Open Access Article
Open Access ArticleGas-phase synthesis of the bicyclic silicon tricarbide molecule (c-SiC3) as a precursor to silicon carbide nanoparticles in space
Shane J.
Goettl
 a,
Kazuumi
Fujioka
a,
Márcio O.
Alves
b,
Mateus X.
Silva†
a,
Kazuumi
Fujioka
a,
Márcio O.
Alves
b,
Mateus X.
Silva†
 b,
Zhenghai
Yang
a,
Surajit
Metya
b,
Zhenghai
Yang
a,
Surajit
Metya
 a,
Iakov A.
Medvedkov
a,
Iakov A.
Medvedkov
 a,
Tosaporn
Sattasathuchana
*a,
Breno R. L.
Galvão
a,
Tosaporn
Sattasathuchana
*a,
Breno R. L.
Galvão
 *b,
Rui
Sun
*b,
Rui
Sun
 *a and
Ralf I.
Kaiser
*a and
Ralf I.
Kaiser
 *a
*a
aDepartment of Chemistry, University of Hawai'i at Mānoa, Honolulu, HI 96822, USA. E-mail: ralfk@hawaii.edu; ruisun@hawaii.edu; tsatta@hawaii.edu
bCentro Federal de Educação Tecnológica de Minas Gerais, Belo Horizonte 30421-169, Brazil. E-mail: brenogalvao@gmail.com
First published on 19th February 2026
Abstract
Silicon–carbon bond couplings represent a fundamental foundation for bottom-up molecular mass growth processes of silicon carbide grains in extraterrestrial environments. Yet, the elementary reaction mechanisms affording the gas-phase preparation of simple silicon- and carbon-containing molecules, which act as central molecular building blocks of silicon carbide grains, remain largely unexplored. Herein, we reveal the barrierless gas-phase preparation of the bicyclic silicon tricarbide molecule (c-SiC3, X1A1) and its linear isomer (l-SiC3, X3Σ−) as prototype silicon carbide grain precursors via the bimolecular reaction between tricarbon (C3, X1Σg+) and silylidyne (SiH, X2Π) under single-collision conditions. With the detection of c-SiC3 and l-SiC3 in the circumstellar envelope of the carbon star IRC+10216, the title reaction offers an entrance point for the exotic silicon chemistry in deep space thus bringing us closer to an understanding of how silicon and carbon chemistries can be coupled in our galaxy.
Introduction
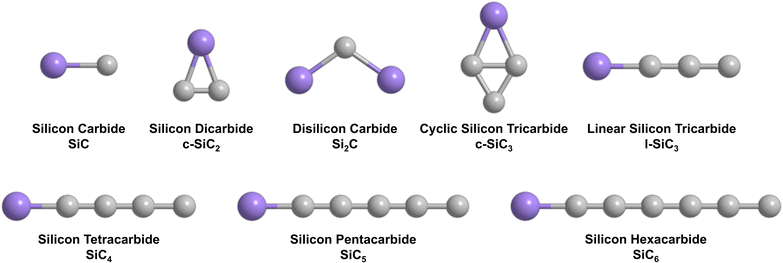
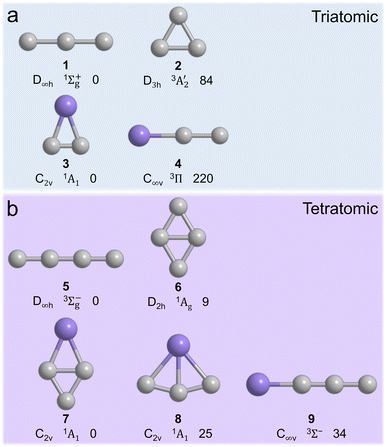
Ever since the detection of the cyclic silicon dicarbide molecule (SiC2, X1A1) in the circumstellar envelope of the asymptotic giant branch (AGB) carbon star IRC+10216 more than 60 years ago,1 silicon–carbon clusters have emerged as fundamental molecular building blocks of silicon carbide nanoparticles in deep space as identified via their 11.3 µm (885 cm−1) emission line (Fig. 1).2 These silicon–carbon clusters are characterized through unique chemical bonding schemes, which are distinct from their isovalent carbon–carbon counterparts. For instance, tricarbon (C3, 1, X1Σg+) is linear and has a singlet ground state, which is favored by 84 kJ mol−1 compared to the equilateral triangle structure in its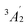 triplet ground state (2).3 However, in case of silicon dicarbide (SiC2), the cyclic isomer (3) is advantaged energetically by 220 kJ mol−1 compared to the linear isomer (4) (Fig. 2a).4 In the tetratomic system (Fig. 2b), tetracarbon (C4) is most stable in its linear geometry and holds a 3Σg− ground state (5) with a low-lying, bicyclic rhombic isomer only 9 kJ mol−1 higher in energy (6).5 Silicon tricarbide (SiC3), on the other hand, features exotic C2v symmetric bicyclic isomers with either bisecting carbon–carbon (7) or silicon–carbon (8) bonds, which are favorable by 34 and 9 kJ mol−1 compared to the linear triplet isomer (9).
triplet ground state (2).3 However, in case of silicon dicarbide (SiC2), the cyclic isomer (3) is advantaged energetically by 220 kJ mol−1 compared to the linear isomer (4) (Fig. 2a).4 In the tetratomic system (Fig. 2b), tetracarbon (C4) is most stable in its linear geometry and holds a 3Σg− ground state (5) with a low-lying, bicyclic rhombic isomer only 9 kJ mol−1 higher in energy (6).5 Silicon tricarbide (SiC3), on the other hand, features exotic C2v symmetric bicyclic isomers with either bisecting carbon–carbon (7) or silicon–carbon (8) bonds, which are favorable by 34 and 9 kJ mol−1 compared to the linear triplet isomer (9).
This stability of cyclic versus acyclic silicon carbides can be in part rationalized through the concept of aromaticity. Natural bond order (NBO) analysis of cyclic silicon dicarbide (3) predicts an optimal Lewis structure with delocalization of two π electrons between the silicon and carbon atoms.6 However, c-SiC2 exhibits a significant ionic character between the silicon atom and the dicarbon moiety7 thus categorizing 3 as polytopic8 with the dicarbon group undergoing a hindered rotation (pinwheel motion).9–11 The classification of silicon tricarbide (7) depicts a delocalization of two π electrons over one or both rings,12,13 while the higher energy linear isomer (9) features characteristic cumulenic bonding.14 The c-SiC3 structure which contains a silicon–carbon bisection (8) represents a fascinating benchmark with π electron delocalization only along the C3 moiety, which does not overlap with the silicon π orbitals or lone pair.15 Overall, the introduction of a single silicon atom into a carbon framework results in fundamental changes in the chemical bonding, aromaticity, molecular structure ((bi)cyclic versus linear), and electronic properties (triplet versus singlet), which are not predictable by Langmuir's concept of isovalency.16
However, despite the critical importance of silicon carbides in astrochemistry,17–20 material sciences (catalysis,21 nanostructures,22,23 semiconductors24), and physical (organic) chemistry from the viewpoint of chemical bonding and molecular structure, the controlled gas-phase preparation of silicon carbides has been scarce. Previous work involved laser ablation of silicon carbides25–28 along with electric discharge of mixtures of silicon- and carbon-containing gases like silane and methane.14,29,30 These approaches generated complex mixtures of reactive organosilicon species, which complicate the recognition of distinct reaction mechanisms of formation of the simple silicon–carbon clusters in the gas phase.12,13 Yet, an elucidation of these formation pathways is imperative for an intimate understanding of the largely elusive coupling of the silicon and carbon chemistries of the interstellar medium at the molecular level, eventually constraining the pathways to bare silicon–carbon molecules in our galaxy.
Herein, we provide compelling evidence on the directed gas-phase synthesis of the bicyclic silicon tricarbide molecule (c-SiC3, 7) along with its linear isomer (l-SiC3, 9) in the gas phase under single-collision conditions from two acyclic reactants, tricarbon (C3, X1Σg−) and the silylidyne radical (SiH, X2Π), by exploiting crossed molecular beam experiments coupled with electronic structure calculations and quasi-classical trajectory (QCT) simulations. The overall barrierless and exoergic nature of this bimolecular reaction provides an unconventional entry point to the synthesis of 7 and 9 even at ultralow temperatures in cold molecular clouds such as G+0.693–0.0027 in the Galactic Center, where the cyclic silicon dicarbide molecule (3) was observed31 and also affords a mechanistical framework rationalizing the astronomical detection of 7 and 9 in the circumstellar envelope of IRC+10216.32,33 Indeed, formation mechanisms of 7 have only been previously explored from the degradation of SiC3H2 isomers via Lyman-α photodissociation in the outer envelopes of carbon AGB stars,34 thus, our study reveals the first account of the bottom-up synthesis of 7via bimolecular reactions in circumstellar envelopes. This represents a fundamental shift in knowledge toward neutral–neutral molecular mass growth processes to silicon carbides, questioning prior viewpoints that silicon carbides can only be formed via photochemical degradation of higher molecular weight silicon- and carbon-containing molecules or at elevated temperatures via ion–molecule reactions.35 The close agreement between experimental chemical dynamics studies and the conclusions from QCT simulations in particular reveals that molecular beam studies merged with dynamics simulations have progressed to such a sophisticated degree that polyatomic reactions involving elements from the third row of the periodic table of the elements can be untangled at the molecular level. This framework thus offers a unified, predictive picture and rare glimpse of the gas-phase chemistry and chemical bonding of isovalent systems involving silicon under single-collision conditions.
Results and discussion
Time-of-flight mass spectra
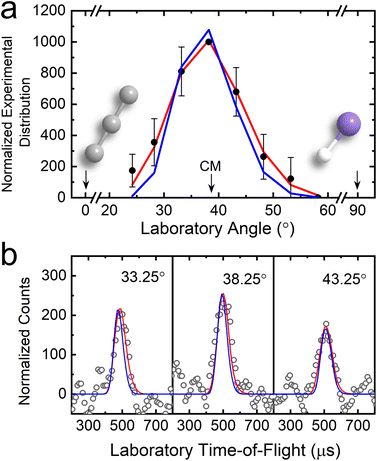
The gas-phase reaction of electronically ground state tricarbon (C3, X1Σg+) with the silylidyne radical (SiH, X2Π) was explored experimentally under single-collision conditions in a crossed molecular beam machine by intersecting supersonic beams of tricarbon and silylidyne radicals perpendicularly at a collision energy of 47.9 ± 0.9 kJ mol−1. The helium-seeded tricarbon beam was generated via laser ablation of graphite (266 nm, 5 mJ pulse−1),36 while silylidyne radicals (SiH, X2Π) were produced via photodissociation of helium-seeded disilane (Si2H6) at a fraction of 0.5%37 (SI). Neutral reaction products were ionized through electron impact ionization with 80 eV electrons within a triply-differentially-pumped quadrupole mass spectrometer (QMS) operated at pressures of a few 10−12 Torr and then mass analyzed. Time-of-flight (TOF) spectra at distinct mass-to-charge ratios (m/z) were probed with reactive scattering signal observed at m/z = 64 (28Si12C3+). Accounting for the tricarbon and silylidyne reactant masses of 36 amu and 29 amu, respectively, the detection at m/z = 64 can be attributed to the formation of SiC3 (64 amu) product(s) along with atomic hydrogen (1 amu) (reaction (1)). No signal was observed at m/z = 65 (28Si12C3H+, 29Si12C3+, 28Si13C12C2+) and 66 (28Si12C3H2+, 29Si12C3H+, 28Si13C12C2H+, 30Si12C3+) revealing that any silyl radicals (SiH3) or silylene (SiH2)—if generated during the photolysis of disilane—do not react with tricarbon. The TOF spectra were then collected at m/z = 64 in 5° steps with respect to the center-of-mass (CM) angle (θCM) (Fig. 3b) and integrated to obtain the laboratory angular distribution (LAD) (Fig. 3a). The TOF spectra are fairly narrow and range only from about 400 to 600 µs, which is also reflected in a narrow LAD range of only 30°. This distribution is nearly symmetric with respect to the θCM and hence indicative of indirect scattering dynamics involving SiC3H reaction intermediate(s) undergoing unimolecular decomposition to SiC3 product(s) plus atomic hydrogen (reaction (1)).| C3 (36 amu) + SiH (29 amu) → SiC3 (64 amu) + H (1 amu) | (1) |
Center-of-mass flux distributions
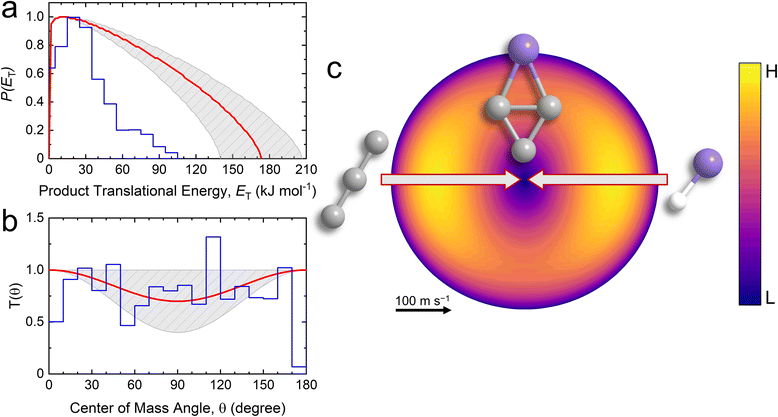
With the detection of SiC3 isomer(s) as a consequence of reactive single collisions between tricarbon and the silylidyne radical via Reaction (1), information on the chemical reaction dynamics can be obtained by converting the laboratory data into the CM reference frame.38 This procedure yields the CM translational energy (P(ET)) and angular (T(θ)) flux distributions along with the associated flux contour map (Fig. 4). The laboratory data could be replicated by a single reaction channel via reaction (1). In detail, the best-fit P(ET) exhibits a maximum translational energy (Emax) of 173 ± 34 kJ mol−1 (Fig. 4a). For those molecules born without internal energy, the reaction energy (ΔrG) is determined to be −125 ± 35 kJ mol−1 by utilizing energy conservation with ΔrG = EC − Emax, where EC is the collision energy (47.9 ± 0.9 kJ mol−1).39 Additionally, the P(ET) features a distribution maximum near zero, which is indicative of a low or no barrier in the exit channel with only a slight electron density rearrangement from the fragmenting SiC3H complex to the final products.40 Further, the T(θ) (Fig. 4b) is forward–backward symmetric and exhibits intensity over the complete angular range, suggestive of indirect reactive scattering dynamics through complex formation (SiC3H); this finding reinforces the conclusions drawn from the nearly symmetric LAD. The higher intensity at the poles (0° and 180°) and the dip at 90° indicates coplanar scattering dynamics, which involves geometric constraints and an emission of the hydrogen atom preferentially perpendicular to the total angular momentum vector within the rotational plane of the fragmenting complex(es).39 These findings are also reflected in the flux contour map (Fig. 4c).Potential energy surface
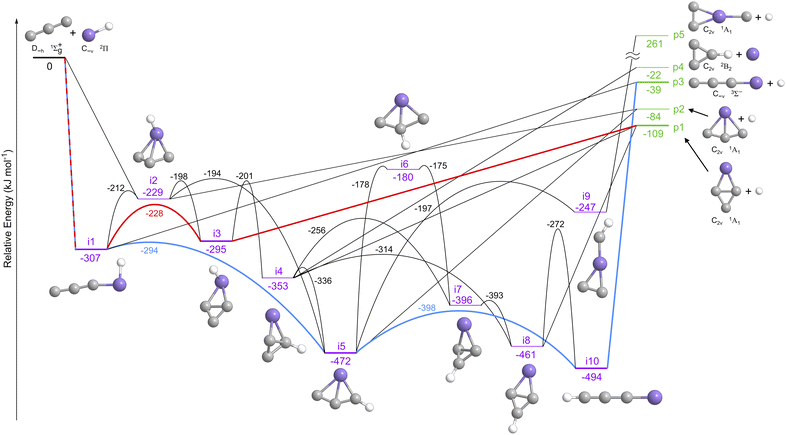
To garner insight into the underlying reaction mechanism(s) involved in the tricarbon–silylidyne system and the gas-phase preparation of SiC3 isomer(s), the experimental results were combined with electronic structure calculations performed at the CCSD(T)-F12/aug-cc-pV(Q+d)Z//TPSSh/cc-pV(T+d)Z + ZPE (TPSSh/cc-pV(T+d)Z) level of theory.41–44 These computations provide zero-point energy (ZPE) corrected energies of the reactants, transition states, and products with an expected accuracy of ± 5 kJ mol−1 (SI). The doublet SiC3H potential energy surface (PES) is compiled in Fig. 5 and comprises ten SiC3H reaction intermediates (i1–i10) and five SiC3 product isomers (p1–p5), four of which (p1–p4) are formed in overall exoergic reactions releasing 109, 84, 39, and 22 kJ mol−1, respectively. The experimentally observed reaction exoergicity of 125 ± 35 kJ mol−1 correlates well with the formation of the thermodynamically most stable carbon–carbon bisected bicyclic isomer c-SiC3 (p1, X1A1, ΔrG = −109 ± 5 kJ mol−1). The energetics of the remaining exoergic hydrogen-atom-loss isomers—silicon–carbon bisected c-SiC3 (p2, X1A1, ΔrG = −84 ± 5 kJ mol−1) and linear SiC3 (p3, X3Σ−, ΔrG = −39 ± 5 kJ mol−1)—lie outside the experimental error bars. Therefore, at least p1 is formed. However, this does not exclude the generation of the C2v symmetric p2 and the linear p3 isomers, since their kinetic energy release could be hidden in the lower energy section of the P(ET).How can the bicyclic product p1 be synthesized as the result of a single collision of two acyclic reactants? The electronic structure calculations identified two barrierless entrance channels via addition of the silylidyne radical (SiH, X2Π) either to one terminal carbon atom of tricarbon (C3, X1Σg+) or to both terminal carbon atoms simultaneously yielding the acyclic and bicyclic doublet intermediates i1 and i2, respectively. The barrierless nature of these entrance channels was verified by optimized PES scans following the reaction coordinate. From these initial collision complexes, there are a plethora of possible pathways to p1. Both intermediates can be interconverted easily via a transition state ranging 212 kJ mol−1 below the energy of the separated reactants. This isomerization involves the simultaneous formation of two silicon–carbon bonds of 196 pm and 231 pm lengths producing a bicyclic structure (i1 → i2). These bonds are slightly elongated compared to the carbon–silicon single bond in methylsilane (CH3SiH3)45 and hence can be classified as sigma bonds. The central silicon–carbon bond in i2 then breaks, and a new bond forms between the outer two carbon atoms generating a diamond-like intermediate (i2 → i3) followed by barrierless hydrogen atom loss to p1. Alternatively, i1 can isomerize in one step to i3via a synchronous formation of a carbon–carbon and carbon–silicon bond (i1 → i3). Both intermediates i4 and i8 may also decompose via hydrogen atom loss to p1, with the most energetically-favorable routes involving a hydrogen migration and ring closure from i1 to i5 followed by C–Si bisecting bond breaking and C–C bisecting bond reforming from i5 to i4 (i1 → i5 → i4 → p1), and [1,2]-hydrogen shift from i4 to i8 (i1 → i5 → i4 → i8 → p1). The unimolecular decomposition of i3, i4, and i8 are all barrierless (loose exit transition states) as verified experimentally in the close-to-zero peaking of the center-of-mass translational energy distribution.
The second bicyclic SiC3 isomer p2 as well as the linear structure p3 may also have been synthesized via the bimolecular reaction of tricarbon with the silylidyne radical. First, p2 can be accessed via unimolecular decomposition of intermediates i2 and/or i5 accompanied by atomic hydrogen loss; the energetically most likely pathways involve first a ring closure from i1 to the bicyclic backbone moiety (i1 → i2 → p2; i1 → i5 → p2) and/or a simple addition–elimination from i2 followed by hydrogen atom loss (i2 → p2). Second, p3 may be prepared via atomic hydrogen loss from intermediates i1 and/or i10via a simple addition–elimination process (i1 → p3) or through the energetically most favorable cyclization–ring-opening route passing through i10 (i1 → i5 → i10 → p3). Overall, the computational results support the experimental detection of at least the thermodynamically most stable bicyclic isomer (p1) accessed via a barrierless, bimolecular, exoergic reaction of electronically ground state tricarbon (C3, X1Σg+) with the silylidyne radical (SiH, X2Π), while both p2 and p3 also feature possible routes under our experimental conditions.
Quasi-classical trajectory simulations
To identify the most likely reaction pathways and illustrate how the reaction progresses, reaction dynamics simulations were performed. 3600 bimolecular collision machine-learned molecular dynamics (MLMD) trajectories were simulated for 15 ps or until dissociation to reactants or products. Of these, 2068 trajectories formed intermediates or products, resulting in product branching ratios of 63% p1, 22% p2, 2% p3, and 13% p4, with no formation of the high-energy product p5. For the reaction pathways, the trajectories show no direct product formation, i.e., all are indirect with finite lifetimes of intermediates before dissociating. Due to the large potential energy release from the bimolecular association, isomerization between intermediates is extremely fast, with more than one million recorded over all trajectories. To quantify precise statistical probabilities of these events, the reaction mechanism can be formulated as a Markov process, where a sequence of transitions between discrete states takes place. The states are defined as critical points on the PES, thus the transitions correspond to isomerization processes. The probability of product formation from each unimolecular state is shown in Fig. 6, where exit channels to p1, p2, and p3 mostly originate from i3, i5, and i10, respectively. From the MLMD simulations and Markov model, the most probable reaction pathway involves the entrance channel to i1, followed by cyclization to i3 and atomic hydrogen loss forming the experimentally observed bicyclic silicon tricarbide molecule (p1). More details regarding the QCT simulations are given in the SI accompanied by Fig. S1–S9 and Tables S1–S6.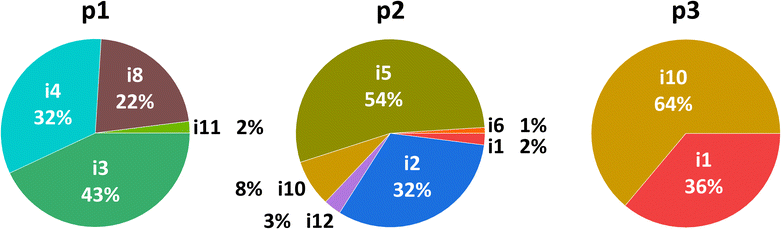 | ||
| Fig. 6 Transition probabilities from different intermediates to products p1, p2, and p3, represented as percentages, shown as pie charts. | ||
Finally, the dynamics were validated against experimental results. Although the experiments do not have information on the time evolution of the intermediates, they contain information on the products such as scattering angles and translational energies. To match the experimental detection, only hydrogen-atom-loss products (p1, p2, and p3) were tallied. As shown in Fig. 4, the scattering angle distributions of the experimental fit and the MLMD agree qualitatively. While the translational energy distributions disagree on maximum energies, they have similar most-probable energies. In general, products with high translational energy are rare and difficult to measure in MD simulations, often leading to mismatches between experimental and MD distributions at the high-energy tail, as reported in similar studies.46–48 Extending the MLMD simulations is not likely to resolve this issue, as the longer the lifetime of an intermediate, the more inelastic the collision becomes, and the less translational energy the products retain. A more solid comparison between the MLMD simulations and the experiment can come by directly forward-convoluting the MLMD simulations' CM distributions to the raw lab data as shown in Fig. 3, where the data agree well.
Quasi-atomic orbital analysis
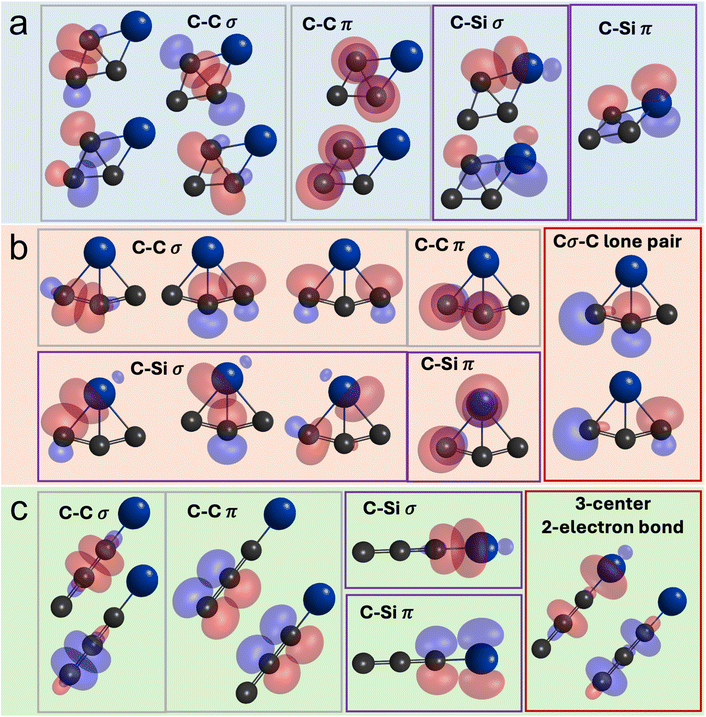
As the hydrogen atom is ejected during the transformation from i3 to p1, multiple structural changes occur within the SiC3 moiety. The carbon–carbon bisecting bond shrinks from 152 to 148 pm while the peripheral carbon–carbon bonds elongate from 135 to 144 pm. In addition, the silicon–carbon bonds are reduced from 197 to 184 pm essentially shifting the bisected bond towards the silicon atom. This results in an increased C–Si–C angle of 47.4° and reduced opposing C–C–C angle of 62.0°. The structural changes could reflect inclusion of the silicon atom within the π electron delocalized system and shift to 2π aromaticity. As for the linear SiC3 isomer, the conversion from i10 to p3 invokes a silicon–carbon bond elongation from 170 to 173 pm, C1–C2 carbon–carbon bond shortening from 133 to 129 pm, and a lengthening of the C2–C3 carbon–carbon bond from 123 to 131 pm. These changes likely indicate a shift from a butadiyne-type structure to cumulenic bonding as the hydrogen atom is ejected. Fundamental insights into the chemical bonding of these SiC3 isomers can be further obtained from quasi-atomic orbital (QUAO) analysis (Fig. 7) highlighting the orbitals that participate in (covalent) bonding interactions.49–54 In this analysis, the kinetic bond order (KBO) represents a measure of the strength of the bonding interaction, while the occupation numbers in two orbitals are the number of electron populations involved in the covalent bond (Table S7). Generally, for all isomers, the strengths of the carbon–silicon σ and π bonds (KBOs = −105 to −63 and −42 to −8 kJ mol−1, respectively) significantly decreases compared to the isovalent carbon–carbon bond interactions (KBOs = −293 to −167 and −71 to −33 kJ mol−1 for σ and π bonds, respectively). In all isomers, the interference kinetic energy, i.e. the fundamental origin of the covalent bond,55–59 is mainly the result of the σ bonding interactions (64–84%), while the π bonds contribute 15–34%. From the QUAO analysis, the stability of the bicyclic SiC3 isomer p1 is based on the strong carbon–silicon σ interactions with contributions of 23, 17, and 11% for isomers p1, p2, and p3, respectively, while minor contributions of 4, 2, and 8% arise from π interactions. In bicyclic p1 and p2, an additional stabilization arises from the lateral overlap between the C–C and Si–C σ interactions. Such an orbital overlap of silicon with the peripheral carbon in p2 is in excellent agreement with the previous theoretical study by Rintelman and Gordon.15 Interestingly, p3 exhibits interactions between three center atoms with electron populations of 1.7 to 2.0, which is indicative of a three-center-two-electron interaction type (Fig. S10 and Table S7).Conclusions
The crossed molecular beam experiments in conjunction with electronic structure calculations and QCT simulations provide a unique glimpse into the distinct dynamics of isovalent systems at the fundamental, microscopic level benchmarked with the gas-phase synthesis of silicon tricarbide molecules in their carbon–carbon (c-SiC3, X1A1, p1) and silicon–carbon (c-SiC3, X1A1, p2) bisected bicyclic forms along with the linear isomer (l-SiC3, X3Σ−, p3) from the bimolecular reaction of the acyclic tricarbon (C3, X1Σg+) and silylidyne radical (SiH, X2Π) reactants. The addition of the silicon atom of the silylidyne radical to a terminal carbon atom of tricarbon proceeds without barrier, followed by exotic ring-formation through doublet silabicyclobutadienyl intermediates (c-SiC3H) either forming p1 or p2 coupled with atomic hydrogen loss, or isomerizing through ring-opening to a 2-propynylidynesilyl structure (l-HC3Si) followed by hydrogen atom loss to p3. While the two most stable structures of silicon tricarbide are bicyclic with 34 and 9 kJ mol−1 lower energy than the linear structure, the ground state of the isovalent tetracarbon molecule is not bicyclic; the linear structure (C4, X3Σg−) is more stable by 9 kJ mol−1 (Fig. 2b). This demonstrates the dramatic effects substituting just one carbon atom with an isovalent silicon atom has on chemical structure and electronic properties. The inherent QUAO analysis and interference kinetic energies of the three SiC3 isomers offer evidence of primary bonding interactions originating from the σ bonding motifs: while carbon–carbon σ bonds contribute about 60% of the interference kinetic energy, the percent contributions of the carbon–silicon σ bonds in bicyclic SiC3 isomers p1 and p2 of 23 and 17%, respectively, are significantly larger than those in the linear isomer p3 (11%).Silicon–carbon bond coupling and the formation of silicon carbide clusters signifies a crucial topic not only for fundamental reaction dynamics and chemical bonding, but also for the astronomy and astrochemistry communities. Recently, dust particles produced from modeling the condensation of silicon carbide (SiC) and 1,3,5-trisiladehydrobenzene (Si3C3) clusters in asymptotic giant branch (AGB) stellar winds of carbon rich stars showed a match with common SiC emission lines observed astronomically.60 These spectral similarities extend to small- and mid-size clusters ranging up to Si16C16,61 further reinforcing the importance of exploring the fundamental formation mechanisms of silicon carbides under astrophysically relevant conditions.
With the tricarbon molecule detected in the circumstellar envelope of the carbon star IRC+10216 (ref. 62) and predicted to exist in cold molecular clouds63 and the silylidyne radical generated via photodissociation and galactic comic ray degradation of silane (SiH4), the title reaction provides a unique entry point into an exotic silicon–carbon chemistry and molecular mass growth processes involving silicon in deep space as evidenced by the formation of the astronomically observed silicon tricarbide molecules p1 and p3. The elucidated bottom-up mechanisms are of fundamental significance to facilitate an understanding of how carbon and silicon chemistries can be coupled and hence challenge conventional wisdom that silicon carbides can only be formed at elevated temperatures via complex ion–molecule reactions or degradation of higher molecular weight silicon carbon molecules.35 This system exemplifies the versatile concept of barrierless and exoergic molecular mass growth processes to silicon carbides from acyclic precursors potentially adaptable to the multifaceted reaction sequence Reaction (2). With the full-scale operation of the Atacama Large Millimeter/Submillimeter Array, the detection of unusual silicon-bearing molecules is expected to increase significantly. Interpreting these observations will depend critically on advances in experimental and computational laboratory astrophysics, as proposed in this work. Such progress is essential to bridge the longstanding gap between observational data and laboratory studies of organosilicon chemistry in the galaxy.
| Cn + SiH → SiCn + H | (2) |
Author contributions
S. J. G. and R. I. K. conceptualized the experiments; S. J. G., Z. Y., S. M., and I. A. M. conducted the experiments; R. I. K. supervised the experiments; M. O. A. and M. X. S. performed the electronic structure calculations; B. R. L. G. supervised the electronic structure calculations; K. F. performed the quasi-classical trajectory simulations; R. S. supervised the quasi-classical trajectory simulations; T. S. performed the quasi-atomic orbital analysis; S. J. G. and K. F. wrote the original draft of the manuscript; S. J. G., K. F., R. I. K., B. R. L. G., R. S., and T. S. revised the draft of the manuscript with input from all authors.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available in the article and the supplementary information (SI). Additional data are available from the corresponding authors upon request. Supplementary information: experimental methods, computational methods, details of AIMD simulations, comparison of different MD methods (Table S1), benchmarking the sGDML potential, energy and force errors (Fig. S1), training set representation (Fig. S2), reaction probability method comparison (Fig. S3), complete MLMD results, reaction probability for different products (Fig. S4), transition probabilities (Table S2), product transition probabilities (Fig. S5), details of the Markov model, Markov digraph (Fig. S6), DFT PES (Fig. S7), product branching ratios (Table S3), chemical concentration curves (Fig. S8), intermediate lifetime distributions (Fig. S9), intermediate lifetime fits (Table S4 and S5), semi-Markov stationary states (Table S6), QUAO analysis of SiC3 isomers, presentation of QUAOs (Fig. S10), QUAO analysis values (Table S7), Cartesian coordinates and vibrational frequencies for all species (Data S1), supplementary references. See DOI: https://doi.org/10.1039/d6sc00002a.Acknowledgements
The experimental studies at the University of Hawaii were supported by the US National Science Foundation CHE 2244717. The computational studies at the University of Hawaii were supported by the US National Science Foundation grant no. 2144031, CAREER: Minimize ab initio Tasks in Dynamics Simulations of Chemical Reactions with Active Machine Learning. The technical support and advanced computing resources from the University of Hawaii Information Technology Services – Research Cyberinfrastructure, funded in part by the National Science Foundation CC* awards 2201428 and 2232862, are gratefully acknowledged. MOV, MXS, and BRLG thank Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) for grant 305211/2024-2, FAPEMIG for grant APQ-02390-25, and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for grant 88887.125122/2025-00.Notes and references
- P. Thaddeus, S. E. Cummins and R. A. Linke, Astrophys. J., 1984, 283, L45–L48 CrossRef CAS.
- R. Papoular, M. Cauchetier, S. Begin and G. LeCaer, Astron. Astrophys., 1998, 329, 1035–1044 CAS.
- H. Fueno and Y. Taniguchi, Chem. Phys. Lett., 1999, 312, 65–70 CrossRef CAS.
- H. S. P. Müller and D. E. Woon, J. Phys. Chem. A, 2013, 117, 13868–13877 CrossRef PubMed.
- A. J. C. Varandas and C. M. R. Rocha, Philos. Trans. R. Soc., A, 2018, 376, 1–46 CrossRef.
- G. Maier, H. Pacl, H. P. Reisenauer, A. Meudt and R. Janoschek, J. Am. Chem. Soc., 1995, 117, 12712–12720 CrossRef CAS.
- T. J. Butenhoff and E. A. Rohlfing, J. Chem. Phys., 1991, 95, 1–8 CrossRef CAS.
- E. Clementi, H. Kistenmacher and H. Popkie, J. Chem. Phys., 1973, 58, 2460–2466 CrossRef CAS.
- S. C. Ross, T. J. Butenhoff, E. A. Rohlfing and C. M. Rohlfing, J. Chem. Phys., 1994, 100, 4110–4126 CrossRef CAS.
- I. M. B. Nielsen, W. D. Allen, A. G. Császár and H. F. Schaefer, III, J. Chem. Phys., 1997, 107, 1195–1211 CrossRef CAS.
- C. M. R. Rocha, H. Linnartz and A. J. C. Varandas, J. Chem. Phys., 2022, 157, 104301 CrossRef CAS PubMed.
- M. C. McCarthy, A. J. Apponi and P. Thaddeus, J. Chem. Phys., 1999, 110, 10645–10648 CrossRef CAS.
- A. J. Apponi, M. C. McCarthy, C. A. Gottlieb and P. Thaddeus, J. Chem. Phys., 1999, 111, 3911–3918 CrossRef CAS.
- M. C. McCarthy, A. J. Apponi, C. A. Gottlieb and P. Thaddeus, Astrophys. J., 2000, 538, 766–772 CrossRef CAS.
- J. M. Rintelman and M. S. Gordon, J. Chem. Phys., 2001, 115, 1795–1803 CrossRef CAS.
- I. Langmuir, J. Am. Chem. Soc., 1919, 41, 868–934 CrossRef CAS.
- N. Job and K. Thirumoorthy, ACS Earth Space Chem., 2024, 8, 467–482 CrossRef CAS.
- J. Bernal, P. Haenecour, J. Howe, T. Zega, S. Amari and L. Ziurys, Astrophys. J., Lett., 2019, 883, L43 CrossRef CAS.
- T. Chen, C. Y. Xiao, A. Li and C. T. Zhou, Mon. Not. R. Astron. Soc., 2021, 509, 5231–5236 CrossRef.
- P. R. Heck, J. Greer, L. Kööp, R. Trappitsch, F. Gyngard, H. Busemann, C. Maden, J. N. Ávila, A. M. Davis and R. Wieler, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 1884–1889 CrossRef CAS PubMed.
- S. R. Kulkarni, V. K. Velisoju, F. Tavares, A. Dikhtiarenko, J. Gascon and P. Castaño, Catal. Rev., 2023, 65, 174–237 CrossRef CAS.
- O. Arayawut, T. Kerdcharoen and C. Wongchoosuk, Nanomaterials, 2022, 12, 1869 CrossRef CAS PubMed.
- F. Ullah, N. Kosar, M. N. Arshad, M. A. Gilani, K. Ayub and T. Mahmood, Opt. Laser Technol., 2020, 122, 105855 CrossRef CAS.
- P. Miró, M. Audiffred and T. Heine, Chem. Soc. Rev., 2014, 43, 6537–6554 RSC.
- R. Suenram, F. Lovas and K. Matsumura, Astrophys. J., Lett., 1989, 342, L103–L105 CrossRef CAS.
- M. A. Capano, J. Appl. Phys., 1995, 78, 4790–4792 CrossRef CAS.
- S. H. Nam and S. M. Park, Appl. Phys. A, 2004, 79, 1117–1120 CrossRef CAS.
- D. Witsch, V. Lutter, A. A. Breier, K. M. T. Yamada, G. W. Fuchs, J. Gauss and T. F. Giesen, J. Phys. Chem. A, 2019, 123, 4168–4177 CrossRef CAS PubMed.
- B. Gans, J. Liévin, P. Halvick, N. L. Chen, S. Boyé-Péronne, S. Hartweg, G. A. Garcia and J. C. Loison, Phys. Chem. Chem. Phys., 2023, 25, 23568–23578 RSC.
- M. C. McCarthy, J. H. Baraban, P. B. Changala, J. F. Stanton, M.-A. Martin-Drumel, S. Thorwirth, C. A. Gottlieb and N. J. Reilly, J. Phys. Chem. Lett., 2015, 6, 2107–2111 CrossRef CAS PubMed.
- S. Massalkhi, I. Jiménez-Serra, J. Martín-Pintado, V. M. Rivilla, L. Colzi, S. Zeng, S. Martín, B. Tercero, P. de Vicente and M. A. Requena-Torres, Astron. Astrophys., 2023, 678, A45 CrossRef CAS.
- A. J. Apponi, M. C. McCarthy, C. A. Gottlieb and P. Thaddeus, Astrophys. J., 1999, 516, L103–L106 CrossRef CAS.
- J. Cernicharo, J. R. Pardo, M. Agúndez, J. P. Fonfría, L. Velilla-Prieto, C. Cabezas, B. Tercero, P. de Vicente and M. Guélin, Astron. Astrophys., 2025, 700, L20 CrossRef CAS.
- T. Yang, L. Bertels, B. B. Dangi, X. Li, M. Head-Gordon and R. I. Kaiser, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 14471–14478 CrossRef CAS PubMed.
- A. Glassgold and G. Mamon, in Chemistry and Spectroscopy of Interstellar Molecules, ed. D. Bohme, University of Tokyo Press, Tokyo, 1992, p. 261 Search PubMed.
- R. I. Kaiser, T. N. Le, T. L. Nguyen, A. M. Mebel, N. Balucani, Y. T. Lee, F. Stahl, P. v. R. Schleyer and H. F. Schaefer III, Faraday Discuss., 2002, 119, 51–66 RSC.
- T. Yang, B. B. Dangi, A. M. Thomas and R. I. Kaiser, Chem. Phys. Lett., 2016, 654, 58–62 CrossRef CAS.
- R. I. Kaiser, C. Ochsenfeld, D. Stranges, M. Head-Gordon and Y. T. Lee, Faraday Discuss., 1998, 109, 183–204 RSC.
- W. B. Miller, S. A. Safron and D. R. Herschbach, Discuss. Faraday Soc., 1967, 44, 108–122 RSC.
- J. Laskin and C. Lifshitz, J. Mass Spectrom., 2001, 36, 459–478 CrossRef CAS PubMed.
- H. J. Werner, P. J. Knowles, G. Knizia, F. R. Manby, M. Schütz, P. Celani, W. Györffy, D. Kats, T. Korona, R. Lindh, A. Mitrushenkov, G. Rauhut, K. R. Shamasundar, T. B. Adler, R. D. Amos, A. Bernhardsson, A. Berning, D. L. Cooper, J. O. Deegan, A. J. Dobbyn, F. Eckert, E. Goll, C. Hampel, A. Hesselmann, G. Hetzer, T. Hrenar, G. Jansen, C. Köppl, Y. Liu, A. W. Lloyd, R. A. Mata, A. J. May, S. J. McNicholas, W. Meyer, M. E. Mura, A. Nicklass, D. P. O'Neill, P. Palmieri, D. Peng, K. Pflüger, R. Pitzer, M. Reiher, T. Shiozaki, H. Stoll, A. J. Stone, R. Tarroni, T. Thorsteinsson and M. Wang, MOLPRO, a package of ab initio programs, Version 2015.1, University of Cardiff, Cardiff, UK, 2015 Search PubMed.
- T. H. Dunning Jr, K. A. Peterson and A. K. Wilson, J. Chem. Phys., 2001, 114, 9244–9253 CrossRef.
- T. B. Adler, G. Knizia and H.-J. Werner, J. Chem. Phys., 2007, 127, 221106 CrossRef PubMed.
- G. Knizia, T. B. Adler and H.-J. Werner, J. Chem. Phys., 2009, 130, 054104 CrossRef PubMed.
- R. W. Kilb and L. Pierce, J. Chem. Phys., 1957, 27, 108–112 CrossRef CAS.
- C. He, K. Fujioka, A. A. Nikolayev, L. Zhao, S. Doddipatla, V. N. Azyazov, A. M. Mebel, R. Sun and R. I. Kaiser, Phys. Chem. Chem. Phys., 2022, 24, 578–593 Search PubMed.
- S. J. Goettl, A. Vincent, M. X. Silva, Z. Yang, B. R. L. Galvão, R. Sun and R. I. Kaiser, Sci. Adv., 2024, 10, eadq5018 Search PubMed.
- S. Doddipatla, C. He, R. I. Kaiser, Y. Luo, R. Sun, G. R. Galimova, A. M. Mebel and T. J. Millar, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 22712–22719 Search PubMed.
- A. C. West, M. W. Schmidt, M. S. Gordon and K. Ruedenberg, J. Chem. Phys., 2013, 139, 234107 CrossRef PubMed.
- A. C. West, M. W. Schmidt, M. S. Gordon and K. Ruedenberg, J. Phys. Chem. A, 2015, 119, 10360–10367 CrossRef CAS.
- A. C. West, M. W. Schmidt, M. S. Gordon and K. Ruedenberg, J. Phys. Chem. A, 2017, 121, 1086–1105 Search PubMed.
- K. Ruedenberg, J. Chem. Phys., 2022, 157, 024111 Search PubMed.
- K. Ruedenberg, J. Chem. Phys., 2022, 157, 210901 CrossRef CAS PubMed.
- D. Del Angel Cruz, K. N. Ferreras, T. Harville, G. Schoendorff and M. S. Gordon, Phys. Chem. Chem. Phys., 2024, 26, 21395–21406 RSC.
- W. Kutzelnigg and W. H. E. Schwarz, Phys. Rev. A, 1982, 26, 2361–2367 Search PubMed.
- G. B. Bacskay, J. Chem. Phys., 2022, 156, 204122 CrossRef CAS PubMed.
- G. Frenking, Mol. Phys., 2023, 121, e2110168 CrossRef.
- L. Zhao, S. Pan, N. Holzmann, P. Schwerdtfeger and G. Frenking, Chem. Rev., 2019, 119, 8781–8845 CrossRef CAS PubMed.
- W. Kutzelnigg, Angew. Chem., Int. Ed., 1973, 12, 546–562 CrossRef.
- R. Wu, C. Zhu, G. Lü, S. Yang, Z. Meng, X. Zhang, X. Lu, J. Yu, W. Chen and M. Long, Res. Astron. Astrophys., 2024, 24, 055002 CrossRef CAS.
- D. Gobrecht, S. Cristallo, L. Piersanti and S. T. Bromley, Astrophys. J., 2017, 840, 117 CrossRef.
- K. W. Hinkle, J. J. Keady and P. F. Bernath, Science, 1988, 241, 1319–1322 CrossRef CAS PubMed.
- A. M. Mebel, M. Agúndez, J. Cernicharo and R. I. Kaiser, Astrophys. J., Lett., 2023, 945, L40 CrossRef CAS.
Footnote |
| † Present address: Departamento de Química, Universidade Federal de Ouro Preto, Minas Gerais 35402-136, Brazil. |
| This journal is © The Royal Society of Chemistry 2026 |