Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Cooperative reactivity of halomethanes and -silanes at an A-frame complex: transannular addition versus bridging tetrylenes
Max Passargusab,
Celine Nieuwland c,
Merle Arrowsmith
c,
Merle Arrowsmith ab,
F. Matthias Bickelhaupt
ab,
F. Matthias Bickelhaupt *cde and
Holger Braunschweig
*cde and
Holger Braunschweig *ab
*ab
aInstitute for Inorganic Chemistry, Julius-Maximilians-Universität Würzburg, Am Hubland, 97074 Würzburg, Germany
bInstitute for Sustainable Chemistry & Catalysis with Boron, Julius-Maximilians-Universität Würzburg, Am Hubland, 97074 Würzburg, Germany
cDepartment of Chemistry and Pharmaceutical Sciences, Amsterdam Institute of Molecular and Life Sciences (AIMMS), Vrije Universiteit Amsterdam, De Boelelaan 1108, 1081 HZ Amsterdam, The Netherlands. E-mail: F.M.Bickelhaupt@vu.nl
dInstitute for Molecules and Materials, Radboud University, Heyendaalseweg 135, 6525 AJ Nijmegen, The Netherlands
eDepartment of Chemical Sciences, University of Johannesburg, Auckland Park, Johannesburg 2006, South Africa
First published on 15th April 2026
Abstract
With CH2X2 (X = Cl, Br, I) and CCl4 the diplatinum(0) complex [(µ-dmpm)2Pt2(nbe)2] (dmpm = bis(dimethylphosphino)methane, nbe = norbornene) undergoes facile cooperative addition of one C–X bond at each Pt centre to yield the methylene-bridged diplatinum(II) A-frame complexes [(µ-dmpm)2Pt2X2(µ-CY2)] (Y![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) H, Cl). In contrast, reactions with Me4−nSiXn (X = Cl, n = 1–3; X = I, n = 1) lead preferentially to transannular oxidative additions of a single Si–X bond over the two metal centres, yielding the complexes [(µ-dmpm)2{PtX}{Pt(SiMe4−nXn−1)}]. In CH2Cl2 [(µ-dmpm)2{PtCl}{Pt(SiCl3)}] undergoes rearrangement to the silylene-bridged [(µ-dmpm)2Pt2Cl2(µ-SiCl2)], while in CH2Br2 oxidation of the platinum centres to Pt(II), Cl–Br exchange, and the insertion of a CH2Br2-derived methylene unit into the Pt–Si bond are observed. Quantum-chemical calculations provide insights into the differences in reactivity between the halomethanes and -silanes.
H, Cl). In contrast, reactions with Me4−nSiXn (X = Cl, n = 1–3; X = I, n = 1) lead preferentially to transannular oxidative additions of a single Si–X bond over the two metal centres, yielding the complexes [(µ-dmpm)2{PtX}{Pt(SiMe4−nXn−1)}]. In CH2Cl2 [(µ-dmpm)2{PtCl}{Pt(SiCl3)}] undergoes rearrangement to the silylene-bridged [(µ-dmpm)2Pt2Cl2(µ-SiCl2)], while in CH2Br2 oxidation of the platinum centres to Pt(II), Cl–Br exchange, and the insertion of a CH2Br2-derived methylene unit into the Pt–Si bond are observed. Quantum-chemical calculations provide insights into the differences in reactivity between the halomethanes and -silanes.
Introduction
The oxidative addition of polar carbon–halogen bonds to electron-rich metal complexes is a key step in many examples of transition metal (TM) catalysis.1 While palladium has been the metal of choice for the vast majority of catalytic cross-coupling processes involving the oxidative addition of organohalides,2 more expensive platinum(0) catalysts have found applications in some specialised cross-coupling reactions, including the coupling of polyfluoroarylimines with ZnMe2,3 thiolates with (hetero)aryl iodides,4 hydrazones with allyl halides,5 or hydrosilanes with iodoalkanes.6Whereas the oxidative addition of carbon–halide bonds and their heavier group 14 congeners at mononuclear Pt(0) complexes has been studied in detail,7,8 studies of E–X (E = group 14 element, X = halide) bond activations by dinuclear platinum complexes, which may offer unique cooperative reactivity and catalytic enhancement,9 remain rare. Our group has studied the oxidative addition of (di)boron halides to the diplatinum(0) complex [(µ-dmpm)2Pt2(nbe)2] (1, dmpm = bis(dimethylphosphino)methane, nbe = norbornene),10–12 which offers a flexible platform for the cooperative oxidative addition of B–X bonds at both Pt centres, generating so-called A-frame complexes13 with a bridging (di)boranediyl apex ligand, like IX (Fig. 1A). A number of cooperative activations of organodihalides by similar dppm-bridged (dppm = bis(diphenylphosphino)methane) dinickel(0) and dipalladium(0) systems have been reported, affording a range of organodiyl-bridged A-frame complexes, like the methylene- or vinylidene-bridged complexes IIX and IIIX.14 To our knowledge, however, there have been no reports of cooperative activation of organodihalides, let alone heavier group 14 dihalides, by dinuclear group 10 systems.
 | ||
| Fig. 1 Cooperative addition of (A) trihaloboranes at the diplatinum(0) complex 1, (B) organodihalides at a dipalladium(0) complex, and (C) di- and tetrahalomethanes and heavier group 14 halides at 1. | ||
In this contribution, we present our investigation into the oxidative addition of di- and tetrahalomethanes at 1 and compare it to that of their heavier group 14 counterparts, underpinning the experimental results by DFT calculations on the origin of the differences in reactivity.
Results and discussion
Synthesis of methylene-bridged diplatinum A-frame complexes
Equimolar reactions of 1 with CH2X2 (X = Cl, Br) in benzene at room temperature (rt) for 5 min yielded the methylene-bridged platinum(II) A-frame complexes [(µ-dmpm)2Pt2X2(µ-CH2)] (2CH2-X) as yellow solids in high isolated yield (75% each, Scheme 1a). To avoid the use of light-sensitive and potentially mutagenic CH2I2, the brown complex 2CH2-I was synthesised quantitatively by treating 2CH2-Br with an excess of trimethylsilyl iodide (Scheme 1b). It is noteworthy that all three complexes 2CH2-X decompose both in solution and when subjected to vacuum to the diplatinum(I) complexes [(µ-dmpm)2Pt2X2], as has also been observed for boranediyl-bridged diplatinum A-frames under comparable conditions.10–12 While we are yet to determine the fate of the lost methylene fragment in these decomposition reactions, its spontaneous release from the A-frame complexes 2CH2-X could potentially be harnessed for metal-mediated C–C bond forming reactions in the presence of suitable substrates, such as azoalkanes, alkenes, alkynes and nucleophilic heterocycles.15 Coupled with a reductive step to reduce the by-product [(µ-dmpm)2Pt2X2] back to a diplatinum(0) complex, such processes could even be made catalytic.While the addition of CCl4 to 1 also led to the instant formation of the corresponding, orange-coloured dichloromethylene-bridged A-frame complex 2CCl2-Cl in near-quantitative yield, the latter was only partially characterised as it decomposed rapidly in solution to a range of unidentified complexes.
The 1H NMR spectra of 2CH2-X show a quintet (2JH–P ≈ 9 Hz) with 195Pt satellites (1JH–Pt = 55–58 Hz) for the protons of the bridging methylene, which is shifted upfield with increasing halide electronegativity (δ = 0.83 (2CH2-Cl), 1.07 (2CH2-Br), 1.47 (2CH2-I) ppm), as in the series of analogous dipalladium A-frames IIX.14 A similar multiplet (δ1H = 1.08 ppm, 2JH–P ≈ 9 Hz, 1JH–Pt ≈ 54 Hz) was observed for the analogous [(µ-dppm)2Pt2Cl2(µ-CH2)], synthesised in 1985 from the reaction of highly explosive diazomethane with the cationic diplatinum complex [(µ-dppm)2{Pt(PPh3)}{PtH}]+, and subsequent recrystallisation from CH2Cl2.16 The chemical shift of the corresponding high-field 13C{1H} NMR multiplet shows a similar trend (δ = −14.5 (2CH2-Cl), −7.6 (2CH2-Br), 4.5 (2CH2-I) ppm), the 1JC–Pt coupling remaining relatively unchanged (1JC–Pt = 613–617 Hz) over the entire series. As expected, the methylene 13C NMR resonance of 2CCl2-Cl is shifted to much lower field at 20.3 ppm. Conversely, the 31P{1H} NMR singlet of 2CH2-X, with its typical higher-order A-frame satellite splitting pattern (1JP–Pt ≈ 2980–3070 Hz, 3JP–Pt ≈ 70 Hz), is shifted upfield upon descending the halide group (from δ = −7.3 (2CH2-Cl) to −13.2 (2CH2-I) ppm), as expected with decreasing halide electronegativity (Table 1). The 31P{1H} NMR resonance of 2CCl2-Cl appears at −8.7 ppm, between those of 2CH2-Cl and 2CH2-Br, and displays a much lower 1JP–Pt coupling of 2750 Hz but significantly higher 3JP–Pt coupling of 150 Hz. An upfield shift is also observed for the 195Pt{1H} NMR triplet of 2CH2-X upon going down the halide group (from δ = −4267 ppm (2CH2-Cl) to −4645 (2CH2-I) ppm), concomitant with a decrease in their 1JPt–P coupling constants (from 1JPt–P = 3081 (2CH2-Cl) to 2981 (2CH2-I) Hz). The 195Pt{1H} NMR triplet of 2CCl2-Cl was detected by 1H–195Pt HMQC at −4270 ppm, close to that of 2CH2-Cl. The analysis of the 195Pt satellite pattern of the 31P{1H} NMR multiplets of 2CH2-X (see Fig. S2 in the SI)17 enables the determination of the 2JPt–Pt coupling constants, which decrease from 842 Hz in 2CH2-Cl to 754 Hz in 2CH2-I, and are similar to that of ICl (2JPt–Pt = 879 Hz).11 We have shown that the value of the 2JPt–Pt constant in the boranediyl-bridged complexes [(µ-dmpm)2Pt2X2(µ-BY)] (X = halide; Y = halide, amino, organo) decreases as the Pt⋯Pt distance increases, which is also the case in the methylene-bridged A-frame series 2CH2-X (vide infra).
| 31PP1 | 31PP2 | 1JP1–Pt1 | 1JP2–Pt2 | nJP1–Pt2 | nJP2–Pt1 | 195PtPt1 | 195PtPt2 | nJPt–Pt | |
|---|---|---|---|---|---|---|---|---|---|
| a n = 3.b n = 2.c n = 1.d n.d. = not determined due to lack of spectral resolution.e P1/P3.f P2/P4. | |||||||||
| 2CH2-Cl | −7.3 | — | 3072 | — | 73a | — | −4267 | — | 842b |
| 2CH2-Br | −9.6 | — | 3036 | — | 71a | — | −4397 | — | 831b |
| 2CH2-I | −13.2 | — | 2982 | — | 68a | — | −4645 | — | 754b |
| 2Cl2-Cl | −8.7 | — | 2750 | — | 150 | — | −4270 | — | n.d.d |
| 2SiMeCl-Cl | −0.1e | −2.8f | 3261 | 3114 | 261a | 203a | −4519 | — | n.d. |
| 2SiCl2-Cl | 1.2 | — | 3098 | — | 242a | — | −4446 | — | 297b |
| 3SiMe3-Cl | −15.6 | −25.7 | 3156 | 2671 | 122b | 48b | −4317 | −4798 | 3060c |
| 3SiMe3-I | −21.4 | −26.5 | 3132 | 2656 | 116b | 57b | −4686 | −4755 | n.d. |
| 3SiMe2Cl-Cl | −17.5 | −26.4 | 3035 | 2581 | 138b | 52b | −4309 | −4748 | 3269c |
| 3SiMeCl2-Cl | −17.7 | −26.0 | 2930 | 2475 | 154b | 57b | −4329 | −4752 | 3738c |
| 3SiCl3-Cl | −19.4 | −26.7 | 2815 | 2434 | 154b | 62b | −4305 | −4791 | 4163c |
| 5SiCl3-Br | −20.7 | −15.0 | 2420 | 2702 | n.d. | n.d. | −4408 | −4421 | — |
| 5SiCl3-Cl | −16.7 | −9.6 | 2420 | 2720 | n.d. | n.d. | −4367 | −4411 | — |
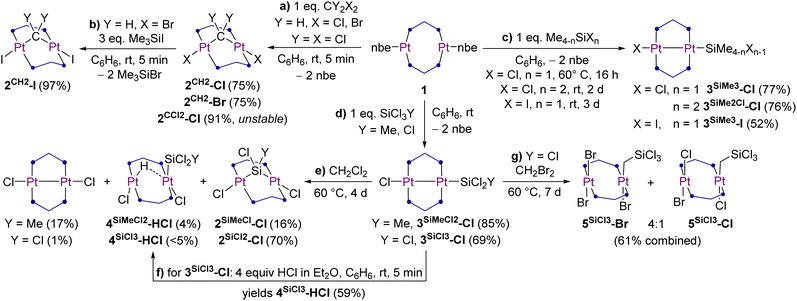
The formation of 2CH2-Cl and 2CH2-Br was accompanied by the precipitation of a yellow solid, which in the case of the reaction with CH2Br2 was identified by single-crystal X-ray diffraction (SCXRD) analysis as the bis(methylene)-bridged diplatinum(IV) complex [(µ-dmpm)2Pt2Br4(µ-CH2)2] (2CH2-Br′) (Fig. 2), in which one CH2Br2 molecule has added on each side of the (µ-dmpm)2Pt2 core. Unfortunately, the reaction of 1 with a large excess of CH2Br2 did not yield 2CH2-Br′ more selectively. Although further characterisation of these side-products was marred by their insolubility in all common organic solvents, it is noteworthy that 2CH2-Br′ is a unique example of a dinuclear group 10 complex bridged by two methylene units.
Crystals of 2CH2-Cl, 2CH2-Br and 2CH2-I were also analysed by SCXRD (Fig. 2 and Table 2, see Fig. S67 and S70 in the SI for 2CH2-Cl and 2CH2-I). The degree of A-frame distortion in these complexes is quantified by their |P–M–M–P|cis and |P–M–M–P|trans torsion angles (M = metal centre), which amount to 0° and 180°, respectively, in the ideal A-frame structure. Complexes 2CH2-X all show a relatively small degree of A-frame distortion (|P–Pt–Pt–P|cis ≤ 19°; |P–Pt–Pt–P|trans ≥ 163°), while the centrosymmetric structure of 2CH2-Br′ provides a near-ideal A-frame (|P–Pt–Pt–P|cis 1.19(5)°; |P–Pt–Pt–P|trans 180°). The distortion in 2CH2-X is higher than in their haloboranediyl-bridged counterparts IX (|P–Pt–Pt–P|cis ≤ 7°; |P–Pt–Pt–P|trans ≥ 173°), as the more acute Pt–C–Pt angle (2CH2-X avg. 100°; IX Pt–B–Pt 111–126°) enforces a shorter Pt⋯Pt distance (2CH2-X avg. 3.15 Å; IX 3.25–3.47 Å).10 Moreover, the 2JPt–Pt coupling constant decreases (from 842 (2CH2-Cl) to 754 (2CH2-I) Hz) in line with the increasing Pt⋯Pt distance (from 3.1475(5) (2CH2-Cl) to 3.1745(5) (2CH2-I) Å). The Pt–C bond lengths in 2CH2-X are all similar (avg. 2.06 Å) and consistent with those previously reported for a similar methylene-bridged complex [(µ-dppm)2Pt2Cl2(µ-CH2)] (2.008(13) Å).18
| P–Pt | Pt–X | Pt⋯Pt | Pt1–E | Pt2–E | Pt–E–Pt | |P–Pt–Pt–P|cis | |P–Pt–Pt–P|trans | |
|---|---|---|---|---|---|---|---|---|
| a X = Cl.b X = Br.c X = I.d E = C.e E = Si. | ||||||||
| 2CH2-Cl | 2.267(2)–2.294(2) | 2.4445(19), 2.4351(19)a | 3.1475(5) | 2.060(7)d | 2.054(7)d | 99.8(3)d | 12.01(7), 18.61(7) | 163.02(7), 166.36(7) |
| 2CH2-Br | 2.2764(17)–2.2864(17) | 2.5606(7), 2.5606(7)b | 3.1604(4) | 2.055(6)d | 2.051(6)d | 100.7(2)d | 6.80(6), 13.54(6) | 169.69(6), 169.98(6) |
| 2CH2-Br′ | 2.3419(16), 2.3417(16) | 2.6099(12), 2.5811(8)b | 3.1816(10) | 2.079(5)d | 2.077(5)d | 99.9(2)d | 1.19(5) | 180 |
| 2CH2-I | 2.279(3), 2.288(3) | 2.7358(7), 2.7129(6)c | 3.1745(5) | 2.057(9)d | 2.063(8)d | 100.8(3)d | 7.7(2), 9.2(1) | 170.4(2), 172.6(1) |
| 2SiCl2-Cl | 2.2928(16)–2.3054(17) | 2.4486(16), 2.4366(16)a | 3.5682(6) | 2.2610(17)e | 2.2541(17)e | 104.42(7)e | 2.28(6), 7.87(6) | 161.05(6), 171.20(6) |
| 3SiMe3-I | 2.2542(19)–2.2714(19) | 2.7402(6)c | 2.7320(5) | — | 2.357(2)e | — | 52.27(7), 54.80(7) | 125.15(7), 127.78(7) |
| 3SiMe2Cl-Cl | 2.2501(19)–2.2796(17) | 2.4419(18)a | 2.6921(7) | — | 2.3316(18)e | — | 53.68(6), 55.83(6) | 125.23(6), 125.26(6) |
| 3SiMeCl2-Cl | 2.2562(19)–2.2798(18) | 2.4402(17)a | 2.6897(6) | — | 2.2960(18)e | — | 52.67(6), 54.89(6) | 126.19(7), 126.24(6) |
| 3SiCl3-Cl | 2.2576(9)–2.2897(8) | 2.4195(9)a | 2.66556(18) | — | 2.2933(9)e | — | 53.14(3), 55.79(3) | 125.39(3), 125.67(3) |
| 4SiCl3-HCl | 2.278(3)–2.330(3) | 2.420(3), 2.397(3)a | 3.1734(9) | — | 2.253(3)e | — | 14.2(1), 28.4(1) | 157.5(1), 159.9(1) |
| 5SiCl3-Br | 2.2959(16)–2.3073(17) | 2.4403(7)–2.4968(7)b | 3.400(1) | 2.097(6)d | — | — | 12.21(7), 12.61(7) | 163.88(7), 171.30(7) |
Transannular oxidative addition of methylhalosilanes
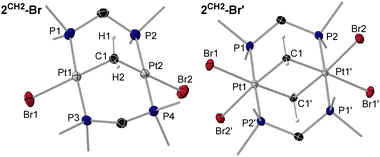
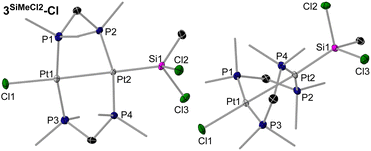
There are no literature precedents for the oxidative addition of heavier group 14 halides at dinuclear platinum complexes. The reaction of 1 with Me4−nSiCln (n = 1–4) provided the transannular oxidative addition products [(µ-dmpm)2{PtX}{Pt(SiMe4−nXn−1)}] (X = Cl, n = 1: 3SiMe3-Cl (brown), n = 2: 3SiMe2Cl-Cl (beige), n = 3: 3SiMeCl2-Cl (yellow), n = 4: 3SiCl3-Cl (yellow); X = I, n = 1: 3SiMe3-I (orange)), in which one Si–X bond has formally added across the central Pt2 unit (Scheme 1c and d).† Except for the reaction with Me3SiCl, which required heating at 60 °C for 16 h, all these reactions proceeded at rt. After workup, the resulting silyldiplatinum complexes were isolated in moderate to excellent yields (69–84%). Unlike the A-frame complexes 2CY2-X the silyldiplatinum complexes 3SiMe(3–n)Xn-X (n = 0–3) were stable in solution over longer periods of time. While mononuclear silyl platinum(II) complexes are key intermediates in the catalytic addition of Si–E (E = H, Si, B) bonds to alkenes and alkynes, as well in the silylation of aryl halides,19 the use of bimetallic complexes or of halosilane precursors in such transformations have not yet been has not been studied.The 29Si{1H} NMR spectra of 3SiMe3-Cl and 3SiMe3-I display a triplet of triplets (2JSi–P ≈ 10 Hz, 3JSi–P ≈ 2–4 Hz) with 195Pt satellites, while 3SiMeCl2-Cl and 3SiCl3-Cl only show coupling to the adjacent phosphines.‡ As expected, the 29Si{1H} NMR resonances of the chloro derivatives are progressively deshielded (from δ = −11 to 45 ppm) as the number of chlorides increases at the silicon centre. The 1JSi–Pt and 2JSi–Pt coupling constants increase with the number of halides at silicon, from 990 and 273 Hz, respectively, in 3SiMe2Cl-Cl to 1629 and 624 Hz, respectively, in 3SiCl3-Cl. The same trends have been observed for the 29Si{1H} NMR chemical shifts and 1JSi–Pt constants in the mononuclear disilylplatinum complexes [(Et3P)2Pt(SiMe3−nCln)2] (n = 0–2). The 31P{1H} NMR spectra of 3SiMe(3–n)Xn-X reflect the asymmetry of the complexes, showing two higher-order multiplets with complex satellite patterns around −26 ppm for the P2PtSi nuclei and −15 to −19 ppm for the P2PtCl nuclei, or −21 ppm for the P2PtI nucleus. The influence of the trans-silyl ligand in the chloro derivatives induces a small but non-negligible upfield shift in the P2PtCl resonance as the degree of chlorination of the silyl ligand increases, as also observed in [(Et3P)2Pt(SiMe3−nCln)Cl] (n = 0–2).20 While the PtSi centres in 3SiMe(3–n)Xn-X all display a similar 195Pt{1H} NMR shift, between −4740 and −4800 ppm, the resonance of the PtI centre in 3SiMe3-I (δ = −4686 ppm) is significantly upfield-shifted compared to that of the PtCl centre in the chloro derivatives (δ ≈ −4300 to −4330 ppm), as already observed in 2CH2-X. In the chloro derivatives the 1JPt–Pt coupling constants increase from 3060 Hz to 4163 Hz, both with the degree of chlorination of the silyl ligand and a shortening of the Pt–Pt bond length (vide infra). These 1JPt–Pt coupling constants are relatively small compared to other unsymmetrical bis(diphosphine)-bridged diplatinum(I) complexes displaying Pt–Pt bonding, such as the stannyl analogue of 3SiCl3-Cl, [(µ-dppm)2{PtCl}{Pt(SnCl3)}] (obtained from the reaction of [(µ-dppm)2Pt2Cl2] with SnCl2, 1JPt–Pt = 8200 Hz),21 [(µ-dppm)2Pt2Cl2] (1JPt–Pt = 8146 Hz),22 or our [(µ-dmpmtab)(µ-dmpm)Pt2Br2] complexes (dmpmtab = tetrazaborolyl-substituted dmpm ligand, 1JPt–Pt = 6670–8510 Hz).23
Colourless crystals of 3SiMe2Cl-Cl, and yellow crystals of 3SiMe3-I, 3SiMeCl2-Cl and 3SiCl3-Cl, provided suitable data for SCXRD analysis (Fig. 3 and 4, left, and Table 2; see Fig. S71 and S72 in the SI for 3SiMe3-I and 3SiMe2Cl-Cl). As exemplified in the right-hand view of complex 3SiMeCl2-Cl in Fig. 3, the (µ-dmpm)2Pt2 frameworks of these complexes are highly distorted (|P–Pt–Pt–P|cis 52–56°, |P–Pt–Pt–P|trans 125–128°). Indeed, a CCDC search shows these are the most distorted [(µ-CH2{PR2}2)2Pt2XY] complexes that have been reported, beside the twisted isomers of our recent [(µ-dmpmtab)(µ-dmpm)Pt2Br2] complexes (|P–Pt–Pt–P|cis 49–52°, |P–Pt–Pt–P|trans 126–132°),23 and [(µ-dmpm)2Pt2Br2] (|P–Pt–Pt–P|cis 49°, |P–Pt–Pt–P|trans 124–136°).10 The Pt–Pt bond lengths range from 2.66556(18) Å in 3SiCl3-Cl to 2.7320(5) Å in 3SiMe3-I, in the typical range for neutral [{µ-CH2(PR2)2}2Pt2XY] complexes (2.62–2.71 Å).10,24 They also decrease with the degree of chlorination of the silyl ligand, as more electron density is pulled out of the Pt–Pt bond. Similarly, the Pt–Si distance also decreases with the degree of silyl chlorination, from 2.2933(9) in 3SiCl3-Cl to 2.357(2) Å in 3SiMe3-I. Similar Pt–Si bond lengths are found in mononuclear, unchelated [trans-(R3P)2PtX(SiR′3)] complexes (2.32–2.39 Å).25
Transannular oxidative additions like these, with concomitant metal–metal bond formation, were first reported by Schmidbaur in 1975 at a cyclometallated dinuclear Au(II) complex using elemental halogens and methyl iodide.26 Other examples of transannular oxidative additions of group 14 halides have been reported for dinuclear Au(I), Au(II), Ir(I) and Rh(I) complexes,27–29 but remain limited to alkyl halides. Similarly, Brown and Puddephatt reported the oxidative addition of alkyl iodides at [(µ-dppm)3Pt2], which yielded the ionic Pt(I)2 complexes [(µ-dppm)2{(η1-dppm)Pt}{PtR}]I, in which a terminally coordinated dppm ligand takes the place of the iodide.30
Solvent-dependent reactivity of 3SiMeCl2-Cl and 3SiCl3-Cl
During the full NMR-spectroscopic characterisation of both 3SiMeCl2-Cl and 3SiCl3-Cl in CD2Cl2, the appearance of two new products was observed after several hours at rt (Scheme 1e). The minor one was identified by SCXRD as 4SiMeCl2-HCl and 4SiCl3-HCl, respectively, the product of the transannular addition of HCl, possibly derived from traces of HCl present from the degradation of the dichloromethane solvent through either hydrolysis, photolysis or Pt-catalysed oxidation with trace oxygen.31 The major one was identified as the rearrangement product 2SiMeCl-Cl and 2SiCl2-Cl, respectively, i.e. the corresponding silylene-bridged A-frame complex. Complexes 4SiMeCl2-HCl and 4SiCl3-HCl were systematically detected in small amounts (<5%) in dichloromethane solutions of 3SiMeCl2-Cl and 3SiCl3-Cl, respectively. For full characterisation 4SiCl3-HCl was synthesised independently by the addition of 1 equiv. HCl in Et2O to 3SiCl3-Cl (Scheme 1f). In the 31P{1H} NMR spectra, 4SiMeCl2-HCl and 4SiCl3-HCl appear as two multiplets around −7 to −9 ppm (P2PtHCl) and −14 ppm (P2PtSiCl), and in the 195Pt{1H} NMR spectra as two multiplets around −4600 and −4700 ppm, as also confirmed by 1H–195Pt{1H} HSQC experiments. The 1H NMR spectrum of 4SiCl3-HCl shows a highly shielded PtH triplet at −15.8 ppm (2JH–P = 13.2 Hz), with satellites to the two Pt nuclei (1JH–Pt = 1193 Hz, 3JH–Pt = 22.8 Hz). These values are similar to those of the mononuclear [trans-(Me3P)2PtHCl], which shows a PtH triplet at −16.2 ppm (2JH–P = 17 Hz), with a slightly larger coupling to the adjacent Pt nucleus (1JH–Pt = 1308 Hz).32The solid-state structure of 4SiCl3-HCl confirms the transannular oxidative addition of HCl at 3SiCl3-Cl (Fig. 4, right). The Pt⋯Pt distance of 3.1734(9) Å is similar to that of the A-frame complexes 2CH2-X, thus precluding any Pt–Pt bonding. As a result, the (µ-dmpm)2Pt2 framework is much less distorted than in 3SiCl3-Cl (|P–Pt–Pt–P|cis ≤ 28.4(1)°, |P–Pt–Pt–P|trans ≥ 157.5(1)°). The bridging hydride, located in the inverse Fourier map and freely refined, bridges very unsymmetrically between the Pt centres (Pt1–H1 1.87(11), Pt2⋯H1 2.23(11) Å). To our knowledge, 4SiCl3-HCl is the first example of a neutral hydridodiplatinum(II) complex, the only reported cationic one being the symmetrical complex [(µ-dmpm)2Pt2Me2(µ-H)]+, with a significantly shorter Pt⋯Pt distance 2.932(1) Å, the hydride not having been located in that structure.33
Whereas 3SiCl3-Cl fully rearranged to the µ-silylene A-frame complex 2SiCl2-Cl over 4 days at 60 °C in CH2Cl2, enabling its isolation in 70% yield, the rearrangement of 3SiMeCl2-Cl to 2SiMeCl-Cl remained incomplete, reaching only 17%, and was accompanied by the formation of the decomposition product [(µ-dmpm)2Pt2Cl2] (Scheme 1e). Like its methylene-bridged analogues, 2SiCl2-Cl proved unstable in solution, decomposing spontaneously to [(µ-dmpm)2Pt2Cl2] with release of the:SiCl2 unit, the exact fate of which was not ascertained. As such, 2SiCl2-Cl may provide an interesting starting point for studying platinum-mediated or -catalysed silylene transfer reactions, which are usually performed using silirane precursors.34
Complex 2SiCl2-Cl shows a 29Si NMR triplet with Pt satellites at 22.5 ppm (2JSi–P = 10.2 Hz, 1JSi–Pt = 1559 Hz), ca. 20 ppm upfield-shifted compared to the terminal silyl resonance of 3SiMeCl2-Cl (δ = 44.6 ppm). The 31P{1H} NMR singlet of 2SiCl2-Cl at 1.2 ppm is ca. 10 ppm upfield-shifted from that of its dichloromethylene-bridged analogue 2CCl2-Cl (δ = −8.7 ppm), as the silylene is more electron-donating than the methylene ligand. Both the 1JP–Pt (3098 (2SiCl2-Cl), 2750 (2CCl2-Cl) Hz) and 3JP–Pt (242 (2SiCl2-Cl), 150 (2CCl2-Cl) Hz) coupling constants increase significantly upon changing from the CCl2 to the SiCl2 bridge. Interestingly, complex 2SiMeCl-Cl displays two 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 31P{1H} NMR triplets at −0.1 and −2.8 ppm, rather than a singlet. This is owed to the asymmetry generated by the SiMeCl bridge, as the methyl and chloride substituents each point to one of the dmpm ligands, thereby creating slightly different magnetic environments. The 195Pt NMR triplet of 2SiCl2-Cl at −4446 ppm is also upfield-shifted from 2CCl2-Cl (δ = −4270 ppm), while its 3JPt–Pt coupling constant is substantially lower than that of all the methylene-bridged A-frames (3JPt–Pt = 297 (2SiCl2-Cl), 750–842 (2SiCH2-X) Hz),§ in line with the larger Pt⋯Pt distance in 2SiCl2-Cl (vide infra). For 2SiMeCl-Cl the asymmetry generated by the SiMeCl bridge results in a dddd resonance at −4519 ppm in the 195Pt{1H} NMR spectrum, with two distinct 1JPt–P (3261, 3114 Hz) and 3JPt–P (261, 203 Hz) coupling constants to each of the dmpm ligands (Fig. 5).
1 31P{1H} NMR triplets at −0.1 and −2.8 ppm, rather than a singlet. This is owed to the asymmetry generated by the SiMeCl bridge, as the methyl and chloride substituents each point to one of the dmpm ligands, thereby creating slightly different magnetic environments. The 195Pt NMR triplet of 2SiCl2-Cl at −4446 ppm is also upfield-shifted from 2CCl2-Cl (δ = −4270 ppm), while its 3JPt–Pt coupling constant is substantially lower than that of all the methylene-bridged A-frames (3JPt–Pt = 297 (2SiCl2-Cl), 750–842 (2SiCH2-X) Hz),§ in line with the larger Pt⋯Pt distance in 2SiCl2-Cl (vide infra). For 2SiMeCl-Cl the asymmetry generated by the SiMeCl bridge results in a dddd resonance at −4519 ppm in the 195Pt{1H} NMR spectrum, with two distinct 1JPt–P (3261, 3114 Hz) and 3JPt–P (261, 203 Hz) coupling constants to each of the dmpm ligands (Fig. 5).
Green crystals of 2SiCl2-Cl were analysed by SCXRD (Fig. 6, left). The much larger covalent radius of the silicon compared to the carbon bridge (rSi = 1.11 Å, rC = 0.76 Å) induces a significant widening of the Pt⋯Pt distance (3.5682(6) Å) compared to 2CH2-Cl (3.1475(5) Å), which in turn releases the strain of the (µ-dmpm)2Pt2 framework, resulting in a nearly ideal A-frame structure (|P–Pt–Pt–P|cis ≤ 7.87(6), |P–Pt–Pt–P|trans ≥ 161.05(6)°). The Pt–Si bonds in 2SiCl2-Cl (2.2610(17), 2.2541(17) Å) are significantly shorter than in the silyl complexes 3SiMe(3–n)Xn-X (2.2933(9)–2.357(2) Å), or other silylene-bridged diplatinum complexes with square-planar Pt coordination environments (2.2998(11)–2.412(2) Å).35,36 This shortening is likely due to the strain imposed by the A-frame and the electron-withdrawing effect of the chlorides trans to the silylene.
While there are numerous examples of silylene-bridged di- and triplatinum(0) complexes, usually synthesised by dehydrogenation of dihydrosilanes or the direct addition of stable silylenes to the Pt(0) precursors,37 our silylene-bridged A-frames are the first examples of µ-silylene diplatinum(II) complexes, obtained by twofold Si–X oxidative addition at two Pt(0) centres. There are, however, a couple of examples of homo- and heterobimetallic µ-silylene platinum(II) complexes obtained by the oxidative addition of the Si–H bond of a hydrosilyl ligand to a Pt centre.36 In these cases, the bridging SiR2 ligand should rather be viewed as a triplet silanediyl ligand with electron-sharing Pt–Si bonds, rather than a singlet silylene with Si→Pt donor–acceptor bonding. The same observation has been made for our BY-bridged A-frame complexes, which should be regarded as boranediyl-rather than borylene-bridged complexes.
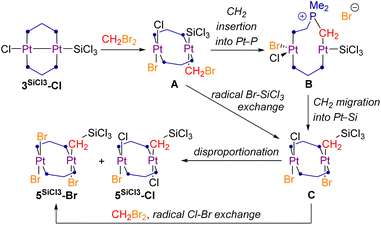
While in CH2Cl2 complex 3SiCl3-Cl rearranges to 2SiCl2-Cl, in CH2Br2 the unsymmetrical silyl(trihalo)diplatinum(II) complexes 5SiCl3-Br and 5SiCl3-Cl are formed, in which a unique twofold oxidative addition of CH2Br2, concomitant with methylene insertion into the Pt–Si bond, and in the case of 5SiCl3-Br an additional Cl–Br exchange, have taken place. The two products were formed in a 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture, as determined by NMR spectroscopy, and could not be separated by fractional crystallisation (Scheme 1g). Both species were detected by HRMS, and 5SiCl3-Br was additionally characterised by SCXRD (vide infra). In the 1H NMR spectrum complexes 5SiCl3-Br and 5SiCl3-Cl show a distinctive P2PtCH2 2H triplet at 1.74 and 2.35 ppm (2JH–P = 8.5 Hz), respectively, corresponding to a shielded 13C{1H} NMR PtCH2 multiplet at −1.2 ppm. In the 29Si{1H} NMR spectrum both compounds overlap at 2.5 ppm. The 31P{1H} NMR spectrum shows two triplets for each complex, reflecting their asymmetry, one set at −15.0 (P2PtBr(CH2SiCl3)) and −20.7 (P2PtBr2) ppm for 5SiCl3-Br (2JP–P = 11.8 Hz), and one at −9.6 (P2PtCl(CH2SiCl3)) and −16.7 (P2PtBrCl) ppm for 5SiCl3-Cl (2JP–P = 15.2 Hz), with 1JP–Pt coupling constants of ca. 2400 Hz for the P2PtX2 moiety and ca. 2700 Hz for the P2PtX(CH2SiCl3) moiety. These assignments are based on comparison with the 31P NMR data of the mononuclear complexes trans-[(Me3P)2PtX2] (X = Cl, δ = −15.8 ppm, 1JP–Pt = 2386 Hz; X = Br, δ = −21.5 ppm, 1JP–Pt = 2324 Hz),38 and trans-[(Me3P)2Pt(CH2SiMe3)Cl] (δ = −14.6 ppm, 1JP–Pt = 2765 Hz).39 The orientation of the Cl and Br ligands at the first Pt centre in 5SiCl3-Cl relative to the CH2SiCl3 ligand of the second Pt centre remains unclear, as no single crystals of 5SiCl3-Cl could be obtained.
1 mixture, as determined by NMR spectroscopy, and could not be separated by fractional crystallisation (Scheme 1g). Both species were detected by HRMS, and 5SiCl3-Br was additionally characterised by SCXRD (vide infra). In the 1H NMR spectrum complexes 5SiCl3-Br and 5SiCl3-Cl show a distinctive P2PtCH2 2H triplet at 1.74 and 2.35 ppm (2JH–P = 8.5 Hz), respectively, corresponding to a shielded 13C{1H} NMR PtCH2 multiplet at −1.2 ppm. In the 29Si{1H} NMR spectrum both compounds overlap at 2.5 ppm. The 31P{1H} NMR spectrum shows two triplets for each complex, reflecting their asymmetry, one set at −15.0 (P2PtBr(CH2SiCl3)) and −20.7 (P2PtBr2) ppm for 5SiCl3-Br (2JP–P = 11.8 Hz), and one at −9.6 (P2PtCl(CH2SiCl3)) and −16.7 (P2PtBrCl) ppm for 5SiCl3-Cl (2JP–P = 15.2 Hz), with 1JP–Pt coupling constants of ca. 2400 Hz for the P2PtX2 moiety and ca. 2700 Hz for the P2PtX(CH2SiCl3) moiety. These assignments are based on comparison with the 31P NMR data of the mononuclear complexes trans-[(Me3P)2PtX2] (X = Cl, δ = −15.8 ppm, 1JP–Pt = 2386 Hz; X = Br, δ = −21.5 ppm, 1JP–Pt = 2324 Hz),38 and trans-[(Me3P)2Pt(CH2SiMe3)Cl] (δ = −14.6 ppm, 1JP–Pt = 2765 Hz).39 The orientation of the Cl and Br ligands at the first Pt centre in 5SiCl3-Cl relative to the CH2SiCl3 ligand of the second Pt centre remains unclear, as no single crystals of 5SiCl3-Cl could be obtained.
The solid-state structure of 5SiCl3-Br (Fig. 6, right) shows two near-parallel square-planar Pt centres (Pt1 ∠ Pt2 ca. 3°) bridged by two dmpm ligands, one bearing two bromides and the other a bromide and a CH2SiCl3 ligand. The Pt⋯Pt distance of 3.400(1) Å is significantly shorter than in the related complex trans-[(µ-dmpm)2Pt2I4] (Pt⋯Pt 3.3477(6) Å).11 The Pt2–C1 bond length of 2.091(6) Å is similar to that of the related trans-[(Me2PhP)2PtCl(CH2SiMe3)] (2.079(14) Å).40
Insertion reactions of unsaturated hydrocarbons into Pt–Si bonds represent a key step in the Pt-catalysed hydrosilylation of alkenes and alkynes.41 To our knowledge, however, there have been no reports of methylene insertions into a Pt–Si bond. Puddephatt and coworkers have studied a similar insertion of methylene into the Pt–aryl bond of a cycloneophylplatinum(II) complex upon addition of CH2X2 (X = Cl, Br, I), which was calculated to proceed via a radical mechanism.42 Others have reported ionic reaction mechanisms of chloromethyl platinum(II) complexes involving the displacement of the chloride anion by various Lewis bases (LB) and formation of new Pt–CH2–LB linkages (E = PR3, SR2, NRn).43
Based on these studies, it is possible that the formation of 5SiCl3-Cl proceeds first via a formal transannular, likely radical oxidative addition of Br˙ and ˙CH2Br, yielding the unstable trans-(bromomethyl)silyl complex A (Scheme 2). Subsequent insertion of the CH2 unit into one of the Pt–P bonds and displacement of the bromide anion yields the phosphonium ylide B (or its dicationic Pt–Pt-bonded analogue if the second bromide also gets displaced). Complex B then undergoes CH2 migration into the Pt–Si bond, with concomitant reformation of the Pt–P bond and bromide addition to Pt2, yielding the silyl(trihalo)diplatinum(II) complex C. Alternatively, a radical Br–SiCl3 exchange at A could lead directly to C. Finally, C undergoes radical or ionic disproportionation to 5SiCl3-Cl and 5SiCl3-Br, or radical Cl–Br exchange to yield the observed excess of 5SiCl3-Br.
Comparative computational study of the reactivity of 1 with CH2Cl2, CCl4 and SiCl4
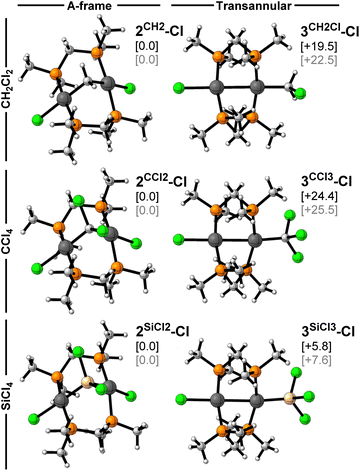
In order to understand the divergent reactivity of 1 towards dihalomethanes and polychlorosilanes, quantum-chemical calculations using density functional theory (DFT) were undertaken. All computations were performed at the ZORA44-BLYP45-D3(BJ)46/TZ2P47 level of theory, using the conductor-like screening model (COSMO)48 to simulate solvation in benzene (see the full computational details in the SI). This level of theory has been proven to be accurate for studying bond activation by group 10 metals and metal–metal interactions, both in previous reports,49,50 as well as in the additional performance test carried out in the current work (see details in the SI). The optimised structures of 2CH2-Cl and 3SiCl3-Cl at this level of theory are in good agreement with the experimental SCXRD-derived structures, notwithstanding small discrepancies attributable to the fact that the computed structures are optimised in solution rather than the solid state (see Fig. S78 in the SI).In order to identify the origin of the divergent reactivity of 1 towards CY2Cl2 (Y = H, Cl) and SiCl4, the relative stability of the two possible isomeric products, i.e. the methylene/silylene-bridged A-frame, and the product of transannular C/Si–Cl addition, was first investigated. The optimised geometries and relative energies are presented in Fig. 7. In all three cases the A-frame complex is the most stable species and thus corresponds to the thermodynamic product. This aligns with the experimentally observed eventual rearrangement of 3SiCl3-Cl to 2SiCl2-Cl upon prolonged heating in CH2Cl2. It is noteworthy that the energetic preference for the A-frame is significantly more pronounced for the halomethanes (ΔG ≥ 19.5 kcal mol−1) than the halosilane (ΔG = 5.8 kcal mol−1), which is consistent with the sole observation of the A-frame product in the former systems.
Although the A-frame product is thermodynamically favoured, all halosilane substrates yield the transannular diplatinum(I) complex at rt, which is therefore the kinetic product, associated with the lowest activation barrier in the selectivity-determining step. This is confirmed by our DFT calculations, which reveal two distinct two-step reaction mechanisms, one for the transannular oxidative addition of SiCl4 to 1, yielding 3SiCl3-Cl (TA pathway in blue, Fig. 8a), and the other for the direct twofold oxidative addition, yielding the A-frame 2SiCl2-Cl (AF pathway in red, Fig. 8a). In both cases SiCl4 first coordinates to 1 with release of the nbe ligands to form a hypervalent reactant complex (RC), a typical feature of silicon chemistry that cannot be accessed by the halocarbon analogues.51 This can occur either on one side of the diplatinum complex, yielding the transannular RC (TA-RC), or in the centre above the Pt⋯Pt unit, yielding the A-frame RC (AF-RC, Fig. 8b). The nature of the [SiCl4⋯1] interaction in both reactant complexes was assessed in more detail using quantitative energy decomposition analysis (see Table S1 in the SI).52 The results show that the electrostatic and orbital interactions are the main stabilising bonding components in both complexes and of similar magnitude. The main difference lies in the higher steric Pauli repulsion experienced by AF-RC as the SiCl4 molecule sits atop the centre of 1, rather than on one side. As a result, the formation of TA-RC is exergonic by ΔG1 = −12.9 kcal mol−1, whereas that of AF-RC is endergonic by ΔG1 = +14.1 kcal mol−1. Note that CCl4 and CH2Cl2 cannot form reactant complexes of this nature because the central carbon atom cannot become hypervalent, and the C–Cl bonds are relatively shorter and too rigid to allow this degree of bond angle deformation. Instead, CCl4 and CH2Cl2 form a reactant complex by coordinating on top of 1 through one of the Cl atoms (see Fig. S79 in the SI), thereby favouring an AF pathway.
Once the RC is formed, both pathways proceed with the selectivity-determining step, the oxidative addition of one Si–Cl bond at one of the Pt centres (Fig. 8a). In the corresponding transition-state (TS) structure of the AF pathway, AF-TS1, the SiCl4 moiety is highly deformed, resulting in a very high overall first activation barrier of ΔG1‡ = +24.3 kcal mol−1. In contrast, the TS structure of the first oxidative addition of the TA pathway, TA-TS1, strongly resembles that of TA-RC, resulting in a very low first activation barrier of ΔG1‡ = +0.9 kcal mol−1. In both cases an intermediate mixed-valence Pt(II)–Pt(0) complex is formed, in which the Pt(0) centre remains dicoordinate and the Pt(II) centre displays a distorted square-planar geometry, with either the silyl (AF-I) or the chloride (TA-I) ligand positioned above the Pt⋯Pt unit. While the formation of AF-I from AF-RC is highly exergonic (ΔG2 = −34.7 kcal mol−1), that of TA-I from TA-RC is only mildly so (ΔG2 = −2.0 kcal mol−1), making the latter process actually reversible. While the AF pathway is thus highly thermodynamically favoured, the TA pathway is kinetically much more accessible, and therefore always preferred.
In the AF pathway AF-I then undergoes a second Si–Cl bond addition at the remaining Pt(0) centre via AF-TS2 yielding the silylene-bridged 2SiCl2-Cl, with a much lower barrier than for the first addition (ΔG2‡ = +7.2 kcal mol−1). In the TA pathway the transfer of the second chloride to the second Pt centre, which yields 3SiCl3-Cl, is virtually barrierless (ΔG2‡ = +0.2 kcal mol−1). Consequently, the TA pathway is kinetically favoured for both reaction steps. Formation of the thermodynamically favoured silylene-bridged A-frame 2SiCl2-Cl could only be achieved by heating 3SiCl3-Cl in CH2Cl2 for four days (see Scheme 1e). The kinetic preference for the TA product is expected to be even more pronounced for the methylsilyl derivatives, as the larger methyl groups would render the formation of AF-RC and AF-TS1 even less favourable. In addition, the non-coordinating and larger methyl groups likely decrease, or even invert, the thermodynamic preference for the AF relative to the TA pathway, as additional steric clashes between the silane and Pt complex methyl groups would induce even larger Pauli repulsion in the AF reactant complexes and transition states (see SI Table S1). As a result, only 3SiCl3-Cl fully rearranges to 2SiCl2-Cl, whereas 3SiMeCl2-Cl rearranges only partially, and 3SiMe2Cl-Cl and 3SiMe3-Cl do not.
Conclusion
We demonstrated in this work the unique cooperative reactivity that a dinuclear platinum complex, [(µ-dmpm)2Pt2(nbe)2] (1), exhibits towards the double activation of C–X and Si–X bonds of CH2X2 (X = Cl, Br, I), CCl4 and Me4−nSiXn (X = Cl, n = 1–4; X = I, n = 1), respectively. Upon reaction with di- and tetrahalomethanes, complex 1 exclusively forms methylene-bridged A-frame structures (2CY2-X), while halosilanes undergo transannular oxidative addition reactions in which one Si–X bond is added across the two metal centres, leading to the formation of unsymmetrical silyldiplatinum(I) complexes (3SiMe(3−n)Xn-X). Upon heating in dichloromethane the transannular addition product of 1 and SiCl4, complex 3SiCl3-Cl, fully rearranges to the corresponding silylene-bridged A-frame complex 2SiCl2-Cl, while its MeSiCl3-derived analogue, 3SiMeCl2-Cl, undergoes only partial rearrangement. In contrast, heating in dibromomethane results in a highly unusual insertion of a CH2Br2-derived methylene unit into the Pt–Si bond (5SiCl3-X).DFT computations reveal that the methylene/silylene-bridged A-frame complexes are always the thermodynamic products, albeit with a much stronger preference for the A-frames in the halomethane-based reactions. For SiCl4 the direct two-step formation of the A-frame complex 2SiCl2-Cl has a much higher activation barrier than the pathway leading to the kinetic transannular addition product 3SiCl3-Cl. The propensity towards rearrangement of the transannular addition product 3SiMe(3−n)Cln-Cl to the A-frame is dictated by the number of methyl groups, as these clash with the dmpm methyl groups during the rearrangement.
The divergent reaction pathways described herein highlight the potential of bimetallic complexes in tuning cooperative bond activations, which could be integrated into new methylene/silylene transfer or silylation reactions. Further investigations to broaden the substrate scope and ultimately harness the cooperativity of these diplatinum complexes in catalytic transformations are underway in our laboratory.
Author contributions
M. P. designed the study, carried out the experimental work and wrote the SI. C. N. carried out and wrote the computational contributions. M. A. wrote the manuscript. H. B. and M. B. provided supervision and funding.Conflicts of interest
The authors declare no conflict of interest.Data availability
CCDC 2406490 (2CH2-Cl), 2406494 (2CH2-Br), 2406498 (2CH2-I), 2406509 (2CH2-Br′), 2406550 (3SiMe3-I), 2406556 (3SiMe2Cl-Cl), 2406558 (3SiMeCl2-Cl), 2406564 (3SiCl3-Cl), 2505905 (4SiCl3-HCl), 2406843 (2SiCl2-Cl) and 2406901 (5SiCl3-Br) contain the supplementary crystallographic data for this paper.54a–kThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: methods, synthetic procedures, NMR spectra, X-ray crystallographic and computational details. See DOI: https://doi.org/10.1039/d6sc00337k.
Acknowledgements
The authors thank Mr Christoph Maler for mass-spectroscopic and Dr Rüdiger Bertermann for NMR-spectroscopic analyses. Financial support from the Julius-Maximilians-Universität Würzburg, as well as funding from the Deutsche Forschungsgemeinschaft (DFG project number 466754611) is gratefully acknowledged. C. N. and F. M. B. thank the Dutch Research Council (NWO) for financial support and SURF Cooperative and VU ADA for the computational resources.References
- Selected reviews: (a) N. Chatani, Chem.-Asian J., 2025, 20, e00808 CrossRef CAS PubMed; (b) K. M. Korch and D. A. Watson, Chem. Rev., 2019, 119, 8192–8228 CrossRef CAS PubMed; (c) D. J. Jones, M. Lautens and G. P. McGlacken, Nat. Catal., 2019, 2, 843–851 CrossRef CAS; (d) D. J. Weix, Acc. Chem. Res., 2015, 48, 1767–1775 CrossRef CAS PubMed; (e) N. Kambe, T. Iwasakia and J. Terao, Chem. Soc. Rev., 2011, 40, 4937–4947 RSC; (f) J. K. Stille and K. S. Y. Lau, Acc. Chem. Res., 1977, 10, 434–442 CrossRef CAS.
- Selected reviews: (a) I. P. Beletskaya and A. V. Cheprakov, Chem. Rev., 2000, 100, 3009–3066 CrossRef CAS PubMed; (b) C. Cordovilla, C. Bartolome, J. M. Martínez-Ilarduya and P. Espinet, ACS Catal., 2015, 5, 3040–3053 CrossRef CAS; (c) F. Bellina, A. Carpita and R. Rossi, Synthesis, 2004, 15, 2419–2440 Search PubMed; (d) R. Chinchilla and C. Nájera, Chem. Rev., 2007, 107, 874–922 CrossRef CAS PubMed.
- T. Wang, B. J. Alfonso and J. A. Love, Org. Lett., 2007, 9, 5629–5631 CrossRef CAS PubMed.
- H. Kuniyasu, F. Yamashita, T. Hirai, J.-H. Ye, S.-I. Fujiwara and N. Kambe, Organometallics, 2006, 25, 566–570 CrossRef CAS.
- J.-T. Hong and H.-Y. Jang, J. Org. Chem., 2011, 76, 6877–6882 CrossRef CAS PubMed.
- H. Inubushi, H. Kondo, A. Lesbani, M. Miyachi, Y. Yamanoi and H. Nishihara, Chem. Commun., 2013, 49, 134–136 RSC.
- (a) S. Kozuch, C. Amatore, A. Jutand and S. Shaik, Organometallics, 2005, 24, 2319–2330 CrossRef CAS; (b) T. Nishida, S. Ogoshi, K. Tsutsumi, Y. Fukunishi and H. Kurosawa, Organometallics, 2000, 19, 4488–4491 CrossRef CAS; (c) A. L. Casado and P. Espinet, Organometallics, 1998, 17, 954–959 CrossRef CAS; (d) H. Kurosawa, H. Kajimaru, S. Ogoshi, H. Yoneda, K. Miki, K. Kasai, S. Murai and I. Ikeda, J. Am. Chem. Soc., 1992, 114, 8417–8424 CrossRef CAS; (e) M. F. Lappert and P. W. Lednor, J. Chem. Soc. Chem. Commun., 1973, 948–949 RSC.
- (a) H. Yamashita, T. Hayashi, T. Kobayashi, M. Tanaka and M. Goto, J. Am. Chem. Soc., 1988, 110, 4417–4418 CrossRef CAS; (b) C. J. Levy, R. J. Puddephatt and J. J. Vittal, Organometallics, 1994, 13, 1559–1560 CrossRef CAS; (c) H. Yamashita, T.-A. Kobayashi, M. Tanaka, J. A. Samuels and W. E. Streib, Organometallics, 1992, 11, 2330–2333 CrossRef CAS; (d) T. A. K. Al-Allaf, J. Organomet. Chem., 1999, 590, 25–35 CrossRef CAS; (e) C. Eaborn, A. Pidcock and B. R. Steele, J. Chem. Soc., Dalton Trans., 1976, 767–776 RSC; (f) J. Kuyper, Inorg. Chem., 1977, 16, 2171–2176 CrossRef CAS; (g) S. M. Nabavizadeh, F. Niroomand Hosseini, N. Nejabat and Z. Parsa, Inorg. Chem., 2013, 52, 13480–13489 CrossRef CAS PubMed.
- (a) M. Navarro, J. J. Moreno, M. Pérez-Jiménez and J. Campos, Chem. Commun., 2022, 58, 11220–11235 RSC; (b) R. Maity, B. S. Birenheide, F. Breher and B. Sarkar, Chem. – Eur. J., 2021, 13, 2337–2370 CAS; (c) B. M. Trost, C.-I. Hung and G. Matac, Angew. Chem., Int. Ed., 2020, 59, 4240–4261 CrossRef CAS PubMed; (d) J. Parka and S. Hong, Chem. Soc. Rev., 2012, 41, 6931–6943 RSC.
- C. Brunecker, J. H. Müssig, M. Arrowsmith, F. Fantuzzi, A. Stoy, J. Böhnke, A. Hofmann, R. Bertermann, B. Engels and H. Braunschweig, Chem. – Eur. J., 2020, 26, 8518–8523 CrossRef CAS PubMed.
- C. Brunecker, M. Arrowsmith, J. H. Müssig, J. Böhnke, A. Stoy, M. Heß, A. Hofmann, C. Lenczyk, C. Lichtenberg, J. Ramler, A. Rempel and H. Braunschweig, Dalton Trans., 2021, 50, 3506–3515 RSC.
- C. Brunecker, M. Arrowsmith, F. Fantuzzi and H. Braunschweig, Angew. Chem., Int. Ed., 2021, 60, 16864–16868 CrossRef CAS PubMed.
- (a) D. M. Hoffmann and R. Hoffmann, Inorg. Chem., 1981, 20, 3543–3555 CrossRef; (b) C. P. Kubiak and R. Eisenberg, J. Am. Chem. Soc., 1977, 99, 6129–6131 CrossRef CAS.
- (a) E. Lumbreras Jr., E. M. Sisler and Q. D. Shelby, J. Organomet. Chem., 2010, 695, 201–205 CrossRef; (b) S. Hinze, J. K. Gong, P. E. Fanwick and C. P. Kubiak, J. Organomet. Chem., 1993, 458, C10–C11 CrossRef CAS; (c) X. L. R. Fontaine, S. J. Higgins and B. L. Shaw, J. Chem. Soc., Dalton Trans., 1988, 1179–1184 RSC; (d) S. J. Young, B. Kellenberger, J. H. Reibenspies, S. E. Himmel, M. Manning, O. P. Anderson and J. K. Stille, J. Am. Chem. Soc., 1988, 110, 5744–5753 CrossRef CAS; (e) X. L. R. Fontaine, S. J. Higgins, B. L. Shaw, M. Thornton-Pett and W. Yichang, J. Chem. Soc., Dalton Trans., 1987, 1501–1507 RSC; (f) A. L. Balch, C. T. Hunt, C.-L. Lee, M. M. Olmstead and J. P. Farr, J. Am. Chem. Soc., 1981, 103, 3764–3772 CrossRef CAS.
- (a) M. Cowie, Can. J. Chem., 2005, 83, 1043–1055 CrossRef CAS; (b) S. J. Trepanier, J. N. L. Dennett, B. T. Sterenberg, R. McDonald and M. Cowie, J. Am. Chem. Soc., 2004, 126, 8046–8058 CrossRef CAS PubMed; (c) M. Akita, R. Hua, S. A. R. Knox, Y. Moro-oka, S. Nakanishi and M. I. Yates, J. Organomet. Chem., 1998, 569, 71–83 CrossRef CAS; (d) T. B. Gunnoe, P. S. White, J. L. Templeton and L. Casarrubios, J. Am. Chem. Soc., 1997, 119, 3171–3172 CrossRef CAS; (e) P. M. Maitlis, J. Organomet. Chem., 1995, 500, 239–249 CrossRef CAS.
- M. P. Brown, J. R. Fisher, R. J. Puddephatt and K. R. Seddon, Inorg. Chem., 1979, 18, 2808–2813 CrossRef CAS.
- (a) M. C. Grossel, J. R. Batson, R. P. Moulding and K. R. Seddon, J. Organomet. Chem., 1986, 304, 391–423 CrossRef CAS; (b) M. P. Brown, R. J. Puddephatt, L. Rashidi and K. R. Seddon, J. Chem. Soc., Dalton Trans., 1978, 516–522 RSC.
- K. A. Azam, A. A. Frew, B. R. Lloyd, L. Manojlovic-Muir, K. W. Muir and R. J. Puddephatt, Organometallics, 1985, 4, 1400–1406 CrossRef CAS.
- (a) R. Yu. Lukin, A. M. Kuchkaev, A. V. Sukhov, G. E. Bekmukhamedov and D. G. Yakhvarov, Polymers, 2020, 12, 2174 CrossRef CAS PubMed; (b) D. Troegel and J. Stohrer, Coord. Chem. Rev., 2011, 255, 1440–1459 CrossRef CAS; (c) A. Hamze, O. Provot, M. Alami and J.-D. Brion, Org. Lett., 2006, 8, 931–934 CrossRef CAS PubMed; (d) F. Ozawa, J. Organomet. Chem., 2000, 611, 332–342 CrossRef CAS; (e) M. Tanaka, Y. Uchimaru and H.-J. Lautenschlager, Organometallics, 1991, 10, 16–18 CrossRef CAS; (f) T. Hayashi, T.-a. Kobayashi, A. M. Kawamoto, H. Yamashlta and M. Tanaka, Organometallics, 1990, 9, 280–281 CrossRef CAS.
- A. Roscher, A. Bockholt and T. Braun, Dalton Trans., 2009, 1378–1382 RSC.
- M. C. Grossel, R. P. Moulding and K. R. Seddon, Inorg. Chim. Acta Lett., 1982, 64, L275–L277 CrossRef CAS.
- K. A. Brittingham and S. Schreiner, J. Chem. Educ., 1995, 72, 941–943 CrossRef CAS.
- M. Passargus, M. Arrowsmith, R. Bertermann, M. Finze and H. Braunschweig, Inorg. Chem., 2025, 64, 21111–21129 CrossRef CAS PubMed.
- N. I. Khan, C. King, J.-C. Wang, S. Wang and J. P. Fackler Jr., Inorg. Chem., 1989, 28, 4656–4662 CrossRef.
- (a) P. Kapoor, K. Lövqvist and Å. Oskarsson, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1995, 51, 611–613 CrossRef; (b) Y.-J. Kim, J.-I. Park, S.-C. Lee, K. Osakada, M. Tanabe, J.-C. Choi, T.-A. Koizumi and T. Yamamoto, Organometallics, 1999, 18, 1349–1352 CrossRef CAS.
- H. Schmidbaur and R. Franke, Inorg. Chim. Acta, 1975, 13, 85–89 CrossRef CAS.
- (a) H. E. Abdou, A. A. Mohamed and J. P. Fackler Jr., Inorg. Chem., 2007, 46, 9692–9699 CrossRef CAS PubMed; (b) J. D. Basil, H. Murray, J. P. Fackler Jr., J. Tocher, A. M. Mazany, B. Trzcinska-Bancroft, H. Knachel, D. Dudis, T. J. Delord and D. O. Marler, J. Am. Chem. Soc., 1985, 107, 6908–6915 CrossRef CAS; (c) H. Schmidbaur and P. Jandik, Inorg. Chim. Acta, 1983, 74, 97–99 CrossRef CAS.
- (a) M. T. Pinillos, A. Elduque, J. A. Lopez, F. J. Lahoz and L. A. Oro, J. Chem. Soc., Dalton Trans., 1991, 1391–1395 RSC; (b) M. A. Ciriano, J. J. Pérez-Torrente and L. A. Oro, J. Organomet. Chem., 1993, 445, 273–281 CrossRef CAS.
- (a) M. P. Brown, A. Yavari, R. H. Hill and R. J. Puddephatt, J. Chem. Soc., Dalton Trans., 1985, 2421–2425 RSC; (b) K. A. Azam, M. P. Brown, R. H. Hill, R. J. Puddephatt and A. Yavari, Organometallics, 1984, 3, 697–702 CrossRef CAS.
- N. S. Lewis, K. R. Mann, J. G. Gordon II and B. Gray, J. Am. Chem. Soc., 1976, 98, 7461–7463 CrossRef CAS.
- (a) I. Fells and E. A. Moelwyn-Hughes, J. Chem. Soc., 1958, 1326–1333 RSC; (b) M. E. Jacox and D. E. Milligan, J. Chem. Phys., 1970, 53, 2688–2701 CrossRef CAS; (c) L. Pinard, J. Mijoin, P. Ayrault, C. Canaff and P. Magnoux, Appl. Catal. B Environ., 2004, 51, 1–8 CrossRef CAS.
- D. L. Packett, C. M. Jensen, R. L. Cowan, C. E. Strouse and W. C. Trogler, Inorg. Chem., 1985, 24, 3578–3583 CrossRef CAS.
- M. P. Brown, S. J. Cooper, A. A. Frew, L. Manojlović-Muir, K. W. Muir, R. J. Puddephatt and M. A. Thomson, J. Chem. Soc., Dalton Trans., 1982, 299–305 RSC.
- (a) Z. Nevárez and K. A. Woerpel, Org. Lett., 2007, 9, 3773–3776 CrossRef PubMed; (b) M. A. Greene, M. Prévost, J. Tolopilo and K. A. Woerpel, J. Am. Chem. Soc., 2012, 134, 12482–12484 CrossRef CAS PubMed; (c) Z. Li and C. He, Eur. J. Org Chem., 2006, 4313–4322 CrossRef CAS.
- (a) M. Tanabe, Y. Nakamura, T.-a. Niwa, M. Sakai, A. Kaneko, H. Toi, K. Okuma, Y. Tsuchido, T.-a. Koizumi, K. Osakada and T. Ide, Organometallics, 2022, 41, 3301–3312 CrossRef CAS; (b) M. Tanabe, J. Jiang, H. Yamazawa, K. Osakada, T. Ohmura and M. Suginome, Organometallics, 2011, 30, 3981–3991 CrossRef CAS; (c) H. Arii, M. Takahashi, M. Nanjo and K. Mochida, Dalton Trans., 2010, 39, 6434–6440 RSC; (d) J. Braddock-Wilking, J. Y. Corey, K. A. Trankler, K. M. Dill, L. M. French and N. P. Rath, Organometallics, 2004, 23, 4576–4584 CrossRef CAS; (e) K. A. Brittingham, T. N. Gallaher and S. Schreiner, Organometallics, 1995, 14, 1070–1072 CrossRef CAS.
- J. Braddock-Wilking, Y. Levchinsky and N. P. Rath, Inorg. Chim. Acta, 2002, 330, 82–88 CrossRef CAS.
- (a) S. Shimada, M. L. N. Rao, Y. Hua Li and M. Tanaka, Organometallics, 2005, 24, 6029–6036 CrossRef CAS; (b) J. Powell, J. F. Sawyer and M. Shiralian, Organometallics, 1989, 8, 577–583 CrossRef CAS.
- R. Favez, R. Roulet, A. A. Pinkerton and D. Schwarzenbach, Inorg. Chem., 1980, 19, 1356–1365 CrossRef CAS.
- R. Kapadia, J. B. Pedley and G. B. Young, Inorg. Chim. Acta, 1997, 265, 235–239 CrossRef CAS.
- B. Jovanović, L. Manojlović-Muir and K. W. Muir, J. Chem. Soc., Dalton Trans., 1974, 195–198 RSC.
- (a) K. M. Lewis and S. Couderc, Molecules, 2022, 27, 4341 CrossRef CAS PubMed; (b) P. Gigler, M. Drees, K. Riener, B. Bechlars, W. A. Herrmann and F. E. Kuhn, J. Catal., 2012, 295, 1–14 CrossRef CAS; (c) I. E. Markó, S. Stérin, O. Buisine, G. Mignani, P. Branlard, B. Tinant and J. P. Declercq, Science, 2002, 298, 204–206 CrossRef PubMed; (d) A. K. Roy and R. B. Taylor, J. Am. Chem. Soc., 2002, 124, 9510–9524 CrossRef CAS PubMed; (e) F. Ozawa, J. Organomet. Chem., 2000, 611, 332–342 CrossRef CAS.
- (a) M. E. Moustafa, P. D. Boyle and R. J. Puddephatt, J. Organomet. Chem., 2021, 941, 121803 CrossRef CAS; (b) M. A. Fard, A. Behnia and R. J. Puddephatt, J. Organomet. Chem., 2019, 890, 32–42 CrossRef CAS.
- (a) R. McCrindle, G. J. Arsenault, A. Gupta, M. J. Hampden-Smith, R. E. Rice and A. J. McAlees, J. Chem. Soc., Dalton Trans., 1991, 949–954 RSC; (b) R. McCrindle, G. Ferguson and A. J. McAlees, J. Chem. Soc., Chem. Commun., 1990, 1524–1526 RSC; (c) R. McCrindle, G. Ferguson, G. J. Arsenault, M. J. Hampden-Smith, A. J. McAlees and B. L. Rub, J. Organomet. Chem., 1990, 390, 121–126 CrossRef CAS; (d) N. W. Alcock, P. G. Pringle, P. Bergamini, S. Sostero and O. Traverso, J. Chem. Soc., Dalton Trans., 1990, 1553–1556 RSC; (e) J. F. Hoover and J. M. Stryker, Organometallics, 1988, 7, 2082–2084 CrossRef CAS; (f) R. McCrindle, G. J. Arsenault and R. Farwaha, J. Organomet. Chem., 1985, 296, C51–C53 CrossRef CAS.
- (a) E. van Lenthe, A. Ehlers and E. J. Baerends, J. Chem. Phys., 1999, 110, 8943–8953 CrossRef CAS; (b) E. van Lenthe, E. J. Baerends and J. G. Snijders, J. Chem. Phys., 1994, 101, 9783–9792 CrossRef CAS.
- (a) A. D. Becke, Phys. Rev. A, 1988, 38, 3098–3100 CrossRef CAS PubMed; (b) C. Lee, W. Yang and R. G. Parr, Phys. Rev. B, 1988, 37, 785–789 CrossRef CAS PubMed; (c) Q. Wu and W. Yang, J. Chem. Phys., 2002, 116, 515–524 CrossRef CAS.
- (a) S. Grimme, J. Comput. Chem., 2004, 25, 1463–1473 CrossRef CAS PubMed; (b) S. Grimme, J. Comput. Chem., 2006, 27, 1787–1799 CrossRef CAS PubMed; (c) S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed; (d) S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456 CrossRef CAS PubMed; (e) A. D. Becke and E. R. Johnson, J. Chem. Phys., 2005, 122, 154101 CrossRef PubMed; (f) E. R. Johnson and A. D. Becke, J. Chem. Phys., 2006, 124, 024101 CrossRef PubMed.
- E. van Lenthe and J. Baerends, J. Comput. Chem., 2003, 24, 1142–1156 CrossRef CAS PubMed.
- (a) A. Klamt and G. Schüürmann, J. Chem. Soc., Perkin Trans. 2, 1993, 799–805 RSC; (b) A. Klamt, J. Phys. Chem., 1995, 99, 2224–2235 CrossRef CAS; (c) C. C. Pye and T. Ziegler, Theor. Chem. Acc., 1999, 101, 396–408 Search PubMed.
- (a) A. Diefenbach and F. M. Bickelhaupt, J. Phys. Chem. A, 2004, 108, 8460–8466 CrossRef CAS; (b) G. T. de Jong, M. Solà, L. Visscher and F. M. Bickelhaupt, J. Chem. Phys., 2004, 121, 9982–9991 CrossRef CAS PubMed; (c) T. Hansen, X. Sun, M. Dalla Tiezza, W.-J. van Zeist, J. N. P. van Stralen, D. P. Geerke, L. P. Wolters, J. Poater, T. A. Hamlin and F. M. Bickelhaupt, Chem. – Eur. J., 2022, 28, e202201093 CrossRef CAS PubMed; (d) J. N. P. van Stralen and F. M. Bickelhaupt, Organometallics, 2006, 25, 4260–4264 CrossRef CAS; (e) N. Arnold, R. Bertermann, F. M. Bickelhaupt, H. Braunschweig, M. Drisch, M. Finze, F. Hupp, J. Poater and J. A. P. Sprenger, Chem. – Eur. J., 2017, 23, 5948–5955 CrossRef CAS PubMed.
- M. B. Brands, J. Nitsch and C. Fonseca Guerra, Inorg. Chem., 2018, 57, 2603–2613 CrossRef CAS PubMed.
- S. C. A. H. Pierrefixe, C. Fonseca Guerra and F. M. Bickelhaupt, Chem. – Eur. J., 2008, 14, 819–828 CrossRef PubMed.
- (a) F. M. Bickelhaupt and E. J. Baerends, in Reviews in Computational Chemistry, ed. K. B. Lipkowitz and D. B. Boyd, Wiley-VCH, New York, 2000, 15, pp. 1–86 Search PubMed; (b) T. A. Hamlin, P. Vermeeren, C. Fonseca Guerra and F. M. Bickelhaupt, in Complementary Bonding Analysis, ed. S. Grabowsky, De Gruyter, Berlin, 2021, 8, pp. 199–212 Search PubMed.
- R. Walsh, Acc. Chem. Res., 1981, 14, 246–252 CrossRef CAS.
- (a) CCDC 2406490: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls4q8; (b) CCDC 2406494: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls4vd; (c) CCDC 2406498: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls4zj; (d) CCDC 2406509: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls5bx; (e) CCDC 2406550: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls6n8; (f) CCDC 2406556: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls6vg; (g) CCDC 2406558: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls6xj; (h) CCDC 2406564: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ls73r; (i) CCDC 2406843: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2lsj31; (j) CCDC 2406901: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2lskzy; (k) CCDC 2505905: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q3ln3.
Footnotes |
| † In order to study silicon–halide bond activation, methylhalosilanes were chosen instead of dihydrodihalosilanes (SiH2X2) as the Si–H bond is significantly weaker than the Si–X bond,53 resulting in preferential Si–H activation in test reactions with SiHCl3 and Me2SiHCl. |
| ‡ The 29Si{1H} NMR signal for 3SiCl3-Cl was too weak to be detected. |
| § The 2JPt–Pt coupling constant of 2CCl2-Cl could not be determined due to poor spectral resolution. |
| This journal is © The Royal Society of Chemistry 2026 |