Open Access Article
Open Access ArticleDimensional engineering in chiral layered hybrid perovskites for high-anisotropy self-powered circularly polarized light detection
Wan Luoab,
Qianxi Wanga,
Xiaoqi Lia,
Fen Zhanga,
Yan Fuab,
Yikun Fuab,
Xiaoyu Zhangab,
Guifu Zhanga,
Xitao Liu *abcd and
Junhua Luo
*abcd and
Junhua Luo bcd
bcd
aState Key Laboratory of Functional Crystals and Devices, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences Fuzhou, Fujian 350002, P.R. China. E-mail: xtliu@fjirsm.ac.cn
bCollege of Chemistry and Materials Science, Fujian Normal University, Fuzhou, Fujian 350007, P.R. China
cUniversity of Chinese Academy of Sciences, Beijing 100049, P.R. China
dFujian College, University of Chinese Academy of Sciences, Fuzhou, Fujian 350002, P.R. China
First published on 24th April 2026
Abstract
Chiral perovskites have emerged as a highly promising family of materials for circularly polarized light (CPL) detection, owing to their unique combination of structural chirality and remarkable optoelectronic performance. However, breaking the linear scaling law between chiroptical activity and intrinsic conductivity with dimensionality remains a substantial challenge toward high-performance CPL detection. Herein, through dimensional engineering involving the incorporation of a large cage cation and a chiral bifunctional bulky cation, a series of chiral layered hybrid perovskites (R/S-BrBA)2EAn−1PbnBr3n+1 (n = 1 to 3; R/S-BrBA+: 3-amino-1-bromobutanium; EA+: ethylammonium) has been successfully constructed. By incorporating oversized EA+ within the layered perovskite lattices, two new pairs of multilayered hybrid perovskites (R/S-BrBA)2EAPb2Br7 (2R/S) and (R/S-BrBA)2EA2Pb3Br10 (3R/S) have been synthesized, which exhibit remarkable semiconducting properties, including small optical absorption edges (2.79 and 2.69 eV) and high photoconductive on/off ratio (>102 and 103). Strikingly, cooperatively driven by the large cation–induced lattice expansion and bifunctional cation–introduced halogen⋯halogen interaction, the chirality transfer from the organic to inorganic sublattices increases, and the chiroptical activity with an asymmetric factor was enhanced by 5.7 times as the n value increased, breaking the linear scaling law. Benefiting from the dimensional engineering, exceptional self-powered CPL detection with an anisotropy factor (gIph) of up to 0.278 has been achieved in a photoelectric device fabricated with 3R single crystals. This study provides a pathway for the development of chiral perovskites that integrate high chiroptical activity and remarkable intrinsic conductivity, thereby enabling high-anisotropy self-powered CPL detection.
Introduction
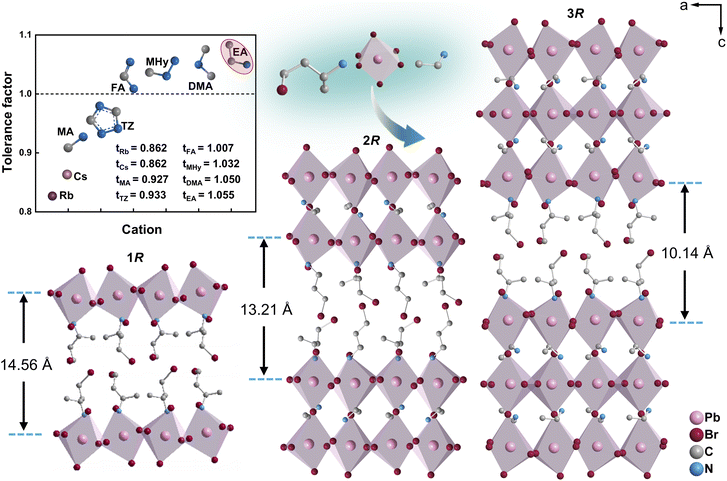
Circularly polarized light (CPL) detection, based on a selective response to the intrinsic handedness of chiral light, is attracting increasing attention for its extensive utilization in diverse applications, including remote sensing, optical sensing, biological imaging, and many other fields.1–4 Compared with conventional achiral substances, chiral semiconductors that possess intrinsic chiroptical anisotropy present a distinct advantage in the direct discrimination of CPL without requiring external optical components such as polarizers or wave plates.5,6 Particularly, the straightforward operation and high detection efficiency of chiral semiconductor devices provide a more efficient and convenient path to realizing CPL detection.7,8 Since the chiroptical properties of chiral hybrid perovskites were unveiled, these extraordinary materials, featuring a unique combination of structural chirality and remarkable optoelectronic performance, have emerged as a fascinating family of chiral semiconductors for CPL detection.9–16 For example, Chen et al. developed a chiral hybrid perovskite R/S-α-PEAPbI3 (PEA+: phenylethylammonium) by incorporating a chiral cation into the perovskite lattice, which facilitated efficient direct CPL detection with a responsivity of 797 mA W−1.17 Despite their tunable band gaps, exceptional optical characteristics, and distinctive low-dimensional architectures, chiral hybrid perovskites still suffer from limited semiconducting properties.18,19Over the past decade, substantial efforts have been devoted to exploring appropriate chiral hybrid perovskites with enhanced intrinsic conductivity for CPL detection through dimensional engineering.20–24 Among them, the chiral layered hybrid perovskites, especially those featuring multilayered architectures, have shown promising potential for CPL detection due to their prominent semiconducting properties, including high carrier mobility, long carrier diffusion length, and suitable bandgaps.15,25,26 As a result, a series of multilayered chiral perovskites have been successfully developed, such as (R/S-MPEA)2MAn−1PbnI3n+1 (n = 1 to 3, MPEA+: methylphenylethylammonium; MA+: methylammonium), (R/S-NEA)2(MA)2Pb3I10 (NEA+: naphthylethylammonium).13,27 However, despite remarkable advancements in the intrinsic conductivity achieved through increasing the inorganic sublattice layer number (n), this enhancement often comes at the expense of a diminished chiroptical response and a reduced dissymmetry factor, impairing the sensitivity and efficacy of CPL detection.28,29 Therefore, overcoming the linear scaling law between chiroptical activity and conductivity, which dictates that an increase in dimensionality leads to higher conductivity but lower chiroptical activity, remains a challenging task in multilayered chiral perovskites.30–36 It is well established that non-covalent interactions, including hydrogen bonding, π–π stacking, and halogen⋯halogen bonding, play a significant role in chiral transfer between the inorganic sublattice and the chiral organic sublattice.37,38 For example, Jooho Moon and others enhanced the chirality transfer by inducing inorganic framework distortion via the introduction of intermolecular halogen⋯halogen bonds.39 Meanwhile, oversized organic cations, such as ethylammonium (EA+), dimethylammonium (DMA+), and guanidinium (GA+), with their larger radii, are expected to enhance the lattice distortion and generate large symmetry breaking and chirality.40 Inspired by these results, the incorporation of bifunctional bulky cations with non-covalent interactions in multilayered chiral perovskites is expected to provide an exciting pathway to develop appropriate chiral hybrid perovskites for CPL detection.
Herein, we designed a series of chiral layered perovskites (R/S-BrBA)2EAn−1PbnBr3n+1 (n = 1 to 3; R/S-BrBA+: 3-amino-1-bromobutanium). Chiroptical activity measurements revealed that this series of materials breaks the scaling law where chiral optical activity linearly decreases with increasing perovskite layer number. The circular dichroism (CD) signal for (R/S-BrBA)2EA2Pb3Br10 (3R/S) is 5.7 times higher than that of its bilayered counterpart (R/S-BrBA)2EAPb2Br7 (2R/S). This phenomenon arises from the distortion index of the outermost inorganic framework, generating stronger local electric field gradients. Benefiting from the enhanced semiconductor properties and chiroptical activity, exceptional self-powered CPL detection with an anisotropy factor up to 0.278 has been achieved in a device fabricated with 3R single crystals, validating a pathway for the development of multilayered chiral perovskites in CPL sensing.
Results and discussion
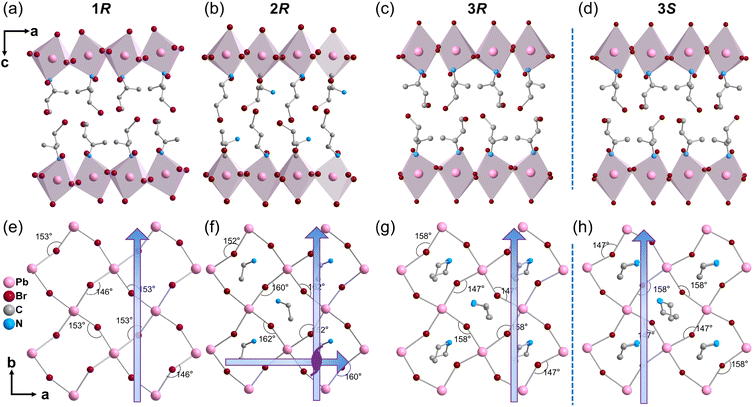
Multilayered perovskite architectures exhibit exceptionally efficient carrier transport, owing to synergistic band alignment and reduced interfacial recombination.41,42 The inset in Fig. 1 shows the tolerance factors for the interlayer cations commonly used in constructing multilayer perovskite. Current research predominantly focuses on chiral perovskites incorporating conventional small cations such as MA+, formamidinium (FA+), and cesium (Cs+).43,44 Herein, we synthesized the chiral monolayered perovskite (R/S-BrBA)2PbBr4 (1R/S) via the bifunctional chiral cation R/S-BrBA.45,46 To obtain multilayer perovskites with enhanced semiconducting properties, we tuned the number of structural layers and their optical activity and further introduced EA as a spacer cation, successfully constructing the quasi-two-dimensional structure 2R/S.Structural characterization revealed that 2R/S exhibits significantly reduced octahedral distortion between layers compared with 1R/S, which directly leads to a weaker CD signal. Subsequently, by precisely adjusting the stoichiometric ratio of lead acetate to EA+, we ultimately obtained chiral trilayered perovskites 3R/S with enhanced interfacial octahedral distortion, whose chiroptical response is markedly stronger than that of the 2R/S (Fig. 1). Bulk 3R/S single crystals with dimensions of 3 × 3 × 0.5 mm3 were successfully prepared by oriented insertion of bromine-substituted chiral R/S-BrBA cations into the [PbBr6]4− inorganic backbone using a controlled cooling process (Fig. S1). The constructed 1R/S, 2R/S, and 3R/S structures are Ruddlesden-Popper type hybrid perovskites with the chemical formula (R–NH3)2An−1MnX3n+1 (R–NH3: organic spacer cation; A: a small cation in the inorganic framework; M: metal cation; X: halide ion; n: layer number), which consists of alternating layers of organic cations and inorganic framework.47 Powder X-ray diffraction (XRD) patterns confirmed the phase purity of 1R/S, 2R/S, and 3R/S (Fig. S2). Single-crystal structures of 2R/S and 3R/S were collected. Further analysis through structural refinement confirmed the high phase purity and thermal stability of 3R/S, with a decomposition temperature reaching 513 K (Fig. S3). X-ray crystallography analysis indicates that 2R/S crystallizes in space group P212121, while the 1R/S and 3R/S compounds crystallize in polar space group P21. Structural analysis reveals an intriguing trend in octahedral distortion at the organic–inorganic interface: as the number of perovskite layers increases from n = 1 to n = 2, the degree of octahedral distortion decreases. However, when the layer number further increases to n = 3, the distortion of the interface octahedra actually increases. This distortion manifests as fluctuations in the Pb–Br bond length ranging from 2.817 to 3.29 Å and in the Br–Pb–Br bond angle ranging from 78.35° to 177.98° (Tables S1–S14). The average bond angle of the Pb–Br–Pb bond in the 1R structure is 149.5°, while it is 159° in the 2R structure and 152.8° in the 3R structure. The Pb–Br–Pb bond network in 3R exhibits helical distortion, resulting in greater spatial asymmetry, which may lead to enhanced chirality transfer (Fig. 2). This enhanced distortion reduces Br⋯Br distances (Fig. S4), thereby significantly strengthening halogen bonding interactions. The organic and inorganic layers were spatially arranged alternately, resulting in the construction of a natural quantum well structure. When the chiral perovskite transitions from n = 1 to n = 3, its interlayer spacing decreases from 14.56 Å to 10.14 Å (Fig. 1). The smaller interlayer spacing enhances electronic coupling between adjacent layers, thereby improving electron mobility and photovoltaic conversion efficiency.48 Additionally, the reduced interlayer spacing strengthens interlayer forces, such as van der Waals forces and hydrogen bonds, which help to enhance the material's thermal stability.49
Two fundamental prerequisites serve as crucial elements for achieving highly sensitive CPL detection: significant chiral light absorption and outstanding semiconductor performance. Table S15 presents some research data on the dimensionality, chiroptical activity, and conductivity of chiral perovskites. The optical properties of perovskites were evaluated using CD spectroscopy. As with other reported 2D chiral hybrid perovskites, CD signals of opposite signs are observed between 1R and 1S, 2R and 2S, and between 3R and 3S near the exciton absorption and interband transitions, respectively.23,50,51 The CD spectrum of 1R/S exhibits distinct peaks with opposite signs at 325 nm and 356 nm. The CD spectrum of 2R/S shows distinct peaks with opposite signs at 395 nm and 413 nm. In contrast, the CD signals of 3R/S are located at 407 nm and 430 nm, respectively, and they exhibit opposite signs at the corresponding wavelengths (Fig. 3a–c and S5). These distinct CD signals arise from the chiral-induced splitting of the degenerate energy states of the inorganic framework, modulated by the chiral organic cations. From the CD spectra, the dimensionless anisotropy factor (gCD) was extracted using eqn (1).
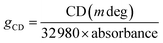 | (1) |
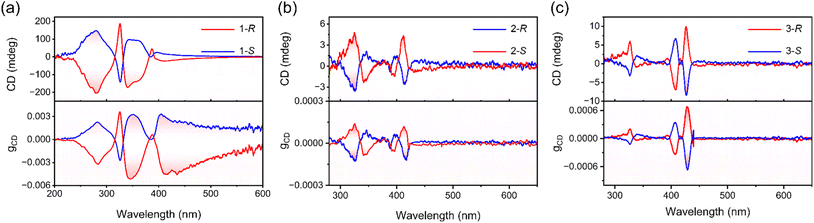 | ||
| Fig. 3 (a) Circular dichroism (CD) and gCD spectra of 1R and 1S films. (b) CD and gCD spectra of 2R and 2S films. (c) CD and gCD spectra of 3R and 3S films. | ||
The gCD of the 1R/S film at 325 nm is 3.3 × 10−3, and the gCD of the 2R/S film at 413 nm is 1.18 × 10−4, while the gCD of the 3R/S film at 430 nm reaches 6.83 × 10−4 (Fig. 3a–c). It is noteworthy that when the structure transitions from n = 2 to n = 3 configuration, the chirality typically weakens.28 However, the gCD of 3R/S is larger than that of 2R/S. This is because the chirality transfer efficiency of perovskites correlates with the degree of distortion in the inorganic framework.52,53 Calculations in this study indicate that the interfacial octahedra directly coordinated to the organic layer in 3R/S exhibit greater distortion. A distortion of the [PbBr6]4− octahedron was quantitatively evaluated by the octahedral band-length variance of Δd:
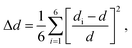 | (2) |
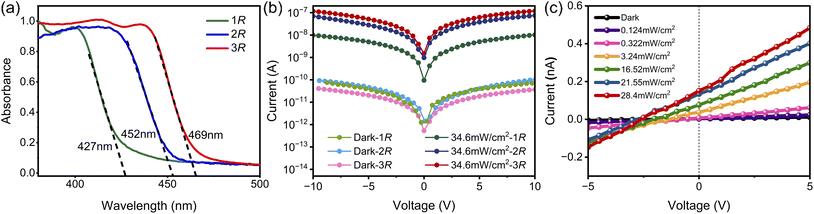
Furthermore, the reduced interlayer distance enhances electronic coupling across the inorganic layers, resulting in a narrow bandgap and consequently improved light absorption, which are beneficial semiconducting characteristics. The application of 2D hybrid perovskites in optoelectronic devices is influenced by their semiconductor and optical properties. Therefore, we further conducted ultraviolet-visible absorption and photoluminescence (PL) measurements to investigate their related properties. As shown in Fig. 4a, the UV-vis absorption spectra of 1R, 2R and 3R have obvious absorption cutoffs at 427 nm, 452 nm and 469 nm, respectively. The corresponding optical band gaps for 1R, 2R, and 3R were estimated from Tauc plots.58 Fig. S8 shows that the optical bandgap of 1R is 3.02 eV, while the optical bandgap of 2R and 3R are 2.79 eV and 2.69 eV, respectively. Their density functional theory calculated structures indicate that they are all direct-bandgap semiconductors. Taking 3R as an example to analyze the origin of their optical properties, the partial density of states profiles show that the valence band maximum is mainly from the Br-4p orbitals, while the conduction band minimum is mainly contributed by the Pb-6p states (Fig. S9–11). These results suggest that the inorganic composition mainly determines the energy band structure of the chiral halide perovskites 3R/S. Furthermore, the photoluminescence spectra of 1R, 2R and 3R exhibit emission peaks at 462 nm, 464 nm and 469 nm, respectively, under 369 nm excitation (Fig. S12). In addition, 1R shows a sub-bandgap emission peak at 590 nm and 2R at 530 nm. We attribute these peaks to self-trapped excitons.59–61 A photodetector based on large single crystals of 1R, 2R, and 3R, at an optical power of 34.6 mW cm−2, was observed to have a larger photocurrent response for 3R than for 2R and 1R, and a current switching ratio of 103 was achieved for 3R (Fig. 4b). The responsivity (R) and detection (D*) of the 3R device were 0.36 A mW−1 and 1.21 × 1013 Jones, respectively (Fig. S13).
The stability of hybrid perovskites significantly impacts their applications, including thermal stability and photocurrent reproducibility. Fig. S14 demonstrates that the powder XRD pattern of crystals stored for 30 days is consistent with the powder XRD pattern of freshly prepared crystals, indicating that 3R exhibits good phase stability. In addition, the stability of the 3R-based planar arrays was further investigated by comparing the I–V curves of fresh samples with those of samples that had been left for 30 days. After 30 days of exposure to air, the dark currents and photocurrents of the samples remained consistent with those of fresh devices (Fig. S15), and the photocurrent performance of 3R did not degrade after several cyclic switching tests (Fig. S16), further demonstrating the robust reproducibility of crystal device photocurrents. Moreover, 3R was found to exhibit an intrinsic bulk photovoltaic effect under zero external bias, delivering a photovoltage of 2.2 V along the b-axis (Fig. 4c and S17). Notably, this photovoltage exceeds values previously reported for layered hybrid perovskites, including (C4H9NH3)2(NH2CHNH2)Pb2Br7 (0.4 V),62 (CH3OC3H9N)2CsPb2Br7 (0.5 V),29 EA4Pb3Br10 (0.5 V),63 and (CPA)2FAPb2Br7 (0.25 V).64 This phenomenon suggests the existence of an inherent electric field and demonstrates that 3R is capable of detecting photons in a self-powered mode without requiring external bias. Conversely, the photovoltages along the a- and c-axes are nearly undetectable. The fact that photovoltage is observed only along the b-axis further validates that the photovoltaic effect is due to the chiral-polar nature.
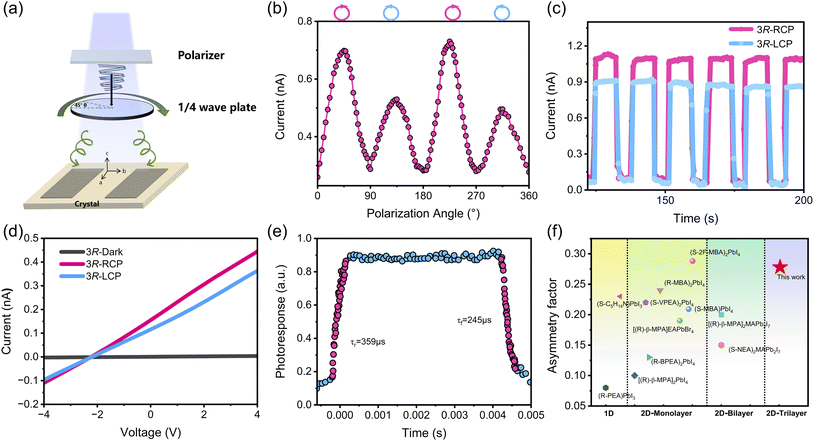
To evaluate the inherent CPL detection capability of 3R, Fig. 5a and S18 schematically depict a planar single-crystal device with Ag electrodes fabricated within the ab plane. That is, carriers will be transported along the b-axis of the polarization direction, leading to the chiral-polar photovoltaic effect when illuminated by CPL. This setup paves the way for further self-powered CPL detection. The device is illuminated with 405 nm polarized light, whose handedness is precisely controlled by a quarter-wave plate. As shown in Fig. 5b, under the same intensity of laser irradiation, the photocurrent of right-handed light (RCP) is much larger than that of left-handed (LCP) light, revealing a significant differentiation ability under RCP and LCP lasers (Vbias = 0 V). Meanwhile, the photocurrent remained almost constant under multiple CPL cycles (Fig. 5c). The device was confirmed to be stable and suitable for detecting CPL. Therefore, the discrimination between RCP and LCP was quantified via the photocurrent anisotropy factor gIph, which was derived from the following equation:
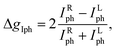 | (3) |
Where IphR and IphL denote the photocurrents under RCP and LCP irradiation. A gIph value of 0.278 was determined, which is an outstanding figure among multilayer chiral perovskite materials, even surpassing the current reported levels of chiral perovskite direct CPL detectors based on semiconductor materials such as [(R)-β-MPA]2MAPb2I7 (0.2),10 (R-α-PEA)2PbI4 (0.23),17 (R/S-MPA)2PbCl4 (0.1),65 and others.13,39,66–69 Some of the reported experimental gIph values for layered CPL detectors are summarized in Table S16 and Fig. 5f. As shown in Fig. 5d, the photocurrent varies with the excitation of RCP or LCP light, and there is still a 2.2 V bulk photovoltaic effect under RCP and LCP light. Under RCP light illumination at 10 V, the rise and decay times of the measurements were about 359 and 245 µs, respectively (Fig. 5e). This rapid response highlights the potential of 3R for use in high-speed detection systems. These benefits allow the well-engineered chiral polarized hybrid perovskite materials to exhibit significant promise in the realm of self-powered intelligent CPL detection.
Conclusions
In summary, this work systematically investigates the structure–property relationships in chiral perovskite single crystals, revealing that the trilayer (3R) configuration exhibits superior optoelectronic performance compared to the monolayer (1R) and bilayer (2R) counterparts. The 3R perovskite demonstrates remarkable photodetection capabilities with a current switching ratio of 103 and a significant bulk photovoltaic effect, generating 2.2 V photovoltage along the b-axis. These properties stem from the unique structural characteristics of 3R, where interfacial octahedral distortion (0.0023 vs. 0.0017 for 2R) and enhanced electronic coupling through halogen bridges facilitate efficient charge separation and transport. Furthermore, the material shows exceptional chiral optical properties with an anisotropy factor gCD of 6.83 × 10−4 at 430 nm, which is 5.7 times higher than that of 2R, enabling sensitive CPL detection. The fabricated self-powered CPL detector achieves an outstanding photocurrent anisotropy factor gIph of 0.278 with fast response times (rise/decay: 359/245 µs), outperforming existing chiral perovskite-based detectors. This work not only offers a new route for designing chiral perovskites but also shows their potential in making high-performance, self-powered optoelectronic devices. It also broadens the possibilities of multilayer chiral perovskites in self-powered CPL detection.Experimental
Materials
Material characterization and theoretical analysis
Powder XRD data were recorded on a Rigaku Miniflex 600 diffractometer (Cu Kα, 2θ = 5–40°, 0.5° min−1). Single-crystal XRD was collected on a SuperNova diffractometer (Mo Kα) and a Bruker D8 diffractometer (Mo Kα). The collected crystal data were refined using Olex2 software based on the F2 full matrix method to obtain the crystal structure. Optical bandgaps were collected at room temperature using a PerkinElmer Lambda-950 spectrophotometer with a spectral scan from 280 to 650 nm at room temperature. CD measurement samples were fabricated on quartz substrates via spin coating. Pre-dried 1R/S, 2R/S, and 3R/S crystals were ground into powder and separately dispersed in anhydrous DMF, and the reaction was conducted in a glove box purged with nitrogen. The resulting precursor solution was spin-coated onto cleaned quartz substrates and annealed on a hot plate, yielding square samples. Measurements were subsequently performed at room temperature using a Jasco J-1500 circular dichroism spectrometer. Photoelectric measurements were performed using planar electrodes. Two symmetrical silver electrodes were sputtered on a single crystal plane with dimensions of 1.0 × 1.0 × 0.3 mm3. The distance between the electrodes was ∼0.6 mm, and the thickness was ∼0.05 mm. I–V curves and I–t curves were measured using a Keithley 6517B electrometer. Under a 405 nm laser beam (THORLABS, LP405-MF300), pure RCP and LCP were obtained by rotating the angle of the quarter-wave plate (from 45 to 135°), respectively. The response times were measured by a high-speed oscilloscope (Tektronix MDO3014) at 10 Vbias voltage.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2469041 (R-BrBA)2EAPb2Br7, 2469438 (S-BrBA)2EA2Pb3Br10, 2520919 (R-BrBA)2EAPb2Br7 and 2520920 (S-BrBA)2EA2Pb2Br7 contain the supplementary crystallographic data for this paper.70a–dThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc00286b.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (52473283, 22193042, 22125110, 22435005), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB1170000), the Self-Deployed Key Project of State Key Laboratory of Functional Crystals and Devices (GNJT-2025-ZD01), the Natural Science Foundation of Fujian Province (2024J010037), and the Self-Deployment Project Research Program of Haixi Institutes, Chinese Academy of Sciences(CXZX-2023-JQ04).References
- Y. Yang, R. C. Da Costa, M. J. Fuchter and A. J. Campbell, Nat. Photonics, 2013, 7, 634–638 CrossRef CAS.
- Y. Dang, X. Liu, B. Cao and X. Tao, Matter, 2021, 4, 794–820 CrossRef CAS.
- L. Xu, X. Wang, W. Wang, M. Sun, W. J. Choi, J.-Y. Kim, C. Hao, S. Li, A. Qu, M. Lu, X. Wu, F. M. Colombari, W. R. Gomes, A. L. Blanco, A. F. De Moura, X. Guo, H. Kuang, N. A. Kotov and C. Xu, Nature, 2022, 601, 366–373 Search PubMed.
- Z. Xie, J. Deng, D. Liu, J. Lin, T. Jiang, X. Wang, W. Liu, L. Ma, F. Song, Z. Xiong, J. Chen, J. Zhang, C. Redshaw, Z. Zhao, X. Feng and B. Z. Tang, Chem. Sci., 2026, 17, 4145–4156 RSC.
- F. J. Rodríguez-Fortuño, N. Engheta, A. Martínez and A. V. Zayats, Nat. Commun., 2015, 6, 8799 CrossRef PubMed.
- J. Schmidt, A. Guggenmos, M. Hofstetter, S. H. Chew and U. Kleineberg, Opt. Express, 2015, 23, 33564 CrossRef CAS PubMed.
- Z. Zhang, J. Wu and H. Lu, Chem. Sci., 2024, 15, 20440–20447 RSC.
- M. Schulz, F. Balzer, D. Scheunemann, O. Arteaga, A. Lützen, S. C. J. Meskers and M. Schiek, Adv. Funct. Mater., 2019, 29, 1900684 CrossRef.
- Y. Zhao, Y. Qiu, J. Feng, J. Zhao, G. Chen, H. Gao, Y. Zhao, L. Jiang and Y. Wu, J. Am. Chem. Soc., 2021, 143, 8437–8445 CrossRef CAS PubMed.
- L. Wang, Y. Xue, M. Cui, Y. Huang, H. Xu, C. Qin, J. Yang, H. Dai and M. Yuan, Angew. Chem. Int. Ed., 2020, 59, 6442–6450 Search PubMed.
- S. You, P. Yu, T. Zhu, Q. Guan, J. Wu, H. Dai, H. Zhong, Z.-K. Zhu and J. Luo, Mater. Horiz., 2023, 10, 5307–5312 RSC.
- X. Shang, L. Wan, L. Wang, F. Gao and H. Li, J. Mater. Chem. C, 2022, 10, 2400–2410 RSC.
- T. Liu, W. Shi, W. Tang, Z. Liu, B. C. Schroeder, O. Fenwick and M. J. Fuchter, ACS Nano, 2022, 16, 2682–2689 CrossRef CAS PubMed.
- T. Zhu, H. Wu, C. Ji, X. Zhang, Y. Peng, Y. Yao, H. Ye, W. Weng, W. Lin and J. Luo, Adv. Opt. Mater., 2022, 10, 2200146 CrossRef CAS.
- W. Wu, X. Shang, Z. Xu, H. Ye, Y. Yao, X. Chen, M. Hong, J. Luo and L. Li, Adv. Sci., 2023, 10, 2206070 CrossRef CAS PubMed.
- X.-F. Luo, J. He, Y. Wang, H. Dai and Z.-G. Wu, Chin. J. Struct. Chem., 2022, 41, 70–79 Search PubMed.
- C. Chen, L. Gao, W. Gao, C. Ge, X. Du, Z. Li, Y. Yang, G. Niu and J. Tang, Nat. Commun., 2019, 10, 1927 CrossRef PubMed.
- L. Mao, C. C. Stoumpos and M. G. Kanatzidis, J. Am. Chem. Soc., 2019, 141, 1171–1190 CrossRef CAS PubMed.
- J. Hu, L. Yan and W. You, Adv. Mater., 2018, 30, 1802041 CrossRef PubMed.
- L. Cong and B.-B. Cui, Sci. Sin.: Chim., 2024, 54, 1194–1211 Search PubMed.
- L. Yang, Y. Gao, Z. Wang, L. Yang and M. Shao, Nat. Commun., 2025, 16, 4161 CrossRef CAS PubMed.
- J. Hu, X. Wen, D. Yang, Y. Chen, Z. Liu and D. Li, Nano Lett., 2024, 24, 1001–1008 CrossRef CAS PubMed.
- J. Ahn, E. Lee, J. Tan, W. Yang, B. Kim and J. Moon, Mater. Horiz., 2017, 4, 851–856 RSC.
- X. Dong, T. Chen, J. Liang, L. Wang, H. Wu, Z. Xu, J. Luo and L.-N. Li, Chin. J. Struct. Chem., 2024, 43, 100256 CAS.
- Z. Zhu, T. Zhu, S. You, P. Yu, J. Wu, Y. Zeng, Y. Jiang, X. Liu, L. Li, C. Ji and J. Luo, Adv. Sci., 2024, 11, 2307593 CrossRef CAS PubMed.
- D. Li, W. Wu, S. Han, X. Liu, Y. Peng, X. Li, L. Li, M. Hong and J. Luo, Chem. Sci., 2021, 12, 3050–3054 RSC.
- S. Ramakrishnan, Y. Dong, Y. Xie, J. L. Shelton, M. P. Hautzinger, D. Nguyen Minh, M. Taddei, X. Zhang, Y. Zhang, D. B. Mitzi, M. A. Haque, J. L. Blackburn, Q. Yu, M. C. Beard and J. M. Luther, J. Am. Chem. Soc., 2025, 147, 44457–44467 CrossRef CAS PubMed.
- G. Long, R. Sabatini, M. I. Saidaminov, G. Lakhwani, A. Rasmita, X. Liu, E. H. Sargent and W. Gao, Nat. Rev. Mater., 2020, 5, 423–439 CrossRef.
- C. Ji, Y. Li, X. Liu, Y. Wang, T. Zhu, Q. Chen, L. Li, S. Wang and J. Luo, Angew. Chem. Int. Ed., 2021, 60, 20970–20976 CrossRef CAS PubMed.
- R. Babu, J. E. Heger, T. Dutta, X. Hu, N. Pradhan, P. Müller-Buschbaum, S. Gómez-Graña and L. Polavarapu, ACS Energy Lett., 2025, 10, 5703–5721 CrossRef CAS PubMed.
- C. Coccia, M. Moroni and L. Malavasi, Molecules, 2023, 28, 6166 CrossRef CAS PubMed.
- A. Dučinskas, M. Jung, Y.-R. Wang, J. V. Milić, D. Moia, M. Grätzel and J. Maier, J. Mater. Chem. C, 2024, 12, 7909–7915 RSC.
- J. M. Hoffman, X. Che, S. Sidhik, X. Li, I. Hadar, J.-C. Blancon, H. Yamaguchi, M. Kepenekian, C. Katan, J. Even, C. C. Stoumpos, A. D. Mohite and M. G. Kanatzidis, J. Am. Chem. Soc., 2019, 141, 10661–10676 CrossRef CAS PubMed.
- C. Asker, C. Pipitone, F. Ursi, K. Chen, A. G. Ricciardulli, E. S. Suena Galindez, S. Luong, P. Samorì, M. Reece, A. Martorana, F. Giannici and O. Fenwick, J. Mater. Chem. A, 2025, 13, 26009–26018 RSC.
- G. Long, C. Jiang, R. Sabatini, Z. Yang, M. Wei, L. N. Quan, Q. Liang, A. Rasmita, M. Askerka, G. Walters, X. Gong, J. Xing, X. Wen, R. Quintero-Bermudez, H. Yuan, G. Xing, X. R. Wang, D. Song, O. Voznyy, M. Zhang, S. Hoogland, W. Gao, Q. Xiong and E. H. Sargent, Nat. Photonics, 2018, 12, 528–533 CrossRef CAS.
- Y.-H. Lee, G. M. Macam, R. A. B. Villaos, L.-Y. Feng, C.-H. Hsu, Z.-Q. Huang, D.-C. Wang, H. Lin and F.-C. Chuang, J. Phys. Chem. C, 2024, 128, 17091–17097 CrossRef CAS.
- J. Son, S. Ma, Y.-K. Jung, J. Tan, G. Jang, H. Lee, C. U. Lee, J. Lee, S. Moon, W. Jeong, A. Walsh and J. Moon, Nat. Commun., 2023, 14, 3124 CrossRef CAS PubMed.
- S. An and P. Xing, Chem. Soc. Rev., 2026, 55, 1460–1493 RSC.
- J. Son, G. Jang, S. Ma, H. Lee, C. U. Lee, S. Yang, J. Lee, S. Moon, W. Jeong, J. H. Park, J. Kim, D. H. Kim, J. Park and J. Moon, Adv. Funct. Mater., 2025, 35, 2413041 CrossRef CAS.
- Y. Fu, Adv. Mater., 2022, 34, 2108556 CrossRef CAS PubMed.
- D. Lin, L. Ma, W. Ni, C. Wang, F. Zhang, H. Dong, G. G. Gurzadyan and Z. Nie, J. Mater. Chem. A, 2020, 8, 25402–25410 RSC.
- J. Lu, W. Chen, C. Zhou, S. Yang, C. Wang, R. Wang, X. Wang, Z. Gan, B. Jia and X. Wen, J. Mater. Chem. C, 2021, 9, 8966–8974 RSC.
- X. Wang, X. Dong, H. Dong, Y. Zhou, C. Ran, L. Song, X. Liu, W. Xu, W. Huang and Z. Wu, ACS Nano, 2024, 18, 26327–26337 CAS.
- L. Tao, W. Tang, M. Yan, L. Ding, J. Wei, L. Wang, L. Li, L. Li, D. Yang and Y. Fang, J. Mater. Chem. C, 2023, 11, 12392–12399 RSC.
- J. Yu, M. Li, Y. Lv, B. Liao, G. Tan, J. Zou, X. Liu, Y. Han and L. Li, Adv. Funct. Mater., 2025, 36, e08904 CrossRef.
- T.-M. Guo, F.-F. Gao, Y.-J. Gong, Z.-G. Li, F. Wei, W. Li and X.-H. Bu, J. Am. Chem. Soc., 2023, 145, 22475–22482 CrossRef CAS PubMed.
- P. Liu, N. Han, W. Wang, R. Ran, W. Zhou and Z. Shao, Adv. Mater., 2021, 33, 2002582 CrossRef CAS PubMed.
- Y. Lu, Y. Zhang, C.-Y. Yang, S. Revuelta, H. Qi, C. Huang, W. Jin, Z. Li, V. Vega-Mayoral, Y. Liu, X. Huang, D. Pohl, M. Položij, S. Zhou, E. Cánovas, T. Heine, S. Fabiano, X. Feng and R. Dong, Nat. Commun., 2022, 13, 7240 CrossRef CAS PubMed.
- H. S. Choi, J. Lin, G. Wang, W. P. D. Wong, I.-H. Park, F. Lin, J. Yin, K. Leng, J. Lin and K. P. Loh, Science, 2024, 384, 60–66 CrossRef CAS PubMed.
- J. Ahn, S. Ma, J.-Y. Kim, J. Kyhm, W. Yang, J. A. Lim, N. A. Kotov and J. Moon, J. Am. Chem. Soc., 2020, 142, 4206–4212 CrossRef CAS PubMed.
- G. Long, C. Jiang, R. Sabatini, Z. Yang, M. Wei, L. N. Quan, Q. Liang, A. Rasmita, M. Askerka, G. Walters, X. Gong, J. Xing, X. Wen, R. Quintero-Bermudez, H. Yuan, G. Xing, X. R. Wang, D. Song, O. Voznyy, M. Zhang, S. Hoogland, W. Gao, Q. Xiong and E. H. Sargent, Nat. Photonics, 2018, 12, 528–533 CrossRef CAS.
- Y. Mao, S. Guo, X. Huang, K. Bu, Z. Li, P. Q. H. Nguyen, G. Liu, Q. Hu, D. Zhang, Y. Fu, W. Yang and X. Lü, J. Am. Chem. Soc., 2023, 145, 23842–23848 CrossRef CAS PubMed.
- J. Son, S. Ma, Y.-K. Jung, J. Tan, G. Jang, H. Lee, C. U. Lee, J. Lee, S. Moon, W. Jeong, A. Walsh and J. Moon, Nat. Commun., 2023, 14, 3124 CrossRef CAS PubMed.
- J. Fu, T. Bian, J. Yin, M. Feng, Q. Xu, Y. Wang and T. C. Sum, Nat. Commun., 2024, 15, 4562 CrossRef CAS PubMed.
- A. G. Doyle and E. N. Jacobsen, Chem. Rev., 2007, 107, 5713–5743 CrossRef CAS PubMed.
- S. An and P. Xing, Chem. Soc. Rev., 2026, 55, 1461–1493 RSC.
- Y. Liu, X. Zhang, X. Xu, J. Dai and Z. Quan, Angew. Chem., Int. Ed., 2025, 64, e202510579 CrossRef CAS PubMed.
- J. Tauc, in Optical properties of solids, ed. S. Nudelman and S. S. Mitra, Springer US, Boston, MA, 1969, pp. 123–136 Search PubMed.
- S. Guo, W. Mihalyi-Koch, Y. Mao, X. Li, K. Bu, H. Hong, M. P. Hautzinger, H. Luo, D. Wang, J. Gu, Y. Zhang, D. Zhang, Q. Hu, Y. Ding, W. Yang, Y. Fu, S. Jin and X. Lü, Nat. Commun., 2024, 15, 3001 CrossRef CAS PubMed.
- J.-C. Blancon, H. Tsai, W. Nie, C. C. Stoumpos, L. Pedesseau, C. Katan, M. Kepenekian, C. M. M. Soe, K. Appavoo, M. Y. Sfeir, S. Tretiak, P. M. Ajayan, M. G. Kanatzidis, J. Even, J. J. Crochet and A. D. Mohite, Science, 2017, 355, 1288–1292 CrossRef CAS PubMed.
- J. Tan, X. Jiang, D. Liu, Z.-K. Tang, E. V. Chulkov and A. S. Vasenko, J. Phys. Chem. Lett., 2025, 16, 7659–7665 CrossRef CAS PubMed.
- Y. Ma, J. Wang, Y. Liu, S. Han, Y. Li, Z. Xu, W. Guo, J. Luo, M. Hong and Z. Sun, J. Mater. Chem. C, 2021, 9, 881–887 RSC.
- S. Wang, X. Liu, L. Li, C. Ji, Z. Sun, Z. Wu, M. Hong and J. Luo, J. Am. Chem. Soc., 2019, 141, 7693–7697 CrossRef CAS PubMed.
- X. Li, F. Wu, Y. Yao, W. Wu, C. Ji, L. Li, Z. Sun, J. Luo and X. Liu, J. Am. Chem. Soc., 2022, 144, 14031–14036 CrossRef CAS PubMed.
- X. Zhang, W. Weng, L. Li, H. Wu, Y. Yao, Z. Wang, X. Liu, W. Lin and J. Luo, Small, 2021, 17, 2102884 Search PubMed.
- Y. Zhao, X. Li, J. Feng, J. Zhao, Y. Guo, M. Yuan, G. Chen, H. Gao, L. Jiang and Y. Wu, Giant, 2022, 9, 100086 Search PubMed.
- Y. Zhao, X. Yin, Z. Gu, M. Yuan, J. Ma, T. Li, L. Jiang, Y. Wu and Y. Song, Adv. Funct. Mater., 2023, 33, 2306199 CrossRef CAS.
- Z. Li, C. Ji, Y. Fan, T. Zhu, S. You, J. Wu, R. Li, Z.-K. Zhu, P. Yu, X. Kuang and J. Luo, J. Am. Chem. Soc., 2023, 145, 25134–25142 CrossRef CAS PubMed.
- Z. Liu, C. Zhang, X. Liu, A. Ren, Z. Zhou, C. Qiao, Y. Guan, Y. Fan, F. Hu and Y. S. Zhao, Adv. Sci., 2021, 8, 2102065 CrossRef CAS PubMed.
- (a) CCDC 2469041: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nw7h9; (b) CCDC 2469438: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2nwn9j; (c) CCDC 2520919: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qm6zk; (d) CCDC 2520920: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qm70m.
| This journal is © The Royal Society of Chemistry 2026 |