Open Access Article
Open Access ArticleTuneable electronic coupling in linked bis(cubane) cobalt-oxo clusters
Vincent J. P.
Maddi
 ab and
T. Don
Tilley
ab and
T. Don
Tilley
 ab
ab
aDepartment of Chemistry, University of California, Berkeley, California 94720, USA. E-mail: tdtilley@berkeley.edu
bChemical Sciences Division, Lawrence Berkeley National Laboratory, Berkeley, California 94720, USA
First published on 21st March 2026
Abstract
A family of cobalt-oxo bis(cubane) complexes wherein each subunit is derived from the Co4O4 cubane, a known water oxidation catalyst, was synthesized. Both 4,4′-bipyrdine and pyrazine were demonstrated to serve as viable bridging ligands. Through an analysis of their half-wave splitting potentials, it was determined that pyrazine-bridged bis(cubane)s exhibit inter-cubane electronic coupling, and that this coupling may be tuned through ligand substitution. Electrostatic contributions to the half-wave splitting potentials were evaluated and found to result in “non-conformist” behavior related to the ion-pairing ability of the electrolytes.
Introduction
Research on catalysts for the oxygen evolution reaction (OER), motivated by the goal of creating an artificial photosynthesis system, has focused on multimetallic cooperativity as playing a key role in overcoming the intrinsic challenges associated with a four-electron, four-proton oxidation process.1–6 This pertains to the Mn4CaO5 cluster of the oxygen-evolving complex (OEC) for water oxidation in photosystem II,7 and to heterogeneous metal oxide catalysts such as cobalt oxyhydroxide (CoOOH).8–11 Several metals working in concert are essential for accumulating and storing the multiple oxidizing equivalents (electron holes) required to drive the four-electron splitting of water.12 Thus, an important challenge for understanding and studying this process is the establishment of a detailed mechanism, which has proven difficult given the complexity of the process.The tetracobalt-oxo cubane Co4O4(Py)4(OAc)4 is a compelling model system for the study of multimetallic cooperativity in water-oxidation catalysis. It bears a structural resemblance to the active edge site in layered CoOOH and has demonstrated competence in the conversion of hydroxide to molecular oxygen.13–17 In this regard, the oxidized (CoIII3CoIV) cubane has been established as an early intermediate in water splitting and has therefore been subjected to substantial interrogation.18–20
Previous studies on establishing an in-depth mechanistic understanding of water-oxidation mediated by the cobalt-oxo cubane21–28 indicate that a key mechanistic step in solution is the disproportionation of two mono-oxidized cubanes to form a highly oxidized intermediate that then rapidly proceeds to the O–O bond-forming step (Scheme 1).29 Accordingly, modeling and modifying this high-valent intermediate, or the electron-transfer reaction that proceeds it, are potentially of great scientific interest. Moreover, such studies into the role of electron-transfer processes in water-oxidation catalysis might provide insight into the mechanism of hole accumulation in heterogenous materials, as has been investigated in hematite catalysts.30
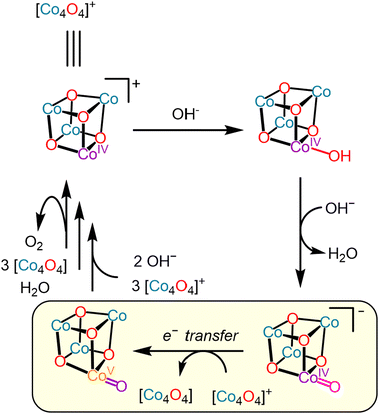 | ||
| Scheme 1 Proposed mechanism for reduction of the oxidized cubane by hydroxide through a highly oxidized intermediate preceded by a disproportionation (highlighted). | ||
Ligand substitutions have allowed electrochemical access to a doubly oxidized cubane (formally CoIII2CoIV2 or CoIII3CoV) at potentials as low as 0.8 V vs. Fc/Fc+.31 Unfortunately, a doubly oxidized cubane has not yet been isolated. A reasonable strategy for accessing and studying the behavior of more highly oxidized clusters would be the linkage of multiple clusters to create systems that can bear a larger redox load but have overall similar degrees of charge distribution. Such expanded clusters would be valuable synthetic targets given the multi-electron nature of water-oxidation.
An important consideration in the design of expanded cobalt-oxo clusters would be to ensure efficient electronic communication between the units. Without such communication each subunit would behave independently, and neither would be able to access the full redox breadth of the assembly; thus, for an expanded cobalt-oxo cluster, it is essential to initially establish the existence of electronic coupling.
Beginning with the Creutz–Taube ion, mixed-valent species have served as valuable tools for studying electron-transfer reactions and electron delocalization in binuclear species and in multimetallic clusters.32–34 Electrochemistry is an established tool for estimating electronic coupling, especially for systems where the mixed-valent state (in which electronic coupling can be measured directly from their intervalence charge transfer bands) cannot be isolated. In general, the half-wave potential splitting, ΔE1/2, the difference between the first and second redox potentials in a mixed-valent species, is proportional to the electronic coupling between the donor and acceptor sites.35 Electrochemistry, however, is not a direct measure of electronic coupling and such data must be handled with care in the absence of other corroborating evidence.
The study presented here describes the synthesis of expanded cobalt-oxo cubanes possessing ditopic bridging ligands and various additional ligand sets. Electrochemical studies on these complexes allow assessment of the extent of electronic communication between the two cubane units. The presence of inter-cubane electronic coupling indicates that these bis(cubane)s display multi-electron chemistry that serves as a model for the encounter complex involving electron transfer between clusters in catalytic oxidation chemistry. The ready accessibility of the doubly oxidized state also indicates the potential utility of bis(cubane)s as electrocatalysts with enhanced charge storage properties.
Results and discussion
Synthesis and characterization of cobalt-oxo bis(cubane)s
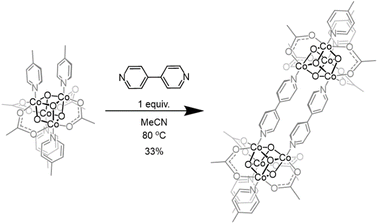
The Co4O4 cubane platform possesses significant synthetic versatility, providing routes to symmetrically and unsymmetrically substituted cubes via “self-assembly” from CoII, pyridine and HOOH reactants, as well as by substitution of the dative, L-type, and anionic, X-type, ligands.31 Therefore, it was presumed that a dimer in which two Co4O4 cubanes share a common bridging bidentate ligand should be accessible. Further, based on previous work involving cubane porous frameworks wherein bridging tris(carboxylate) donors were shown to be prone to decomposition, it was reasoned that bridging L-type donors, binding through the pyridine positions of the cubane, would be the preferred type of bridging ligand.36Initial attempts to prepare a bis(cubane) began with use of 4,4′-bipyridine (bipy) as a bridging ligand. When a solution of the 4-methylpyridine (MePy) substituted cubane, Co4(µ3-O4)(OAc)4(MePy)4, was heated to 80 °C and one equiv of bipy in acetonitrile was added dropwise over 5 h followed by an additional 13 h of heating, a color change from forest green to rust red was observed. The desired bis(cubane) product Co8(µ3-O4)8(OAc)8(MePy)4(µ2-bipy)2 (MePy-OAc-bipy) was isolated by column chromatography followed by crystallization from a solution of CH2Cl2 layered with pentane to give red crystals of MePy-OAc-bipy bis(cubane) in 33% yield (Scheme 2). The dimeric nature of this product was verified by HR-ESI-MS and the symmetry about the bipy ligand in the 1H NMR spectrum. Similar results were obtained using both the pyridine (Py)- and DMAP-substituted mono-cubanes; however, in these cases poor chromatographic separations precluded isolation of pure product.
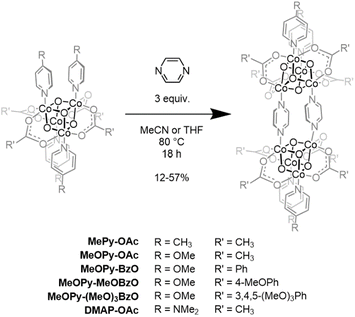
Inspired by the body of work employing pyrazine (Pyz) as a bridging ligand in multimetallic systems,37,38 it was chosen as the next target for the coupling of two cubane units. The Co4(µ3-O4)(OAc)4(MePy)4 cubane and three equivalents of Pyz were dissolved in acetonitrile and sealed in a pressure vessel which was then immersed in an 80 °C oil bath. After heating for 18 h, the mixture was filtered and the resulting red solid was washed three times with acetonitrile to yield pure Co8(µ3-O)8(OAc)8(MePy)4(µ2-Pyz)2 (MePy-OAc) bis(cubane) in 45% yield (Scheme 3; hereafter all bis(cubane)s discussed will possess Pyz bridges, unless otherwise stated). The dimeric structure was corroborated by HR-ESI-MS and a 1H NMR spectrum showing a single Pyz resonance. Previous reports have indicated that substitution of the cubane's L-type ligands occurs primarily when pKa(L′H+) > pKa(LH+) where L is the outgoing ligand and L′ is the incoming ligand.35 The modestly high yield of the pyrazine-bridged bis(cubane) seems to run counter to this trend (pKa-MePy = 6.02; pKa-Pyz = 1.1).39,40 Because no yield could be obtained when more solubilizing ligands like 4-tertbutylpyridine were used it is hypothesized that precipitation of MePy-OAc from the reaction mixture provides a driving force for the ligand exchange equilibrium. Further, it was found that a judicious choice of solvent favoring precipitation of the bis(cubane) relative to its monomeric precursor allowed for the isolation of six bis(cubane)s via this dimerization strategy (Scheme 3). Notably, this procedure is not applicable to cubanes possessing pyridine ligands with electron withdrawing substituents such as 4-cyano-, 4-trifluoromethyl-, or 4-acetyl-pyridine. When subjected to these dimerization conditions, a rust-red precipitate containing cubane subunits (by FT-IR analysis) formed but could not be solubilized in available solvents suggesting that they may correspond to oligomeric products.
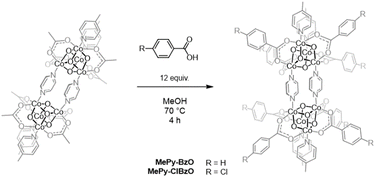
Several additional bis(cubane)s that could not be synthesized by this dimerization strategy were instead prepared by leveraging the fact that benzoate-substituted bis(cubane)s have poor solubility in protic solvents compared to their acetate-substituted analogues (Scheme 4). For example, when a solution of the MePy-OAc bis(cubane) and 12 equiv. of benzoic acid in methanol was heated to reflux for 4 h, a red precipitate formed and was purified chromatographically and by crystallization from CH2Cl2 layered with pentane to give the MePy-BzO bis(cubane) in 16% yield.
In total, a library of nine bis(cubane) complexes encompassing different bridging ligands and a broad range of donor ligands was prepared using strategies of dimerization and X-type ligand substitution.
Electrochemical studies
Cyclic voltammograms (CVs) were collected on each bis(cubane) to obtain qualitative information regarding the extent of electronic coupling between the two cubane cores (Fig. 1a). Initial experiments were conducted with a 100 mM solution of tetrabutylammonium tetrakis(pentafluorophenyl)borate ([n-Bu4N][BArF20]) as the electrolyte in CH2Cl2 (see SI for details). In all cases a reversible event was observed for both the [Co(III)4O4–Pyz–Co(III)4O4]8+/[Co(IV)Co(III)3O4–Pyz–Co(III)4O4]9+ (E1) and [Co(IV)Co(III)3O4–Pyz–Co(III)4O4]9+/[Co(IV)Co(III)3O4–Pyz–Co(IV)Co(III)3O4]10+ (E2) redox couples. The two cubane units are assumed to proceed through sequential oxidations with a mixed-valent intermediate (Fig. 1b). Values for first and second redox potentials, E1 and E2, were determined through differential pulse voltammetry (DPV) due to improved resolution compared to the cyclic voltammograms (Fig. 1c). Values are compiled in Table 1.Despite their hypothetical accessibility based on the electrochemical data, no bis(cubane)s in the mixed-valent [Co(IV)Co(III)7O8]9+ state could be isolated for intervalence charge transfer (ICVT) band analysis. The use of spectroelectrochemistry also proved fruitless in the positive identification of an IVCT band (see SI). However, the electrochemical data was further analyzed to assess the existence of inter-cubane electronic coupling.
The half-wave potential splitting, ΔE1/2, calculated as the difference between E1 and E2, is an established measure for the degree of electronic interaction (electronic coupling) between identical, interconnected, redox-active moieties in mixed-valent systems with well-defined donor, acceptor, and bridge sites such as the two cubane cores in the bis(cubane).34 Larger values of ΔE1/2 arise from greater stabilization of the mixed-valent state which is typically assumed to be the result of resonance stabilization accompanying increased electronic coupling.34,41 Values for ΔE1/2 for bis(cubane)s are compiled in Table 1.
In the case of the MePy-OAc-bipy bis(cubane) only a single redox wave was observed, but use of the Randles–Sevcik equation determined that this corresponds to a net two-electron [Co(III)4O4–bipy–Co(III)4O4]8+/[Co(IV)Co(III)3O4–Pyz–Co(IV)Co(III)3O4]10+ couple (Fig. 2). Therefore, with bipy as a bridging ligand the two cubane cores are electronically independent and exhibit negligible inter-cubane hole delocalization in the singly oxidized state. Longer donor–acceptor distances correlate to weaker electronic coupling and weaker electrostatic interactions (vide infra) and thus it is expected that the bipy-bridged system exhibits a ΔE1/2 value smaller than those of Pyz-bridged analogues.42
As was previously reported in the case of monomeric cubanes,35 more donating ligands lead to a cathodic shift in E1 while more withdrawing ligands lead to an anodic shift implying that E1 is a good proxy for overall ligand donor ability. In mixed-valent systems it is predicted that more strongly donating ligands raise the energy of the d-orbital manifold closer to the π* orbitals of the bridging ligand which then lowers the energy barrier for electron transfer.43 A lower barrier to electron transfer corresponds to stronger electronic coupling, a net stabilization of the mixed-valent state compared to the doubly-oxidized state, and hence an increase in ΔE1/2.35,41 Therefore, a good correspondence between E1 (i.e., ligand donor ability) and ΔE1/2, as is observed in the bis(cubane) system (Fig. 3), should be indicative of electronic coupling. This observed trend is similar to that reported by Kubiak and coworkers for their mixed-valent pyrazine-bridged bis(µ3-oxo-centered triruthenium-acetate) clusters.37,44
While half-wave splitting potentials are frequently used as proxies for electronic coupling (Hab), ΔE1/2 is dependent on several additional factors. Eqn (1) shows the relation between ΔE1/2 and the free energy of comproportionation, ΔGc, while eqn (2) shows the several variables which comprise ΔGc and thus can influence ΔE1/2.
| −ΔGc = nFΔE1/2 | (1) |
| ΔGc = ΔGres + ΔGstat + ΔGind + ΔGex + ΔGel | (2) |
Many of the above mentioned “non-conformist” behaviors arise from changes in the electrostatic term, ΔGel, which is sensitive to medium effects such as solvent and electrolyte. Previously, Lang and coworkers identified “non-conformist” behavior in a series of diferrocenyl functionalized pyrroles that was traced to increased electrostatic repulsion resulting from increased charge localization with more electron withdrawing ligands. By comparing half-wave splitting potentials with direct optical measurements of electronic coupling, Lang and coworkers were able to demonstrate that complexes with larger values of ΔGel experience large changes to ΔE1/2 when changing to an electrolyte with stronger ion pairing which better “shields” the electrostatic repulsion.49 Electrostatic contributions can therefore be gauged (and thus non-conformist behavior ruled out) by varying the electrolyte (e.g., changing [n-Bu4N][BArF20] to [n-Bu4N][PF6]) and determining the value of ΔΔE1/2 (defined as ΔE1/2[BArF20] – ΔE1/2[PF6]).
Electrochemical measurements (CV and DPV) were conducted on each bis(cubane) in a CH2Cl2 solution containing 250 mM [n-Bu4N][PF6] as the electrolyte (see SI for details). The results of these measurements are shown in Table 2.
| Bis(Cubane) | E 1 (mV) | E 2 (mV) | ΔE1/2 (mV) | ΔΔE1/2 (mV) |
|---|---|---|---|---|
| a Only one redox wave identified. | ||||
| MePy-OAc-Bipya | 291 | — | — | — |
| DMAP-OAc | 293 | 420 | 127 | 40 |
| MePy-OAc | 407 | 492 | 85 | 84 |
| MePy-BzO | 453 | 593 | 140 | 20 |
| MePy-ClBzO | 597 | 718 | 121 | 27 |
| MeOPy-OAc | 424 | 494 | 70 | 90 |
| MeOPy-BzO | 453 | 600 | 147 | 5 |
| MeOPy-MeOBzO | 387 | 533 | 146 | 8 |
| MeOPy-(MeO)3BzO | 588 | 680 | 92 | 67 |
The large variance in ΔΔE1/2 immediately suggests a significant electrostatic influence on values of ΔE1/2. One source of this wide range should be attributed to the different hydrodynamic radii of the different bis(cubane)s. DOSY NMR shows that, as representative samples, the MePy-ClBzO bis(cubane) has a slower diffusion coefficient (5.78 × 10−10 m2 s−1) and thus larger hydrodynamic radius than the MePy-OAc bis(cubane) (6.43 × 10−10 m2 s−1). A comparison of acetate-functionalized and benzoate-functionalized bis(cubane)s as a whole indicates that the latter generally have smaller values of ΔΔE1/2 (Fig. 4). Following the electrostatic model of Barrière,50 this is to be expected; analytes with larger radii have larger distances between their charged centers and the counterion experiencing less coulombic stabilization and electrostatic shielding from ion-pairing. For further analysis it is thus appropriate to separate the acetate- and benzoate-functionalized bis(cubane)s.
 | ||
| Fig. 4 Comparing the CV data in [n-Bu4N][BArF20] (gold) and in [n-Bu4N][PF6] (blue) for (a) acetate and (b) benzoate functionalized MePy bis(cubane)s with a Pyz bridge. | ||
When E1 is plotted against ΔE1/2 in the [n-Bu4N][PF6] series (Fig. 5), a much stronger correlation between ligand donor ability and the potential splitting is observed. This supports the assumption that the electronic coupling between the two cubane cores has a greater dependence on the ligand choice than the electrochemical data collected with just the [n-Bu4N][BArF20] electrolyte would suggest. Once the bis(cubane) radii are considered, complexes with less electron-donating ligands have larger values of ΔΔE1/2 implying that, as in the case of Lang's diferrocenyl complexes, these ligands result in greater partial positive charge on the donor–acceptor moieties and thus greater electrostatic repulsion resulting in somewhat “non-conformist” behavior.
One outlier with the [n-Bu4N][PF6] electrolyte is the MeOPy-(MeO)3BzO bis(cubane) (Fig. 6). This complex exhibits the largest ΔΔE1/2 value of all the benzoate bis(cubane)s despite having relatively more electron-donating carboxylates than its nearest neighbor with respect to E1, the MePy-ClBzO bis(cubane). A closer analysis suggests that radius and electron-donor ability are not the only factors at play when determining the electrostatic contribution to ΔE1/2 and that other structural factors related to the trimethoxy groups may be relevant (vide infra).
In addition to having the largest ΔΔE1/2 value, the MeOPy-(MeO)3BzO bis(cubane) also has the largest change in its first oxidation potential between electrolytes, ΔE1 (E1[PF6] − E1[BArF20]), when compared to the other benzoate-substituted bis(cubane)s. Eqn (3) shows the relationship between E1 and the association constant between the singly-oxidized bis(cubane) and the electrolyte anion (KA) relative to the oxidation potential in the idealized absence of ion-pairing  .51
.51
 | (3) |
| Bis(Cubane) | ΔE1 (mV) | ΔΔE1/2 (mV) |
|---|---|---|
| MeOPy-BzO | 21 | 5 |
| MeOPy-MeOBzO | 64 | 8 |
| MePy-BzO | 82 | 20 |
| MePy-ClBzO | 120 | 27 |
| MeOPy-(MeO)3BzO | 161 | 67 |
The combined electrochemical data collected with both [n-Bu4N][BArF20] and [n-Bu4N][PF6] electrolytes provides strong evidence for the existence of electronic coupling between the two cubane cores in the bis(cubane) system. Because ΔGres is known to generally depend on ligand donor properties, a positive correlation (as opposed to no correlation or a slope of zero) between the ligand donor ability (E1) and ΔE1/2 is strongly indicative of an increasing (and thus non-zero) ΔGres across the series. The electrostatic term, ΔGel, is already accounted for in the analysis and it is improbable that ΔGind or ΔGex could be responsible for the large magnitudes of change observed with ligand modification. Thus, it can be concluded that the bis(cubane) system behaves in a fashion similar to that of the analogous mixed-valent pyrazine-bridged bis(µ3-oxo-centered triruthenium-acetate) clusters of Kubiak et al., albeit with smaller magnitudes of electronic coupling, as might be expected when moving from second to first-row transition metals.52
Conclusions
A family of bis(cubane) complexes was synthesized and examined electrochemically to determine the extent of cluster-cluster interaction. As with previously reported mono-cubanes, the bis(cubane) system is synthetically versatile and can be tuned electronically with ligand substitution. The degree of electronic coupling can be tuned via ligand substitution with more strongly donating ligands leading to stronger coupling. Future work will seek to increase electronic coupling by modifying the bridging ligand as well as attempting to build larger clusters capable of greater charge storage. The presence of electronic coupling opens the door to many future investigations with respect to water-oxidation at the bis(cubane) including electron-transfer rate measurements, hole-coupling studies in highly oxidized bis(cubane)s and kinetic studies that would seek to relate the magnitude of electronic coupling to the rate of water oxidation. Furthermore, because the bis(cubane) could bypass intermolecular electron transfer in water-oxidation, a detailed kinetic analysis comparing it to the mono-cubane could aid in determining if the PCET step leading to the high-valent, formally Co(V) intermediate proceeds in a stepwise or concerted fashion and could possibly yield significant rate acceleration in the bis(cubane) case.17,29,53 Towards these ends, the synthesis and isolation of a doubly-oxidized bis(cubane) is subject to ongoing investigation.In addition to directly modifying the electronic properties of the cubane core, the various ligands on the bis(cubane) enable modulation of other factors affecting electrochemistry of the bis(cubane). Electrostatic factors such as ion-pairing were shown to be significant contributors to half-wave potential splitting in these complexes and are highly dependent on analyte size. More strongly coordinating anions could lower the barrier to forming the high-valent intermediate and thus further promote catalysis. The sensitivity of the bis(cubane) redox chemistry to medium effects such as solvent polarity and counterion pairing ability can also be leveraged towards other oxidative transformations, particularly C–H bond activations, which have already been studied for a related mono-cubane.54,55 The ability to activate strong C–H bonds is limited by the bond dissociation energy of the resultant µ3-O–H bond which is, in turn, dependent on the cubane oxidation potential. The sensitivity of the bis(cubane) redox potential to medium effects implies that its reactivity can be tuned through a judicious choice of solvent and counterion enabling more selective substrate oxidations.
Overall, the bis(cubane) system described here provides a discrete, molecular set of electronically coupled multimetallic clusters that afford many future opportunities to increase the understanding of hole transport, storage, and utilization for multi-electron oxidative processes.
Author contributions
The manuscript was written through contributions of all authors. VM performed the experimental work. VM and TDT conceptualized the research. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Supplementary information: experimental details, characterization data, and electrochemical data. See DOI: https://doi.org/10.1039/d5sc09713d.Acknowledgements
This work was funded by the U.S. Department of Energy, Office of Science, Office of Basic Energy Sciences, Chemical Sciences, Geosciences, and Biosciences Division, under Contract DE-AC02-05CH11231. The LBNL Catalysis Laboratory provided HR-ESI-MS instrumentation. NMR spectra were collected at the College of Chemistry Pines Magnetic Resonance Center's Core NMR facility at the University of California, Berkeley. T. D. T. is grateful to Mr and Ms. Chen for their funding of the PMP Tech Chancellor's Chair in Chemistry. The authors thank Drs Hasan Celik, Raynald Giovine, and Pines Magnetic Resonance Center's Core NMR Facility (PMRC Core) for spectroscopic assistance. The instrument used in this work is supported by the National Science Foundation under Grant No. 2018784. The authors Drs Cooper Citek, and Chithra Asokan for assistance with LBNL Catalysis Laboratory instrumentation. Spectroelectrochemical data was collected at the Molecular Foundry supported by the Office of Science, Office of Basic Energy Sciences, of the U.S. Department of Energy under Contract No. DE-AC02-05CH11231, under proposal number MFP-08851 and Dr Steve Shelton is thanked for his assistance. Dr T. Alexander Wheeler is thanked for his thoughtful discussion and consideration of this manuscript.References
- M. D. Kärkäs, O. Verho, E. V. Johnston and B. Åkermark, Chem. Rev., 2014, 114, 11863–12001 CrossRef PubMed.
- N. S. Lewis and D. G. Nocera, Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 15729–15735 CrossRef CAS PubMed.
- G. F. Moore and G. W. Brudvig, Annu. Rev. Condens. Matter Phys., 2011, 2, 303–327 CrossRef CAS.
- R. Eisenberg and H. B. Gray, Inorg. Chem., 2008, 47, 1697–1699 CrossRef CAS PubMed.
- W. Rüttinger and G. C. Dismukes, Chem. Rev., 1997, 97, 1–24 CrossRef PubMed.
- M. G. Walter, E. L. Warren, J. R. McKone, S. W. Boettcher, Q. Mi, E. A. Santori and N. S. Lewis, Chem. Rev., 2010, 110, 6446–6473 CrossRef CAS PubMed.
- Y. Umena, K. Kawakami, J. R. Shen and N. Kamiya, Nature, 2011, 473, 55–60 CrossRef CAS PubMed.
- M. W. Kanan, J. Yano, Y. Surendranath, M. Dincă, V. K. Yachandra and D. G. Nocera, J. Am. Chem. Soc., 2010, 132, 13692–13701 CrossRef CAS PubMed.
- M. Risch, V. Khare, I. Zaharieva, L. Gerencser, P. Chernev and H. Dau, J. Am. Chem. Soc., 2009, 131, 6936–6937 CrossRef CAS PubMed.
- A. M. Ullman, C. N. Brodsky, N. Li, S. L. Zheng and D. G. Nocera, J. Am. Chem. Soc., 2016, 138, 4229–4236 CrossRef CAS PubMed.
- K. Zeng and D. Zhang, Prog. Energy Combust. Sci., 2010, 36, 307–326 CrossRef CAS.
- J. Li, C. A. Triana, W. Wan, D. P. A. Saseendran, Y. Zhao, S. E. Balaghi, S. Heidari and G. R. Patzke, Chem. Soc. Rev., 2021, 50, 2444–2485 RSC.
- A. M. Ullman, Y. Liu, M. Huynh, D. K. Bediako, H. Wang, B. L. Anderson, D. C. Powers, J. J. Breen, H. D. Abruña and D. G. Nocera, J. Am. Chem. Soc., 2014, 136, 17681–17688 CrossRef CAS PubMed.
- J. K. Beattie, T. W. Hambley, J. A. Klepetko, A. F. Masters and P. Turner, Polyhedron, 1998, 17, 1343–1354 CrossRef CAS.
- R. Chakrabarty, S. J. Bora and B. K. Das, Inorg. Chem., 2007, 46, 9450–9462 CrossRef CAS PubMed.
- G. L. Ganga, F. Puntoriero, S. Campagna, I. Bazzan, S. Berardi, M. Bonchio, A. Sartorel, M. Natali and F. Scandola, Faraday Discuss., 2012, 155, 177–190 RSC.
- N. S. McCool, D. M. Robinson, J. E. Sheats and G. C. Dismukes, J. Am. Chem. Soc., 2011, 133, 11446–11449 CrossRef CAS PubMed.
- J. G. McAlpin, T. A. Stich, C. A. Ohlin, Y. Surendranath, D. G. Nocera, W. H. Casey and R. D. Britt, J. Am. Chem. Soc., 2011, 133, 15444–15452 CrossRef CAS PubMed.
- C. N. Brodsky, R. G. Hadt, D. Hayes, B. J. Reinhart, N. Li, L. X. Chen and D. G. Nocera, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 3855–3860 CrossRef CAS PubMed.
- R. G. Hadt, D. Hayes, C. N. Brodsky, A. M. Ullman, D. M. Casa, M. H. Upton, D. G. Nocera and L. X. Chen, J. Am. Chem. Soc., 2016, 138, 11017–11030 CrossRef CAS PubMed.
- X. Li and P. E. M. Siegbahn, J. Am. Chem. Soc., 2013, 135, 13804–13813 CrossRef CAS PubMed.
- A. Fernando and C. M. Aikens, J. Phys. Chem. C, 2015, 119, 11072–11085 CrossRef CAS.
- P. F. Smith, L. Hunt, A. B. Laursen, V. Sagar, S. Kaushik, K. U. D. Calvinho, G. Marotta, E. Mosconi, F. De Angelis and G. C. Dismukes, J. Am. Chem. Soc., 2015, 137, 15460–15468 CrossRef CAS PubMed.
- H. Liu and H. Frei, ACS Catal., 2020, 10, 2138–2147 CrossRef CAS.
- L. P. Wang and T. Van Voorhis, J. Phys. Chem. Lett., 2011, 2, 2200–2204 CrossRef CAS.
- P. F. Smith, C. Kaplan, J. E. Sheats, D. M. Robinson, N. S. McCool, N. Mezle and G. C. Dismukes, Inorg. Chem., 2014, 53, 2113–2121 CrossRef CAS PubMed.
- G. Mattioli, P. Giannozzi, A. Amore Bonapasta and L. Guidoni, J. Am. Chem. Soc., 2013, 135, 15353–15363 CrossRef CAS PubMed.
- R. Ezhov, A. K. Ravari, G. Bury, P. F. Smith and Y. Pushkar, Chem Catal., 2021, 1, 407–422 CAS.
- A. I. Nguyen, M. S. Ziegler, P. Oña-Burgos, M. Sturzbecher-Hohne, W. Kim, D. E. Bellone and T. D. Tilley, J. Am. Chem. Soc., 2015, 137, 12865–12872 CrossRef CAS PubMed.
- C. A. Mesa, L. Francàs, K. R. Yang, P. Garrido-Barros, E. Pastor, Y. Ma, A. Kafizas, T. E. Rosser, M. T. Mayer, E. Reisner, M. Grätzel, V. S. Batista and J. R. Durrant, Nat. Chem., 2020, 12, 82–89 CrossRef CAS PubMed.
- A. I. Nguyen, J. Wang, D. S. Levine, M. S. Ziegler and T. D. Tilley, Chem. Sci., 2017, 8, 4274–4284 RSC.
- M. B. Robin and P. Day, in Adv. Inorg. Chem. Radiochem., ed. H. J. Emeléus and A. G. Sharpe, Academic Press, 1968, vol. 10, pp. 247–422 Search PubMed.
- C. P. Kubiak, Inorg. Chem., 2013, 52, 5663–5676 CrossRef CAS PubMed.
- M. D. Ward, Chem. Soc. Rev., 1995, 24, 121–134 RSC.
- R. F. Winter, Organometallics, 2014, 33, 4517–4536 CrossRef CAS.
- A. I. Nguyen, K. M. Van Allsburg, M. W. Terban, M. Bajdich, J. Oktawiec, J. Amtawong, M. S. Ziegler, J. P. Dombrowski, K. V. Lakshmi, W. S. Drisdell, J. Yano, S. J. L. Billinge and T. D. Tilley, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 11630–11639 CrossRef CAS PubMed.
- T. Ito, T. Hamaguchi, H. Nagino, T. Yamaguchi, H. Kido, I. S. Zavarine, T. Richmond, J. Washington and C. P. Kubiak, J. Am. Chem. Soc., 1999, 121, 4625–4632 CrossRef CAS.
- C. Creutz and H. Taube, J. Am. Chem. Soc., 1969, 91, 3988–3989 CrossRef CAS.
- D. Keyworth, J. Org. Chem., 1959, 24, 1355–1356 Search PubMed.
- K. Clarke and K. Rothwell, J. Chem. Soc., 1960, 1885–1895 RSC.
- N. S. Hush, Electrochim. Acta, 1968, 13, 1005–1023 CrossRef CAS.
- N. S. Hush, Coord. Chem. Rev., 1985, 64, 135–157 CrossRef CAS.
- J. C. Salsman, S. Ronco, C. H. Londergan and C. P. Kubiak, Inorg. Chem., 2006, 45, 547–554 CrossRef CAS PubMed.
- T. Cheng, M. Meng, H. Lei and C. Y. Liu, Inorg. Chem., 2014, 53, 9213–9221 CrossRef CAS PubMed.
- R. J. Crutchley, in Adv. Inorg. Chem., ed. A. G. Sykes, Academic Press, 1994, vol. 41, pp. 273–325 Search PubMed.
- C. E. B. Evans, M. L. Naklicki, A. R. Rezvani, C. A. White, V. V. Kondratiev and R. J. Crutchley, J. Am. Chem. Soc., 1998, 120, 13096–13103 CrossRef CAS.
- D. E. Richardson and H. Taube, Coord. Chem. Rev., 1984, 60, 107–129 CrossRef CAS.
- Y. C. Lin, W. T. Chen, J. Tai, D. Su, S. Y. Huang, I. Lin, J. L. Lin, M. M. Lee, M. F. Chiou, Y. H. Liu, K. S. Kwan, Y. J. Chen and H. Y. Chen, Inorg. Chem., 2009, 48, 1857–1870 CrossRef CAS PubMed.
- S. W. Lehrich, A. Hildebrandt, M. Korb and H. Lang, J. Organomet. Chem., 2015, 792, 37–45 CrossRef CAS.
- F. Barrière, Organometallics, 2014, 33, 5046–5048 CrossRef.
- W. E. Geiger and F. Barrière, Acc. Chem. Res., 2010, 43, 1030–1039 CrossRef CAS PubMed.
- E. J. Piechota and G. J. Meyer, J. Chem. Educ., 2019, 96, 2450–2466 CrossRef CAS.
- M. D. Symes, Y. Surendranath, D. A. Lutterman and D. G. Nocera, J. Am. Chem. Soc., 2011, 133, 5174–5177 CrossRef CAS PubMed.
- J. Amtawong, B. B. Skjelstad, D. Balcells and T. D. Tilley, Inorg. Chem., 2020, 59, 15553–15560 CrossRef CAS PubMed.
- J. Amtawong, A. I. Nguyen and T. D. Tilley, J. Am. Chem. Soc., 2022, 144, 1475–1492 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |