Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Biginelli dihydropyrimidines: a tunable class of alkyl radical precursors
Shahilan
Ratnam
,
Shreya
Unone
,
Nabeel
Alia
,
Enyu Denny
Hafeneger
and
Daniel
Janssen-Müller
 *
*
Institut für Organische und Biomolekulare Chemie, Georg-August-Universität Göttingen, Tammannstraße 2, 37077 Göttingen, Germany. E-mail: djansse@uni-goettingen.de
First published on 13th March 2026
Abstract
Alkyl-substituted dihydropyrimidines (DHPyms), synthesised via the Biginelli reaction, are introduced as tunable alternatives to 4-alkyl Hantzsch dihydropyridines (DHPs) in radical chemistry. Leveraging the modularity of the Biginelli reaction, we systematically explored the redox properties, UV/vis absorption, and synthetic potential of DHPyms in radical-mediated transformations, identifying dimethylamino-substituted DHPym as a highly reactive, bench-stable, and easily synthesised alkyl radical precursor.
Introduction
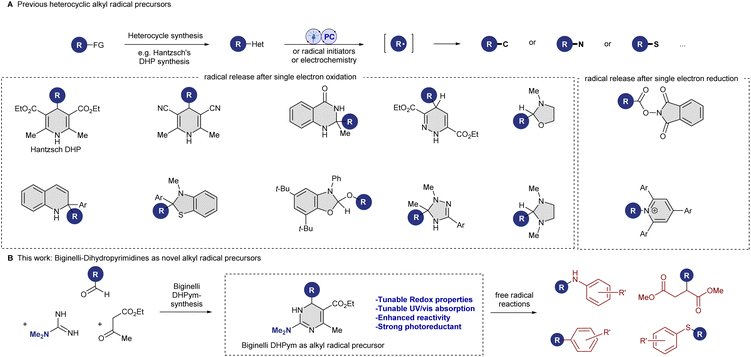
The development and use of organic precursors for the formation of alkyl radicals has emerged as a prominent area of research in organic synthesis. Notably, easily oxidizable redox-active heterocycles1 such as 4-alkyl Hantzsch esters, derived from aldehydes via the Hantzsch dihydropyridine (DHP) synthesis,2 have garnered significant attention due to their utility as precursors for radical-mediated transformations.3 These Hantzsch DHPs have enabled the utilization of aldehydes, a fundamental functional group present in a variety of synthetic and natural compounds, as substrates for alkyl radical formation. Radical formation strategies from these Hantzsch esters include thermal processes using peroxide reagents4 or photochemical activation through photoredox catalysis,5 sometimes in combination with transition-metal catalysis6 or organocatalysis.7 Moreover, photochemical activation without the need for a photocatalyst has been reported for Hantzsch DHPs.8Chemists have recently also investigated different heterocyclic alkyl radical precursors. These alternative redox-active heterocycles include dihydroquinazolinones,9 triazolines,10 and benzothiazolines,11 derived from ketones,12 oxazolidines and imidazolidines derived from aldehydes,13 dihydropyridazines from alkenes,14 dihydroquinolines derived from organometallic reagents,15 and benzoxazolines derived from alcohols (see Scheme 1A).16 A common feature among these auxiliaries is the occurrence of aromatization or resonance as the driving force for the release of the alkyl radical. Mechanistically, these oxidizable auxiliaries all operate through a similar pathway: single-electron oxidation or hydrogen-atom abstraction of the pro-aromatic heterocycle leads to a high-energy intermediate, which induces homolytic C–C bond cleavage, releasing an alkyl radical and often an aromatic heterocycle.
 | ||
| Scheme 1 (A) Previous N-heterocyclic redox-active radical precursors. (B) Biginelli-dihydropyrimidines as alkyl radical precursors. | ||
Motivated by the success of existing heterocyclic auxiliaries and the desire to expand the synthetic toolbox, we sought to investigate the potential of dihydropyrimidines (DHPyms) as novel substitutes for DHPs. DHPyms, readily accessible via the Hideg modification17 of the Biginelli reaction,18 present an intriguing opportunity to broaden the scope of heterocyclic redox auxiliaries and introduce novel variation and tuning possibilities. Inspired by the modularity of the Biginelli reaction (see Scheme 1B), which allows for facile access to structurally diverse compounds, we hypothesized that DHPyms could also act as reducing alkyl radical precursors and offer enhanced reactivity and synthetic versatility, addressing current limitations in radical chemistry. DHPyms have been described in the literature as readily oxidized to aromatic pyrimidines under various conditions, and they usually exist in solution as a mixture of 1,4-dihydropyrimidine and 3,4-dihydropyrimidine tautomers, whose ratio depends on solvent and substitution pattern.19 Kappe reported that by the choice of oxidant, an exocyclic C–C bond could be cleaved (MnO2, CAN, KMnO4) or retained (chloranil and DDQ) during aromatization,19a but so far their use as alkyl radical precursor is not described in the literature.
Results and discussion
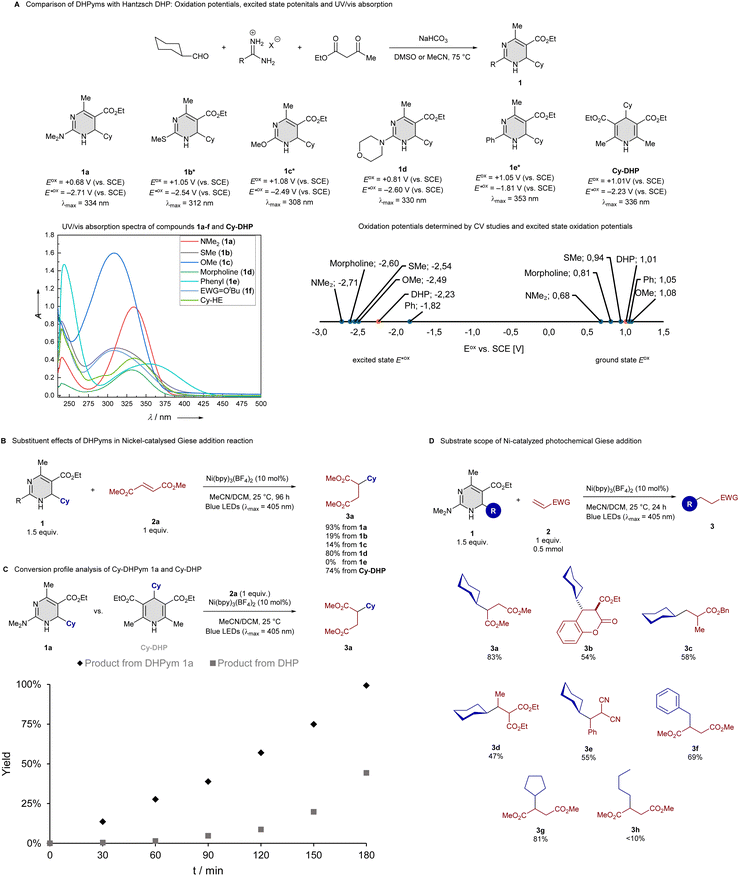
We started our investigation with the synthesis of some DHPyms, which was accomplished via the established Biginelli reaction, providing access to a diverse library with varying substitution patterns. We investigated cyclohexyl-substituted DHPyms 1a–e and compared them with cyclohexyl-substituted Hantzsch DHP by cyclic voltammetry experiments, which revealed a range of oxidation potentials (Eox) for DHPyms, spanning from 0.68 to 1.05 V, markedly lower than the typical Eox range of DHPs (1.0–1.2 V), with amino-substituted DHPym 1a being the most reducing, probably via resonance stabilization of the radical cation through the electron-donating amino-substituent. This observation prompted further investigation into the synthetic utility of DHPyms as alkyl radical precursors and raised the question about their excited state oxidation potential, since DHPs are often used as photoreductants with the capability to generate an alkyl radical. This combination of photoreductant and radical precursor reactivity led to a variety of interesting combinations of photochemistry and low-valent metal catalysis. We therefore measured fluorescence spectra and used the Rehm–Weller equation20 for determining excited state potentials, which revealed that DHPym reagent 1a had an excited state oxidation potential of −2.71 V, much lower than that of the common DHP reagent (see Scheme 2A). It should be noted that
of −2.71 V, much lower than that of the common DHP reagent (see Scheme 2A). It should be noted that 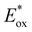 values, calculated from maximum absorption peaks, are overestimated relative to actual photosynthesis conditions at 390 or 405 nm and should be used only for comparative redox assessment of 1a–e. To compare the radical transfer capabilities of DHPyms, we decided to employ them under Melchiorre's photocatalyst-free Giese addition reaction, developed for Hantzsch DHPs and which uses Ni(bpy)3(BF4)2 as an electron-mediator.8s It turned out that amino-substituted DHPyms 1a and 1d were superior to the Hantzsch ester (see Scheme 2C). We then used DHPym 1a as our model reagent to study the conversion profile in comparison to the established DHP. We observed that our DHPym reagent 1a shows a strongly enhanced reactivity, most likely caused by its more negative excited state oxidation potential, which indicates high reducing power.
values, calculated from maximum absorption peaks, are overestimated relative to actual photosynthesis conditions at 390 or 405 nm and should be used only for comparative redox assessment of 1a–e. To compare the radical transfer capabilities of DHPyms, we decided to employ them under Melchiorre's photocatalyst-free Giese addition reaction, developed for Hantzsch DHPs and which uses Ni(bpy)3(BF4)2 as an electron-mediator.8s It turned out that amino-substituted DHPyms 1a and 1d were superior to the Hantzsch ester (see Scheme 2C). We then used DHPym 1a as our model reagent to study the conversion profile in comparison to the established DHP. We observed that our DHPym reagent 1a shows a strongly enhanced reactivity, most likely caused by its more negative excited state oxidation potential, which indicates high reducing power.
Motivated by this enhanced reactivity, we studied the substrate scope of the dimethylamino-substituted DHPym reagent in the Giese addition on 0.5 mmol scale with 24 h reaction time to ensure completion on the larger scale. We found that under Melchiorre's original conditions,8s but using a weaker LED, a variety of electron-deficient alkenes could be employed as well as secondary and benzylic alkyl radicals derived from the DHPym. Slight losses of product 3a compared to the excellent yields form the kinetic study arose from chromatographic purification. Primary alkyl radicals were much less effective, giving less than 10% of hydroalkylation product. For the primary example 3h, a significant amount of aromatized alkyl-pyrimidine was observed together with unreacted DHPym reagent, suggesting that a competing hydrogen-transfer reactivity becomes dominant for primary alkyl-substituted DHPym, which might be controllable through additional fine-tuning of substituents. It has to be noted however that the generation of primary radicals from DHP reagents is a common limitation in a variety of protocols. After having studied the Giese addition with a small selection of examples, we aimed to employ our reagent in some synthetically relevant radical transformation.
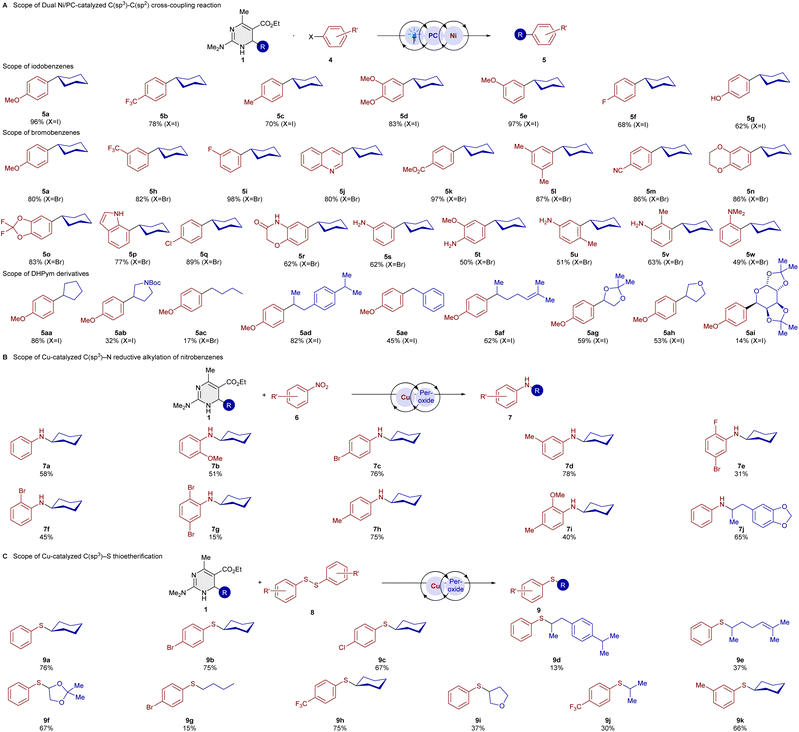
Motivated by reports on the Ni-catalysed cross-coupling with alkyl-DHP reagents,6a,b we discovered that our alkyl-DHPym reagents are also acting as competent coupling partners in a Ni-catalysed cross-coupling.
After reaction optimization (see SI for information), numerous examples of both bromobenzenes and iodobenzenes were tolerated (see Scheme 3A), bearing common functional groups such as ether (5d and 5e), ester (5k), cyano (5m), F (5f and 5i), Cl (5q) or CF3 (5b and 5h). Also for this alkyl radical reaction, an increased rate when compared to the DHP was observed (see SI for information). The cross-coupling with heterocyclic aryl halides such as dioxanes, dioxolanes, indoles, and quinolines gave very good yields. It is worth mentioning that our developed cross-coupling method of aryl iodides using DHPyms works in various solvents with a broad polarity range, from MeOH to PhMe, with very good yields, which can be beneficial for substrates for which solubility in acetonitrile bears a limitation (for further details, see the SI).
A major limitation of previously known methods is the cross-coupling of haloarenes possessing amines, which we could overcome with our method. Since our DHPym 1a has a much lower oxidation potential compared to the Hantzsch ester, we found that under Ni/Ir dual photocatalysis, the cross-coupling of unprotected bromoanilines was performed with a good yield.
Since our DHPym reagents are excellent reductants compared to Hantzsch DHPs, we wondered if they could be applied to the reductive alkylation of nitrobenzene derivatives, which was reported previously for DHPs as radical precursors.4m Our more reducing DHPym reagent turned out to be a competent alkylative reductant in this transformation (Scheme 3B). DHPym 1a with NMe2 and 1b with SMe-substitution had the same yield for aniline 7a in the reductive alkylation with 58%, whereas the OMe-DHPym 1c had a decreased yield with 39%. With dimethylamino-substituted DHPym we went on to study the substrate scope of this transformation. Good to very good yields could be obtained with 7c, 7d, and 7h, which did not show a clear tendency regarding electronic effects. Disubstitution with ortho–para- and ortho–meta-patterns were tolerated as well with good yields. Comparing products 7f and 7g, the disubstituted product had a decreased yield. The helional-derived DHPym reagent resulted in a good yield for 7j. We also became interested in a C–S bond-forming reaction with our DHPym reagents. C–S bond formation with Hantzsch esters is usually achieved with activated sulfur electrophiles such as TsSPh.5t
In our previous study on dihydroquinolines as alkyl radical precursors,15 we achieved a thiolation reaction with less reactive aryl disulfides under copper catalysis. With the even more reducing DHPym reagent 1a, we could achieve much higher yields of thiolation product 9a under similar reaction conditions as for our dihydroquinoline reagent (see Scheme 3C).
When studying the reaction scope of this transformation, we could demonstrate that multiple DHPym precursors to secondary alkyl radical can be utilized with moderate to good yields. A primary radical could be transferred (9g), even though a low yield was obtained.
To validate the formation of an alkyl radical, we added radical scavengers to the Ni-catalysed cross-coupling and we observed that TEMPO and galvinoxyl led to complete shutdown of the reactivity, while BHT led to a slightly reduced yield of 69% (see Scheme 4A). In the case of TEMPO as additive, the expected cyclohexyl TEMPO adduct was formed as evidenced by GC-MS and ESI-HRMS. A radical clock experiment with cyclohexylmethyl-substituted DHPym 1u led to ring-opened cross-coupling product 10, further validating the formation of alkyl radicals (Scheme 4B).
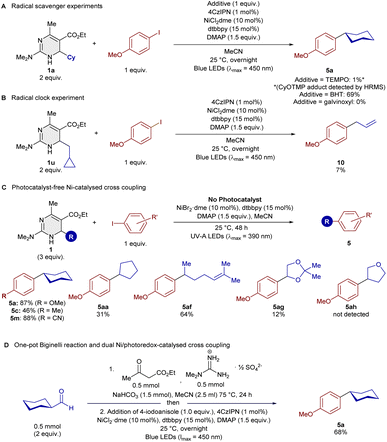 | ||
| Scheme 4 (A) Radical scavenger experiments. (B) Radical clock experiment. (C) Photocatalyst-free Ni-catalysed cross-coupling. (D) One-pot Biginelli cross-coupling reaction. | ||
A separate optimization of the Ni-catalyzed cross-coupling reaction, but without photocatalyst, led to reaction conditions with UV-A irradiation (390 nm), in which an excellent yield of 87% of cross-coupling product 5a was isolated (see Scheme 4C). A small scope study revealed some degree of generality, but addition of the 4CzIPN photocatalyst leads to higher yields whenever compared directly with the photocatalyst-free conditions.
We also attempted a one-pot approach successfully to the cross-coupling of DHPyms, in which cyclohexanecarboxaldehyde was first converted into the DHPym 1avia Biginelli reaction in MeCN before addition of the remaining starting materials for the cross-coupling in the same solvent and flask. Just a simple degassing step was performed after addition of the starting materials for the second step. This approach greatly improves the practicality of the process since the isolation of DHPyms after Biginelli reaction usually requires column chromatography.
Conclusions
In conclusion, our study introduces alkyl DHPyms as novel heterocyclic redox auxiliaries for alkyl radical transfer reactions, which are easily synthesised from aldehyde precursors and are bench-stable solids. The modularity of the Biginelli reaction enables facile access to structurally diverse DHPyms with tunable redox properties, offering exciting opportunities for innovation in radical-mediated transformations. Notably, the cross-coupling with these more-reducing electroauxiliaries turned out to tolerate free unprotected amines as substrates and shows a high degree of generality. The tunable nature of these Biginelli-derived DHPyms, combined with their superior reactivity compared to traditional Hantzsch esters, positions them as versatile tools for radical-mediated transformations under mild conditions. Beyond the demonstrated applications, we envision their utility in late-stage functionalization of complex molecules, such as pharmaceuticals, natural product derivatives, or peptides, where precise alkyl radical delivery enables diversification without disrupting sensitive functional groups. Furthermore, the straightforward one-step synthesis from abundant aldehydes opens avenues for incorporation into total synthesis routes.Author contributions
S. R., S. U., N. A., E. D. H. and D. J.-M. performed experiments. S. R., S. U. and D. J.-M. conducted mechanistic experiments. D. J.-M. conceived the project, supervised the research and wrote the manuscript with contributions from all authors.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2338773–2338775 contain the supplementary crystallographic data for this paper.21a–cThe data that support the findings of this study are available in the supplementary information (SI) of this article. Supplementary information: experimental procedures, characterization data, and NMR spectra. See DOI: https://doi.org/10.1039/d6sc00376a.
Acknowledgements
This work was supported by the Fonds der Chemischen Industrie (scholarships to D. J.-M. and S. R.). Prof. Dr Lutz Ackermann is greatly acknowledged for his continuous support. We thank Dr Christopher Golz for assistance with the X-ray diffraction analysis (both Göttingen University).Notes and references
- For reviews on Hantzsch esters, see: (a) A. Bhunia and A. Studer, Chem, 2021, 7, 2060–2100 CrossRef CAS ; for reviews on electroauxiliaries, see: ; (b) J. Yoshida, K. Kataoka, R. Horcajada and A. Nagaki, Chem. Rev., 2008, 108, 2265–2299 CrossRef CAS PubMed ; for reviews on photocatalysis, see: ; (c) C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Chem. Rev., 2013, 113, 5322–5363 CrossRef CAS PubMed; (d) D. Staveness, I. Bosque and C. R. J. Stephenson, Acc. Chem. Res., 2016, 49, 2295–2306 CrossRef CAS PubMed; (e) J. P. Goddard, C. Ollivier and L. Fensterbank, Acc. Chem. Res., 2016, 49, 1924–1936 CrossRef CAS PubMed; (f) S. Crespi and M. Fagnoni, Chem. Rev., 2020, 120, 9790–9833 CrossRef CAS PubMed.
- A. Hantzsch, Ber. Dtsch. Chem. Ges., 1881, 14, 1637–1638 CrossRef.
- G. Li, L. Wu, G. Lv, H. Liu, Q. Fu, X. Zhang and Z. Tang, Chem. Commun., 2014, 50, 6246–6248 RSC.
- (a) Á. Gutiérrez-Bonet, C. Remeur, J. K. Matsui and G. A. Molander, J. Am. Chem. Soc., 2017, 139, 12251–12258 CrossRef PubMed; (b) X. Liu, R. Liu, J. Dai, X. Cheng and G. Li, Org. Lett., 2018, 20, 6906–6909 CrossRef CAS PubMed; (c) P. Chen, J. H. Fan, W. Q. Yu, B. Q. Xiong, Y. Liu, K. W. Tang and J. Xie, J. Org. Chem., 2022, 87, 5643–5659 CrossRef CAS PubMed; (d) W. Q. Yu, J. H. Fan, P. Chen, B. Q. Xiong, J. Xie, K. W. Tang and Y. Liu, Org. Biomol. Chem., 2022, 20, 1958–1968 RSC; (e) F. T. Xiong, B. H. He, Y. Liu, Q. Zhou and J. H. Fan, J. Org. Chem., 2022, 87, 8599–8610 CrossRef CAS PubMed; (f) J. Wang, Y. B. Pang, N. Tao, R. S. Zeng and Y. Zhao, J. Org. Chem., 2019, 84, 15315–15322 CrossRef CAS PubMed; (g) Y. Liao, Y. Ran, G. Liu, P. Liu and X. Liu, Org. Chem. Front., 2020, 7, 3638–3647 RSC; (h) L. Liu, P. Jiang, Y. Liu, H. Du and J. Tan, Org. Chem. Front., 2020, 7, 2278–2283 RSC; (i) D. Zhang, Z. L. Tang, X. H. Ouyang, R. J. Song and J. H. Li, Chem. Commun., 2020, 56, 14055–14058 RSC; (j) Y. Byun, J. Moon, W. An, N. K. Mishra, H. S. Kim, P. Ghosh and I. S. Kim, J. Org. Chem., 2021, 86, 12247–12256 CrossRef CAS PubMed; (k) Z. Bai, H. Zhang, H. Wang, H. Yu, G. Chen and G. He, J. Am. Chem. Soc., 2021, 143, 1195–1202 CrossRef CAS PubMed; (l) Z. Yao, X. Zhang, Z. Luo, Y. Pan, H. Zhao, B. Li, L. Xu, Q. Shi and Q. H. Fan, Asian J. Org. Chem., 2022, 11, e202100609 CrossRef CAS; (m) H. M. Jiang, J. H. Qin, Q. Sun, D. Zhang, J. P. Jiang, X. H. Ouyang, R. J. Song and J. H. Li, Org. Chem. Front., 2022, 9, 4070–4077 RSC; (n) S.-W. Tian, L.-J. Zhong, J.-H. Fan, K.-W. Tang and Y. Liu, Chem. Commun., 2024, 60, 11568–11571 RSC; (o) L. Zhong, X. Shang, H. Chen, R. Liu, K. Tang, Q. Zhou and Y. Liu, Eur. J. Org Chem., 2025, 28, 2–7 Search PubMed.
- (a) K. Nakajima, S. Nojima, K. Sakata and Y. Nishibayashi, ChemCatChem, 2016, 8, 1028–1032 CrossRef CAS; (b) W. Chen, Z. Liu, J. Tian, J. Li, J. Ma, X. Cheng and G. Li, J. Am. Chem. Soc., 2016, 138, 12312–12315 CrossRef CAS PubMed; (c) H. H. Zhang and S. Yu, J. Org. Chem., 2017, 82, 9995–10006 CrossRef CAS PubMed; (d) F. Gu, W. Huang, X. Liu, W. Chen and X. Cheng, Adv. Synth. Catal., 2018, 360, 925–931 CrossRef CAS; (e) B. R. McDonald and K. A. Scheidt, Org. Lett., 2018, 20, 6877–6881 CrossRef CAS PubMed; (f) J. A. Milligan, J. P. Phelan, V. C. Polites, C. B. Kelly and G. A. Molander, Org. Lett., 2018, 20, 6840–6844 CrossRef CAS PubMed; (g) Q. Y. Wu, Q. Q. Min, G. Z. Ao and F. Liu, Org. Biomol. Chem., 2018, 16, 6391–6394 RSC; (h) S. Liang, R. A. Angnes, C. S. Potnis and G. B. Hammond, Tetrahedron Lett., 2019, 60, 151230 CrossRef; (i) X. Wang, M. Yang, W. Xie, X. Fan and J. Wu, Chem. Commun., 2019, 55, 6010–6013 RSC; (j) S. Liang, T. Kumon, R. A. Angnes, M. Sanchez, B. Xu and G. B. Hammond, Org. Lett., 2019, 21, 3848–3854 CrossRef CAS PubMed; (k) Z. J. Wang, S. Zheng, J. K. Matsui, Z. Lu and G. A. Molander, Chem. Sci., 2019, 10, 4389–4393 RSC; (l) W. Luo, Y. Yang, Y. Fang, X. Zhang, X. Jin, G. Zhao, L. Zhang, Y. Li, W. Zhou, T. Xia and B. Chen, Adv. Synth. Catal., 2019, 361, 4215–4221 CrossRef CAS; (m) Z. Y. Song, C. L. Zhang and S. Ye, Org. Biomol. Chem., 2019, 17, 181–185 RSC; (n) X. Wang, H. Li, G. Qiu and J. Wu, Chem. Commun., 2019, 55, 2062–2065 RSC; (o) H. Chen, D. Anand and L. Zhou, Asian J. Org. Chem., 2019, 8, 661–664 CrossRef CAS; (p) X. Gong, M. Yang, J. B. Liu, F. S. He, X. Fan and J. Wu, Green Chem., 2020, 22, 1906–1910 RSC; (q) J. Dong, F. Yue, W. Xu, H. Song, Y. Liu and Q. Wang, Green Chem., 2020, 22, 5599–5604 RSC; (r) H. W. Du, J. Sun, Q. S. Gao, J. Y. Wang, H. Wang, Z. Xu and M. D. Zhou, Org. Lett., 2020, 22, 1542–1546 CrossRef CAS PubMed; (s) Y. Chen, H. Zhao, D. Cheng, X. Li and X. Xu, Chin. J. Org. Chem., 2020, 40, 1297–1304 CrossRef CAS; (t) J. Li, X. E. Yang, S. Le Wang, L. L. Zhang, X. Z. Zhou, S. Y. Wang and S. J. Ji, Org. Lett., 2020, 22, 4908–4913 CrossRef CAS PubMed; (u) R. A. Angnes, C. Potnis, S. Liang, C. R. D. Correia and G. B. Hammond, J. Org. Chem., 2020, 85, 4153–4164 CrossRef CAS PubMed; (v) Q. Guo, Q. Peng, H. Chai, Y. Huo, S. Wang and Z. Xu, Nat. Commun., 2020, 11, 1463 CrossRef CAS PubMed; (w) F. Yue, J. Dong, Y. Liu and Q. Wang, Org. Biomol. Chem., 2021, 19, 8924–8928 RSC; (x) Z. Liang, K. Lv, S. Zhou, C. Zhu and X. Bao, Org. Chem. Front., 2021, 8, 6499–6507 RSC; (y) X. Chen, X. Luo, K. Wang, F. Liang and P. Wang, Synlett, 2021, 32, 733–737 CrossRef CAS; (z) M.-L. Yang and X.-F. Wu, Green Chem., 2025, 27, 9888–9894 RSC.
- (a) K. Nakajima, S. Nojima and Y. Nishibayashi, Angew. Chem., Int. Ed., 2016, 55, 14106–14110 CrossRef CAS PubMed; (b) Á. Gutierrez-Bonet, J. C. Tellis, J. K. Matsui, B. A. Vara and G. A. Molander, ACS Catal., 2016, 6, 8004–8008 CrossRef PubMed; (c) Y. Luo, Á. Gutiérrez-Bonet, J. K. Matsui, M. E. Rotella, R. Dykstra, O. Gutierrez and G. A. Molander, ACS Catal., 2019, 9, 8835–8842 CrossRef CAS PubMed; (d) Y. Zhang, Y. Tanabe, S. Kuriyama and Y. Nishibayashi, J. Org. Chem., 2021, 86, 12577–12590 CrossRef CAS PubMed; (e) Z. Z. Zhou, X. R. Song, S. Du, K. J. Xia, W. F. Tian, Q. Xiao and Y. M. Liang, Chem. Commun., 2021, 57, 9390–9393 RSC; (f) L. Buzzetti, A. Prieto, S. R. Roy and P. Melchiorre, Angew. Chem., Int. Ed., 2017, 56, 15039–15043 CrossRef CAS PubMed; (g) Y. Zhang, Y. Tanabe, S. Kuriyama and Y. Nishibayashi, Chem.–Eur. J., 2022, 28, e202200727 CrossRef CAS PubMed; (h) A. Dumoulin, J. K. Matsui, Á. Gutiérrez-Bonet and G. A. Molander, Angew. Chem., Int. Ed., 2018, 57, 6614–6618 CrossRef CAS PubMed; (i) H. H. Zhang, J. J. Zhao and S. Yu, J. Am. Chem. Soc., 2018, 140, 16914–16919 CrossRef CAS PubMed; (j) K. Nakajima, X. Guo and Y. Nishibayashi, Chem.–Asian J., 2018, 13, 3653–3657 CrossRef CAS PubMed; (k) J. K. Matsui, Á. Gutiérrez-Bonet, M. Rotella, R. Alam, O. Gutierrez and G. A. Molander, Angew. Chem., Int. Ed., 2018, 57, 15847–15851 CrossRef CAS PubMed; (l) S. O. Badir, A. Dumoulin, J. K. Matsui and G. A. Molander, Angew. Chem., Int. Ed., 2018, 57, 6610–6613 CrossRef CAS PubMed; (m) Z. J. Wang, S. Zheng, E. Romero, J. K. Matsui and G. A. Molander, Org. Lett., 2019, 21, 6543–6547 CrossRef CAS PubMed; (n) K. Zhang, L. Lu, Y. Jia, Y. Wang, F. Lu, F. Pan and W. Xiao, Angew. Chem., Int. Ed., 2019, 58, 13375–13379 CrossRef CAS PubMed; (o) Z. Z. Zhou, R. Q. Jiao, K. Yang, X. M. Chen and Y. M. Liang, Chem. Commun., 2020, 56, 12957–12960 RSC; (p) J. L. Schwarz, H. M. Huang, T. O. Paulisch and F. Glorius, ACS Catal., 2020, 10, 1621–1627 CrossRef CAS; (q) Y. Liu, W. Luo, T. Xia, Y. Fang, C. Du, X. Jin, Y. Li, L. Zhang, W. Lei and H. Wu, Org. Chem. Front., 2021, 8, 1732–1738 RSC; (r) H. M. Huang, P. Bellotti, C. G. Daniliuc and F. Glorius, Angew. Chem., Int. Ed., 2021, 60, 2464–2471 CrossRef CAS PubMed; (s) X. Bai, L. Qian, H. H. Zhang and S. Yu, Org. Lett., 2021, 23, 8322–8326 CrossRef CAS PubMed; (t) S. Xue, B. Limburg, D. Ghorai, J. Benet-Buchholz and A. W. Kleij, Org. Lett., 2021, 23, 4447–4451 CrossRef CAS PubMed; (u) F.-H. Zhang, X. Guo, X. Zeng and Z. Wang, Nat. Commun., 2022, 13, 5036 CrossRef CAS PubMed; (v) S. Xue, À. Cristòfol, B. Limburg, Q. Zeng and A. W. Kleij, ACS Catal., 2022, 12, 3651–3659 CrossRef CAS; (w) F. Cong, R. S. Mega, J. Chen, C. S. Day and R. Martin, Angew. Chem., Int. Ed., 2023, 62, e202214633 CrossRef CAS PubMed; (x) M.-L. Yang and X.-F. Wu, Green Chem., 2025, 27, 9888–9894 RSC; (y) T. Li, L. Luo, X. Cheng and Z. Lu, ACS Catal., 2024, 14, 3278–3286 CrossRef CAS; (z) Y. Liu, Y.-Y. Cheng, J.-X. Yu, C. Wang, H.-L. Hu, G. Liang, F.-X. Li, H.-Y. Hou, X.-N. Guo, C.-H. Tung and L.-Z. Wu, Org. Lett., 2025, 27, 6777–6782 CrossRef CAS PubMed.
- (a) G. Goti, B. Bieszczad, A. Vega-Peñaloza and P. Melchiorre, Angew. Chem., Int. Ed., 2019, 58, 1213–1217 CrossRef CAS PubMed; (b) F. Li, D. Tian, Y. Fan, R. Lee, G. Lu, Y. Yin, B. Qiao, X. Zhao, Z. Xiao and Z. Jiang, Nat. Commun., 2019, 10, 1774 CrossRef PubMed; (c) A. V. Bay, K. P. Fitzpatrick, R. C. Betori and K. A. Scheidt, Angew. Chem., Int. Ed., 2020, 59, 9143–9148 CrossRef CAS PubMed; (d) A. V. Bay, K. P. Fitzpatrick, G. A. González-Montiel, A. O. Farah, P. H. Y. Cheong and K. A. Scheidt, Angew. Chem., Int. Ed., 2021, 60, 17925–17931 CrossRef CAS PubMed; (e) E. Le Saux, D. Ma, P. Bonilla, C. M. Holden, D. Lustosa and P. Melchiorre, Angew. Chem., Int. Ed., 2021, 60, 5357–5362 CrossRef CAS PubMed; (f) A. V. Bay, E. J. Farnam and K. A. Scheidt, J. Am. Chem. Soc., 2022, 144, 7030–7037 CrossRef CAS PubMed; (g) T. Li, L. Luo, X. Cheng and Z. Lu, ACS Catal., 2024, 14, 3278–3286 CrossRef CAS; (h) Y. Liu, Y.-Y. Cheng, J.-X. Yu, C. Wang, H.-L. Hu, G. Liang, F.-X. Li, H.-Y. Hou, X.-N. Guo, C.-H. Tung and L.-Z. Wu, Org. Lett., 2025, 27, 6777–6782 CrossRef CAS PubMed; (i) X. Sun, W. Zhu, Y. Yin, X. Zhao and Z. Jiang, J. Am. Chem. Soc., 2025, 147, 4382–4392 CrossRef CAS PubMed; (j) M.-L. Yang and X.-F. Wu, Green Chem., 2025, 27, 5257–5264 RSC.
- (a) F. F. De Assis, X. Huang, M. Akiyama, R. A. Pilli and E. Meggers, J. Org. Chem., 2018, 83, 10922–10932 CrossRef CAS PubMed; (b) C. Verrier, N. Alandini, C. Pezzetta, M. Moliterno, L. Buzzetti, H. B. Hepburn, A. Vega-Peñaloza, M. Silvi and P. Melchiorre, ACS Catal., 2018, 8, 1062–1066 CrossRef CAS; (c) S. Zhang, Y. Li, J. Wang, X. Hao, K. Jin, R. Zhang and C. Duan, Tetrahedron Lett., 2020, 61, 151721 CrossRef CAS; (d) A. Lipp, S. O. Badir, R. Dykstra, O. Gutierrez and G. A. Molander, Adv. Synth. Catal., 2021, 363, 3507–3520 CrossRef CAS PubMed; (e) L. Liu, Z. Deng, K. Xu, P. Jiang, H. Du and J. Tan, Org. Lett., 2021, 23, 5299–5304 CrossRef CAS PubMed; (f) Y. N. Niu, X. H. Jin, L. L. Liao, H. Huang, B. Yu, Y. M. Yu and D. G. Yu, Sci. China: Chem., 2021, 64, 1164–1169 CrossRef CAS; (g) S. S. Patel, D. Kumar and C. B. Tripathi, Chem. Commun., 2021, 57, 5151–5154 RSC; (h) S. C. Ren, W. X. Lv, X. Yang, J. L. Yan, J. Xu, F. X. Wang, L. Hao, H. Chai, Z. Jin and Y. R. Chi, ACS Catal., 2021, 11, 2925–2934 CrossRef CAS; (i) L. Wang, X. Wang, W. Wang, W. Liu, Y. Liu, H. Xie, O. Reiser, J. Zeng and P. Cheng, J. Nat. Prod., 2021, 84, 2390–2397 CrossRef CAS PubMed; (j) W. Zhou, S. Wu and P. Melchiorre, J. Am. Chem. Soc., 2022, 144, 8914–8919 CrossRef CAS PubMed; (k) F. L. Zeng, K. C. Xie, Y. T. Liu, H. Wang, P. C. Yin, L. B. Qu, X. L. Chen and B. Yu, Green Chem., 2022, 24, 1732–1737 RSC; (l) Z. Yao, J. Yang, Z. Luo, H. Wang, X. Zhang, J. Ye, L. Xu and Q. Shi, Green Chem., 2022, 24, 7968–7973 RSC; (m) S. Xie, D. Li, H. Huang, F. Zhang and Y. Chen, J. Am. Chem. Soc., 2019, 141, 16237–16242 CrossRef CAS PubMed; (n) P. C. Gallage and S. P. Pitre, Green Chem., 2022, 24, 6845–6848 RSC; (o) Á. M. Pálvölgyi, F. Ehrschwendtner, M. Schnürch and K. Bica-Schröder, Org. Biomol. Chem., 2022, 20, 7245–7249 RSC; (p) X. Shan, X. Wang, E. Chen, J. Liu, K. Lu and X. Zhao, J. Org. Chem., 2023, 88, 319–328 CrossRef CAS PubMed; (q) X. Chen, F. Ye, X. Luo, X. Liu, J. Zhao, S. Wang, Q. Zhou, G. Chen and P. Wang, J. Am. Chem. Soc., 2019, 141, 18230–18237 CrossRef CAS PubMed; (r) E. Gandolfo, X. Tang, S. Raha Roy and P. Melchiorre, Angew. Chem., Int. Ed., 2019, 58, 16854–16858 CrossRef CAS PubMed; (s) T. van Leeuwen, L. Buzzetti, L. A. Perego and P. Melchiorre, Angew. Chem., Int. Ed., 2019, 58, 4953–4957 CrossRef CAS PubMed; (t) B. Bieszczad, L. A. Perego and P. Melchiorre, Angew. Chem., Int. Ed., 2019, 58, 16878–16883 CrossRef CAS PubMed; (u) X. Zhao, B. Li and W. Xia, Org. Lett., 2020, 22, 1056–1061 CrossRef CAS PubMed; (v) X. K. He, J. Lu, A. J. Zhang, Q. Q. Zhang, G. Y. Xu and J. Xuan, Org. Lett., 2020, 22, 5984–5989 CrossRef CAS PubMed; (w) I. Kim, S. Park and S. Hong, Org. Lett., 2020, 22, 8730–8734 CrossRef CAS PubMed; (x) S. Patel, A. Chakraborty and I. Chatterjee, Org. Lett., 2023, 25, 8246–8251 CrossRef CAS PubMed; (y) P. C. Gallage, M. G. McKee and S. P. Pitre, Org. Lett., 2024, 26, 1975–1979 CrossRef CAS PubMed.
- (a) P. P. Mondal, A. Pal, A. K. Prakash and B. Sahoo, Chem. Commun., 2022, 58, 13202–13205 RSC; (b) X.-Y. Lv, R. Abrams and R. Martin, Nat. Commun., 2022, 13, 2394 CrossRef CAS PubMed; (c) P. P. Mondal, A. Pal, S. Das, S. M. Vijayan, A. V. Nair, S. Ojha and B. Sahoo, Synlett, 2023, 34, 1241–1246 CrossRef CAS; (d) F. Zhai and R. F. Jordan, ACS Omega, 2017, 2, 3055–3063 CrossRef CAS PubMed; (e) L. Li, L. Fang, W. Wu and J. Zhu, Org. Lett., 2020, 22, 5401–5406 CrossRef CAS PubMed.
- (a) X. Zhou, D. Pyle, Z. Zhang and G. Dong, Angew. Chem., Int. Ed., 2023, 62, e202213691 ( Angew. Chem. , 2023 , 135 , e202213691 ) CrossRef CAS PubMed; (b) Z. Zhang, Q. Zhu, D. Pyle, X. Zhou and G. Dong, J. Am. Chem. Soc., 2023, 145, 21096–21103 CrossRef CAS PubMed; (c) X. Zhou, Y. Xu and G. Dong, J. Am. Chem. Soc., 2021, 143, 20042–20048 CrossRef CAS PubMed; (d) X. Zhou, T. Yu and G. Dong, J. Am. Chem. Soc., 2022, 144, 9570–9575 CrossRef CAS PubMed.
- (a) T. Uchikura, K. Moriyama, M. Toda, T. Mouri, I. Ibáñez and T. Akiyama, Chem. Commun., 2019, 55, 11171–11174 RSC; (b) T. Uchikura, M. Toda, T. Mouri, T. Fujii, K. Moriyama, I. Ibáñez and T. Akiyama, J. Org. Chem., 2020, 85, 12715–12723 CrossRef CAS PubMed.
- S. Miñoza, I. L. Librando and H.-H. Liao, Synlett, 2024, 35, 1072–1088 CrossRef.
- (a) A. Luguera Ruiz, M. La Mantia, D. Merli, S. Protti and M. Fagnoni, ACS Catal., 2022, 12, 12469–12476 CrossRef CAS PubMed; (b) A. Luguera Ruiz, E. Mariani, S. Protti and M. Fagnoni, Org. Chem. Front., 2024, 11, 661–667 RSC.
- (a) S. C. Chen, Q. Zhu, Y. Cao, C. Li, Y. Guo, L. Kong, J. Che, Z. Guo, H. Chen, N. Zhang, X. Fang, J. T. Lu and T. Luo, J. Am. Chem. Soc., 2021, 143, 14046–14052 CrossRef CAS PubMed; (b) S. Chen, Q. Zhu, H. Chen, Z. Chen and T. Luo, Chem.–Eur. J., 2023, 29, e202203425 CrossRef CAS PubMed.
- S. Ratnam, S. Unone and D. Janssen-Müller, Chem.–Eur. J., 2023, 29, e202301685 CrossRef CAS PubMed.
- (a) Z. Dong and D. W. C. MacMillan, Nature, 2021, 598, 451–456 CrossRef CAS PubMed; (b) C. A. Gould, A. L. Pace and D. W. C. MacMillan, J. Am. Chem. Soc., 2023, 145, 16330–16336 CrossRef CAS PubMed.
- (a) P. Biginelli, Ber. Dtsch. Chem. Ges., 1891, 24, 2962–2967 CrossRef; (b) C. O. Kappe, Tetrahedron, 1993, 49, 6937–6963 CrossRef CAS; (c) C. O. Kappe and A. Stadler, in Org. React., Wiley, 2004, pp. 1–116 Search PubMed; (d) S. S. Panda, P. Khanna and L. Khanna, Curr. Org. Chem., 2012, 16, 507–520 CrossRef CAS.
- C. P. Sar, O. H. Oankovszky, G. Jerkovich, I. Pallagi and K. Hideg, ACH – Models Chem., 1994, 131, 363–376 CAS.
- (a) C. O. Kappe, J. Jacques Vanden Eynde, N. Audiart, V. Canonne, S. Michel and Y. Van Haverbeke, Heterocycles, 1997, 45, 1967 CrossRef; (b) B. L. Nilsson and L. E. Overman, J. Org. Chem., 2006, 71, 7706–7714 CrossRef CAS PubMed; (c) J. Klenc, E. Raux, S. Barnes, S. Sullivan, B. Duszynska, A. J. Bojarski and L. Strekowski, J. Heterocycl. Chem., 2009, 46, 1259–1265 CrossRef; (d) L. Wang, Z. G. Ma, X. J. Wei, Q. Y. Meng, D. T. Yang, S. F. Du, Z. F. Chen, L. Z. Wu and Q. Liu, Green Chem., 2014, 16, 3752–3757 RSC; (e) Y. Nishimura, T. Kubo, N. Shibuya and H. Cho, Synthesis, 2023, 55, 1996–2004 CrossRef CAS.
- D. Rehm and A. Weller, Isr. J. Chem., 1970, 8, 259–271 CrossRef CAS.
- (a) CCDC 2338773: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2jhp91; (b) CCDC 2338774: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2jhpb2; (c) CCDC 2338775: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2jhpc3.
| This journal is © The Royal Society of Chemistry 2026 |