Open Access Article
Open Access ArticleCopper-catalyzed enantioselective interrupted azide–alkyne cycloaddition to access axially chiral diaryl ethers
Ruixue Wu,
Lan Chen,
Yihan Chen,
Qimin Wu,
Qirui Chen,
Aijun Lin ,
Hequan Yao
,
Hequan Yao *,
Qiuyu Li* and
Shang Gao
*,
Qiuyu Li* and
Shang Gao *
*
State Key Laboratory of Natural Medicines (SKLNM), Department of Medicinal Chemistry, School of Pharmacy, China Pharmaceutical University, Nanjing, 210009, P. R. China. E-mail: gaoshang1990@cpu.edu.cn; liqiuyu055@163.com; hyao@cpu.edu.cn
First published on 24th April 2026
Abstract
While transition metal-catalyzed enantioselective azide–alkyne cycloadditions are well-established for accessing 1,4-disubstituted triazoles, the direct enantioselective synthesis of chiral 1,4,5-trisubstituted triazoles from terminal alkynes via an interrupted process remains largely underdeveloped. Herein, we report a copper-catalyzed enantioselective interrupted azide–alkyne cycloaddition. This method enables the facile construction of enantioenriched axially chiral diaryl ethers bearing a 1,4,5-trisubstituted triazole moiety directly from terminal alkynes. Mechanistic studies reveal the absence of a further kinetic resolution process involving the product, underscoring the crucial role of our newly developed chiral ligand L5 in achieving excellent enantiocontrol and yield. The combination of Et3N and LiOtBu is essential for the inhibition of protonated byproducts. The orthogonal reactivity of the residual terminal alkyne and amine groups within the products provides versatile platforms for downstream chemistry, enriching the structural diversity of axially chiral diaryl ethers.
Introduction
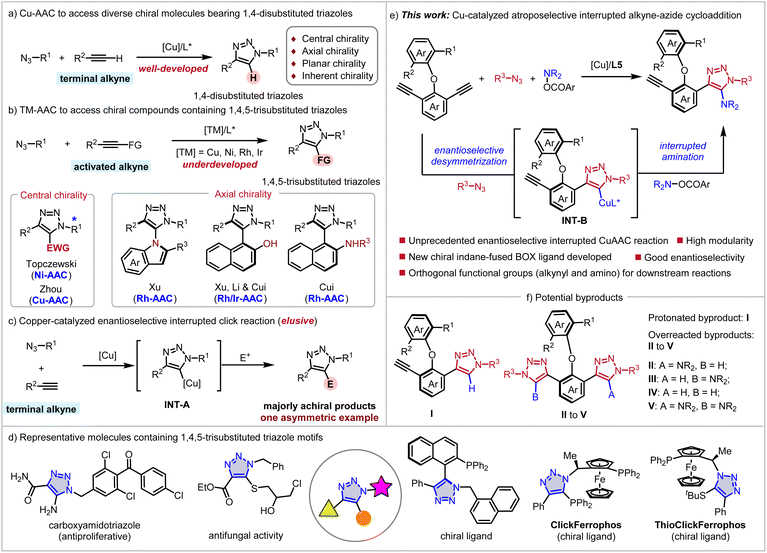
Over the past few decades, transition metal-catalyzed cycloaddition reactions between alkynes and azides have advanced significantly for the synthesis of functionalized triazoles, following the pioneering independent reports by the Meldal and Sharpless groups.1 Among these, copper-catalyzed azide–alkyne cycloadditions (Cu-AAC) have been extensively employed to construct diverse chiral molecules bearing 1,4-disubstituted triazole moieties, including those featuring central, axial, planar, and inherent chirality (Scheme 1a).2 Nevertheless, mechanistic constraints inherently limit this reaction to produce only the 1,4-disubstituted triazoles. Recently, significant progress has been made by the Zhou, Topczewski, Xu, Li, and Cui groups3 towards the asymmetric synthesis of 1,4,5-trisubstituted triazoles containing central and axial chirality (Scheme 1b), utilizing transition-metal catalyzed azide–alkyne cycloadditions (TM-AAC). However, these approaches typically require the use of electronically activated internal alkynes, hampering their application to some extent. In contrast, a copper-catalyzed interrupted click reaction offers a versatile strategy for constructing 1,4,5-trisubstituted triazoles directly from terminal alkynes (Scheme 1c).4 In this process, interception of the cuprate–triazole intermediate (INT-A) by an electrophile enables the introduction of a substituent at the C5-position. Despite this versatility, current interrupted click methodologies mainly yield achiral products. Very recently, Gu and coworkers developed the first asymmetric interrupted CuAAC reaction with terminal alkynes, cyclic diaryliodonium reagents and azides, providing an efficient method to construct atropisomeric biaryl triazoles.4k Given the central role of tri-substituted triazoles as structural scaffolds in bioactive molecules, catalysts, and ligands (Scheme 1d),5 further development of the enantioselective interrupted click reaction constitutes a critical research priority.Diaryl ether skeletons constitute important structural motifs prevalent in diverse natural products, ligands, and bioactive molecules.6 Compared to the well-established synthesis of C–C axially chiral compounds, the development of methods for constructing C–O axial chirality remains challenging.7 This lag stems from inherently lower rotational barriers and the unique dual-axial chirality phenomenon. Recent years have witnessed rapid progress in the atroposelective synthesis of axially chiral diaryl ethers,8 primarily achieved through organocatalyzed9 and transition-metal-catalyzed10 enantioselective desymmetric functionalization of prochiral substrates. Leveraging sequential enantioselective desymmetrization and kinetic resolution processes has enabled the construction of target axially chiral diaryl ethers with excellent enantioselectivities. However, the inevitable formation of double-functionalization byproducts from the prochiral substrates often compromises reaction yield.11 Inspired by these elegant studies, we envisioned that the INT-B might be captured by an electrophile, for example a hydroxylamine ester (Scheme 1e).12 However, to realize this reaction, several challenges must be addressed: (1) how to suppress the formation of byproduct I via protonation of intermediate INT-B (Scheme 1f); (2) the alkyne motif of the product could engage in further reactions with azides and hydroxylamine esters to generate overreacted byproducts II–V. Based on our continuous interest in the transition-metal catalyzed enantioselective desymmetrization and synthesis of axial chirality,13 we report herein an unprecedented copper-catalyzed enantioselective interrupted click reaction. Both protonated and overreacted byproducts I–V were inhibited efficiently via the development of an indane-fused BOX ligand. The axially chiral diaryl ethers containing 1,4,5-trisubstituted triazole motifs were constructed in good yields with excellent enantiopurities. The orthogonal chemical activities of alkynyl and amino motifs provided platforms for further elaboration of products, enabling the enrichment of the structural diversity of products.
Results and discussion
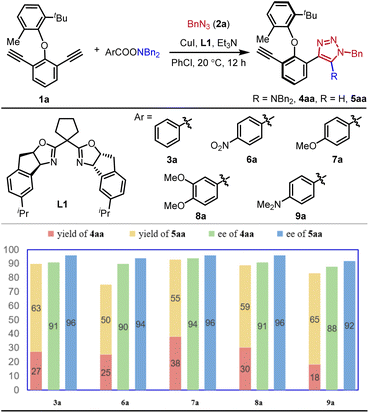
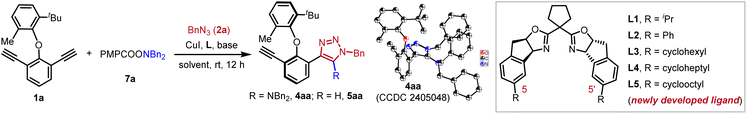
We commenced our studies using alkyne 1a, azide 2a and hydroxylamine ester 3a as the substrates, CuI as the catalyst and Et3N as the additive. Initial screening of the chiral ligand indicated that the indane-fused BOX ligand L1 showed the best performance in the control of enantioselectivity (see the SI for details). The target product 4aa was obtained in 27% yield with 91% ee, accompanied by the formation of protonation byproduct 5aa in 63% yield with 96% ee (Scheme 2). To explore the potential influence of the hydroxylamine ester's acyl group on the yield and enantioselectivity of 4aa, various esters (6a–9a) were evaluated. While 6a, bearing a para-electron-withdrawing group, afforded 4aa in 25% yield with 90% ee, 7a (containing a para-electron-donating group) increased the yield to 38% with a marginal improvement in enantioselectivity. However, further increasing the electron density of the acyl group (8a and 9a) did not enhance the results. The consistently low yield of 4aa is primarily attributed to the predominant formation of the protonated byproduct 5aa. The higher enantiopurity observed for 5aa compared to 4aa suggested the possible involvement of an inefficient kinetic resolution process in the formation of 4aa, and subsequent mechanistic studies confirmed that no kinetic resolution occurred.To suppress the formation of the protonated byproduct 5aa, other reaction parameters were further evaluated. Solvents including THF, toluene, and anisole predominantly yielded 5aa (Table 1, entries 1–3). In contrast, acetone delivered 4aa and 5aa with comparable yields (entry 4). Increasing the loading of Et3N to 10 equivalents marginally improved the yield of 4aa (entry 5). Further base screening revealed that DIPEA or LiOtBu suppressed 5aa formation but significantly reduced either the yield or enantioselectivity of 4aa (entries 6 and 7). Crucially, employing a combination of Et3N (10 equiv) and LiOtBu (0.5 equiv) afforded 4aa in 87% yield with 88% ee, while only trace amounts of 5aa were generated (entry 8). Goldup and co-workers reported that the protonation rate of the Cu(I) triazolide intermediate INT-A shown in Scheme 1c was highly dependent on the acidity of the proton source.14 On this basis, we reasoned that combining tBuOLi with Et3N could prevent the formation of acidic proton shuttles such as [Et3NH]+ species, thereby suppressing the protonation of INT-A and minimizing the formation of byproduct 5aa. Encouraged by this result, additional chiral indane-fused BOX ligands bearing various substituents at the C5- and C5′-positions were evaluated (entries 9–12). Generally, enantioselectivity increased with greater steric bulk of the chiral ligand. Among these, L5 bearing cyclooctyl groups delivered optimal performance, providing 4aa in 83% isolated yield with 93% ee, and no protonated byproduct 5aa was detected. The absolute configuration of 4aa was unambiguously determined by X-ray crystal analysis.
| Entry | Ligand | Solvent | Base | Yield (%) (4aa/5aa) | ee (%) (4aa) |
|---|---|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol and 1.0 equiv), 2a (0.3 mmol and 1.5 equiv), 7a (0.4 mmol and 2.0 equiv) CuI (10 mol%), L (15 mol%), 4 Å MS (100.0 mg) and base (0.4 mmol and 2.0 equiv) in solvent (1.0 mL) at 20 °C under an Ar atmosphere for 12 h.b The yields were determined by 1H NMR with 1,3,5-trimethoxybenzene as the internal standard.c ee values were determined by chiral HPLC analysis on a chiral-stationary-phase.d Et3N (2.0 mmol and 10.0 equiv) was used.e LiOtBu (0.1 mmol and 0.5 equiv) was used.f Isolated yield was listed. | |||||
| 1 | L1 | THF | Et3N | 15/10 | 86 |
| 2 | L1 | PhMe | Et3N | 22/39 | 91 |
| 3 | L1 | PhOMe | Et3N | 14/83 | 87 |
| 4 | L1 | Acetone | Et3N | 42/38 | 87 |
| 5d | L1 | Acetone | Et3N | 46/50 | 86 |
| 6 | L1 | Acetone | DIPEA | 8/— | 86 |
| 7 | L1 | Acetone | LiOtBu | 47/— | 23 |
| 8d,e | L1 | Acetone | Et3N/LiOtBu | 87/— | 88 |
| 9d,e | L2 | Acetone | Et3N/LiOtBu | 48/— | 87 |
| 10d,e | L3 | Acetone | Et3N/LiOtBu | 87/— | 92 |
| 11d,e | L4 | Acetone | Et3N/LiOtBu | 80/— | 93 |
| 12d,e | L5 | Acetone | Et3N/LiOtBu | 91(83)f/— | 93 |
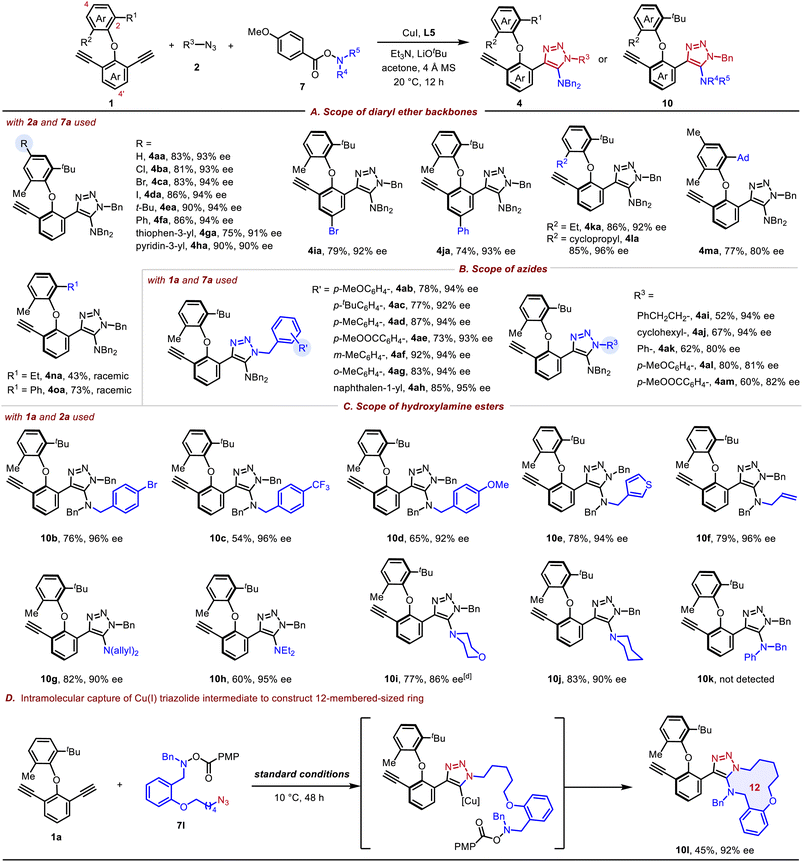
With the optimized conditions in hand, we next explored the substrate scope of prochiral diaryl ether backbones and azides, and the results are summarized in Tables 2A and B. Diaryl ether backbones containing a substituent at the C4- or C4′-position such as halogen, alkyl and aryl groups proceeded smoothly, giving 4ba–4ja in 74–90% yields with 91–94% ee. Switching the methyl group of 1a to ethyl and cyclopropyl groups has no effect on the reaction efficiency, delivering 4ka in 86% yield with 92% ee and 4la in 85% yield with 96% ee. When the tert-butyl group was switched to a 1-adamantanyl group, 4ma was isolated in 77% yield with moderate enantioselectivity. Upon introducing an ethyl or phenyl group at the C2-position, ‘Batman’-type chromatograms15 were observed for products 4na and 4oa during HPLC analysis (see the SI for details), indicating interconversion of enantiomers on the chromatographic timescale. Furthermore, benzyl azides containing an electron-donating group or electron-withdrawing group at the para-, meta- and ortho-positions of the phenyl ring as well as 1-(azidomethyl) naphthalene were suitable substrates, furnishing 4ab–4ah in 73–92% yields with 92–95% ee. In addition, alkyl azides were well compatible, affording 4ai in 52% yield and 4aj in 67% yield with 94% ee. With aryl azides used, the enantioselecitvities decreased slightly, and products 4ak–4am were obtained in 60–80% yields with 80–82% ee. The substrate scope of hydroxylamine esters was further investigated (Table 2C). Hydroxylamine esters containing various substituted benzyl, chain alkyl, allyl as well as cyclic alkyl groups were well compatible with our catalytic system, furnishing the products 10b–10j in 54–82% yields with 86–96% ee. However, aromatic hydroxylamine esters are incompatible with the current reaction system, and no product (10k) was detected. By tethering the azide and hydroxylamine ester motifs within a single molecule, this interrupted click reaction could still proceed smoothly, affording the corresponding 12-membered ring product 10l in 45% yield with 92% ee (Table 2D). However, further attempts to synthesize the six-membered ring failed under this catalytic system.
| a Reaction conditions: 1 (0.2 mmol and 1.0 equiv), 2 (0.3 mmol and 1.5 equiv), 7 (0.4 mmol and 2.0 equiv), Et3N (2.0 mmol and 10.0 equiv), LiOtBu (0.1 mmol and 0.5 equiv), CuI (10 mol%), L5 (15 mol%) and 4 Å MS (100.0 mg) in acetone at 20 °C under an Ar atmosphere for 10 h.b Isolated yields were listed.c ee values were determined by chiral HPLC analysis on a chiral-stationary-phase.d L1 (15 mol%) was used. |
|---|
 |
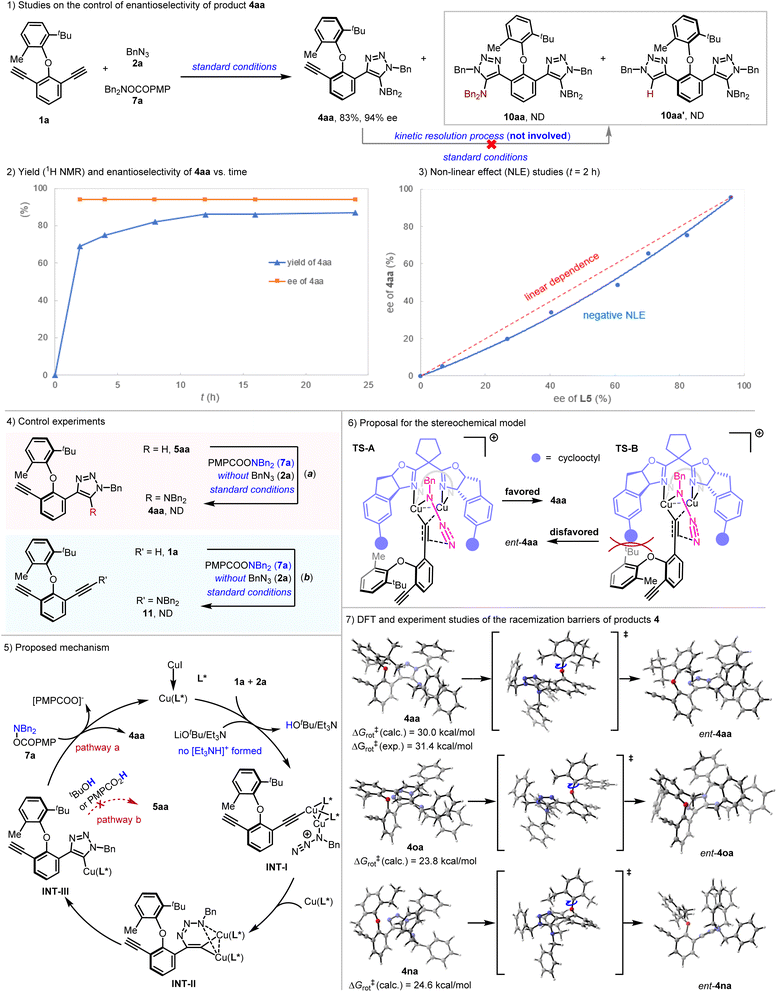
Control experiments were then conducted to elucidate the enantioselectivity control and mechanism of this copper-catalyzed enantioselective interrupted reaction. As shown in Scheme 3(1), no over-reacted byproducts 10aa and 10aa′ were observed under standard conditions. When product 4aa was subjected to the reaction system, neither 10aa nor 10aa′ was detected. Plotting of the yield and enantioselectivity of product 4aa against the reaction time revealed that the reaction rate decreased significantly after 2 hours. The enantioselectivity of compound 4aa was maintained at 94% and did not improve even after extending the reaction time to 24 hours (Scheme 3(2)). Collectively, these results indicate the absence of further kinetic resolution of 4aa. Thus, enantioselectivity is exclusively controlled by the desymmetrization process. Reactions carried out using different enantiopurities of ligand L5 for 2 hours show a negative non-linear effect (NLE),16 hinting that heterochiral dimeric species is more reactive than the corresponding homodimer (Scheme 3(3)). Moreover, no 4aa could be detected without addition of BnN3 (2a) under standard conditions, indicating that 5aa was not the intermediate of this interrupted reaction (Scheme 3(4-a)). In addition, dialkyne 1a did not participate in the reaction with 7a in the absence of 2a, and no ynamine 11 was generated (Scheme 3(4-b)). Therefore, the possibility of a ynamine as an intermediate for this reaction could be ruled out, and the amino motif of the product 4aa should be introduced via the Cu(I) triazolide intermediate (INT-III shown in Scheme 3(5)). Based on previous reports1,17 and our control experiments, a plausible mechanism was proposed and depicted in Scheme 3(5). In the presence of LiOtBu and Et3N, the terminal alkyne groups of 1a react with Cu(L*) to form acetylide intermediate INT-I. Intramolecular migratory insertion of INT-I first forms the six-membered copper metallacycle INT-II, which then undergoes ring contraction to afford the Cu(I) triazolide intermediate INT-III. At this stage, two competitive pathways may occur. In pathway a, trapping of Int-III with hydroxylamine ester 7a directly yields the aminated product 4aa. When a bulky chiral ligand such as L5 is employed, no over-reacted byproducts are formed. Alternatively, pathway b involving protonation could take place in the presence of a proton source. Given that tBuOH and PMPCO2H are the major proton sources present, the protonation of INT-III is expected to be sufficiently slow to shut down pathway b. In addition, the origin of the enantioselectivity observed in this interrupted azide–alkyne cycloaddition in the presence of L5 was rationalized by considering two competing transition-states, TS-A and TS-B. For TS-B, severe repulsion arises between the cyclooctyl group of chiral ligand L5 and the tert-butyl group of substrate 1a (Scheme 3(6)).
Subsequently, the configurational stability of 4 was studied via DFT calculations (Scheme 3(7)). As expected, the rotational barrier of the C(1)–O bond generally increased with greater steric hindrance from the ortho-substituent. Computational studies provided rotational barriers of 23.8 kcal mol−1 for 4oa, 24.6 kcal mol−1 for 4na, and a significantly higher barrier of 30.0 kcal mol−1 for 4aa. The relatively low barriers for 4oa and 4na rationalize their facile racemization. This interpretation is corroborated by thermal racemization studies of enantiopure 4aa, which exhibited a half-life of 32.1 hours at 100 °C in toluene. The experimentally determined rotational barrier of 4aa via a thermal experiment was 31.4 kcal mol−1, which was in agreement with the computed value of 30.0 kcal mol−1.
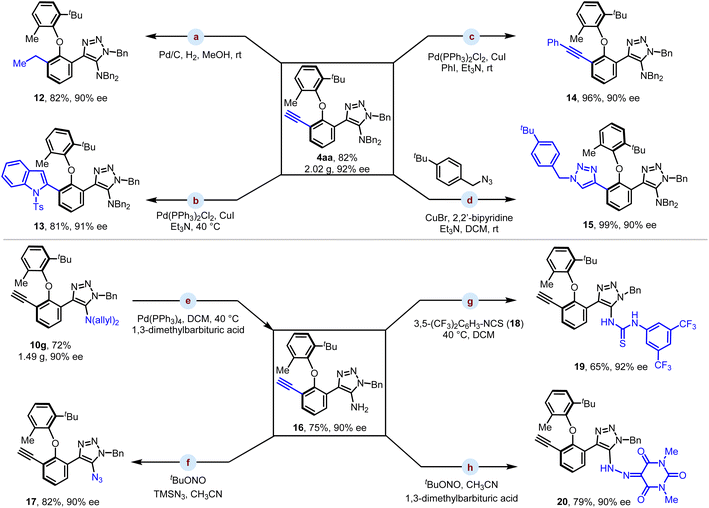
The practical utility of our reaction was further explored via scale-up reactions and derivatization studies of the alkyne and amino motifs of the products (Scheme 4). When the reactions were run on a 4 mmol-scale, the catalyst loading could be further reduced to 2.5 mol% without any loss of efficiency. The products 4aa and 10g were isolated in 82% yield (2.02 g) and 72% yield (1.49 g), respectively, with comparable enantioselectivity. The alkyne group was chemoselectively reduced to an ethyl group with Pd/C under hydrogen balloon conditions without affecting the benzyl group, yielding product 12 in 82% yield and 90% ee (Scheme 4a). In addition, transition-metal catalyzed cross-coupling such as Larock indole synthesis, Sonogashira coupling and alkyne–azide cycloaddition proceeded smoothly, delivering compounds 13–15 in 81–99% yields with 90–91% ee (Scheme 4b–d). Palladium-catalyzed deallylation of 10g proceeded smoothly, giving free amine 16 in 75% yield with 90% ee (Scheme 4e). The amino group could further undergo azidation in the presence of tBuONO and TMSN3, delivering product 17 in 82% yield with 90% ee (Scheme 4f). Treatment of 16 with isothiocyanate 18 afforded thiourea 19, which was isolated in 65% yield with 92% ee (Scheme 4g). In addition, the primary amino motif of 16 could react with barbituric acid and tBuONO in an acetonitrile solvent to generate hydrazone 20 in 79% yield with 90% ee (Scheme 4h).
Conclusions
In summary, we have developed a copper-catalyzed enantioselective desymmetric interrupted alkyne–azide cycloaddition with dialkynes, azides and hydroxylamine esters. This method provides direct access to a series of axially chiral diaryl ethers bearing 1,4,5-trisubstituted triazole motifs in good yields with high modularity. Employing a newly designed indane-fused BOX ligand L5, C–O axially chiral scaffolds are constructed with excellent enantioselectivity. Notably, the over-reacted reactions are effectively blocked using a LiOtBu and Et3N dual-base system. Control experiments demonstrate the absence of kinetic resolution processes in enhancing product enantiopurity. The orthogonal reactivity of the residual alkyne and amino groups in products offers a versatile synthetic handle for downstream diversification.Author contributions
R. W., L. C., Y. C., Q. W., and Q. C. performed all chemical reactions reported, Q. L. contributed to the DFT calculations and A. L., H. Y., Q. L. and S. G. wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI), including detailed experimental procedures and characterization data for new compounds and computational methods. Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc09566b.CCDC 2405048 (4aa) contains the supplementary crystallographic data for this paper.18
Acknowledgements
The authors acknowledge generous financial support from the National Natural Science Foundation of China (22371299 and 22501296), the China Postdoctoral Science Foundation (2024M763657), the Postdoctoral Fellowship Program of CPSF (GZB20250246), the Jiangsu Funding Program for Excellent Postdoctoral Talent (2025ZB316), the Project Program of State Key Laboratory of Natural Medicines (SKLNMZZ202211), the Natural Science Foundation of Jiangsu Province (BK20251564), the Fundamental Research Funds for the Central Universities (2632024ZD09 and 2632025PY03) and Innovation and Entrepreneurship (Shuangchuang) Program of Jiangsu Province (2024). We also thank the Hefei Advanced Computing Center for the computational resources.Notes and references
- For selected reviews and examples, see: (a) P. Thirumurugan, D. Matosiuk and K. Jozwiak, Chem. Rev., 2013, 113, 4905–4979 CrossRef CAS PubMed; (b) J. E. Hein and V. V. Fokin, Chem. Soc. Rev., 2010, 39, 1302–1315 RSC; (c) V. V. Rostovtsev, L. G. Green, V. V. Fokin and K. B. Sharpless, Angew. Chem., Int. Ed., 2002, 41, 2596–2599 CrossRef CAS; (d) C. W. Tornøe, C. Christensen and M. Meldal, J. Org. Chem., 2002, 67, 3057–3064 CrossRef PubMed; (e) H. C. Kolb, M. G. Finn and K. B. Sharpless, Angew. Chem., Int. Ed., 2001, 40, 2004–2021 CrossRef CAS; (f) B. T. Worrell, J. A. Malik and V. V. Fokin, Science, 2013, 340, 457–460 CrossRef CAS PubMed.
- (a) W. D. G. Brittain, B. R. Buckley and J. S. Fossey, ACS Catal., 2016, 6, 3629–3636 CrossRef CAS; (b) C. Wang, F. Zhou and J. Zhou, Chin. J. Org. Chem., 2020, 40, 3065–3073 CrossRef CAS; (c) C.-Q. Qin, C. Zhao, G.-S. Chen and Y.-L. Liu, ACS Catal., 2023, 13, 6301–6309 CrossRef CAS; (d) F. Zhou, C. Tan, J. Tang, Y.-Y. Zhang, W.-M. Gao, H.-H. Wu, Y.-H. Yu and J. Zhou, J. Am. Chem. Soc., 2013, 135, 10994–10997 CrossRef CAS PubMed; (e) T. Song, L. Li, W. Zhou, Z.-J. Zheng, Y. Deng, Z. Xu and L.-W. Xu, Chem.–Eur. J., 2015, 21, 554–558 CrossRef CAS; (f) E.-C. Liu and J. J. Topczewski, J. Am. Chem. Soc., 2019, 141, 5135–5138 CrossRef CAS PubMed; (g) R.-Y. Zhu, L. Chen, X.-S. Hu, F. Zhou and J. Zhou, Chem. Sci., 2020, 11, 97–106 RSC; (h) B. Zu, Y. Guo and C. He, J. Am. Chem. Soc., 2021, 143, 16302–16310 CrossRef CAS PubMed; (i) K. Liao, Y. Gong, R.-Y. Zhu, C. Wang, F. Zhou and J. Zhou, Angew. Chem., Int. Ed., 2021, 60, 8488–8493 CrossRef CAS; (j) Y. Gong, C. Wang, F. Zhou, K. Liao, X.-Y. Wang, Y. Sun, Y.-X. Zhang, Z. Tu, X. Wang and J. Zhou, Angew. Chem., Int. Ed., 2023, 62, e202301470 CrossRef CAS PubMed; (k) M.-Y. Chen, Z. Xu, L. Chen, T. Song, Z.-J. Zheng, J. Cao, Y.-M. Cui and L.-W. Xu, ChemCatChem, 2018, 10, 280–284 CrossRef CAS; (l) X. Han, L. Chen, Y. Yan, Y. Zhao, A. Lin, S. Gao and H. Yao, ACS Catal., 2024, 14, 3475–3483 CrossRef CAS; (m) L. Dai, X. Zhou, J. Guo, Q. Huang and Y. Lu, Chem. Sci., 2024, 15, 5993–5999 RSC; (n) T. Osako and Y. Uozumi, Org. Lett., 2014, 16, 5866–5869 CrossRef CAS PubMed; (o) A. J. Wright, D. L. Hughes, P. C. B. Page and G. R. Stephenson, Eur. J. Org Chem., 2019, 2019, 7218–7227 CrossRef CAS; (p) W.-G. Zhou, L.-L. Xi, M.-R. Zhang, H.-R. Wang, M. An, J.-H. Li and R.-R. Liu, Angew. Chem., Int. Ed., 2025, 64, e202502381 CrossRef CAS PubMed.
- (a) C. Wang, R.-Y. Zhu, K. Liao, F. Zhou and J. Zhou, Org. Lett., 2020, 22, 1270–1274 CrossRef CAS; (b) E.-C. Liu and J. J. Topczewski, J. Am. Chem. Soc., 2021, 143, 5308–5313 CrossRef CAS PubMed; (c) L. Zhou, Y. Li, S. Li, Z. Shi, X. Zhang, C.-H. Tung and Z. Xu, Chem. Sci., 2023, 14, 5182–5187 RSC; (d) X. Zhang, S. Li, W. Yu, Y. Xie, C.-H. Tung and Z. Xu, J. Am. Chem. Soc., 2022, 144, 6200–6207 CrossRef CAS; (e) W.-T. Guo, B.-H. Zhu, Y. Chen, J. Yang, P.-C. Qian, C. Deng, L.-W. Ye and L. Li, J. Am. Chem. Soc., 2022, 144, 6981–6991 CrossRef CAS PubMed; (f) L. Zeng, J. Li, Y. Huang, G. C. Haug, R. S. Paton, F. Zhang and S. Cui, ACS Catal., 2025, 15, 15844–15852 CrossRef CAS; (g) L. Zeng, J. Li and S. Cui, Angew. Chem., Int. Ed., 2022, 61, e202205037 CrossRef CAS; (h) L. Zeng, F. Zhang and S. Cui, Org. Lett., 2023, 25, 443–448 CrossRef CAS PubMed.
- For selected achiral examples, see: (a) W. Hou, X. Zhou, Z. Yang, H. Xia, Y. Wang, K. Xu, S. Hou, S. Zhang, D. Cui, P. Ma, W. Zhou and H. Xu, Angew. Chem., Int. Ed., 2025, 64, e202500942 CrossRef CAS PubMed; (b) W. Wang, X. Peng, F. Wei, C.-H. Tung and Z. Xu, Angew. Chem., Int. Ed., 2016, 55, 649–653 CrossRef CAS; (c) F. Brunelli, C. Russo, M. Giustiniano and G. C. Tron, Chem.–Eur. J., 2024, 30, e202303844 CrossRef CAS PubMed; (d) P. Sun, F. Wei, C.-H. Tung and Z. Xu, Chin. Chem. Lett., 2024, 35, 108478 CrossRef CAS; (e) P. Sun, W. Wang, J. Lai, H. Yan, C.-H. Tung and Z. Xu, Org. Chem. Front., 2023, 10, 1890–1895 RSC; (f) Y. Navarro, J. G. López, M. J. Iglesias and F. L. Ortiz, Org. Lett., 2021, 23, 334–339 CrossRef CAS PubMed; (g) E. M. Larin and M. Lautens, Angew. Chem., Int. Ed., 2019, 58, 13438–13442 CrossRef CAS PubMed; (h) K. P. S. Cheung and G. C. Tsui, Org. Lett., 2017, 19, 2881–2884 CrossRef CAS PubMed; (i) F. Wei, W. Wang, Y. Ma, C.-H. Tung and Z. Xu, Chem. Commun., 2016, 52, 14188–14191 RSC; (j) Q. Cai, J. Yan and K. Ding, Org. Lett., 2012, 14, 3332–3335 CrossRef CAS PubMed For an enantioselective example, see: ; (k) Y. Li, S. Yang, L. Duan and Z. Gu, Nat. Commun., 2026, 17, 944 CrossRef CAS.
- For selected examples, see: (a) M. J. Soltis, H. J. Yeh, K. A. Cole, N. Whittaker, R. P. Wersto and E. C. Kohn, Drug Metab. Dispos., 1996, 24, 799–806 CrossRef CAS; (b) G. Biagi, V. Calderone, I. Giorgi, O. Livi, V. Scartoni, B. Baragatti and E. Martinotti, Eur. J. Med. Chem., 2000, 35, 715–727 CrossRef CAS PubMed; (c) I. Oura, K. Shimizu, K. Ogata and S. Fukuzawa, Org. Lett., 2010, 12, 1752–1755 CrossRef CAS; (d) Y. Morzherin, P. E. Prokhorova, D. A. Musikhin, T. V. Glukhareva and Z. Fan, Pure Appl. Chem., 2011, 83, 715–731 CAS; (e) S. Fukuzawa and H. Oki, Org. Lett., 2008, 10, 1747–1749 CrossRef CAS PubMed.
- For selected examples, see: (a) T. Chen, H. Xiong, J.-F. Yang, X.-L. Zhu, R.-Y. Qu and G.-F. Yang, J. Agric. Food Chem., 2020, 68, 9839–9847 CrossRef CAS PubMed; (b) G. M. Adams and A. S. Weller, Coord. Chem. Rev., 2018, 355, 150–172 CrossRef CAS; (c) K. C. Nicolaou and C. N. C. Boddy, J. Am. Chem. Soc., 2002, 124, 10451–10455 CrossRef CAS PubMed.
- (a) K. Fuji, T. Oka, T. Kawabata and T. Kinoshita, Tetrahedron Lett., 1998, 39, 1373–1376 CrossRef CAS; (b) M. S. Betson, J. Clayden, C. P. Worrall and S. Peace, Angew. Chem., Int. Ed., 2006, 45, 5803–5807 CrossRef CAS; (c) R. Farooqi, A. Mustafai, A. G. Woldegiorgis, X. Lin and P. Wang, ACS Catal., 2025, 15, 7891–7901 CrossRef; (d) A. Naghim, J. Rodriguez, O. Chuzel, G. Chouraqui and D. Bonne, Angew. Chem., Int. Ed., 2024, 63, e202407767 CrossRef CAS PubMed; (e) G.-J. Mei, W. L. Koay, C.-Y. Guan and Y. Lu, Chem, 2022, 8, 1855–1893 CrossRef CAS.
- (a) J. Clayden, C. P. Worrall, W. J. Moran and M. Helliwell, Angew. Chem., Int. Ed., 2008, 47, 3234–3237 CrossRef CAS PubMed; (b) A. Page and J. Clayden, Beilstein J. Org. Chem., 2011, 7, 1327–1331 CrossRef CAS PubMed; (c) A. N. Dinh, R. R. Noorbehesht, S. T. Toenjes, A. C. Jackson, M. A. Saputra, S. M. Maddox and J. L. Gustafson, Synlett, 2018, 29, 2155–2160 CrossRef CAS.
- (a) B. Yuan, A. Page, C. P. Worrall, F. Escalettes, S. C. Willies, J. J. W. McDouall, N. J. Turner and J. Clayden, Angew. Chem., Int. Ed., 2010, 49, 7010–7013 CrossRef CAS; (b) L. Dai, Y. Liu, Q. Xu, M. Wang, Q. Zhu, P. Yu, G. Zhong and X. Zeng, Angew. Chem., Int. Ed., 2023, 62, e202216534 CrossRef CAS PubMed; (c) B.-A. Zhou, X.-N. Li, C.-L. Zhang, Z.-X. Wang and S. Ye, Angew. Chem., Int. Ed., 2024, 63, e202314228 CrossRef CAS; (d) S. Shee, S. S. Ranganathappa, M. S. Gadhave, R. Gogoi and A. T. Biju, Angew. Chem., Int. Ed., 2013, 62, e202311709 CrossRef PubMed; (e) J. Xu, W. Lin, H. Zheng and X. Li, ACS Catal., 2024, 14, 6667–6675 CrossRef CAS; (f) Z. Ye, W. Xie, W. Liu, C. Zhou and X. Yang, Adv. Sci., 2024, 11, 2403125 CrossRef CAS; (g) Y. Wu, X. Guan, H. Zhao, M. Li, T. Liang, J. Sun, G. Zheng and Q. Zhang, Chem. Sci., 2024, 15, 4564–4571 RSC; (h) S. Shee, D. Ramachandran, R. Gogoi and A. T. Biju, ACS Catal., 2025, 15, 13157–13166 CrossRef CAS; (i) J. Che, S. Fang, Z. Liu, X. Luo, Z. Zheng, Y. Guo, H.-L. Hu, W. Chen and T. Wang, ACS Catal., 2025, 15, 19005–19016 CrossRef CAS.
- (a) Y. Wang, R. Mi, S. Yu and X. Li, ACS Catal., 2024, 14, 4638–4647 CrossRef CAS; (b) Z. Han, L. Wei, C. Nian, X. Hu, S. Liu, H. Huang and J. Sun, Sci. China: Chem., 2025, 68, 3996–4011 CrossRef.
- (a) X. Peng, A. Rahim, W. Peng, F. Jiang, Z. Gu and S. Wen, Chem. Rev., 2023, 123, 1364–1416 CrossRef CAS; (b) P. Xu, F. Zhou, L. Zhu and J. Zhou, Nat. Synth., 2023, 2, 1020–1028 CrossRef CAS; (c) X.-P. Zeng, Z.-Y. Cao, Y.-H. Wang, F. Zhou and J. Zhou, Chem. Rev., 2016, 116, 7330–7396 CrossRef CAS PubMed.
- For selected examples, see: (a) Y.-S. Zhu, Y.-L. Guo, Y.-Y. Zhu and B. Su, J. Am. Chem. Soc., 2024, 146, 32283–32292 CrossRef CAS PubMed; (b) S. Wang, L. Shi, X.-Y. Chen and W. Shu, Angew. Chem., Int. Ed., 2023, 62, e202303795 CrossRef CAS PubMed; (c) L. Yu and P. Somfai, Angew. Chem., Int. Ed., 2019, 58, 8551–8555 CrossRef CAS PubMed; (d) Y. Yang, S.-L. Shi, D. Niu, P. Liu and S. L. Buchwald, Science, 2015, 349, 62–66 CrossRef CAS PubMed; (e) S. Zhu, N. Niljianskul and S. L. Buchwald, J. Am. Chem. Soc., 2013, 135, 15746–15749 CrossRef CAS; (f) Y. Miki, K. Hirano, T. Satoh and M. Miura, Angew. Chem., Int. Ed., 2013, 52, 10830–10834 CrossRef CAS.
- (a) Z. Yuan, Z. Feng, Y. Zeng, X. Zhao, A. Lin and H. Yao, Angew. Chem., Int. Ed., 2019, 58, 2884–2888 CrossRef CAS; (b) Z. Yuan, Y. Zeng, Z. Feng, Z. Guan, A. Lin and H. Yao, Nat. Commun., 2020, 11, 2544 CrossRef CAS PubMed; (c) D. Zhang, M. Li, J. Li, A. Lin and H. Yao, Nat. Commun., 2021, 12, 6627 CrossRef CAS; (d) Q. Li, J. Li, J. Zhang, S. Wu, Y. Zhang, A. Lin and H. Yao, Angew. Chem., Int. Ed., 2023, 62, e202313404 CrossRef CAS; (e) Q. Wu, Q. Zhang, S. Yin, A. Lin, S. Gao and H. Yao, Angew. Chem., Int. Ed., 2023, 62, e202305518 CrossRef CAS; (f) X. Li, L. Kong, S. Yin, H. Zhou, A. Lin, H. Yao and S. Gao, Adv. Sci., 2024, 11, 2309706 CrossRef CAS PubMed; (g) B. Gong, Q. Lu, R. Sun, Z. Li, Y. Chen, A. Lin, H. Yao and S. Gao, ACS Catal., 2025, 15, 2351–2359 CrossRef CAS; (h) J. Zhong, M. Li, C. Wu, H. Li, H. Dong, C. Che, A. Lin and H. Yao, Nat. Commun., 2025, 16, 11373 CrossRef CAS PubMed; (i) J. Zhong, M. Li, J. Ma, Z. Zhang, H. Fang, S. Gao, A. Lin and H. Yao, Angew. Chem., Int. Ed., 2026, 65, e21476 CrossRef CAS.
- J. Winn, A. Pinczewska and S. M. Goldup, J. Am. Chem. Soc., 2013, 135, 13318–13321 CrossRef CAS PubMed.
- J.-P. Heeb, J. Clayden, M. D. Smith and R. J. Armstrong, Nat. Protoc., 2023, 18, 2745–2767 CrossRef CAS PubMed.
- For selected reviews, see: (a) D. Guillaneux, S.-H. Zhao, O. Samuel, D. Rainford and H. B. Kagan, J. Am. Chem. Soc., 1994, 116, 9430–9439 CrossRef CAS; (b) T. Satyanarayana, S. Abraham and H. B. Kagan, Angew. Chem., Int. Ed., 2009, 48, 456–494 CrossRef CAS PubMed; (c) D. G. Blackmond, Acc. Chem. Res., 2000, 33, 402–411 CrossRef CAS; (d) X. Zhu, Y. Li and H. Bao, Chin. J. Chem., 2023, 41, 3097–3102 CrossRef CAS.
- For selected examples, see: (a) L. Jin, D. R. Tolentino, M. Melaimi and G. Bertrand, Sci. Adv., 2015, 1, e1500304 CrossRef; (b) M. S. Ziegler, K. V. Lakshmi and T. D. Tilley, J. Am. Chem. Soc., 2017, 139, 5378–5386 CrossRef CAS PubMed.
- CCDC 2405048: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2lqn66.
| This journal is © The Royal Society of Chemistry 2026 |