Open Access Article
Open Access ArticleCucurbit[7]uril as a universal anchor for photoswitchable monolayers on gold
Doroteja
Lončarić
ab,
Eva
Kaletová
a,
Katarina Majerová
Varga
a,
Carina Santos
Hurtado
a,
Milan
Mašát
a,
Ivana
Císařová
b and
Jiří
Kaleta
 *a
*a
aInstitute of Organic Chemistry and Biochemistry of the Czech Academy of Sciences, Flemingovo nám. 2, 160 00 Prague 6, Czech Republic. E-mail: jiri.kaleta@uochb.cas.cz
bDepartment of Inorganic Chemistry, Faculty of Science, Charles University Prague, Hlavova 2030, 128 40 Prague 2, Czech Republic
First published on 22nd January 2026
Abstract
Photoresponsive self-assembled monolayers (SAMs) were fabricated on gold surfaces using cucurbit[7]uril (CB[7])-based host–guest complexes. In this architecture, the macrocyclic CB[7] unit serves as a universal anchoring platform, with one portal binding to the gold surface and the opposite portal encapsulating a photoswitchable molecular rod featuring a pyridinium–adamantyl recognition site. Four distinct molecular rods were synthesized to demonstrate the versatility of this modular approach. In solution, all supramolecular complexes retained the inherent photoresponsive behavior of the parent rods, exhibiting reversible isomerization with high fatigue resistance and minimal perturbation from CB[7] complexation. Upon surface immobilization, the resulting monolayers retained functional photoactivity, as demonstrated on all four systems. These results underscore the potential of CB[7]-anchored assemblies for the fabrication of robust, light-responsive surfaces and molecular devices.
Introduction
The development of functionalized two-dimensional (2-D) surfaces is central to advancing molecular electronics, sensing, and responsive materials.1–6 These systems typically consist of self-assembled monolayers (SAMs) where the constituent molecules feature three critical elements: a functional headgroup (e.g., photoswitch or molecular motor), a linker, and a surface-anchoring unit. While the headgroup is generally transferable between different systems, the design of the linker and anchoring moieties must be carefully tailored to suit the characteristics of specific substrates, such as metallic versus non-metallic surfaces, or solid versus liquid interfaces.2 These non-functional segments are not merely structural supports: they play a crucial role in orienting the headgroup, decoupling it electronically from the substrate, allowing sufficient conformational freedom for switching, and preventing intermolecular quenching or energy transfer.2 If improperly designed, they can dramatically impair or completely disable the functionality of the molecular layer.Despite extensive development of surface functionalization strategies,7–15 challenges remain. Many such approaches involve multistep syntheses and lack modularity, making them less adaptable to diverse molecular functions or new substrate types. To address these limitations, supramolecular methods based on non-covalent interactions have emerged as promising alternatives. In particular, host–guest interactions offer a unique combination of high affinity, selectivity, and reversibility, enabling the construction of modular, tunable, and dynamic molecular architectures.16–21
Herein, we present a supramolecular strategy for constructing light-responsive SAMs on gold surfaces using host–guest complexes formed between rod-like molecules 1–4 and the macrocyclic host cucurbit[7]uril (CB[7]) (Fig. 1). This approach exploits the known ability of CB[7] to physisorb onto gold surfaces via one of its portals, effectively acting as a universal anchoring unit or a molecular pedestal.22–25 To complement this, guest molecules were designed with an adamantyl–pyridinium recognition site that strongly binds within the CB[7] cavity (for reference, the closely related N-(1-adamantyl)pyridinium bromide binds CB[7] with Ka = (1.98 ± 0.42) × 1012 M−1 in aqueous buffer),26 allowing the remainder of the molecule to protrude from the host while the opposite portal of CB[7] remains available for surface anchoring.
To demonstrate the modularity and functional scope of this platform, we incorporated the binding motif into four representative molecular systems: a light-driven molecular motor 1 and three photoswitches 2–4. The formation of discrete 1:1 host–guest complexes 1–4·CB[7] in solution was confirmed, and their fundamental photochemical properties were characterized. These complexes were then immobilized onto gold surfaces to form SAMs, which were investigated using ellipsometry, contact angle measurements, polarization modulation infrared reflection–absorption spectroscopy (PM-IRRAS), and surface-enhanced Raman spectroscopy (SERS). Crucially, the photoresponsive behavior of the guest molecules was retained on the surface, demonstrating that this CB[7]-based supramolecular platform can be used to create robust, light-active monolayers with high modularity and functional fidelity.
Results and discussion
The molecular rods 1–4 were synthesized through two consecutive Sonogashira cross-coupling reactions, starting from the previously reported adamantylated pyridinium 5 (Fig. 1). In the first step, bromine was formally substituted with ethynyltrimethylsilane (6) or ethynyltriisopropylsilane (7), yielding the silyl-protected derivatives 8 and 9 in excellent yields. While both intermediates successfully led to the desired products in subsequent reactions, 9 exhibited greater stability during purification and long-term storage. The final step involved the in situ removal of the silyl protecting group using CsF, followed by cross-coupling with iodides 10–13, affording the target compounds in nearly quantitative yield (as determined by 1H NMR of the crude reaction mixture). However, the isolated yields were approximately 25%, reflecting the challenges of separating poorly soluble cationic species from complex reaction mixtures. Interestingly, all attempts to isolate the intermediate terminal acetylene failed, as its removal of silyl groups consistently led to quantitative oxidative homocoupling, preventing isolation of the desired product.Supramolecular complexes 1–4·CB[7] were quantitatively obtained in preparative quantities through a stoichiometric reaction between 1–4 and CB[7] in DMSO (Fig. 1). Encapsulation was monitored using 1H NMR spectroscopy and progress of representative titration was demonstrated on the most symmetric derivative 2 (Fig. 2). Titration data for the remaining complexes are provided in the SI.
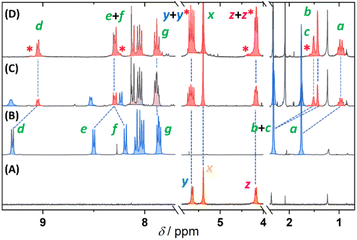 | ||
| Fig. 2 1H NMR spectra of CB[7] (A), 2 (c = 10 mmol L−1) (B), 2 with addition of 0.5 equiv. (C) and 1.0 equiv. (D) of CB[7] recorded in DMSO-d6 at 25 °C. Green asterisks indicate alkali or alkaline-earth metal-promoted trimers. See Fig. 1 for atom labelling. | ||
The attachment of the CB[7] unit resulted in a noticeable upfield shift of the adamantyl hydrogen atoms (Ha-Hc) as well as both resonances of the pyridinium moiety (Hd and He). The shielding effect on other hydrogen atoms progressively diminished with increasing distance from the CB[7] unit, clearly indicating that only the adamantylated pyridinium serves as the binding site.
It is worth noting that 1–4·CB[7] were accompanied by a small but detectable amounts of alkali or alkaline-earth metal-promoted trimers.27 Their presence does not interfere with subsequent reaction, as these species are stable only in non-aqueous environments. The overall binding affinity of structurally related compound with the analogous binding site to CB[7] in DMSO was previously determined to be 104 M−1 using UV-vis spectroscopy.26
The thermal stability of the selected supramolecular complexes 1–2·CB[7], as well as their individual components - the free rods 1 and 2 and the CB[7] macrocycle - was evaluated using differential scanning calorimetry (DSC). Pronounced endothermic transitions observed above 300 °C are attributed to the melting of the samples (Fig. 3). Notably, the melting points of the complexes were higher than those of the free guests and the CB[7] macrocycle, suggesting enhanced solid-state stability resulting from host–guest complexation.
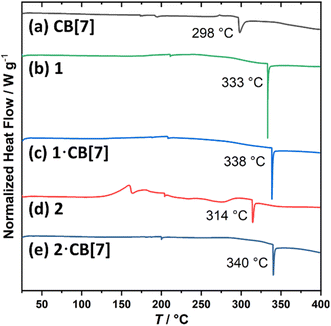 | ||
| Fig. 3 DSC traces of (a) CB[7], (b) 1, (c) 1·CB[7], (d) 2 and (e) 2·CB[7] from 25 to 350 °C, scanned at 10 °C min−1, with labelled melting points. | ||
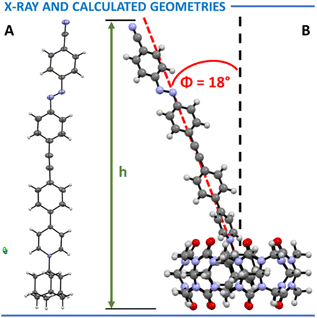
Although crystal structures of one intermediate (compound 8) and one molecular rod (compound 2 in Fig. 4) were successfully obtained, all attempts to crystallize the final complexes were unsuccessful. As a practical and informative alternative, computational modelling was employed to access the desired geometries. The structures of complexes 1–4·CB[7] were optimized using the GFN2-xTB semi-empirical method.28–30Fig. 4 presents 2·CB[7] as a representative example, while the remaining structures are provided in the SI.
In all optimized geometries, the principal molecular axis defined by the triple bonds was tilted by approximately 18° from the normal to the plane formed by the CB[7] oxygen atoms on one portal. These findings are consistent with previous observations made for structurally related systems.31
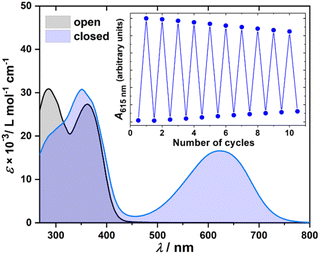
A detailed characterization of the photoswitching behavior in solution was essential before undertaking any photochemical experiments on surfaces. Additionally, it was important to assess whether the switching properties of compounds 1–4 are affected by supramolecular complexation. The proper operation of the light-driven molecular motors and photoswitches, both in the free rods 1–4 and in the supramolecular complexes 1–4·CB[7], was therefore independently evaluated using UV-vis and 1H NMR spectroscopies in DMSO. The well-soluble functional headgroups 10–13 were analyzed in chloroform. UV-vis absorption spectra of supramolecular complexes 1–4·CB[7] (c = 1.3–2.8 × 10−5 mol L−1) were recorded in the presence of an excess of CB[7] (c ≈ 1 mmol L−1) to ensure complete complexation of the molecular rods. Fig. 5 displays the spectra for photochromic 4, which exhibited the most pronounced spectral changes upon photoisomerization. UV-vis spectra for the remaining complexes are provided in the SI.
Basic kinetic parameters like enthalpy (ΔH≠) and Gibbs free energy of activation (ΔG≠), together with a half-life at 20 °C (t1/2) and a corresponding rate constant (k0) characterizing thermal steps of photoswitches and motors are summarized in Table 1. Detailed description of individual systems is provided in the SI.
| Cmpda | Irrad. (nm) | PSSb | Kinetic parameters | |||
|---|---|---|---|---|---|---|
| ΔH‡ (kJ mol−1) | ΔG‡ (kJ mol−1) | k 0 (s−1) | t 1/2 | |||
| a Compounds 1–4 and their CB[7] complexes 1–4·CB[7] were analyzed in DMSO, whereas compounds 10–13 were analyzed in chloroform. b Photostationary states (PSS) achieved at defined wavelengths as determined by 1H NMR. c Half-life (t1/2) of thermal reactions reported at 20 °C. d The value could not be determined because the required irradiation time exceeded the thermal half-life, and the sample could not be cooled due to DMSO freezing. e The value is not available, as the compound does not undergo thermal switching. f PSS was not determined for surface-adsorbed switches and motor. | ||||||
| 1 | 400 | —d | 79 ± 1 | 86 ± 1 | 2.8 ± 0.4 × 10−3 | 4.3 ± 0.6 min |
| 2 | 370 | 74:26 | 99 ± 3 | 101 ± 1 | 6.9 ± 1.4 × 10−6 | 28 ± 6 h |
| 3 | 400 | 50:50 | 97 ± 2 | 95 ± 2 | 8.2 ± 0.4 × 10−5 | 140 ± 5 min |
| 4 | 275 | 5:95 | —e | —e | —e | —e |
| 1·CB[7] | 400 | —d | 89 ± 1 | 87 ± 1 | 2.3 ± 0.1 × 10−3 | 5.1 ± 0.2 min |
| 1·CB[7] (SAM) | 400 | —f | 109 ± 19 | 86 ± 38 | 2.8 ± 0.8 × 10−3 | 4.1 ± 1.2 min |
| 2·CB[7] | 370 | n.d | 101 ± 1 | 101 ± 1 | 6.6 ± 0.6 × 10−6 | 29 ± 3 h |
| 2·CB[7] (SAM) | 370 | —f | 127 ± 14 | 101 ± 30 | 5.9 ± 1.7 × 10−6 | 33 ± 9 h |
| 3·CB[7] | 400 | n.d | 103 ± 1 | 95 ± 1 | 7.0 ± 0.3 × 10−5 | 164 ± 7 min |
| 3·CB[7] (SAM) | 400 | —f | 93 ± 52 | 80 ± 26 | 1.5 ± 0.8 × 10−5 | 76 ± 41 min |
| 4·CB[7] | 275 | 5:95 | —e | —e | —e | —e |
| 10 | 400 | 15:85 | 87 ± 1 | 87 ± 1 | 1.9 ± 0.4 × 10−3 | 6 ± 1 min |
| 11 | 370 | 23:77 | 104 ± 2 | 102 ± 2 | 3.7 ± 0.7 × 10−6 | 52 ± 10 h |
| 12 | 400 | 18:82 | 101 ± 1 | 96 ± 1 | 4.6 ± 0.2 × 10−5 | 257 ± 13 min |
| 13 | 275 | 1:99 | —e | —e | —e | —e |
All systems showed fully reversible switching and rods 1–3 along with complexes 1–3·CB[7] and precursors 10–12 also excellent fatigue resistance since almost no decomposition was detected after more than 10 irradiation cycles. In contrast, compounds 4, 4·CB[7], and 13 showed reduced stability, with photoswitching accompanied by evident photobleaching.
The following conclusions can be drawn from the experimentally determined kinetic parameters. The overcrowded alkene-based molecular motors 1, 1·CB[7], and 10 exhibited half-lives of approximately 5 minutes, whereas the azobenzene derivatives 2, 2·CB[7], and 11 showed significantly longer half-lives of about 1–2 days. In contrast, the diazocine-based systems 3, 3·CB[7], and 12 demonstrated intermediate half-lives in the range of 2–4 hours at 20 °C. Notably, the iodinated precursors 10–12 displayed longer half-lives than their corresponding derivatives 1–3 and their CB[7] complexes, where the iodine atom was replaced with an ethynyl-aryl-pyridinium moiety.
Importantly, the isomerization rates and activation enthalpies remained nearly unchanged upon complexation with CB[7], suggesting that the photoswitchable units are spatially decoupled from the CB[7] binding site and retain their intrinsic photochemical behavior.
In the next step, all four complexes 1–4·CB[7], along with CB[7] alone and 9·CB[7] as references, were adsorbed onto gold surfaces. Self-assembled monolayers (SAMs) composed of CB[7] were formed by immersing the gold substrates in a 0.5 mM aqueous solution for 20 hours. The complexes were subsequently adsorbed from 0.1 mM DMSO solutions over a period of 48 to 72 hours. Importantly, at these concentrations the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 CB[7] complexes remain fully stable in solution, and no measurable dissociation occurs during the adsorption process. All resulting samples were analyzed using contact angle goniometry, ellipsometry, PM-IRRAS, surface-enhanced Raman spectroscopy (SERS), UV-vis spectroscopy, and atomic force microscopy (AFM), with the results discussed in the following sections.
1 CB[7] complexes remain fully stable in solution, and no measurable dissociation occurs during the adsorption process. All resulting samples were analyzed using contact angle goniometry, ellipsometry, PM-IRRAS, surface-enhanced Raman spectroscopy (SERS), UV-vis spectroscopy, and atomic force microscopy (AFM), with the results discussed in the following sections.
Surface hydrophilicity was evaluated using water contact angle goniometry. Measured values ranged approximately between 40–50° (see SI for details), as summarized in Table 2. The observed increase in contact angle relative to the neat, freshly annealed gold substrate (14 ± 2°), as well as substrates immersed in DMSO (22 ± 3°), confirms the formation of an organic layer on the surface.
| Sample | Immer. time (h) | Contact angle (°) | Ellips. thickness (Å) | Layer thickness by AFM (Å) | Length of the guest (Å) | Theor. layer height h (Å) |
|---|---|---|---|---|---|---|
| 1·CB[7] | 72 | 50 ± 4 | 15 ± 2 | 23 ± 2 | 26 | 25 |
| 2·CB[7] | 48 | 45 ± 2 | 16 ± 1 | 24 ± 3 | 27 | 26 |
| 3·CB[7] | 48 | 40 ± 4 | 13 ± 2 | 15 ± 2 | 21 | 20 |
| 4·CB[7] | 48 | 47 ± 2 | 18 ± 1 | 18 ± 1 | 21 | 20 |
| 9·CB[7] | 48 | 40 ± 4 | 17 ± 2 | 20 ± 2 | 19 | 18 |
| CB[7] | 18 | 52 ± 4 | 11 ± 1 | 10 ± 1 | N/A | 9 |
Ellipsometry offered additional indirect evidence for the presence of surface-adsorbed molecular layers. Measurements indicated that complexes 1–4·CB[7] and 9·CB[7] formed films approximately 16 Å thick, whereas neat CB[7], likely oriented horizontally, yielded thinner films around 11 Å. These values are in reasonable agreement with theoretical predictions (Table 2), especially considering the limitations of ellipsometry.32 This technique averages measurements over relatively large surface areas, depends heavily on the assumed refractive index of the film, and is generally known to underestimate actual monolayer thicknesses.30
AFM scratching experiments enabled more precise determination of monolayer thicknesses (see Table 2). In this method, the AFM tip mechanically removed the adsorbed layer from a localized region of the gold substrate, allowing direct measurement of the height difference between the exposed surface and the surrounding intact monolayer. The measured thicknesses closely matched the theoretically predicted heights h, based on the expected molecular orientations shown in Fig. 4B. These data also reflected the relative lengths of the molecular rods: the longest complexes, 1–2·CB[7], formed films approximately 24 Å thick (theoretical ∼26 Å), while the shorter complexes 4·CB[7] and 9·CB[7] yielded films around 19 Å (theoretical ∼19 Å). Bare CB[7] formed the thinnest layer, ∼10 Å, consistent with the expected ∼9 Å for a horizontally aligned macrocycle (see SI for more details). The only notable discrepancy was observed for complex 3·CB[7], which consistently produced films of 15 ± 2 Å, roughly 5 Å shorter than the expected 20 Å. This deviation may result from additional bending of the molecule, potentially due to interactions between the nitrile group in the diazocine unit and the gold surface.
The presence of characteristic vibrational modes associated with specific functional groups (particularly the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C–N stretching vibrations) in 1–4·CB[7], 9·CB[7], and CB[7] was clearly confirmed by PM-IRRAS spectroscopy. In addition, samples containing 1·CB[7] and CB[7] were further analyzed using surface-enhanced Raman spectroscopy (SERS). Representative spectra of 1·CB[7] obtained by both PM-IRRAS and SERS are shown in Fig. 6, while data for the other samples are provided in the SI.
O and C–N stretching vibrations) in 1–4·CB[7], 9·CB[7], and CB[7] was clearly confirmed by PM-IRRAS spectroscopy. In addition, samples containing 1·CB[7] and CB[7] were further analyzed using surface-enhanced Raman spectroscopy (SERS). Representative spectra of 1·CB[7] obtained by both PM-IRRAS and SERS are shown in Fig. 6, while data for the other samples are provided in the SI.
The PM-IRRAS spectrum of 1·CB[7] adsorbed from DMSO displayed a prominent absorption bands near 1750 and 1470 cm−1, which were attributed to the carbonyl and C–N stretching vibrations of the CB[7] macrocycle (Fig. 5).21 Notably, vibrations typically associated with the guest molecule - such as the asymmetric stretching of the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C triple bond or the nitrile group, both expected around 2200 cm−1 based on isotropic KBr pellet spectra - were not detected. This absence can be attributed to the dominant intensity of CB[7] bands, which overshadow the comparatively weaker guest signals due to CB[7]'s high symmetry and the presence of 14 equivalent carbonyl groups. Nonetheless, the persistence of strong carbonyl bands characteristic of CB[7], combined with the known stability of CB[7] complexes in solution, supports the conclusion that 1·CB[7] is indeed present on the gold surface.
C triple bond or the nitrile group, both expected around 2200 cm−1 based on isotropic KBr pellet spectra - were not detected. This absence can be attributed to the dominant intensity of CB[7] bands, which overshadow the comparatively weaker guest signals due to CB[7]'s high symmetry and the presence of 14 equivalent carbonyl groups. Nonetheless, the persistence of strong carbonyl bands characteristic of CB[7], combined with the known stability of CB[7] complexes in solution, supports the conclusion that 1·CB[7] is indeed present on the gold surface.
Owing to the distinct selection rules of Raman spectroscopy, vibrational modes that appear strongly in IR spectra - such as the carbonyl stretching of CB[7] - are less intense or even suppressed in Raman spectra. This attenuation of CB[7] signals in the Raman spectrum enabled the detection of vibrational bands attributed to the guest molecule within the complex. Specifically, bands corresponding to the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C triple bond stretching (∼2200 cm−1) and aromatic ring stretching modes (1508 cm−1, 1570 cm−1, and 1604 cm−1) were clearly observed alongside weaker CB[7] bands in the Raman spectrum of powdered 1·CB[7] (Fig. 6). Additionally, the SERS of 1·CB[7] adsorbed onto a gold surface was acquired (Fig. 6), with distinct bands attributable to the complex. These observations further support the presence of 1·CB[7] on the gold surface.
C triple bond stretching (∼2200 cm−1) and aromatic ring stretching modes (1508 cm−1, 1570 cm−1, and 1604 cm−1) were clearly observed alongside weaker CB[7] bands in the Raman spectrum of powdered 1·CB[7] (Fig. 6). Additionally, the SERS of 1·CB[7] adsorbed onto a gold surface was acquired (Fig. 6), with distinct bands attributable to the complex. These observations further support the presence of 1·CB[7] on the gold surface.
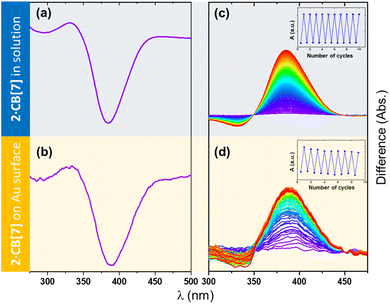
A central question was whether the surface-anchored complexes retain their ability to undergo photoisomerization. To address this, UV–vis absorption spectroscopy was carried out on SAMs of 1–4·CB[7] prepared on semi-transparent gold films (∼5 nm Au on silylated quartz).15,33,34 In all cases, irradiation induced the characteristic spectral changes associated with photoisomerization, followed by thermal and/or photoinduced relaxation (Fig. S51–S57). These surface-based differential spectra closely mirror the behavior observed in solution, where the photoisomerization is unambiguously confirmed by combined NMR and UV-vis spectroscopy. Fig. 7 illustrates a representative example for the azobenzene 2·CB[7] SAM. Because of the inherently low signal-to-noise ratio of surface UV-vis measurements, the data are presented as differential spectra collected before and after UV irradiation. Upon excitation at 400 ± 5 nm, the broad absorption band at ∼390 nm corresponding to π–π* transition decreases, yielding a negative differential feature consistent with trans-to-cis conversion (Fig. 7B). Subsequent thermal relaxation restores the original spectrum (Fig. 7D), in full agreement with the solution behavior (Fig. 7A and C).
Kinetic analysis performed for SAMs of 1–3·CB[7] (see SI for additional details) afforded half-lives comparable to those in solution (Table 1). For 3·CB[7], the surface spectra exhibit only modest amplitude changes, a consequence of its inherently lower molar absorptivities in both isomeric states. Nonetheless, the qualitative photoswitching behavior closely matches the solution data, supporting reliable surface photoisomerization.
To quantify the packing density, the surface coverage of the CB[7]-based SAMs was determined using two independent approaches: AFM counting of individual CB[7] units and UV-vis absorbance analysis (see SI). Both methods yielded consistent values of ca. 60% (±10–15%), in good agreement with the incomplete monolayer coverage typically reported for CB[7] on gold.23,35 The agreement between the two measurements supports the robustness of the SAM preparation and confirms reliable surface attachment of the photoactive complexes.
These observations not only provided further confirmation of the successful immobilization of the complexes on the gold surface but, more importantly, demonstrated the preserved photoactivity of the surface-bound layers. The results also indicate that the molecular anchor effectively decouples the photoresponsive unit from the gold substrate, thereby preventing quenching of the excited state and allowing efficient photoisomerization.
Conclusions
A series of four photoswitchable molecular rods (1–4) were synthesized and complexed with CB[7] to form supramolecular assemblies 1–4·CB[7]. The complexation was followed by 1H NMR spectroscopy. Photochemical studies revealed that the complexes retained the switching behavior of the parent compounds, with minimal impact from CB[7] encapsulation, indicating spatial decoupling of the photoactive core from the binding site.The complexes were immobilized on gold surfaces as self-assembled monolayers (SAMs) and characterized by ellipsometry, AFM, PM-IRRAS, and SERS, confirming their presence and structural integrity. Importantly, the SAMs of all four complexes 1–4·CB[7] exhibited fully reversible photoisomerization upon UV irradiation, clearly demonstrating preserved photoactivity on the surface. These results underline the effectiveness of the molecular architecture in enabling surface-confined photoswitching, offering a robust platform for photoresponsive functional interfaces.
Author contributions
D. L. carried out synthesis and characterization of molecular rods and complexes and performed UV-vis characterization of photoswitches in solution. E. K. performed AFM and UV-vis analysis of SAMs. K. M. V., C. S. H. and M. M. prepared and characterized SAMs. I. C. determined all single-crystal X-ray diffraction structures. J. K. conceived the project.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2457321 (compound 8) and 2457322 (compound 2) contain the supplementary crystallographic data for this paper.36a,bThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: synthetic procedures, copies of 1H and 13C NMR spectra of all new derivatives, UV-vis analysis of photoswitching and thermal steps in solution and within SAMs (1–4, 1–4·CB[7], 10–13), 1H NMR analysis of photoswitching (2–4 and 10–13), quantum yield determination (3 and 4), preparation of gold substrates, characterization of SAMs (AFM visualizations and scratching experiments, ellipsometry, contact angle goniometry, PM-IRRAS, Raman, photoswitching monitored by UV-vis), and ORTEP views of single molecule and packing in two crystal structures (2 and 8), are available in the Supporting Information file (PDF). Optimized geometries of 1–4·CB[7] (xyz). See DOI: https://doi.org/10.1039/d5sc09469k.
Acknowledgements
This work was supported by the Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic (RVO: 61388963), Czech Science Foundation (grant number: 25–16074S), and the Ministry of Education, Youth and Sports (grant number: LTAUSA19120). We are grateful to Dr Lucie Bednárová for help with IR spectra, Dr Anna Šemberová for help with Raman spectra, Dr Lukáš Severa for performing calculations, and Dr Jakub Radek Štoček and Dr Martin Dračínský for help with assigning NMR spectra. Finally, we are grateful to Prof. Eric Masson for providing the CB[7] macrocycle.References
- A. Goulet-Hanssens, F. Eisenreich and S. Hecht, Adv. Mater., 2020, 32(20), 1905966 CrossRef CAS PubMed.
- J. Kaleta, Molecular Switches and Motors in 2-D, in Molecular Photoswitches, ed. Z. L. Pianowski, Wiley-VCH GmbH, 2022 Search PubMed.
- R. Klajn, Pure Appl. Chem., 2010, 82, 2247–2279 Search PubMed.
- Y. Deng, G. Long, Y. Zhang, W. Zhao, G. Zhou, B. L. Feringa and J. Chen, Light:Sci. Appl., 2024, 13(1), 63 CrossRef CAS PubMed.
- H. Chen and J. F. Stoddart, Nat. Rev. Mater., 2021, 6, 804–828 CrossRef CAS.
- T. Li, V. K. Bandari and O. G. Schmidt, Adv. Mater., 2023, 35, 2209088 CrossRef CAS PubMed.
- M. Valášek and M. Mayor, Chem. Eur. J., 2017, 23, 13538–13548 CrossRef PubMed.
- K.-Y. Chen, O. Ivashenko, G. T. Carroll, J. Robertus, J. C. M. Kistemaker, G. London, W. R. Browne, P. Rudolf and B. L. Feringa, J. Am. Chem. Soc., 2014, 136, 3219–3224 CrossRef CAS PubMed.
- R. Löw, T. Rusch, F. Röhrlich, O. Magnussen and R. Herges, Beilstein J. Org.Chem., 2019, 15, 1485–1490 CrossRef PubMed.
- B. Baisch, D. Raffa, U. Jung, O. M. Magnussen, C. Nicolas, J. Lacour, J. Kubitschke and R. Herges, J. Am. Chem. Soc., 2009, 131, 442–443 Search PubMed.
- L. Severa, C. Santos Hurtado, I. Rončević, M. Mašát, G. Bastien, J. R. Štoček, M. Dračínský, V. Houska, E. Kaletová, D. J. Garza, I. Císařová, K. L. A. Cimatu, Z. Bastl and J. Kaleta, Chem. Eur. J., 2024, 30, e2023028 CrossRef PubMed.
- G. Bastien, L. Severa, M. Škuta, C. Santos Hurtado, J. Rybáček, V. Šolínová, I. Císařová, V. Kašička and J. Kaleta, Chem. Eur. J., 2024, 30, e202401889 Search PubMed.
- F. Ishiwari, G. Nascimbeni, H. Tago, Y. Shoji, S. Fujii, M. Kiguchi, T. Tada, M. Zharnikov, E. Zojer and T. Fukushima, J. Am. Chem. Soc., 2019, 141, 5995–6005 Search PubMed.
- S. Das, F. Ishiwari, Y. Shoji, T. Fukushima and M. Zharnikov, J. Phys. Chem. C, 2023, 127, 5178–5185 Search PubMed.
- K. Bezděková, L. Severa, E. Kaletová, K. Majerová Varga, M. Mašát, L.-T. Wu, J.-C. Jiang, I. Císařová and J. Kaleta, Angew. Chem., Int. Ed., 2025, 65, e202513922 Search PubMed.
- F. Tian, D. Jiao, F. Biedermann and O. A. Scherman, Nat. Commun., 2012, 3(1), 1207 Search PubMed.
- C. Santos Hurtado, G. Bastien, I. Rončević, M. Dračínský, E. Tortorici, C. Rogers, J. Michl and J. Kaleta, Chem. Comm., 2024, 60, 960–963 RSC.
- C. Santos Hurtado, G. Bastien, M. Mašát, J. R. Štoček, M. Dračínský, I. Rončević, I. Císařová, C. T. Rogers and J. Kaleta, J. Am. Chem. Soc., 2020, 142, 9337–9351 CrossRef CAS PubMed.
- J. Kaleta, J. Chen, G. Bastien, M. Dračínský, M. Mašát, C. T. Rogers, B. L. Feringa and J. Michl, J. Am. Chem. Soc., 2017, 139, 10486–10498 CrossRef CAS PubMed.
- K. Kim, W. S. Jeon, J.-K. Kang, J. W. Lee, S. Y. Jon, T. Kim and K. Kim, Angew. Chem., Int. Ed., 2003, 42(20), 2293–2296 CrossRef CAS PubMed.
- L. Qi, H. Tian, H. Shao and H.-Z. Yu, J. Phys. Chem. C, 2017, 121(14), 7985–7992 CrossRef CAS.
- A. Corma, H. García, P. Montes-Navajas, A. Primo, J. J. Calvino and S. Trasobares, Chem. - Eur. J., 2007, 13(22), 6359–6364 CrossRef CAS PubMed.
- Q. An, G. Li, C. Tao, Y. Li, Y. Wu and W. Zhang, Chem. Commun., 2008, 17, 1989 RSC.
- E. Blanco, C. Quintana, L. Hernández and P. Hernández, Electroanalysis, 2013, 25(1), 263–268 CrossRef CAS.
- A. Gomez-Casado, P. Jonkheijm and J. Huskens, Langmuir, 2011, 27(18), 11508–11513 CrossRef CAS PubMed.
- S. Liu, C. Ruspic, P. Mukhopadhyay, S. Chakrabarti, P. Y. Zavalij and L. Isaacs, J. Am. Chem. Soc., 2005, 127(45), 15959–15967 CrossRef CAS PubMed.
- D. Lončarić, F. Movahedifar, J. R. Štoček, M. Dračínský, J. Cvačka, G. Shanshan, B. Bythell, I. Císařová, E. Masson and J. Kaleta, Chem. Sci., 2023, 14, 9258–9266 RSC.
- S. Grimme, C. Bannwarth and P. J. Shushkov, Chem. Theory Comput., 2017, 13, 1989–2009 Search PubMed.
- C. Bannwarth, S. Ehlert and S. Grimme, J. Chem. Theory Comput., 2019, 15, 1652–1671 CrossRef CAS PubMed.
- S. Grimme, J. Chem. Theory Comput., 2019, 15, 2847–2862 CrossRef CAS PubMed.
- C. Santos Hurtado, G. Bastien, D. Lončarić, M. Dračínský, I. Císařová, E. Masson and J. Kaleta, Chem. Sci., 2025, 16, 14081–14087 Search PubMed.
- H. Fujiwara, Effect of Roughness on Ellipsometry Analysis, in Spectroscopic Ellipsometry for Photovoltaics, ed. Fujiwara H. and Collins R., Springer Series in Optical Sciences. Springer, Cham, 2018, 212 Search PubMed.
- J. Chen, K.-Y. Chen, G. T. Carroll and B. L. Feringa, Chem. Commun., 2014, 50, 12641–12644 RSC.
- K.-Y. Chen, S. J. Wezenberg, G. T. Carroll, G. London, J. C. M. Kistemaker, T. C. Pijper and B. L. Feringa, J. Org. Chem., 2014, 79, 7032–7040 CrossRef CAS PubMed.
- A. Gomez-Casado, P. Jonkheijm and J. Huskens, Langmuir, 2011, 27(18), 11508–11513 CrossRef CAS PubMed.
- (a) CCDC 2457321: Experimental Crystal Structrue Determination, 2026, DOI:10.5517/ccdc.csd.cc2nh1fn.; (b) CCDC 2457322: Experimental Crystal Structrue Determination, 2026, DOI:10.5517/ccdc.csd.cc2nh1gp.
| This journal is © The Royal Society of Chemistry 2026 |