Open Access Article
Open Access ArticleFrom perovskite top interfaces to metal contacts: stability-driven design for tandem-compatible inverted solar cells
Sangmi Park†
a,
Jong Bin Park†b,
Jung Min Hab,
Hye Seung Kimac,
Myoung Hoon Song *ac and
Han Young Woo
*ac and
Han Young Woo *b
*b
aDepartment of Materials Science and Engineering, Ulsan National Institute of Science and Technology (UNIST), UNIST-gil 50, Ulsan, 44919, Republic of Korea. E-mail: mhsong@unist.ac.kr
bDepartment of Chemistry, Korea University, Seoul 02841, Republic of Korea. E-mail: hywoo@korea.ac.kr
cUNIST InnoCORE AI-driven Space Solar Initiative, Ulsan National Institute of Science and Technology (UNIST), Ulsan, 44919, Republic of Korea. E-mail: mhsong@unist.ac.kr
First published on 24th April 2026
Abstract
Inverted perovskite solar cells (PeSCs) have attracted growing attention, due to their low-temperature processability, reduced hysteresis, and strong compatibility with flexible and tandem device architectures, and have now surpassed conventional devices in terms of both power conversion efficiency and operational stability. Despite these advantages, their long-term operational stability remains critically constrained by the upper device stack, spanning from the perovskite surface through the electron-transporting layer (ETL) to the metal cathode. This review systematically analyzes the dominant origins of instability at these top interfaces in inverted PeSCs and organizes recent progress into three key design directions: (i) perovskite/ETL interface engineering using organic and inorganic modifiers, low-dimensional perovskite capping layers, and thermally conductive interlayers; (ii) C60 replacements or non-fullerene ETLs that integrate defect passivation, interfacial dipole tuning, ion management, and aggregation-resistant morphologies; and (iii) ETL/cathode interlayers that regulate band bending, suppress interfacial recombination, and block volatile halide migration. Finally, the Outlook section discusses emerging opportunities and remaining challenges in further optimizing ETLs and top interlayers, with particular emphasis on advancing the operational stability and commercial viability of inverted PeSCs and their integration into high-performance tandem solar cells.
1. Introduction
Over the past decade, perovskite solar cells (PeSCs) have exhibited a steep increase in power conversion efficiency (PCE), reaching approximately 27.0% for single-junction devices, thereby establishing PeSCs as one of the most promising next-generation photovoltaic technologies.1–5 Early efficiency gains were primarily driven by the conventional n–i–p architecture; however, the inverted p–i–n configuration has rapidly matured and, in many cases, surpassed n–i–p devices in both efficiency and operational stability (Table 1). As a result, inverted PeSCs have attracted growing attention due to their low temperature processability, reduced hysteresis, and strong compatibility with flexible and tandem device architectures.| Structure | Device configuration | Cell area (cm2) | PCE (%) | MPP tracking | Encapsulated | Ref. |
|---|---|---|---|---|---|---|
| a MPP: maximum power point/RT: room temperature. | ||||||
| p–i–n | FTO/CbzNaph: JJ24/Cs0.05FA0.85MA0.1PbI3/EDAI2/C60/SnOX ALD/Ag | 0.0712 | 26.92 | T100: 1000 h (85 °C, 78–80% RH) | O | 8 |
| p–i–n | ITO/NiOx/Me-4PACz/Cs0.05MA0.1FA0.85PbI3/piperazinium diiodide/PC61BM/BCP/Ag | 0.05556 | 26.87 | T96: 2500 h (65 °C, 40 ± 5% RH) | O | 28 |
| p–i–n | ITO/PhPAPy/FA0.9MA0.05Cs0.05PbI3/3MTPAI/C60/BCP/Ag | 0.074 | 26.74 | T95: 2000 h (65 °C, ambient humidity) | O | 29 |
| p–i–n | ITO/D4PA/Cs0.05FA0.85MA0.1PbI3/p-F-PEAI/C60/BCP/Ag | 0.0412 | 26.72 | T97.2: 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 h (RT, ambient humidity) 500 h (RT, ambient humidity) |
O | 30 |
| p–i–n | ITO/PATPA + OMe-PhPACz/FA0.85MA0.1Cs0.05PbI3/piperazinium diiodide/C60/BCP/Cu | 0.0715 | 26.21 | T90: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h (N2 atmosphere) 000 h (N2 atmosphere) |
Not stated | 31 |
| p–i–n | FTO/NiOX/Me-4PACz/Cs0.02(FA0.98MA0.02)0.98Pb(I0.99Br0.01)3/PC61BM/BCP/Ag | 0.09 | 26.17 | T80: 423 h (85 °C, N2 atmosphere) | X | 32 |
| p–i–n | ITO/Me-4PACz/Cs0.05FA0.9MA0.05PbI3/PCNI2-BTI/BCP/Ag | 0.15 | 26.00 | T80: 1280 h (RT, 60 ± 10% RH) | O | 33 |
| p–i–n | ITO/MPAcPA/Cs0.04(MA0.05FA0.95)0.96Pb(I0.95Br0.05)3/SPS/C60/BCP/Ag | 0.0625 | 25.85 | T95: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h (RT, N2 atmosphere) 000 h (RT, N2 atmosphere) |
O | 34 |
| p–i–n | ITO/MPA-CPA/Cs0.15FA0.85PbI2.8Cl0.2/(PiEA)PbI4/PC61BM/BCP/Ag | 0.06 | 25.60 | T93: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h (RT, N2 atmosphere) 000 h (RT, N2 atmosphere) |
Not stated | 35 |
| n–i–p | FTO/SnO2/ZnTPPS/Cs0.05FA0.95PbI3/Spiro-OMeTAD/Au | 0.0618 | 26.66 | T90: 1537 h (not stated) | O | 36 |
| n–i–p | ITO/SnO2 (HS)/FAPbI3/PEAI/Spiro-OMeTAD/Au | 0.08 | 26.61 | T94.9%: 1800 h (not stated) | Not stated | 37 |
| n–i–p | ITO/SnO2/FA0.94Cs0.06PbI3/CISQD/Spiro-OMeTAD/Ag | 0.0590 | 26.34 | T80: 2000 h (25 °C, N2 atmosphere) | X | 38 |
| n–i–p | FTO/SnO2/AlCl3/(FA0.83MA0.06Rb0.12)Pb(I0.94Br0.06)3/PEAI/Spiro-OMeTAD/Au | 0.102 | 26.29 | T80: 502 h (25 °C, N2 atmosphere) | X | 39 |
| n–i–p | FTO/SnO2/FAPbI3/MeO-PEAI/Spiro-OMeTAD/Au | 0.093 | 26.20 | T80: 3000 h (30–45 °C, 20–35% RH) | O | 40 |
| n–i–p | FTO/SnO2/FA0.95MA0.05Pb(I0.95Br0.05)3/MeS-HAI/Spiro-OMeTAD/Au | 0.05 | 25.83 | T98: 400 h (30 °C, N2 atmosphere) | X | 41 |
| n–i–p | ITO/LC-modified SnO2/(FA0.70Cs0.04Rb0.02MA0.25)Pb(I0.87Br0.01Cl0.12)3/PEAI/Spiro-OMeTAD/Ag | 0.10 | 25.30 | T90.17: 1000 h (40 ± 5 °C, N2 atmosphere) | X | 42 |
The operating principle of PeSCs begins with photon absorption in the perovskite layer, generating electron–hole pairs. Owing to the low exciton binding energy of perovskites, these photogenerated species readily dissociate into free carriers at room temperature. The built-in electric field and interfacial energy-level offsets then drive carrier transport, with electrons selectively extracted across the perovskite/electron-transporting layer (ETL) interface to the cathode and holes transported through the hole-transporting layer (HTL) toward the anode. The overall PCE is therefore governed by the balance between charge generation, transport, recombination, and extraction. In particular, interfacial nonradiative recombination, defect-assisted trap states, ion migration, and chemical reactions at selective contacts critically affect both efficiency and operational stability. These considerations underscore the central role of interface engineering in optimizing PeSC performance and durability.
A pivotal factor enabling this progress has been the successful implementation of hole-selective self-assembled monolayers (SAMs) at the bottom electrode/perovskite interface.6–8 SAMs allow precise work function (WF) tuning while minimizing parasitic absorption and simultaneously improving interfacial passivation and charge extraction. These advantages have translated into higher device efficiencies, improved reproducibility, and enhanced stability. Accordingly, extensive SAM-focused studies and reviews have firmly established buried bottom-interface engineering as a cornerstone of high-performance inverted PeSCs.9–13
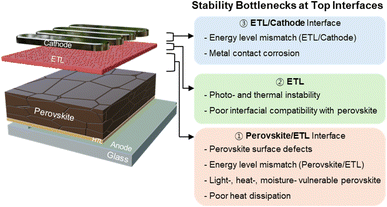
In contrast, despite the substantial advances at the bottom interface, the long-term operational stability of inverted PeSCs is increasingly dictated by processes occurring at the upper device stack, comprising the perovskite top surface, the ETL, and the metal cathode. Recent studies on upper-interface engineering have shown that surface defects,14–16 interfacial chemical reactions,17–19 and suboptimal energy-level alignment20–24 at these junctions can accelerate nonradiative recombination and trigger degradation pathways such as ion migration and electrode corrosion. In addition, commonly used fullerene-based ETLs, including C60 and phenyl-C61-butyric acid methyl ester (PC61BM), often suffer from thermal and morphological instabilities, which further amplify interfacial degradation under prolonged operational stress.25–27 These insights highlight that stability-driven device design must extend beyond bottom-interface optimization to encompass comprehensive control over the perovskite top surface and upper-contact architecture.
In this review, we classify the stability bottlenecks at the upper interfaces of inverted PeSCs into key aspects of perovskite surface defects, energy-level misalignment, inefficient heat dissipation, and the intrinsic instability of ETLs. Correspondingly, we discuss stabilization strategies for the upper device stack across three hierarchical levels: (i) perovskite/ETL interface engineering, (ii) the development of C60-alternative ETLs, and (iii) ETL/cathode interface design (Fig. 1). Finally, we outline future directions toward reliable perovskite photovoltaics, encompassing both single-junction and tandem device architectures. Collectively, this review bridges fundamental interfacial understanding with practical design guidelines for achieving long-term stable PeSCs.
2. Stability issues in top layers of inverted PeSCs
2.1. Surface defects of perovskites
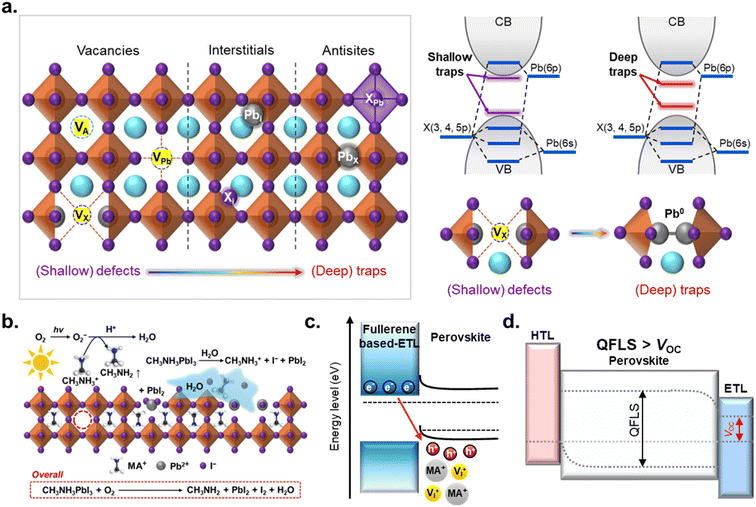
Metal-halide perovskites (MHPs) crystallize in the ABX3 structure, where A is a monovalent cation (methylammonium (MA+), formamidinium (FA+), or cesium (Cs+)), B is a divalent metal cation (Pb2+ or Sn2+), and X is a halide anion (Cl−, Br−, or I−). The A-site cations occupy the cavities of the lattice, while corner-sharing [BX6]4− octahedra form the inorganic framework. Owing to their ionic bonding and mechanically soft lattice, MHP thin films inherently accommodate a high density of point defects, which are preferentially concentrated at surfaces and grain boundaries where structural rigidity is low.43–45 These defects include vacancies (VA, VPb, and VX), interstitials (Ai, Pbi, and Xi), and antisite defects (APb, PbA, AX, PbX, XA, and XPb) (Fig. 2a).46,47 MHPs are often described as defect-tolerant materials because many intrinsic point defects introduce shallow electronic states near the band edges rather than deep mid-gap traps. Nevertheless, even shallow defects can substantially influence nonradiative recombination, ion migration, interfacial energetics, and long-term stability.48,49 Under continuous illumination, electrical bias, or thermal stress, these initially benign defects may evolve into deep traps or activate defect-mediated processes such as ion migration and phase segregation.50 Consequently, defect tolerance in terms of electronic structure does not directly translate into structural or operational stability, and defect formation and passivation remain central challenges in perovskite photovoltaics.Importantly, shallow defects also play a pivotal role in the temporal evolution of degradation. Under-coordinated Pb2+ species, for example, act as nonradiative recombination centers that limit optoelectronic performance and can be irreversibly reduced to metallic Pb0, thereby undermining chemical stability.51–53 Iodide interstitials facilitate halide migration and can accelerate phase transitions, such as the cubic-to-hexagonal transformation, which compromise structural integrity.54 Vacancies further exacerbate degradation by lowering migration barriers, enabling a continuous redistribution of ionic species.55,56 Through these coupled processes, initial surface defects act as nucleation sites for a self-amplifying defect-generation cycle, progressively increasing trap density, enhancing Shockley–Read–Hall recombination, and driving gradual losses in open-circuit voltage (VOC) and fill factor (FF), even when the initial device efficiency is high. Thermal and mechanical factors further intensify defect formation at the perovskite surface. High-temperature annealing promotes the evaporation of volatile MA+/FA+ cations and halide species, leaving PbI2-rich regions and under-coordinated Pb2+ sites. In parallel, residual tensile strain arising from rapid solvent evaporation or thermal expansion mismatch lowers defect formation energies, rendering the lattice more susceptible to vacancy generation during subsequent operation.57–59 As a result, defects in MHPs are not static imperfections but dynamic entities that continuously proliferate throughout the device lifetime.
The vulnerability of surface defects is further amplified by exposure to extrinsic environmental stimuli, particularly at the top surface of inverted PeSCs. Unlike the buried HTL/perovskite interface, the perovskite top surface is directly exposed to oxygen, moisture, and ambient contaminants during and after fabrication. Under illumination, photogenerated electrons can reduce adsorbed O2 to superoxide (O2−), which is a highly reactive species (Fig. 2b).17 O2− extracts protons from MA+, forming methylamine and water, while parallel oxidation pathways generate I2 or HI. The loss of A-site cations destabilizes the perovskite lattice, leading to decomposition into PbI2 and the release of volatile iodine-containing species. Each decomposition step creates additional halide vacancies and under-coordinated Pb2+ sites, thereby feeding back into the intrinsic defect-generation cycle and accelerating long-term degradation. Moisture induces similarly detrimental pathways.60 Water molecules penetrate into grain boundaries and form hydrated intermediates such as MAPbI3·H2O and eventually (MA)4PbI6·2H2O. These hydrated phases distort the [PbI6]4− octahedral framework, weaken A-site binding, and markedly increase ionic mobility. Beyond direct chemical decomposition, hydration significantly lowers the activation energy for halide migration, amplifying secondary defect formation and accelerating the loss of crystallinity.
Taken together, the top surface of inverted PeSCs represents the most vulnerable region where intrinsic defect dynamics and extrinsic degradation pathways converge. Surface defects initiate ionic redistribution, environmental species chemically destabilize the lattice, and illumination and electrical bias further reduce migration barriers. This synergistic interplay leads to a time-dependent expansion of defect populations and cumulative structural decomposition. Ultimately, these processes manifest as reduced carrier lifetimes, enhanced nonradiative recombination, and progressive deterioration of device efficiency and operational stability.
2.2. Energy level alignment between perovskites and the ETL
Fullerene-based ETLs are widely employed in inverted PeSCs due to their high electron mobility and compatibility with low-temperature processing. Nevertheless, voltage losses and stability limitations in fullerene-contacted inverted PeSCs can originate from multiple interfacial factors, including (i) Fermi-level pinning induced by interfacial states that masks the intended band alignment,61,62 (ii) trap-assisted recombination at the perovskite/ETL interface,63,64 and (iii) bias-/illumination-activated interfacial reactions that generate new defect states over time.65,66 In this section, we specifically focus on an alignment-driven instability pathway at the perovskite/ETL interface, where the energetic offset governs interfacial charge accumulation and the subsequent electrostatic and ionic responses during operation.Common fullerene-based ETLs possess relatively deep lowest unoccupied molecular orbital (LUMO) levels (e.g., PC61BM: −3.9 eV, PC71BM: −4.2 eV, C60: −4.1 eV, C70: −4.0 eV, ICBA: −3.7 eV, and Bis-PC60BM: −3.8 eV).67–72 When the ETL LUMO lies substantially below the perovskite conduction band minimum (CBM), a large negative energetic offset (static energetic misalignment) is created, which facilitates electron transfer from the perovskite into the ETL. While such an offset is beneficial for efficient electron extraction, an excessively deep ETL LUMO can lead to pronounced electron accumulation within the ETL and at the perovskite/ETL interface, effectively causing the ETL to function as an electron reservoir.73 The resulting negative charge density on the ETL side electrostatically attracts holes and positively charged ionic species (e.g., MA+, FA+, and VX+) toward the perovskite side of the interface (Fig. 2c). This interfacial charge separation generates a strong local electric field and enhanced band bending, dynamically reshaping the interfacial energy landscape during device operation.74
In MHPs, the resulting interfacial electric field can induce time-dependent redistribution and accumulation of mobile ions; under operating illumination and/or applied bias, these ionic processes are further accelerated, making the interfacial electrostatics increasingly time-dependent (dynamic electrostatics). As a consequence, electron selectivity at the contact is reduced, allowing holes to approach the ETL more readily and thereby enhancing interfacial nonradiative recombination near the junction. Consequently, the enhanced interfacial nonradiative recombination locally reduces the quasi-Fermi level splitting (QFLS) by narrowing the separation between the electron and hole quasi-Fermi levels, which manifests as quasi-Fermi level bending at the interface (Fig. 2d). The externally measured VOC can thus be suppressed beyond what would be expected from the bulk QFLS, resulting in an increased QFLS–qVOC mismatch (q: elementary charge).
Because ionic redistribution occurs on relatively slow timescales, the extent of charge accumulation, band bending, and contact selectivity evolves during operation. This dynamic behavior manifests macroscopically as time-dependent voltage and FF drifts and exacerbated hysteresis. In this regime, inefficient extraction at the perovskite/ETL interface causes photogenerated carriers to accumulate near the junction, further increasing recombination losses and reinforcing the degradation of charge-extraction efficiency. Over extended operation, sustained ion accumulation at the interface can promote additional defect formation by facilitating halide-vacancy generation and ionic rearrangement. Ion-rich interfacial regions also increase chemical reactivity with metal electrodes, such as Ag, enabling interdiffusion and accelerating contact corrosion.75–77 Moreover, in the presence of oxygen and moisture, the elevated interfacial charge and ion density lowers the activation barriers for electrochemical reactions, further amplifying interfacial degradation and long-term instability.
To experimentally substantiate these coupled electrostatic, ionic, and chemical degradation processes at the upper contact, complementary diagnostics that probe both nonradiative losses and operando interfacial evolution are essential. Absolute photoluminescence quantum yield (PLQY) measurements, together with the corresponding QFLS, provide a quantitative metric of radiative efficiency and enable direct comparison with the measured VOC to assess the magnitude and origin of the QFLS–qVOC mismatch.63,64 Electroluminescence (EL) under forward bias further sensitizes contact-related nonradiative recombination and voltage losses.78,79 In addition, transient photovoltage/photocurrent (TPV/TPC), impedance spectroscopy, and transient photoluminescence (TRPL) can resolve the characteristic timescales of charge accumulation and ion redistribution that underpin time-dependent voltage/FF drifts and hysteresis.64,80–82 Finally, operando spectroscopy can directly track the evolution of interfacial energetics, chemical reactions, and ionic redistribution during operation.
2.3. Low thermal conductivity of perovskites
MHPs, owing to their predominantly ionic bonding, are inherently vulnerable to extrinsic stimuli such as moisture, oxygen, heat, and light. While advanced encapsulation technologies can effectively mitigate degradation induced by moisture and oxygen, thermal instability remains a fundamental challenge that cannot be fully addressed by encapsulation alone and instead requires improvements in the intrinsic stability of PeSCs. Under continuous 1-sun illumination, operating device temperatures typically reach 40–65 °C.83 In addition, substantial internal heat is generated through carrier-related processes, including Joule heating, bulk and interfacial recombination, and the thermalization of high-energy photons.84 Heat stress is therefore unavoidable during operation, making effective thermal management and intrinsic thermal robustness essential for long-term device stability.Beyond their chemical sensitivity, perovskites suffer from intrinsically low thermal conductivity, which further aggravates thermal degradation. In these materials, heat transport is dominated by lattice vibrations (phonons) rather than free charge carriers.85 However, a combination of structural and vibrational characteristics severely limits phonon-mediated heat transport, resulting in a lattice thermal conductivity typically below 1.0 W m−1 K−1 at room temperature (Fig. 3a). Inefficient phonon transport in perovskites can be attributed to three primary factors. First, their crystal lattices are mechanically soft and strongly anharmonic, causing phonons to propagate slowly and undergo frequent phonon–phonon scattering.86 This vibrational nonlinearity shortens phonon lifetimes and substantially reduces thermal conductivity. Second, the dominant heat-carrying phonon modes exhibit relatively flat dispersion relations and intrinsically short lifetimes, further suppressing heat transport. The presence of heavy elements such as Pb and I exacerbate these effects by increasing lattice distortion and phonon scattering.87 Third, the high density of defects at surfaces and grain boundaries—including vacancies, dislocations, and dangling bonds—acts as additional phonon-scattering centers, reducing phonon mean free paths and further degrading thermal transport.88–90
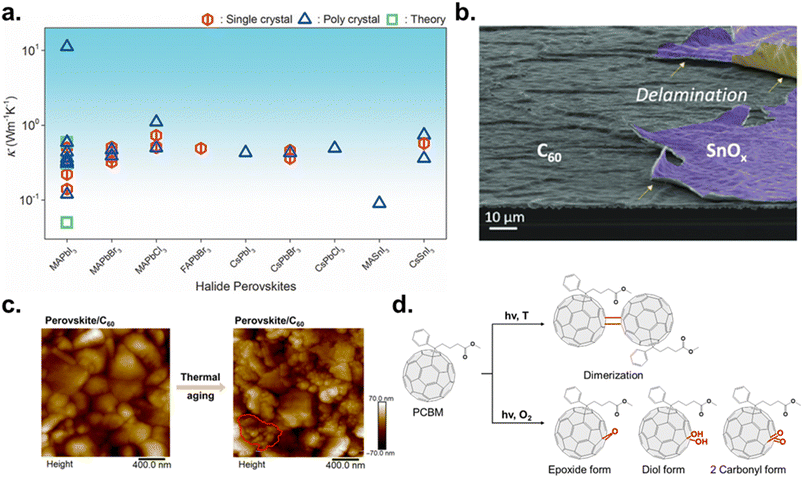 | ||
| Fig. 3 (a) Room temperature experimental and theoretical κ of perovskites. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 85. Copyright 2020, Wiley-VCH GmbH. (b) SEM image of delaminated SnOx on the C60 surface. Reproduced with permission from ref. 99. Copyright 2022, American Chemical Society. (c) Atomic force microscopy (AFM) topography images of a perovskite film covered with C60, before and after thermal aging. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 27. Copyright 2024, the American Association for the Advancement of Science. (d) Schematic of fullerene ETL photoreactions: dimerization of PC61BM under light and thermal stress and photo-oxidation of PC61BM under combined light and oxygen exposure. | ||
As a result of this intrinsically poor heat dissipation, MHPs are prone to local heat accumulation under thermal stress, which activates a cascade of coupled degradation processes. Elevated temperatures promote the decomposition of perovskites into PbX2 and volatile AX-derived species, leading to irreversible loss of the perovskite phase and a concomitant increase in defect density.91,92 The soft and anharmonic lattice also facilitates thermally driven phase transitions, such as the transformation from the photoactive black α-phase to the non-perovskite yellow δ-phase in iodide-based compositions.93,94 Simultaneously, higher temperatures significantly accelerate the migration of mobile ionic species, including halide anions and A-site cations, resulting in halide segregation and local compositional inhomogeneity.18,95,96 Thermally activated ion migration further intensifies interfacial reactions with charge-transport layers and metal electrodes, promoting the formation of interfacial defects and corrosion products that impede charge extraction and exacerbate performance loss. Collectively, these thermally induced processes underscore that inefficient heat dissipation is not merely a secondary issue but a central driver of structural, electronic, and interfacial instability in PeSCs. Consequently, engineering more efficient heat-dissipation pathways and thermally robust interfacial architectures has emerged as a critical strategy for achieving long-term thermal stability in perovskite solar cells.
2.4. Instability of ETL materials
Fullerene-based ETLs, such as C60 and PC61BM, are widely employed in inverted PeSCs owing to their high electron mobility and energetically favorable alignment with the perovskite CBM. However, their intrinsically nonpolar molecular nature results in poor interfacial compatibility with the highly polar perovskite surface, leading to weak interfacial adhesion and substantial interfacial recombination losses.97,98 Although the introduction of polar functional groups has been explored to improve compatibility, such modifications are intrinsically limited in fullerene systems due to their rigid cage structure and synthetic constraints. This weak interfacial compatibility becomes particularly problematic in Si–perovskite tandem architectures, where additional contact layers are required. For example, delamination of tin oxide (SnO2) deposited on PC61BM by atomic layer deposition (ALD) has been observed, originating from the intrinsically weak physisorption-dominated adhesion at the PC61BM/SnO2 interface (Fig. 3b).99 Subsequent thermal processing and sputtering steps, such as indium zinc oxide (IZO) deposition, further exacerbate this issue by introducing mechanical and thermal stress, ultimately promoting stick–slip-type interfacial separation.Beyond interfacial compatibility with adjacent contact layers, fullerene-based ETLs also suffer from intrinsic material instability under prolonged thermal and illumination stress. As shown in Fig. 3c, although C60 initially forms a uniform and fully covered layer on the perovskite surface, extended thermal aging induces severe molecular aggregation, resulting in incomplete surface coverage.27 PC61BM exhibits similar behavior under continuous thermal annealing and light-soaking conditions, where pronounced aggregation and pinhole formation are observed in perovskite/PC61BM films.100,101 This morphological instability arises from the weak perovskite–fullerene interactions and high surface energy of fullerene molecules, which promote self-aggregation during thermal aging. Consequently, incomplete coverage (often <80%) and aggregated morphologies lead to non-uniform interfacial contact, discontinuous electron-transport pathways, increased series resistance (>10 Ω cm2), and enhanced trap-assisted recombination, with carrier lifetimes reduced to tens of nanoseconds or less.102–104 Importantly, repeated reports of morphological instability—stemming from limited solubility, poor film uniformity, and aggregation-induced degradation—persist despite extensive efforts to chemically modify fullerene derivatives.105 This indicates that such instability is not confined to specific molecular structures but is instead an inherent limitation of fullerene-based ETLs. As a result, intrinsic morphological instability represents a major bottleneck for achieving long-term operational stability in inverted PeSCs.
Beyond morphological degradation, fullerene-based ETLs are vulnerable from chemical and photochemical stability perspectives. Even under oxygen-free (inert) conditions, photoexcited fullerenes can undergo photodimerization through cage–cage bonding (Fig. 3d), which alters charge-transport properties and modifies the trap-state distribution within the ETL. These changes can induce early-stage performance fluctuations, such as burn-in behavior, and contribute to long-term degradation.106 Under simultaneous exposure to light and ambient oxygen, functionalized fullerenes such as PC61BM undergo photo-oxidation, forming epoxide and carbonyl defects on the fullerene cage.107–109 Notably, oxidation of less than 1% of the fullerene content has been reported to significantly reduce VOC, short-circuit current density (JSC), and FF by lowering electron mobility and introducing deep trap states, resulting in rapid efficiency loss.110 Similar photo-oxidative degradation pathways have been widely observed across structurally related fullerene derivatives.111 Taken together, the stability limitations of fullerene-based ETLs arise from the coupled effects of poor interfacial compatibility, intrinsic morphological instability, and susceptibility to both oxygen-free photoreactions and photo-oxidation under ambient conditions. These fundamental constraints underscore the need for alternative ETL design strategies that move beyond fullerene-based systems to achieve robust long-term stability in inverted PeSCs.
3. Top layer engineering for inverted PeSCs
Numerous surface defects—vacancies, interstitials, and antisites—can trap charge carriers, promote ion migration, and induce local heating in perovskites. A widely employed strategy to neutralize these ionic trap sites is Lewis acid/base passivation, in which charged molecules or ionic additives provide electrostatic compensation.112–114 In this framework, Lewis acids act as electron-pair acceptors, whereas Lewis bases donate electron density to under-coordinated species. Typical Lewis acids include cationic species and fluorinated aromatics; the strong electronegativity of fluorine withdraws electron density from the aromatic ring, generating partial positive character that can bind with halide anions and thereby mitigate halide-related interfacial defects. Lewis bases encompass N-, O-, S-, and P-donor groups (e.g., –NH2, –CN, C–O, O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S
S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and P
O, and P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), which coordinate with under-coordinated Pb2+ sites. Beyond defect passivation, the intrinsic dipole moments of these molecules—or appropriately engineered substituents—can modulate the interfacial vacuum level, enabling more favorable band alignment between the perovskite and adjacent ETLs. Consequently, functional molecules and ionic additives bearing these groups not only suppress surface defects but also contribute to energy-level alignment, protective capping-layer formation, and improved thermal dissipation. The following section highlights representative organic and inorganic modifiers that leverage these functionalities to simultaneously control defects and optimize perovskite interfaces.
O), which coordinate with under-coordinated Pb2+ sites. Beyond defect passivation, the intrinsic dipole moments of these molecules—or appropriately engineered substituents—can modulate the interfacial vacuum level, enabling more favorable band alignment between the perovskite and adjacent ETLs. Consequently, functional molecules and ionic additives bearing these groups not only suppress surface defects but also contribute to energy-level alignment, protective capping-layer formation, and improved thermal dissipation. The following section highlights representative organic and inorganic modifiers that leverage these functionalities to simultaneously control defects and optimize perovskite interfaces.
3.1. Perovskite/ETL interface engineering
In inverted devices with the structure indium tin oxide (ITO)/NiOx/SAM/FA0.9Cs0.1PbI2.9Br0.1/interlayer/PC61BM/bathocuproine (BCP)/Ag (Fig. 4a), Mo et al. addressed this by introducing a phosphonic-acid–fullerene conjugate (CPPA).118 The phosphonic acid group passivates perovskite surface defects, whereas the fullerene backbone forms a charge-transfer bridge to PC61BM. This single molecule therefore unifies defect passivation, dipole-driven energy-level tuning, and electron bridging, collectively lowering interfacial recombination and accelerating extraction. As a result, both VOC and FF increase relative to using a simple phosphonic-acid layer, and the champion PCE improves from ∼22% to ∼24–25% in FA0.9Cs0.1PbI2.9Br0.1 and Cs0.05(FA0.98MA0.02)0.95Pb(I0.98Br0.02)3 absorbers respectively. Mechanistic characterization using density functional theory (DFT) reinforces this picture: Fig. 4b shows CPPA located precisely at the perovskite/PC61BM contact to create a dedicated electron-transfer channel. Charge-density-difference (Δρ) and electron-localization-function (ELF) maps indicate multisite cooperative binding through O–Pb coordination and OH⋯I hydrogen bonding, accompanied by strengthened conjugation across the PC61BM–perovskite interface. Ultraviolet photoelectron spectroscopy (UPS) further reveals an upward shift of the Fermi level and conduction-band edge, evidencing a reduced electron-extraction barrier, which manifests electrically as decreased series resistance and increased shunt resistance (Fig. 4c). Stability improvements are equally notable, highlighting the synergistic effects of CPPA-enabled top-interface engineering.
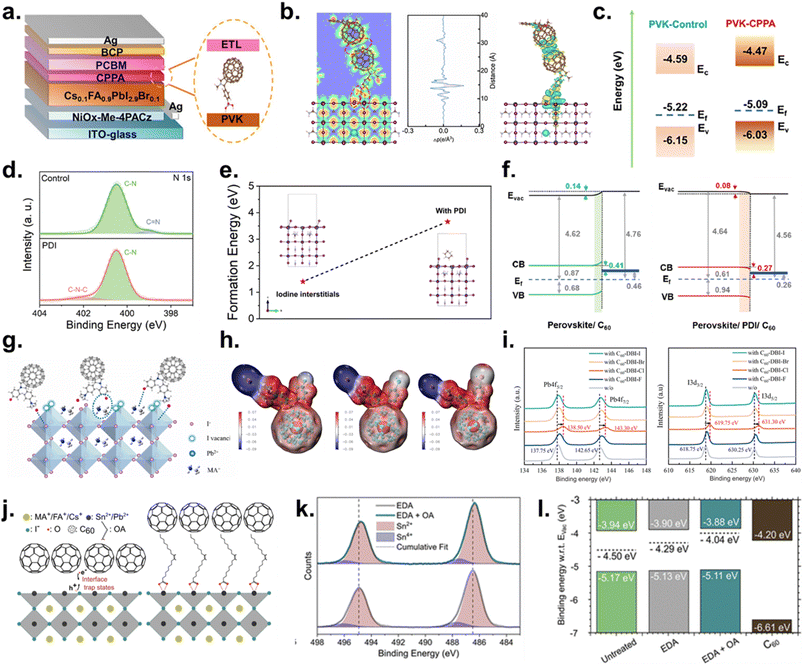 | ||
| Fig. 4 (a) Device architecture and chemical structure of CPPA. (b) DFT Δρ maps for representative adsorption models on the perovskite surface, vertically integrated Δρ profiles, and the ELF of the CPPA/PC61BM/perovskite stack. (c) Energy band diagram. Reproduced with permission from ref. 118. Copyright 2025, Wiley-VCH GmbH. (d) N 1s XPS spectra of control and piperazinium diiodide-treated perovskite films. (e) DFT formation energies of iodine-interstitial defects with and without piperazinium diiodide; the model of piperazinium diiodide adsorption on PbI2-terminated FAPbI3(100). (f) Energy-level diagrams of perovskite/C60 and perovskite/piperazinium diiodide/C60 heterointerfaces, highlighting the reduced extraction barrier with piperazinium diiodide. Reproduced with permission from ref. 22. Copyright 2024, The Royal Society of Chemistry. (g) Schematic of the C60-DBI-I/perovskite interaction. (h) ESP profiles of C60-DBI-F, -Br, and -I. (i) Pb 4f (left) and I 3d (right) XPS spectra of the perovskite films with/without C60-DBI-R. Reproduced with permission from ref. 119. Copyright 2023, Elsevier Ltd. (j) Schematic of perovskite/C60 contact with and without the OA-interlayer. (k) XPS spectra of Sn 3d core levels for perovskite films, without and with OA-post treatment. (l) Band alignment diagrams. Reproduced with permission from ref. 120. Copyright 2024, American Chemical Society. | ||
A second notable example is a dipolar organic salt, piperazinium diiodide, introduced by Zheng et al. as a multifunctional surface modifier that concurrently alleviates deep traps and unfavorable band alignment.22 Unlike conventional ammonium halides that readily induce low-dimensional phases, piperazinium diiodide predominantly remains in its molecular form on the surface, suppressing the deprotonation reaction between A-site cations and the formation of deep-level defects (iodine interstitials) on the surface. X-ray photoelectron spectroscopy (XPS) analysis in Fig. 4d indicates that the control sample exhibits a peak assigned to deprotonated FA (C–N, ∼399 eV), whereas no FA-related signal is detected for the piperazinium diiodide-treated film. Instead, a weak peak corresponding to piperazinium diiodide (C–N–C, ∼402 eV) is observed. According to DFT calculations, the formation energy of iodine interstitials increases from 1.403 eV to 3.459 eV upon piperazinium diiodide adsorption, suggesting that piperazinium diiodide inhibits the formation of surface iodine interstitial defects (Fig. 4e). Depth-profiling UPS reveals a downward band bending in the piperazinium diiodide-treated perovskite, which facilitates electron extraction while suppressing hole backflow. This behavior is attributed to the positively charged –NH2+ moieties in piperazinium diiodide, which act as electron-withdrawing groups and draw electron density from interfacial Pb and I species, thereby establishing an interfacial dipole and an enhanced built-in electric field (Fig. 4f). These interfacial modifications translate directly into record photovoltaic performance. In devices with FTO/SAM/Cs0.05(FA0.98MA0.02)0.95PbI3/interlayer/C60/BCP/Cu, a champion PCE of 26.15% (certified 25.87% and quasi-steady-state 25.52%) is achieved with VOC = 1.18 V and FF = 86.2%, while a 1.028 cm2 device delivers 24.18%, demonstrating scalability. Stability is likewise exceptional, with unencapsulated devices retaining 94.2% of initial PCE after 1000 h of maximum power point (MPP) tracking and 90.4% after 4500 h at 85 °C.
To effectively mitigate traps and hysteresis at the top interface, it is essential to target both positively charged metal defects (under-coordinated Pb2+/Sn2+) and negatively charged defects (VA− and Xi−). In inverted devices configured as FTO/NiOx/MAPbI3/interlayer/PC61BM/SnO2/Ag, Wang et al. addressed this dual requirement by designing a halogen- and quaternary-ammonium-functionalized fullerene (C60-DBI-R) that can passivate both positively and negatively charged defects due to its ionic ammonium-salt motif, providing complementary charge sites at the perovskite interface (Fig. 4g).119 DFT-derived electrostatic potential (ESP) maps show that the negative potential is concentrated on the counter halide in the ammonium-salt group, enabling strong electrostatic binding to under-coordinated metal-cation (Pb2+) defects (Fig. 4h). Meanwhile, the cationic ammonium group (–NRxH4−x+) can electrostatically stabilize negatively charged defect sites and halide-rich surface species, thereby realizing dual-channel defect passivation. This bidirectional repair of incomplete surface coordination suppresses ion migration and reduces trap density which can be identified from the XPS data in Fig. 4i. The Pb 4f and I 3d peak shifts after C60-DBI-R treatment suggest interfacial electrostatic interactions between halide anions and undercoordinated Pb2+ and between ammonium cations and I−. As a result, surface traps and interfacial charge accumulation decrease, mitigating VOC and FF losses and suppressing hysteresis. Performance trends are consistent with this mechanism. Across the halogen series, C60-DBI-F/Cl/Br/I achieves PCEs of 20.31/20.70/19.93/19.44%, respectively, while the unmodified device remains at 18.58%.
In Sn–Pb narrow-bandgap (NBG) devices, the relatively larger conduction band offset at the perovskite/C60 interface can lead to severe misalignment with the ETL. Moreover, surface Sn2+ oxidizes to Sn4+ upon exposure to air and moisture, inducing p-type doping and increasing the minority-carrier density at the perovskite/ETL interface. To address these issues, Maxwell et al. inserted a thin oleic acid (OA) interlayer between the perovskite and C60, which serves as both a buffer and a passivation layer (Fig. 4j).120 XPS shows that OA treatment slightly shifts the Sn 3d3/2 and Sn 3d5/2 peaks to lower binding energy, indicating carboxylic-acid coordination to surface Sn2+, while simultaneously reducing the Sn4+ fraction (Fig. 4k). By suppressing the formation of Sn4+ sites, the OA interlayer strengthens surface n-type character and shifts the Fermi level upward by ∼0.25 eV, thereby lowering the minority-carrier concentration at the C60 interface (Fig. 4l). As a result, OA modification of the NBG top interface enabled the fabrication of monolithic tandem solar cells (ITO/NiOx/Me-4PACz/FA0.8Cs0.2Pb(I0.6Br0.4)3/C60/ALD-SnOx/Au(1 nm)/PEDOT:PSS/Cs0.05FA0.7MA0.25Pb0.5Sn0.5I3/C60/ALD-SnOx/Ag) with a PCE of 27.3% (certified 26.4%) and a VOC of 2.21 V.
Li et al. demonstrated that molecularly tailorable titanium oxide clusters (CTOCs) serve as highly effective inorganic interlayers at the perovskite/ETL interface. CTOCs, constructed from Ti–O–Ti cage frameworks and functionalized with ligands (phenyl (CTOCPh), p-fluorophenyl (CTOCPh-F), and pentafluorophenyl (CTOCPh-5F)), provide atomic-level structural precision and establish strong quadrupole–dipole interactions with the adjacent C60 layer.27 As depicted in Fig. 5a, the CTOC-series clusters exhibit a doughnut-like structure with an inner diameter of 0.83 nm, matching well with the size of a single C60 molecule to restrain the C60 movement under heating. The negatively polarized F-substituted phenyl groups on CTOCs enable Pb–F bonds with the PbI2-rich (100) termination of the perovskite, showing strong binding between perovskite and clusters (Fig. 5b). By forming a lattice-matched cluster/perovskite interface, the interlayer alleviates the strain and lattice distortion that develop during aging. Moreover, the strong electronegativity of the fluoro group modulates the WFs which facilitates charge transfer between the perovskite and C60 (Fig. 5c). When incorporated into inverted PeSCs (ITO/CbzNaph/FA0.80MA0.14Cs0.06PbI3/CTOC derivatives/C60/BCP/Ag), CTOCPh-5F yielded a champion PCE of 25.6% (VOC = 1.18 V; FF = 84.90%), with excellent durability—retaining 98% of initial PCE after 1500 h of MPP tracking and 93% after 1000 h at 85 °C. These results highlight how inorganic molecular clusters can concurrently deliver defect passivation, quadrupole anchoring, and long-term operational stability.
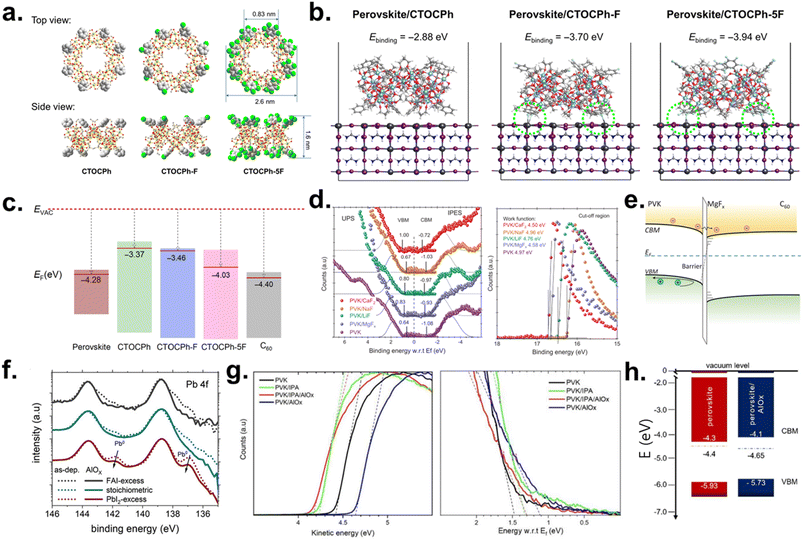 | ||
| Fig. 5 (a) Top- and side-view atomic structures of the TiO2 cluster derivatives. (b) Calculated binding energy for PbI2-rich FAPbI3 (100)/CTOCPh, CTOCPh-F and CTOCPh-5F (c) Energy-level alignment at the perovskite/cluster/C60 interface. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 27. Copyright 2024, American Association for the Advancement of Science. (d) UPS/IPES analysis of energy-level alignment with different alkali/alkaline-earth fluorides. (e) Schematic of the band diagram. Reproduced with permission from ref. 124. Copyright 2022, American Association for the Advancement of Science. (f) XPS Pb 4f spectra of perovskite films with different surface stoichiometries before and after AlOX deposition. (g) UPS spectra near the secondary electron-cutoff (left) and near the WF (right). (h) Schematic of the band diagram. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 125. Copyright 2024, Wiley-VCH GmbH. | ||
Liu et al. further showed that inserting a ∼1 nm ultrathin metal fluoride (MgFx) layer by thermal evaporation at the perovskite/C60 interface effectively prevents direct fullerene–perovskite contact, thereby reducing interfacial nonradiative recombination and lowering mid-gap defect states.124 UPS and low-energy inverse photoemission spectroscopy (LE-IPES) measurements reveal favorable downward band bending upon MgFX insertion, with the perovskite CBM shifting closer to the Fermi level and aligning more optimally with the C60 LUMO (Fig. 5d). This effect is attributed to an interfacial dipole generated by sub-stoichiometric MgFX (x ≈ 1.0 ± 0.2), which lowers the local WF and drives electrons toward C60 (Fig. 5e). The ultrathin MgFx layer still allows efficient electron tunneling, maintaining selective contact functionality. Implementing this modification in monolithic Si–perovskite tandems (Si bottom cell/2PACz/Cs0.05FA0.8MA0.15Pb(I0.755Br0.255)3/metal fluoride/C60/SnO2/IZO/Ag/MgFX) yielded a certified stabilized PCE of 29.3%, with a VOC of 1.92 V and FF of 80.70%. Damp-heat testing (85 °C, 85% RH) demonstrated >95% retention after 1000 h, underscoring the dual performance–stability advantages of inorganic fluoride layers.
Artuk et al. advanced this strategy by introducing an ALD-grown aluminum oxide (AlOX) layer as a universal inorganic interlayer for the perovskite/C60 interface.125 Unlike hygroscopic alkali fluorides (NaF, LiF, and MgFX), ALD-AlOX produces an ultrathin (∼1.4 nm), conformal crystalline layer that chemically interacts with surface Pb-halide species. XPS analysis shows that AlOX and its precursor (trimethylaluminum, TMA) react with residual PbI3− and Pb0, oxidizing Pb0 to Pb2+ and eliminating shallow donor-like states that contribute to nonradiative loss (Fig. 5f). This process reduces the n-type surface character, evidenced by a 0.2 eV upward shift in the valence band maximum (VBM) and an increased WF, producing more balanced band alignment and reduced majority-carrier offsets at the perovskite/C60 junction (Fig. 5g and h). When the dense AlOX layer was applied to monolithic Si–perovskite tandems (Si bottom cell/ITO/Me-4PACz/SiOx-NP/Cs0.05(FA0.9MA0.1)0.95Pb(I0.8Br0.2)3/ALD-AlOX/C60/SnOX/IZO/Ag/LiF), a certified efficiency of 29.9% was achieved with a VOC exceeding 1.92 V. The operational stability tests were conducted for 140 h, after which the champion AlOX device retained 94.5% of its initial efficiency, whereas the device without AlOX retained only 70%.
Taken together, the studies discussed above highlight several practical design strategies for organic and inorganic modifiers that enable high-performance and durable perovskite/ETL contacts. The most effective interlayers are multifunctional, simultaneously (i) passivating defect- and ion-mediated nonradiative recombination pathways, (ii) tuning band alignment through interfacial dipoles or band bending, and (iii) retaining—or even enhancing—efficient interfacial charge transfer rather than acting as an insulating barrier. Organic modifiers offer exceptional tunability in terms of dipole moments, binding motifs, and hydrophobicity; however, their thermal and environmental robustness can be limited. In contrast, inorganic interlayers generally provide stronger interfacial bonding, more effective suppression of ion migration, and improved tolerance to heat and moisture, often with superior process compatibility for tandem architectures and scalable deposition techniques such as ALD or thermal evaporation. From a manufacturability perspective, a critical requirement is achieving nanometer-scale interlayers with conformal, pinhole-free coverage. Such layers must preserve selective contact behavior while minimizing local recombination sites. Encouragingly, several approaches have already demonstrated both large-area scalability and long-term operational stability, suggesting that these emerging design principles can directly inform device architectures that are not only highly efficient at the laboratory scale but also viable for industrial manufacturing.
Surface defect sites, such as vacancies and under-coordinated Pb2+, readily adsorb and activate O2 and H2O molecules.17,126 The resulting hydration distorts the (PbI6)4− octahedral framework, weakens the interactions between the A-site cations and the inorganic lattice, and ultimately increases susceptibility to extrinsic species such as moisture, oxygen, and other ambient gases.127,128 LDP capping layers are most commonly formed using ammonium ligand salts—alkylammonium chains, aromatic ammonium ions, or amidinium cations paired with halides (Fig. 6a).129 These ammonium species bind to the perovskite surface either by filling A-site vacancies or through hydrogen bonding. When the ammonium cation is sufficiently bulky, exceeding the geometrical tolerance factor (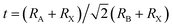 , where RA, RB, and RX are the ionic radii of A, B and X site ions, respectively) threshold of 1, it can substitute A-site cations at the surface and drive a structural reconstruction from the 3D perovskite into low-dimensional phases. These include 2D perovskites—formed by slicing the 3D lattice into layered inorganic slabs (n)—and 1D perovskites consisting of octahedral chains encapsulated by bulky organics and 0D structures composed of isolated Pb-halide octahedra with strong exciton confinement and large bandgaps.130–135
, where RA, RB, and RX are the ionic radii of A, B and X site ions, respectively) threshold of 1, it can substitute A-site cations at the surface and drive a structural reconstruction from the 3D perovskite into low-dimensional phases. These include 2D perovskites—formed by slicing the 3D lattice into layered inorganic slabs (n)—and 1D perovskites consisting of octahedral chains encapsulated by bulky organics and 0D structures composed of isolated Pb-halide octahedra with strong exciton confinement and large bandgaps.130–135
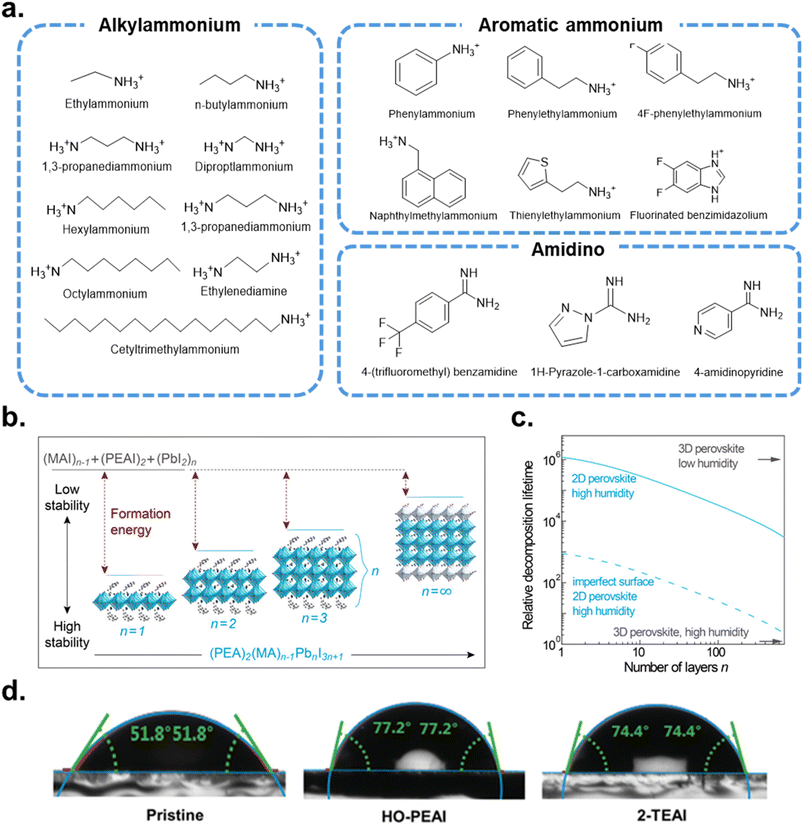 | ||
| Fig. 6 (a) Chemical structures of organic ligands to build LDP capping layers. (b) Energetics of perovskite formation and stability. The unit cell structure of (PEA)2(MA)n−1PbnI3n+1 perovskites with different n values, showing the evolution of dimensionality from 2D (n = 1) to 3D (n = ∞). (c) DFT simulation of the formation energy of perovskites with different n values in different atmospheres. Reproduced with permission from ref. 136. Copyright 2016, American Chemical Society. (d) Measurements of water contact angles after dropping water on the perovskite thin film surfaces without and with HO-PEA and 2-TEAI. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 137. Copyright 2022, Wiley-VCH GmbH. | ||
In general, LDP capping layers enhance PeSC stability through three major mechanisms: (1) higher formation energy, which thermodynamically favors crystalline retention over decomposition; (2) hydrophobic organic spacer layers, which block moisture ingress and suppress ion migration; (3) improved charge transfer and interfacial energetics, arising from reduced defect density and well-defined surface chemistry. When bulky organic cations such as phenylethyl ammonium (PEA+) intercalate between [PbI6]− layers, the phenyl rings of the organic spacers promote strong van der Waals and π–π interactions. These interactions enhance interlayer cohesion and increase the overall formation energy, resulting in a more robust and decomposition-resistant structure (Fig. 6b).136 DFT simulations (Fig. 6c) further show that lower-n 2D perovskites exhibit markedly longer decomposition lifetimes, consistent with enhanced thermodynamic stability. Additionally, the hydrophobic nature of the organic spacer serves as an effective barrier against moisture. The water contact angle of pristine perovskite films (51.8°) increased to 77.2° and 74.4° following 4-hydroxyphenethylammonium iodide (HO-PEAI) and thiopheneethylammonium iodide (2-TEAI) treatments, respectively (Fig. 6d).137 This improvement reflects reduced water affinity and enhanced humidity resistance, underscoring the protective role of LDP layers in mitigating moisture-induced degradation.
Although LDPs provide excellent moisture-resistant capping layers, their integration into inverted device structures is limited by unfavorable energy-level alignment. Due to quantum confinement, LDPs typically retain a similar valence band position while shifting the CBM upward relative to the underlying 3D perovskite (Fig. 7a),138 hindering electron extraction in inverted devices with reduced JSC and FF. Thus, significant efforts have been devoted to enabling the development of stable LDP capping layers in inverted PeSCs without compromising charge extraction.
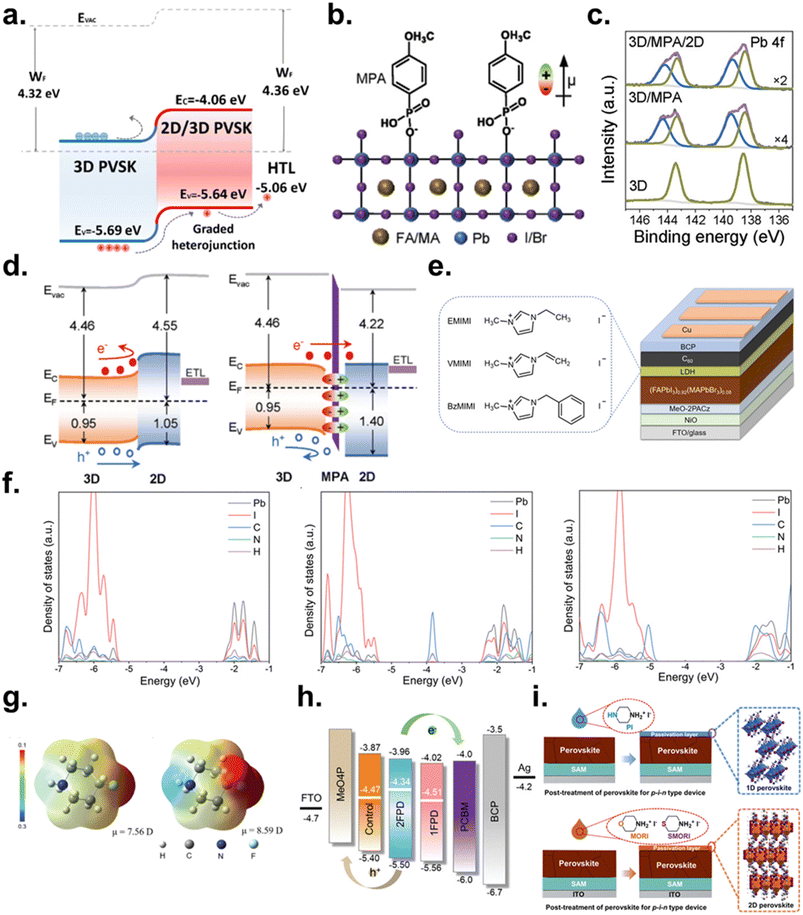 | ||
| Fig. 7 (a) Energy level diagram of a 3D/2D perovskite bilayer. Reproduced with permission from ref. 138. Copyright 2024, Springer Nature. (b) Schematic of interaction between MPA and 3D perovskite, representative of surface dipole moment. (c) XPS Pb 4f core level spectra of 3D, 3D/MPA, and 3D/MPA/2D films. (d) Energy level diagrams of 3D/2D and 3D/MPA/2D perovskite heterointerfaces. Reproduced with permission from ref. 139. Copyright 2023, Wiley-VCH GmbH. (e) Chemical structures of the imidazolium salts and architecture of the inverted PeSCs. (f) Projected density of states of the 1D perovskites (EMIMPbI3, VMIMPbI3, and BzMIMPbI3). Reproduced with permission from ref. 140. Copyright 2022, Wiley-VCH GmbH. (g) ESP maps and electric dipole moment (µ) of 1FPD and 2FPD molecules. (h) Structure and energy level alignment of the inverted PeSCs. Reproduced with permission from ref. 141. Copyright 2024, Wiley-VCH GmbH. (i) Schematic illustration of the post-treatment for the perovskite film using PI, MORI, or SMORI and a single-crystal structure based on PI and MORI. Reproduced with permission from ref. 143. Copyright 2024, American Chemical Society. | ||
To address both interfacial recombination and band offset mismatches between 3D and 2D perovskites, Zang et al. introduced a dipole-active molecular interlayer, 4-methoxyphenylphosphonic acid (MPA), between the 3D perovskite and the top 2D layer (Fig. 7b).139 This layer simultaneously passivates surface defects and tunes the interfacial energetics to facilitate efficient electron extraction. MPA coordinates strongly with under-coordinated Pb2+ through Pb–O–P interactions, effectively suppressing trap states. XPS analysis confirms higher-binding-energy Pb 4f components after MPA treatment, consistent with the formation of Pb–O–P bonds (Fig. 7c). In addition, the dipole introduced by MPA shifts the Fermi level of the 3D perovskite from 4.55 to 4.22 eV, improving its alignment with the adjacent 2D layer (Fig. 7d). The resulting 3D/MPA/2D/PC61BM stack exhibits significantly enhanced electron transport, yielding a PCE of 24.85% (ITO/MeO-2PACz/Cs0.05(MA0.10FA0.90)0.95Pb(I0.90Br0.10)3/MPA/2D perovskite/PC61BM/BCP/Ag).
Zhang et al. further developed an effective interfacial strategy by forming LDP capping layers using 1-ethyl-3-methylimidazolium iodide (EMIMI), 1-vinyl-3-methylimidazolium iodide (VMIMI), and benzylimidazolium iodide (BzMIM) (Fig. 7e).140 The resulting crystals exhibit 1D structures composed of face-sharing polymeric [PbI3]− chains surrounded by organic cations. DFT calculations (Fig. 7f) reveal that in BzMIMPbI3, the valence band originates from the aromatic cation while the conduction band derives from hybridized orbitals of the inorganic chains. This intrinsic spatial separation suppresses electron–hole recombination and extends carrier lifetimes. Moreover, the ultrathin nature of the 1D layer allows tunneling-assisted electron extraction into C60. Devices incorporating BzMIMPbI3 achieved 24.09% PCE, a high VOC of 1.204 V, and >84% FF, while also demonstrating exceptional thermal (92% retention after 1400 h at 85 °C) and operational stability (95% retention after 1008 h under MPP tracking).
Li et al. introduced fluoroalkylamine-based surface molecules (1FPD and 2FPD) (Fig. 7g) to improve charge transport in 2D/3D bilayer PeSCs.141 While 1FPD offered modest passivation, 2FPD induced a well-oriented ferroelectric 2D layer with strong out-of-plane dipoles due to fluorination, leading to a favorable WF shift (4.47 eV → 4.34 eV) and improved band alignment (Fig. 7h). Zhao et al. used 5-(2-hydroxyethyl)-3,4-dimethylthiazolium iodide (HDI) to form a 1D capping layer that converts surface impurities (PbI2 and δ-FAPbI3) into a uniform 1D HDPbI3 phase.142 This induces an n-type-like surface, optimizes band alignment, and enhances electron extraction, enabling a PCE of 25.30% with an FF of 84.21%. Wang et al. replaced the piperazinium iodide (PI) capping layer with morpholine hydriodide (MORI) and thiomorpholine hydriodide (SMORI) to construct a 2D/3D heterostructured capping layer (Fig. 7i).143 SMORI, in particular, provides stronger defect passivation and forms a more favorable n-N homotype (i.e., an n-type/n-type junction with different doping levels) 2D/3D junction. This shifts the surface further n-type, aligning more effectively with the C60 ETL and reducing interfacial recombination. Achieving their seamless integration without efficiency losses would enable the development of devices that combine high performance with exceptional stability.
In summary, LDP capping layers demonstrate that stability and interfacial quality can be co-optimized when surface reconstruction, defect passivation, and moisture resistance are engineered as an integrated interfacial system. The primary trade-off is that quantum confinement in LDPs often raises the CBM relative to that of the 3D absorber, which can impede electron extraction in inverted devices, thereby reducing the JSC and FF. This interfacial trade-off can be deliberately re-tuned using dipole-active interlayers, ultrathin 1D phases that permit tunneling, or junction designs that render the surface more n-type. However, even with these strategies, several practical challenges remain. Nonuniform surface conversion may leave portions of the reactive 3D phase partially exposed, undermining the intended protective function. In addition, excessively thick or poorly oriented capping layers can act as resistive barriers that hinder charge extraction. Furthermore, under prolonged heat and illumination, the interface may evolve over time. Dipole relaxation and ionic redistribution can gradually reintroduce band-offset mismatches and nonradiative recombination pathways, even when the as-fabricated device initially exhibits excellent performance.
A widely adopted approach is integrating high thermal conductivity materials directly into the perovskite matrix. Two-dimensional materials have received particular attention due to their exceptional thermal conductivity and chemical stability. Liu et al. incorporated molecularly functionalized boron nitride (BN-5) nanosheets into the perovskite as local heat-dissipation pathways (Fig. 8a).144 BN exhibits an in-plane thermal conductivity of ∼550 W m−1 K−1, and its incorporation increased the thermal conductivity of the perovskite by 30.7%. Consistent with this, real-time IR thermography confirmed enhanced heat dissipation: BN-5 films cooled more rapidly from 85 °C to ambient (20 °C) than pristine films (Fig. 8b), and under 30 min of 100 mA cm−2 illumination, the temperature rise (ΔT) was significantly lower (21.7 °C vs. 34.6 °C) (Fig. 8c). Beyond thermal benefits, BN-5 interacts with under-coordinated Pb2+ at grain boundaries, enabling a high PCE of 25.36% (ITO/2PACz/Cs0.05FA0.81MA0.14PbI2.55Br0.45/PC61BM/BCP/Cu). Device stability also improved markedly: under 1-sun illumination at 85 °C, the BN-5 device retained over 95% of its initial PCE after 2640 h, whereas the pristine device dropped to 63% within 587 h.
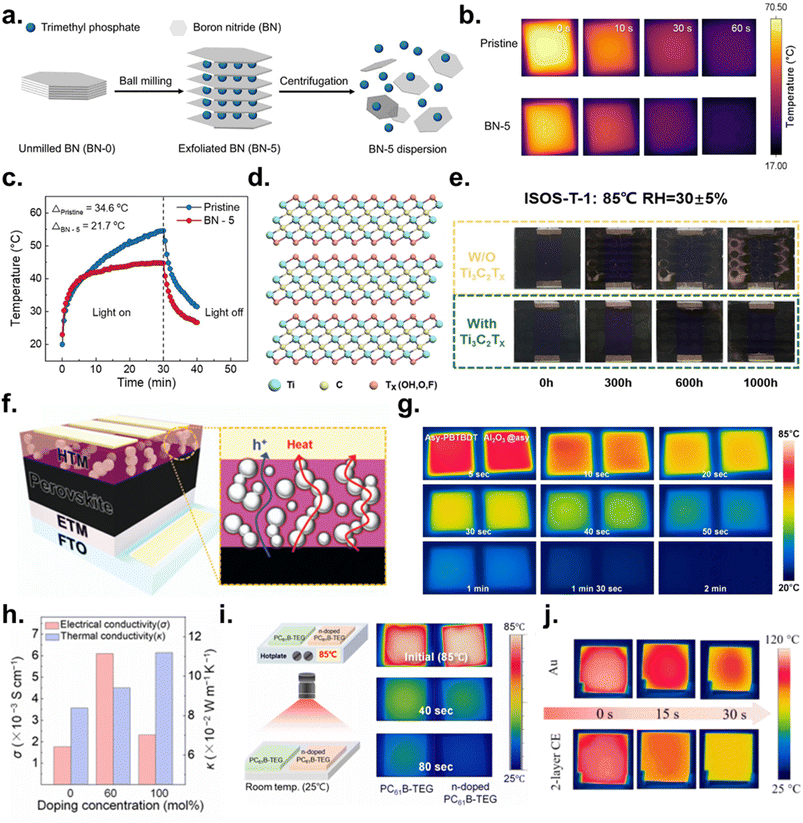 | ||
| Fig. 8 (a) Schematic illustration of the preparation process for BN-5. (b) Comparison of the cooling process between the pristine perovskite film and BN-5 based perovskite films, illustrated through infrared (IR) thermal images. (c) Temperature profile of PeSCs under solar irradiation and after the light is turned off. Reproduced with permission from ref. 144. Copyright 2025, Wiley-VCH GmbH. (d) Schematic diagram of the multilayer Ti3C2TX. (e) Image of PeSC aging evolution under ISOS-T-1 conditions. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 145. Copyright 2025, Springer Nature. (f) Schematic illustration of the PeSC using Al2O3 NPs as thermally conductive materials. (g) IR thermal images of a polymeric HTM (Asy-PBTBDT) and Al2O3@asy-PBTBDT under a plate-cooling test. All samples were prepared on the perovskite layer. Reproduced with permission from ref. 146. Copyright 2020, Royal Society of Chemistry. (h) Electrical and thermal conductivity of PC61B-TEG by varying the N-DMBI doping concentration. (i) IR camera image of PC61B-TEG and n-doped PC61B-TEG. Reproduced with permission from ref. 147. Copyright 2024, Royal Society of Chemistry. (j) IR camera-measured temperature of glass substrates coated with various electrode films. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 148. Copyright 2024, Springer Nature. | ||
Wan et al. applied a similar strategy by introducing Ti3C2TX MXene nanosheets (Fig. 8d).145 With a thermal conductivity of up to 55.8 W m−1 K−1, MXene increased the thermal conductivity of the perovskite to 0.413 W m−1 K−1. Under ISOS-T-1 conditions (85 °C, 30 ± 5% RH), MXene-modified devices showed no degradation after 1000 h, while control devices degraded severely (Fig. 8e). Even under continuous aging, MXene devices preserved 80% of their PCE after 500 h, whereas control devices retained only 58% after 200 h.
A complementary strategy intensifies heat extraction at the perovskite surface. By increasing the thermal conductivity at the top interface, a steeper vertical temperature gradient is created, thereby accelerating heat flux according to Fourier's law (Q = −κ∇T, where Q represents heat flux, κ represents thermal conductivity, and ∇T represents the temperature gradient). Choi et al. implemented this concept using a hybrid interfacial scaffold composed of Al2O3 nanoparticles (NPs) embedded in the polymeric hole-transporting material (HTM) asy-PBTBDT (Al2O3@asy-PBTBDT) (Fig. 8f).146 Due to their high thermal conductivity (20–30 W m−1 K−1), Al2O3 NPs formed efficient heat-dissipation channels. Time-domain thermoreflectance (TDTR) measurements revealed that even 0.1 wt% Al2O3 increased the through-plane thermal conductivity of the HTM from 0.123 to 0.206 W m−1 K−1. Cooling tests confirmed significantly faster temperature decay for Al2O3-containing HTM layers (Fig. 8g). Under harsh conditions (85 °C and 85% RH, no encapsulation), devices with Al2O3@asy-PBTBDT retained 91% of their initial PCE after 31 days—substantially outperforming pristine HTM devices and even encapsulated Spiro-OMeTAD controls. Although the added Al2O3 slightly reduced hole mobility and JSC, devices still achieved high VOC (1.16 V) and 19.9% PCE (FTO/SnO2/(FA0.833MA0.167)Pb(I2.5Br0.5)/Al2O3@asy-PBTBDT/Au). Notably, the strategy was broadly compatible with various HTMs.
A third heat-management mechanism was demonstrated by Park et al., who introduced an n-doped fullerene interlayer (PC61B-TEG) between the perovskite and C60 ETL.147 Doping with 4-(1,3-dimethyl-2,3-dihydro-1H-benzimidazol-2-yl)-N,N-dimethylaniline (N-DMBI) increased free-carrier density, enhancing both electrical and thermal conductivity through carrier-assisted heat transport—analogous to metallic systems. TDTR measurements showed a 25% increase in thermal conductivity upon doping (Fig. 8h). Cooling analysis confirmed faster heat dissipation: the doped film cooled to 30 °C in 80 s, compared to ∼120 s for the undoped film (Fig. 8i). In addition, n-doping shifted the Fermi level upward, improving alignment between PC61B-TEG and the perovskite CBM, leading to more efficient electron extraction, reduced interfacial recombination, and a larger QFLS. Devices achieved 24.42% PCE (ITO/MeO-4PACz/FA0.85Cs0.15PbI3/PC61B-TEG/C60/BCP/Ag) and retained 90% of their initial efficiency over 2400 h at 85 °C. This approach highlights the dual role of conductive interlayers in enhancing both thermal transport and interfacial energetics.
Unlike heat transfer by conduction through the device stack, a significant fraction of heat is dissipated from the device surface into the air via convection and thermal radiation. Yu et al. designed a triple-layer full-carbon electrode (F-CE) in which the top dense carbon layer is intentionally used as a high emissivity radiative-cooling layer.148 The triple-layer F-CE exhibited near black-body-like emission, as quantified using its emissivity (ε ≈ 0.99), exceeding that of Au (0.54) and ITO (0.62). As a result, the F-CE can function as an efficient thermal radiator while retaining electrode functionality. The F-CE reduced the device surface temperature by ∼10 °C compared with Au, indicating more efficient heat dissipation (Fig. 8j).
Recent research trends are increasingly focused on combining multiple thermal-management strategies, including enhanced heat transport within the bulk and across interfaces together with radiative cooling at the device surface. Such integrated approaches can lower the peak operating temperature while simultaneously controlling temperature gradients to facilitate efficient heat dissipation. Under real outdoor operation, however, transient irradiance, wind-dependent convective cooling, partial shading or soiling, and day–night thermal cycling can induce hot-spot formation, localized current crowding, and steep temperature gradients. These effects collectively accelerate ion migration, phase segregation, and interfacial reactions. Therefore, we emphasize that future outdoor-relevant designs should integrate enhanced heat conduction in the bulk and upper interfaces with high-emissivity top-surface concepts, thereby linking thermal management directly to improved long-term operational stability.
3.2. C60-alternative ETLs
This section organizes organic ETL strategies that replace or complement C60, focusing on the physicochemical mechanisms operating at the top perovskite/ETL interface. These mechanisms can be broadly grouped into four functional axes. First, molecular dipoles modulate the electron-extraction barrier by shifting the vacuum level and inducing favorable band bending, while simultaneously blocking holes. Second, defect-control functionalities passivate Pb/Sn, halide, and A-site defects through Lewis acid–base interactions, hydrogen bonding, or ionic coordination, thereby reducing nonradiative recombination and mitigating hysteresis. Third, ion-management mechanisms limit halide and A-site ion migration and prevent interfacial charge accumulation through fixed charges, electrostatic interactions, or ion-blocking barriers. Fourth, morphology and processing control ensures complete coverage, stable packing, and thermal/photostability—achieved through aggregation suppression, surface-energy tuning, and steric or hydrophobic design—which expands the thickness tolerance and improves large-area process reproducibility. Based on these mechanistic axes, we categorize organic ETLs into (i) fullerene derivatives and (ii) non-fullerene (NF) derivatives.Electron-accepting fragments (e.g., PDI, NDI, and NFA derivatives) are covalently coupled or grafted onto the fullerene cage to unify three functions into one molecule: (1) the C60 core provides a continuous electron-transport pathway, (2) imide/NFA functionalities passivate interfacial defects, and (3) the auxiliary fragments tune morphology through π–π interactions and surface-energy control. Carbonyl, nitrile, and halogen groups selectively coordinate with Pb/Sn-centered defects, lowering surface trap density and suppressing interfacial charge buildup. Simultaneously, the auxiliary π-framework promotes face-on orientation and smooth coverage, straightening the electron-extraction pathway and enabling excellent thickness tolerance.
Sun et al. demonstrated this strategy using an NFA fragment grafted onto C60 (Fig. 9a).149 Molecular design favored C60–NFA and NFA–NFA interactions, yielding low surface roughness and pinhole suppression under solution processing (Fig. 9b). Electron-withdrawing terminal groups on the NFA selectively interacted with Pb/I defects, reducing trap density, while the C60 cage ensured efficient transport. As a result, interfacial charge accumulation and ion migration were mitigated, reducing VOC and FF losses and drastically softening the thickness roll-off across several hundred nanometers—significantly improving compatibility with slot-die and blade coating. Using the device stack ITO/SAM/perovskite (Cs0.05(FA0.98MA0.02)0.95Pb(I0.98Br0.02)3)/ETL/BCP/Ag, high efficiency was retained even at large perovskite thicknesses, demonstrating remarkable process tolerance (Fig. 9c). Consistent with this mechanism, C60–NFA hybrids such as BTPC60 retained ∼90% of their initial efficiency after 1500 h under ISOS-D-2 (85 °C, N2), representing a major thermal-stability improvement over pristine C60.
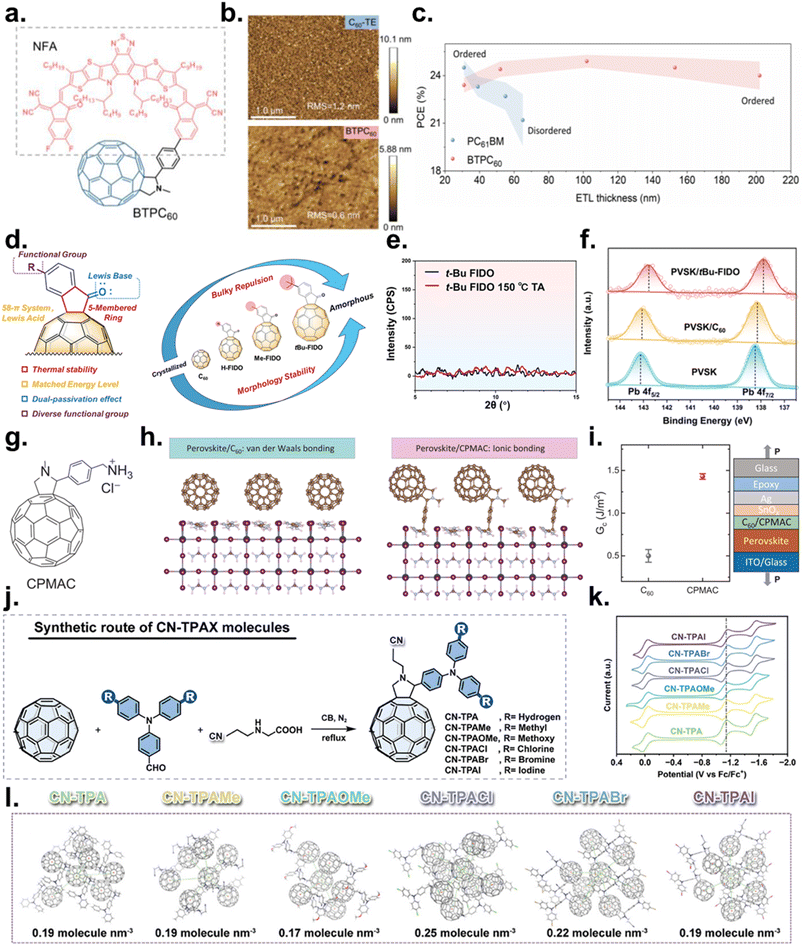 | ||
Fig. 9 (a) Chemical structure of BTPC60. (b) AFM height images of C60-TE and BTPC60 films. (c) PCE versus ETL thickness for PC61BM and BTPC60. Reproduced with permission from ref. 149. Copyright 2024, Wiley-VCH GmbH. (d) Schematic of evaporable fullerene derivatives. (e) GIXRD of thermally deposited films before/after 150 °C annealing: C60, H-FIDO, and tBu-FIDO. (f) XPS spectra of the Pb 4f region. Reproduced with permission from ref. 150. Copyright 2023, American Chemical Society. (g) Structure of CPMAC. (h) Perovskite/C60 (van der Waals) vs. perovskite/CPMAC (ionic) interactions. (i) Comparison of GC of devices to quantify the ETL for C60 or CPMAC. Reproduced with permission from ref. 151. Copyright 2025, American Association for the Advancement of Science. (j) Synthetic route to CN-TPAX (X = H, Me, OMe, Cl, Br, I). (k) CV curves in o-DCB/ACN (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v, 0.1 M Bu4NClO4, 100 mV s−1; Fc/Fc+ reference); Ered1 used to estimate LUMO levels. (l) Representative packing snapshots highlighting cage-to-cage π–π interactions of the six FDs. Reproduced with permission from ref. 152. Copyright 2024, Wiley-VCH GmbH. 1 v/v, 0.1 M Bu4NClO4, 100 mV s−1; Fc/Fc+ reference); Ered1 used to estimate LUMO levels. (l) Representative packing snapshots highlighting cage-to-cage π–π interactions of the six FDs. Reproduced with permission from ref. 152. Copyright 2024, Wiley-VCH GmbH. | ||
Fullerene derivatives can also be engineered to retain smooth amorphous morphology after vacuum deposition, ensuring stable band bending and contact resistance during metal electrode evaporation. Thermally robust substituents preserve film uniformity, while polar groups enable complementary passivation of halide and A-site defects. This enhances compatibility with tandem top cells and encapsulation and reduces variability in large-area vacuum fabrication. Shui et al. developed a thermally evaporable fullerene derivative to address process variability and interfacial instability in tandem stacks (Fig. 9d).150 Indanone substitution and bulky tert-butyl side chains were designed to preserve an amorphous, low-roughness morphology after evaporation. Consistently, grazing incidence X-ray diffraction (GIXRD) shows that t-Bu-FIDO films remain essentially amorphous before and after annealing at 150 °C, indicating suppressed thermally induced crystallization/aggregation (Fig. 9e). In addition, Pb 4f XPS spectra of perovskite/t-Bu-FIDO exhibit a clear shift toward lower binding energy, consistent with carbonyl-Pb2+ coordination and surface defect passivation, which correlates with reduced nonradiative recombination and mitigated hysteresis (Fig. 9f). Using the configuration ITO/NiOx/SAM/perovskite (Cs0.04(FA0.95MA0.05)0.96Pb(I0.95Br0.05)3)/ETL/BCP/Ag, concurrent improvements in VOC and FF translated directly into higher PCE. Compared with solution-processed ETLs, this approach eliminated leakage pathways and thickness nonuniformity, improving line-to-line reproducibility during vacuum processing and offering high compatibility with encapsulation and tandem integration.
In the ionic fullerene strategy, fullerene cores are functionalized with fixed-charge headgroups (e.g., quaternary ammonium and imidazolium) and counter-anions to introduce electrostatic binding and interfacial electron-shuttling. The fixed charges directly neutralize halide vacancy and A-site-derived defects, suppress ion migration, and thereby reduce nonradiative recombination and hysteresis. At the same time, ionic interactions enhance molecular packing and interfacial adhesion, improving fracture toughness and sustaining module-scale reliability under prolonged MPPT operation and combined thermal/illumination stress.
You et al. implemented this strategy by attaching cationic headgroups to C60 to form ionic fullerene salts (Fig. 9g).151 The positively charged termini (e.g., –NH3+) formed strong ionic bonds with surface I− species, selectively neutralizing defect populations and suppressing charge accumulation and ion migration (Fig. 9h). Strengthened intermolecular and interfacial adhesion increased fracture toughness, enabling stable electron extraction with minimal degradation during extended MPPT and thermo-photonic stress. In particular, double-cantilever beam testing showed that the interfacial fracture energy (GC) at the ETL interface increased from 0.50 ± 0.07 J m−2 for C60 to 1.43 ± 0.03 J m−2 for CPMAC, quantitatively confirming the enhanced interfacial mechanical stability (Fig. 9i). This electro-chemo-mechanical synergy also mitigated the instability of thin ETLs, stabilized top-electrode deposition, and reduced performance dispersion in large-area blade-coated mini-modules. Using the architecture FTO/SAM/Cs0.04MA0.14FA0.82Pb(I0.987Br0.013)3/ETL/ALD-SnOX/Ag, replacing C60 with an ionic fullerene yielded PCE ≈ 26%, illustrating that improved contact and energy alignment directly translate into higher efficiency. Stability was likewise enhanced: encapsulated p–i–n mini-modules operated for 2200 h at ∼55 °C under 1-sun MPP retained 91.5% of the initial PCE, outperforming C60-based counterparts that exhibited far greater degradation under identical conditions.
In the case of terminal-group engineering, using a common fullerene core (e.g., methanofullerene and fulleropyrrolidine), terminal substituents are systematically varied to co-optimize LUMO alignment, molecular packing, and electron mobility. Combinations of electron-donating/withdrawing groups, Lewis-basic motifs (pyridyl, carboxylate, and phosphonate), halogen/ionic substitutions, and steric/hydrophobic elements determine dipole moment, packing density, surface energy, and defect passivation capability. Dipole-rich or sterically tailored terminals promote dense packing and uniform film coverage, suppressing pinholes and shunt pathways, while precise energy-level tuning minimizes VOC loss by aligning the LUMO with the perovskite CBM. Modifying solubility and drying kinetics further improves coating reproducibility and thickness tolerance, enabling compatibility with large-area printing and with low-bandgap absorbers such as Sn–Pb alloys.
Liu et al. formalized this “terminal effect” by screening multiple terminal groups on an identical fulleropyrrolidine scaffold (Fig. 9j).152 Donor–acceptor strength governed LUMO alignment with the perovskite CBM, reducing VOC deficit, whereas Lewis-basic termini selectively coordinated with Pb2+ and I− defects, suppressing nonradiative recombination. Cyclic voltammetry (CV) confirmed reversible redox behavior for all six fullerene derivatives (Fig. 9k), while shifts in the first reduction potential correlated with terminal-group electron-donating strength and steric profile, yielding LUMO levels well aligned to the perovskite CBM. Bulky and hydrophobic terminal groups increase the stacking density by balancing steric hindrance and hydrophobic aggregation, which helps sustain a continuous electron-transport pathway and enhances electron mobility (Fig. 9l). Using the device structure ITO/NiOx/perovskite (Cs0.05(FA0.95MA0.05)0.95Pb(I0.95Br0.05)3)/ETL/BCP/Ag, the optimized CN-TPACl terminal design achieved ≈25% steady-state PCE (certified 25.18%). Durability was similarly enhanced: CN-TPACl-based ETLs retained ≈90% of their initial PCE after 1000 h of unencapsulated operation under white LED light in N2, ≈89% after 200 h at 85 °C (N2), and ≈86–88% after 10 cycles of encapsulated light/dark cycling in air, confirming robust long-term operation. These results enable a design guideline for matching terminal groups to specific perovskite compositions and processing routes.
In contrast, NF-ETLs integrate multiple interfacial and transport advantages within a single molecular framework. Their π-accepting backbones and induced dipoles enable precise control over vacuum-level shifting and band bending, thereby lowering the electron-extraction barrier. Heteroatoms, terminal groups, and supramolecular binding motifs further provide multi-site defect passivation and enforce directional molecular orientation, collectively suppressing nonradiative recombination and hysteresis. In parallel, side-chain design and polymerization strategies raise the glass-transition temperature (Tg), stabilize the film morphology, and establish continuous electron-transport pathways, which together improve thickness tolerance and enhance reproducibility in slot-die and printing processes.
Through these combined features, NF-ETLs effectively overcome the structural and interfacial limitations inherent to fullerene ETLs, suppressing recombination and ion migration at the top interface and expanding the process window for large-area and tandem device fabrication. The following section further organizes these advantages into three synergistic design pillars for optimizing NF-ETLs: (1) interfacial dipole and supramolecular engineering, which modulate vacuum-level shifts, band bending, and extraction kinetics; (2) polymerization-driven morphological and transport stabilization, which enhance Tg, suppress aggregation and form continuous electron pathways; and (3) end-group engineering, which simultaneously governs defect passivation, interfacial energetics, and surface hydrophobicity.
Incorporating dipole moments and selective binding motifs into NF-ETLs provides deterministic control over vacuum-level shifts and interfacial band bending. Huang et al. exemplified this strategy by reengineering Y6 into phenanthroline-modified (Y-Phen) and crown-ether-modified (Y-CE) derivatives (Fig. 10a).155 Phenanthroline–Pb2+ and crown-ether-formamidinium iodide (FAI) interactions promoted upright orientation, which can be attributed to enhanced dipole moments (Y6 ≈ 1.07D < Y-Phen ≈ 2.30D < Y-CE ≈ 7.12D), and strengthened π–π stacking. Stepwise WF shifts were observed (Y6 ≈ −0.05/Y-Phen ≈ −0.12/Y-CE ≈ −0.20 eV), indicating progressively deepened band bending and reduced electron-extraction barriers (Fig. 10b).
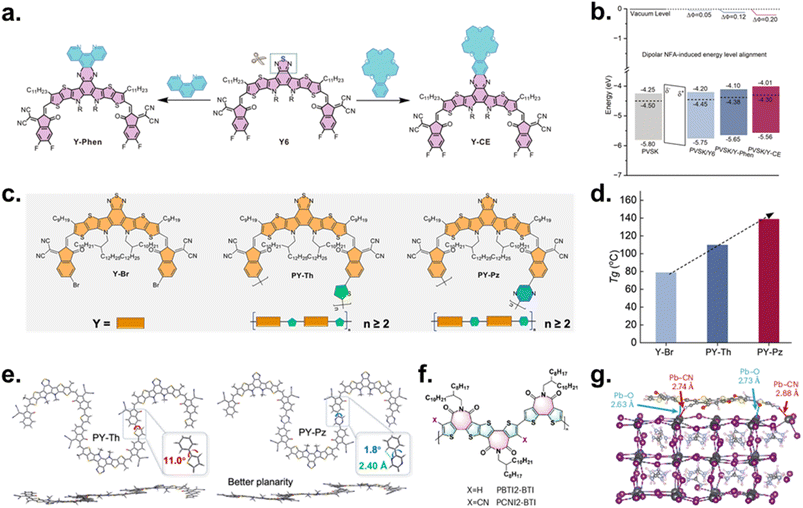 | ||
| Fig. 10 (a) Molecular structures of Y6 derivatives (Y-Phen and Y-CE). (b) Energy-level/band alignment for NFA-coated perovskite films. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 155. Copyright 2025, Springer Nature. (c) Chemical structures of Y-NFA and PY-NFA. (d) Tg of Y–Br, PY–Th, and PY–Pz. (e) DFT-optimized trimer (PY–Th and PY–Pz) structures highlighting planarity. Reproduced with permission from ref. 97. Copyright 2025, Elsevier Ltd. (f) Chemical structures of PBTI2-BTI and PCNI2-BTI. (g) Simulation of coordination effect between PCNI2-BTI and Pb2+ ions. Reproduced with permission from ref. 33. Copyright 2025, Springer Nature. | ||
Polymerizing Y-series NFAs (yielding PY-NFAs) elevates Tg, immobilizes the molecular packing, prevents solution-phase pre-aggregation, and establishes continuous transport pathways with reinforced π–π ordering. Liu et al. designed robust ETL by polymerizing a Y–Br monomer with an electron-rich π-bridge (thiophene, Th) and electron-deficient π-bridge (pyrazine, Pz), yielding two polymers, PY–Th and PY–Pz (Fig. 10c).97 Polymerization increased Tg in the order of Y–Br, PY–Th, and PY–Pz, with Y–Br showing the lowest and PY–Pz the highest Tg (Fig. 10d). Molecular dynamics simulations revealed that both PY–Th and PY–Pz adopt a similar stacking mode, which is favorable for intermolecular electron transport. However, PY–Pz exhibits much higher planarity than PY–Th (dihedral angle 1.8° vs. 11.0°) due to an intramolecular N⋯H non-covalent interaction between Pz and electron-accepting units, suggesting an enhanced electron mobility (Fig. 10e). In devices (ITO/SAM/FA0.95Cs0.05PbI3/ETL/BCP/Ag), PY–Pz delivered FF > 85% and PCE ≈ 25.8% (EQE-integrated ≈ 24.9%). These results show that polymerization enhances both morphological stability and charge transport, mitigating thickness roll-off and enabling scalable fabrication.
Furthermore, introducing Lewis- and hydrogen-bond-capable terminal groups such as –CN and carbonyl provides a unified strategy to achieve multi-site defect passivation, fine-tuned energetic alignment, and hydrophobic top-contact formation. Feng et al. developed an n-type, BTI-based polymer (PCNI2-BTI, −3.95 eV) whose LUMO level closely matches that of the perovskite (−3.91 eV), thereby minimizing the electron-extraction barrier (Fig. 10f).33 A distinctive Pb–O interaction between under-coordinated Pb2+ ions and carbonyl groups is found at the interface between the perovskite and PBTI2-BTI, with a bond distance of 2.63 Å (Fig. 10g). Additionally, Pb–N interaction between the cyano group and Pb2+ is also found, indicating a strong interaction for PCNI2-BTI enabled by the cyano functionality. The devices with PCNI2-BTI (ITO/SAM/Cs0.05FA0.9MA0.05PbI3/ETL/BCP/Ag) delivered a superior performance of VOC = 1.179 V, FF = 86.4%, and PCE = 26.0% (certified 25.4%), with excellent batch-to-batch reproducibility (25.6 ± 0.2%, n = 80) in large-area fabrication. Stability tests under ISOS-L-3 (encapsulated, white LED, ≈65 °C) show significantly slower degradation compared with the devices containing PC61BM and PBTI2-BTI, confirming the superior operational durability of CN/carbonyl-terminated systems.
However, from a commercialization standpoint, replacing C60 requires more than achieving high PCE and stability; the alternative must also simultaneously satisfy (i) cost competitiveness, particularly in light of multi-step synthesis and purification; (ii) kilogram-scale manufacturability with robust batch-to-batch consistency; (iii) compatibility with large-area coating processes (e.g., slot-die or blade coating), including sufficient thickness tolerance and a broad processing window; and (iv) seamless integration with electrode deposition and encapsulation processes. Although NF and polymerized ETLs provide clear advantages in interfacial functionality—such as improved defect passivation, ion migration control, and energy-level alignment—and enhanced morphological stability, persistent challenges related to synthetic complexity, purification, reproducibility, and overall cost may constrain their ability to replace C60 on a one-to-one basis in the near term. Accordingly, in the short term, hybrid ETL strategies that reduce C60 consumption (e.g., ultrathin C60 layers combined with low-cost modifiers or interlayers) or employ more process-tolerant derivatives may represent more pragmatic pathways. Over the mid-to-long term, material families featuring simplified synthetic routes and validated scalability in large-area coating processes are likely to emerge as the most viable candidates.
3.3. ETL/cathode interlayer
In inverted PeSCs, the selection of the cathode metal critically influences electron extraction, interfacial stability, and overall device engineering flexibility. The most commonly used cathode metals are Ag, Cu and Al. Among these, Ag is the predominant choice in high-performance inverted PeSCs because its WF (typically −4.2 to −4.6 eV) aligns well with the LUMO levels of fullerene-based ETLs (−4.0 eV for C60 and –3.8 eV for PC61BM), ensuring a small energy offset and efficient electron collection.157,158 Ag also offers high electrical conductivity, excellent film-forming characteristics, moderate cost relative to Au, and comparatively inert chemical behavior. Cu, with a nominal WF of −4.4 to −4.7 eV, presents a cost-effective alternative and provides strong conductivity and good processability. However, its higher chemical reactivity and susceptibility to oxidation under ambient or thermal stress conditions limit its stability. Al, which has a slightly lower WF (−4.1 to −4.3 eV), can also provide favorable energy-level alignment with conventional ETLs, though its chemical robustness is lower than that of Ag. Although these metals are chosen based on their balance of energy-level alignment, chemical stability, cost, and compatibility with ETLs, all of them—particularly Ag and Cu—are vulnerable to halide-induced corrosion and interfacial degradation in perovskite environments.157 Moreover, the tendency of metal ions to migrate across the interface can distort the local energy landscape and accelerate performance losses. Consequently, advanced ETL/cathode interlayers (CILs) have become essential for tuning effective WF, blocking ion diffusion from both the metal and the perovskite, and stabilizing the cathode contact. Such interlayers are now indispensable for achieving durable, high-efficiency inverted PeSCs.Introducing a dipolar interlayer alleviates these issues by establishing interfacial dipoles that reconfigure the vacuum-level alignment and suppress charge accumulation. Semiconducting interlayers with large intrinsic dipole moments—including ionic molecules, zwitterions, and functional groups capable of coordinating with the metal surface—can orient at the interface to form well-defined electrostatic dipole layers (Fig. 11). These layers shift the local vacuum level and thereby redefine the apparent WF of the electrode. According to the classical dipole-layer model, the magnitude of this shift (Δ) scales with the dipole density (n), dipole moment (p), and dielectric properties of the dipolar medium:160
| Δ = nep/(kk0) |
 | ||
| Fig. 12 The potential distribution of an electric dipole layer by introduction of a CIL. (a) The interfacial dipole-induced Evac shift Δ reduces the apparent WF of the electrode Φ′ = Φ − Δ. Reproduced with permission from ref. 161. Copyright 2020, American Chemical Society. (b) Schematic illustration of cathode materials with a decreased WF and energy level alignment with the ETL. | ||
The use of LiF and BCP has long represented the benchmark for CILs in inverted PeSCs. LiF relies on the strong Li+–F− bond dipole, whose negative pole orients toward the metal surface, thereby downshifting the vacuum level and lowering the apparent WF of the cathode.162,163 Because LiF is insulating, its thickness must remain in the tunneling regime (≤1–2 nm) to prevent electron-blocking. BCP functions through a different mechanism: its phenanthroline nitrogen atoms coordinate with Ag, forming an interfacial dipole with negative charge density near the metal and positive density toward the organic layer.164 Although this interaction reduces the effective WF, the high LUMO energy of BCP also necessitates ultrathin films to ensure efficient electron tunneling. Motivated by the limitations of LiF and BCP, a wide range of innovative CIL materials have been developed, exploiting distinct mechanisms for WF modulation and interface stabilization. Ciro et al. introduced rhodamine 101, an ionic inner-salt dye with a large intrinsic dipole moment.165 When inserted at the PC61BM/Ag interface, rhodamine 101 aligns its dipoles to reduce the Ag WF from −4.5 to −4.3 eV, improving energy-level matching and suppressing recombination. Devices (ITO/NiOx/MAPbI3/PC71BM/rhodamine 101/Ag) attained efficiencies of 16.6%, while the interlayer simultaneously served as a moisture and Ag–halide corrosion barrier.
Organic semiconductors have also been widely explored as dipolar CILs. Lee et al. demonstrated PFN-Br, a conjugated polyelectrolyte with quaternary ammonium–bromide side chains, as an effective dipole-forming interlayer.166 The polymer backbone provides electronic conductivity while the ionic substituents introduce a strong dipole that orients at the cathode interface. PFN-Br reduced the Ag WF from −4.7 to −4.2 eV, enabling better alignment with PC61BM and suppressing interfacial recombination. Wang et al. reported a PDI-based ionene polymer (PNPDI) functionalized with quaternary ammonium–bromide groups, designed to suppress π–π aggregation while embedding strong internal dipoles. Br− counterions generated self-doping, enhancing conductivity and allowing electron-permissive transport (Fig. 13a).167 When blended with Bphen (mixed CIL), the combined dipole strength and ordered PNPDI morphology produced a pronounced reduction in the WF (Fig. 13b). UPS revealed that the WF of Ag reduced from −4.22 eV to −3.60 eV, correlating with increases in VOC, JSC, and FF. Devices (ITO/PTAA/Cs0.05(FAPbI3)0.83(MAPbBr3)0.17/PC61BM/PNPDI + Bphen/Ag) achieved 21.46% PCE and retained 80% of their initial efficiency after 500 h at 85 °C.
 | ||
| Fig. 13 Strategies and effects of CILs for cathode WF-tuning. (a) Schematic illustration of the effect of PNPDI. (b) Energy level diagram of Ag with and without PNPDI or BPhen. Reproduced with permission from ref. 167. Copyright 2023, Elsevier Ltd. (c) UPS spectra and energy level diagram of Ag with and without NDI-ZI. Reproduced with permission from ref. 168. Copyright 2023, Royal Society of Chemistry. (d) Pole figures of vacuum-deposited (VE) or solution-processed (SC) PDIN-S. (e) UPS spectra of Ag with and without PDIN-S in the secondary electron region. Reproduced with permission from ref. 169. Copyright 2024, Elsevier Ltd. (f) UPS spectra of Ag with PDTI1 in the secondary electron region at a bias voltage of −9 V. (g) UPS spectra of Ag without PDTI1 in the secondary electron region at a bias voltage of −9 V. Reproduced with permission from ref. 171. Copyright 2024, Elsevier Ltd. | ||
Noh et al. developed a zwitterionic naphthalene diimide derivative, NDI-ZI, that introduces both dipole engineering and chemical ion capture as a thin CIL.168 The hydrophobic NDI core stacks toward C60, while zwitterionic sulfobetaine groups orient toward Ag, positioning –SO3− groups adjacent to the metal. This configuration upshifted the Ag-side vacuum level (Ag WF from −4.6 to −4.2 eV; Fig. 13c) and reduced the C60/Ag energy offset. Devices (ITO/MeO-2PACz/Cs0.15FA0.85PbI3/C60/NDI-ZI/Ag) exhibited suppressed shunts and leakage, achieving 23.3% PCE and superior thermal/light stability relative to control devices. Packing-induced dipole orientation has also proven decisive. Kim et al. showed that a PDI derivative with dimethylamino terminal groups (PDIN-S) exhibits morphology-dependent dipole orientation: solution processing yields edge-on packing, whereas vacuum deposition produces face-on packing with vertically aligned π–π stacking (Fig. 13d).169 The latter strengthens the perpendicular dipole component, reducing the Ag WF from −4.6 to −4.0 eV (Fig. 13e). Wu et al. synthesized PDI-2N and PDI-4N, bearing two and four dimethylamino groups, respectively.170 PDI-4N exhibited superior solubility, conductivity (6.3 × 10−4 S cm−1), and mobility (1.1 × 10−3 cm2 V−1 s−1) due to stronger intrinsic dipoles and enhanced self-doping. Due to the reduced WF of Ag from −4.20 to −3.60 eV (PDI-2N) and −3.18 eV (PDI-4N), devices using PDI-4N (FTO/MeO-2PACz/Csx(MA0.17FA0.83)(1−x)Pb(I0.83Br0.17)3/PC61BM/PDI-4N/Ag) achieved 21.3% PCE with excellent stability (97% retention after 600 h).
Gao et al. also reported a fused-ring D–A–D PDI derivative (PDTI1) designed for strong dipole formation and high conductivity.171 Terminal dimethylamino groups oriented negative charge toward the metal, reducing the Ag WF from −4.60 to −4.03 eV (Fig. 13f and g). Devices (ITO/Me-4PACz/FA0.7MA0.3PbI3/PEAI/C60/PDTI1/Ag) achieved 24.6% PCE with remarkable thickness tolerance and retained 94% efficiency after 2600 h, outperforming BCP-based devices. Wang et al. further demonstrated carbolong-derived organometallic complexes (a1, a2, and b1) as CILs, where coordinated cation–anion assemblies self-orient to form strong interfacial dipoles.172 Complex b1, containing a phosphonium cation and BF4−, produced the largest WF reduction (Ag: −4.26 → −3.89 eV; Au: −4.70 → −4.11 eV). Imran et al. designed a hybrid Eu-MOF@BCP interlayer, where MOF fragments incorporated into BCP introduce oriented local dipoles that modulate the interfacial electrostatic potential.173 The Eu-MOF/BCP/Cu interface displayed a reduced WF (−4.31 eV) compared to Cu (−4.70 eV) or BCP/Cu (−4.41 eV), enabling improved electron transport and suppressed hole backflow.
Across these material classes, WF modulation consistently arises from the formation of interfacial dipoles—typically originating from intrinsic ionic character or oriented donor–acceptor motifs—that shift the vacuum level and reduce the barrier for electron extraction. These dipole-engineered interfaces provide several universal device benefits: (1) a higher FF via reduced series resistance and enhanced extraction efficiency; (2) increased JSC due to improved electron transport and collection; (3) enhanced operational stability through suppressed ion migration and improved environmental resistance; and (4) reduced hysteresis through more balanced charge transport and fewer interfacial traps.
 | ||
| Fig. 14 Formation of AgI by ion diffusion and its impact on PeSCs. (a) Thermal degradation mechanism. (b) XRD (left) and GIXRD (right) of the peeled Ag electrodes from an MAPbI3-based PeSC after a thermal treatment at 100 °C for 72 h. (c) ToF-SIMS elemental depth profiles of the aged device stack after a thermal treatment at 85 °C for 24 h. Reproduced with permission from ref. 175. Copyright 2017, Wiley-VCH GmbH. (d) Schematic of band energy diagram in the PeSC after ion accumulation at the interfaces. Reproduced with permission from ref. 181. Copyright 2021, Wiley-VCH GmbH. | ||
Several organic CIL materials are built on conjugated cores (e.g., NDI and PDI) functionalized with ionic or coordinating groups that directly interact with metals or halides, thereby suppressing the migration of mobile ionic species such as I− and Ag+ (Fig. 15). By either physically blocking diffusion pathways or chemically immobilizing ions and metal atoms, these CILs effectively interrupt the diffusion–corrosion cycle at the buried metal contact and stabilize inverted PeSCs. One representative strategy relies on dense molecular packing to physically hinder ion diffusion. Ning et al. systematically investigated NDI-BN, an NDI derivative bearing four pyridine side groups that promote strong intermolecular interaction and tight film packing (Fig. 16a).182 The resulting compact interlayer effectively blocked iodide diffusion from the perovskite toward the Ag electrode, as evidenced by suppressed AgI formation in XPS analysis (Fig. 16b).
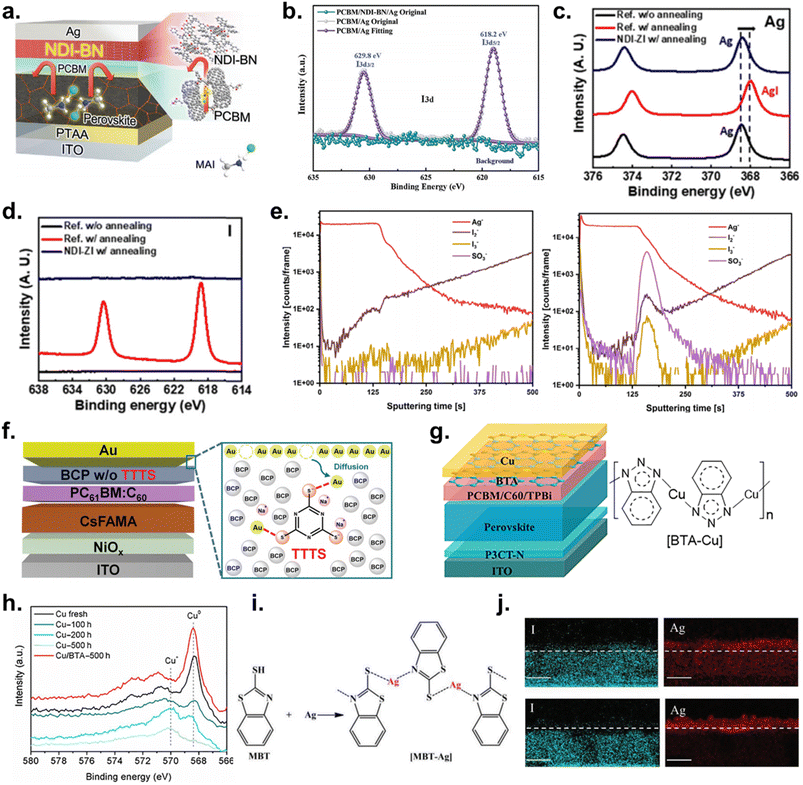 | ||
| Fig. 16 (a) Device adopting NDI-BN and its molecular packing. (b) XPS spectra of Ag with and without NDI-BN after aging for 48 h. Reproduced with permission from ref. 182. Copyright 2020, Wiley-VCH GmbH. (c) Ag 3d and (d) I 3d XPS spectra of the Ag surface with and without NDI-ZI after thermal annealing. (e) ToF-SIMS profiles of control (left) and PDI-C5-S3-adopting (right) PeSCs aged at 60 °C. Reproduced with permission from ref. 183. Copyright 2025, Wiley-VCH GmbH. (f) Device structure and schematic illustration of BCP with TTTS. Reproduced with permission from ref. 185. Copyright 2022, Royal Society of Chemistry. (g) Device structure and schematic illustration of BTA-Cu. (h) Auger electron spectroscopy (AES) results of a Cu electrode with and without BTA aged at 85 °C. Reproduced with permission from ref. 76. Copyright 2020, the American Association for the Advancement of Science. (i) Formation mechanism of MBT-Ag. (j) EDS map of I and Ag from PeSCs aged at 85 °C for 500 h without (top) and with (bottom) MBT. Reproduced with permission from ref. 186. Copyright 2024, Wiley-VCH GmbH. | ||
Beyond physical blocking, chemical ion trapping provides a more active suppression mechanism. Noh et al. introduced NDI-ZI, a zwitterionic NDI derivative containing sulfobetaine side chains.168 The fixed positive and negative charges simultaneously induce a strong interfacial dipole for work-function tuning and act as coulombic ion capture sites for mobile I−/I3− and Ag species. While the NDI backbone ensures electron transport, the ionic side groups chemically immobilize both halides and metal ions, effectively suppressing AgI formation after thermal stress (Fig. 16c and d). This dual functionality illustrates how zwitterionic design enables concurrent electronic optimization and chemical stabilization. A similar zwitterionic concept was extended to PDI systems by Orwat et al., who incorporated sulfobetaine groups into PDIs with systematically varied alkyl chain lengths.183 In these materials, perpendicular dipole alignment at the interface lowers the Ag WF, while sulfonate groups chemically bind mobile Ag+ and halide species. Among the series, PDI-C5-S3 exhibited the optimal balance between dipole strength and ion-capturing ability (Fig. 16e). Wide-bandgap PeSCs employing PDI-C5-S3 (ITO/2PACz/Cs0.18FA0.82Pb(I0.8Br0.2)3/PEAI/PC61BM/PDI-C5-S3/Ag) achieved 19.0% PCE under one-sun illumination and an exceptional 40.7% efficiency under indoor LED lighting, underscoring the importance of precise side-chain engineering. Wang et al. developed a tetramethylammonium-functionalized perylene derivative (TMA-PTC) as a solution-processable CIL.184 The quaternary ammonium cations of TMA-PTC can also attract migrating I− anions by their ionic interactions, thereby suppressing the migration of iodides to the cathode.
Another major class of CIL strategies focuses on direct chemical coordination with metal atoms, immobilizing the electrode surface and preventing metal diffusion. Yang et al. introduced a hybrid BCP:TTTS interlayer, where 1,3,5-triazine-2,4,6-trithiol trisodium salt (TTTS) provides tridentate thiol groups that form strong S–metal coordination bonds with Ag, Au, and Cu (Fig. 16f).185 This chemical anchoring effectively suppresses metal in-diffusion while maintaining favorable energy-level alignment for electron extraction. Similarly, Li et al. employed benzotriazole (BTA) as an anticorrosion interlayer for Cu cathodes.76 The triazole moiety coordinates strongly with Cu to form an insoluble [BTA–Cu] complex, which blocks both halide attack and Cu diffusion (Fig. 16g and h). Devices using BTA-protected Cu cathodes retained over 92% of their initial PCE after 2500 h in air and over 90% after 1000 h at 85 °C. Liu et al. further demonstrated that 2-mercaptobenzothiazole (MBT) forms compact MBT–Ag coordination chains through Ag–S and Ag–N bonds, simultaneously suppressing I− out-diffusion and Ag corrosion (Fig. 16i and j).186 Another structurally elaborate system combines chemical coordination and physical blocking. Yu et al. developed thiol-functionalized covalent organic frameworks (SH-COFs) as crystalline CILs.187 Abundant –SH groups chemically anchor Ag atoms via Ag–S bonds, while the extended COF lattice forms a dense diffusion barrier.
In parallel, inorganic-based CILs provide intrinsically dense and chemically robust barriers. Back et al. introduced an amine-mediated TiOx (AM-TiOx) interlayer, where amine groups electrostatically trap migrating halides and MA+ cations, suppressing ion-induced corrosion at the cathode.188 Devices incorporating AM-TiOx retained nearly 80% of their initial PCE for over 9000 h in N2. Zhu et al. demonstrated a SnO2/W-doped ZnO bilayer, in which SnO2 blocks ion diffusion while W doping stabilizes ZnO surface chemistry, yielding inverted devices with 23.2% PCE and enhanced thermal durability.189 Chen et al. further showed that an ultrathin Ti interlayer exploits low diffusivity of Ti and Ti–N bonding with methylammonium, simultaneously passivating defects and blocking metal diffusion.190
4. Tandem-relevant interlayers
Tandem solar cells consist of multiple subcells with distinct bandgaps, enabling broadband solar absorption through spectral splitting. High-energy photons are absorbed in the wide-bandgap subcells, while lower-energy photons are transmitted to and harvested by the narrow-bandgap subcells. Owing to their high theoretical efficiency (43–46% for perovskite-based two-junction tandem architectures, according to the Shockley–Queisser limit), tandem devices are widely regarded as one of the most promising photovoltaic platforms.191,192MHPs are particularly well suited for tandem integration because their optical bandgap can be tuned via compositional engineering, allowing precise spectral complementarity with various bottom cells. Accordingly, a range of perovskite-based tandem configurations—including Si–perovskite, all-perovskite, organic-perovskite, and CIGS-perovskite structures—have been extensively investigated. Among these, monolithic two-terminal (2T) Si–perovskite tandems currently deliver the highest performance, with a certified PCE of 35.0%.1 In monolithic 2T Si–perovskite tandems, a wide-bandgap perovskite top cell is vertically stacked on a silicon bottom cell and electrically coupled in series through an interconnecting recombination layer. Under front-side illumination, incident light first enters the perovskite subcell, where short-wavelength photons are preferentially absorbed. Longer-wavelength photons are transmitted to the Si bottom cell and absorbed sequentially. Because the two subcells are series-connected, the overall tandem current is limited by the lower photocurrent of the two; therefore, precise spectral management and current matching are essential for optimal device operation.
For tandem integration, the perovskite top cell typically requires a wider optical bandgap (1.65–1.75 eV).193–195 Such wide-bandgap absorbers are commonly realized by increasing the bromide fraction relative to iodide in mixed-halide compositions. However, under illumination, bromide and iodide ions tend to segregate, forming spatially separated Br-rich and I-rich domains. The iodide-rich regions possess a lower bandgap and act as recombination centers, reducing the QFLS and leading to a significant VOC deficit in wide-bandgap devices.196,197 To address this challenge, extensive efforts have focused on suppressing ion migration and stabilizing the mixed-halide lattice through compositional engineering, defect-passivation strategies that reduce halide-vacancy-mediated transport, and interface and dimensional engineering approaches (discussed in earlier sections). Ultimately, achieving intrinsically segregation-resistant wide-bandgap perovskites is a prerequisite for simultaneously maximizing tandem efficiency and ensuring long-term operational stability.
In this section, we discuss tandem-relevant interlayers, particularly for Si–perovskite tandem architectures. These include (i) interconnecting layers (ICLs), (ii) interlayers that enable conformal perovskite coating over textured Si bottom cells, (iii) ETLs with reduced parasitic absorption, and (iv) buffer layers that protect the perovskite stack from sputtering-induced damage during transparent electrode deposition. Consequently, these interlayers critically influence the performance and reliability of monolithic tandem devices (Fig. 17a).
 | ||
| Fig. 17 (a) Interlayers in Si–perovskite tandem solar cells and (b) c-AFM images of Si/ITO/NiOx, Si/ITO/MeO-2PACz, and Si/ITO/NiOx/MeO-2PACz films. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 198. Copyright 2024, Elsevier. (c) Perovskite layers deposited on the micron-scale pyramid texture. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 236. Copyright 2024, John Wiley & Sons. (d) Transmittance of SnO2 QDs and C60 films. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 209. Copyright 2024, Wiley-VCH GmbH (e) Transmittance spectra of films with thin C60 and ALD SnO2 layers. Reproduced with permission from ref. 211. Copyright 2025, Wiley-VCH GmbH. (f) Ion penetration depth distribution induced by sputtering plasma in the perovskite/C60 stack. Reproduced under the terms of the CC-BY Creative Commons Attribution 4.0 International license (https://creativecommons.org/licenses/by/4.0) from ref. 215. Copyright 2024, The Royal Society of Chemistry. | ||
4.1. ICLs between top and bottom subcells
Interconnecting layers (ICLs) are indispensable components in monolithic 2T tandem solar cells, positioned between the wide-bandgap top subcell and the narrow-bandgap bottom subcell to enable an efficient series electrical connection while minimizing optical and electrical losses. Functionally, an effective ICL must simultaneously satisfy three essential requirements: (1) it must provide efficient electrical interconnection by forming low-barrier contacts with adjacent carrier-selective layers and offering a highly effective recombination site for electrons and holes; (2) it must ensure optical interconnection by retaining high transparency, particularly in the NIR region to preserve the bottom-cell photocurrent; and (3) it must serve as a mechanically and chemically robust barrier that prevents solvent penetration and interdiffusion during the sequential processing of the top subcell. In the absence of a properly engineered ICL, direct contact between the two subcells can result in the formation of a parasitic junction, increasing recombination resistance and substantially degrading tandem performance.Two principal ICL design strategies have been developed: tunnel-junction (TJ)-based and recombination-layer (RL)-based architectures. TJ-based ICLs rely on degenerately doped p++/n++ junctions that enable carrier tunneling and recombination with minimal voltage drop, thereby offering highly efficient electrical coupling in principle. However, realizing sufficiently strong and stable doping—particularly in multilayer device stacks fabricated by solution processing—while simultaneously ensuring solvent resistance and structural robustness remains technically challenging. In contrast, RL-based ICLs introduce an explicit recombination medium, such as sputtered TCOs, ultrathin metals (∼1 nm), or conductive polymers, between the charge-selective layers. These structures often enhance recombination efficiency and improve the FF, yet they require meticulous control over sputter-induced damage, shunt formation, and parasitic optical absorption.
Because the two subcells are connected in series, ICL design directly governs both performance and stability. Electrically inefficient or energetically misaligned ICLs increase the effective recombination resistance at the interconnection and may even form a parasitic diode, leading to VOC losses. Ideally, the tandem VOC should approach the sum of the QFLS of the individual subcells. Non-ideal ICLs introduce several loss mechanisms: (i) energy-level misalignment can cause barrier-induced carrier accumulation, leading to interfacial charge build-up, enhanced non-radiative recombination, and reduced subcell QFLS; (ii) insufficient recombination capacity within the ICL can create a recombination-limited bottleneck that lowers the FF; and (iii) structural defects such as pinholes can generate shunt or leakage pathways that degrade both the FF and VOC. From an optical standpoint, absorbing or excessively thick interlayers reduce NIR transmission and limit the tandem JSC, further emphasizing the need for carefully optimized thickness and material selection.
Recent studies illustrate how rational ICL engineering can address these limitations. For instance, Zheng et al. introduced a hybrid NiOX/MeO-2PACz ICL in monolithic Si–perovskite tandems that avoids direct perovskite–TCO contact, thereby suppressing defect-driven shunt and leakage pathways at the interface.198 Conductive atomic force microscope (c-AFM) images indicate that the NiOX/MeO-2PACz hybrid layer exhibited better uniformity and less leakage current than the NiOX and MeO-2PACz films prepared on an ITO substrate (Fig. 17b). By mitigating leakage-related losses and reducing interfacial non-radiative recombination, this strategy achieved a PCE of 28.47% with an FF of 81.8%, alongside improved reproducibility and operational stability. Similarly, An et al. addressed barrier-induced carrier accumulation by employing a SAM (CbzNaph)-anchored MoO3 interconnection structure that balances carrier transport and enhances hole extraction, thereby reducing charge build-up at the series junction.199 Tandem devices incorporating this tailored ICL delivered a PCE of 26.05%, a VOC of 2.21 V, and high operational stability.
Collectively, these advances underscore that ICLs are not merely passive interlayers but decisive functional elements that govern recombination dynamics, voltage additivity, current continuity, and long-term robustness in monolithic tandems. Continued progress in ICL material selection and structural design—aimed at minimizing electrical and optical interconnection losses while enhancing chemical and processing resilience—will be pivotal for realizing high-efficiency, stable 2T tandem solar cells.
4.2. Interlayers on textured Si bottom cells
In Si–perovskite tandem solar cells, microscale pyramid texturing of the Si bottom cell is widely employed to suppress front-surface reflection and enhance light trapping. Random (111)-oriented pyramids, typically 2–4 µm in height, induce multiple internal reflections, thereby increasing the optical path length within Si and boosting the photocurrent of the bottom sub-cell. Such textures are most commonly fabricated by anisotropic alkaline etching using KOH, NaOH, or TMAH, which exploit crystallographic etch-rate selectivity to form ordered pyramidal arrays.200–202Despite their optical advantages, fully textured Si surfaces pose significant challenges for subsequent solution processing of perovskite layers. Achieving conformal coverage of micrometer-scale pyramids remains difficult because the perovskite precursor solution tends to dewet from sharp features, bridge across pyramid valleys, and leave voids or pinholes. These morphological defects can induce shunting pathways and exacerbate interfacial recombination, ultimately degrading device performance and yield (Fig. 17c).236 To address this trade-off between optical light trapping and conformal coating, several strategies have been explored. A straightforward approach involves reducing the pyramid dimensions to sub-micrometer scales (“mild texturing”), thereby decreasing surface roughness and improving precursor wetting.203–205 In this configuration, the perovskite thickness can readily exceed the substrate topography, enabling uniform film formation. However, limiting pyramid height inevitably compromises the maximum achievable light-trapping capability. Alternatively, the original micrometer-scale pyramids can be retained while introducing hierarchical micro/nanotextures to promote improved wetting and nucleation.206 Although this strategy preserves optical benefits, it generally requires additional etching or surface modification steps, increasing process complexity and manufacturing cost.
More recently, inorganic NP interlayers have emerged as an effective and scalable solution to this conformality challenge. For example, Liu et al. introduced an Al2O3 NP interlayer via spray coating to render the textured Si surface superhydrophilic.207 This treatment enhances perovskite precursor spreading and wet-film continuity across pyramid tips and sidewalls. The NPs additionally serve as distributed heterogeneous nucleation sites, mitigating valley-dominant crystallization and promoting more homogeneous perovskite growth. Similarly, Zhang et al. employed SiOX nanospheres as a nano-filler interlayer to locally smooth sharp valleys and steep facets without sacrificing the global light-trapping texture.208 This localized topographical regulation facilitates continuous HTL deposition and enables pinhole-free, conformal perovskite coverage on fully textured substrates. Collectively, these NP-enabled interface engineering strategies demonstrate that the inherent limitations of solution coating on micron-scale pyramids can be overcome by combining enhanced wetting with guided nucleation or texture-aware surface modulation. From an industrial perspective, such inorganic NP interlayers are particularly attractive because they can be implemented via low-temperature, solution-based processes (e.g., spray or colloidal deposition) compatible with large-area manufacturing.
4.3. ETLs on perovskite top cells with reduced parasitic absorption
Although fullerene derivatives such as C60 and its functionalized analogues are widely employed as ETLs in high-efficiency inverted PeSCs, their intrinsic optical absorption presents a critical limitation for Si–perovskite tandem applications. Fullerene-based ETLs exhibit appreciable absorption in the near-UV and blue spectral region (≈330–450 nm), with residual absorption extending into the green region (≈450–550 nm). Even at typical thicknesses of 20–30 nm, illumination through the ETL induces short-wavelength parasitic losses that can reduce the JSC by approximately 1.5 mA cm−2.209 In monolithic tandem architectures, where efficient photon utilization is essential for subcell current matching, such losses directly constrain overall device performance. Consequently, substantial research efforts have focused on minimizing ETL parasitic absorption while preserving efficient electron extraction and interfacial selectivity.One major strategy involves replacing fullerene-based ETLs with highly transparent wide-bandgap metal oxides, most notably SnO2. Owing to its large bandgap (≈3.6–4.5 eV), SnO2 exhibits high optical transparency across the visible spectrum, typically exceeding 80% transmittance between 400 and 800 nm (Fig. 17d).209 This significantly reduces parasitic absorption and increases the fraction of incident photons reaching the perovskite absorber. However, in inverted configurations, SnO2 must be deposited directly onto the perovskite layer, which imposes stringent solvent orthogonality requirements. Most polar solvents effective for dispersing colloidal SnO2 can damage perovskite films, limiting solution processing to a narrow selection of perovskite-compatible solvents. To overcome this constraint, ligand engineering of SnO2 NPs is frequently implemented to enable stable dispersions in benign solvents. Chapagain et al. implemented a ligand exchange strategy in which hydrous SnO2 NPs were capped with acetate, yielding stable ethanol-based inks compatible with perovskite surfaces.210 The use of short-chain acetate ligands minimized electrical losses by limiting the insulating barrier at the interface. In addition, hydrogen bonding between surface-bound acetate groups and ethanol was proposed to enhance colloidal dispersibility, enabling direct deposition onto the perovskite layer without the need for an additional interlayer. Similarly, Kim et al. reported long-term colloidal stability in a low-polarity chloroform/1-butanol cosolvent system, which they attributed to the presence of surface Cl− species.209 They further introduced ethylenediamine (EDA) as a bridging ligand that coordinates to perovskite surface defects while simultaneously anchoring to SnO2 NPs, thereby promoting intimate interfacial contact and improved electronic coupling.
Another effective strategy to suppress parasitic absorption is to reduce the thickness of fullerene-based ETLs while retaining efficient electron extraction and interfacial selectivity, as optical losses scale with both the absorber thickness and the fullerene volume fraction. Recent studies have demonstrated two complementary design approaches that enable thickness- and/or absorption-minimized ETLs without sacrificing device performance. Cui et al. introduced an ultrathin PO-T2T interlayer (∼0.5 nm) to improve heterointerfacial contact and charge selectivity.211 This modification allowed the ETL stack thickness to be reduced from 20 nm C60/20 nm ALD SnO2 to 15 nm C60/17 nm ALD SnO2 without compromising photovoltaic performance. Also the transmittance spectra of the thin films reveal that thinner C60 and ALD SnO2 layers exhibit higher transmittance across the entire wavelength range (Fig. 17e). In parallel, Poma et al. adopted a materials-centric approach by blending an NDI-based polymer (pNDI) with C60 to form a composite ETL (pNDI-C60).212 This hybrid layer not only improved mechanical robustness but also reduced optical absorption relative to neat evaporated C60. Because pNDI exhibits only weak absorption, primarily below ∼400 nm, it effectively dilutes the C60 fraction and lowers the overall absorptance of the ETL. As a result, the pNDI-C60 layer increases transmitted optical power through the ETL stack and alleviates parasitic absorption, even without aggressive thickness reduction.
Taken together, these developments demonstrate that parasitic absorption in ETLs for Si–perovskite tandems can be systematically mitigated either by substituting highly transparent metal oxides such as SnO2 for fullerene derivatives or by rationally engineering thinner and optically diluted fullerene-based layers. In both cases, the central design objective is to balance optical transparency with robust electron extraction and interfacial selectivity, thereby maximizing photocurrent while sustaining high FF and operational stability in tandem architectures.
4.4. Buffer layers on perovskite top cells for sputter damage mitigation
In Si–perovskite tandem photovoltaics, electrode design requirements differ substantially from those of single-junction devices. Because illumination occurs through the top subcell, the front electrode must be highly transparent while retaining low sheet resistance to enable efficient lateral charge collection over large areas. Generally, transparent contacts typically employ sputtered transparent conductive oxides (TCOs), most commonly ITO or IZO, often combined with a metal grid or mesh to further reduce series resistance without incurring excessive optical losses.213,214However, TCO sputtering can significantly degrade the underlying perovskite stack. The plasma environment generates energetic ions, neutral particles, UV radiation, and localized heating, all of which can physically damage soft organic layers such as fullerene-based ETLs, increase defect density, and enhance non-radiative recombination.215,216 In particular, with respect to ion-induced damage, the mechanism is unlikely to involve direct ion penetration into the perovskite. Instead, oxygen, argon, indium, and tin ions are predicted to stop within the C60 overlayer at depths below ∼5 nm, indicating that the damage is more plausibly associated with near-surface collision cascades and recoil-induced defect formation (Fig. 17f).215 Additionally, sputter-induced mechanical stress may cause interfacial delamination or deteriorate contact quality.217 To mitigate these effects, monolithic Si–perovskite tandems typically incorporate a thin buffer layer between the perovskite-side transport layers and the sputtered TCO. Buffer layers can be classified according to their deposition method: solution processing, thermal evaporation, and chemical vapor deposition (CVD), including atomic layer deposition (ALD).
Overall, buffer layer engineering in tandem-compatible perovskite solar cells is not merely a protective strategy but a critical interface optimization problem, requiring simultaneous control of plasma resilience, optical transparency, electrical conductivity, and interfacial energetics to preserve both efficiency and long-term device stability.
5. Outlook and prospects
Based on recent progress, the roles of upper-surface and interface modification strategies can be broadly categorized into four groups: (1) perovskite top-surface defect passivation and improving energy-level alignment at the perovskite/ETL and ETL/cathode interfaces; (2) acting as barrier layers to block moisture and oxygen ingress while suppressing mobile-ion diffusion; (3) enabling internal and interfacial thermal management to facilitate efficient heat dissipation; and (4) replacing thermally and mechanically unstable ETLs, such as C60 and PC61BM. Collectively, these approaches underscore the critical importance of perovskite upper-surface engineering, where tailored interfacial materials can deliver distinct yet complementary functions—from contact optimization to robust barrier effects—ultimately enhancing device stability.Despite this progress, several challenges must be addressed to further advance inverted PeSCs. First, greater attention should be paid to defect generation under operational conditions. Upper-stack stability is governed not only by pre-existing defects but also by defects newly formed under illumination, electric fields, and local heating. These stresses can reconstruct the perovskite top surface and activate metastable pathways, generating fresh nonradiative recombination centers at the perovskite/ETL interface. Future strategies should therefore move beyond static passivation toward interface designs that actively suppress defect formation during operation. In this context, combining operando spectroscopic techniques with time-resolved electrical measurements will be critical for identifying when and where defect-related loss pathways emerge under light and bias.
Second, mechanical integrity at the perovskite/ETL/cathode interfaces remains an underappreciated bottleneck for long-term reliability. The upper stack integrates perovskites with organic and inorganic layers that possess mismatched elastic moduli and thermal-expansion coefficients, rendering the interfaces vulnerable to delamination and microcrack formation during thermal cycling. Such adhesion loss can create local contact failure and diffusion pathways (for O2 and water), thereby accelerating chemical and electrochemical degradation. Accordingly, adhesion should be treated as a key design parameter in future upper-stack engineering. Molecularly engineered interlayers that enhance interfacial bonding while preserving ultrathin charge-transport pathways offer a promising solution.
Third, interfacial damage induced during electrode formation must be carefully considered. As emerging device architectures—including tandem, semitransparent, bifacial, and module-level configurations—expand beyond conventional metal electrodes, the deposition processes themselves can compromise the integrity of carefully engineered upper interfaces. Sputtering of transparent conductive electrodes can cause ion bombardment or plasma-induced damage to the ETL and perovskite surface. Consequently, well-designed perovskite/ETL stacks can fail if electrode deposition introduces new defects or disrupts interfacial continuity. Effective solutions should therefore integrate deposition-compatible interlayers that function as sacrificial or protective buffers without incurring optical or electrical penalties.
Finally, standardized multi-stress testing protocols are urgently needed for the reliable design of inverted PeSCs. Device degradation is often accelerated by synergistic interactions among heat, light, electrical bias, moisture, and oxygen, yet many studies continue to evaluate stability under isolated stress conditions. The establishment of unified, multi-factor evaluation protocols will help identify dominant degradation triggers and guide rational selection of interlayers, ETLs, and electrodes. Such comprehensive reliability assessments will be pivotal for translating laboratory-scale interface innovations into commercially viable technologies.
In summary, PeSCs are transitioning from a laboratory-driven research phase toward commercialization-oriented development. As efficiencies continue to rise, the central question is no longer whether perovskites can achieve high performance, but whether they can sustain reliable operation under realistic conditions and at the manufacturing scale. A deeper understanding of how materials, interfaces, and large-area processes behave in realistic operating and degradation environments is essential for translating record laboratory performance into industrially relevant photovoltaic technologies.
Author contributions
S. P. and J. B. P. mainly contributed to writing and editing the initial manuscript. J. M. H. and H. S. K. revised the manuscript. M. H. S. and H. Y. W. conceived the idea of the review article and supervised it.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analyzed as part of this review.Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (RS-2024-00334832 and RS-2025-24683763), the Korea Institute of Energy Technology Evaluation and Planning (KETEP) grant funded by the Korean government (MOTIE) (20213091010010, Super Solar Cells – Development of double junction solar cells, breakthrough for the theoretical limit of silicon solar cell efficiency (>35%)), the Nano and Material Technology Development Program through the NRF funded by the Ministry of Science and ICT (RS-2025-25442266), and the InnoCORE program of the Ministry of Science and ICT (1.250021.01).References
- National Renewable Energy Laboratory, ‘‘Best Research-Cell Efficiency Chart’’, https://www.nrel.gov/pv/cell-efficiency.html.
- G. Li, Z. Zhang, B. Agyei-Tuffour, L. Wu, T. W. Gries, K. Prashanthan, L. Frohloff, A. Musiienko, J. Li, R. Zhu, L. J. F. Hart, L. Wang, Z. Li, B. Hou, N. Koch, M. Saba, P. R. F. Barnes, J. Nelson, P. J. Dyson, M. K. Nazeeruddin, M. Li and A. Abate, Nat. Photonics, 2026, 20, 55–62 CrossRef CAS PubMed.
- C. Tian, A. Sun, J. Chen, R. Zhuang, C. Chen, J. Zheng, S. Liu, J. Du, Q. Chen, L. Cai, S. Han, F. Tian and C.-C. Chen, Nat. Photonics, 2026, 20, 287–295 CrossRef CAS.
- J. Park, L. Chetan, H. Kim, J.-S. Jee, Y.-S. Gal and S.-H. Jin, Macromol. Res., 2024, 32, 505–513 CrossRef CAS.
- H. Choi, H. Kim, J. Lim, B.-J. Chang and S. Song, Macromol. Res., 2024, 32, 825–832 CrossRef CAS.
- B. Dong, M. Wei, Y. Li, Y. Yang, W. Ma, Y. Zhang, Y. Ran, M. Cui, Z. Su, Q. Fan, Z. Bi, T. Edvinsson, Z. Ding, H. Ju, S. You, S. M. Zakeeruddin, X. Li, A. Hagfeldt, M. Grätzel and Y. Liu, Nat. Energy, 2025, 10, 342–353 CrossRef CAS.
- G. Qu, S. Cai, Y. Qiao, D. Wang, S. Gong, D. Khan, Y. Wang, K. Jiang, Q. Chen, L. Zhang, Y.-G. Wang, X. Chen, A. K. Y. Jen and Z.-X. Xu, Joule, 2024, 8, 2123–2134 CrossRef CAS.
- W. Jiang, G. Qu, X. Huang, X. Chen, L. Chi, T. Wang, C.-T. Wong, F. R. Lin, C. Yang, Q. Jiang, S. Wu, J. Zhang and A. K. Y. Jen, Nature, 2025, 646, 95–101 CrossRef CAS PubMed.
- W. Li, E. Martínez-Ferrero and E. Palomares, Mater. Chem. Front., 2024, 8, 681–699 RSC.
- M. Li, M. Liu, F. Qi, F. R. Lin and A. K. Y. Jen, Chem. Rev., 2024, 124, 2138–2204 CrossRef CAS PubMed.
- J. Suo, B. Yang, D. Bogachuk, G. Boschloo and A. Hagfeldt, Adv. Energy Mater., 2025, 15, 2400205 CrossRef CAS.
- K. Guo, H. Tang, L. Han, R. Qi, H. Yan, G. Gao, W. Lv, M. Li, J. Xia, B. Cai, G. Xing, R. Chen and G. Wu, ACS Energy Lett., 2025, 10, 4882–4910 CrossRef CAS.
- C. E. Puerto Galvis, D. A. González Ruiz, E. Martínez-Ferrero and E. Palomares, Chem. Sci., 2024, 15, 1534–1556 RSC.
- R. Chen, J. Wang, Z. Liu, F. Ren, S. Liu, J. Zhou, H. Wang, X. Meng, Z. Zhang, X. Guan, W. Liang, P. A. Troshin, Y. Qi, L. Han and W. Chen, Nat. Energy, 2023, 8, 839–849 CrossRef CAS.
- X. Qiao, R. Zhu, D. Yan, Z. Su, Z. Zhang, H. Wu, Y. Tan, M. Liang, W. Zuo, J. Zhang, G. Li, X. Gao, M. Saliba and M. Li, Adv. Funct. Mater., 2024, 34, 2409852 CrossRef CAS.
- H. Sun, S. Wang, S. Qi, P. Wang, R. Li, B. Shi, Q. Zhang, Q. Huang, S. Xu, Y. Zhao and X. Zhang, Adv. Funct. Mater., 2023, 33, 2213913 CrossRef CAS.
- N. Aristidou, C. Eames, I. Sanchez-Molina, X. Bu, J. Kosco, M. S. Islam and S. A. Haque, Nat. Commun., 2017, 8, 15218 CrossRef PubMed.
- B. Rivkin, P. Fassl, Q. Sun, A. D. Taylor, Z. Chen and Y. Vaynzof, ACS Omega, 2018, 3, 10042–10047 CrossRef CAS PubMed.
- S. Suragtkhuu, P. Myagmarsereejid, Q. T. Trinh, T. Tsagaantsooj, A. D. Slattery, A. Adenle, S. Purevdorj, E. Campbell, O. E. FitzGerald, T. J. Z. Stock, S. Jamali, T. J. Macdonald, T. Gould, N.-T. Nguyen, C. Adachi, Y. L. Zhong and M. Batmunkh, EES Sol., 2025, 1, 857–867 RSC.
- C. Chen, P. Zhu, X. Dong, Y. Dou, Y. Zhang, J. Liang, R. Mao, Y. Jiang, J. Wang, M. Wang, S. Chen and J. Zhu, Chem. Eng. J., 2024, 489, 151403 CrossRef CAS.
- Z. Liu, H. Li, Z. Chu, R. Xia, J. Wen, Y. Mo, H. Zhu, H. Luo, X. Zheng, Z. Huang, X. Luo, B. Wang, X. Zhang, G. Yang, Z. Feng, Y. Chen, W. Kong, J. Gao and H. Tan, Adv. Mater., 2024, 36, 2308370 CrossRef CAS.
- Y. Zheng, Y. Li, R. Zhuang, X. Wu, C. Tian, A. Sun, C. Chen, Y. Guo, Y. Hua, K. Meng, K. Wu and C.-C. Chen, Energy Environ. Sci., 2024, 17, 1153–1162 RSC.
- J. Warby, F. Zu, S. Zeiske, E. Gutierrez-Partida, L. Frohloff, S. Kahmann, K. Frohna, E. Mosconi, E. Radicchi, F. Lang, S. Shah, F. Peña-Camargo, H. Hempel, T. Unold, N. Koch, A. Armin, F. De Angelis, S. D. Stranks, D. Neher and M. Stolterfoht, Adv. Energy Mater., 2022, 12, 2103567 CrossRef CAS.
- M. Stolterfoht, P. Caprioglio, C. M. Wolff, J. A. Márquez, J. Nordmann, S. Zhang, D. Rothhardt, U. Hörmann, Y. Amir, A. Redinger, L. Kegelmann, F. Zu, S. Albrecht, N. Koch, T. Kirchartz, M. Saliba, T. Unold and D. Neher, Energy Environ. Sci., 2019, 12, 2778–2788 RSC.
- Z. Xing, S. Ma, B.-W. Chen, M. An, A. Fan, X. Hu, Y. Wang, L.-L. Deng, Q. Huang, H. Kanda, F. G. Al-Amri, G. Pozzi, Y. Zhang, J. Xia, J. Wu, X. Guo and M. K. Nazeeruddin, Joule, 2025, 9, 101817 CrossRef CAS.
- J. Yin, X. Shi, L. Wang, H. Yan and S. Chen, Angew. Chem., Int. Ed., 2022, 61, e202210610 CrossRef CAS PubMed.
- F. Li, C. Zhao, Y. Li, Z. Zhang, X. Huang, Y. Zhang, J. Fang, T. Bian, Z. Zeng, J. Yin and A. K. Y. Jen, Sci. Adv., 2024, 10, eadq1150 CrossRef CAS PubMed.
- S. Liu, T. Miao, J. Wang, Y. Zhang, R. Chen, X. Lei, W. Qin, Z. Zhu, L. Lu, Z. Chen, P. Cui, L. Li, M. Li, E. Xu, Y. Shen, S. C. Cho, S. U. Lee, S.-H. Cho, Z. Liu, W. Chen and N.-G. Park, Nat. Energy, 2026, 11, 109–120 CrossRef CAS.
- Y. Feng, Y. Wang, H. Yang, S. Li, J. Luo, X. Han, T. Alshahrani, B. Mai, D. Wang, Z. Ding, W. Sun, C. Sun, Q. Chen, K. Wei, Y. Feng, L. Song, X. Li, X.-Y. Wang, W.-D. Li, Q. An, Y. Jiang and M. Yuan, Angew. Chem., Int. Ed., 2025, 64, e202505876 CrossRef CAS PubMed.
- D. Gao, B. Li, X. Sun, Q. Liu, C. Zhang, L. Qian, Z. Yu, X. Li, X. Wu, B. Liu, N. Wang, F. Vanin, X. Xia, J. Gong, N. Li, X. C. Zeng, Z. a. Li and Z. Zhu, Nat. Photonics, 2025, 19, 1070–1077 CrossRef CAS.
- J. Yang, G. Qu, Y. Qiao, S. Cai, J. Hu, S. Geng, Y. Li, Y. Jin, N. Shen, S. Chen, A. K. Y. Jen and Z.-X. Xu, Nat. Commun., 2025, 16, 6968 CrossRef CAS PubMed.
- J. Zhang, N. Yan, Y. Cao, Y. Li, R. Wang, D. Qi, J. Pi, N. Li, X. Feng, J. Wu, X. Liu, X. Gao, Y. Liu, S. Liu and J. Feng, Adv. Mater., 2026, 38, e11162 CrossRef CAS PubMed.
- K. Feng, G. Wang, Q. Lian, S. Gámez-Valenzuela, B. Li, R. Ding, W. Yang, K. Wang, J. Zeng, Y. Zhang, S. Y. Jeong, B. Xu, A. Ho-Baillie, H. Y. Woo, A. Facchetti and X. Guo, Nat. Mater., 2025, 24, 770–777 CrossRef CAS PubMed.
- Y. Zhang, Y. Liu, Z. Zhao, T. Kong, W. Chen, W. Liu, Y. Rong and D. Bi, Adv. Mater., 2025, 37, 2500501 CrossRef CAS PubMed.
- H. Rao, S. Ye, T. Salim, R. Mayengbam, Y. Guo, M. Feng, L. Xi, Z. Yen, R. Salim, Y. Wang, R. Cai, X. Xiao, B. Wang, H. He, T. C. Sum and Y. M. Lam, Nat. Energy, 2025, 10, 991–1000 CrossRef CAS.
- Z.-Y. Suo, R. Dong, C. Chen, X. Mu, J. Hao and J. Cao, Adv. Mater., 2026, 38, e17731 CrossRef CAS PubMed.
- X. Tang, C. Yang, Y. Xu, J. Xia, B. Li, M. Li, Y. Zhou, L. Jiang, H. Liu, K. Ma, Q. Yu, B. Dong, Y. Liu, O. F. Mohammed and X. Zheng, Nat. Photonics, 2025, 19, 701–708 CrossRef CAS.
- C. Zhang, L. Yang, Y. Wu, K. Wei, D. Liu, J. Hu, W. Wang, S. Pang, B. Xu and J. Zhang, Joule, 2025, 9, 102217 CrossRef CAS.
- K. Huang, W. Wang, A. D. Bui, W. Ji, F. Kremer, Z. Yang, G. Bartholazzi, Y. Yu, O. L. C. Lem, B. He, Z. Su, V. Ahmad, L. Chang, D.-T. Nguyen, Y. Liu, X. Gao, J. Yang, K. R. Catchpole, H. Shen, K. J. Weber and T. Duong, Adv. Sci., 2025, 12, e14595 CrossRef CAS PubMed.
- K. Kim, S. Yang, C. Kim, J. Park, S. Jeong, Y. Kim, J. Park, Z. Sun, M. Kang, B. J. Kang, J. Oh, J. S. Yun, S.-J. Shin, C. Yang and H. Min, Nat. Energy, 2025, 10, 1427–1438 CrossRef CAS.
- W. Jia, R. Sun, J. Qiao, G. Shi, Q. Zhao, Z. Gong, S. Zheng, R. Xu, J. Shang, L. Song, K. Wang, W. Huang, R. Chen, Y. Fang, H. Wang and Z.-Q. Rong, Angew. Chem., Int. Ed., 2025, 64, e202513869 CrossRef CAS PubMed.
- H. Yang, Z. Guo, Z. Xu, W. Cai, S. M. H. Qaid and Z. Zang, Adv. Mater., 2025, 37, e10967 CrossRef CAS.
- Y. Chen and H. Zhou, J. Appl. Phys., 2020, 128, 060903 CrossRef CAS.
- W.-J. Yin, T. Shi and Y. Yan, Appl. Phys. Lett., 2014, 104, 063903 CrossRef.
- X. Zheng, B. Chen, J. Dai, Y. Fang, Y. Bai, Y. Lin, H. Wei, X. C. Zeng and J. Huang, Nat. Energy, 2017, 2, 17102 CrossRef CAS.
- C. Ruan, L. He, L. Zhu, B. Yuan, H. Yang, G. Qin, Y. Chen and Q. Tao, J. Alloys Compd., 2024, 999, 174990 CrossRef CAS.
- J. Wang, L. Bi, Q. Fu and A. K. Y. Jen, Adv. Energy Mater., 2024, 14, 2401414 CrossRef CAS.
- D. Meggiolaro, S. G. Motti, E. Mosconi, A. J. Barker, J. Ball, C. Andrea Riccardo Perini, F. Deschler, A. Petrozza and F. De Angelis, Energy Environ. Sci., 2018, 11, 702–713 RSC.
- G.-W. Kim and A. Petrozza, Adv. Energy Mater., 2020, 10, 2001959 CrossRef CAS.
- H. Baishya, R. D. Adhikari, M. J. Patel, D. Yadav, T. Sarmah, M. Alam, M. Kalita and P. K. Iyer, J. Energy Chem., 2024, 94, 217–253 CrossRef CAS.
- R. Lindblad, D. Bi, B.-w. Park, J. Oscarsson, M. Gorgoi, H. Siegbahn, M. Odelius, E. M. J. Johansson and H. Rensmo, J. Phys. Chem. Lett., 2014, 5, 648–653 CrossRef CAS PubMed.
- R. Lindblad, N. K. Jena, B. Philippe, J. Oscarsson, D. Bi, A. Lindblad, S. Mandal, B. Pal, D. D. Sarma, O. Karis, H. Siegbahn, E. M. J. Johansson, M. Odelius and H. Rensmo, J. Phys. Chem. C, 2015, 119, 1818–1825 CrossRef CAS.
- A. L. Abdelhady, S. N. Afraj, Y. Haruta, M. M. Uddin and M. I. Saidaminov, ACS Nano, 2025, 19, 35276–35305 CrossRef CAS PubMed.
- S. Tan, I. Yavuz, M. H. Weber, T. Huang, C.-H. Chen, R. Wang, H.-C. Wang, J. H. Ko, S. Nuryyeva, J. Xue, Y. Zhao, K.-H. Wei, J.-W. Lee and Y. Yang, Joule, 2020, 4, 2426–2442 CrossRef CAS.
- J.-W. Lee, S.-G. Kim, J.-M. Yang, Y. Yang and N.-G. Park, APL Mater., 2019, 7, 041111 CrossRef.
- E. Bi, Z. Song, C. Li, Z. Wu and Y. Yan, Trends Chem., 2021, 3, 575–588 CrossRef CAS.
- S. Jung, Y. Choi, Y. Kim, Y. Jang, H. Jung, H. Jeon, J. Kim, E. Son, S. Jeong, J. M. Baik, J.-Y. Kim, J. Lu and H. Park, Nano Energy, 2025, 145, 111482 CrossRef CAS.
- D.-J. Xue, Y. Hou, S.-C. Liu, M. Wei, B. Chen, Z. Huang, Z. Li, B. Sun, A. H. Proppe, Y. Dong, M. I. Saidaminov, S. O. Kelley, J.-S. Hu and E. H. Sargent, Nat. Commun., 2020, 11, 1514 CrossRef CAS PubMed.
- J. Zhao, Y. Deng, H. Wei, X. Zheng, Z. Yu, Y. Shao, J. E. Shield and J. Huang, Sci. Adv., 2017, 3, eaao5616 CrossRef PubMed.
- Z. Song, A. Abate, S. C. Watthage, G. K. Liyanage, A. B. Phillips, U. Steiner, M. Graetzel and M. J. Heben, Adv. Energy Mater., 2016, 6, 1600846 CrossRef.
- D. Menzel, A. Al-Ashouri, A. Tejada, I. Levine, J. A. Guerra, B. Rech, S. Albrecht and L. Korte, Adv. Energy Mater., 2022, 12, 2201109 CrossRef CAS.
- F. Zu, P. Amsalem, M. Ralaiarisoa, T. Schultz, R. Schlesinger and N. Koch, ACS Appl. Mater. Interfaces, 2017, 9, 41546–41552 CrossRef CAS PubMed.
- F. Ye, S. Zhang, F. Lang, M. Raoufi, J. Liang, I. Levine, H. Hempel, D. Menzel, F. Zu, S. Albrecht, L. Korte, C. Messmer, J. Schön, S. W. Glunz, T. Unold, N. Koch, D. Neher, D. Ye, Y. Wu and M. Stolterfoht, ACS Energy Lett., 2025, 10, 2942–2951 CrossRef CAS.
- F. Ye, S. Zhang, J. Warby, J. Wu, E. Gutierrez-Partida, F. Lang, S. Shah, E. Saglamkaya, B. Sun, F. Zu, S. Shoaee, H. Wang, B. Stiller, D. Neher, W.-H. Zhu, M. Stolterfoht and Y. Wu, Nat. Commun., 2022, 13, 7454 CrossRef CAS PubMed.
- Z. Ni, H. Jiao, C. Fei, H. Gu, S. Xu, Z. Yu, G. Yang, Y. Deng, Q. Jiang, Y. Liu, Y. Yan and J. Huang, Nat. Energy, 2022, 7, 65–73 CrossRef CAS.
- N. Ahn, K. Kwak, M. S. Jang, H. Yoon, B. Y. Lee, J.-K. Lee, P. V. Pikhitsa, J. Byun and M. Choi, Nat. Commun., 2016, 7, 13422 CrossRef CAS PubMed.
- P. P. Khlyabich, B. Burkhart, A. E. Rudenko and B. C. Thompson, Polymer, 2013, 54, 5267–5298 CrossRef CAS.
- Y. Yang, W. Chen, L. Dou, W.-H. Chang, H.-S. Duan, B. Bob, G. Li and Y. Yang, Nat. Photonics, 2015, 9, 190–198 CrossRef CAS.
- W. Liu, G. Huang, C.-Y. Chen, T. Tan, H. Fuyuki, S. Hu, T. Nakamura, M. A. Truong, R. Murdey, Y. Hashikawa, Y. Murata and A. Wakamiya, Chem. Commun., 2024, 60, 2172–2175 RSC.
- M. N. Thameel, O. M. Dawood, Z. S. Hadawi and A. Y. Ali, Braz. J. Phys., 2024, 54, 76 CrossRef CAS.
- S. Engmann, A. J. Barito, E. G. Bittle, N. C. Giebink, L. J. Richter and D. J. Gundlach, Nat. Commun., 2019, 10, 227 CrossRef PubMed.
- P. Cai, X. Xu, J. Sun, J. Chen and Y. Cao, RSC Adv., 2017, 7, 20440–20450 RSC.
- W. Xu, L. J. F. Hart, B. Moss, P. Caprioglio, T. J. Macdonald, F. Furlan, J. Panidi, R. D. J. Oliver, R. A. Pacalaj, M. Heeney, N. Gasparini, H. J. Snaith, P. R. F. Barnes and J. R. Durrant, Adv. Energy Mater., 2023, 13, 2301102 CrossRef CAS.
- R. Gottesman, P. Lopez-Varo, L. Gouda, J. A. Jimenez-Tejada, J. Hu, S. Tirosh, A. Zaban and J. Bisquert, Chem, 2016, 1, 776–789 CAS.
- S. Guo, X. Sun, C. Ding, R. Huang, M. Tan, L. Zhang, Q. Luo, F. Li, J. Jin and C.-Q. Ma, Energy Technol., 2020, 8, 2000250 CrossRef CAS.
- X. Li, S. Fu, W. Zhang, S. Ke, W. Song and J. Fang, Sci. Adv., 2020, 6, eabd1580 CrossRef CAS PubMed.
- W. Chen, B. Han, Q. Hu, M. Gu, Y. Zhu, W. Yang, Y. Zhou, D. Luo, F.-Z. Liu, R. Cheng, R. Zhu, S.-P. Feng, A. B. Djurišić, T. P. Russell and Z. He, Sci. Bull., 2021, 66, 991–1002 CrossRef CAS PubMed.
- Q. Liang, K. Liu, Y. Han, H. Xia, Z. Ren, D. Li, T. Zhu, L. Cheng, Z. Wang, C. Zhu, P. W. K. Fong, J. Huang, Q. Chen, Y. Yang and G. Li, Nat. Commun., 2025, 16, 190 CrossRef PubMed.
- D. Bi, W. Tress, M. I. Dar, P. Gao, J. Luo, C. Renevier, K. Schenk, A. Abate, F. Giordano, J.-P. Correa Baena, J.-D. Decoppet, S. M. Zakeeruddin, M. K. Nazeeruddin, M. Grätzel and A. Hagfeldt, Sci. Adv., 2016, 2, e1501170 CrossRef PubMed.
- T. Liu, R. A. Scheidt, X. Zheng, S. Joy, Q. Jiang, H. R. Atapattu, M. Chen, H. Pruett, K. Zhu, J. M. Luther, M. C. Beard and K. R. Graham, Cell Rep. Phys. Sci., 2023, 4, 101650 CrossRef CAS.
- A. Zanetta, I. Bulfaro, F. Faini, M. Manzi, G. Pica, M. De Bastiani, S. Bellani, M. I. Zappia, G. Bianca, L. Gabatel, J.-K. Panda, A. E. Del Rio Castillo, M. Prato, S. Lauciello, F. Bonaccorso and G. Grancini, J. Mater. Chem. A, 2023, 11, 12866–12875 RSC.
- H. Chen, C. Liu, J. Xu, A. Maxwell, W. Zhou, Y. Yang, Q. Zhou, A. S. R. Bati, H. Wan, Z. Wang, L. Zeng, J. Wang, P. Serles, Y. Liu, S. Teale, Y. Liu, M. I. Saidaminov, M. Li, N. Rolston, S. Hoogland, T. Filleter, M. G. Kanatzidis, B. Chen, Z. Ning and E. H. Sargent, Science, 2024, 384, 189–193 CrossRef CAS PubMed.
- Fazri, T. A. Rizal, M. Amin and Hamdani, IOP Conf. Ser. Mater. Sci. Eng., 2019, 536, 012082 CrossRef CAS.
- T. Ma, Y. An, Z. Yang, Z. Ai, Y. Zhang, C. Wang and X. Li, Adv. Funct. Mater., 2023, 33, 2212596 CrossRef CAS.
- M. A. Haque, S. Kee, D. R. Villalva, W.-L. Ong and D. Baran, Adv. Sci., 2020, 7, 1903389 CrossRef CAS PubMed.
- A. Bhui, T. Ghosh, K. Pal, K. Singh Rana, K. Kundu, A. Soni and K. Biswas, Chem. Mater., 2022, 34, 3301–3310 CrossRef CAS.
- T. Haeger, R. Heiderhoff and T. Riedl, J. Mater. Chem. C, 2020, 8, 14289–14311 RSC.
- S.-N. Hong, C.-J. Yu, U.-G. Jong, S.-H. Choe and Y.-H. Kye, RSC Adv., 2021, 11, 34015–34023 RSC.
- S. R. Yeandel, M. Molinari and S. C. Parker, Nanoscale, 2018, 10, 15010–15022 RSC.
- H.-S. Kim, S. D. Kang, Y. Tang, R. Hanus and G. Jeffrey Snyder, Mater. Horiz., 2016, 3, 234–240 RSC.
- A. F. Akbulatov, V. M. Martynenko, L. A. Frolova, N. N. Dremova, I. Zhidkov, S. A. Tsarev, S. Y. Luchkin, E. Z. Kurmaev, S. M. Aldoshin, K. J. Stevenson and P. A. Troshin, Sol. Energy Mater. Sol. Cells, 2020, 213, 110559 CrossRef CAS.
- L. Ma, D. Guo, M. Li, C. Wang, Z. Zhou, X. Zhao, F. Zhang, Z. Ao and Z. Nie, Chem. Mater., 2019, 31, 8515–8522 CrossRef CAS.
- S. Kundu and T. L. Kelly, EcoMat, 2020, 2, e12025 CrossRef CAS.
- B. Brunetti, C. Cavallo, A. Ciccioli, G. Gigli and A. Latini, Sci. Rep., 2016, 6, 31896 CrossRef CAS PubMed.
- P.-K. Kung, M.-H. Li, C.-F. Lin and P. Chen, J. Mater. Chem. C, 2024, 12, 11181–11191 RSC.
- D. Zhang, D. Li, Y. Hu, A. Mei and H. Han, Commun. Mater., 2022, 3, 58 CrossRef.
- K.-K. Liu, Z.-F. Yao, D. Wang, X. Huang, Q. Li, Z. Su, Z. Zeng, F. R. Lin, C. Zhao, W. Jiang, M. Liu, D. Lei, S.-W. Tsang and A. K. Y. Jen, Joule, 2025, 9, 101955 CrossRef CAS.
- K. Schutt, M. Davis, M. Li, S. A. Johnson, D. Martinez, J. Titus, T. Leijtens, B. Martin, M. D. McGehee, S. R. Marder, N. Rolston and J. M. Luther, ACS Energy Lett., 2025, 10, 6307–6317 CrossRef CAS PubMed.
- M. De Bastiani, G. Armaroli, R. Jalmood, L. Ferlauto, X. Li, R. Tao, G. T. Harrison, M. K. Eswaran, R. Azmi, M. Babics, A. S. Subbiah, E. Aydin, T. G. Allen, C. Combe, T. Cramer, D. Baran, U. Schwingenschlögl, G. Lubineau, D. Cavalcoli and S. De Wolf, ACS Energy Lett., 2022, 7, 827–833 CrossRef CAS.
- H. Chen, X. Liu, X. Liu, X. Song and W. Zhu, J. Phys. Chem. Lett., 2025, 16, 10080–10086 CrossRef CAS PubMed.
- P. Chen, X. Ma, Z. Wang, N. Yang, J. Luo, K. Chen, P. Liu, W. Xie and Q. Hu, Phys. Chem. Chem. Phys., 2024, 26, 14874–14882 RSC.
- M. Waqas, D. B. Khadka, A. H. H. Khan and Y.-C. Wang, Nanoscale, 2025, 17, 15648–15675 RSC.
- V. S. Katta, M. Waheed and J. H. Kim, Sol. RRL, 2024, 8, 2300908 CrossRef CAS.
- Y.-C. Wang, X. Li, L. Zhu, X. Liu, W. Zhang and J. Fang, Adv. Energy Mater., 2017, 7, 1701144 CrossRef.
- V. V. Ozerova, A. V. Mumyatov, A. E. Goryachev, E. A. Khakina, A. S. Peregudov, S. M. Aldoshin and P. A. Troshin, Inorganics, 2023, 11, 153 CrossRef CAS.
- L. N. Inasaridze, A. I. Shames, I. V. Martynov, B. Li, A. V. Mumyatov, D. K. Susarova, E. A. Katz and P. A. Troshin, J. Mater. Chem. A, 2017, 5, 8044–8050 RSC.
- E. M. Speller, A. J. Clarke, N. Aristidou, M. F. Wyatt, L. Francàs, G. Fish, H. Cha, H. K. H. Lee, J. Luke, A. Wadsworth, A. D. Evans, I. McCulloch, J.-S. Kim, S. A. Haque, J. R. Durrant, S. D. Dimitrov, W. C. Tsoi and Z. Li, ACS Energy Lett., 2019, 4, 846–852 CrossRef CAS PubMed.
- U. Dettinger, H.-J. Egelhaaf, C. J. Brabec, F. Latteyer, H. Peisert and T. Chassé, Chem. Mater., 2015, 27, 2299–2308 CrossRef CAS.
- Y. Zhai, Y. Chang, A. Tang and K. Lu, J. Mater. Chem. A, 2025, 13, 9589–9618 RSC.
- H. K. H. Lee, A. M. Telford, J. A. Röhr, M. F. Wyatt, B. Rice, J. Wu, A. de Castro Maciel, S. M. Tuladhar, E. Speller, J. McGettrick, J. R. Searle, S. Pont, T. Watson, T. Kirchartz, J. R. Durrant, W. C. Tsoi, J. Nelson and Z. Li, Energy Environ. Sci., 2018, 11, 417–428 RSC.
- X. Hou, A. J. Clarke, M. Azzouzi, J. Yan, F. Eisner, X. Shi, M. F. Wyatt, T. J. S. Dennis, Z. Li and J. Nelson, J. Mater. Chem. C, 2022, 10, 7875–7885 RSC.
- S. Wang, A. Wang, X. Deng, L. Xie, A. Xiao, C. Li, Y. Xiang, T. Li, L. Ding and F. Hao, J. Mater. Chem. A, 2020, 8, 12201–12225 RSC.
- Z. Yang, J. Dou, S. Kou, J. Dang, Y. Ji, G. Yang, W.-Q. Wu, D.-B. Kuang and M. Wang, Adv. Funct. Mater., 2020, 30, 1910710 CrossRef CAS.
- C. Li, X. Wang, E. Bi, F. Jiang, S. M. Park, Y. Li, L. Chen, Z. Wang, L. Zeng, H. Chen, Y. Liu, C. R. Grice, A. Abudulimu, J. Chung, Y. Xian, T. Zhu, H. Lai, B. Chen, R. J. Ellingson, F. Fu, D. S. Ginger, Z. Song, E. H. Sargent and Y. Yan, Science, 2023, 379, 690–694 CrossRef CAS PubMed.
- F. Zou, C. Duan, Z. Lin, Z. Zhang, S. Xu, C. Chen, J. Chen, J. Li, S. Zou, L. Ding, H. Luo and K. Yan, Chem. Eng. J., 2024, 491, 152118 CrossRef CAS.
- Y. Ma, J. Gong, P. Zeng and M. Liu, Nano-Micro Lett., 2023, 15, 173 CrossRef CAS PubMed.
- G. Huang, C. Wang, H. Zhang, S. Xu, Q. Xu and Y. Cui, J. Mater. Chem. A, 2018, 6, 2449–2455 RSC.
- H. Mo, D. Li, A. Sergeev, W. K. Yiu, J. Wang, G. Zhang, Z. Yuan, Y. Li, Y. He, T. Zhu, M. Yu Lam, F. J. Angus, W.-D. Li, J. Tang, K. S. Wong, G. Cooke, P. Docampo, J. Popović, G. Li and A. B. Djurišić, Adv. Funct. Mater., 2026, 36, e17140 CrossRef CAS.
- C. Wang, X. Yu, L. Fan, W. Ge, B. Fan, Q. Huang, R. Peng and B. Jin, Carbon, 2023, 215, 118398 CrossRef CAS.
- A. Maxwell, H. Chen, L. Grater, C. Li, S. Teale, J. Wang, L. Zeng, Z. Wang, S. M. Park, M. Vafaie, S. Sidhik, I. W. Metcalf, Y. Liu, A. D. Mohite, B. Chen and E. H. Sargent, ACS Energy Lett., 2024, 9, 520–527 CrossRef CAS.
- Z. Li, B. Li, X. Wu, S. A. Sheppard, S. Zhang, D. Gao, N. J. Long and Z. Zhu, Science, 2022, 376, 416–420 CrossRef CAS PubMed.
- X. Li, W. Zhang, X. Guo, C. Lu, J. Wei and J. Fang, Science, 2022, 375, 434–437 CrossRef CAS PubMed.
- X. Deng, F. Li, Q. Wang, D. Liu, F. Lin, D. Shen, D. Lei, Y.-K. Peng, Z. Zhu and A. K. Y. Jen, Matter, 2021, 4, 3301–3312 CrossRef CAS.
- J. Liu, M. De Bastiani, E. Aydin, G. T. Harrison, Y. Gao, R. R. Pradhan, M. K. Eswaran, M. Mandal, W. Yan, A. Seitkhan, M. Babics, A. S. Subbiah, E. Ugur, F. Xu, L. Xu, M. Wang, A. u. Rehman, A. Razzaq, J. Kang, R. Azmi, A. A. Said, F. H. Isikgor, T. G. Allen, D. Andrienko, U. Schwingenschlögl, F. Laquai and S. De Wolf, Science, 2022, 377, 302–306 CrossRef CAS PubMed.
- K. Artuk, D. Turkay, M. D. Mensi, J. A. Steele, D. A. Jacobs, M. Othman, X. Yu Chin, S.-J. Moon, A. N. Tiwari, A. Hessler-Wyser, Q. Jeangros, C. Ballif and C. M. Wolff, Adv. Mater., 2024, 36, 2311745 CrossRef CAS PubMed.
- Y. Ouyang, L. Shi, Q. Li and J. Wang, Small Methods, 2019, 3, 1900154 CrossRef.
- J. Hidalgo, W. Kaiser, Y. An, R. Li, Z. Oh, A.-F. Castro-Méndez, D. K. LaFollette, S. Kim, B. Lai, J. Breternitz, S. Schorr, C. A. R. Perini, E. Mosconi, F. De Angelis and J.-P. Correa-Baena, J. Am. Chem. Soc., 2023, 145, 24549–24557 CAS.
- U.-G. Jong, C.-J. Yu, G.-C. Ri, A. P. McMahon, N. M. Harrison, P. R. F. Barnes and A. Walsh, J. Mater. Chem. A, 2018, 6, 1067–1074 RSC.
- Y. Zhang, J. Xi, Y. Deng, W. Liu, Z. Li, C. Liu and W. Guo, Adv. Energy Mater., 2024, 14, 2403326 CrossRef CAS.
- P. Liu, X. Li, T. Cai, W. Xing, N. Yang, H. Arandiyan, Z. Shao, S. Wang and S. Liu, Nano-Micro Lett., 2024, 17, 35 CrossRef PubMed.
- U. Gunes, F. V. Yaylali, Z. Gozukara Karabag, X.-X. Gao, O. A. Syzgantseva, A. Karabag, G. B. Yildirim, K. Tsoi, N. Shibayama, H. Kanda, A. I. Rafieh, L. Zhong, A. Züttel, P. J. Dyson, S. Yerci, M. K. Nazeeruddin and G. Gunbas, Cell Rep. Phys. Sci., 2023, 4, 101380 CrossRef CAS.
- H. Lin, C. Zhou, Y. Tian, T. Siegrist and B. Ma, ACS Energy Lett., 2018, 3, 54–62 CrossRef CAS.
- Y. Chen, X. Wang, Y. Wang, X. Liu, Y. Miao and Y. Zhao, Sci. Bull., 2023, 68, 706–712 CrossRef CAS PubMed.
- Y. Han, S. Yue and B.-B. Cui, Adv. Sci., 2021, 8, 2004805 CrossRef CAS PubMed.
- S. Sun, M. Lu, X. Gao, Z. Shi, X. Bai, W. W. Yu and Y. Zhang, Adv. Sci., 2021, 8, 2102689 CrossRef PubMed.
- L. N. Quan, M. Yuan, R. Comin, O. Voznyy, E. M. Beauregard, S. Hoogland, A. Buin, A. R. Kirmani, K. Zhao, A. Amassian, D. H. Kim and E. H. Sargent, J. Am. Chem. Soc., 2016, 138, 2649–2655 CrossRef CAS PubMed.
- B. Li, J. Deng, J. A. Smith, P. Caprioglio, K. Ji, D. Luo, J. D. McGettrick, K. D. G. I. Jayawardena, R. C. Kilbride, A. Ren, S. Hinder, J. Bi, T. Webb, I. Marko, X. Liu, Y. Xiang, J. Reding, H. Li, S. Du, D. G. Lidzey, S. D. Stranks, T. Watson, S. Sweeney, H. J. Snaith, S. R. P. Silva and W. Zhang, Adv. Energy Mater., 2022, 12, 2202868 CrossRef CAS.
- Q. Li, H. Liu and T. Zhou, Sci. China Chem., 2024, 67, 3083–3090 CrossRef CAS.
- X. Zang, S. Xiong, S. Jiang, D. Li, H. Wu, H. Ren, A. Cao, B. Li, Z. Ma, J. Chen, L. Ding, J. Tang, Z. Sun, J. Chu and Q. Bao, Adv. Mater., 2024, 36, 2309991 CrossRef CAS PubMed.
- Y. Zhang, C. Li, E. Bi, T. Wang, P. Zhang, X. Yang and H. Chen, Adv. Energy Mater., 2022, 12, 2202191 CrossRef CAS.
- Z. Li, A. Sun, Y. Zheng, R. Zhuang, X. Wu, C. Tian, C. Tang, Y. Liu, B. Ouyang, J. Du, Z. Li, J. Cai, X. Wu, J. Chen, Y. Hua and C.-C. Chen, Small Methods, 2024, 8, 2400425 CrossRef CAS PubMed.
- Y. Zhao, Y.-C. Ye, X.-M. Chen, J.-G. Wang, L.-J. Zhang, H.-W. Du, C. Zhang, J. Yang, X.-Y. Zhang, M.-L. Xu, Q.-F. Ye, X. Gao, H. Xie, J.-X. Tang and W.-D. Dou, ACS Sustain. Chem. Eng., 2025, 13, 13148–13156 CrossRef CAS.
- T. Wang, L. Bi, L. Yang, Z. Zeng, X. Ji, Z. Hu, S.-W. Tsang, H.-L. Yip, Q. Fu, A. K. Y. Jen and Y. Liu, J. Am. Chem. Soc., 2024, 146, 7555–7564 CrossRef CAS PubMed.
- X. Liu, Q. Li, Y. Zheng, D. Liu, H. Wang, D. Li, M. Song, R. Yang, J. Lin, Y. Shi, P. Wang, Y. Wu, H. G. Yang, S. Yang and Y. Hou, Adv. Funct. Mater., 2026, 36, e17301 CrossRef CAS.
- Z. Wan, R. Wei, Y. Wang, H. Zeng, H. Yin, M. Azam, J. Luo and C. Jia, Nano-Micro Lett., 2025, 18, 18 CrossRef PubMed.
- K. Choi, J. Lee, H. Choi, G.-W. Kim, H. I. Kim and T. Park, Energy Environ. Sci., 2020, 13, 5059–5067 RSC.
- S. Park, S. Y. Jeong, J. Kim, H. Lee, H. S. Kim, Y. W. Noh, Y. I. Kim, S. Cho, J. S. Kang, H. Y. Woo and M. H. Song, Energy Environ. Sci., 2024, 17, 8304–8312 RSC.
- B. Yu, J. Shi, Y. Li, S. Tan, Y. Cui, F. Meng, H. Wu, Y. Luo, D. Li and Q. Meng, Nat. Commun., 2025, 16, 3328 CrossRef CAS PubMed.
- X. Sun, C. Zhang, D. Gao, X. Yu, B. Li, X. Wu, S. Zhang, Y. He, Z. Yu, L. Qian, J. Gong, S. Li, N. Li, Z. Zhu and Z. a. Li, Angew. Chem., Int. Ed., 2025, 64, e202412819 CrossRef CAS PubMed.
- Q.-J. Shui, S. Shan, Y.-C. Zhai, S. Aoyagi, S. Izawa, M. Huda, C.-Y. Yu, L. Zuo, H. Chen, H.-S. Lin and Y. Matsuo, J. Am. Chem. Soc., 2023, 145, 27307–27315 CrossRef CAS PubMed.
- S. You, H. Zhu, Z. Shen, X. Wang, B. Shao, Q. Wang, J. Lu, Y. Yuan, B. D. Dou, E. M. Sanehira, T. Russell, A. Lorenz, Y. Dong, L. Chen, M. Casareto, N. Rolston, M. C. Beard, J. J. Berry, M. Freitag, Y. Yan, O. M. Bakr and K. Zhu, Science, 2025, 388, 964–968 CrossRef CAS PubMed.
- F. Liu, B.-W. Chen, A. Fan, Z.-C. Chen, Y. Pan, Z. Tang, L.-L. Deng, Z. Xing and S.-H. Li, Adv. Funct. Mater., 2025, 35, 2419582 CrossRef CAS.
- Y. Shao, Y. Yuan and J. Huang, Nat. Energy, 2016, 1, 15001 CrossRef CAS.
- J. Lian, B. Lu, F. Niu, P. Zeng and X. Zhan, Small Methods, 2018, 2, 1800082 CrossRef.
- X. Huang, D. Xia, Q. Xie, D. Wang, Q. Li, C. Zhao, J. Yin, F. Cao, Z. Su, Z. Zeng, W. Jiang, W. Kaminsky, K. Liu, F. R. Lin, Q. Feng, B. Wu, S.-W. Tsang, D. Lei, W. Li and A. K. Y. Jen, Nat. Commun., 2025, 16, 1626 CrossRef CAS PubMed.
- Z. Xing, S.-H. Li and S. Yang, Small Struct., 2022, 3, 2200012 CrossRef CAS.
- C.-H. Lin, L. Hu, X. Guan, J. Kim, C.-Y. Huang, J.-K. Huang, S. Singh and T. Wu, Adv. Mater., 2022, 34, 2108616 CrossRef CAS PubMed.
- Y. Li, X. He, R. Zhu, X. Chen, T. Wang, X. Pu, H. Chen, Q. Cao and X. Li, Adv. Funct. Mater., 2025, 35, 2413245 CrossRef CAS.
- S. A. L. Weber, I. M. Hermes, S.-H. Turren-Cruz, C. Gort, V. W. Bergmann, L. Gilson, A. Hagfeldt, M. Graetzel, W. Tress and R. Berger, Energy Environ. Sci., 2018, 11, 2404–2413 RSC.
- H. Ishii, K. Sugiyama, E. Ito and K. Seki, Adv. Mater., 1999, 11, 605–625 CrossRef CAS.
- Q. Chen, C. Wang, Y. Li and L. Chen, J. Am. Chem. Soc., 2020, 142, 18281–18292 CrossRef CAS PubMed.
- J. Seo, S. Park, Y. Chan Kim, N. J. Jeon, J. H. Noh, S. C. Yoon and S. I. Seok, Energy Environ. Sci., 2014, 7, 2642–2646 RSC.
- S.-G. Choi, S.-K. Jung, J.-H. Lee, J.-H. Kim, W. Zheng and J.-W. Lee, ACS Energy Lett., 2024, 9, 5360–5363 CrossRef CAS.
- C. Chen, S. Zhang, S. Wu, W. Zhang, H. Zhu, Z. Xiong, Y. Zhang and W. Chen, RSC Adv., 2017, 7, 35819–35826 RSC.
- J. Ciro, S. Mesa, J. I. Uribe, M. A. Mejía-Escobar, D. Ramirez, J. F. Montoya, R. Betancur, H.-S. Yoo, N.-G. Park and F. Jaramillo, Nanoscale, 2017, 9, 9440–9446 RSC.
- J.-H. Lee, J. Kim, G. Kim, D. Shin, S. Y. Jeong, J. Lee, S. Hong, J. W. Choi, C.-L. Lee, H. Kim, Y. Yi and K. Lee, Energy Environ. Sci., 2018, 11, 1742–1751 RSC.
- D. Wang, C. Kang, T. Ye, D. He, S. Jin, X. Zhang, X. Sun and Y. Zhang, J. Energy Chem., 2023, 82, 334–342 CrossRef CAS.
- Y. W. Noh, J. M. Ha, J. G. Son, J. Han, H. Lee, D. W. Kim, M. H. Jee, W. G. Shin, S. Cho, J. Y. Kim, M. H. Song and H. Y. Woo, Mater. Horiz., 2024, 11, 2926–2936 RSC.
- H. S. Kim, Y. Cho, H. Lee, S. Kim, E. D. Jung, Y. W. Noh, S. Park, S. Cho, B. R. Lee, C. Yang and M. H. Song, Nano Energy, 2024, 125, 109584 CrossRef CAS.
- T. Wu, P. Xu, D. Wang, X. Jiang, F. Guo, S. Gao, Z. Ge and Y. Zhang, Chem. Eng. J., 2023, 454, 140451 CrossRef CAS.
- L. Gao, H. Wang, Q. Guo, Z. Wang, F. Yuan and E. Zhou, Chem. Eng. J., 2024, 480, 148277 CrossRef CAS.
- J. Wang, J. Li, Y. Zhou, C. Yu, Y. Hua, Y. Yu, R. Li, X. Lin, R. Chen, H. Wu, H. Xia and H.-L. Wang, J. Am. Chem. Soc., 2021, 143, 7759–7768 CrossRef CAS PubMed.
- T. Imran, H. S. Aziz, T. Iftikhar, M. Ahmad, H. Xie, Z. Su, P. Yan, Z. Liu, G. Liang, W. Chen and S. Chen, Energy Environ. Sci., 2024, 17, 7234–7246 RSC.
- Y. Kato, L. K. Ono, M. V. Lee, S. Wang, S. R. Raga and Y. Qi, Adv. Mater. Interfaces, 2015, 2, 1500195 CrossRef.
- J. Li, Q. Dong, N. Li and L. Wang, Adv. Energy Mater., 2017, 7, 1602922 CrossRef.
- C. C. Boyd, R. Cheacharoen, T. Leijtens and M. D. McGehee, Chem. Rev., 2019, 119, 3418–3451 CrossRef CAS PubMed.
- S. Svanström, T. J. Jacobsson, G. Boschloo, E. M. J. Johansson, H. Rensmo and U. B. Cappel, ACS Appl. Mater. Interfaces, 2020, 12, 7212–7221 CrossRef PubMed.
- C. Gong, H. Li, H. Wang, C. Zhang, Q. Zhuang, A. Wang, Z. Xu, W. Cai, R. Li, X. Li and Z. Zang, Nat. Commun., 2024, 15, 4922 CrossRef CAS PubMed.
- F. Cheng, S. Zhan, Y. Cai, F. Cao, X. Dai, R. Xu, J. Yin, J. Li, N. Zheng and B. Wu, J. Am. Chem. Soc., 2023, 145, 20081–20087 CrossRef CAS PubMed.
- S. Cao, J. Han, X. Zhao, Y. Yu and P. Gao, Adv. Energy. Sust. Res., 2025, e202500380 Search PubMed.
- M. Kim, H. Jun, H. Lee, H. Nahdi, D. Tondelier, Y. Bonnassieux, J.-É. Bourée and B. Geffroy, Eur. J. Inorg. Chem., 2021, 2021, 4781–4789 CrossRef CAS.
- J. Ning, Y. Zhu, Z. Hu, Y. Shi, M. U. Ali, J. He, Y. He, F. Yan, S. Yang, J. Miao and H. Meng, Adv. Funct. Mater., 2020, 30, 2000837 CrossRef CAS.
- B. Orwat, Z.-E. Shi, C.-H. Ma, K. Jankowska, J. Nawrocik, A. Singh, Y.-H. Zheng, W.-C. Tu, Z. Ling, P. Dąbczyński, M. Rogala, P. Krukowski, P. J. Kowalczyk, P. Data, B. Łuszczyńska, I. Kownacki and C.-P. Chen, Small, 2025, 21, 2411623 CrossRef CAS PubMed.
- C. Wang, P. Liu, H. Ju, Q. Yuan, D. Han, Y. Wang, D.-Y. Zhou and L. Feng, ACS Appl. Mater. Interfaces, 2018, 10, 15933–15942 CrossRef CAS PubMed.
- J. Yang, Q. Cao, T. Wang, B. Yang, X. Pu, Y. Zhang, H. Chen, I. Tojiboyev, Y. Li, L. Etgar, X. Li and A. Hagfeldt, Energy Environ. Sci., 2022, 15, 2154–2163 RSC.
- A. Liu, X. Li, W. Zhang, H. Yang, X. Guo, C. Lu, H. Yuan, W. Ou-Yang and J. Fang, Adv. Funct. Mater., 2024, 34, 2307310 CrossRef CAS.
- B. Yu, K. Wang, Y. Sun and H. Yu, J. Mater. Chem. A, 2024, 12, 27577–27587 RSC.
- H. Back, G. Kim, J. Kim, J. Kong, T. K. Kim, H. Kang, H. Kim, J. Lee, S. Lee and K. Lee, Energy Environ. Sci., 2016, 9, 1258–1263 RSC.
- A. Zhu, H. Gu, W. Li, J. Guo, S. Li, G. Wang, J. Xia, C. Liang, S. Chen and G. Xing, Small, 2025, 21, e11978 CrossRef CAS PubMed.
- T. Chen, T. Shi, X. Li, J. Zheng, W. Fan, B. Ni, Y. Wang, J. Dai and Z. Xiao, Sol. RRL, 2018, 2, 1800167 CrossRef.
- M. H. Futscher and B. Ehrler, ACS Energy Lett., 2016, 1, 863–868 CrossRef CAS.
- J. Lim, N.-G. Park, S. Il Seok and M. Saliba, Energy Environ. Sci., 2024, 17, 4390–4425 RSC.
- J. Liu, Y. He, L. Ding, H. Zhang, Q. Li, L. Jia, J. Yu, T. W. Lau, M. Li, Y. Qin, X. Gu, F. Zhang, Q. Li, Y. Yang, S. Zhao, X. Wu, J. Liu, T. Liu, Y. Gao, Y. Wang, X. Dong, H. Chen, P. Li, T. Zhou, M. Yang, X. Ru, F. Peng, S. Yin, M. Qu, D. Zhao, Z. Zhao, M. Li, P. Guo, H. Yan, C. Xiao, P. Xiao, J. Yin, X. Zhang, Z. Li, B. He and X. Xu, Nature, 2024, 635, 596–603 CrossRef CAS PubMed.
- X. Luo, H. Luo, H. Li, R. Xia, X. Zheng, Z. Huang, Z. Liu, H. Gao, X. Zhang, S. Li, Z. Feng, Y. Chen and H. Tan, Adv. Mater., 2023, 35, 2207883 CrossRef CAS PubMed.
- S. Mariotti, E. Köhnen, F. Scheler, K. Sveinbjörnsson, L. Zimmermann, M. Piot, F. Yang, B. Li, J. Warby, A. Musiienko, D. Menzel, F. Lang, S. Keßler, I. Levine, D. Mantione, A. Al-Ashouri, M. S. Härtel, K. Xu, A. Cruz, J. Kurpiers, P. Wagner, H. Köbler, J. Li, A. Magomedov, D. Mecerreyes, E. Unger, A. Abate, M. Stolterfoht, B. Stannowski, R. Schlatmann, L. Korte and S. Albrecht, Science, 2023, 381, 63–69 CrossRef CAS PubMed.
- E. T. Hoke, D. J. Slotcavage, E. R. Dohner, A. R. Bowring, H. I. Karunadasa and M. D. McGehee, Chem. Sci., 2015, 6, 613–617 RSC.
- T. Huang, S. Tan, S. Nuryyeva, I. Yavuz, F. Babbe, Y. Zhao, M. Abdelsamie, M. H. Weber, R. Wang, K. N. Houk, C. M. Sutter-Fella and Y. Yang, Sci. Adv., 2021, 7, eabj1799 CrossRef CAS PubMed.
- J. Zheng, D. Ning, Y. Li, Y. Wang, Q. Ma, L. Liu, P. Yang, W. He, H. Yang, W. Duan, H. Zhu, S. Chen, A. Lambertz, K. Ding and Y. Mai, Cell Rep. Phys. Sci., 2024, 5, 102114 CrossRef CAS.
- Y. An, N. Zhang, Q. Liu, W. Jiang, G. Du, D. Chen, M. Liu, X. Huang, T. Lei, Q. Qiu, F. R. Lin, X. C. Zeng, A. K. Y. Jen and H.-L. Yip, Nat. Commun., 2025, 16, 2759 CrossRef CAS PubMed.
- Y. Son, J. Lim, A. K. Le, B.-S. Kim, S. Song and H. Kim, Mater. Chem. Front., 2026, 10, 21–51 RSC.
- P. Papet, O. Nichiporuk, A. Kaminski, Y. Rozier, J. Kraiem, J. F. Lelievre, A. Chaumartin, A. Fave and M. Lemiti, Sol. Energy Mater. Sol. Cells, 2006, 90, 2319–2328 CrossRef CAS.
- W. Liang, T. Kho, J. Tong, P. Narangari, S. Armand, M. Ernst, D. Walter, S. Surve, M. Stocks, A. Blakers and K. C. Fong, Sol. Energy Mater. Sol. Cells, 2021, 222, 110909 CrossRef CAS.
- B. Chen, Z. J. Yu, S. Manzoor, S. Wang, W. Weigand, Z. Yu, G. Yang, Z. Ni, X. Dai, Z. C. Holman and J. Huang, Joule, 2020, 4, 850–864 CrossRef CAS.
- Y. Hou, E. Aydin, M. De Bastiani, C. Xiao, F. H. Isikgor, D.-J. Xue, B. Chen, H. Chen, B. Bahrami, A. H. Chowdhury, A. Johnston, S.-W. Baek, Z. Huang, M. Wei, Y. Dong, J. Troughton, R. Jalmood, A. J. Mirabelli, T. G. Allen, E. Van Kerschaver, M. I. Saidaminov, D. Baran, Q. Qiao, K. Zhu, S. De Wolf and E. H. Sargent, Science, 2020, 367, 1135–1140 CrossRef CAS PubMed.
- J. Liu, E. Aydin, J. Yin, M. De Bastiani, F. H. Isikgor, A. U. Rehman, E. Yengel, E. Ugur, G. T. Harrison, M. Wang, Y. Gao, J. I. Khan, M. Babics, T. G. Allen, A. S. Subbiah, K. Zhu, X. Zheng, W. Yan, F. Xu, M. F. Salvador, O. M. Bakr, T. D. Anthopoulos, M. Lanza, O. F. Mohammed, F. Laquai and S. De Wolf, Joule, 2021, 5, 3169–3186 CrossRef CAS.
- Z. Ying, X. Guo, H. Du, X. Li, M. Zhang, Y. Zeng, X. Yang and J. Ye, ACS Energy Lett., 2024, 9, 4018–4023 CrossRef CAS.
- N. Liu, G. Zhang, M. Wei, L. Yang, H. Gu, L. Zeng, X. Zhang, Y. Geng, Y. Zhu, C. Shen, Y. Wu, T. Li, W. Wang, X. Li, K. Qiu, P. Wei, G. Yang, J. Huang and B. Chen, Nat. Commun., 2025, 16, 9435 CrossRef CAS PubMed.
- D. Zhang, T. Wu, B. Li, D. Ding, R. Li, J. Wei, H. Zhang, C. Kan, Y. Yao, P. Hang, Z. Yu, Z. Ni, K. Qiu, D. Yang and X. Yu, Nat. Commun., 2025, 16, 7331 CrossRef CAS PubMed.
- S. Y. Kim, M. Y. Woo, M. J. Jeong, S. W. Jeon, J. W. Ahn, J. H. Park, C. Y. Kim, D. H. Kim, O. J. Oh, G. Yu, S. Lee, C. Kim, D. H. Kim and J. H. Noh, Adv. Energy Mater., 2024, 14, 2402433 CrossRef CAS.
- S. Chapagain, P. S. Chandrasekhar, D. McGott, R. C. Bramante, M. F. A. M. van Hest, M. O. Reese, T. Druffel and C. A. Grapperhaus, ACS Appl. Energy Mater., 2021, 4, 10477–10483 CrossRef CAS.
- H. Cui, G. Li, S. Zhou, C. Wang, X. Yang, S. Liao, G. Chen, S. Du, F. Ye, K. Dong, S. Wang, G. Li, H.-h. Fu, G. Yang, W. Ke and G. Fang, Adv. Funct. Mater., 2025, e22926 Search PubMed.
- M. R. S. Poma, Y. Zhang, M. Li, K. Mao, R. A. DeCrescent, S. Barlow, N. Rolston, S. R. Marder and M. D. McGehee, EES Sol., 2026, 2, 330–337 RSC.
- A. J. Bett, K. M. Winkler, M. Bivour, L. Cojocaru, Ö. Ş. Kabakli, P. S. C. Schulze, G. Siefer, L. Tutsch, M. Hermle, S. W. Glunz and J. C. Goldschmidt, ACS Appl. Mater. Interfaces, 2019, 11, 45796–45804 CrossRef CAS PubMed.
- A. u. Rehman, E. P. Van Kerschaver, E. Aydin, W. Raja, T. G. Allen and S. De Wolf, Prog. Photovoltaics Res. Appl., 2023, 31, 429–442 CrossRef CAS.
- Q. Yang, W. Duan, A. Eberst, B. Klingebiel, Y. Wang, A. Kulkarni, A. Lambertz, K. Bittkau, Y. Zhang, S. Vitusevich, U. Rau, T. Kirchartz and K. Ding, J. Mater. Chem. A, 2024, 12, 14816–14827 RSC.
- M. Härtel, B. Li, S. Mariotti, P. Wagner, F. Ruske, S. Albrecht and B. Szyszka, Sol. Energy Mater. Sol. Cells, 2023, 252, 112180 CrossRef.
- H. Bristow, X. Li, M. Babics, S. Kosar, A. R. Pininti, S. Zhang, B. Vishal, S. Sarwade, A. Razzaq, A. A. Said, G. Lubineau and S. De Wolf, Sol. RRL, 2024, 8, 2400289 CrossRef CAS.
- K. A. Bush, C. D. Bailie, Y. Chen, A. R. Bowring, W. Wang, W. Ma, T. Leijtens, F. Moghadam and M. D. McGehee, Adv. Mater., 2016, 28, 3937–3943 CrossRef CAS PubMed.
- E. Magliano, F. Di Giacomo, H. R. Sathy, S. M. Pourmotlagh, G. Giliberti, D. Becerril Rodriguez, G. Ammirati, P. Mariani, F. Zarotti, F. Matteocci, M. Luce, I. Usatii, E. Bobeico, M. Della Noce, A. Cricenti, F. Cappelluti, L. V. Mercaldo, P. Delli Veneri and A. Di Carlo, ACS Appl. Mater. Interfaces, 2025, 17, 17599–17610 CrossRef CAS PubMed.
- H. H. Park, J. Kim, G. Kim, H. Jung, S. Kim, C. S. Moon, S. J. Lee, S. S. Shin, X. Hao, J. S. Yun, M. A. Green, A. W. Y. Ho-Baillie, N. J. Jeon, T.-Y. Yang and J. Seo, Small Methods, 2020, 4, 2000074 CrossRef CAS.
- D. Song, L. Y. Hsu, C.-M. Tseng and E. W.-G. Diau, Mater. Adv., 2021, 2, 754–759 RSC.
- F. Yousuf, M.-X. Jiang, C.-F. Lin, M.-H. Li, C.-W. Chu, T.-F. Guo and P. Chen, EES Sol., 2025, 1, 796–809 RSC.
- E. Aydin, C. Altinkaya, Y. Smirnov, M. A. Yaqin, K. P. S. Zanoni, A. Paliwal, Y. Firdaus, T. G. Allen, T. D. Anthopoulos, H. J. Bolink, M. Morales-Masis and S. De Wolf, Matter, 2021, 4, 3549–3584 CrossRef CAS.
- Y. Yao, S. Li, H. Xu, Z. Gao, J. Ren, Y. Cui, L. Cai, F. Zhu and Y. Hao, Mater. Today Energy, 2021, 21, 100833 CrossRef CAS.
- C. Weiran, L. Jian, C. Hongzheng and X. Jiangeng, J. Photonics Energy, 2014, 4, 040990 CrossRef.
- G. Huseynova, J.-H. Lee, Y. H. Kim and J. Lee, Adv. Opt. Mater., 2021, 9, 2002040 CrossRef CAS.
- J. Gassmann, J. Brötz and A. Klein, Appl. Surf. Sci., 2012, 258, 3913–3919 CrossRef CAS.
- S. H. Reddy, F. Di Giacomo, F. Matteocci, L. A. Castriotta and A. Di Carlo, ACS Appl. Mater. Interfaces, 2022, 14, 51438–51448 CrossRef CAS PubMed.
- K. O. Sylvester-Hvid, J. Phys. Chem. B, 2006, 110, 2618–2627 CrossRef CAS PubMed.
- C. Gu, J. L. Zhang, S. Sun, X. Lian, Z. Ma, H. Mao, L. Guo, Y. Wang and W. Chen, ACS Appl. Mater. Interfaces, 2020, 12, 22327–22334 CrossRef CAS PubMed.
- T. P. I. Saragi, T. Reichert, A. Scheffler, M. Kussler and J. Salbeck, Synth. Met., 2012, 162, 1572–1576 CrossRef CAS.
- J.-H. Lee, S. Lee, J.-B. Kim, J. Jang and J.-J. Kim, J. Mater. Chem., 2012, 22, 15262–15266 RSC.
- B. Yu, F. Tang, Y. Yang, J. Huang, S. Wu, F. Lu, W. Duan, A. Lambertz, K. Ding and Y. Mai, Adv. Mater., 2023, 35, 2202447 CrossRef CAS PubMed.
- K. O. Brinkmann, T. Gahlmann and T. Riedl, Sol. RRL, 2020, 4, 1900332 Search PubMed.
- M. Kedia, C. Das, M. Kot, Y. Yalcinkaya, W. Zuo, K. Tabah Tanko, P. Matvija, M. Ezquer, I. Cornago, W. Hempel, F. Kauffmann, P. Plate, M. Lira-Cantu, S. A. L. Weber and M. Saliba, Energy Environ. Sci., 2025, 18, 5250–5263 Search PubMed.
- X. Li, Z. Ying, X. Wang, Y. Zeng, X. Yang and J. Ye, Inf. Funct. Mater., 2024, 1, 160–180 Search PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |