Open Access Article
Open Access ArticleEnantioselective organocatalytic radical alkylation enabled by photoexcitation
Yanji Song,
Ruifeng Wang,
Xi Lu,
Yuqiao Zhou ,
Xiaoming Feng
,
Xiaoming Feng ,
Weidi Cao
,
Weidi Cao * and
Xiaohua Liu
* and
Xiaohua Liu *
*
Key Laboratory of Green Chemistry & Technology, Ministry of Education, College of Chemistry, Sichuan University, Chengdu 610064, China. E-mail: liuxh@scu.edu.cn; wdcao@scu.edu.cn
First published on 23rd April 2026
Abstract
Photoinduced single-electron transfer generates radicals that could engage in radical–radical cross-coupling reactions, but organocatalytic photocatalyst-free systems for enantioselective processes remain rare and challenging. Herein, we report a light-driven enantioselective alkylation of 3-hydroxyoxindoles with NHPI esters, providing a new route for a variety of 3-alkyl-3-hydroxy oxindole derivatives. The reaction follows a light-driven electron transfer/radical cross-coupling process by using a readily available chiral guanidine organocatalyst and PPh3 without additional photocatalysts. The mechanistic studies reveal that the guanidine catalyst and phosphine benefit the formation of an excited enol reductant and an NHPI oxidant for electron transfer, respectively, and the chiral bifunctional catalyst also provides an H-bond network to guide outer-sphere radical coupling in an enantioselective manner.
Introduction
Radical cross coupling provides a useful route for carbon–carbon bond formation, which greatly simplifies access to various complex molecules from abundant activated or inert radical precursors.1 For instance, N-(acyloxy)phthalimides (NHPI esters), as redox-active esters introduced by Okada and coworkers,2 can be easily prepared from the corresponding carboxylic acids and N-hydroxyphthalimide (NHPI), and have been exploited as alkyl radical precursors without electron bias.3 The decarboxylative alkylation of NHPI esters via cross-couplings could be achieved by either transition metal complex catalysis4,5 or organocatalysis6–10 with or without light-irradiation (Scheme 1). For instance, Baran and coworkers documented a nickel salt-enabled C(sp3)–C(sp3) cross-coupling with an alkylzinc reagent as the partner.4a,b Difluoromethylenated zinc reagents4c and organo-aluminum reagents,4d as well as protected glycines catalyzed by copper catalysts,5 also participated in the coupling (Scheme 1a). Alternatively, in the absence of an Ir-photocatalyst,6 the coupling reaction could occur via electron transfer methods driven by the photoactivity of electron donor–acceptor (EDA) complexes (Scheme 1b).7 Shang, Fu and coworkers disclosed that a PPh3/NaI combination under blue light irradiation was able to furnish the coupling with silyl enol ethers to synthesize aryl alkyl ketones.8 Later, Zheng's study revealed that photo-driven cross-couplings of NHPI esters with 3-aryl oxindoles and 2-aryl oxindoles were performed in the presence of nBu4NI9 or under catalyst-free conditions,10 respectively (Scheme 1b).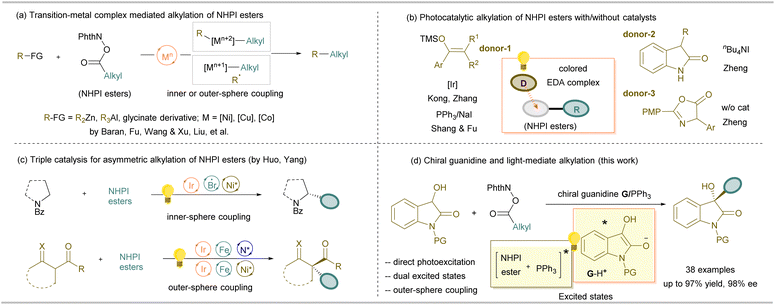 | ||
| Scheme 1 Overview of the decarboxylative alkylation reaction of NHPI esters for Csp3–Csp3 cross-coupling. | ||
Nevertheless, enantioselective control of radical cross-coupling for the generation of stereogenic carbon centers remains challenging. Yang and coworkers utilized triple catalysis of photoredox/Fe with a chiral primary amine or nickel catalyst for the coupling of NHPI esters with carbonyl derivatives (Scheme 1c).11 Huo's group utilized metallaphotoredox catalysis for the enantioselective cross-coupling of carboxylic acid via in situ formed NHPI esters as the precursor.12 However, the progression of asymmetric organocatalytic radical cross coupling has been relatively constrained, and the majority of organocatalytic photochemical systems focused on aminocatalysis, chiral phosphoric acid catalysis, ion-pairing catalysis, and NHC-catalysis, in combination with photocatalysts or exogenous promoters in most cases.13 The enhanced redox activity of excited species under light irradiation enables efficient photoinduced electron transfer processes. Notable examples include Melchiorre's asymmetric β-alkylation of enals via direct excitation of chiral iminium ions,14a Akiyama's alkylation of phosphoric acid-activated imines,14b and our group's alkylation of Lewis acid-activated ketones, which proceed through the generation of excited-state oxidants.14c Additionally, asymmetric α-alkylation of aldehydes14d has been achieved via EDA complex formation.15 Inspired by these advances, we hypothesized that an electron-rich nucleophile in its excited state could serve as a suitable radical coupling partner for electron transfer with NHPI esters (Scheme 1d).
Chiral 3-alkyl-3-hydroxy oxindole skeletons are widely found in bioactive compounds.16 Besides addition reactions of isatin, asymmetric alkylation of 3-hydroxyoxindoles with electrophiles represents a straightforward approach to construct such structures. Nevertheless, the limited diversity of available alkylation reagents significantly restricts structural variation.17 We previously established a chiral guanidine-based polar cascade reaction of 3-hydroxyoxindoles with coumarins.18 This nucleophile readily undergoes base-assisted enolization to form a diol intermediate, which exhibits significant reducing properties and enhanced hydrogen-bonding interactions with the catalyst. We envisioned that direct photoredox radical alkylation of 3-hydroxyoxindoles would provide an efficient route to 3-alkyl-3-hydroxy oxindole derivatives. Herein, we report a chiral guanidine19 and visible-light-driven radical cross-coupling reaction for the decarboxylative alkylation of 3-hydroxyoxindoles with NHPI esters in the absence of external photocatalysts. The chiral catalyst facilitates the formation of an excited state enol intermediate as a reductant to generate an oxindole radical stabilized through a captodative effect, and also enables enantioselective outer-sphere radical cross-coupling through hydrogen bonds,20 giving access to a number of enantiomerically enriched 3,3′-bifunctional oxindoles (Scheme 1d). Moreover, we observed that the assistance of PPh3 could further accelerate the reaction,21 which was proposed to interact with NHPI esters to reach an excited state at the same time, facilitating electron transfer as an electron-shuttle.22 This demonstrates a new double excited state model for light-driven transformation.
Results and discussion
Initially, the irradiation of a mixture of N-benzyl-3-hydroxyoxindole A1 and N-hydroxyphthalimide ester B1 in MeCN using 385 nm light at 20 °C led to the formation of the alkylation product C1 in 18% yield. We next introduced chiral guanidine-amide catalysts developed in our group to promote the asymmetric alkylation (Table 1). The guanidine-amides G1–G5, synthesized from different amino acids with diphenylmethanamine, could accelerate the reaction slightly (28–40% yield), but the enantioselectivities were generally poor (2–21% ee). The sterically hindered aniline-based catalyst G6 gave less improvement in comparison with G3, which has the same guanidine subunit. Another kind of guanidine catalysts bearing a sulfonamide unit (G7–G10) showed better enantioselectivity, and modification of the sulfonamide substituent into a 1-naphthyl group (G8) resulted in 87% ee, but the yield remained unsatisfied.| a Reaction conditions: unless otherwise noted, all reactions were carried out with A1 (0.10 mmol), B1 (1.0 equiv.), and G (10 mol%) in MeCN (0.1 M) at 20 °C under a N2 atmosphere with 10 W LED (λ = 385 nm) irradiation. Isolated yields. Ee values were determined by SFC analysis.b G (10 mol%) and PPh3 (20 mol%) at −20 °C.c MeCN (0.2 M).d 5 W LED.e B1 (2.0 equiv.). |
|---|
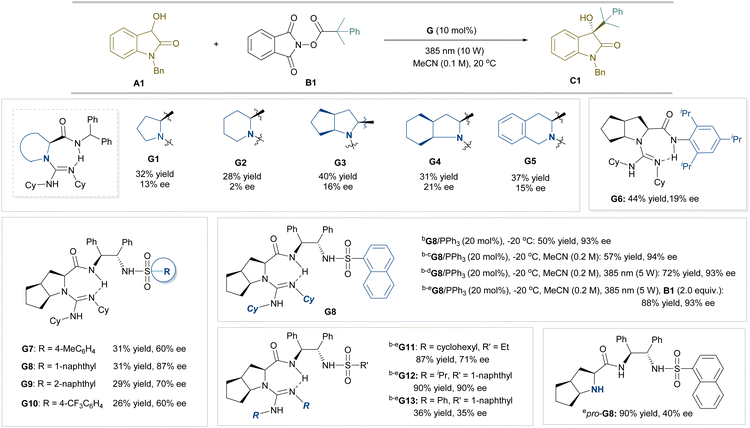 |
Previously, the use of phosphine21 or a Ph3P/NaI system,8,15,23 which are good electron donors for forming EDA complexes, has been developed for alkylation. We therefore attempted to add Ph3P to increase the activity. Delightfully, the combination of guanidine G8 and Ph3P without NaI benefited the reaction, and 50% yield with 93% ee was obtained when the reaction temperature was lowered to −20 °C. After enhancing the concentration of the reaction and reducing the light power, the yield gradually increased to 72% with a maintained ee value. When the amount of NHPI ester B1 was doubled, the product C1 could be isolated in 88% yield with 93% ee.
To probe into the role of subunits of the optimized catalyst, its analogues were tested under the optimized reaction conditions. The guanidine G11, containing ethanesulfonamide instead of naphthalene-1-sulfonamide, decreased the ee value a little (71% ee). Changing the substitutions at the amidine unit from N-cyclohexyl to N-isopropyl (G12) had slightly influence on the outcome, but N-phenyl-based G13 dramatically decreased the reactivity and enantioselectivity. Additionally, the use of an amine procursor without the guanidine unit also resulted in lower enantioselectivity. These results imply the critical role of the basic unit of the catalyst in the reactivity, and the CN3 backbone as well as sulfonamide substitution as the H-bond acceptor and donor in the enantioselectivity.
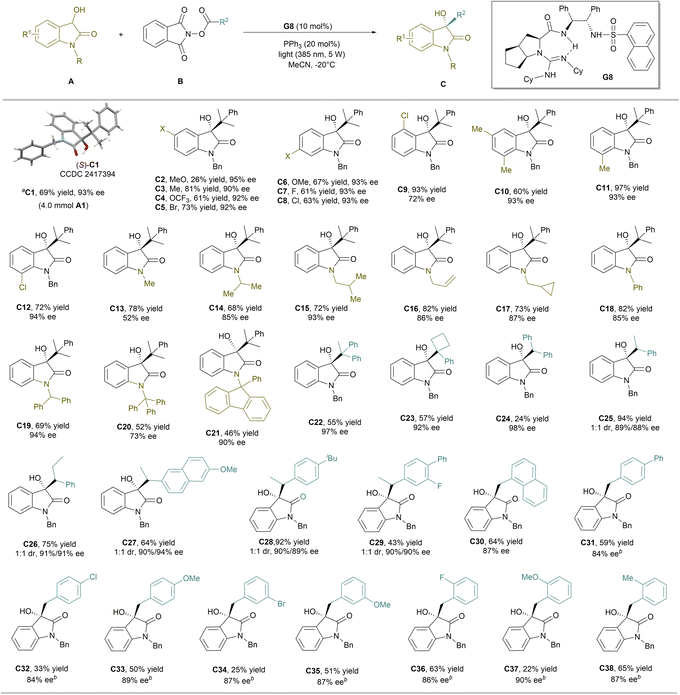
Next, a variety of 3-hydroxyoxindoles with NHPI ester B1 were subjected to the catalyst system of G8/PPh3 under the illumination of light. As shown in Scheme 2, N-benzyl-3-hydroxyindolinones, regardless of the electronic and steric properties of substituents located at the C5–C7 positions on the phenyl group, mostly proceeded well, and the desired products C3–C12 were isolated in moderate to good yields (60–97%) with excellent enantioselectivities (90–94% ee), except for the one bearing a 5-methoxyl substituent (C2). 4-Chloro-subsituted product C9 could be isolated in excellent yield, but the enantioselectivity slightly dropped probably due to the steric hindrance (72% ee). Moreover, 3-hydroxyoxindoles containing different N-substitutions could be well tolerated, delivering a series of chiral 3-alkylated 3-hydroxyindolinones with good results (C14–C21, 46–82% yield, 85–94% ee), except for less hindered N-methyl (C13) and bulky N-triphenylmethyl (C20) substituted ones, whose enantioselectivity decreased a lot.
The catalytic system could be extended to other NHPI esters (Scheme 2). NHPI esters synthesized from bulky tertiary carboxylic acids were compatible in the reaction with 3-hydroxyoxindole A1 (55–57% yield, 92–97% ee), with representative examples such as 1,1-diphenylethyl (C22) and 1-phenylcyclobutyl (C23) substituted ones. The product C24 from a diphenylmethyl-bearing precursor was isolated in up to 98% ee, although the isolated yield was not good, partly due to the isolation problem raised by its solubility. The alkyl/arylmethyl-containing NHPI esters, such as 1-phenylethyl and 1-phenylpropanyl, as well those derived from naproxen, ibuprofen and flurbiprofen, could deliver the related alkylation adducts (C25–C29) in good yields. Although the diastereoselectivities are not good, each diastereomer could be isolated with excellent enantioselectivities (88–94% ee). In addition, NHPI ester derivatives of 2-(naphthalen-1-yl)acetic acid and various substituted 2-phenylacetic acids, regardless of electron-withdrawing or electron-donating groups at ortho-, meta- or para-positions, also performed well to afford the coupling products with moderate to good yields and excellent ee values (C30–C38, 84–90% ee). For benzyl radicals derived from 4-chloro (C32) and 3-bromo (C34) substituents, the reaction system generates oxidation byproduct isatins, resulting in a low yield of the target product. The synthesis of product C37 suffered from a low yield due to the poor reactivity. The absolute configuration of C1 was determined to be (S) by X-ray diffraction analysis.24 To explore the synthetic utility of this methodology, gram-scale synthesis and transformations of the products were conducted. The alkylation of 3-hydroxyoxindole A1 at a 4.0 mmol scale occurred smoothly, giving the product C1 in 69% yield with 93% ee under the standard reaction conditions.
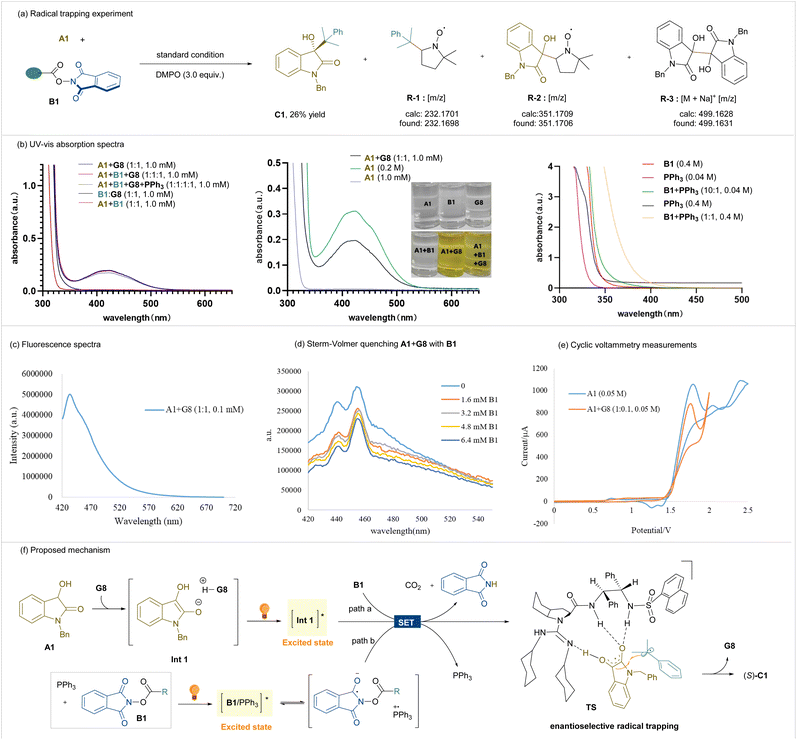
Control experiments and mechanistic studies were carried out in order to reveal the process of the reaction. The radical trapping reaction using 5,5-dimethyl-1-pyrroline N-oxide (DMPO) was carried out under the standard conditions (Scheme 3a). The formation of the product C1 was decreased to 26% yield, and radical species R-1, R-2 and homo-coupling species R-3 were identified based on high-resolution mass spectroscopy (HRMS) analysis. Electron paramagnetic resonance (EPR) measurement using DMPO as a trapping reagent further confirmed the presence of a stable alkyl radical from NHPI ester B1 and radical species derived from 3-hydroxyloxindole (see the SI for details).
The UV-vis absorption spectra of the reaction components were characterized as shown in Scheme 3b. There is no obvious absorption in any single or mixture systems of the two substrates in MeCN in the visible-light region; only A1 at high concentration showed obvious absorption (Scheme 3b, left and middle). The mixture of A1 and G8 exhibited obvious absorption in the visible light band, and the addition of PPh3 contributed little to this absorption (Scheme 3b, middle). This is consistent with the observed solution color change, in that the combination of substrate A1 and the guanidine catalyst led to a yellow solution. We rationalized that this visible-light band is generated from the enolized form of A1, which could be accelerated by the basic guanidine catalyst. This enolization is supported by the 1H NMR spectra analysis, in which the C3-hydrogen of 3-hydroxyl oxindole A1 disappeared upon the addition of guanidine G8 (see the SI for details). The formation of an electron donor–acceptor (EDA) complex between enolized-A1 and NHPI ester B1 is ruled out.8 From the absorption spectra of the B1/PPh3 mixture (Scheme 3b, right), it is revealed that the interaction between NHPI ester and phosphine exists, which could be excited under light irradiation in the reaction. This bathochromic shift is in line with Fu's spectra.8
The excitation of the substrate A1 as well as its interaction with the guanidine catalyst could also been confirmed from fluorescence emission spectra (Scheme 3c). Upon irradiation with 410 nm light, the guanidine-combined system yielded the maximum emission wavelength at 433 nm. The Stern–Volmer quenching studies revealed that redox-active ester B1 effectively quenched the excited state of oxindole-species of A1/G8 (Scheme 3d). Cyclic voltammetry analysis was conducted (Scheme 3e) and it revealed that the oxidation potential of G8/A1 was around 1.61 V. According to the Rehm–Weller equation, the excited state oxidation potential of G8/A1 was calculated [E0ox* = −1.25 V vs. Ag/AgCl in MeCN] (see the SI for details), which is able to reduce B1 [E0red = −1.21 V vs. Ag/AgCl in MeCN]. Finally, light on/off experiments showed that the continuous irradiation was essential for the reaction, and the quantum yield was found to be 5.41 × 10−3, suggesting that a radical chain mechanism via a radical addition pathway may not be the predominant process (see the SI for details).
Based on the mechanistic studies, we proposed a possible mechanism as shown in Scheme 3f. First, the basic guanidine unit promotes the enolization and deprotonation of 3-hydroxyoxindole, forming a zwitterionic intermediate Int 1. Then, this Int 1 absorbs light to reach its excited state and serves as a reductant to take part in the SET process with NHPI ester B1, affording an enol radical and an alkyl radical pair, respectively, after carbon dioxide and isoindoline-1,3-dione are released (path a). With the assistance of PPh3, the complex with B1 is excited, allowing the SET process to generate a radical cation of PPh3 and a radical anion of NHPI ester, simultaneously. The latter species decomposes into an alkyl radical intermediate, and the former species readily undergoes SET as an electron shuttle with excited Int 1 (path b) to yield the enol radical. This pathway might reduce the back-electron transfer between enol and NHPI ester, benefiting the following radical cross-coupling. Overall, the formed enol radical is trapped tightly by an intermolecular H-bond network with the guanidine (H-bond acceptor) and two amides (H-bond donors). As shown in the proposed transition state TS, one face of the enol radical species is blocked by the guanidine unit. The subsequent enantioselective outer-sphere radical coupling occurs at the Re-face, generating the final product (S)-C1 and releasing the chiral guanidine catalyst.
Conclusions
In summary, we have developed a photocatalytic charge-transfer enantioselective radical cross coupling reaction of 3-hydroxyoxindoles with NHPI esters under the catalysis of an organic chiral guanidine with the assistance of a catalytic amount of PPh3. A series of chiral 3-alkyl-substituted 3-hydroxyoxindole derivatives were obtained in good yields with excellent ee values. Detailed mechanistic experiments have elucidated the reaction process, which involved the excitation of the enol intermediate of 3-hydroxyoxindole and chiral guanidine, as well as the complex of NHPI ester with PPh3, respectively, followed by the SET processes as a reactive reductant and oxidant to generate stable radical coupling partners, and an enantioselective radical coupling process between two types of radical species through an outer-sphere mechanism.Author contributions
X. H. L. and W. D. C. guided the research. Y. J. S. conducted the experiments, analyzed the results, and wrote the SI and manuscript. R. F. W. participated in some experiments and the discussions. X. L. repeated some experiments. Y. Q. Z. finished the single crystal diffraction analysis. X. H. L., X. M. F. and W. D. C. helped revise the SI and manuscript. All the authors contributed to the discussion.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2417394 (C1) contains the supplementary crystallographic data for this paper.24Supplementary information (SI): the experimental procedure, 1H, 13C{1H} and 19F{1H} NMR, UPCC and HPLC spectra, and X-ray crystallographic data for C1. See DOI: https://doi.org/10.1039/d6sc00272b.
Acknowledgements
We appreciate the National Natural Science Foundation of China (22188101) for financial support.Notes and references
- (a) A. Studer and D. P. Curran, Angew. Chem., Int. Ed., 2016, 55, 58–102 CrossRef CAS PubMed; (b) J. Xie, H. M. Jin and A. S. K. Hashmi, Chem. Soc. Rev., 2017, 46, 5193–5203 RSC; (c) H. Yi, G. T. Zhang, H. M. Wang, Z. Y. Huang, J. Wang, A. K. Singh and A. W. Lei, Chem. Rev., 2017, 117, 9016–9085 CrossRef CAS PubMed; (d) J. M. Smith, S. J. Harwood and P. S. Baran, Acc. Chem. Res., 2018, 51, 1807–1817 CrossRef CAS PubMed; (e) D. Leifert and A. Studer, Angew. Chem., Int. Ed., 2020, 59, 74–108 CrossRef CAS PubMed; (f) S. Mondal, F. Dumur, D. Gigmes, M. P. Sibi, M. P. Bertrand and M. Nechab, Chem. Rev., 2022, 122, 5842–5976 CrossRef CAS PubMed; (g) G. A. Dawson, E. H. Spielvogel and T. N. Diao, Acc. Chem. Res., 2023, 56, 3640–3653 CrossRef CAS PubMed; (h) Y. X. Cao and G. D. Liu, Adv. Synth. Catal., 2023, 365, 3044–3062 CrossRef CAS; (i) T. Bauer, Y. Z. Hakim and P. Morawska, Molecules, 2023, 28, 6252 CrossRef CAS PubMed; (j) P. Z. Wang, B. Zhang, W. J. Xiao and J. R. Chen, Acc. Chem. Res., 2024, 57, 3433–3448 CrossRef CAS PubMed; (k) Y. L. Yin, M. D. You, X. T. Li and Z. Y. Jiang, Chem. Soc. Rev., 2025, 54, 2246–2274 RSC.
- (a) K. Okada, K. Okamoto and M. Oda, J. Am. Chem. Soc., 1988, 110, 8736–8738 CrossRef CAS; (b) K. Okada, K. Okamoto, N. Morita, K. Okubo and M. Oda, J. Am. Chem. Soc., 1991, 113, 9401–9402 CrossRef CAS.
- (a) S. Murarka, Adv. Synth. Catal., 2018, 360, 1735–1753 CrossRef CAS; (b) S. Crespi and M. Fagnoni, Chem. Rev., 2020, 120, 9790–9833 CrossRef CAS PubMed; (c) S. K. Parida, T. Mandal, S. Das, S. K. Hota, S. D. Sarkar and S. Murarka, ACS Catal., 2021, 11, 1640–1683 CrossRef CAS; (d) S. Q. He, H. C. Li, X. L. Chen, I. B. Krylov, A. O. Terent’ev, L. B. Qu and B. Yu, Chin. J. Org. Chem., 2021, 41, 4661–4689 CrossRef CAS; (e) C. R. A. Nicolas and J. P. Lumb, Beilstein J. Org. Chem., 2024, 20, 346–378 CrossRef PubMed; (f) J. Li, B. Q. Cheng, X. M. Shu, Z. Xu, C. Y. Li and H. H. Huo, Nat. Catal., 2024, 7, 889–899 CrossRef CAS.
- (a) T. Qin, J. Cornella, C. Li, L. R. Malins, J. T. Edwards, S. Kawamura, B. D. Maxwell, M. D. Eastgate and P. S. Baran, Science, 2016, 352, 801–805 CrossRef CAS PubMed; (b) J. Cornella, J. T. Edwards, T. Qin, S. Kawamura, J. Wang, C. M. Pan, R. Gianatassio, M. Schmidt, M. D. Eastgate and P. S. Baran, J. Am. Chem. Soc., 2016, 138, 2174–2177 CrossRef CAS PubMed; (c) J. Echavarren, M. A. Y. Gall, A. Haertsch, D. A. Leigh, V. Marcos and D. J. Tetlow, Chem. Sci., 2019, 10, 7269–7273 RSC; (d) Y. Fu, R. Shang, Z. Z. Wang, G. Z. Wang and B. Zhao, Synlett, 2020, 31, 1221–1225 CrossRef.
- C. Wang, M. Z. Guo, R. P. Qi, Q. Y. Shang, Q. Liu, S. Wang, L. Zhao, R. Wang and Z. Q. Xu, Angew. Chem., Int. Ed., 2018, 57, 15841–15846 CrossRef CAS PubMed.
- W. Kong, C. Yu, H. An and Q. Song, Org. Lett., 2018, 20, 349–352 CrossRef CAS PubMed.
- (a) C. G. S. Lima, T. d. M. Lima, M. Duarte, I. D. Jurberg and M. W. Paixão, ACS Catal., 2016, 6, 1389–1407 CrossRef CAS; (b) G. E. M. Crisenza, D. Mazzarella and P. Melchiorre, J. Am. Chem. Soc., 2020, 142, 5461–5476 CrossRef CAS PubMed; (c) T. Tasnim, M. J. Ayodele and S. P. Pitre, J. Org. Chem., 2022, 87, 10555–10563 CrossRef CAS PubMed.
- M. C. Fu, R. Shang, B. Zhao, B. Wang and Y. Fu, Science, 2019, 363, 1429–1434 CrossRef CAS PubMed.
- (a) Y. Shen, N. Lei, C. Lu, D. Xi, X. Geng, P. Tao, Z. Su and K. Zheng, Chem. Sci., 2021, 12, 15399–15406 RSC; (b) C. Lu, D. Jing, Y. L. Shen, J. J. Luo and K. Zheng, Org. Chem. Front., 2022, 9, 4164–4170 RSC.
- (a) Y. J. Li, J. Yang, X. X. Geng, P. Tao, Y. L. Shen, Z. S. Su and K. Zheng, Angew. Chem., Int. Ed., 2022, 61, e202210755 CrossRef CAS PubMed; (b) C. Lu, R. Chen, R. Wang, D. Jing and K. Zheng, Org. Lett., 2023, 25, 750–755 CrossRef CAS PubMed.
- (a) L. J. Li, J. C. Zhang, W. P. Li, D. Zhang, K. N. Duanmu, H. Yu, Q. Ping and Z. P. Yang, J. Am. Chem. Soc., 2024, 146, 9404–9412 CrossRef CAS PubMed; (b) L. J. Li, J. C. Zhang, J. Y. Tang, H. Yu and Z. P. Yang, Angew. Chem., Int. Ed., 2025, 64, e202506883 CrossRef CAS PubMed.
- T. Li, Z. Xu, Y. L. Huang, W. S. Zu and H. H. Huo, J. Am. Chem. Soc., 2025, 147, 10999–11009 CrossRef CAS PubMed.
- (a) N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 CrossRef CAS PubMed; (b) M. Silvi and P. Melchiorre, Nature, 2018, 554, 41–49 CrossRef CAS; (c) K. Wang and W. Q. Kong, Chin. J. Chem., 2018, 36, 247–256 CrossRef CAS; (d) F. Parsaee, M. C. Senarathna, P. B. Kannangara, S. N. Alexander, P. D. E. Arche and E. R. Welin, Nat. Rev. Chem., 2021, 5, 486–499 CrossRef CAS PubMed; (e) J. Großkopf, T. Kratz, T. Rigotti and T. Bach, Chem. Rev., 2022, 122, 1626–1653 CrossRef PubMed; (f) F. X. Yang, T. Huang, Y. M. Lin and L. Gong, Chem Catal., 2024, 4, 100812 CAS.
- (a) M. Silvi, C. Verrier, Y. P. Rey, L. Buzzetti and P. Melchiorre, Nat. Chem., 2017, 9, 868–873 CrossRef CAS PubMed; (b) T. Uchikura, N. Kamiyama, T. Mouri and T. Akiyama, ACS Catal., 2022, 12, 5209–5216 CrossRef CAS; (c) L. Z. Hou, Y. Q. Zhou, H. Yu, T. Y. Zhan, W. D. Cao and X. M. Feng, J. Am. Chem. Soc., 2022, 144, 22140–22149 CrossRef CAS PubMed; (d) A. Bahamonde and P. Melchiorre, J. Am. Chem. Soc., 2016, 138, 8019–8030 CrossRef CAS PubMed.
- (a) L. Candish, M. Teders and F. Glorius, J. Am. Chem. Soc., 2017, 139, 7440–7443 CrossRef CAS PubMed; (b) E. de P. Beato, D. Spinnato, W. Zhou and P. Melchiorre, J. Am. Chem. Soc., 2021, 143, 12304–12314 CrossRef PubMed; (c) R. H. Li, Y. L. Zhao, Q. K. Shang, Y. Geng, X. L. Wang, Z. M. Su, G. F. Li and W. Guan, ACS Catal., 2021, 11, 6633–6642 CrossRef CAS; (d) X. D. Su, B. B. Zhang, Q. Liu, J. T. Cheng, Z. X. Wang and X. Y. Chen, Org. Lett., 2021, 23, 8262–8266 CrossRef CAS PubMed; (e) S. Shibutani, K. Nagao and H. Ohmiya, Org. Lett., 2021, 23, 1798–1803 CrossRef CAS PubMed; (f) G. Z. Wang, M. C. Fu, B. Zhao and R. Shang, Sci. China: Chem., 2021, 64, 439–444 CrossRef CAS; (g) J. Guo, Y. Xie, Z. M. Lai, J. Weng, A. S. C. Chan and G. Lu, ACS Catal., 2022, 12, 13065–13074 CrossRef CAS; (h) S. P. Panda, S. K. Hota, R. Dash, L. Roy and S. Murarka, Org. Lett., 2023, 25, 3739–3744 CrossRef CAS PubMed; (i) H. Zhao, X. Xi, T. Wu, Z. Chen, M. Wang and W. Yuan, Sci. China: Chem., 2024, 67, 3019–3028 CrossRef CAS; (j) S. Jin, Y. Li, R. Wang and K. Zheng, Eur. J. Org Chem., 2024, 27, e202400798 CrossRef CAS; (k) Z. Q. Zhu, W. Y. Zhang, X. L. Huang, Q. Li, Z. Z. Xu and H. Y. Rao, J. Org. Chem., 2025, 90, 2842–2847 CrossRef CAS PubMed.
- (a) V. U. Khuzhaev, I. Zhalolov, K. K. Turguniv, B. Tashkhodzhaev, M. G. Levkovich, S. F. Arpova and A. S. Shashkov, Chem. Nat. Compd., 2004, 40, 269–272 CrossRef CAS; (b) Varun, Sonam and R. Kakkar, Med. Chem. Commun., 2019, 10, 351–368 RSC; (c) Y. M. Khetmalis, M. Shivani, S. Murugesan and K. V. G. C. Sekhar, Biomed. Pharmacother., 2021, 141, 111842 CrossRef CAS PubMed.
- (a) T. Ishimaru, N. Shibata, J. Nagai, S. Nakamura, T. Toru and S. Kanemasa, J. Am. Chem. Soc., 2006, 128, 16488–16489 CrossRef CAS PubMed; (b) J. R. Chen, X. P. Liu, X. Y. Zhu, L. Li, Y. F. Qiao, J. M. Zhang and W. J. Xiao, Tetrahedron, 2007, 63, 10437–10444 CrossRef CAS; (c) A. V. Malkov, M. A. Kabeshov, M. Bella, O. Kysilka, D. A. Malyshev, K. Pluháčková and P. Kočovský, Org. Lett., 2007, 9, 5473–5476 CrossRef CAS PubMed; (d) D. Sano, K. Nagata and T. Itoh, Org. Lett., 2008, 10, 1593–1595 CrossRef CAS PubMed; (e) M. Retini, G. Bergonzini and P. Melchiorre, Chem. Commun., 2012, 48, 3336–3338 RSC; (f) B. M. Trost and K. Hirano, Org. Lett., 2012, 14, 2446–2449 CrossRef CAS PubMed; (g) Q. L. Wang, L. Peng, F. Y. Wang, M. L. Zhang, L. N. Jia, F. Tian, X. Y. Xu and L. X. Wang, Chem. Commun., 2013, 49, 9422–9424 RSC; (h) R. He, S. C. Wu, H. M. Tang, X. H. Huo, Z. L. Sun and W. B. Zhang, Org. Lett., 2018, 20, 6183–6187 CrossRef CAS PubMed; (i) X. Y. Chen, K. Q. Chen, D. Q. Sun and S. Ye, Chem. Sci., 2017, 8, 1936–1941 RSC; (j) Z. Y. Song, K. Q. Chen, X. Y. Chen and S. Ye, J. Org. Chem., 2018, 83, 2966–2970 CrossRef CAS PubMed; (k) Z. T. Yang, J. H. Zhao, W. L. Yang and W. P. Deng, Org. Lett., 2019, 21, 1015–1020 CrossRef CAS PubMed.
- Y. J. Song, Y. Q. Zhou, Z. Tan, Z. L. Jiao, X. M. Feng and X. H. Liu, Org. Chem. Front., 2024, 11, 6813–6818 RSC.
- (a) S. X. Dong, X. M. Feng and X. H. Liu, Acc. Chem. Res., 2025, 58, 3463–3479 CrossRef CAS PubMed; (b) S. X. Dong, X. M. Feng and X. H. Liu, Chem. Soc. Rev., 2018, 47, 8525–8540 RSC; (c) X. He, Y. H. Fu, R. Y. Xi, C. F. Zhang, K. X. Lan, Z. S. Su, F. Wang, X. M. Feng and X. H. Liu, Angew. Chem., Int. Ed., 2025, 64, e202417636 CrossRef CAS PubMed; (d) S. Xin, J. B. Liao, Q. Tang, X. M. Feng and X. H. Liu, Chem. Sci., 2024, 15, 18557–18563 RSC; (e) Q. P. Chen, Y. Zhang, Y. J. Song, Y. Zhang, Z. S. Su, X. M. Feng and X. H. Liu, Org. Lett., 2024, 26, 3366–3370 CrossRef CAS PubMed; (f) Y. Y. Zhang, L. C. Ning, T. X. Zhu, Z. Xie, S. X. Dong, X. M. Feng and X. H. Liu, Org. Chem. Front., 2024, 11, 2897–2904 RSC; (g) Y. Y. Zhang, J. Wu, L. C. Ning, Q. P. Chen, X. M. Feng and X. H. Liu, Sci. China: Chem., 2023, 66, 526–533 CrossRef CAS; (h) J. Z. Li, L. C. Ning, Q. F. Tan, X. M. Feng and X. H. Liu, Org. Chem. Front., 2022, 9, 6312–6318 RSC.
- (a) A. G. Doyle and E. N. Jacobsen, Chem. Rev., 2007, 107, 5713–5743 CrossRef CAS PubMed; (b) R. S. J. Proctor, A. C. Colgan and R. J. Phipps, Nat. Chem., 2020, 12, 990–1004 CrossRef PubMed; (c) C. C. J. Loh, Nat. Rev. Chem., 2021, 5, 792–815 CrossRef CAS PubMed; (d) D. Parmar, E. Sugiono, S. Raja and M. Rueping, Chem. Rev., 2014, 114, 9047–9153 CrossRef CAS PubMed; (e) A. J. Metrano and S. J. Miller, Acc. Chem. Res., 2019, 52, 199–215 CrossRef CAS PubMed; (f) W. Wen and Q. X. Guo, Acc. Chem. Res., 2024, 57, 776–794 CrossRef CAS PubMed; (g) I. O. Betinol, Y. Kuang, B. P. Mulley and J. P. Reid, Chem. Rev., 2025, 125, 4184–4286 CrossRef CAS PubMed.
- (a) C. Xie, A. J. Smaligo, X. R. Song and O. Kwon, ACS Cent. Sci., 2021, 7, 536–558 CrossRef CAS PubMed; (b) W. Y. Huang and H. Z. Zhang, J. Fluorine Chem., 1990, 50, 133–140 CrossRef CAS; (c) L. Helmecke, M. Spittler, K. Baumgarten and C. Czekelius, Org. Lett., 2019, 21, 7823–7827 CrossRef CAS PubMed; (d) L. Zhao, Y. Huang, Z. Wang, E. Zhu, T. Mao, J. Jia, J. Gu, X. F. Li and C. Y. He, Org. Lett., 2019, 21, 6705–6709 CrossRef CAS PubMed.
- Z. W. Zhong, H. D. Wu, X. F. Chen, Y. Luo, L. Q. Yang, X. M. Feng and X. H. Liu, J. Am. Chem. Soc., 2024, 146, 20401–20413 CrossRef CAS PubMed.
- (a) Y. T. Wang, M. C. Fu, B. Zhao, R. Shang and Y. Fu, Chem. Commun., 2020, 56, 2495–2498 RSC; (b) C. Liu, N. Shen and R. Shang, Org. Chem. Front., 2021, 8, 4166–4170 RSC; (c) J. X. Wang, M. C. Fu, L. Y. Yan, X. Lu and Y. Fu, Adv. Sci., 2024, 11, 2307241 CrossRef CAS PubMed.
- CCDC 2417394: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m4hgr.
| This journal is © The Royal Society of Chemistry 2026 |