Open Access Article
Open Access ArticleFrom alkynes to spiroindole δ-lactams via a copper-nitrenoid intermediate featuring unusual 1,1-addition and high Z-selectivity
Zhiqiang Ren†
 *,
Tianhui Feng†,
Yeyao Li,
Mengyi Chu,
Ye Zheng,
Tianli Gao,
Bo Han
*,
Tianhui Feng†,
Yeyao Li,
Mengyi Chu,
Ye Zheng,
Tianli Gao,
Bo Han ,
Ruili Guo,
Haojie Ma
,
Ruili Guo,
Haojie Ma ,
Ji-Jiang Wang and
Yuqi Zhang
,
Ji-Jiang Wang and
Yuqi Zhang *
*
Department of New Energy & New Functional Materials, Shaanxi Key Laboratory of Chemical Reaction Engineering, School of Chemistry and Chemical Engineerin Yan'an University, Yan'an, 716000, P. R. China. E-mail: renzhiqiang@yau.edu.cn; yqzhang@yau.edu.cn
First published on 31st March 2026
Abstract
This study reports a novel, operationally efficient N-heterocyclic carbene (NHC)-copper-catalysed strategy for the synthesis of spiroindole δ-lactams via the coupling of dioxazolones with alkyne derivatives. The protocol demonstrates high atom economy, excellent product yields, broad functional group tolerance, and outstanding Z-stereoselectivity. Detailed mechanistic investigations, combining density functional theory (DFT) calculations with Kinetic isotope effect (KIE) experiments, reveal that the efficient formation of the copper alkyne intermediate is the key factor governing the progression of this transformation. The observed 1,1-addition of unactivated alkynes, delivering high Z/E stereoselectivity and excellent regioselectivity, is proposed to proceed via a hydrogen atom migration mechanism.
Introduction
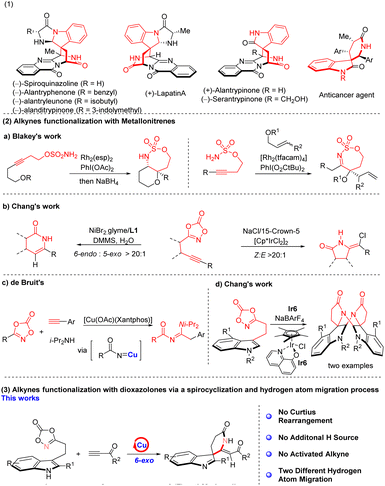
Lactam scaffolds are ubiquitous in agrochemicals, pharmaceuticals, and natural products, serving as privileged structural motifs that confer diverse biological activities and substantial clinical significance.1–3 The alkaloid (−)-spiroquinazoline, distinguished by its unique spiroindole δ-lactam architecture,4–6 together with related congeners, has been reported to display significant anticancer activity7,8 (Scheme 1(1)). Accordingly, establishing operationally simple, efficient, and scalable methodologies for lactam synthesis is a significant objective in contemporary synthetic chemistry, with substantial academic and industrial implications.Metallonitrenes are highly versatile reactive intermediates that have been widely exploited for the efficient assembly of polycyclic lactam frameworks through transition metal-catalyzed (Rh, Ir, Ni, Fe) direct C–N bond-forming reactions with alkynes.9–22 For example, Blakey et al. reported a rhodium-catalyzed strategy for the synthesis of polycyclic lactam frameworks from sulfonamides and alkynes (Scheme 1(2)a);23–26 Similarly, Chang et al. demonstrated that Ir- and Ni-catalyzed reactions of dioxazolones with alkynes proceed with high regioselectivity encompassing both 5-exo and 6-endo cyclization pathways and excellent stereoselectivity, affording defined Z/E configuration (Scheme 1(2)b).27–29 In addition, significant advances have been achieved in the direct formation of N–N, N–C, and N–S bonds through transition metal-catalysed (e.g., Ir, Rh, Fe, Cu, Mn) transformations of dioxazolones via metallonitrene intermediates.30–41 However, the copper-catalyzed construction of polycyclic lactams from alkynes and dioxazolones via metallonitrene intermediates remains unexplored. To date, only de Bruin et al. have reported a related copper-mediated three-component transformation involving dioxazolones, alkynes, and diisopropylamine, leading to the formation of distinctive amidine scaffolds (Scheme 1(2)c).42
Indole derivatives are well recognized for their diverse biological activities and significant medicinal relevance,43–45 and extensive efforts have been devoted to their dearomatization.46–49 In this context, Chang et al. reported an iridium (Ir)-catalyzed transformation of indoles with dioxazolones, leading to the formation of an unusual dimeric framework (Scheme 1(2)c).50 The synthesis of indole-containing polycyclic lactam architectures has garnered significant interest in recent years. In the present study, alkynes were strategically incorporated into the reaction system to enable the construction of a spiroindole δ-lactam framework. Notably, the reaction proceeds without undergoing a Curtius rearrangement and instead furnishes the anti-Markovnikov 1,1-addition product via a 6-exo cyclization pathway, delivering compound 3 with high Z-selectivity (Scheme 1(3)).
Results and discussion
Dioxazolone 1a and methyl propiolate 2a were selected as model substrates for reaction optimization (Table 1). Under the initial conditions using IPrCuCl as the catalyst, AgOAc as the additive, and 1,2-dichloroethane (DCE) as the solvent at 80 °C for 15 min, the desired product 3a was obtained in 77% isolated yield (Table 1, entry 1). The molecular structure of compound 3a was unambiguously established by single-crystal X-ray diffraction (XRD), nuclear magnetic resonance (NMR) spectroscopy, and high-resolution mass spectrometry (HRMS). The Z configuration was primarily determined based on NMR analysis and corroborated by XRD data. Subsequently, the influence of different copper catalysts, including Cu(OAc)2, CuCl, and IMesCuCl, on the reaction outcome was systematically evaluated (Table 1, entries 2–4). It was observed that although the Z/E ratio remained essentially unchanged, the product yield decreased to 20–38%. To further evaluate the influence of additives, various silver salts, including AgNO3, AgOTf, AgSbF6, and AgNTf2, were systematically examined. In all cases, the Z/E ratio was maintained; however, the yields were significantly reduced to 13–26% (Table 1, entries 5–8). To enhance the reaction efficiency, the catalyst loading was increased to 15 mol%, and the additive loading to 30 mol%. Under these optimized conditions, the isolated yield was 88%, while maintaining excellent stereoselectivity (Z/E > 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1; Table 1, entry 9). Furthermore, the choice of solvent significantly influenced the reaction outcome. When DCM or CHCl3 was employed as the solvent, the isolated yield decreased to 67–68% (Table 1, entries 10–11). The reaction temperature was also found to influence the transformation significantly. Lowering the temperature to 60 °C decreased the yield and prolonged the reaction time to 1 h (Table 1, entry 12).
1; Table 1, entry 9). Furthermore, the choice of solvent significantly influenced the reaction outcome. When DCM or CHCl3 was employed as the solvent, the isolated yield decreased to 67–68% (Table 1, entries 10–11). The reaction temperature was also found to influence the transformation significantly. Lowering the temperature to 60 °C decreased the yield and prolonged the reaction time to 1 h (Table 1, entry 12).
| Entry | Catalyst | Additive | Solvent | T/°C | Yield%b | Z/Ec |
|---|---|---|---|---|---|---|
| a Reaction conditions: 1a (0.20 mmol), 2a (0.60 mmol), Cu catalyst (× mol%), Ag salt (2× mol%), 80 °C, 15 min, 0.05 M, under air.b Isolated yields.c Z/E ratio determined by NMR and XRD.d Reaction time: 1 h. Abbreviations: DCE = 1,2-dichloroethane; DCM = dichloromethane. | ||||||
| 1 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 77 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 2 | Cu(OAc)2(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 38 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 3 | CuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 28 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 4 | IMesCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 20 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 5 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 18 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 6 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 15 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 7 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 26 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 8 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 13 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 9 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 88 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1 1 |
| 10 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | CH2Cl2 | 80 | 69 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 11 | IPrCuCl(10 mol%) | AgOAc(20 mol%) | CHCl3 | 80 | 67 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 12d | IPrCuCl(10 mol%) | AgOAc(20 mol%) | DCE | 80 | 68 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
 |
||||||
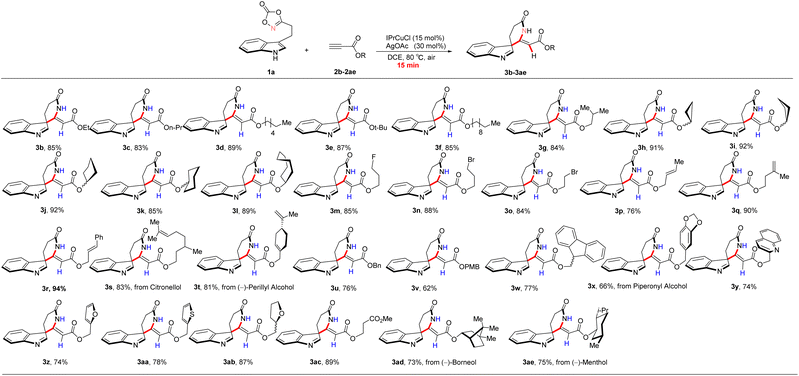
To further optimize and validate the established reaction conditions, a systematic evaluation of substrate scope and functional group tolerance was conducted (Scheme 2). Various ester derivatives were examined to assess the generality of the transformation. Notably, elongation of the alkyl chain (methyl, ethyl, propyl, tert-butyl, sec-butyl, hexyl, nonyl) was well tolerated, affording the corresponding products (3a–3g) in 83–89% isolated yields with excellent stereoselectivity (Z/E > 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Cycloalkyl substituents, including cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and cycloheptyl groups, were also well tolerated under the optimized conditions, delivering the corresponding products (3h–3l) in 75–92% isolated yields. Moreover, a variety of functional groups were compatible with the reaction system. Substrates bearing halogens (Br, F), alkenyl moieties, ether linkages, and ester functionalities provided the desired products in good to excellent yields, for example, compounds 3m–3o (75–88%), 3q–3t (81–94%), 3ab (87%), and 3ac (89%). These transformations proceeded with high efficiency, excellent regioselectivity, and outstanding Z/E stereoselectivity, thereby establishing a robust platform for subsequent functional group derivatization. Both unsubstituted aryl and electron-rich aryl substrates exhibited good reactivity under the optimized conditions, affording product 3u–3aa in 66–78% isolated yields, respectively. To evaluate the influence of chiral induction on the formation of the quaternary carbon center, enantioenriched ester substrates were employed. Under the optimized conditions, products 3ad and 3ae were obtained in 73% and 75% isolated yields, respectively. Notably, NMR analysis revealed no detectable formation of diastereoisomeric mixtures, indicating high stereochemical fidelity during the transformation.
1). Cycloalkyl substituents, including cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and cycloheptyl groups, were also well tolerated under the optimized conditions, delivering the corresponding products (3h–3l) in 75–92% isolated yields. Moreover, a variety of functional groups were compatible with the reaction system. Substrates bearing halogens (Br, F), alkenyl moieties, ether linkages, and ester functionalities provided the desired products in good to excellent yields, for example, compounds 3m–3o (75–88%), 3q–3t (81–94%), 3ab (87%), and 3ac (89%). These transformations proceeded with high efficiency, excellent regioselectivity, and outstanding Z/E stereoselectivity, thereby establishing a robust platform for subsequent functional group derivatization. Both unsubstituted aryl and electron-rich aryl substrates exhibited good reactivity under the optimized conditions, affording product 3u–3aa in 66–78% isolated yields, respectively. To evaluate the influence of chiral induction on the formation of the quaternary carbon center, enantioenriched ester substrates were employed. Under the optimized conditions, products 3ad and 3ae were obtained in 73% and 75% isolated yields, respectively. Notably, NMR analysis revealed no detectable formation of diastereoisomeric mixtures, indicating high stereochemical fidelity during the transformation.
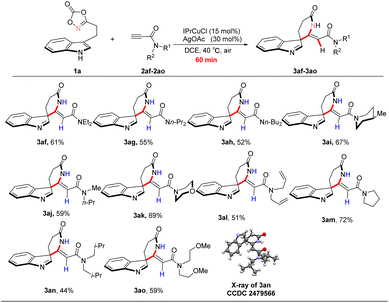
Subsequently, amide derivatives were examined further to expand the substrate scope (Scheme 3). Acyclic amide substrates were poorly compatible under the optimized conditions, affording products 3af–3ah, 3aj, 3al, 3an, and 3ao in moderate yields (44–61%). In contrast, cyclic amide substrates were better tolerated, delivering products 3ai, 3ak, and 3am in improved yields of 62–72%. These results suggest that steric and conformational effects associated with the amide moiety significantly influence the reaction efficiency.
Ketone derivatives were subsequently evaluated further to explore the substrate scope (Scheme 4). The reaction demonstrated broad substrate generality and excellent functional group tolerance, affording products 3ap–3at in 82–94% isolated yields. Notably, the protocol was compatible with various sensitive functional groups, including silyl ethers, benzyl ethers, and alkenes (3au, 3av, 3ay). In addition, cyclic ketone substrates were well accommodated, delivering products 3aw, 3ax, and 3az in good yields.
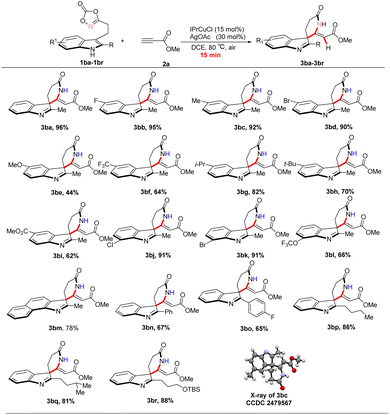
The effect of substituents on the indole framework was further examined (Scheme 5). The parent substrate afforded compound 3ba in 96% isolated yield. Alkyl substituents at the C5 position were well tolerated, providing products 3bb–3bd and 3bg–3bh in excellent yields. In contrast, the presence of a strong electron-donating group at C5 significantly reduced the yield to 44% (3be), whereas electron-withdrawing substituents provided the corresponding products 3bf and 3bi in moderate yields of 62–64%.
At the C7 position, Cl and Br substituents were well tolerated, affording products 3bj–3bk in 91% isolated yields. In contrast, the introduction of a trifluoromethoxy (CF3O) group resulted in a 66% yield. Benzoindole was also compatible under the optimized conditions, serving as a precursor to the structurally complex polycyclic product 3bm in 78% isolated yield. Substitution at the C2 position revealed notable steric effects: bulky aryl substituents led to reduced yields of 65–67% (3bo, 3bp), whereas alkyl groups at C2 were better accommodated, delivering products 3bp–3br in 81–88% isolated yields.
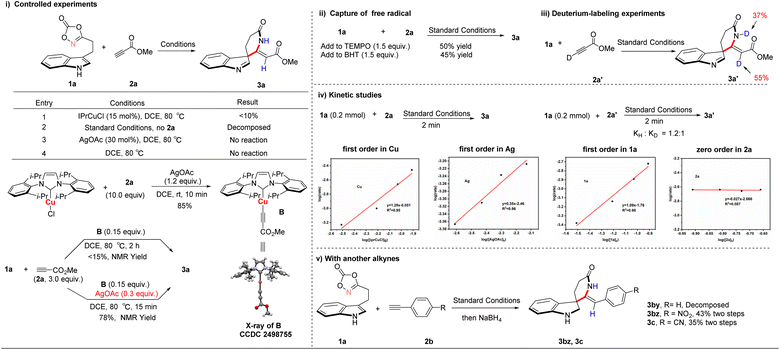
To gain insight into the reaction mechanism (Scheme 6), a series of control experiments was conducted to systematically assess the roles of the catalyst, additive, and methyl propiolate. The results confirmed that IPrCuCl is essential for catalytic activity, while silver acetate functions as a critical additive. To further elucidate the role of silver acetate, complex B was successfully isolated and evaluated. This complex exhibited limited catalytic activity, affording the product in less than 15% yield as determined by NMR analysis. Notably, supplementation with additional silver acetate significantly increased the NMR yield to 78%. This result suggests that silver acetate facilitates the formation of complex B and that the acetate anion plays a crucial role in promoting hydrogen atom transfer (Scheme 6(i)). Furthermore, radical trapping experiments were conducted using TEMPO and BHT. In both cases, the desired products were still obtained, indicating that the transformation does not proceed via a radical-mediated pathway (Scheme 6(ii)). Deuterium labeling experiments provided clear evidence for the migration of the alkynyl hydrogen atom, supporting a 1,1-addition mechanism (Scheme 6(iii)). Kinetic isotope effect (KIE) studies further revealed that copper-alkyne coordination is unlikely to be the rate-determining step. Kinetic analysis established a first-order dependence on the copper catalyst, substrate 1a, and the silver additive, while exhibiting zero-order dependence on alkyne substrate 2a (Scheme 6(iv), see SI for detailed kinetic data). These results indicate that metallonitrene formation constitutes the turnover-limiting step in the catalytic cycle. To further substantiate the critical role of copper catalysis in the spirocyclization process, representative terminal alkynes, including phenylacetylene, p-nitrophenylacetylene, and p-cyanophenylacetylene, were examined (Scheme 6(v)). While the electron-deficient alkynes (p-nitro and p-cyano) generated the corresponding spirocyclic intermediates, these products were found to be unstable under the reaction conditions. Subsequent reduction with NaBH4 afforded the corresponding stable derivatives (3bz, 3c). NMR analysis confirmed the absence of detectable diastereomeric mixtures. These observations further emphasize the critical role of copper catalysis in enabling the spirocyclization transformation.
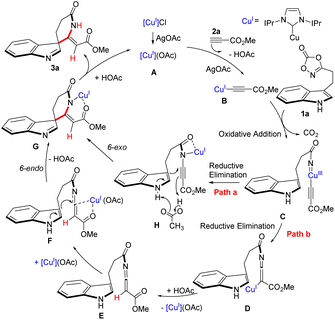
Additional mechanistic insights were obtained from density functional theory (DFT) calculations (Fig. 1; see the SI for computational details). To reduce computational cost while preserving steric and electronic features, the 2,6-diisopropylphenyl in IPrCuCl was replaced with 2,6-dimethylphenyl groups to generate the model complex A′. The DFT results indicate that the reaction of A′ with methyl propiolate to form intermediate B′ is exothermic by 6.60 kcal mol−1. This thermodynamic favorability is consistent with the experimental isolation of intermediate B at ambient temperatures. The initial coordination of the dioxazolone nitrogen atom to the copper center and the subsequent bond formation proceed through an endothermic transition state (TS1). In contrast, extrusion of CO2 to generate the thermodynamically stable chelated intermediate C′ is energetically favorable and exothermic. Notably, the calculated energy difference between the C′ → D′ and C′ → H′ pathways is only 0.01 kcal mol−1, indicating that both pathways are energetically comparable and may competitively operate under the reaction conditions. The formation of intermediate E′ was calculated to be thermodynamically favorable, consistent with time-resolved in situ 1H NMR monitoring (see SI). In contrast, analysis of the H–G transition state indicates that the proton migration step is associated with a relatively high activation barrier, suggesting that hydrogen transfer may be kinetically demanding within the catalytic cycle.
Based on experimental observations and literature reports,42,51–54 a plausible catalytic mechanism is proposed (Scheme 7). Initially, the CuI precatalyst coordinates with AgOAc to generate the active copper complex A. Subsequently, complex A reacts with methyl propiolate in the presence of AgOAc to afford the alkynyl copper intermediate B. Subsequent N–O bond cleavage of the dioxazolone, accompanied by CO2 extrusion, generates a copper-nitrenoid intermediate, which furnishes complex C via metal-nitrenoid insertion. From intermediate C, the catalytic cycle may proceed through reductive elimination to form either species D or H. Protonation of complex D then affords intermediate E, a step associated with hydrogen migration within the spirocyclic framework. To promote the 6-endo cyclization, CuI likely functions as a Lewis acid, facilitating the conversion of intermediate E to F (Path a). Alternatively, complex H may undergo proton migration to generate F (Path b). Intermediate F subsequently undergoes intramolecular 6-endo cyclization to form complex G. Finally, proton migration from G delivers the spiroindole δ-lactam product 3a, thereby completing the catalytic cycle.
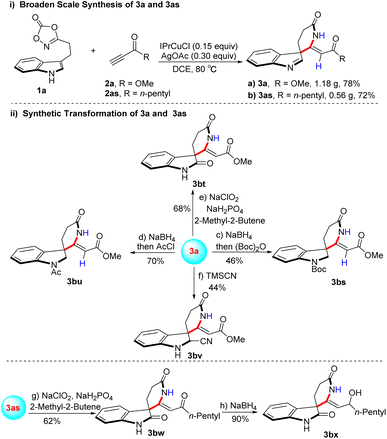
To further evaluate the practical applicability of this methodology, gram-scale synthesis and subsequent synthetic transformation of 3a and 3as were investigated (Scheme 8). Under scaled-up conditions, compounds 3a and 3as were successfully converted into the corresponding dioxazolone derivatives in 78% and 72% isolated yields, respectively (Scheme 8(i)). Compound 3a underwent reduction followed by protection to afford derivative 3bs in 56% isolated yield55. Subsequent acetylation with acetyl chloride (AcCl) proceeded smoothly, delivering compound 3bu in excellent yield (Scheme 8(ii)).56 Oxidation of 3a furnished 3bt in 68% isolated yield,57 while nucleophilic addition of trimethylsilyl cyanide (TMSCN) provided 3bv in 44% isolated yield.58 Similarly, oxidation of 3as afforded 3bw in 62% isolated yield, and subsequent reduction of 3bw gave 3bx in 90% isolated yield.
Conclusions
In summary, we have developed a novel, operationally simple, and efficient NHC copper-catalyzed strategy for the synthesis of spiroindole δ-lactams from dioxazolones and alkyne derivatives. The methodology exhibits high atom economy, consistently excellent yields, broad functional group tolerance, and outstanding Z-selectivity, thereby providing a valuable platform for the construction of structurally complex spirocyclic lactam frameworks. Mechanistic investigations revealed, for the first time, migration of the terminal alkyne hydrogen atom, leading to a distinctive 1,1-addition pathway that enables subsequent indole dearomatization. This transformation not only expands the mechanistic understanding of dioxazolone-alkyne reactivity but also establishes a novel and conceptually distinct synthetic strategy for the construction of spiroindole δ-lactam frameworks.Author contributions
Z. R. and T. F. contributed equally to this work. Z. R. and Y. Z. conceived, designed, and wrote the manuscript. All authors discussed the results and commented on the manuscript.Conflicts of interest
The authors declare no competing interests.Data availability
CCDC 2479571, 2479566, 2479567 and 2498755 contain the supplementary crystallographic data for this paper.59a–dAll other relevant data generated an analysed during this study are included in this article and its supplementary information (SI). Source data are provided with this paper. Supplementary information: experimental, spectroscopic, crystallographic and computational data. See DOI: https://doi.org/10.1039/d5sc09742h.
Acknowledgements
We acknowledge financial support from the National Natural Science Foundation of China (NSFC, No. 22301264). National Science of foundation of Shannxi Province (2023-JC-QN-0135, 2024JC-YBQN-0083), and Research Program of Yan'an University (YAU202407417).Notes and references
- E. Chupakhin, O. Babich, A. Prosekov, L. Asyakina and M. Krasavin, Molecules, 2019, 24, 4165 CrossRef CAS PubMed.
- C. V Galliford and K. A. Scheidt, Angew. Chem., Int. Ed., 2007, 46, 8748–8758 CrossRef PubMed.
- L. Yu, A. Dai, W. Zhang, A. Liao, S. Guo and J. Wu, J. Agric. Food Chem., 2022, 70, 10693–10707 CrossRef CAS PubMed.
- L. Chen, Q. J. Ding, J. J. Liu, S. Yang and Z. M. Zhang, Preparation of spiroindolinone derivatives as antiproliferative and anticancer agents. 2007, US pat. Search PubMed.
- Q. Ding, J. J. Liu and Z. Zhang, Preparation of spiroindolinone derivatives as antitumor agents, WO2007104714A1, 2007.
- M. J. Kornet and A. P. Thio, J. Med. Chem., 1976, 19, 892–898 CrossRef CAS PubMed.
- C. J. Barrow and H. H. Sun, J. Nat. Prod., 1994, 57, 471–476 CrossRef CAS PubMed.
- T. O. Larsen, K. Frydenvang, J. C. Frisvad and C. Christophersen, J. Nat. Prod., 1998, 61, 1154–1157 CrossRef CAS PubMed.
- R. D. Grigg, J. W. Rigoli, S. D. Pearce and J. M. Schomaker, Org. Lett., 2012, 14, 280–283 CrossRef CAS.
- T. R. Roose, D. S. Verdoorn, P. Mampuys, E. Ruijter, B. U. W. Maes and R. V. A. Orru, Chem. Soc. Rev., 2022, 51, 5842–5877 RSC.
- R. H. Morris, Chem. Soc. Rev., 2024, 53, 2808–2827 RSC.
- W. Wang, J. Wang, Y. Wang and Z. Xu, Chin. J. Chem., 2025, 43, 3221–3226 CrossRef CAS.
- J. Miao, Z.-X. Yu, C.-Y. Shi, X.-Q. Zhu, B. Zhou and L.-W. Ye, J. Org. Chem., 2025, 37, 13201–13211 CrossRef PubMed.
- S. Xie, S. Tang, M. Hou, W. Xie, M. Guan, T. Bai, L. He and G. Qiu, Org. Lett., 2024, 26, 11134–11139 CrossRef CAS PubMed.
- E. T. Marris, D. S. Rampon and J. M. Schomaker, Acc. Chem. Res., 2025, 58, 231–249 CrossRef CAS PubMed.
- X. Lai, J.-B. Liu, Y.-C. Wang and G. Qiu, Chem. Commun., 2021, 57, 2077–2080 RSC.
- J.-B. Liu, M. Ren, X. Lai and G. Qiu, Chem. Commun., 2021, 57, 4259–4262 RSC.
- Q. Wang and J. A. May, Org. Lett., 2020, 22, 3039–3044 CrossRef CAS PubMed.
- K. Hong, S. Zhou, W. Hu and X. Xu, Org. Chem. Front., 2020, 7, 1327–1333 RSC.
- J. R. Corbin, D. R. Ketelboeter, I. Fernández and J. M. Schomaker, J. Am. Chem. Soc., 2020, 142, 5568–5573 CrossRef CAS PubMed.
- K. Hong, H. Su, C. Pei, X. Lv, W. Hu, L. Qiu and X. Xu, Org. Lett., 2019, 21, 3328–3331 CrossRef CAS PubMed.
- R. A. Brawn, K. Zhu and J. S. Panek, Org. Lett., 2014, 16, 74–77 CrossRef CAS PubMed.
- A. R. Thornton and S. B. Blakey, J. Am. Chem. Soc., 2008, 130, 5020–5021 CrossRef CAS PubMed.
- A. R. Thornton, V. I. Martin and S. B. Blakey, J. Am. Chem. Soc., 2009, 131, 2434–2435 CrossRef CAS PubMed.
- A. H. Stoll and S. B. Blakey, J. Am. Chem. Soc., 2010, 132, 2108–2109 CrossRef CAS PubMed.
- N. Mace, A. R. Thornton and S. B. Blakey, Angew. Chem., Int. Ed., 2013, 52, 5836–5839 CrossRef CAS PubMed.
- X. Lyu, J. Zhang, D. Kim, S. Seo and S. Chang, J. Am. Chem. Soc., 2021, 143, 5867–5877 CrossRef CAS PubMed.
- S. Y. Hong, J. Son, D. Kim and S. Chang, J. Am. Chem. Soc., 2018, 140, 12359–12363 CrossRef CAS PubMed.
- H. Choi, X. Lyu, D. Kim, S. Seo and S. Chang, J. Am. Chem. Soc., 2022, 144, 10064–10074 CrossRef CAS PubMed.
- S. Gong, S.-H. Wang, Z.-L. Wang and W.-M. He, Adv. Synth. Catal., 2025, 367 DOI:10.1002/adsc.70158.
- K. M. Vliet and B. de Bruin, ACS Catal., 2020, 10, 4751–4769 CrossRef.
- J.-J. Tang, N. Yan, Y. Zhang, Y. Wang, M. Bao and X. Yu, Green Chem., 2023, 25, 7529–7533 RSC.
- Y. Du, X. Yu, J.-J. Tang, Y. Li, J. Fan, F. Li and M. Bao, J. Org. Chem., 2023, 88, 9783–9790 CrossRef CAS PubMed.
- J.-J. Tang, X. Yu, Y. Wang, Y. Yamamoto and M. Bao, Angew. Chem., Int. Ed., 2021, 60, 16426–16435 CrossRef CAS PubMed.
- S. Kim, S. L. Song, J. Zhang, D. Kim, S. Hong and S. Chang, J. Am. Chem. Soc., 2023, 145, 16238–16248 CrossRef CAS PubMed.
- S. Zhou, T. Liu and X. Bao, J. Catal., 2022, 415, 142–152 CrossRef CAS.
- H. Wang, H. Jung, F. Song, S. Zhu, Z. Bai, D. Chen, G. He, S. Chang and G. Chen, Nat. Chem., 2021, 13, 378–385 CrossRef CAS PubMed.
- S.-Y. Zhu, W.-J. He, G.-C. Shen, Z.-Q. Bai, F.-F. Song, G. He, H. Wang and G. Chen, Angew. Chem., Int. Ed., 2024, 63, e202312465 CrossRef CAS PubMed.
- Z. Bai, S. Zhu, Y. Hu, P. Yang, X. Chu, G. He, H. Wang and G. Chen, Nat. Commun., 2022, 13, 6445 CrossRef CAS PubMed.
- E. Lee, Y. Hwang, Y. B. Kim, D. Kim and S. Chang, J. Am. Chem. Soc., 2021, 143, 6363–6369 CrossRef CAS PubMed.
- S. Kim, E. Tufano, M. Barilli, E. Casali, Y. Kim, D. Kim, T. Massimini, G. Zanoni and S. Chang, J. Am. Chem. Soc., 2025, 147, 38772–38784 CrossRef CAS PubMed.
- K. M. Vliet, L. H. Polak, M. A. Siegle, J. I. Vlugt, C. F. Guerra and B. de Bruin, J. Am. Chem. Soc., 2019, 141, 15240–15249 CrossRef PubMed.
- T. P. Singh and O. M. Singh, Mini-Rev. Med. Chem., 2018, 18, 9–25 CrossRef CAS PubMed.
- J. A. Homer and J. Sperry, J. Nat. Prod., 2017, 80, 2178–2187 CrossRef CAS PubMed.
- A. Mehra, V. Sharma, A. Verma, S. Venugopal, A. Mittal, G. Singh and B. Kaur, ChemistrySelect, 2022, 7, e202202361 CrossRef CAS.
- M. Pang, H. Chang, Z. Feng and J. Zhang, Chin. J. Org. Chem., 2023, 43, 1271–1291 CrossRef CAS.
- L.-M. Yu, H. Chen, W. Fang, R. Cai, Y. Tao, Y. Li and H. Dong, Org. Biomol. Chem., 2024, 22, 7074–7091 RSC.
- F. Buttard and X. Guinchard, ACS Catal., 2023, 13, 9442–9475 CrossRef CAS.
- F.-T. Sheng, J.-Y. Wang, W. Tan, Y.-C. Zhang and F. Shi, Org. Chem. Front., 2020, 7, 3967–3998 RSC.
- Y. Hwang, Y. Park, Y. B. Kim, D. Kim and S. Chang, Angew. Chem., Int. Ed., 2018, 57, 13565–13569 CrossRef CAS PubMed.
- T. R. Roose, F. McSorley, B. Groenhuijzen, J. M. Saya, B. U. W. Maes, R. V. A. Orrù and E. Ruijter, J. Org. Chem., 2023, 88, 17345–17355 CrossRef CAS PubMed.
- A. Makarem, R. Berg, F. Rominger and B. F. Straub, Angew. Chem., Int. Ed., 2015, 54, 7431–7435 CrossRef CAS PubMed.
- M. K. Armstrong, M. B. Goodstein and G. Lalic, J. Am. Chem. Soc., 2018, 140, 10233–10241 CrossRef CAS.
- Z. Ren, T. Feng, T. Gao, B. Han, R. Guo, H. Ma, J.-J. Wang and Y. Zhang, Org. Lett., 2024, 26(40), 8532–8536 CrossRef CAS PubMed.
- R. Chouhan, H. Bhattacharyya and S. K. Das, Org. Lett., 2024, 26, 1088–1093 CrossRef CAS PubMed.
- M. Feng, A. J. Fernandes, R. Meyrelles and N. Maulide, Chem, 2023, 9, 1538–1548 CAS.
- X. Zhang, Q. Song, S. Liu, P. Sivaguru, Z. Liu, Y. Yang, Y. Ning, E. A. Anderson, G. D. Ruiter and X. Bi, Nat. Chem., 2025, 17, 215–225 CrossRef CAS PubMed.
- D.-Y. Zhu, L. Fang, H. Han, Y. Wang and J.-B. Xia, Org. Lett., 2017, 19, 4259–4262 CrossRef CAS PubMed.
- (a) CCDC 2479571: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p765b; (b) CCDC 2479566: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p7605; (c) CCDC 2479567: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p7616; (d) CCDC 2498755: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pw50s.
Footnote |
| † F Z.R. and T.F. contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |