Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Activation volumes associated with excited-state electron transfer across amidinium-carboxylate bridge
Daniel
Langford
 a,
René
Weiß
ab,
Marcel
Krug
a,
René
Weiß
ab,
Marcel
Krug
 ab,
Maxence
Urbani
ab,
Maxence
Urbani
 c,
Achim
Zahl
d,
Carolin
Müller
c,
Achim
Zahl
d,
Carolin
Müller
 b,
Timothy
Clark
b,
Timothy
Clark
 b,
Tomás
Torres
b,
Tomás
Torres
 *ce and
Dirk M.
Guldi
*ce and
Dirk M.
Guldi
 *a
*a
aFAU Profile Center Solar, Department of Chemistry and Pharmacy & Interdisciplinary Center for Molecular Materials (ICMM), Friedrich-Alexander-Universität Erlangen-Nürnberg, Egerlandstr. 3, Erlangen 91058, Germany. E-mail: dirk.guldi@fau.de
bDepartment of Chemistry and Pharmacy, Computer-Chemistry Center, Friedrich-Alexander-Universität Erlangen-Nürnberg, Nägelsbachstr. 25, Erlangen 91052, Germany
cInstituto Madrileño de Estudios Avanzados (IMDEA) – Nanociencia, C/ Faraday 9, Madrid 28049, Spain
dDepartment of Chemistry and Pharmacy, Chair of Chemistry of Thin Film Materials, Friedrich-Alexander-Universität Erlangen-Nürnberg, Cauerstr. 3, Erlangen 91058, Germany
eDepartamento de Química Orgánica & Institute for Advanced Research in Chemical Sciences (IAdChem), Universidad Autonoma de Madrid, Spain
First published on 1st April 2026
Abstract
Pressure was used to modulate interactions in an electron donor–acceptor system, composed of a zinc porphyrin (ZnP) and a fullerene (C60), held together by an amidinium-carboxylate salt-bridge (ZnP-H⋯C60). Two different trends evolved in steady-state absorption assays. Volume compression causes an absorbance intensification, and a solvatochromic-like red shift that stems from increased E-field density. Pressure-dependent femtosecond and nanosecond transient-absorption experiments were performed to investigate the activation volumes of the excited-state deactivation processes in ZnP-H⋯C60. Solvent relaxation of S1 is found to have a highly positive ΔVk2‡. The pressure-induced rate attenuation for this process is assumed to be linked to the solvent's viscosity increase. Intersystem crossing to the porphyrin-centered T1 state is free of intrinsic and extrinsic reorganizations and, as such, the activation volume is close to zero. The same applies for the subsequent ground-state deactivation from T1 to S0. Charge-separation to afford (ZnP)˙+⋯H⋯(C60)˙− is linked to a volume compression towards the activated state with ΔVk3‡ = −5.7 ± 2.2 cm3 mol−1. The charge-recombination undergoes, within the experimental margins of error, an equal volume expansion with ΔVk4‡ = +8.6 ± 0.7 cm3 mol−1. This effect is linked to the generation and/or neutralization of charges, best described by the Jung equation for electrostrictive volume changes in dipolar zwitterionic entities. Importantly, volumetric contributions from a possible PT towards the activated state were not observed.
Introduction
Nature has developed various photoenergy–driven reactions that build up the world around us.1–5 Attempts, for example, to mimic nature's light harvesting systems have received remarkable attention in recent research.6–9 one of the cornerstones in the pursuit of transforming our fossil fuel-based society towards renewables. Nature's sophistication in converting photoenergy to chemical fuels in photosynthesis has inspired research on porphyrins10 and porphyrinoid-based11 materials and their integration into electron donor–acceptor systems.Their capability to donate electrons in their photoexcited states renders them a perfect building block for artificial electron donor–acceptor systems. The electron acceptors of choice are fullerenes:12 Various compositions and configurations have been investigated over the years.12 Bridges that connect electron donors and acceptors ranging from covalent bonds,13,14 and through supramolecular binding like π–π interaction,15–19 to neutral hydrogen bonds20–23 and hydrogen bonded salt bridges24–27 have been found to be important.
Electron transfer (ET) in biological systems like proteins is often coupled to a proton transfer (PT) in the hydrogen-bonded peptide environment. Leading examples are cytochrome-c oxidase in cell respiration,28 or photosystems I29 and II30,31 in photosynthesis. Investigations are, however, challenging because of the complexity of the protein's structure and dynamics. Therefore, mimics have been developed and investigated to gather insight into the fundamental processes. Among others,21,32 the amidinium-carboxylate salt bridge stands out. Its chemical versatility, combination with a plethora of electron donors and acceptors, and its structural similarity to arginine–aspartic acid dimers in proteins, are of utmost importance.33
The amidinium-carboxylate salt bridge was used as a blueprint to describe proton-coupled electron transfer (PCET) reactions,34–38 while they were studied with time-resolved spectroscopy experiments.24–26,39–42 Weak electronic couplings between the respective electron donors and acceptors, as seen in temperature-dependent transient-absorption experiments, suggests that the hydrogen-bonding interface is the bottleneck for electron transfer reactions.43 Additionally, the orientation of the salt-bridge was identified to influence the electron transfer (ET) rate significantly. If the salt-bridge is oriented as shown in Fig. 1, that is, amidinium on the electron donor and carboxylate on the electron acceptor, the electron transfer rate was attenuated compared to a hydrogen-bonded heterodimer with carboxylic acids on both the electron donor and electron acceptor.24,39–42 If the salt-bridge is oriented the other way round, that is, the carboxylate on the electron donor and the amidinium on the electron acceptor, a higher electron transfer rate was observed.24,25 The rationales for the attenuation are twofold. First, the adverse effect of the electric field across the bridge reduces the ET rate.33 Second, a coupling between the charge-separation and -recombination rate with the bridging protons' vibrational wavefunction has been reported.43 The notion of ET across the amidinium-carboxylate bridge being coupled to the proton configuration has been identified in temperature-dependent kinetic isotope effects (KIE). In particular, the thermal population of different vibrational modes alters the ET rate.40,42
Questions related to any ET coupling that goes beyond the bridging proton's vibrational configuration – implying that ET is coupled to a proton transfer towards the electron acceptor – could not be finally answered.43 In this notion, the photoexcitation of the electron donor, and/or the charge-separation forming to the one-electron oxidized porphyrin and one-electron reduced fullerene, are expected to come together with a significant change of the amidinium's and carboxylate's acidity/basicity properties.33 Most of these investigations were limited by the spectroscopic features associated with the electron transfer. Proton transfer across the amidinium-carboxylate bridge is often not spectroscopically observable. It is the lack of characteristic features in the observable region that hampers an unambiguous characterization. To date, clarification of the role of the bridging protons during the charge-separation and recombination through amidinium-carboxylate salt bridges remains a challenging task. The key factor for identifying the role of the bridging proton during the observable ET has been postulated to be embodied within reorganization- and solvation free energy changes, as ET or a PCET reaction are expected to yield differently charged products, which can be linked to differently charged intermediate states.35,36,43
According to Marcus–Hush theory, electron-transfer reaction rates k depend on both the free-energy difference ΔG0 and the reorganization energy λ of the reactants (eqn (1)). Reorganization in ET or PCET reaction is, meanwhile, best described by transition-state theory,44 which assumes that an electron donor A and an electron acceptor B are in equilibrium with an activated complex [AB]‡. Charge separation proceeds from [AB]‡ and generates C (eqn (2)). The reaction steps involved, H-bond dissociation, H-bond formation, and redistribution of electronic charges, are traceable by means of the corresponding activation volumes ΔV‡ (eqn (3)). ΔV‡ is governed by two contributions: first, intrinsic volume changes due to contraction, expansion, and reorientation of bonds within electron donating A and electron accepting B.45 Second, electrostrictive volume changes by reason of contraction, expansion, and reorientation of the solvation shell upon charge-separation.46 In general, ΔV‡ is a powerful tool to characterize ligand–substitution reactions in metal complexes.47 Using ΔV‡ to investigate reorganization processes has been a useful tool for characterizing ET reaction, but has found little attention for characterizing PCET reactions.47–49
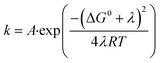 | (1) |
| A + B ⇌ [AB]‡ → C | (2) |
| ΔV‡ = V‡[AB] − (VA + VB) | (3) |
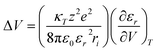 | (4) |
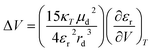 | (5) |
Freely dissolved ions are, however, not formed upon charge-separation in covalently linked or preorganized electron donor–acceptor couples. Instead, the electron donor–acceptor couples become dipolar with zwitterionic character. Here, the Jung formulism is applicable, where volumetric changes induced by a formally spherical zwitterion depend on the radius rd of the dipolar molecule and the dipole moment µd, as described in eqn (5).46,51 Our present work aims to test the effects described by the Jung formulation by determining the activation volumes associated with an excited-state ET reaction across an amidinium-carboxylate bridged zinc porphyrin-fullerene (ZnP-H⋯C60) couple (Fig. 1). This work advances beyond our previous study, which focused on activation volume effects in independently dissolved electron donors and acceptors using Drude–Nernst theory, by investigating a preorganized electron donor–acceptor system through the Jung formalism.49 We establish a methodology to investigate pressure-induced effects in equilibria of supramolecular complexation based on pressure-dependent steady-state absorption and emission experiments. We gather a fully-fledged description of how hydrostatic pressure influences the electronic ground and excited states of (ZnP-H⋯C60) and how pressure influences their dynamics.
Results and discussion
Basic characterization
Previous work on electron donor–acceptor couples featuring porphyrins (ZnP) and fullerenes (C60) has shown that photoexciting ZnPs results either in an electron-transfer or an energy-transfer reaction to C60.13,14,52,53 The solvent polarity is decisive here. In low-polarity solvents like toluene, energy-transfer dominates over electron transfer. In high-polarity solvents, electron-transfer is observed exclusively.13,14,52,53 Importantly, solvents of high polarity like tetrahydrofuran, benzonitrile, or dimethylformamide interfere with intermolecular hydrogen-bonds.10a,54 As such, we took anisole for the current studies. Anisole is sufficiently polar to direct the photoreactivity to electron transfer and allows an effective formation of hydrogen-bonded amidinium-carboxylate bridge.Steady-state absorption and fluorescence spectra of ZnP in anisole under ambient conditions are shown in Fig. 2, while the absorption spectrum of C60 is depicted in Fig. S21 in the SI. For all experiments with ZnP, a 1000-fold excess of 4-dimethylaminopyridine (DMAP) was necessary to suppress any π-stacking (Fig. S22 in the SI). Superimposed on the Soret-band absorption of ZnP at 432 nm (351![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 dm3 mol−1 cm−1) is a well-defined, blue-shifted shoulder with a maximum at 411 nm. Two Q-band absorption maxima evolve at 567 nm (11
000 dm3 mol−1 cm−1) is a well-defined, blue-shifted shoulder with a maximum at 411 nm. Two Q-band absorption maxima evolve at 567 nm (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 dm3 mol−1 cm−1) and 607 nm (5400 dm3 mol−1 cm−1). The Soret-band absorption of ZnP at 432 nm is about 100-times stronger than the absorptions of C60 with 4000 dm3 mol−1 cm−1 (Fig. S21 in the SI). Hence, the Soret-band absorptions were used to photoexcite predominantly ZnP for all pressure-dependent experiments. Fluorescence maxima at 612 and 669 nm were recorded for ZnP upon photoexcitation. The ZnP fluorescence quantum yield was determined using the integrating sphere methodology resulting in values of 4.0% and 3.0% following Soret-band photoexcitation at 430 nm and Q-band photoexcitation at 567 nm, respectively. The fluorescence quantum yields from the two excitations are equal within experimental error.
900 dm3 mol−1 cm−1) and 607 nm (5400 dm3 mol−1 cm−1). The Soret-band absorption of ZnP at 432 nm is about 100-times stronger than the absorptions of C60 with 4000 dm3 mol−1 cm−1 (Fig. S21 in the SI). Hence, the Soret-band absorptions were used to photoexcite predominantly ZnP for all pressure-dependent experiments. Fluorescence maxima at 612 and 669 nm were recorded for ZnP upon photoexcitation. The ZnP fluorescence quantum yield was determined using the integrating sphere methodology resulting in values of 4.0% and 3.0% following Soret-band photoexcitation at 430 nm and Q-band photoexcitation at 567 nm, respectively. The fluorescence quantum yields from the two excitations are equal within experimental error.
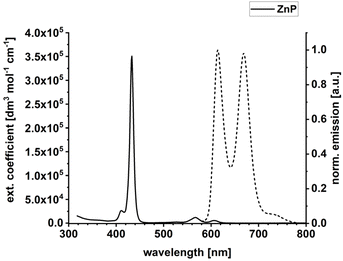 | ||
| Fig. 2 Absorption (solid) and emission (dashed) spectrum of ZnP in anisole. A 1000-fold excess of DMAP vs. ZnP was used in all optical spectroscopy experiments. | ||
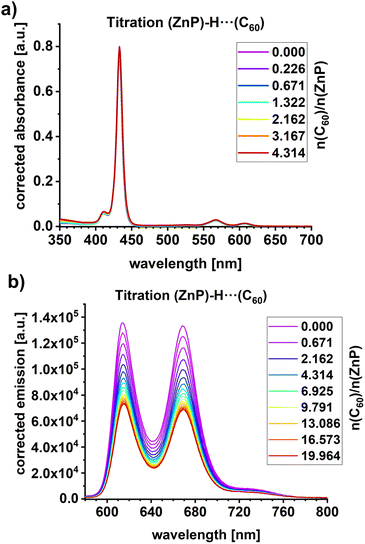
Titration experiments were conducted to investigate the complexation of C60 with ZnP (Fig. 3). Only subtle changes were observed in the absorption spectra upon gradual addition of C60, up to a 4-fold excess relative to ZnP. The mostly unaffected ZnP absorption is attributed to the fact that C60 lacks significant impact on the electronic properties of the Franck–Condon region. Notably, the Q- and B-band transitions are primarily associated with dipole-allowed π–π* excitations localized on ZnP. Time-dependent density functional theory (TDDFT) calculations support this interpretation (SI). Nearly identical π–π* transitions for the ZnP Q- and B-band transitions (S1/2: 2.16 eV; S3/4: 3.12 eV) and ZnP-H⋯C60 (S1/2: 2.17 eV; S3/4: 3.12 eV) are predicted (Fig. S25–S28 in the SI). In contrast, the fluorescence gave rise to a 50% reduction in intensity (Fig. 3b). We take the fluorescence quenching to indicate the presence of a new deactivation pathway in the ZnP-H⋯C60 complex that is not present in ZnP.
Using the steady-state fluorescence titrations (Fig. 3b), the binding constant was determined to be 1.9 × 105 M−1 (Fig. S23 in the SI). This binding constant and the 50% reduction in fluorescence intensity are in line with previously reported data on comparable supramolecular amidinium-carboxylate salt bridged systems.55–57
Job's method of continuous variation was applied to confirm the ZnP-H⋯C60 stoichiometry. Parabolic maximum at a molar ratio of 0.5 (Fig. S24 in the SI) was identified, which confirms a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry. Based on these results, we opted to use a 10-fold excess of C60 for all further experiments, to ensure that, according to the law of mass action, at least 90% of all photoexcited porphyrins are present as ZnP-H⋯C60, as depicted in Fig. 1.
1 stoichiometry. Based on these results, we opted to use a 10-fold excess of C60 for all further experiments, to ensure that, according to the law of mass action, at least 90% of all photoexcited porphyrins are present as ZnP-H⋯C60, as depicted in Fig. 1.
To identify the most stable configuration in solution, we performed DFT calculations considering two primary conformations (see SI for computational details). First, ZnP-H⋯C60 with both bridging protons residing on the porphyrin, forming a positively charged amidinium adjacent to a negatively charged carboxylate (η2-binding motif between the porphyrin amidine group and C60). Second, ZnP⋯H-C60, in which the bridging hydrogen atoms are positioned on either side, forming a charge-neutral amidine-carboxylic acid bridge. DFT predicts ZnP-H⋯C60 to be 4.31 kcal mol−1 more stable than in ZnP⋯H-C60 (see Fig. S29, S30, and Tables S1–S3 in the SI).
Pressure-dependent absorption and emission
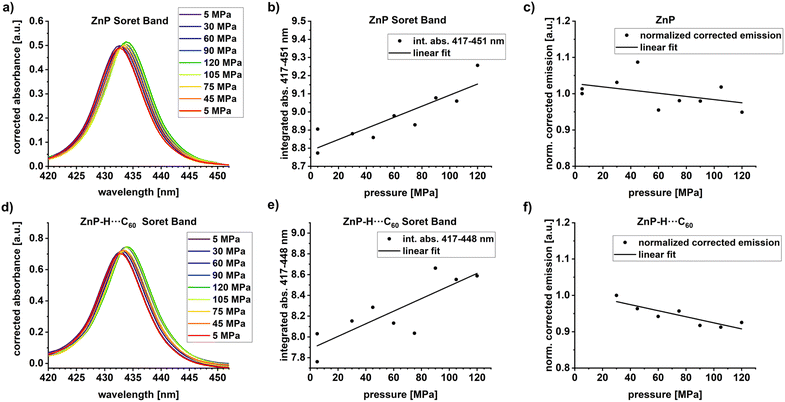
To evaluate the effect of pressure, we started our investigation with pressure dependent steady-state absorption assays.58 Our experimental setup used is described in the SI.49,58 We used 5 × 10−6 M ZnP in anisole and a 10-fold molar excess of C60. Each series of measurements was conducted in the pressurization and depressurization direction to check for the reversibility of the pressure effects. After each new pressure was set, the system was allowed to equilibrate for 15 minutes. All individual spectra were corrected by subtracting either the spectrum of the pure solvent in the ZnP experiments or that of C60 in the experiments with ZnP-H⋯C60. The full absorption spectrum under the given conditions is shown in Fig. S31 in the SI. Magnifications of the Soret-band absorptions of ZnP and ZnP-H⋯C60 are shown in Fig. 4a and d.Pressure induced two different effects on ZnP-H⋯C60 and ZnP. On the one hand, absorption increases as a function of pressure, and, on the other hand, a bathochromic shift from 432 to 434 nm is seen. Neither of these effects is, however, linked to any perturbations of the underlying equilibrium. Instead, we believe that they are caused by a volumetric compression and a density dependent analogue of the solvatochromic effect.59,60 Any volume reduction associated with increasing pressure leads to higher dielectric constants and bathochromic shifts of the S0–S2 transitions of ZnP.59,60 The low intensity/signal-to-noise ratio of the Q-band absorptions under our experimental conditions impedes any meaningful evaluation of a pressure-induced perturbation of the S0–S1 transition.
When analyzing the pressure dependent steady-state fluorescence data it is, however, crucial to consider the absorption intensification. The objective is to subtract any effects due to volumetric compression. We quantified the absorption intensification by linear fits (y = aK + bK·x) of the integrated Soret-band absorption. In doing so, we determined the compression factor K as described in eqn (6), with the highest applied pressures (pmax = 120 MPa) and lowest applied pressure (pmin = 5 MPa for ZnP, 30 MPa for ZnP-H⋯C60).
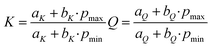 | (6) |
The compression factor K was used to calculate the fluorescence correction factor Ei for each individual measurement i as outlined in eqn (7).
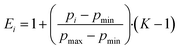 | (7) |
The fluorescence intensities I0,i, as obtained by integrating the measured fluorescence (Fig. S32 and S33 in the SI), were corrected by dividing them with the fluorescence correction factor Ei and gave the corrected fluorescence intensities Ii.
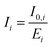 | (8) |
The pressure induced fluorescence quenching factor Q was then calculated from the linear fits (y = aQ + bQ·x) of Iiversus the applied pressure with the same procedure as used for the compression factor K, shown in eqn (6). All results for K and Q are listed in Table 1 and the corresponding plots are shown in Fig. 4.
| ZnP | ZnP-H⋯C60 | |
|---|---|---|
| K | 1.04 ± 0.008 | 1.09 ± 0.02 |
| Q | 0.95 ± 0.04 | 0.90 ± 0.03 |
The compression factor K = 1.04 for ZnP represents a 4% increase of the integrated Soret-band absorption area upon increasing the pressure from 5 to 120 MPa. Correspondingly a K of 1.09 for ZnP-H⋯C60 represents a 9% increase. The fluorescence intensity of ZnP at 120 MPa corresponded to 95% of the intensity at 5 MPa, which relates to lower fluorescence quantum yields; 4.0% at ambient pressure versus 3.8% at 120 MPa. The emission observed for ZnP-H⋯C60 was lowered to 90%. The slightly stronger pressure-induced fluorescence quenching in ZnP-H⋯C60 compared with ZnP will be revisited in the discussions for the activation volumes.
Statistical noise impedes direct data interpretation. At this point, noise-correction was deemed necessary to evaluate any pressure-induced effects. We employed a multi-Gaussian fitting to model the emission spectra. The cumulated fit peaks of the multi-Gaussian fitting – Fig. S35–S41 – resemble the noise-subtracted emission spectra. A closer examination of the normalized cumulative high-energy fluorescence fits reveals a pressure induced bathochromic shift of the 611.4 nm maximum at 30 MPa to 612.2 nm at 120 MPa (Fig. S42). In other words, the emissive S1–S0 transition is also subject to a pressure-induced bathochromic shift, albeit less pronounced than the absorptive S0–S2 transition.59,60
Transient absorption spectroscopy
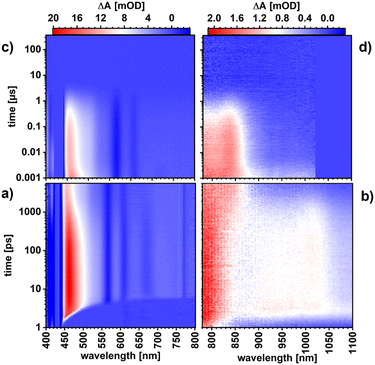
The excited-state characteristics of ZnP and ZnP-H⋯C60 were probed in anisole via femtosecond- and nanosecond transient absorption spectroscopy (fs-TAS and ns-TAS) involving photoexcitation into the Soret-band absorption at 430 nm. The fs-TAS and ns-TAS spectra of ZnP-H⋯C60 and ZnP are shown in Fig. 5 and S43 (in the SI), respectively. The model used for global or target analysis is shown in Fig. 6a.61 The reference experiments with ZnP were evaluated with global analysis based on a sequential four-species model without transitions k3 and k4.61 These transitions were required to fit the raw data of the ZnP-H⋯C60 in a branched target analysis model with five species.61 Corresponding evolution associated species (EAS) from global analysis of ZnP and the species associated spectra (SAS) for ZnP-H⋯C60, together with the time-resolved population profiles are shown in Fig. 6.61,62 | ||
| Fig. 6 (a) Analysis model used for evaluating the transient-absorption spectra of ZnP and ZnP-H⋯C60. General note: Soret-band excitation results in the initial population of S2. Modelling the S2–S1 internal conversion was necessary for a good fit. However, the long optical dispersion through the pressure cell does not allow to assign a clear EAS or SAS for S2.61 (b) Evolution associated spectra (EAS) from a four exponential global analysis of ZnP in anisole at 5 MPa.61 (c) Time profiles depicting the time-resolved population of the corresponding EAS of ZnP from global analysis.61,62 (d) Species associated spectra (SAS) from branched six exponential target analysis of ZnP-H⋯C60 in anisole at 5 MPa.61,62 (e) Time profiles depicting the time-resolved population of the corresponding SAS of ZnP-H⋯C60, from target analysis. A 1000-fold excess of DMAP vs. ZnP was used in all optical spectroscopy experiments.61,62 | ||
The short-lived second singlet excited state (S2) that is populated following Soret-band photoexcitation could not be resolved fully in our pressure dependent fs-TAS. The 3 ps optical dispersion caused by the aqueous medium in the pressure cell (Fig. 5) hampered its full deconvolution. Nevertheless, a fast decay corresponding to an S2–S1 internal conversion with a characteristic lifetime of 1.3 ps for ZnP and 1.2 ps for ZnP-H⋯C60 was required to describe the evolution of the singlet excited state (S1) (Fig. 6). Thus, the first resolved species following Soret-band photoexcitation corresponds to the ZnP-centered S1 state. The corresponding fingerprints are excited-state absorption (ESA) maxima at 464, 588, 639, and 707 nm, a shoulder at 535 nm next to ground state bleaching (GSB) and stimulated emission (SE) minima at 564, 614, and 668 nm (Fig. 6b). This state follows a relaxation process towards the solvated singlet excited state (S1,Solv.). S1,Solv. is discernible by a slight intensification of the 464 nm ESA maximum, which goes together with a reduction of the 464 nm full-width at half maximum (FWHM) from 53 to 50 nm. In the absence of an electron-accepting C60, S1,solv. undergoes intersystem crossing (ISC) to afford the triplet excited state (T1). The T1 fingerprints are a shoulder at 502 nm in the visible region and a new ESA maximum at 838 nm, while the former SE minimum at 668 nm is diminished. T1 deactivates to the ground state by means of oxygen quenching, as the pressure apparatus did not allow these experiments to be performed under deoxygenated conditions. When C60 is present, an additional deactivation pathway from S1,Solv. is present (Fig. 6d and e). It competes with ISC and is identified as charge-separation that affords a charge-separated state (CSS): formation of the radical ion pair state (ZnP)˙+⋯H⋯(C60)˙−. CSS is followed through the characteristic ESA of (ZnP)˙+ at 415 nm,57 while formation of C60˙– at 1050 nm is hampered by the optical absorption of water that was used as pressure medium in the pressure cell with the 6 cm optical path length.14,49
Both the lifetimes at 5 MPa and the activation volumes of the corresponding transitions are summarized in Table 2. The S1 lifetime is 3.8 ps, while for ZnP-H⋯C60 we observed a longer time of 7.3 ps. At this point, we are unable to explain the significantly longer lifetime of S1. S1,Solv features reveal comparable lifetimes of 1.80 ns in ZnP and 1.67 ns in ZnP-H⋯C60. The same applies for the subsequently formed T1, which decays with 1.61 µs or 1.82 µs, respectively. The S1,Solv state lifetime, which reflects the charge-separation to generate (ZnP)˙+⋯H⋯(C60)˙−, is much shorter with 634 ps. The lifetime of CSS is 25.1 ns, during which the singlet ground state (S0) is recovered by means of charge-recombination.
| Lifetimes | Activation volumes [cm3 mol−1] | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| System | S2 | S1 | S1,Solv. | CSS | S1,Solv. | T1 | S2–S1 | S1–S1,Solv. | S1,Solv.–CSS | CSS–S0 | S1,Solv.–T1 | T1–S0 |
| τ 1 [ps] | τ 2 [ps] | τ 3 [ps] | τ 4 [ns] | τ 5 [ns] | τ 6 [µs] | ΔVk1‡ | ΔVk2‡ | ΔVk3‡ | ΔVk4‡ | ΔVk5‡ | ΔVk6‡ | |
| ZnP | 1.3 | 3.8 | — | — | 1.80 | 1.61 | −3.1 ± 0.6 | +26 ± 3 | — | — | 0.0 ± 0.3 | −1.9 ± 1.0 |
| ZnP-H⋯C60 | 1.2 | 7.3 | 634 | 25.1 | 1.67 | 1.82 | −2.6 ± 0.6 | +12 ± 3 | −5.7 ± 2 | +8.6 ± 0.7 | −0.6 ± 1.4 | +0.5 ± 0.4 |
Activation volumes
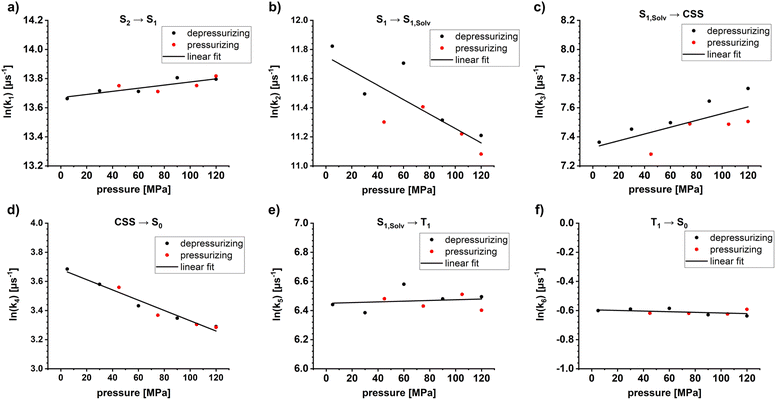
Insights into activation volumes ΔV‡ that correspond to these processes come from investigating the reaction kinetics k as a function of pressure p, as described by the Maxwell relation in eqn (9). We determined the activation volumes by performing fs-TAS and ns-TAS experiments under pressure. ΔV‡ values were calculated as described in the SI.49 The slopes of the ln(k)-p plots for ZnP-H⋯C60 are shown in Fig. 7, and those for ZnP in Fig. S48–S51. All relevant ΔV‡ values are listed in Table 2. The initial process in fs-TAS, the S2–S1 transition, is linked to a slightly negative ΔVk1‡ (Fig. 7a and S48 in the SI). However, the 3 ps optical dispersion failed to capture S2 following photoexcitation into the Soret-band absorption fully – vide supra. Consequently, an unambiguous interpretation of the effects observed is not possible. A positive ΔVk2‡ of more than +10 cm3 mol−1 (Fig. 7b and S49 in the SI) was determined for the subsequent solvent-relaxation process to yield S1,Solv.. This corresponds to a significant rate deceleration as the pressure is increased. We link this effect to the pressure-induced increase in solvent viscosity.59 The spectral features of S1 and S1,Solv. differ, however, only marginally (Fig. 6b and e). This challenges the interpretation of any observable pressure-induced effects. ISC from S1,Solv. to T1 is an activation volume-free process, with ΔVk5‡ values close to zero (Fig. 7e and S50 in the SI). It is porphyrin-centered and not subject to a change in charge density. As such, the intrinsic reorganization should be low and the influence on the solvation shell should be subtle. Deactivation to the ground state should also occur activation volume-free with ΔVk6‡ close to zero (Fig. 7f and S51 in the SI). Charge-separation in ZnP-H⋯C60 occurs with a negative ΔVk3‡ of −5.7 ± 2 cm3 mol−1, while charge-recombination has a comparable but positive ΔVk4‡ of +8.6 ± 0.7 cm3 mol−1 (Fig. 7c and d). A negative activation volume is a result of a pressure-induced acceleration of the charge-separation. This is in accordance with the pressure-induced fluorescence quenching seen for ZnP-H⋯C60 (Fig. 4f and Table 1). An accelerated charge-separation leaves a lower ratio of photoexcited ZnP to undergo fluorescent decay.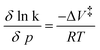 | (9) |
The following factors are considered to evaluate the mechanistic implications of ΔVk3‡ and ΔVk4‡. First, ZnP and C60 are rigid in nature.63 As such, significant bond-length alterations within these electron donors and acceptors are ruled out. A reorganization within the amidinium-carboxylate bridge in the activated state by means of, for example, a proton transfer would result in a neutralization of the charges and to weaker attractive forces across the bridge.24,41 One might expect, at this point, longer N⋯H⋯O bond lengths with positive activation volumes. Neutralization of charges across the amidinium-carboxylate bridge implies a lower dipole moment µd, which is well treated the Jung expression (eqn (5)), and which leads to a volume expansion of the solvation shell.46,51 This conflicts, however, with the negative activation volumes seen in the experiments for ΔVk3‡. Instead, the volume reduction en route to the CSS is likely associated with any of the two following factors. First, oxidation of the porphyrin is linked to a lower electron density at the Zn-center and, in turn, a higher affinity to DMAP as donor ligand. The net effect would be a ZnP-DMAP bond length contraction. Second, charge-separation is linked to the formation of the one-electron oxidized form of the porphyrin and the one-electron reduced form of C60. Considering that electrostrictive volume differences and radii of the dipolar moiety rd are inversely proportional to each other, both hypothetically possible reactions, that is, formation of charged ZnP and C60 and charge-neutralization within the amidinium-carboxylate bridge, contribute to the observable activation volumes. Oppositely charged ZnP and C60 create a dipole moment and, in turn, increase the electrostriction.46,51 Stronger interactions with the solvent dipole moments is the direct consequence and this leads to a volume compression following the Jung equation (eqn 5).46,51 If both charge-carriers are transferred in a single kinetic step, as, for example, via a PCET reaction the opposing volumetric effects will balance out and activation volumes will be negligible.49 Considering these contributions in concert, we conclude that the transition state of charge-separation across the amidinium-carboxylate bridge is dictated exclusively by electron transfer. Herby, the charge-separated state (ZnP)˙+⋯H⋯(C60)˙− is formed without any detectable volumetric contributions stemming from a proton transfer across the amidinium-carboxylate bridge.
The same arguments are applicable for the charge-recombination. Any increase in electron-density at the ZnP-core leads to a lower DMAP affinity and to a ZnP-DMAP bond elongation. Additionally, the neutralization of the opposite charges at the ZnP and C60, which is one-electron oxidized and reduced forms, lowers the previous electrostriction and interactions with the solvent dipole moments as described by the Jung equation.46,51 Both effects result in the observable positive activation volume ΔVk4‡. Consequently, effects from a transfer of electrons also govern the activated state as the bottleneck in charge-recombination. Volumetric contributions from a proton transfer remain non-observable.
Summary
A supramolecular electron donor–acceptor system, consisting of trimesitylene-phenylamidine-zinc porphyrin (ZnP) as electron-donor and noctyl-fulleropyrrolidine-benzoic acid (C60) as electron-acceptor, was synthesized. Studies necessitated a 1000-fold excess of DMAP to avoid intermolecular ZnP π-stacking. DFT calculations suggest that these building blocks form a charged η2-binding motif in their ground state, in which both bridging H-atoms are located at ZnP.Steady-state experiments unveiled a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry with a binding constant of 1.9 × 105 M−1. In the steady-state absorption experiments, two pressure-induced effects were seen. On one hand, an increase in optical density is linked to a volume compression. On the other hand, a bathochromic shift of the absorption is due to an increasing dielectric constant. Fluorescence experiments similarly showed a subtle bathochromic shift together with a small pressure-induced quenching for ZnP-H⋯C60 compared to ZnP.
1 stoichiometry with a binding constant of 1.9 × 105 M−1. In the steady-state absorption experiments, two pressure-induced effects were seen. On one hand, an increase in optical density is linked to a volume compression. On the other hand, a bathochromic shift of the absorption is due to an increasing dielectric constant. Fluorescence experiments similarly showed a subtle bathochromic shift together with a small pressure-induced quenching for ZnP-H⋯C60 compared to ZnP.
Pressure-dependent fs-TAS and ns-TAS were carried out to investigate the activation volumes (ΔV‡) of the excited-state deactivation in ZnP-H⋯C60. S2, which was populated through photoexcitation into the Soret-band absorption, could not be fully registered due to the 3 ps optical dispersion. Transition from S1 towards S1,Solv has a highly positive activation volume. The underlying rate attenuation is linked to a pressure induced viscosity increase. ISC to yield T1 and subsequent ground-state deactivation to S0 are free of any molecular reorganization and the corresponding activation volumes are essentially zero. Charge-separation is linked to a negative activation volume of −5.7 ± 2 cm3 mol−1. Charge-recombination has a comparable, but positive activation volume of +8.6 ± 0.7 cm3 mol−1.
Pressure-dependent reaction kinetics were used to investigate intrinsic as well as electrostrictive volume effects for charge-separation and -recombination in a ZnP-H⋯C60 system across an amidinium-carboxylate bridge, by using the Jung equation for dipolar molecules. Volumetric effects in the activated states for either charge-separation or -recombination are mostly determined by electron transfer. Volumetric contributions from a proton movement are non-observable. Consequently, both activation volumes are determined by electron-transfer processes, while effects induced by a proton-transfer during charge-separation and -recombination are absent.
Our presented work highlights the possibility of using pressure-dependent reactions kinetics to investigate the intrinsic and electrostrictive volume effects in pre-organized electron donor–acceptor systems. This extends our previously reported scope of high-pressure kinetics for studying charge-separation and -recombination reactions based on the Drude–Nernst theory for freely-dissolved reactants to covalently/non-covalently pre-organized electron donor–acceptor systems forming dipolar zwitterions as intermediates, based on the Jung theory.49–51
Conclusions
The activation volumes of the observable electron transfer between ZnP and C60 across an amidinium-carboxylate bridge occur without any volumetric contributions from a proton transfer within the bridging unit towards the formation of the respective activated states. Congruently, the transition states of charge-separation and -recombination are determined by contributions from electron rather than proton transfer.Author contributions
D. L. conducted complexation experiments by means of steady-state absorption and emission spectroscopy. D. L. conducted pressure dependent experiments by means of steady-state absorption, emission, fs- and ns-TA experiments. D. L. main-authored the manuscript. R. W., M. K., C. M. and T. C. contributed with theoretical calculations. R. W. co-authored the manuscript. M. U. synthesized the investigated compounds and characterized them with NMR, MALDI-TOF, ESI-TOF, UV-vis and fluorescence. M. U. co-authored the manuscript. A. Z. contributed with technical support for the pressure equipment. T. T. and D. M. G conceived the project and co-authored the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Experimental data for this article, including ambient pressure steady state absorption and emission, pressure-dependent steady state absorption and emission, and pressure-dependent transient absorption data are available at Zenodo at https://doi.org/10.5281/zenodo.18648422. Calculation data for this article is available at Zenodo at https://doi.org/10.5281/zenodo.19388636. Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc00291a.Acknowledgements
We thank financial support from the Spanish MICIU/AEI/10.13039/501100011033/FEDER, UE (PID2023-151167NB-I00), the Comunidad de Madrid and the Spanish State through the Recovery, Transformation and Resilience Plan [“Materiales Disruptivos Bidimensionales (2D)” (MAD2D-CM) (UAM1)-MRR Materiales Avanzados], and the European Union through the Next Generation EU funds. IMDEA Nanociencia is appreciative of support from the “Severo Ochoa” Programme for Centers of Excellence in R&D (SEV2016-0686, CEX2020-001039 S). T.T. acknowledges the Alexander von Humboldt Foundation (Germany) for the A. v. Humboldt – J. C. Mutis Research Award 2023 (ref. 3.3-1231125-ESP-GSA). D. M. G. acknowledges the Bavarian initiative “Solar Technologies go Hybrid” for support.References
- M. Y. Okamura and G. Feher, Annu. Rev. Biochem., 1992, 61, 861–896 CrossRef CAS PubMed.
- B. A. Barry and G. T. Babcock, Proc. Natl. Acad. Sci. U. S. A., 1987, 84, 7099–7103 Search PubMed.
- B. Kok, B. Forbush and M. McGloin, Photochem. Photobiol., 1970, 11, 457–475 CrossRef CAS PubMed.
- M. K. Chan, J. Kim and D. C. Rees, Science, 1993, 260, 792–794 CrossRef CAS PubMed.
- L. C. Seefeldt, B. M. Hoffman, J. W. Peters, S. Raugei, D. N. Beratan, E. Antony and D. R. Dean, Acc. Chem. Res., 2018, 51, 2179–2186 CrossRef CAS PubMed.
- D. G. Nocera, Acc. Chem. Res., 2012, 45, 767–776 CrossRef CAS PubMed.
- J. Barber, Chem. Soc. Rev., 2009, 38, 185–196 RSC.
- S. J. Mora, E. Odella, G. F. Moore, D. Gust, T. A. Moore and A. L. Moore, Acc. Chem. Res., 2018, 51, 445–453 CrossRef CAS PubMed.
- D. K. Dogutan and D. G. Nocera, Acc. Chem. Res., 2019, 52, 3143–3148 CrossRef CAS PubMed.
- (a) H. Imahori, M. Akiyama and J. Porphyr, Phthalocyanines, 2024, 28, 319–337 CrossRef CAS; (b) A. Cadranel, P. Haines, R. Kaur, A. Menon, P. W. Münich, P. R. Schol and D. M. Guldi, Adv. Energy Mater., 2021, 11, 2002831 CrossRef CAS.
- G. Bottari, G. de la Torre, D. M. Guldi and T. Torres, Coord. Chem. Rev., 2021, 428, 213605 CrossRef CAS.
- (a) H. Imahori and S. Fukuzumi, Adv. Funct. Mater., 2004, 14, 525–536 CrossRef CAS; (b) M. Rudolf, S. V. Kirner and D. M. Guldi, Chem. Soc. Rev., 2016, 45, 612–630 RSC.
- P. A. Liddell, J. P. Sumida, A. N. Macpherson, L. Noss, G. R. Seely, K. N. Clark, A. L. Moore, T. A. Moore and D. Gust, Photochem. Photobiol., 1994, 60, 537–541 CrossRef CAS.
- (a) D. Kuciauskas, S. Lin, G. R. Seely, A. L. Moore, T. A. Moore, D. Gust, T. Drovetskaya, C. A. Reed and P. D. W. Boyd, J. Phys. Chem., 1996, 100, 15926–15932 CrossRef CAS; (b) R. Kaur, F. Possanza, F. Limosani, S. Bauroth, R. Zanoni, T. Clark, G. Arrigoni, P. Tagliatesta and D. M. Guldi, J. Am. Chem. Soc., 2020, 142, 7898–7911 CrossRef CAS PubMed; (c) T. Higashino, T. Yamada, M. Yamamoto, A. Furube, N. V. Tkachenko, T. Miura, Y. Kobori, R. Jono, K. Yamashita and H. Imahori, Angew. Chem., Int. Ed., 2016, 55, 629–633 CrossRef CAS PubMed; (d) G. de la Torre, F. Giacalone, J. L. Segura, N. Martín and D. M. Guldi, Chem. Eur. J., 2005, 11, 1267–1280 CrossRef CAS PubMed.
- P. D. W. Boyd, M. C. Hodgson, C. E. F. Rickard, A. G. Oliver, L. Chaker, P. J. Brothers, R. D. Bolskar, F. S. Tham and C. A. Reed, J. Am. Chem. Soc., 1999, 121, 10487–10495 CrossRef CAS.
- (a) E. M. Pérez and N. Martín, Chem. Soc. Rev., 2015, 44, 6425–6433 RSC; (b) P. Haines, R. Kaur, M. M. Martin, M. B. Minameyer, S. Frühwald, S. Bönisch, D. Lungerich, F. Hampel, A. Görling, T. Drewello, N. Jux and D. M. Guldi, Adv. Energy Mater., 2021, 11, 2100158 CrossRef CAS.
- (a) F. D'Souza, R. Chitta, S. Gadde, M. E. Zandler, A. L. McCarty, A. S. D. Sandanayaka, Y. Araki and O. Ito, Chem. Eur. J., 2005, 11, 4416–4428 CrossRef PubMed; (b) R. Kaur, S. Sen, M. C. Larsen, L. Tavares, J. Kjelstrup-Hansen, M. Ishida, A. Zieleniewska, V. M. Lynch, S. Bähring, D. M. Guldi, J. L. Sessler and A. Jana, J. Am. Chem. Soc., 2020, 142, 11497–11505 CrossRef CAS PubMed.
- (a) Y. Shi, K. Cai, H. Xiao, Z. Liu, J. Zhou, D. Shen, Y. Qiu, Q.-H. Guo, C. Stern, M. R. Wasilewski, F. Diederich, W. A. Goddard III and J. F. Stoddart, J. Am. Chem. Soc., 2018, 140, 13835–13842 CrossRef CAS PubMed; (b) G. Zango, M. Krug, S. Krishna, V. Mariñas, T. Clark, V. Martinez-Diaz, D. M. Guldi and T. Torres, Chem. Sci., 2020, 11, 3448–3459 RSC.
- (a) E. Anaya-Plaza, J. Joseph, S. Bauroth, M. Wagner, C. Dolle, M. Sekita, F. Gröhn, E. Spiecker, T. Clark, A. de la Escosura, D. M. Guldi and T. Torres, Angew. Chem., Int. Ed., 2020, 59, 18786–18794 CrossRef CAS PubMed; (b) Angew. Chem. 2020, 132, 18946–18955 Search PubMed.
- I. Ortmans, B. Elias, J. M. Kelly, C. Moucheron and A.-K. DeMesmaeker, Dalton Trans., 2004, 668–676 RSC.
- F. Wessendorf, J.-F. Gnichwitz, G. H. Sarova, K. Hager, U. Hartnagel, D. M. Guldi and A. Hirsch, J. Am. Chem. Soc., 2007, 129, 16057–16071 CrossRef CAS PubMed.
- F. Wessendorf, B. Grimm, D. M. Guldi and A. Hirsch, J. Am. Chem. Soc., 2010, 132, 10786–10795 CrossRef CAS PubMed.
- A. Pannwitz and O. S. Wenger, Dalton Trans., 2019, 48, 5861–5868 RSC.
- J. A. Roberts, J. P. Kirby and D. G. Nocera, J. Am. Chem. Soc., 1995, 117, 8051–8052 CrossRef CAS.
- J. P. Kirby, J. A. Roberts and D. G. Nocera, J. Am. Chem. Soc., 1997, 119, 9230–9236 CrossRef CAS.
- Y. Deng, J. A. Roberts, S.-M. Peng, C. K. Chang and D. G. Nocera, Angew. Chem., Int. Ed., 1997, 36, 2124–2127 CrossRef CAS.
- (a) S. Vela, S. Bauroth, C. Atienza, A. Molina-Ontoria, D. M. Guldi and N. Martín, Angew. Chem., Int. Ed., 2016, 55, 15076–15080 CrossRef CAS PubMed; (b) S. Vela, S. Bauroth, C. Atienza, A. Molina-Ontoria, D. M. Guldi and N. Martín, Angew. Chem., 2016, 128, 15300–15304 CrossRef.
- G. T. Babcock and M. Wikström, Nature, 1992, 356, 301–309 CrossRef CAS PubMed.
- A. Amunts, O. Drory and N. Nelson, Nature, 2007, 447, 58–63 CrossRef CAS PubMed.
- B. Loll, J. Kern, W. Saenger, A. Zouni and J. Biesiadka, Nature, 2005, 438, 1040–1044 CrossRef CAS PubMed.
- F. Rappaport, M. Guergova-Kuras, P. J. Nixon, B. A. Diner and J. Lavergne, Biochemistry, 2002, 41, 8518–8527 CrossRef CAS PubMed.
- M. Raytchev, E. Mayer, N. Amann, H.-A. Wagenknecht and T. Fiebig, ChemPhysChem, 2004, 5, 706–712 CrossRef CAS PubMed.
- J. C. Lennox, D. A. Kurtz, T. Huang and J. L. Dempsey, ACS Energy Lett., 2017, 2, 1246–1256 CrossRef CAS.
- R. I. Cukier, J. Phys. Chem., 1994, 98, 2377–2381 CrossRef CAS.
- R. I. Cukier, J. Phys. Chem., 1995, 99, 16101–16115 CrossRef CAS.
- R. I. Cukier, J. Phys. Chem., 1996, 100, 15428–15443 CrossRef CAS.
- J.-Y. Fang and S. Hammes-Schiffer, J. Chem. Phys., 1997, 106, 8442–8454 CrossRef CAS.
- I. Rostov and S. Hammes-Schiffer, J. Chem. Phys., 2001, 115, 285–296 CrossRef CAS.
- N. H. Damrauer, J. M. Hodgekiss, J. Rosenthal and D. G. Nocera, J. Phys. Chem. B, 2004, 108, 6315–6321 CrossRef CAS PubMed.
- J. M. Hodgkiss, N. H. Damrauer, S. Pressé, J. Rosenthal and D. G. Nocera, J. Phys. Chem. B, 2006, 110, 18853–18858 CrossRef CAS PubMed.
- E. R. Young, J. Rosenthal and D. G. Nocera, Chem. Commun., 2008, 2322–2324 RSC.
- A. Pressé and R. Silbey, J. Chem. Phys., 2006, 124, 164504 CrossRef PubMed.
- (a) S. Y. Reece, J. M. Hodgkiss, J. Stubbe and D. G. Nocera, Phil. Trans. R. Soc. B, 2006, 361, 1351–1364 CrossRef CAS PubMed; (b) S. Y. Reece and D. G. Nocera, Annu. Rev. Biochem., 2009, 78, 673–699 CrossRef CAS PubMed.
- R. van Eldik, Angew. Chem., Int. Ed., 1986, 25, 673–682 CrossRef.
- D. Sarauli, R. Meier, G.-F. Liu, I. Ivanović-Burmazović and R. van Eldik, Inorg. Chem., 2005, 44, 7624–7633 CrossRef CAS PubMed.
- Y. Marcus, Chem. Rev., 2011, 111, 2761–2783 CrossRef CAS PubMed.
- (a) R. van Eldik, T. Asano and W. J. le Noble, Chem. Rev., 1989, 89, 549–688 CrossRef CAS; (b) A. Drljaca, C. D. Hubbard, R. van Eldik, T. Asano, M. V. Basilevsky and W. J. le Noble, Chem. Rev., 1998, 98, 2167–2290 CrossRef CAS PubMed.
- T. Liu, R. Tyburski, S. Wang, R. Fernández-Terán, S. Ott and L. Hammarström, J. Am. Chem. Soc., 2019, 141, 17245–17259 CrossRef CAS PubMed.
- D. Langford, R. Rohr, S. Bauroth, A. Zahl, A. Franke, I. Ivanović-Burmazović and D. M. Guldi, Nat. Chem., 2025, 17, 847–855 CrossRef CAS PubMed.
- P. Drude and W. Nernst, Z. Phys. Chem., 1894,(15U), 79–85 CrossRef.
- G. Jung, Z. Phys. Chem., 1929, 3B, 204–208 CrossRef.
- S. A. Vail, D. I. Schuster, D. M. Guldi, M. Isosomppi, N. Tkachenko, H. Lemmetyinen, A. Palkar, L. Echegoyen, X. Chen and J. Z. H. Zhang, J. Phys. Chem. B, 2006, 110, 14155–14166 CrossRef CAS PubMed.
- D. M. Martino and H. van Willigen, J. Phys. Chem. A, 2000, 104, 10701–10707 CrossRef CAS.
- A. Osuka, R. Yoneshima, H. Shiratori, T. Okada, S. Taniguchi and N. Mataga, Chem. Commun., 1998, 1567–1568 RSC.
- L. Sánchez, M. Sierra, N. Martín, A. J. Myles, T. J. Dale, J. Rebek Jr, W. Seitz and D. M. Guldi, Angew. Chem., Int. Ed., 2006, 45, 4637–4641 CrossRef PubMed.
- X. Ren, X. Wang, Y. Sun, X. Chi, D. Mangel, H. Wang and S. L. Sessler, Org. Chem. Front., 2019, 6, 584–590 RSC.
- J. Joseph, S. Bauroth, A. Charisiadis, G. Charalambidis, A. G. Coutsolelos and D. M. Guldi, Nanoscale, 2022, 14, 9304–9312 RSC.
- (a) The pressure apparatus was originally developed by the van Eldik group. Its construction details are described in an earlier article. Pictures of the apparatus and details of the measurement procedure are provided in the SI.; (b) M. Spitzer, F. Gartig and R. van Eldik, Rev. Sci. Instrum., 1988, 59, 2092–2093 CrossRef CAS.
- J. F. Skinner, E. L. Cussler and R. M. Fuoss, J. Phys. Chem., 1968, 72, 1057–1064 CrossRef CAS.
- T. G. Politis and H. G. Drickamer, J. Chem. Phys., 1982, 76, 285–291 CrossRef CAS.
- J. J. Snellenburg, S. Laptenok, R. Seger, K. M. Mullen and I. H. M. van Stokkum, J. Stat. Softw., 2012, 49, 1–22 Search PubMed.
- The quality of fit is represented by the residual traces given in Figures S44 – S47 in the SI. The noise is statistical without any distinguishable residual kinetic trace.
- D. M. Guldi, Chem. Soc. Rev., 2002, 31, 22–36 RSC.
| This journal is © The Royal Society of Chemistry 2026 |