Open Access Article
Open Access ArticleSuppressing nonradiative decay via molecular configuration control in Cu(I)–halide clusters enables the fabrication of highly efficient green and green-sensitized blue OLEDs
Xiao
Li
abc,
Sai
Guo
abc,
Xin
Liu
abc,
Yu-Fu
Sun
abc,
Dong-Hai
Zhang
abc,
Hui
Yang
abc,
Jia-Min
Lu
*abc and
Xu-Lin
Chen
 *abc
*abc
aState Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou, Fujian 350002, China. E-mail: xlchem@fjirsm.ac.cn
bClinical Research Institute, The First Affiliated Hospital of Xiamen University, School of Medicine Xiamen University, Xiamen, Fujian 361003, China. E-mail: lujiamin@xmu.edu.cn
cXiamen Key Laboratory of Rare Earth Photoelectric Functional Materials, Xiamen Institute of Rare Earth Materials, Chinese Academy of Sciences, Xiamen, Fujian 361021, China
First published on 6th February 2026
Abstract
Copper(I) complexes are cost-effective and eco-friendly emitters, yet their device applications are hindered by broad emission, by limited film-forming ability, and especially by severe excited-state distortions that typically lead to low emission efficiency in the film state. Herein, to address these challenges, we propose a structural design strategy for highly efficient and sublimable copper(I)-bromide clusters by simultaneously incorporating donor–acceptor bisphosphine ligands and introducing ortho-methyl substitution. This design effectively suppresses intrinsic nonradiative decay by modulating the excited-state geometry, thereby achieving an exceptionally high photoluminescence quantum yield of 99% in doped films. Vacuum-deposited organic light-emitting diodes (OLEDs) using the optimized cluster [dtpb-Ac]2Cu2Br2 as the terminal emitter achieve efficient green emission with a maximum external quantum efficiency (EQE) of 25.1%. Notably, an innovative strategy exploits the intrinsically broad emission of the copper(I)-bromine cluster to sensitize the deep-blue MR-TADF emitter ν-DABNA, achieving high-efficiency green-sensitized blue OLEDs with a maximum EQE of 28.7% and Commission Internationale de l’Eclairage (CIE) coordinates of (0.15, 0.19). As either a green dopant or a sensitizer, the device performance ranks among the best reported for copper(I)-based OLEDs. The current study presents promising molecular design and sensitization strategies to address the key challenges in developing high-performance copper(I)-based OLEDs.
Introduction
Organic light-emitting diodes (OLEDs) have been developed as a leading display technology. In the pursuit of high-performance OLEDs, luminescent materials that comprise emitting layers play a key role and have garnered significant attention. Phosphorescent and thermally activated delayed fluorescence (TADF) materials are the two most promising OLED emitters due to their potential for high exciton utilization efficiency (EUE).1 Phosphorescent platinum-group metal complexes have achieved great success in fabricating green and red pixels for commercial OLEDs. However, these phosphors suffer from high costs and face challenges in achieving high-efficiency blue phosphorescence due to the intrinsic nonradiative metal-centered dd* states.2 Multiresonance (MR) TADF molecules featuring mutually ortho-positioned boron and nitrogen atoms have emerged as ideal terminal emitters for highly efficient narrowband electroluminescence (EL),3 assisted by appropriate phosphorescence or TADF sensitizers for triplet harvesting.4 In these sensitization strategies, a high-performance sensitizer with efficient and rapid triplet-harvesting capability and good spectral overlap with the terminal emitter is essential. In particular, to sensitize deep-blue emitters, the sensitizers should typically be blue-emitting to enable effective Förster resonance energy transfer (FRET) and inhibit back energy transfer.5 However, the pursuit of ideal blue sensitizers encounters significant challenges due to the stringent molecular design principles required.Copper(I) complexes have been extensively investigated as photoluminescence (PL) and EL materials due to their cost-effectiveness, structural diversity, rich photophysical properties, and triplet-harvesting capacity.6 Owing to the low oxidation potential of the Cu+ ion, the lowest-energy optical transitions in most copper(I) complexes are mainly characterized by metal-to-ligand charge transfer (MLCT). The MLCT transitions in copper(I) complexes engender small singlet-triplet energy splitting (ΔEST), thereby imparting TADF characteristics.7 However, these MLCT transitions are associated with large reorganization energies and oxidation at the d10 metal centre, which not only increase nonradiative decay rates (especially in non-rigid matrices) via Jahn–Teller distortion but also lead to broad emission spectra.8 As a result, most copper(I) complexes exhibit low emission efficiencies and broad emission spectra in films, making them unsuitable as terminal emitters for fabricating efficient and high color-purity OLEDs. Moreover, the fully filled 3d orbitals of copper(I) complexes are energetically high. Although nonradiative dd∗ states do not exist as in platinum-group metal complexes, the high-level 3d orbitals in copper(I) complexes inevitably result in relatively low-energy MLCT states, presenting a formidable challenge in achieving blue emission, let alone deep-blue emission.7,9 The EL applications of copper(I) complexes are significantly limited by the aforementioned characteristics of their excited states. Among copper(I) complexes, copper(I)-halide clusters have captured researchers' attention due to their diverse structural and photophysical properties.10 The involvement of halide ligands in the lowest excited states has been shown to facilitate intersystem crossing (ISC), reduce excited-state distortions, and prevent photothermal decomposition in copper(I)-halide clusters.11 However, copper(I)-halide clusters share the same limitations as other copper(I) complexes, including undesired rapid nonradiative decay, broad emission spectra, and the challenge of achieving blue emission.12 The rapid nonradiative decays of intrinsic (metal plus halide)-to-ligand charge transfer [(M + X)LCT] and cluster-centered triplet states (3CC) account for the low emission efficiencies observed in most reported copper(I)-halide clusters in solutions and films.13 Xu and coworkers recently reported that incorporating electron-donating groups into bidentate phosphine ligands is an effective strategy to reduce the population of nonradiative 3CC states in the Cu4I4 cores.14 Apart from their limitations in photoluminescence, many copper(I)-halide clusters are insoluble in common solvents and unstable during sublimation, rendering them impractical for use in fabricating EL devices. Despite some advancements, designing copper(I)-halide clusters for the construction of highly efficient OLEDs, particularly blue devices, based on a deep understanding of the structure–property relationships remains challenging.
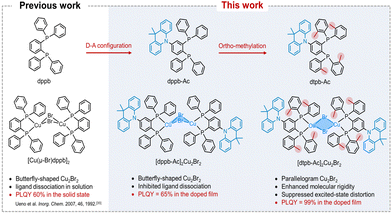
Here, we designed and investigated two copper(I)-bromine clusters, [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2, supported by bisphosphine ligands (dppb-Ac and dtpb-Ac) featuring similar donor (D)-acceptor (A) configurations (Fig. 1). Compared with the previously reported copper(I)-bromine cluster coordinated by the commercially available bisphosphine ligand dppb, incorporation of a dimethylacridine unit into the dppb ligand effectively suppresses dissociation of the ligand from the complex in solution (Fig. S15). The further introduction of ortho-methyl groups on the bisphosphine ligand dppb-AC significantly affected the molecular configuration, transforming the Cu2Br2 core from a “butterfly” shape to a planar parallelogram shape, accompanied by a marked decrease in intramolecular Cu–Cu interaction. Compared with those in [dppb-Ac]2Cu2Br2, the contribution of (M + X)LCT decreased while the contribution of intramolecular charge transfer (ILCT) increased in the emissive state (S1) of [dtpb-Ac]2Cu2Br2. As a result, the distortion and nonradiative decay of the S1 state of [dtpb-Ac]2Cu2Br2 are significantly suppressed. [dtpb-Ac]2Cu2Br2 in doped films exhibits high PLQYs of up to 99%, which is much higher than the corresponding PLQY of 65% observed for [dppb-Ac]2Cu2Br2. The doped OLEDs utilizing [dtpb-Ac]2Cu2Br2 as the terminal emitter displayed bright green emission with maximum external quantum efficiencies (EQEs) reaching 25.1% and corresponding FWHMs exceeding 95 nm. Notably, thanks to the high efficiency and broad emission of [dtpb-Ac]2Cu2Br2, highly efficient green-sensitized deep-blue OLEDs were fabricated by employing the green-emitting [dtpb-Ac]2Cu2Br2 as the sensitizer for the blue MR-TADF emitter ν-DABNA, achieving a maximum EQE of 28.7% and corresponding CIE coordinates of (0.15, 0.19).
Results and discussion
Molecular design and crystal structures
The bidentate phosphine ligands, dppb-Ac and dtpb-Ac, were synthesized through a similar three-step process with respective overall yields of 50% and 38%, as depicted in Fig. S1. The copper(I)–bromine clusters were facilely prepared by reacting CuBr with one equivalent of the corresponding bisphosphine ligand in dichloromethane. Single crystals suitable for X-ray analysis were obtained by diffusing diethyl ether into their filtered dichloromethane solutions. Impressively, these copper(I)–bromine clusters not only exhibit good solubility in various solvents but also demonstrate excellent sublimability. Thermal gravimetric analysis (TGA) and differential scanning calorimetry (DSC) conducted under a nitrogen atmosphere indicated that [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 possess high decomposition temperatures (Td) of 425 and 405 °C and high glass transition temperatures (Tg) of 175 and 138 °C, respectively (Fig. S6). The excellent processability and stability make them stand out among copper-halide clusters, particularly when it comes to purification via thermal gradient sublimation and device fabrication through either vacuum thermal deposition or solution processing. Both newly synthesized clusters were purified through successive recrystallization and sublimation prior to characterization and device fabrication. The molecular structures of [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 were confirmed using 1H-NMR,13C-NMR, elemental analysis, and single-crystal X-ray diffraction analysis (CCDC numbers: 2386729 and 2386762). Detailed synthesis procedures and characterization data are provided in the SI.Single-crystal X-ray diffraction analysis revealed distinct configurations of the two copper(I)–bromine clusters (Fig. 2a). [dppb-Ac]2Cu2Br2 exhibited a butterfly-shaped Cu2Br2 core, with the copper(I) metal centers positioned in close proximity to each other (dCu–Cu = 2.85 Å). The butterfly geometry indicates that the four-membered Cu2Br2 ring is folded along the Cu⋯Cu axis. These configuration characteristics resemble those of the previously reported copper(I)–halide clusters.15 In comparison, when coordinated by the ortho-methylated bisphosphine ligand dtpb-Ac, [dtpb-Ac]2Cu2Br2 adopted a symmetric parallelogram Cu2Br2 core (with the sum of the interior angles being 360°), and the Cu–Cu distance was significantly elongated to 3.10 Å. Each copper(I) atom in these clusters adopted a four-coordinate, tetrahedral-like configuration. The dimethylacridine donor units are nearly orthogonally attached to the bisphosphine ligands, with D–A dihedral angles of 85.63° in [dppb-Ac]2Cu2Br2 and 81.76° in [dtpb-Ac]2Cu2Br2. Besides the molecular configuration, the molecular packings of the copper(I)–bromine clusters are also influenced by ortho-methylation on the bisphosphine ligand. As depicted in Fig. 2c, [dppb-Ac]2Cu2Br2 molecules are densely packed in the crystal lattice, with abundant intermolecular interactions among neighbouring molecules. In contrast, fewer and weaker intermolecular interactions are observed in the crystal lattice of [dtpb-Ac]2Cu2Br2. Compared with [dppb-Ac]2Cu2Br2, the significantly increased intermolecular distance between neighbouring [dtpb-Ac]2Cu2Br2 molecules is expected to more effectively inhibit emission quenching in aggregate states by avoiding short-distance Dexter energy transfer (DET).16
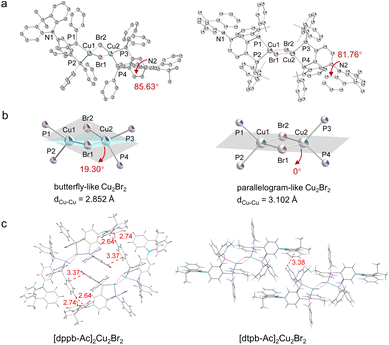 | ||
| Fig. 2 (a) Single-crystal structures; (b) configurations of the Cu2Br2 cores; (c) molecular packing diagrams. | ||
Theoretical calculations
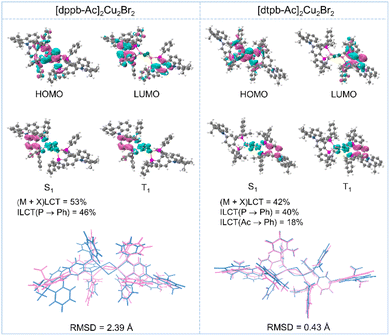
To elucidate the characteristics of the frontier molecular orbitals (FMOs) and excited states of these copper(I)-bromine clusters, we performed density functional theory (DFT) and time-dependent DFT (TD-DFT) calculations using the B3LYP functional and 6-31G* basis set implemented in Gaussian. The highest occupied molecular orbitals (HOMOs) of both clusters are primarily localized on the Cu2Br2 cores with partial extension to the P atoms of the bisphosphine ligands, while the lowest unoccupied molecular orbitals (LUMOs) are mainly distributed over the central phenyl ring of the ligands (Fig. 3). The pronounced spatial separation between the HOMO and LUMO is expected to result in small ΔEST values for these copper(I)-bromine clusters. TD-DFT calculations revealed that the S1 state of [dppb-Ac]2Cu2Br2 is primarily characterized by a (metal + halogen)-to-ligand charge transfer [(M + X)LCT] transition (53%), along with a significant intra-ligand charge transfer (ILCT) component (46%) from the P atoms to the central phenyl rings of the ligands. In comparison, the contributions of the (M + X)LCT and P-to-phenyl ILCT transitions in the S1 state of [dtpb-Ac]2Cu2Br2 decrease to 42% and 40%, respectively, accompanied by a significantly increased ILCT transition (18%) originating from the dimethylacridine units to the central phenyl rings (Fig. 3 and Tables S7, S9). Replacing the partial (M + X)LCT transition with a dominant ILCT character in the emissive state may help suppress nonradiative Jahn–Teller distortions, which are commonly observed in the excited states of d10 metal complexes.17 To gain further insight into the effects of ortho-methylation on the excited-state geometries, the root-mean-square displacements (RMSDs) between the optimized S0 and S1 geometries were calculated to quantify structural changes upon excitation. As depicted in Fig. 3, [dppb-Ac]2Cu2Br2 exhibits pronounced intramolecular motions, such as rotations and twisting, resulting in poor overlap between the S0 (pink) and S1 (blue) geometries and a large RMSD value of 2.39 Å. In contrast, ortho-methylation on the bisphosphine ligand effectively restricts structural relaxation, yielding a markedly smaller RMSD of 0.43 Å for [dtpb-Ac]2Cu2Br2. The effectively restricted excited-state relaxation is expected to contribute to the significantly higher emission efficiencies and narrower-band emission of [dtpb-Ac]2Cu2Br2 in both solution and film states (infra vide).Photophysical properties
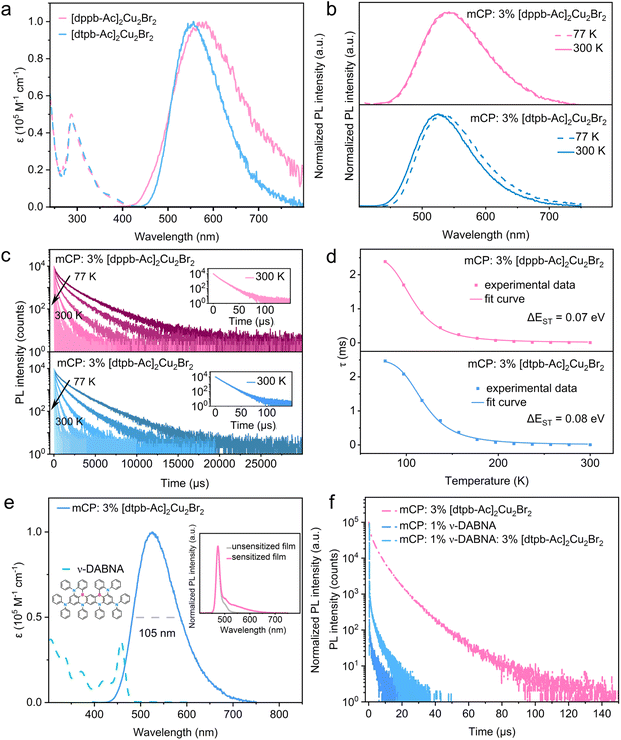
The photophysical properties of both the clusters and the bisphosphine ligands were initially examined in dilute solutions. As shown in Fig. 4a, [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 exhibit similar ultraviolet-visible (UV-vis) absorption profiles in dichloromethane (c = 1 × 10−5 M), featuring strong bands below 340 nm and a weak shoulder band above 340 nm. The high-energy bands (<340 nm), which also appear in the absorption spectra of the respective ligands, can be assigned to the spin-allowed π–π* transitions localized at the bisphosphine ligands. According to theoretical calculations (Fig. 3 and Tables S6–S9), the low-energy shoulder bands (>340 nm) of both clusters likely stem from a combination of (M + X)LCT and ILCT transitions. In degassed solutions, the clusters [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 exhibited yellow and red emissions, with spectral peaks at 574 and 552 nm and full widths at half maximum (FWHM) of 180 and 125 nm, respectively. Compared to [dppb-Ac]2Cu2Br2, [dtpb-Ac]2Cu2Br2 exhibits a significantly blue-shifted and narrower emission band (Fig. 4a). This is attributed to the deeper LUMO energy level, as revealed by cyclic voltammetry measurements (Fig. S7 and Table S5) and the suppressed excited-state structural deformation, as indicated by RMSD calculations (Fig. 3)—both resulting from the introduction of methyl groups onto the bisphosphine ligand. It is worth noting that no emission band from the ligand was observed, indicating that ligand dissociation in solution is effectively suppressed for both [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2.We further investigated the photophysical properties of these two copper(I)-halide clusters dispersed in 1,3-bis(9-carbazolyl)benzene (mCP) films at a doping concentration of 3 wt%. The doped mCP films of [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 exhibited bright yellow and green emissions at room temperature, with emission peaks at 540 and 525 nm, PLQYs of 65% and 99%, and decay times of 7.1 and 8.1 µs, respectively. The onset wavelengths of the room-temperature emission spectra of [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 is very close, while the latter exhibited a much narrower emission profile. The similar emission onsets indicate nearly equal zero–zero energies (E0–0) for the S1 → S0 transitions. The much narrower emission profile of [dtpb-Ac]2Cu2Br2 compared with [dppb-Ac]2Cu2Br2 is attributed to the effectively restricted excited-state distortion of the S1 state through ortho-methylation, as revealed by the theoretical calculations (Fig. 3). To the best of our knowledge, near-unity PLQYs have rarely been reported for copper(I) emitters in evaporated films, due to non-radiative decay caused by their intrinsic excited-state distortions and/or poor sublimability.18 As summarized in Table 1, [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 show comparable radiative rate constants (kr = ΦPL/τ) of 0.92 × 105 and 1.22 × 105 s−1, respectively, while the nonradiative rate constant [knr = (1 − ΦPL)/τ] of the latter is only about one-fortieth of that of the former (0.12 × 104vs. 4.93 × 104 s−1). Consequently, the much higher PLQY of [dtpb-Ac]2Cu2Br2 relative to [dppb-Ac]2Cu2Br2 is primarily attributed to the effective suppression of nonradiative transitions by ortho-methylation on the bisphosphine ligands. Severe nonradiative decay, often driven by structural distortion from a tetrahedral-like ground state (d10, sp3) to a flattened excited state (d9, dsp2) upon photoexcitation, is a well-known challenge in achieving high emission efficiency in copper(I) complexes. The ortho-methyl groups effectively inhibit such excited-state deformations, as supported by theoretical calculations (Fig. 3). The temperature dependence of the emission spectra and decay times was studied for the copper(I)-halide clusters to elucidate the luminescence mechanism. As shown in Fig. 4b, when the temperature decreased from 300 to 77 K, the emission spectra of [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 exhibited a slight redshift. From the onset wavelength of the emission spectra at 300 and 77 K, the S1 and T1 energies and ΔESTs were calculated to be 2.73, 2.70 and 0.03 eV for [dppb-Ac]2Cu2Br2, 2.77, 2.72 and 0.05 eV for [dtpb-Ac]2Cu2Br2, respectively (Table 1). The decay times of [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 increased several hundredfold, from 7.1 and 8.1 µs to 2.39 and 2.47 ms, respectively. These observations suggest that their emissions may originate from two excited states in fast thermal equilibrium, S1 and T1, with an energy spacing of ΔEST. In this case, the temperature dependence of the emission decay time (τ) can be described by the following Boltzmann equation:6b,7,19
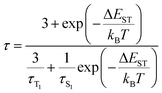 | (1) |
| Compound | λ PL , nm | Φ PL , % | τ , µs | k r , 105 s−1 | k nr , 104 s−1 | S1/T1/ΔESTf, eV |
|---|---|---|---|---|---|---|
| a PL peak. b Photoluminescence quantum yield. c Emission decay time. d Radiative decay rate. e Nonradiative decay rate. f Energy levels and gap between the S1 and T1 states determined from the emission spectra at 77 and 300 K. | ||||||
| [dppb-Ac]2Cu2Br2 | 540 | 65 | 7.1 | 0.92 | 4.93 | 2.73/2.70/0.03 |
| [dtpb-Ac]2Cu2Br2 | 525 | 99 | 8.1 | 1.22 | 0.12 | 2.77/2.72/0.05 |
Electroluminescent properties
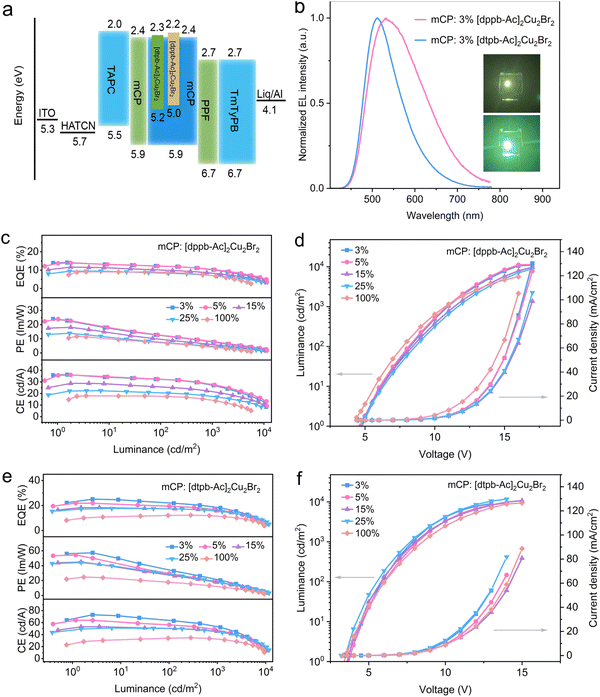
To evaluate the EL performance of the two clusters, OLEDs with [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2 as terminal emitters were fabricated using the following device configuration: ITO/HAT-CN (10 nm)/TAPC (40 nm)/mCP (5 nm)/undoped emitter or an x wt% emitter doped in mCP (30 nm)/PPF (5 nm)/TmPyPB (35 nm)/Liq (1 nm)/Al (100 nm) (x = 3, 5, 15 and 25). Here, HAT-CN, TAPC, PPF, TmPyPB, and Liq served as the hole-injecting, hole-transporting, hole-blocking, electron-transporting, and electron-injecting layers, respectively. mCP, with a high triplet energy (T1 = 2.9 eV), was selected as both the electron-blocking and host material to confine carriers within the emitting layer and ensure efficient exothermic energy transfer from the host to the emitter. The energy level diagram of these devices is shown in Fig. 5a, while the molecular structures of the organic materials are presented in Fig. S18. The device performance was investigated at emitter concentrations of 3%, 5%, 15%, 25%, and 100%. Graphical and numerical data illustrating the device performance are shown in Fig. 5 and Table 2, respectively.| Emitter | Conc.a, wt% | V on , V | EQEmaxc, % | Roll-offd, % | L max , cd m−2 | CEf, cd A−1 | PEg, lm W−1 | λ EL/FWHMh, nm | CIEi, (x, y) |
|---|---|---|---|---|---|---|---|---|---|
| a Doping concentration. b Turn-on voltage at 1 cd m−2. c Maximum EQE. d Efficiency roll-off at 1000 cd m−2. e Maximum luminance. f Maximum current efficiency. g Maximum power efficiency. h EL peak and half-peak width at 1000 cd m−2. i Commission Internatinale de L'Eclairage coordinates measured at 1000 cd m−2. | |||||||||
| [dppb-Ac]2Cu2Br2 | 3 | 4.8 | 14.1/10.3 | 27.2 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 070 070 |
36.3 | 22.8 | 534/145 | (0.38, 0.52) |
| 5 | 4.7 | 14/10.4 | 25 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 370 370 |
36.1 | 23.9 | 535/145 | (0.38, 0.51) | |
| 15 | 4.7 | 11.7/8.4 | 27.8 | 9763 | 28.8 | 18.1 | 545/156 | (0.40, 0.51) | |
| 25 | 4.6 | 9.5/7.1 | 25 | 8885 | 22.4 | 13.9 | 552/165 | (0.42, 0.51) | |
| 100 | 4.4 | 9.2/7.1 | 22.3 | 5616 | 18.1 | 11.8 | 597/162 | (0.48, 0.48) | |
| [dtpb-Ac]2Cu2Br2 | 3 | 3.7 | 25.1/18.6 | 26 | 9053 | 73 | 57.4 | 510/95 | (0.29, 0.52) |
| 5 | 3.6 | 21.9/16.6 | 24.1 | 9378 | 64.1 | 54.4 | 513/95 | (0.29, 0.52) | |
| 15 | 3.5 | 18.4/15.7 | 14.4 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 720 720 |
53 | 45 | 515/106 | (0.32, 0.53) | |
| 25 | 3.4 | 17.6/15.6 | 11.5 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 710 710 |
50.9 | 44.3 | 518/109 | (0.33, 0.53) | |
| 100 | 3.4 | 12.3/11.6 | 6 | 9381 | 34.8 | 24.8 | 535/125 | (0.38, 0.54) | |
A progressive redshift and broadening of the EL spectra were observed in the OLEDs based on both clusters as the emitter concentration increased (Table 2). For [dppb-Ac]2Cu2Br2, the emission peaks shifted from 534 to 597 nm, with the FWHM increasing from 145 to 165 nm. Similarly, for [dtpb-Ac]2Cu2Br2, the emission peaks shifted from 510 to 535 nm and the FWHM increased from 95 to 125 nm as the emitter concentration increased from 3 to 100 wt%. These observations can be attributed to the solid solvation effect, which is common in CT-type emitters.20 At high concentrations, the emissive CT states tend to be stabilized by the highly polar neighbouring molecules. The FWHM values of [dtpb-Ac]2Cu2Br2-based devices are much lower than those of the [dppb-Ac]2Cu2Br2-based devices, primarily due to the effective suppression of excited-state vibrational relaxation through the ortho-methylation strategy. The current density–voltage–luminance characteristics are shown Fig. 5d and f, revealing turn-on voltages of 4.8–4.4 V and maximum luminance of 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 370–5616 cd m−2 for [dppb-Ac]2Cu2Br2-based OLEDs and turn-on voltages of 3.7–3.4 V with a maximum luminance of 11
370–5616 cd m−2 for [dppb-Ac]2Cu2Br2-based OLEDs and turn-on voltages of 3.7–3.4 V with a maximum luminance of 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 710–9053 m−2 for [dtpb-Ac]2Cu2Br2-based OLEDs. The efficiency–luminance curves are shown in Fig. 5c and e. Benefiting from the much higher PLQY of [dtpb-Ac]2Cu2Br2 compared to [dppb-Ac]2Cu2Br2, the 3 wt%-doped device with the former as the emitter exhibited a significantly higher EQEmax of 25.1%, which remained at 18.6% at 1000 cd m−2. In contrast, the device with [dppb-Ac]2Cu2Br2 showed a relatively lower EQEmax of 14.1%, retaining 10.3% at 1000 cd m−2. In addition, a high maximum current efficiency (CEmax) and power efficiency (PEmax) of 73 cd A−1 and 57.4 lm W−1 were achieved for the 3 wt%-doped device based on [dtpb-Ac]2Cu2Br2. To the best of knowledge, the EL efficiency values of [dtpb-Ac]2Cu2Br2 are among the highest reported for copper(I) emitters (Fig. 6f). As the emitter concentration increased, the device efficiencies of both clusters gradually decreased. Interestingly, compared to [dppb-Ac]2Cu2Br2-based devices, [dtpb-Ac]2Cu2Br2-based OLEDs exhibited a much more pronounced decrease in efficiency roll-off with increasing emitter concentration. For instance, the non-doped OLED of [dtpb-Ac]2Cu2Br2 exhibited an EQEmax of 12.3% and retained an EQE of 11.6% at 1000 cd m−2, corresponding to a very small efficiency roll-off of 6%, which is much smaller than the 22.3% roll-off observed in the non-doped device of [dppb-Ac]2Cu2Br2. This is partially attributed to the more effectively separated adjacent molecules in the aggregate state of [dtpb-Ac]2Cu2Br2, as revealed by the crystal packing (Fig. 2c ).
710–9053 m−2 for [dtpb-Ac]2Cu2Br2-based OLEDs. The efficiency–luminance curves are shown in Fig. 5c and e. Benefiting from the much higher PLQY of [dtpb-Ac]2Cu2Br2 compared to [dppb-Ac]2Cu2Br2, the 3 wt%-doped device with the former as the emitter exhibited a significantly higher EQEmax of 25.1%, which remained at 18.6% at 1000 cd m−2. In contrast, the device with [dppb-Ac]2Cu2Br2 showed a relatively lower EQEmax of 14.1%, retaining 10.3% at 1000 cd m−2. In addition, a high maximum current efficiency (CEmax) and power efficiency (PEmax) of 73 cd A−1 and 57.4 lm W−1 were achieved for the 3 wt%-doped device based on [dtpb-Ac]2Cu2Br2. To the best of knowledge, the EL efficiency values of [dtpb-Ac]2Cu2Br2 are among the highest reported for copper(I) emitters (Fig. 6f). As the emitter concentration increased, the device efficiencies of both clusters gradually decreased. Interestingly, compared to [dppb-Ac]2Cu2Br2-based devices, [dtpb-Ac]2Cu2Br2-based OLEDs exhibited a much more pronounced decrease in efficiency roll-off with increasing emitter concentration. For instance, the non-doped OLED of [dtpb-Ac]2Cu2Br2 exhibited an EQEmax of 12.3% and retained an EQE of 11.6% at 1000 cd m−2, corresponding to a very small efficiency roll-off of 6%, which is much smaller than the 22.3% roll-off observed in the non-doped device of [dppb-Ac]2Cu2Br2. This is partially attributed to the more effectively separated adjacent molecules in the aggregate state of [dtpb-Ac]2Cu2Br2, as revealed by the crystal packing (Fig. 2c ).
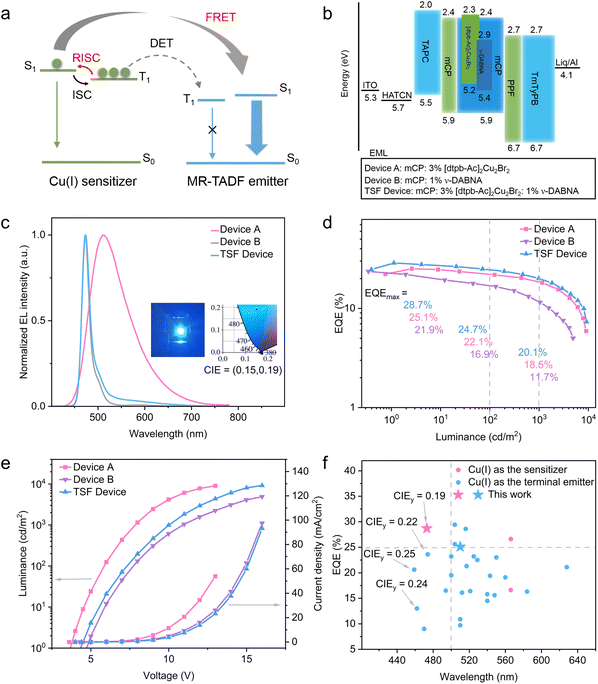 | ||
| Fig. 6 (a) Schematic of the energy transfer mechanism for the copper(I) sensitized system; (b) energy level diagrams; (c) EL spectra; (d) EQE versus luminance curves; (e) current density–voltage–luminance characteristics; (f) EQE versus EL peak wavelength of representative copper(I) complex-based OLEDs.6a,c,d,f,7b,14,15c,19,22 | ||
Although the OLEDs employing [dtpb-Ac]2Cu2Br2 as the emitter exhibited outstanding efficiencies compared to previously reported copper(I) complexes, their emission spectra are very broad—a common shortcoming of copper(I) emitters featuring charge-transfer (CT)-type emissive states. The TADF-sensitized fluorescence (TSF) strategy, originally proposed by Duan4a and Adachi,4b utilizes D–A type TADF molecules as sensitizers for MR-TADF emitters and provides a promising route to simultaneously achieve high EL efficiency and excellent color purity. The efficiency of Förster resonance energy transfer (FRET) from the sensitizer to the emitter partially depends on the spectral overlap between the sensitizer's emission and the emitter's absorption. Although the emission spectra of [dtpb-Ac]2Cu2Br2 in the doped films peak is at above 525 nm, the high-energy edge extends below 430 nm due to the broad spectral width (Fig. 4e). This unique feature, combined with its high-efficiency and short-lived TADF emission, endows [dtpb-Ac]2Cu2Br2 with potential as a sensitizer for blue MR-TADF emitters (Fig. 6a). For instance, as shown in Fig. 4e, there is proper overlap between the absorption spectrum of the representative deep-blue MR-TADF emitter ν-DABNA and the PL spectrum of the sensitizer [dtpb-Ac]2Cu2Br2. The ratio of the overlapping region relative to the total emission band of the copper(I) sensitizer was calculated to be 4.7% for [dtpb-Ac]2Cu2Br2, indicating the possibility of FRET from the sensitizer to the emitter. To preliminarily evaluate the potential of [dtpb-Ac]2Cu2Br2 as a sensitizer, the PL spectrum and PL decay curve of a 1 wt% ν-DABNA: 3 wt% [dtpb-Ac]2Cu2Br2: mCP film (sensitized film) were measured and compared with those of a 3 wt% [dtpb-Ac]2Cu2Br2: mCP film and a 1 wt% ν-DABNA: mCP film (unsensitized film). As shown in Fig. 4e, the sensitized film exhibited narrowband blue emission from ν-DABNA, with only minor emission from [dtpb-Ac]2Cu2Br2. Additionally, compared with the unsensitized film, the sensitized film showed an increased delayed fluorescence ratio in the decay curve (Fig. 4f). These results suggest efficient energy transfer from the sensitizer to ν-DABNA. Using a previously reported method21 (see the SI for details), the FRET rate (kFRET) and efficiency (ηFRET) from the sensitizer [dtpb-Ac]2Cu2Br2 to the terminal emitter ν-DABNA were estimated to be 3.63 × 105 s−1 and 74.8%, respectively. The Förster radius (RFRET) was calculated to be 5.79 nm, while the donor–acceptor distance between [dtpb-Ac]2Cu2Br2 and ν-DABNA was estimated to be 4.84 nm. Given these relatively large distances, it is reasonable to disregard short-range Dexter energy transfer and instead consider long-range FRET as a predominant contributor to the excited-state dynamics of the sensitization system. Encouraged by this finding, we further constructed copper(I)-sensitized-fluorescent OLEDs employing [dtpb-Ac]2Cu2Br2 as the sensitizer, ν-DABNA as the terminal dopant, and mCP as the host material. The vacuum deposited TSF device featured an optimized structure of ITO/HATCN (10 nm)/TAPC (40 nm)/mCP (5 nm)/1 wt% ν-DABNA: 3 wt% [dtpb-Ac]2Cu2Br2: mCP (30 nm)/PPF (5 nm)/TmPyPB (35 nm)/Liq (1 nm)/Al (100 nm) (Fig. 6b). For comparison, two additional devices were fabricated and characterized: Device A, employing 3 wt% [dtpb-Ac]2Cu2Br2 as the terminal emitter, and Device B, an unsensitized reference device with the structure ITO/HATCN (10 nm)/TAPC (40 nm)/mCP (5 nm)/1 wt% ν-DABNA:mCP (30 nm)/PPF (5 nm)/TmPyPB (35 nm)/Liq (1 nm)/Al (100 nm) (Fig. 6b). The device performances are shown in Fig. 6c–e and Table 3. The TSF device achieved highly efficient blue emission, with CIE (x, y) coordinates of (0.15, 0.19). The EL spectrum of the TSF device closely resembled that of the unsensitized Device B, with both peaking at 473 nm and exhibiting nearly identical FWHMs of less than 19 nm (Fig. 6c), which differs significantly from the broad green emission of Device A. A slight low-energy tail observed in the EL spectrum of the TSF device is attributed to a minor emission contribution from the sensitizer. With the assistance of the sensitizer, the TSF device exhibited a maximum external quantum efficiency (EQEmax) of 28.7%, an efficiency roll-off of 30.2% at 1000 cd m−2, and a maximum luminance (Lmax) of 9199 cd m−2, all significantly superior to those of the unsensitized reference device (EQEmax = 22%, roll-off = 46.9%, and Lmax = 4888 cd m−2). These results suggest efficient FRET from the green-emissive copper(I) sensitizer to the deep-blue emitter ν-DABNA. To the best of our knowledge, this copper(I)-sensitized blue OLED not only ranks among the highest-efficiency OLEDs based on copper(I) complexes but also represents the first demonstration of deep-blue electroluminescence with EQE > 20% and CIE-y < 0.2 (Fig. 6f and Table S11).6a,c,d,f,7b,14,15c,19,22
| Device | V on , V | EQEb, % | Roll-offc, % | L max , cd m−2 | CEe, cd A−1 | PEf, Im W−1 | λ EL/FWHMg, nm | CIEh, (x, y) |
|---|---|---|---|---|---|---|---|---|
| a Turn-on voltage at 1 cd m−2. b EQE maximum value at 1000 cd m−2. c Efficiency roll-off at 1000 cd m−2. d Maximum luminance. e Maximum current efficiency. f Maximum power efficiency. g EL peak and half-peak width at 1000 cd m−2. h Commission Internatinale de L'Eclairage coordinates measured at 1000 cd m−2. | ||||||||
| Device B | 4.6 | 22/11.7 | 46.9 | 4888 | 20.1 | 12.6 | 473/18.5 | (0.12, 0.14) |
| TSF device | 4.3 | 28.7/20 | 30.2 | 9199 | 34.4 | 24.6 | 473/18.7 | (0.15, 0.19) |
Conclusions
In summary, we reported two sublimable TADF-emitting copper(I)-bromide clusters, [dppb-Ac]2Cu2Br2 and [dtpb-Ac]2Cu2Br2, coordinated by D–A type bisphosphine ligands. An ortho-methylation strategy was employed to modify the configuration of the Cu2Br2 cluster, thereby effectively suppressing nonradiative decay caused by excited-state distortions—a longstanding challenge in the design of high-efficiency copper(I) emitters. As a result, the optimized cluster [dtpb-Ac]2Cu2Br2 exhibits green emission with a high PLQY of up to 99% in doped films, substantially higher than the PLQY of 65% observed for the control cluster [dppb-Ac]2Cu2Br2. OLEDs doped with [dtpb-Ac]2Cu2Br2 as the terminal emitter exhibited highly efficient green emission, achieving a high maximum EQE of 25.1% and a broad emission with a FWHM greater than 95 nm. Leveraging the high efficiency and broad emission of [dtpb-Ac]2Cu2Br2, we successfully fabricated highly efficient green-sensitized deep-blue OLEDs by utilizing [dtpb-Ac]2Cu2Br2 as the sensitizer for the blue MR-TADF emitter ν-DABNA. The device achieved a maximum EQE reaching 28.7% and corresponding CIE coordinates of (0.15, 0.19). On the one hand, similar molecular design strategies are expected to be suitable for enhancing the emission efficiencies of copper(I) complexes in thin films by suppressing their intrinsic excited-state distortions. On the other hand, the broad emission spectra of copper(I) complexes—traditionally regarded as a limitation—can be harnessed as a beneficial feature to sensitize narrowband blue emitters, even when they exhibit green emission, thereby offering a new design strategy for copper(I)-based optoelectronic devices.Author contributions
X. L. C. and J. M. L. conceived the project. X. L. and X. L. C. designed the experiments and analysed the data. X. L. performed sample preparation, OLED fabrication, and property measurements and drafted the initial manuscript. S. G. conducted theoretical calculations. X. L. (Xin Liu) assisted with synthesis and property measurements. Y. F. S. provided guidance on device fabrication. D. H. Z. contributed to data analysis. H. Y. assisted with single-crystal analysis. X. L. C. and J. M. L. revised the manuscript. All authors discussed and commented on the manuscript.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.CCDC 2386729 and 2386762 contain the supplementary crystallographic data for this paper.23a,b
The data supporting this article are provided in the main manuscript and the supplementary information (SI). Supplementary information: general experimental information, detailed synthetic procedures, crystal structures, thermogravimetric analysis curves, electrochemical data, photophysical characterization, theoretical calculations, and device fabrication and characterization. See DOI: https://doi.org/10.1039/d5sc09307d.
Acknowledgements
This research was made possible as a result of a generous grant from the Major Research Project of Xiamen (Grant No. 3502Z20231052), the Natural Science Foundation of Fujian Province (Grant No. 2024H0034), and the Fujian Science and Technology Innovation Laboratory for Optoelectronic Information of China (Grant No. 2021ZR132 and 2021ZZ115).Notes and references
- (a) M. A. Baldo, D. F. O'Brien, Y. You, A. Shoustikov, S. Sibley, M. E. Thompson and S. R. Forrest, Nature, 1998, 395, 151–154 CrossRef CAS; (b) H. Uoyama, K. Goushi, K. Shizu, H. Nomura and C. Adachi, Nature, 2012, 492, 234–238 CrossRef CAS PubMed; (c) Y. Liu, C. Li, Z. Ren, S. Yan and M. R. Bryce, Nat. Rev. Mater., 2018, 3, 18020 Search PubMed.
- (a) H. Fu, Y. M. Cheng, P. T. Chou and Y. Chi, Mater. Today, 2011, 14, 472–479 Search PubMed; (b) H. Yersin, A. F. Rausch, R. Czerwieniec, T. Hofbeck and T. Fischer, Coord. Chem. Rev., 2011, 255, 2622–2652 CrossRef CAS.
- T. Hatakeyama, K. Shiren, K. Nakajima, S. Nomura, S. Nakatsuka, K. Kinoshita, J. Ni, Y. Ono and T. Ikuta, Adv. Mater., 2016, 28, 2777–2781 Search PubMed.
- (a) D. Zhang, L. Duan, C. Li, Y. Li, H. Li, D. Zhang and Y. Qiu, Adv. Mater., 2014, 26, 5050–5055 Search PubMed; (b) H. Nakanotani, T. Higuchi, T. Furukawa, K. Masui, K. Morimoto, M. Numata, H. Tanaka, Y. Sagara, T. Yasuda and C. Adachi, Nat. Commun., 2014, 5, 4016 Search PubMed.
- (a) C. Y. Chan, M. Tanaka, Y. T. Lee, Y. W. Wong, H. Nakanotani, T. Hatakeyama and C. Adachi, Nat. Photonics, 2021, 15, 203–207 Search PubMed; (b) S. O. Jeon, K. H. Lee, J. S. Kim, S. G. Ihn, Y. S. Chung, J. W. Kim, H. Lee, S. Kim, H. Choi and J. Y. Lee, Nat. Photonics, 2021, 15, 208–215 Search PubMed; (c) D. Zhang and L. Duan, Nat. Photonics, 2021, 15, 173–174 Search PubMed; (d) K. Stavrou, L. G. Franca, A. Danos and A. P. Monkman, Nat. Photonics, 2024, 18, 554–561 Search PubMed; (e) T. Hua, X. Cao, J. Miao, X. Yin, Z. Chen, Z. Huang and C. Yang, Nat. Photonics, 2024, 18, 1161–1169 CrossRef CAS; (f) H. Lee, R. Braveenth, S. Muruganantham, C. Y. Jeon, H. S. Lee and J. H. Kwon, Nat. Commun., 2023, 14, 419 CrossRef CAS PubMed; (g) Z. Xiao, Y. Zou, Z. Chen, J. Miao, Y. Qiu, Z. Huang, X. Cao, X. Peng and C. Yang, Adv. Mater., 2025, 37, 2419601 CrossRef CAS PubMed; (h) Y. Sun, X. F. Fu, C. L. Hou, T. T. Lin, D. H. Zhang, J. Liu, J. X. Hu, F. L. Lin, L. Zhou, L. Meng, X. L. Chen and C. Z. Lu, Angew. Chem., Int. Ed., 2025, 64, e202511921 Search PubMed.
- (a) M. Hashimoto, S. Igawa, M. Yashima, I. Kawata, M. Hoshino and M. Osawa, J. Am. Chem. Soc., 2011, 133, 10348–10351 CrossRef CAS PubMed; (b) M. J. Leitl, V. A. Krylova, P. I. Djurovich, M. E. Thompson and H. Yersin, J. Am. Chem. Soc., 2014, 136, 16032–16038 CrossRef CAS PubMed; (c) R. Hamze, J. L. Peltier, D. Sylvinson, M. Jung, J. Cardenas, R. Haiges, M. Soleilhavoup, R. Jazzar, P. I. Djurovich, G. Bertrand and R. E. Thompson, Science, 2019, 363, 601–606 Search PubMed; (d) S. Shi, M. C. Jung, C. Coburn, A. Tadle, M. R. D. Sylvinson, P. I. Djurovich, S. R. Forrest and M. E. Thompson, J. Am. Chem. Soc., 2019, 141, 3576–3588 Search PubMed; (e) A. M. T. Muthig, O. Mrozek, T. Ferschke, M. Rodel, B. Ewald, J. Kuhnt, C. Lenczyk, J. Pflaum and A. Steffen, J. Am. Chem. Soc., 2023, 145, 4438–4449 CrossRef CAS PubMed; (f) N. Zhang, L. Qu, S. Dai, G. Xie, C. Han, J. Zhang, R. Huo, H. Hu, Q. Chen, W. Huang and H. Xu, Nat. Commun., 2023, 14, 2901 Search PubMed; (g) T. Y. Li, S. J. Zheng, P. I. Djurovich and M. E. Thompson, Chem. Rev., 2024, 124, 4332–4392 Search PubMed.
- (a) X. L. Chen, R. Yu, Q. K. Zhang, L. J. Zhou, X. Y. Wu, Q. Zhang and C. Z. Lu, Chem. Mater., 2013, 25, 3910–3920 CrossRef CAS; (b) L. Zhan, A. Ying, Y. Qi, K. Wu, Y. Tang, Y. Tan, Y. Zou, G. Xie, S. Gong and C. Yang, Adv. Funct. Mater., 2021, 31, 2106345 Search PubMed; (c) Y. Tan, A. Ying, J. Xie, G. Xie and S. Gong, Chem. Sci., 2024, 15, 11382–11390 Search PubMed; (d) T. Y. Li, S. J. Zheng, P. I. Djurovich and M. E. Thompson, Chem. Rev., 2024, 124, 4332–4392 CrossRef CAS PubMed.
- (a) C. T. Cunningham, J. J. Moore, K. L. H. Cunningham, P. E. Fanwick and D. R. McMillin, Inorg. Chem., 2000, 39, 3638–3644 Search PubMed; (b) M. Iwamura, S. Takeuchi and T. Tahara, J. Am. Chem. Soc., 2007, 129, 5248–5256 CrossRef CAS PubMed.
- H. Yersin and U. Monkowius, Adv. Photonics Res., 2025, 6, 2400111 Search PubMed.
- (a) X. Zhang and H. Xu, Angew. Chem., Int. Ed., 2024, 63, e202317597 Search PubMed; (b) Y. Li, X. Zhang, Y. Man, S. Xu, J. Zhang, G. Zhang, S. Chen, C. Duan, C. Han and H. Xu, Adv. Mater., 2023, 35, e2302984 CrossRef PubMed; (c) X. L. Chen, R. Yu, X. Y. Wu, D. Liang, J. H. Jia and C. Z. Lu, Chem. Commun., 2016, 52, 6288–6291 RSC.
- H. Yersin, R. Czerwieniec, M. Z. Shafikov and A. F. Suleymanova, ChemPhysChem, 2017, 18, 3508–3535 CrossRef CAS PubMed.
- R. Czerwieniec, M. J. Leitl, H. H. H. Homeier and H. Yersin, Coord. Chem. Rev., 2016, 325, 2–28 CrossRef CAS.
- P. C. Ford, E. Cariati and J. Bourassa, Chem. Rev., 1999, 99, 3625–3647 CrossRef CAS PubMed.
- (a) M. Xie, C. Han, Q. Liang, J. Zhang, G. Xie and H. Xu, Sci. Adv., 2019, 5, eaav9857 Search PubMed; (b) N. Zhang, H. Hu, L. Qu, R. Huo, J. Zhang, C. Duan, Y. Meng, C. Han and H. Xu, J. Am. Chem. Soc., 2022, 144, 6551–6557 Search PubMed.
- (a) A. Tsuboyama, K. Kuge, M. Furugori, S. Okada, M. Hoshino and M. Ueno, Inorg. Chem., 2007, 46, 1992–2001 CrossRef CAS PubMed; (b) L. Kang, J. Chen, T. Teng, X. L. Chen, R. Yu and C. Z. Lu, Dalton Trans., 2015, 44, 11649–11659 RSC; (c) D. Volz, Y. Chen, M. Wallesch, R. Liu, C. Flechon, D. M. Zink, J. Friedrichs, H. Flugge, R. Steininger, J. Gottlicher, C. Heske, L. Weinhardt, S. Brase, F. So and T. Baumann, Adv. Mater., 2015, 27, 2538–2543 CrossRef CAS PubMed.
- (a) D. Zhang, X. Song, M. Cai and L. Duan, Adv. Mater., 2018, 30, 1705250 Search PubMed; (b) H. Mubarok, A. Amin, T. Lee, J. Jung, J. H. Lee and M. H. Lee, Angew. Chem., Int. Ed., 2023, 62, e202306879 CrossRef CAS PubMed.
- J. H. Jia, D. Liang, R. Yu, X. L. Chen, L. Meng, J. F. Chang, J. Z. Liao, M. Yang, X. N. Li and C. Z. Lu, Chem. Mater., 2019, 32, 620–629 CrossRef.
- R. Hamze, J. L. Peltier, D. Sylvinson, M. Jung, J. Cardenas, R. Haiges, M. Soleilhavoup, R. Jazzar, P. I. Djurovich, G. Bertrand and R. E. Thompson, Science, 2019, 363, 601–606 Search PubMed.
- J. C. Deaton, S. C. Switalski, D. Y. Kondakov, R. H. Young, T. D. Pawlik, D. J. Giesen, S. B. Harkins, A. J. Miller, S. F. Mickenberg and J. C. Peters, J. Am. Chem. Soc., 2010, 132, 9499–9508 CrossRef CAS PubMed.
- (a) J. X. Hu, S. Jiang, D. H. Zhang, T. Zhao, F. L. Lin, L. Meng, X. L. Chen and C. Z. Lu, Adv. Sci., 2023, 10, 2300808 CrossRef CAS PubMed; (b) X. L. Chen, X. D. Tao, Y. S. Wang, Z. Wei, L. Meng, D. H. Zhang, F. L. Lin and C. Z. Lu, CCS Chem., 2023, 5, 589–597 CrossRef CAS.
- Y. Y. Jing, N. Li, X. Cao, H. Wu, J. Miao, Z. Chen, M. Huang, X. Wang, Y. Hu, Y. Zou and C. Yang, Sci. Adv., 2023, 9, eadh8296 CrossRef CAS PubMed.
- (a) M. Osawa, M. Hoshino, M. Hashimoto, I. Kawata, S. Igawa and M. Yashima, Dalton Trans., 2015, 44, 8369–8378 RSC; (b) A. Verma, D. M. Zink, C. Fléchon, J. Leganés Carballo, H. Flügge, J. M. Navarro, T. Baumann and D. Volz, Appl. Phys. A, 2016, 122, 191 CrossRef; (c) J. Zhang, C. Duan, C. Han, H. Yang, Y. Wei and H. Xu, Adv. Mater., 2016, 28, 5975–5979 CrossRef CAS PubMed; (d) D. Di, A. S. Romanov, L. Yang, J. M. Richter, J. P. H. Rivett, S. Jones, T. H. Thomas, M. A. Jalebi, R. H. Friend, M. Linnolahti, M. Bochmann and D. Credgington, Science, 2017, 356, 159–163 Search PubMed; (e) X. Li, J. Zhang, Z. Zhao, X. Yu, P. Li, Y. Yao, Z. Liu, Q. Jin, Z. Bian, Z. Lu and C. Huang, ACS Appl. Mater. Interfaces, 2019, 11, 3262–3270 CrossRef CAS PubMed; (f) M. Klein, N. Rau, M. Wende, J. Sundermeyer, G. Cheng, C.-M. Che, A. Schinabeck and H. Yersin, Chem. Mater., 2020, 32, 10365–10382 Search PubMed; (g) A. Ying, Y. H. Huang, C. H. Lu, Z. Chen, W. K. Lee, X. Zeng, T. Chen, X. Cao, C. C. Wu, S. Gong and C. Yang, ACS Appl. Mater. Interfaces, 2021, 13, 13478–13486 CrossRef CAS PubMed; (h) Q. Gu, F. Chotard, J. Eng, A. M. Reponen, I. J. Vitorica-Yrezabal, A. W. Woodward, T. J. Penfold, D. Credgington, M. Bochmann and A. S. Romanov, Chem. Mater., 2022, 34, 7526–7542 Search PubMed; (i) A. Ruduss, B. Turovska, S. Belyakov, K. A. Stucere, A. Vembris, G. Baryshnikov, H. Agren, J. C. Lu, W. H. Lin, C. H. Chang and K. Traskovskis, ACS Appl. Mater. Interfaces, 2022, 14, 15478–15493 CrossRef CAS PubMed; (j) R. Tang, S. Xu, T. L. Lam, G. Cheng, L. Du, Q. Wan, J. Yang, F. F. Hung, K. H. Low, D. L. Phillips and C. M. Che, Angew. Chem., Int. Ed., 2022, 61, e202203982 CrossRef CAS PubMed; (k) Y. Li, S. Xu, X. Zhang, Y. Man, J. Zhang, G. Zhang, S. Chen, C. Duan, C. Han and H. Xu, Angew. Chem., Int. Ed., 2023, 62, e202308410 CrossRef CAS PubMed; (l) H. J. Wang, Y. Liu, B. Yu, S. Q. Song, Y. X. Zheng, K. Liu, P. Chen, H. Wang, J. Jiang and T. Y. Li, Angew. Chem., Int. Ed., 2023, 62, e202217195 CrossRef CAS PubMed; (m) N. Zhang, Y. Li, S. Han, Y. Wei, H. Hu, R. Huo, C. Duan, J. Zhang, C. Han, G. Xie and H. Xu, Angew. Chem., Int. Ed., 2023, 62, e202305018 CrossRef CAS PubMed; (n) R. Li, A. Ying, Y. Tan, Y. Ai and S. Gong, Chem.–Eur. J., 2024, 30, e202400817 CrossRef CAS PubMed; (o) J. J. Wang, L. Z. Feng, G. Shi, J. N. Yang, Y. D. Zhang, H. Xu, K. H. Song, T. Chen, G. Zhang, X. S. Zheng, F. Fan, Z. Xiao and H. B. Yao, Nat. Photonics, 2023, 18, 200–206 Search PubMed; (p) A. Ying, Y. Tan and S. Gong, Adv. Opt. Mater., 2024, 12, 230333 Search PubMed; (q) Y. Tan, A. Ying, Y. Liu, X. Cai, L. Zhan, Z. Bin, J. You, C. Li and S. Gong, Chem. Eng. J., 2024, 487, 150618 CrossRef CAS.
- (a) CCDC 2386729: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2l3l8l; (b) CCDC 2386762: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2l3mbp.
| This journal is © The Royal Society of Chemistry 2026 |