Open Access Article
Open Access ArticleGeometric orthogonality as a recipe for efficient intramolecular charge generation in core substituted NDI derivatives
Hugh C. Britton†
*ab,
Alberto M. Santa Daría† c,
Chao Lyu†b,
Andras B. Augusztinb,
Lewis M. Cowenb,
Dejan-Krešimir Bučar
c,
Chao Lyu†b,
Andras B. Augusztinb,
Lewis M. Cowenb,
Dejan-Krešimir Bučar b,
Alethea B. Tabor
b,
Alethea B. Tabor b,
Bob C. Schroeder
b,
Bob C. Schroeder b,
Sandra Gómez
b,
Sandra Gómez *d and
Jose M. Marin-Beloqui
*d and
Jose M. Marin-Beloqui *ef
*ef
aSchool of Chemistry and Chemical Engineering, University of Southampton, Southampton, SO17 1BJ, UK. E-mail: H.C.Britton@soton.ac.uk
bDepartment of Chemistry, University College London, London, WC1 0AJ, UK
cUniversidad de Salamanca, Pl. de los Caídos s/n, 37008 Salamanca, Spain
dDepartment of Chemistry, Module 13, Universidad Autónoma de Madrid, 28049 Madrid, Spain. E-mail: sandra.gomezr@uam.es
eDepartment of Physical Chemistry, University of Malaga, Andalucia-Tech Campus de Teatinos s/n, 29071 Málaga, Spain. E-mail: jm.marinbeloqui@uma.es
fMaterials and Nanotechnology College Institute (IMANA), University of Malaga, Campus de Teatinos s/n, 29071 Málaga, Spain
First published on 27th March 2026
Abstract
One of the primary drawbacks of organic materials, compared to their inorganic counterparts in various optoelectronic applications, is their lower charge generation efficiency, which stems from their inherently higher exciton binding energy. Therefore, new out-of-the-box approaches need to be introduced to the field. Herein, we propose a new approach to increase the charge formation of naphthalenediimide (NDI) derivatives by inducing a large torsional angle between the NDI core and the core-attached substituent, deconjugating the resulting extended π-system. To study the extent of this change, transient absorption spectroscopy characterisation has been performed on a set of derivatised NDI molecules where the core-attached substituents have been systematically altered to modulate the resulting torsional angle. The data indicates an enhanced charge generation with core-attached substituents from phenyl to anthracenyl which increase in both size and degree of rotational inhibition. State-of-the-art excited state simulations using the TD-B3LYP/def2-SVP level of theory were performed to calculate absorption spectra and to parametrise potential energy surfaces to run non-adiabatic quantum dynamics simulations for the two extreme NDI systems, showing crucial differences due to the influence of charge transfer states. This opens the possibility for a new family of NDI molecules with implications for a wide range of applications such as photovoltaics, transistors and catalysis.
Introduction
The transfer of electrons is at the heart of a myriad of chemical and physical processes such as photosynthesis, radical reactions, catalysis and organic electronics. However, most organic materials lack the ability to efficiently generate charges upon external stimuli, particularly by photoexcitation. The main reason for this low efficiency is the large exciton binding energy, which hinders the separation of the singlet exciton into free charges. This is a major reason why organic materials, despite their obvious advantageous properties such as flexibility, transparency, and low-cost manufacturing, have not been widely adopted.Efficient generation of charges in organic materials typically relies on using two materials, with one serving as the donor and the other as the acceptor.1–3 But the use of two different materials is accompanied by a myriad of associated problems such as poor miscibility,4 lack of morphology control,5 energy losses6 and stability.7 These problems highlight the utmost importance of new approaches to generate charges and thus obtain organic molecules able to yield free charge carriers without the need for a second material.
To this end, we conduct a detailed investigation of the excited state dynamics, focusing on charge generation enabled by the orthogonality between a central naphthalenediimide (NDI) core and its substituents. The NDI core was selected as it is a building block commonly used in literature due to its ability to generate negative charges. This ability, paired with its outstanding oxidative stability, has granted it a large presence in the field of organic photovoltaics and field effect transistors.8–13 Numerous strategies aimed at enhancing the charge generation in NDI molecules have focused on the substitution at the imide nitrogen.14–17 In instances where substituents have been attached to the NDI naphthalene core, prevalent approaches have predominantly employed electron donating moieties such as thiophenes and amines to facilitate charge generation by raising their highest occupied molecular orbital (HOMO) level.18–22 This reliance has been rooted in electronic effects, diverging from our geometric approach, to decrease the electronic coupling between the core and the substituents. Literature provides an instance in which an anthracene moiety is conjugated to the NDI core; however, an amine functional group is also affixed to the NDI core. Consequently, the predominant contribution to charge generation is attributed to the donor characteristics of the amine group, rather than the anthracene substituent.23 Other examples of NDI core substitution found in literature focus on the attachment of halogen substituents to the NDI core to induce triplet formation by an increase of the spin–orbit coupling through the heavy atom effect.23,24 However, the effects of attaching diverse aromatic groups to the NDI core on the geometry and charge generation have not previously been explored.
Charges generated by the molecules presented in this work offer a distinct advantage over other examples in literature where orthogonal charge formation is shown. In their vast majority, such charges are formed either in larger molecules like polymers,25 where the charge separation is more accessible than in a small molecule, or through strong donor units, again relying on electronic effects.23,26 Accordingly, Wu et al. published a work where they synthesise a strong donor character unit of indacenodithiophene (IDT) sandwiched between two orthogonal NDI units with the ability to generate charges.27 However, the IDT unit large donor strength makes impossible to isolate the role of orthogonality in the charge generation process. In our work, the charge generation occurs in small single-molecules with the orthogonality between the main NDI core and the core substituent as the main relevant actor.
Herein we report a series of NDI core substituted molecules with large aromatic substituents. We synthesised a series of five NDI analogues – NDI-Ph (phenyl), NDI-Th (thiophenyl), NDI-Mes (mesityl), NDI-Nap (naphthalenyl) and NDI-An (anthracenyl) – with a range of aromatic groups of differing sizes (Scheme 1). We anticipated that as the size of the aromatic moieties increased, the steric hindrance between the NDI and the flanking substituents would also increase, thus inducing a larger orthogonality and reduced π-orbital overlap. Furthermore, in some cases configurationally stable biaryl atropisomers could result, particularly for large analogues or with large ortho-substituents. The core substitution with larger aromatic moieties introduces a significant steric hindrance between the substituent and the imide carbonyls, resulting in a nearly orthogonal torsional angle between both aromatic units and a dramatically reduced π-orbital overlap.
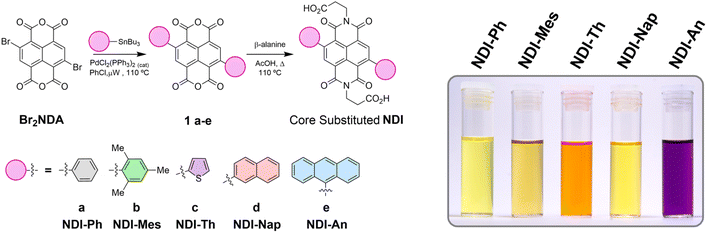 | ||
| Scheme 1 Synthesis of core-substituted NDI dicarboxylic acids NDI-Ph, NDI-Mes, NDI-Th, NDI-Nap and NDI-An. Insert shows the range of colours of the final products. | ||
Unravelling the identity of the charged states observed in the different NDI derivatives upon excitation, presents a challenge. The main reason for this is that the differences between charge transfer and separated charge states are often minimal in terms of their spectral characteristics.28 Results indicate, however, that we cannot associate these charges to twisted intramolecular charge transfer (TICT) states. Molecules that undergo TICT change their ground state geometry, upon excitation, to a twisted geometry in favour of a more energetically favourable charge transfer (CT) state.29–31 The NDI derivatives herein presented maintain their orthogonality in ground and excited states. CT generation in TICT molecules are very solvent sensitive, as the viscosity and intermolecular interactions play a major role in the geometry evolution upon excitation.32,33 Although some TICT systems can exhibit ultrafast charge transfer, their dynamics generally rely on significant excited-state geometry relaxation, which can constitute an efficiency loss pathway, unlike the largely rigid orthogonal structures studied here.
Transient absorption spectroscopy (TAS) was chosen to characterise the charge generation process in the presented series of NDI derivatives. In short, TAS is a technique with two different light sources, where one excites the sample whereas the other one probes the photogenerated excited species. Moreover, creating a delay between the two light sources is possible to obtain information about the time evolution of the photogenerated excited species. This technique is widely used in the field to study the excited state behaviour in organic molecules34,35 as the TAS signal is proportional to the number of charges created due to the Beer–Lambert law.36–38
The implications of the geometric orthogonality approach for charge generation extend beyond applications where charges are the ultimate goal, such as catalysis, photovoltaics, and transistors. Charges also serve as key intermediates in processes like singlet fission, thermally activated delayed fluorescence, and triplet formation, enabling diverse applications including LEDs and bioimaging.39–42
Results
Synthetic procedures
As limited studies have previously been reported on aromatic core-substituted NDIs, it was necessary to develop a flexible and robust synthetic route that would deliver the NDI analogues in good yield and high purity. Initially the syntheses of the substituted NDIs were attempted by reacting known dibromide dianhydride Br2NDA with β-alanine in AcOH to give imide Ala2NDI in >70% (SI Scheme S1).43–45 Unfortunately, Ala2NDI was highly insoluble in all organic solvents, preventing subsequent introduction of aryl substituents directly to the NDI core via standard Pd-mediated approaches. This was attributed to well-known insolubility of NDI core substrates exacerbated by the carboxylic acid side chains (chosen to allow subsequent NDI functionalisation for a variety of applications).10,46–48This necessitated a change in the synthetic approach. The anhydride core Br2NDA was observed to be notably more soluble in a range of organic solvents. However, the sensitive anhydride and bromide functionalities precluded the use of bases in coupling reactions and attempted coupling under standard Suzuki and Sonagashira conditions resulted in decomposition and byproducts.49,50 On the other hand, the neutral Stille reaction conditions provided a viable solution to this issue and following precedent, microwave coupling conditions at high temperature to aid solubility, delivered known 1a and the novel anhydrides 1b–e in 51–95% yields.51–55 The aromatic core-substituted NDI anhydrides 1a–e were highly coloured, most notably a brilliant purple for 1e containing flanking anthracene units. In the final step, the desired imide was introduced through condensation with β-alanine to yield NDI-Ph, NDI-Mes, NDI-Th, NDI-Nap and NDI-An in 66–88% yields spanning a similar range of vibrant colours.
Spectroscopic analysis
Fig. 1 shows the ground state absorbance of the various NDI derivatives in 2-methyltetrahydrofuran (2-MeTHF) solutions. It is possible to see the characteristic NDI core bands with little variations among them (375, 377 and 379 nm for NDI-Ph, NDI-Nap and NDI-Th, respectively). For NDI-An, the NDI core transitions are slightly obscured by the anthracene bands. | ||
| Fig. 1 Ground state absorbance of 2-methyltetrahydrofuran (2-MeTHF) solutions of NDI-Ph (black), NDI-Nap (red), NDI-Th (purple) and NDI-An (blue). | ||
In addition, there is another band at longer wavelengths which is associated in literature with NDI CT bands.15,23,56 We see a larger displacement to the red of this band when the size of the carbon-only substituent is increased phenyl < naphthalene < anthracene. The lower energy absorption band for the thiophene substituted molecule is positioned between the anthracene and the naphthalene. The presence of this longer wavelength band is in line with a more effective separation of the electronic distribution between HOMO and the lowest unoccupied molecular orbital (LUMO) levels, associated with CT states. This effective separation stabilises the LUMO and raises the HOMO levels, and, therefore, decreases the bandgap of this electronic transition.57–59 The larger red-shift for NDI-Th can be explained firstly in light of the electron rich nature of the thiophene ring. This larger donor nature leads to more efficient orbital mixing with the electron deficient NDI core, resulting in a reduced optical bandgap. Secondly, due to the smaller five-membered thiophene ring, the steric hindrance between the thiophene and NDI core is reduced, leading to a more planar overall structure, facilitating electron delocalisation.
To confirm the assignment of these bands to CT states, we represented Mulliken dependence60–62 of the CT frequency with the oxidation potential of the substituents obtained from literature63–65 (Fig. S2). We have used oxidation potential since the NDI donor remains constant, and the oxidation potential represents a close measurement of the donor strength.66 A linear dependence between the CT frequency and the oxidation potential is associated with an unequivocal CT nature of the corresponding absorption band.67 However, as seen in our case, the dependence was not well fitted to a line, indicating that the charged species associated with these bands did not share the same origin. This deviation from linear dependence could be originated by a significant decoupling of the NDI core and the substituent. This decoupling is in line with the small displacement (0.03 eV from NDI-Ph to NDI-Th) seen for the NDI core bands in the ground state absorbance spectra.
To investigate this decoupling between the donor moiety and the acceptor NDI core, we performed cyclic voltammetry (CV) on the NDI compounds (see SI). The reduction of all five compounds led to the same electron affinity (EA), 3.8 eV, which we can approximate to the LUMO level. This EA is similar to other pristine-like NDI molecules in literature.66 The results of the Mulliken dependence indicated that the obtention of the ionization potential from the EA and the bandgap would not be precise, since the CT band seems to be originated by different species. The same EA for all the molecules is consistent with a large decoupling between the NDI core and the substituent, or that it is just extended to the first couple of carbons of the substituent.
We then conducted photoluminescence (PL) studies (Fig. S3). Upon excitation at 365 nm, NDI-Ph and NDI-Nap exhibit a fluorescence band near 420 nm, along with a second emission band at ca. 550 nm observed for NDI-Nap, NDI-An, and NDI-Th, while NDI-Ph lacks this red-shifted emission. This red-shifted emission intensifies with larger substituents.
While NDI derivatives are known to form aggregates that could influence optical properties, we confirmed the absence of such effects in our system. To exclude potential aggregation effects in our characterisation, we measured the absorbance of NDI-An (the paradigmatic system in this study) across a concentration range spanning two orders of magnitude, including the concentration where the further characterisation is performed (Fig. S3b). The spectra show no changes in the relative absorbance of the NDI core and the CT band, nor any baseline variations. This result confirms the absence of aggregation within the concentration range investigated along this work.
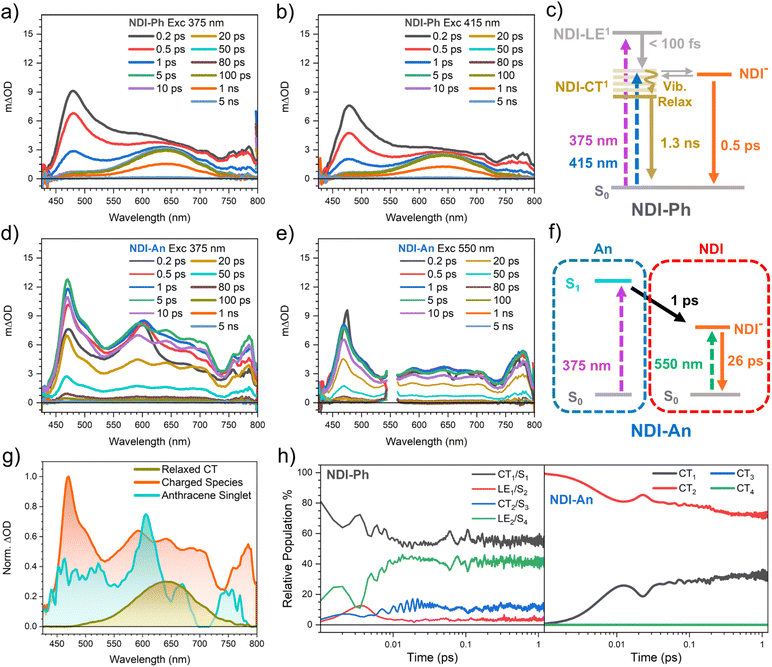
To better understand how the optical properties relate to charge behaviour, fs-TAS was performed in 2-MeTHF solutions. Fig. 2 shows the results with the smallest and the largest substituent, NDI-Ph and NDI-An, respectively. The NDI-Nap and NDI-Th, the intermediate size molecules are shown in the SI. Two different excitation wavelengths were selected for the TAS experiments: one matching the absorbance of the NDI core (at 375 nm) and the other one exciting the lower absorbance band associated to the CT transition (in the range of 420–550 nm).
Upon exciting NDI-Ph at 375 nm, a large band at 480 nm that extends in the 550–800 nm region with a broad absorption is observed. This band decays with a lifetime of 0.5 ps to leave only the broad band centred at 650 nm with a lifetime of 1.3 ns. Upon excitation of NDI-Ph at the band at 415 nm, a similar TA spectrum was obtained with identical bands (see Fig. 2a and b).
On the other hand, upon excitation of NDI-An at 375 nm (Fig. 2d), where anthracene and the NDI core both absorb, a narrow band is observed at 600 nm at 0.2 ps. This 600 nm narrow band decays with a lifetime of 1 ps. In addition, there is a spectral feature with a band at 470 nm that extends into the infrared. This 470 nm feature rises at the same time the 600 nm band decays. This 470 nm band is almost identical to NDI-Ph TA spectrum at early times. However, in this case, the spectral feature at 470 nm is much more defined, with additional bands at 590, 638, 700 and 785 nm. When exciting the NDI-An band at 550 nm, only the defined feature with the 470 nm band is seen. Under both excitation wavelengths the defined 470 nm feature fully decays with a lifetime of 26 ps.
To help elucidate the photoexcitation mechanism we performed global analysis on the TA data for all the molecules studied (Fig. S2). This analysis provided three different species: a broad band centred at 650 nm, the multiband feature with the maximum absorbance at 470–480 nm and the narrow band at 600 nm. First, we can rule out the presence of triplet species as, their absorbance spectrum does not match 3NDI absorbance in the literature, neither Tn nor T1.15,24,68 Also, the triplet lifetime is expected to be much larger (microsecond timescale) due to the forbidden triplet to singlet ground state transition. The defined multiband TA feature with its maximum at ca. 470 nm, is assigned to a NDI charged species due to its similarity to the NDI radical anion spectrum measured in literature.15,69 Then, the next species, the sharp band at 600 nm can be either associated with the absorbance of the anthracene or NDI core singlet exciton due to its similarity to the anthracene15,70,71 and pale NDI24,72 singlet absorbance seen in literature.15,68,69 The ground state absorbance of pristine NDI and anthracene molecules in the same region makes difficult to calculate the percentage of light absorbed by either unit. However, the similarity of the ground state absorbance of NDI-An to the anthracene absorbance, with that particular vibronic progression, we can infer that a larger portion of light is going to be absorbed by the anthracene unit. This larger absorption of the anthracene unit suggests the 600 nm band to be associated with the anthracene singlet. Finally, the global analysis provides a featureless broad band at 650 nm that does not appear for NDI-An which position is slightly displaced (0.16 eV) relative to the localised NDI singlet exciton seen in literature: a sharp band at 600 nm.24,72 However, this state is accessible by excitation at lower energies NDI-Ph and NDI-Nap, confirming it does not originated from the pristine NDI core singlet. We instead assign this species to a relaxed singlet with a significant CT character. While the electronic density is primarily localised on the NDI core, it is expected to extend to the first carbon atoms of the phenyl and naphthalene substituents, in line with a larger coupling of these substituents and the NDI core (see in the Calculations section). The substantial CT character, combined with an extended conjugation facilitated by the substituent's carbon atoms, explains both the observed red shift and the broader spectral profile compared to the unsubstituted NDI singlet. The lifetime of this species, which matches the measured PL lifetime (Fig. S3), hence, suggests this species as the origin for the PL for NDI-Ph and NDI-An. The assignment for the PL to a state with a large CT character, as opposed to a localised state (LE), is in agreement with a less vibronically defined PL spectrum and the red-shift seen for our compounds in comparison to the pristine NDI PL seen in literature.24,72 Therefore, we can safely assign our band at 650 nm to a relaxed singlet with a large CT character. Therefore, we hypothesise that upon excitation of the NDI-Ph at 375 nm, the NDI core singlet is swiftly, in less than 100 fs since it is not seen in the fs-TAS, transformed into a hot CT state. This hot CT state would be in equilibrium with the NDI charged species, but upon relaxation, this equilibrium is no longer possible. Then, this relaxed CT state decays into ground state with 1 ns lifetime. This relaxation in the ps timescale, which hinders the transfer to another state, agrees with other molecules seen in literature.73,74 The direct excitation of the CT state, at 415 nm, populates the hot CT state, and the TA spectrum gave similar results. The NDI-Nap shows a similar trend to NDI-Ph, but NDI-Nap shows a lower weight of the hot CT state when excited at longer wavelengths (Fig. S5). This lower weight of the hot CT state is coherent with a more stable CT state given by the larger extension for the conjugation. In addition, it is possible to see a ps decay of the NDI-Nap relaxed CT state in line with a vibrational relaxation process. Therefore, the NDI-Nap data further supports the assignment of this 650 nm band to a relaxed CT state.
Following the assignment made in the last paragraph, the lack of NDI singlet excitons upon exciting NDI-An became apparent, while there is a considerable formation of singlet excitons, with large CT character, in NDI-Ph, with the NDI-Nap remaining as an intermediate case. In addition, this assignment also explains the differences seen for different excitation wavelengths for NDI-An: exciting the NDI-An anthracene moiety, the anthracene singlet exciton is formed, which is swiftly transformed into charged NDI moiety in the hundreds of femtosecond timescale. The fast generation of charges seen for all the molecules studied in this work is remarkable, but it is a sensible affirmation if we see other more conjugated NDI molecules in literature.75–77 As discussed earlier, there appears to be a pattern in the charge generation depending on the substituent where the NDI derivative with the largest substituent, NDI-An, generates a larger portion of charges in comparison with the NDI derivative with the smaller phenyl substituent, NDI-Ph.
Theoretical calculations
We performed calculations to better understand the behaviour of these molecules. First, we have performed time-dependent density-functional theory (TD-DFT) calculations using TD-B3LYP-D3/def2-SVP method on all molecules. Interestingly, in all cases the LUMO orbital has exactly the same topology, strongly localised on the NDI core unit (Fig. S15 and S16). This is a clear indication of the decoupling of the NDI core and the substituent, and agrees with the experimental CV data, demonstrating the validity of the calculations. To quantify the electronic coupling associated with charge-transfer processes in the NDI-H simplified models, we employed the generalized Mulliken–Hush (GMH) and fragment charge difference (FCD) schemes as implemented in Q-Chem (more information can be found in the SI). These calculations yielded electronic coupling values (HDA, FCD/GMH) of 0.353/0.326 and 0.031/0.032 eV for NDI-Ph and NDI-An, respectively. As expected, these values correspond to the largest and smallest couplings among the series, respectively. Our calculated NDI-Ph coupling values completely match with others, calculated from experimental data, found in literature.23 In contrast, in the same reference, Gurzadyan et al. find a coupling value for a structurally similar system to NDI-An that falls within the same range as our value for NDI-Ph. In their system, the intrinsic donor character of anthracene is masked by the more strongly donating amine substituent. Consequently, all their H2N-NDI derivatives exhibit nearly identical ground-state absorption spectra, indicating the amine dominates the electronic properties. The low coupling value we calculate for NDI-An is, in fact, consistent with systems where coupling is deactivated by meta-substitution or orthogonal geometries, further validating the accuracy of our computational approach.78–80To confirm the assignment done in the fs-TAS section, excited state simulations, upon excitation of the lowest energy band, were performed using the ORCA81 and Q-Chem82 software packages at the TD-B3LYP/def2-SVP level of theory. These calculations provided the necessary parameters for subsequent non-adiabatic quantum dynamics simulations. To perform non-adiabatic quantum dynamic calculations, the ML-MCTDH83 method was used as implemented in the Quantics package,84 after constructing potential energy surfaces based on linear vibronic coupling models (see SI for calculation details).85,86 As shown in Fig. 2h, NDI-Ph showed the presence of LE singlet states alongside CT states whereas NDI-An showed only CT states. NDI-Ph shows a dependent exchange of two different states, namely S1 and S4, with LE and CT character, respectively, which represent the largest population in the first femtoseconds (95% of the population upon excitation). According to the topology of this S1 state (πAQbenz, Fig. S11), the CT character is not that pronounced as for NDI-An, which agrees with the relaxed CT state seen in fs-TAS. In addition, there is a certain population of a purer CT character (HOMO strongly localised over the phenyl group) state, S3, which can be associated with the charged species seen in the fs-TAS. On the other hand, there is only two singlets, with CT character, with enough population on the calculated dynamics of NDI-An, which interconvert one into another (CT1 and CT2). Then, according to these calculations CT1 and CT2 coexist in the first picosecond, but the CT2 is transforming into the CT1. According to the topology of these states (Fig. S14), the CT1 and CT2 are very similar, with most of the electronic density localised over the anthracene moiety, but CT1 shows the anthracene accommodating the positive charge with the NDI core oxygen through space. Therefore, the dynamics seen for NDI-An upon excitation of the CT band show the excitation of the anthracene moiety to then stabilise the positive charge through space with the NDI oxygen. Interestingly, despite finding isoenergetic triplet energy levels in our calculations (Fig. S17), the calculated dynamics did not show any population in the picosecond timescale of triplet states unlike in other NDI derivatives from literature.24 Spin–orbit coupling (SOC) calculations on NDI-Ph and NDI-An (Fig. S18) confirm the lack of triplet state population due to a very low SOC between excited singlet and triplet states.
Crystallographic structural analyses
The differences in excited state dynamic with the substituent size led us to further study their molecular structure using single crystal X-ray diffraction analyses, which was then further probed using computational approaches (see the Discussion section). Single crystals of NDI-Th, NDI-Ph and NDI-An were grown through solvent diffusion of diethyl-ether into THF solutions of the NDI derivatives. X-ray diffraction studies have shown that NDI-Th and NDI-Ph crystallise as isomorphous THF solvates in the monoclinic P21/n space group. The crystallisation of NDI-An resulted in the concomitant formation of two solvates – a diethyl-ether solvate and a diethyl-ether-THF solvate. Both crystallise in the triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group (see Section 6 in the SI document).
space group (see Section 6 in the SI document).
In all crystal structures, the NDI derivatives feature a non-planar conformation with various degrees of tilting between the NDI core and the flanking substituents. Specifically, the disordered thiophenyl groups in NDI-Th are tilted about 69° (in both occupancy sites), the phenyl groups in NDI-Ph flank the NDI core at a 65° angle, while the anthracenyl groups in both discovered NDI-An crystal forms assume angles in the range of 84–88°, almost perpendicular to the NDI core (Fig. 3). We note that no crystal form screens have been pursued to evaluate the existence of NDI derivatives with ‘flat’ molecular structures. But based on the crystallographic and computational analyses, we reason that, in the solid state, the studied NDI derivatives (and particularly those with bulkier substituents) favour more orthogonal arrangements of the NDI core and the substituent. It is therefore, expected that, in solution, the media where the spectroscopic characterisation has been made, the orthogonal conformation still persists. The largely orthogonal conformation of the studied molecules is consistent with the charge generation discussed previously. Free rotation, with a subsequent increase of electronic coupling, would reduce free charge generation efficiency and/or increase charge recombination losses.
Discussion
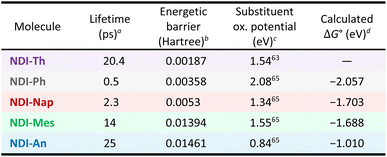
To calculate the extent of the orthogonality impact in the charge generation, further calculations were performed to quantify the steric hindrance of the substituent on these molecules (Fig. 4a). The calculations show the expected behaviour with a larger rotational energy barrier with a larger substituent. Interestingly, the calculated potential scan is not fully symmetrical. This is due to the rapid localisation of the electronic density on one of the oxygens, and therefore creating an asymmetry in the NDI core. That is also the reason why the energy minimum, for instance, for the NDI-Ph, is not exactly zero but around 60° (matching the XRD data). Of all the NDI derivatives, the NDI-Th displayed the lowest rotational energy barrier due the smaller size of the thienyl substituent leading to the lowest steric hindrance. This is in stark contrast to NDI-An which presents the highest energetic barrier to rotation, due to the significant steric hindrance between the NDI core and the anthracene moiety, largely preventing free rotation (Fig. 4a).The increase in the oxidation potential, due to larger conjugation of the substituent,87 and, therefore, the stronger donor character, may emerge as an explanation for more efficient charge formation, either in place of or alongside orthogonality. To rule out this possibility we synthesised an additional NDI molecule with a mesityl group as substituent (NDI-Mes, see its spectroscopic analysis in Fig. S5). The mesityl has a considerably lower oxidation potential than anthracene, actually its oxidation potential, directly related with the donor strength, places it below the naphthalene.64,65 However, despite its lower oxidation potential and a much lower conjugation length it possesses a large steric hindrance (with a similar calculated steric hindrance to NDI-An, Fig. 4a).
We have conducted TAS characterisation of this novel molecule under identical conditions to the other four (Fig. 4b and S5). As seen, right upon excitation, only the feature with a band at 480 nm which extends to the NIR, associated previously with charges, appears. This feature decays with a 14 ps lifetime. Interestingly, the NDI-Mes charges have a less defined spectrum. We hypothesise that the origin for this less defined spectrum is a lower effective charge separation, i.e., shorter distance between charges, for this state than for NDI-An.35 This is in agreement with the much lower size of the mesityl group in comparison to anthracene which makes the separation of the positive and negative electronic densities less effective. However, in this case, there is a signal originated at large times (over 50 ps) which is assigned to NDI triplet state.15,24,68 In previously reported systems, such triplets typically originate from the higher-lying local singlet state via efficient intersystem crossing (ISC), which can occur in less than 200 fs. In NDI-Mes, however, triplet formation does not proceed through ISC from a local state. Instead, it occurs via charged species, a mechanism also observed in other NDIs functionalized with amines or imide-substituted group.15,72 Nevertheless, the ISC efficiency in NDI-Mes is notably lower, as evidenced by the significantly weaker triplet signal compared to those earlier examples. This conclusion rests on the reasonable assumption that the relative absorptivity of the excited species in NDI-Mes is comparable to that in the previously reported NDI derivatives.
As stated before, the introduction of this mesityl substituted molecule to this work neglects the oxidation potential, as experimental figure of merit for the donor strength, of the substituents as the main origin for the charge generation process (Fig. S7). The use of the pure donor oxidation potential as donor strength character was pertinent since the molecules showed considerable decoupling between the donor and acceptor moieties, as indicated by the CV measurements and calculations. Indeed, the CV revealed only a 50 meV variation in reduction potential across the molecules of greatest interest, which could still introduce some error into the relationship.
Since substituent donor strength was not enough to justify these molecules behaviour, we focused on the substituent orthogonality. To quantify the orthogonality and the charge generation relationship we need a suitable way to assess a charge generation yield. However, the TA spectra reveal no detectable NDI singlet exciton signal at even the shortest measured delay (200 fs). Given an NDI singlet lifetime of 1 ps (a literature-based conservative estimation for solvent variation),24,72 at 200 fs, a significant portion of the initial NDI singlet population, calculated to be ca. 80%, should still remain. This lack of signal suggests an ultrafast charge generation process, thereby negating its suitability as a comparative metric. Then, we have used the charge lifetime as better approximation to distinguish between them. Still, charge lifetime does not express, by itself, the charge generation yield but it is directly related to the charge stability and is a valuable comparison for application matters. This approximation is particularly valuable for the NDI-An, since co-excitation remains an issue to properly characterise the charge generation yield. Fig. 5 shows the relationship between the charge lifetime and the rotational energetic barrier. As seen, there is a correlation between the charge lifetime and the orthogonality. A monotonic increase in the calculated rotational barrier (Fig. 4a) increases the observed charged-state lifetime (Fig. 5), suggesting that restricted rotation may play an important role in stabilizing the CT state.
The NDI-Th does not follow the trend as the charge generation process in this molecule is not driven by geometry constraints but rather on electronic effects given by the large donor nature of the thiophene (as explained in the ground state absorbance discussion). The NDI-Th results suggest that charge generation in common literature NDI examples are not driven by molecular geometry but by electronic effects by introduction of either a donor or an acceptor atom/group.
We also performed calculations to investigate the relationship between the experimental charge decay rate with the ΔG (Table 1), following Marcus theory formalism (Fig. S8). We have calculated the ΔG as the electronic difference of the minimum of the S1 (with large CT character in all cases) and the S0 of truncated derivatives, to optimise the computation time. We estimate that changes in geometry from these two states would be minimal, and, therefore, changes in entropy should be negligible. This ΔG is compared to the experimental charge relaxation rate for all compounds. Performing the calculations in vacuum typically leads to larger reorganisation energies. However, applying the same vacuum conditions to all compounds preserves a consistent systematic error and allows us to isolate the influence of backbone torsion, minimizing any contributions from solvent effects. In this work, the rates follow a trend in the normal region of the Marcus theory with a faster decay rate for a larger −ΔG. To place these observations within the Marcus framework, it is useful to recall that the intramolecular charge-transfer rate depends not only on the free energy and reorganisation energy but also on the electronic coupling. Although the free-energy variations follow the expected trend, they cannot fully explain the large differences in charge relaxation. The geometric orthogonality modulates orbital overlap and thus the electronic coupling, providing a clearer explanation for the charge-distribution efficiency than free-energy changes alone.
| a Decay rate from charged species in TAS.b Calculated energetic barrier from unrestricted calculation at 30° between the NDI core and the substituent.c Oxidation potentials taken from literature vs. Ag/AgCl. These values are also in agreement with potentials under other conditions.64d Calculation details are provided in SI. |
|---|
 |
As explained in the introduction, to unequivocally unravel the identity of the charges generated in these molecules represents a challenge due to their similarities in terms of spectral properties.28 The small size of the molecules presented in this work seems to be insufficient to generate a separated charge species. However, the NDI-An and, to a lesser extent, NDI-Mes charges spectra show a larger definition and a slight shift in comparison with NDI-Ph and NDI-Nap charges. This suggests the formation of a larger effective separation of this CT state if not the formation of separated charges in line with other examples in literature where the difference between the CT and the separated charges are also a slight shift in the spectrum.28
The present work about intramolecular charge generation underpins a broad range of applications. Intramolecular charge generation usually constitutes the initial step in photocatalysis, generating the reactive species required for the reaction of interest.88,89 Efficient charge generation is also central to photovoltaics, where charges must be created and extracted before recombination occurs. This is especially critical in single-component photovoltaics, which avoid the donor:acceptor interfacial recombination that typically limits device performance.90,91 Furthermore, radicals play an important role in a large number of biological processes, and, hence, a fast radical formation is of great interest in the field of bioapplications.42,92
Conclusions
In conclusion, we have studied the implications of torsion-modulated on intramolecular charge generation in NDI molecules, focusing on the steric hindrance with lower donor strength building blocks. The solution study in TAS probes the formation of these charges prior the signal detection limit (hundreds of femtoseconds). A larger rotational barrier of the substituent correlates with longer-lived charged states, since reduced electronic coupling stabilises this species. In addition, the lifetime of the as-created charges is increased with the rotational barrier following an exponential growth, with an R2 value of 0.885. The introduction of this new recipe for charge generation is the first step in the production of a new family of molecules for single-component devices with better industrial scalability as it does not rely in very complex donor–acceptor design. However, the implications of this new approach for charge generation does not stop here as it has implications for a much broader range of applications from bioapplications, organic electronics to catalysis.Author contributions
HCB, CL, ABA and LMC have prepared the compounds. AMSD and SG have performed the theoretical calculations. DKB performed the crystallographic measurements and analyses. JMMB has performed the spectroscopic characterisation and analysis. HCB, ABT, BCS, SG and JMMB have elaborated the manuscript. JMMB supervised the project.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2413777–2413780 contain the supplementary crystallographic data for this paper.104a–dData for this article, including NMR, UV-vis, PL, psTAS and Raman spectra are available at UCL Research Data Repository at https://doi.org/10.5522/04/28203680. The data supporting this article have been included as part of the supplementary information (SI). The authors have cited additional references within the SI.93–103 Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc00456c.
Acknowledgements
JMMB wants to acknowledge the Spanish University Ministry and the European Union for his Maria Zambrano fellowship with NextGen-Eu funding and the University of Malaga for the Young Researchers Projects (1-2021_17). JMMB also wants to thank the Research Central Services (SCAI) of the University of Málaga for access to the EVI, EEL, and MENL to perform ground-state and transient spectroscopic characterization. Simulations were performed using the high performance computing resources of the Castilla y León Supercomputing Center (SCAYLE), funded by the European Regional Development Fund (ERDF). AMS thanks the Spanish Ministry of Science and Innovation (MCIN/AEI/10.13039/501100011033) grant no. PID2020-113147GA-I00. SG thanks the EPSRC under the COSMOS programme grant (EP/X026973/1) and the Spanish Ministry of Science and Innovation (MCIN/AEI/10.13039/501100011033) grant PID2024-162002NB-I00. BCS acknowledges funding from the UK Research and Innovation for Future Leaders Fellowship (MR/S031952/1). This work was supported by the CRUK City of London Centre Award (C7893/A26233) and (CTRQQR-2021/100004).Notes and references
- J. Nelson, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 67, 155209 CrossRef.
- X. Lan, X. Liu, Y. Zhang, Q. Li, J. Wang, Q. Zhang and G. Bai, ACS Catal., 2021, 11, 7429–7441 CrossRef CAS.
- J. Zaumseil, R. H. Friend and H. Sirringhaus, Nat. Mater., 2006, 5, 69–74 CrossRef CAS.
- H. B. Naveed and W. Ma, Joule, 2018, 2, 621–641 CrossRef CAS.
- A. Guerrero and G. Garcia-Belmonte, Nano–Micro Lett., 2017, 9, 10 CrossRef PubMed.
- M. S. Vezie, M. Azzouzi, A. M. Telford, T. R. Hopper, A. B. Sieval, J. C. Hummelen, K. Fallon, H. Bronstein, T. Kirchartz, A. A. Bakulin, T. M. Clarke and J. Nelson, ACS Energy Lett., 2019, 4, 2096–2103 CrossRef CAS.
- D. Baran, R. S. Ashraf, D. A. Hanifi, M. Abdelsamie, N. Gasparini, J. A. Röhr, S. Holliday, A. Wadsworth, S. Lockett, M. Neophytou, C. J. M. Emmott, J. Nelson, C. J. Brabec, A. Amassian, A. Salleo, T. Kirchartz, J. R. Durrant and I. McCulloch, Nat. Mater., 2017, 16, 363–369 CrossRef CAS PubMed.
- J. Hamonnet, M. Nakano, K. Nakano, H. Sugino, K. Takimiya and K. Tajima, Chem. Mater., 2017, 29, 9618–9622 CrossRef CAS.
- E. Ahmed, G. Ren, F. S. Kim, E. C. Hollenbeck and S. A. Jenekhe, Chem. Mater., 2011, 23, 4563–4577 CrossRef CAS.
- N. Zhou and A. Facchetti, Mater. Today, 2018, 21, 377–390 CrossRef CAS.
- J. H. Oh, S. Suraru, W. Lee, M. Könemann, H. W. Höffken, C. Röger, R. Schmidt, Y. Chung, W. Chen, F. Würthner and Z. Bao, Adv. Funct. Mater., 2010, 20, 2148–2156 CrossRef CAS.
- B. A. Jones, A. Facchetti, T. J. Marks and M. R. Wasielewski, Chem. Mater., 2007, 19, 2703–2705 CrossRef CAS.
- B. J. Jung, K. Lee, J. Sun, A. G. Andreou and H. E. Katz, Adv. Funct. Mater., 2010, 20, 2930–2944 CrossRef CAS.
- N. T. La Porte, J. A. Christensen, M. D. Krzyaniak, B. K. Rugg and M. R. Wasielewski, J. Phys. Chem. B, 2019, 123, 7731–7739 CrossRef CAS PubMed.
- X. Chen, A. A. Sukhanov, M. Taddei, B. Dick, J. Zhao, V. K. Voronkova and M. Di Donato, J. Phys. Chem. Lett., 2022, 13, 8740–8748 CrossRef CAS PubMed.
- S. Chakraborty, D. Ray, V. K. Aswal and S. Ghosh, Chem.–Eur. J., 2018, 24, 16379–16387 CrossRef CAS PubMed.
- M. K. Gish, A. L. Jones, J. M. Papanikolas and K. S. Schanze, J. Phys. Chem. C, 2018, 122, 18802–18808 CrossRef CAS.
- K. Cai, J. Xie, X. Yang and D. Zhao, Org. Lett., 2014, 16, 1852–1855 CrossRef CAS PubMed.
- K. Cai, J. Xie and D. Zhao, J. Am. Chem. Soc., 2014, 136, 28–31 CrossRef CAS PubMed.
- K. Kranthiraja, D. X. Long, V. G. Sree, W. Cho, Y.-R. Cho, A. Zaheer, J.-C. Lee, Y.-Y. Noh and S.-H. Jin, Macromolecules, 2018, 51, 5530–5536 CrossRef CAS.
- Y. Zhao, X. Li, Z. Wang, W. Yang, K. Chen, J. Zhao and G. G. Gurzadyan, J. Phys. Chem. C, 2018, 122, 3756–3772 CrossRef CAS.
- P. Pahlavanlu, A. J. Tilley, B. T. McAllister and D. S. Seferos, J. Org. Chem., 2017, 82, 12337–12345 CrossRef CAS PubMed.
- K. Chen, J. Zhao, X. Li and G. G. Gurzadyan, J. Phys. Chem. A, 2019, 123, 2503–2516 CrossRef CAS PubMed.
- O. Yushchenko, G. Licari, S. Mosquera-Vazquez, N. Sakai, S. Matile and E. Vauthey, J. Phys. Chem. Lett., 2015, 6, 2096–2100 CrossRef CAS PubMed.
- J. Shaikh, D. G. Congrave, A. Forster, A. Minotto, F. Cacialli, T. J. H. Hele, T. J. Penfold, H. Bronstein and T. M. Clarke, Chem. Sci., 2021, 12, 8165–8177 RSC.
- S. Kasemthaveechok, L. Abella, M. Jean, M. Cordier, T. Roisnel, N. Vanthuyne, T. Guizouarn, O. Cador, J. Autschbach, J. Crassous and L. Favereau, J. Am. Chem. Soc., 2020, 142, 20409–20418 CrossRef CAS PubMed.
- Y. Wu, S. Schneider, Y. Yuan, R. M. Young, T. Francese, I. F. Mansoor, P. J. Dudenas, Y. Lei, E. D. Gomez, D. M. DeLongchamp, M. C. Lipke, G. Galli, M. R. Wasielewski, J. B. Asbury, M. F. Toney and Z. Bao, Adv. Energy Mater., 2022, 12, 2103957 CrossRef CAS.
- G. J. Moore, F. Günther, K. M. Yallum, M. Causa’, A. Jungbluth, J. Réhault, M. Riede, F. Ortmann and N. Banerji, Nat. Commun., 2024, 15, 9851 CrossRef CAS PubMed.
- H. Langhals, S. Kinzel and A. Obermeier, J. Org. Chem., 2024, 89, 2138–2154 CrossRef CAS PubMed.
- A. M. El-Zohry, E. A. Orabi, M. Karlsson and B. Zietz, J. Phys. Chem. A, 2021, 125, 2885–2894 CrossRef CAS PubMed.
- Y. Yao, P. Ding, C. Yan, Y. Tao, B. Peng, W. Liu, J. Wang, M. A. Cohen Stuart and Z. Guo, Angew. Chem., Int. Ed., 2023, 62, e202218983 CrossRef CAS PubMed.
- E. Wang, J. W. Y. Lam, R. Hu, C. Zhang, Y. S. Zhao and B. Z. Tang, J. Mater. Chem. C, 2014, 2, 1801 RSC.
- M. A. Haidekker and E. A. Theodorakis, Org. Biomol. Chem., 2007, 5, 1669–1678 RSC.
- J. M. Marin-Beloqui, D. G. Congrave, D. T. W. Toolan, S. Montanaro, J. Guo, I. A. Wright, T. M. Clarke, H. Bronstein and S. D. Dimitrov, J. Am. Chem. Soc., 2023, 145, 3507–3514 CrossRef CAS PubMed.
- J. M. Marin-Beloqui, K. J. Fallon, H. Bronstein and T. M. Clarke, J. Phys. Chem. Lett., 2019, 10, 3813–3819 CrossRef CAS PubMed.
- J. Marin-Beloqui, G. Zhang, J. Guo, J. Shaikh, T. Wohrer, S. M. Hosseini, B. Sun, J. Shipp, A. J. Auty, D. Chekulaev, J. Ye, Y.-C. Chin, M. B. Sullivan, A. J. Mozer, J.-S. Kim, S. Shoaee and T. M. Clarke, J. Phys. Chem. C, 2022, 126, 2708–2719 CrossRef CAS.
- T. M. Clarke, A. Ballantyne, S. Shoaee, Y. W. Soon, W. Duffy, M. Heeney, I. McCulloch, J. Nelson and J. R. Durrant, Adv. Mater., 2010, 22, 5287–5291 CrossRef CAS PubMed.
- C. G. Shuttle, B. O'Regan, A. M. Ballantyne, J. Nelson, D. D. C. Bradley and J. R. Durrant, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 78, 113201 CrossRef.
- E. Busby, J. Xia, Q. Wu, J. Z. Low, R. Song, J. R. Miller, X.-Y. Zhu, L. M. Campos and M. Y. Sfeir, Nat. Mater., 2015, 14, 426–433 CrossRef CAS PubMed.
- J. S. Ward, R. S. Nobuyasu, A. S. Batsanov, P. Data, A. P. Monkman, F. B. Dias and M. R. Bryce, Chem. Commun., 2016, 52, 2612–2615 RSC.
- S. D. Dimitrov, S. Wheeler, D. Niedzialek, B. C. Schroeder, H. Utzat, J. M. Frost, J. Yao, A. Gillett, P. S. Tuladhar, I. McCulloch, J. Nelson and J. R. Durrant, Nat. Commun., 2015, 6, 6501 CrossRef CAS PubMed.
- X. Cui, Z. Zhang, Y. Yang, S. Li and C. Lee, Exploration, 2022, 2, 20210264 CrossRef CAS PubMed.
- A. Sarkar, T. Behera, R. Sasmal, R. Capelli, C. Empereur-mot, J. Mahato, S. S. Agasti, G. M. Pavan, A. Chowdhury and S. J. George, J. Am. Chem. Soc., 2020, 142, 11528–11539 CrossRef CAS PubMed.
- A. Sarkar, R. Sasmal, A. Das, A. Venugopal, S. S. Agasti and S. J. George, Angew. Chem., Int. Ed., 2021, 60, 18209–18216 CrossRef CAS PubMed.
- M. Sasikumar, Y. V. Suseela and T. Govindaraju, Asian J. Org. Chem., 2013, 2, 779–785 CrossRef CAS.
- T. A. Welsh, O. Matsarskaia, R. Schweins and E. R. Draper, New J. Chem., 2021, 45, 14005–14013 RSC.
- X. Chen, Y. He, M. U. Ali, Y. He, Y. Zhu, A. Li, C. Zhao, I. F. Perepichka and H. Meng, Sci. China Chem., 2019, 62, 1360–1364 CrossRef CAS.
- B. Reiß and H.-A. Wagenknecht, Beilstein J. Org. Chem., 2019, 15, 2043–2051 CrossRef PubMed.
- K. Sonogashira, Y. Tohda and N. Hagihara, Tetrahedron Lett., 1975, 16, 4467–4470 CrossRef.
- N. Miyaura, K. Yamada and A. Suzuki, Tetrahedron Lett., 1979, 20, 3437–3440 CrossRef.
- M. Frigoli, M. Caldara, J. Royakkers, J. W. Lowdon, T. J. Cleij, H. Diliën, K. Eersels and B. van Grinsven, Microchem. J., 2024, 200, 110433 CrossRef CAS.
- J. Royakkers, K. Guo, D. T. W. Toolan, L. Feng, A. Minotto, D. G. Congrave, M. Danowska, W. Zeng, A. D. Bond, M. Al-Hashimi, T. J. Marks, A. Facchetti, F. Cacialli and H. Bronstein, Angew. Chem., Int. Ed., 2021, 60, 25005–25012 CrossRef CAS PubMed.
- P. Piyakulawat, A. Keawprajak, A. Chindaduang, M. Hanusch and U. Asawapirom, Synth. Met., 2009, 159, 467–472 CrossRef CAS.
- J. K. Stille, Angew Chem. Int. Ed. Engl., 1986, 25, 508–524 CrossRef.
- P. Espinet and A. M. Echavarren, Angew. Chem., Int. Ed., 2004, 43, 4704–4734 CrossRef CAS PubMed.
- S. Kuila, A. Ghorai, P. K. Samanta, R. B. K. Siram, S. K. Pati, K. S. Narayan and S. J. George, Chem.–Eur. J., 2019, 25, 16007–16011 CrossRef CAS PubMed.
- H. A. M. van Mullekom, J. A. J. M. Vekemans and E. W. Meijer, Chem.–Eur. J., 1998, 4, 1235–1243 CrossRef CAS.
- B. Grimm, C. Risko, J. D. Azoulay, J.-L. Brédas and G. C. Bazan, Chem. Sci., 2013, 4, 1807 RSC.
- L. Pandey, C. Risko, J. E. Norton and J.-L. Brédas, Macromolecules, 2012, 45, 6405–6414 CrossRef CAS.
- Y. S. Rosokha, S. V. Lindeman, S. V. Rosokha and J. K. Kochi, Angew. Chem., Int. Ed., 2004, 43, 4650–4652 CrossRef CAS PubMed.
- H. T. Chifotides, B. L. Schottel and K. R. Dunbar, Angew. Chem., Int. Ed., 2010, 49, 7202–7207 CrossRef CAS PubMed.
- B. Han, J. Lu and J. K. Kochi, Cryst. Growth Des., 2008, 8, 1327–1334 CrossRef CAS.
- M. B. Camarada, P. Jaque, F. R. Díaz and M. A. del Valle, J. Polym. Sci., Part B: Polym. Phys., 2011, 49, 1723–1733 CrossRef CAS.
- L. Eberson and K. Nyberg, J. Am. Chem. Soc., 1966, 88, 1686–1691 CrossRef CAS.
- W. C. Neikam and M. M. Desmond, J. Am. Chem. Soc., 1964, 86, 4811–4814 CrossRef CAS.
- S. B. Schmidt, T. Biskup, X. Jiao, C. R. McNeill and M. Sommer, J. Mater. Chem. C, 2019, 7, 4466–4474 RSC.
- S. V. Rosokha and J. K. Kochi, in Halogen Bonding, Springer Berlin Heidelberg, Berlin, Heidelberg, pp. , pp. 137–160 Search PubMed.
- A. Aster, C. Rumble, A.-B. Bornhof, H.-H. Huang, N. Sakai, T. Šolomek, S. Matile and E. Vauthey, Chem. Sci., 2021, 12, 4908–4915 RSC.
- D. Gosztola, M. P. Niemczyk, W. Svec, A. S. Lukas and M. R. Wasielewski, J. Phys. Chem. A, 2000, 104, 6545–6551 CrossRef CAS.
- J. Sepioł, J. Lumin., 1986, 36, 115–120 CrossRef.
- B. Manna, R. Ghosh and D. K. Palit, J. Phys. Chem. C, 2015, 119, 10641–10652 CrossRef CAS.
- P. Ganesan, J. Baggerman, H. Zhang, E. J. R. Sudhölter and H. Zuilhof, J. Phys. Chem. A, 2007, 111, 6151–6156 CrossRef CAS PubMed.
- A. Singh, L. Weatherill, A. Agarwal, S. M, C. Tonnele, D. Casanova, R. Singh, F. Dias and I. Laskar, ChemRxiv, 2025, preprint, DOI:10.26434/chemrxiv-2025-pgpn8.
- K. Wang, X. You, X. Miao, Y. Yi, S. Peng, D. Wu, X. Chen, J. Xu, M. Y. Sfeir and J. Xia, J. Am. Chem. Soc., 2024, 146, 13326–13335 CrossRef CAS PubMed.
- M. E. El-Khouly, C. A. Wijesinghe, V. N. Nesterov, M. E. Zandler, S. Fukuzumi and F. D'Souza, Chem.–Eur. J., 2012, 18, 13844–13853 CrossRef CAS PubMed.
- Y. Mori, Y. Sakaguchi and H. Hayashi, J. Phys. Chem. A, 2002, 106, 4453–4467 CrossRef CAS.
- M. P. Debreczeny, W. A. Svec, E. M. Marsh and M. R. Wasielewski, J. Am. Chem. Soc., 1996, 118, 8174–8175 CrossRef CAS.
- S. Ito, T. Nagami and M. Nakano, J. Phys. Chem. A, 2016, 120, 6236–6241 CrossRef CAS PubMed.
- E. W. Castner, D. Kennedy and R. J. Cave, J. Phys. Chem. A, 2000, 104, 2869–2885 CrossRef CAS.
- Y. Hou, I. Kurganskii, A. Elmali, H. Zhang, Y. Gao, L. Lv, J. Zhao, A. Karatay, L. Luo and M. Fedin, J. Chem. Phys., 2020, 152, 114701 CrossRef CAS PubMed.
- F. Neese, Wiley Interdiscip. Rev. Comput. Mol. Sci., 2012, 2, 73–78 CrossRef CAS.
- Y. Shao, Z. Gan, E. Epifanovsky, A. T. B. Gilbert, M. Wormit, J. Kussmann, A. W. Lange, A. Behn, J. Deng, X. Feng, D. Ghosh, M. Goldey, P. R. Horn, L. D. Jacobson, I. Kaliman, R. Z. Khaliullin, T. Kuś, A. Landau, J. Liu, E. I. Proynov, Y. M. Rhee, R. M. Richard, M. A. Rohrdanz, R. P. Steele, E. J. Sundstrom, H. L. Woodcock, P. M. Zimmerman, D. Zuev, B. Albrecht, E. Alguire, B. Austin, G. J. O. Beran, Y. A. Bernard, E. Berquist, K. Brandhorst, K. B. Bravaya, S. T. Brown, D. Casanova, C.-M. Chang, Y. Chen, S. H. Chien, K. D. Closser, D. L. Crittenden, M. Diedenhofen, R. A. DiStasio, H. Do, A. D. Dutoi, R. G. Edgar, S. Fatehi, L. Fusti-Molnar, A. Ghysels, A. Golubeva-Zadorozhnaya, J. Gomes, M. W. D. Hanson-Heine, P. H. P. Harbach, A. W. Hauser, E. G. Hohenstein, Z. C. Holden, T.-C. Jagau, H. Ji, B. Kaduk, K. Khistyaev, J. Kim, J. Kim, R. A. King, P. Klunzinger, D. Kosenkov, T. Kowalczyk, C. M. Krauter, K. U. Lao, A. D. Laurent, K. V. Lawler, S. V. Levchenko, C. Y. Lin, F. Liu, E. Livshits, R. C. Lochan, A. Luenser, P. Manohar, S. F. Manzer, S.-P. Mao, N. Mardirossian, A. V. Marenich, S. A. Maurer, N. J. Mayhall, E. Neuscamman, C. M. Oana, R. Olivares-Amaya, D. P. O'Neill, J. A. Parkhill, T. M. Perrine, R. Peverati, A. Prociuk, D. R. Rehn, E. Rosta, N. J. Russ, S. M. Sharada, S. Sharma, D. W. Small, A. Sodt, T. Stein, D. Stück, Y.-C. Su, A. J. W. Thom, T. Tsuchimochi, V. Vanovschi, L. Vogt, O. Vydrov, T. Wang, M. A. Watson, J. Wenzel, A. White, C. F. Williams, J. Yang, S. Yeganeh, S. R. Yost, Z.-Q. You, I. Y. Zhang, X. Zhang, Y. Zhao, B. R. Brooks, G. K. L. Chan, D. M. Chipman, C. J. Cramer, W. A. Goddard, M. S. Gordon, W. J. Hehre, A. Klamt, H. F. Schaefer, M. W. Schmidt, C. D. Sherrill, D. G. Truhlar, A. Warshel, X. Xu, A. Aspuru-Guzik, R. Baer, A. T. Bell, N. A. Besley, J.-D. Chai, A. Dreuw, B. D. Dunietz, T. R. Furlani, S. R. Gwaltney, C.-P. Hsu, Y. Jung, J. Kong, D. S. Lambrecht, W. Liang, C. Ochsenfeld, V. A. Rassolov, L. V. Slipchenko, J. E. Subotnik, T. Van Voorhis, J. M. Herbert, A. I. Krylov, P. M. W. Gill and M. Head-Gordon, Mol. Phys., 2015, 113, 184–215 CrossRef CAS.
- H. Wang and M. Thoss, J. Chem. Phys., 2003, 119, 1289–1299 CrossRef CAS.
- G. A. Worth, Comput. Phys. Commun., 2020, 248, 107040 CrossRef.
- H. Köuppel, W. Domcke and L. S. Cederbaum, Multimode Molecular Dynamics Beyond the Born-Oppenheimer Approximation, in Advances in Chemical Physics, ed. I. Prigogine and S.A. Rice, 1984, pp. 59–246 Search PubMed.
- A. Martín Santa Daría, L. González-Sánchez and S. Gómez, Phys. Chem. Chem. Phys., 2024, 26, 174–184 RSC.
- J. L. Bredas, R. Silbey, D. S. Boudreaux and R. R. Chance, J. Am. Chem. Soc., 1983, 105, 6555–6559 CrossRef CAS.
- M. Fumanal, A. Ortega-Guerrero, K. M. Jablonka, B. Smit and I. Tavernelli, Adv. Funct. Mater., 2020, 30, 2003792 CrossRef CAS.
- F. Chen, T. Ma, T. Zhang, Y. Zhang and H. Huang, Adv. Mater., 2021, 33, 2005256 CrossRef CAS PubMed.
- S. M. Menke, N. A. Ran, G. C. Bazan and R. H. Friend, Joule, 2018, 2, 25–35 CrossRef CAS.
- H. B. Naveed, K. Zhou and W. Ma, Acc. Chem. Res., 2019, 52, 2904–2915 CrossRef CAS PubMed.
- J. Liu, M. Wu, R. Zhang and Z. P. Xu, View, 2021, 2, 20200139 CrossRef CAS.
- A. C. Thompson, H. M. Grimm, A. Gray Bé, K. J. McKnight and J. J. Reczek, Synth. Commun., 2015, 45, 1127–1136 CrossRef CAS.
- Y. Yu, J. Wang, Y. Cui, Z. Chen, T. Zhang, Y. Xiao, W. Wang, J. Wang, X.-T. Hao and J. Hou, J. Am. Chem. Soc., 2024, 146, 8697–8705 CrossRef CAS PubMed.
- P. Tang, T. Furuya and T. Ritter, J. Am. Chem. Soc., 2010, 132, 12150–12154 CrossRef CAS PubMed.
- Y. Chen, M. Chen and Y. Liu, Angew. Chem., Int. Ed., 2012, 51, 6181–6186 CrossRef CAS PubMed.
- N. N. Sergeeva, A. Scala, M. A. Bakar, G. O'Riordan, J. O'Brien, G. Grassi and M. O. Senge, J. Org. Chem., 2009, 74, 7140–7147 CrossRef CAS PubMed.
- CrysAllisPro 1.171.42.60a, Rigaku, 2022. Search PubMed.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Adv., 2015, 71, 3–8 CrossRef PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- G. M. Sheldrick, Acta Crystallogr., Sect. C: Struct. Chem., 2015, 71, 3–8 Search PubMed.
- C. B. Hübschle, G. M. Sheldrick and B. Dittrich, J. Appl. Crystallogr., 2011, 44, 1281–1284 CrossRef PubMed.
- B. Rees, L. Jenner and M. Yusupov, Acta Crystallogr., Sect. D: Biol. Crystallogr., 2005, 61, 1299–1301 CrossRef PubMed.
- (a) CCDC 2413777: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m0qs5; (b) CCDC 2413778: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m0qt6; (c) CCDC 2413779: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m0qv7; (d) CCDC 2413780: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m0qw8.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |