Open Access Article
Open Access ArticleLinear and branched supramolecular polymers formed from isomeric monomers as revealed by solution viscoelasticity
Yufuka Furukawa†
a,
Ren Sato†b,
Natsumi Fukaya a,
Takashi Kajitanic,
Takuya Katashima*b and
Kazunori Sugiyasu
a,
Takashi Kajitanic,
Takuya Katashima*b and
Kazunori Sugiyasu *a
*a
aDepartment of Polymer Chemistry, Graduate School of Engineering, Kyoto University, Kyotodaigaku-katsura, Saikyo-ku, Kyoto 615-8510, Japan. E-mail: sugiyasu.kazunori.8z@kyoto-u.ac.jp
bDepartment of Bioengineering, Graduate School of Engineering, The University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo 113-8656, Japan. E-mail: katashima@g.ecc.u-tokyo.ac.jp
cCore Facility Center, Research Infrastructure Management Center, Institute of Science Tokyo, 4259 Nagatsuta, Midori-ku, Yokohama 226-8501, Japan
First published on 14th April 2026
Abstract
Supramolecular polymers (SPs) assembled through non-covalent interactions provide a promising platform for designing soft materials with dynamic and tunable properties. However, the viscoelastic properties of their solutions remain poorly understood, largely due to the tendency of SP chains to bundle into thick supramolecular fibers, often resulting in precipitation or gelation. In this study, we show that bundle formation can be effectively suppressed by appropriate design of the surface of SPs through alteration of their side chains. Structural analyses revealed that the resulting SPs are homogeneously solvated, permitting their rheological characterization. Notably, we found that even subtle modifications, such as positional isomerism of the side chains, can dramatically alter the physical properties of SP solutions. Our findings, therefore, highlight the critical role of side chains in governing hierarchical structures and macroscopic properties of SPs, offering a design strategy for engineering functional supramolecular materials.
Introduction
A discotic molecule equipped with properly designed hydrogen bonding sites at its periphery can undergo spontaneous self-assembly to form a one-dimensional (1D) columnar polymeric array, thereby forming a supramolecular polymer (SP) chain.1 Supramolecular polymerization of such monomers is well understood on the basis of a nucleation–elongation mechanism.2 In addition, under kinetic control, living supramolecular polymerization is possible, which permits precise control of the polymer chain length3 and the creation of block architectures.4 As such, SPs can nowadays be rationally designed from the monomer level. However, at the polymer level, the design principles that govern the behavior of SP chains in solution, particularly with respect to their viscoelastic properties, remain poorly understood.2,5–11 ‡One difficulty in investigating the solution viscoelasticity of SPs lies in their propensity to bundle into thick 1D fibers, rather than to exist as individually solvated polymeric chains (Fig. 1a). This bundling behavior is probably associated with a polymerization mechanism that resembles the crystallization of a small molecule, i.e., a nucleation–elongation process. Consequently, SPs in solution often form precipitates or colloidal dispersions that take the form of physical gels, commonly referred to as low-molecular-weight gels (LMWGs).12 Rheological studies of LMWGs have been extensively conducted to assess their mechanical stability13 and to determine critical gelation parameters, including the gelation temperature,14 concentration,15 and time.16 Although LMWGs hold promise for a wide range of applications,12,17 their inherent bundling and gelation phenomena significantly impede the investigation of SP chains within the framework of conventional polymer physics.
Tuning the solvent polarity based on solubility parameters would prevent SP chains from bundling.18 As an alternative strategy, we recently demonstrated that bundling of SPs can be effectively suppressed by an appropriate design of the surface of the SP chain.19 Specifically, by introducing a degree of randomness into the lengths of the alkyl side chains covering the SP surface, we were able to obtain a homogeneous solution of individually separated SP chains, without gelation, even at relatively high concentrations (∼mM). This, in turn, permitted solution spinning of the resulting SP. We anticipated that this strategy might allow us to study the solution viscoelasticity of SPs. To expand our molecular-design concept, we employed a branched alkyl chain. Branched alkyl groups have previously been used as side chains to improve the solubilities of rigid π-conjugated polymers.20 Moreover, branched alkyl chains have been used as “entropic ligands” on nanocrystal surfaces, where the ligands harvest conformational entropy in solution and disrupt the crystalline interdigitation typically observed with linear alkyl chains, thereby enhancing the colloidal solubility.21 We hoped that incorporating such side chains onto the surface of SPs might similarly suppress the bundling of the SP chains.
In this study, we designed three monomers based on triphenylamine with distinct alkyl side chains:22,23 D345, which carries linear dodecyl (D) chains, and DT34 and DT35, which carry branched 2-decyltetradecyl (DT) chains (Fig. 1b). The numerical subscripts indicate the positions of the alkyl chains on the benzamide groups. Whereas the SPs derived from D345 showed heavy bundling and formed an LMWG, those consisting of DT34 and DT35 were homogeneously soluble in aliphatic solvents. Although common characterization techniques for SPs—such as variable-temperature absorption spectroscopy, atomic force microscopy, and X-ray scattering—could not clearly distinguish the SPs formed from DT34 and DT35, the flow dynamics of these solutions showed marked differences. Rheological investigations revealed that whereas the SP from DT34 showed a similar behavior to a “linear” covalent polymer in exhibiting reptation-driven relaxation, the SP from DT35 showed “branched” covalent polymer-like dynamics, dominated by arm retraction. These findings demonstrate that engineering the surface of SPs—even by such subtle modifications as positional isomerism of the side chains—can dramatically influence the physical properties of SP chains.
Results
Characterization of D345 gel
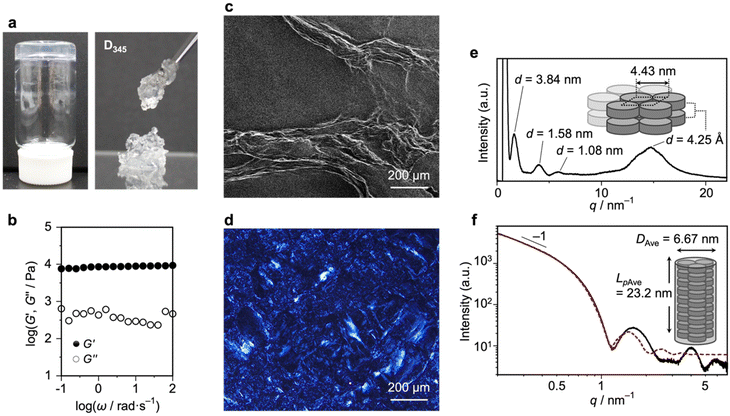
We first investigated the supramolecular polymerization of D345 as a reference monomer bearing commonly used linear alkyl chains (i.e., dodecyl chains). D345 was dissolved in hot dodecane to form a 10 mM solution, and the solution was subsequently cooled to ambient temperature. As observed for other discotic monomers,24 D345 formed an LMWG (Fig. 2a; left). Complementary oscillatory rheology measurements revealed that the storage modulus (G′) exceeded the loss modulus (G″) across the entire frequency range examined (0.1–100 rad s−1), confirming the solid-like nature of this material (Fig. 2b). However, as often observed for LMWGs, the gel formed by D345 was mechanically fragile and, upon the application of a mechanical stress, it readily collapsed and released the solvent (Fig. 2a; right).Scanning electron microscopy (SEM) of the dried gel revealed the formation of thick supramolecular fibers composed of bundled SPs (Fig. 2c). Polarized optical microscopy (POM) images of the gel exhibited birefringence at the microscopic scale, indicating the presence of large crystalline aggregates (Fig. 2d). Under diluted conditions (∼5.0 × 10−5 M), the supramolecular polymers of D345 precipitated, most likely due to their crystalline nature SI; Fig. S1. To further characterize the structure of the supramolecular fibers in the gel state, wide-angle X-ray scattering (WAXS) and small-angle X-ray scattering (SAXS) measurements were performed. The WAXS profile of the D345 gel showed scattering peaks at q = 1.67 (d = 3.84 nm), 4.08 (d = 1.58 nm), and 5.83 nm−1 (d = 1.08 nm) (Fig. 2e). The ratio of these q values approximates to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) √7
√7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) √12, which is consistent with a hexagonal packing of the SPs with a calculated diameter of 4.43 nm (the diameter of D345 estimated from molecular model is 5.0 nm (Fig. S3a)). A peak at q = 14.71 nm−1 (d = 4.25 Å) was attributed to the distance between nitrogen centers of the stacked triphenylamine units, in agreement with previous reports.22 The stacking appeared to be stabilized by intermolecular hydrogen bonding of the amide groups, as suggested by the observation of a N–H stretching vibration at 3265 cm−1 in the FT-IR spectrum (SI; Fig. S2). The SAXS profile of D345 exhibited a slope of −1 in the low-q region, characteristic of a cylindrical structure (Fig. 2f).25 A cylindrical model fitted to the SAXS data yielded an average diameter (DAve) of 6.67 nm and an average persistence length (LpAve) of 23.2 nm (SI; Table S1). Collectively, these results show that the SPs of D345, which are covered with linear dodecyl chains, are prone to bundle into thick supramolecular fibers, thereby forming an organogel.
√12, which is consistent with a hexagonal packing of the SPs with a calculated diameter of 4.43 nm (the diameter of D345 estimated from molecular model is 5.0 nm (Fig. S3a)). A peak at q = 14.71 nm−1 (d = 4.25 Å) was attributed to the distance between nitrogen centers of the stacked triphenylamine units, in agreement with previous reports.22 The stacking appeared to be stabilized by intermolecular hydrogen bonding of the amide groups, as suggested by the observation of a N–H stretching vibration at 3265 cm−1 in the FT-IR spectrum (SI; Fig. S2). The SAXS profile of D345 exhibited a slope of −1 in the low-q region, characteristic of a cylindrical structure (Fig. 2f).25 A cylindrical model fitted to the SAXS data yielded an average diameter (DAve) of 6.67 nm and an average persistence length (LpAve) of 23.2 nm (SI; Table S1). Collectively, these results show that the SPs of D345, which are covered with linear dodecyl chains, are prone to bundle into thick supramolecular fibers, thereby forming an organogel.
Characterization of SPs of DT34 and DT35
The D345 gel retained its shape for over a month without flowing (Fig. 3e). In sharp contrast to this observation, solutions of both DT34 and DT35 (10 mM in dodecane) did not undergo gelation and remained in a fluid state (Fig. 3a–d). At first glance, a noticeable difference was observed in the viscosities of these solutions (discussed later). POM observations revealed no evidence of the presence of large aggregates (SI; Fig. S4), suggesting that SP chains of DT34 and DT35 remained well dispersed in the solutions.The WAXS patterns of the solutions showed a reflection at q = 14.52 nm−1 (d = 4.34 Å) for DT34 and at q = 14.52 nm−1 (d = 4.32 Å) for DT35, corresponding to the N–N stacking distance (Fig. 4b). In addition, FT-IR spectra of DT34 and DT35 showed a N–H stretching band at 3304 cm−1 and 3300 cm−1, confirming the formation of intermolecular hydrogen bonds (SI; Fig. S5). These results suggest that both DT34 and DT35 form 1D polymeric arrays, as was the case for D345. In fact, the SAXS profiles of 10 mM solutions of these two SPs exhibited a slope of −1 in the low-q region (q < 0.5 nm−1), indicative of cylindrical structures (Fig. 4c). Importantly, the SAXS data fitted to a cylindrical model gave LpAve = 17.7 nm and DAve = 2.81 nm for DT34 and LpAve = 16.9 nm and DAve = 2.65 nm for DT35 (SI; Table S1). The diameters of the SPs formed from DT34 and DT35, therefore, matched the dimensions of the aromatic cores in the monomers (Fig. 4a and SI; S3b), indicating that these SPs exist as individually solvated chains in solution without bundling. In fact, unlike the case of D345 (Fig. 2e), no crystalline diffraction peaks attributable to the hexagonal packing of the SP chains were observed in the WAXS profiles of the SPs (Fig. 4b). The longer LpAve of D345 compared to those of DT34 and DT35 is consistent with the bundled, and thus stiff supramolecular fibers consisting of D345. Fig. 4d and e show atomic-force microscopy (AFM) images of the SP chains. We surmise that the observed network-like structures were formed during the sample preparation process, specifically, during spin-coating onto the silicon wafer. Importantly, the SPs of both DT34 and DT35 had average heights of approximately 2.5 nm (Fig. 4d and e, bottom, and SI; S6), which is in good agreement with the SAXS analysis. These results suggest that the introduction of the 2-decyltetradecyl group effectively prevents the SP chains from bundling, and that the position of the alkyl chain has a minimal impact on the primary structure of the SP.
To elucidate the supramolecular polymerization mechanism, variable-temperature absorption spectral measurements were conducted for solutions of DT34 and DT35 at various concentrations (3.0 × 10−5 to 20 × 10−5 M: SI; Fig. S7 and S8). The dissociation curves of DT34 and DT35 were analyzed based on an equilibrium (EQ) model26 (Fig. 4f and g) and the van't Hoff plots27 (SI; Fig. S9–S11). The fitting details are described in the SI, and the resulting thermodynamic parameters are summarized in Tables S2 and S4. In brief, the supramolecular polymerizations of both DT34 and DT35 were characterized by stronger gains in elongation enthalpy (ΔHe = −191.1 and −155.9 kJ mol−1, respectively) compared with typical benzene-1,3,5-tricarboxamide (BTA)-based SPs (ΔHe = −60.1 kJ mol−1 in heptane),27b suggesting that the SPs of DT34 and DT35 form through relatively strong monomer–monomer interactions associated with the larger core. The changes in the Gibbs free energy (ΔGe) at 298 K, determined from the EQ model, were −34.4 kJ mol−1 for DT34 and –35.2 kJ mol−1 for DT35 (SI; Table S2). The van't Hoff plots provided a consistent ΔGe values (SI; Table S4). Given the lack of substantial difference in the ΔGe values of DT34 and DT35, we infer that positional isomerism of the alkyl chains exerts a minimal influence on the monomer–monomer interactions. We were unable to determine the thermodynamic parameters for D345 because of the precipitation (see above).
Viscoelastic behaviors of SPs of DT34 and DT35
The introduction of 2-decyltetradecyl groups as side chains suppressed the bundling of SPs and the gelation; consequently, homogeneous solutions of individually solvated SPs were obtained, which permitted their investigation within the framework of conventional polymer physics. Fig. 5 shows the angular frequency (ω) dependences of the storage modulus (G′) and loss modulus (G″) of the dodecane solutions of DT34 and DT35. The crossover points between G′ and G″ are clearly observable, marking a transition from elastic to viscous behavior, and indicating the onset of a terminal relaxation regime. Our analysis began with an examination of the similarities between the two systems (Fig. 5a and b). In both DT34 and DT35, the value of G′ leveled off at higher frequencies, known as the rubbery plateau; this typically arises from entanglements of sufficiently long polymer chains.We investigated the concentration dependence of the plateau modulus (Gp), defined as the value of G′ at the minimum of tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ (SI; Fig. S12 and S13), in the range of 5–10 mM, where sufficient torque was obtained across the rubbery plateau. The Gp values for DT34 and DT35 were comparable and increased with concentration, following the power-law relationship Gp ∼ c1.5. According to well-established experimental and theoretical studies on entangled covalent polymers, Gp is primarily governed by the density of entanglements and, consequently, exhibits a concentration dependence described by the relationship Gp ∼ cα. Here, the value of α depends on the flexibility of the polymer chain. For instance, the value of α for a flexible polymer has been reported to be 2,28 reflecting the probability of the two-body contacts required to form an entanglement point.29 When the flexibility decreases, and the persistence length becomes comparable to the contour length (referred to as a semi-flexible polymer), entanglement is suppressed. Accordingly, the concentration dependence follows a weaker power-law relationship, with α reported to be 1.5.30 The concentration dependence of Gp observed for both DT34 and DT35 (Gp ∼ c1.5, see above) suggests that these SPs can, therefore, be classified as semi-flexible polymers.
δ (SI; Fig. S12 and S13), in the range of 5–10 mM, where sufficient torque was obtained across the rubbery plateau. The Gp values for DT34 and DT35 were comparable and increased with concentration, following the power-law relationship Gp ∼ c1.5. According to well-established experimental and theoretical studies on entangled covalent polymers, Gp is primarily governed by the density of entanglements and, consequently, exhibits a concentration dependence described by the relationship Gp ∼ cα. Here, the value of α depends on the flexibility of the polymer chain. For instance, the value of α for a flexible polymer has been reported to be 2,28 reflecting the probability of the two-body contacts required to form an entanglement point.29 When the flexibility decreases, and the persistence length becomes comparable to the contour length (referred to as a semi-flexible polymer), entanglement is suppressed. Accordingly, the concentration dependence follows a weaker power-law relationship, with α reported to be 1.5.30 The concentration dependence of Gp observed for both DT34 and DT35 (Gp ∼ c1.5, see above) suggests that these SPs can, therefore, be classified as semi-flexible polymers.
All the results described above indicated that the SPs consisting of DT34 and DT35 are similar in terms of their structures and stabilities. However, the flow properties of the SPs of DT34 and DT35 differed markedly (Fig. 3c and d), suggesting that there are distinct underlying mechanisms governing their macroscopic behavior. We therefore subsequently directed our attention to the behavior of these SPs in the lower-frequency regime.
In the case of DT34, a power-law dependence (G′ ∼ ω1, G″ ∼ ω2 as indicated in Fig. 5a) was observed immediately after the rubbery plateau, toward the terminal relaxation regime; this is a characteristic of an entangled “linear” covalent polymer (see, TOC figure).31 That is, the SP chains do not pass through one another, and the chain motion is constrained in the 1D direction along the polymer backbone: a so-called reptation mode. This reptation mode relaxes stress while progressively disentangling the SP chains. The relaxation time can be estimated as the reciprocal of the frequency at the crossover point (τcross); for the SPs of DT34, the τcross value was approximately 1 s.
In contrast, both G′ and G″ of DT35 gradually approached a power-law dependence toward the terminal relaxation regime, without a pronounced maximum in G″. That is, the relaxation dynamics were characterized by a broad and continuous spectrum extending from the high-frequency domain to the terminal relaxation regime. This characteristic resembles the relaxation of entangled “branched” covalent polymers (Fig. 5d).28b,32 The branch points immobilize chain segments, and the reptation mode is no longer dominant in the relaxation mechanism. Consequently, stress relaxation occurs instead through arm retraction, a process in which a polymer chain withdraws along its own contour toward the branch point. As such, the observed broad spectrum of values of G″ is attributable to the variety of time scales associated with localized motions, including arm retraction. Notably, τcross of the SPs consisting of DT35 was approximately 2 × 104 s, representing a value that is four orders of magnitude greater than that for DT34.
Discussion
Despite the recent developments in the mechanistic understanding of supramolecular polymerization mechanisms, the design principles that govern the behavior of SP chains in solution, particularly with respect to their viscoelastic properties, remain poorly understood. In this study, we focused on the side-chain structures of SPs. The reference monomer D345, bearing the commonly used linear dodecyl chains, formed SPs that bundled into thick supramolecular fibers, which, as expected, led to gelation of solutions at concentrations above 1 mM. In contrast, the newly designed DT34 and DT35, possessing branched racemic 2-decyltetradecyl chains, gave homogeneous solutions containing well-solvated SPs, even at higher concentrations of the order of 10 mM. Common characterization techniques such as SAXS, AFM, and temperature-dependent absorption spectroscopy were unable to distinguish the SPs of DT34 and DT35. Intriguingly, however, the flow properties of these SPs differed markedly, and rheological analyses provided the following insights.(1) Both DT34- and DT35-based SPs behave like covalent polymers in that their chain scission and recombination processes are too slow to contribute to stress relaxation. This behavior contrasts with the dynamics of “living” polymers33 and the “phantom-crossing”9 mechanisms proposed by Cates and Shikata, respectively, to explain the terminal relaxation dynamics of wormlike micelles and certain supramolecular polymers (such as that of BTA).9,10 In these previously investigated systems, the lifetime of the main chain was shorter than the global chain dynamics (reptation time), permitting chain breakage and recombination (or crossing) events to govern the terminal relaxation. In this context, it is noteworthy that Van Zee and co-workers recently demonstrated, through passive microrheological measurements, that biphenyltetracarboxamide (BPTA)-based SPs also exhibit a covalent polymer-like behavior.11 Such “non-living” SPs remain rare, and the ability to control their relaxation modes by molecular design might open new avenues for SP-based materials. To this end, in addition to controlling monomer–monomer interactions (i.e., ΔHe), a strategic control of SP–SP interactions could play a key role in material design, as exemplified by the contrasting behaviour of D345.
(2) The subtle structural difference between DT34 and DT35 led to significant variations in their solution viscoelastic behavior. Rheological characterization suggested that SPs formed from DT35 contain branching points. Given the 1D columnar structure of the SP, these branching points are probably formed through lateral interaction between two SP chains. Note that the alkyl chains of DT35 are symmetrically substituted with respect to the amide bond, and, as such, should produce a relatively uniform SP surface (Fig. 5e). We infer that, although not as prominent as D345, such surface uniformity facilitates SP–SP interactions, thereby promoting the formation of a branching point. In contrast, the alkyl chains of DT34 are asymmetrically substituted relative to the amide bond at 3,4-positions. This asymmetry introduces conformational diversity of the alkyl chains through rotation of the benzamide group, which is distributed randomly at the periphery of DT34 monomer,19 thereby leading to a disordered SP surface (Fig. 5d). We surmise that such surface disorder suppresses SP–SP interactions, yielding a situation distinct from that observed for DT35.
Conclusions
As a step toward advancing this research field by molecular-level understanding, our findings highlight the critical role of side chains in tuning the hierarchical assembly and macroscopic properties of SPs. We believe that controlling SP–SP interactions by molecular design might permit precise and engineering and processing of SP-based functional soft materials.19Author contributions
K. S. conceived the project. Y. F. synthesized the monomers, characterized their supramolecular polymerization, and prepared solutions of supramolecular polymers. R. S. conducted viscoelastic measurements. Y. F. and R. S. contributed equally. T. K. conducted WAXS and SAXS measurements. All authors discussed the results. N. F., T. K. and K. S. wrote the paper with input from all the authors.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this study are provided in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc09763k.Acknowledgements
The work is supported by the Japan Society for the Promotion of Science (JSPS) KAKENHI grant no. JP25K18071 (N. F.), no. JP22H02134 (K. S.); no. JP23K17941 (K. S.); no. JP24H01712 (K. S.), in a Grant-in-Aid Scientific Research for Transformative Research Areas (A) “Materials Science of Meso-Hierarchy”; and by Japan Science and Technology Agency (JST) grant no. JPMJCR23L2 (K. S., T. K. & T. K.) in Precise Material Science for Degradation and Stability, CREST. K. S. acknowledges financial support from The Murata Science Foundation, Sekisui Chemical Grant Program, The Mitsubishi Foundation, Masuyakinen Basic Research Foundation, Fujimori Science and Technology Foundation, and The Samco Foundation. We are grateful to Prof. Kazuo Tanaka (Kyoto University) and Assist. Prof. Masayuki Gon (Kyoto University) for use of the sputter coater for SEM measurements.Notes and references
- L. Brunsveld, B. J. B. Folmer, E. W. Meijer and R. P. Sijbesma, Supramolecular Polymers, Chem. Rev., 2001, 101, 4071–4098 CrossRef CAS PubMed.
- T. F. A. De Greef, M. M. J. Smulders, M. Wolffs, A. P. H. J. Schenning, R. P. Sijbesma and E. W. Meijer, Supramolecular Polymerization, Chem. Rev., 2009, 109, 5687–5754 CrossRef CAS PubMed.
- (a) S. Ogi, K. Sugiyasu, S. Manna, S. Samitsu and M. Takeuchi, Living supramolecular polymerization realized through a biomimetic approach, Nat. Chem., 2014, 6, 188–195 CrossRef CAS PubMed; (b) J. Kang, D. Miyajima, T. Mori, Y. Inoue, Y. Itoh and T. Aida, A rational strategy for the realization of chain-growth supramolecular polymerization, Science, 2015, 347, 646–651 CrossRef CAS PubMed; (c) S. Ogi, V. Stepanenko, K. Sugiyasu, M. Takeuchi and F. Würthner, Mechanism of Self-Assembly Process and Seeded Supramolecular Polymerization of Perylene Bisimide Organogelator, J. Am. Chem. Soc., 2015, 137, 3300–3307 CrossRef CAS PubMed; (d) M. Wehner and F. Würthner, Supramolecular polymerization through kinetic pathway control and living chain growth, Nat. Rev. Chem., 2020, 4, 38–53 CrossRef CAS; (e) K. Sugiyasu, A case study of monomer design for controlled/living supramolecular polymerization, Polym. J., 2021, 53, 865–875 CrossRef CAS.
- (a) S. H. Jung, D. Bochicchio, G. M. Pavan, M. Takeuchi and K. Sugiyasu, A Block Supramolecular Polymer and Its Kinetically Enhanced Stability, J. Am. Chem. Soc., 2018, 140, 10570–10577 CrossRef CAS PubMed; (b) W. Wagner, M. Wehner, V. Stepanenko and F. Würthner, Supramolecular Block Copolymers by Seeded Living Polymerization of Perylene Bisimides, J. Am. Chem. Soc., 2019, 141, 12044–12054 CrossRef CAS PubMed; (c) A. Sarkar, R. Sasmal, C. Empereur-mot, D. Bochicchio, S. V. K. Kompella, K. Sharma, S. Dhiman, B. Sundaram, S. S. Agasti, G. M. Pavan and S. J. George, Self-Sorted, Random, and Block Supramolecular Copolymers via Sequence Controlled, Multicomponent Self-Assembly, J. Am. Chem. Soc., 2020, 142, 7606–7617 CrossRef CAS PubMed; (d) N. Sasaki, J. Kikkawa, Y. Ishii, T. Uchihashi, H. Imamura, M. Takeuchi and K. Sugiyasu, Multistep, site-selective noncovalent synthesis of two-dimensional block supramolecular polymers, Nat. Chem., 2023, 15, 922–929 CrossRef CAS PubMed.
- T. Haino, Molecular-recognition-directed formation of supramolecular polymers, Polym. J., 2013, 45, 363–383 CrossRef CAS.
- (a) R. K. Castellano, R. Clark, S. L. Craig, C. Nuckolls and J. Rebek, Emergent mechanical properties of self-assembled polymeric capsules, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 12418–12421 CrossRef CAS PubMed; (b) T. Haino, A. Watanabe, T. Hirao and T. Ikeda, Supramolecular Polymerization Triggered by Molecular Recognition between Bisporphyrin and Trinitrofluorenone, Angew. Chem., Int. Ed., 2012, 51, 1473–1476 CrossRef CAS PubMed; (c) B. Zheng, F. Wang, S. Dong and F. Huang, Supramolecular polymers constructed by crown ether-based molecular recognition, Chem. Soc. Rev., 2012, 41, 1621–1636 RSC; (d) D. Xia and M. Xue, A supramolecular polymer gel with dual-responsiveness constructed by crown ether based molecular recognition, Polym. Chem., 2014, 5, 5591–5597 RSC; (e) L. Wang, L. Cheng, G. Li, K. Liu, Z. Zhang, P. Li, S. Dong, W. Yu, F. Huang and X. Yan, A Self-Cross-Linking Supramolecular Polymer Network Enabled by Crown-Ether-Based Molecular Recognition, J. Am. Chem. Soc., 2020, 142, 2051–2058 CrossRef CAS PubMed.
- (a) R. P. Sijbesma, F. H. Beijer, L. Brunsveld, B. J. B. Folmer, J. H. K. K. Hirschberg, R. F. M. Lange, J. K. L. Lowe and E. W. Meijer, Reversible Polymers Formed from Self-Complementary Monomers Using Quadruple Hydrogen Bonding, Science, 1997, 278, 1601–1604 CrossRef CAS PubMed; (b) B. J. B. Folmer, R. P. Sijbesma and E. W. Meijer, Unexpected Entropy-Driven Ring-Opening Polymerization in a Reversible Supramolecular System, J. Am. Chem. Soc., 2001, 123, 2093–2094 CrossRef CAS PubMed; (c) V. Berl, M. Schmutz, M. J. Krische, R. G. Khoury and J.-M. Lehn, Supramolecular Polymers Generated from Heterocomplementary Monomers Linked through Multiple Hydrogen-Bonding Arrays—Formation, Characterization, and Properties, Chem.–Eur. J., 2002, 8, 1227–1244 CrossRef CAS PubMed.
- (a) W. Weng, Z. Li, A. M. Jamieson and S. J. Rowan, Control of Gel Morphology and Properties of a Class of Metallo-Supramolecular Polymers by Good/Poor Solvent Environments, Macromolecules, 2009, 42, 236–246 CrossRef CAS; (b) T. Vermonden, M. J. van Steenbergen, N. A. M. Besseling, A. T. M. Marcelis, W. E. Hennink, E. J. R. Sudhölter and M. A. Cohen Stuart, Linear Rheology of Water-Soluble Reversible Neodymium(III) Coordination Polymers, J. Am. Chem. Soc., 2004, 126, 15802–15808 CrossRef CAS PubMed.
- (a) D. Ogata, T. Shikata and K. Hanabusa, Chiral Amplification of the Structure and Viscoelasticity of a Supramolecular Polymeric System Consisting of N,N′,N″-Tris(3,7-Dimethyloctyl)Benzene-1,3,5-Tricarboxamide and n-Decane, J. Phys. Chem. B, 2004, 108, 15503–15510 CrossRef CAS; (b) T. Shikata, D. Ogata and K. Hanabusa, Viscoelastic Behavior of Supramolecular Polymeric Systems Consisting of N,N′,N″-Tris(3,7-dimethyloctyl)benzene-1,3,5-tricarboxamide and n-Alkanes, J. Phys. Chem. B, 2004, 108, 508–514 CrossRef CAS.
- (a) P. Terech, V. Schaffhauser, P. Maldivi and J. M. Guenet, Living polymers in organic solvents, Langmuir, 1992, 8, 2104–2106 CrossRef CAS; (b) C. Dammer, P. Maldivi, P. Terech and J.-M. Guenet, Rheological Study of a Bicopper Tetracarboxylate/Decalin Jelly, Langmuir, 1995, 11, 1500–1506 CrossRef CAS; (c) G. Ducouret, C. Chassenieux, S. Martins, F. Lequeux and L. Bouteiller, Rheological characterisation of bis-urea based viscoelastic solutions in an apolar solvent, J. Colloid Interface Sci., 2007, 310, 624–629 CrossRef CAS PubMed; (d) S. Seiffert and J. Sprakel, Physical chemistry of supramolecular polymer networks, Chem. Soc. Rev., 2012, 41, 909–930 RSC.
- E. Vereroudakis, N. A. Burger, L. Bouteiller, B. Loppinet, E. W. Meijer, D. Vlassopoulos and N. J. Van Zee, Counterintuitive Viscoelasticity of Supramolecular Polymer Networks Driven by Coassembly with Water Molecules, Macromolecules, 2024, 57, 9030–9040 CrossRef CAS.
- (a) N. M. Sangeetha and U. Maitra, Supramolecular gels: functions and uses, Chem. Soc. Rev., 2005, 34, 821–836 RSC; (b) E. R. Draper and D. J. Adams, Low-Molecular-Weight Gels: The State of the Art, Chem, 2017, 3, 390–410 CrossRef CAS; (c) D. K. Smith, Supramolecular gels – a panorama of low-molecular-weight gelators from ancient origins to next-generation technologies, Soft Matter, 2024, 20, 10–70 RSC.
- (a) L. Chen, J. Raeburn, S. Sutton, D. G. Spiller, J. Williams, J. S. Sharp, P. C. Griffiths, R. K. Heenan, S. M. King, A. Paul, S. Furzeland, D. Atkins and D. J. Adams, Tuneable mechanical properties in low molecular weight gels, Soft Matter, 2011, 7, 9721–9727 RSC; (b) W. E. M. Noteborn, D. N. H. Zwagerman, V. S. Talens, C. Maity, L. van der Mee, J. M. Poolman, S. Mytnyk, J. H. van Esch, A. Kros, R. Eelkema and R. E. Kieltyka, Crosslinker-Induced Effects on the Gelation Pathway of a Low Molecular Weight Hydrogel, Adv. Mater., 2017, 29, 1603769 CrossRef PubMed; (c) A. Dawn and H. Kumari, Low Molecular Weight Supramolecular Gels Under Shear: Rheology as the Tool for Elucidating Structure–Function Correlation, Chem.–Eur. J., 2018, 24, 762–776 CrossRef CAS PubMed; (d) R. Laishram, S. Sarkar, I. Seth, N. Khatun, V. K. Aswal, U. Maitra and S. J. George, Secondary Nucleation-Triggered Physical Cross-Links and Tunable Stiffness in Seeded Supramolecular Hydrogels, J. Am. Chem. Soc., 2022, 144, 11306–11315 CrossRef CAS PubMed.
- S. Liu, W. Yu and C. Zhou, Solvents effects in the formation and viscoelasticity of DBS organogels, Soft Matter, 2013, 9, 864–874 RSC.
- F. Lortie, S. Boileau, L. Bouteiller, C. Chassenieux, B. Demé, G. Ducouret, M. Jalabert, F. Lauprêtre and P. Terech, Structural and Rheological Study of a Bis-urea Based Reversible Polymer in an Apolar Solvent, Langmuir, 2002, 18, 7218–7222 CrossRef CAS.
- S. Panja and D. J. Adams, Gel to gel transitions by dynamic self-assembly, Chem. Commun., 2019, 55, 10154–10157 RSC.
- (a) K. J. Skilling, F. Citossi, T. D. Bradshaw, M. Ashford, B. Kellam and M. Marlow, Insights into low molecular mass organic gelators: a focus on drug delivery and tissue engineering applications, Soft Matter, 2014, 10, 237–256 RSC; (b) B. O. Okesola and D. K. Smith, Applying low-molecular weight supramolecular gelators in an environmental setting – self-assembled gels as smart materials for pollutant removal, Chem. Soc. Rev., 2016, 45, 4226–4251 RSC; (c) T. Christoff-Tempesta, A. J. Lew and J. H. Ortony, Beyond Covalent Crosslinks: Applications of Supramolecular Gels, Gels, 2018, 4, 40 CrossRef PubMed; (d) S. Panja and D. J. Adams, Stimuli responsive dynamic transformations in supramolecular gels, Chem. Soc. Rev., 2021, 50, 5165–5200 RSC; (e) D. K. Smith, Supramolecular Gels as Active Tools for Reaction Engineering, Angew. Chem., Int. Ed., 2025, 64, e202502053 CrossRef CAS PubMed.
- J. J. B. van der Tol, G. Vantomme and E. W. Meijer, Solvent-Induced Pathway Complexity of Supramolecular Polymerization Unveiled Using the Hansen Solubility Parameters, J. Am. Chem. Soc., 2023, 145, 17987–17994 CrossRef CAS PubMed.
- T. Shimada, Y. Watanabe, T. Kajitani, M. Takeuchi, Y. Wakayama and K. Sugiyasu, Individually separated supramolecular polymer chains toward solution-processable supramolecular polymeric materials, Chem. Sci., 2023, 14, 822–826 RSC.
- (a) F. Wudl and G. Srdanov, Conducting polymer formed of poly(2-methoxy,5-(2′-ethyl-hexyloxy)-p-phenylenevinylene), US Pat., US5189136A, 1993 Search PubMed; (b) X. Zhan, Z. a. Tan, B. Domercq, Z. An, X. Zhang, S. Barlow, Y. Li, D. Zhu, B. Kippelen and S. R. Marder, A High-Mobility Electron-Transport Polymer with Broad Absorption and Its Use in Field-Effect Transistors and All-Polymer Solar Cells, J. Am. Chem. Soc., 2007, 129, 7246–7247 CrossRef CAS PubMed; (c) I. Osaka, R. Zhang, J. Liu, D.-M. Smilgies, T. Kowalewski and R. D. McCullough, Highly Stable Semiconducting Polymers Based on Thiazolothiazole, Chem. Mater., 2010, 22, 4191–4196 CrossRef CAS; (d) T. Lei, J.-H. Dou and J. Pei, Influence of Alkyl Chain Branching Positions on the Hole Mobilities of Polymer Thin-Film Transistors, Adv. Mater., 2012, 24, 6457–6461 CrossRef CAS PubMed; (e) F. Zhang, Y. Hu, T. Schuettfort, C.-a. Di, X. Gao, C. R. McNeill, L. Thomsen, S. C. B. Mannsfeld, W. Yuan, H. Sirringhaus and D. Zhu, Critical Role of Alkyl Chain Branching of Organic Semiconductors in Enabling Solution-Processed N-Channel Organic Thin-Film Transistors with Mobility of up to 3.50 cm2 V–1 s–1, J. Am. Chem. Soc., 2013, 135, 2338–2349 CrossRef CAS PubMed; (f) J. Mei and Z. Bao, Side Chain Engineering in Solution-Processable Conjugated Polymers, Chem. Mater., 2014, 26, 604–615 CrossRef CAS; (g) K. Kawabata, M. Saito, N. Takemura, I. Osaka and K. Takimiya, Effects of branching position of alkyl side chains on ordering structure and charge transport property in thienothiophenedione- and quinacridone-based semiconducting polymers, Polym. J., 2017, 49, 169–176 CrossRef CAS; (h) A. Shinohara, C. Pan, Z. Guo, L. Zhou, Z. Liu, L. Du, Z. Yan, F. J. Stadler, L. Wang and T. Nakanishi, Viscoelastic Conjugated Polymer Fluids, Angew. Chem., Int. Ed., 2019, 58, 9581–9585 CrossRef CAS PubMed.
- (a) Y. Yang, H. Qin, M. Jiang, L. Lin, T. Fu, X. Dai, Z. Zhang, Y. Niu, H. Cao, Y. Jin, F. Zhao and X. Peng, Entropic Ligands for Nanocrystals: From Unexpected Solution Properties to Outstanding Processability, Nano Lett., 2016, 16, 2133–2138 CrossRef CAS PubMed; (b) O. Elimelech, O. Aviv, M. Oded, X. Peng, D. Harries and U. Banin, Entropy of Branching Out: Linear versus Branched Alkylthiols Ligands on CdSe Nanocrystals, ACS Nano, 2022, 16, 4308–4321 CrossRef CAS PubMed; (c) S. Yamashita, Y. Ito, H. Kamiya and Y. Okada, Surface coverage can control the dispersibility of TiO2 and ZrO2 nanoparticles in hydrophobic solvents: Comparison of linear and branched ligands, Adv. Powder Technol., 2024, 35, 104277 CrossRef CAS.
- (a) J. J. Armao IV, M. Maaloum, T. Ellis, G. Fuks, M. Rawiso, E. Moulin and N. Giuseppone, Healable Supramolecular Polymers as Organic Metals, J. Am. Chem. Soc., 2014, 136, 11382–11388 CrossRef PubMed; (b) E. Moulin, J. J. Armao IV and N. Giuseppone, Triarylamine-Based Supramolecular Polymers: Structures, Dynamics, and Functions, Acc. Chem. Res., 2019, 52, 975–983 CrossRef CAS PubMed.
- (a) K. Kubo, Y. Moriyama, S. Ujiie and A. Mori, Synthesis and Properties of New Liquid Crystalline Organogelators with a Tris[4-(benzoylamino)phenyl]amine Core, Chem. Lett., 2015, 44, 984–986 CrossRef CAS; (b) T. Kim, T. Mori, T. Aida and D. Miyajima, Dynamic propeller conformation for the unprecedentedly high degree of chiral amplification of supramolecular helices, Chem. Sci., 2016, 7, 6689–6694 RSC.
- (a) J. J. van Gorp, J. A. J. M. Vekemans and E. W. Meijer, C3-Symmetrical Supramolecular Architectures: Fibers and Organic Gels from Discotic Trisamides and Trisureas, J. Am. Chem. Soc., 2002, 124, 14759–14769 CrossRef CAS PubMed; (b) M. Ikeda, M. Takeuchi and S. Shinkai, Unusual emission properties of a triphenylene-based organogel system, Chem. Commun., 2003, 1354–1355 RSC; (c) M. Shirakawa, S. Kawano, N. Fujita, K. Sada and S. Shinkai, Hydrogen-Bond-Assisted Control of H versus J Aggregation Mode of Porphyrins Stacks in an Organogel System, J. Org. Chem., 2003, 68, 5037–5044 CrossRef CAS PubMed; (d) T. Kishida, N. Fujita, K. Sada and S. Shinkai, Sol–Gel Reaction of Porphyrin-Based Superstructures in the Organogel Phase: Creation of Mechanically Reinforced Porphyrin Hybrids, J. Am. Chem. Soc., 2005, 127, 7298–7299 CrossRef CAS PubMed.
- (a) Y. Kudo, M. Sakuragi, S. Hashida, R. Kuwahara, T. Ishi-I, H. Masunaga and K. Sakurai, Flexibility and local structure of a worm-like cylinder of self-assembled discotic triazine triamide, Polym. J., 2010, 42, 812–817 CrossRef CAS; (b) D. McDowall, D. J. Adams and A. M. Seddon, Using small angle scattering to understand low molecular weight gels, Soft Matter, 2022, 18, 1577–1590 RSC.
- H. M. M. ten Eikelder, A. J. Markvoort, T. F. A. de Greef and P. A. J. Hilbers, An Equilibrium Model for Chiral Amplification in Supramolecular Polymers, J. Phys. Chem. B, 2012, 116, 5291–5301 CrossRef CAS PubMed.
- (a) P. Jonkheijm, P. van der Schoot, A. P. H. J. Schenning and E. W. Meijer, Probing the Solvent-Assisted Nucleation Pathway in Chemical Self-Assembly, Science, 2006, 313, 80–83 CrossRef CAS PubMed; (b) M. M. J. Smulders, A. P. H. J. Schenning and E. W. Meijer, Insight into the Mechanisms of Cooperative Self-Assembly: The “Sergeants-and-Soldiers” Principle of Chiral and Achiral C3-Symmetrical Discotic Triamides, J. Am. Chem. Soc., 2008, 130, 606–611 CrossRef CAS PubMed.
- (a) L. A. Holmes, S. Kusamizu, K. Osaki and J. D. Ferry, Dynamic mechanical properties of moderately concentrated polystyrene solutions, J. Polym. Sci., Part A-2, 1971, 9, 2009–2021 CrossRef CAS; (b) V. R. Raju, E. V. Menezes, G. Marin, W. W. Graessley and L. J. Fetters, Concentration and molecular weight dependence of viscoelastic properties in linear and star polymers, Macromolecules, 1981, 14, 1668–1676 CrossRef CAS.
- M. Doi, S. F. Edwards and S. F. Edwards, The Theory of Polymer Dynamics, Clarendon Press, 1988 Search PubMed.
- (a) D. C. Morse, Viscoelasticity of Concentrated Isotropic Solutions of Semiflexible Polymers. 2. Linear Response, Macromolecules, 1998, 31, 7044–7067 CrossRef CAS; (b) D. C. Morse, Viscoelasticity of Concentrated Isotropic Solutions of Semiflexible Polymers. 1. Model and Stress Tensor, Macromolecules, 1998, 31, 7030–7043 CrossRef CAS; (c) V. Shankar, M. Pasquali and D. C. Morse, Theory of linear viscoelasticity of semiflexible rods in dilute solution, J. Rheol., 2002, 46, 1111–1154 CrossRef CAS.
- R. G. Ricarte and S. Shanbhag, A tutorial review of linear rheology for polymer chemists: basics and best practices for covalent adaptable networks, Polym. Chem., 2024, 15, 815–846 RSC.
- (a) T. Masuda, Y. Ohta and S. Onogi, Rheological Properties of Anionic Polystyrenes. III. Characterization and Rheological Properties of Four-Branch Polystyrenes, Macromolecules, 1971, 4, 763–768 CrossRef CAS; (b) W. W. Graessley, T. Masuda, J. E. L. Roovers and N. Hadjichristidis, Rheological Properties of Linear and Branched Polyisoprene, Macromolecules, 1976, 9, 127–141 CrossRef CAS; (c) W. W. Graessley and J. Roovers, Melt Rheology of Four-Arm and Six-Arm Star Polystyrenes, Macromolecules, 1979, 12, 959–965 CrossRef CAS; (d) L. J. Fetters, A. D. Kiss, D. S. Pearson, G. F. Quack and F. J. Vitus, Rheological behavior of star-shaped polymers, Macromolecules, 1993, 26, 647–654 CrossRef CAS.
- M. E. Cates, Reptation of living polymers: dynamics of entangled polymers in the presence of reversible chain-scission reactions, Macromolecules, 1987, 20, 2289–2296 CrossRef CAS.
Footnotes |
| † Y. F. and R. S. contributed equally. |
| ‡ It should be noted that there is another class of SPs, formed from ditopic monomers.1,5 These monomers self-assemble through host–guest complexation,6 complementary hydrogen bonding,7 or metal–ligand interaction8 and their polymerization process follows an isodesmic model.2 In principle, supramolecular polymerization of this type cannot be controlled kinetically. The viscoelastic properties of these polymers in solution have been extensively studied, owing to their homogeneous solubility in water8 and organic solvents.6,7 As such, this class of SPs differs from those examined in the present study and is not discussed in the context of this manuscript. |
| This journal is © The Royal Society of Chemistry 2026 |