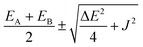Open Access Article
Open Access ArticleControlling triplet-pair formation in acene-bridged trimers through locally excited–charge-transfer state mixing†
Ebin
Sebastian
 a,
Daniel G.
Congrave‡
a,
Daniel G.
Congrave‡
 bc,
Jeroen
Royakkers‡
bc,
Jeroen
Royakkers‡
 b,
Stephanie
Montanaro
b,
Huaxi
Huang
d,
Ashish
Sharma
a,
Julia
Osmólska
b,
Stephanie
Montanaro
b,
Huaxi
Huang
d,
Ashish
Sharma
a,
Julia
Osmólska
 a,
He
Zhu
b,
Chanakarn
Phansa
a,
Jurjen
Winkel
a,
Oliver
Millington
a,
He
Zhu
b,
Chanakarn
Phansa
a,
Jurjen
Winkel
a,
Oliver
Millington
 b,
Murad J. Y.
Tayebjee
b,
Murad J. Y.
Tayebjee
 e,
Luis M.
Campos
e,
Luis M.
Campos
 d,
Hugo
Bronstein
d,
Hugo
Bronstein
 *ab and
Akshay
Rao
*ab and
Akshay
Rao
 *a
*a
aCavendish Laboratory, University of Cambridge, Cambridge, CB3 0HE, UK. E-mail: ar525@cam.ac.uk; hab60@cam.ac.uk
bYusuf Hamied Department of Chemistry, University of Cambridge, Cambridge, CB2 1EW, UK
cChemistry Research Labloratory, University of Oxford, Oxford, OX1 3TA, UK
dDepartment of Chemistry, Columbia University, New York, New York 10027, USA
eSchool of Photovoltaic and Renewable Energy Engineering, UNSW Sydney, Sydney, 2052, NSW, Australia
First published on 23rd March 2026
Abstract
Singlet fission (SF) offers a pathway to surpass conventional efficiency limits in photovoltaics, yet how bridge–chromophore coupling governs locally excited/charge-transfer state (LE–CT) mixing and thereby the multiexciton yield remains elusive. We design three systems, TIPS-BT0, TIPS-TAT, and Encap-TAT, that progressively increase bridge–chromophore coupling. Steady-state spectroscopy and transient absorption reveal that TIPS-BT0 and TIPS-TAT undergo efficient SF, forming correlated triplet pairs (1TT) and, in TIPS-TAT, long-lived triplets. By contrast, Encap-TAT loses vibronic structure and relaxes ultrafast into a broad, red-shifted intramolecular exciplex-like (Ex) state. Quantum chemical analysis shows that the Ex state originates from strong hybridization between local excitations (LEs) and bridge–chromophore CT configurations, driven by large electron and hole transfer integrals and a small LE–CT energy gap. Such strong LE–CT hybridisation suppresses well-defined 1TT formation, favouring intramolecular Ex emission and enhancing photoluminescence quantum yield (≃80.5% vs. ≃38.5% for TIPS-TAT). These findings establish a unifying framework in which the balance between multiexciton generation and exciplex emission in acene-bridged trimers is governed by the degree of LE–CT mixing. Tuning bridge energetics and LE–CT offsets enables deliberate routing of excited-state pathways toward efficient triplet production or bright exciplex emission, providing actionable design rules for next-generation singlet-fission and optoelectronic architectures.
Introduction
Singlet fission (SF) is a spin-conserving multiexciton generation process in which one singlet exciton (S1) splits into two triplet excitons (T1), potentially doubling the exciton yield per absorbed photon and enabling photovoltaics to transcend the Shockley–Queisser limit.1–5 Although crystalline acenes and their derivatives first showcased intermolecular SF, their limited exciton diffusion, aggregation, and heterogeneous packing hinder practical device integration.6–8 In contrast, covalent intramolecular SF (iSF) molecules and polymers have emerged as a powerful alternative, offering precise control over geometry, solution processability, and electronic coupling between chromophores (Fig. 1a).9–18A central challenge in iSF design lies in balancing electronic interactions. The chromophores must couple strongly enough to form a correlated triplet pair (1TT), yet remain sufficiently decoupled to allow triplet separation and avoid losses to excimer or charge-transfer (CT) traps.19–23 π-Conjugated bridges are therefore widely employed, not only to enforce distance and connectivity12,20,24–28 but also to actively tune the electronic landscape through resonance with chromophore frontier orbitals.29–31 Strategies spanning phenylene spacers, extended acenes, and macrocyclic frameworks have been explored to tailor molecular geometry, connectivity, and coupling (Fig. 1a).13,32–39 In particular, rigid macrocyclic/cyclophane platforms enable precise cofacial arrangements that tune electronic coupling, resulting in singlet fission with strongly geometry-dependent rates and, in some cases, long-lived multiexciton signatures.40–43
From a singlet-fission perspective, however, the role of CT-mediated interactions is nuanced. Moderate exciton–CT hybridisation can be advantageous, lowering the barrier to accessing CT-mediated 1TT configurations and facilitating spin–space decoherence required for triplet separation (Fig. 1b).26,34–36,44–46 Yet excessive CT admixture can destabilize the 1TT channel, funneling population into excimer-like manifolds that suppress free-triplet yield.47–50 Particularly intriguing are cases where CT states localized on the bridge–chromophore axis hybridize with the local excitation of the fission-active unit. Such mixing may open superexchange pathways into 1TT, or, if too strong, collapse the exciton into a delocalized LE–CT hybrid that bypasses multiexciton formation. Despite extensive efforts to tune electronic coupling through bridge design, the impact of bridge–chromophore resonance on LE–CT mixing and its consequences for singlet fission is not fully understood.
To address this gap, we employ a trimeric chromophore–bridge–chromophore scaffold as a minimal platform to tune LE–CT mixing while preserving a well-defined intramolecular SF motif on the terminal tetracenes. Relative to directly linked dimers, the bridge provides an independent handle over CT energetics and transfer integrals.35,51 It can also modulate the triplet–triplet exchange interaction that governs whether 1TT remains bound or separates into long-lived triplets. TIPS-anthracene (TIPS-A) was chosen as the bridge because its higher triplet energy avoids acting as a triplet sink or heterofission channel, while its conjugated framework enables systematic control of electronic communication and CT-mediated superexchange. We further introduce Encap-tetracene (Encap-T) to reduce LE–CT detuning and increase bridge–chromophore resonance while preventing aggregation and improving solubility.52 Accordingly, TIPS-TAT features a large CT–LE offset (ECT ≈ 3.05 eV; ELE ≈ 2.30 eV; ΔECT–LE = 0.75) and weak LE–CT mixing, whereas Encap-TAT has a smaller gap (ECT ≈ 2.85 eV; ELE ≈ 2.50 eV; ΔECT–LE = 0.35 eV) and stronger hybridisation. By mapping their optical signatures and fission dynamics, we show how increasing bridge resonance can either support long-lived triplets via CT-assisted superexchange or suppress iSF through exciplex-like trapping.
Results and discussion
Molecular synthesis and structures
To investigate how bridge–chromophore CT state alignment modulates the intramolecular singlet fission (iSF) landscape, we compared two trimeric scaffolds, TIPS-TAT and Encap-TAT (Fig. 2). Both architectures incorporate a TIPS-anthracene (TIPS-A) bridge; however, in Encap-TAT the local excited-state energy of the bridge is more closely aligned with that of the Encap-tetracene (Encap-T) chromophores. In addition, the charge-transfer (CT) state in Encap-TAT lies energetically closer to the local exciton (LE) state compared to TIPS-TAT, thereby enhancing the possibility of LE–CT hybridization. The synthesis of the dimer TIPS-BT0 (Fig. 2a) and the trimer TIPS-TAT (Fig. 2b) has been reported previously.51,53 Unlike the trimeric systems, TIPS-BT0 is a directly linked TIPS-tetracene dimer without a bridging unit and thus serves as a baseline reference for iSF. Building on these precedents, Encap-TAT (Fig. 2c) was prepared via a convergent route culminating in a Suzuki cross-coupling onto a rigid macrocyclic scaffold (Schemes S1–S3). The macrocycle was deliberately chosen to suppress aggregation and finely tune the energetic alignment between bridge and chromophore units.52 2-(Ethyl)hexyl alkyl chains ensure sufficient solubility.Across all three molecular architectures, from the dimer TIPS-BT0 to the trimers TIPS-TAT and Encap-TAT, DFT calculations suggest that the dihedral angle (θ) between each tetracene unit and adjacent arene units is consistently ∼32–33° (Fig. S1). This intrinsic twist and bulky solubilizing substituents suppress face-to-face π–π stacking while retaining orbital overlap sufficient for electronic communication (Fig. S2–S4). By combining structural rigidity with controlled connectivity, these scaffolds establish a well-defined platform for probing how bridge-mediated electronic alignment governs intramolecular singlet fission efficiency.
The TIPS-BT0 dimer (Fig. 2a, d and Table S1) exhibits the canonical TIPS-tetracene vibronic progression, with a sharp 0–0 band near 550 nm (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 182 cm−1) and a 0–1 shoulder at ∼510 nm (19
182 cm−1) and a 0–1 shoulder at ∼510 nm (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 608 cm−1). Compared to the monomer, enhanced 0–1 intensity signals weak H-type excitonic coupling between the tetracene units. In addition, a modest red shift of the excitonic band (∼500 cm−1) and the emergence of a CT-derived absorption feature near 429 nm (23
608 cm−1). Compared to the monomer, enhanced 0–1 intensity signals weak H-type excitonic coupling between the tetracene units. In addition, a modest red shift of the excitonic band (∼500 cm−1) and the emergence of a CT-derived absorption feature near 429 nm (23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 310 cm−1), which borrows oscillator strength through LE–CT mixing is observed.57 This reveals the presence of LE–CT interactions mediated by frontier orbital overlap between the chromophores.58–60 The PL spectrum retains a well-resolved vibronic progression with a small Stokes shift (∼196 cm−1), confirming localized S1 → S0 emission.
310 cm−1), which borrows oscillator strength through LE–CT mixing is observed.57 This reveals the presence of LE–CT interactions mediated by frontier orbital overlap between the chromophores.58–60 The PL spectrum retains a well-resolved vibronic progression with a small Stokes shift (∼196 cm−1), confirming localized S1 → S0 emission.
Building on this reference case, incorporation of a TIPS-A bridge in the TIPS-TAT trimer introduces new spectral features (Fig. 2e). The asymmetric architecture relaxes symmetry restrictions present in homodimers, allowing both the lower-energy (S0 → S1) and higher-energy (S0 → S2) transitions (Tables S2–S3 and Fig. 1C).57 The S0 → S1 transition (∼547 nm, 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 282 cm−1) retains the tetracene vibronic progression but shows enhanced 0–1 intensity, consistent with weak H-type excitonic coupling mediated through the bridge and exhibits a modest red shift (∼400 cm−1) relative to the TIPS-T monomer. A distinct (S0 → S2) band at 471 nm (21
282 cm−1) retains the tetracene vibronic progression but shows enhanced 0–1 intensity, consistent with weak H-type excitonic coupling mediated through the bridge and exhibits a modest red shift (∼400 cm−1) relative to the TIPS-T monomer. A distinct (S0 → S2) band at 471 nm (21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 230 cm−1) is red-shifted by ∼471 cm−1 relative to isolated TIPS-A, while a CT-derived band emerges near 408 nm with enhanced oscillator strength from LE–CT coupling.57 Together, these signatures confirm appreciable orbital overlap across the trimer backbone and modest LE–CT mixing. The PL spectrum preserves the tetracene-like vibronic structure, indicating that despite through-bridge interactions, emission remains dominated by localized TIPS-T excitons.
230 cm−1) is red-shifted by ∼471 cm−1 relative to isolated TIPS-A, while a CT-derived band emerges near 408 nm with enhanced oscillator strength from LE–CT coupling.57 Together, these signatures confirm appreciable orbital overlap across the trimer backbone and modest LE–CT mixing. The PL spectrum preserves the tetracene-like vibronic structure, indicating that despite through-bridge interactions, emission remains dominated by localized TIPS-T excitons.
By contrast, Encap-TAT (Fig. 2c, f and S5) displays qualitatively different optical behavior. Fusing Encap-T units that are more closely aligned in energy with the TIPS-A bridge erases monomeric vibronic progression, yielding a broad, vibronically unresolved absorption envelope (27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–21,000 cm−1). The PL spectrum is likewise broad, red-shifted, featureless with a large Stokes shift (∼1932 cm−1). These trends are consistent with a substantially reduced LE–CT separation in Encap-TAT (ΔECT–LE ≈ 0.35) relative to TIPS-TAT (≈0.75 eV), which enhances LE–CT hybridisation and favors relaxation into an intramolecular exciplex-like minimum.56,61 Overall, the progression from TIPS-TAT to Encap-TAT shows that decreasing LE–CT separation drives increasingly strong hybridization, with Encap-TAT's PL consistent with intramolecular excimer/exciplex-like emission.
000–21,000 cm−1). The PL spectrum is likewise broad, red-shifted, featureless with a large Stokes shift (∼1932 cm−1). These trends are consistent with a substantially reduced LE–CT separation in Encap-TAT (ΔECT–LE ≈ 0.35) relative to TIPS-TAT (≈0.75 eV), which enhances LE–CT hybridisation and favors relaxation into an intramolecular exciplex-like minimum.56,61 Overall, the progression from TIPS-TAT to Encap-TAT shows that decreasing LE–CT separation drives increasingly strong hybridization, with Encap-TAT's PL consistent with intramolecular excimer/exciplex-like emission.
In contrast to the broadened, featureless absorption of Encap-TAT, Encap-TA displays a structured spectrum with well-resolved vibronic features. Two lower-energy peaks at ∼512 nm (19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 531 cm−1) and ∼479 nm (20
531 cm−1) and ∼479 nm (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 878 cm−1), assigned to the S0 → S1 progression (Table S4) are weaker than a higher-energy band at ∼453 nm (22
878 cm−1), assigned to the S0 → S1 progression (Table S4) are weaker than a higher-energy band at ∼453 nm (22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 076 cm−1). This inversion of intensity reflects contributions from both S1 and S2 transitions and is consistent with an intermediate-coupling regime, moderate H-type excitonic coupling, together with differences in transition-dipole strengths for the Encap-T chromophore and the TIPS-A bridge (Fig. S6 and Table S2). A modest red shift in both LE derived (Frenkel-type) bands, peak broadening, and the appearance of a CT feature near ∼385 nm (25
076 cm−1). This inversion of intensity reflects contributions from both S1 and S2 transitions and is consistent with an intermediate-coupling regime, moderate H-type excitonic coupling, together with differences in transition-dipole strengths for the Encap-T chromophore and the TIPS-A bridge (Fig. S6 and Table S2). A modest red shift in both LE derived (Frenkel-type) bands, peak broadening, and the appearance of a CT feature near ∼385 nm (25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 974 cm−1) further indicate LE–CT coupling (in the diabatic basis) that yields LE–CT mixing and appreciable bridge–chromophore electronic communication.61,62 Notably, relative to Encap-TA, the Encap-TAT trimer exhibits additional red shifts of the S0 → S1/S2 envelope and the CT band, signalling much stronger LE–CT hybridisation in the trimer.
974 cm−1) further indicate LE–CT coupling (in the diabatic basis) that yields LE–CT mixing and appreciable bridge–chromophore electronic communication.61,62 Notably, relative to Encap-TA, the Encap-TAT trimer exhibits additional red shifts of the S0 → S1/S2 envelope and the CT band, signalling much stronger LE–CT hybridisation in the trimer.
While the Encap-TA absorption remains vibronically resolved, its PL collapses into a broad, featureless envelope that is markedly broader and more red-shifted than the monomer (Fig. 3c). The monomer's 0–0 fluorescence at ∼500 nm (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 012 cm−1) is only ∼7 nm (∼270 cm−1) red-shifted from its 0–0 absorption, whereas the Encap-TA PL peaks at ∼555 nm (∼18
012 cm−1) is only ∼7 nm (∼270 cm−1) red-shifted from its 0–0 absorption, whereas the Encap-TA PL peaks at ∼555 nm (∼18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 018 cm−1), exhibiting a ∼43 nm (∼1513 cm−1) Stokes shift and loss of vibronic structure. These pronounced red shifts and broadened, structureless PL in both Encap-TA and Encap-TAT are consistent with emission from a relaxed excited-state minimum that lies below the initially prepared Franck–Condon bright state. This behaviour is characteristic of an intramolecular exciplex-like (CT-stabilised) emissive state, i.e., emission from an adiabatic surface with enhanced CT character upon structural/solvent relaxation.47,63–66
018 cm−1), exhibiting a ∼43 nm (∼1513 cm−1) Stokes shift and loss of vibronic structure. These pronounced red shifts and broadened, structureless PL in both Encap-TA and Encap-TAT are consistent with emission from a relaxed excited-state minimum that lies below the initially prepared Franck–Condon bright state. This behaviour is characteristic of an intramolecular exciplex-like (CT-stabilised) emissive state, i.e., emission from an adiabatic surface with enhanced CT character upon structural/solvent relaxation.47,63–66
The experimental absorption spectrum of the heterodimer Encap-TA was quantitatively reproduced by a full vibronic Frenkel-exciton model (Fig. 3f and see Section 1.12 in the SI), which decomposes the response into lower (E−) and upper (E+) excitonic branches. The best fit was obtained using a transition-dipole ratio of µ2/µ1 = 0.75 and J = 334 cm−1. In a heterodimer, the loss of inversion symmetry “lights up” both excitons, so E− and E+ each carry oscillator strength (Fig. 1c). By contrast, a symmetric homodimer supports one bright and one dark exciton (Fig. S7).61,67–69 In our fit, E− captures the red-shifted S1 manifold (Table S2), while E+ accounts for the enhanced exciton-like (LE-derived) higher-energy feature near ∼480 nm. The charge-transfer (CT) contribution is modelled as a single Lorentzian centered at ∼26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1 (∼385 nm), shown as the purple dotted trace in Fig. 3d (Section 1.12, SI).
000 cm−1 (∼385 nm), shown as the purple dotted trace in Fig. 3d (Section 1.12, SI).
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) A˙+–B˙− and |AC〉
A˙+–B˙− and |AC〉 ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) A˙−–B˙+ (Section 1.8–1.11, SI). Diagonalization results in four adiabatic states (ψ1–ψ4) whose LE-subspace (excitonic) vs. CT-subspace weights, obtained from the eigenvector coefficients, are reported in Table 1 and correlated with the spectra in Fig. 3e and f. In this Hamiltonian, LE–CT coupling is mediated by the electron- and hole-transfer integrals (te, th). In both dimers, te and th exceed the LE–LE coulombic coupling (JCoul), indicating that charge-mediated superexchange coupling rather than direct coulomb coupling governs interunit communication (Tables S5–S8). LE–CT mixing refers exclusively to the LE vs. CT composition of these adiabatic eigenstates (ψ1–ψ4).
A˙−–B˙+ (Section 1.8–1.11, SI). Diagonalization results in four adiabatic states (ψ1–ψ4) whose LE-subspace (excitonic) vs. CT-subspace weights, obtained from the eigenvector coefficients, are reported in Table 1 and correlated with the spectra in Fig. 3e and f. In this Hamiltonian, LE–CT coupling is mediated by the electron- and hole-transfer integrals (te, th). In both dimers, te and th exceed the LE–LE coulombic coupling (JCoul), indicating that charge-mediated superexchange coupling rather than direct coulomb coupling governs interunit communication (Tables S5–S8). LE–CT mixing refers exclusively to the LE vs. CT composition of these adiabatic eigenstates (ψ1–ψ4).
| TIPS-TA | Encap-TA | |||||
|---|---|---|---|---|---|---|
| State | Energy (cm−1) | LE (%) | CT (%) | Energy (cm−1) | LE (%) | CT (%) |
| ψ 1 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 226.95 226.95 |
95.3 | 4.7 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 359.78 359.78 |
83.3 | 16.7 |
| ψ 2 | 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 742.32 742.32 |
82.8 | 17.2 | 21![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 745.28 745.28 |
81.4 | 18.6 |
| ψ 3 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 848.05 848.05 |
2.9 | 97.1 | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 056.15 056.15 |
31.1 | 68.9 |
| ψ 4 | 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 609.39 609.39 |
19.0 | 81.0 | 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 849.95 849.95 |
4.3 | 95.7 |
For the Encap-TA dimer, the two lowest adiabatic states are LE-dominant but carry substantial CT content (ψ1, ψ2 ≈ 83–81% LE/17–19% CT; Tables 1 and S9). Both are stabilized relative to their diabatic LE site energies (−804.0 cm−1 for ψ1) and (−663.9 cm−1 for ψ2), consistent with strong CT-mediated mixing and the red-shifted LE envelope (Fig. 3e). Concomitantly, a nominal CT state (ψ3) is mixed down and acquires appreciable LE character (∼31%), evidencing pronounced LE–CT hybridization. In parallel, level repulsion destabilizes the diabatic CT manifold by several hundred cm−1 leaving the CT states high in energy yet optically active via LE–CT mixing. Despite the CT adiabats lying several thousand cm−1 above the LE manifold, this superexchange is sufficient to (i) stabilize the bright LE pair (spectral red shift), (ii) activate CT-derived absorption via oscillator-strength borrowing, and (iii) broaden/soften vibronic structure mirroring the observed absorption and PL trends of Encap-TA (and, amplified further, in Encap-TAT).
In contrast, TIPS-TA displayed weaker LE and CT mixing. ψ1 remains highly localized (∼95% LE) and ψ2 shows only modest CT admixture (∼17% CT), while the upper pair is largely CT-like (ψ3–ψ4 ≳ 81–97% CT; Tables 1 and S10). Spectroscopically, optical features of the TIPS-TAT dimer are consistent with the electronic structure derived from the TIPS-TA model, comprising two stabilized LE-derived (exciton-like) absorption bands with only modest CT admixture, a weak intensity-borrowed CT band, and comparatively narrower vibronic signatures (Fig. 3f). The difference relative to Encap-TA arises from smaller effective LE–CT coupling and a larger LE–CT energy separation, which together suppress hybridization into the lowest bright states. Taken together, these results highlight starkly different mixing regimes. Encap-TA exhibits strong CT-mediated superexchange that injects appreciable CT character into the lowest LE-derived (exciton-like) adiabatic state, while TIPS-TA supports a more localized lowest state with only minor CT admixture.
Impact of LE–CT hybridisation on intramolecular singlet fission
In this section, we investigate how strong excitonic hybridization between local exciton (LE) states and bridge– chromophore charge-transfer (CT) states reshape the excited-state manifold and perturb the kinetic landscape of intramolecular singlet fission. To probe these effects, we performed both femtosecond and nanosecond transient absorption (fs/nsTA) measurements on TIPS-BT0, TIPS-TAT, and Encap-TAT in toluene (c ≈ 100 µM) at room temperature. Fig. 4a, d and g shows the fsTA contour maps (ΔT/T as a function of probe wavelength and pump-probe delay) for the three systems following photoexcitation at 530 nm. Target/global analysis of the fsTA datasets using the kinetic schemes described in Section 1.7 in the SI yields the evolution- and species-associated spectra summarized in Fig. 4.70,71Upon photoexcitation, TIPS-BT0 (Fig. 4a–c) exhibits the characteristic S1 → Sn photoinduced absorption (PIA) bands of TIPS-T or tetracene dimer chromophores centered at ≃657 nm and ≃446 nm, accompanied by ground-state bleach (GSB) features near ≃541 nm and ≃430 nm.48,72–74 Target analysis reveals that the initially populated S1 state decays with a lifetime of 1.39 ± 0.03 ps, concomitant with the rise of a correlated triplet pair (1TT) PIA, centred at 520 nm, spanning the 570–615 nm region as well as 657 nm. The 1TT population persists beyond the temporal window of the fsTA measurement. In the nanosecond transient absorption (nsTA) data (Fig. 5a, instrument response ≃1–2 ns), the same 1TT-associated PIA features are observed at early times. These then decay monoexponentially with a lifetime of τTT ≃ 8.7 ± 0.4 ns, ultimately leaving only a weak residual signal assigned to long-lived triplet (T1) state PIA (Fig. S8–S10). The absence of correlated triplet pair dissociation into free triplets (1TT → 2T1) is attributed to strong electronic communication between the chromophores and a high triplet–triplet exchange interaction. This elevated exchange coupling stabilizes the bound 1TT state and suppresses spin decoherence, thereby preventing its evolution into two non-interacting triplet excitons.75,76
The fsTA response of the TIPS-TAT trimer shows qualitatively similar characteristic S1 state features, with S1 → Sn PIA spanning from ∼590 nm to 850 nm, a GSB centred around ∼544 nm and prominent PIA bands below ∼475 nm region (Fig. 4d–f and S11). While the singlet-state features resemble those of the monomer, the increased spectral broadening may arise from modest CT admixture/exciton–CT hybridisation in the initially accessed adiabatic manifold. Additionally, introducing a TIPS-A bridge that is closely aligned in energy substantially alters the ensuing excited-state kinetics. The rise of the 1TT precursor in TIPS-TAT, tracked by PIA bands at 517 nm and 572–702 nm, occurs with τSF ≃ 11.2 ± 0.06 ps, in line with the previous reports,28,51 but significantly slower than the 1.39 ps observed for TIPS-BT0. This deceleration reflects the longer centre-to-centre separation and added torsional flexibility introduced by the anthracene linker. Once formed, the 1TT state in TIPS-TAT is remarkably long-lived (τTT ≃ 127.56 ± 1 ns), ∼15× longer than in the directly linked dimer and it undergoes partial separation of the correlated pair (1TT → 2T1) to yield free triplets (Fig. 5b, d and S12).
Quantitatively, from the fitted reversible S1 ⇌ 1TT kinetics, we estimate correlated triplet-pair yields of ϕTT = 96.97% (TIPS-BT0) and 98.02% (TIPS-TAT). Using the triplet-plateau analysis (SI Section 1.13), the yields of persistent isolated triplets are ∼15.5% (TIPS-BT0) and ∼58.8% (TIPS-TAT). Thus, while 1TT formation is near-quantitative in both TIPS-BT0 and TIPS-TAT, triplet separation is markedly more efficient in TIPS-TAT, underscoring the sensitivity of free-triplet generation to bridge-controlled exciton–CT mixing. This pronounced stabilization of the 1TT manifold highlights how bridge-resonant coupling can both extend multiexciton lifetimes and boost free-triplet yields, even while moderating the initial fission rate. In the case of TIPS-TAT, the presence of the bridge reduces the triplet–triplet exchange interaction between the terminal TIPS-T units, thereby facilitating spin decoherence and promoting the formation of spatially separated, long-lived free triplets.
In stark contrast, Encap-TAT exhibits a fundamentally different early-time TA signature. Upon 530 nm excitation, its S1 → Sn photoinduced absorption (PIA) no longer displays well-resolved peaks characteristic of the monomer Encap-T (Fig. 4g–i and S13–S15). Instead, it emerges as a broad, red-shifted, structureless band spanning ≃550–850 nm, with the ground-state bleach confined to ≲525 nm. This simultaneously red-shifted, unstructured PIA is consistent with substantial CT admixture in the lowest adiabatic manifold, as quantified for Encap-TA (≈17–19% CT) and expected to be enhanced in Encap-TAT given the smaller LE–CT detuning and stronger bridge–chromophore coupling. We therefore assign this feature to a singlet exciton with significant CT admixture, hereafter denoted as S1 (CT). The initially populated S1 (CT) feature decays with τEx ≃ 2.95 ± 0.04 ps into a second state whose PIA is broader and completely unstructured across the probe window. The decay of the initially populated state is markedly faster than in TIPS-TAT, and the resulting spectral profile closely resembles the intramolecular excimer-like states reported in related chromophoric systems.48,63–66,77,78 This is markedly different from the well-defined 1TT signature seen in TIPS-BT0 or TIPS-TAT and similar tetracene derivatives.7,73
We therefore refer to this broad, unstructured transient state as an intramolecular exciplex-like state (Ex) under dilute conditions. Here, Ex denotes a relaxed minimum on an exciton–CT-hybrid adiabatic surface with substantial bridge–chromophore CT character. We attribute Ex formation to structural and solvent relaxation on this hybrid surface, which progressively stabilises a more CT-rich minimum along the nuclear coordinate. Such relaxation can access a distribution of closely spaced relaxed minima (exciplex-like conformers) on the excited-state landscape, consistent with the progressive broadening and loss of vibronic structure observed in the transient absorption.47,64 The Ex-state decays with a lifetime of ≃ 16.9 ± 0.2 ns back to the ground state, leaving only a very weak residual PIA attributable to long-lived T1 excitons (Fig. S16 and Table S11). The assignment of this exciplex-like state, as a CT-stabilised relaxed minimum on an exciton–CT hybrid surface, is further supported by solvent-dependent steady-state photo-physics. In Encap-TAT, the PL maximum exhibits a pronounced red shift with increasing solvent polarity, whereas TIPS-TAT shows no such solvatochromic response (Fig. S17). Polarity stabilises CT states and enhances LE–CT mixing, whereas higher concentrations can introduce intermolecular excimer/aggregation pathways. Accordingly, measurements were performed under dilute conditions (c ≈ 100 µM) to minimise intermolecular contributions, and solvent-dependent TA data are provided in the SI (Section 4, Fig. S18–S25) to support the CT-rich character of the Ex state. These solvent-dependent fsTA and nsTA measurements (SI Section 4, Fig. S18–S25) show that increasing polarity accelerates Ex formation and shortens its lifetime for Encap-TAT. In contrast, for the SF-active TIPS-TAT, increasing polarity primarily accelerates 1TT formation without inducing comparable long-time quenching.
To further clarify the nature of this second component, we directly compared Encap-TAT with its single-arm analogue, Encap-TA (Fig. S26–30). In Encap-TA, the initially populated S1 (CT) state decays in ≃2.25 ± 0.06 ps into an equally broad, featureless PIA band spanning the entire probe window, virtually identical in spectral shape and kinetics to the second component seen in Encap-TAT. This broad PIA then relaxes to the ground state with a long-lived residual (τ2 ≃ 16–18 ns), consistent with a small population of free T1 formed by intersystem crossing. Crucially, the close correspondence between Encap-TA and Encap-TAT excludes a conventional multiexciton (1TT) assignment, since the triplet of TIPS-A lies far too high in energy to sustain a bound triplet pair with Encap-T.
Instead, we ascribe both decay components to a strongly mixed LE–CT hybrid state, hereafter denoted as the intramolecular exciplex-like (Ex) state. This state arises from nuclear and structural relaxation in the excited state, aided by stabilization of the CT energy and significant electron- and hole-transfer integrals coupling the bridge and chromophores. These factors hybridise the local excitons with CT configurations into a continuous, unstructured excited-state manifold.56,79–82 Importantly, such strong LE–CT hybridization suppresses the formation of well-defined triplet pairs localized on the terminal Encap-T units. The Ex-state instead forms on ultrafast timescales, outcompeting correlated triplet-pair formation, while simultaneously enhancing radiative decay pathways. This is consistent with the higher photo-luminescence quantum yield observed for Encap-TAT (80.5%) compared to TIPS-TAT (∼38.5%).
To rationalize the contrasting excited-state pathways, we summarize the role of LE–CT hybridization using the mechanistic schematic in Fig. 6. Under weak LE–CT mixing (top), exemplified by TIPS-TAT, the CT configuration associated with the bridge–chromophore pair remains largely virtual and mediates population transfer from S1 to the correlated triplet pair 1TT via a CT-assisted superexchange pathway, followed by formation of separated triplets (T1 + T1). In contrast, under strong LE–CT mixing (bottom), exemplified by Encap-TAT, reduced LE–CT detuning and enhanced coupling yield a CT-enriched singlet S1(CT) that relaxes into an intramolecular exciplex-like minimum (Ex). This trap diverts population away from 1TT formation and increases non-productive loss, consistent with the broadened, red-shifted spectral signatures and pronounced solvatochromic PL red shift observed for Encap-TAT.
Conclusions
By constructing the TIPS-BT0 → TIPS-TAT → Encap-TAT series and combining steady-state/ultrafast spectroscopy with a four-state LE/CT diabatic Hamiltonian analysis, we show that bridge–chromophore energetic alignment governs exciton–CT hybridisation in the adiabatic excited states and thereby dictates the photophysical outcome in intramolecular singlet fission (iSF). In TIPS-TAT, a larger LE–CT energy offset limits the CT admixture while retaining sufficient LE ↔ CT coupling to enable CT-assisted superexchange into 1TT, yielding long-lived, separable triplets. Although both TIPS-BT0 and TIPS-TAT exhibit near-quantitative correlated triplet-pair formation (ϕTT > 95%), the isolated triplet yield differs substantially (∼16% for TIPS-BT0vs. ∼59% for TIPS-TAT), demonstrating that triplet separation is highly sensitive to bridge-controlled exciton–CT mixing. In contrast, in Encap-TAT a smaller LE–CT gap increases CT character and stabilises a CT-rich exciplex-like relaxed minimum, which competes with and suppresses iSF. For device applications, iSF scaffolds should therefore be engineered to maintain a sufficient LE–CT offset (avoiding excessive CT stabilisation) while keeping LE ↔ CT coupling in the moderate regime that supports CT-mediated superexchange, alongside efficient pathways for triplet separation/harvesting. Overall, tuning the LE–CT energy gap and bridge–chromophore coupling strength provides a practical design lever to direct molecular assemblies toward either correlated triplet formation or exciplex-like relaxation and emission.Author contributions
A. R. and H. B. conceived and supervised the project. E. S. carried out the experimental investigations, theoretical calculations and prepared the initial draft of the manuscript. J. W. performed the photoluminescence quantum yield (PLQY) measurements. D. G. C., J. R., S. M., O. M., and H. H. were involved in the synthesis and characterization of the materials. All authors contributed to the discussion of the results and to the final version of the manuscript. All authors except Chanakarn Phansa have given approval to the final version of the manuscript.Conflicts of interest
All experimental/computational data and procedures are available in the SI.Data availability
The data underlying all figures and tables in this article are publicly available from the University of Cambridge repository at https://doi.org/10.17863/CAM.129000. This repository includes the underlying datasets and the analysis outputs used to generate the figures. Any analysis code and scripts used for data processing, fitting, and figure generation are also available in the same repository. Additional supporting data are provided in the supplementary information (SI). Supplementary information: additional experimental details, characterization data, tables and figures. See DOI: https://doi.org/10.1039/d6sc00278a.Acknowledgements
E. S. acknowledges funding from UKRI Postdoctoral Individual Fellowships (Grant No. EP/Y026659/1). D. G. C. would like to acknowledge the Herchel Smith Fund for an early career fellowship, and the Royal Society (URF\R1\241806). L. M. C. thanks the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences for funding under Award Number DE-SC0022036. This work was supported by a UKRI Frontier Research Grant (EP/Y015584/1), an Australian Research Council Grant (FT230100002) and an Engineering Physical Sciences Research Council Programme Grant (EP/W017091/1).References
- D. N. Congreve, J. Lee, N. J. Thompson, E. Hontz, S. R. Yost, P. D. Reusswig, M. E. Bahlke, S. Reineke, T. Van Voorhis and M. A. Baldo, Science, 2013, 340, 334–337 CrossRef CAS PubMed.
- M. B. Smith and J. Michl, Chem. Rev., 2010, 110, 6891–6936 CrossRef CAS PubMed.
- A. Rao and R. H. Friend, Nat. Rev. Mater., 2017, 2, 1–12 Search PubMed.
- J. Xia, S. N. Sanders, W. Cheng, J. Z. Low, J. Liu, L. M. Campos, T. J. Sun Xia, W. Cheng, J. Liu, T. Sun, S. N. Sanders, J. Z. Low, L. M. Campos and J. Xia, Adv. Mater., 2017, 29, 1601652 CrossRef PubMed.
- A. Kunzmann, M. Gruber, R. Casillas, J. Zirzlmeier, M. Stanzel, W. Peukert, R. R. Tykwinski and D. M. Guldi, Angew. Chem., Int. Ed., 2018, 57, 10742–10747 CrossRef CAS PubMed.
- O. V. Mikhnenko, P. W. M. Blom and T. Q. Nguyen, Energy Environ. Sci., 2015, 8, 1867–1888 RSC.
- S. T. Roberts, R. E. McAnally, J. N. Mastron, D. H. Webber, M. T. Whited, R. L. Brutchey, M. E. Thompson and S. E. Bradforth, J. Am. Chem. Soc., 2012, 134, 6388–6400 CrossRef CAS PubMed.
- S. W. Eaton, L. E. Shoer, S. D. Karlen, S. M. Dyar, E. A. Margulies, B. S. Veldkamp, C. Ramanan, D. A. Hartzler, S. Savikhin, T. J. Marks and M. R. Wasielewski, J. Am. Chem. Soc., 2013, 135, 14701–14712 CrossRef CAS PubMed.
- B. S. Basel, J. Zirzlmeier, C. Hetzer, B. T. Phelan, M. D. Krzyaniak, S. R. Reddy, P. B. Coto, N. E. Horwitz, R. M. Young, F. J. White, F. Hampel, T. Clark, M. Thoss, R. R. Tykwinski, M. R. Wasielewski and D. M. Guldi, Nat. Commun., 2017, 8, 1–8 Search PubMed.
- S. N. Sanders, E. Kumarasamy, A. B. Pun, M. T. Trinh, B. Choi, J. Xia, E. J. Taffet, J. Z. Low, J. R. Miller, X. Roy, X. Y. Zhu, M. L. Steigerwald, M. Y. Sfeir and L. M. Campos, J. Am. Chem. Soc., 2015, 137, 8965–8972 CrossRef CAS PubMed.
- J. Kim, D. C. Bain, V. Ding, K. Majumder, D. Windemuller, J. Feng, J. Wu, S. Patil, J. Anthony, W. Kim and A. J. Musser, Nat. Chem., 2024, 16, 1680–1686 CrossRef CAS PubMed.
- K. C. Krishnapriya, P. Roy, B. Puttaraju, U. Salzner, A. J. Musser, M. Jain, J. Dasgupta and S. Patil, Nat. Commun., 2019, 10, 1–8 Search PubMed.
- G. He, E. M. Churchill, K. R. Parenti, J. Zhang, P. Narayanan, F. Namata, M. Malkoch, D. N. Congreve, A. Cacciuto, M. Y. Sfeir and L. M. Campos, Nat. Commun., 2023, 14, 1–10 Search PubMed.
- E. Busby, J. Xia, Q. Wu, J. Z. Low, R. Song, J. R. Miller, X. Y. Zhu, L. M. Campos and M. Y. Sfeir, Nat. Mater., 2015, 14, 426–433 CrossRef CAS PubMed.
- A. B. Pun, S. N. Sanders, E. Kumarasamy, M. Y. Sfeir, D. N. Congreve, L. M. Campos, A. B. Pun, S. N. Sanders, E. Kumarasamy, L. M. Campos, M. Y. Sfeir and D. N. Congreve, Adv. Mater., 2017, 29, 1701416 CrossRef PubMed.
- T. Yamakado, S. Takahashi, K. Watanabe, Y. Matsumoto, A. Osuka, S. Saito, T. Yamakado, S. Takahashi, K. W. Atanabe, Y. M. Atsumoto, P. R. A. Osuka and S. Saito, Angew. Chem., Int. Ed., 2018, 57, 5438–5443 CrossRef CAS PubMed.
- L. Schaufelberger, J. T. Blaskovits, R. Laplaza, K. Jorner and C. Corminboeuf, Angew. Chem., Int. Ed., 2025, 64, e202415056 CrossRef CAS PubMed.
- S. Nakamura, H. Sakai, M. Fuki, R. Ooie, F. Ishiwari, A. Saeki, N. V. Tkachenko, Y. Kobori and T. Hasobe, Angew. Chem., Int. Ed., 2023, 62, e202217704 Search PubMed.
- A. M. Levine, G. He, G. Bu, P. Ramos, F. Wu, A. Soliman, J. Serrano, D. Pietraru, C. Chan, J. D. Batteas, M. Kowalczyk, S. J. Jang, B. L. Nannenga, M. Y. Sfeir, E. H. R. Tsai and A. B. Braunschweig, J. Phys. Chem. C, 2021, 125, 12207–12213 CrossRef CAS PubMed.
- A. B. Pun, A. Asadpoordarvish, E. Kumarasamy, M. J. Y. Tayebjee, D. Niesner, D. R. McCamey, S. N. Sanders, L. M. Campos and M. Y. Sfeir, Nat. Chem., 2019, 11, 821–828 CrossRef CAS PubMed.
- Y. Hong, M. Rudolf, M. Kim, J. Kim, T. Schembri, A. M. Krause, K. Shoyama, D. Bialas, M. I. S. Röhr, T. Joo, H. Kim, D. Kim and F. Würthner, Nat. Commun., 2022,(13), 1–11 Search PubMed.
- T. Ullrich, P. Pinter, J. Messelberger, P. Haines, R. Kaur, M. M. Hansmann, D. Munz and D. M. Guldi, Angew. Chem., 2020, 132, 7980–7988 Search PubMed.
- E. A. Margulies, Y. L. Wu, P. Gawel, S. A. Miller, L. E. Shoer, R. D. Schaller, F. Diederich and M. R. Wasielewski, Angew. Chem., Int. Ed., 2015, 54, 8679–8683 CrossRef CAS PubMed.
- C. Hetzer, D. M. Guldi and R. R. Tykwinski, Chem.–Eur. J., 2018, 24, 8245–8257 CrossRef CAS PubMed.
- B. S. Basel, R. M. Young, M. D. Krzyaniak, I. Papadopoulos, C. Hetzer, Y. Gao, N. T. La Porte, B. T. Phelan, T. Clark, R. R. Tykwinski, M. R. Wasielewski and D. M. Guldi, Chem. Sci., 2019, 10, 11130–11140 RSC.
- E. A. Margulies, C. E. Miller, Y. Wu, L. Ma, G. C. Schatz, R. M. Young and M. R. Wasielewski, Nat. Chem., 2016, 8, 1120–1125 CrossRef CAS PubMed.
- M. J. Y. Tayebjee, S. N. Sanders, E. Kumarasamy, L. M. Campos, M. Y. Sfeir and D. R. McCamey, Nat. Phys., 2017, 13, 182–188 Search PubMed.
- K. R. Parenti, R. Chesler, G. He, P. Bhattacharyya, B. Xiao, H. Huang, D. Malinowski, J. Zhang, X. Yin, A. Shukla, S. Mazumdar, M. Y. Sfeir and L. M. Campos, Nat. Chem., 2023, 15, 339–346 CrossRef CAS PubMed.
- T. Wang, H. Liu, X. Wang, L. Tang, J. Zhou, X. Song, L. Lv, W. Chen, Y. Chen and X. Li, J. Mater. Chem. A, 2023, 11, 8515–8539 RSC.
- W. Kim and A. J. Musser, Adv. Phys.:X, 2021, 6, 1918022 Search PubMed.
- W. B. Davis, W. A. Svec, M. A. Ratner and M. R. Wasielewski, Nature, 1998, 396, 60–63 CrossRef CAS.
- K. Majumder, S. Mukherjee, J. Park, W. Kim, A. J. Musser and S. Patil, Angew. Chem., Int. Ed., 2024, 63, e202408615 CrossRef CAS PubMed.
- M. Majdecki, C. H. Hsu, C. H. Wang, E. H. C. Shi, M. Zakrocka, Y. C. Wei, B. H. Chen, C. H. Lu, S. Da Yang, P. T. Chou and P. Gaweł, Angew. Chem., Int. Ed., 2024, 63, e202401103 CrossRef CAS PubMed.
- E. Kumarasamy, S. N. Sanders, M. J. Y. Tayebjee, A. Asadpoordarvish, T. J. H. Hele, E. G. Fuemmeler, A. B. Pun, L. M. Yablon, J. Z. Low, D. W. Paley, J. C. Dean, B. Choi, G. D. Scholes, M. L. Steigerwald, N. Ananth, D. R. McCamey, M. Y. Sfeir and L. M. Campos, J. Am. Chem. Soc., 2017, 139, 12488–12494 CrossRef CAS PubMed.
- T. Wang, S. Zhang, Y. T. Ding, B. Y. Zhang, B. Yu, R. Xu, Z. X. Liu, C. L. Sun, C. Zhang, Q. Wang and H. L. Zhang, J. Mater. Chem. C, 2025, 13, 9576–9583 RSC.
- T. C. Berkelbach, M. S. Hybertsen and D. R. Reichman, J. Chem. Phys., 2013, 138, 114103 CrossRef PubMed.
- B. S. Basel, C. Hetzer, J. Zirzlmeier, D. Thiel, R. Guldi, F. Hampel, A. Kahnt, T. Clark, D. M. Guldi and R. R. Tykwinski, Chem. Sci., 2019, 10, 3854–3863 RSC.
- L. C. Lin, T. Smith, Q. Ai, B. K. Rugg, C. Risko, J. E. Anthony, N. H. Damrauer and J. C. Johnson, Chem. Sci., 2023, 14, 11554–11565 RSC.
- B. Carlotti, I. K. Madu, H. Kim, Z. Cai, H. Jiang, A. K. Muthike, L. Yu, P. M. Zimmerman and T. Goodson, Chem. Sci., 2020, 11, 8757–8770 RSC.
- H. M. Bergman, G. R. Kiel, R. J. Witzke, D. P. Nenon, A. M. Schwartzberg, Y. Liu and T. D. Tilley, J. Am. Chem. Soc., 2020, 142, 19850–19855 CrossRef CAS PubMed.
- D. Bansal, A. Kundu, V. P. Singh, A. K. Pal, A. Datta, J. Dasgupta and P. Mukhopadhyay, Chem. Sci., 2022, 13, 11506–11512 RSC.
- W. Ishii, M. Fuki, E. M. Bu Ali, S. Sato, B. Parmar, A. Yamauchi, C. H. Mulyadi, M. Uji, S. Medina Rivero, G. Watanabe, J. Clark, Y. Kobori and N. Yanai, J. Am. Chem. Soc., 2024, 146, 25527–25535 CrossRef CAS PubMed.
- A. Mohanty, V. P. Singh, C. M. Hussain, M. Dey, D. Ghosh, P. Mukhopadhyay and J. Dasgupta, Chem. Sci., 2025, 16, 21368–21378 RSC.
- A. Neef, S. Beaulieu, S. Hammer, S. Dong, J. Maklar, T. Pincelli, R. P. Xian, M. Wolf, L. Rettig, J. Pflaum and R. Ernstorfer, Nature, 2023, 616, 275–279 CrossRef CAS PubMed.
- H. Miyamoto, K. Okada, K. Tada, R. Kishi and Y. Kitagawa, Molecules, 2024, 29, 5449 CrossRef CAS PubMed.
- Y. Bo, Y. Hou, D. A. X. Lavergne, T. Clark, M. J. Ferguson, R. R. Tykwinski and D. M. Guldi, Nat. Commun., 2025, 16, 1–12 Search PubMed.
- Y. J. Bae, D. Shimizu, J. D. Schultz, G. Kang, J. Zhou, G. C. Schatz, A. Osuka and M. R. Wasielewski, J. Phys. Chem. A, 2020, 124, 8478–8487 CrossRef CAS PubMed.
- C. B. Dover, J. K. Gallaher, L. Frazer, P. C. Tapping, A. J. Petty, M. J. Crossley, J. E. Anthony, T. W. Kee and T. W. Schmidt, Nat. Chem., 2018, 10, 305–310 CrossRef CAS PubMed.
- X. Feng and A. I. Krylov, Phys. Chem. Chem. Phys., 2016, 18, 7751–7761 RSC.
- S. Valianti and S. S. Skourtis, J. Phys. Chem. Lett., 2022, 13, 939–946 CrossRef CAS PubMed.
- K. R. Parenti, G. He, S. N. Sanders, A. B. Pun, E. Kumarasamy, M. Y. Sfeir and L. M. Campos, J. Phys. Chem. A, 2020, 124, 9392–9399 CrossRef CAS PubMed.
- J. Royakkers, A. Minotto, D. G. Congrave, W. Zeng, A. Patel, A. D. Bond, D. K. Bučar, F. Cacialli and H. Bronstein, J. Org. Chem., 2020, 85, 207–214 CrossRef CAS PubMed.
- A. B. Pun, S. N. Sanders, M. Y. Sfeir, L. M. Campos and D. N. Congreve, Chem. Sci., 2019, 10, 3969–3975 RSC.
- K. A. Kistler, C. M. Pochas, H. Yamagata, S. Matsika and F. C. Spano, J. Phys. Chem. B, 2012, 116, 77–86 CrossRef CAS PubMed.
- F. Gao, Y. Zhao and W. Liang, J. Phys. Chem. B, 2011, 115, 2699–2708 CrossRef CAS PubMed.
- N. J. Hestand and F. C. Spano, Chem. Rev., 2018, 118, 7069–7163 CrossRef CAS PubMed.
- T. J. H. Hele, E. G. Fuemmeler, S. N. Sanders, E. Kumarasamy, M. Y. Sfeir, L. M. Campos and N. Ananth, J. Phys. Chem. A, 2019, 123, 2527–2536 CrossRef CAS PubMed.
- E. Sebastian and M. Hariharan, J. Am. Chem. Soc., 2021, 143, 13769–13781 CrossRef CAS PubMed.
- N. J. Hestand and F. C. Spano, Acc. Chem. Res., 2017, 50, 341–350 CrossRef CAS PubMed.
- N. J. Hestand and F. C. Spano, J. Chem. Phys., 2015, 143, 244707 CrossRef PubMed.
- D. Bialas, C. Brüning, F. Schlosser, B. Fimmel, J. Thein, V. Engel and F. Würthner, Chem.–Eur. J., 2016, 22, 15011–15018 CrossRef CAS PubMed.
- P. Ottiger, H. Köppel and S. Leutwyler, Chem. Sci., 2015, 6, 6059–6068 RSC.
- R. Jing, Y. Li, K. Tajima, Y. Wan, N. Fukui, H. Shinokubo, Z. Kuang and A. Xia, J. Phys. Chem. Lett., 2024, 15, 1469–1476 CrossRef CAS PubMed.
- E. Sebastian, J. Sunny and M. Hariharan, Chem. Sci., 2022, 13, 10824–10835 RSC.
- R. M. Young and M. R. Wasielewski, Acc. Chem. Res., 2020, 53, 1957–1968 CrossRef CAS PubMed.
- J. Hoche, M. Flock, X. Miao, L. N. Philipp, M. Wenzel, I. Fischer and R. Mitric, Chem. Sci., 2021, 12, 11965–11975 RSC.
- S. F. Völker, A. Schmiedel, M. Holzapfel, K. Renziehausen, V. Engel and C. Lambert, J. Phys. Chem. C, 2014, 118, 17467–17482 CrossRef.
- J. Seibt, P. Marquetand, V. Engel, Z. Chen, V. Dehm and F. Würthner, Chem. Phys., 2006, 328, 354–362 CrossRef CAS.
- E. Kirchner, D. Bialas and F. Würthner, Chem.–Eur. J., 2019, 25, 11294–11301 CrossRef CAS PubMed.
- J. J. Snellenburg, S. Laptenok, R. Seger, K. M. Mullen and I. H. M. van Stokkum, J. Stat. Software, 2012, 49, 1–22 Search PubMed.
- I. H. M. Van Stokkum, D. S. Larsen and R. Van Grondelle, Biochim. Biophys. Acta Bioenerg., 2004, 1657, 82–104 CrossRef CAS PubMed.
- H. L. Stern, A. J. Musser, S. Gelinas, P. Parkinson, L. M. Herz, M. J. Bruzek, J. Anthony, R. H. Friend and B. J. Walker, Proc. Natl. Acad. Sci. U. S. A., 2015, 112, 7656–7661 CrossRef CAS PubMed.
- Z. Wang, H. Liu, X. Xie, C. Zhang, R. Wang, L. Chen, Y. Xu, H. Ma, W. Fang, Y. Yao, H. Sang, X. Wang, X. Li and M. Xiao, Nat. Chem., 2021, 13, 559–567 CrossRef CAS PubMed.
- Y. Bo, Y. Hou, D. Thiel, R. Weiß, T. Clark, M. J. Ferguson, R. R. Tykwinski and D. M. Guldi, J. Am. Chem. Soc., 2023, 145, 18260–18275 CrossRef CAS PubMed.
- O. Millington, S. Montanaro, A. Sharma, S. A. Dowland, J. Winkel, J. Grune, A. Leventis, T. Bennett, J. Shaikh, N. Greenham, A. Rao and H. Bronstein, J. Am. Chem. Soc., 2024, 146, 29664–29674 Search PubMed.
- V. Abraham and N. J. Mayhall, J. Phys. Chem. Lett., 2021, 12, 10505–10514 Search PubMed.
- R. A. Krueger and G. Blanquart, J. Phys. Chem. A, 2019, 123, 1796–1806 CrossRef CAS PubMed.
- J. Choi, S. Kim, M. Ahn, J. Kim, D. W. Cho, D. Kim, S. Eom, D. Im, Y. Kim, S. H. Kim, K. R. Wee and H. Ihee, Commun. Chem., 2023, 6, 1–11 Search PubMed.
- N. J. Hestand, H. Yamagata, B. Xu, D. Sun, Y. Zhong, A. R. Harutyunyan, G. Chen, H. L. Dai, Y. Rao and F. C. Spano, J. Phys. Chem. C, 2015, 119, 22137–22147 CrossRef CAS.
- H. Yamagata, J. Norton, E. Hontz, Y. Olivier, D. Beljonne, J. L. Brédas, R. J. Silbey and F. C. Spano, J. Chem. Phys., 2011, 134, 204703 Search PubMed.
- S. Lukman, K. Chen, J. M. Hodgkiss, D. H. P. Turban, N. D. M. Hine, S. Dong, J. Wu, N. C. Greenham and A. J. Musser, Nat. Commun., 2016,(7), 1–13 Search PubMed.
- Y. Ishino, K. Miyata, T. Sugimoto, K. Watanabe, Y. Matsumoto, T. Uemura and J. Takeya, Phys. Chem. Chem. Phys., 2014, 16, 7501–7512 RSC.
Footnotes |
| † Chanakarn Phansa could not be contacted to confirm the final author list prior to acceptance. |
| ‡ These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |