Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Reversible iodine capture by nonporous adaptive hybrid[3]arene crystals
Ruike Zhang,
Wenqian Liu and
Jiong Zhou *
*
Department of Chemistry, College of Sciences, Northeastern University, Shenyang 110819, China. E-mail: zhoujiong@mail.neu.edu.cn
First published on 14th April 2026
Abstract
Iodine capture is of great importance for environmental protection and chemical safety, motivating the development of efficient materials for iodine adsorption. Herein, we report an adsorption strategy that uses nonporous adaptive crystals based on novel hybrid[3]arene 1 (1α). 1α exhibits the ability to effectively capture iodine from both iodine vapor and iodine dissolved in n-hexane, reaching maximum capacities of 1.87 g g−1 and 0.74 g g−1, respectively. The adsorption of iodine molecules by 1α is driven by C–H⋯I and O–H⋯I interactions. Notably, 1α can be effectively recycled at least 10 times without a significant reduction of iodine capture capacity. It is anticipated that this work will provide valuable insights into the design of nonporous adaptive crystals as versatile platforms for radioactive iodine capture with recyclability.
Introduction
Radioactive iodine, a byproduct of the nuclear fission process, poses a weighty and long-lasting threat to the environment and human health due to its extended half-life and volatile nature.1,2 The potential release of radioactive iodine can occur during nuclear incidents or leakages, and once released, this hazardous substance can readily permeate and accumulate within food chains, ultimately endangering human life.3–9 While nuclear energy is often regarded as a sustainable and relatively eco-friendly resource, particularly in the context of mitigating global warming caused by excessive fossil fuel consumption, the associated risks of nuclear leakage cannot be overlooked.10–13 Given the severe and enduring consequences of radioactive iodine contamination, there is an urgent necessity to advance the development of innovative materials that can efficiently capture and securely store this radioactive element.14,15Nowadays, considerable research efforts have been directed towards developing advanced materials capable of efficiently capturing and storing iodine in various forms.16–19 Among them, porous materials including metal–organic frameworks,20–22 covalent organic frameworks,23–25 porous polymer networks,26,27 and hyper-cross-linked polymers are used for iodine capture applications.28 These materials exhibit high specific surface areas and tunable pore environments, which facilitate selective molecular recognition and exhibit strong adsorption capacities.29 However, their practical applications are limited by adsorption reliability and long-term stability.30,31
To overcome the problems in practical applications, the development of stable and efficient adsorbents is essential. Previous studies have shown that dense organic crystals can undergo guest-induced structural adjustments despite lacking permanent porosity, indicating that lattice adaptability is an intrinsic feature of certain organic solids.32 Building on these foundational observations, recent efforts have sought to systematize and extend this behaviour into what are now described as nonporous adaptive crystals (NACs), which provide a versatile platform for selective molecular capture.33,34 These crystals initially possess no intrinsic pores, but are capable of undergoing structural transformations in response to guest molecules.35–39 Consequently, NACs can dynamically form adaptable voids or channels that selectively accommodate specific guest molecules based on their sizes, shapes, and chemical characteristics.40–43 This dynamic structural adaptability, facilitated by supramolecular host–guest interactions, renders NACs promising materials for molecular capture applications.44–52 Notably, by harnessing non-covalent interactions between iodine molecules and polar groups, these crystals can spontaneously form optimized binding sites for efficient iodine capture.53–55
Herein, we designed and synthesized a novel hybrid[3]arene 1, characterized by a combination of one 4,4′-biphenol diethyl ether unit and two 3,4,5-trimethoxyphenol units, connected through methylene groups. Based on this unique structure of 1, we constructed NACs of hybrid[3]arene (1α), which demonstrated iodine capture capability. 1α effectively captured both iodine vapor and iodine dissolved in n-hexane. Structural analysis revealed that 1α underwent adaptive reorganization into an orderly structure, which was stabilized by C–H⋯I and O–H⋯I interactions upon iodine capture. Remarkably, 1α maintained the iodine capture efficiency for at least 10 times without significant performance loss.
Results and discussion
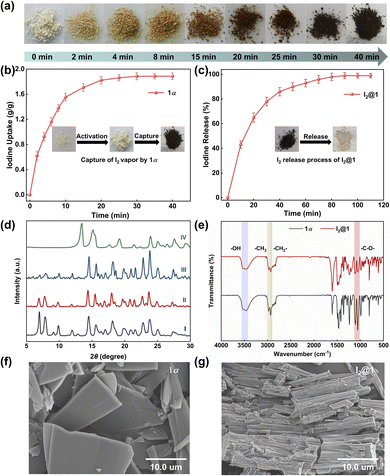
Generally, 1 was synthesized by 4,4′-biphenol diethyl ether (1.0 eq.), 3,4,5-trimethoxyphenol (2.0 eq.), and paraformaldehyde (3.0 eq.) in chloroform, catalyzed by trifluoroacetic acid (TFA, 10.0 eq.). The reaction mixture was refluxed at 65 °C for 50 min, generating 1 as a white solid with a yield of 60% (Fig. 1a). The chemical constitution of 1 was characterized by 1H NMR, 13C NMR, high-resolution mass spectrometry and single crystal X-ray diffraction (SCXRD) analysis (Fig. S1–S3 and Table S1). As shown in Fig. 1b and c, the 1 molecule consisted of one 4,4′-biphenol diethyl ether unit and two 3,4,5-trimethoxyphenol units. These units were bridged by methylene linkers, resulting in a twisted trigonal prismatic structure. Specifically, the biphenyl moiety within the 4,4′-biphenol diethyl ether unit exhibited a twisted conformation, where the two phenyl rings were rotated at a dihedral angle of 40.7° (Fig. S4). Furthermore, the two 3,4,5-trimethoxyphenol units were mutually inclined and located in distinct planes.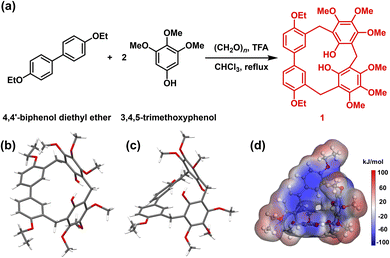 | ||
| Fig. 1 (a) Synthetic method of hybrid[3]arene (1). Crystal structure of 1 on (b) top view and (c) side view. (d) Electrostatic potential map of 1. | ||
The hydroxyl substituents on these phenolic rings were tilted and displaced from the aromatic plane. The electrostatic potential of 1 was partitioned into negative charge regions on the benzene rings and positive charge regions on the methoxy and ethoxy functional groups (Fig. 1d). Electron-rich benzene rings can enhance iodine adsorption by interacting with the localized positive electrostatic potential regions of iodine molecules. Prior to the experiment of iodine capture, 1 was dried under vacuum at 150 °C for 4 h to get activated crystalline 1 (1α). Thermogravimetric analysis (TGA, Fig. S5) verified that the solvent was removed. The powder X-ray diffraction (PXRD, Fig. S6) and N2 absorption–desorption curve showed that 1α was a nonporous crystal, with a Brunauer–Emmett–Teller surface area of 1.03 m2 g−1 (Fig. S7).
To evaluate the adsorption effect of the newly synthesized 1α on iodine, the iodine vapor capture experiment was conducted at 1 bar. As depicted in Fig. 2a, when 1α was exposed to iodine vapor at 77 °C, the color of the solid progressively darkened over time. As illustrated in Fig. 2b, the iodine uptake rate of 1α increased nearly linearly during the first 10 min, and then reached the adsorption saturation state at about 40 min with a maximum iodine uptake capacity of 1.87 g g−1. Although the iodine uptake capacity of 1α was comparable to that of many nonporous solids, the adsorption rate of 1α was faster than that of many previously reported nonporous materials (Table S3). Likewise, the iodine release experiment was investigated at 125 °C, and almost all the iodine was removed at 110 min (Fig. 2c). The time-dependent PXRD patterns of 1α changed over time, but remained unchanged after 10 min (Fig. 2d, S6, and S8), reflecting structural adaptation of 1α to guest uptake.
To elucidate the changes in the crystal morphology and elemental distribution of 1α after iodine capture, analyses including scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS) mapping, and Fourier transform infrared spectroscopy (FT-IR) were undertaken. SEM images showed that 1α exhibited a regular block-like particle morphology with smooth surfaces (Fig. 2f). Notably, after iodine loading, the microstructure of the iodine-loaded 1α (I2@1) displayed an elongated rod-like stacking arrangement (Fig. 2g), which may provide more accessible sites for iodine adsorption, thereby contributing to the high capture capacity. EDS elemental mapping confirmed that iodine was uniformly distributed in I2@1 (Fig. S9–S11). The FT-IR spectra of 1α and I2@1 showed only slight variations upon iodine loading. The peaks located near 3470 cm−1 and 2990–2998 cm−1, assigned to O–H and C–H stretching vibrations, respectively, remained basically unchanged, while the band at ∼1100 cm−1 showed a small decrease in intensity after iodine uptake, but recovered to a certain extent after release (Fig. 2e and S19). The thermal stability and iodine adsorption capacity of 1α were evaluated by TGA. Obviously, 1α exhibited no significant weight loss below 300 °C, confirming its high thermal stability. However, the weight loss of I2@1 in the range of 25–220 °C was about 66 wt%, corresponding to an uptake of approximately 4.9 mol/1α (Fig. S12). Additionally, the Raman spectrum of I2@1 in the low-frequency region (40–200 cm−1) showed a peak at 167 cm−1 (Fig. S13), which corresponded to the stretching vibration of iodine molecules.
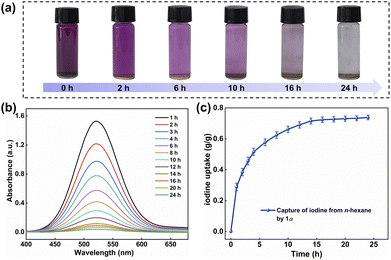
Given the excellent solubility of iodine in most organic solvents, the effective capture of iodine from solvents is also important.56–59 Therefore, we investigated the iodine capture performance of 1α in n-hexane. At 25 °C, 5 mg of 1α was added into the solution of iodine in n-hexane (5 mmol L−1, 3 mL). As shown in Fig. 3a, the color of 1α changed from white to yellow within 24 h, while the color of the solution transitioned from deep purple to transparent. The UV-vis spectra of the solution over the adsorption period were exhibited in Fig. 3b. After 24 h, the uptake of iodine from n-hexane by 1α reached saturation, with a maximum capacity of 0.74 g g−1 (Fig. 3c). What's more, 1α could be easily recycled after iodine capture. When I2@1 was immersed in methanol, the iodine was rapidly released and the solution turned brown within 2 h (Fig. S16).
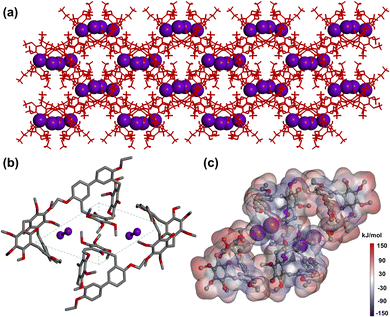
To elucidate the structural features after guest uptake, single crystals of iodine-loaded 1 (I2@1) were obtained by slowly evaporating a chloroform solution containing 1 and iodine. In I2@1, each 1 molecule adopted a slightly distorted geometry upon I2 capture. These molecules aligned cooperatively to form continuous channels within the crystal lattice (Fig. 4a). Fig. 4b illustrated the interaction between I2 and 1. The iodine molecule was not located in the cavity of 1 molecule, but was situated among three adjacent 1 molecules, forming a host–guest complex with a ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Furthermore, in the structure of I2@1, four 1 molecules adopted a centrosymmetric parallelogram arrangement, thereby creating cavities capable of accommodating I2 molecules. The main driving force for the formation of the host–guest complex after iodine capture came from the C–H⋯I and O–H⋯I interactions between 1 molecule and I2 molecule (C–H⋯I distances: 2.373 Å, 2.841 Å; O⋯I distances: 3.137 Å, 3.295 Å, 3.479 Å; Fig. S15). To further analyze these interactions, Hirshfeld surface analysis was performed for I2@1, which showed C–H⋯I and O–H⋯I interactions (Fig. S15). The electrostatic potential map of I2@1 revealed that the iodine molecule tended to be attracted by the oxygen atoms of the ethoxy groups (Fig. 4c).
1. Furthermore, in the structure of I2@1, four 1 molecules adopted a centrosymmetric parallelogram arrangement, thereby creating cavities capable of accommodating I2 molecules. The main driving force for the formation of the host–guest complex after iodine capture came from the C–H⋯I and O–H⋯I interactions between 1 molecule and I2 molecule (C–H⋯I distances: 2.373 Å, 2.841 Å; O⋯I distances: 3.137 Å, 3.295 Å, 3.479 Å; Fig. S15). To further analyze these interactions, Hirshfeld surface analysis was performed for I2@1, which showed C–H⋯I and O–H⋯I interactions (Fig. S15). The electrostatic potential map of I2@1 revealed that the iodine molecule tended to be attracted by the oxygen atoms of the ethoxy groups (Fig. 4c).
Recyclability is a pivotal metric for evaluating the performance of adsorbents.60,61 For practical applications, the iodine adsorbent must perform well over multiple cycles without degradation. The iodine molecule was released by heating I2@1 at 125 °C. As indicated in 1H NMR, PXRD and FT-IR (Fig. S17–S19), the structure of I2@1 was restored to the original 1α after iodine release. Satisfactorily, the newly formed 1α could continue to be reused for capturing iodine and could be recycled at least 10 times without significant loss of performance (Fig. 5a). The recycling experiments indicated that 1α exhibited exceptional reusability across multiple cycles, thereby reinforcing its potential as an efficient iodine adsorbent in practical applications (Fig. 5b).
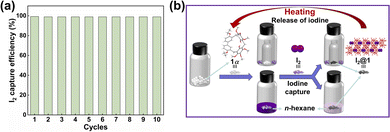 | ||
| Fig. 5 (a) The reusability of 1α in iodine capture. (b) Schematic diagrams represent the capture of iodine using 1α and the subsequent release of iodine. | ||
Conclusions
In conclusion, we synthesized a novel hybrid[3]arene 1. The NACs derived from 1 (1α) exhibited reversible iodine capture behaviour. It was found that 1α enabled capture capacity for both iodine vapor and iodine in n-hexane, with maximum capture capacities of 1.87 g g−1 and 0.74 g g−1, respectively. The single crystal structure and electrostatic potential analysis suggested that the capture ability mainly came from C–H⋯I and O–H⋯I interactions between 1 molecule and iodine molecule. In addition, during the adsorption process, the morphology of 1α tended to form a rod-like stacked multilayer structure, which was favourable for iodine uptake by 1α. Furthermore, the reversible transitions between guest-free and guest-loaded structures improved the recyclability of 1α, which retained its capture efficiency over 10 cycles without noticeable loss in performance. Given the uncomplicated synthesis, favourable adsorption behaviour, and good recyclability of 1α, this system enriches the study of hybrid[3]arene-based NACs for iodine capture. It is expected that this work will provide a general strategy for constructing hybridarene-based NACs as efficient adsorbents for radioactive iodine adsorption, and be applied to related environmental remediation.Author contributions
R. Z. and J. Z. designed the experiments. R. Z. performed most experiments. R. Z. and W. L. analysed data. R. Z. and J. Z. wrote the manuscript. All authors agreed with the results and discussions presented in the manuscript.Conflicts of interest
The authors declare no conflicts of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures and characterization data. See DOI: https://doi.org/10.1039/d6sc00236f.CCDC 2296036 and 2493736 contain the supplementary crystallographic data for this paper.62a,b
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22101043), the Fundamental Research Funds for the Central Universities (N25LPY027), and Northeastern University. Special thanks are due to the instrumental or data analysis from Analytical and Testing Center, Northeastern University.Notes and references
- X. Zhang, J. Maddock, T. M. Nenoff, M. A. Denecke, S. Yang and M. Schröder, Chem. Soc. Rev., 2022, 51, 3243–3262 RSC.
- S. U. Nandanwar, K. Coldsnow, V. Utgikar, P. Sabharwall and D. Eric Aston, Chem. Eng. J., 2016, 306, 369–381 CrossRef CAS.
- C. Muhire, A. Tesfay Reda, D. Zhang, X. Xu and C. Cui, Chem. Eng. J., 2022, 431, 133816 CrossRef CAS.
- N. Arora, T. Debnath, M. C. Senarathna, R. M. Johnson, I. G. Roske, G. A. Cisneros and R. A. Smaldone, Chem. Sci., 2024, 15, 3571–3577 RSC.
- A. Al-Mamoori, M. Alsalbokh, S. Lawson, A. A. Rownaghi and F. Rezaei, Chem. Eng. J., 2020, 391, 123583 CrossRef CAS.
- Z. Yu, Y. Lu, Y. Lu, T. Xiong, Y. Gao and X. Xiao, J. Mater. Chem. A, 2025, 13, 37358–37366 RSC.
- G. Chen, L. Wu, Z. Wang, R. Liu, C. Tan, Y. Tan, Y. Liu, M. He, H. Zhang, Y. Huang, J. Zhu, T. Duan and L. Zhu, Chem. Eng. J., 2025, 507, 160829 CrossRef CAS.
- S. Maji and R. Natarajan, Small, 2023, 19, 2302902 CrossRef CAS PubMed.
- J. Chang, H. Li, J. Zhao, X. Guan, C. Li, G. Yu, V. Valtchev, Y. Yan, S. Qiu and Q. Fang, Chem. Sci., 2021, 12, 8452–8457 RSC.
- K. W. Chapman, P. J. Chupas and T. M. Nenoff, J. Am. Chem. Soc., 2010, 132, 8897–8899 CrossRef CAS.
- M. V. Ravikumar and V. Lakshmi, J. Hazard. Mater., 2025, 500, 140421 CrossRef CAS.
- N. Brown, Z. Alsudairy, R. Behera, F. Akram, K. Chen, K. Smith-Petty, B. Motley, S. Williams, W. Huang, C. Ingram and X. Li, Green Chem., 2023, 25, 6287–6296 RSC.
- S. Maji, S. Pal and R. Natarajan, Small, 2025, 21, 2502761 CrossRef CAS.
- D. K. L. Harijan, V. Chandra, T. Yoon and K. S. Kim, J. Hazard. Mater., 2018, 344, 576–584 CrossRef CAS PubMed.
- D. F. Sava, M. A. Rodriguez, K. W. Chapman, P. J. Chupas, J. A. Greathouse, P. S. Crozier and T. M. Nenoff, J. Am. Chem. Soc., 2011, 133, 12398–12401 CrossRef CAS PubMed.
- X.-H. Xu, Y.-X. Li, L. Zhou, N. Liu and Z.-Q. Wu, Chem. Sci., 2022, 13, 1111–1118 RSC.
- M. El-Shahat, A. E. Abdelhamid and R. M. Abdelhameed, Carbohydr. Polym., 2020, 231, 115742 CrossRef CAS.
- B. Valizadeh, T. N. Nguyen, B. Smit and K. C. Stylianou, Adv. Funct. Mater., 2018, 28, 1801596 CrossRef.
- B. Mishra, S. Dutta, U. Pal, S. Rana, S. K. Mishra, T. Saha-Dasgupta and P. Pachfule, Small, 2025, 21, 2411199 CrossRef CAS PubMed.
- H. Furukawa, K. E. Cordova, M. O'Keeffe and O. M. Yaghi, Science, 2013, 341, 1230444 CrossRef PubMed.
- A. Karmakar, P. Samanta, A. V. Desai and S. K. Ghosh, Acc. Chem. Res., 2017, 50, 2457–2469 CrossRef CAS PubMed.
- S. Horike and S. Kitagawa, Nat. Mater., 2022, 21, 983–985 CrossRef CAS PubMed.
- S. Fajal, W. Mandal, A. Torris, D. Majumder, S. Let, A. Sen, F. Kanheerampockil, M. M. Shirolkar and S. K. Ghosh, Nat. Commun., 2024, 15, 1278 CrossRef CAS.
- N. Farooq, M. A. Malik and A. A. Hashmi, Chem. Eng. J., 2024, 498, 154894 CrossRef CAS.
- T. Skorjanc, D. Shetty and A. Trabolsi, Chem, 2021, 7, 882–918 CAS.
- E. R. Pezoulas, B. Tajdini, Y. Ko, A. A. Uliana, R. Giovine, H. Furukawa, H. Vatankhah, J. Börgel, K. C. Kim, C. Bellona and J. R. Long, J. Am. Chem. Soc., 2025, 147, 21832–21843 CrossRef CAS PubMed.
- M. Sun, J. Chen, T. Zhang, W. Xu, J. He, Y. Zhang, H. Liu, S. Zhang, J. Wang, X. Li, Y. Yang and H. Qiu, Chem. Sci., 2025, 16, 11858–11869 RSC.
- S. Bera, S. Sau, F. Banerjee, N. Kumar and S. K. Samanta, Sep. Purif. Technol., 2025, 352, 128123 CrossRef CAS.
- B. T. Benkhaled, A. Chaix, C. Gomri, S. Buys, N. Namar, N. Sehoulia, R. Jadhav, J. Richard, L. Lichon, C. Nguyen, M. Gary-Bobo and M. Semsarilar, ACS Appl. Mater. Interfaces, 2023, 15, 42942–42953 CrossRef CAS PubMed.
- M. Jia, S. Rong, P. Su and W. Li, Chem. Eng. J., 2022, 437, 135432 CrossRef CAS.
- W. Zhou, Y. Li, L. Xiong, W. Wang, R. Guan, Z. Chen, D. Yuan, E.-Q. Gao and D. Zhang, J. Am. Chem. Soc., 2025, 147, 35664–35674 CrossRef CAS PubMed.
- J. L. Atwood, L. J. Barbour, A. Jerga and B. L. Schottel, Science, 2002, 298, 1000–1002 CrossRef CAS PubMed.
- Y. Chao, T. U. Thikekar, W. Fang, R. Chang, J. Xu, N. Ouyang, J. Xu, Y. Gao, M. Guo, H. Zuilhof and A. C. H. Sue, Angew. Chem., Int. Ed., 2022, 61, e202204589 CrossRef CAS PubMed.
- Z. Hassan, J. Lahann and S. Bräse, Adv. Funct. Mater., 2024, 34, 2410027 CrossRef CAS.
- M. Rahmani, C. R. M. O. Matos, S.-Q. Wang, A. A. Bezrukov, A. C. Eaby, D. Sensharma, Y. Hjiej-Andaloussi, M. Vandichel and M. J. Zaworotko, J. Am. Chem. Soc., 2023, 145, 27316–27324 CrossRef CAS PubMed.
- S. P. Yelgaonkar, G. Campillo-Alvarado and L. R. MacGillivray, J. Am. Chem. Soc., 2020, 142, 20772–20777 CrossRef CAS PubMed.
- J. Chen, S. Wu, Y. Wang and J. Zhou, Chin. Chem. Lett., 2025, 36, 110102 CrossRef CAS.
- J.-R. Wu and Y.-W. Yang, Angew. Chem., Int. Ed., 2021, 60, 1690–1701 CrossRef CAS PubMed.
- J. Chen, W. Zhang, W. Yang, F. Xi, H. He, M. Liang, Q. Dong, J. Hou, M. Wang, G. Yu and J. Zhou, Nat. Commun., 2024, 15, 1260 CrossRef CAS PubMed.
- Z.-Y. Zhang and C. Li, Acc. Chem. Res., 2022, 55, 916–929 CrossRef CAS PubMed.
- M. Yan, Y. Wang, J. Chen and J. Zhou, Chem. Soc. Rev., 2023, 52, 6075–6119 RSC.
- M. Yin, C. Yang, D. Tang, S. Huang, X. Lou, R. Cui, C. Ye, J. Chen and T. Qiu, Chem. Eng. J., 2024, 495, 153508 CrossRef CAS.
- W. Yang, W. Zhang, J. Chen and J. Zhou, Chin. Chem. Lett., 2024, 35, 108740 CrossRef CAS.
- A. Dey, S. Chand, B. Maity, P. M. Bhatt, M. Ghosh, L. Cavallo, M. Eddaoudi and N. M. Khashab, J. Am. Chem. Soc., 2021, 143, 4090–4094 CrossRef CAS PubMed.
- J. Zhou, G. Yu, Q. Li, M. Wang and F. Huang, J. Am. Chem. Soc., 2020, 142, 2228–2232 CrossRef CAS PubMed.
- X.-N. Han, Y. Han and C.-F. Chen, Chem. Soc. Rev., 2023, 52, 3265–3298 RSC.
- Y. Wang, S. Wu, S. Wei, Z. Wang and J. Zhou, Chem. Mater., 2024, 36, 1631–1638 CrossRef CAS.
- W. Yang, K. Samanta, X. Wan, T. U. Thikekar, Y. Chao, S. Li, K. Du, J. Xu, Y. Gao, H. Zuilhof and A. C. H. Sue, Angew. Chem., Int. Ed., 2020, 59, 3994–3999 CrossRef CAS PubMed.
- Y. Wang, Z. Wang, S. Wei, S. Wu, M. Wang, G. Yu, P. Chen, X. Liu and J. Zhou, Mater. Chem. Front., 2024, 8, 2273–2281 RSC.
- D. Pei, W. Guo, P. Liu, T. Xue, X. Meng, X. Shu, J. Nie and Y. Chang, Chem. Eng. J., 2022, 433, 134463 CrossRef CAS.
- M. Yan, Y. Wang and J. Zhou, Cell Rep. Phys. Sci., 2023, 4, 101637 CrossRef CAS.
- M. Wang, S. Fang, S. Yang, Q. Li, N. M. Khashab, J. Zhou and F. Huang, Mater. Today Chem., 2022, 24, 100919 CrossRef CAS.
- D. Luo, Y. He, J. Tian, J. L. Sessler and X. Chi, J. Am. Chem. Soc., 2022, 144, 113–117 CrossRef CAS PubMed.
- K. Jie, Y. Zhou, E. Li, Z. Li, R. Zhao and F. Huang, J. Am. Chem. Soc., 2017, 139, 15320–15323 CrossRef CAS PubMed.
- Z. Wang, J. Li, Z. Du, L. Pang and C. Liu, Chem. Eng. J., 2025, 509, 161326 CrossRef CAS.
- M. Yadollahi, H. Hamadi and V. Nobakht, J. Hazard. Mater., 2020, 399, 122872 CrossRef CAS PubMed.
- M. Alsalbokh, N. Fakeri, S. Lawson, A. A. Rownaghi and F. Rezaei, Chem. Eng. J., 2021, 415, 128968 CrossRef CAS.
- G. Matthys, A. Laemont, N. De Geyter, R. Morent, R. Lavendomme and P. Van Der Voort, Small, 2024, 20, 2404994 CrossRef CAS PubMed.
- M. Dalapati, R. Singha, P. Maity, D. Manna and D. Samanta, Small, 2025, 21, 2504242 CrossRef CAS PubMed.
- M. Liang, J. Chen, Y. Chi, H. Zhu and J. Zhou, ACS Appl. Mater. Interfaces, 2026, 18, 3313–3321 CrossRef CAS PubMed.
- M. Wang, Q. Li, E. Li, J. Liu, J. Zhou and F. Huang, Angew. Chem., Int. Ed., 2021, 60, 8115–8120 CrossRef CAS PubMed.
- (a) CCDC 2296036: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2h26ph; (b) CCDC 2493736: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2ppy3g.
| This journal is © The Royal Society of Chemistry 2026 |