Open Access Article
Open Access ArticleInterfacial failure mechanisms and design principles in solid-state sodium batteries
Mingyue
Wang†
*,
Qing
Zhong†
,
Yue
Wang†
,
Xu
Liu
,
Dongyang
Zhang
* and
Shujiang
Ding
 *
*
School of Chemistry, Engineering Research Center of Energy Storage Materials and Devices, Ministry of Education, National Innovation Platform (Center) for Industry-Education Integration of Energy Storage Technology, Xi'an Jiaotong University, Xi'an 710049, P. R. China. E-mail: mywang@xjtu.edu.cn; dingsj@xjtu.edu.cn; zdysun@xjtu.edu.cn
First published on 28th January 2026
Abstract
Solid-state sodium batteries (SSSBs) have attracted increasing attention as a promising alternative for large-scale energy storage owing to their intrinsic safety, material abundance, and potential cost advantages. Significant progress has been made in developing diverse solid-state electrolytes, including polymers, inorganic ceramics, and hybrid systems, many of which exhibit impressive bulk ionic conductivity. However, the translation of these materials-level properties into durable, high-performance solid-state sodium batteries remains limited, indicating that bulk ion transport alone does not govern practical cell behavior. In this review, we adopt an interface-centered and issue-driven perspective to analyze the key challenges in SSSBs. Rather than providing a materials-category-based summary, we focus on dominant interfacial failure mechanisms and their sodium origins. Chemical and electrochemical instability, electrical blocking associated with space-charge effects and grain boundaries, mechanical degradation arising from elastic and thermal mismatch, and defect-assisted sodium dendrite penetration are discussed within a unified mechanistic framework. These interfacial processes are shown to be intrinsically coupled, collectively controlling effective ion transport, critical current density, and long-term cell stability. Building on this understanding, we critically assess why high bulk ionic conductivity has not translated into robust full-cell performance and emphasize the limitations of conductivity as a single performance metric. We further discuss general design principles for interface engineering across different electrolyte families and revisit lessons from technologically mature sodium battery systems to clarify realistic pathways toward practical implementation. By linking interfacial chemistry, defect physics, and mechanical properties, this Review aims to provide a coherent framework and forward-looking guidance for the rational design of next-generation SSSBs.
1 Introduction
1.1 From liquid electrolytes to solid electrolytes
Rechargeable secondary batteries have become a crucial energy storage and conversion solution supporting renewable energy integration over the past decade.1–3 Among the various types, lithium-ion batteries (LIBs) stand out due to their superior energy density, high power delivery, and extended cycling stability, leading to their extensive use in everyday applications like mobile devices and electric transportation. Despite their success, the continued advancement of LIBs is constrained by several limitations—such as scarce lithium reserves, geographically imbalanced lithium ore distribution, and increasing raw material expenses (Fig. 1a)—posing barriers to long-term scalability.4,5 These challenges, coupled with the rising demand for efficient energy storage across multiple industries, have accelerated research into alternative battery chemistries beyond conventional lithium-ion systems.6,7 Recently, sodium-based batteries have emerged as a promising candidate, drawing significant scientific and industrial interest due to their cost-effectiveness, the widespread availability of sodium, and operational mechanisms analogous to those of lithium-ion technologies.8,9 While sodium-ion batteries (SIBs) generally exhibit lower theoretical energy densities compared to LIBs owing to the greater atomic mass of sodium, they offer compelling benefits for large-scale applications such as grid-level energy storage, where safety, longevity, and economic feasibility are prioritized over maximum energy density.10,11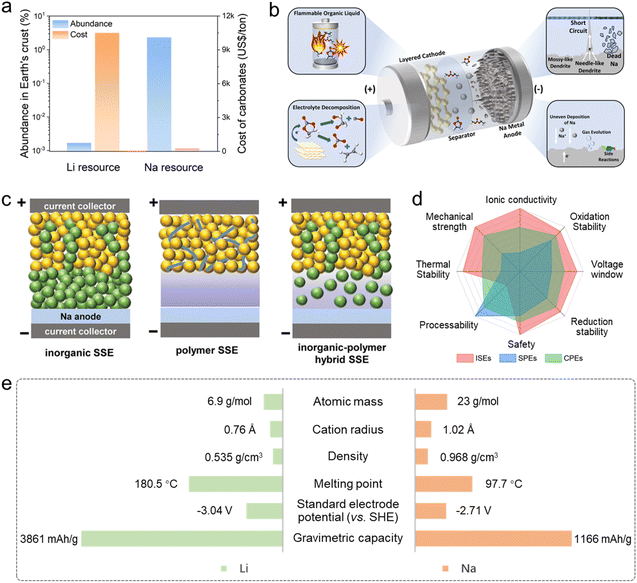 | ||
| Fig. 1 (a) Abundance and costs of Li and Na resources. (b) Challenges in sodium batteries arising from organic liquid electrolytes.35 Copyright 2023, The Royal Society of Chemistry. (c) Schematic diagram of inorganic SSE, polymer SSE and inorganic–polymer hybrid SSE based SSMBs.36 Copyright 2023, Wiley-VCH. (d) Characteristics of ISEs, SPEs and composite polymer electrolytes (CPEs). (e) Comparison of physicochemical properties between Li and Na elements. | ||
The electrolyte is a critical component in sodium batteries, serving as the medium for sodium ion transport between the cathode and anode, thereby significantly affecting the energy density and rate performance of batteries. Electrolyte systems are typically classified into three types: aqueous, organic liquid, and solid-state electrolytes (SSEs).12 Although aqueous electrolytes offer advantages such as high safety and stable cycling behavior, their narrow electrochemical stability window limited by water decomposition restricts the operating voltage to below 2 V, which inherently limits the achievable energy density.13,14 Only under highly concentrated salt conditions, for example with 17 mol per L NaClO4, can voltages beyond 2 V be attained, leading to the failure of achieving practical scalability of the aqueous systems.15 Similar to those in lithium batteries, organic liquid electrolytes based on carbonate or ether solvents are commonly employed in sodium batteries.16,17 Early research primarily focused on carbonate solvents like ethylene carbonate (EC) and propylene carbonate (PC), owing to their high dielectric constants and wide electrochemical windows.18 By 2014, researchers had demonstrated that by tailoring the solvation structure of ether-based electrolytes, reversible co-intercalation of Na+ ions along with ether molecules into graphite anodes could be achieved—a process not feasible with conventional carbonate electrolytes.19 Moreover, ether-based electrolytes exhibit superior low-temperature performance and high-rate capability due to their strong reductive stability and low desolvation energy.20 Despite ongoing advancements in liquid electrolyte formulations, the inherent flammability and thermal instability of organic electrolytes remain major concerns, often leading to thermal runaway and associated safety hazards in real-world applications. Numerous incidents in recent years—including fires and explosions during battery operation or charging—have been attributed to such risks, particularly under abusive conditions involving mechanical damage (e.g., crushing or puncture), electrical abuse (e.g., overcharging or short circuits), or excessive heat buildup, as shown in Fig. 1b.21,22
SSEs have emerged as a promising substitute for conventional liquid electrolytes, offering enhanced safety and improved electrochemical performance in battery systems.23 Notably, β″-Al2O3 (BASE) solid electrolytes represent one of the earliest and most technologically mature sodium-ion conducting ceramics. Since the 1960s, BASE has been successfully employed in high-temperature molten sodium batteries, including Na–S, Na–NiCl2 (ZEBRA), and Na–FeCl2 systems, where it serves as a robust separator enabling fast Na+ transport at operating temperatures of 250–350 °C.24 These battery systems have reached commercial deployment for stationary energy storage, highlighting the exceptional chemical stability, high ionic conductivity, and long-term reliability of BASE electrolytes under harsh operating conditions. In recent years, extensive research has led to the development of various SSE types aimed at broadening the applicability of SSSBs.25,26 Currently, SSEs for sodium batteries are primarily categorized into inorganic, organic (polymer) and inorganic–organic types (Fig. 1c), and the corresponding characteristics are summarized in Fig. 1d.27,28 However, achieving both effective interface contact and chemical stability of the electrolyte–electrode interface is vital for high-performance SSSBs. Two central challenges therefore dominate the design of efficient SSSBs: (i) developing SSEs with high ionic conductivity at ambient temperatures, and (ii) engineering well-matched electrode–electrolyte interfaces.29–31 In recent years, substantial progress has been achieved in boosting ionic conductivity through compositional modifications, composite integration, and innovative structural designs, with some advanced SSEs now reaching conductivities comparable to those of liquid electrolytes.32–34
1.2 Similarities and differences with solid-state Li batteries
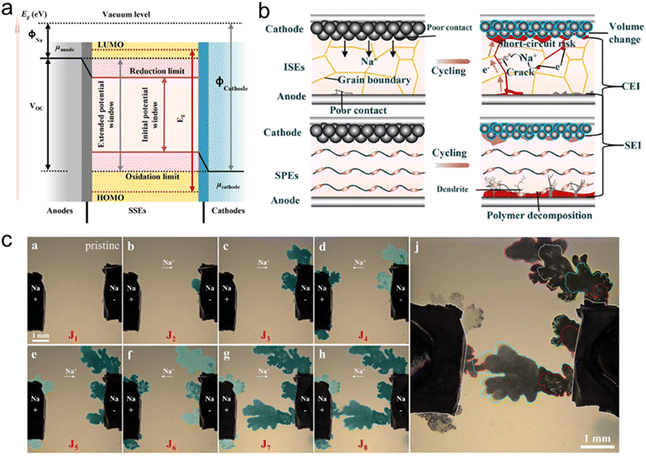
Owing to the similar electrochemical properties between lithium and sodium, SSSBs exhibit numerous functional similarities with solid-state lithium batteries (SSLBs). As a result, much of the fundamental understanding and experimental methodologies developed for SSSBs over recent years can be partially adapted to accelerate the advancement of SSSB technology.37,38 Similar to their lithium counterparts, a typical SSSB consists of an anode, an SSE and a cathode. The working principles rely on the directional movement of cations coupled with redox reactions to enable energy storage and delivery.39 The SSE functions as both an ionic conductor, facilitating Na+ ion transport between electrodes, and as a physical barrier that prevents electrical shorting.40,41 Oxidation occurs at the anode, releasing electrons into the external circuit and generating Na+ ions that migrate through the SSE toward the cathode. At the cathode, these ions undergo electrochemical insertion into the host lattice, completing the energy release process.Although SSSBs operate on the same “rocking chair” principle as SSSBs, and many similar materials for electrodes and electrolytes developed for SSSBs can be applied to SSSBs, significant distinctions exist between the two systems. These differences primarily stem from the distinct physicochemical characteristics of sodium and lithium, as illustrated in Fig. 1e. One of the most influential factors is the ionic radius, which directly affects ion diffusion kinetics and storage mechanisms. The larger ionic radius of Na+ (1.02 Å) compared to Li+ (0.76 Å), along with its lower charge density, makes Na+ migration more challenging within the crystal lattice of SSEs.42 For instance, in inorganic solid electrolytes such as NASICON-type materials, wider conduction pathways are required for Na+ to avoid increased energy barriers caused by steric constraints.43,44 Moreover, certain materials widely used in SSSBs—such as the LiCoO2 cathode and Li7La3Zr2O12 electrolyte—are not suitable for use in SSSBs.45,46 A major hurdle in the development of SSSBs is the optimization of electrode–electrolyte interfaces. The larger size of Na+ ions promotes the formation of a dense and thick space charge layer at the interface, leading to significantly higher interfacial impedance compared to Li+-based systems. In composite organic-inorganic electrolytes, the transfer of Na+ across the internal phase boundary faces a greater energy barrier due to weaker coordination between Na+ and the polymer matrix, as well as reduced lattice compatibility with inorganic fillers—both of which are less favorable than in lithium systems—resulting in a pronounced ion transport bottleneck.47,48 Additionally, mismatches in thermal expansion coefficients between sodium-based components are more pronounced; for example, the difference between sodium-β-Al2O3 and metallic sodium can increase the likelihood of interfacial delamination under thermal cycling, further exacerbating impedance growth.49,50 Chemical compatibility between the electrolyte and electrodes is another concern, for example, sulfide-based SSEs tend to react adversely with metallic sodium anodes.51,52 All in all, while SSSBs share fundamental similarities with SSSBs—enabling the leveraging of existing research—their unique challenges arising from the physicochemical properties of sodium make interface engineering and material selection more complex. These distinctions demand focused attention to overcome the obstacles impeding the performance and commercialization of SSSBs.
Despite the rapidly growing body of literature on solid-state electrolytes for sodium batteries, a number of fundamental questions remain insufficiently addressed. In particular, the performance gap between impressive ionic conductivity values reported for many sodium solid electrolytes and the poor durability and rate capability of full solid-state sodium batteries persists as a central bottleneck. This discrepancy suggests that materials chemistry alone is not the limiting factor, and that interfacial phenomena, mechanical compatibility, and sodium-specific electrochemical behaviors play a decisive role in determining practical cell performance.53,54 In this Review, rather than providing a purely materials-category-based summary, we adopt a problem-driven and concept-oriented perspective to critically analyze solid-state sodium battery systems. We emphasize three overarching themes. First, we examine the fundamental differences between sodium and lithium solid-state batteries that extend beyond simple ionic radius considerations, with particular focus on interfacial chemistry, space-charge effects, and failure mechanisms unique to sodium systems. Second, we critically assess why high bulk ionic conductivity has not translated into robust full-cell performance, highlighting the limitations of conductivity as a single performance metric and underscoring the importance of interfacial resistance, critical current density, and realistic full-cell validation. Third, we revisit technologically mature systems such as β″-Al2O3-based high-temperature molten sodium batteries, extracting key lessons from their long-term success and discussing the challenges associated with translating these advantages to lower-temperature solid-state configurations. By integrating insights across organic, inorganic, and hybrid electrolyte systems, and by explicitly linking ion transport, interfacial chemistry, and mechanical properties, this review aims to provide a coherent framework for understanding the persistent challenges in solid-state sodium batteries. We further identify near-term research priorities and realistic technological pathways, clarifying where progress is fundamentally materials-limited versus engineering-limited, and highlighting application scenarios in which sodium-based solid-state systems may offer advantages over their lithium counterparts.
2 Mechanisms and challenges of solid-state electrolytes
2.1 Transport mechanisms of Na-ions in solid-state electrolytes
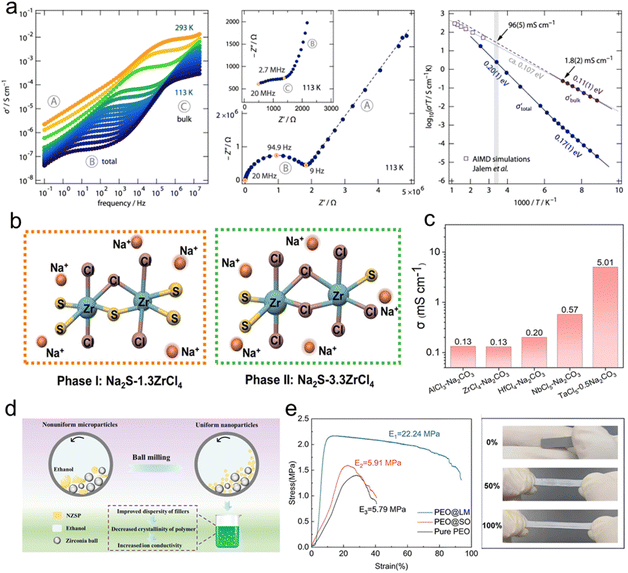
The transport behavior of sodium ions in SSEs directly determines the overall ionic conductivity and electrochemical performance of the SSSBs. Typically, the ion transport mechanism is closely tied to the specific type of SSE and temperature,25,55 as provided in Fig. 2a. Clarifying these region-specific transport mechanisms is not only essential for understanding the fundamental principles of ion conduction in solid systems, but also provides critical guidance for the structural design and performance optimization of SSEs.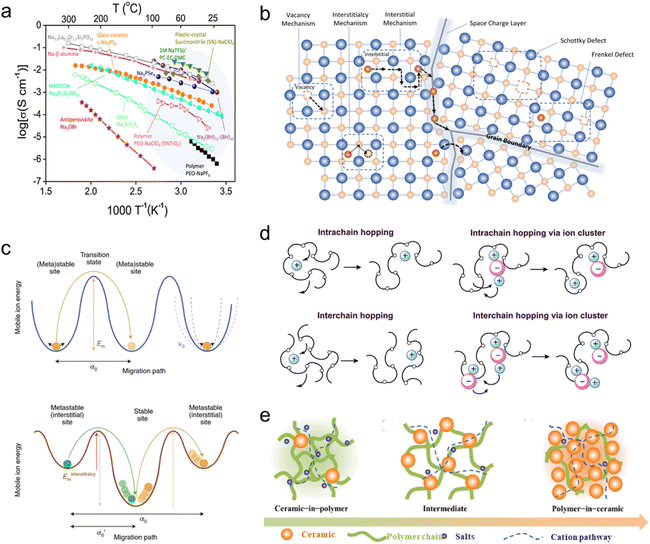 | ||
| Fig. 2 (a) Temperature-dependent ionic conductivities of typical SSEs for sodium batteries.25 Copyright 2018, Elsevier. (b) Na-ion transport mechanism in ISEs.35 Copyright 2023, The Royal Society of Chemistry. (c) The energy profiles associated with cation migration via direct vacancy or interstitial hopping and correlated hopping.69 Copyright 2022, Springer Nature. (d) Na-ion transport mechanism in SPEs66 and (e) inorganic/organic hybrid SSEs.70 Copyright 2015, The Royal Society of Chemistry. Copyright 2018, Elsevier. | ||
| σ = σ0e(−EA/kBT) | (1) |
| σ = σ0T−1/2e−B/(T−T0) | (2) |
Beyond theoretical modelling, electrochemical impedance spectroscopy (EIS) is widely used to experimentally determine the ionic conductivity of SSEs. In this method, the SSE is placed between two ion-blocking electrodes (e.g., stainless steel), allowing the calculation of σ using the formula:74
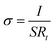 | (3) |
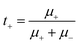 | (4) |
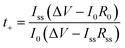 | (5) |
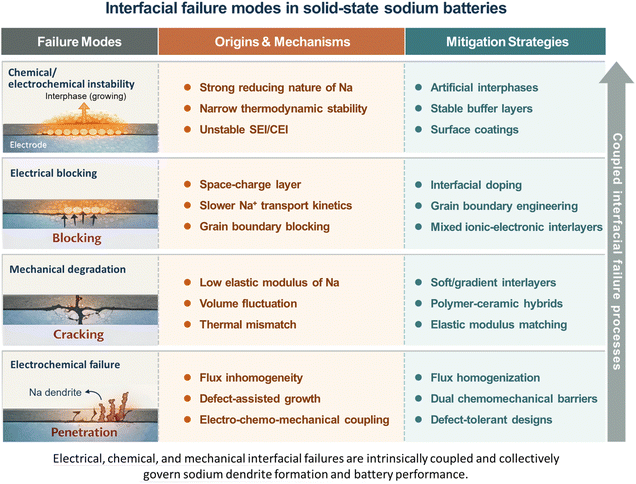
2.2 Interfacial failure modes in solid-state sodium batteries
Although interfacial issues are widely recognized as the primary bottleneck in solid-state sodium batteries, their underlying mechanisms are often discussed in a fragmented and qualitative manner. In practice, interfacial failure rarely originates from a single factor; instead, it emerges from the coupled effects of interfacial chemistry, defect-mediated ion transport, and mechanical mismatch.76 Sodium-specific properties—including its stronger reducing character, lower elastic modulus, and slower ion mobility compared to lithium—amplify these couplings and give rise to failure modes that are distinct from those in lithium-based solid-state systems. From a chemical perspective, many sodium solid electrolytes are thermodynamically unstable against sodium metal or high-voltage cathodes, leading to continuous interphase growth and increasing interfacial resistance.77 Electrically, space-charge layers and grain boundary barriers dominate ion transport, explaining why high bulk ionic conductivity often fails to translate into high effective conductivity at the cell level. Mechanically, volume fluctuations during cycling and mismatched elastic moduli induce interfacial debonding and crack formation, which further localize current density and accelerate degradation. These chemical, electrical, and mechanical processes are intrinsically coupled and collectively promote electrochemical failure, such as sodium dendrite nucleation and penetration under practical current densities. Fig. 3 summarizes the dominant interfacial failure modes in solid-state sodium batteries, their sodium-specific origins, and corresponding mitigation strategies. This framework highlights that addressing interfacial challenges requires integrated design approaches rather than isolated materials optimization.3 Solid-state electrolyte design
3.1 Organic solid-state electrolytes
Organic solid-state electrolytes (OSE) generally consist of an organic compound matrix and sodium salts.102 Most polymer electrolytes still suffer from low ionic conductivity at room temperature and unsatisfactory Na+ transference numbers, which significantly limit their practical application.74,103,104 According to the type of organic matrix, OSE can mainly be divided into polymer SSEs and gel SSEs.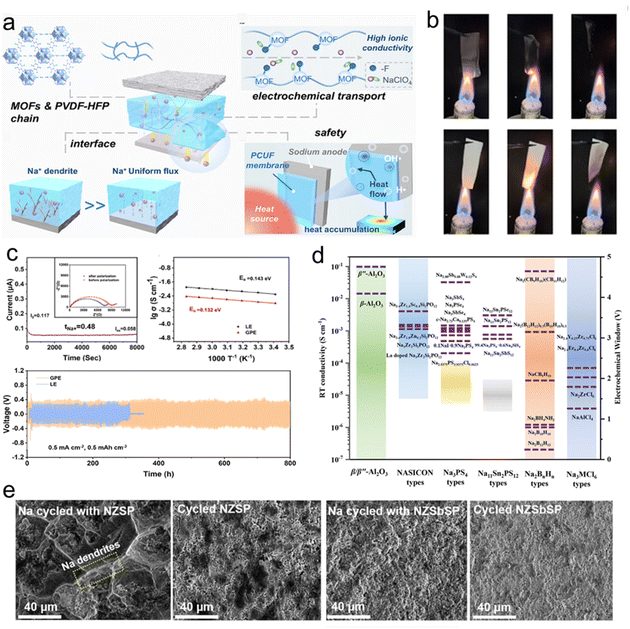 | ||
| Fig. 5 (a) Schematic illustration of the design principle of PCUF SSEs. (b) Ignition experiments of PH and PCUF membranes.109 Copyright 2025, Wiley-VCH. (c) Chronoamperometry curve and impedance variation, liner fitting plots for σ and voltage–time curves of the GPE and LE.122 Copyright 2025, Wiley-VCH. (d) Ionic conductivities of ISSEs and the corresponding electrochemical stability windows.61 Copyright 2023, Wiley-VCH. (e) SEM images of sodium metal, NZSP and NZSbSP in corresponding symmetric cells cycled at 60 °C.127 Copyright 2025, Elsevier. | ||
3.2 Inorganic solid-state electrolytes
ISEs are based on inorganic compounds such as crystals and glassy states. They possess advantages such as high ionic conductivity, good chemical stability, and high pressure resistance. According to their crystal structure and composition, they can be classified into various types (Fig. 5d).To improve both ionic conductivity and mechanical performance, various strategies have been explored, including the substitution of Al3+ with foreign cations such as Li+, Mg2+, Nb5+, Ti4+ and Zr4+, which modify the Na+ content in the conduction layers.134–140 For instance, Chen et al.141 doped β-Al2O3 with a 0.5 wt% Cu2+ stabilizing agent, boosting ionic conductivity to 1.2 × 10−3 S cm−1. They further introduced an In2S3 interlayer between the sodium anode and the electrolyte, forming a composite layer composed of an Na–In alloy and Na2S that exhibited mixed ionic–electronic conductivity. This strategy improved interfacial electrochemical stability and enabled efficient Na+ transport. Various aliovalent dopants and secondary phases have been introduced to stabilize the β″ phase and enhance ionic conductivity; however, these compositional optimizations do not fundamentally resolve the interfacial and mechanical challenges encountered in lower-temperature solid-state configurations.142–146 Traditional solid-state synthesis of Na-β-Al2O3 typically involves high sintering temperatures and prolonged processing times, often leading to decomposition of the desired β′′-Al2O3 phase. To overcome these limitations, alternative fabrication techniques such as co-precipitation, sol–gel processes, microwave-assisted heating, and mechanochemical synthesis have been developed.139,144,147–152 Despite these advances, achieving highly homogeneous and conductive β′′-Al2O3 remains difficult due to issues such as sodium volatilization, abnormal grain growth, moisture sensitivity from boundary-phase NaAlO2 formation, and residual impurities.59 Process improvements like the double-zeta sintering method and spark plasma sintering (SPS) can enhance densification; however, the former demands extreme temperatures (up to 1600 °C), while the latter requires expensive equipment, limiting scalability.153–155
To address these challenges, extensive efforts have focused on compositional tuning and microstructural engineering. Representative approaches include aliovalent cation substitution to optimize lattice dimensions and grain boundary chemistry, as well as surface modification strategies aimed at stabilizing the Na/NASICON interface.167–169 Akbar et al. introduced Sb3+ into the NZSP lattice to tailor the crystal structure and facilitate ion mobility.170 The resulting Na3.1Zr1.9Sb0.1Si2PO12 (NZSbSP) exhibited a room-temperature ionic conductivity of 5.1 × 10−4 S cm−1, improved structural stability, lowered grain boundary resistance, and effectively suppressed Na dendrite formation (Fig. 5e).127 While such methods can reduce grain boundary impedance and improve cycling stability under moderate conditions, the resulting gains in full-cell performance remain incremental, and stable operation at practically relevant current densities remains difficult to achieve. Overall, NASICON-type electrolytes illustrate a broader challenge in solid-state sodium batteries: excellent bulk transport properties alone are insufficient to guarantee robust cell-level performance. Further progress will therefore depend less on continued optimization of lattice conductivity and more on interface engineering, defect control, and mechanically compliant cell architectures capable of sustaining uniform Na+ flux under realistic operating conditions.
 | ||
| Fig. 6 (a) Conductivity of Na2.9Sb0.9W0.1S4.178 Copyright 2025, American Chemical Society. (b) The scheme diagram of Na2S–1.3ZrCl4 SSEs and Na2S–3.3ZrCl4 SSEs.180 Copyright 2025, Wiley-VCH. (c) RT ionic conductivities of MCly–xNa2CO3 samples (M = Ta, Nb, Hf, Zr, and Al).181 Copyright 2025, The Royal Society of Chemistry. (d) Schematic illustration of the inorganic–organic hybrid SSE.182 Copyright 2023, Wiley-VCH. (e) Mechanical properties of PEO@LM electrolyte.183 Copyright 2024, Wiley-VCH. | ||
Despite their outstanding ionic transport properties, sulfide-based electrolytes face significant challenges related to environmental instability. They are highly hygroscopic and prone to hydrolysis upon exposure to moisture, susceptible to oxidation when paired with high-voltage cathodes, and may undergo reduction reactions in contact with metallic sodium, severely restricting their practical deployment. Guo et al. introduced a multi-cation co-substitution strategy using Sn, W, Ca, and Ti at the Sb site in Na3−□Sb1−4x(SnWCaTi)xS4.179 This approach not only generated a high concentration of sodium vacancies but also increased the configurational entropy of the lattice, stabilizing the crystal structure. The resulting high-entropy electrolyte achieved a room-temperature ionic conductivity of 6.3 × 10−3 S cm−1 with a remarkably low activation energy of 0.15 eV. Besides, the ionic conductivity remained at 5.3 × 10−3 S cm−1 after 10 minutes in air with 50% relative humidity, and no structural degradation was observed, even upon direct exposure to water.
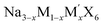 , where M denotes a transition metal, lanthanide, or element from the boron/nitrogen group, M′ refers to Zr or its substituted variants, and X stands for one or more halogen elements.35 Owing to the higher electronegativity of halide ions relative to O2− and S2−, halide electrolytes generally demonstrate superior oxidative stability. Among them, Na2ZrCl6 is the most studied chloride-based sodium conductor, albeit with a modest ionic conductivity of approximately 10−5 S cm−1.59 Recent studies indicate that incorporating oxygen can effectively improve ion transport.186 Ruoff et al. employed mechanochemical synthesis using Na2O to introduce oxygen into NaAlCl4, achieving an optimized conductivity exceeding 1 × 10−4 S cm−1.187 Hydride-based electrolytes, which consist of Na+ ions paired with complex anions such as (BH4)−, (NH2)−, (AlH4)−, and (B10H10)2−, are notable for their excellent reductive stability, mechanical flexibility, and low mass density.188 In particular, borohydride-based SSEs exhibit favorable Na+ conduction and good compatibility with metallic sodium anodes, though their application is limited by insufficient thermal and oxidative resilience.189
, where M denotes a transition metal, lanthanide, or element from the boron/nitrogen group, M′ refers to Zr or its substituted variants, and X stands for one or more halogen elements.35 Owing to the higher electronegativity of halide ions relative to O2− and S2−, halide electrolytes generally demonstrate superior oxidative stability. Among them, Na2ZrCl6 is the most studied chloride-based sodium conductor, albeit with a modest ionic conductivity of approximately 10−5 S cm−1.59 Recent studies indicate that incorporating oxygen can effectively improve ion transport.186 Ruoff et al. employed mechanochemical synthesis using Na2O to introduce oxygen into NaAlCl4, achieving an optimized conductivity exceeding 1 × 10−4 S cm−1.187 Hydride-based electrolytes, which consist of Na+ ions paired with complex anions such as (BH4)−, (NH2)−, (AlH4)−, and (B10H10)2−, are notable for their excellent reductive stability, mechanical flexibility, and low mass density.188 In particular, borohydride-based SSEs exhibit favorable Na+ conduction and good compatibility with metallic sodium anodes, though their application is limited by insufficient thermal and oxidative resilience.189
Growing attention has been directed toward hybrid inorganic systems. Dong et al. developed a new class of sulfide–chloride composite electrolytes (Na–Zr–S–Cl), yielding two phases: a chlorine-deficient form (Na2S·1.3ZrCl4) and a chlorine-rich counterpart (Na2S·3.3ZrCl4), as illustrated in Fig. 6b.180 The latter delivered a high ionic conductivity of 4.89 × 10−4 S cm−1, whereas the former showed superior chemical stability. Besides, the strategic integration of multiple anions to form amorphous frameworks has proven effective in simultaneously enhancing various electrolyte characteristics. Wang et al. synthesized a series of amorphous SSEs (MCly–xNa2CO3, M = Ta, Nb, Hf, Zr, Al) via high-energy ball milling.181 The TaCl5·1.2Na2CO3 electrolyte achieved a high ionic conductivity of 1.11 × 10−3 S cm−1 at room temperature (Fig. 6c).
3.3 Inorganic–organic hybrid solid-state electrolytes
Inorganic–organic hybrid SSEs are composite systems formed by combining inorganic and organic components through either physical blending or chemical linkage, aiming to leverage the mechanical flexibility of polymers and the high ionic conductivity of inorganic phases to achieve complementary performance enhancements.190 In these systems, polymers such as PEO and PVDF-HFP serve as continuous frameworks that provide structural flexibility and interfacial adaptability, while dispersed inorganic fillers, such as NASICON-type ceramics or sulfide-based conductors, act as ion-conducting reinforcements that establish efficient Na+ transport networks, thereby enhancing overall ionic mobility.191,192 By adjusting the filler loading, particle size, and dispersion homogeneity, the interfacial resistance between the two phases can be effectively minimized. For instance, Wang et al. engineered a bilayer architecture comprising a PEO layer adjacent to the sodium metal anode and a polyacrylonitrile (PAN)-based layer facing the high-voltage cathode,182 as displayed in Fig. 6d. This approach introduced additional Na+ conduction pathways and promoted segmental motion of PEO chains, yielding a thin (25 µm), lightweight (1.65 mg cm−2) and mechanically robust (13.84 MPa) CPE with an ionic conductivity of 1.62 × 10−4 S cm−1.Although conventional inorganic fillers enhance stiffness to help suppress dendrite penetration, they often compromise material toughness and lack the ability to heal existing defects or mitigate “dead sodium” formation. To address this, modern designs exploit the rigidity of inorganic components to hinder dendrite propagation, while utilizing the elasticity of the organic phase to accommodate volume fluctuations during cycling, thus simultaneously improving ion transport efficiency and interfacial durability.193,194 Suo et al. incorporated liquid metal (GalnSn) nanoparticles into a PEO matrix, creating a highly resilient PEO@LM composite with electric-field-driven self-healing functionality.183 The liquid metal filler reduced PEO crystallinity via hydrogen bonding interactions, boosting ionic conductivity to 1.62 × 10−4 S cm−1 and expanding the electrochemical window to 5.2 V, which enabled self-healing at the electrolyte–anode interface through electric-field-induced migration (Fig. 6e). Metal–organic frameworks (MOFs) represent another promising class of inert fillers for CPEs, owing to their well-defined porosity, large surface area, abundant surface functional groups, and excellent thermal and chemical stability.195 Tian et al. incorporated a 3D Cu-MOF into a PEO-based electrolyte, where open metal sites competitively coordinated with both TFSI− anions and ether oxygens in PEO, weakening the binding between Na+ and the polymer chain.196 This facilitated ion release and migration, leading to a room-temperature conductivity of 1.03 × 1−4 S cm−1, a cation transference number of 0.58, and effective suppression of dendritic plating.
4 Interface engineering
Interfacial challenges remain a critical bottleneck in advancing SSSBs.197,198 The anode–electrolyte and cathode–electrolyte interfaces are two core components of SSSBs, both of which face common issues that restrict battery performance.199,200 However, the two interfaces also have distinct problems due to differences in potential environments and material characteristics.201,2024.1 Anode–electrolyte interface
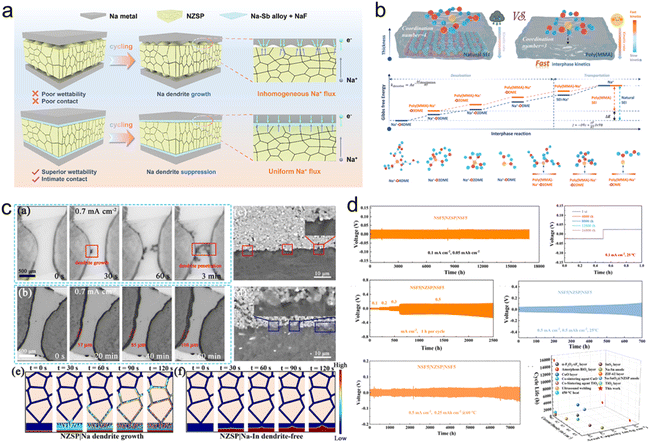
For SSSBs, the anode/electrolyte interface is critical, primarily concerning the physical and chemical contact between sodium metal or alloy anodes and solid electrolytes, which must facilitate efficient ion conduction pathways. Achieving reversible sodium plating and stripping without dendrite formation remains a key challenge.203,204 Research indicates that polymer-based electrolytes generally offer more stable and closer interfacial contact with sodium anodes compared to inorganic solid electrolytes, where dendritic growth is prevalent, which often leads to grain boundary penetration. Furthermore, repeated volume expansion and contraction of the sodium anode during cycling can degrade interfacial adhesion, resulting in poor long-term cycling stability.205,206 Therefore, effective interfacial engineering between sodium anodes and inorganic solid electrolytes is essential to minimize interfacial resistance and improve overall battery performance.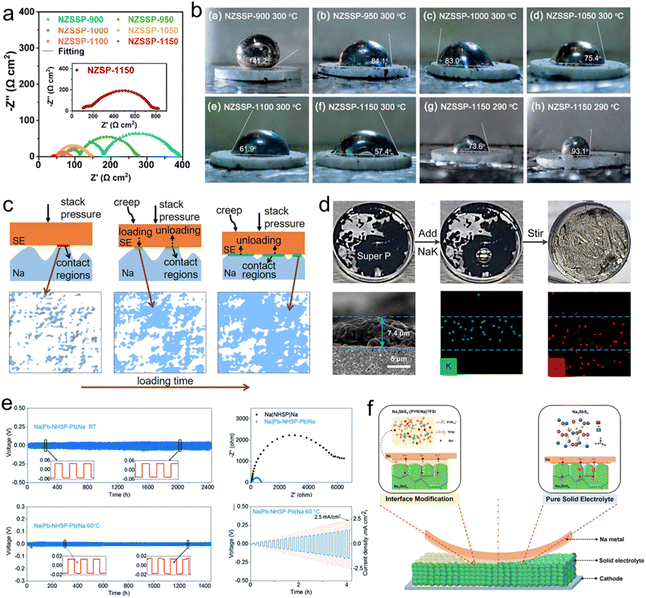 | ||
| Fig. 7 (a) EIS spectra of Na/NZSSP/Na symmetrical cells. (b) Pictures of Na on different SSEs.169 Copyright 2023, American Chemical Society. (c) Surface evolution with time due to creep.209 Copyright 2021, American Chemical Society. (d) Fabrication and SEM images of the C@Na–K interface.210 Copyright 2023, American Chemical Society. (e) The electrochemical performance of Pb decorated Na3Hf2Si2PO12.211 Copyright 2022, The Royal Society of Chemistry. (f) Schematic diagram of the Na3SbS4/Na interface with the (PYR/Na)TFSI interlayer.212 Copyright 2022, Elsevier. | ||
 | ||
| Fig. 8 (a) Schematic of the mechanism for Na plating/stripping at the Na/NZSP and Na/SbF3-NZSP interface.215 Copyright 2025, Wiley-VCH. (b) Schematic illustration of different coordination structures and desolvation energy barriers of Na+ at the natural SEI and poly(MMA) interface.217 Copyright 2025, Wiley-VCH. (c) Sodium deposition behavior of Na|NZSP|Na and Na–In|NZSP|Na–In at a current density of 0.7 mA cm−2.218 Copyright 2025, Wiley-VCH. (d) Electrochemical performance of an NZSP-based symmetrical cell.219 Copyright 2024, American Chemical Society. | ||
4.2 Cathode–electrolyte interface
The physical contact between the cathode and SSE at the microscopic level typically remains limited to point-to-point interaction, resulting in elevated interfacial impedance and sluggish kinetics of sodium ion migration. Furthermore, repeated volume changes of cathodes during sodium ion insertion and extraction processes can induce mechanical separation at the cathode–electrolyte interface, exacerbating interfacial degradation.222 This deterioration further increases resistance and accelerates capacity fading, ultimately reducing the cycle life of SSSBs. Besides, the cathode–electrolyte interface operates under high electrochemical potentials, creating favorable conditions for redox side reactions. These approaches aim to suppress undesirable side reactions at high voltages, accommodate volumetric strain during cycling, reduce interfacial resistance, prevent loss of ionic conductivity due to interface separation or insulating byproduct formation and thereby enhance overall cycling stability (Fig. 9a).223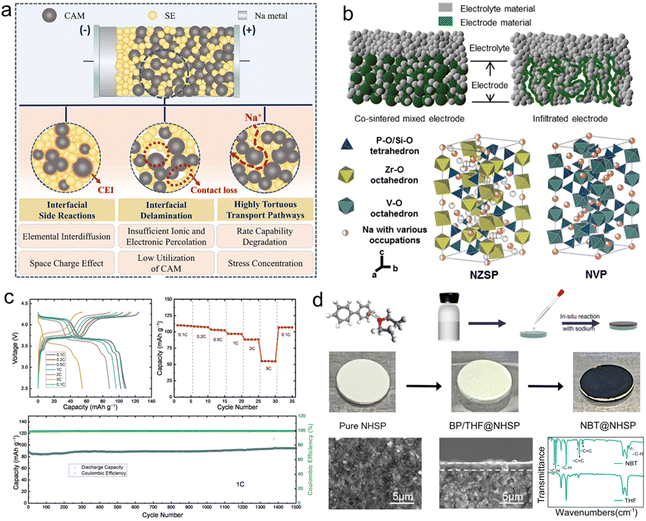 | ||
| Fig. 9 (a) Interfacial issues between cathodes and SSEs.223 Copyright 2025, Wiley-VCH. (b) Design and fabrication of chemically infiltrated SSSBs with the Na3V2P3O12 cathode and Na3.4Zr2Si2.4P0.6O12 electrolyte.225 Copyright 2019, Elsevier. (c) Electrochemical performance of NVPF in a hybrid SSE.227 Copyright 2023, Wiley-VCH. (d) Construction of NBT on an SSE.228 Copyright 2022, American Chemical Society. | ||
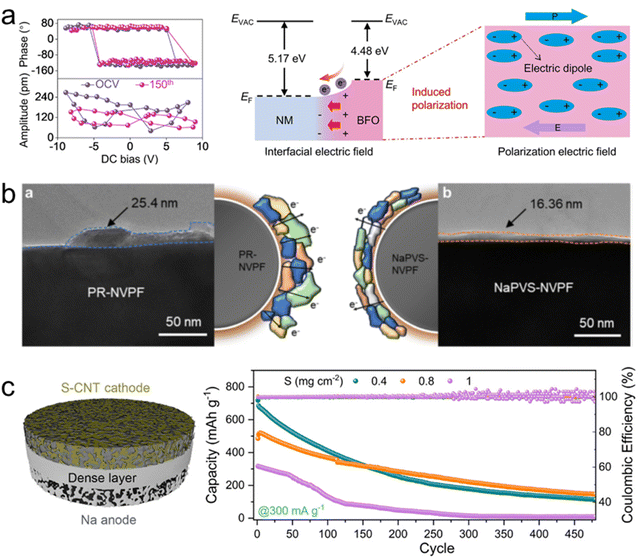 | ||
| Fig. 10 (a) Analysis of the synergistic ferroelectric–magnetic effect on the cathode–electrolyte interface.231 Copyright 2025, Wiley-VCH. (b) TEM images of pristine-NVPF and 5 wt% NaPVS-NVPF cathode particles after 50 cycles.232 Copyright 2025, Springer Nature. (c) Electrochemical performance of the monolithic electrolyte.234 Copyright 2021, American Chemical Society. | ||
5 Conclusions and outlook: from materials discovery to deployable solid-state sodium batteries
Despite substantial progress in the development of solid-state electrolytes for sodium batteries, the translation of promising materials properties into durable, high-performance full cells remains limited. This gap reflects not only unresolved materials challenges, but also a broader mismatch between laboratory-scale performance metrics and practical cell-level requirements. In particular, the widespread emphasis on achieving high bulk ionic conductivity has often obscured more critical factors, including interfacial resistance, mechanical integrity, critical current density, and long-term stability under realistic operating conditions.Looking forward, it is instructive to distinguish between challenges that are fundamentally materials-limited and those that are predominantly engineering-limited. For polymer-based electrolytes, low room-temperature conductivity and insufficient oxidative stability remain intrinsic materials constraints, suggesting that their near-term deployment is most realistic in moderately elevated temperature regimes or hybrid configurations. In contrast, many inorganic electrolytes, including sulfides and NASICON-type ceramics, already exhibit adequate ionic conductivity, but suffer from severe interfacial and processing challenges, indicating that further progress will depend primarily on interface engineering, microstructural control, and scalable fabrication strategies rather than continued bulk material optimization.
β″-Al2O3-based electrolytes provide an instructive example of this distinction. Their long-standing success in high-temperature molten sodium batteries demonstrates that exceptional ionic conductivity and chemical stability can be achieved when materials properties are well matched with operating conditions. However, attempts to translate these advantages to lower-temperature solid-state configurations reveal that interfacial impedance, mechanical brittleness, and thermal mismatch rapidly become dominant limitations. These observations highlight that the viability of solid-state sodium batteries is inherently application-dependent, and that direct extrapolation from lithium-based solid-state paradigms may be neither necessary nor optimal.
From a technological perspective, the most realistic deployment pathways for solid-state sodium batteries in the next 5–10 years are likely to emerge in application spaces where sodium chemistry offers intrinsic advantages. These include large-scale stationary energy storage, high-temperature or thermally managed systems, and cost-sensitive applications that can tolerate moderate energy density in exchange for improved safety, durability, and resource sustainability. In such contexts, hybrid solid-state architectures, graded interfaces, and composite electrolytes that balance mechanical compliance with ionic transport may offer more immediate impact than fully inorganic, room-temperature sodium-metal configurations.
More broadly, sodium-based solid-state systems should not be viewed merely as lower-cost analogues of lithium solid-state batteries. Instead, their future competitiveness will depend on identifying operating regimes and cell designs that exploit the distinct electrochemical, mechanical, and thermodynamic characteristics of sodium. Achieving this goal will require a shift in research emphasis—from maximizing isolated materials metrics toward integrated cell-level design principles that explicitly couple ion transport, interfacial chemistry, and mechanical stability. By aligning materials development with realistic application targets, solid-state sodium batteries may ultimately establish technological niches that are complementary to, rather than directly competitive with, their lithium counterparts.
Author contributions
Mingyue Wang: conceptualization, writing – original draft, supervision, writing – review & editing. Qing Zhong: writing – original draft, writing – review & editing. Yue Wang & Xu Liu: formal analysis, investigation. Dongyang Zhang: investigation, supervision. Shujiang Ding: conceptualization, funding acquisition, supervision.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analyzed as part of this review.Acknowledgements
This work is financially supported by the Key Projects of Shaanxi Province (2023GXLH-001), Xi'an Association for Science and Technology Young Talent Support Program Project (0959202513149) and Innovation Capability Support Plan of Shaanxi (no. 2022TD-27).References
- L. Miao, Z. Guo and L. Jiao, Insights into the design of mildly acidic aqueous electrolytes for improved stability of Zn anode performance in zinc-ion batteries, Energy Mater., 2023, 3, 300014 CAS.
- G. Tang, Y. Li, J. Wu, Q. Kang and L. Ma, Engineering heterostructured electrocatalysts for enhanced sulfur redox kinetics in metal-sulfur batteries, Nano Res. Energy, 2025, 4, e9120179 CrossRef.
- J. Xu, Q. Kang, B. Peng, Z. Zhuang, D. Wang and L. Ma, Engineering single-atom catalysts for sulfur electrochemistry in metal–sulfur batteries, J. Energy Chem., 2025, 106, 768–790 CrossRef CAS.
- B. Zhu, W. Zhang, Z. Jiang, J. Chen, Z. Li, J. Zheng, N. Wen, R. Chen, H. Yang, W. Zong, Y. Dai, C. Ye, Q. Zhang, T. Qiu, Y. Lai, J. Li and Z. Zhang, Sodium compensation: a critical technology for transforming batteries from sodium-starved to sodium-rich systems, Chem. Sci., 2024, 15, 14104–14121 RSC.
- Y. Zheng, L. Li, Y. Jiao, Y. Zhang, B. Peng, J. Xu and L. Ma, Recent advances in metal nitrides as highly-efficient electrocatalysts for lithium-sulfur batteries, Nano Res., 2025, 18, 94907542 CrossRef.
- L. Ma, Q. Wu, M. He, J. Wu, B. Peng, J. Xu, J. Liu and Z. Jin, Binary Metal Alloy Electrocatalyst Synergistically Accelerates the Bidirectional Polysulfide Conversions in Lithium–Sulfur Batteries, Nano Lett., 2025, 25, 1939–1947 CrossRef CAS PubMed.
- L. Ma, G. Zhu, Z. Wang, A. Zhu, K. Wu, B. Peng, J. Xu, D. Wang and Z. Jin, Long-Lasting Zinc–Iodine Batteries with Ultrahigh Areal Capacity and Boosted Rate Capability Enabled by Nickel Single-Atom Electrocatalysts, Nano Lett., 2023, 23, 5272–5280 CrossRef CAS PubMed.
- M. Wang, Y. Wang, D. Zhang, X. Kong, Q. Rong, L. Zhou, J. Wang and S. Ding, Recent advances in emerging three-dimensional carbon-based architectures for ion transfer and storage in advanced metal-ion batteries, J. Energy Storage, 2025, 128, 117173 CrossRef CAS.
- M. Wang, H. Zhang, J. Cui, S. Yao, X. Shen, T. J. Park and J.-K. Kim, Recent advances in emerging nonaqueous K-ion batteries: from mechanistic insights to practical applications, Energy Storage Mater., 2021, 39, 305–346 CrossRef.
- Y. Li, Y. Mei, R. Momen, B. Song, Y. Huang, X. Zhong, H. Ding, W. Deng, G. Zou, H. Hou and X. Ji, Boosting the interfacial dynamics and thermodynamics in polyanion cathode by carbon dots for ultrafast-charging sodium ion batteries, Chem. Sci., 2024, 15, 349–363 RSC.
- M. Wang, H. Zhao, Y. Wang, N. Li, X. Liu, D. Zhang, K. Xi and S. Ding, Melt-carbonization of liquid metal organic frameworks reconfigured hard carbon surface with boosted sodium storage, Sci. China Chem., 2025 DOI:10.1007/s11426-025-2880-y.
- D. Ruan, Z. Cui, J. Fan, D. Wang, Y. Wu and X. Ren, Recent advances in electrolyte molecular design for alkali metal batteries, Chem. Sci., 2024, 15, 4238–4274 RSC.
- Z. Xing, W. Zhao, B. Yu, Y. Wang, L. Zhou, P. Xiong, M. Chen and J. Zhu, Electrolyte Design Strategies for Aqueous Sodium-Ion Batteries: Progress and Prospects, Small, 2024, 20, 2405442 CrossRef CAS.
- X. Ma, Y. Liu, Y. Zhang and Y. Gong, Layered sodium titanate with a matched lattice: a single ion conductor in a solid-state sodium metal battery, Chem. Sci., 2023, 14, 13812–13824 RSC.
- T. Jin, X. Ji, P.-F. Wang, K. Zhu, J. Zhang, L. Cao, L. Chen, C. Cui, T. Deng, S. Liu, N. Piao, Y. Liu, C. Shen, K. Xie, L. Jiao and C. Wang, High-Energy Aqueous Sodium-Ion Batteries, Angew. Chem., Int. Ed., 2021, 60, 11943–11948 CrossRef CAS.
- H. Darjazi, M. Falco, F. Colò, L. Balducci, G. Piana, F. Bella, G. Meligrana, F. Nobili, G. A. Elia and C. Gerbaldi, Electrolytes for Sodium Ion Batteries: The Current Transition from Liquid to Solid and Hybrid systems, Adv. Mater., 2024, 36, 2313572 CrossRef CAS.
- C. Li, H. Xu, L. Ni, B. Qin, Y. Ma, H. Jiang, G. Xu, J. Zhao and G. Cui, Nonaqueous Liquid Electrolytes for Sodium-Ion Batteries: Fundamentals, Progress and Perspectives, Adv. Energy Mater., 2023, 13, 2301758 CrossRef CAS.
- J. Wu, Z. Song, Q. Huang, K. Zhu, D. Lin, X. Chen, X. Li, X. Chen, X. Li, L. Ma and Y. Chen, Electrolyte Engineering for Room-Temperature Sodium–Sulfur Batteries: Challenges, Strategies, and Future Perspectives, Adv. Mater., 2026, 38, e14290 CrossRef CAS PubMed.
- B. Jache and P. Adelhelm, Use of Graphite as a Highly Reversible Electrode with Superior Cycle Life for Sodium-Ion Batteries by Making Use of Co-Intercalation Phenomena, Angew. Chem., Int. Ed., 2014, 53, 10169–10173 CrossRef CAS PubMed.
- S. Tang, J. Li, Q. Yuan, C. Wang, T. Wang, W. Xiang, Y. Xiao, S. Xu and J. S. Yu, Anion screening in ether-based electrolytes for boosted sodium storage at low-temperature, Energy Storage Mater., 2025, 81, 104459 CrossRef.
- X. Yu, R. Chen, L. Gan, H. Li and L. Chen, Battery Safety: From Lithium-Ion to Solid-State Batteries, Engineering, 2023, 21, 9–14 CrossRef.
- X. Yi, H. Fu, A. M. Rao, Y. Zhang, J. Zhou, C. Wang and B. Lu, Safe electrolyte for long-cycling alkali-ion batteries, Nat. Sustain., 2024, 7, 326–337 CrossRef.
- X. Guo, Z. Xie, R. Wang, J. Luo, J. Chen, S. Guo, G. Tang, Y. Shi and W. Chen, Interface-Compatible Gel-Polymer Electrolyte Enabled by NaF-Solubility-Regulation toward All-Climate Solid-State Sodium Batteries, Angew. Chem., Int. Ed., 2024, 63, e202402245 CrossRef CAS PubMed.
- J. T. Kummer and N. Weber, Battery having a molten alkali metal anode and a molten sulfur cathode, US Patent, 3413150, 1968 Search PubMed.
- W. Hou, X. Guo, X. Shen, K. Amine, H. Yu and J. Lu, Solid electrolytes and interfaces in all-solid-state sodium batteries: Progress and perspective, Nano Energy, 2018, 52, 279–291 CrossRef CAS.
- H. Ahmad, K. T. Kubra, A. Butt, U. Nisar, F. J. Iftikhar and G. Ali, Recent progress, challenges, and perspectives in the development of solid-state electrolytes for sodium batteries, J. Power Sources, 2023, 581, 233518 CrossRef CAS.
- B. Pandit, M. Johansen, B. P. Andersen, C. S. Martínez-Cisneros, B. Levenfeld, D. B. Ravnsbæk and A. Varez, All-solid-state sodium-ion batteries operating at room temperature based on NASICON-type NaTi2(PO4)3 cathode and ceramic NASICON solid electrolyte: A complete in situ synchrotron X-ray study, Chem. Eng. J., 2023, 472, 144509 CrossRef CAS.
- Y. Shuai, J. Lou, X. Pei, C. Su, X. Ye, L. Zhang, Y. Wang, Z. Xu, P. Gao, S. He, Z. Wang and K. Chen, Constructing an In Situ Polymer Electrolyte and a Na-Rich Artificial SEI Layer toward Practical Solid-State Na Metal Batteries, ACS Appl. Mater. Interfaces, 2022, 14, 45382–45391 CrossRef CAS PubMed.
- Z. Yang, L. Chen, H. Jiang, X. Liang, J. Wei, Z. Xie, B. Tang and Z. Zhou, SnF2-Induced Highly Current-Tolerant Solid Electrolytes for Solid-State Sodium Batteries, Adv. Funct. Mater., 2023, 33, 2306558 CrossRef CAS.
- D. Li, C. Sun, C. Wang, J. Li, Z. Wang and H. Jin, Regulating Na/NASCION electrolyte interface chemistry for stable solid-state Na metal batteries at room temperature, Energy Storage Mater., 2023, 54, 403–409 CrossRef.
- P. Zou, C. Wang, Y. He and H. L. Xin, Making Plasticized Polymer Electrolytes Stable Against Sodium Metal for High-Energy Solid-State Sodium Batteries, Angew. Chem., Int. Ed., 2024, 63, e202319427 CrossRef CAS PubMed.
- T. Liu, P. Xiang, Y. Li, Z. Li, H. Sun, J. Yang, Z. Tian and X. Yao, In Situ Forming Na–Sn Alloy/Na2S Interface Layer for Ultrastable Solid State Sodium Batteries, Adv. Funct. Mater., 2024, 34, 2316528 CrossRef CAS.
- Q. Wang, C. Yu, L. Li, X. Liu, X. Zhang, G. Gao, Y. Wang and G. Li, Sc-doping in Na3Zr2Si2PO12 electrolytes enables preeminent performance of solid-state sodium batteries in a wide temperature range, Energy Storage Mater., 2023, 54, 135–145 CrossRef.
- J. Guo, F. Feng, S. Zhao, R. Wang, M. Yang, Z. Shi, Y. Ren, Z. Ma, S. Chen and T. Liu, Achieving Ultra-Stable All-Solid-State Sodium Metal Batteries with Anion-Trapping 3D Fiber Network Enhanced Polymer Electrolyte, Small, 2023, 19, 2206740 CrossRef CAS PubMed.
- J. Huang, K. Wu, G. Xu, M. Wu, S. Dou and C. Wu, Recent progress and strategic perspectives of inorganic solid electrolytes: fundamentals, modifications, and applications in sodium metal batteries, Chem. Soc. Rev., 2023, 52, 4933–4995 RSC.
- Y. Dong, P. Wen, H. Shi, Y. Yu and Z.-S. Wu, Solid-State Electrolytes for Sodium Metal Batteries: Recent Status and Future Opportunities, Adv. Funct. Mater., 2024, 34, 2213584 CrossRef CAS.
- Y. Zhu and Y. Mo, Materials Design Principles for Air-Stable Lithium/Sodium Solid Electrolytes, Angew. Chem., Int. Ed., 2020, 59, 17472–17476 CrossRef CAS PubMed.
- X. Feng, H. Fang, N. Wu, P. Liu, P. Jena, J. Nanda and D. Mitlin, Review of modification strategies in emerging inorganic solid-state electrolytes for lithium, sodium, and potassium batteries, Joule, 2022, 6, 543–587 CrossRef CAS.
- M. Jin, S. Cheng, Z. Yang, Y. Luo and Y. Guo, A novel high-voltage solid electrolyte of Na3B24H23 for 4 V all-solid-state sodium battery, Chem. Eng. J., 2023, 455, 140904 CrossRef CAS.
- Y. Hu, J. Fu, J. Xu, J. Luo, F. Zhao, H. Su, Y. Liu, X. Lin, W. Li, J. T. Kim, X. Hao, X. Yao, Y. Sun, J. Ma, H. Ren, M. Yang, Y. Huang and X. Sun, Superionic amorphous NaTaCl6 halide electrolyte for highly reversible all-solid-state Na-ion batteries, Matter, 2024, 7, 1018–1034 CrossRef CAS.
- X. Lin, Y. Zhao, C. Wang, J. Luo, J. Fu, B. Xiao, Y. Gao, W. Li, S. Zhang, J. Xu, F. Yang, X. Hao, H. Duan, Y. Sun, J. Guo, Y. Huang and X. Sun, A Dual Anion Chemistry-Based Superionic Glass Enabling Long-Cycling All-Solid-State Sodium-Ion Batteries, Angew. Chem., Int. Ed., 2024, 63, e202314181 CrossRef CAS PubMed.
- V. Thangadurai and B. Chen, Solid Li- and Na-Ion Electrolytes for Next Generation Rechargeable Batteries, Chem. Mater., 2022, 34, 6637–6658 CrossRef CAS.
- C. Li, R. Li, K. Liu, R. Si, Z. Zhang and Y.-S. Hu, NaSICON: A promising solid electrolyte for solid-state sodium batteries, Interdiscip. Mater., 2022, 1, 396–416 Search PubMed.
- J. A. S. Oh, X. Xu, Z. Zeng, K. Wang, N. Y. J. Tan, E. Kok, J. Huang and L. Lu, Thin NASICON Electrolyte to Realize High Energy Density Solid-State Sodium Metal Battery, Energy Environ. Mater., 2023, 6, e12472 CrossRef CAS.
- C. Heubner, B. Matthey, T. Lein, F. Wolke, T. Liebmann, C. Lämmel, M. Schneider, M. Herrmann and A. Michaelis, Insights into the electrochemical Li/Na-exchange in layered LiCoO2 cathode material, Energy Storage Mater., 2020, 27, 377–386 CrossRef.
- P. Yang, Z. Wu, Y. Liang, H. Chen, C. Lin, J. Qiu, J. Meng, Y. He and S. Zhang, Engineering ion transport in all-solid-state sodium-ion batteries: fundamentals, strategies, and perspectives, Prog. Mater. Sci., 2025, 154, 101503 CrossRef CAS.
- H. Chen, H. Zang, J. Du, H. Zhang, X. Xu, P. Zhang, X. Li, J. Han, X.-L. Li and Q.-C. Wang, Leveraging Lewis Acid–Modulated Polymer–Inorganic Interface for High–Performance Solid–State Sodium Batteries, Adv. Funct. Mater., 2025, 35, 2504177 CrossRef CAS.
- H. Li, J. Huang, Y. Wu, T. Wang, S. Wu, S. Li and T. Yan, Organic Electrolyte Composed of Strongly and Weakly Coordinating Molecules for Sodium-Ion Battery. A Molecular Dynamics Simulation Study, Energy Environ. Mater., 2026, e70147 Search PubMed.
- T. Deng, X. Ji, L. Zou, O. Chiekezi, L. Cao, X. Fan, T. R. Adebisi, H.-J. Chang, H. Wang, B. Li, X. Li, C. Wang, D. Reed, J.-G. Zhang, V. L. Sprenkle, C. Wang and X. Lu, Interfacial-engineering-enabled practical low-temperature sodium metal battery, Nat. Nanotechnol., 2022, 17, 269–277 CrossRef CAS PubMed.
- R. Fang, Y. Li, N. Wu, B. Xu, Y. Liu, A. Manthiram and J. B. Goodenough, Ultra-Thin Single-Particle-Layer Sodium Beta-Alumina-Based Composite Polymer Electrolyte Membrane for Sodium-Metal Batteries, Adv. Funct. Mater., 2023, 33, 2211229 CrossRef CAS.
- Z. Lu, H. Yang, G. Wu, P. Shan, H. Lin, P. He, J. Zhao, Y. Yang and H. Zhou, A “Liquid-In-Solid” Electrolyte for High-Voltage Anode-Free Rechargeable Sodium Batteries, Adv. Mater., 2024, 36, 2404569 CrossRef CAS PubMed.
- A. Nasu, T. Inaoka, F. Tsuji, K. Motohashi, A. Sakuda, M. Tatsumisago and A. Hayashi, Formation of Passivate Interphases by Na3BS3-Glass Solid Electrolytes in All-Solid-State Sodium-Metal Batteries, ACS Appl. Mater. Interfaces, 2022, 14, 24480–24485 CrossRef CAS PubMed.
- P. Song, S. Chen, J. Guo, J. Wu, Q. Lu, H. Xie, Q. Wang and T. Liu, Electrostatic Regulation of Na+ Coordination Chemistry for High-Performance All-Solid-State Sodium Batteries, Nano-Micro Lett., 2025, 18, 72 CrossRef PubMed.
- F. Chen, Y. Xie, Z. Yu, N. Li, Y. Qiao and X. Ding, Robust Janus-faced quasi-solid-state honeycomb-mimicking electrolytes for the fast transport and adequate supply of sodium ions, Chem. Sci., 2026 10.1039/D5SC08536E.
- S. Miao, Y. You, Z. Wang, Y. Zhao, H. Fan, X. Ai, Y. Cao and Y. Fang, Non-flammable electrolytes for high-safety sodium-ion batteries, Chem. Soc. Rev., 2025, 54, 9289–9316 RSC.
- X. Xu, Z. Ai, Z. Xiu, Y. Shao, Y. Wu and X. Hao, Enhancing sodium ionic conductivity: An interface bridging strategy for Na3Zr2Si2PO12 solid-state electrolyte, J. Energy Storage, 2025, 115, 116046 CrossRef CAS.
- H. Kwak, J.-S. Kim, D. Han, J. S. Kim, J. Park, G. Kwon, S.-M. Bak, U. Heo, C. Park, H.-W. Lee, K.-W. Nam, D.-H. Seo and Y. S. Jung, Boosting the interfacial superionic conduction of halide solid electrolytes for all-solid-state batteries, Nat. Commun., 2023, 14, 2459 CrossRef CAS PubMed.
- W. Wang, W. Yuan, Z. Zhao, D. Zou, P. Zhang, Z. Shi, J. Weng and P. Zhou, Enhanced ionic conductivity of Cu-doped NASICON solid electrolyte for solid-state sodium batteries, J. Electroanal. Chem., 2023, 937, 117405 CrossRef CAS.
- G. Liu, J. Yang, J. Wu, Z. Peng and X. Yao, Inorganic Sodium Solid Electrolytes: Structure Design, Interface Engineering and Application, Adv. Mater., 2024, 36, 2311475 CrossRef CAS PubMed.
- D. Lee, A. Kumar Kakarla, S. Sun, P. Joohyun Kim and J. Choi, Inorganic Solid-State Electrolytes for Solid-State Sodium Batteries: Electrolyte Design and Interfacial Challenges, Chemelectrochem, 2025, 12, e202400612 CrossRef CAS.
- Y. Yang, S. Yang, X. Xue, X. Zhang, Q. Li, Y. Yao, X. Rui, H. Pan and Y. Yu, Inorganic All-Solid-State Sodium Batteries: Electrolyte Designing and Interface Engineering, Adv. Mater., 2024, 36, 2308332 CrossRef CAS PubMed.
- M. Akbar, M. Kim, I. Moeez, A. H. Umar Bhatti, Y. H. Kim, J. Jeong, J.-Y. Kim, J.-H. Park, S. Yu and K. Y. Chung, Dendrite-free Sb-doped NASICON-type Na3Zr2Si2PO12 solid-electrolyte for stable solid-state sodium batteries, Chem. Eng. J., 2025, 504, 158860 CrossRef CAS.
- A. Tiwari, D. Meghnani, R. Mishra, R. K. Tiwari, A. Patel and R. K. Singh, Impact of sintering temperature and ascertaining hopping bottlenecks for ions in sodium superionic conducting electrolyte for sodium-ion batteries, J. Power Sources, 2023, 580, 233365 CrossRef CAS.
- Z. Li, J. Chen, Q. Fang, J. Fu, Y. Ren, X. Wang and X. Guo, Modulating Electric Double Layers via Polymer Electric Dipole Effects to Enhance Stability in Quasi-Solid-State Sodium Metal Batteries, Angew. Chem., Int. Ed., 2025, 64, e202505509 CrossRef CAS PubMed.
- F. Cheng, J. Hu, W. Zhang, B. Guo, P. Yu, X. Sun and J. Peng, Reviving ether-based electrolytes for sodium-ion batteries, Energy Environ. Sci., 2025, 18, 6874–6898 RSC.
- Z. Xue, D. He and X. Xie, Poly(ethylene oxide)-based electrolytes for lithium-ion batteries, J. Mater. Chem. A, 2015, 3, 19218–19253 RSC.
- M. Yahia, M. Enterría, C. Pozo-Gonzalo and N. Ortiz-Vitoriano, A New Quasi-Solid Polymer Electrolyte for Next-Generation Na–O2 Batteries: Unveiling the Potential of a Polyamide-Polyether System, Adv. Sci., 2025, 12, e04490 CrossRef CAS PubMed.
- T. Hou, D. Wang, B. Jiang, Y. Liu, J. Kong, Y. He, Y. Huang and H. Xu, Ion bridging enables high-voltage polyether electrolytes for quasi-solid-state batteries, Nat. Commun., 2025, 16, 962 CrossRef CAS PubMed.
- T. Famprikis, P. Canepa, J. A. Dawson, M. S. Islam and C. Masquelier, Fundamentals of inorganic solid-state electrolytes for batteries, Nat. Mater., 2019, 18, 1278–1291 CrossRef CAS PubMed.
- L. Chen, Y. Li, S.-P. Li, L.-Z. Fan, C.-W. Nan and J. B. Goodenough, PEO/garnet composite electrolytes for solid-state lithium batteries: From “ceramic-in-polymer” to “polymer-in-ceramic”, Nano Energy, 2018, 46, 176–184 CrossRef CAS.
- H. Wang, Y. Sun, Q. Liu, Z. Mei, L. Yang, L. Duan and H. Guo, An asymmetric bilayer polymer-ceramic solid electrolyte for high-performance sodium metal batteries, J. Energy Chem., 2022, 74, 18–25 CrossRef CAS.
- P. Liu, L. Miao, Z. Sun, X. Chen, Y. Si, Q. Wang and L. Jiao, Inorganic–Organic Hybrid Multifunctional Solid Electrolyte Interphase Layers for Dendrite-Free Sodium Metal Anodes, Angew. Chem., Int. Ed., 2023, 62, e202312413 CrossRef CAS PubMed.
- X. Zheng, C. Shi, Z. Li, Z. Zhang, L. Yang, Q. Fang and J. Tang, In-Situ formation Inorganic/Organic solid electrolyte interphase and sodium affinity sites for improved sodium metal anodes, Chem. Eng. J., 2025, 506, 159713 CrossRef CAS.
- Y. Lu, L. Li, Q. Zhang, Z. Niu and J. Chen, Electrolyte and Interface Engineering for Solid-State Sodium Batteries, Joule, 2018, 2, 1747–1770 CrossRef CAS.
- J. Evans, C. A. Vincent and P. G. Bruce, Electrochemical measurement of transference numbers in polymer electrolytes, Polymer, 1987, 28, 2324–2328 CrossRef CAS.
- C. Sun, J. Liu, Y. Gong, D. P. Wilkinson and J. Zhang, Recent advances in all-solid-state rechargeable lithium batteries, Nano Energy, 2017, 33, 363–386 CrossRef CAS.
- Z.-H. Luo, Y.-F. Du, Z.-J. Jiang, D. Zhang, N.-L. Shen, J.-X. Guo, F. Jiang, X. Liu, X. Zhang, X.-G. Qi, X. Shen, Y. Ma, Y. Zhong, J. He, Y. Wu and X.-B. Cheng, High-Entropy Inorganic Solid Electrolyte Interphase Enables Thermally Safe Sodium-Ion Battery with Deep Sodium Storage, ACS Nano, 2025, 19, 39946–39960 CrossRef CAS.
- S. Lou, F. Zhang, C. Fu, M. Chen, Y. Ma, G. Yin and J. Wang, Interface Issues and Challenges in All-Solid-State Batteries: Lithium, Sodium, and Beyond, Adv. Mater., 2021, 33, 2000721 CrossRef CAS PubMed.
- C. Delmas, D. Carlier and M. Guignard, The Layered Oxides in Lithium and Sodium-Ion Batteries: A Solid-State Chemistry Approach, Adv. Energy Mater., 2021, 11, 2001201 CrossRef CAS.
- Q. Zhou, J. Ma, S. Dong, X. Li and G. Cui, Intermolecular Chemistry in Solid Polymer Electrolytes for High-Energy-Density Lithium Batteries, Adv. Mater., 2019, 31, 1902029 CrossRef CAS.
- L. Xiang, X. Li, J. Xiao, L. Zhu and X. Zhan, Interface issues and challenges for NASICON-based solid-state sodium-metal batteries, Adv. Powder Mater., 2024, 3, 100181 CrossRef.
- Y. Liu, H. Mao, R. Bai, S. Weng, Q. Zhang, X. Rong, X. Chen, C. Zhang, S. Han, F. Ding, X. Wang, Y. Lu, J. Zhao, F. Wei, L. Chen and Y.-S. Hu, Designing an isotropic epilayer for stable 4.2 V solid-state Na batteries, Nat. Energy, 2025 DOI:10.1038/s41560-025-01857-y.
- Y. Zhao, C. Wang, Y. Dai and H. Jin, Homogeneous Na+ transfer dynamic at Na/Na3Zr2Si2PO12 interface for all solid-state sodium metal batteries, Nano Energy, 2021, 88, 106293 CrossRef CAS.
- Y. Lu, C.-Z. Zhao, H. Yuan, X.-B. Cheng, J.-Q. Huang and Q. Zhang, Critical Current Density in Solid-State Lithium Metal Batteries: Mechanism, Influences, and Strategies, Adv. Funct. Mater., 2021, 31, 2009925 CrossRef CAS.
- K. Takada, Progress and prospective of solid-state lithium batteries, Acta Mater., 2013, 61, 759–770 CrossRef CAS.
- A. Banerjee, X. Wang, C. Fang, E. A. Wu and Y. S. Meng, Interfaces and Interphases in All-Solid-State Batteries with Inorganic Solid Electrolytes, Chem. Rev., 2020, 120, 6878–6933 CrossRef CAS PubMed.
- S. Qu, T. Niu, X. Qiao, Y. Shen, G. Cai, X. Wang, Y. Wang, Z. Zhou, S. Zhang, Z. Zhang, G. Li, G. Cai and J. Sun, Dual Optimization of Electrolyte and Interface in Na-β″-Al2O3 via Ga3+ Doping for Advanced Solid-State Sodium Batteries, Adv. Mater., 2025, 37, e03562 CrossRef CAS PubMed.
- P. Wang, Y. Niu, K. Zhang, W. Hou, X. Yao and Y. Xu, Adapting Crystal Structure and Grain Boundaries through Sm3+ Doping in Na3Zr2Si2PO4 for Boosting Applicability in Sodium Solid-State Batteries, ACS Appl. Mater. Interfaces, 2024, 16, 2877–2887 CrossRef CAS PubMed.
- X. Bai, Y. Meng, H. Chen, D. Zhou and L.-Z. Fan, Regulation of Carrier Transport at Grain Boundaries of Na3Zr2Si2PO12 Solid Electrolyte with Ionic Conductor Filler for Superior Solid-State Sodium Batteries, Adv. Funct. Mater., 2025, e19793 CrossRef.
- J. Liu, L. Pei and J. Li, Enhancing interfacial stability between NASICON-type solid-state electrolytes and lithium metal anode via mixed ionic-electronic conductive interlayer engineering, Chem. Eng. J., 2025, 519, 165208 CrossRef CAS.
- P. Hu, Y. Zhang, X. Chi, K. Kumar Rao, F. Hao, H. Dong, F. Guo, Y. Ren, L. C. Grabow and Y. Yao, Stabilizing the Interface between Sodium Metal Anode and Sulfide-Based Solid-State Electrolyte with an Electron-Blocking Interlayer, ACS Appl. Mater. Interfaces, 2019, 11, 9672–9678 CrossRef CAS.
- F. Li, M. Hou, L. Zhao, D. Zhang, B. Yang and F. Liang, Electrolyte and interface engineering for solid-state sodium batteries, Energy Storage Mater., 2024, 65, 103181 CrossRef.
- G. Du, S. Wang, Z. Tong, X. Ji, X. Wei, Q. Zha, T. Zhai and H. Li, Dual thermal-stimulated self-adhesive mixed-phase interface to enable ultra-long cycle life of solid-state sodium metal batteries, Energy Environ. Sci., 2025, 18, 3689–3698 RSC.
- J. Hou, T. Zhu, G. Wang, R. Cheacharoen, W. Sun, X. Lei, Q. Yuan, D. Sun and J. Zhao, Composite electrolytes and interface designs for progressive solid-state sodium batteries, Carbon Energy, 2024, 6, e628 CrossRef CAS.
- Z. L. Dong, Y. Gan, V. Martins, X. Wang, B. Fu, E. Jin, Y. Gao, Y. Hu, X. Lin, Y. Yuan, C. Turner, X. Pang, H. Abdolvand, Y. Huang, T.-K. Sham and Y. Zhao, Novel Sulfide-Chloride Solid-State Electrolytes with Tunable Anion Ratio for Highly Stable Solid-State Sodium-Ion Batteries, Adv. Mater., 2025, 37, 2503107 CrossRef CAS PubMed.
- C. Liu, S. Jia, T. Yang, J. Liu, X. Zhou, Z. Wang, H. Dong, Z. Shi, Y. Zhang and Z. Chen, Scalable and Ultrathin Dual Entangled Network Polymer Electrolytes for Safe Solid-State Sodium Batteries, Angew. Chem., Int. Ed., 2025, 64, e202505938 CrossRef CAS.
- C. Zhao, L. Liu, X. Qi, Y. Lu, F. Wu, J. Zhao, Y. Yu, Y.-S. Hu and L. Chen, Solid-State Sodium Batteries, Adv. Energy Mater., 2018, 8, 1703012 CrossRef.
- A. Wang, Q. Zhang, W. Li, K. Zhang, C. Dong, Y. Wang, X. Xu, Y. Liu, Z. Ren, F. Shen, Z. Song and X. Han, Electrochemical–Mechanical Evolution of Dendrites and Cracks in Na3Zr2Si2PO12 Ceramic Solid Electrolytes, Adv. Energy Mater., 2025, 15, e02156 CrossRef CAS.
- W. Shan, J. Chen, X. Wei, B. V. R. Chowdari, X. Wu and Z. Wen, Enhancing the Dendritic Tolerance of NASICON-Based Electrolytes by Grain Boundary Engineering, ACS Appl. Mater. Interfaces, 2025, 17, 56144–56152 CrossRef PubMed.
- S. Yang, N. Li, E. Zhao, C. Wang, J. He, X. Xiao, D. Fang, Q. Ni, X. Han, X. Xue, L. Chen, N. Li, J. Li, T. Guo, Y. Su and H. Jin, Imaging dendrite growth in solid-state sodium batteries using fluorescence tomography technology, Sci. Adv., 2024, 10, eadr0676 CrossRef CAS PubMed.
- L. Geng, D. Xue, J. Yao, Q. Dai, H. Sun, D. Zhu, Z. Rong, R. Fang, X. Zhang, Y. Su, J. Yan, S. J. Harris, S. Ichikawa, L. Zhang, Y. Tang, S. Zhang and J. Huang, Morphodynamics of dendrite growth in alumina based all solid-state sodium metal batteries, Energy Environ. Sci., 2023, 16, 2658–2668 RSC.
- S. Zhao, H. Che, S. Chen, H. Tao, J. Liao, X.-Z. Liao and Z.-F. Ma, Research progress on the solid electrolyte of solid-state sodium-ion batteries, Electrochem. Energy Rev., 2024, 7, 3 CrossRef CAS.
- C. Zhao, L. Liu, X. Qi, Y. Lu, F. Wu, J. Zhao, Y. Yu, Y. S. Hu and L. Chen, Solid-state sodium batteries, Adv. Energy Mater., 2018, 8, 1703012 CrossRef.
- Z.-Y. Li, Z. Li, J.-L. Fu and X. Guo, Sodium-ion conducting polymer electrolytes, Rare Met., 2023, 42, 1–16 CrossRef.
- Y. L. Ni'Mah, M.-Y. Cheng, J. H. Cheng, J. Rick and B.-J. Hwang, Solid-state polymer nanocomposite electrolyte of TiO2/PEO/NaClO4 for sodium ion batteries, J. Power Sources, 2015, 278, 375–381 CrossRef.
- H. Chen, H. Zang, J. Du, H. Zhang, X. Xu, P. Zhang, X. Li, J. Han, X. L. Li and Q. C. Wang, Leveraging Lewis Acid–Modulated Polymer–Inorganic Interface for High–Performance Solid–State Sodium Batteries, Adv. Funct. Mater., 2025, 2504177 CrossRef CAS.
- B. Yi, Z. Wei, S. Yao, S. Zhao, Z. Gao, S. Savilov, G. Chen, Z. X. Shen and F. Du, Challenges and prospectives of sodium-containing solid-state electrolyte materials for rechargeable metal batteries, Mater. Sci. Eng. R Rep., 2025, 163, 100949 Search PubMed.
- J. Hou, T. Zhu, G. Wang, R. Cheacharoen, W. Sun, X. Lei, Q. Yuan, D. Sun and J. Zhao, Composite electrolytes and interface designs for progressive solid-state sodium batteries, Carbon Energy, 2024, 6, e628 CrossRef CAS.
- S. Wang, X. Lu, T. Zhang, Y. Kang, Y. Shi, Y. Tian, Z. Chen, H. Wang, Q. Ji and W. Huang, Nonflammable Polyfluorides-Anchored Quasi-Solid Electrolytes by Chemical-Crosslinking for High-Safety Sodium Metal Battery, Adv. Funct. Mater., 2025, 2507147 CrossRef CAS.
- J. Yang, H. Zhang, Q. Zhou, H. Qu, T. Dong, M. Zhang, B. Tang, J. Zhang and G. Cui, Safety-enhanced polymer electrolytes for sodium batteries: recent progress and perspectives, ACS Appl. Mater. Interfaces, 2019, 11, 17109–17127 CrossRef CAS PubMed.
- X. Wang, C. Zhang, M. Sawczyk, J. Sun, Q. Yuan, F. Chen, T. C. Mendes, P. C. Howlett, C. Fu and Y. Wang, Ultra-stable all-solid-state sodium metal batteries enabled by perfluoropolyether-based electrolytes, Nat. Mater., 2022, 21, 1057–1065 CrossRef CAS PubMed.
- X. Dong, X. Liu, H. Li, S. Passerini and D. Bresser, Single-ion conducting polymer electrolyte for superior sodium-metal batteries, Angew. Chem., 2023, 135, e202308699 CrossRef.
- X. Dong, Y. Zhang, Z. You, Y. Chen, X. Wu and Z. Wen, Polyanion-Induced Single Na-Ion Polymer Electrolytes for Ultra-Stable Sodium Metal Batteries, Adv. Funct. Mater., 2024, 34, 2405931 CrossRef CAS.
- Y. Zhong, Y. Wang, L. Ma, P. He, J. Fang and J. Li, Endogenous triboelectricity enhancing the piezoelectric performance of fluorinated polyimide foam for sound detection, Nano Energy, 2025, 141, 111129 CrossRef CAS.
- G. Feuillade and P. Perche, Ion-conductive macromolecular gels and membranes for solid lithium cells, J. Appl. Electrochem., 1975, 5, 63–69 CrossRef CAS.
- S. Wang, Y. Jiang and X. Hu, Ionogel-based membranes for safe lithium/sodium batteries, Adv. Mater., 2022, 34, 2200945 CrossRef CAS PubMed.
- M. Menisha, S. Senavirathna, K. Vignarooban, N. Iqbal, H. Pitawala and A. Kannan, Synthesis, electrochemical and optical studies of poly (ethylene oxide) based gel-polymer electrolytes for sodium-ion secondary batteries, Solid State Ionics, 2021, 371, 115755 CrossRef CAS.
- M. L. Lehmann, G. Yang, J. Nanda and T. Saito, Well-designed crosslinked polymer electrolyte enables high ionic conductivity and enhanced salt solvation, J. Electrochem. Soc., 2020, 167, 070539 CrossRef.
- S. Janakiraman, A. Surendran, R. Biswal, S. Ghosh, S. Anandhan and A. Venimadhav, Electrospun electroactive polyvinylidene fluoride-based fibrous polymer electrolyte for sodium ion batteries, Mater. Res. Express, 2019, 6, 086318 Search PubMed.
- Y. Zhong, L. Ma, P. He, Q. Zhao and J. Li, Hybrid Piezo/Triboelectric Nanogenerators Based on Nanofibrous Aerogels for Energy Harvesting and Respiratory Monitoring, ACS Sens., 2025, 10, 8839–8851 CrossRef CAS PubMed.
- J. Zheng, W. Li, X. Liu, J. Zhang, X. Feng and W. Chen, Progress in gel polymer electrolytes for sodium-ion batteries, Energy Environ. Mater., 2023, 6, e12422 CrossRef CAS.
- H. Yang, Y. Chen, W. Tian, S. Yuan, P. Liu, Q. Wang, T. Jin and L. Jiao, Co-sustained Release Strategy of Nonflammable Gel Polymer Electrolytes Enables Long-Life Sodium Metal Batteries, Angew. Chem., Int. Ed., 2025, e202506349 CAS.
- W. Liang, K. Zhao, L. Ouyang, M. Zhu and J. Liu, A review of functional group selection and design strategies for gel polymer electrolytes for metal batteries, Mater. Sci. Eng. R Rep., 2025, 164, 100973 CrossRef.
- S. Zhao, Y. Shen, H. Che, M. Jabeen, C. Lu, X. Z. Liao and Z. F. Ma, Cellulose Derivative and Polyionic Liquid Crosslinked Network Gel Electrolytes for Sodium Metal Quasi-Solid-State Batteries, Adv. Funct. Mater., 2025, 35, 2422162 CrossRef CAS.
- Y. Xu, D. Chen, L. Zheng, H. Fu, X. Ye, Y. Zhang, Z. Chen, Y. Zhong, X. Wang and J. Tu, Ester-ether Synergistic Interactions Regulating the Solvation Structure of Gel Polymer Electrolytes Enables Stable Sodium Metal Batteries, Energy Storage Mater., 2025, 104533 Search PubMed.
- S. Zhao, Y. Shen, H. Che, X.-Z. Liao, X.-M. Zhang and Z.-F. Ma, Ionic conductive membrane suitable for sodium metal batteries, J. Membr. Sci., 2024, 702, 122792 CrossRef CAS.
- M. Akbar, I. Moeez, A. H. U. Bhatti, Y. H. Kim, M. Kim, J.-Y. Kim, J. Jeong, J. H. Park and K. Y. Chung, Antimony-doped NASICON-type solid electrolyte with homogeneous sodium-ion flux for high-temperature solid-state sodium batteries, Chem. Eng. J., 2025, 164300 CrossRef CAS.
- G. Rankin and H. Merwin, The Ternary System CaO-Al2O3-MgO, J. Am. Chem. Soc., 1916, 38, 568–588 CrossRef CAS.
- X. Lu, G. Xia, J. P. Lemmon and Z. Yang, Advanced materials for sodium-beta alumina batteries: Status, challenges and perspectives, J. Power Sources, 2010, 195, 2431–2442 CrossRef CAS.
- X.-S. Li, J. Liang, X. Cao, S.-Y. Zhu, Y.-F. Bai, J.-W. Sun, H.-B. Luo and J. Kong, Research progress of inorganic solid electrolyte materials for all-solid-state sodium-ion batteries, Rare Met., 2025, 44, 2871–2899 CrossRef CAS.
- Y. Yang, S. Yang, X. Xue, X. Zhang, Q. Li, Y. Yao, X. Rui, H. Pan and Y. Yu, Inorganic all-solid-state sodium batteries: electrolyte designing and interface engineering, Adv. Mater., 2024, 36, 2308332 CrossRef CAS.
- A. V. Virkar and R. S. Gordon, Fracture Properties of Polycrystalline Lithia-Stabilized β “-Alumina, J. Am. Ceram. Soc., 1977, 60, 58–61 CrossRef CAS.
- A. P. de Kroon, G. W. Schaefer and F. Aldinger, Direct Synthesis of Binary K-. beta.-and K-. beta.''-Alumina. 1. Phase Relations and Influence of Precursor Chemistry, Chem. Mater., 1995, 7, 878–887 Search PubMed.
- Z. Wang, X. Feng, T. Zhang, Z. Xie, F. Song and Y. Li, Preparation and characterization of CoO-doped and Li2O-stabilized Na-β″-Al2O3 solid electrolyte via a solid-state reaction method, Ceram. Int., 2020, 46, 24668–24673 Search PubMed.
- G. Chen, J. Lu, X. Zhou, L. Chen and X. Jiang, Solid-state synthesis of high performance Na-β″-Al2O3 solid electrolyte doped with MgO, Ceram. Int., 2016, 42, 16055–16062 CrossRef CAS.
- D. Xu, H. Jiang, Y. Li, L. Li, M. Li and O. Hai, The mechanical and electrical properties of Nb2 O5 doped Na-β″-Al2O3 solid electrolyte, Eur. Phys. J. Appl. Phys., 2016, 74, 10901 Search PubMed.
- S.-T. Lee, D.-H. Lee, J.-S. Kim and S.-K. Lim, Influence of Fe and Ti addition on properties of Na+-β/β″-alumina solid electrolytes, Met. Mater. Int., 2017, 23, 246–253 CrossRef CAS.
- G. Chen, J. Lu, L. Li, L. Chen and X. Jiang, Microstructure control and properties of β″-Al2O3 solid electrolyte, J. Alloys Compd., 2016, 673, 295–301 CrossRef CAS.
- H. Li, X. Jiang, J. Zhang and J. Zhang, Effect of excess Na2O on the sintering behavior and properties of sodium beta-alumina solid electrolyte, Mater. Today Commun., 2023, 34, 105203 Search PubMed.
- K. Zhao, Y. Liu, S. Zeng, J. Yang, Y. Liu, Z. Zhan and L. Song, Preparation and characterization of a ZrO2–TiO2-co-doped Na-β′′-Al2O3 ceramic thin film, Ceram. Int., 2016, 42, 8990–8996 CrossRef CAS.
- H. Chen, T. Wang, Z. Wang, A. Li, S. Cen, Z. Mao, J. Chen, X. Wang and C. Dong, Unlocking Solid-State Sodium–Metal Batteries at −15 °C by Electrolyte Optimization and Interface Regulation, ACS Appl. Mater. Interfaces, 2024, 17, 1119–1126 CrossRef PubMed.
- L. Ghadbeigi, A. Szendrei, P. Moreno, T. D. Sparks and A. V. Virkar, Synthesis of iron-doped Na-β″-alumina+ yttria-stabilized zirconia composite electrolytes by a vapor phase process, Solid State Ionics, 2016, 290, 77–82 CrossRef CAS.
- S.-J. Shan, L.-P. Yang, X.-M. Liu, X.-L. Wei, H. Yang and X.-D. Shen, Preparation and characterization of TiO2 doped and MgO stabilized Na–β″-Al2O3 electrolyte via a citrate sol–gel method, J. Alloys Compd., 2013, 563, 176–179 Search PubMed.
- D.-H. Lee, S.-T. Lee, J.-S. Kim and S.-K. Lim, Analysis of properties of partially stabilized zirconia-doped Na+-beta-alumina prepared by calcining-cum-sintering process, Mater. Res. Bull., 2017, 96, 143–148 CrossRef CAS.
- D. Butts, J. R. Schoiber, C. Choi, G. N. J. Redhammer, N. Hüsing, S. Donne and B. Dunn, Fe-Substituted Sodium β″-Al2O3 as a High-Rate Na-Ion Electrode, Chem. Mater., 2021, 33, 6136–6145 Search PubMed.
- S.-T. Lee, D.-H. Lee, S.-M. Lee, S.-S. Han, S.-H. Lee and S.-K. Lim, Effects of calcium impurity on phase relationship, ionic conductivity and microstructure of Na+-β/β″-alumina solid electrolyte, Bull. Mater. Sci., 2016, 39, 729–735 Search PubMed.
- G. Zhang, Z. Wen, X. Wu, J. Zhang, G. Ma and J. Jin, Sol–gel synthesis of Mg2+ stabilized Na-β″/β-Al2O3 solid electrolyte for sodium anode battery, J. Alloys Compd., 2014, 613, 80–86 Search PubMed.
- C. Zhu and J. Xue, Structure and properties relationships of beta-Al2O3 electrolyte materials, J. Alloys Compd., 2012, 517, 182–185 CrossRef CAS.
- J. Lin, Z. Wen, X. Wang, S. Song and Y. Liu, Mechanochemical synthesis of Na-β/β″-Al2O3, J. Solid State Electrochem., 2010, 14, 1821–1827 CrossRef CAS.
- J. Wang, X.-P. Jiang, X.-L. Wei, H. Yang and X.-D. Shen, Synthesis of Na-β″-Al2O3 electrolytes by microwave sintering precursors derived from the sol–gel method, J. Alloys Compd., 2010, 497, 295–299 Search PubMed.
- S. C. Ligon, G. Blugan, M.-C. Bay, C. Battaglia, M. V. Heinz and T. Graule, Performance analysis of Na-β″-Al2O3/YSZ solid electrolytes produced by conventional sintering and by vapor conversion of α-Al2O3/YSZ, Solid State Ionics, 2020, 345, 115169 CrossRef CAS.
- D. Li, X. Wang, Q. Guo, X. Yu, S. Cen, H. Ma, J. Chen, D. Wang, Z. Mao and C. Dong, Atomically bonding Na anodes with metallized ceramic electrolytes by ultrasound welding for high-energy/power solid-state sodium metal batteries, Carbon Energy, 2023, 5, e299 Search PubMed.
- K. Li, Y. Yang, X. Zhang and S. Liang, Highly oriented β″-alumina ceramics with excellent ionic conductivity and mechanical performance obtained by spark plasma sintering technique, J. Mater. Sci., 2020, 55, 8435–8443 CrossRef CAS.
- K. Koganei, T. Oyama, M. Inada, N. Enomoto and K. Hayashi, C-axis oriented β″-alumina ceramics with anisotropic ionic conductivity prepared by spark plasma sintering, Solid State Ionics, 2014, 267, 22–26 Search PubMed.
- D. Xu, H. Jiang, M. Li, O. Hai and Y. Zhang, Synthesis and characterization of Y2O3 doped Na–β″-Al2O3 solid electrolyte by double zeta process, Ceram. Int., 2015, 41, 5355–5361 Search PubMed.
- R. Li, D. Jiang, P. Du, C. Yuan, X. Cui, Q. Tang, J. Zheng, Y. Li, K. Lu, X. Ren, S. Gao and X. Zhan, Negating Na‖Na3Zr2Si2PO12 interfacial resistance for dendrite-free and “Na-less” solid-state batteries, Chem. Sci., 2022, 13, 14132–14140 RSC.
- C. Lou, W. Zhang, J. Liu, Y. Gao, X. Sun, J. Fu, Y. Shi, L. Xu, H. Luo, Y. Chen, X. Gao, X. Kuang, L. Su and M. Tang, The glass phase in the grain boundary of Na3Zr2Si2PO12, created by gallium modulation, Chem. Sci., 2024, 15, 3988–3995 Search PubMed.
- A. K. Dey, S. S. Selvasundarasekar, S. Kundu, A. K. Mandal, A. Das and S. K. Pramanik, 2D organic nanosheets of self-assembled guanidinium derivative for efficient single sodium-ion conduction: rationalizing morphology editing and ion conduction, Chem. Sci., 2024, 15, 16321–16330 Search PubMed.
- K. B. Hueso, M. Armand and T. Rojo, High temperature sodium batteries: status, challenges and future trends, Energy Environ. Sci., 2013, 6, 734–749 RSC.
- C. Bao, B. Wang, P. Liu, H. Wu, Y. Zhou, D. Wang, H. Liu and S. Dou, Solid electrolyte interphases on sodium metal anodes, Adv. Funct. Mater., 2020, 30, 2004891 Search PubMed.
- Z. Gao, Y. Bai, J. Feng, J. Yang, P. Liu, H. Yuan, X. Guan, F. R. Wang and Y. Huang, Controlling sodium dendrite growth via grain boundaries in Na3Zr2Si2PO12 electrolyte, Adv. Energy Mater., 2024, 14, 2304488 CrossRef CAS.
- Y. Li, M. Li, Z. Sun, Q. Ni, H. Jin and Y. Zhao, Recent advance on NASICON electrolyte in solid-state sodium metal batteries, Energy Storage Mater., 2023, 56, 582–599 CrossRef.
- Y. Shao, G. Zhong, Y. Lu, L. Liu, C. Zhao, Q. Zhang, Y.-S. Hu, Y. Yang and L. Chen, A novel NASICON-based glass-ceramic composite electrolyte with enhanced Na-ion conductivity, Energy Storage Mater., 2019, 23, 514–521 Search PubMed.
- Y. Li, C. Sun, Z. Sun, M. Li, H. Jin and Y. Zhao, Boosting Na-O Affinity in Na3Zr2Si2PO12 Electrolyte Promises Highly Rechargeable Solid-State Sodium Batteries, Adv. Funct. Mater., 2024, 34, 2403937 CrossRef CAS.
- B. Wei, S. Huang, X. Wang, M. Liu, C. Huang, R. Liu and H. Jin, Intermediate phase induced in situ self-reconstruction of amorphous NASICON for long-life solid-state sodium metal batteries, Energy Environ. Sci., 2025, 18, 831–840 RSC.
- B. Xun, J. Wang, Y. Sato, S. Jia, S. Ohno, H. Akamatsu and K. Hayashi, Bifunctional Al dopant for enhancing bulk and grain boundary conductivities in sodium ion conducting NASICON ceramics, Adv. Energy Mater., 2025, 15, 2402891 Search PubMed.
- S. Liu, C. Zhou, Y. Wang, W. Wang, Y. Pei, J. Kieffer and R. M. Laine, Ce-substituted nanograin Na3Zr2Si2PO12 prepared by LF-FSP as sodium-ion conductors, ACS Appl. Mater. Interfaces, 2019, 12, 3502–3509 CrossRef PubMed.
- D. Zuo, L. Yang, Z. Zou, S. Li, Y. Feng, S. J. Harris, S. Shi and J. Wan, Ultrafast synthesis of NASICON solid electrolytes for sodium-metal batteries, Adv. Energy Mater., 2023, 13, 2301540 Search PubMed.
- H. Liu, Y. Xing, N. Chen, J. Wu, Y. Li and C. Zhang, Engineering the NASICON Electrolyte/Na Anode Interface by Tuning the Phase of Electrolyte for Solid-State Sodium Battery, Chem. Mater., 2023, 35, 8686–8694 Search PubMed.
- M. Akbar, M. Kim, I. Moeez, A. H. U. Bhatti, Y. H. Kim, J. Jeong, J.-Y. Kim, J.-H. Park, S. Yu and K. Y. Chung, Dendrite-free Sb-doped NASICON-type Na3Zr2Si2PO12 solid-electrolyte for stable solid-state sodium batteries, Chem. Eng. J., 2025, 504, 158860 CrossRef CAS.
- H. Nguyen, A. Banerjee, X. Wang, D. Tan, E. A. Wu, J.-M. Doux, R. Stephens, G. Verbist and Y. S. Meng, Single-step synthesis of highly conductive Na3PS4 solid electrolyte for sodium all solid-state batteries, J. Power Sources, 2019, 435, 126623 CrossRef CAS.
- L. Zhang, D. Zhang, K. Yang, X. Yan, L. Wang, J. Mi, B. Xu and Y. Li, Vacancy-contained tetragonal Na3SbS4 superionic conductor, Adv. Sci., 2016, 3, 1600089 CrossRef PubMed.
- Z. Zhang, E. Ramos, F. Lalère, A. Assoud, K. Kaup, P. Hartman and L. F. Nazar, Na 11 Sn 2 PS 12: a new solid state sodium superionic conductor, Energy Environ. Sci., 2018, 11, 87–93 RSC.
- A. Hayashi, K. Noi, A. Sakuda and M. Tatsumisago, Superionic glass-ceramic electrolytes for room-temperature rechargeable sodium batteries, Nat. Commun., 2012, 3, 856 CrossRef PubMed.
- Z. L. Dong, Y. Yuan, V. Martins, E. Jin, Y. Gan, X. Lin, Y. Gao, X. Hao, Y. Guan and J. Fu, Structural insight and modulating of sulfide-based solid-state electrolyte for high-performance solid-state sodium sulfur batteries, Nano Energy, 2024, 128, 109871 CrossRef CAS.
- A. Nasu, T. Otono, T. Takayanagi, M. Deguchi, A. Sakuda, M. Tatsumisago and A. Hayashi, Utilizing reactive polysulfides flux Na2Sx for the synthesis of sulfide solid electrolytes for all-solid-state sodium batteries, Energy Storage Mater., 2024, 67, 103307 CrossRef.
- A. Hayashi, N. Masuzawa, S. Yubuchi, F. Tsuji, C. Hotehama, A. Sakuda and M. Tatsumisago, A sodium-ion sulfide solid electrolyte with unprecedented conductivity at room temperature, Nat. Commun., 2019, 10, 5266 CrossRef CAS PubMed.
- J. Königsreiter, B. Gadermaier and H. M. R. Wilkening, Unveiling Ultra-High Ionic Conductivity in W-Doped Na3SbS4: Grain Boundary Effects and Pure Bulk Transport, J. Am. Chem. Soc., 2025, 174, 20092–20097 CrossRef PubMed.
- Y. Guo, K. Liu, C. Li, D. Song, H. Zhang, Z. Wang, Y. Yan, L. Zhang and S. Dai, A Sulfide-Based Solid Electrolyte With High Humid Air Tolerance for Long Lifespan All-Solid-State Sodium Batteries, Adv. Energy Mater., 2024, 14, 2401504 CrossRef CAS.
- Z. L. Dong, Y. Gan, V. Martins, X. Wang, B. Fu, E. Jin, Y. Gao, Y. Hu, X. Lin and Y. Yuan, Novel Sulfide-Chloride Solid-State Electrolytes with Tunable Anion Ratio for Highly Stable Solid-State Sodium-Ion Batteries, Adv. Mater., 2025, 2503107 CrossRef CAS PubMed.
- X. Wang, S. Zhang, J. Yue, X. Wang, Y. Xu, Y. Gong, L. Zhou, C. Zhao, J. Liang and X. Zhu, A New Class of Carbonate-Oxychloride Solid Electrolytes for High-Performance Sodium-Ion All-Solid-State Batteries, Energy Environ. Sci., 2025, 18, 9927–9938 RSC.
- T. Wang, M. Zhang, K. Zhou, H. Wang, A. Shao, L. Hou, Z. Wang, X. Tang, M. Bai and S. Li, A hetero-layered, mechanically reinforced, ultra-lightweight composite polymer electrolyte for wide-temperature-range, solid-state sodium batteries, Adv. Funct. Mater., 2023, 33, 2215117 CrossRef CAS.
- J. Suo, Y. Jia, X. Zhu, S. Liu, X. Tang, F. Liang, L. Wang and J. Lu, A Novel Super-Toughness and Self-Healing Solid Polymer Electrolyte for Solid Sodium Metal Batteries, Adv. Mater., 2024, 36, 2409587 Search PubMed.
- I. A. Ojelade, E. Truong, I. P. Oyekunle, T. P. Poudel, Y. Chen, M. J. Deck, Y. Jin, B. Ogbolu, P. K. Ojha, M. M. Islam, T. N. D. D. Gamaralalage, J. S. R. Vellore Winfred and Y.-Y. Hu, Break it down to speed it up: Na2O–NaTaCl6, Chem. Sci., 2025, 16, 19857–19866 RSC.
- L. Xiang, Y. Gao, Y. Ding, X. Li, D. Jiang, C. Wu, X. Zhan and L. Zhu, Self-forming Na3P/Na2O interphase on a novel biphasic Na3Zr2Si2PO12/Na3PO4 solid electrolyte for long-cycling solid-state Na-metal batteries, Energy Storage Mater., 2024, 73, 103831 Search PubMed.
- T. Dai, S. Wu, Y. Lu, Y. Yang, Y. Liu, C. Chang, X. Rong, R. Xiao, J. Zhao and Y. Liu, Inorganic glass electrolytes with polymer-like viscoelasticity, Nat. Energy, 2023, 8, 1221–1228 Search PubMed.
- E. Ruoff, S. Kmiec and A. Manthiram, Enhanced Interfacial Conduction in Low-Cost NaAlCl4 Composite Solid Electrolyte for Solid-State Sodium Batteries, Adv. Energy Mater., 2024, 14, 2402091 CrossRef CAS.
- Y. Sadikin, M. Brighi, P. Schouwink and R. Černý, Superionic conduction of sodium and lithium in anion-mixed hydroborates Na3BH4B12H12 and (Li0. 7Na0. 3) 3BH4B12H12, Adv. Energy Mater., 2015, 5, 1501016 CrossRef.
- W. S. Tang, M. Matsuo, H. Wu, V. Stavila, A. Unemoto, S.-i. Orimo and T. J. Udovic, Stabilizing lithium and sodium fast-ion conduction in solid polyhedral-borate salts at device-relevant temperatures, Energy Storage Mater., 2016, 4, 79–83 CrossRef.
- Y. Lu, L. Li, Q. Zhang, Y. Cai, Y. Ni and J. Chen, High-performance all-solid-state electrolyte for sodium batteries enabled by the interaction between the anion in salt and Na3SbS4, Chem. Sci., 2022, 13, 3416–3423 RSC.
- S. Kmiec, E. Ruoff, J. Darga, A. Bodratti and A. Manthiram, Scalable Glass-Fiber-Polymer Composite Solid Electrolytes for Solid-State Sodium–Metal Batteries, ACS Appl. Mater. Interfaces, 2023, 15, 20946–20957 CrossRef CAS PubMed.
- Y. Zheng, Y. Yao, J. Ou, M. Li, D. Luo, H. Dou, Z. Li, K. Amine, A. Yu and Z. Chen, A review of composite solid-state electrolytes for lithium batteries: fundamentals, key materials and advanced structures, Chem. Soc. Rev., 2020, 49, 8790–8839 RSC.
- H. An, M. Li, Q. Liu, Y. Song, B. Deng, X. Liu and J. Wang, Hybrid electrolyte enables solid-state sodium batteries sustaining 50,000 cycles, Nat Sustainability, 2025, 1–11 Search PubMed.
- H. Tian, J. Li, Y. Cai, X. Yao and Z. Su, Dendrite-free, interfacially compatible Na 2 ZrCl 6 composite halide solid-state electrolyte for solid-state sodium-ion batteries, J. Mater. Chem. A, 2025, 13, 15178–15188 RSC.
- M. Wang, H. Zhao, B. Du, X. Lu, S. Ding and X. Hu, Functions and applications of emerging metal–organic-framework liquids and glasses, Chem. Commun., 2023, 59, 7126–7140 RSC.
- W. Tian, G. Lin, S. Yuan, T. Jin, Q. Wang and L. Jiao, Competitive Coordination and Dual Interphase Regulation of MOF-Modified Solid-State Polymer Electrolytes for High-Performance Sodium Metal Batteries, Angew. Chem., Int. Ed., 2025, 64, e202423075 CrossRef CAS PubMed.
- L. Xiang, D. Jiang, Y. Gao, C. Zhang, X. Ren, L. Zhu, S. Gao and X. Zhan, Self-Formed Fluorinated Interphase with Fe Valence Gradient for Dendrite-Free Solid-State Sodium-Metal Batteries, Adv. Funct. Mater., 2024, 34, 2301670 Search PubMed.
- R. Wang, W. Feng, X. Yu, Q. Shi, P. Wang, Y. Liu, J. Zhang and Y. Zhao, Stable zero-sodium-excess solid-state batteries enabled by interphase stratification, eScience, 2024, 4, 100274 CrossRef.
- Y. Lu, Y. Cai, Q. Zhang, L. Liu, Z. Niu and J. Chen, A compatible anode/succinonitrile-based electrolyte interface in all-solid-state Na–CO2 batteries, Chem. Sci., 2019, 10, 4306–4312 RSC.
- Y. Wang, Z. Wang, F. Zheng, J. Sun, J. A. S. Oh, T. Wu, G. Chen, Q. Huang, M. Kotobuki, K. Zeng and L. Lu, Ferroelectric Engineered Electrode-Composite Polymer Electrolyte Interfaces for All-Solid-State Sodium Metal Battery, Adv. Sci., 2022, 9, 2105849 CrossRef CAS PubMed.
- E. J. Kim, P. R. Kumar, Z. T. Gossage, K. Kubota, T. Hosaka, R. Tatara and S. Komaba, Active material and interphase structures governing performance in sodium and potassium ion batteries, Chem. Sci., 2022, 13, 6121–6158 RSC.
- C. Ma, Z. Fu, Y. Fan, H. Li, Z. Ma, W. Jiang, G. Han, H. Ben and H. Xiong, Synergistic interface and structural engineering for high initial coulombic efficiency and stable sodium storage in metal sulfides, Chem. Sci., 2024, 15, 8966–8973 RSC.
- T. Ortmann, T. Fuchs, J. K. Eckhardt, Z. Ding, Q. Ma, F. Tietz, C. Kübel, M. Rohnke and J. Janek, Deposition of Sodium Metal at the Copper-NaSICON Interface for Reservoir-Free Solid-State Sodium Batteries, Adv. Energy Mater., 2024, 14, 2302729 Search PubMed.
- L. Li, R. Xu, L. Zhang, Z. Zhang, M. Yang, D. Liu, X. Yan and A. Zhou, O-Tailored Microstructure-Engineered Interface toward Advanced Room Temperature All-Solid-State Na Batteries, Adv. Funct. Mater., 2022, 32, 2203095 CrossRef CAS.
- G. Zhao, L. Xu, J. Jiang, Z. Mei, Q. An, P. Lv, X. Yang, H. Guo and X. Sun, COFs-based electrolyte accelerates the Na+ diffusion and restrains dendrite growth in quasi-solid-state organic batteries, Nano Energy, 2022, 92, 106756 CrossRef CAS.
- L. Gao, J. Chen, Q. Chen and X. Kong, The chemical evolution of solid electrolyte interface in sodium metal batteries, Sci. Adv., 2022, 8, eabm4606 CrossRef CAS PubMed.
- C. Hänsel, P. V. Kumar and D. Kundu, Stack Pressure Effect in Li3PS4 and Na3PS4 Based Alkali Metal Solid-State Cells: The Dramatic Implication of Interlayer Growth, Chem. Mater., 2020, 32, 10501–10510 CrossRef.
- S. Wenzel, T. Leichtweiss, D. A. Weber, J. Sann, W. G. Zeier and J. Janek, Interfacial Reactivity Benchmarking of the Sodium Ion Conductors Na3PS4 and Sodium β-Alumina for Protected Sodium Metal Anodes and Sodium All-Solid-State Batteries, ACS Appl. Mater. Interfaces, 2016, 8, 28216–28224 CrossRef CAS PubMed.
- X. Zhang, Q. J. Wang, B. Peng and Y. Wu, Pressure-Driven and Creep-Enabled Interface Evolution in Sodium Metal Batteries, ACS Appl. Mater. Interfaces, 2021, 13, 26533–26541 Search PubMed.
- J. Suo, Q. Zhao, H. Tian, L. Wang, L. Dai, J. Luo and S. Liu, Designing a Quasi-Liquid Alloy Interface for Solid Na-Ion Battery, ACS Nano, 2023, 17, 10229–10235 CrossRef CAS PubMed.
- S. Cai, H. Tian, J. Liu, S. Liu, L. Dai, J. Xu, L. Kong and L. Wang, Tuning Na3Hf2Si2PO12 electrolyte surfaces by metal coating for high-rate and long cycle life solid-state sodium ion batteries, J. Mater. Chem. A, 2022, 10, 1284–1289 RSC.
- Y. Li, S. Halacoglu, V. Shreyas, W. Arnold, X. Guo, Q. Dou, J. B. Jasinski, B. Narayanan and H. Wang, Highly efficient interface stabilization for ambient-temperature quasi-solid-state sodium metal batteries, Chem. Eng. J., 2022, 434, 134679 CrossRef CAS.
- C. Yuan, R. Li, X. Zhan, V. L. Sprenkle and G. Li, Stabilizing Metallic Na Anodes via Sodiophilicity Regulation: A Review, Materials, 2022, 15, 4636 CrossRef CAS PubMed.
- E. Jin, J. Su, H. Hou, P. Pirayesh, Y. Wang, Y. Yuan, H. Yan, G. Popov, L. V. Goncharova, S. Ketabi, F. Dai, C. Cao, L. Chen and Y. Zhao, Electro-Chemo-Mechanically Stable and Sodiophilic Interface for Na Metal Anode in Liquid-based and Solid-State Batteries, Adv. Mater., 2024, 36, 2406837 Search PubMed.
- X. Luo, J. Mo, X. Xu, F. Li, W. Fan, Y. Wu, J. Zhao, J. Liu and Y. Huo, Interface-Targeting Integrated Sandwich-Structured Na3Zr2Si2PO12 Composite Electrolyte for Ultra-Long Cycle Life Sodium Metal Batteries, Angew. Chem., Int. Ed., 2025, 64, e202510960 CrossRef CAS PubMed.
- T. Yang, S. Qin, S. Gao, X. Wang, D. Luo, Y. Shi, Q. Ma, X. Zhang, Y. Zhang and Z. Chen, Electroinitiated interfacial healing for external pressure-free solid-state sodium metal batteries, Nat. Commun., 2025, 16, 9613 Search PubMed.
- Y. Zhang, D. Tie, Z. Xiong, X. Lin, S. Liu, Q. Tan, A. Vlad, M. Xu, Y.-S. Hu and Y. Qi, Fast-Charging Hard Carbons: A Fully Organic SEI Enables Low-Coordination Interfacial Environments and Fast Na+ Desolvation, Angew. Chem., Int. Ed., 2025, 64, e202516068 CrossRef CAS PubMed.
- S. Xu, C. Xie, R. Wang, H. Sun, D. Sun, X. Meng, H. Zhang, L. Che, Y. Tang and H. Wang, Dispersed Sodophilic Phase Induced Bulk Phase Reconstruction of Sodium Metal Anode for Highly Reversible Solid-State Sodium Batteries, Adv. Funct. Mater., 2025, 52, e14032 Search PubMed.
- Y. Yang, G. Chang, Z. Liu, X. Chen, C. Huang, Y. Qian, Z. Liu, Q. Tang, A. Hu and X. Chen, Stabilized Solid–Solid Interface for Solid-State Sodium Batteries Using Gradient Ion-Electron Conductive Phases Modified Sodium Metal Anode, ACS Mater. Lett., 2024, 6, 3564–3571 CrossRef CAS.
- Z. Sun, Y. Zhao, Q. Ni, Y. Liu, C. Sun, J. Li and H. Jin, Solid-State Na Metal Batteries with Superior Cycling Stability Enabled by Ferroelectric Enhanced Na/Na3Zr2Si2PO12 Interface, Small, 2022, 18, 2200716 CrossRef CAS PubMed.
- X. Li, Q. Zhou, J. Wu, C. Yuan, K. Lu, X. Zhan and L. Zhu, An in Situ Formed Multifunctional Interphase with High Dendrite Tolerance for Long-Life Solid-State Sodium–Metal Batteries, ACS Appl. Energy Mater., 2023, 6, 10333–10339 Search PubMed.
- L. Zhou, J. D. Bazak, C. Li and L. F. Nazar, 4 V Na Solid State Batteries Enabled by a Scalable Sodium Metal Oxyhalide Solid Electrolyte, ACS Energy Lett., 2024, 9, 4093–4101 Search PubMed.
- G. Sun, Y. Ma, H. Zhang, S. Wu, S. Chu, S. Passerini and Y. Ma, Fabrication of Composite Cathode for All-Solid-State Sodium Batteries, Adv. Energy Mater., 2025, e03756 CrossRef.
- Y.-F. Zhu, Y. Xiao, S.-X. Dou, Y.-M. Kang and S.-L. Chou, Spinel/Post-spinel engineering on layered oxide cathodes for sodium-ion batteries, eScience, 2021, 1, 13–27 Search PubMed.
- T. Lan, C.-L. Tsai, F. Tietz, X.-K. Wei, M. Heggen, R. E. Dunin-Borkowski, R. Wang, Y. Xiao, Q. Ma and O. Guillon, Room-temperature all-solid-state sodium batteries with robust ceramic interface between rigid electrolyte and electrode materials, Nano Energy, 2019, 65, 104040 Search PubMed.
- H. Yamauchi, J. Ikejiri, F. Sato, H. Oshita, T. Honma and T. Komatsu, Pressureless all-solid-state sodium-ion battery consisting of sodium iron pyrophosphate glass-ceramic cathode and β″-alumina solid electrolyte composite, J. Am. Ceram. Soc., 2019, 102, 6658–6667 CrossRef CAS.
- D. Wang, M. Takiyama, J. Hwang, K. Matsumoto and R. Hagiwara, A Hexafluorophosphate-Based Ionic Liquid as Multifunctional Interfacial Layer between High Voltage Positive Electrode and Solid-State Electrolyte for Sodium Secondary Batteries, Adv. Energy Mater., 2023, 13, 2301020 Search PubMed.
- W. Meng, J. Liu, L. Wang, L. Dai and S. Liu, In Situ Construction of a Liquid Film Interface with Fast Ion Transport for Solid Sodium-Ion Batteries, Nano Lett., 2022, 22, 5214–5220 Search PubMed.
- C. M. Costa, Y.-H. Lee, J.-H. Kim, S.-Y. Lee and S. Lanceros-Méndez, Recent advances on separator membranes for lithium-ion battery applications: From porous membranes to solid electrolytes, Energy Storage Mater., 2019, 22, 346–375 CrossRef.
- J. Wu, S. Liu, F. Han, X. Yao and C. Wang, Lithium/Sulfide All-Solid-State Batteries using Sulfide Electrolytes, Adv. Mater., 2021, 33, 2000751 CrossRef CAS PubMed.
- H. Zhang, Y. Zhang, D. Yan, P. Lv, C. Yu, H. Zheng, L. Yan, Z. Cheng, H. Y. Yang and Y. Bai, Activating Ferroelectric-Magnetic Synergistic Effects at Cathode-Electrolyte Interfaces Toward Superfast and Stable Sodium Storage, Adv. Mater., 2025, 37, e02846 CrossRef CAS PubMed.
- J.-C. Liu, T. You, Y.-F. Zhao, F.-Q. Liu, J.-D. Li, L.-L. Wang, C. Wang and L. Li, Multifunctional sulfonate additive induced CEI layer enables ultra-stable PEO based solid-state sodium batteries, Rare Met., 2025, 44, 3817–3826 CrossRef CAS.
- M. Hou, J. Zi, L. Zhao, Y. Zhou, F. Li, Z. Xie, D. Zhang, B. Yang and F. Liang, Enhancement of interfacial sodium ion transport stability in quasi-solid-state sodium-ion batteries using polyethylene glycol, Mater. Chem. Front., 2023, 7, 2027–2037 Search PubMed.
- L. Lu, Y. Lu, J. A. Alonso, C. A. López, M. T. Fernández-Díaz, B. Zou and C. Sun, A Monolithic Solid-State Sodium–Sulfur Battery with Al-Doped Na3.4Zr2(Si0.8P0.2O4)3 Electrolyte, ACS Appl. Mater. Interfaces, 2021, 13, 42927–42934 CrossRef CAS PubMed.
- H. Lai, Y. Li, J. Wang, W. Li, X. Wu and Z. Wen, Design of solid-state sodium-ion batteries with high mass-loading cathode by porous-dense bilayer electrolyte, J. Materiomics, 2021, 7, 1352–1357 CrossRef.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |