Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Recent advances in MOF-based single-atom photocatalysts for CO2 to solar fuel conversion under sunlight irradiation
Adnan
Majeed
 ,
Minh-Khoa
Duong
,
Van-Duc
Nguyen
,
Minh-Khoa
Duong
,
Van-Duc
Nguyen
 and
Trong-On
Do
and
Trong-On
Do
 *
*
Department of Chemical Engineering, Laval University, 1065 Avenue de la Médecine, Quebec, QC G1V 0A6, Canada. E-mail: Trong-On.Do@gch.ulaval.ca
First published on 27th March 2026
Abstract
Solar-driven photocatalytic CO2 conversion offers a sustainable route to address energy demand and carbon emissions. Metal–organic frameworks (MOFs) have emerged as promising photocatalyst platforms due to their tunable structures and well-defined coordination environments, while covalent organic frameworks (COFs) also offer ordered porosity and tunable electronic structures that support single-atom catalysts. Incorporation of single-atom active sites in MOFs and COFs further enhances site utilization, charge separation, and photocatalytic performance. This review offers a unified perspective on MOF-based single-atom photocatalysts for CO2 conversion by focusing on the synergistic interaction between atomically dispersed metal sites and MOFs. Unlike previous reports, it systematically compares different single-atom incorporation strategies and directly correlates atomic coordination environments with photocatalytic performance. By linking the structure, coordination, and activity, this work provides clear design guidelines for developing efficient and durable solar-driven CO2 reduction systems.
1 Introduction
Due to rapid growth and increasing industrialization, a serious energy crisis has emerged, leading to widespread water,1,2 soil,3,4 and air5,6 pollution.7 The energy demand is expected to escalate slightly; it will double by 2050 and triple before the end of this century.8,9 Such growth will place enormous strain on existing energy systems. Although fossil fuels contribute heavily to meeting the fuel requirements of the world, their continuous use contributes largely to rising global warming.10 Moreover, owing to the ample use of mineral fuel, greenhouse gases such as carbon dioxide (CO2) have escalated significantly.11 The International Energy Agency believed that CO2 emissions from energy production would peak in about 2025 based on the recent publication of World Energy Outlook 2023.12,13 Despite the projected peak, atmospheric CO2 concentrations continued to increase, surpassing 430 ppm by the end of April 2025, with an estimated growth rate of more than 2 ppm per year.4,14,15 This rapid rise in CO2 plays a significant role in enhancing the process of global warming and other natural disasters like the melting of polar ice, acidification of the oceans, and climate instability.16,17 Consequently, the urgent reduction of atmospheric CO2 concentration has become a critical global priority.18 To address this dilemma, much effort has been invested to establish efficient CO2 transformation and reduction methods, i.e., thermocatalysis,19 electrocatalysis,20 and photocatalysis.22The reduction of CO2 itself is quite difficult because of the large amount of energy needed to overcome the extremely stable C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond with a bond energy of around 805 kJ mol−1.21 There are many methods for the reduction of CO2, but using a photocatalyst is quite appealing due to an eco-friendly process, in which CO2 can be reduced to various value-added products22 such as HCOOH,23 CO,24 CH4,25 CH3OH,26etc. The use of sunlight is especially promising, as it is abundant and readily available.27 In nature, photosynthesis is a well-known biological process that reduces CO2 under sunlight, converting it into chemical forms. In photocatalytic reduction reactions, the reduced product can be C1 or C2+ molecules, as presented in Fig. 1.28 Each molecule faces its own thermodynamic and kinetic barriers during reduction.29 The process is quite efficient depending upon the nature of the photocatalyst used, which may increase the yields.30 The most important factors are the efficiency of absorption of photons and the transfer characteristics of charges in the photocatalyst.31 Despite major efforts to optimize these factors, the overall activity of photocatalytic materials remains insufficient to meet the growing demands of the environmental and energy sectors.32 Consequently, for the evolution of novel photocatalytic materials, it would be imperative to combine high absorption and efficient charge separation.33,34
O bond with a bond energy of around 805 kJ mol−1.21 There are many methods for the reduction of CO2, but using a photocatalyst is quite appealing due to an eco-friendly process, in which CO2 can be reduced to various value-added products22 such as HCOOH,23 CO,24 CH4,25 CH3OH,26etc. The use of sunlight is especially promising, as it is abundant and readily available.27 In nature, photosynthesis is a well-known biological process that reduces CO2 under sunlight, converting it into chemical forms. In photocatalytic reduction reactions, the reduced product can be C1 or C2+ molecules, as presented in Fig. 1.28 Each molecule faces its own thermodynamic and kinetic barriers during reduction.29 The process is quite efficient depending upon the nature of the photocatalyst used, which may increase the yields.30 The most important factors are the efficiency of absorption of photons and the transfer characteristics of charges in the photocatalyst.31 Despite major efforts to optimize these factors, the overall activity of photocatalytic materials remains insufficient to meet the growing demands of the environmental and energy sectors.32 Consequently, for the evolution of novel photocatalytic materials, it would be imperative to combine high absorption and efficient charge separation.33,34
 | ||
| Fig. 1 Schematic representation of the possible photocatalytic CO2 reduction pathways producing C1 and C2+ products. Redrawn using ChemDraw.28,29,35 | ||
Single-atom catalysts (SACs) have recently attracted much interest as highly promising photocatalysts owing to their distinctive atomic properties, such as maximum metal utilization efficiency and high catalytic activity.36,37 Single atoms (SAs) in the photocatalytic system can also create isolated reaction sites, which can not only provide more active sites but also increase light harvesting and charge separation.38 These findings collectively confirm that isolated metal sites enhance both light absorption and charge transfer, which are crucial for high-performance SA photocatalysis. In addition, their coordinatively unsaturated configuration provides well-defined active sites for reactant adsorption and activation. Such features enable improved photocatalytic performance while retaining high atomic utilization efficiency.39 SAC models were first developed by Zhang et al. to describe the high activity and stability of isolated Pt atoms on iron oxides, attributable to metal-support interactions.40,41 The unsaturated sites, quantum size effects, and strong metal-support interactions of atomic-sized SACs enable catalytic activity similar to that of homogeneous catalysts and heterogeneous stability.42,43 However, it should be pointed out that atomic-scale metal dispersal could raise surface free energy owing to the inherent metallic atom clustering nature.44,45 Hence, the process of immobilizing individual atoms on practical surfaces is now a current research field of interest in chemistry, environmental science, and materials science.46 Therefore, supporting SAs on carefully designed substrates is crucial to fully exploit the catalytic capacity of SACs.47,48
In recent years, metal–organic frameworks (MOFs) have emerged as highly effective supports for SACs because of their structural flexibility and synergistic catalytic properties.49 The high density of coordination sites within MOF architectures allows SACs to be anchored at fixed positions, effectively preventing atom migration and aggregation.50,51 Supporting SACs on MOFs not only reduces aggregation but also promotes synergistic catalysis in the composite material.52 Consequently, SACs derived from MOFs provide an efficient bridge between homogeneous and heterogeneous catalysis,53 as illustrated in Fig. 2. In addition to MOFs, covalent organic frameworks (COFs) have recently attracted attention as alternative platforms for single-atom photocatalysts due to their well-defined porous structures, crystallinity, and tunable π-conjugated backbones. Their predesignable architectures enable precise control over light absorption, electronic structure, and charge separation behavior, making them suitable for stabilizing isolated metal sites for photocatalytic CO2 reduction.54 The incorporation of single metal atoms into COFs not only facilitates CO2 activation but also promotes efficient electron transfer from the conjugated network to the catalytic centers. Moreover, the photocatalytic performance of COFs can be systematically tuned through the selection of precursors, linkage chemistry, framework topology, and the nature of the anchored metal atoms.55 However, despite these advances, MOF-based systems remain the primary focus of this review due to their diverse coordination environments and well-established structural tunability. Nonetheless, recent reviews have highlighted some progress in MOF-based photocatalysts for CO2 reduction. However, limited attention has been given to MOF-based SACs (MOF@SACs), including both MOF-supported and MOF-derived structures, in the context of photochemical energy conversion. Building on our previous review,18 this work strives to illustrate structure–activity relationships and outline future directions for MOF-based single-atom catalysts toward efficient and sustainable solar-driven applications and product conversion. This review is solely focused on the reduction of CO2 by MOF@SACs.
2 Fundamentals of MOF-based single-atom photocatalysts for CO2 reduction
In MOF-based single-atom photocatalysts, this process is governed by the synergistic interplay between the light-harvesting framework and atomically dispersed metal active sites (Fig. 3),57 which facilitate efficient charge transfer and suppress recombination.56 The MOF-based photocatalytic CO2 reduction reaction is catalyzed by a series of elementary reactions, similar to those in heterogeneous catalysis that involve the adsorption of CO2, surface reactions by charge transfer, and desorption of products.57,58 CO2 adsorption is a step that cannot be underestimated, because the linear geometry of the molecule and the high thermodynamic stability of CO2, which is due to strong C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O double bonds, make the activation of CO2 intrinsically hard.59,60 The well-defined pores, high surface area, and adjustable chemical environment in MOF-based photocatalysts help enrich CO2 around active sites to reduce diffusion barriers and maximize local reactant concentration.61,62 A CO2 activation reaction has to ensure that the conduction band edge of the photocatalyst is more negative than the CO2 reduction potential, whereas the valence band edge is sufficiently positive to maintain water oxidation.63,64 Proper band alignment is therefore essential to ensure that photogenerated carriers possess adequate driving force to initiate and sustain redox reactions.65 The activation of CO2 at the reaction interface typically occurs via interaction with *CO2 (commonly referred to as the rate-limiting step), which has a very negative reduction potential.66 The molarity of many photocatalysts is insufficient to activate this one-electron step effectively; therefore, proton-assisted multi-electron transfer mechanisms are preferred, which decrease the energy barrier for the activation of CO2.67 Subsequent reaction pathways diverge toward the formation of C1 or C2+ products (Fig. 1). C1 products typically originate from key intermediates such as *COOH and *OCHO, whereas C2+ products require C–C coupling steps involving species such as *CO, *CHO, or *COCO.68,69 Even though C2+ products are more energy-rich and possess more practical utility, their production is kinetically more difficult and necessitates the fine control of intermediate adsorption strength, surface mobility, and coupling probability.64,70
O double bonds, make the activation of CO2 intrinsically hard.59,60 The well-defined pores, high surface area, and adjustable chemical environment in MOF-based photocatalysts help enrich CO2 around active sites to reduce diffusion barriers and maximize local reactant concentration.61,62 A CO2 activation reaction has to ensure that the conduction band edge of the photocatalyst is more negative than the CO2 reduction potential, whereas the valence band edge is sufficiently positive to maintain water oxidation.63,64 Proper band alignment is therefore essential to ensure that photogenerated carriers possess adequate driving force to initiate and sustain redox reactions.65 The activation of CO2 at the reaction interface typically occurs via interaction with *CO2 (commonly referred to as the rate-limiting step), which has a very negative reduction potential.66 The molarity of many photocatalysts is insufficient to activate this one-electron step effectively; therefore, proton-assisted multi-electron transfer mechanisms are preferred, which decrease the energy barrier for the activation of CO2.67 Subsequent reaction pathways diverge toward the formation of C1 or C2+ products (Fig. 1). C1 products typically originate from key intermediates such as *COOH and *OCHO, whereas C2+ products require C–C coupling steps involving species such as *CO, *CHO, or *COCO.68,69 Even though C2+ products are more energy-rich and possess more practical utility, their production is kinetically more difficult and necessitates the fine control of intermediate adsorption strength, surface mobility, and coupling probability.64,70
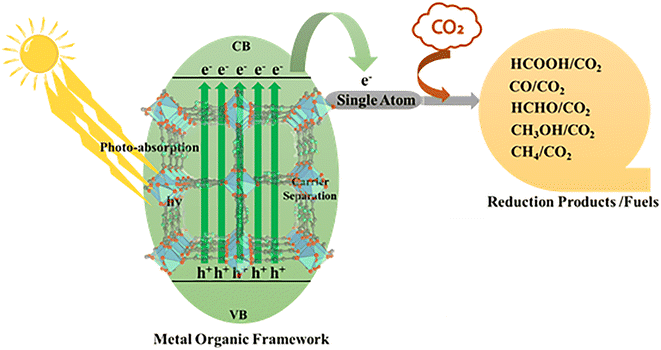 | ||
| Fig. 3 Schematic of MOF-based single-atom photocatalytic CO2 reduction under solar light irradiation. | ||
The single-atom (SA) active sites attached to MOFs are important in controlling such elementary steps.46,71 They have the benefit of allowing the control of the local electronic structure and coordination environment with a high level of precision, which directly affects the CO2 adsorption strength, the stability of intermediates, and charge-transfer kinetics.72 Besides, the charge transfer of SAs with the MOF host using metal–ligands can create new electronic states to absorb more visible light and improve the separation of charge carriers generated by photoexcitation.73 Notably, the atomic dispersion of the metal center optimizes the utilization of the metal and avoids the formation of metallic clusters, which are likely to be recombination centers.74,75 These properties enable MOF-based single-atom (MOF@SA) photocatalysts to act as highly efficient platforms towards selective CO2 activation and conversion.76
The porous structure of MOFs offers spatial confinement effects that can stabilize chemical intermediates and encourage particular reaction routes in addition to electrical regulation. Fine control over reaction kinetics and product selectivity is made possible by the diffusion of reactants and intermediates within MOF channels and customized coordination environments surrounding SAs.77,78 However, excessive intermediate binding can delay product desorption and block sites, emphasizing the significance of balancing adsorption and desorption.79 Thus, SA photocatalysts based on MOFs have to be rationally designed, that needs to take into account light absorption, charge separation, band alignment, active-site accessibility, and intermediate binding strength.80 Through combining tunable MOF structured frameworks with atomically dispersed metal sites, the systems offer an unprecedented platform to resolve the inherent drawbacks of CO2 activation, rejection of charge recombination, and reaction directionalities to products of interest.81 The knowledge of these core concepts forms a good basis for rational development of next-generation MOF@SA photocatalysts, which are effective in reducing CO2 under sunlight. SA photocatalysts based on MOFs have clear benefits over traditional ones due to the ability to control the binding energy of the reaction intermediate by the number of metal atoms in the crystal, which increases the catalytic efficiency. Nonetheless, overly powerful binding of intermediates is counterproductive because it prevents the desorption of products and reduces the turnover. The development of an optimal, moderate binding strength in MOF@SACs architectures, thus, is a major challenge.
3 Synthetic strategies for single-atom incorporation in MOFs
Both homogeneous and heterogeneous catalysts can be used in both industrial practice and fundamental research. In homogeneous catalysis, the catalyst and the reactants are in the same phase, which makes the active site well-defined and the reaction mechanism clear; however, poor stability and separation make such an application not practicable.82,83 In contrast, heterogeneous catalysts are more stable and easily recoverable; but in many cases, their catalytic activity is complicated by surface heterogeneity. This heterogeneity is due to different atomic structures, defects, and variation of particle sizes.84 It is so complex that it is difficult to spot the same active sites, and comparison of catalytic performance is then not reliable. In order to overcome these shortcomings, converting metal nanoparticles into single-atom catalysts (SACs) has been a viable approach.85 With the combination of molecular-level specificity of homogeneous catalysts and the stability and reusability of heterogeneous systems, SACs allow customizable reactivity, enhanced metal productivity, and atomic-scale photocatalysis.86 Overall, recent years have seen massive progress in the field of synthetic chemistry, which has greatly increased the pace at which SA catalysts can be developed.43,71,87,88 These active sites have a high inherent surface energy and therefore can migrate and cluster or form nanoparticles during synthesis and further processing due to a large quantity of active sites per mole compared to bulk materials.89Supported catalysts often have a blend of metal nanoparticles and atomically dispersed metal atoms, and high and uniform loadings of active sites are hard to reach.90 Scientists have thus concentrated on synthetic techniques, which maintain the distant togetherness of metal precursors, avoid agglomeration of these during thermal or chemical treatments, and provide sufficient anchoring spots to retain solitary metal atoms.91 The introduction of MOFs as host matrices with added guest species is an effective way of increasing the charge-carrier separation in photocatalysis. This is enhanced by the interfacial interaction or heterojunctions, which develop internal electric fields that facilitate effective separation and transport of charges. MOF-derived carbon supports are promising when constructed on mixtures of MOFs and porous crystalline materials.92 They are self-assembled using metal ions/clusters with organic linkers, thereby offering high surface area, defined pores, tunable chemistry, and numerous coordination sites.93 MOF-based carbon materials have been demonstrated to be superior SAC supports, which enhance catalyst stability, ease recovery, and allow SACs with homogeneous and heterogeneous catalysis to be combined.41,94
Four major methods are used to produce MOF@SA catalysts: intrinsic linker stabilization, post-synthesis ligand modification, metal node stabilization, and nanopore confinement.95 SAs may be immobilized through coordination with metal nodes or organic ligands, whereas confinement within MOF pores further suppresses aggregation by physically separating metal precursors of appropriate size. The combination of these techniques enables this to be controlled in terms of atom dispersion, stability, and catalysis.96 Heterogeneous catalysis is said to involve coordinatively unsaturated metal sites in MOFs, which are considered to be the chief active centers. Their electrophilic nature imparts Lewis acidity, enabling efficient electron acceptance and favorable interactions with nucleophilic or reactive species, thereby enhancing catalytic performance.97 Other sites that can be post-synthetically modified to add more catalytic centers are provided by unsaturated metal ions or cluster nodes.98 This is particularly useful with MOF-based photocatalysis, in which it enhances light absorption, charge separation, and surface redox reactions. The use of these unsaturated metal nodes provides an opportunity to allow isolated metal atoms to increase the catalytic ability of MOFs.99 Node-anchored species can remain stably dispersed at the atomic level without aggregating into nanoparticles, due to strong coordination bonds between the SAs and the inorganic metal clusters.100 This anchoring strategy also optimizes the local environment of the active sites, thereby facilitating the photocatalytic activation of CO2.81,101 Several strategies have been developed to modulate the local structure of SA active sites and evaluate their catalytic performance for CO2 conversion. These approaches include the design of molecular catalysts, carbonization of functionalized precursors, and tuning the coordination number of metal centers supported on MOFs.102 The coordination environment of SAs plays a crucial role in determining the catalytic activity of a material.103 When SAs are anchored between the layers of a catalyst, they can promote charge separation and enable efficient interlayer charge transfer, which improves overall catalytic performance.104
3.1 Metal node anchoring strategy
Metal node anchoring represents an effective strategy for stabilizing single atoms in MOFs, as summarized in recent studies on MOF-based single-atom catalysts.105–107 Metal clusters at MOF nodes often contain coordinatively unsaturated sites terminated by inorganic ligands such as –OH or –OH2. These oxygen-containing groups provide strong coordination environments for anchoring isolated metal atoms in the node region rather than on organic linkers. Abdel-Mageed and co-workers demonstrated that Cu single atoms can be anchored at defective Zr6-oxo nodes of UiO-66 through coordination with terminal –OH or –OH2 ligands. Spectroscopic and microscopic characterization confirmed that the Cu species remain atomically dispersed without aggregation, resulting in fully exposed and stable active sites108 (Fig. 4). Geng and collaborators utilized a node metal ion-exchange strategy that has been demonstrated in ZIF-8, where partial replacement of Zn2+ ions by Sn2+ ions leads to the formation of isolated Sn species at the metal nodes, while retaining the structural integrity of the framework.109 | ||
| Fig. 4 (a) Crystal structure of defect-free UiO-66 (yellow spheres indicate pore space). (b) Zr6-oxide cluster showing defect sites capped by –OH/–OH2 and acetate groups (C, black; O, red; Zr, blue; H are omitted). (c) PXRD patterns of UiO-66 and Cu/UiO-66 compared with the simulated UiO-66 pattern, confirming framework integrity. (d) DRIFTS spectra of UiO-66 and Cu/UiO-66 showing changes in –OH vibrations after Cu anchoring. Reproduced with permission from ref. 108. (Copyright 2019 American Chemical Society). (e) Schematic preparation of Sn-doped ZIF-8. (f) Sn 3d XPS spectra of Sn-ZIF-8 with different tin contents. Reproduced with permission from ref. 109. (Copyright 2020 Wiley-VCH). | ||
Zhu Gao and co-workers developed an in situ co-doping strategy for MIL-125(Ti) by introducing Zn(II) and pyrrolic N during hydrothermal synthesis. Zn partially substituted Ti at the metal nodes, generating non-equivalent active sites, while pyrrolic N coordinated with Ti clusters to form Ti–N bonds. XPS confirmed Ti3+ formation, oxygen vacancy generation, and N–Ti coordination (Fig. 5a–d). A slight reduction in surface area indicated node-related defects. The node-anchored Zn and N species improved charge separation and transfer, as evidenced by reduced PL intensity, lower charge-transfer resistance, increased photocurrent, and stronger ESR signals. As a result, MIL-125(N–Ti9Zn1) exhibited a photocatalytic CH3CHO degradation rate nearly ten times higher than that of pristine MIL-125(Ti), demonstrating the effectiveness of metal-node anchoring in enhancing photocatalytic performance. Fig. 5e shows the schematic illustration of photogenerated charge separation and band-structure modulation induced by Zn and N doping at the MOF metal nodes.110
 | ||
| Fig. 5 (a) XPS survey spectra of MIL-125(Ti), MIL-125(Ti9Zn1), and MIL-125(N–Ti9Zn1). High-resolution (b) Ti 2p and (c) O 1s XPS spectra of MIL-125(Ti) and MIL-125(N–Ti9Zn1) and (d) N 1s XPS spectra of MIL-125(N–Ti9Zn1). (e) Schematic illustration of photogenerated charge separation and band-structure modulation induced by Zn and N doping at the MOF metal nodes. Reproduced with permission from ref. 110. (Copyright 2020 Elsevier). | ||
In another study, the Di Chen group introduced photo-deposition as an effective strategy for incorporating single-atom sites into MOFs, particularly Zr-based MOFs such as UiO-66 NU-1000, and MOF-808, which possess coordinatively unsaturated Zr6-oxo nodes terminated by –OH/–OH2 ligands. Under light irradiation, metal precursors (e.g., Cu2+, Fe3+, Ni2+, and Au3+) can be reduced and stabilized as atomically dispersed species at the metal nodes, typically with loadings below 1 wt%, as confirmed by HAADF-STEM (Fig. 6). The high surface area (often >2000 m2 g−1) and mesoporous channels (∼2–3 nm) of these MOFs further suppress aggregation and enable the confined integration of single atoms and atomically modified metal nanoparticles in proximity. This node-anchored photodeposition approach provides precise control over atomic dispersion and multisite construction, offering a versatile synthetic pathway for MOF-based single-atom photocatalysts.111 Youssef et al. reported a method for incorporating SACs into MOFs via surface functionalization and postmetalation. UiO-66 was first modified with a silane-based photoinitiator, followed by surface-initiated photopolymerization of bipyridine derivatives to form a polymer shell. Postmetalation with Re(CO)5Cl generated well-dispersed rhenium single-atom sites anchored on the MOF surface. The HAADF-STEM and EDX analyses confirmed the formation and uniform distribution of the SA shell around the UiO-66 particles (Fig. 7A–C). This hybrid material retained the MOF's structural integrity and porosity (Fig. 7D and E), showed enhanced CO2 adsorption and charge separation, and exhibited high photocatalytic activity, highlighting the potential of surface-initiated polymerization and postmetalation for preparing efficient single-atom MOF catalysts.112
 | ||
| Fig. 6 (a) Schematic of the synthesis of (AuCu-NU-1000) ACN, showing CO2 conversion sites. (b) SEM image of ACN. (c) HAADF-STEM image of ACN with a zoomed-in view of a single nanoparticle. (d) HAADF-STEM of an enlarged nanoparticle with elemental maps of Au, Cu, and their overlap. (e) Schematic of the overall catalytic mechanism of a single-atom-based MOF. Reproduced with permission from ref. 111. (Copyright 2025 American Chemical Society). | ||
 | ||
| Fig. 7 (A) HAADF-STEM of UiO-66-poly[Re] showing a 15 nm poly[Re] shell. (B) Zoomed view of the shell. (C) EDX elemental maps for N (red), O (yellow), Zr (green), and Re (blue). (D) Photopolymerization of A2bpy on UiO-66-SPI. (E) Functionalization of UiO-66-poly[bpy] with Re(CO)5Cl to form UiO-66-poly[Re]. Reproduced with permission from ref. 112. (Copyright 2025 American Chemical Society). | ||
3.2 MOF-derived carbon-based single-atom catalysts
The preparation of MOF-derived carbon supports involves the synthesis of suitable MOF precursors followed by controlled pyrolysis under a defined atmosphere. During thermal treatment, organic ligands decompose to form a porous carbon matrix, while coordinated metal centers evolve into atomically dispersed M–Nx active sites.113 The pyrolysis temperature plays a critical role: moderate temperatures enhance graphitization and conductivity, whereas excessively high temperatures may reduce heteroatom content (e.g., nitrogen) and promote metal aggregation. Therefore, optimization of carbonization temperature is essential to balance conductivity and active site preservation. For Zn-containing MOFs, temperatures above 900 °C are typically used to remove Zn species and generate metal-free porous carbon. In contrast, transition-metal-based systems are commonly carbonized at 700–1000 °C to stabilize isolated metal atoms (e.g., Fe–N4, Ni–N4, and Pt–N4) within nitrogen-doped carbon frameworks. The carbonization atmosphere also influences the final structure: inert gases (Ar or N2) prevent oxidation, while NH3 can enhance nitrogen doping and increase coordination sites.114,115 Using MOF-derived carbon-based SACs has emerged as a dominant strategy for constructing highly stable and conductive SACs. They effectively overcome the structural instability of pristine MOFs at high temperatures and in harsh chemical environments.53,116 As a result, they exhibit enhanced stability and promising catalytic performance. Their morphology and pore structure can be precisely controlled through MOF precursor design, and the coordination environment of metal sites can be tuned via linkers and pyrolysis conditions. The inherent porous architecture provides high surface area and efficient mass and charge transport, while MOF cavities and unsaturated sites enable construction of well-defined single-atom active centers.113 Additionally, in situ characterization is needed to accurately probe active sites under working conditions and guide the design of more efficient MOF-derived SACs.117Biao Hu et al. demonstrated a MOF-derived strategy for incorporating single and dual metal atoms into a photocatalytic carbon nitride matrix. Using NH2-MIL-68(In) as a precursor, atomically dispersed Co and In sites were successfully anchored on a CN support via a straightforward pyrolysis approach. High-resolution HAADF-STEM, EXAFS, and XANES analyses confirmed the formation of Co–N4 and In–N5 coordination structures without nanoparticle aggregation (Fig. 8). The Co1In1 system derived from MOF precursors exhibits enhanced photocatalytic performance, where the MOF structure enables uniform dispersion of dual-metal sites and facilitates efficient charge transfer.118
 | ||
| Fig. 8 (a) Schematic of Co1In1/CN synthesis. (b) TEM, (c) HAADF-STEM with elemental mapping, (d) AC-STEM, and (e) corresponding atomic intensity profiles. Reproduced with permission from ref. 118. (Copyright 2025 Elsevier). | ||
3.3 Ultrathin MOF nanosheet engineering
The ultrathin nanosheets of MOFs have open pores on both sides, high accessibility of active sites, and shorter pathways for charge diffusion.119 Quan Zuo and colleagues reported an effective strategy for stabilizing single atoms using ultrathin MOF nanosheets as supports.120 Compared with bulk MOFs, two-dimensional nanosheets provide open pores on both sides,121 enabling high SA loading and improved contact with CO2 molecules, while the ultrathin thickness shortens charge diffusion pathways and suppresses electron–hole recombination.122 Through a bottom-up synthetic approach, Co single atoms were pre-coordinated within porphyrin linkers and assembled into ultrathin MOF nanosheets (Co-MNSs), avoiding the low yield and complexity of exfoliation methods (Fig. 9a). The resulting Co-MNSs exhibited an ultrathin thickness of ∼2.4 nm with micron-sized lateral dimensions (Fig. 9b–d), delivering abundant exposed active sites. As a result, Co-MNSs achieved efficient visible-light-driven CO2-to-CO conversion with high activity and selectivity, demonstrating the advantages of ultrathin MOF nanosheets for single-atom photocatalysis.120 | ||
| Fig. 9 (a) Schematic of ultrathin Co single-atom MOF nanosheet synthesis. (b and c) SEM and TEM images. (d) AFM height profile. (e) HAADF-STEM image with EDS elemental mapping. Reproduced with permission from ref. 120. (Copyright 2023 Springer Nature). | ||
3.4 Pre-synthetic incorporation strategies
Pre-synthetic incorporation of single-atom catalysts into MOFs allows uniform atomic dispersion and strong coordination with organic linkers or metal nodes, effectively suppressing aggregation and improving catalytic performance for CO2 reduction. Unlike post-synthetic methods, the Shankar group introduced metal precursors during MOF formation, enabling better control over the coordination environment and electronic structure. In that study, electrodeposition-assisted MOF growth was achieved, in which Fe single atoms are incorporated onto carbon-modified nickel nanosheets (C–Ni NSs) through an electrochemical process. In this method, Fe3+ ions are reduced to Fe2+ in the presence of benzenetricarboxylate (BTC3−), promoting MOF growth directly on the electrode surface, as confirmed by various characterization techniques (Fig. 10a–f). The in situ coordination stabilizes isolated metal sites and strengthens metal-support interactions, resulting in durable and efficient catalytic centers, suggesting the potential of this approach for designing MOF-based SACs for CO2 reduction.123 | ||
| Fig. 10 SEM (a), TEM (b), and high-resolution TEM (c) images of the catalyst; the inset highlights the dispersed Fe single atoms on the C–Ni nanosheets. Elemental mapping (d) and the corresponding EDX spectrum (e) confirm the elemental distribution of the Fe-SACs/C–Ni@3 electrode along with the Fe 2p spectrum and (f) a schematic illustration of the pre-synthetic electrodeposition strategy used for MOF-based single-atom catalysts. Reproduced with permission from ref. 123. (Copyright 2024 Royal Society of Chemistry). | ||
3.5 Linker engineering and metallation
In addition to metal clusters, the organic linkers in MOFs possess strong anchoring ability and can effectively coordinate with single-atom metal sites.100 Porphyrins, which contain four pyrrolic nitrogen atoms arranged in square-planar or tetrahedral geometries, are especially effective for trapping metal SAs through coordination interactions arising from their lone-pair electrons.124 Zhang et al. reported that exciton migration in natural photosynthesis mainly occurs in highly ordered porphyrin-like pigments. Inspired by this, MOF-525, with the formula Zr6O4(OH)4(TCPP-H2)3 was synthesized. This MOF integrates Zr6 clusters and porphyrin units into a 3D network. Coordinatively unsaturated Co sites were introduced into the porphyrin units to form MOF-525-Co (Fig. 11a). In this structure, each Co active site is simultaneously exposed to CO2 molecules while being spatially isolated, preventing aggregation. These unsaturated Co sites act as highly efficient catalytic centers and enhance CO2 adsorption through open Co porphyrin sites, enabling effective CO2 activation. Moreover, the presence of Co promotes directional energy migration within the MOF, significantly suppressing electron–hole recombination and providing long-lived electrons for the reduction of adsorbed CO2 molecules.51 | ||
| Fig. 11 (a) 3D network of MOF-525-Co, showing its highly porous framework with incorporated single Co active sites. Reproduced with permission from ref. 51. (Copyright 2016 Wiley-VCH). (b) Schematic illustration of PCN-808-BDBR synthesis, where [Ru(bpy)3]2+ active sites are installed on the organic linkers, forming a stable Zr6-based MOF with mesopores. Reproduced with permission from ref. 126. (Copyright 2020 American Chemical Society). (c) Schematic illustration of the formation of Ni-BDC nanosheets (NSs). (I) Three-dimensional crystalline structure of Ni-BDC viewed along the [010] direction, illustrating the layer stacking arrangement. Nickel, oxygen, carbon, and hydrogen atoms are represented in green, orange, gray, and pink, respectively. (II) CO2 molecules replace coordinated H2O and bind to Ni1 sites, breaking the interlayer hydrogen bonding. (III) The chemically adsorbed CO2 stabilizes the exfoliated single-layer Ni-BDC nanosheets, exposing the active (200) planes. Reproduced with permission from ref. 127. (Copyright 2021 WILEY-VCH). | ||
One of the most common organic linkers, the porphyrin unit, has been regarded as a key active site in photoredox reactions. In the recent past, the Deng group determined that the photocatalytic activity increases as the distance between the interactive sites reduces and developed a formula that quantitatively explains this association.125 The porphyrin MOFs that were used as representatives in this case are MOF-525, PCN-221, PCN-222, PCN-223, PCN-224, and AlPMOF, which possess different intermolecular distances at the interactive sites. Nevertheless, excessive proximity of porphyrin active sites will decrease the size of the pore of MOFs, thereby inhibiting reaction kinetics. To overcome this, the authors developed a family of MOFs by creating vacancies by the partial removal of porphyrin linkers to form mesopores to favour the diffusion of the substrates. The results highlight the importance of the spatial arrangement of linker active sites, showing that shorter distances can theoretically maximize catalytic efficacy. Also, active sites on organic linkers could be introduced through postsynthetic modification. Pang et al. synthesized a mesoporous MOF, PCN-808, based on Zr6-oxo clusters and tetratopic carboxylate ligands. Using a linker installation procedure, the pore environment of PCN-808 was modified by precisely incorporating a linear ruthenium metalloligand [Ru(bpy)3]2+ (H2BDBR) into the framework, producing PCN-808-BDBR as an effective photocatalyst (Fig. 11b).126
The synthesis relies on a CO2-assisted exfoliation strategy in which supercritical CO2 acts as both a chemical modulator and a structural driving force. In bulk Ni-BDC, adjacent coordination layers are held together by hydrogen bonding between coordinated and lattice water molecules. When exposed to supercritical CO2, these water molecules are partially displaced and replaced by CO2, which chemically adsorbs onto coordinatively unsaturated Ni sites. This adsorption weakens the interlayer hydrogen bonds and induces effective delamination of the 3D framework into single-layer nanosheets (Fig. 11c). Simultaneously, the strong interaction between CO2 and Ni stabilizes the exposed metal centers, preserving the ultrathin layered structure, and results in 2D MOFs with a high surface area, abundant active sites, and enhanced accessibility for CO2 activation.127
3.6 Pore confinement strategy
MOFs are porous materials with well-defined and tunable pore structures. When the pore size matches the diameter of a metal complex ion, the precursor can be introduced during MOF growth, allowing the complex ion to be trapped within the MOF cages.128 Due to this size matching, at most one metal precursor can be confined within each pore, effectively separating the metal species at the atomic level.129 A subsequent mild pyrolysis step decomposes the precursor without damaging the overall framework of the MOF, while the resulting single metal atoms remain stabilized through interactions with the surrounding ligands. In this way, pore confinement acts as a pre-separation strategy that suppresses metal agglomeration during heat treatment by providing spatial isolation of the metal centers.130Shenghua Chen and coworkers reported a pore-confinement strategy in which a molecular N-heterocyclic carbene-ligated Cu complex was incorporated into an MOF during framework assembly. Because the size of the Cu-NHC complex matches the pore dimensions of the MOF, each cavity can host only one metal center, which ensures atomic-level separation and prevents Cu aggregation. The surrounding organic linkers and confined pore environment stabilize the Cu atoms through strong coordination and spatial restriction (Fig. 12a). This approach produces uniformly dispersed single-atom Cu sites embedded within the MOF, providing a well-defined and robust catalytic platform with high activity and stability.131 Bao et al. anchored Cu species onto open Zr6-oxo clusters of the Zr-based MOF PCN-222 via post-synthetic ion exchange, confining Cu within the MOF nanopores to generate strong metal-support interactions (SMSIs) (Fig. 12b). The resulting Cu@PCN-222 catalyst shows high CO2 hydrogenation activity toward methanol, achieving a space-time yield of 437.4 mg MeOH gcat−1 h−1 with good stability over 100 h. Detailed experimental and theoretical analyses reveal that Zr4+–O2−(–Cu2+) and Zr4+–Cu2+ interfacial sites act as highly efficient centers for CO2 and H2 adsorption and activation, while pore confinement modulates the electronic structure of Cu, favoring H2 activation and methanol selectivity.132
 | ||
| Fig. 12 (a) Schematic of the preparation of SAC 2Bn-Cu@UiO-67. Reproduced with permission from ref. 131. (Copyright 2022 Wiley-VCH). (b) Schematic diagram of the fabrication of Cu/PCN-222. Reproduced with permission from ref. 132. (Copyright 2025 Elsevier). (c) Schematic diagram of the fabrication of Ni-74-Am. Reproduced with permission from ref. 133. (Copyright 2024 WILEY-VCH). | ||
3.7 Defect engineering and vacancy control
Structural defects, including vacancies and interstitials, can adjust the coordination environment of metal centers and regulate their electronic properties. These defects provide additional active sites, enhance the stabilization of isolated metal atoms against aggregation, and modify the adsorption behavior of reactants, leading to improved catalytic performance.134,135 Yong-Li Dong et al. reported the construction of a defect-rich, amorphous Ni-MOF-74 (Ni-74-Am) by regulating synthesis temperature and solvent environment to induce ligand deficiencies (Fig. 12c). The resulting material contains abundant coordinatively unsaturated Ni sites and a mesoporous framework, which significantly improves photogenerated charge separation, carrier transport, and CO2 activation. Owing to these structural and electronic advantages, Ni-74-Am achieves a high CO evolution rate (1.38 mmol g−1 h−1) with 94% selectivity under visible-light irradiation, markedly outperforming crystalline Ni-MOF-74. This study highlights ligand-defect engineering as an effective strategy for enhancing MOF-based photocatalytic CO2 reduction.133 Wu et al. developed a Zr-based MOF-supported single-atom Cu catalyst (Cu@UiO-66-(COOH)2) by introducing controlled linker defects into the UiO-66 framework (Fig. 13a and b). The missing linkers create coordinatively unsaturated Zr6 nodes terminated with –OH/–H2O groups, which serve as strong anchoring sites for isolated Cu atoms and prevent their aggregation during synthesis and catalysis. Owing to the uniform atomic dispersion and strong metal-support interaction, the catalyst exhibits excellent activity and selectivity for the catalytic transfer hydrogenation of biomass-derived levulinic acid to γ-valerolactone. The high performance is attributed to the synergistic effect between Cu+1 single-atom sites responsible for catalytic activation and the acidic Zr6 nodes and carboxyl groups that promote intermediate transformation, resulting in high stability and recyclability.136 | ||
| Fig. 13 (a) Schematic structures of defective UiO-66 isoreticular MOFs with different functional groups, highlighting the Zr6 secondary building unit of UiO-66-(COOH)2, where unsaturated metal sites are occupied by –OH/–H2O (H atoms are omitted for clarity). (b) Illustration of the synthesis of single-atom Cu@UiO-66-(COOH)2, in which Cu precursors coordinate to dangling oxygen species on defective Zr nodes and are subsequently reduced to form isolated Cu sites. Reproduced with permission from ref. 136. (Copyright 2023 Elsevier). (c) Scheme of showing Ni2+ decoration in UiO-66-NH2via an efficient microwave-assisted method. Reproduced with permission from ref. 137. (Copyright 2021 American Chemical Society). | ||
Xing Ma et al. reported a microwave-assisted strategy for the synthesis of high-loading SACs on Zr-based MOFs. Metal salts such as NiCl2·6H2O, CoCl2·6H2O, CuCl2·2H2O, and RuCl3 were dispersed with the MOF in acetonitrile and rapidly heated to 85 °C for 30 min under microwave irradiation. The –O/OHx groups at defect sites of the Zr6-oxo clusters serve as anchoring points, stabilizing single metal atoms and preventing aggregation. Using this method, Ni loading reached 4.83 wt%, significantly higher than that in conventional solvothermal routes (1.01–1.37 wt%). Post-synthetic treatments, such as sulfidation or hydroxylation, allow modulation of the metal coordination environment, yielding atomically dispersed, highly accessible single-atom sites for photocatalysis137 (Fig. 13c).
Different incorporation strategies offer distinct benefits. Table 1 shows the overview of the main methods used to incorporate and stabilize single-atom catalysts (SACs) within MOFs. Anchoring single atoms at metal nodes provides high stability. Linker metallation allows control over electronic conductivity. Pore confinement suppresses aggregation during thermal treatment. However, these methods often suffer from low metal loading or limited scalability. Future work must address controllable high-density single-atom incorporation without sacrificing framework integrity or photocatalytic activity.
| MOF single atom system | MOF platform/support | Single atoms (SAs) | SA incorporation strategy | Ref. |
|---|---|---|---|---|
| Cu-UiO-66 | UiO-66 | Cu | Metal-node anchoring | 108 |
| Sn-ZIF-8 | ZIF-8 | Sn | Node ion exchange | 109 |
| Zn, N-MIL-125(Ti) | MIL-125(Ti) | Zn | In situ co-doping | 110 |
| M-Zr-MOFs | UiO-66, MOF-808, and NU-1000 | Cu, Fe, Ni, and Au | Node photodeposition | 111 |
| Re-UiO-66 | UiO-66-poly[bpy] | Re | Surface functionalization + postmetalation | 112 |
| Co, InCN | NH2-MIL-68 → CN | Co and In | MOF-derived pyrolysis | 118 |
| Co-MOF nanosheets | Co-MOF | Co | Ultrathin 2D MOF assembly | 120 |
| Fe-MOF/C–Ni NS | Fe-SACs/C–Ni NS | Fe | Pre-synthetic incorporation | 123 |
| Co-MOF-525 | MOF-525 | Co | Linker metallation | 51 |
| Co-PCN series | PCN-221-224 | Co | Linker distance engineering | 125 |
| Ru-PCN-808 | PCN-808 | Ru | Postsynthetic linker installation | 126 |
| Ni-BDC nanosheets | Ni-BDC | Ni | CO2-assisted exfoliation | 127 |
| Cu-UiO-67 | UiO-67 | Cu | Nanopore confinement | 131 |
| Cu-PCN-222 | PCN-222 | Cu | Ion exchange + confinement | 132 |
| Ni-MOF-74-Am | Ni-MOF-74-Am | Ni | Defect engineering | 133 |
| Cu-UiO-66-(COOH)2 | UiO-66-(COOH)2 | Cu | Defect-assisted anchoring | 136 |
| M-UiO-66-NH2 | UiO-66-NH2 | Ni, Co, Cu, and Ru | Microwave-assisted anchoring | 137 |
4 Characterization techniques of MOF-based SACs
The catalytic activity of SACs can only be fully understood through precise structural characterization of the isolated metal species. SACs differ fundamentally from conventional nanoparticle catalysts due to their atomic dispersion and unique electronic environments and therefore require advanced characterization techniques.138 A combination of spectroscopic, microscopic, and structural methods is commonly employed to elucidate their atomic structure, coordination environment, and electronic properties.139 Techniques such as X-ray absorption spectroscopy (XAS), including XANES and EXAFS, and X-ray photoelectron spectroscopy (XPS) provide valuable information on bonding states and local coordination.106,140 Meanwhile, isolated metal atoms can be directly visualized using high-resolution imaging techniques, particularly aberration-corrected HAADF-STEM and scanning tunneling microscopy (STM). These recent advances in electron microscopy, spectroscopy, and in situ techniques have significantly improved the understanding of the dynamic structural evolution of SACs under reaction conditions.141 Furthermore, density functional theory (DFT) calculations provide deeper insights into active-site geometry and reaction mechanisms, thereby enabling a more comprehensive understanding of their catalytic performance, particularly in the CO2 reduction reaction.1424.1 X-ray absorption spectroscopy (XPS)
High-energy X-rays generated at synchrotron radiation facilities provide powerful tools for probing materials and surface chemical processes at the atomic scale.143 Within XAS, X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) are widely employed to monitor oxidation states, coordination environments, and bond distances, allowing detailed insight into dynamic structural changes.144,145 The position and spectral features of the XANES region are closely associated with the oxidation state and local electronic structure of the absorbing atom, while variations in edge energy offer a reliable means to evaluate changes in metal valence.140 In contrast, EXAFS analysis yields quantitative information on the local coordination environment, including coordination numbers, neighboring atomic species, and interatomic bond lengths. Importantly, the absence of metal–metal scattering paths in EXAFS spectra serves as direct evidence for the formation of isolated single-atom sites.146 Together, XANES and EXAFS provide complementary and comprehensive structural and electronic information essential for understanding the nature and stability of single-atom catalysts. For instance, Co K-edge X-ray absorption spectroscopy was used to determine the coordination environment of Co single atoms in a MOF. EXAFS Fourier and wavelet transform analyses showed the absence of Co–Co scattering paths, confirming atomic dispersion of Co as isolated sites. EXAFS fitting indicated a Co–N4 coordination with a bond length of approximately 1.95 Å, consistent with a square-planar geometry and coordinatively unsaturated active sites. XANES spectra differed markedly from those of metallic Co, evidencing Co–N bonding, while the characteristic pre-edge feature arising from a 1s → 4p transition further supported the square-planar symmetry. The agreement between experimental and simulated XANES spectra validated the isolated nature and electronic structure of the Co single-atom sites (Fig. 14a–e).51 | ||
| Fig. 14 (a) Experimental Co K-edge XANES spectrum of MOF-525-Co (orange) compared with the corresponding theoretical spectrum; the non-convoluted calculated spectrum is shown for clarity. (b) Fourier-transformed magnitudes of Co K-edge EXAFS spectra (without phase correction) for Co foil, Co@C, and MOF-525-Co. (c) Normalized Co K-edge XANES spectra of MOF-525-Co and reference samples. (d) Wavelet transform of the k3-weighted Co K-edge EXAFS signal of MOF-525-Co, showing Co–N contributions and the absence of Co–Co scattering. (e) Wavelet transform of the k3-weighted Co K-edge EXAFS signal of Co foil, displaying characteristic Co–Co coordination features. Reproduced with permission from ref. 51. (Copyright 2016 Wiley-VCH). | ||
Jiao et al. demonstrated that in a MOF-derived FeSA–N–C catalyst, the Fe K-edge XANES spectrum shows the absorption threshold positioned between those of Fe2O3 and Fe foil, indicating positively charged Feδ+ species stabilized by nitrogen coordination (Fig. 15a). EXAFS analysis further resolved the Fe–N scattering paths with an average bond distance of ∼1.44–1.95 Å and revealed no detectable Fe–Fe contributions at ∼2.13 Å, confirming the formation of atomically dispersed Fe sites (Fig. 15b). Wavelength transform analysis of the EXAFS signal also supported the absence of Fe–Fe interactions and highlighted the localized coordination environment of Fe atoms. These combined X-ray measurements provide direct evidence of isolated Fe–N4 single-atom sites and are widely employed to verify the formation, electronic structure, and stability of single-atom active centers in MOF-derived SACs (Fig. 15c).147 Jianfei Sui and coworkers determined that the Pt K-edge XANES spectra of Pt1/SnO2/UiO-66-NH2 indicate that the Pt single atoms carry a partial positive charge between 0 and +2. EXAFS analysis shows a prominent peak at ∼1.63 Å, corresponding to Pt–O bonds, while no Pt–Pt (∼2.49 Å) or Pt–Cl (∼1.9 Å) signals are observed. EXAFS fitting suggests that each Pt atom is coordinated by approximately four oxygen atoms (Fig. 15d).148
 | ||
| Fig. 15 (a) Fe K-edge XANES spectra of FeSA–N–C, FeTPPCl, FePc, Fe2O3, and Fe foil. (b) Fourier-transformed EXAFS (FT-EXAFS) spectra of the same samples. (c) EXAFS fitting for FeSA–N–C (inset: structural model, Fe red, N blue, and C gray). Reproduced with permission from ref. 147. (Copyright 2018 WILEY-VCH). (d) FT k2-weighted χ(k) EXAFS spectra of Pt1/SnO2/UiO-66-NH2, PtO2, K2PtCl4, and Pt foil. Reproduced with permission from ref. 148. (Copyright 2022 WILEY-VCH). | ||
4.2 X-ray photoelectron spectroscopy (XPS)
It is a powerful surface-sensitive technique for analyzing elemental composition and oxidation states in heterogeneous catalysts. Advanced in situ XPS measurements, particularly when combined with synchrotron radiation, provide deeper insight into the nature, stability, and evolution of active sites such as single atoms under working conditions.137,149 For example, Weiwei Wang et al. employed XPS to analyze UiO-66(Zr/Ce), a system that reveals characteristic peaks associated with metal oxygen coordination and ligand environment, confirming successful incorporation of active sites. Deconvolution of the C 1s spectrum reveals contributions from sp2-hybridized C–C bonds and C–O species associated with the UiO-66 framework. The N 1s spectrum can be resolved into multiple components corresponding to metal–nitrogen coordination (N–Zr/Ce), C–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, and sp2-hybridized N–H species. Such detailed spectral analysis underscores the capability of XPS to identify metal-support interactions and local electronic environments, making it a crucial tool for characterizing MOF-based and single-atom catalysts (Fig. 16a–c).150 Ali M. Abdel-Mageed et al. analyzed the chemical states of Cu species in fresh and spent Cu/UiO-66 catalysts. The Zr 3d spectra showed nearly identical binding energies for the fresh and used samples, indicating that the Zr4+ framework remained structurally stable during the reaction. In contrast, the Cu 2p3/2 peak shifted slightly to higher binding energy after CO oxidation, accompanied by the appearance of characteristic satellite peaks, confirming the partial oxidation of Cu species from Cu0/Cu+ to Cu2+ under reaction conditions. These results demonstrate the effectiveness of XPS in tracking oxidation state changes and metal-support stability in MOF-supported catalysts.108
C, and sp2-hybridized N–H species. Such detailed spectral analysis underscores the capability of XPS to identify metal-support interactions and local electronic environments, making it a crucial tool for characterizing MOF-based and single-atom catalysts (Fig. 16a–c).150 Ali M. Abdel-Mageed et al. analyzed the chemical states of Cu species in fresh and spent Cu/UiO-66 catalysts. The Zr 3d spectra showed nearly identical binding energies for the fresh and used samples, indicating that the Zr4+ framework remained structurally stable during the reaction. In contrast, the Cu 2p3/2 peak shifted slightly to higher binding energy after CO oxidation, accompanied by the appearance of characteristic satellite peaks, confirming the partial oxidation of Cu species from Cu0/Cu+ to Cu2+ under reaction conditions. These results demonstrate the effectiveness of XPS in tracking oxidation state changes and metal-support stability in MOF-supported catalysts.108
 | ||
| Fig. 16 (a) Spectrum confirming the presence of C, N, O, Zr, and Ce; (b) high-resolution C 1s spectrum; (c) high-resolution N 1s spectrum. Reproduced with permission from ref. 150. (Copyright 2023 American Chemical Society). (d) Spectrum of Cu/UiO-66 (the Zr 3d region) and (e) Cu 2p region recorded after CO oxidation. Reproduced with permission from ref. 108. (Copyright American 2019 Chemical Society). | ||
4.3 Electron microscopy techniques
Electron microscopy techniques such as SEM, TEM, and HAADF-STEM are widely used to examine the morphology, microstructure, and elemental distribution of catalysts.151 When combined with EDS, these methods enable nanoscale mapping of metal species within MOF structures. In particular, aberration-corrected HAADF-STEM enables the direct visualization of isolated metal atoms, as the image contrast scales with the atomic number (Z2).152,153 Yu-Chen Hao et al. investigated the morphology and atomic dispersion of Ir1/A-aUiO and Pd1/A-aUiO using SEM, TEM, HAADF-STEM, and aberration-corrected HAADF-STEM. SEM and TEM images show that the A-aUiO particles exhibit a polyhedral morphology with sizes around 100–200 nm, which is preserved after Ir and Pd incorporation (Fig. 17a and b). HAADF-STEM analysis reveals no formation of metal nanoparticles, while aberration-corrected HAADF-STEM images display isolated bright spots corresponding to atomically dispersed Ir and Pd species (Fig. 17c and d). Elemental mapping further confirms the uniform distribution of Zr, O, C, Ir, and Pd within the MOF matrix, demonstrating the successful formation of single-atom catalytic centers.154 Yan Che et al. reported the preparation of Cu-DOTA@D-MIL via defect engineering of MIL-125-NH2 and site-specific anchoring of DOTA, followed by Cu metalation. SEM and TEM images (Fig. 17e–i) show that the disk-like morphology is retained after modification, while HRTEM and elemental mapping (Fig. 17j and k) confirm atomically dispersed Cu without nanoparticle formation.155 | ||
| Fig. 17 (a) SEM image of Ir1/A-aUiO particles. (b) Corresponding EDS mapping (c) and (d) Ir1/A-aUiO and Pd1/A-aUiO, indicating the atomic dispersion of metal species in A-aUiO matrices. Reproduced with permission from ref. 154. (Copyright 2021 Springer Nature). (e) SEM images of D-MIL, DOTA@D-MIL, and Cu-DOTA@D-MIL; scale bar: 5 µm. (f–i) TEM images of MIL-125-NH2, D-MIL, DOTA@D-MIL, and Cu-DOTA@D-MIL; the scale bar is 50 nm. (j and k) HRTEM and elemental mapping of Cu-DOTA@D-MIL. Reproduced with permission from ref. 155. (Copyright 2026 American Chemical Society). | ||
The Tan group reported the amorphous carbon structure of MnSA/NC and MnSA/SNC (Fig. 18a and b) without any observable lattice fringes, indicating the absence of crystalline manganese species. No Mn nanoparticles or clusters were detected in the TEM images, suggesting the lack of metal aggregation. To further verify the dispersion of Mn species, aberration-corrected high-angle annular dark-field scanning transmission electron microscopy (AC-HAADF-STEM) was employed. As shown in Fig. 18c, isolated bright spots are clearly observed and can be attributed to individual Mn atoms uniformly dispersed on the carbon matrix, confirming their atomic dispersion. In addition, the corresponding energy-dispersive X-ray spectroscopy (EDS) elemental mapping images reveal a homogeneous distribution of Mn along with C, N, and S (for MnSA/SNC) throughout the structure, further demonstrating the successful formation of Mn single-atom sites on the MOF-derived carbon support.156 In another study, Shenghua Chen et al. showed that HAADF-STEM images of 2Bn-Cu@UiO-67 exhibit an octahedral morphology with particle sizes around 60 nm, differing from that of 2Bn@UiO-67 due to variations in aggregation (Fig. 18d). The images also display numerous bright spots corresponding to atomically dispersed Cu and Zr sites, confirming the absence of Cu nanoparticles (Fig. 18e). EDX elemental mapping demonstrates a uniform distribution of Cu throughout the UiO-67 framework, indicating successful incorporation of Cu and its coexistence with Zr.131 Advanced spectroscopic and microscopic techniques provide strong evidence for the atomic dispersion and coordination environment of SA sites. However, most analyses are performed ex situ, offering limited insight into dynamic structural changes under operating conditions. In situ characterization is therefore essential for correlating the active-site structure with photocatalytic performance.
 | ||
| Fig. 18 (a) and (b) HRTEM images (the inset image shows the corresponding SAED pattern) and (c) (AC-HAADF-STEM) images of MnSA/NC and MnSA/SNC.156 (Copyright 2021 American Chemical Society). (d and e) AC HAADF-STEM image. Reproduced with permission from ref. 131. (Copyright 2022 WILEY-VCH). | ||
5 Applications of MOF-based SACs for photocatalytic CO2 reduction
Photocatalytic CO2 reduction requires catalysts with suitable band structures to enable efficient light absorption, charge separation, and surface redox reactions. However, limited CO2 adsorption and complex multi-step reaction pathways often result in low efficiency and poor product selectivity.157 Integrating SACs into MOFs offers an effective strategy to overcome these challenges. The strong interfacial interaction between MOFs and isolated metal atoms enhances charge separation, increases active site utilization, and suppresses charge recombination.158 Consequently, MOF@SACs have emerged as promising platforms for improving photocatalytic efficiency and selectivity in CO2 reduction. Metalloporphyrin-based MOFs with atomically dispersed metal centers provide valuable model systems for understanding structure–activity relationships in photocatalytic CO2 reduction.Co SAs embedded in porphyrin-based MOFs, such as MOF-525-Co, illustrate how the coordination environment affects photocatalytic performance. The coordinatively unsaturated Co sites maximize metal utilization and expose accessible active centers, enhancing CO2 adsorption and activation. Consequently, MOF-525-Co delivers CO and CH4 production rates of 200.6 and 36.7 µmol g−1 h−1, respectively, representing 3.13- and 1.93-fold improvements compared with pristine MOF-525 (Fig. 19a). The superior activity is attributed to the strong CO2 binding of isolated Co atoms and efficient charge transfer through the porphyrin framework.51 The role of the metal type and coordination environment is further highlighted by Cu and Ni single atoms in MOFs. Cu single atoms anchored onto UiO-66-NH2via amino groups using a photoinduced deposition method show enhanced charge separation and increased electron density at the Cu sites, which favors multi-electron CO2 reduction. This structural design results in CH3OH and C2H5OH production rates of 50.33 and 40.22 µmol g−1 h−1, respectively, significantly higher than those of their nanoparticle-based counterparts159 (Fig. 19b). Similarly, Ni single atoms incorporated into a hierarchically porous ZIF-8 framework (Ni/SOM-ZIF-8) illustrate the importance of combining atomic dispersion with macro-microporosity. The ordered porosity facilitates CO2 diffusion and adsorption, while isolated Ni atoms optimize reaction energetics. This results in a CO production rate of 4.2 mmol g−1 h−1 with 94% electron selectivity, roughly 47 times higher than that of pristine ZIF-8, and an apparent quantum yield of 0.71% under 400 nm irradiation (Fig. 19c).160
 | ||
| Fig. 19 (a) Time-dependent evolution of CO and CH4 over MOF-525-Co (green), MOF-525-Zn (orange), and MOF-525 (purple) photocatalysts, and the H6TCPP ligand (pink). Reproduced with permission from ref. 51. (Copyright 2016 WILEY-VCH). (b) Production rate of liquid fuels of Cu SAs/UiO-66-NH2 and comparative yield rates of C2H5OH and CH3OH formation using different catalysts. Reproduced with permission from ref. 159. (Copyright 2020 American Chemical Society). (c) Photocatalytic CO2 reduction performance of ZIF-8, Ni/ZIF-8, SOM-ZIF-8, Ni/SOM-ZIF-8, Co/SOM-ZIF-8, and Cu/SOM-ZIF-8; AQY and UV-vis absorption spectrum of Ni/SOM-ZIF-8. Reproduced with permission from ref. 160. (Copyright 2023 American Chemical Society). | ||
The UiO-66-NH2-Co/EDTA photocatalyst highlights the impact of metal coordination and chelation on CO2 reduction. Co SAs are uniformly incorporated into the UiO-66-NH2 framework using EDTA as a chelating agent, creating accessible active sites that facilitate CO2 adsorption, activation, and conversion to syngas. This design achieves CO and H2 generation rates of 776.40 and 1217.29 µmol g−1 h−1, nearly double those of EDTA-free UiO-66-NH2-Co, with a CO/H2 ratio of about 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, making it well-suited for industrial syngas applications (Fig. 20a–c).161 In the case of Fe@MIL-OV-T, isolated Fe sites are introduced into a MIL-125-NH2-based framework through Schiff-base functionalization in an oxygen-vacancy-rich structure. This combination of defect engineering and atomic dispersion promotes efficient CO2 photoreduction, achieving a CH3OH production rate of 3.96 mmol g−1 h−1 with 98% selectivity under visible light. Even without a sacrificial agent, the catalyst retains a CH3OH production rate of 0.59 mmol g−1 h−1, demonstrating its intrinsic efficiency (Fig. 20d–f).162
2, making it well-suited for industrial syngas applications (Fig. 20a–c).161 In the case of Fe@MIL-OV-T, isolated Fe sites are introduced into a MIL-125-NH2-based framework through Schiff-base functionalization in an oxygen-vacancy-rich structure. This combination of defect engineering and atomic dispersion promotes efficient CO2 photoreduction, achieving a CH3OH production rate of 3.96 mmol g−1 h−1 with 98% selectivity under visible light. Even without a sacrificial agent, the catalyst retains a CH3OH production rate of 0.59 mmol g−1 h−1, demonstrating its intrinsic efficiency (Fig. 20d–f).162
 | ||
| Fig. 20 (a) Schematic illustration of the charge-transfer mechanism in UiO-66-NH2-EDTA-Co during photocatalytic CO2 reduction to syngas under light irradiation. (b) Gas production rates of UiO-66-NH2-Co and the samples U-E-0.4, U-E-0.6, and U-E-1.2. (c) Control experiments for the composite sample U-E-0.6 using different solvent systems (MeCN/TEOA/H2O, MeCN/TEOA, and MeCN/TEA). Reproduced with permission from ref. 161. (Copyright 2024 American Chemical Society). (d) Schematic illustration of the synthesis of Fe@MIL-OV-T. (e) Photocatalytic CH3OH yields over MIL-125-NH2, MIL-OV-300, and Fe@MIL-OV-300. (f) CH3OH generation rates of Fe@MIL-OV-300 under different reaction conditions. Reproduced with permission from ref. 162. (Copyright 2025 Elsevier). (g) Photocatalytic performances of the RuS-SAs@Zn-PCN-222 series under standard conditions: catalyst (3 mg), NH3BH3 (10 mg), methanol (5 mL), light irradiation (λ > 420 nm), reaction time (0.5 h). Reproduced with permission from ref. 163. (Copyright 2025 Elsevier). | ||
Ru SAs immobilized on Zr6O8 clusters in the RuS-SAs@Zn-PCN-222 MOF further illustrate the role of metal-MOF electronic interactions. Stabilized by thiol (–SH) groups, the Ru sites enhance electron transfer and CO2 adsorption, promoting efficient coupling with surface H* species. This results in a formate (HCOO–) production rate of 54.4 mmol g−1 h−1, nearly 100% selectivity, and a turnover frequency of 440 h (Fig. 20g).163 Defect-engineered NH2-UiO-66 (A-aUiO) with isolated Ir single atoms at missing-linker sites shows that the combination of atomic dispersion and framework defects can dramatically improve activity and selectivity. Under visible-light irradiation (λ > 420 nm), Ir1/A-aUiO produces HCOOH at 0.51 mmol g−1 h−1 with 98–100% selectivity, while Ir clusters and nanoparticles produce more H2 and CO (Fig. 21a–c). The positively charged Ir sites (+3 to +4) reduce activation energy (9.8 kJ mol−1, Fig. 21e) and suppress side reactions. Incorporating Ir1/A-aUiO into a gas-permeable MOF membrane using a gas-membrane-gas (GMG) setup increases the formate yield to 3.38 mmol g−1 h−1, over six times higher than that of the particle-in-solution configuration, while retaining high selectivity and stability (Fig. 21f). Similar results are observed for Ir1/aMIL, demonstrating the synergy between MOF matrices and SACs in selective CO2 photoreduction.164
 | ||
| Fig. 21 (a) UV-Vis diffuse reflectance spectra showing enhanced light absorption of defect-engineered A-aUiO. (b) HCOOH evolution over time for A-aUiO, Ir1/A-aUiO, Irx/A-aUiO, and IrNPs/A-aUiO catalysts. (c) HCOOH and H2 selectivity as a function of Ir loading in Ir/A-aUiO catalysts. (d) Ir L-edge XANES spectra of Ir/A-aUiO with different Ir loadings; Ir foil and IrO2 are shown as references. The shift of the first XANES peak indicates decreasing metallicity with lower Ir loading. (e) Apparent activation energies (Eapp) for HCOOH formation over different catalysts. (f) HCOOH yields on A-aUiO and aMIL with and without Ir single atoms. Reproduced with permission from ref. 164. (Copyright 2021 Springer Nature). | ||
5.1 MOF-based SA nanocomposites
Despite the atomically dispersed catalytic sites and adjustable coordination spheres of MOF single-atom catalysts, their photocatalytic activities are fraught with challenges such as low electrical conductivity, limited photo-absorption ranges, rapid charge recombination rates, and instability.39 These challenges are effectively overcome by hybrid MOF-based composite systems and conductive supports, which help in improving photo-absorption ranges, facilitating charge separation and transportation, enhancing CO2 adsorption and activation abilities, and improving structural stability.165 The composite synergistic effect also promotes the control of reaction routes and the modulation of electronic structures for enhanced activities and stability. The strategy of making composites is crucial for fully exploring the potential of single-atom photocatalysts based on MOFs for solar-driven CO2 reduction.166X. Li et al. synthesized a hybrid Co-metalloporphyrin (CoTCPPNa4) ⊂ NH2-MIL-101(Fe) (CTN ⊂ NM) via a one-pot approach. The optimized 4%-CTN ⊂ NM composite (Co/Fe = 0.40) achieves a HCOOH production rate of 119.11 µmol g−1 h−1 without sacrificial agents, a 4.2-fold enhancement over the pristine MOF (Fig. 22a–c). With sacrificial agents, the rate increases to 179.87 µmol g−1 h−1, while the catalyst retains its activity over multiple cycles, confirming excellent stability and structural integrity.167Table 2 shows the summary of recently reported MOF@SA photocatalysts for CO2 reduction. Beyond the photocatalytic reduction of CO2 to CO, the generated CO can be directly utilized in downstream organic transformations to produce high-value compounds. Monticelli et al. demonstrated that rapid and complete CO2 to CO conversion (<10 min, >80% yield) enables immediate application of CO in various carbonylation reactions, including the synthesis of amides, esters, ketones, aldehydes, carboxylic acids, and ynones. The study further extended this strategy to isotopically labeled CO ([13C], [14C], [11C]), allowing the straightforward preparation of radiolabeled pharmaceuticals with high isotopic purity and good radiochemical yields. These findings highlight the practical relevance of CO2 photoreduction for integrated catalytic systems and emphasize the importance of achieving high CO2 to CO conversion to maximize the efficiency of subsequent chemical transformation.168 Similarly, X. L. Ma et al. determined that the integration of CO2 photoreduction and subsequent carbonylation in a continuous tandem photocatalytic system highlights a practical route for sustainable chemical synthesis. By encapsulating CdS QDs within PCN-Co, the photogenerated electrons are efficiently transferred to Co sites, reducing CO2 to CO, while the holes oxidize alcohol substrates to generate α-hydroxybenzyl radicals that undergo C–C coupling. The produced CO can be immediately utilized in downstream carbonylation reactions catalyzed by Pd/PCN-Zn, enabling the gram-scale synthesis of high-purity amides and pinacols. This dual functionality addresses key challenges in solar-driven CO2 reduction, including the efficient utilization of photogenerated holes, high selectivity toward valuable products, and the integration of oxidation and reduction processes within a single, continuous-flow system. These findings demonstrate the feasibility of combining CO2 valorization with fine chemical synthesis, paving the way for practical applications of photochemical CO2 conversion.169
 | ||
| Fig. 22 (a) Schematic representation for producing CTN ⊂ NM MOF structures via the one-pot method. (b) HCOOH generation rates of NM, CTN, CTN/NM, and CTN ⊂ NM in different systems. (c) Cyclic catalytic experiment with 4%-CTN ⊂ NM. Reproduced with permission from ref. 167. (Copyright 2026 Elsevier). | ||
| Photocatalyst | Single-atom | Light source | Product | Photocatalytic activity | Ref. |
|---|---|---|---|---|---|
| MOF-525-Co | Co | 300 W Xe lamp (400 nm < λ < 800 nm) | CO and CH4 | CO (200.6 µmol g−1 h−1) | 51 |
| CH4 (36.7 µmol g−1 h−1) | |||||
| Cu-SAs/UiO-66-NH2 | Cu | 300 W Xe lamp (λ > 400 nm) | CH3OH and C2H5OH | CH3OH (5.33 µmol g−1 h−1) | 159 |
| C2H5OH (4.22 µmol g−1 h−1) | |||||
| Ni/SOM-ZIF-8 | Ni | 300 W Xe lamp (λ > 400 nm) | CO | CO (4.2 mmol g−1 h−1) | 160 |
| UiO-66/EDTA-Co | Co | 100 W Xe lamp | CO and H2 | CO (776.4 µmol g−1 h−1) | 161 |
| H2 (1217.29 µmol g−1 h−1) | |||||
| Fe@MIL-OV-T | Fe | 300 W Xe lamp (420 nm < λ < 800 nm) | CH3OH | CH3OH (3.96 mmol g−1 h−1) | 162 |
| RuS-SAs@Zn-PCN-222 | Ru | 300 W Xe lamp (λ > 420 nm) | HCOO− | HCOO− (54.4 mmol g−1 h−1) | 163 |
| CTN ⊂ NM | Co | 300 W Xe lamp (λ > 420 nm) | HCOOH | 179.87 (µmol g−1 h−1) | 167 |
5.2 COF-based single-atom photocatalysts
Covalent organic frameworks (COFs) provide an excellent platform for stabilizing SACs, allowing precise control over the coordination environment of metal centers. For instance, Pan et al. designed Co-THD-COF with bioinspired N, S-coordination sites that anchored single Co atoms, facilitating CO2 adsorption, improving charge separation, and lowering the energy barrier for *COOH intermediate formation. This system achieved a remarkable CO generation rate of 9357 µmol g−1 h−1 with 95.1% selectivity, even in natural seawater, demonstrating the potential of COF-based SACs as highly efficient photocatalysts (Fig. 23a–f).170 COFs with SA active sites, such as the MoN2-modified COF (Mo-COF), show significantly enhanced photocatalytic CO2 reduction performance. The Mo-COF produced C2H4 at a rate of 3.57 µmol g−1 h−1 with 32.92% selectivity, along with CH4 (1.08 µmol g−1 h−1) and CO (6.19 µmol g−1 h−1) under visible light (Fig. 24a). Structural characterization confirmed SA-Mo incorporation without cluster formation via HAADF-STEM (Fig. 24b) and EXAFS (Fig. 24c and d). Photoelectrochemical measurements revealed improved charge separation and electron transfer in Mo-COF, evidenced by higher photocurrent response and smaller Nyquist arc radius compared to the pristine COF (Fig. 24e and f).171 | ||
| Fig. 23 Photocatalytic CO2 reduction by Co-THD-COF: (a) CO and H2 production rates and CO selectivity with different sacrificial agents; (b) time-resolved CO2RR performance of THD-COF and Co-THD-COF; (c) CO2 photoreduction under various control conditions; (d) GC-MS spectra from 13CO2 reduction; (e) CO and H2 production on M-THD-COFs (M = Co, Cu, Fe, Ni); (f) CO and H2 generation over five consecutive cycles (error bars from three replicates) Reproduced with permission from ref. 170. (Copyright 2024 Elsevier). | ||
 | ||
| Fig. 24 (a) Time courses of photocatalytic product formation. (b) Aberration-corrected HAADF-STEM image of Mo-COF showing single-atom Mo sites. (c) EXAFS analysis of Mo-COF (e); (d) Wavelet transform (WT) analysis of Mo foil, Mo-CO, and MoO3. (e) Transient photocurrent response and (f) electrochemical impedance spectra (EIS) of the COF and Mo-COF under visible light irradiation (λ ≥ 420 nm). Reproduced with permission from ref. 171. (Copyright 2021 Elsevier). | ||
Covalent organic frameworks (COFs) with anchored SA cobalt sites demonstrate excellent potential for photocatalytic CO2 reduction. The in situ coordination of Co with interlayer nitrogen atoms in keto-enamine TpPa-1 COFs ensures uniform dispersion of SAs, enhancing visible light absorption and charge separation. This design significantly increases CO production to 414.5 µmol g−1 h−1 with 99.45% selectivity (Fig. 25a–e), two orders of magnitude higher than that of pristine COFs, while retaining stability over multiple cycles. The formation of *COOH intermediates during the reaction confirms the single-atom Co sites as active centers, providing high efficiency and selectivity in CO2 reduction.172
 | ||
| Fig. 25 Photocatalytic CO2 reduction performance under visible light irradiation for TpPa-1 COFs and Co/TpPa-1 composites at different concentrations: (a) CO yield, (b) CH4 yield, (c) comparison of CO selectivity among SAC photocatalysts, (d) production rate after 3 h of visible light exposure, and (e) cycling stability test of the 0.05 mM Co/TpPa-1 composite. Reproduced with permission from ref. 172. (Copyright 2025 American Chemical Society). | ||
6 Future directions
MOF-based single-atom photocatalysts have shown considerable potential for solar-driven CO2 conversion, yet several challenges remain. Establishing clear structure–activity relationships is essential, particularly by linking single-atom coordination environments with charge-transfer behavior and product selectivity. Precise control over metal-framework electronic interactions will be critical for further performance optimization. Limited solar-light utilization continues to restrict overall efficiency. Future designs should focus on extending light absorption while suppressing charge recombination through rational linker engineering and interfacial charge-transfer modulation, without compromising structural stability. Long-term durability and scalability also require greater attention, as framework degradation, metal migration, and limited single-atom loading hinder practical application. Developing robust MOF architectures that support high single-atom densities under prolonged irradiation is therefore necessary. Finally, in situ and operando characterization should be more widely employed to track dynamic changes in active sites and reaction intermediates. Such insights, combined with theoretical modeling, will guide the rational design of next-generation MOF-based single-atom photocatalysts for efficient and sustainable CO2 conversion.7 Conclusion
MOF-based single-atom photocatalysts have emerged as a highly promising class of materials for solar-driven CO2 reduction, offering unique advantages over conventional nanoparticle systems. By confining isolated metal atoms within precisely defined coordination environments, MOF platforms enable near-complete metal utilization, enhanced exposure of active sites, and fine regulation of electronic structures. These features collectively lead to improved light harvesting, more efficient charge separation, and superior control over reaction intermediates and product selectivity. While MOF-based single-atom photocatalysts have demonstrated superior activity, stability, and selectivity, covalent organic frameworks (COFs) provide an additional platform with tunable porosity and electronic properties for stabilizing single-atom sites, offering further opportunities to enhance photocatalytic CO2 conversion. Throughout this review, it is evident that MOF-supported single-atom catalysts consistently outperform their nanoparticle and cluster counterparts in terms of activity, selectivity, and stability. For example, node-anchored and linker-coordinated SA systems frequently exhibit multi-fold enhancements in CO, CH4, CH3OH, or HCOO− production rates, while simultaneously suppressing competing hydrogen evolution reactions. Comparative analysis across reported systems clearly demonstrates that atomically dispersed metal centers facilitate optimized adsorption energies for key intermediates such as *COOH, *CO, and *OCHO, thereby lowering reaction energy barriers and steering reaction pathways toward desired C1 or C2+ products.In contrast to nanoparticle catalysts, which often suffer from metal aggregation, heterogeneous active sites, and severe charge recombination, single-atom incorporation within MOFs offers clear catalytic advantages. Strategies such as metal-node anchoring, linker metallation, pore confinement, and defect engineering enable precise control over atomic dispersion, electronic structure, and charge-transfer pathways, while MOF-derived and composite systems further enhance conductivity and light utilization. These findings underscore the importance of rationally designing and combining anchoring strategies to maximize photocatalytic efficiency. Nevertheless, challenges related to limited solar-light utilization, framework stability under prolonged irradiation, and high-density single-atom stabilization remain. Addressing these issues will require advanced in situ and operando characterization, coupled with theoretical modeling, to establish reliable structure–activity relationships. Overall, MOF-based single-atom photocatalysts represent a promising platform for efficient and sustainable solar-driven CO2 conversion.
Author contributions
Adnan Majeed: writing the original draft and software. Minh-Khoa Duong: writing, review and editing. Van-Duc Nguyen: data curation. Trong-On Do: conceptualization, resources, supervision, and overall guidance.Conflicts of interest
The authors declare no conflicts of interest.Data availability
No primary research results, software, or code have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
This work was supported by the Natural Science and Engineering Research Council of Canada (NSERC) through the Discovery Grant. We also acknowledge the partial financial support of the CGCC Center through FRQNT-Strategic Clusters, Canada (RS-265155).References
- M. Alshabib and S. A. Onaizi, A review on phenolic wastewater remediation using homogeneous and heterogeneous enzymatic processes: Current status and potential challenges, Sep. Purif. Technol., 2019, 219, 186–207 CrossRef CAS.
- M. Alshabib and S. A. Onaizi, Effects of surface active additives on the enzymatic treatment of phenol and its derivatives: a mini review, Curr. Pollut. Rep., 2019, 5, 52–65 CrossRef CAS.
- Y. Ren, G. Wang, X. Bai, Y. Su, Z. Zhang and J. Han, Research progress on remediation of organochlorine pesticide contamination in soil, Environ. Geochem. Health, 2024, 46, 25 CrossRef CAS PubMed.
- A. M. Alkadhem, M. A. Elgzoly and S. A. Onaizi, Novel amine-functionalized magnesium oxide adsorbents for CO2 capture at ambient conditions, J. Environ. Chem. Eng., 2020, 8, 103968 CrossRef CAS.
- N. Shehzad, M. Tahir, K. Johari, T. Murugesan and M. Hussain, A critical review on TiO2 based photocatalytic CO2 reduction system: Strategies to improve efficiency, J. CO2 Util., 2018, 26, 98–122 CrossRef CAS.
- M. Zhou, Z. Qu, J. Zhang, H. Jiang, Z. Tang and R. Chen, Boosting CO 2 chemical fixation over MOF-808 by the introduction of functional groups and defective Zr sites, Chem. Commun., 2024, 60, 3170–3173 RSC.
- C. I. Ezugwu, S. Liu, C. Li, S. Zhuiykov, S. Roy and F. Verpoort, Engineering metal-organic frameworks for efficient photocatalytic conversion of CO2 into solar fuels, Coord. Chem. Rev., 2022, 450, 214245 CrossRef CAS.
- S. K. Sharma, A. Kumar, G. Sharma, T. Wang, A. Iglesias-Juez and P. Dhiman, Recent advances in g-C3N4/Metal organic frameworks heterojunctions for high-performance photocatalytic environmental remediation and energy production, J. Mol. Liq., 2023, 382, 121890 Search PubMed.
- T. R. Cook, D. K. Dogutan, S. Y. Reece, Y. Surendranath, T. S. Teets and D. G. Nocera, Solar energy supply and storage for the legacy and nonlegacy worlds, Chem. Rev., 2010, 110, 6474–6502 CrossRef CAS PubMed.
- D. Senthil Raja, H.-W. Lin and S.-Y. Lu, Synergistically well-mixed MOFs grown on nickel foam as highly efficient durable bifunctional electrocatalysts for overall water splitting at high current densities, Nano Energy, 2019, 57, 1–13 Search PubMed.
- D. J. Soeder, Greenhouse gas sources and mitigation strategies from a geosciences perspective, Adv. Geo-Energy Res., 2021, 5, 274–285 CrossRef CAS.
- N. Prasetya, N. F. Himma, P. D. Sutrisna, I. G. Wenten and B. P. Ladewig, A review on emerging organic-containing microporous material membranes for carbon capture and separation, Chem. Eng. J., 2020, 391, 123575 Search PubMed.
- T. Li, P. Wang, M. He, T. Zhang, C. Yang and Z. Li, Metal-organic frameworks for photocatalytical carbon dioxide reduction reaction, Coord. Chem. Rev., 2024, 521, 216179 Search PubMed.
- E. Rabei and S. A. Onaizi, Photocatalytic conversion of carbon dioxide using metal organic framework-based materials: A review on recent advances and future outlooks, J. Mater. Sci., 2025, 1–42 Search PubMed.
- I. E. Agency, Global Energy Review 2025, 2025 Search PubMed.
- A. Wagner, C. D. Sahm and E. Reisner, Towards molecular understanding of local chemical environment effects in electro-and photocatalytic CO2 reduction, Nat. Catal., 2020, 3, 775–786 CrossRef CAS.
- A. Shahzadi, M. A. Iqbal, A. Majeed, I. Yasmeen, M. Akmal, S. Ejaz, S. Fatima, M. N. Arshad and M. Asad, Progress in mechanochemical synthesis of catalysts for the CO 2 processes: a step towards carbon neutrality, RSC Adv., 2026, 16, 7371–7388 RSC.
- D. T. Nguyen and T. O. Do, Comprehensive review for an efficient charge transfer in single atomic site catalyst/organic polymers toward photocatalytic CO2 reduction, Adv. Mater. Interfaces, 2023, 10, 2201413 CrossRef CAS.
- Y.-B. Huang, J. Liang, X.-S. Wang and R. Cao, Multifunctional metal–organic framework catalysts: synergistic catalysis and tandem reactions, Chem. Soc. Rev., 2017, 46, 126–157 Search PubMed.
- X. Kang, B. Wang, K. Hu, K. Lyu, X. Han, B. F. Spencer, M. D. Frogley, F. Tuna, E. J. McInnes and R. A. Dryfe, Quantitative electro-reduction of CO2 to liquid fuel over electro-synthesized metal–organic frameworks, J. Am. Chem. Soc., 2020, 142, 17384–17392 CrossRef CAS PubMed.
- O. Ola and M. M. Maroto-Valer, Review of material design and reactor engineering on TiO2 photocatalysis for CO2 reduction, J. Photochem. Photobiol., C, 2015, 24, 16–42 CrossRef CAS.
- Z. Kuspanov, B. Bakbolat, A. Baimenov, A. Issadykov, M. Yeleuov and C. Daulbayev, Photocatalysts for a sustainable future: Innovations in large-scale environmental and energy applications, Sci. Total Environ., 2023, 885, 163914 CrossRef CAS PubMed.
- Y. Tamaki, T. Morimoto, K. Koike and O. Ishitani, Photocatalytic CO2 reduction with high turnover frequency and selectivity of formic acid formation using Ru (II) multinuclear complexes, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 15673–15678 CrossRef CAS PubMed.
- S. Wang and X. Wang, Photocatalytic CO2 reduction by CdS promoted with a zeolitic imidazolate framework, Appl. Catal., B, 2015, 162, 494–500 CrossRef CAS.
- X. Li, Y. Sun, J. Xu, Y. Shao, J. Wu, X. Xu, Y. Pan, H. Ju, J. Zhu and Y. Xie, Selective visible-light-driven photocatalytic CO2 reduction to CH4 mediated by atomically thin CuIn5S8 layers, Nat. Energy, 2019, 4, 690–699 CrossRef CAS.
- A. Lais, M. Gondal, M. Dastageer and F. Al-Adel, Experimental parameters affecting the photocatalytic reduction performance of CO2 to methanol: a review, Int. J. Energy Res., 2018, 42, 2031–2049 CrossRef CAS.
- Z. Jiang, X. Xu, Y. Ma, H. S. Cho, D. Ding, C. Wang, J. Wu, P. Oleynikov, M. Jia and J. Cheng, Filling metal–organic framework mesopores with TiO2 for CO2 photoreduction, Nature, 2020, 586, 549–554 CrossRef CAS PubMed.
- Y. Wang, E. Chen and J. Tang, Insight on reaction pathways of photocatalytic CO2 conversion, ACS Catal., 2022, 12, 7300–7316 CrossRef CAS PubMed.
- P. Cai, W. Weng, Y. Han, X. Li, Z. Lu and Z. Wen, Boosting multi-carbon products selectivity of carbon dioxide reduction via bifunctional cyclodextrin-modification on copper/copper (I) oxide electrocatalysts, J. Colloid Interface Sci., 2025, 680, 453–458 CrossRef CAS PubMed.
- Y. Qian, F. Zhang, D. J. Kang and H. Pang, A Review of Metal–Organic Framework-Based Compounds for Environmental Applications, Energy Environ. Mater., 2023, 6, e12414 CrossRef CAS.
- H. Tong, S. Ouyang, Y. Bi, N. Umezawa, M. Oshikiri and J. Ye, Nano-photocatalytic materials: possibilities and challenges, Adv. Mater., 2012, 24, 229–251 CrossRef CAS PubMed.
- H. Li, Y. Zhou, W. Tu, J. Ye and Z. Zou, State-of-the-art progress in diverse heterostructured photocatalysts toward promoting photocatalytic performance, Adv. Funct. Mater., 2015, 25, 998–1013 CrossRef CAS.
- S. Zhu and D. Wang, Photocatalysis: basic principles, diverse forms of implementations and emerging scientific opportunities, Adv. Energy Mater., 2017, 7, 1700841 CrossRef.
- N. N. Vu, S. Kaliaguine and T. O. Do, Critical aspects and recent advances in structural engineering of photocatalysts for sunlight-driven photocatalytic reduction of CO2 into fuels, Adv. Funct. Mater., 2019, 29, 1901825 CrossRef.
- J. Zhang, J. Jiang, Y. Lei, H. Liu, X. Tang, H. Yi, X. Huang, S. Zhao, Y. Zhou and F. Gao, Photocatalytic CO2 reduction reaction: Influencing factors, reaction pathways and dominant catalysts, Sep. Purif. Technol., 2024, 328, 125056 CrossRef CAS.
- C. Zhu, S. Fu, Q. Shi, D. Du and Y. Lin, Single-atom electrocatalysts, Angew. Chem., Int. Ed., 2017, 56, 13944–13960 CrossRef CAS PubMed.
- J. Liu, Catalysis by supported single metal atoms, ACS Catal., 2017, 7, 34–59 CrossRef CAS.
- X. Su, X.-F. Yang, Y. Huang, B. Liu and T. Zhang, Single-atom catalysis toward efficient CO2 conversion to CO and formate products, Acc. Chem. Res., 2018, 52, 656–664 CrossRef PubMed.
- C. Gao, J. Low, R. Long, T. Kong, J. Zhu and Y. Xiong, Heterogeneous single-atom photocatalysts: fundamentals and applications, Chem. Rev., 2020, 120, 12175–12216 CrossRef CAS PubMed.
- B. Qiao, A. Wang, X. Yang, L. F. Allard, Z. Jiang, Y. Cui, J. Liu, J. Li and T. Zhang, Single-atom catalysis of CO oxidation using Pt1/FeO x, Nat. Chem., 2011, 3, 634–641 CrossRef CAS PubMed.
- X.-F. Yang, A. Wang, B. Qiao, J. Li, J. Liu and T. Zhang, Single-atom catalysts: a new frontier in heterogeneous catalysis, Acc. Chem. Res., 2013, 46, 1740–1748 CrossRef CAS PubMed.
- F. Chen, X. Jiang, L. Zhang, R. Lang and B. Qiao, Single-atom catalysis: Bridging the homo-and heterogeneous catalysis, Chin. J. Catal., 2018, 39, 893–898 CrossRef CAS.
- Y. Chen, S. Ji, C. Chen, Q. Peng, D. Wang and Y. Li, Single-atom catalysts: synthetic strategies and electrochemical applications, Joule, 2018, 2, 1242–1264 CrossRef CAS.
- R. Qin, P. Liu, G. Fu and N. Zheng, Strategies for stabilizing atomically dispersed metal catalysts, Small Methods, 2018, 2, 1700286 CrossRef.
- X. Li, L. Liu, X. Ren, J. Gao, Y. Huang and B. Liu, Microenvironment modulation of single-atom catalysts and their roles in electrochemical energy conversion, Sci. Adv., 2020, 6, eabb6833 CrossRef CAS PubMed.
- Y. Zhao, W. J. Jiang, J. Zhang, E. C. Lovell, R. Amal, Z. Han and X. Lu, Anchoring sites engineering in single-atom catalysts for highly efficient electrochemical energy conversion reactions, Adv. Mater., 2021, 33, 2102801 CrossRef CAS PubMed.
- Q. Pang, B. Tu and Q. Li, Metal–organic frameworks with multicomponents in order, Coord. Chem. Rev., 2019, 388, 107–125 CrossRef CAS.
- M. Ahmad, J. Chen, J. Liu, Y. Zhang, Z. Song, S. Afzal, W. Raza, L. Zeb, A. Mehmood and A. Hussain, Metal-organic framework-based single-atom electro-/photocatalysts: Synthesis, energy applications, and opportunities, Carbon Energy, 2024, 6, e382 CrossRef CAS.
- L. Zeng, X. Guo, C. He and C. Duan, Metal–organic frameworks: versatile materials for heterogeneous photocatalysis, ACS Catal., 2016, 6, 7935–7947 CrossRef CAS.
- A. Dhakshinamoorthy, A. M. Asiri and H. Garcia, Metal–organic framework (MOF) compounds: photocatalysts for redox reactions and solar fuel production, Angew. Chem., Int. Ed., 2016, 55, 5414–5445 CrossRef CAS PubMed.
- H. Zhang, J. Wei, J. Dong, G. Liu, L. Shi, P. An, G. Zhao, J. Kong, X. Wang and X. Meng, Efficient visible-light-driven carbon dioxide reduction by a single-atom implanted metal–organic framework, Angew. Chem., 2016, 128, 14522–14526 CrossRef.
- Z. Song, L. Zhang, K. Doyle-Davis, X. Fu, J. L. Luo and X. Sun, Recent advances in MOF-derived single atom catalysts for electrochemical applications, Adv. Energy Mater., 2020, 10, 2001561 CrossRef CAS.
- S. Ma, W. Han, W. Han, F. Dong and Z. Tang, Recent advances and future perspectives in MOF-derived single-atom catalysts and their application: a review, J. Mater. Chem. A, 2023, 11, 3315–3363 RSC.
- Z.-X. Pan, S. Yang, X. Chen, J.-X. Luo, R.-Z. Zhang, P. Yang, Q. Xu and J.-Y. Yue, Covalent organic framework with bioinspired N, S-anchored single atom sites for photocatalytic CO2 reduction reaction, Chem. Eng. J., 2024, 493, 152798 CrossRef CAS.
- M. E. G. Carmo, L. Spies, G. N. Silva, O. F. Lopes, T. Bein, J. Schneider and A. O. T. Patrocinio, From conventional inorganic semiconductors to covalent organic frameworks: advances and opportunities in heterogeneous photocatalytic CO 2 reduction, J. Mater. Chem. A, 2023, 11, 13815–13843 RSC.
- S. S. Tan, L. Zou and E. Hu, Photocatalytic reduction of carbon dioxide into gaseous hydrocarbon using TiO2 pellets, Catal. Today, 2006, 115, 269–273 CrossRef CAS.
- R. Li, W. Zhang and K. Zhou, Metal–organic-framework-based catalysts for photoreduction of CO2, Adv. Mater., 2018, 30, 1705512 CrossRef PubMed.
- Z. Kovacic, B. Likozar and M. Hus, Photocatalytic CO2 reduction: a review of ab initio mechanism, kinetics, and multiscale modeling simulations, ACS Catal., 2020, 10, 14984–15007 CrossRef CAS.
- X. Li, J. Yu, D. J. Gosztola, H. C. Fry and P. Deria, Wavelength-dependent energy and charge transfer in MOF: a step toward artificial porous light-harvesting system, J. Am. Chem. Soc., 2019, 141, 16849–16857 CrossRef CAS PubMed.
- J. Feng, S. Chen, Z. Lu and J. Bai, Metal–Organic Frameworks for Photocatalytic CO2 Reduction: Progress and Prospects, ACS Appl. Mater. Interfaces, 2025, 17, 60028–60054 CrossRef CAS PubMed.
- Y. Wang, D. He, H. Chen and D. Wang, Catalysts in electro-, photo-and photoelectrocatalytic CO2 reduction reactions, J. Photochem. Photobiol., C, 2019, 40, 117–149 CrossRef.
- E. Gong, S. Ali, C. B. Hiragond, H. S. Kim, N. S. Powar, D. Kim, H. Kim and S.-I. In, Solar fuels: research and development strategies to accelerate photocatalytic CO 2 conversion into hydrocarbon fuels, Energy Environ. Sci., 2022, 15, 880–937 RSC.
- Z.-H. Xue, D. Luan, H. Zhang and X. W. D. Lou, Single-atom catalysts for photocatalytic energy conversion, Joule, 2022, 6, 92–133 CrossRef CAS.
- W. Tu, Y. Zhou and Z. Zou, Photocatalytic conversion of CO2 into renewable hydrocarbon fuels: state-of-the-art accomplishment, challenges, and prospects, Adv. Mater., 2014, 26, 4607–4626 CrossRef CAS PubMed.
- J. Wu, Y. Huang, W. Ye and Y. Li, CO2 reduction: from the electrochemical to photochemical approach, Adv. Sci., 2017, 4, 1700194 CrossRef PubMed.
- C. W. Lee, N. H. Cho, S. W. Im, M. S. Jee, Y. J. Hwang, B. K. Min and K. T. Nam, New challenges of electrokinetic studies in investigating the reaction mechanism of electrochemical CO 2 reduction, J. Mater. Chem. A, 2018, 6, 14043–14057 RSC.
- L. Zuo, Y. Deng, L. Chen, T. He, J. Yang and J. Zhang, Fundamental insights into photoelectrochemical carbon dioxide reduction: elucidating the reaction pathways, ACS Catal., 2024, 14, 16795–16833 CrossRef CAS.
- H.-w. Zhu, R.-t. Guo, C. Liu, H.-f. Cui, M.-y. Liu and W.-g. Pan, Recent progress on photocatalytic reduction of CO2 to C2+ products, J. Mater. Chem. A, 2024, 21677–21703 RSC.
- B. Rhimi, M. Zhou, Z. Yan, X. Cai and Z. Jiang, Cu-based materials for enhanced C2+ product selectivity in photo-/electro-catalytic CO2 reduction: challenges and prospects, Nano-Micro Lett., 2024, 16, 64 CrossRef CAS PubMed.
- V. N. Gopalakrishnan, J. Becerra, E. F. Pena, M. Sakar, F. Béland and T.-O. Do, Porphyrin and single atom featured reticular materials: recent advances and future perspective of solar-driven CO 2 reduction, Green Chem., 2021, 23, 8332–8360 RSC.
- X. Liang, S. Yao, Z. Li and Y. Li, Challenge and Chance of Single Atom Catalysis: The Development and Application of the Single Atom Site Catalysts Toolbox: Pioneer Account, Acc. Chem. Res., 2025, 58, 1607–1619 CrossRef CAS PubMed.
- Y. Chen, J. Lin, B. Jia, X. Wang, S. Jiang and T. Ma, Isolating single and few atoms for enhanced catalysis, Adv. Mater., 2022, 34, 2201796 CrossRef CAS PubMed.
- C. A. Trickett, A. Helal, B. A. Al-Maythalony, Z. H. Yamani, K. E. Cordova and O. M. Yaghi, The chemistry of metal–organic frameworks for CO2 capture, regeneration and conversion, Nat. Rev. Mater., 2017, 2, 1–16 Search PubMed.
- H. Liu, M. Cheng, Y. Liu, J. Wang, G. Zhang, L. Li, L. Du, G. Wang, S. Yang and X. Wang, Single atoms meet metal–organic frameworks: collaborative efforts for efficient photocatalysis, Energy Environ. Sci., 2022, 15, 3722–3749 RSC.
- Y. Luo, X. Wang, F. Gao, L. Jiang, D. Wang and H. Pan, From single atom photocatalysts to synergistic photocatalysts: design principles and applications, Adv. Funct. Mater., 2025, 35, 2418427 CrossRef CAS.
- Z. Liang, C. Qu, D. Xia, R. Zou and Q. Xu, Atomically dispersed metal sites in MOF-based materials for electrocatalytic and photocatalytic energy conversion, Angew. Chem., Int. Ed., 2018, 57, 9604–9633 CrossRef CAS PubMed.
- H. Ali, O. Iqbal, B. Al Alwan, A. Abdulrahman, Y. Orooji, E. S. Al-Farraj, M. Sadiq, S. Imran, A. M. Abu-Dief and D. Yue, Morphology-controlled metal-organic frameworks for enhanced photocatalytic performance, Coord. Chem. Rev., 2025, 541, 216822 CrossRef CAS.
- Z. Lei, Y. Xue, W. Chen, W. Qiu, Y. Zhang, S. Horike and L. Tang, MOFs-based heterogeneous catalysts: new opportunities for energy-related CO2 conversion, Adv. Energy Mater., 2018, 8, 1801587 CrossRef.
- A. Wang, J. Li and T. Zhang, Heterogeneous single-atom catalysis, Nat. Rev. Chem., 2018, 2, 65–81 CrossRef CAS.
- T. Irie, K. Sasaki, S. Das and Y. Negishi, Materials Innovation and the Changing Face of Photocatalytic and Electrocatalytic Carbon Dioxide Reduction Research: From Metal Nanoclusters to Extended Frameworks, Angew. Chem., Int. Ed., 2025, e202515667 CAS.
- W. Gao, H. Chi, Y. Xiong, J. Ye, Z. Zou and Y. Zhou, Comprehensive insight into construction of active sites toward steering photocatalytic CO2 conversion, Adv. Funct. Mater., 2024, 34, 2312056 CrossRef CAS.
- S. Ji, Y. Chen, X. Wang, Z. Zhang, D. Wang and Y. Li, Chemical synthesis of single atomic site catalysts, Chem. Rev., 2020, 120, 11900–11955 CrossRef CAS PubMed.
- J. Li, M. F. Stephanopoulos and Y. Xia, Introduction: heterogeneous single-atom catalysis, Chem. Rev., 2020, 120, 11699–11702 CrossRef CAS PubMed.
- H. Zhang, G. Liu, L. Shi and J. Ye, Single-atom catalysts: emerging multifunctional materials in heterogeneous catalysis, Adv. Energy Mater., 2018, 8, 1701343 CrossRef.
- J. Xi, H. S. Jung, Y. Xu, F. Xiao, J. W. Bae and S. Wang, Synthesis strategies, catalytic applications, and performance regulation of single-atom catalysts, Adv. Funct. Mater., 2021, 31, 2008318 CrossRef CAS.
- W. Guo, Z. Wang, X. Wang and Y. Wu, General design concept for single-atom catalysts toward heterogeneous catalysis, Adv. Mater., 2021, 33, 2004287 CrossRef CAS PubMed.
- N. Cheng, L. Zhang, K. Doyle-Davis and X. Sun, Single-atom catalysts: from design to application, Electrochem. Energy Rev., 2019, 2, 539–573 CrossRef.
- Z. Liang, J. Shen, X. Xu, F. Li, J. Liu, B. Yuan, Y. Yu and M. Zhu, Advances in the development of single-atom catalysts for high-energy-density lithium–sulfur batteries, Adv. Mater., 2022, 34, 2200102 CrossRef CAS PubMed.
- N. Zheng and T. Zhang, Preface: single-atom catalysts as a new generation of heterogeneous catalysts, Natl. Sci. Rev., 2018, 5, 625 CrossRef.
- K. Qi, M. Chhowalla and D. Voiry, Single atom is not alone: Metal–support interactions in single-atom catalysis, Mater. Today, 2020, 40, 173–192 CrossRef CAS.
- Z. Li, S. Ji, Y. Liu, X. Cao, S. Tian, Y. Chen, Z. Niu and Y. Li, Well-defined materials for heterogeneous catalysis: from nanoparticles to isolated single-atom sites, Chem. Rev., 2019, 120, 623–682 CrossRef PubMed.
- A. J. Howarth, A. W. Peters, N. A. Vermeulen, T. C. Wang, J. T. Hupp and O. K. Farha, Best practices for the synthesis, activation, and characterization of metal–organic frameworks, Chem. Mater., 2017, 29, 26–39 CrossRef CAS.
- B. Liu, H. Shioyama, T. Akita and Q. Xu, Metal-organic framework as a template for porous carbon synthesis, J. Am. Chem. Soc., 2008, 130, 5390–5391 CrossRef CAS PubMed.
- H. Xiong, A. K. Datye and Y. Wang, Thermally stable single-atom heterogeneous catalysts, Adv. Mater., 2021, 33, 2004319 CrossRef CAS PubMed.
- Y. Yu, Y. Cheng, L. Tan, X. Liu, Z. Li, Y. Zheng, T. Wu, Y. Liang, Z. Cui and S. Zhu, Theory-screened MOF-based single-atom catalysts for facile and effective therapy of biofilm-induced periodontitis, Chem. Eng. J., 2022, 431, 133279 CrossRef CAS.
- W. Xue, X. Song and D. Mei, Theoretical insights into CO oxidation over MOF-808-encapsulated single-atom metal catalysts, J. Phys. Chem. C, 2021, 125, 17097–17108 CrossRef CAS.
- L. Zhu, X.-Q. Liu, H.-L. Jiang and L.-B. Sun, Metal–organic frameworks for heterogeneous basic catalysis, Chem. Rev., 2017, 117, 8129–8176 CrossRef CAS PubMed.
- A. Corma, H. Garcia and F. Llabrés i Xamena, Engineering metal organic frameworks for heterogeneous catalysis, Chem. Rev., 2010, 110, 4606–4655 CrossRef CAS PubMed.
- T. Drake, P. Ji and W. Lin, Site isolation in metal–organic frameworks enables novel transition metal catalysis, Acc. Chem. Res., 2018, 51, 2129–2138 CrossRef CAS PubMed.
- C.-C. Hou, H.-F. Wang, C. Li and Q. Xu, From metal–organic frameworks to single/dual-atom and cluster metal catalysts for energy applications, Energy Environ. Sci., 2020, 13, 1658–1693 RSC.
- W. Morris, B. Volosskiy, S. Demir, F. Gándara, P. L. McGrier, H. Furukawa, D. Cascio, J. F. Stoddart and O. M. Yaghi, Synthesis, structure, and metalation of two new highly porous zirconium metal–organic frameworks, Inorg. Chem., 2012, 51, 6443–6445 CrossRef CAS PubMed.
- X. Y. Dong, Y. N. Si, Q. Y. Wang, S. Wang and S. Q. Zang, Integrating single atoms with different microenvironments into one porous organic polymer for efficient photocatalytic CO2 reduction, Adv. Mater., 2021, 33, 2101568 CrossRef CAS PubMed.
- Y. Yu, Z. Zhu and H. Huang, Surface engineered single-atom systems for energy conversion, Adv. Mater., 2024, 36, 2311148 CrossRef CAS PubMed.
- J. Shen, C. Luo, S. Qiao, Y. Chen, Y. Tang, J. Xu, K. Fu, D. Yuan, H. Tang and H. Zhang, Single-atom Cu channel and N-vacancy engineering enables efficient charge separation and transfer between C3N4 interlayers for boosting photocatalytic hydrogen production, ACS Catal., 2023, 13, 6280–6288 CrossRef CAS.
- H. Huang, K. Shen, F. Chen and Y. Li, Metal–organic frameworks as a good platform for the fabrication of single-atom catalysts, ACS Catal., 2020, 10, 6579–6586 CrossRef CAS.
- L. Jiao and H.-L. Jiang, Metal-organic-framework-based single-atom catalysts for energy applications, Chem, 2019, 5, 786–804 CAS.
- L. Zou, Y. S. Wei, C. C. Hou, C. Li and Q. Xu, Single-atom catalysts derived from metal–organic frameworks for electrochemical applications, Small, 2021, 17, 2004809 Search PubMed.
- A. M. Abdel-Mageed, B. Rungtaweevoranit, M. Parlinska-Wojtan, X. Pei, O. M. Yaghi and R. J. r. Behm, Highly active and stable single-atom Cu catalysts supported by a metal–organic framework, J. Am. Chem. Soc., 2019, 141, 5201–5210 CrossRef CAS PubMed.
- W. Geng, W. Chen, G. Li, X. Dong, Y. Song, W. Wei and Y. Sun, Induced CO2 electroreduction to formic acid on metal–organic frameworks via node doping, ChemSusChem, 2020, 13, 4035–4040 Search PubMed.
- Z. Gao, J. Wang, Y. Muhammad, Y. Zhang, S. J. Shah, Y. Hu, Z. Chu, Z. Zhao and Z. Zhao, Enhanced moisture-resistance and excellent photocatalytic performance of synchronous N/Zn-decorated MIL-125(Ti) for vaporous acetaldehyde degradation, Chem. Eng. J., 2020, 388, 124389 Search PubMed.
- D. Chen, B. Chu, F. Li, Y.-T. Zheng, Y. Lu, B. Shao, L. Li, N.-Y. Huang and Q. Xu, Synergistic Catalysis by Cu Single Atoms and Atomically Cu-Doped Au Nanoparticles in a Metal–Organic Framework for Photocatalytic CO2 Reduction to C2H6, J. Am. Chem. Soc., 2025, 147, 22705–22713 CrossRef CAS PubMed.
- A. Youssef, I. Telegeiev, J. Dhainy, N. Alboudone, O. I. Lebedev, J. El Fallah, M. Hmadeh and M. El-Roz, Poly-Single-Atom Photocatalysts on UiO-66 Scaffolds for Superior CO2 Conversion under Visible Light, ACS Appl. Mater. Interfaces, 2025, 17, 32248–32259 Search PubMed.
- K. Shen, X. Chen, J. Chen and Y. Li, Development of MOF-derived carbon-based nanomaterials for efficient catalysis, ACS Catal., 2016, 6, 5887–5903 Search PubMed.
- P. Zhang, F. Sun, Z. Xiang, Z. Shen, J. Yun and D. Cao, ZIF-derived in situ nitrogen-doped porous carbons as efficient metal-free electrocatalysts for oxygen reduction reaction, Energy Environ. Sci., 2014, 7, 442–450 Search PubMed.
- J. Wei, Y. Hu, Y. Liang, B. Kong, J. Zhang, J. Song, Q. Bao, G. P. Simon, S. P. Jiang and H. Wang, Nitrogen-doped nanoporous carbon/graphene nano-sandwiches: Synthesis and application for efficient oxygen reduction, Adv. Funct. Mater., 2015, 25, 5768–5777 CrossRef CAS.
- M. Cui, B. Xu, X. Shi, Q. Zhai, Y. Dou, G. Li, Z. Bai, Y. Ding, W. Sun and H. Liu, Metal–organic framework-derived single-atom catalysts for electrocatalytic energy conversion applications, J. Mater. Chem. A, 2024, 12, 18921–18947 RSC.
- Z. Zhai, W. Yan, L. Dong, S. Deng, D. P. Wilkinson, X. Wang, L. Zhang and J. Zhang, Catalytically active sites of MOF-derived electrocatalysts: synthesis, characterization, theoretical calculations, and functional mechanisms, J. Mater. Chem. A, 2021, 9, 20320–20344 RSC.
- B. Hu, Z. Li, B. Wang, L. Chen, X. Wang, X. Hu, Z. Bai, Y. Li, G. Chen, X. Luo and S.-F. Yin, Construction of Co-In dual single-atom catalysts for photocatalytic CO2 reduction into CH4, Appl. Catal., B, 2025, 371, 125196 CrossRef CAS.
- M. Zhao, Y. Huang, Y. Peng, Z. Huang, Q. Ma and H. Zhang, Two-dimensional metal–organic framework nanosheets: synthesis and applications, Chem. Soc. Rev., 2018, 47, 6267–6295 RSC.
- Q. Zuo, R. Cui, L. Wang, Y. Wang, C. Yu, L. Wu, Y. Mai and Y. Zhou, High-loading single cobalt atoms on ultrathin MOF nanosheets for efficient photocatalytic CO2 reduction, Sci. China: Chem., 2023, 66, 570–577 Search PubMed.
- M. Ding, R. W. Flaig, H.-L. Jiang and O. M. Yaghi, Carbon capture and conversion using metal–organic frameworks and MOF-based materials, Chem. Soc. Rev., 2019, 48, 2783–2828 RSC.
- X. Jiao, K. Zheng, L. Liang, X. Li, Y. Sun and Y. Xie, Fundamentals and challenges of ultrathin 2D photocatalysts in boosting CO 2 photoreduction, Chem. Soc. Rev., 2020, 49, 6592–6604 RSC.
- A. Shankar, S. Marimuthu and G. Maduraiveeran, High-valent iron single-atom catalysts for improved overall water splitting via a reduced energy barrier and stabilization of the active center, J. Mater. Chem. A, 2024, 12, 121–127 Search PubMed.
- T. Ying, X. Shi, C. Yi, J. Hu, D. Li, R. Zhu and Y. Wang, Unveiling the cutting-edge innovations in single atom catalysts: Preparation, mechanism, performance, and applications, Nano Res., 2025, 18, 94907528 CrossRef.
- X. Gong, Y. Shu, Z. Jiang, L. Lu, X. Xu, C. Wang and H. Deng, Metal–organic frameworks for the exploitation of distance between active sites in efficient photocatalysis, Angew. Chem., 2020, 132, 5364–5369 CrossRef.
- J. Pang, Z. Di, J.-S. Qin, S. Yuan, C. T. Lollar, J. Li, P. Zhang, M. Wu, D. Yuan, M. Hong and H.-C. Zhou, Precisely Embedding Active Sites into a Mesoporous Zr-Framework through Linker Installation for High-Efficiency Photocatalysis, J. Am. Chem. Soc., 2020, 142, 15020–15026 CrossRef CAS PubMed.
- H. Chang, Y. Zhou, S. Zhang, X. Zheng and Q. Xu, CO2-induced 2D Ni-BDC metal–organic frameworks with enhanced photocatalytic CO2 reduction activity, Adv. Mater. Interfaces, 2021, 8, 2100205 Search PubMed.
- G. Cai, P. Yan, L. Zhang, H.-C. Zhou and H.-L. Jiang, Metal–organic framework-based hierarchically porous materials: synthesis and applications, Chem. Rev., 2021, 121, 12278–12326 CrossRef CAS PubMed.
- N. Stock and S. Biswas, Synthesis of metal-organic frameworks (MOFs): routes to various MOF topologies, morphologies, and composites, Chem. Rev., 2012, 112, 933–969 CrossRef CAS PubMed.
- N. Wang, Q. Sun and J. Yu, Ultrasmall metal nanoparticles confined within crystalline nanoporous materials: a fascinating class of nanocatalysts, Adv. Mater., 2019, 31, 1803966 CrossRef PubMed.
- S. Chen, W. H. Li, W. Jiang, J. Yang, J. Zhu, L. Wang, H. Ou, Z. Zhuang, M. Chen and X. Sun, MOF encapsulating N-heterocyclic carbene-ligated copper single-atom site catalyst towards efficient methane electrosynthesis, Angew. Chem., 2022, 134, e202114450 CrossRef.
- C. Bao, Y.-F. Huo, Y.-T. Li, S.-Q. Yang, W. Li and T.-L. Hu, Anchoring Cu on Zirconium-oxo nodes in a pore-confined metal-organic framework for CO2 hydrogenation to methanol, Chem. Eng. J., 2025, 503, 158610 CrossRef CAS.
- Y. L. Dong, Y. Jiang, S. Ni, G. W. Guan, S. T. Zheng, Q. Guan, L. M. Pei and Q. Y. Yang, Ligand defect-induced active sites in Ni-MOF-74 for efficient photocatalytic CO2 reduction to CO, Small, 2024, 20, 2308005 CrossRef CAS PubMed.
- K. A. Adegoke and N. W. Maxakato, Porous metal-organic framework (MOF)-based and MOF-derived electrocatalytic materials for energy conversion, Mater. Today Energy, 2021, 21, 100816 CrossRef CAS.
- J.-K. Sun, Y.-W. Pan, M.-Q. Xu, L. Sun, S. Zhang, W.-Q. Deng and D. Zhai, Heteroatom doping regulates the catalytic performance of single-atom catalyst supported on graphene for ORR, Nano Res., 2024, 17, 1086–1093 Search PubMed.
- Y. Wu, H. Wang, J. Peng, J. Zhang and M. Ding, Single-atom Cu catalyst in a zirconium-based metal–organic framework for biomass conversion, Chem. Eng. J., 2023, 454, 140156 Search PubMed.
- X. Ma, H. Liu, W. Yang, G. Mao, L. Zheng and H.-L. Jiang, Modulating Coordination Environment of Single-Atom Catalysts and Their Proximity to Photosensitive Units for Boosting MOF Photocatalysis, J. Am. Chem. Soc., 2021, 143, 12220–12229 Search PubMed.
- P. Qi, J. Wang, X. Djitcheu, D. He, H. Liu and Q. Zhang, Techniques for the characterization of single atom catalysts, RSC Adv., 2022, 12, 1216–1227 RSC.
- W. Song, C. Xiao, J. Ding, Z. Huang, X. Yang, T. Zhang, D. Mitlin and W. Hu, Review of carbon support coordination environments for single metal atom electrocatalysts (SACS), Adv. Mater., 2024, 36, 2301477 CrossRef CAS PubMed.
- A. Iglesias-Juez, G. L. Chiarello, G. S. Patience and M. O. Guerrero-Pérez, Experimental methods in chemical engineering: X-ray absorption spectroscopy—XAS, XANES, EXAFS, Can. J. Chem. Eng., 2022, 100, 3–22 Search PubMed.
- X. Li, X. Yang, J. Zhang, Y. Huang and B. Liu, In situ/operando techniques for characterization of single-atom catalysts, ACS Catal., 2019, 9, 2521–2531 CrossRef CAS.
- X. Wen, X. Geng, G. Su, Y. Li, Q. Li, Y. Yi and L. Liu, Machine learning-driven design of single-atom catalysts for carbon dioxide valorization to high-value chemicals: a review of photocatalysis, electrocatalysis, and thermocatalysis, Green Chem., 2025, 4898–4925 Search PubMed.
- Z. Song, J. Li, K. D. Davis, X. Li, J. Zhang, L. Zhang and X. Sun, Emerging Applications of Synchrotron Radiation X-Ray Techniques in Single Atomic Catalysts, Small Methods, 2022, 6, 2201078 CrossRef CAS PubMed.
- Y.-X. Li, N.-S. Chiu, W.-H. Lee, S. Bauer and B. Davis, X-ray Absorption Near Edge Structure (XANES) and Extended X-ray Absorption Fine Structure (EXAFS) Spectra of Pt—Sn—Alumina Catalysts, Characterization and Catalyst Development, ACS Publications, 1989, pp. 328–341 Search PubMed.
- J. Rockenberger, U. Zum Felde, M. Tischer, L. Tröger, M. Haase and H. Weller, Near edge X-ray absorption fine structure measurements (XANES) and extended X-ray absorption fine structure measurements (EXAFS) of the valence state and coordination of antimony in doped nanocrystalline SnO 2, J. Chem. Phys., 2000, 112, 4296–4304 CrossRef CAS.
- A. Gaur, B. D. Shrivastava and H. Nigam, Proc. Indian Natl. Sci. Acad., 2013, 921–966 Search PubMed.
- L. Jiao, G. Wan, R. Zhang, H. Zhou, S. H. Yu and H. L. Jiang, From metal–organic frameworks to single-atom Fe implanted N-doped porous carbons: efficient oxygen reduction in both alkaline and acidic media, Angew. Chem., 2018, 130, 8661–8665 Search PubMed.
- J. Sui, H. Liu, S. Hu, K. Sun, G. Wan, H. Zhou, X. Zheng and H. L. Jiang, A general strategy to immobilize single-atom catalysts in metal–organic frameworks for enhanced photocatalysis, Adv. Mater., 2022, 34, 2109203 Search PubMed.
- L. Chen, X. Ding, Z. Wang, S. Xu, Q. Jiang, C. Dun and J. J. Urban, Advances in in situ/operando techniques for catalysis research: enhancing insights and discoveries, J. Surf. Sci. Technol., 2024, 2, 9 CrossRef CAS.
- W. Wang, S. Song, P. Wang, M. He, Z. Fang, X. Yuan, H. Li, C. Li, X. Wang and Y. Wei, Chemical bonding of g-C3N4/UiO-66 (Zr/Ce) from Zr and Ce single atoms for efficient photocatalytic reduction of CO2 under visible light, ACS Catal., 2023, 13, 4597–4610 CrossRef CAS.
- Y. Su, G. Yuan, J. Hu, W. Feng, Q. Zeng, Y. Liu and H. Pang, Recent progress in strategies for preparation of metal-organic frameworks and their hybrids with different dimensions, 2023 Search PubMed.
- X. Wang, Y. Guo, X. Cui, Z. Wang, Z. Zhang, L. Tosheva and H. Guo, Graphene oxide membranes using MOF@ Chitosan core-shell nanoparticles as dual modulators for dye separation, Chem. Synth., 2024, 4, 27 CAS.
- Y. Yao, Z. Huang, P. Xie, L. Wu, L. Ma, T. Li, Z. Pang, M. Jiao, Z. Liang and J. Gao, High temperature shockwave stabilized single atoms, Nat. Nanotechnol., 2019, 14, 851–857 CrossRef CAS PubMed.
- Y.-C. Hao, L.-W. Chen, J. Li, Y. Guo, X. Su, M. Shu, Q. Zhang, W.-Y. Gao, S. Li and Z.-L. Yu, Metal-organic framework membranes with single-atomic centers for photocatalytic CO2 and O2 reduction, Nat. Commun., 2021, 12, 2682 CrossRef CAS PubMed.
- Y. Che, D. Chen, B. Wang, M. Li, Y. Zhao, G. Zhu and H. Xing, Site-Specific Asymmetric Coordination Engineering in Defective Metal–Organic Frameworks Stabilizes Cu (I) Active Sites for Selective CO2-to-Methanol Photocatalysis, J. Am. Chem. Soc., 2026, 148(5), 5764–5773 CrossRef CAS PubMed.
- H.-Y. Tan, S.-C. Lin, J. Wang, C.-J. Chang, S.-C. Haw, K.-H. Lin, L. D. Tsai, H.-C. Chen and H. M. Chen, MOF-templated sulfurization of atomically dispersed manganese catalysts facilitating electroreduction of CO2 to CO, ACS Appl. Mater. Interfaces, 2021, 13, 52134–52143 CrossRef CAS PubMed.
- W. Che, S. Zhao, W. J. Byun, T. Tao, J. P. Jeon, Q. Zhao, Y. Shao, J. Li, J. Kim and J. S. Lee, From carbon nitrides to COFs: opportunities and prospects in photocatalytic CO2 reduction, Adv. Mater., 2025, 37, e06961 CrossRef CAS PubMed.
- S. Ajmal, J. Huang, J. Guo, M. Tabish, M. A. Mushtaq, M. M. Alam and G. Yasin, Substrate engineering of single atom catalysts enabled next-generation electrocatalysis to power a more sustainable future, Catalysts, 2025, 15, 137 CrossRef CAS.
- G. Wang, C.-T. He, R. Huang, J. Mao, D. Wang and Y. Li, Photoinduction of Cu single atoms decorated on UiO-66-NH2 for enhanced photocatalytic reduction of CO2 to liquid fuels, J. Am. Chem. Soc., 2020, 142, 19339–19345 CrossRef CAS PubMed.
- Z. Liu, Z. Chen, M. Li, J. Li, W. Zhuang, X. Yang, S. Wu and J. Zhang, Construction of single Ni atom-immobilized ZIF-8 with ordered hierarchical pore structures for selective CO2 photoreduction, ACS Catal., 2023, 13, 6630–6640 CrossRef CAS.
- T.-A. Quach, M.-K. Duong, S. Mohan and T.-O. Do, Cobalt-Atom Active Sites Grafted UiO-66-NH2 via EDTA for the Enhanced Production of Syngas in CO2 Photoreduction, ACS Appl. Energy Mater., 2024, 7(19), 8797–8806 CrossRef CAS.
- Y. Che, C. Zhu, X. Liu, M. Li, X. Liu and H. Xing, Synergy of single-atom iron and titanium-oxo cluster at oxygen vacancy in MIL-125-NH2 to achieve high-performance CO2-to-CH3OH photocatalysis, Chem. Eng. J., 2025, 162811 CrossRef CAS.
- S. Li, W. Deng, Q. Mo, H. Song, C. Chen and L. Zhang, Engineering S-coordinated Ru Single-Atoms in a Porphyrinic Metal–Organic Framework for CO2 Photoreduction, Chin. J. Struct. Chem., 2025, 100841 Search PubMed.
- Y.-C. Hao, L.-W. Chen, J. Li, Y. Guo, X. Su, M. Shu, Q. Zhang, W.-Y. Gao, S. Li, Z.-L. Yu, L. Gu, X. Feng, A.-X. Yin, R. Si, Y.-W. Zhang, B. Wang and C.-H. Yan, Metal-organic framework membranes with single-atomic centers for photocatalytic CO2 and O2 reduction, Nat. Commun., 2021, 12, 2682 CrossRef CAS PubMed.
- B. Hu, Z. Li, B. Wang, L. Chen, X. Wang, X. Hu, Z. Bai, Y. Li, G. Chen and X. Luo, Construction of Co-In dual single-atom catalysts for photocatalytic CO2 reduction into CH4, Appl. Catal., B, 2025, 371, 125196 CrossRef CAS.
- Y.-J. Lin, F.-Y. Wu, Y. Wu and F.-X. Xiao, Single-atom alloys for photocatalytic applications: material innovation and light-driven reactivity, J. Mater. Chem. A, 2025,(13), 36056–36071 RSC.
- X. Li, Z. Lv, T. Wu, B. Ji, W. Wu, Y. Zhong, H. Xu, Z. Mao and L. Zhang, Structural-coupling driven cobalt metalloporphyrin⊂MIL-101 (Fe) hybrids for efficient photocatalytic CO2 reduction, J. Colloid Interface Sci., 2026, 139853 CrossRef CAS PubMed.
- S. Monticelli, A. Talbot, P. Gotico, F. Caillé, O. Loreau, A. Del Vecchio, A. Malandain, A. Sallustrau, W. Leibl and A. Aukauloo, Unlocking full and fast conversion in photocatalytic carbon dioxide reduction for applications in radio-carbonylation, Nat. Commun., 2023, 14, 4451 CrossRef CAS PubMed.
- X. L. Ma, W. X. Shi, S. Guo, Q. P. Zhao, W. Lin, T. B. Lu and Z. M. Zhang, Gram-Scale Green-Synthesis of High Purity Pinacols and Amides by Continuous Tandem Photocatalysis via a Negative Carbon Emission Process, Adv. Mater., 2025, 2506133 CrossRef CAS PubMed.
- Z.-X. Pan, S. Yang, X. Chen, J.-X. Luo, R.-Z. Zhang, P. Yang, Q. Xu and J.-Y. Yue, Covalent organic framework with bioinspired N,S-anchored single atom sites for photocatalytic CO2 reduction reaction, Chem. Eng. J., 2024, 493, 152798 CrossRef CAS.
- M. Kou, W. Liu, Y. Wang, J. Huang, Y. Chen, Y. Zhou, Y. Chen, M. Ma, K. Lei, H. Xie, P. K. Wong and L. Ye, Photocatalytic CO2 conversion over single-atom MoN2 sites of covalent organic framework, Appl. Catal., B, 2021, 291, 120146 CrossRef CAS.
- Y. Liu, L. Shao, L. Ding, X. Chen, Y. Bao and W. Chen, In Situ Anchoring of Co Single Atoms within Keto-Enamine COFs via the Coordination of an Interlayer N Atom with Co for the Enhanced Photocatalytic CO2 Reduction Efficiency, ACS Appl. Mater. Interfaces, 2025, 17, 26722–26730 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |