Open Access Article
Open Access ArticleControl over stereogenic centres beyond tetracoordination
Anton
Budeev
 and
Christof
Sparr
and
Christof
Sparr
 *
*
Department of Chemistry, University of Basel, St. Johanns-Ring 19, Basel 4056, Switzerland. E-mail: christof.sparr@unibas.ch
First published on 18th February 2026
Abstract
In contrast to conventional tetracoordinate stereogenic centres exhibiting twofold stereogenicity, penta- and hexacoordinate stereogenic centres may encode more than two stereoisomers, expanding the stereochemical space accessible to these species. This more complex stereoisomerism is characteristic to compounds of a variety of main-group elements and transition metals having a suitable set of ligands. Despite these unique hallmarks and the prospective applications in medicinal chemistry, catalysis, and information science, this area of stereoselective synthesis awaits widespread exploration. In this review, the fundamentals of the higher-order stereogenicity of penta- and hexacoordinate stereogenic centres will thus be summarized and an overview of emerging applications and their stereoselective synthesis will be provided.
Introduction
The ubiquitous tetracoordinate stereogenic centres, mainly represented by the tetrasubstituted carbon, are known to exist in two stereoisomeric forms per stereogenic centre. The number of stereoisomers of a molecule containing these stereogenic centres is thus described by the Le Bel–van 't Hoff rule as 2n, where 2 is the stereogenicity, or the number of stereoisomers per one stereogenic centre, and n represents the number of stereogenic centres.1,2 Depending on symmetry, the number of meso isomers needs to be subtracted from this calculated value. Although the Le Bel–van 't Hoff rule was initially described for carbon-based stereogenic centres, it is generally applicable to all types of twofold stereogenic elements. The principle can also be used to predict the number of stereoisomers encoded by a stereogenic axes (atropisomers) and other irreducible stereogenic units with two states. In contrast to twofold stereogenic structures, there are molecular systems that have more than a pair of stereoisomers arising from a single irreducible stereogenic unit, i.e., they have higher-order stereogenicity. The number of stereoisomers for such systems can be calculated using an extended Le Bel–van 't Hoff rule, as m =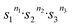 … where s is the stereogenicity and n the number of stereogenic units with that specific stereogenicity. These stereogenic units are most commonly represented by stereogenic centres beyond tetracoordination3–6 and several atropisomers,7–9 although they can also be encountered in systems with stereogenic units that span over larger parts of a molecule.10–13 Notably, higher-order stereogenic units encode a significantly expanded stereochemical space, enabling access to distinct three-dimensional molecular configurations with the potential for a diversity of applications. However, the stereoselective control over these increasingly complex systems remained underdeveloped. In this review, the stereochemistry of various higher-order stereogenic centres will be discussed along with their stereoselective synthesis, whereas solvent and anion effects on stereoisomerisation processes as well as associative isomerisations fall outside the scope of this review and are not addressed. Unlike tetrahedral geometries, other stereogenic centres can possess coordination numbers greater than four, thereby expanding the range of possible configurations and increasing the number of stereoisomers per one stereogenic centre. These stereogenic centres are typically represented by penta- and hexacoordinate species. Since hexacoordinate stereogenic centres may result in an immense stereochemical complexity, our discussion will start with pentacoordinate stereogenic centres.
… where s is the stereogenicity and n the number of stereogenic units with that specific stereogenicity. These stereogenic units are most commonly represented by stereogenic centres beyond tetracoordination3–6 and several atropisomers,7–9 although they can also be encountered in systems with stereogenic units that span over larger parts of a molecule.10–13 Notably, higher-order stereogenic units encode a significantly expanded stereochemical space, enabling access to distinct three-dimensional molecular configurations with the potential for a diversity of applications. However, the stereoselective control over these increasingly complex systems remained underdeveloped. In this review, the stereochemistry of various higher-order stereogenic centres will be discussed along with their stereoselective synthesis, whereas solvent and anion effects on stereoisomerisation processes as well as associative isomerisations fall outside the scope of this review and are not addressed. Unlike tetrahedral geometries, other stereogenic centres can possess coordination numbers greater than four, thereby expanding the range of possible configurations and increasing the number of stereoisomers per one stereogenic centre. These stereogenic centres are typically represented by penta- and hexacoordinate species. Since hexacoordinate stereogenic centres may result in an immense stereochemical complexity, our discussion will start with pentacoordinate stereogenic centres.
Pentacoordinate stereogenic centres
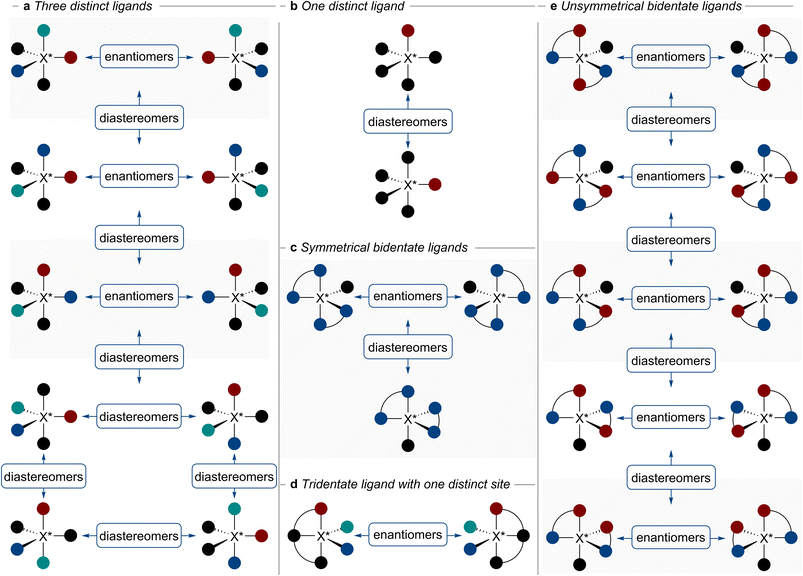
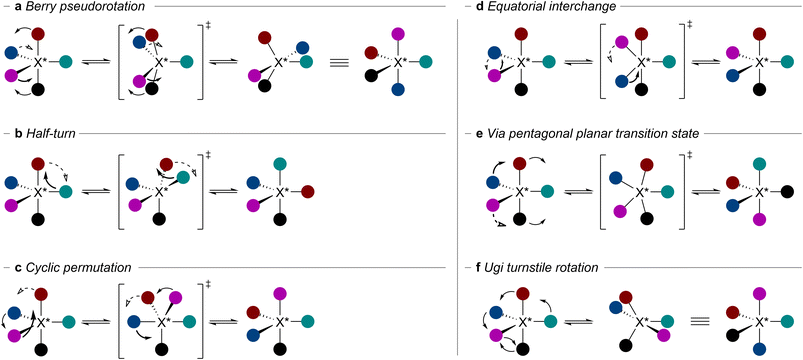
Pentacoordinate compounds are often species of main-group elements that typically adopt geometries that span a continuum between the idealised trigonal bipyramidal and square pyramidal arrangements.14,15 For both idealised geometries, ligands occupy two distinct positions. In a trigonal bipyramidal geometry, two ligands occupy axial (also called apical) and the other three occupy equatorial positions. In the square pyramid, one ligand occupies axial positions and four ligands occupy basal sites (Fig. 1a). The stereochemistry of pentacoordinate species is characterised by a vastly increased number of stereoisomers emerging from one stereogenic centre. Remarkably, up to 20 stereoisomers, 10 pairs of diastereomeric enantiomers, may emerge from a single stereogenic centre in a trigonal bipyramid (Fig. 1b and c).16 It is worth noting that in trigonal bipyramidal systems, exchange between positions of the same type (e.g. equatorial) leads to enantiomers, while exchange between sites of different types (equatorial with axial) gives rise to diastereomers. In pentacoordinate systems with exclusively monodentate ligands, enantiomerism is observed when at least three of the five substituents are distinct (Fig. 2a). In the simplest case, when only one of the five ligands is different, two achiral diastereomeric configurations are observed (Fig. 2b). This situation changes when two pairs of ligands are connected to form a spirocyclic system. Now, a single distinct ligand is sufficient to introduce threefold stereogenicity with two diastereomers: an achiral one in which the distinct ligand occupies the apical position and another in which the distinct ligand occupies the equatorial position, forming a pair of enantiomers (Fig. 2c). In the case of a tridentate ligand with one of the axial substituents different from the other and two distinct equatorial ligands, only one pair of enantiomers is typically observed. This is due to ligand rigidity which usually prevents the tridentate ligand from simultaneously occupying two equatorial sites (Fig. 2d). Returning to the case with two bidentate ligands, if they are unsymmetrical, the number of stereoisomers increases dramatically, with up to ten possible stereoisomers (Fig. 2e). In practice, the number of stereoisomers that are isolable or observable under standard conditions is usually lower due to the configurational instability of certain stereoisomers. This property is common for pentacoordinate species of main group elements, with configurational stability being dependent on the central element, its oxidation state, and the ligands. Therefore, some structures (Fig. 2 and 3) can thus be viewed as intermediates or even transition states in the intramolecular stereoisomerisation processes.Several mechanisms have been proposed to explain these stereoisomerisation processes. In 1960, Berry proposed the first mechanism, in which two equatorial ligands exchange positions with two axial ones via a square pyramidal transition state (Fig. 3a).17 Geometrically, the angle between two equatorial ligands is increased from 120° to 180° and the angle between two axial ligands is decreased from 180° to 120°. As a result of a single Berry pseudorotation, a diastereomer of the starting stereoisomer is formed. Notably, all stereoisomers in a trigonal bipyramid are connected through a series of one to five Berry pseudorotations.18,19
In 1969, four other stereoisomerisation mechanisms were proposed by Muetterties.19 The half-turn mechanism is characterised by an exchange of axial and equatorial ligands by a 180° rotation (Fig. 3b). In the cyclic permutation mechanism, a face of the trigonal bipyramid connecting one axial and two equatorial ligands is rotated by 120° (Fig. 3c). In both processes, axial and equatorial positions are exchanged, leading to diastereomerisation. In another process, interchange between two equatorial ligands by a 180° rotation results in enantiomerisation (Fig. 3d). Finally, stereoisomerisation can occur via a planar pentagonal transition state, enabling isomerisation between any two stereoisomers of a pentacoordinate stereogenic centre in a single process (Fig. 3e). The latter two processes are considered implausible since they proceed via planar transition states of high energy.19
A turnstile rotation mechanism was suggested by Ugi in 1970.20,21 In this mechanism, a trio of two equatorial and one axial ligand and a pair of two remaining ligands undergo a relative 60° rotation (Fig. 3f). Shortly after this proposal, Yates and later Kutzelnigg suggested that the turnstile rotation is not an independent mechanism but rather describes the same motion as Berry pseudorotations.22,23 Later, in 2010, Lammertsma and Couzijn confirmed this notion in a theoretical study by the analysis of the structural changes along the stereomutational intrinsic reaction coordinate using internal coordinates.24 They concluded that Ugi's turnstile rotation is an alternative way of describing the Berry pseudorotation. Moreover, the mechanisms (Fig. 3b and c) represent sequences of two and three Berry pseudorotations, respectively. It is worth noting that, in the case of five different ligands, a minimum of five Berry pseudorotations is required for enantiomerisation. However, for specific diastereomers of bidentate ligands, direct enantiomerisation via a single Berry pseudorotation is feasible. Notably, a mathematical approach of polytopal rearrangement model has been successfully applied to describe stereoisomerization processes in pentacoordinate systems and has been reviewed comprehensively elsewhere.25 Many pentacoordinate compounds, especially those with five monodentate ligands, readily undergo Berry pseudorotations, making it challenging to isolate or even observe the compound in a specific configuration.
For example, the barrier for pseudorotation in PF5 was calculated to be only ∼15 kJ mol−1.26 The introduction of bulky bidentate ligands can help stabilise the configuration to some extent, as demonstrated by pentacoordinate silicates bearing two bidentate biaryl ligands (Fig. 4a).27 However, the most general way to stabilise a pentacoordinate system is by the use of Martin's hexafluorocumyl alcohol ligand (Fig. 4b).28 This ligand enhances stability through multiple factors, one of which is apicophilicity, the tendency for more electronegative substituents in a trigonal bipyramidal geometry to occupy axial positions. This phenomenon can be attributed to the nature of the apical bonding in the pentacoordinate system, which is characterised by a 3-centre-4-electron bond. In this arrangement, electron density is primarily localised on the two axial substituents, making configurations where more electronegative substituents occupy axial positions more stable.29,30 Consequently, a positive charge develops on the central atom of the 3-centre-4-electron bond, which is stabilised by the electropositive equatorial carbon ligand. Another stabilising factor is the Thorpe-Ingold effect, which occurs due to the presence of two geminal trifluoromethyl groups, favouring the formation of a cyclic system.31 Finally, the five-membered ring connecting axial and equatorial sites of the trigonal bipyramid accommodates intrinsically unstrained bond angles (∼90°) between the apical and the equatorial bonds in the ring.28 These factors contribute not only to the stabilisation of the ground states, but also to the kinetic stabilisation of usually unstable pentacoordinate systems. For example, using these ligands, otherwise highly unstable derivatives of hypercoordinate bromine,32 sulfur,33 phosphorus and silicon34 have been prepared and isolated (Fig. 4c).
In the case of pentacoordinate siliconates bearing Martin's ligands, only the most stable diastereomer can normally be observed, making the system apparently twofold stereogenic (Fig. 4d).27,35 In contrast, for pentacoordinate phosphoranes, up to four stereoisomers can be readily distinguished, as demonstrated by Akiba and co-workers (Fig. 4e).3,4,36,37 For the more thermodynamically stable trans isomer, where the electronegative oxygen atoms occupy both axial positions, a high enantiomerisation barrier of 142 kJ mol−1 was measured. Remarkably, also the cis diastereomer, where one oxygen occupies an axial and the other an equatorial position, was sufficiently stable to be isolated. The barrier of interconversion from cis to trans was found to be 92 kJ mol−1, indicating a slow diastereomerisation at room temperature. The barrier for enantiomerisation between the two enantiomers of the cis isomer was much lower (42 kJ mol−1), making it impossible to separately isolate them at ambient temperature. It should be noted that absolute configuration in pentacoordinate stereogenic centres, similarly to tetracoordinate stereogenic centres, is described using the Cahn–Ingold–Prelog (CIP) rules.38 Importantly, enantiomerisation between the two isomers of cis phosphorane requires only one Berry pseudorotation, whereas interconversion between trans enantiomers does not proceed directly, but involves seven pseudorotations and passes through rather unstable intermediates with the key step being the enantiomerisation between the R-cis and S-cis stereoisomers (Fig. 4f).
Pentacoordinate stereogenic centres are of intrinsic interest, not only for their unique properties but also because they have been proposed as potential therapeutics, as well as key intermediates in biological processes and numerous chemical reactions.39–52 However, their enantiopure synthesis remained underexplored. In 1977, Martin and co-workers prepared the first enantioenriched sulfurane based on a chiral auxiliary approach (Fig. 5a).53 Starting from a cumyl Grignard reagent, the stereoselective substitution reaction with menthyl-(S)-benzenesulfinate followed by acidic work-up afforded (S)-sulfoxide with 96% ee. Treatment of that sulfoxide with acetyl chloride gave chlorosulfurane with high enantioenrichment (95% ee). Later, Drabowicz and Martin synthesised optically active sulfuranes bearing tridentate ligands using a resolution procedure with enantiopure 2,2′-dihydroxy-1,1′-binaphthol as a chiral host (Fig. 5b).54 In another study, Drabowicz and Martin described an enantioselective intramolecular condensation of a sulfoxide diol, resulting in a spirosulfurane with two bidentate ligands, albeit with low enantioselectivity (Fig. 5c).55
Following the synthesis of optically active pentacoordinate sulfuranes, another class of pentacoordinate compounds, Martin's phosphoranes, was also prepared in enantioenriched form.56 In 1994, Akiba and co-workers reported the resolution of a racemic mixture of phosphorane derivative using (−)-menthol as a chiral auxiliary (Fig. 6a).57 A diastereomeric mixture of phosphoranes was resolved by fractional crystallisation. The following auxiliary cleavage with LiAlH4 provided phosphoranes with the phosphorus as the single stereogenic centre with high enantiomeric excess. Later, they used (R)-1-phenylethan-1-amine as a chiral auxiliary to obtain a highly enantioenriched hydridophosphorane (Fig. 6b).58 Notably, auxiliary cleavage with MeLi proceeded via the phosphoranide ion and when the reaction was performed at low temperature, no enantiomerisation was observed for this seemingly configurationally labile species. Despite fundamental prospects, the catalytic stereoselective synthesis of pentacoordinate stereogenic centres remained an unaddressed challenge. Recently, we devoted our efforts to investigate the feasibility of catalyst stereocontrol over pentacoordinate stereogenic centres with the possibility for diastereodivergent catalysis (Fig. 7).6 Starting from a stereodynamic hydridophosphorane (ΔG‡93°C = 72 kJ mol−1), a dynamic kinetic resolution process was investigated. Dehydrogenation catalysed by a superbasic bifunctional iminophosphorane thiourea furnished the trans-dioxophosphorane in high yield and with high levels of enantio- and diastereoselectivity. As anticipated, the enantiomer of the trans-product was obtained by using the enantiomeric catalyst. Notably, for a stereodivergent approach, palladium on charcoal was found as an alternative manifold for the dehydrogenation to afford the cis-dioxophosphorane, featuring stereodynamic enantiomerisation, with excellent diastereoselectivity. Control experiments showed that the trans-selective dehydrogenation does not proceed via the cis-diastereomer, suggesting the occurrence of distinct transition states for the trans- and cis-selective dehydrogenations. Therefore, since catalyst control over pentacoordinate phosphorus stereogenic centres is feasible, it is expected that a significantly increased stereochemical space of diverse penta- and hexacoordinate main group species will be selectively accessible by catalysis.
Hexacoordinate stereogenic centres
Hexacoordinate compounds are primarily represented by transition metal complexes and some main group complexes.59–61 Two major types of geometry are known for hexacoordinate compounds: octahedral and trigonal prismatic (Fig. 8a).62 Due to the eclipsing of three pairs of ligands in the trigonal prism, repulsion between ligands is maximised, making this configuration less favourable compared to the octahedral geometry. As a result, the majority of reported hexacoordinate complexes adopt an octahedral geometry. In the simplest case with six monodentate ligands, where only two are distinct, two achiral diastereomers are observed (Fig. 8b). Similar to pentacoordinate stereogenic centres, hexacoordinate ones can exhibit higher-order stereogenicity. With three different ligands, up to eight stereoisomers are viable, represented as four diastereomeric pairs of enantiomers (Fig. 8c). In the case of six distinct ligands, a total of thirty stereoisomers are possible (Fig. 8d). Chelation is also a very common phenomenon in hexacoordinate systems. The IUPAC suggests the following nomenclature for these compounds: the absolute configuration is assigned by the polyhedral symbol, the configuration index and the chirality descriptors.63 However, the use of descriptors for assignment of the relative position of ligands (cis/trans and mer/fac) and the Δ/Λ nomenclature for the description of absolute configuration remained common. With three symmetric bidentate ligands, hexacoordinate stereogenic centre exhibits twofold stereogenicity (Fig. 9a). The introduction of one unsymmetric ligand results in a similar situation (Fig. 9b). With two unsymmetric ligands, the complex possesses sixfold stereogenicity (Fig. 9c). Three diastereomers are described as cis–cis, cis–trans and trans–cis, respectively. For the description of diastereomers in complexes with three unsymmetric ligands, fac (sometimes cis) is used for C3 symmetrical facial diastereomer, where three distinct termini of the bidentate ligand are arranged in all-cis arrangement on a trigonal face of the coordination tetrahedron (Fig. 9d). | ||
| Fig. 8 (a) Common hexacoordinate compounds. (b and c) The stereochemical relationship among isomers of an octahedral hexacoordinate stereogenic centre with six monodentate ligands. | ||
Alternatively, the meridional diastereomer with C1 symmetry (mer or trans) has two of the distinct termini in trans-relation. Interestingly, these complexes have a reduced number of stereoisomers compared to those with two unsymmetric bidentate ligands, with an overall four stereoisomers per stereogenic centre. Among more unusual coordination topologies of stereogenic-at-metal complexes, octahedral complexes with unsymmetric tripodal tetradentate ligand64 and tetrahedral complexes were reported.65 Various mechanisms have been suggested to explain the stereoisomerisation in hexacoordinate systems. Two types of mechanisms can be devised: dissociative, in which isomerisation takes place by the cleavage of the metal-ligand bond and isomerisation by a twisting motion without bond cleavage. In 1912, Alfred Werner proposed the bond rupture mechanism, based on the premise that isomerisation is initiated by a metal-ligand bond cleavage, resulting in a pentacoordinate intermediate.66 The following reattachment of the bidentate ligand provides the rearranged stereoisomer. This mechanism can proceed via trigonal bipyramidal and square pyramidal intermediates or transition states.67–69 In the first case, the initial dissociation of one of the metal-ligand bonds provides a square pyramid with a monodentate ligand, which can be either in equatorial or in axial position (Fig. 10a). The following half Berry pseudorotation yields a trigonal bipyramid, while another half Berry pseudorotation and ligand reattachment provide the hexacoordinate octahedral complex in stereoisomeric form. Notably, it was found that stereoisomerisation via trigonal bipyramid can only take place between certain stereoisomers.67–69
Alternatively, this mechanism can proceed via square pyramidal intermediates (Fig. 10b). Starting from a square pyramidal-basal geometry, instead of half Berry pseudorotation, the basal intermediate undergoes rearrangement to render the square pyramidal-axial intermediate. From these species, two mechanisms have been suggested proceeding through the rotation of the ligand trans to the unidentate ligand. In the first, called primary process, the site trans to the unidentate ligand migrates to the free coordination site, yielding basal ligands in cis-arrangement. Alternatively, the whole bidentate ligand trans to the unidentate ligand can rotate to give basal ligands in trans-arrangement in a secondary process. It is worth noting that both primary and secondary processes can take place from all square pyramidal intermediates.
 | ||
| Fig. 10 Bond rupture mechanisms for the stereoisomerisation of hexacoordinate stereogenic centres: (a) through trigonal bipyramid intermediates; (b) through square pyramid intermediates. | ||
As an alternative to Werner's bond rupture mechanism, Thomas in 1921 suggested an intermolecular dissociative mechanism.70 In this mechanism, both metal-ligand bonds are subsequently cleaved, resulting in a free ligand and a complex that now attains either cis- or trans-configured square planar or tetrahedral geometry. Reattachment of the free ligand results in isomerisation, potentially resulting in all four stereoisomers of the octahedral complex.
Another type of mechanism proceeding without ligand dissociation suggests that stereoisomerisation occurs through a twisting motion of ligands and proceeds through a trigonal prismatic transition state. Two mechanisms have been proposed. In the Bailar twist, three bidentate ligands span the edges between the square faces of the trigonal prism (Fig. 11a).71 In the Rây–Dutt transition state, two of the bidentate ligands occupy the opposite edges of the two trigonal faces and one ligand connects the two remaining corners (Fig. 11b).72 There are two ways to describe the pathway to the Bailar or Rây–Dutt transition state.
 | ||
| Fig. 11 Non-dissociative mechanisms for stereoisomerisation of octahedral tris-chelate complexes: (a) Bailar twist; (b) Rây–Dutt twist. | ||
Bailar originally suggested that a trigonal face of the octahedral complex is rotated by 60°, leading either to the Bailar or Rây–Dutt trigonal prismatic transition state, depending on the face chosen.73 Rây and Dutt and then Springer and Sievers74 illustrated the motion by holding one chelating ligand fixed and rotating the square face spanned by the two remaining bidentate ligands by 45° to reach the trigonal prismatic transition state, followed by another 45° turn to furnish the rearranged product. It is apparent that both mechanisms are essentially identical, as they only appear different because they view the intramolecular twisting process from two different perspectives.73
There are a total of six stereoisomers for the trigonal prismatic transition states of homoleptic tris-chelate complexes with unsymmetric bidentate ligands: two symmetrical isomers and two diastereomeric pairs of enantiomers.68,69 Similar to the bond rupture mechanisms, stereoisomerisation is only accessible among particular stereoisomers. Through the Bailar transition state, only enantiomerisation but not diastereomerisation occurs for both the fac- and mer-diastereomers. Conversely, through the Rây–Dutt transition state, diastereomerisation with inversion of configuration is possible for both diastereomers and enantiomerisation is possible for the mer-diastereomer.
Tris-chelate octahedral species with bidentate ligands are commonly represented by β-diketonate complexes. Configurational stability varies significantly among them, depending on the central atom, solvent and electronic properties of the ligand. For instance, AlIII, GaIII and InIIIcomplexes are known to be stereodynamic at room temperature,75 whereas RhIII, RuIII, CoIII and CrIII are often configurationally stable and can be resolved into their diastereomers and partly into their enantiomers.76 This property is crucial and has to be taken into account when designing stereogenic-at-metal complexes for application in catalysis and medicinal chemistry (vide infra).
It is not a trivial task to distinguish between the different mechanisms when studying any particular case of stereoisomerisation in hexacoordinate stereogenic centres.77 Only the information on the relative rates of interconversion between the stereoisomers can be obtained from experimentally accessible microscopic rate constants. Various intermediates or transition states can explain these relative rates. However, the distinction between inter- and intramolecular mechanisms can be made relatively easily by comparing ligand exchange and stereoisomerisation rates using isotopic labelling studies.
The stereoisomerisation reactions of Co(mhd)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 69 (mhd = 5-methylhexane-2,4-dionate) and Co(bzac)3
69 (mhd = 5-methylhexane-2,4-dionate) and Co(bzac)3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 78 (bzac = benzoylacetonate) have been extensively studied experimentally by a combination of polarimetry and NMR spectroscopy. The stereoisomerisation pathways in these complexes (Fig. 12) can be described by six first-order rate constants, because the enantiomeric pathways such as Λ-mer to Λ-fac and Δ-mer to Δ-fac have identical rates.
78 (bzac = benzoylacetonate) have been extensively studied experimentally by a combination of polarimetry and NMR spectroscopy. The stereoisomerisation pathways in these complexes (Fig. 12) can be described by six first-order rate constants, because the enantiomeric pathways such as Λ-mer to Λ-fac and Δ-mer to Δ-fac have identical rates.
After several independent kinetic measurements, microscopic rate constants were determined for both complexes, showing that diastereomerisation primarily occurs under inversion of configuration and the direct enantiomerisation of the mer-isomer is faster than that of the fac-diastereomer. These observations indicate the Rây–Dutt and Bailar twist mechanisms as major diastereomerisation pathways. For the mer-isomer, isomerisation is possible via both the Rây–Dutt and the Bailar transition states, which can explain its faster enantiomerisation rate compared to a fac-isomer, which can only racemise via the Rây–Dutt twist. However, the observed behaviour can also be explained by a combination of the square pyramidal-axial and trigonal bipyramid-axial bond rupture mechanisms. To differentiate between bond rupture and intramolecular mechanisms, pre-exponential potential Arrhenius factors were analysed. Large values would favour the bond rupture mechanism, whereas the twisting motions are expected to have relatively low factors, as the energy needs to be distributed into specific vibrational modes. As a result, the bond rupture mechanism was deemed the dominant isomerisation pathway for both complexes.
Stereoisomerically pure hexacoordinate metal complexes have found numerous applications in catalysis and medicinal chemistry.79–81 Octahedral complexes with well-defined geometry have, for instance, been identified in metalloenzymes such as hydrogenases.82 In laboratory settings, enantioenriched stereogenic-at-metal complexes are an emerging area of research, particularly in the field of stereoselective catalysis. Two major catalytic modes have been explored – using the ligand sphere for hydrogen bonding catalysis and the stereogenic metal centre itself as a Lewis acid catalyst. In 2008, Ganzmann and Gladysz reported the use of enantioenriched Werner complexes Δ-[Co(en)3](BArF)3 for enantioselective Michael addition of malonates to enones (Fig. 13a).83 Despite only moderate enantioselectivity being achieved, this is the first example of hydrogen bond catalysis by the ligand sphere of a stereogenic-at-metal complex. The introduction of additional stereogenic centres into the ligands allowed to achieve higher enantioselectivity.84 In a series of contributions by the Meggers group, stereogenic metal centres have been utilised as Lewis acid catalysts.79,80 Friedel–Crafts addition of indoles to α,β-unsaturated 2-acylimidazoles were catalysed efficiently by a stereogenic-at-metal iridium complex (Fig. 13b).85 Notably, while configurationally stable (as discussed above), the complex is substitutionally labile due to the presence of two weakly coordinating acetonitrile ligands, which contributes to its reactivity. By another strategy, Meggers used a similar iridium complex for a combination of stereoselective Lewis acid and photoredox catalysis.86 An intermediate enolate complex of the iridium catalyst thereby acted as an efficient photosensitiser, generating reactive electrophilic radicals. For medicinal chemistry, hexacoordinate stereogenic centres were found to selectively bind to DNA through hydrophobic and electrostatic interactions, acting as highly specific intercalators.87–89 The sixfold stereogenic rhodium(III) complex [Rh(MGP)2phi]5+ (MGP = (4-guanidylmethyl)-1,10-phenantroline, phi = phenanthrene-9,10-diimine) can bind to different base sequences depending on the enantiomer. While the Δ-enantiomer displays an affinity for the 5′-CATCTG-3′ sequence, the Λ-enantiomer specifically targets the 5′- CATATG-3′ sequence (Fig. 13c).90
Furthermore, Meggers and co-workers demonstrated the utility of higher-order stereogenic octasporine ruthenium complexes as potent inhibitors of protein kinases. Notably, the complex can exist in up to 20 stereoisomeric forms (10 diastereomeric pairs of enantiomers), and was used in this study as a single stereoisomer (Fig. 13d).91 These applications highlight the importance of the synthetic methods used to obtain these complexes.
Historically, the first preparation of compounds with enantioenriched hexacoordinate stereogenic centers was achieved by Werner via the recrystallisation of a cationic cobalt complex, [Co(en)3]X3, with an enantiopure counterion. The Δ-isomer was crystallised with silver D-tartrate, whereas the Λ-isomer remained in the supernatant (Fig. 14).92 The Lacour group advanced this strategy for the resolution of various other complexes by the stereoselective partitioning between two phases using the lipophilic tris(tetrachloro-benzenediolato)phosphate (TRISPHAT) ion.93–95 The resolution of higher-order hexacoordinate stereogenic centres often requires the separation of diastereomers before the isolation of enantiomers.96 Alternatively, a chiral auxiliary approach can be employed, followed by separation via column chromatography or by utilising the solubility differences of the diastereomeric complexes. Subsequent stereospecific substitution by a ligand provides the stereoisomerically enriched complex with the metal as the only stereogenic centre.97 As a first step, unsymmetric bidentate ligands are introduced to a common metal precursor, such as RhCl3 (Fig. 15).98 A racemic mixture of the most thermodynamically favoured diastereomer is obtained. A chiral ligand is subsequently introduced by the substitution of the labile ligands, followed by resolution either by column chromatography or based on their different solubilities in EtOH. Finally, the chiral auxiliary ligand is replaced by labile ligands under acidic conditions. In a similar fashion, stereoisomerically enriched complexes of Ir,85 Ru,99 Os,100 Fe101 and Co102 were obtained. Besides the salicyloxazoline auxiliary, oxazolines,100 salicylthiazolines85 and amino acid derivatives103 have been shown as efficient resolving agents.97 Furthermore, a kinetic resolution approach can be utilised to separate the enantiomers of the hexacoordinate metal stereogenic centres, as was shown by Baslé and co-workers (Fig. 16).104 A chiral NHC-carboxylate silver complex was used for the transmetalation of a racemic [Ir(µ-Cl)(ppy)2]2 complex, yielding a chiral NHC-Ir(III) complex in >99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 d.r. and the enantioenriched bis-cyclometalated Ir(III) complex in 97
1 d.r. and the enantioenriched bis-cyclometalated Ir(III) complex in 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 e.r. besides that, L-proline was used as the chiral ligand for the resolution of rac-[Ir(µ-Cl)(ppy)2]2105 To eliminate the need for the separation of diastereomers, a metal stereogenic centre can be formed stereoselectively either under kinetic106 or thermodynamic107 control, starting from a complex with a chiral auxiliary as the single bidentate ligand. The latter process begins with the introduction of a chiral auxiliary to [RuCl2(C6H6)]2, followed by photolysis to yield a substitutionally labile complex as a mixture of diastereomers (Ru2, Fig. 17). Subsequently, substitution with the first bipyridine ligand occurs under thermodynamic control, providing Λ-Ru3 as a single diastereomer. The following in situ introduction of the second bipyridine ligand proceeded stereospecifically to yield Λ-Ru4 in a remarkable 250
3 e.r. besides that, L-proline was used as the chiral ligand for the resolution of rac-[Ir(µ-Cl)(ppy)2]2105 To eliminate the need for the separation of diastereomers, a metal stereogenic centre can be formed stereoselectively either under kinetic106 or thermodynamic107 control, starting from a complex with a chiral auxiliary as the single bidentate ligand. The latter process begins with the introduction of a chiral auxiliary to [RuCl2(C6H6)]2, followed by photolysis to yield a substitutionally labile complex as a mixture of diastereomers (Ru2, Fig. 17). Subsequently, substitution with the first bipyridine ligand occurs under thermodynamic control, providing Λ-Ru3 as a single diastereomer. The following in situ introduction of the second bipyridine ligand proceeded stereospecifically to yield Λ-Ru4 in a remarkable 250![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereomeric ratio. The final substitution required acidic conditions for the auxiliary cleavage and proceeded with complete retention of configuration, resulting in a heteroleptic ruthenium complex with 250
1 diastereomeric ratio. The final substitution required acidic conditions for the auxiliary cleavage and proceeded with complete retention of configuration, resulting in a heteroleptic ruthenium complex with 250![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 e.r.107 Notably, the chiral ligand was used only in the first substitution step, and following stereospecific substitutions proceeded smoothly with achiral ligands. Interestingly, the introduction of an additional transiently coordinating thioether ligand to the salicyloxazoline chiral auxiliary led to a stereoselectivity switch under kinetic control in the second substitution step, providing the product with inversion of the configuration at ruthenium.108 Moreover, a catalytic enantioselective synthesis of [Ru(bpy)3](PF6)2 was achieved by the Meggers group by combining the stereoselective binding of a chiral auxiliary to a metal complex with its stereoselective substitution in a single reaction (Fig. 18).109 Starting from the trans-[Ru(bpy)2(MeCN)2](PF6)2, a chiral sulfoxide was used as a nucleophilic catalyst. The reaction is triggered by the substitution of one MeCN ligand by a sulfur centre, leading to destabilisation of the remaining MeCN ligand via the sulfoxide's trans-effect. Following reorganisation of the bipyridine ligands under deprotonation of the phenolic moiety provides the cis-complex with sulfoxide now as a bidentate ligand with exceptional diastereoselectivity. Finally, stereospecific substitution of the sulfoxide yielded the enantioenriched homoleptic [Ru(bpy)3]2+ complex.
1 e.r.107 Notably, the chiral ligand was used only in the first substitution step, and following stereospecific substitutions proceeded smoothly with achiral ligands. Interestingly, the introduction of an additional transiently coordinating thioether ligand to the salicyloxazoline chiral auxiliary led to a stereoselectivity switch under kinetic control in the second substitution step, providing the product with inversion of the configuration at ruthenium.108 Moreover, a catalytic enantioselective synthesis of [Ru(bpy)3](PF6)2 was achieved by the Meggers group by combining the stereoselective binding of a chiral auxiliary to a metal complex with its stereoselective substitution in a single reaction (Fig. 18).109 Starting from the trans-[Ru(bpy)2(MeCN)2](PF6)2, a chiral sulfoxide was used as a nucleophilic catalyst. The reaction is triggered by the substitution of one MeCN ligand by a sulfur centre, leading to destabilisation of the remaining MeCN ligand via the sulfoxide's trans-effect. Following reorganisation of the bipyridine ligands under deprotonation of the phenolic moiety provides the cis-complex with sulfoxide now as a bidentate ligand with exceptional diastereoselectivity. Finally, stereospecific substitution of the sulfoxide yielded the enantioenriched homoleptic [Ru(bpy)3]2+ complex.
 | ||
| Fig. 15 Chiral auxiliary approach for the resolution of racemic mixtures of hexacoordinate stereogenic centres. | ||
 | ||
| Fig. 17 Chiral auxiliary-mediated stereoselective substitution under thermodynamic control for the synthesis of heteroleptic ruthenium complex. | ||
Several important factors had to be taken into consideration to render this catalytic process feasible. Firstly, the cis-configuration of the complex is favoured not only thermodynamically, but also by the condition that the sulfoxide ligand is only able to coordinate in a bidentate fashion to the cis-complex. Secondly, the reaction with sulfoxide is significantly faster than with bpy, which is monoprotonated under the acidic reaction conditions and thus less reactive. Additionally, acidic conditions promote the substitution of the sulfoxide, thus allowing a full catalytic cycle. Notably, only 10% of the starting material is dissolved in ethylene glycol under the reaction conditions, ensuring that the catalyst (20 mol%) is always present in excess. Therefore, despite being the first catalytic method to control octahedral stereogenic centres, the solubility effect and the need for precise pH control somewhat limit the generality of this protocol.
A unique feature of oxidation state-dependent stereodynamics of some metal stereogenic centres has been utilised for the stereoselective preparation of hexacoordinate complexes. Based on the interconversion between stereodynamic Co(II) and configurationally stable Co(III) species, a deracemisation approach has been developed for the synthesis of Co(III) complexes in enantioenriched form. Deracemisation is the process of converting a racemic mixture into a single enantiomer without altering the structure of the molecule. This process is inherently endergonic, as the racemate has higher entropy due to a greater number of states compared to the enantioenriched compound (ΔS > 0). Consequently, deracemisations have a positive free enthalpy (ΔG = −T·ΔS > 0). To make this a thermodynamically favourable process with a net ΔG < 0, energy must be supplied either through a coupled exergonic stoichiometric reaction110,111 or by photonic energy.110–114 Additionally, the presence of a chiral catalyst is required to control the direction of deracemisation. Based on these principles, the photocatalytic deracemisation of Co(acac)3 has been developed by Hamada and Sakaki (Fig. 19a).115,116 The stereogenic-at-metal and chiral auxiliary-supported Ru(II) photocatalyst is responsible for both the conversion of photonic energy and the discrimination between enantiomers. The reaction involves the excitation of the photocatalyst by visible light, followed by the quenching of its excited state by Co(acac)3, which generates the Ru(III) photocatalyst and the Co(II) complex, Co(acac)3−. The cobalt centre is rendered stereodynamic and can equilibrate either through direct epimerisation in the presence of the chiral photocatalyst or via a Co(acac)2 intermediate. Stereoinduction may arise from the differing stabilities of diastereomeric Ru(III)–Co(II) contact ion pairs. Finally, Co(II) is likely oxidised by Ru(III) via single-electron transfer, yielding Co(acac)3 in enantioenriched form (51% ee), thus regenerating the Ru(II) photocatalyst. This pioneering method addressed twofold stereogenicity of the homoleptic cobalt complex with symmetric ligands.
 | ||
| Fig. 19 (a) Photocatalytic deracemisation of Co(acac)3. (b) Unsymmetric congeners for catalyst-control over higher-order stereogenicity. | ||
Inspired by this strategy, our group investigated the photocatalytic deracemisation of Co(III) complexes with fourfold stereogenicity (Fig. 19b).5 A series of homoleptic Co(III) complexes with unsymmetrical β-diketonate ligands (Fig. 12) was used in these studies under similar conditions. Starting from the rac-mer complex, enantioenrichments of up to 88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 e.r. for mer- and up to 82
12 e.r. for mer- and up to 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 e.r. for fac-diastereomer were obtained. Interestingly, in some cases, a distribution of diastereomers distinct from the thermodynamic ratio was observed upon reaching the photostationary state. Complementary to direct deracemisation process, a two-step strategy utilizing chiral auxiliary has been developed by the Meggers group for preparation of enantioenriched ruthenium complexes starting from a racemic mixture.117
18 e.r. for fac-diastereomer were obtained. Interestingly, in some cases, a distribution of diastereomers distinct from the thermodynamic ratio was observed upon reaching the photostationary state. Complementary to direct deracemisation process, a two-step strategy utilizing chiral auxiliary has been developed by the Meggers group for preparation of enantioenriched ruthenium complexes starting from a racemic mixture.117
In summary, three major approaches exist for the preparation of enantioenriched complexes of hexacoordinate stereogenic centres. The earliest approaches rely on the resolution of stereoisomeric mixtures by using chiral counterions for the fractional crystallisation or specific phase partitioning. A versatile and general approach involves the use of a chiral auxiliary, enabling the separation of isomers by column chromatography or based on differences in solubility.
Further variations include kinetic resolutions or a sequence of stereoselective or stereospecific substitutions controlled by a preinstalled auxiliary. Finally, first examples of direct catalytic strategies, including deracemisation, emerged, which involve the use of transient auxiliaries and photocatalysis.
Conclusions
Penta- and hexacoordinate stereogenic centres possessing higher-order stereogenicity encode a significantly increased stereochemical space. This unique propensity suggests their potential use in diverse areas where having more than two isomeric states per one stereogenic centre is beneficial. For instance, applications in molecular information science, storage or molecular switching can be envisioned. Catalysis is another intriguing application of configurationally-defined stereogenic centres beyond tetracoordination, with the possibility for more than two isomers of a catalyst structure per one stereogenic centre. Despite this potential, only a few methods exist for stereoselective synthesis with enantio- and diastereoselectivity to control the configuration of one stereogenic centre. With the feasibility of catalyst-stereocontrol over pentacoordinate stereogenic centres, it is expected that considerable efforts will be devoted to the stereoselective synthesis of higher-order stereogenic centres and methods that allow to selectively navigate, by means of diastereodivergent catalysis, the significantly increased stereochemical space created by stereogenic centers beyond tetracoordination.118Author contributions
Anton Budeev: conceptualization, draft preparation, reviewing & editing, visualization. Christof Sparr: supervision, reviewing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This project has received funding from the European Research Council (ERC) under the European Union's Horizon 2020 research and innovation program (grant agreement No. 101002471) and from the Swiss National Science Foundation (10001653). We thank Dr T. A. Schmidt and the other members of the Sparr group for helpful discussions.Notes and references
- J. A. Le Bel, Bull. Soc. Chim. Fr., 1874, 22, 337–347 Search PubMed.
- J. H. van ’t Hoff, Arch. Neerl. Sci. Exactes Nat., 1874, 9, 445–454 Search PubMed.
- S. Kojima, K. Kajiyama, M. Nakamoto and K. Akiba, J. Am. Chem. Soc., 1996, 118, 12866–12867 CrossRef CAS.
- S. Kojima, K. Kajiyama, M. Nakamoto, S. Matsukawa and K. Akiba, Eur. J. Org Chem., 2006, 2006, 218–234 CrossRef.
- T. A. Schmidt and C. Sparr, Chem. Commun., 2022, 58, 12172–12175 RSC.
- A. Budeev, J. Dong, D. Häussinger and C. Sparr, Nat. Commun., 2023, 14, 8013 CrossRef CAS PubMed.
- T. A. Schmidt and C. Sparr, Acc. Chem. Res., 2021, 54, 2764–2774 CrossRef CAS PubMed.
- T. A. Schmidt, S. Schumann, A. Ostertag and C. Sparr, Angew. Chem., Int. Ed., 2023, 62, e202302084 CrossRef CAS PubMed.
- X. Wu, R. M. Witzig, R. Beaud, C. Fischer, D. Häussinger and C. Sparr, Nat. Catal., 2021, 4, 457–462 CrossRef CAS.
- T. A. Schmidt and C. Sparr, Angew. Chem., Int. Ed., 2021, 60, 23911–23916 CrossRef CAS PubMed.
- L. Greb and J.-M. Lehn, J. Am. Chem. Soc., 2014, 136, 13114–13117 CrossRef CAS PubMed.
- J.-H. Li, X.-K. Li, J. Feng, W. Yao, H. Zhang, C.-J. Lu and R.-R. Liu, Angew. Chem., Int. Ed., 2024, 63, e202319289 CrossRef CAS PubMed.
- S. Jia, Y. Hao, Y. Li and Y. Lan, Nat. Rev. Chem., 2025, 9, 617–633 CrossRef PubMed.
- E. L. Muetterties and R. A. Schunn, Q. Rev., Chem. Soc., 1966, 20, 245–299 RSC.
- R. R. Holmes and J. A. Deiters, J. Am. Chem. Soc., 1977, 99, 3318–3326 CrossRef CAS.
- J. D. Dunitz and V. Prelog, Angew Chem. Int. Ed. Engl., 1968, 7, 725–726 CrossRef CAS.
- R. S. Berry, J. Chem. Phys., 1960, 32, 933–938 CrossRef CAS.
- P. C. Lauterbur and F. Ramirez, J. Am. Chem. Soc., 1968, 90, 6722–6726 CrossRef CAS.
- E. L. Muetterties, J. Am. Chem. Soc., 1969, 91, 4115–4122 CrossRef CAS.
- I. Ugi, D. Marquarding, H. Klusacek, P. Gillespie and F. Ramirez, Acc. Chem. Res., 1971, 4, 288–296 CrossRef CAS.
- P. Gillespie, P. Hoffman, H. Klusacek, D. Marquarding, S. Pfohl, F. Ramirez, E. A. Tsolis and I. Ugi, Angew Chem. Int. Ed. Engl., 1971, 10, 687–715 CrossRef CAS.
- J. A. Altmann, K. Yates and I. G. Csizmadia, J. Am. Chem. Soc., 1976, 98, 1450–1454 CrossRef CAS.
- W. Kutzelnigg and J. Wasilewski, J. Am. Chem. Soc., 1982, 104, 953–960 CrossRef CAS.
- E. P. A. Couzijn, J. C. Slootweg, A. W. Ehlers and K. Lammertsma, J. Am. Chem. Soc., 2010, 132, 18127–18140 CrossRef CAS PubMed.
- P. J. Canfield, J. R. Reimers and M. J. Crossley, ACS Org. Inorg. Au, 2024, 4, 356–372 CrossRef CAS PubMed.
- A. Caligiana, V. Aquilanti, R. Burcl, N. C. Handy and D. P. Tew, Chem. Phys. Lett., 2003, 369, 335–344 CrossRef CAS.
- L. J. P. van der Boon, L. van Gelderen, T. R. de Groot, M. Lutz, J. C. Slootweg, A. W. Ehlers and K. Lammertsma, Inorg. Chem., 2018, 57, 12697–12708 CrossRef CAS PubMed.
- J. C. Martin, Science, 1983, 221, 509–514 CrossRef CAS PubMed.
- E. L. Muetterties, W. Mahler and R. Schmutzler, Inorg. Chem., 1963, 2, 613–618 CrossRef CAS.
- E. L. Muetterties, W. Mahler, K. J. Packer and R. Schmutzler, Inorg. Chem., 1964, 3, 1298–1303 CrossRef CAS.
- R. M. Beesley, C. K. Ingold and J. F. Thorpe, J. Chem. Soc. Trans., 1915, 107, 1080–1106 RSC.
- T. T. Nguyen and J. C. Martin, J. Am. Chem. Soc., 1980, 102, 7382–7383 CrossRef CAS.
- J. C. Martin and E. F. Perozzi, Science, 1976, 191, 154–159 CrossRef CAS PubMed.
- E. F. Perozzi and J. C. Martin, J. Am. Chem. Soc., 1979, 101, 1591–1593 CrossRef CAS.
- T. Deis, J. Maury, F. Medici, M. Jean, J. Forte, N. Vanthuyne, L. Fensterbank and G. Lemière, Angew. Chem., 2022, 134, e202113836 CrossRef.
- K. Kajiyama, M. Yoshimune, M. Nakamoto, S. Matsukawa, S. Kojima and K. Akiba, Org. Lett., 2001, 3, 1873–1875 CrossRef CAS PubMed.
- K. Kajiyama, M. Yoshimune, S. Kojima and K. Akiba, Eur. J. Org Chem., 2006, 2006, 2739–2746 CrossRef.
- N. G. Connelly, T. Damhus, R. M. Hartshorn and A. T. Hutton, Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005, Royal Society of Chemistry, Cambridge, UK, 2005, pp. 175–193 Search PubMed.
- V. V. Negrebetsky and A. A. Korlyukov, in Organogermanium Compounds: Theory, Experiment, and Applications, ed. V. Y. Lee, John Wiley & Sons, Inc., Hoboken, NJ, 1st edn, 2023, ch. 15, vol. 2, pp. 629–666 Search PubMed.
- K. Akiba, Heteroat. Chem., 2011, 22, 207–274 CrossRef CAS.
- F. H. Westheimer, Acc. Chem. Res., 1968, 1, 70–78 CrossRef CAS.
- E. Procházková, R. Navrátil, Z. Janeba, J. Roithová and O. Baszczyňski, Org. Biomol. Chem., 2019, 17, 315–320 RSC.
- T. Kawashima, K. Kato and R. Okazaki, J. Am. Chem. Soc., 1992, 114, 4008–4010 CrossRef CAS.
- N. Kano, X. J. Hua, S. Kawa and T. Kawashima, Tetrahedron Lett., 2000, 41, 5237–5241 CrossRef CAS.
- K. Ando, J. Org. Chem., 1999, 64, 6815–6821 CrossRef CAS PubMed.
- H. J. Bestmann and F. Seng, Angew Chem. Int. Ed. Engl., 1963, 2, 393 CrossRef.
- T. Kawashima, T. Soda and R. Okazaki, Angew Chem. Int. Ed. Engl., 1996, 35, 1096–1098 CrossRef CAS.
- F. Ramirez, Acc. Chem. Res., 1968, 1, 168–174 CrossRef CAS.
- E. J. Miller, W. Zhao, J. D. Herr and A. T. Radosevich, Angew. Chem., Int. Ed., 2012, 51, 10605–10609 CrossRef CAS PubMed.
- A. Calcatelli, R. M. Denton and L. T. Ball, Org. Lett., 2022, 24, 8002–8007 CrossRef CAS PubMed.
- I. Granoth, J. Chem. Soc. Perkin Trans. I, 1982, 735–740 RSC.
- N. Satish Kumar, P. Kommana, J. J. Vittal and K. C. Kumara Swamy, J. Org. Chem., 2002, 67, 6653–6658 CrossRef CAS PubMed.
- J. C. Martin and T. M. Balthazor, J. Am. Chem. Soc., 1977, 99, 152–162 CrossRef CAS.
- J. Drabowicz and J. C. Martin, Tetrahedron Asymmetry, 1993, 4, 297–300 CrossRef CAS.
- J. Drabowicz and J. C. Martin, Pure Appl. Chem., 1996, 68, 951–956 CrossRef CAS.
- D. Krasowska, J. Chrzanowski, P. Kiełbasiński and J. Drabowicz, Molecules, 2016, 21, 1573 CrossRef PubMed.
- S. Kojima, K. Kajiyama and K. Akiba, Tetrahedron Lett., 1994, 35, 7037–7040 CrossRef CAS.
- S. Kojima, K. Kajiyama and K. Akiba, Bull. Chem. Soc. Jpn., 1995, 68, 1785–1797 CrossRef CAS.
- V. P. Solov’ev, M. S. Stuklova, E. V. Koltunova and N. N. Kochanova, Russ. J. Coord. Chem., 2003, 29, 660–668 CrossRef.
- M. Dudev, J. Wang, T. Dudev and C. Lim, J. Phys. Chem. B, 2006, 110, 1889–1895 CrossRef CAS PubMed.
- A. Werner, Helv. Chim. Acta, 1918, 1, 5–32 CrossRef CAS.
- R. Hoffmann, J. M. Howell and A. R. Rossi, J. Am. Chem. Soc., 1976, 98, 2484–2492 CrossRef CAS.
- N. G. Connelly, T. Damhus, R. M. Hartshorn and A. T. Hutton, Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005, Royal Society of Chemistry, Cambridge, UK, 2005, pp. 174–198 Search PubMed.
- M. Carmona, R. Rodríguez, V. Passarelli, F. J. Lahoz, P. García-Orduña and D. Carmona, J. Am. Chem. Soc., 2018, 140, 912–915 CrossRef CAS PubMed.
- K. Endo, Y. Liu, H. Ube, K. Nagata and M. Shionoya, Nat. Commun., 2020, 11, 6263 CrossRef CAS PubMed.
- A. Werner, Ber. Dtsch. Chem. Ges., 1912, 45, 3061–3070 CrossRef.
- C. S. Springer, J. Am. Chem. Soc., 1973, 95, 1459–1467 CrossRef CAS.
- E. L. Muetterties, J. Am. Chem. Soc., 1968, 90, 5097–5102 CrossRef CAS.
- J. G. Gordon II and R. H. Holm, J. Am. Chem. Soc., 1970, 92, 5319–5332 CrossRef.
- W. Thomas, J. Chem. Soc. Trans., 1921, 119, 1140–1145 RSC.
- J. C. Bailar, J. Inorg. Nucl. Chem., 1958, 8, 165–175 CrossRef CAS.
- P. Rây and N. K. Dutt, J. Indian Chem. Soc., 1943, 20, 81–92 Search PubMed.
- J. E. Brady, Inorg. Chem., 1969, 8, 1208–1209 CrossRef CAS.
- C. S. Springer Jr and R. E. Sievers, Inorg. Chem., 1967, 6, 852–854 CrossRef.
- R. C. Fay and T. S. Piper, Inorg. Chem., 1964, 3, 348–356 CrossRef.
- R. C. Fay, A. Y. Girgis and U. Klabunde, J. Am. Chem. Soc., 1970, 92, 7056–7060 CrossRef CAS.
- N. Serpone and D. G. Bickley, in Progress in Inorganic Chemistry: Inorganic Reaction Mechanisms: Part II, ed. J. O. Edwards, John Wiley & Sons, Inc, New York, 1972, ch. 7, vol. 17, pp. 391–566 Search PubMed.
- R. C. Fay and A. Y. Girgis, J. Am. Chem. Soc., 1970, 92, 7061–7072 CrossRef CAS.
- E. Meggers, Angew. Chem., Int. Ed., 2017, 56, 5668–5675 CrossRef CAS PubMed.
- P. S. Steinlandt, L. Zhang and E. Meggers, Chem. Rev., 2023, 123, 4764–4794 CrossRef CAS PubMed.
- P. Dey, P. Rai and B. Maji, ACS Org. Inorg. Au, 2022, 2, 99–125 CrossRef CAS PubMed.
- S. Shima, O. Pilak, S. Vogt, M. Schick, M. S. Stagni, W. Meyer-Klaucke, E. Warkentin, R. K. Thauer and U. Ermler, Science, 2008, 321, 572–575 CrossRef CAS PubMed.
- C. Ganzmann and J. A. Gladysz, Chem. – Eur. J., 2008, 14, 5397–5400 CrossRef CAS PubMed.
- K. G. Lewis, S. K. Ghosh, N. Bhuvanesh and J. A. Gladysz, ACS Cent. Sci., 2015, 1, 50–56 CrossRef CAS.
- H. Huo, C. Fu, K. Harms and E. Meggers, J. Am. Chem. Soc., 2014, 136, 2990–2993 CrossRef CAS.
- H. Huo, X. Shen, C. Wang, L. Zhang, P. Röse, L.-A. Chen, K. Harms, M. Marsch, G. Hilt and E. Meggers, Nature, 2014, 515, 100–103 CrossRef CAS.
- K. E. Erkkila, D. T. Odom and J. K. Barton, Chem. Rev., 1999, 99, 2777–2796 CrossRef CAS.
- R. H. Terbrueggen and J. K. Barton, Biochemistry, 1995, 34, 8227–8234 CrossRef CAS.
- S. J. Franklin and J. K. Barton, Biochemistry, 1998, 37, 16093–16105 CrossRef CAS.
- R. H. Terbrueggen, T. W. Johann and J. K. Barton, Inorg. Chem., 1998, 37, 6874–6883 CrossRef CAS PubMed.
- L. Feng, Y. Geisselbrecht, S. Blanck, A. Wilbuer, G. E. Atilla-Gokcumen, P. Filippakopoulos, K. Kräling, M. A. Celik, K. Harms, J. Maksimoska, R. Marmorstein, G. Frenking, S. Knapp, L.-O. Essen and E. Meggers, J. Am. Chem. Soc., 2011, 133, 5976–5986 CrossRef CAS PubMed.
- A. Werner, Ber. Dtsch. Chem. Ges., 1912, 45, 121–130 CrossRef CAS.
- J. Lacour, C. Ginglinger, C. Grivet and G. Bernardinelli, Angew Chem. Int. Ed. Engl., 1997, 36, 608–610 CrossRef CAS.
- F. Favarger, C. Goujon-Ginglinger, D. Monchaud and J. Lacour, J. Org. Chem., 2004, 69, 8521–8524 CrossRef CAS.
- J. Lacour, C. Goujon-Ginglinger, S. Torche-Haldimann and J. J. Jodry, Angew. Chem., Int. Ed., 2000, 39, 3695–3697 CrossRef CAS PubMed.
- A. Werner, Ber. Dtsch. Chem. Ges., 1911, 44, 2445–2455 CrossRef.
- L. Zhang and E. Meggers, Chem. Soc. Rev., 2025, 54, 1986–2005 RSC.
- J. Ma, X. Shen, K. Harms and E. Meggers, Dalton Trans., 2016, 45, 8320–8323 RSC.
- Y. Zheng, Y. Tan, K. Harms, M. Marsch, R. Riedel, L. Zhang and E. Meggers, J. Am. Chem. Soc., 2017, 139, 4322–4325 CrossRef CAS PubMed.
- G. Wang, Z. Zhou, X. Shen, S. Ivlev and E. Meggers, Chem. Commun., 2020, 56, 7714–7717 RSC.
- Y. Hong, L. Jarrige, K. Harms and E. Meggers, J. Am. Chem. Soc., 2019, 141, 4569–4572 CrossRef CAS PubMed.
- S.-Y. Yao, M. Villa, Y. Zheng, A. Fiorentino, B. Ventura, S. I. Ivlev, P. Ceroni and E. Meggers, Nat. Commun., 2025, 16, 6635 CrossRef CAS PubMed.
- C. Wang, L.-A. Chen, H. Huo, X. Shen, K. Harms, L. Gong and E. Meggers, Chem. Sci., 2015, 6, 1094–1100 RSC.
- R. Manguin, D. Pichon, R. Tarrieu, T. Vives, T. Roisnel, V. Dorcet, C. Crévisy, K. Miqueu, L. Favereau, J. Crassous, M. Mauduit and O. Baslé, Chem. Commun., 2019, 55, 6058–6061 RSC.
- S.-Y. Yao, Y.-L. Ou and B.-H. Ye, Inorg. Chem., 2016, 55, 6018–6026 CrossRef CAS PubMed.
- M. Kraack, K. Harms and E. Meggers, Organometallics, 2013, 32, 5103–5113 CrossRef CAS.
- L. Gong, S. P. Mulcahy, D. Devarajan, K. Harms, G. Frenking and E. Meggers, Inorg. Chem., 2010, 49, 7692–7699 CrossRef CAS PubMed.
- L.-A. Chen, J. Ma, M. A. Celik, H.-L. Yu, Z. Cao, G. Frenking, L. Gong and E. Meggers, Chem. – Asian J., 2012, 7, 2523–2526 CrossRef CAS PubMed.
- L. Gong, Z. Lin, K. Harms and E. Meggers, Angew. Chem., Int. Ed., 2010, 49, 7955–7957 CrossRef CAS PubMed.
- M. Huang, T. Pan, X. Jiang and S. Luo, J. Am. Chem. Soc., 2023, 145, 10917–10929 CrossRef CAS.
- K.-K. Qiao, G.-S. Feng and L. Shi, J. Catal., 2023, 422, 99–116 CrossRef CAS.
- J. Großkopf and T. Bach, Angew. Chem., Int. Ed., 2023, 62, e202308241 CrossRef PubMed.
- J. Wang, X. Lv and Z. Jiang, Chem. – Eur. J., 2023, 29, e202204029 CrossRef CAS PubMed.
- C. Onneken, T. Morack, J. Soika, O. Sokolova, N. Niemeyer, C. Mück-Lichtenfeld, C. G. Daniliuc, J. Neugebauer and R. Gilmour, Nature, 2023, 621, 753–759 CrossRef CAS.
- T. Hamada, H. Ohtsuka and S. Sakaki, Chem. Lett., 2000, 29, 364–365 CrossRef.
- T. Hamada, H. Ohtsuka and S. Sakaki, J. Chem. Soc., Dalton Trans., 2001, 928–934 RSC.
- D. Baran, S. I. Ivlev and E. Meggers, Organometallics, 2021, 41, 52–59 CrossRef.
- D. Moser, T. A. Schmidt and C. Sparr, JACS Au, 2023, 3, 2612–2630 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |