Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Formation of cyclopentanes and cyclopropanes through alkylation of benzylic anions using ethers, thioethers and alcohols as substrates under Grubbs–Stoltz (Et3SiH/KOtBu) conditions
Alexander J.
Stewart†
a,
Daniela
Dimitrova†
a,
Scott T. M.
Logan†
a,
Cassie
Pratley
 a,
Jonathan D.
Bell
a,
Katy
McGonigal
a,
Anna
Lauer
a,
Sabine
Fenner
b,
Simon M.
Nicolle
b,
Stuart G.
Leach
b and
John A.
Murphy
a,
Jonathan D.
Bell
a,
Katy
McGonigal
a,
Anna
Lauer
a,
Sabine
Fenner
b,
Simon M.
Nicolle
b,
Stuart G.
Leach
b and
John A.
Murphy
 *a
*a
aDepartment of Pure and Applied Chemistry, University of Strathclyde, 295 Cathedral Street, Glasgow G1 1XL, UK. E-mail: john.murphy@strath.ac.uk
bGSK Medicines Research Centre, Gunnels Wood Road, Stevenage, Herts SG1 2NY, UK
First published on 10th March 2026
Abstract
Reaction of diarylmethanes with the Grubbs–Stoltz reagent (KOtBu + Et3SiH) using THF as solvent led to diarylcyclopentanes through an unprecedented double-alkylation reaction, with four of the carbons of the cyclopentane coming from THF. In like manner, reaction of diarylmethanes with the same reagent in 1,4-dioxane as solvent led to double-alkylation to form diarylcyclopropanes, with two of the cyclopropane carbons coming from 1,4-dioxane. Monoalkylated substrates that were likely intermediates on the dialkylation pathway were subjected to the same conditions, leading to cyclisations to form cyclopentanes and cyclopropanes. The cyclisation chemistry also extended to formation of monoarylcyclopropanes from reaction of the corresponding benzylpotassium reagents with ethers, alcohols, sulfides, sulfoxides and sulfones.
Introduction
Alkylations of carbanions under basic conditions routinely use very reactive electrophiles such as alkyl halides or sulfonate esters. The high reactivity of these reagents makes them convenient to use, but also underpins their ability to alkylate biomolecules, leading to their classification as major safety hazards. Ethers are much less electrophilic – indeed they are usually not recognised as electrophiles in the absence of protic or Lewis acids that activate them to C–O bond cleavage. If ethers could routinely be used as alkylating agents under defined basic conditions, this would be a very useful advance. THF has been deconstructed in many ways in the literature1,2 but it has never been subject to a double nucleophilic substitution by carbanions. Substitution of a single C–O bond cleavage has been reported using conventional powerful Lewis acids.3,4 Madsen et al. showed,5 using organomagnesium compounds as both Lewis acids and nucleophiles, that allylic and benzylic Grignard reagents such as 1 open THF and 3,3-dimethyloxetane to form alcohol products e.g.2 and 3 respectively (Scheme 1A). In these cases, the Lewis acidic properties of the magnesium atom are harnessed to assist ring-opening.5 Very recently, Chiba et al. have generated MgH2in situ from magnesium halides and sodium hydride and used it to add to 1,1-diarylethenes (e.g.4) to generate benzylic organomagnesium intermediates 5 that reacted with cyclic ethers to form alcohol products 6.6 We now report activation of ethers in the presence of benzylic potassium reagents; here, THF and dioxane each act as dual electrophiles in forming cyclopentanes and cyclopropanes respectively.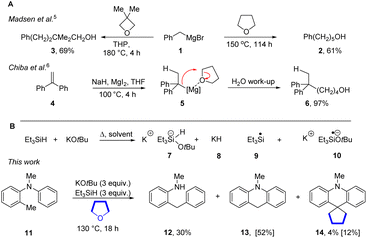 | ||
| Scheme 1 (A) Prior reactions of benzylic organomagnesium reagents; (B) our discovery of cyclopentane formation from THF (NMR yields in brackets). | ||
Results and discussion
The Grubbs–Stoltz reagent, formed by heating a mixture of KOtBu and Et3SiH,7 has been widely investigated since its introduction in 2013. This reagent is most unusual since it simultaneously generates a number of reactive intermediates, 7–10, that perform hydride transfer, hydrogen atom transfer, deprotonation, radical chemistry, silylation and electron transfer chemistry (Scheme 1B). Simultaneous exposure of substrates to such a variety of reactive entities is very unusual, and has been shown to lead to diverse and novel outcomes.7–19 In the course of our studies, we observed the rearrangement of the N-methyl-N-o-tolylaniline 11 to diarylmethane 12 and dihydroacridine 13; the mechanisms of the formation of 12 and 13 have been investigated and reported.16,17 However, in addition to these products, and relevant to this paper, cyclopentane 14 (12%) was also formed (Our reactions were routinely performed in sealed vessels).The reaction of 11 was repeated with THF-d8 as solvent and led to 14-d8 (13%) confirming the origin of the cyclopentane. In a further experiment, this time using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of THF and THF-d8, the product 14 was a mixture of d8 and d0 isotopologues, with no evidence of H/D exchange. If such exchange had been seen, it would have been evidence of THF-derived radicals playing a part in the reaction. The result instead now focused our thoughts on a double aliphatic nucleophilic substitution of THF.
1 ratio of THF and THF-d8, the product 14 was a mixture of d8 and d0 isotopologues, with no evidence of H/D exchange. If such exchange had been seen, it would have been evidence of THF-derived radicals playing a part in the reaction. The result instead now focused our thoughts on a double aliphatic nucleophilic substitution of THF.
Our working hypothesis was that dihydroacridine 13 was a likely precursor of 14. In support of this, when the dihydroacridine 13 was heated with KOtBu and Et3SiH in THF, the cyclopentane 14 (12%) was again formed (Scheme 2A). In addition, a trace of the dimer 15 was detected by GCMS, indicating a slower background radical chemistry.13–17 Abstraction of an H-atom from dihydroacridine 13e.g. by triethylsilyl radicals 9 would form a stabilised acridinyl radical 16. Such radicals could then dimerise to afford 15. Interestingly, when 2-methylTHF was used as solvent, the reaction afforded only the dimeric product 15 (64%) and when tetrahydropyran (THP) was used as solvent, no alkylation was detected and product 15 was again seen (20%). The different behaviour of 2-methylTHF was surprising.20 The inertness of THP to this reaction allowed us to use it as solvent in some later reactions.21 Before studying intermediates in this reaction, we tested dihydrobenzofuran 17 and phthalan 18 as substrates (Scheme 2B). We recognised that oxygen-containing heterocycles fused to arene rings can undergo electron transfer from 10 leading to reduction to their arene radical anions, followed by C–O bond cleavage.7 Both led to complex mixtures.
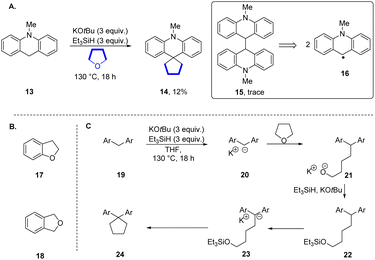 | ||
| Scheme 2 (A) N-Methyldihydroacridine 13 as substrate. (B) Dihydrobenzofuran 17 and phthalan 18. (C) Mechanistic proposal for formation of cyclopentane 24. | ||
Our proposed mechanism for cyclopentane formation is shown in Scheme 2C. Deprotonation of diarylmethane 19 would afford diarylmethyl potassium 20 which reacts with THF to afford intermediate alkoxide 21. To proceed to the cyclopentane, this alkoxide would require protection as silyl ether 22. It is known that when alkoxides are treated with triethylsilane or other silanes, silyl ethers are formed.22–24 The silyl alkyl ether component of 22 can then behave as an alkylating agent, reacting intramolecularly in this case with the anion in 23 to form the cyclopentane 24.
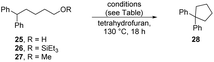
To test this proposal, alcohol 25, silyl ether 26 and methyl ether 27 were prepared and subjected to the reaction conditions (Table 1, entries 1–3). All three compounds were converted to the cyclopentane 28 (27–77% yields). The formation of cyclopentanes in this way is novel – the cyclisations of silyl ether 24 and methyl ether 27 proceed with very unusual but credible leaving groups.
| Entry | R | Solvent | Et3SiH | Base | Et3SiOtBu | 28 (yield) | 26 (yield) |
|---|---|---|---|---|---|---|---|
| a From this reaction, alcohol 25 (50%) was also isolated. | |||||||
| 1 | H | THF | 3 equiv. | KOtBu – 3 equiv. | — | 50% | — |
| 2 | SiEt3 | THF | 3 equiv. | KOtBu – 3 equiv. | — | 77% | — |
| 3 | Me | THF | 3 equiv. | KOtBu – 3 equiv. | — | 27% | — |
| 4 | H | THF | 3 equiv. | KOtBu – 4 equiv. | 3 equiv. | 17% | — |
| 5 | SiEt3 | THF | — | KOtBu – 3 equiv. | — | — | 36%a |
| 6 | H | THF | 3 equiv. | NaOtBu – 4 equiv. | — | — | 3% |
| 7 | H | THF | 3 equiv. | LiOtBu – 4 equiv. | — | — | 76% |
| 8 | H | THF | 3 equiv. | NaH – 4 equiv. | — | 26% | 36% |
| 9 | H | THF | 3 equiv. | KH – 4 equiv. | — | 67% | — |
| 10 | H | 2-MeTHF | 3 equiv. | KOtBu – 4 equiv. | — | 49% | — |
A byproduct in these reactions was Et3SiOtBu (Scheme 1B). This compound was tested for its effect on the cyclisation (entry 4). Specifically, this was to probe whether this silyl ether would provide some Lewis acid assistance to the cyclisation. However, this led to a lower yield (17%) for the cyclisation and hence does not facilitate the reaction.
Interestingly, when the silyl ether 26 was subjected to reaction with KOtBu (entry 5), but in the absence of triethylsilane, no cyclisation was observed. This could be due to either or both of two reasons: (i) the silyl ether substrate 26 could be attacked by the excess KOtBu to form Et3SiOtBu and the diphenylbutoxide anion derived from the substrate. The alkoxide would then not function as an electrophile, and no cyclisation would be seen [This would be consistent with the isolation of 25 (50%) from this reaction]. The alternative possibility was that KOtBu was not a strong enough base to deprotonate 26, but that when cyclopentanes had successfully formed in previous cases, that this had resulted from the generation of a stronger base, i.e. KH, 8, through interaction of KOtBu with Et3SiH.22–24 It has been shown that LiOtBu reacts with triethylsilane to liberate LiH25 and we have previously shown13 that inorganic potassium amides also react with triethylsilane to afford KH; this species would be a much stronger base than KOtBu and should certainly deprotonate 26.
Accordingly, the role of base was explored with alcohol 25 using NaOtBu, LiOtBu, NaH and KH (entries 6–9 respectively). NaOtBu reacted little, perhaps due to insolublilty; LiOtBu converted the alcohol 25 to its silyl ether 26. The stronger base, NaH, formed a mixture of silyl ether 26 and cyclopentane 28 while KH made a more effective conversion to the cyclopentane product. We have recently observed that potassium salts demonstrate special reactivity relative to their sodium and lithium counterparts in a number of reaction types, and in some cases, we have identified the specific interactions responsible.26,27
Finally, 2-MeTHF was studied (entry 10) as an alternative solvent for the intramolecular alkylation of alcohol 25. Having seen the dramatic differences between THF and 2-methylTHF in the reaction of dihydroacridine 13 above, we were not confident of the outcome. However, in the complex reaction seen with Grubbs–Stoltz conditions, the interplay of kinetics and thermodynamics of the different possible processes, mean that it was valuable to explore this. In effect, the reaction with 25 afforded 28 (49%), similar to the yield seen with THF as solvent (55%, entry 1). So, this shows that THF and 2-MeTHF were equally suitable solvents for reactions that involve intramolecular alkylation.
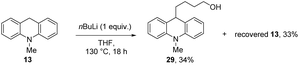
In our case, K+ or an organopotassium species would be a much milder Lewis acid than magnesium ions5,6 and our substrates show that magnesium ions are not required for these reactions. Based on these thoughts, a further experiment with base was undertaken. nBuLi was examined as base, using N-methyldihydroacridine 13 as substrate and alcohol 29 (34%) was isolated following workup; here, the reaction was likely assisted by the Lewis acidity of the lithium species (Scheme 3). Further reaction of the alkoxide was not achieved in the absence of silane.
Returning to Grubbs–Stoltz conditions (KOtBu + Et3SiH), substrates 29 and 30 were then subjected to the cyclisation reaction, leading to the cyclopentanes 14 and 31 in the yields shown in Scheme 4. We did not isolate other products from the reaction, but diaryl ethers have previously been shown to undergo cleavage of Ar–O bonds, likely resulting following electron transfer from radical anion 10.7 Electron transfer to the oxygen heterocycle of 30 should be easier than to the more electron-rich nitrogen heterocycle of 29, and this could underpin the higher yield of cyclopentane from substrate 29.
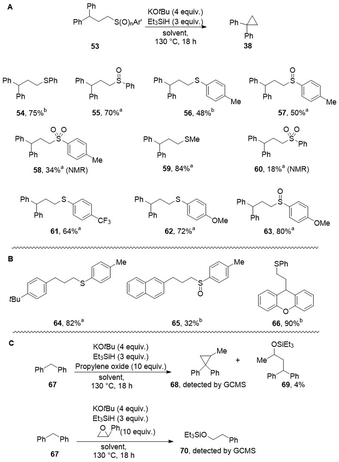
Our first encounter with cyclopropane formation arose when 1,4-dioxane was used as solvent. This converted benzylpyridines 32 and 33 to products 34 and 35 (Scheme 5A). Again, the cyclopropane formation likely arose through sequential substitution reactions on ether C–O bonds. By analogy with the THF case, our proposal was that initial alkylation had afforded a 3,3-diarylpropyl alkyl ether. This led us to select 3,3-diarylpropanols (36, R![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ar′) as substrates for cyclopropane formation, leading to 34, 35, 38 and 39 in moderate to excellent yields (Scheme 5B). Clearly, diphenylmethane anions have the right balance of nucleophilicity and basicity to succeed in substitution reactions with the ethers. To extend the scope, simpler benzyl anions were investigated; hydroxypropylbenzenes (36, R
Ar′) as substrates for cyclopropane formation, leading to 34, 35, 38 and 39 in moderate to excellent yields (Scheme 5B). Clearly, diphenylmethane anions have the right balance of nucleophilicity and basicity to succeed in substitution reactions with the ethers. To extend the scope, simpler benzyl anions were investigated; hydroxypropylbenzenes (36, R![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) H) were then chosen as substrates, affording arylcyclopropanes 41–50 in a range of yields (15–89% yield) (The simplest substrate (36, Ar = Ph, R
H) were then chosen as substrates, affording arylcyclopropanes 41–50 in a range of yields (15–89% yield) (The simplest substrate (36, Ar = Ph, R![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) H) cyclised cleanly to 40, giving an NMR yield (83%) but the product was too volatile to get an isolated yield.).
H) cyclised cleanly to 40, giving an NMR yield (83%) but the product was too volatile to get an isolated yield.).
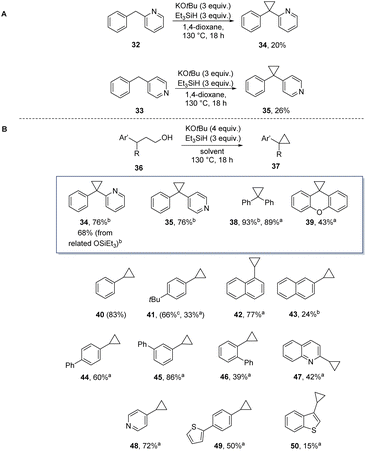 | ||
| Scheme 5 Cyclopropane formation (A) from dioxane solvent and (B) from hydroxypropyl arenes under Grubbs–Stoltz conditions. aIn THF as solvent, bin dioxane, cin THP. | ||
We had previously tested whether cyclopentanations could be achieved by simply using a strong base, KH; this was now investigated for cyclopropanations. Accordingly, substrates 51 (R = Ph, Me, Et) were treated with commercial KH, with THF as solvent, respectively affording product 38 in 49, 30 and 69% yield (Table 2, entries 1–3). Thus, KH as strong base can achieve these reactions. The yields are lower than when KH acts in conjunction with Et3SiH, as seen in comparing entries 4 and 5. Although we don’t have evidence for the cause of this at this stage, it may relate to aggregation state of KH. Chiba et al. have recently shown that the presence of additives can affect both the aggregation state and the reactivity of NaH.29
| Entry | R | Solvent | Et3SiH | Base | Temp °C | Duration | 38 (yield) |
|---|---|---|---|---|---|---|---|
| a See ref. 28 for formation of cyclopropanes through displacement of aryloxide leaving groups. | |||||||
| 1 | Me | THF | — | KH – 3 equiv. | 130 °C | 18 h | 49% |
| 2 | Et | THF | — | KH – 3 equiv. | 130 °C | 18 h | 30% |
| 3 | Ph | THF | — | KH – 3 equiv. | 130 °C | 18 h | 69%a |
| 4 | SiEt3 | THF | — | KH – 3 equiv. | 130 °C | 18 h | 34% |
| 5 | SiEt3 | THF | 3 equiv. | KH – 3 equiv. | 130 °C | 18 h | 70% |
| 6 | H | THF | 3 equiv. | KH – 4 equiv. | 130 °C | 18 h | 82% |
| 7 | H | THF | 3 equiv. | KH – 4 equiv. | 100 °C | 18 h | 67% |
| 8 | H | THF | 3 equiv. | KH – 4 equiv. | 80 °C | 18 h | 70% |
| 9 | H | THF | 3 equiv. | KH – 4 equiv. | 60 °C | 18 h | 51% |
| 10 | H | THF | 3 equiv. | KH – 4 equiv. | 80 °C | 3 h | 42% |
Entries 6–9 examine the effect of temperature on the cyclisation of 51, R![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) H. The reaction is clearly more effective at 130 °C than at lower temperatures. Entry 10 shows the outcome of the reaction at 80 °C but for a 3 hour duration (cf. entries 6 and 10).
H. The reaction is clearly more effective at 130 °C than at lower temperatures. Entry 10 shows the outcome of the reaction at 80 °C but for a 3 hour duration (cf. entries 6 and 10).
The role of Et3SiH was next examined in Table 3 (Et3SiH appeared in Table 2, entry 5, yield 70%). Replacement of Et3SiH by Me2PhSi led to a similar outcome (76% yield) in the formation of cyclopropane 38 (R![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) H). However, more bulky silanes iPr3SiH and tBuMe2SiH led to silylation of the alcohol as silyl ether 52, but not to cyclisation, likely due to steric hindrance in the cyclisation step. When tri-n-butylstannane was used instead of a silane, then only starting alcohol 51 was isolated.
H). However, more bulky silanes iPr3SiH and tBuMe2SiH led to silylation of the alcohol as silyl ether 52, but not to cyclisation, likely due to steric hindrance in the cyclisation step. When tri-n-butylstannane was used instead of a silane, then only starting alcohol 51 was isolated.
Having seen that ethers behaved favourably, thioethers were next assayed. Scheme 6 shows that cyclopropanes were again formed from sulfides, sulfoxides and sulfones. Thioethers are not viewed as electrophiles in the absence of specific activations. However, conversion of sulfides 53 to cyclopropanes 38 was also achieved. Scheme 6 shows yields (48% to 90%) of cyclopropanes derived from substrates including sulfoxides 55, 57, and 63. Sulfones 58, 60 also underwent cyclopropane formation on treatment by base in the presence of silane. The sulfones were generally less successful than the corresponding sulfides and sulfoxides, likely due to the relative ease of deprotonation α-to sulfones in competition with deprotonation of the benzylic C–H sites.
Our recent results with the Grubbs–Stoltz reagent have underlined the competition between the different types of processes that can result from intermediates that are generated, notably radical chemistry and electron transfer chemistry. Besides the formation of 15, mentioned earlier in relation to Scheme 2, the SI file exemplifies some side-reactions that can be attributed to competing processes (see pp. S45–46).
The rates of formations of cycloalkanes of different ring sizes by intramolecular displacement by nucleophiles have been studied for a number of different classes of compounds. In each case, there is a balance between enthalpic and entropic considerations. Accordingly, having witnessed formation of three-membered and five-membered ring originally arising from use of tetrahydrofuran and 1,4-dioxane as solvents, we were interested in exploring possible formation of 4-membered and 6-membered rings. In fact, these efforts led to only traces of cyclisation and the details of products are more fully discussed in the SI file (see page S57 et seq.) In these cases, the appearance of alternative products likely results from slower cyclisation kinetics compared to the 3- and 5-membered rings discussed in this paper.30,31 We also examined two epoxides as substrates, namely propylene oxide and styrene oxide, under our Grubbs–Stoltz conditions (Scheme 6C). Propylene oxide afforded a low yield of silyl ether 69 and trace quantities of cyclopropane 68 were detected by GCMS. With styrene oxide, silyl ether 70 was detected, arising from reductive opening of the styrene oxide. At least with these examples, it thus appears that reactions other than SN2 attack by benzylic anions occur preferentially.
Conclusions
Initial observations showed diarylcyclopentane formation on treatment of diarylmethanes with the Grubbs–Stoltz reagent (KOtBu + Et3SiH) when THF was used as solvent. The transformation featured two sequential substitution reactions involving cleavage of C–O bonds, with four of the carbons of the cyclopentane coming from THF. Similar reactivity was shown by 1,4-dioxane as solvent, where double-alkylation led to diarylcyclopropanes, with two of the cyclopropane carbons coming from 1,4-dioxane. In these cases, the intermediate alkoxides react with the silane to form silyl ethers, thereby allowing R3SiO− anions to act as leaving groups in the cyclisation steps. With appropriate substrates, deprotonation of benzylic C–H bonds with potassium bases afforded anions that underwent intramolecular substitution reactions at ethers or silyl ethers to form cyclopentanes and cyclopropanes. The chemistry also extended to formation of cyclopropanes from substitution reactions at sulfides, sulfoxides and sulfones. Our examples show relatively simple cyclopropanes and cyclopentanes – in view of the importance of these ring systems, we are currently examining the expansion of the scope of this chemistry.Author contributions
AJS, DD, STML, CP, JDB, KMcG, ALK performed the experiments. SF, SMN, SGL and JAM supervised the research. All authors analysed and commented on the results. AJS, DD and STML drafted the manuscript. All authors contributed to the final draft and approved the paper.Conflicts of interest
CP and DD are now employees of AstraZeneca and may or may not own stock options.Data availability
Supplementary information: the experimental procedures and spectroscopic data in support of the reported results. See DOI: https://doi.org/10.1039/d5sc10055k.Acknowledgements
The authors thank the EPSRC for funding via Prosperity Partnership (EP/S035990/1), and GSK (CP) and the University of Strathclyde for funding of studentships (AJS, SL). We thank GSK and EPSRC for an i-CASE award to DD (EP/S513908/1). Additionally, we thank Craig Irving, Patricia Keating, Dr Graeme Anderson and Dr Jessica Bame for NMR and mass spectrometry assistance.Notes and references
- J. Clayden, Deconstructing THF, Nat. Chem., 2010, 2, 523–524, DOI:10.1038/nchem.712.
- R. E. Mulvey, V. L. Blair, W. Clegg, A. R. Kennedy, J. Klett and L. Russo, Cleave and Capture Chemistry illustrated through Bimetallic-Induced Fragmentation of Tetrahydrofuran, Nat. Chem., 2010, 2, 588–591, DOI:10.1038/nchem.667.
- P. I. Dalko and Y. Langlois, Stereoselective Synthesis of Quaternary Benzylic Carbons Using C2 Symmetric Imidazolines and Tetrahydrofuran as Electrophile, J. Org. Chem., 1998, 63, 8107–8117, DOI:10.1021/jo980289r.
- S. M. Kunnari, R. Oilunkanieme, R. S. Laitinen and M. Ahlgrén, An Unexpected Tetrahydrofuran Ring Opening: Synthesis and Structural Characterization of Ph3PO(CH2)4TeBr4, J. Chem. Soc., Dalton Trans., 2001, 3417–3418, 10.1039/B108790H.
- S. H. Christensen, T. Holm and R. Madsen, Ring-opening of Cyclic Ethers with Carbon–Carbon Bond Formation by Grignard Reagents, Tetrahedron, 2014, 70, 4942–4946, DOI:10.1016/j.tet.2014.05.026.
- N. Chaisan, E. Y. K. Tan and S. Chiba, Hydroalkylation of 1,1-Diarylalkenes Mediated by Magnesium Hydride in Ethereal Solvents, Helv. Chim. Acta, 2024, 107, e202300212, DOI:10.1002/hlca.202300212.
- A. Fedorov, A. A. Toutov, N. A. Swisher and R. H. Grubbs, Lewis-Base Silane Activation: from reductive Cleavage of Aryl Ethers to Selective ortho-Silylation, Chem. Sci., 2013, 4, 1640–1645, 10.1039/C3SC22256J.
- A. A. Toutov, M. Salata, A. Fedorov, Y. F. Yang, Y. Liang, R. Cariou, K. N. Betz, E. P. A. Couzijn, J. W. Shabaker, K. N. Houk and R. H. Grubbs, A Potassium tert-Butoxide and Hydrosilane System for ultra-Deep Desulfurization of Fuels, Nat. Energy, 2017, 2, 17008, DOI:10.1038/nenergy.2017.8.
- W. B. Liu, D. P. Schuman, Y. F. Yang, A. A. Toutov, Y. Liang, H. F. T. Klare, N. Nesnas, M. Oestreich, D. G. Blackmond, S. C. Virgil, S. Bannerjee, R. N. Zare, R. H. Grubbs, K. N. Houk and B. M. Stoltz, Potassium tert-Butoxide-Catalyzed Dehydrogenative C–H Silylation of Heteroaromatics: A Combined Experimental and Computational Mechanistic Study, J. Am. Chem. Soc., 2017, 139, 6867–6879, DOI:10.1021/jacs.6b13031.
- S. Banerjee, Y. F. Yang, I. D. Jenkins, Y. Liang, A. A. Toutov, W. B. Liu, D. P. Schuman, R. H. Grubbs, B. M. Stoltz, E. H. Krenske, K. N. Houk and R. N. Zare, Ionic and Neutral Mechanisms for C–H Bond Silylation of Aromatic Heterocycles Catalyzed by Potassium tert-Butoxide, J. Am. Chem. Soc., 2017, 139, 6880–6887, DOI:10.1021/jacs.6b13032.
- A. J. Smith, A. Young, S. Rohrbach, E. F. O’Connor, M. Allison, H. S. Wang, D. L. Poole, T. Tuttle and J. A. Murphy, Electron-Transfer and Hydride-Transfer Pathways in the Stoltz–Grubbs Reducing System (KOtBu/Et3SiH), Angew. Chem., Int. Ed., 2017, 56, 13747–13751, DOI:10.1002/anie.201707914.
- P. Asgari, Y. Hua, A. Bokka, C. Thiamsiri, W. Prasitwatcharakorn, A. Karedath, X. Chen, S. Sardar, K. Yum, G. Leem, B. S. Pierce, K. Nam, J. Gao and J. Jeon, Catalytic Hydrogen Atom Transfer from Hydrosilanes to Vinylarenes for Hydrosilylation and Polymerization, Nat. Catal., 2019, 2, 164–173, DOI:10.1038/s41929-018-0217-z.
- F. Palumbo, S. Rohrbach, T. Tuttle and J. A. Murphy, N-Silylation of Amines Mediated by Et3SiH/KOtBu, Helv. Chim. Acta, 2019, 102, e1900235, DOI:10.1002/hlca.201900235.
- A. J. Smith, D. Dimitrova, J. N. Arokianathar, K. F. Clark, D. L. Poole, S. G. Leach and J. A. Murphy, Et3SiH + KOtBu Provide Multiple Reactive Intermediates that Compete in the Reactions and Rearrangements of Benzylnitriles and Indolenine, Chem. Sci., 2020, 11, 12364–12370, 10.1039/D0SC04244G.
- A. J. Smith, D. Dimitrova, J. N. Arokianathar, K. Kolodziejczak, A. Young, M. Allison, D. L. Poole, S. G. Leach, J. A. Parkinson, T. Tuttle and J. A. Murphy, New Reductive Rearrangement of N-arylindoles Triggered by the Grubbs–Stoltz Reagent Et3SiH/KOtBu, Chem. Sci., 2020, 11, 3719–3726, 10.1039/D0SC00361A.
- J. N. Arokianathar, K. Kolodziejczak, F. E. Bugden, K. F. Clark, T. Tuttle and J. A. Murphy, Benzylic C−H Functionalisation by [Et3SiH+KOtBu] leads to Radical Rearrangements in o-tolyl Aryl Ethers, Amines and Sulfides, Adv. Synth. Catal., 2020, 362, 2260–2267, DOI:10.1002/adsc.202000356.
- K. Kolodziejczak, A. J. Stewart, T. Tuttle and J. A. Murphy, Radical and Ionic Mechanisms in Rearrangements of o-Tolyl Aryl Ethers and Amines Initiated by the Grubbs–Stoltz Reagent, Et3SiH/KOtBu, Molecules, 2021, 26, 6879, DOI:10.3390/molecules26226879.
- D. Jenkins and E. H. Krenske, Mechanism of the Stoltz–Grubbs (KOtBu/Et3SiH) Silylation: Single-Electron Transfer is the Missing Link between the Heterolytic and Radical Pathways, Angew. Chem., Int. Ed., 2025, 64, e202517336, DOI:10.1002/anie.202517336.
- P. St. Onge, H. Nugraha and S. G. Newman, Hydroalkylation of Vinylarenes by TransitionMetal-Free in situ Generation of Benzylic Nucleophiles Using Tetramethyldisiloxane and Potassium tert-Butoxide, Angew. Chem., Int. Ed., 2025, 64, e202421077, DOI:10.1002/anie.202421077.
- As the Grubbs-Stoltz reaction mixture produces radicals, we suggest that 2-methylTHF may undergo easy abstraction of the methine C–H, to afford a tertiary α-alkoxyalkyl radical. In turn this radical would likely undergo deprotonation under the basic conditions of the reaction to afford a radical anion that could undergo fragmentation; see T. Hokamp, A. Dewanji, M. Lübbesmeyer, C. Mück-Lichtenfeld, E.-U. Würthwein and A. Studer, Radical Hydrodehalogenation of Aryl Bromides and Chlorides with Sodium Hydride and 1,4-Dioxane, Angew. Chem., Int. Ed., 2017, 56, 13275–13278, DOI:10.1002/anie.201706534.
- Resistance of THP to ring-opening reactions has been noted in other contexts: R. A. Dastidar, M. S. Kim, P. Zhou, Z. Luo, C. Shi, K. J. Barnett, D. J. McClelland, E. Y.-X. Chen, R. C. Van Lehn and G. W. Huber, Catalytic production of tetrahydropyran (THP): a biomass-derived, economically competitive solvent with demonstrated use in plastic dissolution, Green Chem . , 2022, 24, 9101–9113, 10.1039/D2GC03475A.
- A. Weickgenannt and M. Oestreich, Potassium tert-Butoxide-Catalyzed Dehydrogenative Si-O Coupling: Reactivity Pattern and Mechanism of an Underappreciated Alcohol Protection, Chem.–Asian J., 2009, 4, 406–410, DOI:10.1002/asia.200800426.
- A. A. Toutov, K. N. Betz, M. C. Haibach, A. M. Romine and R. H. Grubbs, Sodium Hydroxide Catalyzed Dehydrocoupling of Alcohols with Hydrosilanes, Org. Lett., 2016, 18, 5776–5779, DOI:10.1021/acs.orglett.6b01687.
- A. DeLucia, N. Das and A. K. Vannucci, Mild Synthesis of Silyl Ethers via Potassium Carbonate Catalyzed Reactions between Alcohols and Hydrosilanes, Org. Biomol. Chem., 2018, 16, 3415–2418, 10.1039/C8OB00464A.
- T. Yoshida, L. Ilies and E. Nakamura, Silylation of Aryl Halides with Monoorganosilanes Activated by Lithium Alkoxide, Org. Lett., 2018, 20, 2844–2847, DOI:10.1021/acs.orglett.8b00818.
- G. Nocera, I. Robb, K. F. Clark, T. M. McGuire, L. Evans, S. Chiba and J. A. Murphy, Reductive activation of arenes by potassium metal with potassium salts, Org. Chem. Front., 2024, 11, 5099–5106, 10.1039/D4QO01027B.
- G. Nocera, A. Young, F. Palumbo, K. J. Emery, G. Coulthard, T. McGuire, T. Tuttle and J. A. Murphy, Electron Transfer Reactions: KOtBu (but not NaOtBu) Photoreduces Benzophenone under Activation by Visible Light, J. Am. Chem. Soc., 2018, 140, 9751–9757, DOI:10.1021/jacs.8b06089.
- J. L. Crompton, J. R. Frost, S. M. Rowe, K. E. Christensen and T. J. Donohoe, Synthesis of Cyclopropanes via Hydrogen-Borrowing Catalysis, Org. Lett., 2023, 25, 5253–5257, DOI:10.1021/acs.orglett.3c01768.
- P. C. Too, G. H. Chan, Y. L. Tnay, H. Hirao and S. Chiba, Hydride Reduction by a Sodium Hydride–Iodide Composite, Angew. Chem., Int. Ed., 2016, 55, 3719–3723, DOI:10.1002/anie.201600305.
- March’s Advanced Organic Chemistry, ed. M. B. Smith, J. Wiley & Sons Inc., 8th edn, 2020, pp. 284–288 Search PubMed.
- A. Di Martino, C. Galli, P. Gargano and L. Mandolini, Ring-closure Reactions. Part 23.’ Kinetics of Formation of Three- to Seven-membered-ring N-Tosylazacyclo-alkanes. The Role of Ring Strain in Small- and Common-sized ring Formation, J. Chem. Soc. Perkin Trans. 2, 1985, 1345–1349, 10.1039/P29850001345.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |