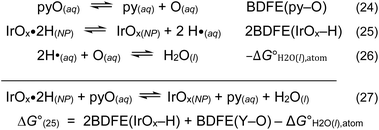Open Access Article
Open Access ArticleNon-ideal stoichiometry and thermochemistry of aqueous iridium oxide nanoparticles in proton-coupled electron transfer and oxygen-atom transfer
Justin L. Lee†
 ab,
Saeed Saeed†
ab,
Saeed Saeed† a and
James M. Mayer
a and
James M. Mayer *a
*a
aDepartment of Chemistry, Yale University, New Haven, Connecticut 06520-8107, USA. E-mail: james.mayer@yale.edu
bDepartment of Chemistry, College of the Holy Cross, Worcester, MA 01610, USA
First published on 11th April 2026
Abstract
Reported here are reactions of aqueous colloidal IrOx nanoparticles (NPs) with proton-coupled electron transfer (PCET) and oxygen-atom transfer (OAT) organic reagents, determining the reaction stoichiometries and thermochemistry. IrOx NPs have attracted much attention for their high electrocatalytic activity, but understanding of their fundamental reaction chemistry is limited. This IrOx NP model system is simple, with UV-vis titrations demonstrating reversible interconversion between predominantly IrIV and predominantly IrIII NPs. This simplicity allows studies that reveal their complex non-idealities. The NP redox chemistry has a “super-Nernstian” stoichiometry of ∼1.3H+ per 1e− transferred during both PCET and OAT reactions, as measured with electrochemistry and chemical methods. Spectroelectrochemistry revealed a broad distribution of surface IrOx–H bond dissociation free energies (BDFEs), becoming weaker as more H is added. Such variation in binding strengths—a non-ideal binding isotherm—is common for surface adsorbates. For IrOx, the variation of BDFE(IrO–H)s is fit well to a Frumkin isotherm with a width of 6.5 kcal mol−1. For OAT from the reactive oxygen atoms of IrOx NPs, bracketing experiments gave 93 ± 24 kcal mol−1 for the average BDFE(OxIr–O), with a predicted spread much larger than that for the BDFE(IrO–H). Taken together, the results show the importance of non-ideal stoichiometry and thermochemistry for IrOx NPs, and they open a path to more complete models to understand catalytic redox reactions at such surfaces.
1. Introduction
Redox reactions at metal-oxide/aqueous interfaces are essential to energy conversion, wastewater treatment, catalysis, corrosion, geochemistry, etc. These processes involve multiple steps on complex surfaces, and understanding them at atomistic detail is challenging. While much has been learned from experimental and computational studies of ideal surfaces, most surfaces have defects, irregularities, amorphous regions, and varying M:O:H stoichiometries.1–5 In addition, electrochemical studies have often implicated >1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+/e− stoichiometries, termed ‘super-Nernstian’ behaviour, as discussed below.6–9
1H+/e− stoichiometries, termed ‘super-Nernstian’ behaviour, as discussed below.6–9
Because of the complexity of oxide/water interfaces, direct experimental measurements of surface reactions are needed. Our approach is to study individual reaction steps on high surface area materials, especially colloidal nanoparticles (NPs).10–14 While such measurements are an average over the diverse NP surfaces, or perhaps for that reason, some organizing principles are beginning to emerge.
Examined here are redox reactions of aqueous colloidal iridium oxide (IrOx) nanoparticles (NPs).15 IrOx materials have long been known to undergo facile proton-coupled electron transfer (PCET)/hydrogen atom transfer (HAT) reactions. We have also recently reported oxygen atom transfer (OAT) reactions from these IrOx NPs to molecular substrates.14 These are some of the most fundamental reaction steps for redox-active oxides, and OAT is a rare example of a multi-electron process at a NP. Scheme 1 shows OAT with an O vacancy  , but lattice and adsorbed oxygen species can be difficult to distinguish for hydrous oxides (see below).16
, but lattice and adsorbed oxygen species can be difficult to distinguish for hydrous oxides (see below).16
A central parameter for surface reactions is the free energy of adsorption of the various intermediates. According to the Sabatier principle, a core intuition in heterogeneous catalysis, substrates and products should not bind too strongly or too weakly.17,18 We have advocated that surface binding energies be quantified similarly to molecular bond strengths, as bond dissociation free energies (BDFEs) (eqn (1)).19 Such BDFEs are central to molecular HAT and OAT reactions.20–23 The heterogeneous catalysis/surface science literature uses the similar 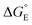 parameter that is defined vs. H2 or O2 and is in the opposite direction (eqn (2) and (3), E = H or O).16,17,24,25
parameter that is defined vs. H2 or O2 and is in the opposite direction (eqn (2) and (3), E = H or O).16,17,24,25 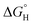 is often used as the ‘descriptor’ for multistep catalytic reactions, for instance in ‘volcano plots’ for the electrochemical hydrogen evolution reaction (HER).26–29
is often used as the ‘descriptor’ for multistep catalytic reactions, for instance in ‘volcano plots’ for the electrochemical hydrogen evolution reaction (HER).26–29
[surface] − E → [surface] + E![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ΔG° = BDFE([Surf] − E) ΔG° = BDFE([Surf] − E)
| (1) |
 | (2) |
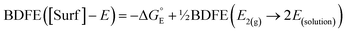 | (3) |
Surface BDFEs and the Sabatier principle are, however, challenging to use for real surfaces. Even for flat, single crystal surfaces, surface − E BDFEs vary with surface coverage (θE), typically decreasing at higher ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ.1,2,24,30–32 Complex surfaces have various types of surface sites, with different BDFEs. Surfaces thus typically deviate from ‘ideal’ behaviour, with adsorption not well described by the ideal Langmuir isotherm.5 Understanding non-idealities is particularly important for IrOx and related oxides33,34 because their amorphous, hydrous, or semicrystalline forms can have higher OER activity than single-crystal surfaces.35–37
θ.1,2,24,30–32 Complex surfaces have various types of surface sites, with different BDFEs. Surfaces thus typically deviate from ‘ideal’ behaviour, with adsorption not well described by the ideal Langmuir isotherm.5 Understanding non-idealities is particularly important for IrOx and related oxides33,34 because their amorphous, hydrous, or semicrystalline forms can have higher OER activity than single-crystal surfaces.35–37
This study uses chemical and electrochemical methods to determine the stoichiometries and thermochemistry of redox reactions of IrOx NPs. Studies were limited to IrIII–IrIV interconversions to enable reversibility (avoiding catalysis). This report starts with various aspects of PCET reactions, then moves to OAT, and discusses possible links between these reactions.
2. Results
2.1 Synthesis & characterization of IrOx NPs
The IrOx NPs used in this study were prepared following a procedure from Mallouk et al.,15 by first solvolysis of K2IrCl6 in basic water, followed by nitric acid-facilitated hydrolysis (SI Section S2, Fig. S1). The resulting dark purple colloidal solutions were 1.68 ± 0.01 mM [Ir] (by ICP-MS, see SI Section S2 and Fig. S3) and were used without further purification. Characterization of these NPs was reported in our recent OAT study.14 The studies here were performed at pH 1.86 unless otherwise specified (pH adjusted with aqueous HNO3 or NaOH, or by dilution with 18 MΩ H2O). The IrOx NPs were diluted 2-fold with 18 MΩ H2O in UV-vis spectroscopic and pH titration experiments ([Ir] = 0.84 ± 0.01 mM), while other experiments used the as-prepared colloids. The optical feature at λmax = 568 nm (top trace in Fig. 1A) is a useful spectroscopic handle for quantitative studies. | ||
| Fig. 1 (A) Spectra from a UV-vis titration of IrOx + H2Q. (B) Beer's law plot of corrected A568 vs. µmol of H2Q added. | ||
TEM images showed the NPs to be approximately spherical in shape, with an average diameter of 1.7 ± 0.2 nm (Fig. S1 and S2A). This corresponds to ∼73 Ir atoms per average NP, most of which are estimated to be on the surface (∼63 Ir, ∼86%, SI Section S2). The large surface-to-core atom ratio is valuable for reactivity and catalytic studies. For simplicity, the as-prepared NPs are called IrOx here, even though they likely have hydrous surfaces and an IrOxHy composition.
2.2 PCET reactivity for Ir4+/3+Ox(H+)m NPs
IrO2 is known to access higher oxidation state, catalytically active surface species by PCET.38–42 Murray and Mallouk15,30 have shown the PCET behaviour of aqueous IrOx NPs by cyclic voltammetry (CV) and rotated disk voltammetry. This section describes thermal PCET reactions of IrOx NPs, their redox stoichiometry and reversibility, CVs as a function of pH, and finally the e−-to-H+ stoichiometry.A similar titration monitored by 1H NMR spectroscopy showed the same endpoint. With increasing H2Q added, NMR spectra showed its conversion to 1,4-benzoquinone (Q; Fig. S5). The [Q] increased linearly until the endpoint, and H2Q was only observed past the endpoint, when there was no further formation of Q. Just as in the UV-vis experiment, the initial dark purple colour was bleached at the NMR endpoint (by eye).
H2Q is a 2e−, 2H+ PCET reagent, equally well described as a donor of two hydrogen atoms. The 0.70 ± 0.01 µmol H2Q endpoint thus has 1.40 µmol of reducing equivalents. The UV-vis titrations started with 1.68 µmol of Ir (2 mL of the 2-fold diluted 1.68 ± 0.01 mM as-prepared colloids), so the titration endpoint corresponds to 0.8 reducing equivalents per iridium. This ∼80% reactivity is consistently observed with various PCET, ET, and OAT reagents (vide infra).
Based on these observations and the NP oxidation reactions described below, we assign the Ir oxidation state of the as-prepared NPs to be Ir(3.8+). IrOx materials are often found in such non-integer average oxidation states, likely with a delocalized electronic structure, as IrO2 is metallic.43 The colourless reduced form at the end of the titration is assigned as essentially all IrIII, as anticipated from the coordination chemistry of iridium and lack of low-energy LMCT or MLCT transitions.44,45 The 568 nm absorbance has been suggested to be due to an Ir/Ir intervalence charge transfer, for instance on the basis of TD-DFT calculations.46–48 The slight red-shift of the spectrum as the NPs are reduced is perhaps due to the shifts of their reduction potentials and/or to their slight growth, as suggested by the TEM images discussed below.
TEM images after reduction by H2Q showed IrOx NPs with a diameter of 1.8 ± 0.2 nm (Fig. S2B). This is within error of the as-prepared NPs, perhaps with a slight expansion. A slight elongation of the Ir–O bonds would be expected upon reduction due to the increase in the ionic radius. For instance, a 0.05 Å increase in the Ir–O bond length was reported for an electrodeposited IrO2 film upon reduction.49
Analogous spectrophotometric redox titrations of the IrOx NPs were performed with ascorbic acid and sodium ascorbate (AscH2 and AscH−), with the same spectral changes as in Fig. 1A. The same spectra were also obtained in titrations with [CoII(bpy)3]2+, generally considered to be a mild, 1e−, outer-sphere reductant (Fig. S7).50
The endpoints of the AscH2 and Asc− titrations were the same as those with H2Q (Fig. S8 and S9), as expected because these are all overall 2e− reductants. With the CoII solution, the endpoint was at twice the number of moles of reagent, within our experimental uncertainty. This is consistent with the reported 1e− nature of this reductant.50 The redox titrations with these reagents thus agree and well define the redox changes of the IrOx NPs, specifically the number of electrons being added or removed. Discussion of the proton stoichiometries of these reactions, specifically how many H+ are taken up by the IrOx NPs upon reduction, is deferred to Section 2.2.3 below.
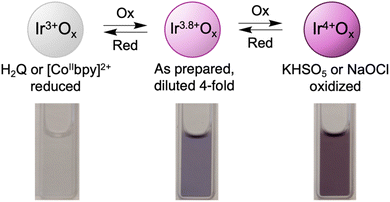
Oxone oxidized the as-prepared IrOx NPs, as indicated by a darkening of the purple suspensions. Titration of the as-prepared NPs with Na2SO4 caused little change in the optical spectra (Fig. S11), implicating KHSO5 as the reactive material. Scheme 2 shows images of the suspensions at different iridium oxidation levels. While quantitative titrations were not possible due to the instability of the particles with excess oxone (Fig. S12), the results are consistent with eqn (4) (which does not indicate the O and H stoichiometries).
| Ir+3.8Ox (as-prepared) + SO52− → Ir+4Ox + SO42− | (4) |
Sodium iodate readily oxidized the colourless H2Q-reduced IrOx NPs but appeared unreactive with the as-prepared NPs. Consistent with this limited reactivity, IO3− oxidized the H2Q-reduced IrOx NPs only to an absorbance close to that of as-prepared NPs. Iodate is primarily in its IO3− form at pH 1.86, based on its pKa of 0.8.51 Chemically reduced NPs (generated by treatment with 0.60 µmol H2Q [1.2 µmol reducing equivalents]) were re-oxidized by 0.24 µmol IO3−, consistent with the 5e− redox chemistry in eqn (5) (see below). The reduction of periodate stops at I2 because the Ir3.8Ox NPs are not strong enough reductants to form I− or I3−. This was confirmed by the oxidation of I− by as-prepared NPs (see below).
| IO3− + 5e− + 6H+ → ½I2 + 3H2O | (5) |
The IrOx NPs can undergo multiple reduction–oxidation cycles with H2Q and IO3− (Fig. 2). This is consistent with the reversibility of the Ir4+/3+ couple electrochemically (see ref. 30 and 15 and the next section). After a few cycles, a distinct feature at λmax = 454 nm appeared that is characteristic of I2,52,53 consistent with eqn (5). The optical titrations and the cycling in Fig. 2A also show a slightly shifted λmax for the IO3−-oxidized NPs, from the original 568 nm to 575 nm. The magnitude of the absorbance remained relatively constant over the four cycles shown in Fig. 2B, while the bleach upon addition of H2Q was less complete in later cycles. These changes could be due to the underlying I2 absorbance and/or to small changes in the NPs upon repeated redox cycling.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 proton-to-electron stoichiometry.54 However, hydrous forms of IrOx (and many other redox-active oxides6,55 typically have >59 mV pH−1 slopes (see the next section)).30,56–58 Such super-Nernstian slopes have typically been interpreted as >1
1 proton-to-electron stoichiometry.54 However, hydrous forms of IrOx (and many other redox-active oxides6,55 typically have >59 mV pH−1 slopes (see the next section)).30,56–58 Such super-Nernstian slopes have typically been interpreted as >1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+/e− ratios, but not always.59,60 To directly measure the stoichiometry for the NPs examined here, we applied our pH-monitoring approach previously developed for colloidal Au and TiO2 NPs.11,13
1H+/e− ratios, but not always.59,60 To directly measure the stoichiometry for the NPs examined here, we applied our pH-monitoring approach previously developed for colloidal Au and TiO2 NPs.11,13The >1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
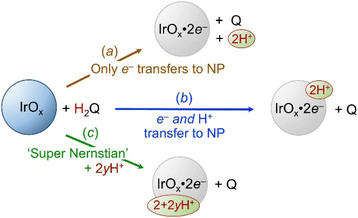
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+/e− stoichiometry of our IrOx NP redox reactions was measured by chemical titrations with concurrent pH measurements, using [H+] = 10−pH. If titrations with H2Q added only electrons to the IrOx NPs, one H+ would be released from H2Q per e− (Scheme 3(a), brown top reaction). Alternatively, if H2Q transferred H˙ (e− + H+) to IrOx, there would be no change in [H+] (blue reaction, (b)). ‘Super-Nernstian’ behaviour, uptake of greater than 1H+ per e−, would be indicated by a decrease in [H+] (green reaction, (c)).
1H+/e− stoichiometry of our IrOx NP redox reactions was measured by chemical titrations with concurrent pH measurements, using [H+] = 10−pH. If titrations with H2Q added only electrons to the IrOx NPs, one H+ would be released from H2Q per e− (Scheme 3(a), brown top reaction). Alternatively, if H2Q transferred H˙ (e− + H+) to IrOx, there would be no change in [H+] (blue reaction, (b)). ‘Super-Nernstian’ behaviour, uptake of greater than 1H+ per e−, would be indicated by a decrease in [H+] (green reaction, (c)).
For these experiments, the colloid was initially adjusted with NaOH(aq) to pH 2.90–3.10 ([H+]bulk = 0.79–1.26 mM), so that changes of ±0.1 µmol in the amount of H+ in the bulk solution could be detected with sufficient accuracy. An aliquot of H2Q was added to this solution; after a few minutes, both the pH and UV-vis spectra were measured, and the cycle was repeated. The spectra (Fig. S6) showed the titration endpoint. The experiment was performed with three separate solutions to ensure reproducibility.
Experimentally, addition of H2Q to these IrOx NPs resulted in an increase in pH, i.e., a decrease in the amount of H+ in solution. This is shown by the green squares in Fig. 3. After the optical titration endpoint, there was little change in the pH with additional H2Q. The data showed that, for each reducing equivalent transferred from H2Q to the NPs, the IrOx took up 1.28H+. In addition to the one H+ per e− from H2Q, IrOx absorbed 0.28 additional H+, in other words, super-Nernstian behaviour (eqn (6)).
| IrOx + ½H2Q + 0.28H+ → [IrOx·1e−, 1.28H+] + ½Q | (6) |
Experiments with the other reductants used above were consistent with the 1.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+
1H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− ratio but the complex speciation of those reagents and their products prevented quantitative analysis of the observed pH changes (see Section S4, Fig. S10). The 1.3
e− ratio but the complex speciation of those reagents and their products prevented quantitative analysis of the observed pH changes (see Section S4, Fig. S10). The 1.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+
1H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− results agree with the pH electrochemical results from Murray and from our laboratory (see the next section). These results are clearly different from the pure ET and 1
e− results agree with the pH electrochemical results from Murray and from our laboratory (see the next section). These results are clearly different from the pure ET and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1e−,H+ PCET pathways in Scheme 3 (see Section 3.1 below). The “extra” protons in the H2Q and other reactions are provided by the pH 1.86 solution, which results from the nitric acid addition in one step of the synthesis.
1e−,H+ PCET pathways in Scheme 3 (see Section 3.1 below). The “extra” protons in the H2Q and other reactions are provided by the pH 1.86 solution, which results from the nitric acid addition in one step of the synthesis.
 | ||
| Fig. 4 (A) CVs of aqueous IrOx NPs from pH 1.58–3.37; the + indicates the open circuit potential. (B) Pourbaix plot of E½ values vs. pH for the IrIV/III redox couple. | ||
The E½ values from the CVs shifted with pH from 1.5–4 by 78.5 mV per pH unit (Fig. 4B). This slope was in excellent agreement with the 75 mV per pH reported by Murray et al. for phosphate-buffered IrOx NPs in the pH 1–6 range.30 The close agreement of Murray's and our slopes indicates that this deviation is not a measurement error from the broad waves. These results implicate a PCET process, consistent with the chemical titrations reported above.
The observed E½ vs. pH slopes are ∼30% larger than the 59.2 mV pH−1 expected for 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+
1H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− transfers at 298 K. Such ‘super-Nernstian’ slopes, >59.2 mV pH−1, are common for hydrous metal oxides and typically interpreted as a >1H+ per e− stoichiometry. From this perspective, the measured slope of 78.5 mV pH−1 translates into 1.33H+/e− (eqn (7)). This is in excellent agreement with the proton stoichiometry experiments described in the last section, which gave 1.28H+/e−. This agreement is strong confirmation of the proton stoichiometry explanation for the super-Nernstian slope. This result is placed in a broader context in the Discussion below.
e− transfers at 298 K. Such ‘super-Nernstian’ slopes, >59.2 mV pH−1, are common for hydrous metal oxides and typically interpreted as a >1H+ per e− stoichiometry. From this perspective, the measured slope of 78.5 mV pH−1 translates into 1.33H+/e− (eqn (7)). This is in excellent agreement with the proton stoichiometry experiments described in the last section, which gave 1.28H+/e−. This agreement is strong confirmation of the proton stoichiometry explanation for the super-Nernstian slope. This result is placed in a broader context in the Discussion below.
| IrIV [in IrOx] + e− + 1.3H+ ⇌ IrIII [in IrOx](H+)1.3 | (7) |
2.3 PCET thermochemistry of [IrOx]–H
 , a 1e− + 1H+ reduction potential E°, and an X–H BDFE, since 1e− + 1H+ ≡ H˙ (eqn (8)). The
, a 1e− + 1H+ reduction potential E°, and an X–H BDFE, since 1e− + 1H+ ≡ H˙ (eqn (8)). The 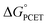 and −BDFE are simply related by the constant CG, which is 52.8 kcal mol−1 in water (eqn (9), for E° vs. NHE at pH 0, or for E° vs. RHE at any aH+).19
and −BDFE are simply related by the constant CG, which is 52.8 kcal mol−1 in water (eqn (9), for E° vs. NHE at pH 0, or for E° vs. RHE at any aH+).19For 1H+/1e− reactions:
| X + e− + H+ → X–H X + H˙ → X–H | (8) |
 | (9) |
The IrOx NP system, however, transfers 1.3 protons per electron, based on the electrochemical and pH studies in the last two sections. [Such behaviour has been termed super-Nernstian (see reviews6) and has been previously reported for hydrous IrOx and generally.] The non-1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry means that the ΔG° for reaction (7) is not simply a BDFE. What follows are our initial ideas; we have not found any related thermochemical analyses of super-Nernstian reactions in the literature. We expect to return to this topic in a future publication.
1 stoichiometry means that the ΔG° for reaction (7) is not simply a BDFE. What follows are our initial ideas; we have not found any related thermochemical analyses of super-Nernstian reactions in the literature. We expect to return to this topic in a future publication.
For a super-Nernstian reaction that transfers 1 + s protons per electron, the free energy ΔG10 can be represented as a BDFE plus s times the proton chemical potential µH+ = −2.303RT × pH (eqn (10)).
For (1 + s)H+/1e− reactions:
 | (10) |
For the IrOx NP system discussed here, the sµH+ term is quite small. With s = 0.3 and pH = 1.83, sµH+ = 0.8 kcal mol−1 (35 meV). Thus, the ΔG10 and BDFE for IrOx–H are numerically close, roughly within the uncertainties of the analysis. To further illustrate that this is a small effect, we return to the E/pH dashed line in Fig. 3B above, with a 78.5 mV pH−1 slope and a y intercept (E°) of 0.975 V vs. NHE. Requiring a fixed 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 PCET slope of 0.059 V pH−1 moves the intercept only 1.7 kcal mol−1 (74 meV) lower at most (Fig. S14). We also emphasize that all the thermochemical studies in this report were carried out at pH 1.86, so that the sµH+ term is constant and does not affect relative energies. Based on this analysis, the thermodynamic discussion that follows will ignore the 1.33H+/e− stoichiometry and use the normal BDFE parameter.
1 PCET slope of 0.059 V pH−1 moves the intercept only 1.7 kcal mol−1 (74 meV) lower at most (Fig. S14). We also emphasize that all the thermochemical studies in this report were carried out at pH 1.86, so that the sµH+ term is constant and does not affect relative energies. Based on this analysis, the thermodynamic discussion that follows will ignore the 1.33H+/e− stoichiometry and use the normal BDFE parameter.
 | (11) |
To experimentally measure the binding isotherm for hydrogen on IrOx NPs, we adapted a spectroelectrochemical approach previously developed for nickel oxide electrodes.32 Briefly, an IrOx NP colloid was poised at a specific electrochemical potential by controlled potential electrolysis, and after equilibration the UV-vis spectrum was recorded (Fig. 5B). These measurements were conducted at various Eapp from 0.53 to 1.13 V vs. NHE at pH 1.86; as indicated by dashed and coloured vertical lines in Fig. 5A. Applying more anodic potentials resulted in oxidation beyond Ir4.0+. Further details are discussed in Section S5.
 | ||
| Fig. 5 (A) CV of IrOx NPs. Coloured lines indicated applied potential steps at 0.025 V increments. (B) Optical spectra at different applied potentials. The colours of the absorbance spectra correspond to the colour of the applied potentials in Fig. 5A. (C) Plot of hydrogen coverage and Ir oxidation state as a function of applied potential and BDFE([IrOx]–H). Coloured points indicate experimental data fitted to the Frumkin isotherm (black line; eqn (15)). The purple dashed line corresponds to the H-binding isotherm as described by the Langmuir isotherm (eqn (11)) and Nernst equation (eqn (17) below). | ||
The 568 nm absorbance decreased at more cathodic Eapp, where the NPs were more reduced, and increased at anodic Eapp (Fig. 5B). These changes were fully reversible. Waiting for equilibration and the reversibility of the absorbance show that these experiments measure thermodynamic properties. As established above, the A568nm directly gives [IrIV] or, equivalently, the average oxidation state n of Ir (Irn+) (eqn (12)). This is readily converted to the fraction of Ir ions that are formally IrIV, f(IrIV), and to the coverage of H (eqn (13) and (14)).
| A568 = ε568[IrIV] | (12) |
 | (13) |
| θH = 1 − f(IrIV) = f(IrIII) | (14) |
The experimental isotherm, the dependence of θH on Eapp, is shown as the coloured points in Fig. 5C. The measured isotherm is quite broad, needing a change of 320 mV to move from 10% to 90% θH. The ideal Langmuir isotherm is much narrower, making this change over only 113 mV (the purple dashed line in Fig. 5C). The broadness of the isotherm is the primary origin of the broad CV waves in Fig. 4A.
The measurements fit well to a non-ideal Frumkin isotherm (black line, eqn (15)).63,64 The century-old Frumkin isotherm simply adds a term linear in θH to the Langmuir isotherm (eqn (11)). The conclusion that surface binding is non-ideal is consistent with the discussion above about real surfaces, especially small nanoparticles.5
 | (15) |
As a check, the midpoint of the fit (0.87 V) is within 40 mV of the E1/2 measured by CV of the colloid at pH 1.86, as it should be. In addition, the isotherm in Fig. 5C is consistent with the as-prepared IrOx NPs being mostly, but not completely, reduced by excess I− (Fig. S13). The E°(I2/I−) of ∼0.62 V lies close to the bottom end of the isotherm, and the effective potential was higher due to the excess of I− over I2. The isotherm also predicts θH = 0.2 and Ir3.8+ for the as-prepared NPs, from their open circuit potential (OCP) value (Fig. 5C), in agreement with the titrations above. All the results are consistent with this Frumkin isotherm.
Fitting the data to the Frumkin equation gave C = 0.28 V, with free energy FC = 0.28 eV (6.5 kcal mol−1). In some presentations, C is replaced by the unitless parameter g = FC/RT (=11 in this case).65,66 Given the width of the isotherm, the BDFE([IrOx]–H) should be considered to have a range from 73 to 79 kcal mol−1, with a midpoint of 76 kcal mol−1.
2.4 OAT reactivity of IrOx NPs
We recently reported that IrOx NPs can oxidize substrates by oxygen atom transfer, including phosphines to phosphine oxides and a thioether to its sulfoxide (and slowly to the sulfone).14 Here we extend those studies and compare them with the PCET chemistry described above.While PAr3 and H2Q are both 2e− reductants, phosphines accept O atoms while H2Q typically donates H atoms. 18O studies showed that OAT from IrOx to PAr3 forms some sort of vacant site, which is rapidly filled by a water molecule.14 The process of O-removal and H2O addition apparently forms the same product as the reduction by H2Q.14,67 To confirm this, we repeated the proton stoichiometry titration using PAr3 instead of H2Q. The pH and [H+] change with PAr3 (Fig. S18) were essentially identical to those for H2Q (Fig. 3).
Overall, the similarities between the PAr3 and H2Q reactions reaffirm the implicit assumption above that these reactions are all under thermodynamic control. These are two quite different reagents, but they add the same number of reducing equivalents and form the same IrOxHy NP product.
In the other direction, the as-prepared NPs are reduced by PAr3 and methionine (Fig. S21–S25),14 and also by excess nitrite (NaNO2, Fig. S26). Nitrate formation was demonstrated by 15N NMR (15NO3− from 15NO2−, Fig. S27). However, titration with nitrite did not show a sharp endpoint and nitrite is a complicated reagent that can disproportionate in acid solutions.
Many of these net OAT reactions are listed in order of decreasing estimated Y–O BDFEs in Scheme 4 (mostly converted from BDEs: see SI Section S7). As expected, the reagents with high affinities to accept an O atom reduce the IrOx NPs (arrows pointing to the right), and those that have weak Y–O bonds are good O-atom donors and oxidize the NPs (arrows to the left).
These reagents cover a huge range of driving forces for the OAT reactions, at least 87 kcal mol−1. This range of ΔG corresponds to a change in Keq of ≥1064. The results in Scheme 4 provide some insights into the OAT thermochemistry of the IrOx NPs, as discussed below.
In contrast, no reaction was observed between the IrOx NPs and pyridine derivatives. The as-prepared IrOx NPs were not reduced by pyridine (py) or 4-CN-pyridine, and the H2Q-reduced NPs were not oxidized by pyridine-N-oxide (pyO, Fig. S29–S31). The lack of reaction with both py and pyO indicates a kinetic barrier for OAT. In contrast, the HAT/PCET reactions of these IrOx NPs all seem to follow their thermochemistry without substantial kinetic limitation.
3. Discussion
3.1 IrOx NPs: a simple model system that reveals complexities
From one perspective, this colloidal IrOx NP system is simple. Chemical reactions reversibly cycle the NPs between predominantly IrIV and predominantly IrIII (for the 80% of the Ir that is redox active). The reaction stoichiometry sets the redox level of the product NPs, so the same thermodynamic product is formed regardless of the reagent. The same product is formed in PCET, HAT, and OAT reactions that give the same average Irn+. This reversibility and generality have enabled understanding of the intrinsic underlying complexity of the IrOx NPs.Based on our titrations and measurements of total Ir by ICP-MS, 80% of the Ir ions in these NPs are redox active. Gambarella, Murray, et al. reported the same result for IrOx NPs at pH 1.4, using coulometry (though almost all the Ir was electroactive at higher pH values).30 We previously estimated that the ∼1.7 nm IrOx NP diameter corresponds to 4–5 unit cells across and roughly 86% of the Ir ions are on the surface.14 While there are uncertainties in both the percent electroactive and the percent at the surface, the values are consistent with the redox reactions and H addition primarily involving surface sites. This is consistent with the lack of a change in size by TEM.14 However, oxides in water often have a hydrous, partly amorphous shell, which muddies the definition of a surface atom.
All the chemical reactions studied here, PCET, HAT, and OAT, have the same 1.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 proton-to-electron stoichiometry, or H+ per redox equivalent. The H+
1 proton-to-electron stoichiometry, or H+ per redox equivalent. The H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− ratio is important because it determines how the reduction potential varies with pH. This follows from the Nernst or Langmuir equations, with [XHm(m–n)+]/[X] ≡ (θ/1 − θ) (eqn (16)–(21)). Examples with integer H+
e− ratio is important because it determines how the reduction potential varies with pH. This follows from the Nernst or Langmuir equations, with [XHm(m–n)+]/[X] ≡ (θ/1 − θ) (eqn (16)–(21)). Examples with integer H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− ratios are widely recognized, in Pourbaix's comprehensive Atlas and elsewhere.68,69 E/pH slopes that are not integer multiples of 59 mV pH−1 (m/n) have also long been known and are typically interpreted as a non-integer H+
e− ratios are widely recognized, in Pourbaix's comprehensive Atlas and elsewhere.68,69 E/pH slopes that are not integer multiples of 59 mV pH−1 (m/n) have also long been known and are typically interpreted as a non-integer H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− stoichiometry. However, other effects could lead to such super-Nernstian slopes. For example, recent papers by Suntivich and co-workers assumed 1
e− stoichiometry. However, other effects could lead to such super-Nernstian slopes. For example, recent papers by Suntivich and co-workers assumed 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+
1H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− ratios and attributed super-Nernstian slopes to changes in E°.7,34,70 For our IrOx NPs, the non-integer stoichiometry was established by a direct chemical method, the change in pH upon reaction (Fig. 3), in addition to electrochemistry. The 1.3
e− ratios and attributed super-Nernstian slopes to changes in E°.7,34,70 For our IrOx NPs, the non-integer stoichiometry was established by a direct chemical method, the change in pH upon reaction (Fig. 3), in addition to electrochemistry. The 1.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio must be the thermodynamic preference of the IrOx, although the origin of this preference is not known. For these NPs, we take full H coverage, θH = 1, to be 1.3H+ for every IrIII.
1 ratio must be the thermodynamic preference of the IrOx, although the origin of this preference is not known. For these NPs, we take full H coverage, θH = 1, to be 1.3H+ for every IrIII.
| For: X + mH+ + ne− → XHm(m–n)+ | (16) |
 | (17) |
 | (18) |
 | (19) |
The IrOx NPs studied here have non-ideal H adsorption in terms of both stoichiometry and thermochemistry. The adsorption energies follow a Frumkin isotherm rather than a Langmuir isotherm. θH as a function of pH and Eapplied does not follow eqn (18) and (19). We have not found literature that addresses this case of both non-integer stoichiometry and a non-ideal binding isotherm, and we are not prepared to do this fully here. Still, the model system studies reported here show large non-idealities that are not captured by current models.
To simplify the thermochemical discussions that follow, the remainder of this Discussion will assume a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+
1H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e− stoichiometry. As explained above, this assumption has little numerical effect on the parameters because the pH was kept constant at 1.83 that is close to the standard state of pH 0.
e− stoichiometry. As explained above, this assumption has little numerical effect on the parameters because the pH was kept constant at 1.83 that is close to the standard state of pH 0.
3.2 H-atom binding energies
The measured Frumkin isotherm is about 3 times broader than ideal Langmuir behaviour, with a long linear middle portion. The ratio of IrIII to IrIV in the NPs changes much more slowly with potential than in ideal behaviour. The non-ideality of these IrOx NPs contradicts the common assumption that surface concentrations are good proxies for surface activities, and the common assumption of a single free energy of adsorption. The measured isotherm implies a range of IrOx–H BDFEs, from 73–79 kcal mol−1.
The non-ideality of the binding energies is not surprising, as discussed in the next section.5 To quote and expand Gileadi's colourful analogy:66
“Solid surfaces are rarely homogeneous. There are active sites on which the standard free energy of adsorption is high, and there are less-active sites…. This is much like people entering a movie theater with unmarked seats: the best seats are taken first, and the worst remain for late-comers. Some like to sit together while others prefer to leave empty seats”.66
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1H+
1H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) e−).34
e−).34The generality of the Frumkin isotherm is perhaps surprising. It seems to apply equally well to the highly ordered RuO2(110) films, to hydrous, partly amorphous IrO2 and Co/phosphate films, and the IrOx NPs studied here. The 1.7 nm IrOx NPs are only a few unit cells across, and a high fraction of surface sites are edges, corners, and defects, if they are faceted at all. Not-so-different results have been obtained with oxides, various single crystal metal surfaces,72 and metal nanoparticles.24 The Frumkin isotherm was developed based on inter-adsorbate interactions, but these observations suggest a generality that transcends any single cause.
The Frumkin isotherm is the first-order approximation to a Langmuir isotherm66 and can be thought of as the first term of a Taylor series expansion in θ. Taking a0 in eqn (20) as the Langmuir isotherm (eqn (18)), and x as a small perturbation from that function, the Frumkin linear Cθ term is simply the a1x term. From this perspective, any small effect that causes deviation from ideality should, to a first approximation, yield a Frumkin isotherm. The observation of a Frumkin isotherm does not provide insight into the origin of the non-ideality.
| f(x) = a0 + a1x + a2x2 + a3x3 + … | (20) |
The breadth of the isotherm is likely due to a variety of factors. These include (some of these overlap): (i) a diversity of surface sites, especially due to the small size of the NPs, (ii) the range of NP sizes, (iii) other surface heterogeneities, for instance due to surface O/OH/OH2 groups, (iv) inter-adsorbate repulsion (as in Frumkin's original derivation), (v) changes in the structure and bonding of the nanoparticles upon H addition, (vi) variability in the points of zero charge, both among different NPs and upon H addition, and (vii) the presence of surface dipoles. The very broad isotherm for H on CeO2 nanoparticles was suggested to be due to increasing lattice strain as individual Ce4+ ions are reduced to localized and form much larger Ce3+ ions,26 but this is not the case for IrOx due to its metallic bonding and delocalized electrons.
Still, it is remarkable that the isotherm data follow a single, smooth curve. On an irregular surface such as a small NP, one might have expected a subset of the sites to have much higher or lower affinities for H, which would have led to a more irregular experimental isotherm. Instead, for most materials, the Frumkin C(θ − 0.5) seems to well describe an average over all the potential non-idealities.
3.3 Connecting H- and O-atom thermochemistry
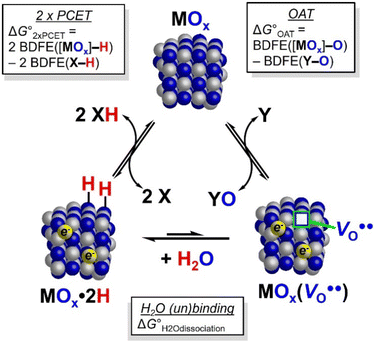
OAT and HAT/PCET are two quite different classes of reactions, yet the products are related simply by a water molecule, as illustrated in Scheme 5.67 This stoichiometric connection has long been recognized but is often not highlighted in discussion.67 Both double H-atom addition and removal of an O-atom result in a 2e− reduction. OAT formally gives a vacancy in the NP , which in aqueous media is rapidly filled by H2O. This forms the same MOx·2H as HAT/PCET to the NP. This equivalence is evident for the IrOx reactions with H2Q and PAr3 discussed above, in their identical titration endpoints and amounts of proton uptake.
, which in aqueous media is rapidly filled by H2O. This forms the same MOx·2H as HAT/PCET to the NP. This equivalence is evident for the IrOx reactions with H2Q and PAr3 discussed above, in their identical titration endpoints and amounts of proton uptake.
The triangle scheme in Scheme 5 also serves as a thermochemical cycle between PCET and OAT reactivity. Because it is a closed loop, the sum of the three ΔG°s around the cycle must be zero, as shown in eqn (21), moving counterclockwise from the oxidized form at the top. This equation allows the calculation of any free energy from the other two. While more work is needed to experimentally define the ΔG°(OAT to Y) and ΔG°(H2O binding to  ), this connection between HAT and OAT provides valuable insights.
), this connection between HAT and OAT provides valuable insights.
 | (21) |
The ΔG° for H2O dissociation (bottom of Scheme 5) is thus a key parameter in connecting HAT and OAT thermochemistry.58 Unfortunately, we have found little information about H2O binding energies in aqueous solutions when the O being lost is formally a lattice oxygen [the product of OAT] (SI Section S7). Calculations of H2O binding energies for an ideal, oxidized, stoichiometric (IrO2)115(H2O)88 nanoparticle in vacuo gave  values from −26 to −36 kcal mol−1.76 The binding energy for H2O to a bare RuO2 surface was calculated to be −33 kcal mol−1.77
values from −26 to −36 kcal mol−1.76 The binding energy for H2O to a bare RuO2 surface was calculated to be −33 kcal mol−1.77
Following this kind of analysis, the OAT thermochemistry of the IrOx NPs is bracketed by the ladder of reactions in Scheme 4 above (see SI Section S7). This approach has long been used in molecular and solid-state chemistries. For instance, Soper et al. bracketed the BDE of a VV![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O bond in [(Phisq)(Phibq)VIV(O)Cl] to be 73 ± 14 kcal mol−1 (ref. 78) by reacting its complex with seven OAT reagents with 60 < BDE[Y–O] < 133 kcal mol−1.21,79 The free energies for loss of O2 from solid binary metal oxides as a function of temperature were summarized in an ‘Ellingham diagram’ 80 years ago.80
O bond in [(Phisq)(Phibq)VIV(O)Cl] to be 73 ± 14 kcal mol−1 (ref. 78) by reacting its complex with seven OAT reagents with 60 < BDE[Y–O] < 133 kcal mol−1.21,79 The free energies for loss of O2 from solid binary metal oxides as a function of temperature were summarized in an ‘Ellingham diagram’ 80 years ago.80
For IrOx NPs, the driving force for the OAT reactions includes both the OxIr–O BDFE and the ΔG° for H2O binding. This H2O binding is specifically to the oxygen vacancy resulting from Ir–O bond cleavage (Scheme 5). With this approach, the ladder of OAT reactions in Scheme 4 brackets the combination of the BDFE(OxIr–O) and the  (eqn (22), in kcal mol−1). Unfortunately, this analysis gives only a broad range because reagents with intermediate Y–O BDFEs, such as pyO, were kinetically unreactive. If
(eqn (22), in kcal mol−1). Unfortunately, this analysis gives only a broad range because reagents with intermediate Y–O BDFEs, such as pyO, were kinetically unreactive. If  is taken to be −30 kcal mol−1, BDFE(Ir–O) is estimated to be 93 ± 24 kcal mol−1 (eqn (23))
is taken to be −30 kcal mol−1, BDFE(Ir–O) is estimated to be 93 ± 24 kcal mol−1 (eqn (23))
 | (22) |
| 117 > BDFE(OxIr–O) > 70, or BDFE(OxIr–O) = 93 ± 24 kcal mol−1 | (23) |
Thermochemical cycles such as Scheme 5 can be used in multiple ways. For instance, Scheme 6 shows how the ΔG° for the pyridine N-oxide (pyO(aq)) oxidation of fully reduced IrOxHn NPs (eqn (25)) can be determined without knowledge of the  . The O removed from pyO is transferred to solution (eqn (24)) and then picks up two H from the reduced IrOx NPs (eqn (25) and (26)) to form the product water. The ΔG° (eqn (27)) is estimated to be −13 kcal mol−1. More details and references are given in SI Section S7, together with a related calculation for OAT to methionine.
. The O removed from pyO is transferred to solution (eqn (24)) and then picks up two H from the reduced IrOx NPs (eqn (25) and (26)) to form the product water. The ΔG° (eqn (27)) is estimated to be −13 kcal mol−1. More details and references are given in SI Section S7, together with a related calculation for OAT to methionine.
Under the pH 1.83 reaction conditions, the product py of reaction (27) will be protonated, which will add more driving force. Yet this reaction does not proceed. The absence of reaction is therefore due to a kinetic barrier (ΔG‡) and not due to the reaction being uphill. This contrasts with the HAT/PCET reactions above that seem to parallel the thermochemistry in all cases.
3.4 Non-ideality in O-atom thermochemistry
The next layer of the thermochemical analysis of O-atom transfer reactions is the likelihood of broad ranges of the OAT free energy parameters. Scheme 5 and eqn (21) show that OAT is related to twice the HAT energetics, so to a first approximation the spread of Ir–O BDFEs should be twice that of the IrO–H BDFEs, or ca. ∼13 kcal mol−1. This follows from an O-atom being a 2e− oxidant vs. the 1e− reductant H˙.The range of H2O binding energies noted above will also contribute to the non-ideality of O-atom binding. The calculations of the ideal (IrO2)115(H2O)88 NP mentioned above found eleven different Ir binding sites for water and that hydrogen-bonding networks were important.76 The diversity of sites, H-bonding networks, and binding energies is likely even larger for our hydrous IrOx NPs in aqueous solution. Perhaps most importantly, we have yet no information about which of the possible water dissociations form a vacancy that can accept an O atom.
A combined surface-science and DFT study examined 4-layer (9 Å) films of rutile IrO2(100) grown on an Ir(111) crystal surface.81 The surface was “oxygen-rich”, with roughly one O per Ir beyond the stoichiometric IrO2(100) surface, in the less-stable atop binding configuration. BDFE(Ir–O) for these extra O atoms was very small, decreasing from 23–18 kcal mol−1 with increasing O coverage. Considering all the surface Ir–O bonds, the calculations showed a 47 kcal mol−1 binding energy range.
These calculations show the remarkable diversity and complexity of IrO2 single crystal and idealized NP surfaces. For our IrOx NPs, the chemistry of Ir–O bonds will be more complex than that of IrO–H bonds.
4. Conclusions
The redox reactions of aqueous colloidal IrOx NPs are non-ideal in both their stoichiometry and thermochemistry.These 1.7 nm-diameter NPs in acidic water have been developed as a simple model system for interfacial reactivity at a redox-active metal oxide. Focusing only on the IrIV/IrIII redox couple, various PCET/HAT (e− + H+) and oxygen-atom transfer (OAT) reactions are reversible, with ∼80% of the Ir ions electroactive. The colloids can be set at any average Ir redox level, and the same thermodynamic product is formed with each reagent.
The reactions of the IrOx NPs all have the same ‘super-Nernstian’ stoichiometry, with 1.3 protons adding to the IrOx surface with each electron added. The non-integer stoichiometry was demonstrated electrochemically and by directly measuring the H+ taken up upon reduction. Non-integer PCET is common for hydrous oxides,30,56–58 but its origin is not well understood.
Spectroelectrochemical studies showed that changes in the [IrIV]/[IrIII] ratio in the NPs required much larger changes in applied potential than predicted by the Nernst equation. The data fit well to a non-ideal Frumkin isotherm. Approximating the reactions as H-atom transfers (1H+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1e−), the distribution of BDFE([IrOx]–H) has a midpoint of 76 kcal mol−1 and a spread of 6.5 kcal mol−1 as a function of H saturation (θH). The isotherm for O-atom removal from the IrOx NPs is estimated to be much wider than that for HAT, due in part to OAT being a formal 2e− transfer.
1e−), the distribution of BDFE([IrOx]–H) has a midpoint of 76 kcal mol−1 and a spread of 6.5 kcal mol−1 as a function of H saturation (θH). The isotherm for O-atom removal from the IrOx NPs is estimated to be much wider than that for HAT, due in part to OAT being a formal 2e− transfer.
The non-ideal stoichiometry and thermochemistry are core features of the redox chemistry of this ‘simple’ IrOx NP model system and many others. Oxide–water interfaces should not be assumed to follow ideal Nernstian or Langmuirian behaviour (as is often done). The thermochemistry very nicely fits a simple two-parameter Frumkin isotherm, and the reason for the generality of this isotherm is discussed. The accuracy of the mathematically simple Frumkin isotherm provides quantitative understanding and provides a path forward to more realistic models. New models are needed because the non-ideality of (nano)materials is likely an advantage in many applications.
Author contributions
The conception of the project was by J. L. L. and J. M. M., and the experiments and analyses were performed by J. L. L. and S. S. J. M. M. was the research advisor, assisting with project goal selection, project focus, and experiment interpretation. All authors participated in manuscript preparation and editing, with final editing by J. M. M.Conflicts of interest
There are no conflicts to declare.Data availability
The datasets supporting this article have been uploaded as part of the supplementary information (SI). Supplementary information: reagents, NP synthesis, characterization of NPs, UV-vis and NMR spectra, thermochemical tables of Y–O substrates. See DOI: https://doi.org/10.1039/d6sc00492j.Acknowledgements
This research made extensive use of Yale University's Chemical and Biophysical Instrumentation Center (CBIC; RRID:SCR_021738; equipment was purchased with funds from Yale University). We also thank the Yale Institute for Nanoscience and Quantum Engineering (YINQE) and Dr Sungwoo Sohn, as well as the Yale Analytical and Stable Isotope Center (YASIC) and Jonas Karosas. The work was funded predominantly by the U.S. Department of Energy, Office of Science, Basic Energy Sciences (award No. DE-SC0021298).Notes and references
- G. D. Halsey, The Role of Surface Heterogeneity in Adsorption, in Advances in Catalysis, Elsevier, 1952, vol. 4, pp 259–269, DOI:10.1016/S0360-0564(08)60616-1.
- M. Boudart, Kinetics on Ideal and Real Surfaces, AIChE J., 1956, 2(1), 62–64, DOI:10.1002/aic.690020113.
- G. E. Brown, V. E. Henrich, W. H. Casey, D. L. Clark, C. Eggleston, A. Felmy, D. W. Goodman, M. Grätzel, G. Maciel, M. I. McCarthy, K. H. Nealson, D. A. Sverjensky, M. F. Toney and J. M. Zachara, Metal Oxide Surfaces and Their Interactions with Aqueous Solutions and Microbial Organisms, Chem. Rev., 1999, 99(1), 77–174, DOI:10.1021/cr980011z.
- O. Q. Carvalho, P. Adiga, S. K. Murthy, J. L. Fulton, O. Y. Gutiérrez and K. A. Stoerzinger, Understanding the Role of Surface Heterogeneities in Electrosynthesis Reactions, iScience, 2020, 23(12), 101814, DOI:10.1016/j.isci.2020.101814.
- J. M. Mayer, Site Heterogeneity and Broad Surface-Binding Isotherms in Modern Catalysis: Building Intuition beyond the Sabatier Principle, J. Catal., 2024, 439, 115725, DOI:10.1016/j.jcat.2024.115725.
- L. D. Burke and M. E. G. Lyons, Electrochemistry of Hydrous Oxide Films, in Modern Aspects of Electrochemistry, ed.R. E. White, J. O. Bockris and B. E. Conway, Springer US, Boston, MA, 1986, vol. 18, pp 169–248, DOI:10.1007/978-1-4613-1791-3.
- D.-Y. Kuo, J. K. Kawasaki, J. N. Nelson, J. Kloppenburg, G. Hautier, K. M. Shen, D. G. Schlom and J. Suntivich, Influence of Surface Adsorption on the Oxygen Evolution Reaction on IrO2 (110), J. Am. Chem. Soc., 2017, 139(9), 3473–3479, DOI:10.1021/jacs.6b11932.
- C. Liang, Y. Katayama, Y. Tao, A. Morinaga, B. Moss, V. Celorrio, M. Ryan, I. E. L. Stephens, J. R. Durrant and R. R. Rao, Role of Electrolyte pH on Water Oxidation for Iridium Oxides, J. Am. Chem. Soc., 2024, 146(13), 8928–8938, DOI:10.1021/jacs.3c12011.
- N. P. Holzapfel, V. Augustyn and V. Balland, Fundamentals of Proton-Insertion Coupled Electron Transfer (PICET) in Metal Oxides for Aqueous Batteries, ACS Energy Lett., 2025, 10(3), 1143–1164, DOI:10.1021/acsenergylett.4c03076.
- C. N. Valdez, A. M. Schimpf, D. R. Gamelin and J. M. Mayer, Proton-Controlled Reduction of ZnO Nanocrystals: Effects of Molecular Reductants, Cations, and Thermodynamic Limitations, J. Am. Chem. Soc., 2016, 138(4), 1377–1385, DOI:10.1021/jacs.5b12182.
- J. L. Peper, N. E. Gentry, B. Boudy and J. M. Mayer, Aqueous TiO2 Nanoparticles React by Proton-Coupled Electron Transfer, Inorg. Chem., 2022, 61(2), 767–777, DOI:10.1021/acs.inorgchem.1c03125.
- R. G. Agarwal and J. M. Mayer, Coverage-Dependent Rate-Driving Force Relationships: Hydrogen Transfer from Cerium Oxide Nanoparticle Colloids, J. Am. Chem. Soc., 2022, 144(45), 20699–20709, DOI:10.1021/jacs.2c07988.
- N. E. Gentry, A. Kurimoto, K. Cui, J. L. Cleron, C. M. Xiang, S. Hammes-Schiffer and J. M. Mayer, Hydrogen on Colloidal Gold Nanoparticles, J. Am. Chem. Soc., 2024, 146(21), 14505–14520, DOI:10.1021/jacs.4c00507.
- J. L. Lee, N. E. Gentry, J. L. Peper, S. Hetzel, C. Quist, F. S. Menges and J. M. Mayer, Oxygen Atom Transfer Reactions of Colloidal Metal Oxide Nanoparticles, ACS Nano, 2025, 19(10), 10289–10300, DOI:10.1021/acsnano.4c17955.
- Y. Zhao, E. A. Hernandez-Pagan, N. M. Vargas-Barbosa, J. L. Dysart and T. E. Mallouk, A High Yield Synthesis of Ligand-Free Iridium Oxide Nanoparticles with High Electrocatalytic Activity, J. Phys. Chem. Lett., 2011, 2(5), 402–406, DOI:10.1021/jz200051c.
- J. K. Nørskov, T. Bligaard, A. Logadottir, S. Bahn, L. B. Hansen, M. Bollinger, H. Bengaard, B. Hammer, Z. Sljivancanin, M. Mavrikakis, Y. Xu, S. Dahl and C. J. H. Jacobsen, Universality in Heterogeneous Catalysis, J. Catal., 2002, 209(2), 275–278, DOI:10.1006/jcat.2002.3615.
- A. J. Medford, A. Vojvodic, J. S. Hummelshøj, J. Voss, F. Abild-Pedersen, F. Studt, T. Bligaard, A. Nilsson and J. K. Nørskov, From the Sabatier Principle to a Predictive Theory of Transition-Metal Heterogeneous Catalysis, J. Catal., 2015, 328, 36–42, DOI:10.1016/j.jcat.2014.12.033.
- H. Ooka, J. Huang and K. S. Exner, The Sabatier Principle in Electrocatalysis: Basics, Limitations, and Extensions, Front. Energy Res., 2021, 9, 654460, DOI:10.3389/fenrg.2021.654460.
- R. G. Agarwal, S. C. Coste, B. D. Groff, A. M. Heuer, H. Noh, G. A. Parada, C. F. Wise, E. M. Nichols, J. J. Warren and J. M. Mayer, Free Energies of Proton-Coupled Electron Transfer Reagents and Their Applications, Chem. Rev., 2022, 122(1), 1–49, DOI:10.1021/acs.chemrev.1c00521.
- F. R. Mayo, Rate Constants in the Gas-Phase Oxidation of Alkanes and Alkyl Radicals, in Advcances in Chemistry, Oxidation of organic compounds, American Chemical Society, Washington, DC, 1968, vol. 2 Search PubMed.
- R. H. Holm, Metal-Centered Oxygen Atom Transfer Reactions, Chem. Rev., 1987, 87(6), 1401–1449 CrossRef CAS.
- J. M. Mayer, Understanding Hydrogen Atom Transfer: From Bond Strengths to Marcus Theory, Acc. Chem. Res., 2011, 44(1), 36–46, DOI:10.1021/ar100093z.
- C. T. Campbell, Energies of Adsorbed Catalytic Intermediates on Transition Metal Surfaces: Calorimetric Measurements and Benchmarks for Theory, Acc. Chem. Res., 2019, 52(4), 984–993, DOI:10.1021/acs.accounts.8b00579.
- M. García-Diéguez, D. D. Hibbitts and E. Iglesia, Hydrogen Chemisorption Isotherms on Platinum Particles at Catalytic Temperatures: Langmuir and Two-Dimensional Gas Models Revisited, J. Phys. Chem. C, 2019, 123(13), 8447–8462, DOI:10.1021/acs.jpcc.8b10877.
- Z. Paal and P. G. Menon, Hydrogen Effects in Catalysis, CRC Press, 2020, DOI:10.1201/9781003065807.
- B. E. Conway and J. O. Bockris, Electrolytic Hydrogen Evolution Kinetics and Its Relation to the Electronic and Adsorptive Properties of the Metal, J. Chem. Phys., 1957, 26(3), 532–541, DOI:10.1063/1.1743339.
- T. Bligaard, J. K. Nørskov, S. Dahl, J. Matthiesen, C. H. Christensen and J. Sehested, The Brønsted–Evans–Polanyi Relation and the Volcano Curve in Heterogeneous Catalysis, J. Catal., 2004, 224(1), 206–217, DOI:10.1016/j.jcat.2004.02.034.
- J. Zheng, W. Sheng, Z. Zhuang, B. Xu and Y. Yan, Universal Dependence of Hydrogen Oxidation and Evolution Reaction Activity of Platinum-Group Metals on pH and Hydrogen Binding Energy, Sci. Adv., 2016, 2(3), e1501602, DOI:10.1126/sciadv.1501602.
- N. Dubouis and A. Grimaud, The Hydrogen Evolution Reaction: From Material to Interfacial Descriptors, Chem. Sci., 2019, 10(40), 9165–9181, 10.1039/C9SC03831K.
- A. A. Gambardella, N. S. Bjorge, V. K. Alspaugh and R. W. Murray, Voltammetry of Diffusing 2 Nm Iridium Oxide Nanoparticles, J. Phys. Chem. C, 2011, 115(44), 21659–21665, DOI:10.1021/jp206987z.
- R. G. Agarwal, H. J. Kim and J. M. Mayer, Nanoparticle O-H Bond Dissociation Free Energies from Equilibrium Measurements of Cerium Oxide Colloids, J. Am. Chem. Soc., 2021, 143(7), 2896–2907, DOI:10.1021/jacs.0c12799.
- H. Noh and J. M. Mayer, Medium-Independent Hydrogen Atom Binding Isotherms of Nickel Oxide Electrodes, Chem, 2022, 8(12), 3324–3345, DOI:10.1016/j.chempr.2022.08.018.
- C. Liang, R. R. Rao, K. L. Svane, J. H. L. Hadden, B. Moss, S. B. Scott, M. Sachs, J. Murawski, A. M. Frandsen, D. J. Riley, M. P. Ryan, J. Rossmeisl, J. R. Durrant and I. E. L. Stephens, Unravelling the Effects of Active Site Density and Energetics on the Water Oxidation Activity of Iridium Oxides, Nat. Catal., 2024, 7(7), 763–775, DOI:10.1038/s41929-024-01168-7.
- L. Zhang, J. Kloppenburg, C.-Y. Lin, L. Mitrovic, S. Gelin, I. Dabo, D. G. Schlom, J. Suntivich and G. Hautier, Atomistic Understanding of Hydrogen Coverage on RuO2 (110) Surface under Electrochemical Conditions from Ab Initio Statistical Thermodynamics, J. Phys. Chem. C, 2025, 129(8), 4043–4051, DOI:10.1021/acs.jpcc.4c07229.
- T. Reier, D. Teschner, T. Lunkenbein, A. Bergmann, S. Selve, R. Kraehnert, R. Schlögl and P. Strasser, Electrocatalytic Oxygen Evolution on Iridium Oxide: Uncovering Catalyst-Substrate Interactions and Active Iridium Oxide Species, J. Electrochem. Soc., 2014, 161(9), F876–F882, DOI:10.1149/2.0411409jes.
- H. Yu, F. Liao, W. Zhu, K. Qin, J. Shi, M. Ma, Y. Li, M. Fang, J. Su, B. Song, L. Li, R. R. Zairov, Y. Ji, M. Shao and Q. Shao, Two-Dimensional Amorphous Iridium Oxide for Acidic Oxygen Evolution Reaction, ChemCatChem, 2023, 15(19), e202300737, DOI:10.1002/cctc.202300737.
- R. V. Mom, L. J. Falling, O. Kasian, G. Algara-Siller, D. Teschner, R. H. Crabtree, A. Knop-Gericke, K. J. J. Mayrhofer, J.-J. Velasco-Vélez and T. E. Jones, Operando Structure–Activity–Stability Relationship of Iridium Oxides during the Oxygen Evolution Reaction, ACS Catal., 2022, 12(9), 5174–5184, DOI:10.1021/acscatal.1c05951.
- J. Gao, Y. Liu, B. Liu and K. W. Huang, Progress of Heterogeneous Iridium-Based Water Oxidation Catalysts, ACS Nano, 2022, 16(11), 17761–17777, DOI:10.1021/acsnano.2c08519.
- R. Sharma, M. A. Karlsen, P. Morgen, J. Chamier, D. B. Ravnsbæk and S. M. Andersen, Crystalline Disorder, Surface Chemistry, and Their Effects on the Oxygen Evolution Reaction (OER) Activity of Mass-Produced Nanostructured Iridium Oxides, ACS Appl. Energy Mater., 2021, 4(3), 2552–2562, DOI:10.1021/acsaem.0c03127.
- C. Bozal-Ginesta, R. R. Rao, C. A. Mesa, X. Liu, S. A. J. Hillman, I. E. L. Stephens and J. R. Durrant, Redox-State Kinetics in Water-Oxidation IrOx Electrocatalysts Measured by Operando Spectroelectrochemistry, ACS Catal., 2021, 11(24), 15013–15025, DOI:10.1021/acscatal.1c03290.
- C. Bozal-Ginesta, R. R. Rao, C. A. Mesa, Y. Wang, Y. Zhao, G. Hu, D. Antón-García, I. E. L. Stephens, E. Reisner, G. W. Brudvig, D. Wang and J. R. Durrant, Spectroelectrochemistry of Water Oxidation Kinetics in Molecular versus Heterogeneous Oxide Iridium Electrocatalysts, J. Am. Chem. Soc., 2022, 144(19), 8454–8459, DOI:10.1021/jacs.2c02006.
- G. Kwon, S. H. Chang, J. E. Heo, K. J. Lee, J. K. Kim, B. G. Cho, T. Y. Koo, B. J. Kim, C. Kim, J. H. Lee, S. M. Bak, K. A. Beyer, H. Zhong, R. J. Koch, S. Hwang, L. M. Utschig, X. Huang, G. Hu, G. W. Brudvig, D. M. Tiede and J. Kim, Experimental Verification of Ir 5d Orbital States and Atomic Structures in Highly Active Amorphous Iridium Oxide Catalysts, ACS Catal., 2021, 11(15), 10084–10094, DOI:10.1021/acscatal.1c00818.
- J. M. Kahk, C. G. Poll, F. E. Oropeza, J. M. Ablett, D. Céolin, J.-P. Rueff, S. Agrestini, Y. Utsumi, K. D. Tsuei, Y. F. Liao, F. Borgatti, G. Panaccione, A. Regoutz, R. G. Egdell, B. J. Morgan, D. O. Scanlon and D. J. Payne, Understanding the Electronic Structure of IrO2 Using Hard-X-Ray Photoelectron Spectroscopy and Density-Functional Theory, Phys. Rev. Lett., 2014, 112(11), 117601, DOI:10.1103/PhysRevLett.112.117601.
- L. J. Yellowlees and K. G. Macnamara, Iridium, in Comprehensive Coordination Chemistry II, 2003, vol. 6, pp. 147–246 Search PubMed.
- R. H. Crabtree, High Oxidation State Complexes of Rhodium and Iridium, in Comprehensive Coordination Chemistry III, 2021, pp. 159–170 Search PubMed.
- H. Ooka, Y. Wang, A. Yamaguchi, M. Hatakeyama, S. Nakamura, K. Hashimoto and R. Nakamura, Legitimate Intermediates of Oxygen Evolution on Iridium Oxide Revealed by in Situ Electrochemical Evanescent Wave Spectroscopy, Phys. Chem. Chem. Phys., 2016, 18(22), 15199–15204, 10.1039/C6CP02385A.
- S. Gottesfeld, J. D. E. McIntyre, G. Beni and J. L. Shay, Electrochromism in Anodic Iridium Oxide Films, Appl. Phys. Lett., 1978, 33(2), 208–210, DOI:10.1063/1.90277.
- Z. Pavlovic, C. Ranjan, Q. Gao, M. Van Gastel and R. Schlögl, Probing the Structure of a Water-Oxidizing Anodic Iridium Oxide Catalyst Using Raman Spectroscopy, ACS Catal., 2016, 6(12), 8098–8105, DOI:10.1021/acscatal.6b02343.
- Y. Mo, I. C. Stefan, W. B. Cai, J. Dong, P. Carey and D. A. Scherson, In Situ Iridium LIII-Edge X-Ray Absorption and Surface Enhanced Raman Spectroscopy of Electrodeposited Iridium Oxide Films in Aqueous Electrolytes, J. Phys. Chem. B, 2002, 106(14), 3681–3686, DOI:10.1021/jp014452p.
- Y. Sasaki, H. Kato and A. Kudo, [Co(bpy)3)]3+/2+ and [Co(phen)3]3+/2+ Electron Mediators for Overall Water Splitting under Sunlight Irradiation Using Z-Scheme Photocatalyst System, J. Am. Chem. Soc., 2013, 135(14), 5441–5449, DOI:10.1021/ja400238r.
- D. Dalzel Perrin, Dissociation Constants of Inorganic Acids and Bases in Aqueous Solution, Pergamon, New York, New York, 1982 Search PubMed.
- A. D. Awtrey and R. E. Connick, The Absorption Spectra of I2, I3−, I−, IO3−, S4 O6= and S2 O3=. Heat of the Reaction I3− = I2 + I−, J. Am. Chem. Soc., 1951, 73(4), 1842–1843, DOI:10.1021/ja01148a504.
- T. Labasque, C. Chaumery, A. Aminot and G. Kergoat, Spectrophotometric Winkler Determination of Dissolved Oxygen: Re-Examination of Critical Factors and Reliability, Mar. Chem., 2004, 88(1–2), 53–60, DOI:10.1016/j.marchem.2004.03.004.
- M. F. Yuen, I. Lauks and W. C. Dautremont-Smith, pH Dependent Voltammetry of Iridium Oxide Films, Solid State Ionics, 1983, 11(1), 19–29, DOI:10.1016/0167-2738(83)90058-9.
- S. Fleischmann, J. B. Mitchell, R. Wang, C. Zhan, D. E. Jiang, V. Presser and V. Augustyn, Pseudocapacitance: From Fundamental Understanding to High Power Energy Storage Materials, Chem. Rev., 2020, 120(14), 6738–6782, DOI:10.1021/acs.chemrev.0c00170.
- L. D. Burke and T. A. M. Twomey, Influence of Ph on the Redox Behaviour of Hydrous Nickel Oxide, J. Electroanal. Chem. Interfacial Electrochem., 1982, 134(2), 353–362, DOI:10.1016/0022-0728(82)80013-2.
- C. Bock and V. I. Birss, Anion and Water Involvement in Hydrous Ir Oxide Redox Reactions in Acidic Solutions, J. Electroanal. Chem., 1999, 475(1), 20–27, DOI:10.1016/S0022-0728(99)00330-7.
- K. A. Stoerzinger, R. R. Rao, X. R. Wang, W. T. Hong, C. M. Rouleau and Y. Shao-Horn, The Role of Ru Redox in pH-Dependent Oxygen Evolution on Rutile Ruthenium Dioxide Surfaces, Chem, 2017, 2(5), 668–675, DOI:10.1016/j.chempr.2017.04.001.
- L. D. Burke and D. P. Whelan, A Voltammetric Investigation of the Charge Storage Reactions of Hydrous Iridium Oxide Layers, J. Electroanal. Chem. Interfacial Electrochem., 1984, 162(1–2), 121–141 CrossRef CAS.
- D.-Y. Kuo, J. K. Kawasaki, J. N. Nelson, J. Kloppenburg, G. Hautier, K. M. Shen, D. G. Schlom and J. Suntivich, Influence of Surface Adsorption on the Oxygen Evolution Reaction on IrO2 (110), J. Am. Chem. Soc., 2017, 139(9), 3473–3479, DOI:10.1021/jacs.6b11932.
- M. A. Petit and V. Plichon, Anodic Electrodeposition of Iridium Oxide Films, J. Electroanal. Chem., 1998, 444(2), 247–252, DOI:10.1016/S0022-0728(97)00570-6.
- C. Vanleugenhaghe, G. Valensi, M. Pourbaix and M. Pourbaix, Iodine, in Atlas of Electrochemical Equilibria in Aqueous Solutions, NACE International, 1974, pp. 614–626 Search PubMed.
- E. Gileadi, Adsorption in Electrochemistry, in Electrosorption, Springer, 1967, pp. 1–18 Search PubMed.
- A. J. Bard and L. R. Faulkner, Double-Layer Structure and Adsorption, in Electrochemical Methods: Fundamentals and Applications, John Wiley & Sons, Inc., 2001, pp. 534–579 Search PubMed.
- P. Delahay, Double Layer and Electrode Kinetics, Interscience Publishers, 1965 Search PubMed.
- E. Gileadi, Electrode Kinetics for Chemists, Chemical Engineers, and Materials Scientists, John Wiley & Sons, 1996, pp. 262–266, The discussion here, and the quoted text, are from Search PubMed.
- T. Norby, M. Widerøe, R. Glöckner and Y. Larring, Hydrogen in Oxides, Dalton Trans., 2004,(19), 3012–3018, 10.1039/B403011G.
- M. Pourbaix, Atlas of Electrochemical Equilibria in Aqueous Solutions, National Association of Corrosion Engineers, 1974 Search PubMed.
- B. D. McCarthy and J. L. Dempsey, Decoding Proton-Coupled Electron Transfer with Potential-pKa Diagrams, Inorg. Chem., 2017, 56(3), 1225–1231, DOI:10.1021/acs.inorgchem.6b02325.
- B. Hu, D.-Y. Kuo, H. Paik, D. G. Schlom and J. Suntivich, Enthalpy and Entropy of Oxygen Electroadsorption on RuO2(110) in Alkaline Media, J. Chem. Phys., 2020, 152(9), 094704, DOI:10.1063/1.5139049.
- Grokipedia v2.0., https://grokipedia.com/page/alexander_frumkin, accessed January 14, 2026 Search PubMed.
- K. Christmann, Interaction of Hydrogen with Solid Surfaces, Surf. Sci. Rep., 1988, 9(1–3), 1–163, DOI:10.1016/0167-5729(88)90009-X.
- R. Rizo, E. Sitta, E. Herrero, V. Climent and J. M. Feliu, Towards the Understanding of the Interfacial pH Scale at Pt(111) Electrodes, Electrochim. Acta, 2015, 162, 138–145, DOI:10.1016/j.electacta.2015.01.069.
- B. Moss, K. L. Svane, D. Nieto-Castro, R. R. Rao, S. B. Scott, C. Tseng, M. Sachs, A. Pennathur, C. Liang, L. I. Oldham, E. Mazzolini, L. Jurado, G. Sankar, S. Parry, V. Celorrio, J. M. Dawlaty, J. Rossmeisl, J. R. Galán-Mascarós, I. E. L. Stephens and J. R. Durrant, Cooperative Effects Drive Water Oxidation Catalysis in Cobalt Electrocatalysts through the Destabilization of Intermediates, J. Am. Chem. Soc., 2024, 146(13), 8915–8927, DOI:10.1021/jacs.3c11651.
- N. J. Gibson, G. A. Parada, B. Q. Mercado and J. M. Mayer, Colloidal Hexagonal Tungsten Oxide Nanorods: Synthesis, Characterization, and Proton-Coupled Electron Transfer, Inorg. Chem., 2026, 65(4), 2659–2668, DOI:10.1021/acs.inorgchem.5c06029.
- D. González, M. Sodupe, L. Rodríguez-Santiago and X. Solans-Monfort, Surface Morphology Controls Water Dissociation on Hydrated IrO2 nanoparticles, Nanoscale, 2021, 13(34), 14480–14489, 10.1039/d1nr03592d.
- R. Mu, D. C. Cantu, X. Lin, V. A. Glezakou, Z. Wang, I. Lyubinetsky, R. Rousseau and Z. Dohnálek, Dimerization Induced Deprotonation of Water on RuO2(110), J. Phys. Chem. Lett., 2014, 5(19), 3445–3450, DOI:10.1021/jz501810g.
- A. G. Hill, M. C. Castillo, J. Bacsa, K. S. Otte and J. D. Soper, Redox-Active Ligands Permit Multielectron O2 Homolysis and O-Atom Transfer at Exceptionally High-Valent Vanadyl Complexes, J. Am. Chem. Soc., 2025, 147(16), 13356–13369, DOI:10.1021/jacs.4c18305.
- R. H. Holm and J. P. Donahue, A Thermodynamic Scale for Oxygen Atom Transfer Reactions, Polyhedron, 1993, 12, 571–589 CrossRef CAS.
- H. J. T. Ellingham, Transactions and Communications, J. Chem. Technol. Biotechnol., 1944, 63(5), 125–160, DOI:10.1002/jctb.5000630501.
- R. Rai, T. Li, Z. Liang, M. Kim, A. Asthagiri and J. F. Weaver, Growth and Termination of a Rutile IrO2(100) Layer on Ir(111), Surf. Sci., 2016, 652, 213–221, DOI:10.1016/j.susc.2016.01.018.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |