Open Access Article
Open Access ArticleHigh brightness in bis(tri-isopropylsilyl)ethynyl-functionalized polycyclic aromatic hydrocarbons: localized representation versus Clar's model
Kais
Dhbaibi
 a,
Huong
Le Thi
ab,
Jérôme
Marrot
a,
Huong
Le Thi
ab,
Jérôme
Marrot
 a,
Masahiro
Hayakawa
a,
Masahiro
Hayakawa
 c,
Masashi
Mamada
c,
Masashi
Mamada
 c and
Michel
Frigoli
c and
Michel
Frigoli
 *a
*a
aUMR CNRS 8180, UVSQ, Institut Lavoisier de Versailles, Université Paris-Saclay, 45 Avenue des Etats-Unis, 78035 Versailles Cedex, France. E-mail: michel.frigoli@uvsq.fr
bDepartment of Advanced Materials Science and Nanotechnology, University of Science and Technology of Hanoi (USTH), Vietnam Academy of Science and Technology (VAST), 18 – Hoang Quoc Viet, Ha Noi 11307, Vietnam
cDepartment of Chemistry, Graduate School of Science, Kyoto University, Kitashirakawa Oiwake-cho, Sakyo-ku, Kyoto 606–8502, Japan
First published on 2nd February 2026
Abstract
Recent studies on acenes have highlighted a localized representation (LR) of aromatic sextets, challenging Clar's classical model of migrating sextets. Here, we extend this framework to peri-fused systems by investigating bis(tri-isopropylsilyl)ethynyl (TIPS)-functionalized polycyclic aromatic hydrocarbons (TIPS-PAHs) namely TIPS-anthanthrene (TIPS-ATT), TIPS-pyranthrene (TIPS-PYR) and TIPS-dibenzo[pyranthrene] (TIPS-DBPYR) as test beds for LR. Crystal structures provide direct evidence for determining the Fries canonical structures, with bond lengths and bond length alternation (BLA) at the zig-zag edge offering robust criteria for positioning aromatic sextets. While NICS and HOMA indices are broadly consistent with these assignments, their interpretation should be considered with some caution. Optical studies reveal progressive bathochromic shifts (491–599 nm) with increasing conjugation, but absorption strength and fluorescence quantum yield depend critically on the mode of π-extension: catacondensation enhances brightness, whereas linear extension diminishes it. Remarkably, TIPS-PYR combines a record molar absorption coefficient (>200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1) with a high fluorescence quantum yield (ΦF = 0.93), yielding a molar brightness comparable to giant nanoribbons. Stability assays show that TIPS-ATT and TIPS-PYR are bench stable, whereas TIPS-DBPYR, despite reduced fluorescence efficiency, exhibits enhanced photostability relative to TIPS-pentacene (TIPS-PEN) and satisfies the energetic criterion for singlet fission. These findings establish LR as a robust framework for describing peri-fused PAHs, identify TIPS-PYR as an exceptionally bright dye, and position TIPS-DBPYR as a promising singlet fission material.
000 M−1 cm−1) with a high fluorescence quantum yield (ΦF = 0.93), yielding a molar brightness comparable to giant nanoribbons. Stability assays show that TIPS-ATT and TIPS-PYR are bench stable, whereas TIPS-DBPYR, despite reduced fluorescence efficiency, exhibits enhanced photostability relative to TIPS-pentacene (TIPS-PEN) and satisfies the energetic criterion for singlet fission. These findings establish LR as a robust framework for describing peri-fused PAHs, identify TIPS-PYR as an exceptionally bright dye, and position TIPS-DBPYR as a promising singlet fission material.
Introduction
Over the past three decades, polycyclic aromatic hydrocarbons (PAHs) have been the focus of intensive investigation, spanning both fundamental questions of aromaticity and their emerging optoelectronic applications.1–13 This modern interest is based on century old foundations, from Scholl's pioneering synthetic strategies to Clar's influential structural and aromaticity models.14–16 Clar's sextet model, in particular, has long provided a powerful tool for rationalizing the interplay between aromatic stabilization, molecular stability and optical properties.16 For planar PAHs with a unique Clar structure such as phenanthrene, pyrene and triphenylene, the representation is straightforward (Fig. 1a).16,17 For systems describable by multiple Clar structures, Clar introduced the concept of migrating sextets, exemplified in acenes, chrysene, anthanthrene (ATT) (Fig. 1a) and related PAHs.16,18 Yet the case of acenes has recently challenged the classical Clar picture. We recently proposed the localized representation (LR) of acenes, in which the aromatic sextet is centred in the middle of the molecules (Fig. 1b).19 Accordingly, acenes can be viewed as symmetric linear extensions of benzene. The validity of this LR is supported by bond length alternation (BLA) analyses, which show that the zig-zag edges of acenes closely resemble those of ortho-quinoid acenothiophenes.19,21 The LR highlights that acenes are aromatic compounds with a 4n + 2 π-electron count along their periphery. Thus, despite their polycyclic structure, they can also be regarded as 4n + 2 annulenes, not only by virtue of the alternating single and double bonds along the zig-zag edge, but also because this connectivity gives rise to a predominantly global perimetric ring current, as evidenced by the anisotropy of the induced current density (ACID) plots.19 Complementary current density analyses based on the pseudo-π approach further indicate that, in addition to this dominant global current, acenes exhibit weaker (semi)local contributions.22,23 It should be emphasized that unlike the classical representation of acenes based on a single Kekulé structure,24,25 which no longer provides an adequate account of their ring current behaviour, the LR systematically incorporates all these components of the 4n + 2 diatropic ring current (see the SI).26 Moreover, resonance energy data derived from experimental heats of formation, which increase nearly linearly with ring number, underscore that stabilization is not fully delocalized across the backbone but instead reflects localized contributions.27 In this way, the LR reconciles bond length alternation with energetic resonance stabilization and with both global and subsidiary current density contributions, characteristic of acenes.28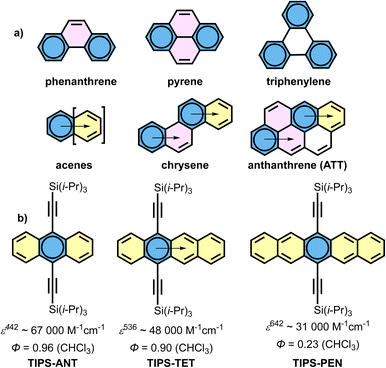 | ||
| Fig. 1 (a) Clar representation of the selected PAHs; (b) localised representation (LR)19 and optical properties of TIPS-acenes in chloroform solution.20 | ||
Acenes represent a particularly remarkable family of PAHs as each additional fused ring containing a cis-diene induces an exceptionally large bathochromic shift of nearly 100 nm in the absorption maximum. For instance, when functionalized with TIPS-ethynyl groups at the central ring, the absorption maxima are 442 nm for TIPS-anthracene (TIPS-ANT), 536 nm for TIPS-tetracene (TIPS-TET) and 642 nm for TIPS-pentacene (TIPS-PEN) in diluted chloroform solution (Fig. 1b).20 However, both the molar absorption coefficient (ε) and the fluorescence quantum yield (ΦF) decrease with increasing ring number, correlating with a progressive loss of aromatic stabilization from the dilution of the central aromatic sextet by the successive addition of cis-diene fragments (Fig. 1b). This pattern is also observed in the [m,2]peri-acenoacene family developed by Wu and coworkers, where ε and ΦF decrease as the molecular structure becomes more laterally extended.29 In this series, although the substituents are not located in the same position and therefore they are not strictly comparable, ε decreases roughly from 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 for mesityl-anthanthrene (Mes-ATT) to 35
000 M−1 cm−1 for mesityl-anthanthrene (Mes-ATT) to 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 for peri-pentacenopentacene (PPP) and ΦF decreases from 56 to 32%, respectively. Recently, we synthesized TIPS-PPP, which exhibits a 26-fold increase in stability compared to TIPS-PEN,28 maintaining the same 2D π–π brickwork molecular packing and a 6-fold increase in charge transport in organic field-effect transistor (OFET) devices.19,30 With absorption at 726 nm and a ε of 85
000 M−1 cm−1 for peri-pentacenopentacene (PPP) and ΦF decreases from 56 to 32%, respectively. Recently, we synthesized TIPS-PPP, which exhibits a 26-fold increase in stability compared to TIPS-PEN,28 maintaining the same 2D π–π brickwork molecular packing and a 6-fold increase in charge transport in organic field-effect transistor (OFET) devices.19,30 With absorption at 726 nm and a ε of 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1, the optical properties of TIPS-PPP suggest that TIPS-ATT, a smaller analogue, may have a ε exceeding 100
000 M−1 cm−1, the optical properties of TIPS-PPP suggest that TIPS-ATT, a smaller analogue, may have a ε exceeding 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1, a remarkable value for a small PAH (Fig. 2). The optical properties of TIPS-ATT, beyond its absorption spectrum, remain unknown despite its use as a donor in OPV devices and as an active layer in OFETs.31,32 Even more striking, Morin and coworkers recently reported dibenzo[a,l]peri-tetracenotetracene derivatives (DBPTT) with an ε of 168
000 M−1 cm−1, a remarkable value for a small PAH (Fig. 2). The optical properties of TIPS-ATT, beyond its absorption spectrum, remain unknown despite its use as a donor in OPV devices and as an active layer in OFETs.31,32 Even more striking, Morin and coworkers recently reported dibenzo[a,l]peri-tetracenotetracene derivatives (DBPTT) with an ε of 168![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 at 611 nm when functionalized with TIPS groups, although fluorescence data were not reported.33 This finding suggests that TIPS-pyranthrene (TIPS-PYR), a smaller analogue, could possess an even stronger ε, making it a very high brightness dye if combined with high fluorescence quantum yield (Fig. 2).31
000 M−1 cm−1 at 611 nm when functionalized with TIPS groups, although fluorescence data were not reported.33 This finding suggests that TIPS-pyranthrene (TIPS-PYR), a smaller analogue, could possess an even stronger ε, making it a very high brightness dye if combined with high fluorescence quantum yield (Fig. 2).31
 | ||
| Fig. 2 Localized representations of the molecules investigated in this study and their associated optical properties. | ||
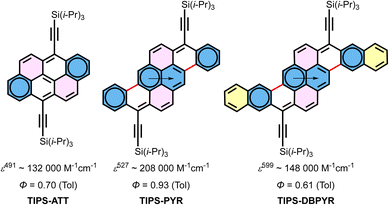
In this context, TIPS-ATT and TIPS-PYR, the latter of which can be regarded as cata-extensions of the ATT core along the a,j faces, emerge as particularly valuable cases due to their multiple Clar structures, making them ideal test beds for the localized representation (LR). To further extend this framework, we also investigated TIPS-dibenzo[pyranthrene] (TIPS-DBPYR), a lateral expansion of the PYR core (Fig. 2), in order to elucidate the impact of cata versus linear extension in PAHs on their optoelectronic properties. This study reports their synthesis, optoelectronic properties, stability, and crystal structures. The latter were analysed in depth using structural parameters such as bond lengths and bond length alternation (BLA), along with “aromaticity indices” including nucleus-independent chemical shift (NICS)34 and the harmonic oscillator model of aromaticity (HOMA).35
Results and discussions
Synthesis
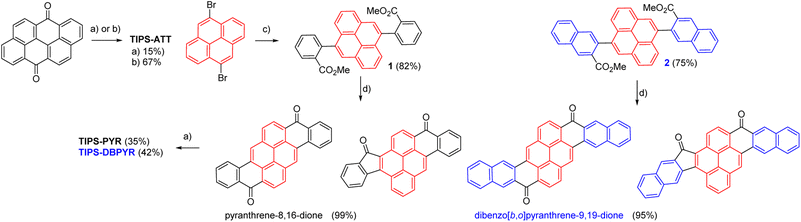
TIPS-ATT can be synthesized in a single step from anthanthrone (Scheme 1).30 Using the classical lithium salt of TIPS, the yield was limited to 15%, reflecting the well-known competition between 1,2- and 1,4-addition pathways on anthanthrone.36 To favour the desired 1,2-addition, we carried out the reaction in the presence of LaCl3 LiCl, as previously applied in the synthesis of TIPS-PPP, which significantly improved the yield.19 Pyranthrone is a commercially available product that can be synthesized by two main methods: the first starts with 2,2′-dimethyl-[1,1′-bianthracene]-9,9′,10,10′-tetraone as the starting material and the second starts with pyrene-1,6-diylbis(phenylmethanone), which then undergoes a Scholl reaction. In the Scholl reaction, when starting from pyrene-1,6-diylbis(naphthalen-2-ylmethanone), cyclization occurs specifically at position 1 of the naphthalene ring.37 Consequently, none of these methods is suitable to obtain the extended version of pyranthrone. Given that the first two Friedel–Crafts acylations of pyrene occur at positions 1 and 6, compounds 1 and 2 emerged as convenient precursors for obtaining large diones (Scheme 1). These two compounds were obtained in good yield through a Suzuki reaction between 5,10-dibromopyrene and the corresponding boronic esters.38 The double intramolecular Friedel–Crafts acylation/cyclization using triflic acid as an activator proceeded smoothly.39–42 However, infrared spectra of both insoluble compounds revealed the presence of two carbonyl stretches: more intense bands at 1641 and 1648 cm−1, characteristic of a six-membered ring (6-MR) aromatic ketone, and less intense bands at 1704 and 1691 cm−1, respectively, indicative of a five-membered ring (5-MR) aromatic ketone (see the SI). Despite this, the two targeted diones having two 6-MRs are likely the major products, as the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretch of 5-MR is typically much more intense compared to that of 6-MR (see the SI).38 The regioselectivity reflects a competition between electronic and steric factors. The intrinsic reactivity of the 1,6-positions in pyrene rationalizes their role as the preferred electrophilic sites, even though they are also the most sterically hindered. Following the first cyclization at C1, the electrophilic susceptibility of C6 is attenuated, thereby shifting partial activation toward C4, which represents the most sterically accessible alternative site. Addition of TIPS-acetylide to either the pyranthrone or dibenzopyranthrone mixture afforded TIPS-PYR and TIPS-DBPYR, respectively, in moderate yields. The structures of all three compounds were unambiguously confirmed by NMR spectroscopy, high-resolution mass spectrometry (HRMS) and single crystal X-ray crystallography (see the SI).
O stretch of 5-MR is typically much more intense compared to that of 6-MR (see the SI).38 The regioselectivity reflects a competition between electronic and steric factors. The intrinsic reactivity of the 1,6-positions in pyrene rationalizes their role as the preferred electrophilic sites, even though they are also the most sterically hindered. Following the first cyclization at C1, the electrophilic susceptibility of C6 is attenuated, thereby shifting partial activation toward C4, which represents the most sterically accessible alternative site. Addition of TIPS-acetylide to either the pyranthrone or dibenzopyranthrone mixture afforded TIPS-PYR and TIPS-DBPYR, respectively, in moderate yields. The structures of all three compounds were unambiguously confirmed by NMR spectroscopy, high-resolution mass spectrometry (HRMS) and single crystal X-ray crystallography (see the SI).
X-ray crystallographic analysis and localised representation
The crystal structures of TIPS-ATT and TIPS-PYR have already been determined.29,30 However, the reliability factor (R-factor) exceeds 0.1, precluding meaningful discussion of bond lengths. Attempts to obtain higher quality crystals of TIPS-ATT were unsuccessful, whereas crystals of sufficient quality were obtained for TIPS-PYR (R = 0.060) and TIPS-DBPYR (R = 0.057), enabling reliable structural analysis. For structural comparison, we therefore refer to the crystal structures of Mesityl-ATT and the parent ATT.27,43Since the LR of acenes has been rationalized by analogy with the bond lengths observed in both acenes and acenothiophenes,19 we begin with pyrene, phenanthrene and chrysene as reference systems, together with naphthalene (Fig. 3). In the crystal structures, the bonds depicted in blue are shorter than their neighbours. By analogy with acenes, these shortened bonds can be assigned as formal double bonds, thereby giving rise to a specific Kekulé resonance structure (Fig. 3 and 4). This latter corresponds to the Fries structure, in which the maximum number of rings share a double bond at the fusion and can therefore be formally represented as having six internal π-electrons.44,45 Consequently, the preferred representation of PAHs using single and double bonds should be the Fries structure, since it corresponds most closely to the crystallographic data. While Graovac et al. introduced the Kekulé index and suggested that the Fries structure has the dominant contribution in the ground state,46 the Fries structure of naphthalene likely accounts for 50% of the ground state. This is because the rings of naphthalene can be regarded as a resonance hybrid between a delocalized aromatic sextet and a cis-diene, in agreement with the Clar model (Fig. 3). This interpretation is supported by bond length alternation analysis: the peripheral BLA of naphthalene corresponds to the average of an aromatic sextet (0) and a cis-diene (0.074, as in anthracene), yielding a value of 0.037, which is in close agreement with the observed BLA of 0.043.19 It should be noted that Fries assumed, in the case of anthracene, that the oscillation of double bonds was confined to the central ring while the outer rings retained fixed double bonds.43 This arrangement corresponds directly to the LR of anthracene (Fig. 1).
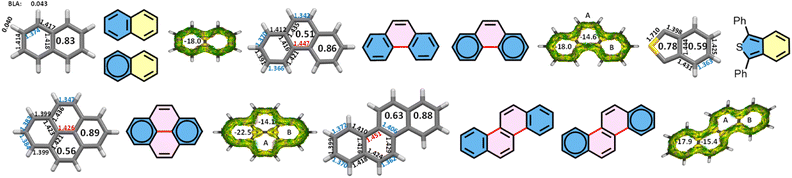 | ||
| Fig. 3 Crystal structures of naphthalene,47 pyrene,48 phenanthrene,49 chrysene50 and 1,3-diphenylbenzo[c]thiophene21 (phenyl rings removed) with bond lengths and HOMA values inside the rings. The deduced Fries structures and the corresponding Clar structures and LR for chrysene. ACID plots and NICS(1.7)πzz inside the rings of molecules with optimized structures. | ||
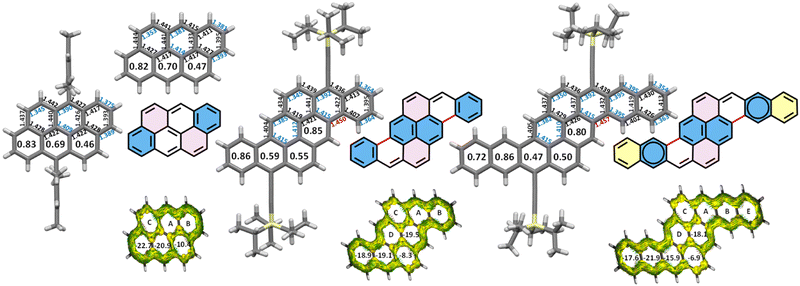 | ||
| Fig. 4 Crystal structures of Mes-ATT,27ATT,43TIPS-PYR and TIPS-DBPYR with bond lengths and HOMA values inside rings, deduced Fries structures and ACID plots and NICS(1.7)πzz inside the rings of parent PAHs with optimized structures. | ||
Pyrene and phenanthrene each possess a single Clar structure, whereas chrysene has three. For pyrene and phenanthrene, there is broad consensus that the Clar structure with two aromatic sextets located in the terminal rings provides the most accurate description.18 Pyrene belongs to the D2h point group, and as a consequence, the bond lengths in the outer rings B are inherently delocalized. This delocalization makes the Clar structure particularly evident: the outer rings B are represented as aromatic sextets, while ring A contains a localized double bond of 1.347 Å. The bond length distribution thus provides direct structural support for the Clar structure of pyrene. Accordingly, HOMA and NICS values are highest for rings B. Notably, the single bond highlighted in red is as short as 1.426 Å. Pyrene, with 16 π-electrons (4n), exhibits a strong global diatropic ring current (clockwise) of 14 π-electrons (4n + 2), together with two weaker local ring currents of 6 π-electrons, each around rings B, as revealed by the acid plot.
For phenanthrene, the Fries structure can be deduced from the alternating lengths of the external bonds in rings B. These bond lengths are very similar to those of naphthalene, with the notable exception of the C2–C3 bond, which measures 1.391 Å in phenanthrene versus 1.414 Å in naphthalene (Fig. 3), resulting in a slightly higher HOMA value. Owing to this shorter bond, three adjacent bonds within rings B fall within a narrow range (1.366–1.391 Å; BLA = 0.025 Å), lower than that observed in naphthalene (BLA = 0.040 Å). Since naphthalene is represented as a resonance hybrid between a delocalized aromatic sextet and a cis-diene (Fig. 3), rings B of phenanthrene are most appropriately described as delocalized aromatic sextets. The outer bond lengths of rings A, which closely resemble those observed in pyrene, further support the placement of the aromatic sextet in rings B. As in pyrene, the NICS and HOMA values for rings B corresponding to the aromatic sextet are higher than those for rings A. The red bond connecting the two phenyl rings measures 1.447 Å and a comparison with the corresponding bond in pyrene (1.426 Å) illustrates how single bond lengths are strongly modulated by their molecular environment, just as the lengths of single and double bonds are modulated in acenes.19 Phenanthrene, with 14 π-electrons, sustains a global diatropic ring current of 14 π-electrons in the ACID plot.
Chrysene can be analyzed in the same way. The bond lengths in rings B closely resemble those in phenanthrene, thereby supporting the placement of the aromatic sextets in the terminal rings, as confirmed by comparable HOMA and NICS values. This interpretation is further reinforced by the single bond connecting rings B and A (highlighted in red, 1.451 Å), which is nearly identical to the corresponding bond in phenanthrene (1.447 Å) and consistent with their cata-condensed arrangement. Moreover, while the BLA of the outer bonds in rings B is slightly lower than in phenanthrene, leading to a higher HOMA value, similar outer bond lengths are also observed in the 6-MR of benzo[c]thiophene (Fig. 3).21 Accordingly, chrysene can be represented as one LR. While phenanthrene and pyrene can be considered as biphenyls linked by one or two ethenyl spacers, respectively, chrysene can be considered a trans-stilbene with two ethenyl bridges. Chrysene, with 18 π-electrons, shows a global ring current of 18 π-electrons in the ACID plot.
The Clar representation of ATT already indicated that rings C contain localized bonds since Clar originally regarded ATT as an extension of pyrene (Fig. 1a).16 This assignment is consistent with the highest BLA at the periphery and the lowest HOMA and NICS values observed for rings C (Fig. 4). However, as discussed above, smaller cata-condensed PAHs are more appropriately described as arenes bridged by two ethenyl spacers, in line with the general tendency of nature to favour symmetry. From this perspective, ATT should therefore be regarded not as an extension of pyrene but rather as an extension of chrysene. Accordingly, the LR of ATT can be directly deduced from that of chrysene and both HOMA and NICS values fully support this assignment. More specifically, the BLA of the outer bonds in rings B is similar to or lower than that observed in chrysene with both molecules sharing the same symmetry. In contrast, the reduced HOMA values originate from relatively long intra-annular bonds, in comparison to pyrene, which exhibits the same structural motif. Nevertheless, the NICS value of ring B remains comparable to that of pyrene, consistent with its designation as an aromatic sextet within an identical environment of two neighbouring rings. The BLA at the periphery of rings A is significantly lower than in chrysene, reflecting their central position and the resulting higher degree of conjugation, as observed in acenes.19 Nevertheless, the BLA of rings A (0.034–0.037 Å) remains higher than that of the second ring of pentacene (0.026 Å), indicating that the bonds in rings A are localized. Owing to this relatively low BLA and the fact that each ring A is fused to three different neighbours, their NICS values are higher than those of pyrene, in which the corresponding rings are fused to only two different neighbours. Although ATT is an aromatic compound with 22 π electrons, the ACID plot reveals two distinct 18 π-electron ring current circuits when only the symmetry allowed pathways are considered: one following the periphery and the other bifurcated, corresponding to the chrysene subunit of the π-structure. This dual circuit confirms that ATT is best described as an extended version of chrysene. The chrysene contribution is further evident in the shape of the HOMO orbital (see the SI).
Considering all of the above, the Fries structure of TIPS-PYR together with its molecular symmetry and the structural parameters at the zig-zag edge (bond lengths and BLA values) and HOMA index, places the aromatic sextets on rings B and D (Fig. 2). Although the HOMA and NICS values of rings B are comparable to or higher than those of phenanthrene, which correspond to a phenanthrene-type aromatic sextet, the NICS values of rings A are very similar to those of rings B and could be misinterpreted as a migration of the sextet between rings A and B. This apparent “magnetic delocalisation” results from the local environment of ring A: its three non-equivalent neighbours reinforce its local magnetic susceptibility. The combined evidence from the structure (bond lengths, BLA and HOMA) therefore continues to support the LR with sextets localized on rings B and D (Fig. 2). Moreover, PYR can be regarded as an extended fulminene ([6]-phenacene): a fulminene core bridged by two ethenyl spacers. PYR is an aromatic system with 30 π-electrons (4n + 2), but the ACID plot reveals two distinct 26 π-electron ring current circuits: one delocalized along the periphery and another bifurcated pathway around the fulminene subunit. Moreover, fulminene itself can be seen as a 2,6-diphenylnaphthalene bridged by two ethenyl spacers.18
In the case of TIPS-DBPYR, all structural parameters (bond lengths and BLA) and “aromaticity indices” (HOMA and NICS) consistently place the aromatic sextets on rings B and D. Thus, the LR of TIPS-DBPYR can be seen in Fig. 2.
Ultimately, crystal structures provide a direct means of identifying the Fries structures. Bond lengths and bond length alternation (BLA) analysis at the zig-zag edge offer a robust criterion for positioning aromatic sextets. In the present series, NICS and HOMA values are largely consistent with these structural attributions, except in the case of PYR, where NICS overestimates the local aromaticity of a ring due to the high number of neighboring rings. HOMA may likewise underestimate local aromaticity when internal bonds are elongated by conjugation, as observed for the central ring in pentacene.51 These discrepancies highlight the need to interpret local aromaticity indices with caution, and the placement of aromatic sextets cannot be deduced from a single criterion such as NICS or HOMA in molecules that possess more than one Clar structure.
It should be noted that in phenanthrene, chrysene and anthanthrene, electrophilic substitution occurs preferentially at the central rings bearing double bonds, whereas in pyrene and in the acenes, the most reactive sites correspond to the rings hosting aromatic sextets.52,53 This demonstrates that the reactivity of PAHs cannot be rationalized solely in terms of their ground state electronic structures. While the computed thermodynamic stability of PAH adduct intermediates and the average local ionization energy (ALIE) provide a reasonable prediction of reactivity,51 the calculated spin density distribution of the triplet state (the singlet state being inaccessible for closed-shell systems) may serve as an even more reliable predictor of reactivity, as it likely resembles the electronic structure of the transition state.54 For ATT, PYR and DBPYR, the most reactive site is ring A, which bears the highest spin density at the zig-zag edge, as illustrated in Fig. 5. The Clar structure of the diradical is in good agreement with these calculations.
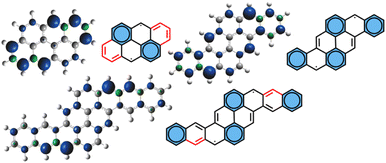 | ||
| Fig. 5 Triplet spin density and Clar structures showing the main delocalization along red highlighted bonds. | ||
Electrooptical properties, stability and theoretical calculations
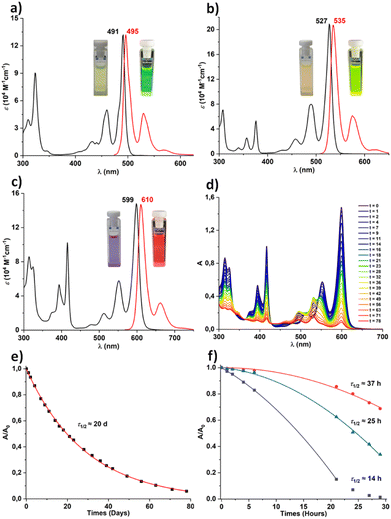
The optical properties and stability were investigated in toluene solution (Fig. 6 and Table 1). The smaller analogue, TIPS-ATT, exhibits an absorption maximum at 491 nm, while TIPS-PYR is red shifted by 36 nm to 527 nm. A further linear extension from TIPS-PYR to TIPS-DBPYR produces an even more pronounced red shift of 72 nm, yielding an absorption maximum at 599 nm. Thus, each additional ring induces a red shift of approximately 36 nm, in sharp contrast to the ∼100 nm increments typically observed in acenes. These progressive bathochromic shifts arise from the concomitant destabilization of the HOMO and stabilization of the LUMO, amounting to 0.09 eV from TIPS-ATT to TIPS-PYR and 0.15 eV from TIPS-PYR to TIPS-DBPYR, thereby narrowing the frontier orbital gap (Table 1). The calculated HOMO–LUMO gaps for the three molecules are in very good agreement with the optical band gap (Eoptg) obtained from the absorption onset using the tangent method (Table 1). As expected, TIPS-ATT exhibits a higher ε than the laterally extended TIPS-PPP (85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1), reaching a remarkable value of 132
000 M−1 cm−1), reaching a remarkable value of 132![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1. Even more striking, TIPS-PYR displays an ε exceeding 200
000 M−1 cm−1. Even more striking, TIPS-PYR displays an ε exceeding 200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1 underscoring the exceptional light harvesting capability of this system. In contrast, lateral extension of the PYR core to TIPS-DBPYR reduces ε to 148
000 M−1 cm−1 underscoring the exceptional light harvesting capability of this system. In contrast, lateral extension of the PYR core to TIPS-DBPYR reduces ε to 148![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1, highlighting the sensitivity of absorption strength to the mode of π-extension (cata vs. linear). Within this series, the fraction of rings that can be described as Clar sextets increases upon cata extension (2/6 in ATTvs. 3/8 in PYR) correlating with the observed enhancement in ε, whereas this fraction decreases with the introduction of cis-diene units upon linear extension (3/8 PYRvs. 3/10 DBPYR), which in turn correlates with a reduction in ε. TD-DFT calculations of these allowed S0 → S1 transitions reproduce this trend, with TIPS-PYR exhibiting the highest oscillator strength (f = 1.80), while TIPS-ATT shows the lowest (f = 0.98). In relative terms, however, the calculated intermediate f value of 1.62 for TIPS-DBPYR appears to be significantly overestimated compared to the experimental value. Among the three compounds, TIPS-PYR shows the highest absolute quantum yield of fluorescence (ΦF = 0.93) in deoxygenated solution, while TIPS-DBPYR has the lowest (ΦF = 0.61). By contrast, TIPS-ATT has an intermediate value (ΦF = 0.70). Notably, the formal molar fluorescence brightness of TIPS-PYR (εΦF = 193
000 M−1 cm−1, highlighting the sensitivity of absorption strength to the mode of π-extension (cata vs. linear). Within this series, the fraction of rings that can be described as Clar sextets increases upon cata extension (2/6 in ATTvs. 3/8 in PYR) correlating with the observed enhancement in ε, whereas this fraction decreases with the introduction of cis-diene units upon linear extension (3/8 PYRvs. 3/10 DBPYR), which in turn correlates with a reduction in ε. TD-DFT calculations of these allowed S0 → S1 transitions reproduce this trend, with TIPS-PYR exhibiting the highest oscillator strength (f = 1.80), while TIPS-ATT shows the lowest (f = 0.98). In relative terms, however, the calculated intermediate f value of 1.62 for TIPS-DBPYR appears to be significantly overestimated compared to the experimental value. Among the three compounds, TIPS-PYR shows the highest absolute quantum yield of fluorescence (ΦF = 0.93) in deoxygenated solution, while TIPS-DBPYR has the lowest (ΦF = 0.61). By contrast, TIPS-ATT has an intermediate value (ΦF = 0.70). Notably, the formal molar fluorescence brightness of TIPS-PYR (εΦF = 193![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1) is comparable to that reported for giant nanoribbons (GNRs).55TIPS-PYR thus stands out as one of the brightest dyes among polybenzenoid compounds. By contrast, TIPS-ATT and TIPS-PYR display εΦF values of approximately 90
000 M−1 cm−1) is comparable to that reported for giant nanoribbons (GNRs).55TIPS-PYR thus stands out as one of the brightest dyes among polybenzenoid compounds. By contrast, TIPS-ATT and TIPS-PYR display εΦF values of approximately 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1, which are comparable to those of well-established fluorescent benchmarks such as pyrromethene dyes and perylenediimides.56,57 It should be noted that peri-acenoacenes generally display lower ΦF values compared to their acene counterparts. TIPS-ANT exhibits a ΦF of 0.96, whereas TIPS-ATT shows a reduced value of 0.70 and similarly TIPS-PEN (ΦF = 0.44) is reduced relative to TIPS-PPP (ΦF = 0.27) in diluted toluene solution.19 The reduction of ΦF for TIPS-ATT can be attributed to the competition between fluorescence and intersystem crossing (ISC) from the first excited singlet state (S1), a process that has previously been demonstrated in ATT derivatives.58,59 For all compounds, the fluorescence quantum yield (ΦF) increased under deoxygenated conditions, indicating the quenching of S1 by molecular oxygen (3O2).60 This effect was most pronounced for TIPS-DBPYR, which can be ascribed to its comparatively smaller radiative decay rate constant and larger molecular size (Table 1).60 As recently demonstrated for perylenediimides, all compounds studied here are intrinsically capable of producing singlet oxygen (1O2) from both S1 and the first triplet excited state (T1).60 From S1, this process is energetically allowed since the gap ΔE = E(S1)–E(T1) exceeds 0.976 eV, while from T1, it is enabled because E(T1) lies above the excitation energy of 3O2 → 1O2 (0.976 eV) (Table 1). In other words, under illumination, these molecules act as photosensitizers, generating reactive 1O2 with TIPS-DBPYR likely being the most efficient. Moreover, according to the calculation, energetically, only TIPS-DBPYR satisfies the singlet fission criterion with E(S1) ≥ 2E(T1) providing a near isoenergetic driving force for the formation of the correlated triplet pair. Given that TIPS-DBPYR also exhibits markedly greater photostability than TIPS-PEN (vide infra), it emerges as a particularly promising candidate for singlet fission applications.61
000 M−1 cm−1, which are comparable to those of well-established fluorescent benchmarks such as pyrromethene dyes and perylenediimides.56,57 It should be noted that peri-acenoacenes generally display lower ΦF values compared to their acene counterparts. TIPS-ANT exhibits a ΦF of 0.96, whereas TIPS-ATT shows a reduced value of 0.70 and similarly TIPS-PEN (ΦF = 0.44) is reduced relative to TIPS-PPP (ΦF = 0.27) in diluted toluene solution.19 The reduction of ΦF for TIPS-ATT can be attributed to the competition between fluorescence and intersystem crossing (ISC) from the first excited singlet state (S1), a process that has previously been demonstrated in ATT derivatives.58,59 For all compounds, the fluorescence quantum yield (ΦF) increased under deoxygenated conditions, indicating the quenching of S1 by molecular oxygen (3O2).60 This effect was most pronounced for TIPS-DBPYR, which can be ascribed to its comparatively smaller radiative decay rate constant and larger molecular size (Table 1).60 As recently demonstrated for perylenediimides, all compounds studied here are intrinsically capable of producing singlet oxygen (1O2) from both S1 and the first triplet excited state (T1).60 From S1, this process is energetically allowed since the gap ΔE = E(S1)–E(T1) exceeds 0.976 eV, while from T1, it is enabled because E(T1) lies above the excitation energy of 3O2 → 1O2 (0.976 eV) (Table 1). In other words, under illumination, these molecules act as photosensitizers, generating reactive 1O2 with TIPS-DBPYR likely being the most efficient. Moreover, according to the calculation, energetically, only TIPS-DBPYR satisfies the singlet fission criterion with E(S1) ≥ 2E(T1) providing a near isoenergetic driving force for the formation of the correlated triplet pair. Given that TIPS-DBPYR also exhibits markedly greater photostability than TIPS-PEN (vide infra), it emerges as a particularly promising candidate for singlet fission applications.61
| Compounds | TIPS-ATT | TIPS-PYR | TIPS-DBPYR |
|---|---|---|---|
| a Absorption maximum. b Molar absorption coefficient. c Emission maximum. d Quantum yield of fluorescence determined in air and under N2. e Molar fluorescence brightness calculated under N2. f Fluorescence lifetime. g Radiative rate constant calculated as kr = ΦF/τF. h Non-radiative rate constant calculated as knr = (1 – ΦF)/τF. i Optical band gap estimated from absorption onset (tangent). j Deduced from the intersection between absorption and emission spectra. k TD-DFT calculation at the M062X/6-311+G(d,p) level using optimized geometry calculated at the B3LYP/6311 + G(d,p) level. l Oscillator strength. m Calculated at the B3LYP/6311 + G(d,p) level. n Biradical character calculated at the BS-UHF/6-31G(d,p) level. | |||
| λ max [nm]a | 491 | 527 | 599 |
| ε max [M−1 cm−1]b | 132![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
208![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
148![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
| ε 365 [M−1 cm−1]b | 1000 | 7100 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
| λ em [nm]c | 495 | 535 | 610 |
| Φ F (air)/ΦF (N2) [%]d | 0.64/0.70 | 0.86/0.93 | 0.51/0.61 |
| Φ F × ε max (N2)e | 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
193![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
| τ F (air)/τF (N2) [ns]f | 2.37/2.65 | 2.20/2.52 | 5.34/6.24 |
| k r (N2) [s−1] g | 2.64 × 108 | 3.69 × 108 | 9.77 × 107 |
| k nr (N2) [s−1]h | 1.13 × 108 | 2.06 × 107 | 6.25 × 107 |
| E g opt | 2.48 (499) | 2.30 (538) | 2.02 (614) |
| E(S1) [eV] (nm)j | 2.51 (493) | 2.33 (531) | 2.05 (604) |
| E(S1) [eV] (nm)k | 2.58 (480) | 2.40 (517) | 2.10 (591) |
| f , | 0.98 | 1.80 | 1.62 |
| E(T1) [eV] (nm)k | 1.35 (918) | 1.29 (961) | 1.03 (1204) |
| ΔE([S1]–[T1]) [eV]k | 1.23 | 1.11 | 1.07 |
| HOMO/LUMO [eV]l | −5.15/−2.67 | −5.06/−2.76 | −4.91/−2.91 |
| H–L gap [eV] (nm)m | 2.48 (500) | 2.30 (540) | 2.00 (620) |
| y 0 | 0.30 | 0.34 | 0.42 |
Stability tests were performed in toluene solution ([C] = 1 × 10−5 M) under ambient laboratory conditions with permanent light exposure. The stability studies reveal that while TIPS-ATT and TIPS-PYR remain intact under these conditions, TIPS-DBPYR undergoes slow degradation with a half-life (τ1/2) of 20 days (Fig. 6d and e and SI). The monoexponential decay at 599 nm confirms a first-order kinetic process, excluding the involvement of secondary degradation pathways that could accelerate 1O2 generation. It should be noted that TIPS-DBPYR is 20 times more stable than TIPS-PEN (τ1/2 = 1.4 days) studied under the same conditions.19 As illustrated in Fig. 6d, the original absorption bands of TIPS-DBPYR progressively decrease, while new bands emerge at 531, 495, and 462 nm. The intensities of these new bands grow steadily up to 28 days before subsequently declining. The progressive decrease of the three bands in their relative intensities with energy is characteristic of acene-like chromophores. The new spectral features are tentatively attributed to endoperoxide formation localized on one of the aromatic sextets (rings B) (see the SI). TD-DFT calculations on this degradation product further support this assignment (see the SI). By contrast, exposure to two 6 W bench lamps at 365 nm resulted in degradation of all compounds (Fig. 6f and SI). The decay monitored at each compound's λmax could well be fitted with a quadratic function, indicating an acceleration of the degradation process under these conditions (Fig. 5e). This can result from the formation of ketone-based photoproducts, as previously shown in TIPS-PPP.19 Among the series, TIPS-ATT proved to be the most robust (τ1/2 = 37 h), TIPS-DBPYR was the least stable (τ1/2 = 14 h) and TIPS-PYR exhibited intermediate stability (τ1/2 = 25 h). By analogy with acenes, where stability diminishes as the number of fused rings increases relative to the single Clar sextet, the predicted stability order is TIPS-PYR (3/8) > TIPS-ATT (2/6) > TIPS-DBPYR (3/10) (Fig. 2). Experimentally, however, an inversion is observed between TIPS-ATT and TIPS-PYR. Under our test conditions, this discrepancy indicates that factors beyond the Clar sextet count must be considered. Although a clear correlation exists between biradical character (y0)62,63 and thus the HOMO–LUMO gap (Table 1) and stability, the dominant factor in this series is likely photochemical: TIPS-PYR and TIPS-DBPYR exhibit ε at 365 nm that are approximately seven and eleven times higher, respectively, than that of TIPS-ATT, thereby accounting for their reduced stability (Table 1). However, there is no strict correlation between ε and stability, only a general trend. Additional insight is provided by the nature of the oxidation photoproducts. Upon photooxidation, TIPS-PYR derivatives gain an extra Clar sextet (increasing from three to four), leading to stabilization that is more significant than in the case of TIPS-ATT, whose Clar sextet count remains unchanged (two to two) (Fig. 5). Accordingly, the observed stability trend can be rationalized by the combined influence of photophysical properties (ε and τF) and the relative thermodynamic stability of the resulting photoproducts.64
Conclusions
This work demonstrates that chrysene, anthanthrene and pyranthrene can be consistently represented as localized structures with the aromatic sextets positioned on the outer rings. By analogy with phenanthrene and pyrene, which can be regarded as biphenyls with one or two ethenyl spacers, respectively, chrysene, anthanthrene and pyranthrene may be viewed as derivatives of trans-stilbene, chrysene and fulminene cores, each extended by two ethenyl spacers. Crystal structures provide direct evidence of determining the Fries structures. Representing polybenzenoid compounds with single and double bonds should therefore follow the Fries model. Bond lengths and bond length alternation (BLA) at the zig-zag edge offer robust criteria for positioning aromatic sextets. While NICS and HOMA indices are broadly consistent with these assignments, their interpretation requires caution. Photophysical studies reveal clear design rules: cata-condensation enhances brightness, as exemplified by TIPS-PYR with record molar brightness, whereas linear extension reduces emission efficiency but potentially enables singlet fission functionality, as in TIPS-DBPYR. Stability trends are further rationalized by the interplay between the molar absorption coefficient, radiative lifetime and the relative thermodynamic stabilization of oxidation photoproducts. Beyond highlighting the localized structure of peri-fused systems, this study identifies TIPS-PYR as an exceptionally bright dye and TIPS-DBPYR as a potentially robust singlet fission material, thereby expanding the molecular design space for next-generation optoelectronic devices.Author contributions
K. D. and K. L. T. conducted the synthesis and the study of the optical properties and managed the data. J. M. and M. H. obtained the crystal structures and managed the data. M. M. performed theoretical calculations, managed the data and carried out the review. M. F. contributed to conceptualization, writing, review and editing and project administration. All authors have read and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2503087, 2503088 and 2504051 contain the supplementary crystallographic data for this paper.65a–cThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedures, characterization data for the synthesized compounds and theoretical calculations. See DOI: https://doi.org/10.1039/d5sc09423b.
Acknowledgements
The synchrotron X-ray crystallography experiments were performed at the BL40XU beamline of SPring-8, with the approval of the Japan Synchrotron Radiation Research Institute (JASRI; Project No. 2024B1520).Notes and references
- M. Randić, Chem. Rev., 2003, 103, 3449–3606 CrossRef PubMed
.
- Z. Chen, C. S. Wannere, C. Corminboeuf, R. Puchta and P. von R. Schleyer, Chem. Rev., 2005, 105, 3842–3888 CrossRef PubMed
.
- A. T. Balaban, P. v. R. Schleyer and H. S. Rzepa, Chem. Rev., 2005, 105, 3436–3447 CrossRef PubMed
.
- M. Bendikov, F. Wudl and D. F. Perepichka, Chem. Rev., 2004, 104, 4891–4946 CrossRef PubMed
.
- J. E. Anthony, Angew. Chem., Int. Ed., 2008, 47, 452–483 Search PubMed
.
- J. Mei, Y. Diao, A. L. Appleton, L. Fang and Z. Bao, J. Am. Chem. Soc., 2013, 135, 6724–6746 Search PubMed
.
- Q. Ye and C. Chi, Chem. Mater., 2014, 26, 4046–4056 Search PubMed
.
- G. Schweicher, G. Garbay, R. Jouclas, F. Vibert, F. Devaux and Y. H. Geerts, Adv. Mater., 2020, 32, 1905909 Search PubMed
.
- C. Tönshoff and H. F. Bettinger, Chem.–Eur. J., 2021, 27, 3193–3212 Search PubMed
.
- N. Zeitter, N. Hippchen, A. Weidlich, P. Jäger, P. Ludwig, F. Rominger, A. Dreuw, J. Freudenberg and U. H. F. Bunz, Chem.–Eur. J., 2023, 29, e202302323 CrossRef PubMed
.
- N. H. Zeitter Nikolai, P. Baur, T. V. Unterreiner, F. Rominger, J. Freudenberg and U. H. F. Bunz, Org. Mater., 2024, 06, 12–17 Search PubMed
.
- C. Zong, S. Yang, Y. Sun, L. Zhang, J. Hu, W. Hu, R. Li and Z. Sun, Chem. Sci., 2022, 13, 11442–11447 Search PubMed
.
- X. Liu, B. Chen, M. Wang, X. Gu and L. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202416160 CrossRef PubMed
.
- R. Scholl, C. Seer and R. Weitzenböck, Berichte Dtsch. Chem. Ges., 1910, 43, 2202–2209 Search PubMed
.
- E. Clar and Fr. John, Berichte Dtsch. Chem. Ges. B Ser., 1930, 63, 2967–2977 Search PubMed
.
-
E. Clar and R. Schoental, Polycyclic Hydrocarbons, London, Academic Press INC., 1964, vol. 1 Search PubMed
.
- M. Solà, Front. Chem., 2013, 1, 22, DOI:10.3389/fchem.2013.00022
.
- G. Portella, J. Poater and M. Solà, J. Phys. Org. Chem., 2005, 18, 785–791 Search PubMed
.
- T. Jousselin-Oba, M. Mamada, K. Wright, J. Marrot, C. Adachi, A. Yassar and M. Frigoli, Angew. Chem., Int. Ed., 2022, 61, e202112794 CrossRef
.
- Y. Duan, G. Zhang, X. Liu, F. Shi, T. Wang, H. Yan, H. Xu and L. Zhang, J. Org. Chem., 2022, 87, 8841–8848 CrossRef PubMed
.
- X. Shi, T. Y. Gopalakrishna, Q. Wang and C. Chi, Chem.–Eur. J., 2017, 23, 8525–8531 CrossRef PubMed
.
- P. W. Fowler and W. Myrvold, J. Phys. Chem. A, 2011, 115, 13191–13200 CrossRef PubMed
.
- S. Fias, P. W. Fowler, J. L. Delgado, U. Hahn and P. Bultinck, Chem.–Eur. J., 2008, 14, 3093–3099 Search PubMed
.
- O. L. Griffith, A. G. Jones, J. E. Anthony and D. L. Lichtenberger, J. Phys. Chem. C, 2010, 114, 13838–13845 CrossRef
.
- M. R. Ajayakumar, J. Ma and X. Feng, Eur. J. Org Chem., 2022, 2022, e202101428 CrossRef
.
- D. W. Szczepanik, M. Solà, T. M. Krygowski, H. Szatylowicz, M. Andrzejak, B. Pawełek, J. Dominikowska, M. Kukułka and K. Dyduch, Phys. Chem. Chem. Phys., 2018, 20, 13430–13436 Search PubMed
.
- P. von R. Schleyer, M. Manoharan, H. Jiao and F. Stahl, Org. Lett., 2001, 3, 3643–3646 Search PubMed
.
- J. E. Anthony, J. S. Brooks, D. L. Eaton and S. R. Parkin, J. Am. Chem. Soc., 2001, 123, 9482–9483 Search PubMed
.
- Y. Gu, Y. G. Tullimilli, J. Feng, H. Phan, W. Zeng and J. Wu, Chem. Commun., 2019, 55, 5567–5570 Search PubMed
.
- S. Zhang, F. Talnack, T. Jousselin-Oba, V. Bhat, Y. Wu, Y. Lei, Y. Tomo, H. Gong, L. Michalek, D. Zhong, C. Wu, A. Yassar, S. Mannsfeld, C. Risko, M. Frigoli and Z. Bao, J. Mater. Chem. C, 2023, 11, 8992–9001 Search PubMed
.
- L. Zhang, B. Walker, F. Liu, N. S. Colella, S. C. B. Mannsfeld, J. J. Watkins, T.-Q. Nguyen and A. L. Briseno, J. Mater. Chem., 2012, 22, 4266–4268 Search PubMed
.
- L. Zhang, A. Fonari, Y. Zhang, G. Zhao, V. Coropceanu, W. Hu, S. Parkin, J.-L. Brédas and A. L. Briseno, Chem.–Eur. J., 2013, 19, 17907–17916 Search PubMed
.
- F. Lirette, A. Darvish, Z. Zhou, Z. Wei, L. Renn, M. A. Petrukhina, R. T. Weitz and J.-F. Morin, Chem. Sci., 2023, 14, 10184–10193 Search PubMed
.
- R. Gershoni-Poranne and A. Stanger, Chem. Soc. Rev., 2015, 44, 6597–6615 Search PubMed
.
- T. M. Krygowski, J. Chem. Inf. Comput. Sci., 1993, 33, 70–78 CrossRef
.
- P. Ribar, L. Valenta, T. Šolomek and M. Juríček, Angew. Chem., Int. Ed., 2021, 60, 13521–13528 Search PubMed
.
- R. Scholl and C. Seer, Adv. Cycloaddit., 1912, 394, 111–177 Search PubMed
.
- J. Lee and J. Park, Org. Lett., 2015, 17, 3960–3963 CrossRef
.
- T. Jousselin-Oba, K. Sbargoud, G. Vaccaro, F. Meinardi, A. Yassar and M. Frigoli, Chem.–Eur. J., 2017, 23, 16184–16188 CrossRef
.
- K. Sbargoud, M. Mamada, T. Jousselin-Oba, Y. Takeda, S. Tokito, A. Yassar, J. Marrot and M. Frigoli, Chem.–Eur. J., 2017, 23, 5076–5080 CrossRef PubMed
.
- T. Jousselin-Oba, M. Mamada, A. Okazawa, J. Marrot, T. Ishida, C. Adachi, A. Yassar and M. Frigoli, Chem. Sci., 2020, 11, 12194–12205 Search PubMed
.
- T. Jousselin-Oba, M. Mamada, J. Marrot, A. Maignan, C. Adachi, A. Yassar and M. Frigoli, J. Am. Chem. Soc., 2019, 141, 9373–9381 CrossRef PubMed
.
- J. H. Smith, D. Čavlović, L. T. Lackovic, M. Medina Lopez, E. Meirzadeh, M. L. Steigerwald, X. Roy, C. P. Nuckolls and S. R. Docherty, J. Am. Chem. Soc., 2025, 147, 111–117 CrossRef CAS PubMed
.
- K. Fries, R. Walter and K. Schilling, Adv. Cycloaddit., 1935, 516, 248–285 CAS
.
- A. Ciesielski, T. M. Krygowski and M. K. Cyrański, Symmetry, 2010, 2, 1390–1400 Search PubMed
.
- A. Graovac, I. Gutman, M. Randic and N. Trinajstic, J. Am. Chem. Soc., 1973, 95, 6267–6273 Search PubMed
.
- J. Oddershede and S. Larsen, J. Phys. Chem. A, 2004, 108, 1057–1063 CrossRef CAS
.
- M. Su, Y.-N. Jing, H. Bao and W.-M. Wan, Mater Chem Front, 2020, 4, 2435–2442 RSC
.
- T. Friščić, R. W. Lancaster, L. Fábián and P. G. Karamertzanis, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 13216–13221 CrossRef
.
- J. Harada, N. Yoneyama, S. Sato, Y. Takahashi and T. Inabe, Cryst. Growth Des., 2019, 19, 291–299 CrossRef CAS
.
- G. Portella, J. Poater, J. M. Bofill, P. Alemany and M. Solà, J. Org. Chem., 2005, 70, 2509–2521 Search PubMed
.
- I. A. Titaley, D. M. Walden, S. E. Dorn, O. M. Ogba, S. L. Massey Simonich and P. H.-Y. Cheong, Environ. Sci. Technol., 2019, 53, 1595–1607 CrossRef CAS PubMed
.
- B. K. Shah, D. C. Neckers, J. Shi, E. W. Forsythe and D. Morton, J. Phys. Chem. A, 2005, 109, 7677–7681 CrossRef CAS
.
- G. Markert, E. Paenurk and R. Gershoni-Poranne, Chem.–Eur. J., 2021, 27, 6923–6935 Search PubMed
.
- R. K. Dubey, M. Marongiu, S. Fu, G. Wen, M. Bonn, H. I. Wang, M. Melle-Franco and A. Mateo-Alonso, Chem, 2023, 9, 2983–2996 CAS
.
- W. P. Partridge, N. M. Laurendeau, C. C. Johnson and R. N. Steppel, Opt. Lett., 1994, 19, 1630–1632 CrossRef CAS
.
- O. Nagler, A.-M. Krause, K. Shoyama, M. Stolte, R. K. Dubey, L. Liu, Z. Xie and F. Würthner, Org. Lett., 2022, 24, 6839–6844 Search PubMed
.
- Y. Jue Bae, M. D. Krzyaniak, M. B. Majewski, M. Desroches, J.-F. Morin, Y.-L. Wu and M. R. Wasielewski, ChemPlusChem, 2019, 84, 1432–1438 Search PubMed
.
- D. J. Stewart, J. Shi, T. R. Naranjo, T. A. Grusenmeyer, J. M. Artz, C. L. McCleese, R. M. O'Donnell, T. M. Cooper, W. M. Shensky and J. E. Haley, Phys. Chem. Chem. Phys., 2018, 20, 28412–28418 RSC
.
- Y. Bo, N. Zink-Lorre, R. Weiß, Á. Sastre-Santos, T. Clark, F. Fernández-Lázaro and D. M. Guldi, Artif. Photosynth, 2025, 1, 226–236 CrossRef CAS
.
- M. Dvořák, S. K. K. Prasad, C. B. Dover, C. R. Forest, A. Kaleem, R. W. MacQueen, A. J. I. Petty, R. Forecast, J. E. Beves, J. E. Anthony, M. J. Y. Tayebjee, A. Widmer-Cooper, P. Thordarson and T. W. Schmidt, J. Am. Chem. Soc., 2021, 143, 13749–13758 CrossRef
.
- K. Yamaguchi, Chem. Phys. Lett., 1975, 33, 330–335 CrossRef CAS
.
- M. Nakano, R. Kishi, T. Nitta, T. Kubo, K. Nakasuji, K. Kamada, K. Ohta, B. Champagne, E. Botek and K. Yamaguchi, J. Phys. Chem. A, 2005, 109, 885–891 CrossRef CAS PubMed
.
- J. Ly, K. Martin, S. Thomas, M. Yamashita, B. Yu, C. A. Pointer, H. Yamada, K. R. Carter, S. Parkin, L. Zhang, J.-L. Bredas, E. R. Young and A. L. Briseno, J. Phys. Chem. A, 2019, 123, 7558–7566 Search PubMed
.
-
(a)
CCDC 2503087: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q0nr5
; (b) CCDC 2503088: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q0ns6
; (c) CCDC 2504051: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q1nv9
.
| This journal is © The Royal Society of Chemistry 2026 |