Open Access Article
Open Access ArticleAnion-sublattice engineering of Li3PS4:Br and I incorporation enhances ionic conductivity and Li-metal compatibility
Tej P. Poudel†
abc,
Michael J. Deck†bc,
Ifeoluwa P. Oyekunlebc,
Pawan K. Ojha bc,
Bright O. Ogbolubc,
Islamiyat Ojeladebc,
Thilina N. D. D. Gamaralagalebc,
Erica Truongbc,
Yongkang Jinbc,
Amirhossein Zareihassangheshlaghib and
Yan-Yan Hu
bc,
Bright O. Ogbolubc,
Islamiyat Ojeladebc,
Thilina N. D. D. Gamaralagalebc,
Erica Truongbc,
Yongkang Jinbc,
Amirhossein Zareihassangheshlaghib and
Yan-Yan Hu *abc
*abc
aMaterials Science and Engineering Program, Florida State University, Tallahassee, FL 32310, USA. E-mail: yhu@fsu.edu
bDepartment of Chemistry and Biochemistry, Florida State University, Tallahassee, FL 32306, USA
cCenter for Interdisciplinary Magnetic Resonance, National High Magnetic Field Laboratory, Tallahassee, FL 32310, USA
First published on 27th April 2026
Abstract
All-solid-state batteries (ASSBs) are promising to enhance the safety and energy density of rechargeable batteries. Li3PS4 remains one of the most viable solid electrolytes (SEs) for all-solid-state lithium batteries. However, its low ionic conductivity and poor stability limit its commercial use. In this work, we introduce lithium bromide (LiBr) and lithium iodide (LiI) into the anionic sublattice of Li3PS4 to induce local disorder. An eightfold increase in ionic conductivity to 4.36 mS cm−1 at 25 °C is achieved with the 2Li3PS4:LiBr:LiI composition. 2Li3PS4:LiBr:LiI also exhibits a high room temperature critical current density of 0.92 mA cm−2 and improved electrochemical stability against lithium metal. The half-cell fabricated with 2Li3PS4:LiBr:LiI as the separator and TiS2 as the active cathode material shows significantly better rate and long-term cycling performance compared to cells based on Li3PS4. Solid-state NMR and Raman spectroscopy indicate the formation of bridging PS43− tetrahedra facilitated by the incorporation of I− and Br−. This study further highlights the advantages of strategic halide incorporation in thiophosphate electrolytes to enhance the performance of Li3PS4-based SEs and, in turn, ASSBs.
Introduction
All-solid-state batteries (ASSBs) are a promising way to improve the energy density and safety of rechargeable batteries. Solid electrolyte(s) (SEs) are a critical component in ASSBs. Li3PS4 remains one of the most promising solid electrolytes for all-solid-state lithium batteries, primarily due to its cost-effectiveness, mechanical processability, and compatibility with lithium metal. However, its practical application faces key challenges: relatively low ionic conductivity (<0.5 mS cm−1) and stability issues. Ongoing research with doping, glass–ceramic approaches, and interface engineering has made progress in addressing these challenges.1–5 With continued improvements, Li3PS4 (or modified variants) could form the backbone of next-generation solid-state batteries – offering high energy density, safety, and potentially long cycle life.Our previous work has demonstrated that incorporating mixed anions, such as O and Br, into the LPS structural framework significantly enhances Li+ ionic conductivity, stability, and critical current density.1,2 The improvement in Li+ transport in the mixed-anion SEs is attributed to increased nanoscale local disorder, which creates a flattened energy landscape.6–8 Further introduction of local disorder, i.e., the incorporation of mixed anions such as halide combinations, can yield higher-performing SEs, as is the case for Li5.3PS4.3Cl0.3Br0.7.9
Herein, we synthesize and investigate the Li+ dynamics, electrochemical performance, and local structures of xLi3PS4:yLiBr:zLiI. Electrochemical impedance spectroscopy (EIS) determined a maximum ionic conductivity of 4.36 mS cm−1 at 25 °C and a minimum activation energy of 0.29 eV for sintered 2Li3PS4:LiBr:LiI. Symmetrical galvanostatic cycling revealed significantly improved electrochemical stability against Li metal with a high critical current density (CCD). Compared to sintered 2Li3PS4:LiBr:LiI (LPSBI-sin), as-milled LPSBI (LPSBI-as) has a lower ionic conductivity of 1.4 mS cm−1 and lower CCD; however, it has a better electrochemical stability against Li metal. When assembled into a stacked pellet using the Li|LPSBI-as|LPSBI-sin|LPSBI-as|Li setup, the CCD is further improved. Cyclic voltammograms indicate that the effective electrochemical stability window is retained upon incorporation of halide salts into LPS. In ASSB half-cells using TiS2 as cathode active material (CAM), the LPSBI-containing cell showed significantly improved rate performance and long-term cycling when compared to that using LPS. Powder X-ray diffraction (PXRD) indicates the absence of long-range order, and solid-state nuclear magnetic resonance spectroscopy (NMR) and Raman spectroscopy were used to elucidate the local environments of Li and P. This work highlights the use of mixed-halide lithium thiophosphate SEs to increase local disorder and enhance SE functional properties, thus facilitating the commercialization of ASSBs.
Materials and methods
Material synthesis
Li2S, P2S5, LiBr, and LiI, were purchased from Sigma-Aldrich and stored in an argon-filled glovebox (Vacuum Technology). xLi3PS4:yLiBr:zLiI was synthesized through a high-energy ball milling technique followed by low-temperature heat treatment. First, the precursors were hand-ground to ensure a homogeneous mixture, followed by high-energy ball milling in a SPEX8000M for 20 h. The samples after ball milling were named “as-milled”. The samples were then either pelletized in a 6 mm mold into ∼1 mm thick pellets under ∼300 MPa pressure or directly transferred into an evacuated quartz tube. Sintering was carried out at 200 °C for 12 h at a ramping rate of 1 °C min−1. The sintered pellet samples are named “sintered” hereafter. All synthesis processes were performed in an argon-filled glovebox (MBRAUN).Material characterization
The DC polarization technique was also utilized to measure the effective ionic and electronic conductivity of the catholytes prepared for the TiS2 half-cell cycling, according to the literature.10 Effective electronic conductivity was measured using a SS|2SE:TiS2|SS cell setup. 100 mg of the catholyte was pressed under 250 MPa for 10 s. The effective ionic conductivity of catholytes was measured utilizing a Li–In|LPSCl|2SE:TiS2|LPSCl|Li–In cell setup. 100 mg of catholyte was first pressed in the split cell at 250 MPa for 10 s, followed by pressing 60 mg of Li6PS5Cl on each side of the catholyte pellet at 250 MPa for another 10 s. Li–In alloy was used as the Li source, while Li6PS5Cl layers were used as the electron-blocking electrodes. The calculated resistance from the current plateau is used to calculate the effective conductivity, σeff, according to 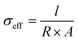 where l is the thickness of the catholyte pellet, R is the resistance, and A is the area of the electrodes.
where l is the thickness of the catholyte pellet, R is the resistance, and A is the area of the electrodes.
Results and discussion
Superionic conductivity in mixed-halide lithium thiophosphates
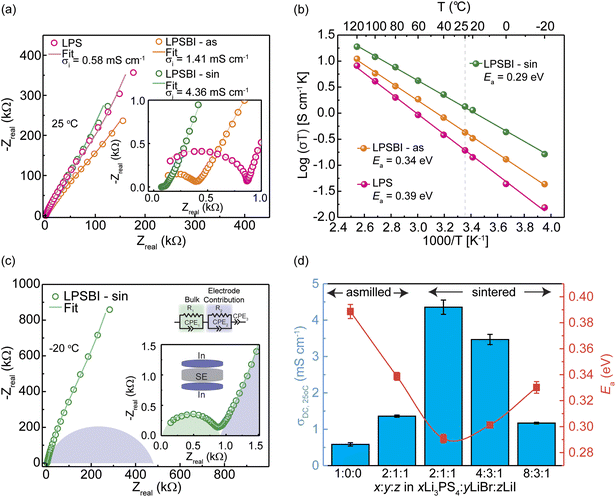
Table S1 shows the various compositions of xLi3PS4:yLiBr:zLiI and the corresponding conductivities measured at 22 °C. The highest conductivity (1.41 mS cm−1) was achieved for 2Li3PS4:LiBr:LiI (LPSBI-as) upon one-step ball milling synthesis, which was chosen as a model milled sample and denoted LPSBI-as. We sintered the milled SEs at 200 °C. Upon pellet sintering, a threefold increase in conductivity (4.36 mS cm−1) was achieved for the best conductive milled composition (2Li3PS4:LiBr:LiI), denoted as LPSBI-sin.Fig. 1a shows the Nyquist plots and equivalent circuit model fit of the fabricated SEs at 25 °C. From the resistances obtained from the fittings, 20-h high-energy milled Li3PS4 (LPS-as), LPSBI-as, and LPSBI-sin have room-temperature ionic conductivities of 0.58, 1.41, and 4.36 mS cm−1, respectively. Variable temperature EIS was performed, and Arrhenius-type plots were obtained (Fig. 1b and S1). The Li+ migration barrier (Ea), as determined from variable-temperature EIS measurements and an Arrhenius-type equation15 (Fig. 1b), is 0.39 eV for LPS-as, 0.34 eV for LPSBI-as, and 0.29 eV for LPSBI-sin, with the data listed in Table 1. The enhanced ionic conductivity upon halide introduction is due to increased local disorder at the nanoscale, which lowers the energy barrier for Li+ conduction.16 The Nyquist plot at −20 °C, with an exemplary plot presented in Fig. 1c, shows only 1 semicircle at high frequency and indicates that the bulk and grain boundary conduction cannot be separated for all samples except for 4Li3PS4:3LiBr:LiI and 8Li3PS4:3LiBr:LiI. The high-frequency impedance response is assigned to the bulk, with a capacitance of ∼79 pF.17,18 The medium-frequency semicircle is assigned to the grain boundary (GB) impedance response due to a capacitance of 0.3 nF for 4Li3PS4:3LiBr:LiI and 27 nF for 8Li3PS4:3LiBr:LiI. A low-frequency semicircle is assigned to the blocking electrode contribution, as is often the case for halide-containing SEs.19 Fig. 1d shows the conductivity values for other as-milled and sintered compositions, confirming the maximum ionic conductivity and minimum Ea achieved in 2LPS:LiBr:LiI. Furthermore, sintered 2LPS:LiBr:LiI has the lowest Arrhenius prefactor among the compositions, indicating that Ea correlates strongly with conductivity (Fig. S1). Fig. S1 also shows the Nyquist plots of the less-conductive samples, their fits, and Arrhenius-type plots. The direct current (DC) polarization method with blocking electrodes was used to determine the electronic conductivity, as shown in Fig. S2. LPSBI-sin has an electronic conductivity of 1.82 × 10−9 S cm−1.
| Composition | σDC,25°C [mS cm−1] | Ea,DC [eV] | Log(σ0) [S cm−1 K−1] |
|---|---|---|---|
| Li3PS4-as-milled | 0.58 ± 0.05 | 0.388 ± 0.005 | 5.85 ± 0.09 |
| 2Li3PS4:LiBr:LiI – as-milled | 1.41 ± 0.03 | 0.339 ± 0.003 | 5.36 ± 0.05 |
| 2Li3PS4:LiBr:LiI – sintered | 4.36 ± 0.20 | 0.291 ± 0.003 | 5.02 ± 0.05 |
| 4Li3PS4:3LiBr:LiI – sintered | 3.47 ± 0.14 | 0.301 ± 0.002 | 5.11 ± 0.04 |
| 8Li3PS4:3LiBr:1LiI – sintered | 1.17 ± 0.02 | 0.330 ± 0.004 | 5.11 ± 0.07 |
Electrochemical performance of mixed-halide lithium thiophosphates
Comparing the 1st cycle of each composition (Fig. S4d) and starting with the cathodic sweep, a cathodic peak at around 1.8 V is attributed to S reduction. LPS-as exhibits the largest current for this peak. The cathodic peak at 1.8 V is smaller for LPSBI samples because they contain less sulfur. At lower voltages, the SE redox product, P, is lithiated to form Li3P. For the anodic sweep, the peak starting at ∼1.8 V vs. Li–In is assigned Sx2− → Sx.11 The onset oxidation voltage, i.e., the oxidation of PS43−, begins at the voltage of ∼2.4 V vs. Li–In for all compositions and is consistent with previous reports for Li3PS4.1,2,11
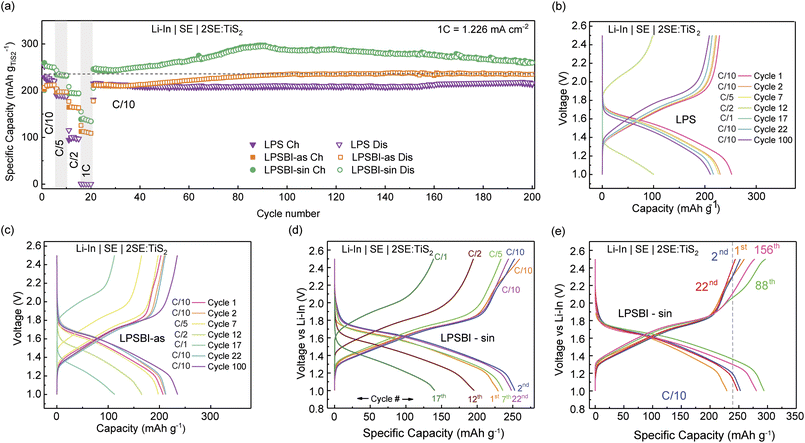
To determine the reversible redox reactions of the SEs that contribute to overall capacity, identical cells as those from CV were assembled and galvanostatically cycled (Fig. S5). Over 85 cycles, the LPSBI-sin cell shows a higher charge/discharge capacity compared to the other cells. The LPSBI-sin cell shows an improved 1st discharge capacity of ∼175 mAh gSE−1 compared to 145 mAh gSE−1 for the LPS-as cell and 112 mAh gSE−1 for the LPSBI-as cell. The irreversible capacity plateaus for the 1st cycle are the formation of a passivation layer, as extensively reported in the literature, and likely contribute to our TiS2-containing cells' (see below) capacity being slightly greater than the theoretical value for Cycles 1 and 2.1,2,11 From dQ/dV analysis (Fig. S6), it is evident that a greater amount of I− oxidation is observed for LPSBI-as, which starts at ∼2.25 V vs. Li–In; this correlates well with the 1st charge capacity of the LPSBI-as cell being significantly larger than the 1st discharge, as well as when compared to the other SEs. The oxidation voltage of I− was determined by using LiI as the active cathode, shown in Fig. S7. Notably, there is no indication of Br− oxidation at >3 V vs. Li–In, consistent with our previous studies on halide-containing SEs.1,12 On discharge, the plateau at 1.8 V is assigned to lithiation of elemental sulfur impurities from our synthesis process, as we typically observe and have reported for other Li3PS4-based solid electrolytes (as is also seen in our CV measurements).1,2,11 In agreement with the cyclic voltammograms, the quasi-plateau at 1.2 V is assigned to the reduction of the SE to form Li2S + P, and the plateau at <0.5 V is assigned to lithiation of reduction products such as P to form Li3P. Both plateaus remain reversible, indicating the effective stability window is larger than the thermodynamic window of the SE.22 This aligns well with previous experimental reports on stability windows of thiophosphates.11,23 On the first charge, we see capacity plateaus at 1.8 V, 2.25 V, and 2.4 V vs. Li–In. At 1.8 V, this is assigned to Sx2− → Sx.11 The plateau at 2.25 V is assigned to the oxidation of I− (Fig. S5). The capacity plateau at 2.4 V is assigned to the oxidation of PS43−, based on previous reports.11 However, this plateau disappears for later cycles, indicating the formation of a passivation layer after the first cycle.
Over long-term cycling at C/10 (see voltage profile of selected cycles in Fig. 3e), the LPSBI-sin cell shows a steady increase in capacity up to ∼300 mAh g−1 until cycle 88 and then gradually decreases to 236 mAh g−1 at cycle 200. A potential reason for this is I− oxidation, which is not fully reversible (Fig. S8). I− oxidation is observed at ∼2.25 V vs. Li–In, which is also seen from the dQ/dV of 3SE:SP-containing cells (Fig. S6). Our dQ/dV results on long-term cycling show that the I− oxidation peak is observed from the 1st cycle to the 88th cycle, but it is lacking for the 156th cycle (Fig. S8). Furthermore, this assignment is confirmed by comparing our galvanostatic cycling results to our previous work using 2(3Li3PS4:LiBr):TiS2 catholytes,1 where we do not observe any capacity at ∼2.25 V vs. Li–In. Besides the I− oxidation peak, we observe the onset of another peak for the first cycle at ∼2.4 V, which disappears after the first cycle. This peak for the first charge can be assigned to the oxidation of PS43−.11 The LPSBI-as-containing cell shows an increase in capacity for the same reasons; however, the maximum capacity achieved is ∼240 mA h g−1, which is significantly lower than that of the LPSBI-sin-containing cell. Conversely, the LPS-containing cell shows no capacity increase upon cycling and shows stable cycling at C/10.
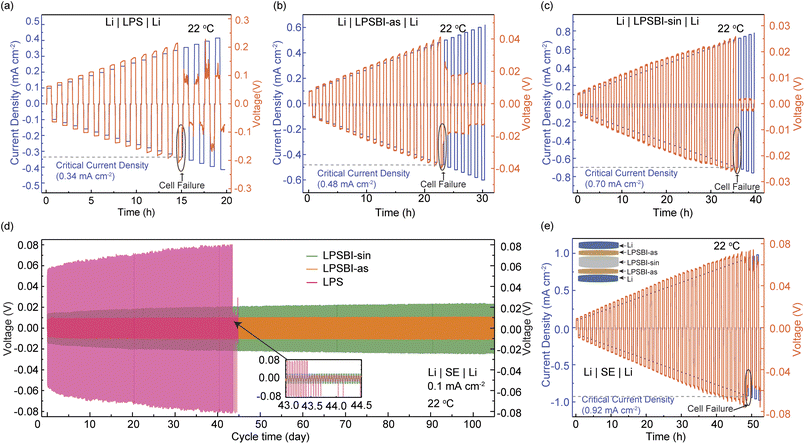
The trends in the stability of the SEs and the effective partial ionic and electronic conductivity of the catholytes closely match the trends in cycling capacity (Fig. S9). All catholytes exhibit a high effective electronic conductivity of ≥95 mS cm−1, with ionic conductivity being the kinetic rate-limiting factor. The LPSBI-sin-containing catholyte shows the highest effective ionic conductivity at 0.026 mS cm−1, compared to LPS (0.001 mS cm−1) and LPSBI-as (0.010 mS cm−1).
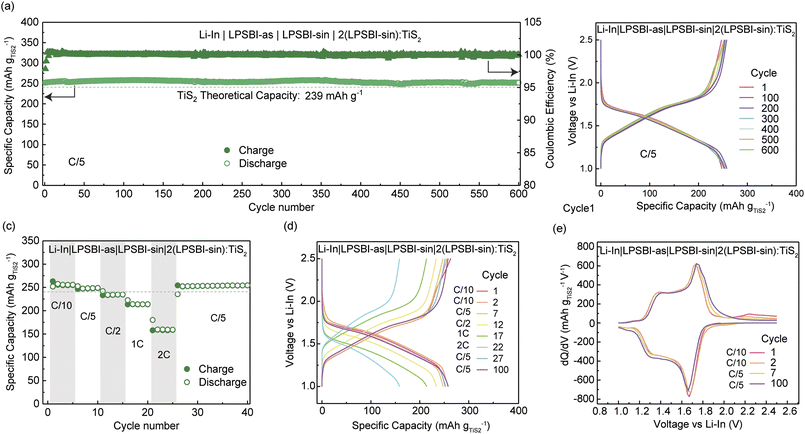
Owing to the better stability of LPSBI-as against Li-metal, and the higher CCD achieved with the sandwiched structure Li|LPSBI-as|LPSBI-sin|LPBSBI-as|Li, we assembled ASSB half-cells with two separator layers. A thin layer (25 mg) of LPSBI-as was placed towards the Li–In anode, while LPSBI-sin separator (75 mg) was used towards the cathode. For the catholyte, 2(LPSBI-sin):TiS2 was used. The performance of the cells showing high rate-capability and stable cycling is presented in Fig. 4. The first discharge capacity of 252 mAh g−1 and charge capacity of 253 mAh g−1 are achieved at C/5 at room temperature (Fig. 4a and b). As shown in Fig. 4a and b, a very high-capacity retention (>99%) after 600 cycles indicates the high stability and excellent performance of the engineered cell configuration (Li–In|LPSBI-as|LPSBI-sin|2(LPSBI-sin):TiS2). As shown in Fig. 4c, a high reversible capacity of ∼155 mAh g−1 is achieved at 2C, followed by excellent retention at C/5. Fig. 4d shows the voltage profiles of selected cycles, and Fig. 4e shows the differential capacity. The capacity from the Ti3+/4+ peak at ∼1.7 V vs. Li–In is prominent, as expected, in addition to another peak at higher voltage ( ∼2.25 V vs. Li–In), which can be assigned to I− oxidation.23
Structural characterization
X-ray diffraction
To determine the long-range structure, we performed powder X-ray diffraction measurements of the three model SEs. The diffraction patterns (Fig. 5) reveal low crystallinity, even for the low-temperature sintered LPSBI-sin. For LPS, small peaks of β-Li3PS4 can be identified. A slight trace of LiBr impurity is observed for both LPSBI-as and LPSBI-sin samples, while the LiI impurity is not visible in the powder XRD pattern. This suggests that both LiBr and LiI are incorporated into the LPS framework to get the new glass–ceramic phase, responsible for superionic conduction in LPSBI.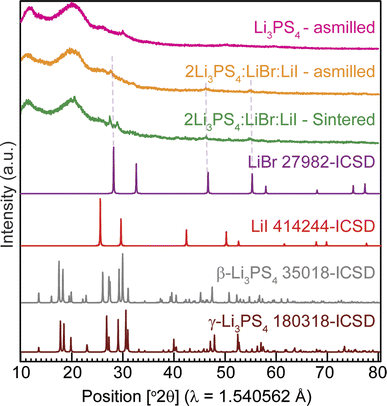 | ||
| Fig. 5 XRD patterns of LPS, LPSBI-as, and LPSBI-sin and comparison with reference patterns of β-Li3PS4, γ-Li3PS4, LiBr, and LiI from ICSD. | ||
Nuclear magnetic resonance (NMR) spectroscopy
Since the SEs of interest lack any long-range ordering evident from XRD, we examine the short-range structures using solid-state NMR (Fig. 6). Starting with LPS, the main phase, as determined from 31P MAS NMR spectra (Fig. 6a), is located at ∼83 ppm and assigned to a glassy PS43− unit.1,2,25 A secondary phase is also observed at ∼106 ppm, which is assigned to P2S64−.1,25 The 31P MAS NMR spectra are not significantly different for LPSBI-as compared to those of LPS. With the sintering, LPSBI-sin shows the formation of PS43−–Br−/I− at ∼81 and ∼78 ppm, along with the formation of P2S74−–Br−/I− at around ∼91 and ∼89 ppm.26–28 In addition, a trace amount of β-PS43− at ∼85.5 ppm and glassy-PS43− at ∼82.5 ppm are observed.1,2,25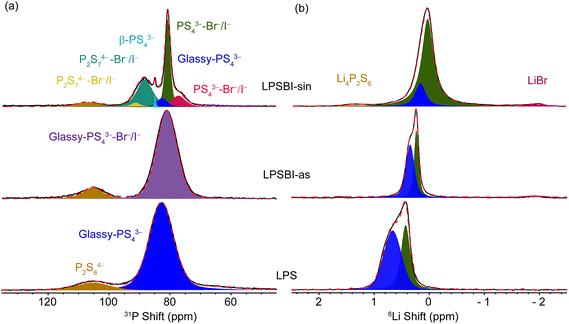 | ||
| Fig. 6 Local structural characterization of LPS, LPSBI-as, and LPSBI-sin solid electrolytes with (a) 31P and (b) 6Li MAS NMR. | ||
To understand the local lithium environment of LPS, LPSBI-as, and LPSBI-sin, we performed 6Li MAS NMR, as shown in Fig. 6b. We observed two distinct resonances corresponding to distinct lithium environments in all three electrolytes, consistent with previous reports.1,2 The variation in peak position and broadening suggests differences in the lithium local environments and dynamics. The resonance shown in blue (Fig. 6b) is predominant in LPSBI-as and LPS. The intensity of this resonance in LPSBI-sin was reduced significantly, making the resonance in green the dominant Li local environment. It has been reported that the Li site, as indicated by the green resonance (upfield), is more mobile than the blue one (downfield).1,2 With the incorporation of Br− and I− into the LPS framework, further changes in the lithium environment are indicated by shifts in the peaks towards lower ppm values.1
Raman spectroscopy
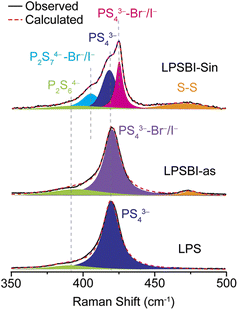
Raman spectroscopy is highly informative for understanding the connectivity of thiophosphate polyhedra (Fig. 7). For LPS, two components were fitted and assigned to glassy-PS43− and P2S64−. LPSBI-as shows no significant changes, but with slightly more P2S64− and a new peak at ∼475 cm−1 assigned to S–S stretching.27 With the sintering, LPSBI-sin shows a new peak around 405 cm−1 assigned to P2S74−–Br−/I−,26,27 in addition to the peaks from PS43− and P2S64−.NMR relaxometry
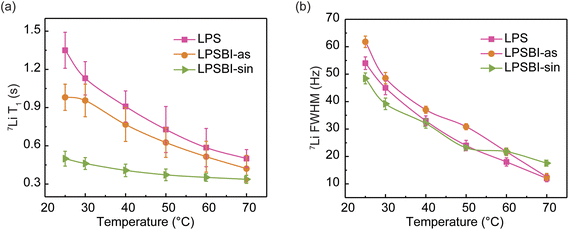
To probe the lithium-ion dynamics, variable-temperature 7Li MAS NMR spectra and T1 relaxation times were acquired. Representative spectra are presented in Fig. S13a, S14a and S15a. Fig. 8 presents the 7Li spin-lattice relaxation times (T1) and linewidths as a function of temperature. Fig. S13b, S14b and S15b present the variable-temperature 31P MAS NMR spectra, and Fig. S16 presents the 31P T1 alongside the line broadening of the SEs. With increasing temperature (Fig. 8b), motional line narrowing becomes evident in the 7Li MAS NMR spectra of LPS, LPSBI-as, and LPSBI-sin, originating from the rapid averaging of Li–Li dipolar interactions as Li-ion mobility increases at higher temperatures.29The 7Li T1 NMR relaxation time for LPS, LPSBI-as, and LPSBI-sin decreases with increasing temperature (Fig. 8a); therefore, a shorter T1 signifies faster Li+ motion. Notably, the shorter T1 for LPSBI-sin at room temperature compared to LPSBI-as and LPS indicates enhanced Li+ mobility for LPSBI-sin. This observation correlates with the trend in ionic conductivity. The 31P NMR spectrum of LPSBI-sin (Fig. S15b) reveals the presence of multiple distinct phosphorus local environments. As the temperature increases, the line width narrows, suggesting motional averaging. The decrease in T1 relaxation time of P2S74−–Br−/I− and PS43−–Br−/I− relative to PS43−, as observed in the 31P VT-NMR experiments, also suggests increased anion motion in LPSBI, which may facilitate fast Li+ ion conduction.30
Conclusion
In this work, Li3PS4 with halide incorporation, such as Br− and I−, has been shown to significantly enhance ion transport. LPSBI-sin exhibits an ionic conductivity of 4.36 mS cm−1 at 25 °C with an activation energy of 0.29 eV. Multinuclear solid-state NMR and Raman spectroscopy show a significant change in the Li+ local environment upon the incorporation of halides. With Br− and I− interspersed within the framework composed of PS43− and P2S74−, we observe enhanced dynamics of both Li+ and the anion matrix.Using a stacked cell with LPSBI-as and LPSBI-sin SEs shows improved cycling performance compared to single-layer SE-containing cells. It demonstrates how cell engineering with different SEs can boost the performance of ASSBs. A maximum critical current density of 0.92 mA cm−2 and a specific capacity of around 260 mAh g−1 are achieved using TiS2 as the CAM, showing high-rate performance and over 99% coulombic efficiency and very high-capacity retention (>99%) over 600 cycles. This work shows that the synergistic effects of mixing halides in Li3PS4 can significantly enhance Li+ dynamics and electrochemical performance.
Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: electrochemical characterization, electrochemical performance tests, structural characterizations, electron microscopy, X-ray diffraction, and variable-temperature NMR. See DOI: https://doi.org/10.1039/d6sc00740f.Acknowledgements
The authors would like to acknowledge the support from the National Science Foundation under grant no. DMR-2319151. All solid-state NMR experiments were performed at the National High Magnetic Field Laboratory, which is supported by National Science Foundation Cooperative Agreement No. DMR-1644779 and DMR-2128556*. T. P. Poudel and Y.-Y. Hu would like to thank Dr J. S. Raaj Vellore Winfred from the Materials Characterization Lab at the Department of Chemistry and Biochemistry at Florida State University for his help in acquiring the Raman data. The authors would like to thank Dr Xinsong Lin for his assistance with the lab powder X-ray characterization at the Department of Chemistry and Biochemistry at Florida State University.References
- T. P. Poudel, M. J. Deck, P. Wang and Y.-Y. Hu, Adv. Funct. Mater., 2024, 34, 2309656 CrossRef CAS.
- M. J. Deck, P.-H. Chien, T. P. Poudel, Y. Jin, H. Liu and Y.-Y. Hu, Adv. Energy Mater., 2024, 14, 2302785 CrossRef CAS.
- P. Wang, S. Patel, J. E. Roberts, B. E. Francisco and Y.-Y. Hu, ACS Mater. Lett., 2024, 6, 2059–2064 CrossRef CAS.
- H. Stöffler, T. Zinkevich, M. Yavuz, A. Senyshyn, J. Kulisch, P. Hartmann, T. Adermann, S. Randau, F. H. Richter, J. Janek, S. Indris and H. Ehrenberg, J. Phys. Chem. C, 2018, 122, 15954–15965 CrossRef.
- Z. Liu, W. Fu, E. A. Payzant, X. Yu, Z. Wu, N. J. Dudney, J. Kiggans, K. Hong, A. J. Rondinone and C. Liang, J. Am. Chem. Soc., 2013, 135, 975–978 CrossRef CAS PubMed.
- Y. Zeng, B. Ouyang, J. Liu, Y.-W. Byeon, Z. Cai, L. J. Miara, Y. Wang and G. Ceder, Science, 2022, 378, 1320–1324 CrossRef CAS PubMed.
- P. Minnmann, L. Quillman, S. Burkhardt, F. H. Richter and J. Janek, J. Electrochem. Soc., 2021, 168, 040537 CrossRef CAS.
- M. J. Deck and Y.-Y. Hu, J. Mater. Res., 2023, 38, 2631–2644 CrossRef CAS.
- S. V. Patel, S. Banerjee, H. Liu, P. Wang, P.-H. Chien, X. Feng, J. Liu, S. P. Ong and Y.-Y. Hu, Chem. Mater., 2021, 33, 1435–1443 CrossRef CAS.
- S. Ohno, C. Rosenbach, G. F. Dewald, J. Janek and W. G. Zeier, Adv. Funct. Mater., 2021, 31, 2010620 CrossRef CAS.
- G. F. Dewald, S. Ohno, M. A. Kraft, R. Koerver, P. Till, N. M. Vargas-Barbosa, J. Janek and W. G. Zeier, Chem. Mater., 2019, 31, 8328–8337 CrossRef CAS.
- T. P. Poudel, E. Truong, I. P. Oyekunle, M. J. Deck, B. Ogbolu, Y. Chen, P. K. Ojha, T. N. D. D. Gamaralalage, S. V. Patel, Y. Jin, D. Hou, C. Huang, T. Li, Y. Liu, H. Xiong and Y.-Y. Hu, ACS Energy Lett., 2025, 10, 40–47 CrossRef CAS.
- T. P. Poudel, I. P. Oyekunle, M. J. Deck, Y. Chen, D. Hou, P. K. Ojha, B. O. Ogbolu, C. Huang, H. Xiong and Y.-Y. Hu, Chem. Sci., 2025, 16, 2391–2401 RSC.
- I. P. Oyekunle, E. Truong, T. P. Poudel, Y. Chen, Y. Jin, I. A. Ojelade, M. J. Deck, B. Ogbolu, M. M. Islam, P. K. Ojha, J. S. R. V. Winfred, D. Hou, H. Xiong, C. Huang and Y.-Y. Hu, Chem. Sci., 2025, 16, 10372–10385 RSC.
- R. B. Nuernberg, Ionics, 2020, 26, 2405–2412 CrossRef CAS.
- S. V. Patel, E. Truong, H. Liu, Y. Jin, B. L. Chen, Y. Wang, L. Miara, R. Kim and Y.-Y. Hu, Energy Storage Mater., 2022, 51, 88–96 CrossRef.
- I. Hanghofer, M. Brinek, S. L. Eisbacher, B. Bitschnau, M. Volck, V. Hennige, I. Hanzu, D. Rettenwander and H. M. R. Wilkening, Phys. Chem. Chem. Phys., 2019, 21, 8489–8507 RSC.
- B.-Y. Chang, J. Electrochem. Sci. Technol., 2020, 11, 318–321 CrossRef CAS.
- R. Schlem, A. Banik, S. Ohno, E. Suard and W. G. Zeier, Chem. Mater., 2021, 33, 327–337 CrossRef CAS.
- Y. Lu, C. Zhao, H. Yuan, X. Cheng, J. Huang and Q. Zhang, Adv. Funct. Mater., 2021, 31, 2009925 CrossRef CAS.
- F. Han, Y. Zhu, X. He, Y. Mo and C. Wang, Adv. Energy Mater., 2016, 6, 1501590 CrossRef.
- S. Banerjee, M. L. H. Chandrappa and S. P. Ong, ACS Appl. Energy Mater., 2022, 5, 35–41 CrossRef CAS.
- B. T. Leube, C. Robert, D. Foix, B. Porcheron, R. Dedryvère, G. Rousse, E. Salager, P.-E. Cabelguen, A. M. Abakumov, H. Vezin, M.-L. Doublet and J.-M. Tarascon, Nat. Commun., 2021, 12, 5485 CrossRef CAS PubMed.
- A. Shastri, N. Rons, Q.-P. Ding, S. J. Kmiec, M. Olson, Y. Furukawa and S. W. Martin, Solid State Ionics, 2023, 402, 116363 CrossRef CAS.
- X. Chi, Y. Zhang, F. Hao, S. Kmiec, H. Dong, R. Xu, K. Zhao, Q. Ai, T. Terlier, L. Wang, L. Zhao, L. Guo, J. Lou, H. L. Xin, S. W. Martin and Y. Yao, Nat. Commun., 2022, 13, 2854 CrossRef CAS PubMed.
- R. Rajagopal, Y. Subramanian and K.-S. Ryu, ACS Sustainable Chem. Eng., 2023, 11, 2692–2703 CrossRef CAS.
- A. Kato, M. Yamamoto, F. Utsuno, H. Higuchi and M. Takahashi, Commun. Mater., 2021, 2, 1–8 Search PubMed.
- H. Tsukasaki, S. Mori, S. Shiotani and H. Yamamura, Solid State Ionics, 2018, 317, 122–126 CrossRef CAS.
- B. B. Duff, S. J. Elliott, J. Gamon, L. M. Daniels, M. J. Rosseinsky and F. Blanc, Chem. Mater., 2023, 35, 27–40 CrossRef CAS PubMed.
- P. Wang, H. Liu, S. Patel, J. E. Roberts, Y. Chen, B. Ogbolu, B. E. Francisco and Y.-Y. Hu, Adv. Energy Mater., 2024, 14, 2401549 CrossRef CAS.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |