Open Access Article
Open Access ArticlePd-N-C shelled Pd nanoparticle catalysts for high-performance hydrogen peroxide electrosynthesis
Jiao
Dong†
a,
Zixiang
Su†
bc,
Yanyan
Jia†
d,
Runjia
Xing†
a,
Sixuan
She
e,
Daqin
Guan
f,
Sheng
Dai
 d,
Jinling
Wang
a,
Manqing
Chai
a,
Zhenshan
Hou
d,
Jinling
Wang
a,
Manqing
Chai
a,
Zhenshan
Hou
 a,
Zhi-Qiang
Wang
a,
Zhi-Qiang
Wang
 *a,
Hehe
Wei
*a,
Hehe
Wei
 *a,
P.
Hu
*a,
P.
Hu
 ag and
Xue-Qing
Gong
ag and
Xue-Qing
Gong
 *b
*b
aState Key Laboratory of Green Chemical Engineering and Industrial Catalysis, Center for Computational Chemistry and Research Institute of Industrial Catalysis, School of Chemistry and Molecular Engineering, East China University of Science and Technology, 130 Meilong Road, Shanghai, 200237, China. E-mail: zhiqiangwang@ecust.edu.cn; weihh@ecust.edu.cn
bState Key Laboratory of Synergistic Chem-Bio Synthesis, School of Chemistry and Chemical Engineering, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai, 200240, China. E-mail: xqgong@sjtu.edu.cn
cNational Engineering & Technology Research Center of Scattered Metals, First Rare Materials Co., Limited, Baijia Industrial Park 27-9B, Qingyuan, Guangdong 511517, China
dKey Laboratory for Advanced Materials and Feringa Nobel Prize Scientist Joint Research Center, School of Chemistry and Molecular Engineering, East China University of Science and Technology, 130 Meilong Road, Shanghai, 200237, China
eDepartment of Applied Physics, The Hong Kong Polytechnic University, Hung Hom, Kowloon, Hong Kong SAR, China
fWA School of Mines: Minerals, Energy and Chemical Engineering (WASM-MECE), Curtin University, Perth, WA 6102, Australia
gSchool of Physical Science and Technology, Shanghai Tech University, 393 Middle Huaxia Road, Shanghai, 201210, China
First published on 8th April 2026
Abstract
Metal-nitrogen-carbon (M-N-C) catalysts have attracted widespread attention due to their potential in promoting the electrochemical oxygen reduction reaction (ORR) for the selective production of hydrogen peroxide (H2O2). However, the effects of their diverse structures and complex compositions on the catalytic performance remain poorly understood. Herein, systematic theoretical calculations reveal that the Pd-N-C based single-atom catalyst featuring a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of pyridinic and pyrrolic nitrogen adopts a centrosymmetric PdN4 structure (PdSAN2-2C), and the Pd dz2 orbital can strongly interact with the O 2p orbital of the adsorbed OOH intermediate, thereby strengthening its adsorption and facilitating subsequent conversion to H2O2. Guided by the theoretical insights, the PdSAN2-2C catalyst and a novel Pd@PdSAN2-2C core–shell catalyst with Pd nanoparticles encapsulated by an ultrathin PdSAN2-2C shell are synthesized, and the latter exhibits a remarkable H2O2 selectivity of 97% and a high yield of 35.88 mol gcat−1 h−1 at an industrially relevant current density of 200 mA cm−2, along with superior operational stability. This combined theoretical and experimental study provides useful guidance for the rational design of high-efficiency M-N-C catalysts for selective electrocatalysis.
1 ratio of pyridinic and pyrrolic nitrogen adopts a centrosymmetric PdN4 structure (PdSAN2-2C), and the Pd dz2 orbital can strongly interact with the O 2p orbital of the adsorbed OOH intermediate, thereby strengthening its adsorption and facilitating subsequent conversion to H2O2. Guided by the theoretical insights, the PdSAN2-2C catalyst and a novel Pd@PdSAN2-2C core–shell catalyst with Pd nanoparticles encapsulated by an ultrathin PdSAN2-2C shell are synthesized, and the latter exhibits a remarkable H2O2 selectivity of 97% and a high yield of 35.88 mol gcat−1 h−1 at an industrially relevant current density of 200 mA cm−2, along with superior operational stability. This combined theoretical and experimental study provides useful guidance for the rational design of high-efficiency M-N-C catalysts for selective electrocatalysis.
Introduction
Nitrogen-modified-carbon supported metal single-atom catalysts (M-N-C) have shown significant promise for the oxygen reduction reaction (ORR) owing to their structural tunability,1–3 excellent electrical conductivity and low cost.4–8 The varying types of nitrogen coordination (such as pyridinic, pyrrolic and graphitic nitrogen) as well as the electronic properties of the active metal sites in M-N-C catalysts are expected to be critically important for the two-electron (2e−) ORR toward hydrogen peroxide (H2O2) production.9–11 In particular, significant research efforts have been dedicated to elucidating how the pyridinic- and pyrrolic-nitrogen coordination environments affect the active sites and enhance the catalytic selectivity toward the 2e− ORR.12–16 Through combined theoretical and experimental studies, Chen et al. reported that pyrrole-type CoN4 single atom catalysts exhibit a remarkable H2O2 selectivity of 94% at 0.3 V versus the reversible hydrogen electrode (vs. RHE) in a flow cell,12 and it was mainly attributed to the increased adsorption strength of OOH, the key intermediate species for H2O2 formation, as a result of charge redistribution among the multiple d orbitals of Co after its adsorption. In contrast, Wang et al. found that pyridine-type CoN4 catalysts exhibit a much lower H2O2 selectivity of only 29% at 0.4 V vs. RHE. This decrease in selectivity was potentially attributed to the modulation of the metal center by pyridinic N, which leads to strong H2O2 adsorption and subsequent dissociation.13 Although these studies illustrated that pyrrole-type M-N-C single-atom catalysts generally exhibit higher 2e− ORR selectivity than pyridine-type catalysts, the synergistic effects of different nitrogen coordination environments on the active metal center remain elusive, and the related catalytic mechanisms are also controversial.11–19Beyond nitrogen coordination, the selection of the metal center is another crucial determinant of 2e− ORR selectivity and activity. Several studies have demonstrated that atomically dispersed metal centers coordinated with nitrogen-doped carbon matrices may exhibit distinct catalytic performances in H2O2 production. Notably, it has been suggested that single-atom Pd-N-C catalysts are highly efficient for the 2e− ORR to H2O2.18–21 For example, Jiang et al. synthesized a series of single atom catalysts including Pd-N-C, Co-N-C, and Mn-N-C, and they demonstrated that the corresponding H2O2 selectivity at 0.82 V vs. RHE was 90.3%, 74.8% and 39.8% for these catalysts, respectively.18 Wang et al. confirmed the outstanding performance of the single-atom Pd-N-C catalyst, which achieved approximately 95% H2O2 selectivity and an onset potential of ∼0.8 V vs. RHE.19 Generally, these findings underscored that both the local nitrogen coordination environment and the identity of the active metal center can tailor the 2e− ORR performance. Notably, under alkaline conditions, the 2e− ORR usually exhibits enhanced activity and selectivity towards H2O2 due to the pH effect,22,23 which can help optimize the intermediate adsorption strength24 as well as the thermodynamics of ORR processes.25 Therefore, unraveling the distinct nitrogen coordination motifs and electronic characteristics of catalytic centers at the atomic level is imperative for enabling efficient H2O2 electrosynthesis under industrially relevant alkaline conditions.
In this work, by using integrated density functional theory (DFT) calculations and experimental investigations, we elucidated the atomic-level interplay between nitrogen coordination symmetry and d-orbital engineering in Pd-N-C catalysts for highly efficient H2O2 production via the 2e− ORR. Our calculated results revealed a synergistic modulation mechanism where a balanced 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of pyridinic to pyrrolic N coordination induces a centrosymmetric PdN4 structure and promotes coupling between the Pd dz2 orbital and the 2p orbital of the terminal O atom (OT) of the OOH species binding with Pd through such OT. This configuration enhances OOH adsorption and reduces the 2e− ORR overpotential (η) to 0.54 V, which is significantly lower than that of previously reported Pd-N-C catalysts (η = 0.77 V).19 Guided by the theoretical insights, we employed a dynamic coordination and selective etching (DC–SE) approach for the syntheses of atomically dispersed Pd sites with targeted nitrogen coordination ratios and an interface-constrained self-assembly (ICSA) strategy for the construction of a novel core–shell type catalyst featuring metallic Pd core encapsulated by an ultrathin (∼1.0 nm) N-coordinated Pd shell with a 1
1 ratio of pyridinic to pyrrolic N coordination induces a centrosymmetric PdN4 structure and promotes coupling between the Pd dz2 orbital and the 2p orbital of the terminal O atom (OT) of the OOH species binding with Pd through such OT. This configuration enhances OOH adsorption and reduces the 2e− ORR overpotential (η) to 0.54 V, which is significantly lower than that of previously reported Pd-N-C catalysts (η = 0.77 V).19 Guided by the theoretical insights, we employed a dynamic coordination and selective etching (DC–SE) approach for the syntheses of atomically dispersed Pd sites with targeted nitrogen coordination ratios and an interface-constrained self-assembly (ICSA) strategy for the construction of a novel core–shell type catalyst featuring metallic Pd core encapsulated by an ultrathin (∼1.0 nm) N-coordinated Pd shell with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 coordination of pyridinic and pyrrolic nitrogen (Pd@PdSAN2-2C). The Pd@PdSAN2-2C catalyst delivered exceptional H2O2 electrosynthesis performance with a remarkable selectivity of 97%. Moreover, it delivered a high H2O2 yield of 35.88 mol gcat−1 h−1 at an industrially relevant current density of 200 mA cm−2 and produced 34.27 g L−1 H2O2 within 1080 min, demonstrating superior durability and thereby outperforming previously reported catalysts.2,19,26–31 This work also provides valuable insights into designing high-performance single-atom catalysts through modulating atomic-level coordination symmetry and active-site orbital levels.
1 coordination of pyridinic and pyrrolic nitrogen (Pd@PdSAN2-2C). The Pd@PdSAN2-2C catalyst delivered exceptional H2O2 electrosynthesis performance with a remarkable selectivity of 97%. Moreover, it delivered a high H2O2 yield of 35.88 mol gcat−1 h−1 at an industrially relevant current density of 200 mA cm−2 and produced 34.27 g L−1 H2O2 within 1080 min, demonstrating superior durability and thereby outperforming previously reported catalysts.2,19,26–31 This work also provides valuable insights into designing high-performance single-atom catalysts through modulating atomic-level coordination symmetry and active-site orbital levels.
Methods
Computational methods
All the structural optimizations and total energy calculations in this work were performed with spin-polarized DFT by using the Vienna Ab initio Simulation Package (VASP).32–35 The Perdew–Burke–Ernzerhof (PBE) functional within the generalized gradient approximation (GGA)36,37 and the projector augmented wave (PAW) method38,39 were applied throughout the geometric, electronic and energetic calculations. A cut-off energy of 400 eV was used, and the convergence criteria of energy and force in the calculations were set as 10−4 eV Å−1 and 0.05 eV Å−1, respectively.For the construction of the PdSANC model catalyst, we built a p(7 × 7) single-layer graphene structure, where two adjacent C atoms were replaced with one Pd atom and four C atoms around the Pd atoms were further substituted with four N atoms, and one O was also included on the surface to form a C–O–C group. Both pyridinic N and pyrrolic N were considered. To model the core–shell structure of Pd@PdSAN2-2C, we built the surface slab of Pd(111) covered by a PdSAN2-2C layer with minimal lattice mismatch. Specifically, the complex slab contains p(9 × 9) PdSAN2-2C and p(8 × 8) Pd(111), which were determined to give a lattice mismatch rate of 0.29%. Vacuum gaps of ∼10 Å for PdSANC surfaces and ∼20 Å for Pd@PdSAN2-2C surfaces were used to eliminate the interaction between neighboring slabs. The Brillouin zones were sampled with k-point meshes of 2 × 2 × 1 and 1 × 1 × 1 Gamma grids for PdSANC and Pd@PdSAN2-2C surfaces, respectively. The calculated ORR mechanisms and other computational details are provided in the SI.
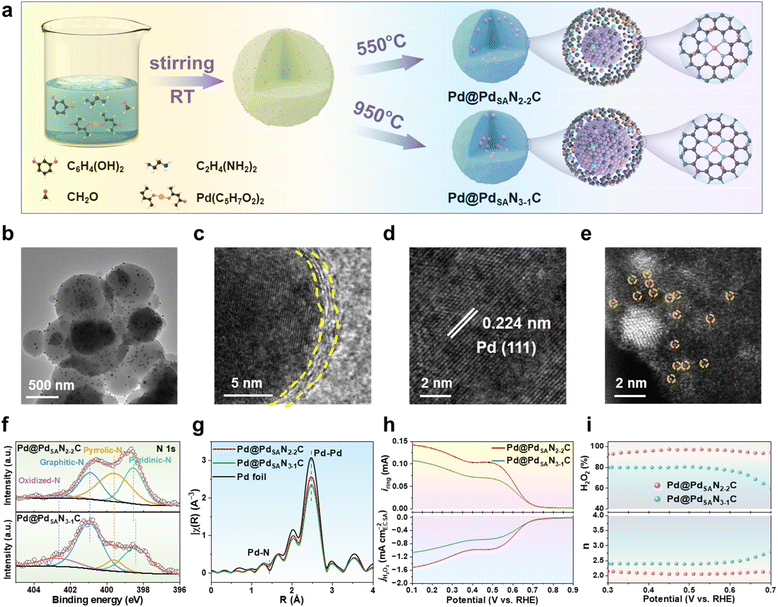
Synthesis of core–shell Pd@PdSANC catalysts
The Pd@PdSANC catalysts were synthesized using an interface-constrained self-assembly strategy.40 In a typical synthesis, 90 mL of ethanol, 45 mL of ultrapure water, and 0.45 mL of ethylenediamine were added to a 250 mL beaker and stirred at a rate of 560 rpm. for 30 min. Subsequently, 0.36 g of resorcinol was gently introduced into the above solution and stirred for another 30 min. Concurrently, 0.02 g of Pd(acac)2 was dissolved in 10 mL of ethanol by ultrasonication at 40 °C. Subsequently, 0.45 mL of formaldehyde and the prepared Pd solution were added into the above beaker and stirred for 24 h at room temperature. The resulting precursors were collected through centrifugation, rinsed three times with ultrapure water and ethanol, and dried at 65 °C. Next, the dried precursors were placed in an alumina boat and pyrolyzed in a tube furnace under a constant N2 flow at temperatures of 550 and 950 °C for 2 h to synthesize Pd@PdSAN2-2C and Pd@PdSAN3-1C, respectively.The PdSANC materials were synthesized by a dynamic coordination and selective etching process, which involved the co-condensation of Pd precursors (0.02 g Pd(acac)2) with phenolic resin precursors (0.36 g resorcinol and 0.45 mL formaldehyde) and 0.45 mL ethylenediamine, followed by nitrogen-coordination-driven pyrolysis (550 and 950 °C for 2 h with a N2 flow) and acid etching (50 mL nitrohydrochloric acid at 50 °C for 24 h), which finally generated PdSAN2-2C and PdSAN3-1C, respectively. More details of synthesis are provided in the SI.
Electrochemical measurements
All electrochemical measurements were conducted on a CHI 760E electrochemical workstation at room temperature. The experimental setup comprised a RRDE, an Ag/AgCl reference electrode (saturated by KCl), and a graphite rod as the counter electrode. The RRDE assembly (IPS Elektroniklabor) consisted of a glass carbon rotating disk electrode (disk area of 0.196 cm2) and a Pt ring (ring area of 0.159 cm2). Notably, the experimentally determined apparent collection efficiency under the ferrocyanide/ferricyanide half-reaction system agreed well with the theoretical value of 34.4%. All potentials were calibrated to the RHE according to the equation ERHE = EAg/AgCl + 0.199V + 0.059 × pH – iR × 85%, where pH was measured using a pH meter.For the preparation of catalytic ink, 4 mg of as-prepared catalyst and 1.5 mg of XC-72R were dispersed in a mixture containing 870 µL of isopropanol, 100 µL ultrapure water and 30 µL Nafion solution (5 wt%), followed by ultrasonication for 30 min to ensure a homogeneous dispersion. Then, 2.5 µL of ink was added dropwise onto the glassy carbon disk electrode and dried by rotating the electrode at 500 rpm, yielding a film-coated electrode.
The RRDE measurements were performed in O2-saturated 0.1 M KOH aqueous solution, over a potential range from 1.1 to 0.1 V vs. RHE, at a scan rate of 10 mV s−1 and a rotation speed of 1600 rpm. During these measurements, the ring electrode was held at a constant potential of 1.2 V vs. RHE. Prior to each ORR test, the catalyst was activated by 30 cycles of potential cycling between 1.1 and 0.1 V vs. RHE at a scan rate of 50 mV s−1 in N2-saturated 0.1 M KOH solution. Subsequently, the electrolyte was purged with O2 for at least 30 min to ensure optimal ORR conditions. The LSV curves for the ORR were obtained by subtracting the currents measured in N2-saturated 0.1 M KOH, effectively eliminating capacitive currents. The H2O2 reduction performance was tested in N2-saturated 0.1 M KOH containing 10 mM H2O2. The stability of the as-prepared catalysts was recorded by current–time (i–t) chronoamperometric measurements at 0.1 V vs. RHE in O2-saturated 0.1 M KOH at 1600 rpm. for 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s. The H2O2 selectivity and the electron transfer number (n) were determined using the following equations:
000 s. The H2O2 selectivity and the electron transfer number (n) were determined using the following equations:
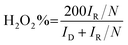 | (1) |
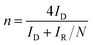 | (2) |
Results and discussion
Theoretical exploration of PdSANC catalysts with specific N ratios for the 2e− ORR
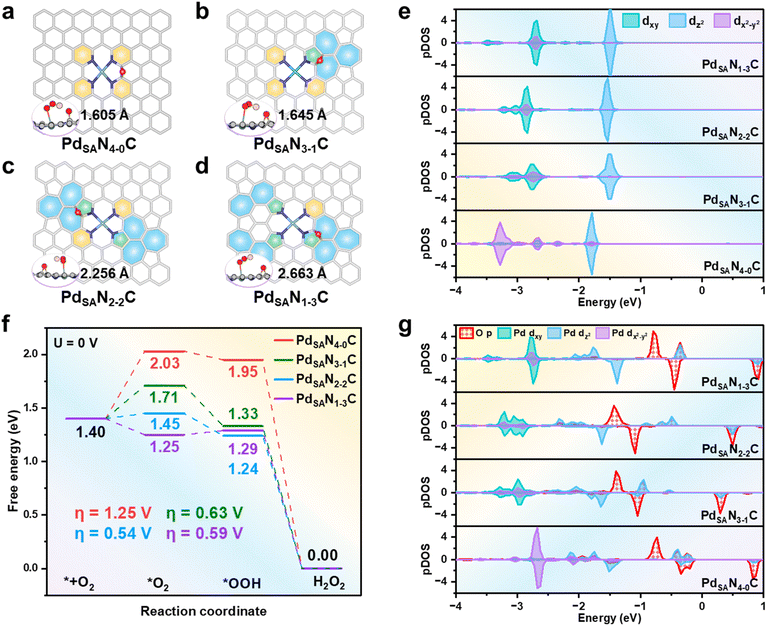
It is well known that the surface structure and composition of M-N-C catalysts, especially the ratio of pyridinic to pyrrolic N, can significantly influence their ORR activity.11,16,18 Moreover, as shown in previous studies, the presence of surface O species (particularly in the form of C–O–C groups) may also improve the 2e− ORR activity.3,31,41–43 Therefore, in this work, we first constructed a Pd-N-C catalyst modified with four pyridinic N atoms and one adsorbed O atom and progressively replaced the pyridinic N with the pyrrolic one, i.e., PdSAN4-0C, PdSAN3-1C, PdSAN2-2C and PdSAN1-3C (where 4-0, 3-1, 2-2 and 1-3 denote the numbers of pyridinic and pyrrolic N atoms, see Fig. 1a–d). For each PdSANC catalyst, multiple possible C–O–C configurations at different carbon sites were examined (Fig. S1–S4 and Tables S1–S4), and the thermodynamically most stable structure was selected for all subsequent calculations. Our calculations showed that the PdSAN4-0C structure exhibits a high degree of both axial and central symmetries. Then, the introduction of pyrrolic N effectively alters the surface symmetry, resulting in asymmetric PdSAN3-1C and PdSAN1-3C structures, while the PdSAN2-2C structure is still centrosymmetric around the Pd center. These differences in nitrogen ratios and surface symmetries can modulate the valence state of the single Pd atom. Specifically, the calculated density of states (DOS) (Fig. 1e) illustrated that the highest occupied energy levels are from the dz2 orbital in the single-atom Pd of the various PdSANC catalysts, while the introduction of pyrrolic N can modify the d-orbital configurations of the single Pd atom by causing the disappearance of dx2−y2 and the appearance of dxy at high energy levels.To investigate the activity and selectivity of the ORR on the PdSAN4-0C, PdSAN3-1C, PdSAN2-2C and PdSAN1-3C catalysts, we systematically calculated the Gibbs free energy changes (ΔG) for the key steps in both 2e− and 4e− ORR processes (2e− associative pathway: O2 + 2(H2O + e−) → H2O2 + 2OH−; 4e− associative pathway O2 + 4(H2O+ e−) → 2H2O + 4OH−) on these catalysts (Fig. S5–S10). The computational details are presented in SI Methods: ORR mechanism and calculation details. It is widely accepted that the catalytic selectivity towards H2O2 formation over H2O is critically governed by the thermodynamics of the key intermediates, and a key descriptor for predicting this selectivity is the Gibbs free energy of *O (ΔG(*O)).44,45 This is because, to make H2O2 production thermodynamically favorable, the O–O bond scission for the surface *OOH intermediate must be suppressed, and this can be achieved when the ΔG(*O) is higher than 3.52 eV (ΔGH2O2).44–47 Our results showed that the calculated ΔG(*O) values for the PdSANC catalysts are all higher than 3.52 eV (the corresponding ΔG(*O) values are 3.63, 4.24, 4.81 and 4.92 eV for PdSAN4-0C, PdSAN3-1C, PdSAN2-2C and PdSAN1-3C, see Fig. S1), indicating high selectivity for H2O2 production on these catalysts. At the same time, it can be noticed that the steps with the maximum ΔG (ΔGmax) during the 2e− ORR are identical at various PdSANC catalysts, i.e., the potential determining step (PDS) is the first step: O2 + H2O + e− + * → *OOH + OH−. The corresponding overpotentials (η) were calculated to be 1.25 V, 0.63 V, 0.54 V and 0.59 V for the PdSAN4-0C, PdSAN3-1C, PdSAN2-2C and PdSAN1-3C catalysts, respectively, clearly showing that PdSAN2-2C shows the highest 2e− ORR activity (Fig. 1f).
To understand the differences in 2e− ORR activity and selectivity among these PdSANC catalysts, we performed DOS and Bader charge analyses to investigate the adsorption of key intermediates during the reaction processes on these catalyst surfaces. The calculated results revealed that on all the PdSANC catalysts, the *OOH intermediate binds to the Pd sites (with the O atom directly bonding with Pd being defined as OT). Although a weak hydrogen-bond interaction between the H atom in *OOH and the O atom in the C–O–C group can help stabilize *OOH, this effect is common to all PdSANC catalysts, and the C–O–C group does not alter the Bader charge of the Pd center (Fig. 1a–d, S11 and S12). The stability of the resulting Pd–OT bond mainly originates from electron transfer from the dz2 orbital of Pd to the OT 2p orbital (see Fig. 1e and g and S10 and Tables S5 and S6). The amount of electron transfer in this process follows the order: PdSAN2-2C > PdSAN1-3C > PdSAN3-1C ≈ PdSAN4-0C (with transferred charges calculated to be 0.225, 0.159, 0.103 and 0.108 |e|, respectively, see Table S5). In addition, we also found that on the surfaces where both pyridinic- and pyrrolic-N exist, in comparison with the PdSAN4-0C surface, the energy level of the dxy orbital is lowered upon OOH adsorption, which may further indicate the enhancement of OOH adsorption.
Furthermore, we observed that as the content of pyrrolic N increases, the calculated Bader charge on the single atom Pd becomes more positive (0.709, 0.702, 0.712, and 0.741 |e| for PdSAN4-0C, PdSAN3-1C, PdSAN2-2C and PdSAN1-3C, respectively, see Fig. S13). It clearly showed that a moderate (neither too high nor too low) positive Bader charge on the Pd site is favorable for OOH adsorption. Nevertheless, the PdSAN2-2C surface still exhibits the largest Bader charge change for the Pd after OOH adsorption, reaching 0.119 |e| (Fig. S13). Finally, we further calculated the electrostatic interaction energies (Einter) between the OT atom of the adsorbed OOH species and the Pd active site, which again confirms that the PdSAN2-2C surface interacts most strongly with the *OOH species (Einter values for PdSAN4-0C, PdSAN3-1C, PdSAN2-2C and PdSAN1-3C are −1.27 eV, −1.28 eV, −1.50 eV and −1.35 eV, respectively, see Table S7), in excellent agreement with the calculated 2e− ORR activity of the PdSANC catalysts.
Interestingly, our results can reveal a direct linear correlation between electron transfer and electrostatic interaction across the different PdSANC configurations: systems exhibiting larger electron transfer consistently display stronger electrostatic interactions (Fig. S14). Specifically, different N-coordination environments modulate the energy level distribution of Pd d-orbitals; the resulting changes further tune the ability of Pd to transfer electrons to *OOH, thereby strengthening the electrostatic interaction between Pd and the terminal O atom (OT) in *OOH and ultimately governing the adsorption strength of *OOH. From the above discussion, we can conclude that the centrosymmetric PdSAN2-2C surface, with its balanced pyridinic-to-pyrrolic N ratio, can regulate the d-orbital distribution of the Pd center to transfer more electrons and achieve stronger electrostatic stabilization and help it maintain a moderate Bader charge, thereby facilitating OOH adsorption and leading to excellent 2e− ORR catalytic activity.
Syntheses and characterization of novel Pd-N-C catalysts
Inspired by the theoretical predictions of the optimal nitrogen coordination ratio for the electrochemical synthesis of hydrogen peroxide via the 2e− ORR, we tentatively synthesized two prototypical single-atom PdSANC catalysts, PdSAN2-2C (with high activity) and PdSAN3-1C (with low activity), both featuring the coexistence of pyridinic and pyrrolic nitrogen species. To rationally engineer these PdSANC catalysts, we developed two distinct synthetic strategies. Firstly, a dynamic coordination and selective etching approach40 was used to construct these single-atom PdSANC catalysts with atomically dispersed Pd-N sites and tunable pyridinic-to-pyrrolic N distributions. Transmission electron microscopy (TEM) and high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) images confirmed the atomic dispersion of Pd in the as-prepared PdSANC catalysts (Fig. S15–S17). X-ray photoelectron spectroscopy (XPS) analysis revealed that the PdSAN2-2C and PdSAN3-1C catalysts synthesized in this way indeed possess optimized pyridinic-to-pyrrolic nitrogen ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 3
1 and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively (Fig. S18). Notably, the PdSAN2-2C catalyst exhibited approximately 64% higher ring current (iring) and 16% higher disk current density (jdisk) than PdSAN3-1C, with these performance enhancements being consistently observed across a broad potential window ranging from 0.3 to 0.6 V vs. RHE. Furthermore, PdSAN2-2C exhibited exceptional electrocatalytic performance, with a hydrogen peroxide selectivity close to 93% and an electron transfer number (n) of 2.2, which is also a significant improvement over PdSAN3-1C (77% selectivity, n = 2.5) as evidenced by the rotating ring-disk electrode (RRDE) measurement (Fig. S19–S22). The general consistency between experimental results and theoretical calculations suggested that the nitrogen coordination engineering strategy for the construction of a centrosymmetric structure can potentially give rise to enhanced 2e− ORR activity and selectivity.
1, respectively (Fig. S18). Notably, the PdSAN2-2C catalyst exhibited approximately 64% higher ring current (iring) and 16% higher disk current density (jdisk) than PdSAN3-1C, with these performance enhancements being consistently observed across a broad potential window ranging from 0.3 to 0.6 V vs. RHE. Furthermore, PdSAN2-2C exhibited exceptional electrocatalytic performance, with a hydrogen peroxide selectivity close to 93% and an electron transfer number (n) of 2.2, which is also a significant improvement over PdSAN3-1C (77% selectivity, n = 2.5) as evidenced by the rotating ring-disk electrode (RRDE) measurement (Fig. S19–S22). The general consistency between experimental results and theoretical calculations suggested that the nitrogen coordination engineering strategy for the construction of a centrosymmetric structure can potentially give rise to enhanced 2e− ORR activity and selectivity.
Secondly, an interface-constrained self-assembly strategy (Fig. 2a) was used to form a N-containing resin precursor on Pd, followed by pyrolysis under N2 to generate a structure in which Pd nanoparticles are encapsulated by an ultrathin Pd-N-C shell. By varying the pyrolysis temperature, the pyridinic/pyrrolic N distribution in the shell can be tuned. Specifically, at relatively lower temperatures, pyrrolic-N in pentagonal ring structures is more readily retained within the forming carbon matrix. As the temperature increases, additional dehydrogenation and structural rearrangement favor the formation of thermodynamically more stable pyridinic-N sites in hexagonal rings, leading to a corresponding increase in the proportion of pyridinic-N. Intriguingly, the as-prepared catalyst gave the conformation with a PdSANC shell encapsulating the Pd nanoparticle; i.e., a Pd@PdSANC core–shell architecture was obtained through this synthesis strategy (Fig. 2b–e). The TEM measurement revealed that the core–shell Pd@PdSANC catalysts exhibit a nanosphere morphology, showing Pd nanocrystals with an average size of 21.9 nm (Fig. 2b and S23–S25) encapsulated by a PdSANC shell with a thickness of 1.0 nm (Fig. 2c–e). The core–shell structure was further confirmed by HAADF-STEM images (Fig. S26 and S27). Moreover, the uniform distribution of Pd, N, O, and C atoms in the shell of Pd@PdSANC was evidenced by STEM energy dispersive X-ray spectroscopy (EDS) mapping (Fig. S28 and S29). Overall, these findings suggested that following an interface-constrained self-assembly strategy, the structural evolution of the catalyst mainly originates from the suppression of Pd aggregation mediated by oxygen-containing functional groups. This modulation also facilitates lattice matching between metallic Pd and graphitic carbon domains during pyrolysis, thus promoting the epitaxial growth of nitrogen-doped carbon shells.
Then, the electronic properties of the Pd@PdSANC catalysts were analyzed using XPS (Fig. 2f and S30–S35) and X-ray absorption spectroscopy (XAS). The deconvoluted C 1s XPS spectra revealed peaks at 284.8 eV (sp2-hybridized C–C), 285.9 eV (C–O/C–N), 287.3 eV (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 288.9 eV (COOH), and 290.7 eV (π–π*) (Fig. S33a). The peak positions for both Pd@PdSAN2-2C and Pd@PdSAN3-1C remained virtually unchanged, with their corresponding peak area ratios also showing no significant variation. Combined with the Raman spectra (Fig. S33b), the ID/IG values of Pd@PdSAN2-2C and Pd@PdSAN3-1C are both less than 1, indicating that the carbon defect contents of both are relatively low.48–50 Therefore, we can expect that the main source of its 2e− ORR activity is not the carbon framework itself. The high-resolution N 1s XPS spectra of Pd@PdSAN2-2C (Fig. 2f) show three prominent subpeaks at 398.4, 399.6 and 400.9 eV, corresponding to pyridinic-, pyrrolic- and graphitic-N, respectively. The combined atomic content of pyridinic- and pyrrolic-N in Pd@PdSAN2-2C was determined to be 4.17 at%, with their atomic ratio approximating 0.8 (close to 1
O), 288.9 eV (COOH), and 290.7 eV (π–π*) (Fig. S33a). The peak positions for both Pd@PdSAN2-2C and Pd@PdSAN3-1C remained virtually unchanged, with their corresponding peak area ratios also showing no significant variation. Combined with the Raman spectra (Fig. S33b), the ID/IG values of Pd@PdSAN2-2C and Pd@PdSAN3-1C are both less than 1, indicating that the carbon defect contents of both are relatively low.48–50 Therefore, we can expect that the main source of its 2e− ORR activity is not the carbon framework itself. The high-resolution N 1s XPS spectra of Pd@PdSAN2-2C (Fig. 2f) show three prominent subpeaks at 398.4, 399.6 and 400.9 eV, corresponding to pyridinic-, pyrrolic- and graphitic-N, respectively. The combined atomic content of pyridinic- and pyrrolic-N in Pd@PdSAN2-2C was determined to be 4.17 at%, with their atomic ratio approximating 0.8 (close to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, see Fig S35 and Table S8). In contrast, Pd@PdSAN3-1C exhibited a ratio of 3
1, see Fig S35 and Table S8). In contrast, Pd@PdSAN3-1C exhibited a ratio of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for these two nitrogen species. Hence, although the two Pd@PdSANC catalysts demonstrated a similar carbon shell with similar carbon species and carbon defects, accompanied by atomically dispersed Pd, the Pd@PdSAN2-2C and Pd@PdSAN3-1C catalysts featured distinctive functional moieties in the carbon shell, including pyridinic- and pyrrolic-N, with a ratio close to 1
1 for these two nitrogen species. Hence, although the two Pd@PdSANC catalysts demonstrated a similar carbon shell with similar carbon species and carbon defects, accompanied by atomically dispersed Pd, the Pd@PdSAN2-2C and Pd@PdSAN3-1C catalysts featured distinctive functional moieties in the carbon shell, including pyridinic- and pyrrolic-N, with a ratio close to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 3
1 and 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, respectively. Complementary XAS results provided critical information on the Pd chemical states. In the X-ray absorption near-edge structure (XANES) analysis, the absorption edges of these Pd@PdSANC catalysts shift to higher energy compared with the metallic Pd foil, indicating partially oxidized Pd (Fig. S36). Extended X-ray absorption fine structure (EXAFS) analysis further reveals a pronounced metal peak at ∼2.5 Å, corresponding to Pd–Pd coordination (Fig. 2g), together with an additional feature at ∼1.5 Å that can be assigned to the Pd–N coordination1–3 (Fig. S37 and S38). It should be noted that because the bond lengths of Pd–N and Pd–O are nearly identical, structural assignment based solely on EXAFS fitting carries uncertainty. To further support the presence of Pd–N coordination, the color change observed during catalyst preparation and the UV-vis results (Fig. S39 and S40) were provided. These results indicated that, at the early stage of the liquid-phase synthesis, ethylenediamine effectively replaces the original acetylacetonate ligands and preferentially pre-coordinates with the Pd centers, thereby providing the basis for the formation of Pd–N coordination in the final catalyst. The fitted Pd–N coordination number is 3.89 (Table S9), suggesting the presence of Pd–N bonds within the PdN4 structures. The integration of the morphological characterization and XPS analysis of Pd 3d gave compelling evidence for the existence of a core–shell structure, where the oxidized Pd species with nitrogen coordination constitute the PdSANC shell, while the metallic Pd forms the encapsulated Pd nanoparticle core.51
1, respectively. Complementary XAS results provided critical information on the Pd chemical states. In the X-ray absorption near-edge structure (XANES) analysis, the absorption edges of these Pd@PdSANC catalysts shift to higher energy compared with the metallic Pd foil, indicating partially oxidized Pd (Fig. S36). Extended X-ray absorption fine structure (EXAFS) analysis further reveals a pronounced metal peak at ∼2.5 Å, corresponding to Pd–Pd coordination (Fig. 2g), together with an additional feature at ∼1.5 Å that can be assigned to the Pd–N coordination1–3 (Fig. S37 and S38). It should be noted that because the bond lengths of Pd–N and Pd–O are nearly identical, structural assignment based solely on EXAFS fitting carries uncertainty. To further support the presence of Pd–N coordination, the color change observed during catalyst preparation and the UV-vis results (Fig. S39 and S40) were provided. These results indicated that, at the early stage of the liquid-phase synthesis, ethylenediamine effectively replaces the original acetylacetonate ligands and preferentially pre-coordinates with the Pd centers, thereby providing the basis for the formation of Pd–N coordination in the final catalyst. The fitted Pd–N coordination number is 3.89 (Table S9), suggesting the presence of Pd–N bonds within the PdN4 structures. The integration of the morphological characterization and XPS analysis of Pd 3d gave compelling evidence for the existence of a core–shell structure, where the oxidized Pd species with nitrogen coordination constitute the PdSANC shell, while the metallic Pd forms the encapsulated Pd nanoparticle core.51
Intriguingly, the H2O2 selectivity of Pd@PdSAN2-2C was the highest among those of the various PdSANC and Pd@PdSAN3-1C catalysts, accompanied by significantly enhanced intrinsic activity (Fig. 2h and S20, S41–S46), indicating its superior ORR performance. Specifically, the Pd@PdSAN2-2C catalyst achieved approximately 97% hydrogen peroxide selectivity and an electron transfer number of 2.1 across a very broad potential range from 0.3 to 0.7 V vs. RHE, outperforming those of previously reported catalysts2,19,26–31 (Fig. 2i, S47 and S48, Table S10). Furthermore, RRDE measurement revealed that the Pd@PdSAN2-2C catalyst also exhibited accelerated electron transfer kinetics and exceptional mass activity (Fig. S49 and S50). However, RRDE tests are often affected by O2 diffusion. In practical applications using a flow-cell measurement, which effectively overcomes the influence of mass transfer, it can be anticipated that Pd@PdSAN2-2C would achieve more efficient H2O2 synthesis.
Theoretical evidence for the high performance of Pd@PdSANC in the 2e− ORR
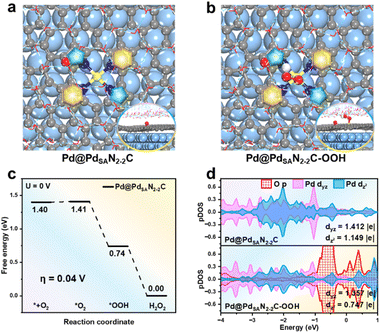
To elucidate the reason for the enhanced 2e− ORR activity of the Pd@PdSANC catalysts, we modeled and calculated the characteristic Pd@PdSAN2-2C structure with a minimal lattice mismatch rate of 0.29% (Table S11). It was constructed with a metallic Pd slab as the core covered by a PdSANC single layer containing pyridinic and pyrrolic N at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio (Fig. 3a). We then systematically calculated the Gibbs free energy changes for the key steps of the 2e− and 4e− ORR processes on this Pd@PdSAN2-2C catalyst (Fig. 3b and c, S51 and S52). The results showed that the ΔG(*O) on Pd@PdSAN2-2C is 4.36 eV (Fig. S52), which is greater than the benchmark of 3.52 eV,44–47 indicating a preference for the 2e− ORR pathway and H2O2 production. We also found that the calculated η of such a core–shell Pd@PdSAN2-2C catalyst (η = 0.04 V) is much lower than that of the PdSAN2-2C catalyst (η = 0.54 V) and the Pd-N-C materials reported in the literature as well (η = 0.77 V),19 which aligns well with our experimental results.
1 ratio (Fig. 3a). We then systematically calculated the Gibbs free energy changes for the key steps of the 2e− and 4e− ORR processes on this Pd@PdSAN2-2C catalyst (Fig. 3b and c, S51 and S52). The results showed that the ΔG(*O) on Pd@PdSAN2-2C is 4.36 eV (Fig. S52), which is greater than the benchmark of 3.52 eV,44–47 indicating a preference for the 2e− ORR pathway and H2O2 production. We also found that the calculated η of such a core–shell Pd@PdSAN2-2C catalyst (η = 0.04 V) is much lower than that of the PdSAN2-2C catalyst (η = 0.54 V) and the Pd-N-C materials reported in the literature as well (η = 0.77 V),19 which aligns well with our experimental results.
To further understand the fundamental reasons behind the high activity and selectivity of the Pd@PdSAN2-2C catalyst in the 2e− ORR process, we first carried out DOS calculations for the key intermediates on the surface. The results demonstrated that during the formation of the bond between the *OOH species and the single-atom Pd on the Pd@PdSAN2-2C surface, the electron occupation of the Pd dz2 orbital is significantly reduced by 0.402 |e| (Fig. 3d), which is nearly twice that determined for the PdSAN2-2C catalyst (0.225 |e|, Table S5). Furthermore, Bader charge analysis revealed that, although the single atom Pd sites in both PdSAN2-2C and Pd@PdSAN2-2C catalysts possess similar Bader charges (0.712 and 0.718 |e|, respectively), the change in Bader charge of the shell Pd single atoms after OOH adsorption is more pronounced for the latter (0.119 and 0.125 |e| for PdSAN2-2C and Pd@PdSAN2-2C, respectively, Fig. S53). Notably, further calculations revealed that the core Pd cluster also promotes the electrostatic interaction between the single-atom Pd site in the PdSAN2-2C shell and the OT in *OOH (the corresponding electrostatic interaction energy is −2.43 eV, see Fig. S53). To further investigate the role of the metallic Pd core, we calculated the d-band center of Pd on the PdSAN2-2C and Pd@PdSAN2-2C surfaces, as well as that of the shell (PdSAN2-2C(Pd@PdSAN2-2C)) and the core (Pd(Pd@PdSAN2-2C)) in Pd@PdSAN2-2C. The results indicate that the single-atom Pd in the PdSAN2-2C(Pd@PdSAN2-2C) shell has the lowest d-band center (Table S12), suggesting that the metallic Pd core can modulate the d-band center of the surface single-atom Pd via charge transfer, thereby optimizing the OOH adsorption and enhancing the 2e− ORR performance of Pd@PdSAN2-2C.
Structure–activity relationship of the Pd@PdSAN2-2C catalyst
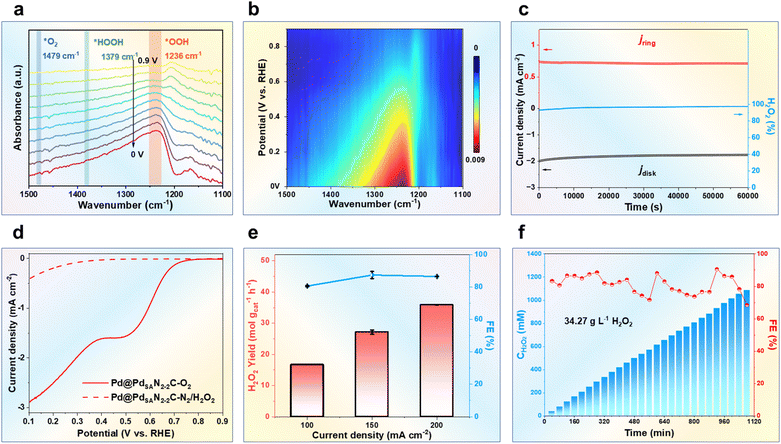
Detailed experimental tests were conducted to better illuminate the catalytic activity of the Pd@PdSAN2-2C catalyst. In situ attenuated total reflection Fourier-transform infrared spectra (ATR-FTIR) were obtained for the ORR process at various potentials ranging from 0.9 to 0.0 V vs. RHE, and they revealed three distinct peaks, where the peak at 1236 cm−1 corresponds to the O-O stretching of the adsorbed OOH, while the peaks at about 1397 cm−1 and 1457 cm−1 may originate from the adsorbed H2O2 and O2, respectively (Fig. 4a). Most importantly, the intensity of the broad *OOH peak increased as the applied potential decreased from 0.9 to 0.0 V vs. RHE (Fig. 4b), confirming that the 2e− ORR pathway indeed occurs on the Pd@PdSAN2-2C surface.In addition, to elucidate the effect of the Pd single atom within the PdSANC shell on the ORR activity and selectivity, a comparative study was performed by utilizing commercial Pd/C and the PdSANC catalysts. The results showed that the exposed Pd nanoparticles in commercial Pd/C mainly facilitate the 4e− ORR pathway (Fig. S54), while the abundant surface Pd single atoms in the PdSANC shell of the Pd@PdSAN2-2C catalyst serve as key active sites to promote the ORR process for H2O2 production. This conclusion was further confirmed by the SCN− poisoning experiment. Upon the addition of 10 mM KSCN, the disk current density decreased immediately by 9.6% (Fig. S55), further indicating that the Pd species are the main catalytic sites. This is in line with the electronic properties illustrated by the theoretical calculations discussed earlier.
Furthermore, the long-term stability of the Pd@PdSAN2-2C catalyst was evaluated using chronoamperometry assessments at a constant potential of 0.1 V vs. RHE. Over 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 s of continuous 2e− ORR operation, both the disk and ring currents remained largely unchanged, and the H2O2 selectivity remained above 97% without any obvious decrease (Fig. 4c). The very small current density of the H2O2 reduction reaction (H2O2RR) observed in Pd@PdSAN2-2C (Fig. 4d) indicated its negligible activity for the H2O2RR.
000 s of continuous 2e− ORR operation, both the disk and ring currents remained largely unchanged, and the H2O2 selectivity remained above 97% without any obvious decrease (Fig. 4c). The very small current density of the H2O2 reduction reaction (H2O2RR) observed in Pd@PdSAN2-2C (Fig. 4d) indicated its negligible activity for the H2O2RR.
To rigorously assess the electrocatalytic performance of the Pd@PdSAN2-2C catalyst for H2O2 synthesis under actual operating conditions, we conducted tests using an advanced three-phase flow cell setup (Fig. S56 and S57). The results further demonstrated that Pd@PdSAN2-2C exhibited a high catalytic activity, with a H2O2 yield exceeding 15 mol gcat−1 h−1 at current densities from 100 to 200 mA cm−2. Specifically, the H2O2 yield of Pd@PdSAN2-2C reached 35.88 mol gcat−1 h−1 at 200 mA cm−2, with a H2O2 Faradaic efficiency (FE) of 86.55% and a mass activity of 870 AH2O2gcat−1. Compared with those of most reported electrocatalysts, this result demonstrates the relatively superior performance of our material, particularly in terms of H2O2 yield (Fig. 4e and S58, Table S13). Notably, even in 1.0 M KOH, Pd@PdSAN2-2C demonstrated a superior H2O2 formation activity of 50 mol gcat−1 h−1 and a H2O2 FE of approximately 90% (Fig. S59). These results suggested that the core–shell Pd@PdSAN2-2C catalyst with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of pyridinic to pyrrolic N indeed exhibits a remarkable H2O2 generation rate at industrially relevant current densities.
1 ratio of pyridinic to pyrrolic N indeed exhibits a remarkable H2O2 generation rate at industrially relevant current densities.
Moreover, the large-scale continuous productivity and electrochemical stability of Pd@PdSAN2-2C were further evaluated in 0.1 M KOH at 200 mA cm−2. During the test, the potential remained stable at around −0.1 V vs. RHE (Fig. S60), and the H2O2 FE remained higher than 80%. Continuous electrolysis at 200 mA cm−2 for 1080 min gave a sustained high H2O2 yield, achieving a concentration of up to 34.27 g L−1 (Fig. 4f). Post-test XRD patterns, TEM images and XPS spectra of the catalyst showed neglectable changes (Fig. S61–S64). These findings demonstrated that the Pd@PdSAN2-2C catalyst can maintain excellent H2O2 generation performance even under scaled-up operation conditions, highlighting its superior efficacy and versatility.
Conclusions
In summary, we have systematically investigated the effects of local structures and nitrogen environments in Pd-N-C catalysts on the electrochemical 2e− ORR for H2O2 formation through combined theoretical and experimental studies. DFT calculation results revealed that the PdSAN2-2C catalyst, featuring a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio of pyridinic to pyrrolic N atoms, exhibited superior activity for the 2e− ORR. This exceptional activity was attributed to the centrosymmetric structure of PdN4 and the involvement of the dz2 orbital of the single Pd atom in the formation of stable Pd-O bonds with the 2p orbital of OT species in the adsorbed OOH intermediate. Furthermore, experimental efforts enabled the successful synthesis of both the PdSAN2-2C catalyst and a complex one with a core–shell nanostructure, where the exposed two-dimensional structure involving atomically dispersed Pd atoms with tailored nitrogen coordination serves as the shell and the Pd nanoparticle as the core. The Pd@PdSAN2-2C catalyst exhibited excellent 2e− ORR performance for H2O2 production, achieving a selectivity of 97% and a robust ring current across a broad potential window from 0.1 to 0.7 V vs. RHE in alkaline media. Our calculated results further illustrated that the high performance of the Pd@PdSAN2-2C catalyst for the 2e− ORR can be again attributed to the centrosymmetric PdN4 structure in the shell, which maintains a structure similar to that of PdSAN2-2C, as well as the existence of the Pd core that can modulate the d-orbital arrangement of the single Pd atom in the shell. Within a gas diffusion electrode integrated flow-cell setup, Pd@PdSAN2-2C achieved a high H2O2 yield of 35.88 mol gcat−1 h−1 at 200 mA cm−2 with over 80% selectivity, alongside a cumulative H2O2 concentration of 34.27 g L−1 over 1080 min, surpassing previously reported metal single-atom electrocatalysts. This work not only provides a deeper understanding of the structural and electronic features of M-N-C materials in electrocatalytic reactions, but it also offers valuable guidance for the development of highly efficient electrocatalysts for H2O2 production.
1 ratio of pyridinic to pyrrolic N atoms, exhibited superior activity for the 2e− ORR. This exceptional activity was attributed to the centrosymmetric structure of PdN4 and the involvement of the dz2 orbital of the single Pd atom in the formation of stable Pd-O bonds with the 2p orbital of OT species in the adsorbed OOH intermediate. Furthermore, experimental efforts enabled the successful synthesis of both the PdSAN2-2C catalyst and a complex one with a core–shell nanostructure, where the exposed two-dimensional structure involving atomically dispersed Pd atoms with tailored nitrogen coordination serves as the shell and the Pd nanoparticle as the core. The Pd@PdSAN2-2C catalyst exhibited excellent 2e− ORR performance for H2O2 production, achieving a selectivity of 97% and a robust ring current across a broad potential window from 0.1 to 0.7 V vs. RHE in alkaline media. Our calculated results further illustrated that the high performance of the Pd@PdSAN2-2C catalyst for the 2e− ORR can be again attributed to the centrosymmetric PdN4 structure in the shell, which maintains a structure similar to that of PdSAN2-2C, as well as the existence of the Pd core that can modulate the d-orbital arrangement of the single Pd atom in the shell. Within a gas diffusion electrode integrated flow-cell setup, Pd@PdSAN2-2C achieved a high H2O2 yield of 35.88 mol gcat−1 h−1 at 200 mA cm−2 with over 80% selectivity, alongside a cumulative H2O2 concentration of 34.27 g L−1 over 1080 min, surpassing previously reported metal single-atom electrocatalysts. This work not only provides a deeper understanding of the structural and electronic features of M-N-C materials in electrocatalytic reactions, but it also offers valuable guidance for the development of highly efficient electrocatalysts for H2O2 production.
Author contributions
The manuscript was written through contributions from all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
Supplementary information (SI): data related to the ORR mechanism, computational details, chemicals and reagents, physicochemical characterization and electrochemical tests. See DOI: https://doi.org/10.1039/d5sc09543c.Acknowledgements
This work was supported by the National Key R&D Program of China (2024YFA1509901, 2023YFA1508500, and 2021YFA1500700) and the National Nature Science Foundation of China (22572049 and 22203030).References
- H. Liu, L. Jiang, Y. Sun, J. Khan, B. Feng, J. Xiao, H. Zhang, H. Xie, L. Li, S. Wang and L. Han, Adv. Energy Mater., 2023, 13, 2301223 CrossRef CAS.
- B. Yue, L. Lei, H. Xie, Y. Si, Q. Yang and X. Liu, ACS Appl. Mater. Interfaces, 2023, 15, 33665–33674 CrossRef CAS PubMed.
- Q. Zhang, X. Tan, N. M. Bedford, Z. Han, L. Thomsen, S. Smith, R. Amal and X. Lu, Nat. Commun., 2020, 11, 4181 CrossRef PubMed.
- D. Zhang, F. She, J. Chen, L. Wei and H. Li, J. Am. Chem. Soc., 2025, 147, 6076–6086 CrossRef CAS PubMed.
- H. T. Chung, D. A. Culle, D. Higgins, B. T. Snee, E. F. Holb, K. L. More and P. Zelenay, Science, 2017, 357, 479–484 CrossRef CAS PubMed.
- K. Kumar, L. Dubau, F. Jaouen and F. Maillard, Chem. Rev., 2023, 123, 9265–9326 CrossRef CAS PubMed.
- L. Yan, P. Li, Q. Zhu, A. Kumar, K. Sun, S. Tian and X. Sun, Chem, 2023, 9, 280–342 CAS.
- H. Huang, M. Sun, S. Li, S. Zhang, Y. Lee, Z. Li, J. Fang, C. Chen, Y.-X. Zhang, Y. Wu, Y. Che, S. Qian, W. Zhu, C. Tang, Z. Zhuang, L. Zhang and Z. Niu, J. Am. Chem. Soc., 2024, 146, 9434–9443 CrossRef CAS PubMed.
- Z. Levell, S. Yu, R. Wang and Y. Liu, J. Am. Chem. Soc., 2025, 147, 603–609 CrossRef CAS PubMed.
- Y. Wang, R. Shi, L. Shang, G. I. N. Waterhouse, J. Zhao, Q. Zhang, L. Gu and T. Zhang, Angew. Chem., Int. Ed., 2020, 59, 13057–13062 CrossRef CAS PubMed.
- J. Gao and B. Liu, ACS Mater. Lett., 2020, 2, 1008–1024 CrossRef CAS.
- S. Chen, T. Luo, X. Li, K. Chen, J. Fu, K. Liu, C. Cai, Q. Wang, H. Li, Y. Chen, C. Ma, L. Zhu, Y.-R. Lu, T.-S. Chan, M. Zhu, E. Cortés and M. Liu, J. Am. Chem. Soc., 2022, 144, 14505–14516 CrossRef CAS PubMed.
- W. Wang, Y. Hu, P. Li, Y. Liu and S. Chen, ACS Catal., 2024, 14, 5961–5971 CrossRef CAS.
- N. Zhang, T. Zhou, M. Chen, H. Feng, R. Yuan, C. Zhong, W. Yan, Y. Tian, X. Wu, W. Chu, C. Wu and Y. Xie, Energy Environ. Sci., 2020, 13, 111–118 RSC.
- B. Ni, P. Shen, G. Zhang, J. Zhao, H. Ding, Y. Ye, Z. Yue, H. Yang, H. Wei and K. Jiang, J. Am. Chem. Soc., 2024, 146, 11181–11192 CAS.
- H. Xu, D. Cheng, D. Cao and X. C. Zeng, Nat. Catal., 2024, 7, 207–218 CrossRef CAS.
- J. Gao, H. Yang, X. Huang, S.-F. Hung, W. Cai, C. Jia, S. Miao, H. M. Chen, X. Yang, Y. Huang, T. Zhang and B. Liu, Chem, 2020, 6, 658–674 CAS.
- K. Jiang, S. Back, A. J. Akey, C. Xia, Y. Hu, W. Liang, D. Schaak, E. Stavitski, J. K. Nørskov, S. Siahrostami and H. Wang, Nat. Commun., 2019, 10, 3997 CrossRef PubMed.
- N. Wang, X. Zhao, R. Zhang, S. Yu, Z. H. Levell, C. Wang, S. Ma, P. Zou, L. Han, J. Qin, L. Ma, Y. Liu and H. L. Xin, ACS Catal., 2022, 12, 4156–4164 CrossRef CAS.
- Y. L. Wang, S. Gurses, N. Felvey, A. Boubnov, S. S. Mao and C. X. Kronawitter, ACS Catal., 2019, 9, 8453–8463 CrossRef CAS.
- Z. Wei, B. Deng, P. Chen, T. Zhao and S. Zhao, Chem. Eng. J., 2022, 428, 131112 CrossRef CAS.
- T. Zhang, W. Wang, W. Liu, Z. Guo and J. Liu, Nat. Commun., 2025, 16, 5240 CrossRef CAS PubMed.
- T. Wu, M. Sun and B. Huang, Mater. Today Energy, 2019, 12, 426e430 Search PubMed.
- J. S. Spendelow and A. Wieckowski, Phys. Chem. Chem. Phys., 2007, 9, 26542675 RSC.
- S. C. Perry, D. Pangotra, L. Vieira, L.-I. Csepei, V. Sieber, L. Wang, C. P. León and F. C. Walsh, Nat. Rev. Chem., 2019, 3, 442–458 CrossRef CAS.
- B. Xia, Q. Huang, K. Wu, L. Jiang, M. Li, L. Yu, S. Ding, Z. Nie, D. Hua, J. Duan and S. Chen, AIChE J., 2023, 69, e18022 CrossRef CAS.
- R. Shen, R. Shen, W. Chen, Q. Peng, S. Lu, L. Zheng, X. Cao, Y. Wang, W. Zhu, J. Zhang, Z. Zhuang, C. Chen, D. Wang and Y. Li, Chem, 2019, 5, 2099–2110 CAS.
- Y. Sun, L. Silvioli, N. R. Sahraie, W. Ju, J. Li, A. Zitolo, S. Li, A. Bagger, L. Arnarson, X. Wang, T. Moeller, D. Bernsmeier, J. Rossmeisl, F. Jaouen and P. Strasser, J. Am. Chem. Soc., 2019, 141, 12372–12381 CrossRef CAS PubMed.
- J. Du, S. Jiang, R. Zhang, P. Wang, C. Ma, R. Zhao, C. Cui, Y. Zhang and Y. Kang, ACS Catal., 2023, 13, 6887–6892 CrossRef CAS.
- S. Zhang, Z. Tao, M. Xu, L. Kan, C. Guo, J. Liu, L. He, M. Du and Z. Zhang, Small, 2024, 20, 2310468 CrossRef CAS PubMed.
- Q. Chang, P. Zhang, A. H. B. Mostaghimi, X. Zhao, S. R. Denny, J. H. Lee, H. Gao, Y. Zhang, H. L. Xin, S. Siahrostami, J. G. Chen and Z. Chen, Nat. Commun., 2020, 11, 2178 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169 CrossRef CAS PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B:Condens. Matter Mater. Phys., 1993, 47, 558 CrossRef CAS PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 49, 14251 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- J. P. Perdew, J. A. Chevary, S. H. Vosko, K. A. Jackson, M. R. Pederson, D. J. Singh and C. Fiolhais, Phys. Rev. B:Condens. Matter Mater. Phys., 1992, 46, 6671 CrossRef CAS PubMed.
- M. P. Teter, M. C. Payne and D. C. Allan, Phys. Rev. B, 1989, 40, 12255–12263 CrossRef PubMed.
- P. E. Blöchl, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 50, 17953 CrossRef PubMed.
- G. Kresse and D. Joubert, Phys. Rev. B:Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- Z. Su, H. Wei, L. Zhang, H. Li, P. Liu, Z. Hou and X. Gong, Electrochim. Acta, 2023, 468, 143180 CrossRef CAS.
- Y. Yuan, J. Ma, B. Kang and J. Y. Lee, ACS Catal., 2025, 15, 4039–4050 CrossRef CAS.
- C. Tang, L. Chen, H. Li, L. Li, Y. Jiao, Y. Zheng, H. Xu, K. Davey and S.-Z. Qiao, J. Am. Chem. Soc., 2021, 143, 7819–7827 CrossRef CAS PubMed.
- E. Jung, H. Shin, B.-H. Lee, V. Efremov, S. Lee, H. S. Lee, J. Kim, W. H. Antink, S. Park, K.-S. Lee, S.-P. Cho, J. S. Yoo, Y.-E. Sung and T. Hyeon, Nat. Mater., 2020, 19, 436–442 CrossRef CAS PubMed.
- X. Guo, S. Lin, J. Gu, S. Zhang, Z. Chen and S. Huang, ACS Catal., 2019, 9, 11042–11054 CrossRef CAS.
- Y. Zou, X. Guo, X. Bian, Y. Zhang, W. Lin, S. Huang, Z. Chen and K. Ding, Appl. Surf. Sci., 2022, 592, 153233 CrossRef CAS.
- X. Chen, H. Zhu, J. Zhu and H. Zhang, Chem. Eng. J., 2023, 451, 138998 CrossRef CAS.
- Q. Liu, X. Bian, S. Xie, W. Ruan, W. Chen, X. Guo and K. Ding, Int. J. Hydrogen Energy, 2024, 51, 1413–1420 CrossRef CAS.
- Q. Wu, H. Zou, X. Mao, J. He, Y. Shi, S. Chen, X. Yan, L. Wu, C. Lang, B. Zhang, L. Song, X. Wang, A. Du, Q. Li, Y. Jia, J. Chen and X. Yao, Nat. Commun., 2023, 14, 6275 CrossRef CAS PubMed.
- H. Gong, L. Wei, S. Chen, Z. Chen, T. F. Jaramillo and Z. Bao, Nano Res., 2023, 16, 11556–11563 CrossRef CAS.
- J. Su, L. Jiang, B. Xiao, Z. Liu, H. Wang, Y. Zhu, J. Wang and X. Zhu, Small, 2024, 20, 2310317 CrossRef CAS PubMed.
- S. Wei, A. Li, J.-C. Liu, Z. Li, W. Chen, Y. Gong, Q. Zhang, W.-C. Cheong, Y. Wang, L. Zheng, H. Xiao, C. Chen, D. Wang, Q. Peng, L. Gu, X. Han, J. Li and Y. Li, Nat. Nanotechnol., 2018, 13, 856–861 CrossRef CAS PubMed.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |