.
Introduction
Acetylene (C2H2) and ethylene (C2H4) are pivotal feedstocks in the chemical industry.1 C2H2 is typically generated by partial oxidation of methane (CH4) or thermal cracking of hydrocarbons, which co-produces significant amounts of carbon dioxide (CO2).2–5 Similarly, C2H4 is obtained via steam cracking or ethane (C2H6) dehydrogenation, where C2H6 and trace C2H2 will unavoidably be produced as by-products.6,7 These impurities severely compromise the purity and productivity of polymer-grade C2H4 or high-purity C2H2, necessitating highly efficient separation processes.8,9 However, the similar physicochemical properties of these gas molecules make separations (C2H2/CO2/CH4, C2H2/C2H4/C2H6) particularly difficult (Table S2). Conventional methods like catalytic hydrogenation and cryogenic distillation are complex and energy-intensive, thus driving the urgent need for efficient, alternative separation technologies.10–12Adsorptive separation using porous materials presents a promising, energy-efficient alternative to traditional cryogenic distillation.13–16 Among various adsorbents, metal–organic frameworks (MOFs) have garnered immense attention due to their designable pore structures and tunable surface functionalities. In particular, anion-pillared MOFs (APMOFs) have emerged as an important subclass of MOF adsorbents. These materials utilize inorganic anions (e.g., SiF62−, GeF62−, NbOF52−, MoO42−) as structural pillars to construct robust frameworks with strong electrostatic potential fields and abundant electronegative atoms that act as hydrogen-bond (H-bond) acceptors, enabling highly selective recognition of C2H2 with strong positive H atoms and C2H6 with more H atoms. Initially, APMOFs are constructed from linear bidentate ligands to form one-dimensional (1D) channels.17–20 The subsequent introduction of tri- and tetra-dentate ligands created more open APMOFs with a “cage + window” structure, which provides high capacity from large cages and high selectivity from narrow windows. Certainly, these APMOFs have been considered as valuable candidates capable of addressing the “trade-off” between capacity and selectivity in gas separation.
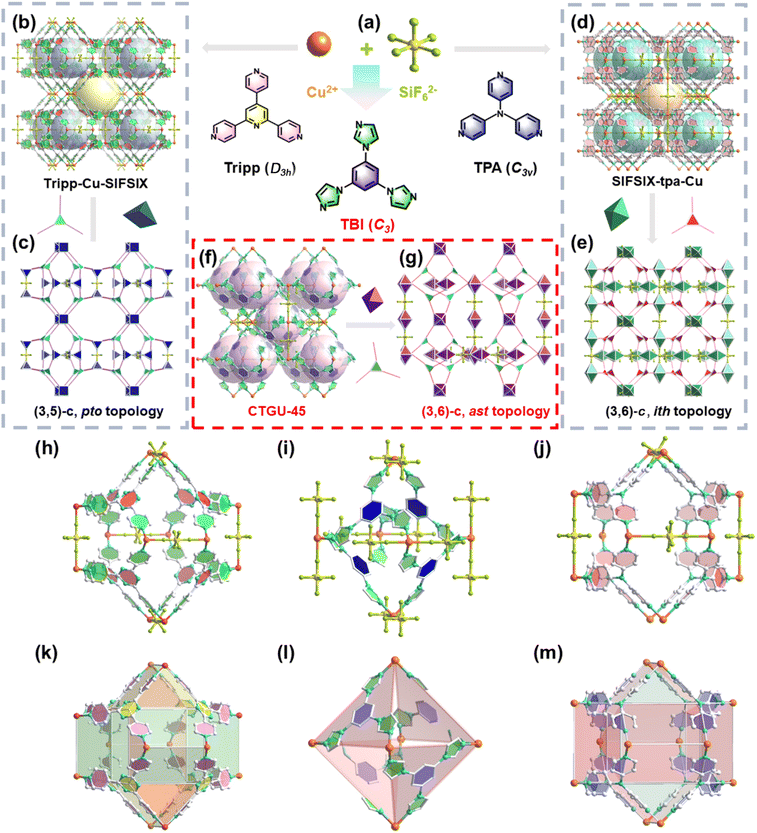
To date, only three highly symmetrical tridentate modules (Tripp, 2,4,6-tris(4-pyridyl)pyridine; TPA, tri(pyridin-4-yl)amine; and TPT, 2,4,6-tri(4-pyridyl)-1,3,5-triazine) have been successfully employed to construct APMOFs and used for gas separation, such as C2H2/CO2, CO2/N2, C3H4/C3H6, Xe/Kr, and C6 hydrocarbons. These ligands share critical structural features: their terminal coordinating units are all pyridine rings, which enforce a nearly ideal equilateral triangular arrangement of the three coordination N atoms. Although limited rotation of the pyridine rings is possible, the spatial positions of these key N donors remain essentially fixed, enabling a predictable assembly of frameworks. Interestingly, replacing the terminal pyridyl moiety with less symmetric imidazole rings would introduce significant conformational flexibility. The free rotation around the C–N single bonds linking the central benzene ring to the imidazole arms results in variable spatial orientations of the coordination N atoms, thereby breaking the inherent topological symmetry and creating opportunities for constructing APMOFs with unique pores. Moreover, certain challenging separations, e.g., one-step C2H4 purification from ternary C2 mixtures, are seldom realized in APMOFs owing to intrinsic polarity mismatches, suggesting that a reconfiguration of functional sites within APMOFs is required. Since the coordination geometry and configuration of inorganic anions are relatively well-defined, the linker desymmetrisation offers a powerful means to systematically reorganize the functional sites within APMOFs. By changing the symmetry of the organic linkers, it becomes possible to create frameworks in which the chemical environment around each coordination site can be independently tuned. This controlled asymmetry allows for the introduction of complementary binding sites, modulation of local polarity, and the rational design of preferential adsorption domains, ultimately enhancing the selectivity and efficiency of challenging gas separations.
Keep this in mind, herein, by utilizing a tridentate TIB linker (1,3,5-tri(1H-imidazol-1-yl)benzene) with C3 symmetry, a novel SiF62− anion hybrid Cu-MOF (SIFSIX-Cu-TIB, CTGU-45, CTGU = China Three Gorges University) has been successfully constructed. Single-crystal X-ray diffraction (SCXRD) analysis reveals that the activated CTGU-45 features uniform cage-like pores with an internal cage diameter of approximately 7.6 Å and a pore window size of about 6.5 Å. In contrast to most reported cage-structured APMOFs, the main pore surface of CTGU-45 is defined solely by TIB ligands and Cu(II) nodes, resulting in a low-polar pore environment that provides optimized adsorption space for C2H6 molecules. This establishes a foundation for the one-step production of high-purity C2H4 from ternary C2 hydrocarbon mixtures (C2H2/C2H4/C2H6). Meanwhile, the marginalized F atoms of the inorganic SiF62− pillars serve as strong basic sites, engaging in pronounced interactions with the acidic H atoms of C2H2, thereby facilitating efficient C2H2/CO2, C2H2/CO2/CH4, and C2H2/C2H4 separations (Scheme 1). The outstanding separation performances, as well as the underlying adsorbate–adsorbent interactions, have been comprehensively verified by a combination of experimental static adsorption/dynamic breakthrough tests and theoretical calculations. This work not only creates CTGU-45 as an efficient adsorbent for challenging C2 hydrocarbons separation but also demonstrates the great potential of ligand engineering to break the inherent limitations of conventional APMOFs, opening up new avenues for developing advanced adsorbents.
Results and discussion
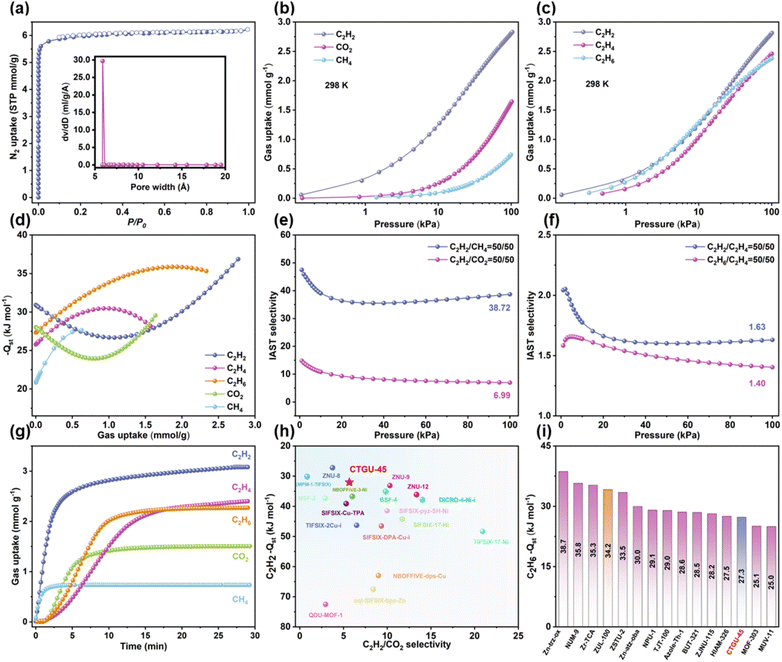
Blue octahedral single crystals of CTGU-45 were successfully synthesized by reacting Cu(OAc)2·H2O and TIB ligand in a mixed solvent of N,N-dimethylacetamide (DMA)/H2O, with HBF4 added in a sealed glass tube (Fig. S3). SCXRD analysis revealed that CTGU-45 crystallizes in a three-dimensional (3D) framework with the cubic system space group Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) n. Each Cu(II) cation is coordinated in a square-planar geometry by four N atoms from imidazole groups of four distinct tridentate TIB ligands (Fig. S2). The SiF62− anions as 2-connected nodes occupy the two axial positions to complete the octahedral coordination. Notably, the SiF62− anions originated from the in situ etching of the borosilicate glass vessel by HF (from HBF4 hydrolysis). Due to the free rotation of the C–N bonds between the central benzene and imidazole moieties, the TIB ligands in CTGU-45 exhibit substantial disorder. Upon removal of the minor disordered components, the real structure shows a uniform cage-like architecture with a pore diameter of approximately 7.6 Å (Fig. 1f). Each incomplete octahedral cage displays four identical triangular windows consisting of three TIB linkers and three Cu2+ ions. The effective aperture of these windows, after considering the van der Waals radii of H atoms, is only about 6.5 Å, allowing the entry of C1–C2 hydrocarbon molecules (Fig. S1). These cages are further linked by SiF62− anion, whose F atoms act as potential Lewis basic sites, which may work synergistically with the confined space and aromatic surface to enhance the binding of gas molecules. In contrast to most reported APMOFs, which typically consist of bidentate ligands and anion pillars forming 1D linear channels (e.g., SIFSIX-1-Cu,21 SIFSIX-3-Ni22), the structure presented here features a well-defined windowed cage architecture. Furthermore, APMOFs constructed from tridentate ligands are exceptionally rare. To our knowledge, only three tridentate ligands (Tripp, TPA, and TPT) have been reported for constructing APMOFs, such as Tripp-Cu-SIFSIX23 (Fig. 1b) and SIFSIX-Cu-TPA (Fig. 1d),24 both of which exhibit two distinct types of cages. Tripp-Cu-SIFSIX displays octahedra cages (∼7.2 Å) and icositetrahedron cages (∼8.9 Å), while SIFSIX-Cu-TPA possesses icosahedral cages (∼6.0 Å) and tetrahedral cages (∼4.7 Å). Though the pyridine rings of Tripp (D3h, excluding the non-coordinating pyridine N atom) and TPA (C3v) ligands could rotate around C(pyridine)–C(pyridine) or N–C(benzene) single bonds, their three pyridyl N donors were locked into a fixed, equilateral triangular geometry. In contrast, the rotation of C(benzene)–N(imidazole) bonds in the TIB (C3) linkers results in dynamic positions of coordination imidazole N atoms, breaking the perfect symmetry, as presented in Fig. 1a and S4. Such ligand asymmetry easily leads to a distorted coordination geometry and complicates the crystallization process, hindering the predictable assembly of conventional networks. Concretely, in the Tripp-Cu-SIFSIX, SIFSIX-Cu-TPA, and CTGU-45, all the organic linkers act as 3-connected topological nodes with D3h (Tripp/TPA-node) and C3 (TIB-node) symmetry, respectively. Meanwhile, the coordination numbers of Cu2+ are either 5 or 6, exhibiting square pyramidal (C4v) or octahedral (D4h) coordination geometries. The combination of organic nodes and metal nodes ultimately gives rise to (3,5)-c or (3,6)-c topological frameworks in three APMOFs (Fig. 1c/g/e). It is noteworthy that although Cu2+ acts as a 6-c node in both SIFSIX-Cu-TPA and CTGU-45, the four pyridine rings coordinated to the Cu2+ center in the former exhibit relative dihedral angles close to 90°, whereas the four imidazole rings coordinated to Cu2+ in the latter are severely twisted. This causes the 6-c nodes in CTGU-45 to link adjacent nodes in a more distorted fashion, breaking the D4h extension symmetry of the 6-c nodes observed in SIFSIX-Cu-TPA. This, together with the 3-c nodes with C3 symmetry, facilitates the formation of a geometrically self-consistent fundamental cage-like network in the absence of SiF62− anions (Fig. S4). The foregoing analysis has validated the critical influence of ligand symmetry on the formation of anionic hybrid frameworks. Overall, compared to Tripp-Cu-SIFSIX and SIFSIX-Cu-TPA, CTGU-45 shares the common feature of incorporating SiF62− anions, whose electronegative F atoms serve as Lewis-basic sites for interacting with acidic H atoms of C2H2. However, a fundamental distinction lies in their pore architecture: in conventional APMOFs, the SiF62− anions are integral components that participate directly in cage formation, resulting in highly polar pore surfaces dominated by F atoms (Fig. 1h/k and j/m). In contrast, in CTGU-45, the pore cages are constructed exclusively by the TIB ligands and Cu(II) nodes, while the SiF62− anions merely serve as inter-cage pillars (determined by the topology, Fig. 1i/l). This unique arrangement creates a low-polar pore surface rich in aromatic rings and N atoms, which not only provides optimal van der Waals interactions with highly polarizable C2H6 molecules but also enables synergistic binding through multiple C–H⋯π and C–H⋯N interactions. This distinctive pore environment, combined with the selective C–H⋯F binding sites at the SiF62− pillars, allows CTGU-45 to achieve cooperative adsorption of both C2H2 and C2H6, thereby providing a robust foundation for one-step C2H4 purification from ternary mixtures. The phase purity of the as-synthesized CTGU-45 was confirmed by powder X-ray diffraction (PXRD), as the experimental pattern matches well with simulated ones (Fig. S5). The thermogravimetric analysis (TGA) revealed the high thermal stability of activated CTGU-45, which remains stable up to 350 °C (Fig. S6). The initial weight loss below 100 °C is ascribed to solvent removal, while the subsequent major loss above 350 °C corresponds to the decomposition of the MOF framework. The N2 adsorption isotherm of CTGU-45 at 77 K displayed a typical Type I profile, and the corresponding HK pore size distribution suggested ultra-micropores of 6.1 Å, collectively confirming its microporous characteristic. The N2 adsorption uptake of 125.5 cm3 g−1 at 1 kPa and the calculated BET surface area of 544.8 m2 g−1 further corroborated its porous structure (Fig. 2a). Moreover, the experimental pore volume of CTGU-45 was calculated to be 0.21 cm3 g−1, which is consistent with the theoretical value from crystal structure (0.24 cm3 g−1). Combined with its excellent thermal stability, these integrated properties collectively endorse CTGU-45 as a highly promising adsorbent for gas separation applications.
n. Each Cu(II) cation is coordinated in a square-planar geometry by four N atoms from imidazole groups of four distinct tridentate TIB ligands (Fig. S2). The SiF62− anions as 2-connected nodes occupy the two axial positions to complete the octahedral coordination. Notably, the SiF62− anions originated from the in situ etching of the borosilicate glass vessel by HF (from HBF4 hydrolysis). Due to the free rotation of the C–N bonds between the central benzene and imidazole moieties, the TIB ligands in CTGU-45 exhibit substantial disorder. Upon removal of the minor disordered components, the real structure shows a uniform cage-like architecture with a pore diameter of approximately 7.6 Å (Fig. 1f). Each incomplete octahedral cage displays four identical triangular windows consisting of three TIB linkers and three Cu2+ ions. The effective aperture of these windows, after considering the van der Waals radii of H atoms, is only about 6.5 Å, allowing the entry of C1–C2 hydrocarbon molecules (Fig. S1). These cages are further linked by SiF62− anion, whose F atoms act as potential Lewis basic sites, which may work synergistically with the confined space and aromatic surface to enhance the binding of gas molecules. In contrast to most reported APMOFs, which typically consist of bidentate ligands and anion pillars forming 1D linear channels (e.g., SIFSIX-1-Cu,21 SIFSIX-3-Ni22), the structure presented here features a well-defined windowed cage architecture. Furthermore, APMOFs constructed from tridentate ligands are exceptionally rare. To our knowledge, only three tridentate ligands (Tripp, TPA, and TPT) have been reported for constructing APMOFs, such as Tripp-Cu-SIFSIX23 (Fig. 1b) and SIFSIX-Cu-TPA (Fig. 1d),24 both of which exhibit two distinct types of cages. Tripp-Cu-SIFSIX displays octahedra cages (∼7.2 Å) and icositetrahedron cages (∼8.9 Å), while SIFSIX-Cu-TPA possesses icosahedral cages (∼6.0 Å) and tetrahedral cages (∼4.7 Å). Though the pyridine rings of Tripp (D3h, excluding the non-coordinating pyridine N atom) and TPA (C3v) ligands could rotate around C(pyridine)–C(pyridine) or N–C(benzene) single bonds, their three pyridyl N donors were locked into a fixed, equilateral triangular geometry. In contrast, the rotation of C(benzene)–N(imidazole) bonds in the TIB (C3) linkers results in dynamic positions of coordination imidazole N atoms, breaking the perfect symmetry, as presented in Fig. 1a and S4. Such ligand asymmetry easily leads to a distorted coordination geometry and complicates the crystallization process, hindering the predictable assembly of conventional networks. Concretely, in the Tripp-Cu-SIFSIX, SIFSIX-Cu-TPA, and CTGU-45, all the organic linkers act as 3-connected topological nodes with D3h (Tripp/TPA-node) and C3 (TIB-node) symmetry, respectively. Meanwhile, the coordination numbers of Cu2+ are either 5 or 6, exhibiting square pyramidal (C4v) or octahedral (D4h) coordination geometries. The combination of organic nodes and metal nodes ultimately gives rise to (3,5)-c or (3,6)-c topological frameworks in three APMOFs (Fig. 1c/g/e). It is noteworthy that although Cu2+ acts as a 6-c node in both SIFSIX-Cu-TPA and CTGU-45, the four pyridine rings coordinated to the Cu2+ center in the former exhibit relative dihedral angles close to 90°, whereas the four imidazole rings coordinated to Cu2+ in the latter are severely twisted. This causes the 6-c nodes in CTGU-45 to link adjacent nodes in a more distorted fashion, breaking the D4h extension symmetry of the 6-c nodes observed in SIFSIX-Cu-TPA. This, together with the 3-c nodes with C3 symmetry, facilitates the formation of a geometrically self-consistent fundamental cage-like network in the absence of SiF62− anions (Fig. S4). The foregoing analysis has validated the critical influence of ligand symmetry on the formation of anionic hybrid frameworks. Overall, compared to Tripp-Cu-SIFSIX and SIFSIX-Cu-TPA, CTGU-45 shares the common feature of incorporating SiF62− anions, whose electronegative F atoms serve as Lewis-basic sites for interacting with acidic H atoms of C2H2. However, a fundamental distinction lies in their pore architecture: in conventional APMOFs, the SiF62− anions are integral components that participate directly in cage formation, resulting in highly polar pore surfaces dominated by F atoms (Fig. 1h/k and j/m). In contrast, in CTGU-45, the pore cages are constructed exclusively by the TIB ligands and Cu(II) nodes, while the SiF62− anions merely serve as inter-cage pillars (determined by the topology, Fig. 1i/l). This unique arrangement creates a low-polar pore surface rich in aromatic rings and N atoms, which not only provides optimal van der Waals interactions with highly polarizable C2H6 molecules but also enables synergistic binding through multiple C–H⋯π and C–H⋯N interactions. This distinctive pore environment, combined with the selective C–H⋯F binding sites at the SiF62− pillars, allows CTGU-45 to achieve cooperative adsorption of both C2H2 and C2H6, thereby providing a robust foundation for one-step C2H4 purification from ternary mixtures. The phase purity of the as-synthesized CTGU-45 was confirmed by powder X-ray diffraction (PXRD), as the experimental pattern matches well with simulated ones (Fig. S5). The thermogravimetric analysis (TGA) revealed the high thermal stability of activated CTGU-45, which remains stable up to 350 °C (Fig. S6). The initial weight loss below 100 °C is ascribed to solvent removal, while the subsequent major loss above 350 °C corresponds to the decomposition of the MOF framework. The N2 adsorption isotherm of CTGU-45 at 77 K displayed a typical Type I profile, and the corresponding HK pore size distribution suggested ultra-micropores of 6.1 Å, collectively confirming its microporous characteristic. The N2 adsorption uptake of 125.5 cm3 g−1 at 1 kPa and the calculated BET surface area of 544.8 m2 g−1 further corroborated its porous structure (Fig. 2a). Moreover, the experimental pore volume of CTGU-45 was calculated to be 0.21 cm3 g−1, which is consistent with the theoretical value from crystal structure (0.24 cm3 g−1). Combined with its excellent thermal stability, these integrated properties collectively endorse CTGU-45 as a highly promising adsorbent for gas separation applications.
Single-component adsorption isotherms of CH4, CO2 and C2–C3 hydrocarbons on CTGU-45 were measured at 273, 283 and 298 K (Fig. 2b, c and S7–S18). CTGU-45 exhibited an obviously preferential adsorption of C2H2 over CO2 and CH4, with an uptake of 2.83 mmol g−1 at 298 K and 101 kPa, significantly exceeding those of CO2 (1.64 mmol g−1) and CH4 (0.74 mmol g−1). Notably, the uptakes of C2H2 (2.00 mmol g−1) and C2H6 (1.88 mmol g−1) are significantly higher than that of C2H4 (1.75 mmol g−1) at 30 kPa, thus showing great potential for one-step C2H4 purification. The C2H2 uptake capacity of CTGU-45 is higher than that of many reported APMOFs, such as NBOFFIVE-dps-Cu (1.65 mmol g−1),25 sql-SIFSIX-bpe-Zn (1.79 mmol g−1),26 DICRO-4-Ni-I (1.92 mmol g−1),27 ZNU-15 (1.61 mmol g−1),28 BSF-2 (1.85 mmol g−1),29 and BSF-4 (2.38 mmol g−1).30
Meanwhile, the CO2 adsorption amount is markedly lower than that of top-performing APMOFs, including SIFSIX-Cu-TPA (4.79 mmol g−1),24 QDU-MOF-1 (5.70 mmol g−1),31 TIFSIX-2-Cu-I (4.3 mmol g−1),32 ZNU-9 (4.32 mmol g−1),33 and ZNU-12 (4.33 mmol g−1).34 According to the virial equation, the isosteric heat of adsorption (Qst) values at near-zero loading for C2H2, C2H4, C2H6, CO2, and CH4 on CTGU-45 are 30.9, 25.8, 27.3, 28.0, and 20.9 kJ mol−1, respectively (Fig. 2d and S24–S28). The higher Qst value for C2H2 and C2H6, consistent with the steeper slope of its single-component adsorption isotherm, indicates a stronger binding affinity between the framework and C2H2 and C2H6 molecules. Overall, except for ZNU-8 (27.2 kJ mol−1)33 and MPM-1-TIFSIX (30.1 kJ mol−1),35 CTGU-45 exhibits the lowest Qst value for C2H2 among reported APMOFs, which is significantly lower than that of QDU-MOF-1 (72.52 kJ mol−1),31 TIFSIX-17-Ni (48.3 kJ mol−1),36 NBOFFIVE-dps-Cu (53.6 kJ mol−1),37 SIFSIX-Cu-TPA (39.1 kJ mol−1)24 and so on (Fig. 2h). Given that only one APMOF (ZUL-100)33 has been reported for C2H4 purification from C2 mixtures, the C2H6 Qst value of CTGU-45 (27.3 kJ mol−1) is not only lower than that of ZUL-100 (34.2 kJ mol−1),33 but also lower than those of most previously reported C2H2/C2H6-selective MOFs for one-step C2H4 purification, such as Zn-trz-ox (38.7 kJ mol−1),38 NUM-9 (35.8 kJ mol−1),39 Zn-atz-oba (30.0 kJ mol−1),4 TJT-100 (29.0 kJ mol−1),40 and BUT-321 (28.5 kJ mol−1)41 (Fig. 2i). This low Qst value ensures easy desorption and low-energy regeneration, which might significantly reduce the operating cost in practical separation cycles.
In addition, since practical gas separation is a non-equilibrium process, the kinetic adsorption behavior should be evaluated synchronously. Therefore, we studied the kinetic diffusion rate of C2H2, C2H4, C2H6, CO2, and CH4 in CTGU-45. As shown in Fig. 2g, the time required to reach adsorption equilibrium follows the order: Tads (CH4) < Tads (C2H2) < Tads (CO2) < Tads (C2H6) < Tads (C2H4). In detail, the calculated diffusion time constants (Dc/rc2) of these gases on CTGU-45 are 0.0380 (CH4), 0.2628 (C2H2), 0.0284 (CO2), 0.0141 (C2H6), and 0.0104 s−1 (C2H4), respectively (Fig. S29). Interestingly, in the desorption kinetics tests (Fig. S30), while CH4 and CO2 still exhibited the fastest desorption, the desorption rate sequence of the C2 gases (Tdes (C2H4) < Tdes (C2H6) < Tdes (C2H2)) was exactly opposite to their respective adsorption rate sequence. This observation directly reflects the relative strength of their interactions with the framework, consistent with the assessments discussed earlier.
To further assess the separation potential of CTGU-45, the adsorption selectivity for equimolar C2H2/CO2, C2H2/CH4, C2H2/C2H4, and C2H6/C2H4 mixtures at 298 K was calculated based on the Ideal Adsorbed Solution Theory (IAST) after fitting isotherms to the single-site/dual-site Langmuir–Freundlich equation with excellent accuracy (Fig. S19–S23, R2 > 0.99999). As illustrated in Fig. 2e and f, the IAST selectivity of CTGU-45 at 298 K and 101 kPa reached 6.99 for C2H2/CO2 and 38.72 for C2H2/CH4; notably, the selectivity for C2H2/C2H4 (50/50, v/v) (1.63) and C2H6/C2H4 (50/50, v/v) (1.40) further underscore its potential for one-step C2H4 purification. The C2H2/CO2 selectivity is moderate among benchmark APMOFs, being comparable to those of NBOFFIVE-3-Ni (6.0) and TIFSIX-2-Cu-I (6.5), and higher than SIFSIX-Cu-TPA (5.3), QDU-MOF-1 (2.95), and ZNU-8 (3.74) (Fig. 2h). Meanwhile, the moderate C2H2/CO2 selectivity, combined with the high C2H2/CH4 selectivity, suggests that CTGU-45 is a viable candidate for separating C2H2/CO2 and C2H2/CH4 mixtures. Furthermore, the calculated C2H2 and CO2 uptake (2.20 and 0.32 mmol g−1) in the equimolar mixture closely matches the corresponding single-component adsorption capacities at 50 kPa (2.37 and 1.02 mmol g−1), confirming the consistency between mixture prediction and gas adsorption measurement.
Grand Canonical Monte Carlo (GCMC) simulations and density functional theory with dispersion corrections (DFT-D) calculations were employed to elucidate the gas adsorption mechanisms in CTGU-45, revealing two primary binding sites within the framework: one near the SiF62− anion pillars and another inside the pore cavity (Fig. 3). In general, the region adjacent to the SiF62− pillars serves as a strong adsorption site for CO2, CH4, C2H2, and C2H4, whereas the pore cavity functions as the preferred strong binding region for C2H6. Taking C2H2 as a representative case, the first corresponds to a strong adsorption site, where the C2H2 molecule was confined by two SiF62− with a binding energy of −53.42 kJ mol−1. Specifically, each H atom of C2H2 forms two C–H⋯F hydrogen bonds (H-bonds) with F atoms from two adjacent SiF62− pillars, resulting in a total of four H-bonds with distances ranging from 2.34 to 3.69 Å. These are complemented by C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C⋯H interactions (distances: 2.58–3.45 Å) between the π-electrons of C2H2 and H atoms of imidazole rings. The second site corresponds to a weaker interaction environment within the pore cavity, involving C
C⋯H interactions (distances: 2.58–3.45 Å) between the π-electrons of C2H2 and H atoms of imidazole rings. The second site corresponds to a weaker interaction environment within the pore cavity, involving C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C⋯H (distances: 3.85–3.94 Å), C–H (C2H2)⋯N(imidazole) (2.91–3.82 Å), and C–H (C2H2)⋯π (imidazole/benzene) interactions with the distances of 3.10–3.92 Å. The binding energy for this weak-interaction site is −19.92 kJ mol−1, substantially lower than that of the above-mentioned site. A similar distribution of adsorption sites was observed for CO2, CH4, and C2H4 with strong binding near the SiF62− pillars and weaker interactions in the pore cavity. For CO2, the electronegative O atoms participated in C
C⋯H (distances: 3.85–3.94 Å), C–H (C2H2)⋯N(imidazole) (2.91–3.82 Å), and C–H (C2H2)⋯π (imidazole/benzene) interactions with the distances of 3.10–3.92 Å. The binding energy for this weak-interaction site is −19.92 kJ mol−1, substantially lower than that of the above-mentioned site. A similar distribution of adsorption sites was observed for CO2, CH4, and C2H4 with strong binding near the SiF62− pillars and weaker interactions in the pore cavity. For CO2, the electronegative O atoms participated in C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯H and C
O⋯H and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯N contacts with H and N atoms of the imidazole rings, as well as C
O⋯N contacts with H and N atoms of the imidazole rings, as well as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O⋯π interactions with the aromatic systems. Simultaneously, its electropositive C atom interacts with F atoms of the SiF62− pillars via O
O⋯π interactions with the aromatic systems. Simultaneously, its electropositive C atom interacts with F atoms of the SiF62− pillars via O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C⋯F interactions. In the case of CH4, C2H4, and C2H6, adsorption was governed by C–H⋯F and C–H⋯N interactions with the pillar F and imidazole N atoms, respectively, along with C–H⋯π contacts involving the aromatic rings of the TIB ligands. Notably, C2H6 exhibits its strongest adsorption within the pore cavity rather than near the anion pillars. Consequently, the binding energies of CO2 and CH4 at both types of adsorption sites are substantially lower than those of C2H2, while those of C2H2 and C2H6 are higher than those of C2H4. This energetic trend (C2H2 > CO2 > CH4; C2H2 > C2H6 > C2H4) further rationalizes the preferential adsorption of C2H2 over CO2 and CH4, and of C2H2 and C2H6 over C2H4 by CTGU-45. The stronger intermolecular interactions between the framework and C2H2/C2H6 directly correlate with its higher adsorption affinity, which is consistent with the observation on Qst and single-component adsorption data.
C⋯F interactions. In the case of CH4, C2H4, and C2H6, adsorption was governed by C–H⋯F and C–H⋯N interactions with the pillar F and imidazole N atoms, respectively, along with C–H⋯π contacts involving the aromatic rings of the TIB ligands. Notably, C2H6 exhibits its strongest adsorption within the pore cavity rather than near the anion pillars. Consequently, the binding energies of CO2 and CH4 at both types of adsorption sites are substantially lower than those of C2H2, while those of C2H2 and C2H6 are higher than those of C2H4. This energetic trend (C2H2 > CO2 > CH4; C2H2 > C2H6 > C2H4) further rationalizes the preferential adsorption of C2H2 over CO2 and CH4, and of C2H2 and C2H6 over C2H4 by CTGU-45. The stronger intermolecular interactions between the framework and C2H2/C2H6 directly correlate with its higher adsorption affinity, which is consistent with the observation on Qst and single-component adsorption data.
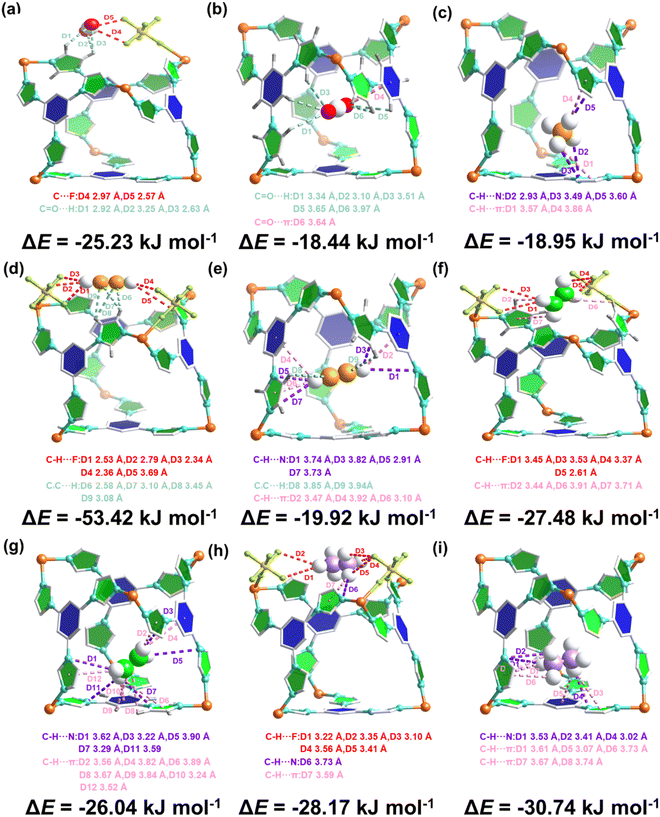 | ||
| Fig. 3 Optimal binding sites and adsorption configurations of (a and b) CO2, (c) CH4, (d and e) C2H2, (f and g) C2H4, and (h and i) C2H6 molecules within CTGU-45 defined by DFT-D calculations. | ||
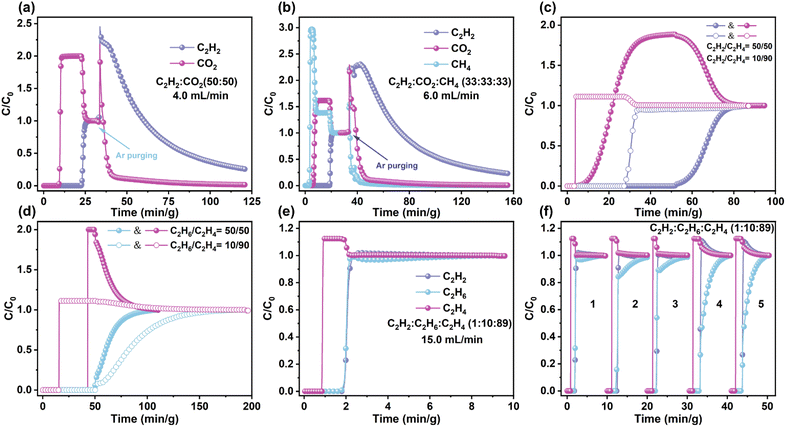
Dynamic breakthrough experiments were conducted to further evaluate the practical separation performance of CTGU-45 for C2H2/CO2, C2H2/CO2/CH4, and C2 hydrocarbon mixtures. As depicted in Fig. 4a, CTGU-45 can achieve efficient separation of a C2H2/CO2 (50/50, v/v) mixture at a total gas flow rate of 4.0 mL min−1, wherein CO2 gases were detected in ∼9 min g−1 while C2H2 breakthrough occurred at ∼22 min g−1. Based on the integration of the curve, the dynamic CO2 and C2H2 capture amounts of CTGU-45 were calculated to be 6.10 and 47.68 cm3 g−1, corresponding to approximately 26 and 90% of their respective static uptake values at 50 kPa (23.04 and 53.12 cm3 g−1), respectively. After adsorption saturation, the column was regenerated by purging with helium at 10 mL min−1 under ambient temperature. The desorption profiles indicated rapid release of both gases, with CO2 completely desorbed within 23 min g−1. Beyond this point, high-purity C2H2 (>95.2%) was obtained at a productivity of 22.1 L kg−1 (Fig. 4a). Subsequently, we further investigated the separation capacity of CTGU-45 for actual ternary C2H2/CO2/CH4 mixtures (33/33/33, v/v/v) with flow rates of 6 and 3 mL min−1. Owing to its higher C2H2 uptakes and selectivity over CO2 and CH4, efficient separation of C2H2 from C2H2/CO2/CH4 mixtures can be fulfilled by CTGU-45 under different temperatures (298 and 308 K, Fig. S31).
As illustrated in Fig. 4b, CH4 first eluted through the fixed bed at 3 min g−1, followed by CO2 at 4 min g−1, whereas the C2H2 was retained in the packed column over 18 min g−1. Despite CO2 and CH4 exhibiting a faster adsorption rate than C2H2 in CTGU-45, the stronger host–guest interactions between the framework and C2H2 (C–H⋯F, C–H⋯N, and C–H⋯π interactions) confer thermodynamic dominance in the actual separation process, leading to preferential C2H2 capture and the longest breakthrough time. The desorption curve further confirmed that polymer-grade C2H2 can be recovered at a productivity (>95.2%) of 19.7 L kg−1.
For C2H2/C2H4 and C2H6/C2H4 mixtures with different compositions (50/50 or 10/90, v/v), C2H4 first eluted from the packed column while C2H2 or C2H6 remained strongly retained within the CTGU-45, demonstrating its ability to directly produce polymer-grade C2H4 products. In detail, C2H4 molecules broke through the fixed bed and were detected at 4.2 (3.7) and 42.9 (15.5) min g−1 for the 50/50 (10/90) C2H2/C2H4 and C2H6/C2H4 mixtures, respectively, whereas C2H2 and C2H6 were adsorbed in the column over 51.7 (27.6) and 49.9 (51.4) min g−1. As depicted in Fig. 4c and d, the productivity of 99.9% pure C2H4 from the 50/50 (10/90) C2H2/C2H4 and C2H6/C2H4 mixtures reached 28.6 (47.7) and 6.0 (33.3) L kg−1, respectively. Building on the excellent separation performance observed for these binary mixtures, breakthrough experiments were further conducted using a ternary C2H2/C2H6/C2H4 (1/10/89, v/v/v) mixture. Notably, C2H4 eluted (0.9 min g−1) prior to both C2H2 (1.8 min g−1) and C2H6 (1.7 min g−1), enabling the direct production of high-purity C2H4 (Fig. 4e). After calculation, 9.57 L kg−1 of C2H4 with ≥99.95% purity can be directly obtained in a single cycle (298 K, 15 mL min−1). For both ternary mixtures, CTGU-45 maintains reliable efficiency in C2H2 and C2H4 purification with steady gas productivities when the operating temperature is raised from 298 to 308 K, or when the flow rate is decreased from 6 to 3 mL min−1 (15 to 14 mL min−1, Fig. S31). Moreover, repeated breakthrough experiments over five consecutive cycles showed no discernible decay in either C2H4 purity or productivity (9.24–11.0 L kg−1), confirming the stable one-step C2H4 purification capability of CTGU-45 (Fig. 4f and S32). Finally, PXRD measurements reveal that CTGU-45 displays moderate chemical stability across a wide pH range (1–13) and in aqueous environments (Fig. S33). The combination of high separation efficiency, excellent adaptability, good thermal/chemical stability, and mild regeneration requirements (room-temperature He purging) underscores the potential of CTGU-45 for practical C2H2 and C2H4 purification.
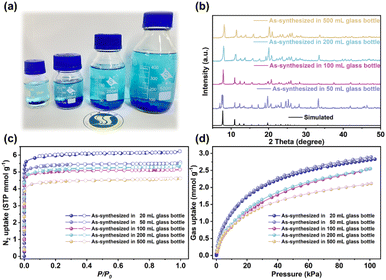
In addition to the impressive separation properties, CTGU-45 crystals can be successfully synthesized at scale-up levels using glass containers with volumes ranging from 50 to 500 mL (Fig. 5a). PXRD measurements confirm the basic phase purity of the CTGU-45 sample obtained via scaled-up synthesis (Fig. 5b). Although the N2 adsorption capacity of samples synthesized in 50, 100, and 200 mL vessels were only slightly lower than that of the 20 mL reference (Fig. 5c), and the sample prepared in a 500 mL bottle exhibits a 25.2% reduction in N2 uptake. To further assess the impact on functional performance, C2H2 adsorption isotherms were measured at 298 K (Fig. 5d). The results indicate that the C2H2 uptake of samples from 20 and 50 mL vessels is nearly identical, while those from 100 and 200 mL vessels show an approximate 8.8% decrease. In contrast, the sample synthesized in the 500 mL bottle exhibits a 25.4% reduction in C2H2 adsorption capacity, consistent with the trend observed in N2 adsorption. Through additional analysis of the N2 ads/des–orption isotherms, pore size distributions (Fig. S34), and PXRD patterns of samples synthesized at different scales, we found that the decrease in adsorption capacity may arise not only from undesired structural defects but also from the presence of by-products (amorphous, non-porous, and non-adsorbing). Since MOF synthesis is a complex self-assembly process governed by both thermodynamic and kinetic factors, changes in the scale of the reaction system—that is, the microscopic reaction conditions—can perturb the reaction pathway, leading to the formation of unknown by-products. To mitigate the formation of amorphous by-products and structural defects during industrial production for CTGU-45, future efforts might focus on two key strategies: (i) the implementation of continuous-flow synthesis or microwave-assisted heating can ensure uniform nucleation and growth conditions, thereby suppressing kinetic traps that lead to amorphous phases (ii) dynamic adjustment of reaction parameters (e.g., reactant concentration, temperature gradients, local mass transfer); to steer the self-assembly toward the desired crystalline product.
Conclusions
In summary, we reported a SiF62− embedded MOF, CTGU-45, featuring abundant electronegative F atoms and a low-polar cage environment, which enables one-step C2H4 purification via the simultaneous removal of C2H2 and C2H6. CTGU-45 exhibits high C2H2 and C2H6 uptake, markedly exceeding those for CO2, CH4, and C2H4. The low-polar pore environment effectively addresses the critical challenge of achieving C2H6-selective adsorption in C2H6/C2H4 separation, a behavior that is exceedingly rare among reported APMOFs. As a result, ∼9.57 L kg−1 of polymer grade C2H4 (≥99.95%) can be directly produced from ternary C2H2/C2H6/C2H4 (1/10/89, v/v/v) mixtures in a single step. DFT calculations reveal that strong C–H⋯F interactions between the inorganic SiF62− pillars and C2H2 enable CTGU-45 to not only efficiently purify C2H2 from C2H2/CO2/CH4 mixtures but also, in synergy with its C2H6-trapping capability, to achieve one-step production of high-purity C2H4 from ternary C2 hydrocarbon mixtures. In addition, the low C2H6 Qst (27.3 kJ mol−1) ensures facile regeneration with low energy input. Notably, CTGU-45 can be synthesized on a scalable level (up to 500 mL) with well-retained adsorption capacity. Overall, this work demonstrates that breaking ligand symmetry in APMOFs can fundamentally alter the spatial role of anion pillars, thereby reshaping pore surface chemistry and enabling rare one-step C2H4 purification from C2 hydrocarbon mixtures.Author contributions
J.-J. Wu, P.-D. Zhang, X.-Q. Wu, and D.-S. Li conceived this project and designed the experiments. J.-J. Wu, S.-H. Huang, W.-W. Dong and Y.-P. Wu conducted the experiments. J.-J. Wu, P.-D. Zhang and X.-Q. Wu drafted the work. D.-S. Li revised and finalised the manuscript. All authors approved the submitted version.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are openly available in the supporting information (SI). Supplementary information: experimental procedures, TGA curves, gas adsorption/separation isotherms, crystallographic data, and theoretical computational details. See DOI: https://doi.org/10.1039/d6sc00819d.CCDC 2522768 contains the supplementary crystallographic data for this paper.42
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (No. 22401172, No. 22371165), the 111 Project (D20015), the project of Hubei Three Gorges Laboratory (Z2022078, SK213002, SC232018, SC240013), Natural Science Foundation of Hubei Province (2024AFB021, 2022CFB326, Z2023203), Natural Science Foundation Innovation and Development Joint Fund of Hubei Province (2024AFD197).Notes and references
- K. M. Carsch, H. Z. H. Jiang, R. A. Klein, A. S. Rosen, P. S. Summerhill, J. L. Peltier, A. J. Huang, R. A. Murphy, M. N. Dods, H. A. Silva, Z. Hasanbasri, H. Kwon, S. L. Karstens, Y. Yabuuchi, J. Börgel, J. W. Taylor, K. R. Meihaus, K. C. Bustillo, A. M. Minor, K. A. Persson, C. M. Brown, R. D. Britt, N. P. Stadie and J. R. Long, Science, 2025, 390, 808–812 CrossRef CAS PubMed.
- I. J. Vitórica-Yrezábal, C. A. McAnally, M. P. Snelgrove, M. R. Warren, A. H. Hill, S. P. Thompson, M. Quinn, S. Mottley, S. Mottley, A. J. Fletcher and L. Brammer, Nat. Chem., 2025, 17, 1705–1711 CrossRef PubMed.
- Z. Zhou, T. Ma, H. Zhang, S. Chheda, H. Li, K. Wang, S. Ehrling, R. Giovine, C. Li, A. H. Alawadhi, M. M. Abduljawad, M. O. Alawad, L. Gagliardi, J. Sauer and O. M. Yaghi, Nature, 2024, 635, 96–101 CrossRef CAS PubMed.
- J.-W. Cao, S. Mukherjee, T. Pham, Y. Wang, T. Wang, T. Zhang, X. Jiang, H.-J. Tang, K. A. Forrest, B. Space, M. J. Zaworotko and K.-J. Chen, Nat. Commun., 2021, 12, 6507 CrossRef CAS PubMed.
- C. Lu, S. Liu, Z. Wang, X. Wei, X. Chen, X. Wang, J. Pang, S. Geng, X. Lu, J. Duan, F. Dai and X.-H. Bu, Adv. Mater., 2025, 38, e14488 CrossRef PubMed.
- L. Li, R.-B. Lin, R. Krishna, H. Li, S. Xiang, H. Wu, J. Li, W. Zhou and B. Chen, Science, 2018, 362, 443–446 CrossRef CAS PubMed.
- Y. Yang, L. Li, R.-B. Lin, Y. Ye, Z. Yao, L. Yang, F. Xiang, S. Chen, Z. Zhang, S. Xiang and B. Chen, Nat. Chem., 2021, 13, 933–939 CrossRef CAS PubMed.
- P. Ajayan, W. Wang, Y. Chen, X. Bu and P. Feng, Adv. Mater., 2024, 36, 2408042 CrossRef CAS PubMed.
- B. E. R. Snyder, A. B. Turkiewicz, H. Furukawa, M. V. Paley, E. O. Velasquez, M. N. Dods and J. R. Long, Nature, 2023, 613, 287–291 CrossRef CAS PubMed.
- L. Li, L. Guo, D. H. Olson, S. Xian, Z. Zhang, Q. Yang, K. Wu, Y. Yang, Z. Bao, Q. Ren and J. Li, Science, 2022, 377, 335–339 CrossRef CAS PubMed.
- G.-R. Si, X.-J. Kong, L.-H. Xie, Y.-T. Zhang, T. He and J.-R. Li, Sci. Bull., 2025, 70, 3475–3478 CrossRef CAS PubMed.
- Z.-H. Guo, L.-Q. Yang, Q.-G. Zhai, G.-P. Yang and Y.-Y. Wang, Angew. Chem., Int. Ed., 2025, 64, e202519278 CrossRef CAS PubMed.
- L. Wang, H. Chen, C. Lou, G. Xiong, Y. Jiang, B. Chen and Y. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202519160 CrossRef.
- Z.-S. Wang, M.-Y. Zhou, D.-Y. Hu, X.-X. Chen, J.-X. Chen, X.-W. Zhang, D.-D. Zhou, J.-P. Zhang and X.-M. Chen, J. Am. Chem. Soc., 2025, 147, 47977–47984 CrossRef CAS PubMed.
- Y. Xiao, A. N. Hong, Z. Jia, X. Bu and P. Feng, J. Am. Chem. Soc., 2025, 147, 42786–42795 CAS.
- T. Wu, D. Zhou, Z. Yang, Y. Liu, P. Yin, K. Lu, F. Jiang, X. Suo, L. Yang, X. Cui and H. Xing, J. Am. Chem. Soc., 2025, 148, 5299–5306 CrossRef PubMed.
- Z. Ji, Y. Zhou, C. Chen, D. Yuan, M. Wu and M. Hong, Angew. Chem., Int. Ed., 2024, 63, e202319674 CrossRef CAS PubMed.
- Y. Jiang, Y. Hu, B. Luan, L. Wang, R. Krishna, H. Ni, X. Hu and Y. Zhang, Nat. Commun., 2023, 14, 401 CrossRef PubMed.
- A. Cadiau, K. Adil, P. M. Bhatt, Y. Belmabkhout and M. Eddaoudi, Science, 2016, 353, 137–140 CrossRef CAS PubMed.
- F. Zheng, L. Guo, R. Chen, L. Chen, Z. Zhang, Q. Yang, Y. Yang, B. Su, Q. Ren and Z. Bao, Angew. Chem., Int. Ed., 2022, 61, e202116686 CrossRef CAS PubMed.
- X. Cui, K. Chen, H. Xing, Q. Yang, R. Krishna, Z. Bao, H. Wu, W. Zhou, X. Dong, Y. Han, B. Li, Q. Ren, M. J. Zaworotko and B. Chen, Science, 2016, 353, 141–144 CrossRef CAS PubMed.
- H.-M. Wen, C. Liao, L. Li, L. Yang, J. Wang, L. Huang, B. Li, B. Chen and J. Hu, Chem. Commun., 2019, 55, 11354–11357 RSC.
- M. Lusi, P. B. A. Fechine, K.-J. Chen, J. J. Perry and M. J. Zaworotko, Chem. Commun., 2016, 52, 4160–4162 RSC.
- H. Li, C. Liu, C. Chen, Z. Di, D. Yuan, J. Pang, W. Wei, M. Wu and M. Hong, Angew. Chem., Int. Ed., 2021, 60, 7547–7552 CrossRef CAS PubMed.
- M. Shivanna, K.-i. Otake, B.-Q. Song, L. M. van Wyk, Q.-Y. Yang, N. Kumar, W. K. Feldmann, T. Pham, S. Suepaul, B. Space, L. J. Barbour, S. Kitagawa and M. J. Zaworotko, Angew. Chem., Int. Ed., 2021, 60, 20383–20390 CrossRef CAS PubMed.
- M.-Y. Gao, A. A. Bezrukov, B.-Q. Song, M. He, S. J. Nikkhah, S.-Q. Wang, N. Kumar, S. Darwish, D. Sensharma, C. Deng, J. Li, L. Liu, R. Krishna, M. Vandichel, S. Yang and M. J. Zaworotko, J. Am. Chem. Soc., 2023, 145, 11837–11845 CrossRef CAS PubMed.
- H. S. Scott, M. Shivanna, A. Bajpai, D. G. Madden, K.-J. Chen, T. Pham, K. A. Forrest, A. Hogan, B. Space, J. J. Perry Iv and M. J. Zaworotko, ACS Appl. Mater. Interfaces, 2017, 9, 33395–33400 CrossRef CAS PubMed.
- H. Chen, Y. He, Y. Han, J. Hu, J. Li, Y. Jiang, B. Keshta, L. Wang and Y. Zhang, Chin. J. Struct. Chem., 2025, 44, 100508 CAS.
- Y. Zhang, J. Hu, R. Krishna, L. Wang, L. Yang, X. Cui, S. Duttwyler and H. Xing, Angew. Chem., Int. Ed., 2020, 59, 17664–17669 CrossRef CAS PubMed.
- L. Wang, W. Sun, Y. Zhang, N. Xu, R. Krishna, J. Hu, Y. Jiang, Y. He and H. Xing, Angew. Chem., Int. Ed., 2021, 60, 22865–22870 CrossRef CAS PubMed.
- H.-Y. Li, Z.-Z. Xue, S.-D. Han, G.-M. Wang and T. He, Sep. Purif. Technol., 2025, 357, 130094 CrossRef CAS.
- K.-J. Chen, H. S. Scott, D. G. Madden, T. Pham, A. Kumar, A. Bajpai, M. Lusi, K. A. Forrest, B. Space, J. J. Perry and M. J. Zaworotko, Chem, 2016, 1, 753–765 Search PubMed.
- Y. Zhang, W. Sun, B. Luan, J. Li, D. Luo, Y. Jiang, L. Wang and B. Chen, Angew. Chem., Int. Ed., 2023, 62, e202309925 CrossRef CAS PubMed.
- Y. Zhang, Y. Han, B. Luan, L. Wang, W. Yang, Y. Jiang, T. Ben, Y. He and B. Chen, J. Am. Chem. Soc., 2024, 146, 17220–17229 CrossRef CAS PubMed.
- P. S. Nugent, V. L. Rhodus, T. Pham, K. Forrest, L. Wojtas, B. Space and M. J. Zaworotko, J. Am. Chem. Soc., 2013, 135, 10950–10953 CrossRef CAS PubMed.
- S. Mukherjee, N. Kumar, A. A. Bezrukov, K. Tan, T. Pham, K. A. Forrest, K. A. Oyekan, O. T. Qazvini, D. G. Madden, B. Space and M. J. Zaworotko, Angew. Chem., Int. Ed., 2021, 60, 10902–10909 CrossRef CAS PubMed.
- N. Kumar, S. Mukherjee, N. C. Harvey-Reid, A. A. Bezrukov, K. Tan, V. Martins, M. Vandichel, T. Pham, L. M. van Wyk, K. Oyekan, A. Kumar, K. A. Forrest, K. M. Patil, L. J. Barbour, B. Space, Y. Huang, P. E. Kruger and M. J. Zaworotko, Chem, 2021, 7, 3085–3098 CAS.
- T. Wu, C. Yu, R. Krishna, Z. Qiu, H. Pan, P. Zhang, X. Suo, L. Yang, X. Cui and H. Xing, AIChE J., 2024, 70, e18312 CrossRef CAS.
- S.-Q. Yang, F.-Z. Sun, P. Liu, L. Li, R. Krishna, Y.-H. Zhang, Q. Li, L. Zhou and T.-L. Hu, ACS Appl. Mater. Interfaces, 2021, 13, 962–969 CrossRef CAS PubMed.
- H.-G. Hao, Y.-F. Zhao, D.-M. Chen, J.-M. Yu, K. Tan, S. Ma, Y. Chabal, Z.-M. Zhang, J.-M. Dou, Z.-H. Xiao, G. Day, H.-C. Zhou and T.-B. Lu, Angew. Chem., Int. Ed., 2018, 57, 16067–16071 CrossRef CAS PubMed.
- P.-D. Zhang, X.-Q. Wu, Q. Shuai, J. Yu, X. Zhang and J.-R. Li, ACS Mater. Lett., 2024, 6, 4632–4638 CrossRef CAS.
- CCDC 2522768: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qp4m6.
| This journal is © The Royal Society of Chemistry 2026 |