Open Access Article
Open Access ArticleSilver-catalysed intermolecular benzylic-selective C–H amidation via nitrene transfer
Subir
Panja
,
Tuan Anh
Trinh
 ,
Ethan M.
Warrington
,
Derek B.
Hu
,
Leah C.
Garman
,
Ilia A.
Guzei
and
Jennifer M.
Schomaker
,
Ethan M.
Warrington
,
Derek B.
Hu
,
Leah C.
Garman
,
Ilia A.
Guzei
and
Jennifer M.
Schomaker
 *
*
Department of Chemistry, University of Wisconsin–Madison, 1101 University Avenue, Madison, WI 53706, USA
First published on 17th March 2026
Abstract
Transition metal-catalysed C–H functionalisation is a powerful strategy to upgrade simple hydrocarbons to versatile synthetic building blocks and is a useful tool for the late-stage functionalisation of complex molecules. In this study, we report an intermolecular, non-directed amidation of benzylic C–H bonds via a nitrene transfer pathway. This operationally simple method uses inexpensive silver-based catalysts, only a small excess of substrate and displays broad substrate scope that includes arenes, biaryls, heteroarenes, and complex molecules. Changes to the AgNTf2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) tert-butylterpyridine ligand ratio furnish dimeric or trimeric Ag complexes as the proposed active catalysts; these show differing reactivity and selectivity dependent on the nature of the substrate's benzylic C–H bond.
tert-butylterpyridine ligand ratio furnish dimeric or trimeric Ag complexes as the proposed active catalysts; these show differing reactivity and selectivity dependent on the nature of the substrate's benzylic C–H bond.
Introduction
Transition metal-catalysed C–H functionalisation is a powerful method to efficiently increase the molecular complexity of abundant hydrocarbons.1 Amines are found in pharmaceuticals, agrochemicals, natural products, and in key ligands for catalysis; thus, there is significant interest in methods for the selective introduction of C–N bonds into readily available precursors.2 Traditional functional group interconversions,3 reductive amination,4 or alkene hydroaminations5 often involve multiple steps, require precious metal catalysts or are limited in scope. In contrast, nitrene transfer (NT) enables direct conversions of diverse C–H bonds to new C–N bonds6 using metals that include Rh,7a–c Ru,7d–f Ir,7g Cu,7h,i Fe,7j–m Co,7o,p Mn7q and Ag,7r–v among others. While recent reports from the Zhang,7o,q White,7m,q Chang,8a Du Bois,7b,8b Pérez,7r,8c Dauban,8e Arnold8e,f and our own group (among others),7t–v,8g have driven progress in the field, intermolecular NT into benzylic C–H bonds9via a nitrene transfer process using Rh,8b,9a Ag,9b,c Co,9d Cu,9e Mn,9f Fe9g catalysts can be challenging. Depending on the metal, disadvantages include high cost,8b,d,9a lack of site- and chemo-selectivity,7g,h the need for pre-oxidized nitrene precursors,9f difficult protein purification and aqueous conditions.9g Other drawbacks include long reaction times, modest yields and the use of large excesses of the hydrocarbon substrate (>5 equiv.), which limits application to late-stage functionalisation. Thus, simple methods for catalyst-controlled, selective amidations under mild conditions and in high yield are in demand for substrates that contain multiple reactive C–H sites.10Our group has developed a suite of methods for silver-catalysed NT with the capacity for catalyst-controlled, chemo- and regiodivergent C–H amidations.7t–v,11–13 The site-selectivity of intramolecular NT is tunable via modifications to the nitrene precursor, ligand, Ag![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ligand ratio, or tether length between the nitrogen and the targeted C–H bond. In contrast to NT catalysts with fixed and/or bespoke ligand scaffolds, Ag(I) complexes with simple N-donor ligands can adopt diverse geometries in solution, including linear, tetrahedral, square planar, or seesaw.14 This unusual feature enables control over the steric environment of the reactive Ag-supported nitrenoid species and precise tuning of chemo-, site-, and stereoselectivity, typically aided by directing Ag⋯π and arene–arene interactions between the catalyst and the substrate.11–13
ligand ratio, or tether length between the nitrogen and the targeted C–H bond. In contrast to NT catalysts with fixed and/or bespoke ligand scaffolds, Ag(I) complexes with simple N-donor ligands can adopt diverse geometries in solution, including linear, tetrahedral, square planar, or seesaw.14 This unusual feature enables control over the steric environment of the reactive Ag-supported nitrenoid species and precise tuning of chemo-, site-, and stereoselectivity, typically aided by directing Ag⋯π and arene–arene interactions between the catalyst and the substrate.11–13
Results and discussion
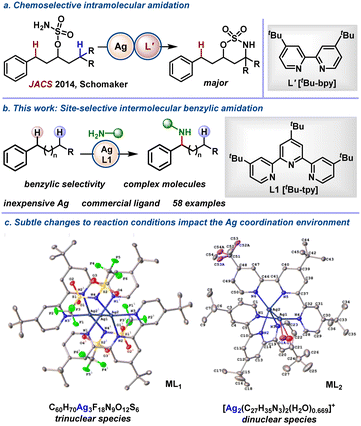
Our previous work on intramolecular NT reactions between two competing C–H bonds showed that the site-selectivity could be tuned through the ligand identity. For example, a silver salt supported by 2 equivalents of a bidentate N-donor ligand, such as tert-butylbipyridine (tBu-bpy), preferred reaction at a more electron-rich C–H (Scheme 1a)12a over a benzylic C–H bond; however, changing to a fluxional tris(2-pyridylmethyl)amine (tpa) ligand favored benzylic C–H amidation. In another system, altering the ligand on Ag from tBu-bpy to a terpyridine (tpy) favored chemoselective C–H bond amidation over aziridination in homoallenic carbamate precursors (not shown).13b Terpyridines have low-lying LUMOs, provide tight chelation with cationic Ag(I) in a stable co-planar conformation and increase the coordination number at the metal, all factors we have observed to impact site-selectivity.15a,b We were curious if the selectivity observed with Ag salts supported by multidentate terpyridine-based ligands might be extended to intermolecular amidations of benzylic C–H bonds. In this work, we present a mild, practical Ag-catalysed method for amidation of benzylic C(sp3)–H bonds in arenes, biaryls, heteroarenes, and complex molecules (Scheme 1b). The protocol does not use costly metals or ligands, proceeds quickly at room temperature, and avoids the need for pre-oxidization of the nitrene precursor. An intriguing effect on the geometry of the proposed active Ag catalysts was noted when changing the ratios of AgNTf2 and L1 (Scheme 1c), with ML1 and ML2 displaying differences in reactivity and selectivity, depending on the substrate (Scheme 2, vide infra).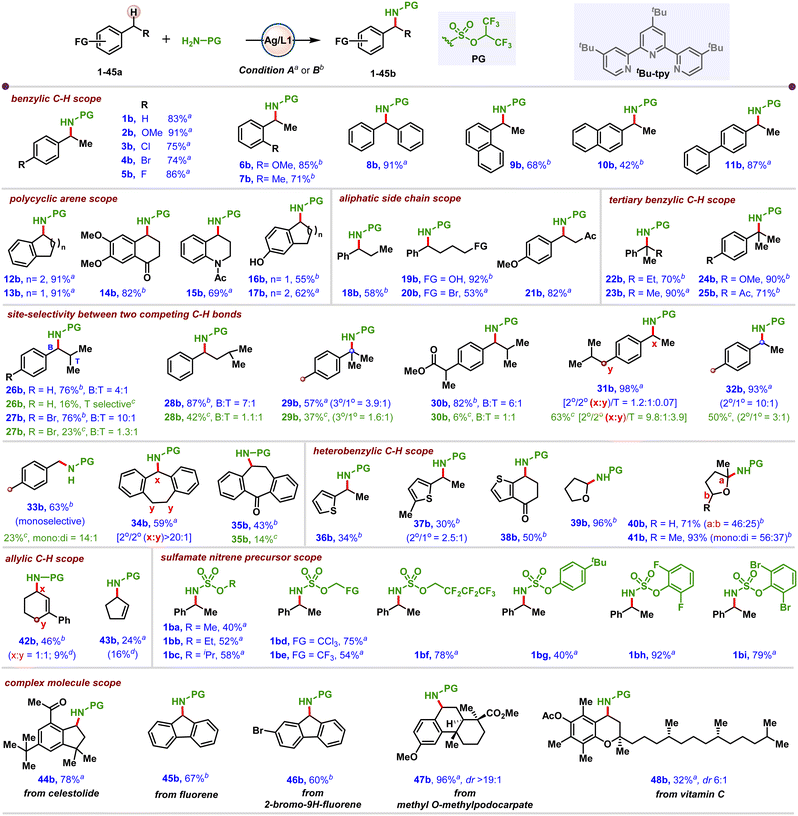 | ||
| Scheme 2 Scope of benzylic-selective intermolecular Ag-catalysed C–H amidation. aCondition A: arene (2 equiv.), HfpsNH2 (0.025 mmol), AgNTf2 (20 mol%), tBu-tpy (12 mol%), PhIO (3 equiv.), 4 Å MS (25 mg), σ-DCB (0.05 M), 4 h, rt, under air. Yields determined from crude NMR of the reaction mixture using 1,3,5-(MeO)3C6H3 as internal standard. bCondition B: arene (5 equiv.), HfpsNH2 (0.025 mmol), AgNTf2 (10 mol%), tBu-tpy (12 mol%), pentafluoroiodosobenzene (PFIOB, 2 equiv.), 4 Å MS (25 mg), PhH (0.1 M), 4 h, rt, under air. Yields and selectivity are determined from the crude NMR of the reaction mixture using 1,3,5-(MeO)3C6H3 as internal standard. cRh-catalysed conditions from ref. 16. dAziridination product. | ||
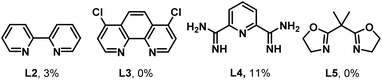
Treatment of ethylbenzene 1a with 1,1,1,3,3,3-hexa-fluoropropan-2-yl sulfamate (HfspNH2) as the nitrene precursor using condition A afforded an 85% yield of 1b (Table 1, entry 1), which was lowered to 45% in TFE (entry 2). AgOTf and AgTFA proved less effective (entries 3–4), while lower and higher catalyst loadings also decreased the yields of 1b (entries 5–6). The identity of the ligand was key in controlling the efficiency and selectivity of the NT event. Screening of a series of bipyridine, phenanthroline, terpyridine, and other bidentate/tridentate ligands (L2–L5, Table 1; see the SI, Table S16 for additional ligands), showed that tButpy (L1) was optimal. The use of 4 Å molecular sieves (MS) as an additive was best (entries 7–8). Changing the solvent to MeCN (entry 9) gave poor yield, while changing the reaction time decreased the yield of 1b (entries 10–11). Finally, control experiments confirmed that both AgNTf2 and the ligand are essential for product formation (entries 12–13). For full optimization studies, see Tables S1–S17 in the SI.
| Entry | Change from condition A | 1b % yield | 1b′ % yield |
|---|---|---|---|
| a Standard conditions: condition A: arene (2 equiv.), HfpsNH2 (0.05 mmol), AgNTf2 (20 mol%), tBu-tpy (12 mol%), PhIO, (3 equiv.), 4 Å molecular sieves (50 mg), σ-DCB (0.05 M), 4 h, rt, under air. b Yields determined by crude NMR of the reaction mixture using 1,3,5-(MeO)3C6H3 as an internal standard. ML1 = [Ag3(tBu-tpy)2(NTf2)3]. | |||
| 1 | None | 85 | 10 |
| 2 | TFE | 45 | 11 |
| 3 | AgOTf instead of AgNTf2 | 38 | 20 |
| 4 | AgTFA instead of AgNTf2 | 29 | 3 |
| 5 | AgNTf2 (5 mol%) | 41 | 11 |
| 6 | AgNTf2 (25 mol%) | 76 | 14 |
| 7 | 3 Å MS | 63 | 4 |
| 8 | 5 Å MS | 45 | 2 |
| 9 | MeCN | 20 | 5 |
| 10 | 2 h reaction time | 29 | <1 |
| 11 | 8 h reaction time | 60 | 8 |
| 12 | No Ag catalyst | 0 | 2 |
| 13 | No ligand | 0 | — |
|
|||
The Ag complex formed under reaction condition A was subjected to SC-XRD; the resulting ML1 shows an unexpected linear arrangement of three Ag atoms (Scheme 1c). Resubjecting ML1 to condition A for the reaction of 1a to 1b, minus the addition of AgNTf2 and L1 (see details in S4.6.2 of the SI), gave results that were similar to condition A only. However, biased substrates with competing benzylic and tertiary C(sp3)–H bonds gave poor yield with ML1 (see SI, Tables S18–S33); thus, a second set of conditions was developed (condition B, Scheme 2). The solvent was switched to PhH using AgNTf2 (10 mol%), L1 (12 mol%), PFIOB (2 equiv.), and 4 Å MS at rt for 4 h to form ML2 (Scheme 1c) as the proposed active catalyst (see Section 4.6.2 in the SI for further details).
With conditions A and B in hand, the scope of the amidation was explored (Scheme 2). An electron-rich MeO- substituted arene gave excellent yields (2b), while halogenated arenes were also well-tolerated (3a–5a), offering valuable opportunities for subsequent functionalisations. Installing an ortho-OMe group in 6a gave an excellent 85% yield of 6b, while a Me group resulted in a 71% yield of 7b due to stereoelectronic factors. Diphenylmethane 8a afforded a 91% yield of 8b. The scope was extended to bicyclic arenes and biaryl derivatives, including 1-ethyl naphthalene 9a, 2-ethyl naphthalene 10a, and 4-phenyl ethylbenzene 11a, which delivered good-to-excellent yields of 9b–11b. Polycyclic arenes 1,2,3,4-tetrahydronaphthalene 12a, 2,3-dihydro-1H-indene 13a, and electron-poor 6,7-dimethoxy-3,4-dihydronaphthalen-1(2H)-one derivative 14a also performed well under this amidation strategy (12b–14b). An N-protected amine derivative reacts smoothly (15a), while free amine inhibits the reaction (see the SI for details). Phenolic –OH groups were also tractable under the reaction conditions (16b–17b). Arenes 18a–21a bearing longer side chains gave good yields of 18b–21b; notably, aliphatic –OH and –Br groups were well-tolerated under the reaction conditions. Tertiary benzylic C–H bonds in 22a–25a were amidated to furnish 22b–25b in excellent yields, suggesting that the ML2 catalytic species proposed to form under condition B is better able to tolerate steric congestion as compared to ML1.
To further challenge our NT protocol, substrates bearing both benzylic and tertiary alkyl C–H bonds (26a–28a), as well as those containing competing C–H sites (3° vs. 1° in 29a, 3° vs. 2° in 30a, 2° vs. 2° in 31a, 2° vs. 1° in 32a and 1° vs. 1° in 33a), were evaluated. For the purposes of comparison, results are also shown for reported Rh-catalysed NT.16 Precursors 26a–30a afforded good site-selectivity, although as previously mentioned, it was necessary to employ a second set of reaction conditions (condition B, see Section 3.2 in the SI for further details) to achieve good results. The less bulky ML2 that is proposed to form under these conditions appears to better tolerate steric congestion than the complex ML1. Competing 2° vs. 2° benzylic C–H bonds in 31a gave no selectivity in 31b, while a 2° benzylic C–H was heavily favored over the primary C–H bond in 32a.
As expected, the doubly activated C–H bond in 34a preferentially delivers 34b, while blocking this site shifts the product to 35b, albeit in lower yield. Thiophene derivatives 36a–38a were tolerated, giving modest yields of 36b–38b. Amidation at the α-oxygenated carbon of tetrahydrofuran derivatives 39a–41a furnished excellent yields of 39b–41b, representing a facile way to activate this position towards further substitution via an oxonium ion.17
In addition to benzylic C–H amidation, allylic C–H bonds in 42a–43a could be functionalised to deliver 42b–43b, albeit with minor amounts of the aziridination byproduct. The identity of the nitrene precursor was briefly explored, with aliphatic and aromatic side chains on the sulfamate giving the products in good to excellent yields (1ba–1bi). Finally, to demonstrate the potential of this chemistry as a tool for late-stage intermolecular NT in complex molecules, several biorelevant scaffolds including celestolide, fluorene derivatives, methyl o-methylpodocarpate and vitamin C were examined (44a–48a). The experimental results showed that the trinuclear metal–ligand complex (ML1) prefers unhindered benzylic C–H bonds, while the dinuclear complex ML2 performs better for NT into more sterically crowded C–H sites (see Section 4.8 in the SI for more details).
The nitrogen protecting group of 1b was easily removed through a mild hydrolysis to deliver 1bj (Scheme 3a) in 87% yield. The intermolecular NT chemistry was readily scalable and demonstrated excellent yields using condition A (Scheme 3b and Section 4.2 in the SI).
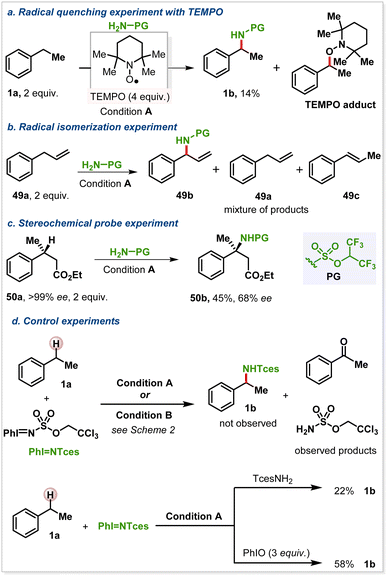
We conducted additional studies to better understand the behavior of this unusual intermolecular Ag-catalysed NT reaction. First, the reaction was performed in the presence of various radical quenchers (Scheme 4a). A TEMPO ((2,2,6,6-tetramethylpiperidin-1-yl)oxyl) adduct was detected in the reaction of 1a to 1b (HRMS: m/z for [M + H]+ calc 262.2165; found, 262.1340), formed by the interaction of TEMPO and benzyl radical (Scheme 4a). Coupling this result with the observed isomerization of the double bond of 49a (Scheme 4b) and a stereochemical probe experiment of 50a (Scheme 4c) suggested the potential for a relatively long-lived radical species under these reaction conditions.
Interestingly, treatment of 1a with PhI![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NTces (Scheme 4d) did not give the desired 1b; rather, only a mixture of acetophenone and TcesNH2 was observed (see Section 4.9 in the SI for further details). We found that the addition of either TcesNH2 or 3 equiv. PhIO partially restored the desired reactivity; however, efforts to establish a causal relationship between the rates of formation of PhI
NTces (Scheme 4d) did not give the desired 1b; rather, only a mixture of acetophenone and TcesNH2 was observed (see Section 4.9 in the SI for further details). We found that the addition of either TcesNH2 or 3 equiv. PhIO partially restored the desired reactivity; however, efforts to establish a causal relationship between the rates of formation of PhI![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NTces and 1b were unsuccessful due to the heterogeneity of the reaction mixture and our inability to detect an iminoiodinane intermediate by NMR. It is possible that an equilibrium exists between TcesNH2 and PhI
NTces and 1b were unsuccessful due to the heterogeneity of the reaction mixture and our inability to detect an iminoiodinane intermediate by NMR. It is possible that an equilibrium exists between TcesNH2 and PhI![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NTces that heavily favors the former, where excess oxidant helps drive the equilibrium toward the desired reactivity and ‘recycle’ TcesNH2. However, if the reaction proceeds through a typical metal-nitrene species derived from an intermediate iminoiodinane, a preformed iminoiodinane should also perform the desired nitrene transfer. These unexpected observations suggest that the reaction mechanism may involve additional processes beyond the simple generation and transfer of an iminoiodinane species, which is the generally accepted mechanistic pathway for these types of nitrene transfer reactions. Further investigation is needed to conclusively establish the mechanism.
NTces that heavily favors the former, where excess oxidant helps drive the equilibrium toward the desired reactivity and ‘recycle’ TcesNH2. However, if the reaction proceeds through a typical metal-nitrene species derived from an intermediate iminoiodinane, a preformed iminoiodinane should also perform the desired nitrene transfer. These unexpected observations suggest that the reaction mechanism may involve additional processes beyond the simple generation and transfer of an iminoiodinane species, which is the generally accepted mechanistic pathway for these types of nitrene transfer reactions. Further investigation is needed to conclusively establish the mechanism.
Conclusions
In summary, we have demonstrated a benzylic-selective, non-directed Ag-catalysed intermolecular benzylic amidation of arenes, biaryls, heteroarenes and complex molecules containing these motifs. Three main design principles in this strategy include: (i) targeting the weakest C–H bond via a stepwise HAA, (ii) using bulky ligands to favor 2° benzylic sites, and (iii) leveraging the steric differences between di- and trinuclear Ag complexes to further fine-tune the site-selectivity. The method uses inexpensive silver salts, commercially available ligands and proceeds at rt. Ongoing efforts are directed toward exploring the role of non-covalent interactions between the catalyst and substrate, including π⋯π and Ag⋯π interactions, that may aid in directing the selectivity of the NT event and stabilize the transition state.14Author contributions
The manuscript was written by S. P. and J. M. S. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2492278 and 2492279 contain the supplementary crystallographic data for this paper.18a,bThe data that support the findings in this work are available within the paper and have been included as part of the supporting information (SI). Supplementary information: experimental procedures, full optimization of the methodology, characterization data and spectra for the synthesis of the precursors and products of the silver-catalysed nitrene transfer reactions. Full SC-XRD information is provided for ML1 (CCDC 2492278) and ML2 (CCDC 2492279). Copies of NMR and HPLC spectra are also included. See DOI: https://doi.org/10.1039/d5sc10184k.
Acknowledgements
J. M. S. is grateful to the NSF (CHE-2247217) for funding this research. Financial support was also provided by a Dickinson Graduate Fellowship and a Doban Mentorship Award to T. A. T. and an ACS DOC SURF Fellowship and a Hilldale Research Fellowship to E. M. W. Dr Heike Hofstetter, Dr Cathy Clewett and Dr Gabi Carosio at UW-Madison are thanked for help with NMR techniques. Dr Martha Vestling at UW-Madison is thanked for mass spectrometry characterization.Notes and references
- (a) H. M. L. Davies, J. D. Bois and J.-Q. Yu, Chem. Soc. Rev., 2011, 40, 1855–1856 Search PubMed; (b) J. Yamaguchi, A. D. Yamaguchi and K. Itami, Angew Chem. Int. Ed. Engl., 2012, 51, 8960–9009 CrossRef; (c) L. Guillemard, N. Kaplaneris, L. Ackermann and M. J. Johansson, Nat. Rev. Chem., 2021, 5, 522–545 CrossRef CAS.
- (a) S. A. Lawrence, Amines: synthesis, properties and applications, Cambridge University Press, New York, NY, 2004 Search PubMed; (b) M. B. Smith, Compendium of organic synthetic methods, Wiley, New York, NY, 2009, vol. 12 Search PubMed; (c) W. S. Emerson, Organic reactions, Wiley, New York, NY, 2004 Search PubMed; (d) I. Schmeltz and D. Hoffmann, Chem. Rev., 1977, 77, 295–311 Search PubMed.
- (a) F. Khan, M. Dlugosch, X. Liu and M. G. Banwell, Acc. Chem. Res., 2018, 51, 1784–1795 CrossRef CAS PubMed; (b) J.-Q. Chen, J.-H. Li and Z.-B. Dong, Adv. Synth. Catal., 2020, 362, 3311–3331 CrossRef CAS; (c) P. A. Forero-Cortes and A. M. Haydl, Org. Process Res. Dev., 2019, 23, 1478–1483 CrossRef CAS; (d) L. Song, L. Cai, L. Gong and E. V. Van der Eycken, Chem. Soc. Rev., 2023, 52, 2358–2376 RSC.
- O. I. Afanasyev, E. Kuchuk, D. L. Usanov and D. Chusov, Chem. Rev., 2019, 119, 11857–11911 CAS.
- (a) J.-H. Xie, S.-F. Zhu and Q.-L. Zhou, Chem. Rev., 2011, 111, 1713–1760 CrossRef CAS PubMed; (b) L. Huang, M. Arndt, K. Gooßen, H. Heydt and L. J. Gooßen, Chem. Rev., 2015, 115, 2596–2697 CrossRef CAS.
- For selected reviews on transition metal-catalyzed nitrene transfer, see: (a) D. N. Zalatan and J. Du Bois, Curr. Chem., 2009, 292, 347–378 Search PubMed; (b) P. Muller and C. Fruit, Chem. Rev., 2003, 103, 2905–2920 Search PubMed; (c) Z. G. Li and C. He, Eur. J. Org Chem., 2006, 4313–4322 CrossRef CAS; (d) J. A. Halfen, Curr. Org. Chem., 2005, 9, 657–669 CrossRef CAS; (e) G. Dequirez, V. Pons and P. Dauban, Angew. Chem., Int. Ed., 2012, 51, 7384–7395 Search PubMed; (f) F. Collet, R. H. Dodd and P. Dauban, Chem. Commun., 2009, 34, 5061–5074 RSC; (g) L. Degennaro, P. Trinchera and R. Luisi, Chem. Rev., 2014, 114, 7881–7929 CrossRef CAS; (h) M. Ju and J. M. Schomaker, Nat. Rev. Chem., 2021, 5, 580–594 CrossRef CAS PubMed.
- For selected references, see: (a) F. Collet, C. Lescot, C. G. Liang and P. Dauban, Dalton Trans., 2010, 39, 10401–10413 RSC; (b) J. L. Roizen, M. E. Harvey and J. Du Bois, Acc. Chem. Res., 2012, 45, 911–922 CrossRef CAS; (c) A. Fanourakis, B. D. Williams, K. Paterson and R. J. Phipps, J. Am. Chem. Soc., 2021, 143, 10070–10076 CrossRef CAS; (d) S.-M. Au, W.-H. Fung, M.-C. Cheng, C.-M. Che and S.-M. Peng, Chem. Commun., 1997, 1655–1656 RSC; (e) M. E. Harvey, D. G. Musaev and J. Du Bois, J. Am. Chem. Soc., 2011, 133, 17207 CrossRef CAS; (f) Y. Nishioka, T. Uchida and T. Katsuki, Angew. Chem., Int. Ed., 2013, 52, 1739–1742 CrossRef CAS; (g) S. Y. Hong, Y. Park, Y. Hwang, Y. B. Kim, M.-H. Baik and S. Chang, Science, 2018, 359, 1016–1021 Search PubMed; (h) D. A. Evans, M. M. Faul and M. T. Bilodeau, J. Org. Chem., 1991, 56, 6744–6746 CrossRef CAS; (i) F. Dielmann, D. M. Andrada, G. Frenking and G. Bertrand, J. Am. Chem. Soc., 2014, 136, 3800–3802 CrossRef CAS; (j) S. Kuppuswamy, T. M. Powers, B. M. Johnson, M. W. Bezpalko, C. K. Brozek, B. M. Foxman, L. A. Berben and C. M. Thomas, Inorg. Chem., 2013, 52, 4802–4811 CrossRef CAS; (k) E. R. King, E. T. Hennessy and T. A. Betley, J. Am. Chem. Soc., 2011, 133, 4917–4923 CrossRef CAS; (l) J. J. Tang, X. Yu, Y. Wang, Y. Yamamoto and M. Bao, Angew. Chem., 2021, 133, 16562–16571 CrossRef; (m) S. M. Paradine and M. C. White, J. Am. Chem. Soc., 2012, 134, 2036 CrossRef CAS; (n) W. C. Lee and X. P. Zhang, Nat. Chem., 2023, 15, 1499–1500 CrossRef; (o) H.-J. Lu, H.-L. Jiang, L. Wojtas and X. P. Zhang, Angew. Chem., Int. Ed., 2010, 49, 10192–10196 CrossRef CAS; (p) H. J. Lu, V. Subbarayan, J. R. Tao and X. P. Zhang, Organometallics, 2010, 29, 389–393 CrossRef CAS; (q) S. M. Paradine, J. R. Griffin, J. Zhao, A. L. Petronico, S. M. Miller and M. C. White, Nat. Chem., 2015, 7, 987–994 CrossRef CAS PubMed; (r) M. R. Fructos, S. Trofimenko, M. M. Diaz-Requejo and P. J. Perez, J. Am. Chem. Soc., 2006, 128, 11784 CrossRef CAS PubMed; (s) Y. Cui and C. He, Angew. Chem., Int. Ed., 2004, 43, 4210 CrossRef CAS; (t) J. M. Alderson, J. R. Corbin and J. M. Schomaker, Acc. Chem. Res., 2017, 50, 2147–2158 Search PubMed; (u) W. Liu, I. Choi, E. E. Zerull and J. M. Schomaker, ACS Catal., 2022, 12, 5527–5539 CrossRef CAS; (v) E. E. Zerull, L. E. Vine and J. M. Schomaker, Synlett, 2021, 32, 30–44 CrossRef.
- For selected references, see: (a) Y. Park, Y. Kim and S. Chang, Chem. Rev., 2017, 117, 9247–9301 CrossRef CAS PubMed; (b) K. W. Fiori and J. Du Bois, J. Am. Chem. Soc., 2007, 129, 562 CrossRef CAS PubMed; (c) B. P. Gomez-Emeterio, J. Urbano, M. M. Diaz-Requejo and P. J. Perez, Organometallics, 2008, 27, 4126–4130 CrossRef CAS; (d) F. Collet, C. Lescot and P. Dauban, Chem. Soc. Rev., 2011, 40, 1926–1936 RSC; (e) T. K. Hyster, C. C. Farwell, A. R. Buller, J. A. McIntosh and F. H. Arnold, J. Am. Chem. Soc., 2014, 136, 15505–15508 CrossRef CAS PubMed; (f) A. Das, S. Gao, S. V. Athavale, E. Alfonzo, Y. Long and F. H. Arnold, Methods Enzymol., 2023, 693, 1–30 CAS; (g) R. M. Ward and J. M. Schomaker, Handbook of CH-Functionalization (CHF), John Wiley & Sons, Chichester, UK, 2022, vol. 13, pp. 1–37 Search PubMed.
- (a) A. Nasrallah, V. Boquet, A. Hecker, P. Retailleau, B. Darses and P. Dauban, Angew. Chem., Int. Ed., 2019, 58, 8192–8196 CrossRef CAS PubMed; (b) A. Beltrana, C. Lescot, M. Mar Díaz-Requejoa, P. J. Perez and P. Dauban, Tetrahedron, 2013, 69, 4488–4492 CrossRef; (c) N. S. Dolan, R. J. Scamp, T. Yang, J. F. Berry and J. M. Schomaker, J. Am. Chem. Soc., 2016, 138, 14658–14667 Search PubMed; (d) H. Lu, V. Subbarayan, J. Tao and X. P. Zhang, Organometallics, 2010, 29, 389–393 Search PubMed; (e) R. T. Gephart and T. H. Warren, Organometallics, 2012, 31, 7728–7752 CrossRef CAS; (f) J. R. Clark, K. Feng, A. Sookezian and M. C. White, Nat. Chem., 2018, 10, 583–591 Search PubMed; (g) C. K. Prier, R. K. Zhang, A. R. Buller, S. B. Chen and F. H. Arnold, Nat. Chem., 2017, 9, 629–634 Search PubMed.
- For selected examples, see: (a) E. Brunard, V. Boquet, E. V. Elslande, T. Saget and P. Dauban, J. Am. Chem. Soc., 2021, 143, 6407–6412 Search PubMed; (b) W. Liu, Z. Ren, A. T. Bosse, K. Liao, E. L. Goldstein, J. Bacsa, D. G. Musaev, B. M. Stoltz and H. M. L. Davies, J. Am. Chem. Soc., 2018, 140, 12247–12255 Search PubMed.
- (a) T. A. Trinh, S. Cherempei, D. S. Rampon and J. M. Schomaker, Chem. Sci., 2025, 16, 4796–4805 Search PubMed; (b) Y. Fu, E. E. Zerull, J. M. Schomaker and P. Liu, J. Am. Chem. Soc., 2022, 144, 2735–2746 CrossRef CAS.
- (a) M. Huang, T. Yang, J. D. Paretsky, J. F. Berry and J. M. Schomaker, J. Am. Chem. Soc., 2017, 139, 17376–17386 CrossRef CAS; (b) E. Z. Schroeder, J. H. Kim, Y. Fu, W. Liu, P. Liu and J. M. Schomaker, Chem Catal., 2025, 5, 101388 Search PubMed.
- (a) J. M. Alderson, A. M. Phelps, R. J. Scamp, N. S. Dolan and J. M. Schomaker, J. Am. Chem. Soc., 2014, 136(48), 16720–16723 Search PubMed; (b) J. W. Rigoli, C. D. Weatherly, J. M. Alderson, B. T. Vo and J. M. Schomaker, J. Am. Chem. Soc., 2013, 135, 17238–17241 Search PubMed.
- For selected references, see: (a) F. Hung-Low, A. Renz and K. K. Klausmeyer, Polyhedron, 2009, 28, 407 CrossRef CAS; (b) F. Hung-Low, A. Renz and K. K. Klausmeyer, J. Chem. Crystallogr., 2011, 41, 1174 CrossRef CAS; (c) J. Du, T. Hu, S. Zhang, Y. Zeng and X. Bu, CrystEngComm, 2008, 10, 1866 Search PubMed; (d) H. Zhang, L. Chen, H. Song and G. Zi, Inorg. Chim. Acta, 2011, 366, 320 Search PubMed; (e) K. Xiong, M. Wu, Q. Zhang, W. Wei, M. Yang, F. Jiang and M. Hong, Chem. Commun., 2009, 1840 Search PubMed; (f) F. Hung-Low, A. Renz and K. K. Klausmeyer, J. Chem. Crystallogr., 2009, 39, 438 Search PubMed; (g) N. Schultheiss, D. R. Powell and E. Bosch, Inorg. Chem., 2003, 42, 5304 Search PubMed.
- (a) C. Weib, Y. Heb, X. Shia and Z. Song, Coord. Chem. Rev., 2019, 385, 1–19 Search PubMed; (b) R. Abhijnakrishna, K. Magesh, A. Ayushi and S. Velmathi, Coord. Chem. Rev., 2023, 496, 215380 CrossRef CAS.
- E. N. Bess, R. J. DeLuca, D. J. Tindall, M. S. Oderinde, J. L. Roizen, J. Du Bois and M. S. Sigman, J. Am. Chem. Soc., 2014, 136, 5783–5789 CrossRef CAS PubMed.
- (a) I. Buslov and X. Hu, Adv. Synth. Catal., 2014, 356, 3325–3330 Search PubMed; (b) T. Fuchigami, T. Sato and T. Nonaka, J. Org. Chem., 1986, 51, 366–369 Search PubMed.
- (a) CCDC 2492278: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pnf2w; (b) CCDC 2492279: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pnf3x.
| This journal is © The Royal Society of Chemistry 2026 |