Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Mechanism governing the formation of atomically precise dithiolate-protected gold nanoclusters
Sara
Yoshikawa†
ab,
Tokuhisa
Kawawaki†
 *ab,
Sakiat
Hossain
b and
Yuichi
Negishi
*ab,
Sakiat
Hossain
b and
Yuichi
Negishi
 *ab
*ab
aInstitute of Multidisciplinary Research for Advanced Materials, Tohoku University, Katahira 2-1-1, Aoba-ku, Sendai 980-8577, Japan. E-mail: tokuhisa.kawawaki.d8@tohoku.ac.jp; yuichi.negishi.a8@tohoku.ac.jp
bCarbon Value Research Center, Research Institute for Science and Technology, Tokyo University of Science, Kagurazaka, Shinjuku-ku, Tokyo 162-8601, Japan
First published on 13th March 2026
Abstract
Atomically precise metal nanoclusters (NCs) stabilized by organic ligands are promising functional materials in various fields owing to their unique geometric and electronic structures. However, many such NCs exhibit insufficient stability, e.g., processes such as alloying can induce structural destabilization. Gold (Au) NCs can be protected by introducing multi-site thiolates (SR), which form exceptionally strong Au–S bonds, thus enhancing the stability of the NCs and expanding their practical applicability. However, multi-site SR protection using bidentate ligands often leads to undesirable polymerization due to inter-NC cross-linking. The present study addresses this issue by elucidating the mechanism governing the formation of Au NCs co-protected by both bidentate (SR′S) and monodentate (SR) ligands. The key impacts of ligand flexibility and site-specific exchange kinetics are identified, thereby providing crucial insights to support the strategic design and synthesis of stable, multi-site SR-protected Au NCs with rigid, well-defined architectures.
Introduction
Ligand-protected metal nanoclusters (NCs) with atomically precise composition exhibit unique geometric and electronic structures distinct from those of the corresponding bulk metals or larger metal nanoparticles (NPs).1–9 These structural features endow them with exceptional properties and wide applicability in diverse areas, including catalysis, luminescence, and magnetic materials.10–17 Notably, the high-performance properties of such NCs are directly correlated with their precise atomic arrangements, which can be elucidated through single-crystal X-ray diffraction (SC-XRD) analysis and theoretical calculations. This structural clarity is crucial because it enables a fundamental understanding of structure–property relationships to support the rational design of next-generation metal NCs with enhanced functionality.7,18For example, thiolate (SR)-protected gold (Au) NCs (i.e., Aum(SR)n, where m denotes the number of Au atoms, and n represents the number of SR ligands) have high stability owing to the strong Au–S bonds; they have also shown high activity in various catalytic reactions.19,20 Lee et al. demonstrated that doping [Au25(SC6H13)18]− (SC6H13 = 1-hexanethiolate) with a single equivalent of Pt to form [Au24Pt(SC6H13)18]0 significantly enhanced the NC's hydrogen evolution reaction (HER) activity, with approximately twice the reaction rate of commercial Pt NP catalysts.21 Our group conducted a comprehensive study investigating how various structural parameters of Aum(SR)n NCs (e.g., the number of constituent atoms, ligand chain length, and alloying) influenced their HER activity.22 On this basis, we designed a novel bimetallic NC, [Au24Pt(TBBT)12(PDT)3]0 (TBBT = 4-tert-butylbenzenethiolate; PDT = 1,3-propanedithiolate) by optimizing the structural features to promote the HER. The catalytic activity of [Au24Pt(TBBT)12(PDT)3]0 is approximately five-fold higher than that of its monodentate SR-protected analogue, [Au24Pt(SC6H13)18]0 and [Au24Pt(TBBT)18]0.23
In addition to NCs protected solely by monodentate SRs, Au NCs co-protected by monodentate SRs and bidentate SRs (SR′S)—denoted as Aum(SR)n(SR′S)l—can be synthesized via ligand-exchange reactions.23,24 Precise control over the reaction conditions, particularly temperature and the ligand molar ratio, allows for the isolation of Aum(SR)n(SR′S)l NCs. Notably, the prepared [Au24Pt(SR)12(SR′S)3]0 retains a regular icosahedral Au12Pt core that is structurally analogous to that of prototypical Au24M(SR)18 NCs (Fig. 1).25–27 Additionally, [Au24Pt(SR)12(SR′S)3]0 has a significantly constrained surface structure due to the cross-linking of staple motifs by the SR′S ligands. This structural constraint exposes the metal core atoms to the external environment (Fig. 1(a)). Such accessible Au12M cores are expected to facilitate substrate access, thereby supporting catalytic activity for the HER, as well as a diverse range of other catalytic reactions.
However, the precise formation mechanism of [Au24Pt(SR)12(SR′S)3]0 remains elusive. This species is unique in that it is obtained exclusively through a shell (staple) structural transformation, which avoids the common pitfalls of size conversion or polymerization (Fig. 1(b, c) and 2). This study included a multi-faceted investigation into the synthesis of [Au24Pt(SR)12(SR′S)3]0 and explored strategies to increase the incorporation of SR′S ligands. The results indicate that the successful creation of Au NCs co-protected by SR and SR′S depends on two critical factors: (1) the selection of SR′S ligands with appropriate S–S bridging distances, and (2) the regulation of internal core Au–staple S distances by incorporating monodentate SRs with optimized binding affinities for the Au surface. These insights were implemented to synthesize novel [Au24Pt(SR)10(SR′S)4]0 NCs, which have a higher degree of SR′S incorporation than reported thiolate-protected NCs to date.23 These findings can inform the strategic design and synthesis of novel Aum(SR)n(SR′S)l architectures with broad functional applicability in various fields.
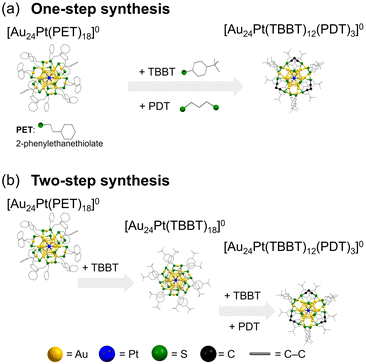 | ||
| Fig. 2 Schematic of the (a) one-step and (b) two-step synthetic methods for preparing [Au24Pt(TBBT)12(PDT)3]0 from [Au24Pt(PET)18]0. | ||
Results and discussion
Influence of monothiolate on the synthesis of mono/dithiolate-co-protected Au NCs
[Au11(PPh3)7(X)3]0 (PPh3 = triphenylphosphine) is an established Au NC protected by monodentate phosphines (PR) that is relatively unstable.28–30 However, its stability can be enhanced significantly upon protection with bidentate phosphines (PR′P), which stabilizes the [Au13(DPPE)5X2]3+ (DPPE = 1,2-bis(diphenylphosphino)ethane) core with a regular icosahedral geometry (Fig. S1).31,32 Meanwhile, owing to the superior binding affinity of SR toward Au, various Aum(SR)n NCs remain stable even when protected by monodentate SR ligands. Therefore, Au NCs protected by SR′S ligands are expected to exhibit high stability. Nevertheless, the synthesis of such Aum(SR′S)l NCs have not yet been realized, likely because introducing SR′S ligands frequently causes ligand-exchange-induced size/structural transformation (LEIST),33–38 resulting in the formation of Au NCs with unintended nuclearities (Fig. 1(b)). Moreover, the strong Au–S bonds of SR′S promote inter-NC linkages, leading to polymerization (Fig. 1(c)). Recent studies have demonstrated that the use of SR′S ligands with a carbon chain length of six (i.e., 1,6-hexanedithiol) causes the formation of disulfide bonds between sulfur termini, thereby linking Aum(SR)n units.39 Consequently, to our knowledge, there are no reported examples of isolated Aum(SR′S)l protected by solely SR′S ligands.Recently, Au NCs co-protected by both monodentate and bidentate SRs have been reported, such as [Au24Pt(TBBT)12(SR′S)3]0 and [Au28(TBBT)18(SR′S)1]0.23,24 In the case of [Au24Pt(TBBT)12(SR′S)3]0, the combination of a rigid Au12Pt core and the presence of monodentate SR ligands suppresses LEISTs and prevents NC polymerization. Instead, this configuration limits the structural changes to only the transformation of staple (shell) motifs via SR′S-mediated cross-linking (Fig. 1(a)). Thus, it is crucial to elucidate the role of monodentate SRs in the synthesis of Aum(SR)n(SR′S)l to comprehensively understand these synthetic design strategies.
Previous studies have shown that when [Au24Pt(PET)18]0 (PET = 2-phenylethanethiolate) serves as a precursor for ligand exchange with bulky TBBT, all ligands are substituted while leaving the underlying framework intact, resulting in the selective synthesis of [Au24Pt(TBBT)18]0 (Fig. 2(b)).40 This stability is particularly noteworthy given that the same reaction using [Au25(PET)18]− leads to structural transformations into Aum(SR)n NCs with varying nuclearities, such as Au28(TBBT)20, Au23(TBBT)17, and Au20(TBBT)16.41–44 This structural integrity is attributed to the inherently robust framework of the Au12Pt core in [Au24Pt(SR)18]0.
In this study, [Au24Pt(SR)18]0 was selected as a precursor for the introduction of SR′S ligands because it does not undergo unintended structural core rearrangements. Geometric investigations of the S–S distances in [Au24Pt(SR)18]0 suggested that SR′S ligands with a carbon chain length of two or three (i.e., S(CH2)xS, where x = 2 or 3) are sterically appropriate for bridging adjacent SR′S ligands.23 However, the addition of 1,3-propanedithiol (PDTH2) to [Au24Pt(PET)18]0 resulted in the formation of insoluble materials, rather than the desired SR′S-protected NCs. This was likely due to (i) the high stability of the Au–S bonds in the PET ligands, which may inhibit the exchange process, or (ii) the onset of inter-NC polymerization induced by an excess of PDT (Fig. 1(c)).
In contrast, the reaction of [Au24Pt(TBBT)18]0 with PDTH2 readily yielded [Au24Pt(TBBT)12(PDT)3]0 (Fig. 2(b)). To better understand this reactivity, the structural differences between [Au24Pt(PET)18]0 and [Au24Pt(TBBT)18]0 were examined. Although these NCs shared essentially the same framework, they exhibited distinct structural distortions. Analysis of the average bond lengths (Fig. S2) revealed that the Ausurface–Ssurface (i.e., the Au–S bond between the surface Au of the Au12Pt core and staple S) was longer in [Au24Pt(TBBT)18]0 than in [Au24Pt(PET)18]0. Consequently, [Au24Pt(TBBT)18]0 had a more compact Au12Pt core, with the Au2(SR)3 staples positioned further from the core surface than in [Au24Pt(PET)18]0. The difference in the pKa values of the aromatic TBBT and aliphatic PET ligands suggested that TBBT has a lower electron-donating ability of the S atoms.40 This leads to weaker Au–S bonds in [Au24Pt(TBBT)18]0, which facilitate the displacement of Au2(SR)3 staples from the metal core. These structural and electronic factors account for the differing chemical and thermodynamic stabilities of the two NCs, with [Au24Pt(TBBT)18]0 exhibiting lower stability. Indeed, [Au24Pt(TBBT)18]0 can only be obtained via ligand exchange and cannot be synthesized through conventional co-reduction methods. These results indicate that ligand exchange with SR′S ligands is promoted by (i) maintaining an appropriate S–S distance within the NCs and (ii) using a monodentate SR ligand with a relatively weak Au–S bond to facilitate the exchange process (Fig. 2).
Furthermore, the reaction proceeded when using 4-isopropylbenzenethiolate (IPBT), an aromatic ligand similar to TBBT, to form [Au24Pt(IPBT)12(PDT)3]0 (Fig. S3). In contrast, the reaction barely proceeded when using SC6H13,23 an aliphatic ligand similar to PET. These observations suggest that aromatic ligands (e.g., TBBT and IPBT), which have weaker Au–S bonds than those of aliphatic ligands (e.g., PET, SC6H13, and SC12H25 [1-dodecanethiolate]), are more readily substituted by PDT ligands.
Reaction tracking from [Au24Pt(TBBT)18]0 or [Au24Pt(PET)18]0 to [Au24Pt(TBBT)12(PDT)3]0
Our group previously established a methodology for the atomically precise separation of various SR-protected NCs using reverse-phase high-performance liquid chromatography (RP-HPLC).45–47 This separation is primarily driven by the charge state, ligand types, and alloy configurations of the NCs. It is also possible to characterize the electronic structures of Aum(SR)n by using a photodiode array (PDA) detector integrated within the HPLC system (Fig. S4). These methods were applied to investigate the chemical composition, elemental distribution, and electronic structures of the prepared materials by coupling RP-HPLC with electrospray-ionization mass spectrometry (ESI-MS).48,49 Previous research on this reaction system demonstrated that for [Au24Pt(PET)18]0, a ligand-exchange reaction at 25 °C with a [TBBTH]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [PET] ratio of approximately 250
[PET] ratio of approximately 250![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 over more than 10 hours yielded [Au24Pt(TBBT)18]0.42 The present study aimed to prepare [Au24Pt(TBBT)12(PDT)3]0via ligand-exchange synthesis with a [PDTH2]
1 over more than 10 hours yielded [Au24Pt(TBBT)18]0.42 The present study aimed to prepare [Au24Pt(TBBT)12(PDT)3]0via ligand-exchange synthesis with a [PDTH2]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [TBBTH]
[TBBTH]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [PET] ratio of 12
[PET] ratio of 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 239
239![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
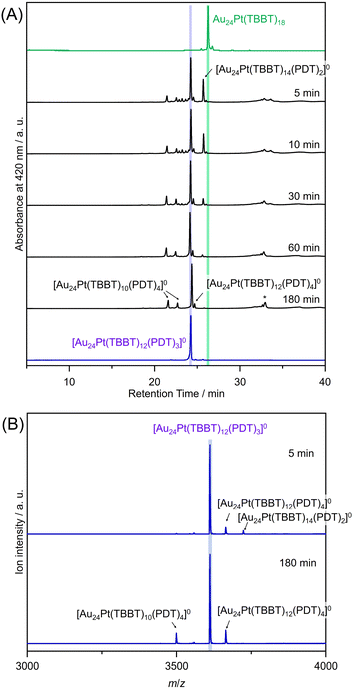
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at 25 °C. Fig. 3 and 4 show chromatograms highlighting the retention times of the precursor [Au24Pt(PET)18]0 (10.0 min), the fully-TBBT-exchanged [Au24Pt(TBBT)18]0 (27.0 min), and the target product [Au24Pt(TBBT)12(PDT)3]0 (24.2 min). The appearance of a single peak in each chromatogram confirms that these NCs were obtained with high purity.
1 at 25 °C. Fig. 3 and 4 show chromatograms highlighting the retention times of the precursor [Au24Pt(PET)18]0 (10.0 min), the fully-TBBT-exchanged [Au24Pt(TBBT)18]0 (27.0 min), and the target product [Au24Pt(TBBT)12(PDT)3]0 (24.2 min). The appearance of a single peak in each chromatogram confirms that these NCs were obtained with high purity.
The shift in retention times can be explained by the surface polarity of the NCs. The TBBT ligands have lower polarity than the precursor PET ligands owing to their structural symmetry and the steric bulk of the tert-butyl groups surrounding the sulfur atoms. In reverse-phase separation mode, higher polarity species elute earlier (i.e., with shorter retention times), whereas lower polarity (more hydrophobic) species are retained longer (Fig. S4). Thus, the systematic increase in the number of TBBT ligands resulted in a corresponding shift toward longer retention times in the experimental data.
The structural flexibility of PDT is critical for these transformations. It is reasonable to presume that PDT coordinated to Au24Pt(TBBT)12(PDT)3 in a folded conformation, which facilitated the cross-linking required to form Au24Pt(TBBT)10(PDT)4. The importance of backbone flexibility and length is further supported by comparative experiments. The addition of HS(CH2)2SH to Au24Pt(TBBT)12(PDT)3 yielded no reaction, whereas the use of HS(CH2)4SH generated a 4-SR′S-substituted product (refer to “Synthesis of other dithiolate-protected Au NCs” section). Additionally, the lack of a reaction between rigid 1,3-benzenedithiol (BDTH2) and Au24Pt(TBBT)18 indicated that a specific flexible chain length is essential for effective SR′S cross-linking.23
After 180 minutes of reaction, the peak corresponding to Au24Pt(TBBT)14(PDT)2 disappeared, and peaks attributable to the Au24Pt(TBBT)10(PDT)4 and Au24Pt(TBBT)12(PDT)4 byproducts were observed (Fig. 3). The formation of Au24Pt(TBBT)12(PDT)4 was attributed to trace amounts of (PDTH)2, which is a disulfide-bonded impurity present in the PDTH2 precursor. This (PDTH)2 species could induce inter-NC cross-linking, rather than the intra-NC bridging observed with monomeric PDT.
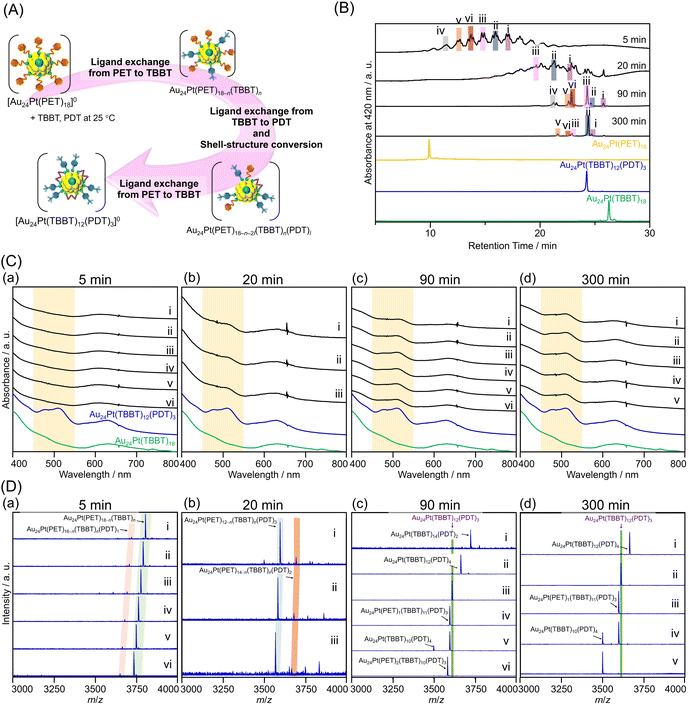
Fig. 4B–D present the RP-HPLC, UV-vis, and ESI-MS results over time after the simultaneous addition of PDTH2 and TBBTH to [Au24Pt(PET)18]0. After 5 minutes (Fig. 4(a)), the RP-HPLC chromatogram (Fig. 4B) contained multiple peaks at retention times longer than that of the precursor [Au24Pt(PET)18]0. These peaks were interpreted as intermediate species formed during the ligand-exchange of PET with TBBT. At this stage, the optical absorption spectra lacked the characteristic features of [Au24Pt(SR)12(SR′S)3]0 that are typically observed in the 450–550 nm range (Fig. 4C(a)). This suggests that the initial phase of the reaction was dominated by PET-to-TBBT ligand exchange, with negligible PDT-mediated cross-linking. This was further supported by ESI-MS analysis of the representative peaks (Fig. 4D(a); i–vi), which were assigned to Au24Pt(PET)18−n(TBBT)n (n = 3–8). These results confirmed that there was negligible PDT substitution during the early stages of the reaction. Previous studies examining the ligand-exchange reaction between [Au25(PET)18]− and PDTH2 indicated that the process typically yielded a polydisperse mixture of Au25(PET)18−2l(PDT)l (l = 2–6), rather than a discrete product.23,50 In the present study, the molar ratio of PDT to TBBT was low ([PDTH2]![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [TBBTH] ratio of 1
[TBBTH] ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20), and as a result, PET–PDT exchange products such as Au24Pt(PET)18−2l(PDT)l were scarcely detected. These observations support the hypothesis that PDT substitution occurs preferentially at Au–TBBT sites, rather than Au–PET sites, consistent with the mechanistic behavior discussed above.
20), and as a result, PET–PDT exchange products such as Au24Pt(PET)18−2l(PDT)l were scarcely detected. These observations support the hypothesis that PDT substitution occurs preferentially at Au–TBBT sites, rather than Au–PET sites, consistent with the mechanistic behavior discussed above.
Fig. 4(b) shows the results of the reaction after 20 minutes. The optical absorption spectra of the peaks with longer retention times in Fig. 4B reveal a broadening and splitting of the features in the 450–550 nm region (Fig. 4C(b)). This spectral evolution was primarily attributed to the emergence of the characteristic peaks of [Au24Pt(SR)12(SR′S)3]0, which originate from the staple structures cross-linked by PDT. ESI-MS analysis of peaks (i–iii) identified the species as Au24Pt(PET)1(TBBT)11(PDT)3, Au24Pt(PET)2(TBBT)10(PDT)3, and Au24Pt(PET)3(TBBT)9(PDT)3, respectively (Fig. 4D(b)). Based on these observations, it is presumed that subsequent ligand substitution to form PDTl (l ≤ 3) proceeds once the (PET)2n (n ≤ 3) core sites with appropriate S–S distances are replaced by TBBT. Our group previously demonstrated that during the ligand exchange of SC12H25 with [Au24Pd(PET)18]0, the SC12H25 ligands first preferentially replaced the PET ligand at the core site, yielding the monosubstituted isomer [Au24Pd(PET)17(SC12H25)]0.48 Thus, in the present study, the following three-step mechanism is proposed. First, the chain-like PDTH2 coordinates to the core-site TBBT as an S–(CH2)3–SH group. This is followed by rapid replacement by TBBT ligands that coordinate to the core at sites with optimal S–S distances. Finally, the conversion of the staple structure occurs nearly simultaneously with these exchange events. This mechanism is consistent with the experimental observation that both Au24Pt(PET)16−n(TBBT)n(PDT)1 (13 ≥ n ≥ 4) and Au24Pt(PET)12−n(TBBT)n(PDT)3 (n = 3, 5, 6) were present as early as 5 minutes after reaction initiation.
Furthermore, after 90 minutes of reaction (Fig. 4(c)), the optical absorption spectrum exhibited complete splitting of the peak in the 450–550 nm region, suggesting that the conversion to the cross-linked staple structure was nearly complete following PDT introduction (Fig. 4C(c)). Peaks were assigned via ESI-MS analysis (Fig. 4D(c)) as Au24Pt(TBBT)14(PDT)2 (i), Au24Pt(TBBT)12(PDT)4 (ii), Au24Pt(TBBT)10(PDT)4 (v), and various Au24Pt(PET)12−n(TBBT)n(PDT)3 intermediates (iii, iv and vi). These results indicated that after 90 minutes, the system had largely transitioned to thermodynamically stable cross-linked configurations, although some residual PET ligands remained within the TBBT/PDT framework.
The results after 300 minutes of reaction are presented in Fig. 4(d). Peaks (i–v) were observed at four distinct retention times (Fig. 4B). All corresponding optical absorption spectra showed complete splitting of the peak around 450–550 nm, consistent with the observations after 90 minutes (Fig. 4C(d)). ESI-MS assignments confirmed the formation of Au24Pt(PET)12−n(TBBT)n(PDT)3 (ii–iv) and Au24Pt(TBBT)n(PDT)4 (where n = 10 or 12; i, and v) (Fig. 4D(d)). However, in contrast to the 90-minute samples, Au24Pt(TBBT)14(PDT)2 was no longer detected. This suggested that as the reaction approached equilibrium, the intermediate products converged to the target Au24Pt(TBBT)12(PDT)3.
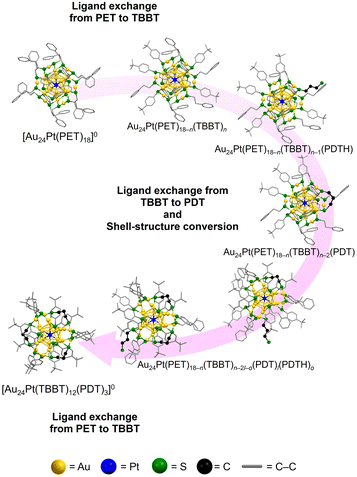
Based on the comprehensive analysis of the experimental data, the following mechanism is proposed for the ligand exchange of [Au24Pt(PET)18]0 using PDT and TBBT to obtain Au24Pt(TBBT)12(PDT)3 (Fig. 4A and 5). (1) Initial ligand exchange: the reaction is initiated with the preferential exchange of PET ligands for TBBT. (2) PDT coordination: ligand exchange occurs between the newly substituted TBBT and PDTH at the core-site S atom. (3) Staple conversion: rapid ligand exchange occurs between TBBT—which provides a sterically appropriate S–S distance—and the opposite terminal S of the already-ligated PDTH; this process facilitates the conversion of the shell structure to the cross-linked staple configuration. (4) Final substitution: all remaining PET ligands undergo exchange with TBBT, resulting in the stable [Au24Pt(TBBT)12(PDT)3]0 (Fig. 4A and 5). This proposed mechanism was further validated by observing the same behavior during the ligand exchange from [Au24Pt(SC6H13)18]0 to [Au24Pt(TBBT)12(PDT)3]0 (Fig. S6–S11), suggesting that the preference for TBBT-mediated core substitution and subsequent PDT cross-linking are key characteristics supporting the formation of [Au24Pt(TBBT)12(PDT)3]0. [Au24Pt(TBBT)12(PDT)3]0 obtained via this reaction mechanism was confirmed to possess high thermal stability (Fig. S12).
Synthesis of other dithiolate-protected Au NCs
Based on the findings discussed above, the syntheses of novel Aum(SR)n(SR′S)l NCs were further explored. First, PDT was introduced as an SR′S ligand to determine the possibility of achieving higher substitution numbers. Structurally, [Au24Pt(TBBT)12(PDT)3]0 has three additional core sites available for further SR′S substitution (Fig. S13 and S14). If all available core sites are occupied, [Au24Pt(TBBT)18−2l(PDT)l]0 (l = 4–6) can form via a similar reaction pathway. However, although [Au24Pt(TBBT)10(PDT)4]0 formed relatively easily, it could not be isolated.Additionally, the effect of the SR′S ligand alkyl chain length was investigated. When TBBTH and HS(CH2)xSH (x = 2, 4, 6) were reacted with [Au24Pt(PET)18]0, the process yielded insoluble precipitates, and no SR′S-protected NCs were obtained. Additionally, SR′S cross-linking was attempted using BDTH2, which has the same carbon chain length as PDTH2 but features a rigid benzene ring. This also failed to yield SR′S-protected NCs. The S–S distances bridged by PDT in [Au24Pt(TBBT)12(PDT)3]0 were similar (4.443, 4.497, and 4.497 Å) (Fig. S13). Thus, it is reasonable to conclude that [Au24Pt(TBBT)12(PDT)3]0 exhibits high stability due to the presence of these three core sites with S–S distances that are optimally suited for PDT coordination.
Considering these findings, we posited that introducing additional SR′S ligands into [Au24Pt(TBBT)12(PDT)3]0 would require SR′S ligands with an S–S distance more compatible than that of PDT. To evaluate Aum(SR)n(SR′S)l with a higher degree of substitution, [Au24Pt(TBBT)12(PDT)3]0 NCs were prepared with SR′S ligands of varying chain lengths. The addition of 1,2-ethanedithiol (HS(CH2)xSH; x = 2) to [Au24Pt(TBBT)12(PDT)3]0 did not initiate a reaction; however, the use of 1,4-butanedithiol (HS(CH2)xSH; x = 4) yielded a relatively stable product 1 that could be isolated (Fig. 6(a)). ESI-MS analysis identified the resulting product as [Au24Pt(TBBT)10(PDT)3(S(CH2)4S)1]0, in which two additional TBBT units were replaced by S(CH2)4S ligands (Fig. 6(b)). These results also suggest that both the carbon chain length and the inherent flexibility of the SR′S ligand are critical factors for increasing the number of SR′S ligands on the NC surface.
 | ||
| Fig. 6 (a) UV-vis spectrum and (b) negative-ion ESI mass spectrum of 1. In (a), the absorbance spectra of Au24Pt(TBBT)12(PDT)3 are also shown for comparison. | ||
Conclusions
This study provided the following key insights regarding the transformation of [Au24Pt(PET)18]0 into [Au24Pt(TBBT)12(PDT)3]0:(1) Direct substitution of PET with PDT ligands is hindered by the strong Au–PET bonds, ultimately rendering the formation of [Au24Pt(PET)12(PDT)3]0 unfavorable.
(2) Aromatic ligands (e.g., TBBT and IPBT), which have weaker Au–S bonds than aliphatic ligands (e.g., PET, SC6H13, SC12H25), readily undergo substitution with the aliphatic PDT ligand.
(3) In the intermediate species Au24Pt(PET)18−n(TBBT)n, PDT facilitates intra-NC bridging at S sites with appropriate S–S distances, simultaneously inducing a transformation of the staple structure.
(4) PDT ligands that fail to support intra-NC bridging tend to induce inter-NC cross-linking, leading to polymerization.
These synthetic insights were implemented to synthesize [Au24Pt(TBBT)10(PDT)3(S(CH2)4S)1]0, which incorporates more SR′S ligands than in any previously reported material.23 It would be useful to estimate these substitution sites using theoretical calculations in the future.51
Indeed, Au25(SR)18 is recognized as a stable framework that can further incorporate various heterometals (e.g., Ir, Rh, Pt, Pd, Ag, Cu, Hg, or Cd); however, excessive or highly dissimilar doping often compromises the material's geometric and electronic stability. The Aum(SR)n(SR′S)l synthesized in this work have a rigid staple structure, which is expected to mitigate the instabilities typically associated with NC alloying. Consequently, the novel MyAu25−y(SR)12(SR′S)3 template can enable the development of NCs with enhanced functionality. This research provides valuable insights to support the design of novel metal NCs and their alloyed derivatives featuring rigid, cross-linked staple architectures.52,53
Experimental
Synthesis of 1
In the optimized synthesis, 8.6 mg [Au24Pt(TBBT)12(PDT)3]0 was dissolved in 4.0 mL toluene, to which 863 µL TBBT (98%) and 51.3 µL HS(CH2)4SH were added at room temperature, and the mixture was stirred for 3 hours to allow the ligand-exchange reaction to proceed. The product was washed sequentially with ultrapure water and a mixture of ultrapure water and methanol and then extracted with dichloromethane. Finally, the products were separated by PTLC (dichloromethane![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) hexane = 4
hexane = 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6), and the second layer was scraped off and extracted with dichloromethane to obtain [Au24Pt(TBBT)10(PDT)3(S(CH2)4S)1]0.
6), and the second layer was scraped off and extracted with dichloromethane to obtain [Au24Pt(TBBT)10(PDT)3(S(CH2)4S)1]0.
Author contributions
T. Kawawaki and Y. Negishi designed the experiments and conducted the measurements with S. Yoshikawa and S. Hossain. T. Kawawaki, S. Hossain and Y. Negishi wrote the paper. All authors approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Relevant data are available from the corresponding authors (T. Kawawaki and Y. Negishi) upon reasonable request.All data supporting the findings of this work are provided in the supplementary information (SI). Supplementary information: additonal experimental details, characterization and figures. See DOI: https://doi.org/10.1039/d6sc00460a.
Acknowledgements
The authors thank Ms Ayaka Ikeda and Miyu Sera (Tokyo University of Science) for their technical assistance. This work was based on results obtained from a project commissioned by JST FOREST Program, Grant Number JPMJFR245R and the Environment Research and Technology Development Fund (JPMEERF20255RA2) of the Environmental Restoration and Conservation Agency. It was also supported by the Japan Society for the Promotion of Science (JSPS) through KAKENHI grants (grant numbers 23H00289, 22K19012, and 24K01459) and “Crossover Alliance to Create the Future with People, Intelligence”. Funding from the Carbon Recycling Fund Institute, the Japan Gas Association, the Iwatani Naoji Foundation, the Ichimura Foundation for New Technology, the Suzuki Foundation, the Japan Keirin Aurtorace Foundation and Tobe-Maki Foundation are gratefully acknowledged.Notes and references
- I. Chakraborty and T. Pradeep, Chem. Rev., 2017, 117, 8208 CrossRef CAS PubMed.
- R. Jin, G. Li, S. Sharma, Y. Li and X. Du, Chem. Rev., 2021, 121, 567 CrossRef CAS PubMed.
- M. R. Narouz, K. M. Osten, P. J. Unsworth, R. W. Y. Man, K. Salorinne, S. Takano, R. Tomihara, S. Kaappa, S. Malola, C.-T. Dinh, J. D. Padmos, K. Ayoo, P. J. Garrett, M. Nambo, J. H. Horton, E. H. Sargent, H. Häkkinen, T. Tsukuda and C. M. Crudden, Nat. Chem., 2019, 11, 419 CrossRef CAS PubMed.
- C. A. Hosier and C. J. Ackerson, J. Am. Chem. Soc., 2019, 141, 309 CrossRef CAS PubMed.
- Y. Pei, P. Wang, Z. Ma and L. Xiong, Acc. Chem. Res., 2019, 52, 23 CrossRef CAS PubMed.
- B. Bhattarai, Y. Zaker, A. Atnagulov, B. Yoon, U. Landman and T. P. Bigioni, Acc. Chem. Res., 2018, 51, 3104 CrossRef CAS PubMed.
- Q. Tang, G. Hu, V. Fung and D.-e. Jiang, Acc. Chem. Res., 2018, 51, 2793 CrossRef CAS PubMed.
- T. Kawawaki, Y. Mitomi, N. Nishi, R. Kurosaki, K. Oiwa, T. Tanaka, H. Hirase, S. Miyajima, Y. Niihori, D. J. Osborn, T. Koitaya, G. F. Metha, T. Yokoyama, K. Iida and Y. Negishi, Nanoscale, 2023, 15, 7272 RSC.
- S. Sharma, K. K. Chakrahari, J.-Y. Saillard and C. W. Liu, Acc. Chem. Res., 2018, 51, 2475 CrossRef CAS PubMed.
- J. Fang, B. Zhang, Q. Yao, Y. Yang, J. Xie and N. Yan, Coord. Chem. Rev., 2016, 322, 1 CrossRef CAS.
- V. Truttmann, H. Drexler, M. Stöger-Pollach, T. Kawawaki, Y. Negishi, N. Barrabés and G. Rupprechter, ChemCatChem, 2022, 14, e202200322 CrossRef CAS PubMed.
- S. Funaki, T. Kawawaki, T. Okada, K. Takemae, S. Hossain, Y. Niihori, T. Naito, M. Takagi, T. Shimazaki, S. Kikkawa, S. Yamazoe, M. Tachikawa and Y. Negishi, Nanoscale, 2023, 15, 5201 RSC.
- Y.-Z. Huang, R. K. Gupta, G.-G. Luo, Q.-C. Zhang and D. Sun, Coord. Chem. Rev., 2024, 499, 215508 CrossRef CAS.
- Z. Chen, D. Zuo, L. Zhao, Y. Chen, F. Sun, L. Wang, H. Shen and Q. Tang, Chem. Sci., 2025, 16, 10397 RSC.
- X.-X. Lai, J. Wei, T.-T. Liu, J. Li, J.-J. Li, X.-K. Wan and Q.-M. Wang, Nat. Commun., 2025, 16, 7862 CrossRef CAS PubMed.
- J.-H. Huang, Y. Cui, Z.-Y. Wang and S.-Q. Zang, Acc. Chem. Res., 2025, 58, 1249 CrossRef CAS.
- C. Cesari, J.-H. Shon, S. Zacchini and L. A. Berben, Chem. Soc. Rev., 2021, 50, 9503 RSC.
- O. López-Estrada, N. Mammen, L. Laverdure, M. M. Melander, H. Häkkinen and K. Honkala, ACS Catal., 2023, 13, 8997 CrossRef.
- T. Song, X. Liu, H. Wang, Q. Yuan, X. Cai, W. Ding and Y. Zhu, Coord. Chem. Rev., 2025, 543, 216922 CrossRef CAS.
- T. Kawawaki, Y. Kataoka, M. Hirata, Y. Iwamatsu, S. Hossain and Y. Negishi, Nanoscale Horiz., 2021, 6, 409 RSC.
- K. Kwak, W. Choi, Q. Tang, M. Kim, Y. Lee, D.-e. Jiang and D. Lee, Nat. Commun., 2017, 8, 14723 CrossRef PubMed.
- B. Kumar, T. Kawawaki, N. Shimizu, Y. Imai, D. Suzuki, S. Hossain, L. V. Nair and Y. Negishi, Nanoscale, 2020, 12, 9969 RSC.
- M. Sera, S. Hossain, S. Yoshikawa, K. Takemae, A. Ikeda, T. Tanaka, T. Kosaka, Y. Niihori, T. Kawawaki and Y. Negishi, J. Am. Chem. Soc., 2024, 146, 29684 CrossRef CAS PubMed.
- W. Fan, N. Yan, J. Zha, W. Gu, Q. You, Y. Yang, S. Zhuang and Z. Wu, J. Phys. Chem. Lett., 2023, 14, 3216 CrossRef CAS PubMed.
- M. Zhu, C. M. Aikens, F. J. Hollander, G. C. Schatz and R. Jin, J. Am. Chem. Soc., 2008, 130, 5883 CrossRef CAS PubMed.
- M. W. Heaven, A. Dass, P. S. White, K. M. Holt and R. W. Murray, J. Am. Chem. Soc., 2008, 130, 3754 CrossRef CAS PubMed.
- J. Akola, M. Walter, R. L. Whetten, H. Häkkinen and H. Grönbeck, J. Am. Chem. Soc., 2008, 130, 3756 CrossRef CAS PubMed.
- R. H. Adnan, J. M. L. Madridejos, A. S. Alotabi, G. F. Metha and G. G. Andersson, Adv. Sci., 2022, 9, 2105692 CrossRef CAS PubMed.
- V. G. Albano, P. L. Bellon, M. Manassero and M. Sansoni, J. Chem. Soc. D, 1970, 1210 RSC.
- F. Cariati and L. Naldini, Inorg. Chim. Acta, 1971, 5, 172 CrossRef CAS.
- Y. Shichibu and K. Konishi, Small, 2010, 6, 1216 CrossRef CAS PubMed.
- K. Konishi, M. Iwasaki and Y. Shichibu, Acc. Chem. Res., 2018, 51, 3125 CrossRef CAS PubMed.
- M. S. Bootharaju, C. P. Joshi, M. J. Alhilaly and O. M. Bakr, Chem. Mater., 2016, 28, 3292 CrossRef CAS.
- X. Kang and M. Zhu, Chem. Mater., 2019, 31, 9939 CrossRef CAS.
- M. Rambukwella and A. Dass, Langmuir, 2017, 33, 10958 CrossRef CAS PubMed.
- Y. Wang and T. Bürgi, Nanoscale Adv., 2021, 3, 2710 RSC.
- C. Zeng, Y. Chen, A. Das and R. Jin, J. Phys. Chem. Lett., 2015, 6, 2976 CrossRef CAS PubMed.
- M. P. Maman, A. S. Nair, H. Cheraparambil, B. Pathak and S. Mandal, J. Phys. Chem. Lett., 2020, 11, 1781 CrossRef CAS PubMed.
- Y. Saito, Y. Shichibu and K. Konishi, Nanoscale, 2021, 13, 9971 RSC.
- S. Hossain, Y. Imai, D. Suzuki, W. Choi, Z. Chen, T. Suzuki, M. Yoshioka, T. Kawawaki, D. Lee and Y. Negishi, Nanoscale, 2019, 11, 22089 RSC.
- C. M. Aikens, Acc. Chem. Res., 2018, 51, 3065 CrossRef CAS PubMed.
- Y. Negishi, H. Horihata, A. Ebina, S. Miyajima, M. Nakamoto, A. Ikeda, T. Kawawaki and S. Hossain, Chem. Sci., 2022, 13, 5546 RSC.
- C. Zeng, C. Liu, Y. Chen, N. L. Rosi and R. Jin, J. Am. Chem. Soc., 2014, 136, 11922 CrossRef CAS PubMed.
- A. George, A. Sundar, A. S. Nair, M. P. Maman, B. Pathak, N. Ramanan and S. Mandal, J. Phys. Chem. Lett., 2019, 10, 4571 CrossRef CAS PubMed.
- V. L. Jimenez, M. C. Leopold, C. Mazzitelli, J. W. Jorgenson and R. W. Murray, Anal. Chem., 2003, 75, 199 CrossRef CAS PubMed.
- D. M. Black, N. Bhattarai, S. B. H. Bach and R. L. Whetten, J. Phys. Chem. Lett., 2016, 7, 3199 CrossRef CAS PubMed.
- Y. Negishi, S. Hashimoto, A. Ebina, K. Hamada, S. Hossain and T. Kawawaki, Nanoscale, 2020, 12, 8017 RSC.
- Y. Niihori, Y. Kikuchi, A. Kato, M. Matsuzaki and Y. Negishi, ACS Nano, 2015, 9, 9347 CrossRef CAS PubMed.
- C. Zeng, C. Liu, Y. Pei and R. Jin, ACS Nano, 2013, 7, 6138 CrossRef CAS PubMed.
- V. R. Jupally, R. Kota, E. V. Dornshuld, D. L. Mattern, G. S. Tschumper, D.-e. Jiang and A. Dass, J. Am. Chem. Soc., 2011, 133, 20258 CrossRef CAS PubMed.
- T. Ubayasena, B. Bhattarai, B. Yoon, U. Landman and T. P. Bigioni, J. Phys. Chem. C, 2025, 129, 878 CrossRef CAS.
- Z. Gan, N. Xia and Z. Wu, Acc. Chem. Res., 2018, 51, 2774 CrossRef CAS PubMed.
- S. Yang, J. Chai, Y. Song, J. Fan, T. Chen, S. Wang, H. Yu, X. Li and M. Zhu, J. Am. Chem. Soc., 2017, 139, 5668 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |