Open Access Article
Open Access ArticleTris((4-BMes2)phenyl)methanide: a carbanion with a delocalised triple quinoidal structure
Yufeng Zhang abc,
Johannes Krebsb,
Alexandra Friedrich
abc,
Johannes Krebsb,
Alexandra Friedrich b,
Shigehiro Yamaguchi
b,
Shigehiro Yamaguchi c,
Ivo Krummenacher
c,
Ivo Krummenacher b,
Holger Braunschweig
b,
Holger Braunschweig b,
Todd B. Marder
b,
Todd B. Marder *b and
Lei Ji
*b and
Lei Ji *a
*a
aState Key Laboratory of Flexible Electronics (LOFE) & Institute of Flexible Electronics (IFE), Northwestern Polytechnical University, 127 West Youyi Road, Xi'an, 710072, China. E-mail: iamlji@nwpu.edu.cn
bInstitute for Inorganic Chemistry and Institute for Sustainable Chemistry & Catalysis with Boron, Julius-Maximilians-Universität Würzburg, Am Hubland, 97074 Würzburg, Germany. E-mail: todd.marder@uni-wuerzburg.de
cDepartment of Chemistry, Graduate School of Science, Integrated Research Consortium on Chemical Sciences (IRCCS) and Institute of Transformative Bio-Molecules (WPI-ITbM), Nagoya University, Furo Chikusa, Nagoya 464-8601, Japan
First published on 21st March 2026
Abstract
Herein, we report that incorporation of strongly electron-accepting BMes2 (Mes = 2,4,6-trimethylphenyl) groups at the three para-positions of a trigonal [CPh3]− framework leads to extensive delocalisation of the negative charge over the three aryl branches and onto the boron centres. Single-crystal structure analysis reveals that all three branches of CB-Mes-1 exhibit short C(center)–C(phenylene) and C(phenylene)–B bonds, and significant C–C bond length alternation within the central phenylene ring, indicative of a unique triple quinoidal structure in a single system. The HOMO of CB-Mes-1 is delocalised over the whole molecule, further indicating its quinoidal structure. Upon deprotonation of the neutral HC[(C6H4)-4-B(Mes)2]3 precursor (CB-Mes), the resulting CB-Mes-1 shows a 439 nm (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1) red shift in its absorption from 316 nm in the UV to 755 nm in the NIR. Compared with trigonal N-centred analogues and [CPh3]−, CB-Mes-1 displays a significantly raised HOMO energy level, accompanied by a dramatic red shift of the maximum absorption band with a large oscillator strength.
400 cm−1) red shift in its absorption from 316 nm in the UV to 755 nm in the NIR. Compared with trigonal N-centred analogues and [CPh3]−, CB-Mes-1 displays a significantly raised HOMO energy level, accompanied by a dramatic red shift of the maximum absorption band with a large oscillator strength.
Introduction
Unveiling electron delocalisation is fundamental to understanding the structure, reactivity, and properties of organic compounds.1–13 Trimethylenemethane (TMM), first recognised by Moffitt and Coulson in 1948, is the most typical representative of trigonal systems.14 It features high symmetry and reactivity, along with a triplet open-shell diradical ground state.15 Thus, TMM is a representative non-Kekulé system characterised by the presence of at least two atoms not involved in π-bonding, resulting in four unpaired π electrons that are localised rather than delocalised over the entire molecule (Fig. 1a).16 Shiotani et al. reported that the TMM radical cation shows an apparent D3h structure. Nevertheless, this is a result of intramolecular dynamics among the three energetically equivalent C2v structures, and its unpaired electron primarily resides in the π orbitals of the two equivalent terminal carbon atoms.17 This finding highlights the fact that high molecular symmetry does not necessarily ensure uniform electron delocalisation.Compared to TMM, the trigonal triphenylmethyl [CPh3] cation, radical, and anion systems (A–C, respectively, Fig. 1b) are more stable owing to the resonance stabilisation arising from charge delocalisation over the phenyl rings, and offer more peripheral sites for functionalisation.18,19 In the unsubstituted CPh3 systems, the distances from the central carbon atom to its three neighbouring carbon atoms in these molecules exhibit appreciable double-bond character.20–22 For [CPh3]− (C), as it interacts with metal cations in most cases, the central carbon atom adopts sp2 hybridisation and exhibits delocalisation of the negative charge over the phenyl rings.23–25 However, the “free” [CPh3]− ([CPh3−][Me4N+]) exhibits a distinct geometry in the solid state; one phenyl ring is nearly coplanar with the central carbon framework (11.5°), one falls within the expected range (26.1°), and the third is much more twisted with respect to the central plane (61.1°). As a result, the co-planar ring displays a large extent of charge delocalisation, whereas charge delocalisation is nearly shut off for the highly twisted phenyl ring.26 In terms of the functionalised trigonal [CPh3]+ trityl system, three electron-donating dimethylamino groups at the para-positions of the phenyl rings in A give rise to a triply quinoidal structure, in which the C–N bond lengths are 1.364 Å (D).27 The central carbon atom of CPh3 systems can be replaced by a boron atom and the aryl rings modified with electron donors. For example, in the trigonal carbazolyl-substituted triarylborane E, the ground-state dipole moment of E is 1.7 D, and it exhibits a symmetry lower than D3 in the ground state, as two branches of the molecule contribute only marginally to the overall dipole moment.28 This demonstrates that the electron-donating influence of the carbazolyl substituents does not distribute evenly over the three aryl moieties in this case. The strong electron-donating O− group at the para-position of the phenyl ring attached to the boron centre in F, leads to a quinoidal structure, as observed in a few other cases.29 Likewise, the carbanion [Mes2BCH2]− shows B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond character (Mes = 2,4,6-trimethylphenyl).22 The NPh3 core is isoelectronic with [CPh3]− and, with three BAr2 (Ar = –Mes (G), –FMes, and –C6F5) groups as strong acceptors, those compounds exhibit delocalised HOMOs.30,31
C double bond character (Mes = 2,4,6-trimethylphenyl).22 The NPh3 core is isoelectronic with [CPh3]− and, with three BAr2 (Ar = –Mes (G), –FMes, and –C6F5) groups as strong acceptors, those compounds exhibit delocalised HOMOs.30,31
However, no study has clarified how the central negative charge in [CPh3]− responds to a simultaneous reduction in the electron density of all three aryl substituents (Fig. 1c). Herein, we installed three strongly π-accepting BMes2 groups32–37 at the para-positions of the phenyl rings in the trigonal [CPh3]− framework via synthesis of the neutral compound HC[1,4-C6H4B(Mes2)]3, CB-Mes, and, following deprotonation, its corresponding carbanion CB-Mes-1 (Fig. 1c). Structural and electronic properties were elucidated by single-crystal X-ray diffraction, photophysical measurements, and DFT calculations. The results demonstrate that the pz-electrons on the central carbon atom are fully delocalised over the entire molecule, giving rise to a triple quinoidal resonance structure.
Results and discussion
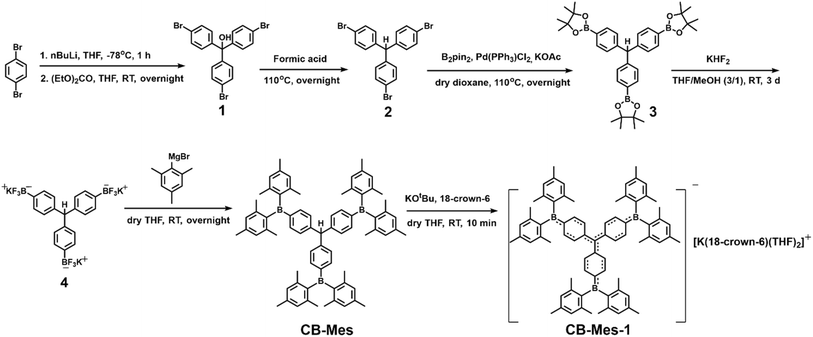
The synthetic routes to CB-Mes and CB-Mes-1 are shown in Scheme 1. Compounds 1–3 were synthesised according to literature procedures.38,39 Compound 3 was transformed into its potassium trifluoroborate salt 4 in 94% yield using KHF2 under an ambient atmosphere in a THF/methanol mixture. It was then reacted with the Grignard reagent MesMgBr to give the neutral CB-Mes compound in 14% yield. Owing to the strong electron accepting ability of the BMes2 moieties, the central carbon atom was easily deprotonated by treatment with KOtBu in THF, affording the corresponding carbanion (CB-Mes-1). This process was monitored by in situ 1H NMR spectroscopy (Fig. S1). The 1H NMR spectra of CB-Mes and CB-Mes-1 reveal two doublets and one singlet in the aromatic region with an intensity ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, consistent with C3 symmetry in solution. Interestingly, all proton signals of CB-Mes-1 shift downfield compared to those of CB-Mes. The upfield shift of the 11B NMR signal of CB-Mes-1 (δ = 66.1 ppm) relative to that of CB-Mes (δ = 73.3 ppm) reveals an increase in the electron density at the three boron centres, indicating the possibility of a triple quinoidal resonance structure.
2, consistent with C3 symmetry in solution. Interestingly, all proton signals of CB-Mes-1 shift downfield compared to those of CB-Mes. The upfield shift of the 11B NMR signal of CB-Mes-1 (δ = 66.1 ppm) relative to that of CB-Mes (δ = 73.3 ppm) reveals an increase in the electron density at the three boron centres, indicating the possibility of a triple quinoidal resonance structure.
Single crystals suitable for X-ray diffraction analysis were obtained by slow evaporation of a THF solution of CB-Mes and by diffusion of pentane into a THF solution of CB-Mes-1, respectively. Their structures are shown in Fig. 2, and selected bond lengths and angles are given in Table 1. CB-Mes and CB-Mes-1 adopt familiar propeller-like geometries, and crystallise in the monoclinic Cc and triclinic P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space groups, respectively. The torsion angles between the planes of the phenylene rings describing their relative orientations in CB-Mes are 80.6(1)°, 88.3(1)°, and 83.5(1)°, which significantly decrease in CB-Mes-1 to 52.5(1)°, 50.8(1)°, and 45.6(1)° (Table 1). In addition, a much smaller deviation between the central carbon atom (C1) and the plane defined by C2, C26, and C50 was observed in CB-Mes-1 (0.030(3) Å), compared to CB-Mes (0.451(4) Å). The average C(central)–C(phenylene) bond length (a) shortens from 1.522(3) Å (CB-Mes) to 1.453(2) Å (CB-Mes-1), and the sum of the C–C1–C angle increases from 334.8(2)° (CB-Mes) to 359.9(2)° (CB-Mes-1). These results indicate that the central carbon atom is sp3-hybridised in CB-Mes, but sp2-hybridized in CB-Mes-1, as reported for unsubstituted [CPh3]−.38 The average C(phenylene)–B bond appears to be slightly shortened from 1.553(6) Å in CB-Mes to 1.545(3) Å in CB-Mes-1, although the two values are within 3 esd's of one another. This value is slightly larger than that of linear boron-containing anions because, in CB-Mes-1, the electrons are distributed over the three BMes2 groups.29,40 This C(phenylene)–B bond is also longer than those in the one-electron-reduced 2-(BMes2)pyrene (1.514(6) Å) and the two-electron-reduced 2,7-bis(BMes2)pyrene (1.510(3) Å), in which the negative charge is predominantly localised on the boron centres, but is comparable to those observed in the one-electron-reduced species of the latter (1.547(6) Å) and in 1,4-bis(BMes2)phenylene (1.532(4) Å).2 Compared to CB-Mes, the mean bond lengths b and d (Table 1; for labelling, see Fig. 2a) in CB-Mes-1 become longer, whereas the average length of the c bonds decreases. Considering the short C(central)–C(phenylene) and C(phenylene)–B bonds and the significant C–C bond length alternation within the three phenyl rings, CB-Mes-1 could be described as having a triple quinoidal structure. The optimised structures of CB-Mes and CB-Mes-1 obtained from density functional theory (DFT) calculation show similar characteristics to those observed in their single crystal structures (Table 1).
space groups, respectively. The torsion angles between the planes of the phenylene rings describing their relative orientations in CB-Mes are 80.6(1)°, 88.3(1)°, and 83.5(1)°, which significantly decrease in CB-Mes-1 to 52.5(1)°, 50.8(1)°, and 45.6(1)° (Table 1). In addition, a much smaller deviation between the central carbon atom (C1) and the plane defined by C2, C26, and C50 was observed in CB-Mes-1 (0.030(3) Å), compared to CB-Mes (0.451(4) Å). The average C(central)–C(phenylene) bond length (a) shortens from 1.522(3) Å (CB-Mes) to 1.453(2) Å (CB-Mes-1), and the sum of the C–C1–C angle increases from 334.8(2)° (CB-Mes) to 359.9(2)° (CB-Mes-1). These results indicate that the central carbon atom is sp3-hybridised in CB-Mes, but sp2-hybridized in CB-Mes-1, as reported for unsubstituted [CPh3]−.38 The average C(phenylene)–B bond appears to be slightly shortened from 1.553(6) Å in CB-Mes to 1.545(3) Å in CB-Mes-1, although the two values are within 3 esd's of one another. This value is slightly larger than that of linear boron-containing anions because, in CB-Mes-1, the electrons are distributed over the three BMes2 groups.29,40 This C(phenylene)–B bond is also longer than those in the one-electron-reduced 2-(BMes2)pyrene (1.514(6) Å) and the two-electron-reduced 2,7-bis(BMes2)pyrene (1.510(3) Å), in which the negative charge is predominantly localised on the boron centres, but is comparable to those observed in the one-electron-reduced species of the latter (1.547(6) Å) and in 1,4-bis(BMes2)phenylene (1.532(4) Å).2 Compared to CB-Mes, the mean bond lengths b and d (Table 1; for labelling, see Fig. 2a) in CB-Mes-1 become longer, whereas the average length of the c bonds decreases. Considering the short C(central)–C(phenylene) and C(phenylene)–B bonds and the significant C–C bond length alternation within the three phenyl rings, CB-Mes-1 could be described as having a triple quinoidal structure. The optimised structures of CB-Mes and CB-Mes-1 obtained from density functional theory (DFT) calculation show similar characteristics to those observed in their single crystal structures (Table 1).
| Compound | CB-Mes | CB-Mes-1 |
|---|---|---|
a Esd's of the mean values were calculated from the esd's of the individual values using the formula 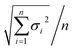 . Values obtained from DFT calculations (calc.) employed model compounds CB-Mes′ and CB-Mes-1′ in which the Mes groups were replaced with phenyl groups. Bond types a, b, c, d, and e are shown in Fig. 2a.b Values are given for the major part (68% occupancy) of the disorder. . Values obtained from DFT calculations (calc.) employed model compounds CB-Mes′ and CB-Mes-1′ in which the Mes groups were replaced with phenyl groups. Bond types a, b, c, d, and e are shown in Fig. 2a.b Values are given for the major part (68% occupancy) of the disorder. |
||
| C1–C2 | 1.518(5) | 1.446(4) |
| C1–C26 | 1.527(5) | 1.449(4) |
| C1–C50 | 1.521(4) | 1.463(4) |
| Mean bond length a | 1.522(3) | 1.453(2) |
| C–C (calc.) | 1.531 | 1.449 |
| C2–C3 | 1.395(5) | 1.431(4) |
| C26–C27 | 1.380(5) | 1.409(4) |
| C50–C51 | 1.397(5) | 1.417(4) |
| C7–C2 | 1.395(5) | 1.411(4) |
| C31–C26 | 1.400(5) | 1.423(4) |
| C55–C50 | 1.386(5) | 1.409(4) |
| Mean bond length b | 1.392(2) | 1.417(2) |
| C–C (calc.) | 1.402 | 1.420 |
| C3–C4 | 1.377(6) | 1.370(4) |
| C27–C28 | 1.397(6) | 1.376(4) |
| C51–C52 | 1.377(5) | 1.370(4) |
| C6–C7 | 1.388(5) | 1.366(4) |
| C30–C31 | 1.375(5) | 1.374(4) |
| C54–C55 | 1.384(5) | 1.374(4) |
| Mean bond length c | 1.383(2) | 1.372(2) |
| C–C (calc.) | 1.393 | 1.386 |
| C4–C5 | 1.411(5) | 1.412(4) |
| C28–C29 | 1.379(6) | 1.419(4) |
| C52–C53 | 1.409(5) | 1.402(4) |
| C5–C6 | 1.410(5) | 1.410(4) |
| C29–C30 | 1.413(6) | 1.404(5) |
| C53–C54 | 1.403(5) | 1.414(4) |
| Mean bond length d | 1.404(2) | 1.410(2) |
| C–C (calc.) | 1.411 | 1.413 |
| B1–C5 | 1.555(6) | 1.547(5) |
| B2–C29 | 1.544(17)b | 1.545(5) |
| B3–C53 | 1.561(5) | 1.542(5) |
| Mean bond length e | 1.553(6) | 1.545(3) |
| B–C (calc.) | 1.567 | 1.535 |
| C7–C2–C1–C26 | 88.0(4) | 27.1(5) |
| C7–C2–C1–C26 (calc.) | 80.4 | 30.6 |
| C31–C26–C1–C50 | 72.4(4) | 28.4(4) |
| C31–C26–C1–C50 (calc.) | 80.7 | 30.7 |
| C55–C50–C1–C2 | 81.8(4) | 27.3(5) |
| C55–C50–C1–C2 (calc.) | 81.2 | 30.5 |
| C2–C1–C26 | 112.2(3) | 120.4(3) |
| C2–C1–C26 (calc.) | 112.9 | 120.0 |
| C2–C1–C50 | 111.1(3) | 120.3(3) |
| C2–C1–C50 (calc.) | 112.9 | 120.0 |
| C26–C1–C50 | 111.5(3) | 119.2(3) |
| C26–C1–C50 (calc.) | 112.8 | 120.0 |
| sum of the C–C1–C angles | 334.8(2) | 359.9(2) |
| sum of the C–C1–C angles (calc.) | 338.6 | 360.0 |
| ∠(R1,R2) | 80.6(1) | 52.5(1) |
| ∠(R2,R3) | 88.3(1) | 50.8(1) |
| ∠(R1,R3) | 83.5(1) | 45.6(1) |
As the NPh3 core is isoelectronic with [CPh3]−, we selected model compounds N-(1,4-C6H4-BPh2)3 (H) and N-[1,4-C6H4–B(C6F5)2]3 (I)31 as representative N-containing analogues (Fig. S4) for comparison. DFT, TD-DFT, and NBO calculations on H and I were conducted at the same level as for CB-Mes′ and CB-Mes-1′, and the data are summarised in Table S3. The calculated e bond length in CB-Mes-1′ (1.535 Å) is shorter than that in H (1.564 Å). In addition, the strong electron-withdrawing effect of the –C6F5 groups leads to a shortening of the e bond from 1.564 Å in H to 1.539 Å in I, whereas the C/Ncentral–Cphenylene (a) bond lengths show only slight changes between H and I. These results indicate that the electron-donating ability of the central carbanion is significantly stronger than that of the N atom. Natural bond orbital (NBO) calculations reveal the presence of a C(phenylene)-to-B donor–acceptor interaction in CB-Mes-1′ associated with a second-order energy of 150.7 kcal mol−1. Consistent with these structural trends, the Wiberg bond index (WBI) values of the e bond in CB-Mes′ (0.908), H (0.918), and I (0.974) are all smaller than that in CB-Mes-1′ (1.021). Moreover, the WBI value of the a bond (1.867) in CB-Mes-1′ is almost twice those observed in CB-Mes′ (0.986), H (1.013), and I (1.016). Natural population analysis (NPA) calculations on CB-Mes reveal that the charge on the boron atom is 0.881 e−, which decreases to 0.802 e− upon deprotonation. However, H and I exhibit boron charges that are 0.072 e− and 0.079 e−, respectively, more positive than that of CB-Mes-1′. These indicate increased electron densities in the C(phenylene)–B bonds and strong C(phenylene)–B interactions in CB-Mes-1′.
The torsion angles between the three phenyl rings in [CPh3]− show significant differences (62.8(1)°, 51.9(1)°, and 43.8(1)°, Table S2) and, while two of them are fairly similar to that in CB-Mes-1, the third one is somewhat larger. Moreover, the bond lengths within the phenyl ring with the smallest torsion angle in [CPh3]− exhibit a pronounced difference relative to the other two rings. These observations indicate that the electrons are not equally distributed among the three branches. In contrast, the three p-BMes2 groups in CB-Mes-1 exert a remarkable influence on the molecular geometry, enhancing the uniformity of electron delocalisation. Compared to [CPh3]−, CB-Mes-1 exhibits decreased torsion angles and increased C–C bond length alternation in the three phenyl groups, attributed to its triple quinoidal resonance structure. In addition, the C(phenylene)–B bonds in CB-Mes-1 are significantly shorter than those in linear donor-π-BMes2 structures, such as (p-Me2N-phenyl)dimesitylborane, illustrating that, although each boron center carries only one-third of the negative charge, the electron-donating ability of the carbanion is stronger than that of NMe2.41
The harmonic oscillator model of aromaticity (HOMA) was calculated for CB-Mes′ and CB-Mes-1′ based on their DFT-optimised geometries to elucidate variations of aromaticity. The HOMA value of the central phenyl rings in CB-Mes-1′ (0.82) is smaller than that in CB-Mes′ (0.96), further verifying its triple quinoidal resonance structure.
Cyclic voltammetry (CV) and differential pulse voltammetry (DPV) measurements in THF (Fig. S5) reveal three reversible reductions at −2.72, −2.83, and −2.89 V (vs. Fc/Fc+ in THF), which can be ascribed to the sequential reduction of the three BMes2 groups in CB-Mes. The small redox potential gaps suggest a lack of substantial electron delocalisation in the neutral species. We also attempted to record the CV of CB-Mes-1; however, unfortunately, it is highly unstable in common electrolyte solutions such as [nBu4N]+[PF6]− and [nBu4N]+[B(C6F5)4]−.
The UV-vis absorption spectra of CB-Mes and CB-Mes-1 were measured in dry THF solutions under an argon atmosphere. CB-Mes shows two absorption bands in the ultraviolet region, with the maximum at 316 nm (ε = 5.4 × 104 M−1 cm−1), which results from the three BMes2 groups (Fig. 3a and b).42 Its fluorescence spectrum displays a single, broad emission band at 386 nm, with an absolute fluorescence quantum yield (Φ) of 0.23 in THF (Fig. 3c and Table S4). As the solvent polarity increases, the absorption of CB-Mes shows only slight changes, whereas the emission spectra exhibit a pronounced solvent-dependent behaviour, with the maximum shifting from 361 nm (hexane) to 399 nm (acetonitrile), indicating a charge transfer process in CB-Mes. For the anion CB-Mes-1, a broad absorption band is observed at 755 nm (ε = ca. 104 M−1 cm−1), which is significantly red-shifted compared to CB-Mes. This is attributed to its triple quinoidal resonance structure. Compared with calculated values for N-centred model compounds H and I, CB-Mes-1 displays a significantly raised HOMO energy level (−2.89 eV vs. −7.19 eV for H and −8.21 eV for I; see Table S3), accompanied by a dramatic red shift of the maximum absorption band with a large oscillator strength.
 | ||
| Fig. 3 (a) Absorption spectra of CB-Mes and CB-Mes-1 in THF; (b) normalised absorption and (c) emission spectra of CB-Mes in different solvents. | ||
Density functional theory (DFT) calculations on CB-Mes′ and CB-Mes-1′ were performed at the ωB97X-D/6-31G+(d,p) level (Fig. 4). For computational simplicity, the Mes groups in CB-Mes and CB-Mes-1 were replaced with phenyl groups. Due to the C3 symmetry of the optimised structures, the LUMO and LUMO + 1 of both molecules are degenerate and delocalised over the entire molecule. The HOMO of CB-Mes′ is mainly localised on the three central phenyl rings with a small contribution from the central CH group, whereas in CB-Mes-1′ there is a large contribution from the central carbon atom and delocalisation through the phenylene rings and onto the three boron atoms as well, further supporting the quinoidal structure. The HOMO of CB-Mes-1′ is similar to that of N[C6H4B(Mes)2]3.30,31,43 The positions of the absorption bands are accurately reproduced by time-dependent density functional theory (TD-DFT) calculations at the B3LYP/6-31+G(d,p) level. CB-Mes′ (312 nm) and CB-Mes-1′ (745 nm) exhibit strongly allowed transitions from S0 to the lowest degenerate levels (S1,2), which are attributed to the degenerate HOMO → LUMO and HOMO → LUMO + 1 transitions (Tables S5 and S6).
Conclusion
In summary, we have demonstrated that installing three strongly electron-accepting BMes2 substituents at the para-positions of a trigonal CPh3− framework induces a remarkable structural and electronic evolution upon deprotonation of neutral CB-Mes. The resulting anion, CB-Mes-1, exhibits nearly similar geometries in its three branches, all of them showing shortened key C–C and C–B bonds and bond length alternation in the central phenyl rings, collectively indicative of a triple quinoidal delocalised structure. DFT calculations and spectroscopic evidence support an extensive redistribution of the negative charge over the entire π-framework, in stark contrast to the localised charge observed in classical CPh3− species in the solid state. In addition, this special geometry leads to a significant red shift (18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1) in the absorption of CB-Mes-1, formed by deprotonation of CB-Mes. These findings not only fill a missing conceptual gap regarding electron distribution in functionalised trigonal carbanions, but also provide a general molecular design principle for creating electronically adaptive anionic π-architectures.
400 cm−1) in the absorption of CB-Mes-1, formed by deprotonation of CB-Mes. These findings not only fill a missing conceptual gap regarding electron distribution in functionalised trigonal carbanions, but also provide a general molecular design principle for creating electronically adaptive anionic π-architectures.
Author contributions
T. B. M., L. J., and Y. Z. contributed to the conception and design of the experiments and wrote the manuscript. Y. Z. synthesized and characterized the compounds and conducted the photophysical measurements and theoretical studies. J. K. performed the single-crystal X-ray diffraction data collection and A. F. refined the structures and analysed the resulting data. I. K. performed the electrochemistry supervised by H. B. S. Y. provided valuable advice during data analysis and manuscript preparation and T. B. M., L. J., and S. Y. supervised the project. All authors analysed and discussed the results and participated in preparing the final draft of the manuscript.Conflicts of interest
The authors declare no conflicts.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: including experimental details, synthesis, NMR spectra, UV-vis spectra, computational details, crystallographic information, and additional characterization data. See DOI: https://doi.org/10.1039/d5sc10121b.CCDC 2504657 (for CB-Mes) and 2504659 (for CB-Mes-1) contain the supplementary crystallographic data for this paper.44a,b
Acknowledgements
We are grateful for generous financial support by the Nature Science Foundation of China (Grant No. 62174137), the Nature Science Basic Research Program of Shaanxi (Grant No. 2025JC-JCQN-047), the Fundamental Research Funds for the Central Universities, and Northwestern Polytechnical University. Y. Z gratefully acknowledges the China Scholarship Council for a scholarship to spend one year at Julius-Maximilians-Universität Würzburg and the Japan Society for the Promotion of Science (JSPS) (P25341) for a postdoctoral fellowship. T. B. M. thanks the Julius-Maximilians-Universität Würzburg for support. The authors thank Prof. Dr Bernd Engels (Julius-Maximilians-Universität) for helpful discussions regarding the DFT calculation and Prof. Dr John Evans (Durham University) for helpful discussions regarding the calculation of standard uncertainty for averaged crystallographic values.References
- L. B. Ren, Y. Han, L. Y. Jiao, Y. Zou and J. S. Wu, Highly Strained, Fully π-Conjugated Porphyrin Cyclophanes: Template-Free Synthesis and Global Aromaticity, Angew. Chem., Int. Ed., 2025, 64, e202418532 Search PubMed.
- L. Ji, R. M. Edkins, A. Lorbach, I. Krummenacher, C. Brückner, A. Eichhorn, H. Braunschweig, B. Engels, P. J. Low and T. B. Marder, Electron Delocalisation in Reduced Forms of 2-(Bmes2)Pyrene and 2,7-Bis(Bmes2)Pyrene, J. Am. Chem. Soc., 2015, 137, 6750–6753 Search PubMed.
- Y. L. Wu, M. Frasconi, D. M. Gardner, P. R. McGonigal, S. T. Schneebeli, M. R. Wasielewski and J. F. Stoddart, Electron Delocalisation in a Rigid Cofacial Naphthalene-1,8:4,5-Bis(Dicarboximide) Dimer, Angew. Chem., Int. Ed., 2014, 53, 9476–9481 CrossRef CAS PubMed.
- T. Nishiuchi, S. Aibara and T. Kubo, Synthesis and Properties of a Highly Congested Tri(9-Anthryl)Methyl Radical, Angew. Chem., Int. Ed., 2018, 57, 16516–16519 Search PubMed.
- A. Das, B. J. Elvers, N. Chrysochos, S. K. I. Uddin, T. Gangber, I. Krummenacher, D. Borah, A. Mishra, M. Shanmugam, C. B. Yildiz, H. Braunschweig, C. Schulzke and A. Jana, Dianionic and Neutral Diboron-Centered Classical Diradicaloids, J. Am. Chem. Soc., 2024, 146, 9004–9011 CrossRef CAS PubMed.
- L. Wu, X. Zhang, M. Moos, I. Krummenacher, M. Dietz, A. Jayaraman, R. Bertermann, Q. Ye, M. Finze, M. Wenzel, R. Mitric, C. Lambert, H. Braunschweig and L. Ji, Full Electron Delocalisation across the Cluster in 1,12-BisBmes2-p-Carborane Radical Anion, J. Am. Chem. Soc., 2024, 146, 17956–17963 Search PubMed.
- Z. Yuan, J. C. Collings, N. J. Taylor, T. B. Marder, C. Jardin and J. F. Halet, Linear and Nonlinear Optical Properties of Three-Coordinate Organoboron Compounds, J. Solid State Chem., 2000, 154, 5–12 CrossRef CAS.
- W. Kaim and A. Schulz, p-Phenylenediboranes: Mirror Images of p-Phenylenediamines, Angew. Chem., Int. Ed., 1984, 23, 615–616 CrossRef.
- A. Schulz and W. Kaim, Bor-Organische Redoxsysteme, Chem. Ber., 1989, 122, 1863–1868 CrossRef CAS.
- J. Fiedler, S. Zalis, A. Klein, F. M. Hornung and W. Kaim, Electronic Structure of π-Conjugated Redox Systems with Borane/Borataalkene End Groups, Inorg. Chem., 1996, 35, 3039–3043 CrossRef CAS.
- S. Zalis and W. Kaim, Electronic Structures of Two Kinds of Oligoborane Radical Anions: Mixed-Valence Description of a Reduced p-Phenylenediborane and Spin Density Distributions in Distorted Octahedral Clusters [B6Hal6]˙−, Hal = Cl, Br, I, Main Group Chem., 2007, 5, 267–276 CrossRef.
- W. Kaim, N. S. Hosmane, S. Záliš, J. A. Maguire and W. N. Lipscomb, Boron Atoms as Spin Carriers in Two- and Three-Dimensional Systems, Angew. Chem., Int. Ed., 2009, 48, 5082–5091 CrossRef CAS PubMed.
- A. Lichtblau, W. Kaim, A. Schulz and T. Stahl, Organoboron Substituent Effects in EPR/ENDOR Spectroscopy. Radical Ions with B–C and B–N Bonds, J. Chem. Soc., Perkin Trans. 2, 1992, 1497–1501 RSC.
- C. A. Coulson, Free Valence in Organic Reactions, J. Chim. Phys. Phys.-Chim. Biol., 1948, 45, 243–248 CrossRef CAS.
- P. Dowd, Trimethylenemethane, J. Am. Chem. Soc., 1966, 88, 2587–2589 CrossRef CAS.
- L. V. Slipchenko and A. I. Krylov, Electronic Structure of the Trimethylenemethane Diradical in Its Ground and Electronically Excited States: Bonding, Equilibrium Geometries, and Vibrational Frequencies, J. Chem. Phys., 2003, 118, 6874–6883 Search PubMed.
- K. Komaguchi, M. Shiotani and A. Lund, An ESR Study of Trimethylenemethane Radical Cation, Chem. Phys. Lett., 1997, 265, 217–223 CrossRef CAS.
- S. Baghery, M. Zarei, M. A. Zolfigol, S. Mallakpour and V. Behranvand, Application of Trityl Moieties in Chemical Processes: Part I, J. Iran. Chem. Soc., 2020, 17, 2737–2843 Search PubMed.
- B. M. Trost, [3+2] Cycloaddition Approaches to 5-Membered Rings Via Trimethylenemethane and Its Equivalents, Angew. Chem., Int. Ed., 1986, 25, 1–20 Search PubMed.
- J. S. Alexander and K. Ruhlandt-Senge, Barium Triphenylmethanide: An Examination of Anion Basicity, Angew. Chem., Int. Ed., 2001, 40, 2658–2660 CrossRef CAS PubMed.
- M. Gomberg, An Instance of Trivalent Carbon Triphenylmethyl, J. Am. Chem. Soc., 1900, 22, 757–771 CrossRef.
- H. Nishimae, H. Kurata and M. Oda, Arylbis(9-Anthryl)Methyl Cations: Highly Crowded, near Infrared Light Absorbing Hydrocarbon Cations, Angew. Chem., Int. Ed., 2004, 43, 4947–4950 Search PubMed.
- S. Harder, S. Müller and E. Hübner, Syntheses and Crystal Structures of Simple Dibenzylcalcium Complexes: Useful Reagents in the Preparation of Calcium Compounds, Organometallics, 2004, 23, 178–183 CrossRef CAS.
- M. M. Olmstead and P. P. Power, The Isolation and X-Ray Structures of Lithium Crown Ether Salts of the Free Phenyl Carbanions [CHPh2]− and [CPh3]−, J. Am. Chem. Soc., 1985, 107, 2174–2175 CrossRef CAS.
- D. E. Smiles, G. Wu and T. W. Hayton, Synthesis of Uranium-Ligand Multiple Bonds by Cleavage of a Trityl Protecting Group, J. Am. Chem. Soc., 2014, 136, 96–99 CrossRef CAS PubMed.
- S. Harder, Schlenk's Early “Free” Carbanions, Chem.–Eur. J., 2002, 8, 3229–3232 CrossRef CAS PubMed.
- S. Lovell, B. J. Marquardt and B. Kahr, Crystal Violet's Shoulder, J. Chem. Soc., Perkin Trans. 2, 1999, 2241–2247 RSC.
- R. Stahl, C. Lambert, C. Kaiser, R. Wortmann and R. Jakober, Electrochemistry and Photophysics of Donor-Substituted Triarylboranes: Symmetry Breaking in Ground and Excited State, Chem.–Eur. J., 2006, 12, 2358–2370 CrossRef CAS PubMed.
- P. Y. Feng, Y. H. Liu, T. S. Lin, S. M. Peng and C. W. Chiu, Redox Chemistry of a Hydroxyphenyl-Substituted Borane, Angew. Chem., Int. Ed., 2014, 53, 6237–6240 CrossRef CAS PubMed.
- N. S. Makarov, S. Mukhopadhyay, K. Yesudas, J.-L. Brédas, J. W. Perry, A. Pron, M. Kivala and K. Müllen, Impact of Electronic Coupling, Symmetry, and Planarization on One-and Two-Photon Properties of Triarylamines with One, Two, or Three Diarylboryl Acceptors, J. Phys. Chem. A, 2012, 116, 3781–3793 CrossRef CAS PubMed.
- R. F. Jin, X. F. Zhang, W. M. Xiao and D. M. Luo, Theoretical Investigations of the Photophysical Properties of Star-Shaped π-Conjugated Molecules with Triarylboron Unit for Organic Light-Emitting Diodes Applications, Int. J. Mol. Sci., 2017, 18, 2178 CrossRef PubMed.
- J. C. Doty, B. Babb, P. J. Grisdale, M. Glogowski and J. L. R. Williams, Boron Photochemistry IX. Synthesis and Fluorescent Properties of Dimesityl-Phenylboranes, J. Organomet. Chem., 1972, 38, 229–236 CrossRef CAS.
- C. D. Entwistle and T. B. Marder, Boron Chemistry Lights the Way: Optical Properties of Molecular and Polymeric Systems, Angew. Chem., Int. Ed., 2002, 41, 2927–2931 CrossRef CAS PubMed.
- C. D. Entwistle and T. B. Marder, Applications of Three-Coordinate Organoboron Compounds and Polymers in Optoelectronics, Chem. Mater., 2004, 16, 4574–4585 CrossRef CAS.
- L. Ji, S. Griesbeck and T. B. Marder, Recent Developments in and Perspectives on Three-Coordinate Boron Materials: A Bright Future, Chem. Sci., 2017, 8, 846–863 RSC.
- S. Y. Li, Z. B. Sun and C. H. Zhao, Charge-Transfer Emitting Triarylborane π-Electron Systems, Inorg. Chem., 2017, 56, 8705–8717 CrossRef CAS PubMed.
- A. Wakamiya and S. Yamaguchi, Designs of Functional π-Electron Materials Based on the Characteristic Features of Boron, Bull. Chem. Soc. Jpn., 2015, 88, 1357–1377 CrossRef CAS.
- G. Li, W. B. Yang, S. H. Wang, T. Liu, C. Q. Yan, G. Li, Y. Zhang, D. D. Li, X. Y. Wang, P. Hao, J. W. Li, L. J. Huo, H. Yan and B. Tang, Methane-Perylene Diimide-Based Small Molecule Acceptors for High Efficiency Non-Fullerene Organic Solar Cells, J. Mater. Chem. C, 2019, 7, 10901–10907 RSC.
- C. Kleeberg, On the Structural Diversity of K(18-Crown-6)EPh3 Complexes (E = C, Si, Ge, Sn, Pb): Synthesis, Crystal Structures and NOESY NMR Study, Dalton Trans., 2013, 42, 8276–8287 Search PubMed.
- M. M. Olmstead, P. P. Power, K. J. Weese and R. J. Doedens, Isolation and X-Ray Crystal-Structure of the Boron Methylidenide Ion [Mes2BCH2]− (Mes = 2,4,6-Me3C6H2): A Boron-Carbon Double Bonded Alkene Analog, J. Am. Chem. Soc., 1987, 109, 2541–2542 CrossRef CAS.
- Z. Yuan, C. D. Entwistle, J. C. Collings, D. Albesa-Jové, A. S. Batsanov, J. A. K. Howard, N. J. Taylor, H. M. Kaiser, D. E. Kaufmann, S. Y. Poon, W. Y. Wong, C. Jardin, S. Fathallah, A. Boucekkine, J.-F. Halet and T. B. Marder, Synthesis, Crystal Structures, Linear and Nonlinear Optical Properties, and Theoretical Studies of (p-R-Phenyl)-, (p-R-Phenylethynyl)-, and (E)-2-(p-R-Phenyl)Ethenyl Dimesitylboranes and Related Compounds, Chem.–Eur. J., 2006, 12, 2758–2771 CrossRef CAS PubMed.
- M. E. Long, Absorption and Emission of Substituted Dimesitylboranes, J. Lumin., 1978, 16, 177–189 CrossRef CAS.
- For a related study of dipolar, quadrupolar, and octupolar amino donor – B(Mes)2 acceptor systems and their one- and two-photon optical properties, see: J. C. Collings, S.-Y. Poon, C. Le Droumaguet, M. Charlot, C. Katan, L. O. Pålsson, A. Beeby, J. A. Mosely, H. M. Kaiser, D. E. Kaufmann, W.-Y. Wong, M. Blanchard-Desce and T. B. Marder, The Synthesis and One- and Two-Photon Optical Properties of Dipolar, Quadrupolar and Octupolar Donor-Acceptor Molecules Containing Dimesitylboryl Groups, Chem.–Eur. J., 2009, 15, 198–208 Search PubMed.
- (a) CCDC 2504657: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q29dj; (b) CCDC 2504659: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q29gl.
| This journal is © The Royal Society of Chemistry 2026 |