Open Access Article
Open Access ArticleOverriding the radical polarity matching principle: selective chlorohydroxylation of electron-deficient alkenes enabled by Ce photocatalysis
Luchuan Ou†
a,
Yihang Bai†a,
Shanshuo Liu†c,
Zongling Kanc,
Yu Lan *abd,
Jiali Zhua,
Shihan Liue,
Zhigang Ren*c,
Shi-Jun Li
*abd,
Jiali Zhua,
Shihan Liue,
Zhigang Ren*c,
Shi-Jun Li *ab and
Linbin Niu*ab
*ab and
Linbin Niu*ab
aCollege of Chemistry, State Key Laboratory of Antiviral Drugs and Pingyuan Laboratory, Zhengzhou University, Zhengzhou, Henan 450001, P. R. China. E-mail: lishijunzong@zzu.edu.cn; nlb@zzu.edu.cn
bState Key Laboratory of Antiviral Drugs, Henan Normal University, Xinxiang, Henan 453007, P. R. China
cDepartment of Infectious Diseases, State Key Laboratory of Antiviral Drugs, Pingyuan Laboratory, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, P. R. China. E-mail: fccrenzg@zzu.edu.cn
dSchool of Chemistry and Chemical Engineering, Chongqing Key Laboratory of Chemical Theory and Mechanism, Chongqing University, Chongqing 401331, P. R. China. E-mail: lanyu@cqu.edu.cn
eCollege of Chemistry and Molecular Sciences, Henan University, Kaifeng 475004, P. R. China
First published on 28th April 2026
Abstract
Radical addition to alkenes generally follows the polarity matching principle; for instance, the electrophilic chlorine radical favors electron-rich double bonds over electron-deficient ones in alkenes. Herein, we achieve a one-step selective aerobic chlorohydroxylation of alkenes via Ce-LMCT-generated chlorine radicals. The protocol exhibits a broad substrate scope, notably extending to electron-deficient, amide-containing alkenes—a class of substrates historically challenging for the addition of electrophilic radicals due to the polarity matching principle. Theoretical calculations reveal that the radical adduct generated from the chlorine radical and amide-containing alkene is stabilized by the amide group, which helps the system to override the radical polarity matching principle.
Introduction
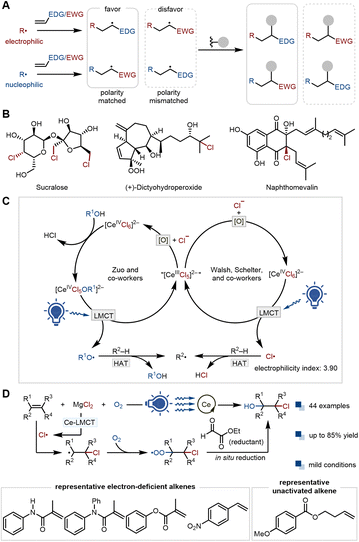
Radical addition to alkenes is a fundamental transformation in radical-mediated organic synthesis. Generally, a nucleophilic radical prefers to add to an electron-deficient alkene rather than an electron-rich one. Conversely, an electrophilic radical favors addition to electron-rich alkenes (Fig. 1A).1 This polarity matching rule governing radical addition to unsaturated bonds is important for designing highly selective radical relay reactions.2 However, the scope of these radical reactions may be limited by their reliance on the addition step. To circumvent the polarity matching rule, methods that allow, for example, the effective addition of electrophilic radicals to electron-deficient alkenes are highly desirable, as they would provide access to novel synthetic pathways.3C(sp3)–Cl bonds not only are extensively present in natural products, pharmaceuticals, agrochemicals, and functional materials,4 but also serve as an indispensable synthon in modern organic synthesis (Fig. 1B). Numerous catalytic strategies have been developed to construct C(sp3)–Cl bonds,5 among which chlorine radical-mediated approaches show intriguing reactivity and selectivity.6 The electrophilic chlorine radical (electrophilicity index: 3.90) exhibits a strong preference for electron-rich alkenes, which enables the elegant, anti-Markovnikov selective synthesis of various C(sp3)–Cl-containing chemicals.7 For instance, the Lin group achieved the dichlorination of electron-rich alkenes to vicinal dichlorides via electrocatalysis;8 the West group recently reported anti-Markovnikov hydrochlorination of electron-rich alkenes for the production of alkyl chlorides using iron photocatalysis.9 Nevertheless, the selective chlorohydroxylation of electron-deficient alkenes via chlorine radical addition remains under-explored.
Visible light-induced organic transformations have been rapidly developed and are highly attractive in organic synthesis due to their safety and environmental friendliness.10 As one of the prevalent electron transfer mechanisms under visible light, LMCT has garnered widespread attention from organic synthetic chemists. This process typically involves using specific wavelengths of light to excite the electrons from ligands to vacant metal orbitals, thereby generating ligand-derived radicals that can be utilized to facilitate various types of chemical reactions, such as addition to unsaturated bonds or activation of inert chemical bonds.11 To date, a wide range of metals have demonstrated the capability to mediate LMCT reactions.12 Among them, cerium, the most abundant lanthanide element in the earth's crust, possesses a unique electronic configuration [Xe] 4f15d16 s2.13 Cerium photocatalysis via the LMCT mechanism has flourished over the past decade.14 The Zuo group presented a highly representative paradigm: the cerium–alcohol complex generated in situ from cerium salts and alcohols undergoes homolysis to produce electrophilic alkoxy radicals under visible light irradiation. Alkoxy radical can abstract the hydrogen atom of C(sp3)–H bonds to generate nucleophilic alkyl radicals, which tend to react with electron-deficient unsaturated bonds, thereby fulfilling C(sp3)–H functionalization (Fig. 1C).15 In contrast, Walsh, Schelter, and their colleagues found that visible light-induced homolysis of cerium–chloride complexes generates chlorine radicals, which subsequently abstract hydrogen atoms from alkanes to form alkyl radicals (Fig. 1C).16 Notably, reports in which Ce-LMCT-generated chlorine radicals add to electron-deficient alkenes to form C(sp3)–Cl bonds are scarce.17
Thus, we sought to study the reaction of chlorine radicals with electron-deficient alkenes under the conditions of Ce-LMCT, aiming to unlock the selective chlorohydroxylation of electron-deficient alkenes. O2 was expected to intercept the radical adduct from the thermodynamically unfavorable addition of chlorine radical to electron-deficient alkenes.18 Theoretically, this step is also reversible, indicating that overcoming this reversibility to ensure the success of the desired one-step aerobic chlorohydroxylation may rely on a suitable reducing reagent to promptly and irreversibly convert the O–O intermediate into the alcohol.19 Herein, by combining ethyl glyoxylate as a unique reducing reagent and aerobic Ce-LMCT photocatalysis, we successfully unlock the selective chlorohydroxylation of electron-deficient alkenes in a single step. Moreover, using this system, unactivated alkyl alkenes can also be well tolerated.
Results and discussion
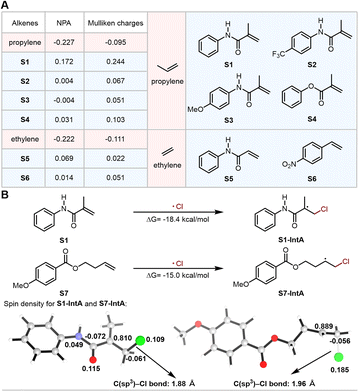
Given the ubiquitous presence of amide groups in natural products and pharmaceutical molecules,20 incorporating amide functionalities into the unsaturated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds of alkenes could further improve the potential value of the corresponding chlorohydrin products. Additionally, the electron-withdrawing character of the carbonyl in the amide group renders this kind of alkene electron-poor.21 Then, we evaluated the electronic properties of the alkenes containing amide groups using DFT calculations (Fig. 2A). Both the Mulliken and natural population analysis charge calculations of amide-containing alkenes exhibited more positive charges compared to those of propylene and ethylene. In addition to amide-containing alkenes, phenyl methacrylate and 4-nitrostyrene also show similar charge characteristics to the amide-containing alkenes. Furthermore, the spin density analysis also revealed that the amide group can effectively stabilize the alkyl radical S1-IntA generated from the addition of chlorine radical to N-phenylmethacrylamide (S1), which helps to override the radical polarity matching principle.
C double bonds of alkenes could further improve the potential value of the corresponding chlorohydrin products. Additionally, the electron-withdrawing character of the carbonyl in the amide group renders this kind of alkene electron-poor.21 Then, we evaluated the electronic properties of the alkenes containing amide groups using DFT calculations (Fig. 2A). Both the Mulliken and natural population analysis charge calculations of amide-containing alkenes exhibited more positive charges compared to those of propylene and ethylene. In addition to amide-containing alkenes, phenyl methacrylate and 4-nitrostyrene also show similar charge characteristics to the amide-containing alkenes. Furthermore, the spin density analysis also revealed that the amide group can effectively stabilize the alkyl radical S1-IntA generated from the addition of chlorine radical to N-phenylmethacrylamide (S1), which helps to override the radical polarity matching principle.
Subsequently, S1 was selected as the model substrate to investigate the conditions of the aerobic chlorohydroxylation. The target product, 3-chloro-2-hydroxy-2-methyl-N-phenylpropanamide 2a, was obtained in 78% isolated yield when employing MgCl2 as the chlorine source and ethyl glyoxylate (R1) as an in situ reductant (Scheme 1, Entry 1). A series of control experiments revealed that blue light irradiation, R1, Ce catalyst, and O2 are indispensable for the generation of 2a (Entries 2–6). The reaction under an air atmosphere gave a decreased yield of 60% (Entry 7). We further observed that the addition of 4 Å molecular sieves (4 Å MS, 20.0 mg) to the system resulted in a lower isolated yield of 54% (Entry 8); adding two equivalents of H2O did not inhibit the formation of product (Entry 9), and H2O could not replace R1 in this reaction (Entry 10). These results suggested that the hydrogen in the hydroxyl group is derived from R1. It was worth noting that ethyl pyruvate R2 failed to afford the chlorohydrin (Entry 11), whereas oxalic acid 1-ethyl ester R3 succeeded (Entry 12).
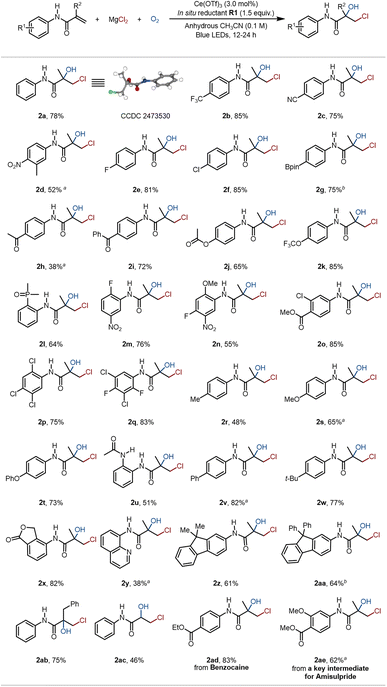
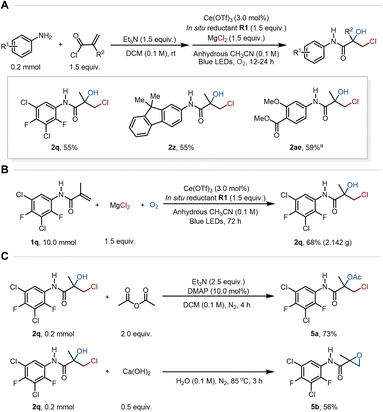
With the optimal conditions in hand, the reaction scope of the terminal alkenes containing amide scaffolds was systematically examined (Fig. 3). Under our standard conditions, a series of N-phenylmethacrylamides more electron-deficient than S1 were converted to the corresponding chlorohydrins in moderate to good yields (2b–2q), successfully accommodating not only strong electron-withdrawing groups (e.g., –CF3, –CN, –NO2; 2b–2d) but also medicinally relevant motifs (e.g., fluoro, trifluoromethoxy, phosphoryl; 2e, 2k, 2l). Most notably, even alkenes bearing multiple electron-withdrawing substituents (2m–2q) proved to be competent substrates. These results collectively indicated that our protocol effectively overcomes the reactivity constraints imposed by the classic polarity matching principle for radical additions. In addition, a range of alkenes with different electron-donating groups, such as –Me, –OMe, –OPh, –NHCOMe, –Ph, and –t-Bu, smoothly underwent this protocol to afford diverse chlorohydrin products (2r–2w). Given that the introduction of heterocycles is an effective strategy for developing lead compounds and drug candidates in medicinal chemistry, we evaluated relevant heterocyclic scaffolds, such as phthalide and quinoline, and successfully afforded the desired products 2x and 2y. The compatibility of the fluorene scaffold, which is of high interest in materials science, was also demonstrated, giving chlorohydrins 2z and 2aa. This result further highlights the excellent functional group tolerance of this method. The one-step chlorohydroxylation was also successful on substrates where the methyl group of S1 was replaced by benzyl or removed, delivering 2ab and 2ac, respectively. This method also efficiently enabled the selective transformation of the alkenes derived from commercial drug Benzocaine and from the key intermediate for Amisulpride into the desired chlorohydrins 2ad and 2ae in good yields, further underscoring its robustness.
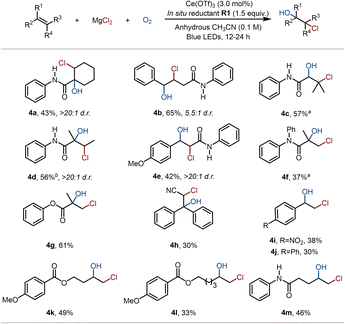
Besides the terminal amide-based alkenes, the substrate scope was also found to cover diverse internal alkenes (Fig. 4), such as those derived from 1-cyclohexene-1-carboxylic acid (4a), trans-styrylacetic acid (4b), 3,3-dimethylacrylic acid (4c), tiglic acid (4d), and trans-4-methoxycinnamic acid (4e), all of which afforded the corresponding chlorohydrins in moderate to good yields. Notably, the replacement of the N–H moiety in S1 with an N–Ph group was tolerated, furnishing product 4f in 37% yield. Inspired by this result, the amide was replaced with an ester group, which provided product 4g in 61% yield, indicating that the amide scaffold is not essential for the reaction. Furthermore, the broad substrate scope was underscored by the successful conversion of diverse alkene types, including electron-deficient styrenes (4h and 4i), conjugated aryl alkenes (4j) and unactivated alkyl alkenes (4k–4m).
Encouraged by the positive experimental results discussed above, we further demonstrated the synthetic utility of this methodology. A one-pot procedure was developed, wherein anilines bearing different functional groups were condensed with methacryloyl chloride to generate the amide-containing alkene substrates in situ, and then directly subjected to chlorohydroxylation without purification (Fig. 5A; 55–59% yields for 2q, 2z, and 2ae). This process effectively simplifies the synthetic workflow by eliminating the need for column chromatography isolation of the intermediate alkene. Furthermore, a gram-scale reaction was conducted smoothly under the standard conditions, delivering the product 2q in 68% yield (Fig. 5B). The product from this gram-scale experiment was then used in subsequent transformations. It was successfully converted to the ester 5a in 73% yield and epoxide product 5b in 56% yield, respectively (Fig. 5C).
To gain insight into the mechanism, several mechanistic experiments were conducted. Light on/off experiments confirmed that continuous blue light irradiation is essential for the reaction to proceed (Fig. 6A). UV-vis spectroscopy was also employed to gain insight into the in situ generation of the Ce-based light-absorbing system (Fig. 6B). It was interesting that irradiation of a Ce(OTf)3 and MgCl2 mixture with blue light under O2 for 1 hour produced a Ce(IV)-based absorption peak in the blue light region, whereas no such peak was observed under N2 atmosphere. Consequently, O2 can be identified as promoting the in situ generation of the Ce(IV)-based light-absorbing system from Ce(III) under blue light irradiation.15a,16a Furthermore, 18O-labelling experiments also demonstrated that the oxygen atom incorporated into the product is indeed derived from O2 (Fig. S10). The inhibition of the chlorohydroxylation of alkenes by both 2,6-di-tert-butyl-4-methylphenol (BHT) and 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO) suggested that the reaction proceeds via a radical pathway (Fig. 6C). Notably, dichlorination of alkenes was observed when using stoichiometric Ce(OTf)4 and MgCl2 under blue LEDs irradiation (Fig. 6D). N-Fluorobenzenesulfonimide (NFSI) was further found to be able to complete the catalytic cycle of Ce. These results indicated that chlorine radicals are crucial intermediates generated by the Ce photocatalysis.16a,22 Last but not least, the direct use of Ce(OTf)4 or (Et4N)2CeCl6 as catalysts also afforded the desired chlorohydroxylation in good yields.
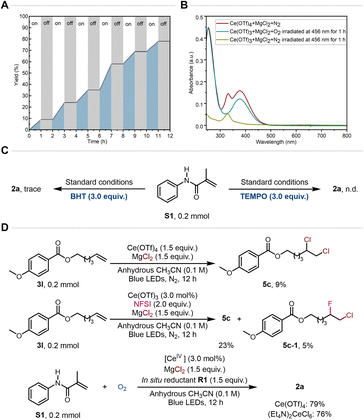 | ||
| Fig. 6 Mechanistic studies. (A) Light on/off experiments. (B) UV-vis studies. (C) Radical inhibition experiments. (D) Determination of chlorine radical generation via Ce(IV)–Cl LMCT. | ||
Conclusion
In conclusion, we have developed a one-step aerobic selective chlorohydroxylation protocol that overcomes the limitations imposed by the polarity matching principle for the chlorine radical addition to electron-deficient alkenes. The practical utility and broad application value of this protocol are evidenced by its excellent functional group tolerance, successful late-stage functionalization of commercial drug derivatives, gram-scale synthesis, and one-pot procedure. Further efforts toward developing novel chlorine radical transformation platforms are ongoing in our laboratory.Author contributions
Conceptualization: L. O., Y. B., S. L., Z. K., Y. L., J. Z., Z. R., S.-J. L., L. N.; methodology: L. O., Y. B., Y. L., J. Z., S.-J. L., L. N.; investigation: L. O., Y. B., S. L., Z. K., Y. L., J. Z., Z. R., S. L., L. N.; supervision: Y. L., Z. R., S.-J. L., L. N.; writing—original draft: L. O., Y. B., Y. L., S.-J. L., L. N.; writing—review & editing: L. O., Y. B., S. L., Z. K., Y. L., J. Z., S. L., Z. R., S.-J. L., L. N.Conflicts of interest
The authors declare that they have no conflict of interest.Data availability
CCDC 2473530 contains the supplementary crystallographic data for this paper.23The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc00166a.
Acknowledgements
This project was supported by the National Natural Science Foundation of China (Grants No. 22571284, 22531008, and 22101265); Henan Provincial Joint Fund for Science and Technology Research and Development (Grant No. 242301420053 and 222301420006); the Changjiang (Yangtze River) Scholar Programme of the Ministry of Education of China; the Ministry of Science and Technology of the People's Republic of China; Open Research Fund of State Key Laboratory of Coordination Chemistry, School of Chemistry and Chemical Engineering, Nanjing University; Open Cooperation Foundation of the Department of Chemical Science of Henan University (Grant No. DCSHENU2401); Postdoctoral Fellowship Program of CPSF (Grant No. GZC20250669); The authors are also grateful to the supports from Center of Advanced Analysis & Gene Sequencing of Zhengzhou University and Prof. Zhiguo Zhang.Notes and references
- (a) B. Giese, Angew. Chem., Int. Ed., 1983, 22, 753–764 CrossRef; (b) M. S. Kharasch, E. V. Jensen and W. H. Urry, Science, 1945, 102, 128–130 CrossRef CAS PubMed; (c) J. J. A. Garwood, A. D. Chen and D. A. Nagib, J. Am. Chem. Soc., 2024, 146, 28034–28059 CAS.
- (a) A. Ruffoni, R. C. Mykura, M. Bietti and D. Leonori, Nat. Synth., 2022, 1, 682–695 CrossRef CAS; (b) A. L. J. Beckwith, Chem. Soc. Rev., 1993, 22, 143–151 RSC.
- (a) E. Yoshioka, S. Kohtani, K. Sawai, Kentefu, E. Tanaka and H. Miyabe, J. Org. Chem., 2012, 77, 8588–8604 CrossRef CAS PubMed; (b) J. Liu, S. Wu, J. Yu, C. Lu, Z. Wu, X. Wu, X.-S. Xue and C. Zhu, Angew. Chem., Int. Ed., 2020, 59, 8195–8202 CrossRef CAS PubMed; (c) H. Wang, H. Liu, M. Wang, M. Huang, X. Shi, T. Wang, X. Cong, J. Yan and J. Wu, iScience, 2021, 24, 102693 CrossRef CAS PubMed; (d) S. Paul, D. Filippini and M. Silvi, J. Am. Chem. Soc., 2023, 145, 2773–2778 CrossRef CAS PubMed; (e) Y.-Y. He, Z.-K. Yang, T. Matsuyama, Y. Wei, L. Qin, Z. Zhang, M. Uchiyama and X.-Q. Hu, Org. Lett., 2025, 27, 4170–4175 CrossRef CAS PubMed; (f) J. A. Leitch, A. L. Fuentes de Arriba, J. Tan, O. Hoff, C. M. Martínez and D. J. Dixon, Chem. Sci., 2018, 9, 6653–6658 RSC.
- (a) B. M. Mikhailov, Russ. Chem. Rev., 1971, 40, 983–996 CrossRef; (b) Y. Ren, Y. Ge, Q. Yan, S. Chen, Y. Li, L. Li, Z.-Q. Liu and Z. Li, J. Org. Chem., 2021, 86, 12460–12466 CrossRef CAS PubMed; (c) Z. D. Miles, S. Diethelm, H. P. Pepper, D. M. Huang, J. H. George and B. S. Moore, Nat. Chem., 2017, 9, 1235–1242 CrossRef CAS PubMed; (d) G. W. Gribble, Acc. Chem. Res., 1998, 31, 141–152 CrossRef CAS; (e) I. R. Vitorino, J. D. N. Santos, G. Crespo, I. Pérez-Victoria, J. Martín, L. Rodriguez, M. C. Ramos, T. P. Martins, P. N. Leão, F. Vicente, V. Vasconcelos, O. M. Lage and F. Reyes, Microb. Biotechnol., 2025, 18, e70076 CrossRef CAS PubMed; (f) X. Dong, J. L. Roeckl, S. R. Waldvogel and B. Morandi, Science, 2021, 371, 507–514 CrossRef CAS PubMed.
- (a) D. A. Petrone, J. Ye and M. Lautens, Chem. Rev., 2016, 116, 8003–8104 CrossRef CAS PubMed; (b) Y. Cai, M. Dawor, G. Gaurav and T. Ritter, J. Am. Chem. Soc., 2025, 147, 18438–18444 CrossRef CAS PubMed; (c) L. N. Pham, A. Olding, C. C. Ho, A. C. Bissember and M. L. Coote, Angew. Chem., Int. Ed., 2025, 64, e202415792 CrossRef CAS; (d) X.-J. Tang and W. R. Dolbier Jr, Angew. Chem., Int. Ed., 2015, 54, 4246–4249 CrossRef CAS PubMed; (e) M. Jeon, D. Maiti, G. Kang, J. Kim and I. Choi, Angew. Chem., Int. Ed., 2026, 65, e16250, DOI:10.1002/anie.202516250; (f) X. Liu, Y. Kou, H. Wu, T.-X. Liu, Q. Liu, Z. Zhang, X. Zhang and G. Zhang, Sci. Adv., 2025, 11, eadt2715 CrossRef CAS PubMed.
- (a) D. Wu, W. Fan, L. Wu, P. Chen and G. Liu, ACS Catal., 2022, 12, 5284–5291 CrossRef CAS; (b) M. Sadeghi, Adv. Synth. Catal., 2024, 366, 2898–2918 CrossRef CAS; (c) A. T. Nguyen, H. Kang, T. G. Luu, S.-E. Suh and H.-K. Kim, Bull. Korean Chem. Soc., 2024, 45, 738–758 CrossRef CAS; (d) H.-L. Cui, Org. Biomol. Chem., 2024, 22, 1580–1601 RSC.
- (a) J. Kim, X. Sun, B. A. van der Worp and T. Ritter, Nat. Catal., 2023, 6, 196–203 CrossRef CAS; (b) X.-X. He, H.-H. Chang, Y.-X. Zhao, X.-J. Li, S.-A. Liu, Z.-L. Zang, C.-H. Zhou and G.-X. Cai, Chem. Asian J., 2023, 18, e202200954 CrossRef CAS PubMed.
- N. Fu, G. S. Sauer and S. Lin, J. Am. Chem. Soc., 2017, 139, 15548–15553 CrossRef CAS PubMed.
- K.-J. Bian, D. N. Jr, Y. Chen, Y.-C. Lu, S.-C. Kao, X.-W. Chen, A. A. Martí and J. G. West, Nat. Synth., 2025, 4, 314–326 CrossRef CAS PubMed.
- Y. Chen, L.-Q. Lu, D.-G. Yu, C.-J. Zhu and W.-J. Xiao, Sci. China Chem., 2019, 62, 24–57 CrossRef CAS.
- A. M. May and J. L. Dempsey, Chem. Sci., 2024, 15, 6661–6678 RSC.
- (a) F. Juliá, ChemCatChem, 2022, 14, e202200916 CrossRef; (b) Y. Yang, X. Huang and Y. Jin, Chem. Commun., 2025, 61, 1944–1961 RSC; (c) Y. Abderrazak, A. Bhattacharyya and O. Reiser, Angew. Chem., Int. Ed., 2021, 60, 21100–21115 CrossRef CAS.
- Y. Wang, S. Liu, J. Han, L. Wang and J. Chen, Synthesis, 2023, 55, 2860–2872 CrossRef CAS.
- V. Nair and A. Deepthi, Chem. Rev., 2007, 107, 1862–1891 CrossRef CAS PubMed.
- (a) A. Hu, J.-J. Guo, H. Pan and Z. Zuo, Science, 2018, 361, 668–672 CrossRef CAS; (b) L. Chang, Q. An, L. Duan, K. Feng and Z. Zuo, Chem. Rev., 2022, 122, 2429–2486 CrossRef CAS PubMed; (c) Q. An, L. Chang, H. Pan and Z. Zuo, Acc. Chem. Res., 2024, 57, 2915–2927 CrossRef CAS PubMed.
- (a) Q. Yang, Y.-H. Wang, Y. Qiao, M. Gau, P. J. Carroll, P. J. Walsh and E. J. Schelter, Science, 2021, 372, 847–852 CrossRef CAS; (b) Q. Yang, E. Song, Y. Wu, C. Li, M. R. Gau, J. M. Anna, E. J. Schelter and P. J. Walsh, J. Am. Chem. Soc., 2025, 147, 2061–2076 CrossRef CAS PubMed.
- (a) D. Shao, Y. Wu, S. Hu, W. Gao, Y. Du, X. Jia, S. Liu, M. Zhou and J. Chen, ACS Sustain. Chem. Eng., 2022, 10, 10294–10302 CrossRef CAS; (b) T. K. Dinda, A. Manna and P. Mal, ACS Catal., 2024, 14, 7664–7673 CrossRef CAS.
- S. S. Stahl, Angew. Chem., Int. Ed., 2004, 43, 3400–3420 CrossRef CAS PubMed.
- (a) B. Yang and Z. Lu, Chem. Commun., 2017, 53, 12634–12637 RSC; (b) X. Sun, X. Li, S. Song, Y. Zhu, Y.-F. Liang and N. Jiao, J. Am. Chem. Soc., 2015, 137, 6059–6066 CrossRef CAS PubMed; (c) B. Yang and Z. Lu, ACS Catal., 2017, 7, 8362–8365 CrossRef CAS.
- (a) Q.-L. Zhang, W. Liu, Y. Zhou and F.-L. Zhang, Green Chem., 2023, 25, 5735–5740 RSC; (b) S. Mahesh, K.-C. Tang and M. Raj, Molecules, 2018, 23, 2615 CrossRef PubMed.
- A. E. Reed, R. B. Weinstock and F. Weinhold, J. Chem. Phys., 1985, 83, 735–746 CrossRef CAS.
- P. Lian, W. Long, J. Li, Y. Zheng and X. Wan, Angew. Chem., Int. Ed., 2020, 59, 23603–23608 CrossRef CAS PubMed.
- CCDC 2473530: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p0x9y.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |