Open Access Article
Open Access ArticleActive control of forward/backward charge transfer in Z-scheme water splitting: manipulating electrostatic affinity/repulsion between photocatalyst surface and electron mediator
Ren
Itagaki
a,
Akinobu
Nakada
 *a,
Hajime
Suzuki
*a,
Hajime
Suzuki
 ab,
Osamu
Tomita
ab,
Osamu
Tomita
 a and
Ryu
Abe
a and
Ryu
Abe
 *a
*a
aDepartment of Energy and Hydrocarbon Chemistry, Graduate School of Engineering, Kyoto University, Nishikyo-ku, Kyoto 615-8510, Japan. E-mail: nakada@scl.kyoto-u.ac.jp; ryu-abe@scl.kyoto-u.ac.jp
bPrecursory Research for Embryonic Science and Technology (PRESTO), Japan Science and Technology Agency (JST), 4-1-8 Honcho, Kawaguchi, Saitama 332-0012, Japan
First published on 16th February 2026
Abstract
Z-scheme water splitting using semiconductor photocatalysts is a promising strategy for achieving sustainable solar hydrogen production. However, in Z-scheme systems, competition for backward electron transfer, which exerts a substantial influence on the overall quantum efficiency, is thermodynamically unavoidable. In this study, a rational strategy is proposed to overcome the backward electron transfer in Z-scheme water-splitting systems by manipulating the electrostatic affinity/repulsion between photocatalyst surfaces and electron mediators. A designed cationic/neutral charge-switchable [Co(bpc)2]+/0 complex selectively suppressed the backward electron transfer caused by the electrostatic repulsion between the oxidised [Co(bpc)2]+ form and positively surface-charged H2-evolving photocatalyst, to which the forward electron transfer from the reduced [Co(bpc)2]0 form should be negligibly influenced by electrostatic interactions. This selective suppression of backward electron transfer enabled by charge-switchable [Co(bpc)2]+/0 is unique and could not be achieved using conventional cationic (e.g. Fe3+/2+) or anionic (e.g. IO3−/I−) redox mediators. As a result, the [Co(bpc)2]+/0 complex mediator provided the best photocatalytic performance for a benchmark H2-evolving SrTiO3:Rh photocatalyst among the conventional redox mediators and yielded a much improved apparent quantum efficiency of 2.7% for overall water splitting using SrTiO3:Rh and Bi4TaO8Cl photocatalysts. This study establishes a molecular design principle for redox mediators to improve Z-scheme water splitting, shifting the focus beyond the conventional emphasis on engineered photocatalyst materials.
Introduction
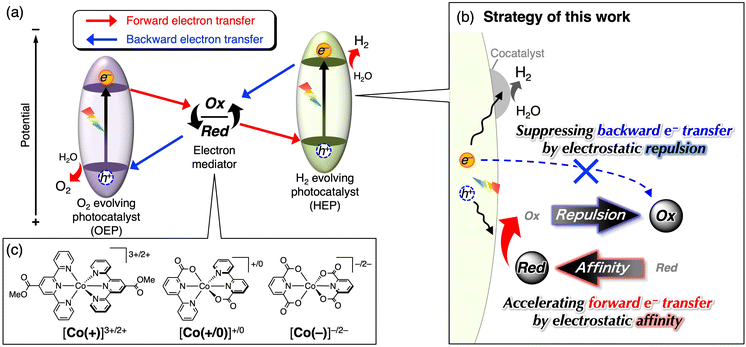
Solar-driven water splitting using semiconductor photocatalysts has been extensively studied as a promising technology for the sustainable production of hydrogen—a clean and renewable energy carrier.1–5 To achieve the practical production of solar hydrogen, it is imperative to efficiently utilise the visible light region, as this accounts for almost half of the solar energy that reaches the Earth's surface.6 Numerous efforts have been made to develop visible light-responsive semiconductor materials as photocatalysts.7 However, only a limited number of studies have reported successful overall water splitting by employing a single photocatalyst material.To overcome this limitation, considerable attention has been directed toward Z-scheme water-splitting systems inspired by natural photosynthesis.7,8 In a typical Z-scheme configuration, two distinct semiconductor photocatalysts, one responsible for H2 evolution and the other responsible for O2 evolution, are interconnected via a reversible electron mediator (redox couple) in solution. To date, various semiconductor photocatalysts, which could not solely split water, have been applied to Z-scheme water splitting because each photocatalyst only needs to drive H2 or O2 evolution with the redox species in the Z-scheme system.
However, in addition to the aforementioned benefits, the Z-scheme system has an inherent drawback, that is, competitive backward electron transfer reactions.8 The electron mediator must have reversible redox properties with a redox potential between the conduction band minimum (CBM) of the O2-evolving photocatalyst (OEP) and the valence band maximum (VBM) of the H2-evolving photocatalyst (HEP), as shown in Fig. 1a. Although an appropriate redox potential enables electron transportation from photoexcited OEP to HEP (red arrows in Fig. 1a), electron transfer in the backward direction, i.e., from HEP to OEP (blue arrows in Fig. 1a), is possible. In Z-scheme systems, competition for backward electron transfer, which exerts a substantial influence on the overall quantum efficiency, is thermodynamically unavoidable. Therefore, a substantial body of research has been dedicated to mitigating backward electron transfer, and most studies have focused on surface modifications, including cocatalyst loading.9–13
Regarding the electron mediator species, some redox compounds, such as IO3−/I−, Fe3+/2+, [Fe(CN)6]3−/4−, [Co(bpy)3]3+/2+, and polyoxometalates (POMs), have been applied to Z-scheme water splitting.14–18 The selection of electron mediators for combination with semiconductor photocatalysts has mainly been based on their redox potentials and stability at the pH of the reaction solution. However, clear guidelines for electron mediator selection with respect to resistance toward backward electron transfer are lacking, and thus choices have largely relied on “invisible” empirical compatibility in the specific combination of mediator and photocatalyst material.
In this study, a rational strategy to address the persistent challenge of backward electron transfer was developed with specific focus on the electrostatic affinity and repulsion between the photocatalyst surface and electron mediators (Fig. 1b). Three cobalt complexes with different charges were prepared: cationic [Co(tpyCO2Me)2]3+/2+ (Co(+)), anionic [Co(dipic)2]−/2− (Co(−)), and cationic/neutral [Co(bpc)2]+/0 (Co(+/0)) (Fig. 1c). Because inorganic semiconductor particles have specific surface charges that can be tuned by changing the solution pH,19 the effects of various combinations of mediator/surface charges on forward and backward electron transfer were systematically investigated. In particular, we aimed to actively control the electrostatic affinity/repulsion caused by photoinduced electron transfer to suppress backward electron transfer using the neutral-to-cationic charge-switching nature of a novel [Co(bpc)2]+/0 complex.
Results and discussion
Properties of cobalt complexes as a redox mediator
The homoleptic CoII complexes (Fig. 1c) with tridentate ligands—methyl 2,2′:6′,2″-terpyridine-4′-carboxylate (tpyCO2Me), 2,2′-bipyridine-6-carboxylate (bpc−), or dipicolinate (dipic2−)—were synthesised by mixing CoCl2·6H2O with the corresponding ligand (2 eq.) in methanol, as well as tetramethylammonium hydroxide in the case for carboxylate ligands. The corresponding oxidised forms (CoIII) were prepared by reacting the CoII complexes with a nitrosonium ion (NO+). The Co complexes showed negligible absorption in the visible region (λ > 400 nm), except for [Co(+)]2+, which showed weak absorption attributed to metal-to-ligand charge transfer arising from the lower π* level of tpyCO2Me (Fig. S1).20 Even for [Co(+)]2+, the molar extinction coefficient was not large (∼2000 M−1 cm−1). Therefore, the inner filter effect (shielding of light absorption of the photocatalyst) by the Co complexes would be small.The redox potential of an electron mediator for Z-scheme water splitting must be more positive than the conduction band minimum (CBM) of the OEP and more negative than the valence band maximum (VBM) of the HEP (Fig. 1a). The cobalt complexes [Co(+)]2+, [Co(+/0)]0, and [Co(−)]2− each exhibited a single redox wave corresponding to the metal-centred Co3+/2+ at E1/2 = +0.17, +0.25, and +0.53 V vs. Ag/AgCl, respectively (Fig. 2a). The redox peak separations (ΔE) for [Co(+)]2+ and [Co(+/0)]0 were ca. 70 mV, which is close to ideal reversibility. In contrast, the ΔE for the [Co(−)]−/2− redox reaction is approximately as large as 780 mV, suggesting slow electron transfer kinetics and/or the existence of protonation/deprotonation processes. However, the redox reaction was repeatable including [Co(−)]−/2− (Fig. S2), and the E1/2 value was constant over a range of pH values for all three Co complexes (Fig. 2b). Importantly, all CoIII complexes are suitable as redox mediators because their E1/2 values are between the CBM of representative OEP (e.g., WO3,13 BiVO4,21 and Bi4TaO8Cl22) and the VBM of HEP (e.g., SrTiO3:Rh23 and TaON24), as summarised in Fig. 2b and c.
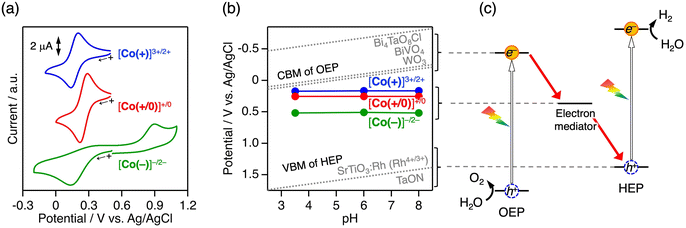 | ||
| Fig. 2 (a) Cyclic voltammograms of the cobalt complexes (0.3 mM) in aqueous NaCl (0.1 M) solution at pH 3.5 (adjusted with HCl) and (b) potentials of cobalt complexes as the redox mediator, CBM of OEP (WO3, BiVO4, and Bi4TaO8Cl), and VBM of HEP (SrTiO3:Rh and TaON), in which the band potentials of semiconductor photocatalysts are assumed to follow Nernst relationship.25–27 (c) Energy diagram of Z-scheme water splitting. | ||
Manipulation of electrostatic interactions on redox-mediated H2 evolution
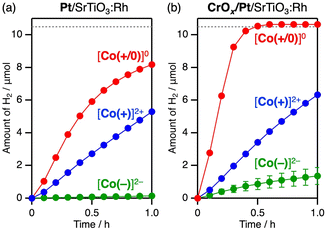
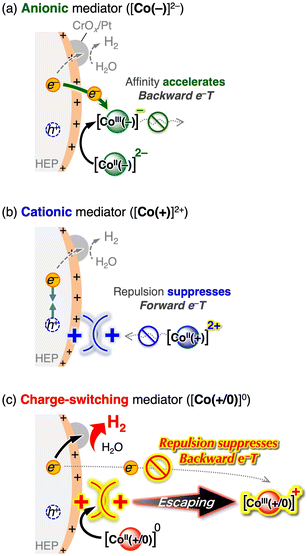
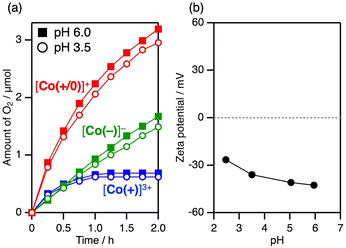
Fig. 3 shows the time course of visible-light-driven H2 evolution on M/SrTiO3:Rh (M = Pt or CrOx/Pt) photocatalysts using three different CoII complexes as electron donors. Among the three CoII complexes, [Co(+/0)]0 induced the most efficient H2 evolution, although the driving force for electron transfer was similar to that of [Co(+)]2+. The CrOx shell coating on the Pt cocatalyst (Fig. S3) further improved the H2 evolution rate by 2.1 times when using [Co(+/0)]0 (red lines in Fig. 3a and b). The CrOx shell coated on the Pt cocatalyst has been known to block the penetration of redox species into the Pt catalyst centre while allowing H+.9,28 Thus, the CrOx coating improved the H2 evolution activity by suppressing backward electron transfer (re-reduction of the oxidised electron donor) on the cocatalyst core. The use of [Co(−)]2− resulted in negligible H2 evolution activity over Pt/SrTiO3:Rh, whereas the CrOx overlayer significantly improved the activity by 16.5 times, although it was still the lowest among the three complexes. In contrast, the CrOx coating had only a minor effect (approximately 1.2-fold enhancement) on the rate of photocatalytic activity when [Co(+)]2+ was used.Interestingly, the degree of impact of the CrOx-shell coating on improving H2 evolution activity differed significantly depending on the Co complex used ([Co(−)]2− (16.5 times) > [Co(+/0)]0 (2.1 times) > [Co(+)]2+ (1.2 times)). Electrostatic interactions between the photocatalyst surface and Co complex with different charges were considered to understand the different efficacies of the CrOx shells in suppressing backward electron transfer. Although no significant adsorption of Co complexes onto CrOx/Pt/SrTiO3:Rh was observed (Fig. S4), dynamic electrostatic interaction in the reaction dispersion cannot be excluded. After serving as an electron donor, the total valence states of the oxidised Co complexes [Co(+)]3+, [Co(+/0)]+, and [Co(−)]− are +3, +1, and −1, respectively. Because the surface zeta potential of M/SrTiO3:Rh is positive (+43 mV) at pH 3.5, only [Co(−)]− could be electrostatically attracted to the positively charged photocatalyst surface (Fig. 4a). Therefore, [Co(−)]− would have greater opportunities for backward electron transfer, resulting in negligible H2 evolution activity on Pt/SrTiO3:Rh. Once the backward electron transfer on Pt was protected by the CrOx shell, [Co(−)]2− provided improved activity because the electrostatic affinity between [Co(−)]2− and the positively charged photocatalyst favours forward electron transfer. However, even if the Pt surface is protected by the CrOx shell, there are still significant opportunities for backward electron transfer on the SrTiO3:Rh surface before the photoexcited electrons reach the Pt cocatalyst core (Fig. 4a). Therefore, [Co(−)]2− still exhibited low activity even when a CrOx-shell coating was applied. In contrast, [Co(+)]3+/2+ experiences electrostatic repulsion with the photocatalyst in both its oxidised and reduced forms. This electrostatic repulsion possibly inhibited not only the backward electron transfer but also the forward electron transfer (Fig. 4b), resulting in a negligible effect of the CrOx coating when [Co(+)]2+ was used as the mediator. Notably, the superior performance of [Co(+/0)]0 should originate from its “charge-switching” nature. Regarding the backward electron transfer, [Co(+/0)]+ induces electrostatic repulsion, as in the case of [Co(+)]3+. In contrast, neutral [Co(+/0)]0, which does not induce electrostatic interactions, should not inhibit the forward electron transfer, in stark contrast to the case of [Co(+)]2+. Consequently, it is supposed that the “charge-switching” [Co(+/0)]0 enables selective suppression of backward electron transfer without decelerating forward electron transfer with a positively charged photocatalyst (Fig. 4c).
The hypothesis that electrostatic interaction manipulates backward electron transfer was further investigated by adding oxidised redox mediators to H2 evolution photocatalysis. Table 1 shows the rates of H2 evolution over CrOx/Pt/SrTiO3:Rh photocatalysts with various reversible electron donors in the presence and absence of the corresponding oxidised compound. In addition to the conventional Fe3+/2+ system,29 the H2 evolution rates obtained using Fe2+, [Co(+)]2+, or [Co(+/0)]0 did not significantly decrease upon the addition of the corresponding cationic electron acceptor (i.e., Fe3+, [Co(+)]3+, or [Co(+/0)]+). In contrast, pronounced reductions in photocatalytic activity were observed in anionic redox systems, including I−,30 [Co(−)]2−, and [SiVW11O40]6−.31 The decreased H2 evolution rate can be attributed to the competition between the reducing protons and oxidised redox compounds (Fig. 4a). Based on the above overall trend, we can conclude that the positively charged surface of CrOx/Pt/SrTiO3:Rh (zeta potential of +43 mV) is beneficial for suppressing backward electron transfer from the positively charged redox species. Notably, [Co(+/0)]0 provided higher H2 evolution rate than cationic electron donors. [Co(+/0)]0 and [Co(+)]2+ have similar redox potentials but may have different steric effects from their ligands, which would affect the photocatalytic performance. However, introducing [Co(tpy)2]2+, which is a cationic complex similar to [Co(+)]2+ but without methyl ester ligand, resulted in an activity similar to that of [Co(+)]2+ and much lower than that of [Co(+/0)]0. Therefore, the superior performance of [Co(+/0)]0 as the mediator for H2 evolution should originate from its “charge-switching” nature that enabled selective suppression of backward electron transfer without decelerating forward electron transfer with a positively charged photocatalyst (Fig. 4c). Therefore, [Co(+/0)]0 most effectively exploits the intrinsic potential of CrOx/Pt/SrTiO3:Rh for H2 evolution, outperforming various well-known conventional reversible electron donors. Under these conditions, an apparent quantum efficiency (AQE) of 5.1% at 430 nm was achieved for H2 evolution using CrOx/Pt/SrTiO3:Rh with [Co(+/0)]0 at pH 3.5.
| Charge of redox species | Donor | E Donor/Vb | Additive | Rate of H2 evolution/µmol h−1 | Retention ratio of H2 evolution rate |
|---|---|---|---|---|---|
| a Standard conditions: CrOx/Pt/SrTiO3:Rh (10 mg) in an aqueous solution (70 mL, pH 3.5) containing an electron donor (0.3 mM) in the absence or presence of the corresponding oxidised form as an additive (0.3 mM) under visible light irradiation (λ = 430 nm). b vs. Ag/AgCl. c Performed at pH 2.4. | |||||
| Cationic | Fe2+c | 0.56 (ref. 32) | — | 9.8 | 0.94 |
| Fe3+ | 9.2 | ||||
| [Co(+)]2+ | 0.17 | — | 7.7 | 0.95 | |
| [Co(+)]3+ | 7.3 | ||||
| [Co(tpy)2]2+ | 0.10 (ref. 33) | — | 5.5 | 0.98 | |
| [Co(tpy)2]3+ | 5.4 | ||||
| Neutral/Cationic | [Co(+/0)]0 | 0.25 | — | 27.8 | 0.97 |
| [Co(+/0)]+ | 27.1 | ||||
| Anionic | I− | 0.67 (ref. 34) | — | 9.2 | 0.09 |
| IO3− | 0.8 | ||||
| [Co(−)]2− | 0.53 | — | 2.7 | 0.44 | |
| [Co(−)]− | 1.2 | ||||
| [SiVW11O40]6− | 0.48 (ref. 31) | — | 7.1 | 0.42 | |
| [SiVW11O40]5− | 3.0 | ||||
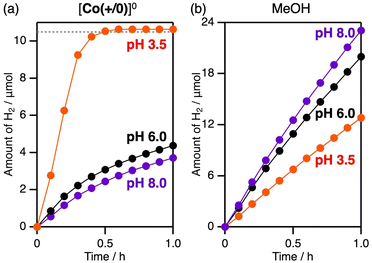
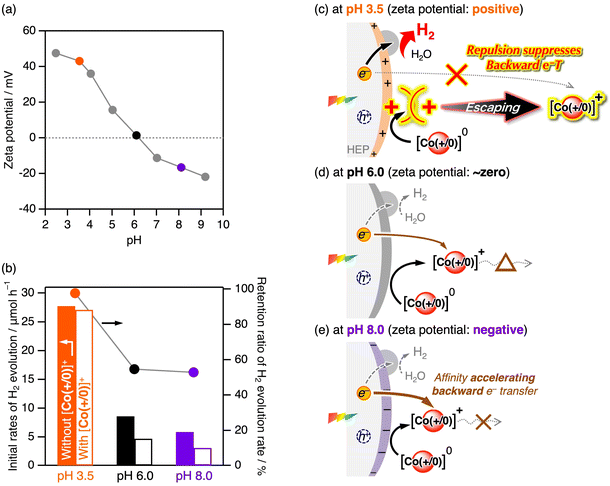
Upon increasing the solution pH, the photocatalytic H2 evolution activity of CrOx/Pt/SrTiO3:Rh with [Co(+/0)]0 decreased (Fig. 5a). This trend could not be simply explained by the decreased H+ concentration because the activity increased at high pH when MeOH was used as an irreversible sacrificial reductant (Fig. 5b). Therefore, the pH dependence should be derived based on the electron transfer process with the electron mediator.
The surface zeta potential of CrOx/Pt/SrTiO3:Rh decreased with increasing solution pH and flipped to negative at pH 6.0 (Fig. 6a). Importantly, the inhibitory effects caused by the addition of [Co(+/0)]+ also largely depended on the solution pH (Fig. 6b). As discussed above, the rate of H2 evolution remained almost unchanged when [Co(+/0)]+ was added at pH 3.5 (zeta potential = +43 mV). However, the durability against backward electron transfer weakened as the pH increased to 6.0 (zeta potential = ∼0) and 8.0 (zeta potential = −16 mV), where electrostatic repulsion with [Co(+/0)]+ was not expected. Similar relationship between zeta potential and photocatalytic activity was observed when using ZrO2/TaON (Fig. S5), which is another benchmark HEP.35 Although photoexcitation can shift the zeta potential to some extent, the overall trend suggests the importance of manipulating electrostatic interaction by appropriately tuning the surface charge and the “charge-switchable” redox mediator.
In summary, the electrostatic affinity/repulsion between the redox mediators and photocatalyst surface plays an important role in forward and backward electron transfer when using reversible electron donors (CoII complexes) for photocatalytic H2 evolution (Fig. 4 and 6). The reduced and oxidised forms of [Co(+)]3+/2+ are both cationic complexes, whereas those of [Co(−)]−/2− are both anionic complexes, indicating that their charged nature (cationic or anionic) is maintained during redox events. For instance, if the photocatalyst surface charge is positive, electrostatic repulsion with cationic [Co(+)]3+/2+ is unfavourable for accelerating forward electron transfer, but favourable for suppressing backward electron transfer (Fig. 4b). This situation is reversed when anionic [Co(−)]−/2− is used or when the photocatalyst has a negative surface charge, i.e., accelerating both forward and unfavourable backward electron transfer (Fig. 4a). Therefore, when [Co(+)]3+/2+ and [Co(−)]−/2− are used, enhancement of forward electron transfer inevitably occurs at the expense of increased backward electron transfer, resulting in an inherent trade-off. Notably, although many redox mediators have been employed in Z-scheme water splitting (e.g., Fe3+/2+, IO3−/I−, [Fe(CN)6]3−/4−, [Co(bpy)3]3+/2+, and anionic polyoxometalates), all conventional redox mediators are both cationic or anionic pairs. In contrast, “charge switching” [Co(+/0)]+/0 can cancel out the trade-off, and only the backward electron transfer with the cationic oxidised form [Co(+/0)]+ is affected by the electrostatic interaction and has a chance to be suppressed when the surface charge is positive (Fig. 4c and 6c). Therefore, we can conclude that [Co(+/0)]0 provides distinct H2 evolution activities for positively charged SrTiO3:Rh and TaON (enabled in acidic conditions).
Z-scheme water splitting using Co complexes as a redox mediator
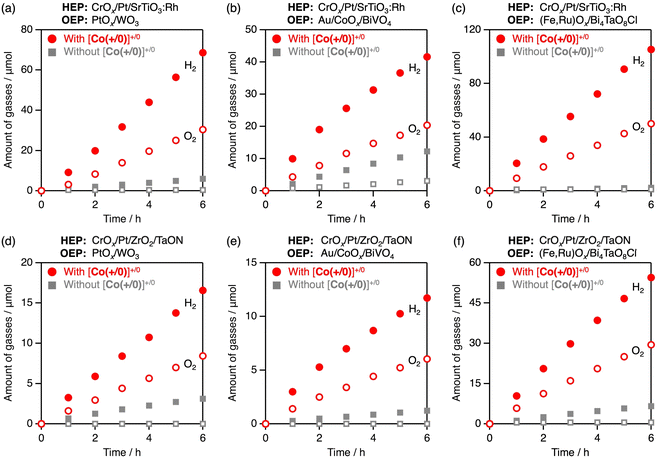
Prior to demonstrating Z-scheme overall water splitting, photocatalytic water oxidation was tested with the oxidised form of the three CoIII complexes using PtOx/WO3 (ref. 36) as an OEP. Among the three CoIII complexes, [Co(+/0)]+ provided the best water oxidation activity for PtOx/WO3 (Fig. 7a), despite having a smaller thermodynamic driving force for electron transfer than [Co(−)]− (Fig. 2). Considering the electrostatic interactions, the zeta potential of WO3 always remained negative (Fig. 7b), and the rate of O2 evolution remained unchanged, regardless of the solution pH. Negligible pH dependency of photocatalytic O2 evolution was also found using AgNO3 as a sacrificial electron acceptor (Fig. S6). Hence, the backward electron transfer with [Co(+/0)]+/0 does not seem to be influenced by the solution pH during the photocatalytic water oxidation over WO3. Similar to the case of H2 evolution, a trade-off between forward and backward electron transfer was considered due to electrostatic effects when cationic [Co(+)]3+/2+ or anionic [Co(−)]−/2− mediators were employed. For example, [Co(−)]− induced stable water oxidation without a decrease in the O2 evolution rate, although the rate was low. This is reasonable considering that the surface charge of PtOx/WO3 is negative (Fig. 7b), and both forward and backward electron transfers decelerate in the anionic [Co(−)]−/2− system. In the case of the charge-switchable [Co(+/0)]+/0 system, forward electron transfer from PtOx/WO3 to [Co(+/0)]+ is potentially favoured due to its electrostatic affinity, whereas the generated [Co(+/0)]0 is not influenced by the electrostatic interactions. This explains the superior O2 evolution activity of [Co(+/0)]+ as an electron acceptor.When BiVO4 or Bi4TaO8Cl was applied as the OEP with the aid of a [Co(+/0)]+ acceptor, the O2 evolution activity was influenced by the solution pH (Fig. S7). At pH 3.5, both BiVO4 and Bi4TaO8Cl have positive charges, which leads to electrostatic repulsion with [Co(+/0)]+ and was therefore unfavourable for electron transfer. Consequently, they showed decreased O2 evolution activity compared to conditions where the surface charge was negative (at pH 6 for BiVO4 and pH 8 for Bi4TaO8Cl; Fig. S7). The best AQE for O2 evolution was 4.7% at 430 nm, achieved using (Fe,Ru)Ox/Bi4TaO8Cl with [Co(+/0)]+ at pH 8.0.
Overall water splitting into H2 and O2 proceeded in the presence of [Co(+/0)]+/0 as a redox mediator in combination with CrOx/Pt/SrTiO3:Rh and PtOx/WO3 (entry 1, Table 2). On the other hand, simultaneous H2 and O2 generation was not observed in the absence of CrOx/Pt/SrTiO3:Rh, PtOx/WO3, or Co complexes (entries 2–4). Therefore, we can conclude that the Co complexes act as redox mediators in Z-scheme water splitting with CrOx/Pt/SrTiO3:Rh and PtOx/WO3 as H2- and O2-evolving photocatalysts, respectively. The overall water splitting proceeded stably for several cycles of photocatalysis tests (Fig. S8).
| Entry | HEP | OEP | Mediator | H2 rate/µmol h−1 | O2 rate/µmol h−1 | CoII![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CoIII CoIII |
|---|---|---|---|---|---|---|
| a Standard conditions: CrOx/Pt/SrTiO3:Rh (70 mg) and PtOx/WO3 (70 mg) in an aqueous solution (pH 3.5, 70 mL) containing an electron mediator (0.3 mM) under visible-light irradiation (λ = 430 nm). b N.D. = not detected. | ||||||
| 1 | CrOx/Pt/SrTiO3:Rh | PtOx/WO3 | [Co(+/0)]+ | 11.7 | 5.4 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9 |
| 2 | — | PtOx/WO3 | [Co(+/0)]+ | N.D.b | 4.5 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 3 | CrOx/Pt/SrTiO3:Rh | — | [Co(+/0)]+ | N.D. | N.D. | 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
| 4 | CrOx/Pt/SrTiO3:Rh | PtOx/WO3 | — | 1.0 | 0.5 | — |
| 5 | CrOx/Pt/SrTiO3:Rh | PtOx/WO3 | [Co(+)]3+ | 3.8 | 1.8 | 0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
| 6 | CrOx/Pt/SrTiO3:Rh | PtOx/WO3 | [Co(−)]− | 1.2 | 2.3 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
Among the three Co complexes, “charge switching” [Co(+/0)]+/0 induced the best water splitting performance (entries 1, 5 and 6 in Table 2). This trend is reasonable considering their performance in half-reactions (Fig. 3b and 7a). During steady Z-scheme operation with the [Co(+)]3+/2+ mediator, the oxidised form (CoIII; [Co(+)]3+) was dominant in the solution (entry 5 in Table 2) because water oxidation photocatalysis was much slower than H2 evolution (Fig. 3b and 7a). In contrast, for the anionic [Co(−)]−/2− mediator, CoII was dominant species under steady state conditions (entry 6 in Table 2), because the existence of the [Co(−)]− species significantly drops the H2 evolution activity (Table 1). In the [Co(+/0)]+/0 system, the CoIII/CoII balance was partially improved to some extent (CoII![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CoIII = 1
CoIII = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9, see entry 1 in Table 2). This improvement arises from the charge-switchable nature of the mediator, which alleviates the trade-off between forward and backward electron transfer in both H2- and O2-evolving reactions, as discussed above.
9, see entry 1 in Table 2). This improvement arises from the charge-switchable nature of the mediator, which alleviates the trade-off between forward and backward electron transfer in both H2- and O2-evolving reactions, as discussed above.
The AQE of the overall water splitting was recorded to be 2.0% at 405 nm when using the [Co(+/0)]+/0 mediator. Although the O2 evolution activity of the PtOx/WO3 photocatalyst still has considerable room for improvement, this performance represents the highest level reported to date for Z-scheme systems based on SrTiO3:Rh and WO3 photocatalysts using conventional redox mediators (Table S1).31 The [Co(+/0)]+/0 mediator was also applicable to Z-scheme overall water splitting under various combinations of HEP (SrTiO3:Rh, TaON) and OEP (WO3, BiVO4, Bi4TaO8Cl) (Fig. 8). Among these combinations, the use of SrTiO3:Rh and Bi4TaO8Cl resulted in the best water-splitting performance, with an AQE of 2.7% at 430 nm.
Conclusions
This study demonstrates the strong impact of manipulating the electrostatic interactions between photocatalyst surfaces and electron mediators to enhance the efficiency of Z-scheme water splitting systems. The designed charge-switchable [Co(bpc)2]+/0 complex represented an effective redox mediator, outperforming conventional cationic and anionic mediators. By selectively suppressing backward electron transfer without hindering forward electron transfer, the [Co(bpc)2]+/0 mediator improves the performance in both half-reactions and overall Z-scheme water splitting, with an apparent quantum efficiency of 2.7% for overall water splitting using SrTiO3:Rh and Bi4TaO8Cl photocatalysts (improved from 1.6%12 using Fe3+/2+). To date, in contrast to the rational engineering of photocatalyst materials itself, the selection of the redox mediator has largely relied on “invisible” empirical compatibility in specific combinations of photocatalyst materials. This work provides clear design guidelines for the selection of electron mediators from the perspective of resistance toward backward electron transfer. The design of metal complex mediators has room for tuning their important properties such as redox potentials to further push up the Z-scheme water splitting performance, contributing to the advancement of solar hydrogen production technologies.Experimental section
General procedure
1H NMR (400 MHz) spectra were recorded using a JEOL ECX400 spectrometer. Crystallographic data were collected at 143 K on a Rigaku Saturn 724+ with Varimax equipment using graphite monochromated Mo-Kα radiation (λ = 0.71073 Å). Powder X-ray diffraction patterns were recorded on MiniFlex II (Rigaku, X-ray source: Cu Kα). UV-visible absorption spectra were recorded at room temperature using a Shimadzu U1800 spectrometer. UV-visible diffuse reflectance spectra were recorded at room temperature on a Shimadzu U2600i spectrometer equipped with an integrating sphere. Cyclic voltammetry measurements were conducted on a BAS model 600F electrochemical analyser with a glassy carbon working electrode (ϕ = 3 mm) and platinum wire counter electrode. The reference electrode, made of a silver wire, was inserted into a small glass tube fitted with a porous Vycor frit at the tip and filled with an aqueous solution containing 3 M NaCl. Zeta potentials were measured using a zeta potential analyser (Zetasizer Nano ZS, Malvern Instruments) in an aqueous solution under dark conditions. The pH of the solution was adjusted using HCl and NaOH. Transmission electron microscopy (TEM) images were obtained using a JEOL JEM-2100F microscope.The amount of adsorbed cobalt complex was determined as follows. A suspension of CrOx/Pt/SrTiO3:Rh (1.5 mg) was dispersed in a 10 mL aqueous solution (pH 3.5, adjusted with HCl) containing cobalt complexes (ca. 0.3 mM) and stirred for 5 h in dark under an Ar atmosphere. The amount of adsorbed cobalt complexes was determined by UV-vis absorption spectroscopy. A cobalt complex solution without CrOx/Pt/SrTiO3:Rh was used as the blank control. Since there were cases where the chemical species changed before and after the experiment (CoII → CoIII species), we estimated the adsorption amount by fitting the absorption spectra using the molar extinction coefficient and Lambert–Beer law for each complex (eqn (1) and Fig. S4).
| Fitting = ε(CoII)[CoII] + ε(CoIII)[CoIII] | (1) |
Synthesis of redox mediator
[CoII(tpyCO2Me)2]Cl2 ([Co(+)]2+), [CoIII(tpyCO2Me)2]Cl3 ([Co(+)]3+), Na2[Co(dipic)2] ([Co(−)]2−), and Na[Co(dipic)2] ([Co(−)]−) were synthesised according to a previously reported procedure.37,38 K6[SiVW11O40]·6H2O and K5[SiVW11O40]·14H2O were prepared according to previously reported methods.31,39[CoII(bpc)2]0 ([Co(+/0)]0) was synthesised as follows: a 50 mL flask was charged with [2,2′-bipyridine]-6-carboxylic acid (bpcH, 1.0 g, 5.0 mmol, BLD pharm, >97.0%) and MeOH (33 mL, Wako Pure Chemical Industries Ltd, >99.8%). To this solution, tetramethylammonium hydroxide in MeOH (2.2 mL, 5.0 mmol, Sigma-Aldrich, 25 wt%) was added, affording a pale-yellow solution. CoCl2·6H2O (594 mg, 2.5 mmol, Kanto Chemical Co. Inc., >99.0%) was then added, and the reaction mixture was heated at 65 °C under an Ar atmosphere overnight. The mixture was subsequently cooled to room temperature, and the precipitate was collected by filtration and washed with a small amount of MeOH. The obtained crude solid was dissolved in MeOH (ca. 160 mL) in a 200 mL flask and heated until almost complete dissolution. The hot solution was filtered, and the filtrate was placed in a freezer to promote crystallisation. The resulting red crystals were collected via filtration, washed three times with cold MeOH, and dried under vacuum to obtain [CoII(bpc)2] (855.6 mg, 65% yield). 1H NMR (400 MHz, D2O, Fig. S9a): δ 98.68 (s, 2H), 95.57 (s, 2H), 94.74 (s, 2H), 77.42 (s, 2H), 39.86 (s 2H), 24.16 (s, 2H), 17.38 (s, 2H). Anal. Calcd. for C22H14N4O4Co·H2O: C, 55.59; H, 3.39; N, 11.79; found: C, 55.81; H, 3.19; N, 11.76.
[CoIII(bpc)2]+ ([Co(+/0)]+) was synthesised via chemical oxidation as follows: a 30 mL vial was charged with [CoII(bpc)2]0 (400 mg, 0.8 mmol) and MeCN (16.8 mL, Wako Pure Chemical Industries Ltd, > 99.5%). To this solution, NO+BF4− (153.2 mg, 1.3 mmol, Sigma-Aldrich, >95.0%) was added, and the mixture was stirred at room temperature for more than 1 h. The solvent was removed under reduced pressure, and the residue was dissolved in a minimal amount of H2O (ca. 2 mL). A saturated aqueous solution of KPF6 (Wako Pure Chemical Industries Ltd, >99.5%) was added to this solution, and the resulting mixture was filtered through a membrane filter and washed with a small amount of water. The residue was then treated with Amberlite IRA-900J ion-exchange resin (Cl form) at room temperature for more than 30 min in water. After removing the resin by filtration, the filtrate was evaporated under reduced pressure. The product was dried under vacuum to obtain [CoIII(bpc)2]+ as a possible salt form [CoIII(bpc)2]Cl0.85(PF6)0.15 (335.7 mg, 73% yield). 1H NMR (400 MHz, D2O, Fig. S9b): δ 9.10 (d, 2H), 8.98 (t, 2H), 8.63 (dd, 4H), 8.26 (t, 2H), 7.43 (t, 2H), 7.30 (d, 2H). Anal. Calcd. for C22H14N4O4CoCl0.85(PF6)0.15·2H2O: C, 48.47; H, 3.33; N, 10.28; found: C, 48.39; H, 3.13; N, 10.32. A summary of X-ray diffraction structural analysis for the MeOH-recrystallised single-crystal and crystallographic data is shown in Fig. S10 and Table S2.
Synthesis of semiconductor photocatalysts
SrTiO3:Rh,29 ZrO2/TaON,35 surface-modified WO3,13 BiVO4,40 and Bi4TaO8Cl22 were prepared according to previously reported procedures, and their synthesis was confirmed by XRD and UV-visible differential reflectance spectroscopy (Fig. S11).CrOx/Pt/SrTiO3:Rh was synthesised as follows. SrCO3 (1.72 g, 11.6 mmol, Wako Pure Chemical Industries Ltd, >99.99%), rutile-TiO2 (0.859 g, 10.8 mmol, Wako Pure Chemical Industries Ltd, >99.9%), and Rh2O3 (13.79 mg, 0.05 mmol, Wako Pure Chemical Industries Ltd, >99.99%) were used as starting materials. These were mixed in a mortar with an Sr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ti
Ti![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Rh atomic ratio of 1.07
Rh atomic ratio of 1.07![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.99
0.99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.01. The mixture was then transferred to an alumina crucible and subjected to preliminary calcination at 773 K in air for 2 h. After regrinding in a mortar, the mixture was subjected to main calcination at 1273 K in air for 10 h.29 Pt cocatalyst loading onto SrTiO3:Rh was performed by a photodeposition method.29 First, the synthesised SrTiO3:Rh powder was dispersed in a 10 vol% aqueous methanol solution containing H2PtCl6 (Wako Pure Chemical Industries Ltd, >99.9%) as a Pt precursor (0.3 wt% as metal). This suspension was irradiated with visible light (λ > 420 nm) for 3 h at room temperature under an Ar atmosphere to photodeposit Pt. A 300 W Xenon lamp with an L-42 cutoff filter was used for light irradiation. The cocatalyst-loaded photocatalysts were collected by filtration, washed with water, and dried at room temperature in air. In the same manner, a CrOx shell was coated onto Pt/SrTiO3:Rh using K2CrO4 (Wako Pure Chemical Industries Ltd, >99.0%) as a Cr precursor (0.45 wt% as metal) according to a previously reported procedure.41
0.01. The mixture was then transferred to an alumina crucible and subjected to preliminary calcination at 773 K in air for 2 h. After regrinding in a mortar, the mixture was subjected to main calcination at 1273 K in air for 10 h.29 Pt cocatalyst loading onto SrTiO3:Rh was performed by a photodeposition method.29 First, the synthesised SrTiO3:Rh powder was dispersed in a 10 vol% aqueous methanol solution containing H2PtCl6 (Wako Pure Chemical Industries Ltd, >99.9%) as a Pt precursor (0.3 wt% as metal). This suspension was irradiated with visible light (λ > 420 nm) for 3 h at room temperature under an Ar atmosphere to photodeposit Pt. A 300 W Xenon lamp with an L-42 cutoff filter was used for light irradiation. The cocatalyst-loaded photocatalysts were collected by filtration, washed with water, and dried at room temperature in air. In the same manner, a CrOx shell was coated onto Pt/SrTiO3:Rh using K2CrO4 (Wako Pure Chemical Industries Ltd, >99.0%) as a Cr precursor (0.45 wt% as metal) according to a previously reported procedure.41
CrOx/Pt/ZrO2/TaON was synthesised as follows. Zirconia-modified tantalum oxynitride (ZrO2/TaON) was synthesised through a nitridation method.35 First, Ta2O5 (1.33 g, 3.0 mmol, Kojundo Chemical Laboratory Co., Ltd, >99.9%) and ZrO(NO3)2·2H2O (0.16 g, 0.6 mmol, Kanto Chemical Co. Inc., >99.0%) were mixed with a small amount of MeOH. This mixture was then calcined in air at 1073 K for 2 h in an alumina crucible to obtain the ZrO2/Ta2O5 composite. Subsequently, this composite was subjected to nitridation under an NH3 gas flow (12 mL min−1) at 1123 K for 15 h to convert it into ZrO2/TaON. The Pt cocatalyst was loaded onto ZrO2/TaON via an impregnation method.35 The ZrO2/TaON powder was then dispersed in an aqueous solution containing H2PtCl6 as the Pt precursor (1.0 wt% as metal). After impregnation, the sample was dried and calcined at 473 K for 1 h under a H2 flow. A CrOx shell was coated onto Pt/ZrO2/TaON by the photodeposition method using K2CrO4 as the Cr precursor (1.5 wt% as metal) following the same procedure as for Pt/SrTiO3:Rh.
PtOx/WO3 was synthesised as follows. WO3 powder (10 g, Kojundo Chemical Laboratory Co., Ltd, >99.99%) was used as the starting material. The WO3 powder was first thermally treated in air at 973 K for 2 h to improve its crystallinity. The powder was then suspended in water (500 mL) by ultrasonication for 15 min, and fine particles that did not precipitate were removed by decantation. This separation process was repeated three times, and the resulting powder was filtered and air-dried to afford large WO3 particles. The WO3 powder (1.0 g) was impregnated with 0.63 mL of an aqueous solution containing 110 mM CsCl (Wako Pure Chemical Industries Ltd, >99.0%), and the pH was adjusted to 1 using HCl (Wako Pure Chemical Industries Ltd, 6 M). The mixture was dried and calcined at 773 K for 30 min in air. Subsequently, Cs-modified WO3 was suspended and stirred for 15 min in an aqueous solution (50 mL) containing 50 mM FeSO4 and 1 M H2SO4. After treatment, the suspension was decanted to separate the solid product and then dried in air.13 Then, a PtOx cocatalyst was loaded onto the surface-modified WO3 by an impregnation method. The Fe–H–Cs–WO3 powder was dispersed in an aqueous solution of H2PtCl6 as the Pt precursor (0.5 wt% as metal). After impregnation, the sample was dried and calcined at 823 K for 30 min in air to afford PtOx/WO3.31
Au/CoOx/BiVO4 was synthesised as follows. BiVO4 was synthesised via a hydrothermal process according to a previously reported procedure.40 The starting materials, NH4VO3 (1.17 g, 10 mmol, Wako Pure Chemical Industries Ltd, >99.0%) and Bi(NO3)3·5H2O (4.85 g, 10 mmol, Wako Pure Chemical Industries Ltd, >99.5%), were dissolved in a 2.0 M HNO3 solution (Wako Pure Chemical Industries Ltd, 69%), whose pH value was then adjusted to be about 0.5 using an NH3 solution (Wako Pure Chemical Industries Ltd, 28.0%). The mixed solution was strongly stirred until a light-yellow precipitate was observed. This precipitate was further aged for 2 h and then transferred to a Teflon-lined stainless-steel autoclave for 10 h of hydrothermal treatment at 473 K. Au and CoOx cocatalysts were loaded onto BiVO4via a photodeposition method.42 First, BiVO4 powder was dispersed in an aqueous solution containing HAuCl4 (Wako Pure Chemical Industries Ltd, >99.0%) as a Au precursor (0.8 wt% as metal). This suspension was irradiated for 2 h at 283 K under an Ar atmosphere to photodeposit Au. Second, through filtration, washing, and drying, the as-obtained powder was dispersed in a sodium buffer solution (pH 6.0, 50 mM) containing CoSO4 (Wako Pure Chemical Industries Ltd, >99.5%) as a Co precursor (0.1 wt% as metal). This suspension was irradiated for 1 h at 283 K under an Ar atmosphere to photodeposit CoOx. A 300 W xenon lamp with full-spectral irradiation was used for light irradiation.
(Fe,Ru)Ox/Bi4TaO8Cl was synthesized as follows. Bi4TaO8Cl was synthesized via a flux method according to a previously reported procedure.22 Bi2O3 (Wako Pure Chemical Industries Ltd, >99.9%), Ta2O5 (Wako Pure Chemical Industries Ltd, >99.9%), and BiOCl (Wako Pure Chemical Industries Ltd, >95.0%) were mixed at the stoichiometric molar ratio required for Bi4TaO8Cl. The mixture was combined with a eutectic mixture of NaCl (Wako Pure Chemical Industries Ltd, >99.5%) and CsCl (35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 65) as a flux, at a solute concentration (Bi4TaO8Cl/(Bi4TaO8Cl + flux)) of 5 mol%. The mixture (25 g) was placed in an alumina crucible and heated to 1023 K at a rate of 20 K h−1. The product was thoroughly washed with deionised water and collected via filtration. The obtained powder samples were then dried at room temperature. Co-loading of the (Fe,Ru)Ox cocatalyst was performed by simultaneous impregnation using FeCl3 (Wako Pure Chemical Industries Ltd, >99.0%) and RuCl3 (Wako Pure Chemical Industries Ltd, 85 wt%) precursors (0.5 and 0.9 wt% for Fe and Ru, respectively, i.e., 10 mol% each for Bi4TaO8Cl).12 A proper amount of the photocatalyst powder was dispersed in an aqueous solution containing the defined molar quantities of FeCl3 and RuCl3, followed by solvent evaporation in a water bath and heating at 573 K for 1 h under an Ar flow (20 mL min−1).
65) as a flux, at a solute concentration (Bi4TaO8Cl/(Bi4TaO8Cl + flux)) of 5 mol%. The mixture (25 g) was placed in an alumina crucible and heated to 1023 K at a rate of 20 K h−1. The product was thoroughly washed with deionised water and collected via filtration. The obtained powder samples were then dried at room temperature. Co-loading of the (Fe,Ru)Ox cocatalyst was performed by simultaneous impregnation using FeCl3 (Wako Pure Chemical Industries Ltd, >99.0%) and RuCl3 (Wako Pure Chemical Industries Ltd, 85 wt%) precursors (0.5 and 0.9 wt% for Fe and Ru, respectively, i.e., 10 mol% each for Bi4TaO8Cl).12 A proper amount of the photocatalyst powder was dispersed in an aqueous solution containing the defined molar quantities of FeCl3 and RuCl3, followed by solvent evaporation in a water bath and heating at 573 K for 1 h under an Ar flow (20 mL min−1).
Photocatalytic reactions
Photocatalytic reactions were carried out in a Pyrex glass reactor under an Ar flow (20 mL min−1). For the half-reaction of photocatalysis, a HEP (10 mg) or an OEP (35 mg) was dispersed in an aqueous solution (70 mL) containing an electron donor or acceptor (0.3 mM). The pH of the solution was adjusted by adding HCl or NaOH. For Z-scheme water splitting, HEP (70 mg) and OEP (70 mg) were dispersed in an aqueous solution (70 mL, pH 3.5 adjusted with HCl) containing an electron mediator (0.3 mM). The reaction dispersion was irradiated with visible light (λ = 430 nm; 195 mW cm−2) using an LED lamp (CL-H1-430-9-1-B and CL-H1LCB02, Asahi Spectra Co.). The generated gases were detected using a thermal conductivity detector (TCD) gas chromatograph (GC 3210D, GL Sciences, thermal conductivity detector, column packing: 5 Å molecular sieves, and Ar carrier gas) connected to the reaction system.The apparent quantum efficiency (AQE) for H2 evolution or Z-scheme water splitting was measured using the same experimental setup with bandpass filters and estimated according to eqn (2).
| AQE = n × (amount of evolved H2)/(amount of incident photons) | (2) |
Author contributions
R. I. and A. N. designed the study, with advice from H. S., O. T., and R. A. All remaining experiments were performed by R. I. All authors discussed the results. R. I. and A. N. wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the main text and the supplementary information (SI). Supplementary information: UV-vis absorption spectra, cyclic voltammograms, photocatalytic activities for H2 evolution, zeta potential, 1H NMR spectra, powder XRD patterns, and UV-vis absorption spectra. See DOI: https://doi.org/10.1039/d5sc10049f.CCDC 2528130 contains the supplementary crystallographic data for this paper.43
Acknowledgements
This work was supported by a JSPS KAKENHI grant (JP24K01603) and Grant-in-Aid for Transformative Research Areas “Concerto Photocatalysis” (JP23H03830 and JP23H03832). R. I. acknowledges support from the JSPS Fellowship for Young Scientists (JP23KJ1351). The authors are grateful to Profs. Kazuhiko Semba and Takayuki Tanaka (Kyoto University) for their kind support for single-crystal X-ray crystal structural analysis. Our acknowledgement is extended to Prof. Takaaki Toriyama (Kyushu University) for his support with TEM analysis. We express our gratitude to Mr Harutaka Ninomiya (Kyoto University) for his assistance in the synthesis of polyoxometalates.References
- A. Fujishima and K. Honda, Nature, 1972, 238, 37–38 CrossRef CAS PubMed.
- R. Abe, Bull. Chem. Soc. Jpn., 2011, 84, 1000–1030 CrossRef CAS.
- A. Kudo and Y. Miseki, Chem. Soc. Rev., 2009, 38, 253–278 RSC.
- F. E. Osterloh, Chem. Soc. Rev., 2013, 42, 2294–2320 RSC.
- S. Chen, T. Takata and K. Domen, Nat. Rev. Mater., 2017, 2, 17050 CrossRef CAS.
- C. Chen, W. Ma and J. Zhao, Chem. Soc. Rev., 2010, 39, 4206–4219 RSC.
- Q. Wang and K. Domen, Chem. Rev., 2020, 120, 919–985 CrossRef CAS PubMed.
- Y. Wang, H. Suzuki, J. Xie, O. Tomita, D. J. Martin, M. Higashi, D. Kong, R. Abe and J. Tang, Chem. Rev., 2018, 118, 5201–5241 CrossRef CAS PubMed.
- M. Shi, X. Wu, Y. Zhao, R. Li and C. Li, J. Am. Chem. Soc., 2025, 147, 3641–3649 CrossRef CAS.
- H. Matsuoka, T. Inoue, H. Suzuki, O. Tomita, S. Nozawa, A. Nakada and R. Abe, Sol. RRL, 2023, 7, 2300431 CrossRef CAS.
- H. Kato, Y. Sasaki, A. Iwase and A. Kudo, Bull. Chem. Soc. Jpn., 2007, 80, 2457–2464 CrossRef CAS.
- A. Nakada, H. Suzuki, J. J. M. Vequizo, K. Ogawa, M. Higashi, A. Saeki, A. Yamakata, H. Kageyama and R. Abe, ACS Appl. Mater. Interfaces, 2019, 11, 45606–45611 CrossRef CAS PubMed.
- Y. Miseki and K. Sayama, Catal. Sci. Technol., 2019, 9, 2019–2024 RSC.
- R. Abe, K. Sayama, K. Domen and H. Arakawa, Chem. Phys. Lett., 2001, 344, 339–344 CrossRef CAS.
- H. Kato, M. Hori, R. Konta, Y. Shimodaira and A. Kudo, Chem. Lett., 2004, 33, 1348–1349 CrossRef CAS.
- Y. Miseki, S. Fujiyoshi, T. Gunji and K. Sayama, J. Phys. Chem. C, 2017, 121, 9691–9697 CrossRef CAS.
- Y. Sasaki, H. Kato and A. Kudo, J. Am. Chem. Soc., 2013, 135, 5441–5449 CrossRef CAS PubMed.
- K. Tsuji, O. Tomita, M. Higashi and R. Abe, ChemSusChem, 2016, 9, 2201–2208 CrossRef CAS PubMed.
- Y. Sasaki, H. Nemoto, K. Saito and A. Kudo, J. Phys. Chem. C, 2009, 113, 17536–17542 CrossRef CAS.
- I. Krivokapic, M. Zerara, M. L. Daku, A. Vargas, C. Enachescu, C. Ambrus, P. Tregenna-Piggott, N. Amstutz, E. Krausz and A. Hauser, Coord. Chem. Rev., 2007, 251, 364–378 CrossRef CAS.
- J. Resasco, H. Zhang, N. Kornienko, N. Becknell, H. Lee, J. Guo, A. L. Briseno and P. Yang, ACS Cent. Sci., 2016, 2, 80–88 CrossRef CAS PubMed.
- R. Itagaki, A. Nakada, H. Suzuki, O. Tomita, H.-C. Chang and R. Abe, J. Am. Chem. Soc., 2025, 147, 15567–15577 CrossRef CAS PubMed.
- Z. Pan, J. J. M. Vequizo, H. Yoshida, J. Li, X. Zheng, C. Chu, Q. Wang, M. Cai, S. Sun, K. Katayama, A. Yamakata and K. Domen, Angew. Chem., Int. Ed., 2025, 64, e202414628 CrossRef CAS PubMed.
- W.-J. Chun, A. Ishikawa, H. Fujisawa, T. Takata, J. N. Kondo, M. Hara, M. Kawai, Y. Matsumoto and K. Domen, J. Phys. Chem. B, 2003, 107, 1798–1803 CrossRef CAS.
- M. Anik, Electrochim. Acta, 2009, 54, 3943–3951 CrossRef CAS.
- F. Ambrosio, J. Wiktor and A. Pasquarello, ACS Energy Lett., 2018, 3, 829–834 CrossRef CAS.
- J. M. Bolts and M. S. Wrighton, J. Phys. Chem., 1976, 80, 2641–2645 CrossRef CAS.
- H. Suzuki, Y. Okada, S. Kise, X. Sui, S. Nozawa, O. Tomita and R. Abe, ACS Catal., 2025, 15, 4870–4879 CrossRef CAS.
- Y. Sasaki, A. Iwase, H. Kato and A. Kudo, J. Catal., 2008, 259, 133–137 CrossRef CAS.
- R. Abe, K. Shinmei, N. Koumura, K. Hara and B. Ohtani, J. Am. Chem. Soc., 2013, 135, 16872–16884 CrossRef CAS PubMed.
- O. Tomita, H. Naito, A. Nakada, M. Higashi and R. Abe, Sustainable Energy Fuels, 2022, 6, 664–673 RSC.
- A. J. Bard, R. Parsons and J. Jordan, Standard Potentials in Aqueous Solution, New York, 1985 Search PubMed.
- T. M. Suzuki, S. Yoshino, K. Sekizawa, Y. Yamaguchi, A. Kudo and T. Morikawa, Appl. Catal., B, 2022, 316, 121600 CrossRef CAS.
- R. Abe, K. Sayama and H. Sugihara, J. Phys. Chem. B, 2005, 109, 16052–16061 CrossRef CAS PubMed.
- K. Maeda, M. Higashi, D. Lu, R. Abe and K. Domen, J. Am. Chem. Soc., 2010, 132, 5858–5868 CrossRef CAS PubMed.
- Y. Miseki and K. Sayama, RSC Adv., 2014, 4, 8308 RSC.
- E. C. Constable, K. Harris, C. E. Housecroft, M. Neuburger and J. A. Zampese, CrystEngComm, 2010, 12, 2949–2961 RSC.
- M. K. Tse, H. Jiao, G. Anilkumar, B. Bitterlich, F. G. Gelalcha and M. Beller, J. Organomet. Chem., 2006, 691, 4419–4433 CrossRef CAS.
- H. Ninomiya, O. Tomita, H. Suzuki, A. Nakada and R. Abe, ACS Mater. Lett., 2024, 6, 2474–2478 CrossRef CAS.
- Y. Qi, J. Zhang, Y. Kong, Y. Zhao, S. Chen, D. Li, W. Liu, Y. Chen, T. Xie, J. Cui, C. Li, K. Domen and F. Zhang, Nat. Commun., 2022, 13, 484 CrossRef CAS PubMed.
- K. Maeda, K. Teramura, D. Lu, N. Saito, Y. Inoue and K. Domen, Angew. Chem., Int. Ed., 2006, 45, 7806–7809 CrossRef CAS PubMed.
- Y. Qi, Y. Zhao, Y. Gao, D. Li, Z. Li, F. Zhang and C. Li, Joule, 2018, 2, 2393–2402 CrossRef CAS.
- CCDC 2528130: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qvqlx.
| This journal is © The Royal Society of Chemistry 2026 |