Open Access Article
Open Access ArticleFrom centrosymmetric (C4H8N5)(SbF4) to polar (C4H7N4O)(SbF4): a new UV nonlinear optical material achieved by functional group modulation
Zhi-Xiang
Wang
ac,
Chun-Li
Hu
a,
Chuan-Fu
Sun
 *b,
Jiang-Gao
Mao
*b,
Jiang-Gao
Mao
 a and
Fang
Kong
a and
Fang
Kong
 *ab
*ab
aState Key Laboratory of Functional Crystals and Devices, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou 350002, P. R. China. E-mail: kongfang@fjirsm.ac.cn
bState Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences, Fuzhou 350002, P. R. China. E-mail: cfsun@fjirsm.ac.cn
cCollege of Chemistry, Fuzhou University, Fuzhou 350108, P. R. China
First published on 2nd February 2026
Abstract
The design and synthesis of new ultraviolet nonlinear optical (NLO) materials is challenging due to the constraint relationship between their NLO efficiency and optical bandgap. Using a functional group modulation strategy, the π-conjugated organic cations and the distorted [SbF4]− anions are assembled in both antiparallel and parallel arrangement modes, resulting in two new organic–inorganic hybrid fluoroantimonites, namely, centrosymmetric (C4H8N5)(SbF4) and polar (C4H7N4O)(SbF4). The polar (C4H7N4O)(SbF4) demonstrates excellent comprehensive properties, including a strong SHG effect (4.2 × KDP), a wide bandgap (4.40 eV) and a short phase matchable wavelength (263 nm). This SHG intensity is the largest among the organic–inorganic hybrid perfluoroantimonites with an optical bandgap >4.20 eV. The SHG density calculation indicates that the NLO performance of (C4H7N4O)(SbF4) mainly comes from the parallel-arranged organic groups, with their contribution accounting for 89.49%. This work proves that the arrangement of π-conjugated organic ligands can be modulated to a parallel mode by adjusting the number of hydrogen bond donors. Combined with a wide HOMO–LUMO gap, the functional group modulation method is an effective strategy for designing and synthesizing ultraviolet NLO materials.
Introduction
Ultraviolet nonlinear optical (UV NLO) materials play a significant role in modern science and technology due to their ability to extend the wavelength range of lasers to the ultraviolet region through the second harmonic generation (SHG) process. Broad applications can be found in optical sensors, quantum communication, biological imaging, etc.1–7 It is well known that excellent UV nonlinear crystals should meet the following conditions: a broad bandgap, a strong SHG response, an appropriate birefringence, and excellent physicochemical stability.2,8–13 However, due to the bandgap being inversely proportional to the SHG efficiency, it is very difficult to combine the above excellent properties in a single compound.Fluoroantimonites have been regarded as a potential UV NLO material system,14,15 because (i) Sb3+ ions with stereochemically active lone pairs usually can undergo second-order Jahn–Teller distortion, resulting in high anisotropy, (ii) the presence of fluorine elements causes a blue shift in the bandgap. Many inorganic fluoroantimonites have been reported as promising UV NLO materials, such as Rb2Sb(P2O7)F (5.1 × KDP, 4.76 eV) and CsSbF2SO4 (3.0 × KDP, 4.76 eV).16,17 However, recent studies on UV NLO fluoroantimonites were mainly focused on metal sulfates and phosphates. Although these tetrahedral groups have a positive effect on the large bandgaps of the materials, the non-polar configuration limits the contribution of SHG intensity.18–22 For such compounds an SHG response that is four times that of KDP is usually difficult to reach, such as Rb2SO4·(SbF3)2 (0.5 × KDP, 4.69 eV), NH4SbF2SO4 (0.7 × KDP, 4.67 eV), K2Sb(P2O7)F (4.0 × KDP, 4.74 eV), etc.23–25 Thus, it is necessary to expand the types of functional groups beyond tetrahedra to enhance the SHG response of fluoroantimonites.
Recently, it has been found that compared with traditional inorganic π-conjugated units (BO33−, B3O63−, etc), organic π-conjugated units not only possess greater hyperpolarizability and anisotropy26–30 but also exhibit rich structural tunability (Fig. 1). If they can be introduced into fluoroantimonites, it is expected to break the current situation of generally low SHG intensity of UV transparent fluoroantimonites. Lately, Zhang and his coworkers reported a new oxyfluoroantimonite, namely, (C5H5NO)(Sb2OF4), which can exhibit a large SHG response and a wide bandgap.31 This work demonstrates the feasibility of exploring UV NLO materials in fluoroantimonites with organic π-conjugated groups. However, it is the only case breaking through 4 times that of KDP among the reported UV transparent organic–inorganic hybrid fluoroantimonites. Furthermore, noncentrosymmetric (NCS) fluoroantimonites with organic π-conjugated groups are also scarce, although such compounds contain two kinds of SHG functional units. The reason can be ascribed to the anti-parallel arrangement of the organic section, especially for those with large hyperpolarizability.
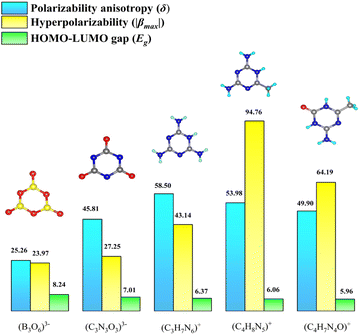 | ||
| Fig. 1 The calculated values of polarizability anisotropy, hyperpolarizability and HOMO–LUMO gap for optically active groups. | ||
It is reported that hydrogen bonding can optimize the arrangement of organic units.32–34 For example, by regulating the degree of intermolecular hydrogen bonds, compound TM1 crystallizes in the NCS space group (P1), showing a relatively large SHG response (3 × KDP), while compound TM2 crystallizes in the centrosymmetry (CS) space group (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) in an antiparallel arrangement.35 Inspired by these studies, our strategy is to modulate the type of functional group to change the number of the hydrogen bond donors (HBDs) and construct a new hydrogen bond network, with the aim of regulating the macroscopic symmetry of the structure (Fig. 2). After thorough screening, (C3H7N6)2(SbF5)·H2O with a melamine cation came into our focus.36 On the one hand, melamine has enough amino functional groups, which can be replaced by non-HBDs, such as methyl and carbonyl groups. On the other hand, the modulated cations, such as (C4H8N5)+ and (C4H7N4O)+, can exhibit higher hyperpolarizability than that of (C3H7N6)+ while their HOMO–LUMO gaps still remain at a relatively high level (Fig. 1). It is expected to obtain a new UV NLO material through a functional group modulation strategy in organic–inorganic hybrid fluoroantimonites.
) in an antiparallel arrangement.35 Inspired by these studies, our strategy is to modulate the type of functional group to change the number of the hydrogen bond donors (HBDs) and construct a new hydrogen bond network, with the aim of regulating the macroscopic symmetry of the structure (Fig. 2). After thorough screening, (C3H7N6)2(SbF5)·H2O with a melamine cation came into our focus.36 On the one hand, melamine has enough amino functional groups, which can be replaced by non-HBDs, such as methyl and carbonyl groups. On the other hand, the modulated cations, such as (C4H8N5)+ and (C4H7N4O)+, can exhibit higher hyperpolarizability than that of (C3H7N6)+ while their HOMO–LUMO gaps still remain at a relatively high level (Fig. 1). It is expected to obtain a new UV NLO material through a functional group modulation strategy in organic–inorganic hybrid fluoroantimonites.
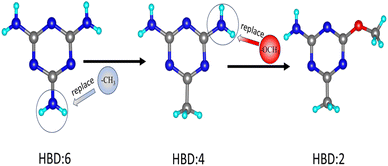 | ||
| Fig. 2 The hydrogen bond donor (HBD) number decreases when the amino group has been replaced with methyl and methoxy groups. | ||
According to the above strategic guidance, two novel organic–inorganic hybrid fluoroantimonites, namely, (C4H8N5)(SbF4) and (C4H7N4O)(SbF4), have been successfully obtained by replacing the amino groups in melamine with methyl and carbonyl groups. With the decrease in hydrogen bonds between the organic cations, the structure undergoes a transformation from CS (P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) ) to NCS (Pc). As expected, (C4H7N4O)(SbF4) demonstrates an excellent comprehensive performance including a strong response (4.2 × KDP), a wide bandgap (4.40 eV), and a short UV cut-off edge (263 nm). It represents the second UV transparent organic–inorganic hybrid fluoroantimonite with SHG intensity higher than 4 × KDP. Herein, we report the syntheses, structures and optical properties of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4).
) to NCS (Pc). As expected, (C4H7N4O)(SbF4) demonstrates an excellent comprehensive performance including a strong response (4.2 × KDP), a wide bandgap (4.40 eV), and a short UV cut-off edge (263 nm). It represents the second UV transparent organic–inorganic hybrid fluoroantimonite with SHG intensity higher than 4 × KDP. Herein, we report the syntheses, structures and optical properties of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4).
Results and discussion
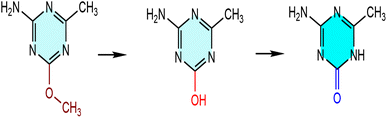
Since methyl groups are hydrophobic, if two of the hydrophilic amino groups on melamine molecules are replaced by methyl groups, it will reduce the solubility of the organic molecules in water, which is not conducive to crystal growth. So, the methoxy group with both hydrophilic and hydrophobic properties has been used to replace the amino group. It's worth noting that the methoxy group can undergo a demethylation reaction in an acidic environment to form a hydroxyl group, which then leads to enol isomerization and the formation of a ketone structure eventually37,38 (Fig. 3). The demethylation of the methoxy group can be proved by the 1H NMR spectrum (Fig. S1). No peak is observed at δ 3.60 ppm, which indicates that the methoxy group is no longer in the organic framework. A sharp single peak and a wide single peak appears at δ 2.10 ppm and 7.00 ppm, with the integral area corresponding to 3 hydrogen protons and 2 hydrogen protons, which can be respectively attributed to the proton signals of the methyl and amino groups.Both (C4H8N5)(SbF4) and (C4H7N4O)(SbF4) single crystals were obtained by volatilizing aqueous solutions at room temperature (Fig. S2). Details of the synthesis procedure are outlined in the SI under the section titled “Syntheses”. The purity of these crystals was verified using X-ray diffraction (XRD), as depicted in Fig. S3. Crystallographic details of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4) can be found in Table S1. Thermogravimetric (TG) tests show that (C4H7N4O)(SbF4) is thermally stable at temperatures up to 105 °C and decomposes slowly when the temperature exceeds this value. (C4H8N5)(SbF4) starts to decompose when the temperature exceeds 185 °C (Fig. S4). We believe that the significant difference in their thermal stability is mainly caused by the difference of hydrogen bond numbers in their unit cell.
(C4H8N5)(SbF4) crystallizes in the triclinic system with the space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) (no. 2), with the lattice parameters a = 5.0998(3) Å, b = 9.4607(5) Å, c = 10.3378(6) Å, α = 68.330(5)°, β = 77.847(5)°, γ = 85.276(5)° and V = 453.13(5) Å3. There is one (C4H8N5)+ cation and one SbF4− anion in its asymmetric unit. Antimony is four-coordinated, producing a [SbF4]− seesaw, with the bond length of Sb–F ranging from 1.9161(16) to 2.0646(16) Å. The bond-valence-sum results show that the bond valence is 3.046, indicating an oxidation state of +3. (C4H8N5)+ cations are connected to [SbF4]− by N–H⋯F hydrogen bonding with the hydrogen bond length ranging from 1.757 to 2.056 Å (Fig. 4c). The adjacent (C4H8N5)+ cations are then connected by a trans-arrangement of N–H⋯N hydrogen bonds. The ordered arrangement of cations and anions interspersed along the c-axis forms the structure of (C4H8N5)(SbF4) (Fig. 4d).
(no. 2), with the lattice parameters a = 5.0998(3) Å, b = 9.4607(5) Å, c = 10.3378(6) Å, α = 68.330(5)°, β = 77.847(5)°, γ = 85.276(5)° and V = 453.13(5) Å3. There is one (C4H8N5)+ cation and one SbF4− anion in its asymmetric unit. Antimony is four-coordinated, producing a [SbF4]− seesaw, with the bond length of Sb–F ranging from 1.9161(16) to 2.0646(16) Å. The bond-valence-sum results show that the bond valence is 3.046, indicating an oxidation state of +3. (C4H8N5)+ cations are connected to [SbF4]− by N–H⋯F hydrogen bonding with the hydrogen bond length ranging from 1.757 to 2.056 Å (Fig. 4c). The adjacent (C4H8N5)+ cations are then connected by a trans-arrangement of N–H⋯N hydrogen bonds. The ordered arrangement of cations and anions interspersed along the c-axis forms the structure of (C4H8N5)(SbF4) (Fig. 4d).
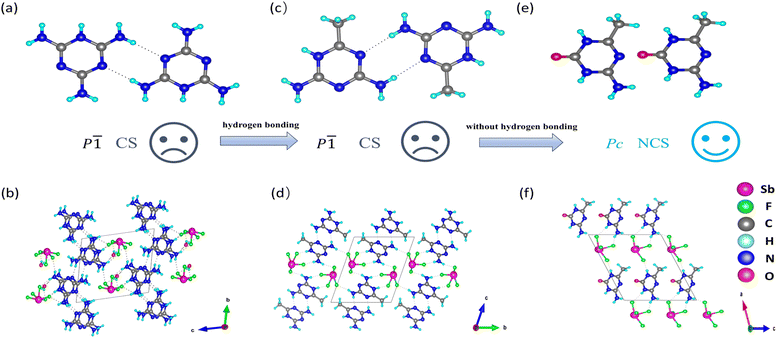 | ||
| Fig. 4 The arrangement patterns of (C3N6H7)+ (a), (C4H8N5)+ (c) and (C4H7N4O)+ (e). The crystal structures of (C3N6H7)2(SbF5)·H2O (b), (C4H8N5)(SbF4) (d) and (C4H7N4O)(SbF4) (f). | ||
(C4H7N4O)(SbF4) crystallizes in the monoclinic space group Pc (no. 7). The lattice parameters are a = 11.4719(5) Å, b = 4.6982(2) Å, c = 9.1703(4) Å, α = γ = 90°, β = 113.104(5)° and V = 454.61(4) Å3. Its asymmetric unit contains one SbF4− anion and one (C4H7N4O)+ cation. The Sb–F distance is in the range of 1.927(3) to 2.068(3) Å. The bond-valence-sum results show that the bond valence is 2.987, suggesting that the oxidation state is +3 (Table S2). C4H7N4O+ cations are connected to [SbF4]−via N–H⋯F hydrogen bonding (Fig. 4f) with the hydrogen bond length ranging from 1.820 to 1.946 Å. Each SbF4− anion is fixed by three (C4H7N4O)+ cations and arranged along the c-axis, forming a new three-dimensional network. The dipole moment of the [SbF4] seesaw in (C4H7N4O)(SbF4) was calculated. Its local dipole moment is 9.18–9.98 D and the net dipole moment for a unit cell is 16.40 D, which implies its polar nature.
From (C3N6H7)2(SbF5)·H2O to (C4H8N5)(SbF4) and then to (C4H7N4O)(SbF4), the crystal structure realizes the transformation from a CS to a NCS structure. To better illustrate this transition, we make a detailed comparison of the three crystals. We can see that in their basic structural unit, the anions do not change much except from [SbF5]2− to [SbF4]−, and the difference is mainly in the arrangement of cations. As shown in Table S3, the bond lengths and bond angles of the N–H⋯F hydrogen bonds in these three crystals change very little, but in (C4H7N4O)(SbF4), the numbers of both N–H⋯F and N–H⋯N hydrogen bonds have decreased. Due to the large number of hydrogen bond donors (–NH2 and –NH+), it can be seen that in (C3N6H7)2(SbF5)·H2O and (C4H8N5)(SbF4), even though a methyl group has been replaced by an amino group, organic cations can still be connected by N–H⋯N hydrogen bonds, forming a trans-arrangement (Fig. 4a and c), which is not favourable for the formation of NCS structures. However, in (C4H7N4O)(SbF4), after undergoing enol tautomerism, another amino group in the organic molecule is substituted by an oxygen atom. The number of hydrogen bond donors is reduced, and the four strong electronegative F atoms in the anions are sufficient to anchor them all, which causes the connection between the organic cations to be shielded, breaking the original trans-arrangement (Fig. 4e).
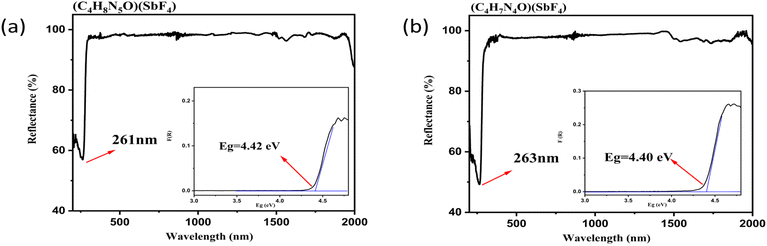
The UV-vis-NIR diffuse reflectance spectra (Fig. 5a and b) show that the UV cutoff edges of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4) are 261 nm and 263 nm, respectively, corresponding to experimental bandgaps of 4.42 eV and 4.40 eV. In addition, it is worth noting that the UV cutoff edge of the template (C3N6H7)2(SbF5)·H2O is 261 nm with an experimental bandgap of 4.74 eV, which can indicate that our modification of the organic groups has almost no effect on the UV cutoff edge, and this is conducive to the design of new UV transmittable crystals.
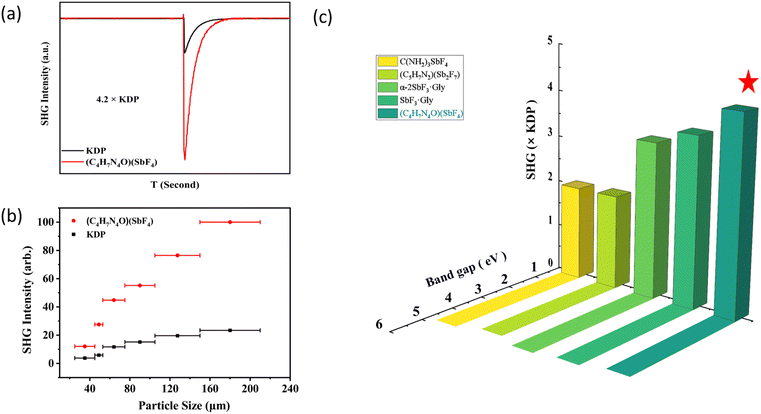
Based on the Kurtz–Perry method,39–42 the powder SHG measurement of (C4H7N4O)(SbF4) was accomplished under 1064 nm laser irradiation, and standard KH2PO4 (KDP) crystals were used as a reference. As shown in Fig. 6a, (C4H7N4O)(SbF4) displays a strong SHG effect of 4.2 × KDP. Meanwhile, as the particle size increases, the intensity of SHG also increases, which indicates that it has phase matching (PM) capability (Fig. 6b). To evaluate the excellent properties of (C4H7N4O)(SbF4), we further compared it with some reported UV inorganic antimonates and UV organic–inorganic hybrid fluoroantimonates (Table S4) such as NaSbF4 (3.2 × KDP), (NH4)SbCl2(SO4) (1.7 × KDP), CsSbF2SO4 (3 × KDP), and Rb2SO4·(SbF3)2 (2.2 × KDP) and C(NH2)3SbF4 (2.0 × KDP), SbF3·Gly (3.6 × KDP), (C5H7N2)Sb2F7 (2.0 × KDP), and α-2SbF3·Gly (3.3 × KDP).17,23,31,43–47 Among organic–inorganic hybrid antimony-based UV NLO materials, its SHG response is only lower than that of (C5H5NO)(Sb2OF4). So, (C4H7N4O)(SbF4) possesses the largest SHG intensity in organic–inorganic hybrid perfluoroantimonites with a bandgap >4.20 eV (Fig. 6c). The r-on-1 test scheme was used to detect the powder LIDT of (C4H7N4O)(SbF4) and AgGaS2, yielding values of 65 and 4 MW cm−2, indicating that the LIDT of (C4H7N4O)(SbF4) is about 16 times that of AgGaS2.
It is known that appropriate birefringence is essential for efficient phase matching during the SHG process. High birefringence exacerbates walk-off issues, while low birefringence makes phase matching challenging to achieve.48–50 Thus, the birefringence was analyzed (Fig. 7). Fig. 7a shows the calculated birefringence curves of (C4H7N4O)(SbF4) and its birefringence was calculated to be 0.155 at 546 nm, which falls between that of (C3N6H7)2(SbF5)·H2O (0.380@550 nm) and (C4H8N5)(SbF4) (0.059@546 nm) (Fig. S5). From Fig. 7b we can find that the shortest phase matching wavelength of (C4H7N4O)(SbF4) reaches 248 nm. However, because its UV cut off edge was measured to be 263 nm (Fig. 5b), so its phase matching limit should be 263 nm and (C4H7N4O)(SbF4) is a full-wavelength phase matching compound.
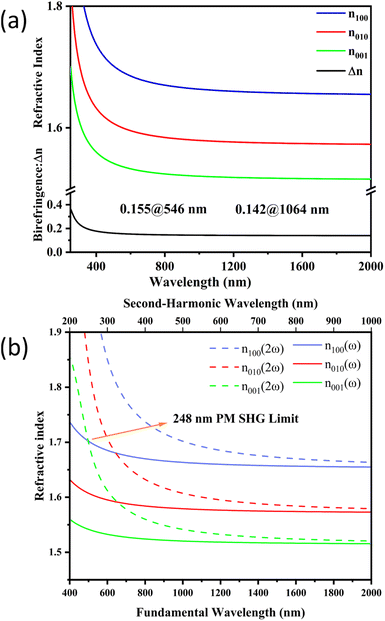 | ||
| Fig. 7 The calculated birefringence curves of (C4H7N4O)(SbF4) (a) and the calculated shortest phase matching wavelength of (C4H7N4O)(SbF4) (b) based on the calculated refractive indices. | ||
The infrared (IR) spectra of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4) were analyzed in the wavelength range of 4000–400 cm−1 at room temperature. Fig. S6 shows the IR spectra of the compounds. The peaks at 3375 and 3183 cm−1 correspond to N–H vibrations. The absorption peaks observed at 2932–2769 cm−1 and 1032–722 cm−1 can be attributed to C–H vibrations. Sharp peaks between 1760 and 1600 cm−1 are associated with C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O vibration. The peaks around 1535–1308 cm−1 and 1082 cm−1 are assigned to –CH3 bending and C–N vibrations, respectively. In addition, the peaks of Sb–F vibrations can be observed at 600–450 cm−1 (Table S5). The above attributions are consistent with the reports of the previous compounds.51
O vibration. The peaks around 1535–1308 cm−1 and 1082 cm−1 are assigned to –CH3 bending and C–N vibrations, respectively. In addition, the peaks of Sb–F vibrations can be observed at 600–450 cm−1 (Table S5). The above attributions are consistent with the reports of the previous compounds.51
To further understand the structure–property relationship of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4), theoretical calculation was performed using density functional theory (DFT).52 The calculation results along the high-symmetry points of the first Brillouin zone show that (C4H8N5)(SbF4) and (C4H7N4O)(SbF4) are indirect band gap compounds with theoretical bandgaps of 3.23 eV and 2.89 eV (Fig. S7), respectively. Both experimental bandgaps are larger than the calculated values, which might be due to the limitation of the generalized gradient approximation method. Therefore, in an effort to analyse the optical properties of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4) accurately, the scissor operator with 1.19 eV and 1.51 eV was utilized.
The partial and total density of states (DOS) of (C4H8N5)(SbF4) and (C4H7N4O)(SbF4) are shown in Fig. 8a and S8. For (C4H8N5)(SbF4), the top of the valence band (VB) is mainly occupied by Sb-5s5p and F-2p, whereas the bottom of the conduction band (CB) is primarily contributed by N-2p and C-2p, indicating that the bandgap of (C4H8N5)(SbF4) is dominated by Sb, F, N and C atoms. The VB near the Fermi level of (C4H7N4O)(SbF4) is predominately composed of Sb-5s5p and F-2p, while the CB bottom is mainly consisted of N-2p and C-2p. It indicates that the bandgap of (C4H7N4O)(SbF4) is primarily determined by Sb, F, N and C atoms. According to the SHG density data, its NLO performance mainly comes from organic assembly (Fig. 8b and c), with its contribution accounting for as high as 89.49%. Although the inorganic [SbF4] unit contributes little (10.50%) to the SHG effect, it is crucial for the formation of the NCS structure due to its polar nature.
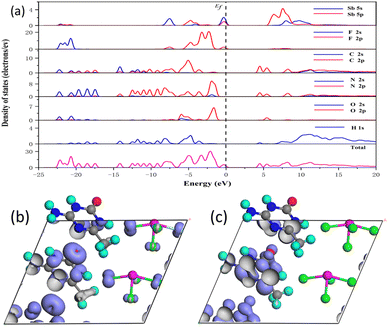 | ||
| Fig. 8 The total and partial density of states (a) and SHG density of d11 in the VB (b) and the CB (c) for (C4H7N4O)(SbF4). | ||
Conclusions
In conclusion, we have created an NCS organic–inorganic hybrid fluoroantimonite with excellent SHG performance by adopting a functional group modulation strategy. The inorganic part was focused on the [SbF4]− group while the organic part was changed from (C4H8N5)+ to (C4H7N4O)+. When the HBD number was decreased from 5 to 4, the arrangement of the organic units was changed from anti-parallel mode to parallel mode. The polarity of organic and inorganic groups has been combined mutually, resulting in the polar structure of (C4H7N4O)(SbF4). As we expected, (C4H7N4O)(SbF4) features a strong SHG intensity of about 4.2 × KDP, a wide bandgap of 4.40 eV and a short PM wavelength of 263 nm. Compared with the reported work, the SHG intensity of (C4H7N4O)(SbF4) is the largest among the organic–inorganic hybrid perfluoroantimonites with an optical bandgap >4.20 eV. Our work not only provides a promising candidate for UV NLO materials but also offers an effective strategy for novel SHG materials design.Author contributions
Zhi-Xiang Wang: investigation, formal analysis, experimental manipulation, writing – original draft. Chun-Li Hu: theoretical calculations. Chuan-Fu Sun: resources, formal analysis, writing – review & editing. Jiang-Gao Mao: supervision, resources, funding acquisition. Fang Kong: conceptualization, project administration, writing – review & editing. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the findings of this study are available in the supplementary information (SI) of this article. Supplementary information: experimental section, computational method, crystal data, important bond distances and bond valences, elemental analysis, PXRD patterns, TGA curves, IR spectra, UV-visible-NIR diffuse reflectance spectra and band structures. See DOI: https://doi.org/10.1039/d5sc09256f.CCDC 2474438 and 2474439 contain the supplementary crystallographic data for this paper.53a,b
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 22475215 and 22375201), the Natural Science Foundation of Fujian Province (Grant No. 2024J010039 and 2023j01216) and the Self-deployed Key Project of State Key Laboratory of Functional Crystals and Devices (GNJT-2025-ZD01).References
- D. Dou, C. Wei, B. Zhang, D. Yang and Y. Wang, Angew. Chem., Int. Ed., 2025, 64, e202504761 CrossRef CAS PubMed.
- F. Gong, X. Jiang, K. Duanmu, C. Wu, Z. Lin, Z. Huang, M. G. Humphrey and C. Zhang, Angew. Chem., Int. Ed., 2025, e21786 Search PubMed.
- C. Jin, Y. Li, J. S. Kim, J. H. Lim, H. Huang, C. A. Chen, J. Lee, Y. Heo, B. Zhang, J. I. Jang and K. M. Ok, Angew. Chem., Int. Ed., 2025, 64, e202512618 CrossRef CAS PubMed.
- P. F. Li, C. L. Hu, J. G. Mao and F. Kong, Coord. Chem. Rev., 2024, 517, 216000 Search PubMed.
- X. Long, F. Li, H. Qiu, X. Hou, J. Lu, J. Li, Z. Yang, J. Cao, S. Pan and M. Mutailipu, Angew. Chem., Int. Ed., 2025, e22210 Search PubMed.
- H. Qiu, S. Pan and M. Mutailipu, Fundam. Res., 2025, 5, 640–653 CrossRef CAS PubMed.
- H. Tian, C. Lin, Y. Zhou, X. Zhao, H. Fan, T. Yan, N. Ye and M. Luo, Angew. Chem., Int. Ed., 2023, 62, e202304858 CrossRef CAS PubMed.
- Z. Chen, C. Li, X. Wu, J. Lu, Z. Yang, X. Hou and M. Mutailipu, Aggregate, 2025, 6, e70134 CrossRef CAS.
- T. T. He, F. Kong and J. G. Mao, J. Synth. Cryst., 2025, 54(10), 1811–1822 Search PubMed.
- H. Liu, X. Zhai, X. Li, H. Wu, Z. Hu, J. Wang, Y. Wu and H. Yu, Angew. Chem., Int. Ed., 2025, 64, e202502252 CrossRef CAS PubMed.
- J. Lu, X. Liu, M. Zhao, X. B. Deng, K. X. Shi, Q.-R. Wu, L. Chen and L.-M. Wu, J. Am. Chem. Soc., 2021, 143, 3647–3654 CrossRef CAS PubMed.
- X. Wen, Y. Yan, J. Lu, X. Shi, P. Tang, J. Chen, G. Yang, G. Peng, H. Yu, H. Zhang, Z. Hu, J. Wang and N. Ye, Angew. Chem., Int. Ed., 2025, 64, e202424153 CrossRef CAS PubMed.
- M. Yan, C.-L. Hu, R.-L. Tang, W. D. Yao, W. Liu and S.-P. Guo, Chem. Sci., 2024, 15, 8500–8505 RSC.
- X. Dong, L. Huang, H. Zeng, Z. Lin, K. M. Ok and G. Zou, Angew. Chem., Int. Ed., 2025, 64, e202509917 CrossRef CAS PubMed.
- J. H. Wu, B. Zhang, T.-K. Jiang, F. Kong and J. G. Mao, Chin. J. Struct. Chem., 2023, 42, 100016 CAS.
- X. Dong, H. Huang, L. Huang, Y. Zhou, B. Zhang, H. Zeng, Z. Lin and G. Zou, Angew. Chem., Int. Ed., 2024, 63, e202318976 Search PubMed.
- X. Dong, L. Huang, C. Hu, H. Zeng, Z. Lin, X. Wang, K. M. Ok and G. Zou, Angew. Chem., Int. Ed., 2019, 58, 6528–6534 CrossRef CAS PubMed.
- X. Chen, Y. Li, J. Luo and S. Zhao, Chin. J. Struct. Chem., 2023, 42, 100044 CAS.
- P. F. Li, C. L. Hu, Y. F. Li, J. G. Mao and F. Kong, J. Am. Chem. Soc., 2024, 146, 7868–7874 CrossRef CAS PubMed.
- Y. Li, F. Liang, S. Zhao, L. Li, Z. Wu, Q. Ding, S. Liu, Z. Lin, M. Hong and J. Luo, J. Am. Chem. Soc., 2019, 141, 3833–3837 CrossRef CAS PubMed.
- H. Liu, H. Wu, Z. Hu, J. Wang, Y. Wu, P. S. Halasyamani and H. Yu, ACS Mater. Lett., 2022, 5, 155–161 CrossRef.
- X. Zhang, B. Xu, D. Xiao, X. Zhang, P. Gong and Z. Lin, Inorg. Chem. Front., 2025, 12, 7566–7572 RSC.
- Q. Wang, L. Wang, X. Zhao, L. Huang, D. Gao, J. Bi, X. Wang and G. Zou, Inorg. Chem. Front., 2019, 6, 3125–3132 RSC.
- F. He, Y. Ge, X. Zhao, J. He, L. Huang, D. Gao, J. Bi, X. Wang and G. Zou, Dalton Trans., 2020, 49, 5276–5282 RSC.
- S. V. Krivovichev and L. Bindi, Angew. Chem., Int. Ed., 2020, 60, 3854–3855 CrossRef.
- R. N. Li, Z. C. Wu, W. D. Yao, Y. F. Fu, Y. M. Zhang, W. Liu, J. Wu and S. P. Guo, Adv. Funct. Mater., 2025, e25860 CrossRef.
- R. Wei, H. Huang, D. Yang, Y. Wang and B. Zhang, Adv. Opt. Mater., 2024, 12, 2401814 CrossRef CAS.
- J. Wu, Y. Guo, J. L. Qi, W. D. Yao, S. X. Yu, W. Liu and S. P. Guo, Angew. Chem., Int. Ed., 2023, 62, e202301937 Search PubMed.
- J. H. Wu, C. L. Hu, Y. F. Li, J. G. Mao and F. Kong, Chem. Sci., 2024, 15, 8071–8079 Search PubMed.
- Z. P. Zhang, X. Liu, R. X. Wang, S. Zhao, W. J. He, H. Y. Chen, X. B. Deng, L. M. Wu, Z. Zhou and L. Chen, Angew. Chem., Int. Ed., 2024, 63, e202408551 CrossRef CAS PubMed.
- L. Qi, X. Jiang, K. Duanmu, C. Wu, Z. Lin, Z. Huang, M. G. Humphrey and C. Zhang, J. Am. Chem. Soc., 2024, 146, 9975–9983 Search PubMed.
- D. Yang, H. Sha, Z. Wang, R. Su, C. He, B. Su, X. Yang and X. Long, Adv. Opt. Mater., 2024, 12, 2401762 Search PubMed.
- L. Yang, H. Mi, Q. Li, D. Yang, H. Sha, Z. Wang, R. Su, B. Su and C. He, Mater. Today Chem., 2025, 48, 102931 Search PubMed.
- Y. Wu, Y. Li, Y. Zhang, B. Teng, X. Jiang, C. Hu, S. Sun, L. Cao, J. Ma, K. Xu, D. Xu, Z. Lin and D. Zhong, Adv. Funct. Mater., 2025, 35, 2503125 CrossRef CAS.
- R. Zhao, T. Zhu, S. Wang, C. Jarrett-Wilkins, A. M. Najjarian, A. J. Lough, S. Hoogland, E. H. Sargent and D. S. Seferos, Chem. Sci., 2022, 13, 12144–12148 RSC.
- Y. Shen, B. Chen, H. Chen and J. Luo, Inorg. Chem., 2022, 61, 14242–14246 CrossRef CAS PubMed.
- M. Bocus, E. Van Den Broeck, X. Wu, M. Bal, J. Bomon, L. Vanduyfhuys, B. F. Sels, B. U. W. Maes and V. Van Speybroeck, Nat. Catal., 2025, 8, 33–45 CrossRef CAS.
- Y. Lyu, M. Peng, Y. Hu, X. Hu, Y. Zhang, P. Zhao, T. Wang, S. Xu, K. Zhang and L. Bian, Cell. Biomater., 2025, 1, 100124 CrossRef.
- D. Chu, K. Zhang, C. Xie, K. T. Butler, Z. Yang and S. Pan, ACS Mater. Lett., 2024, 6, 1094–1102 CrossRef CAS.
- Y. Kang, C. Yang, J. Zhang and Q. Wu, Chin. Chem. Lett., 2026, 37, 111385 CrossRef CAS.
- H. Yu, N. Z. Koocher, J. M. Rondinelli and P. S. Halasyamani, Angew. Chem., Int. Ed., 2018, 57, 6100–6103 CrossRef CAS PubMed.
- X. Zhang, J. Q. Shen, H. X. Lv, P. H. Guo, Y. G. Chen, C. L. Hu and X. M. Zhang, Inorg. Chem. Front., 2025, 12, 5430–5438 RSC.
- G. Zhang, J. Qin, T. Liu, Y. Li, Y. Wu and C. Chen, Appl. Phys. Lett., 2009, 95, 261104 CrossRef.
- F. He, Q. Wang, C. Hu, W. He, X. Luo, L. Huang, D. Gao, J. Bi, X. Wang and G. Zou, Cryst. Growth Des., 2018, 18, 6239–6247 CrossRef CAS.
- M. Zhang, B. Zhang, D. Yang and Y. Wang, Inorg. Chem., 2021, 60, 18483–18489 CrossRef CAS PubMed.
- Z. Bai, J. Lee, C. L. Hu, G. Zou and K. M. Ok, Chem. Sci., 2024, 15, 6572–6576 Search PubMed.
- Y. Kuk, Z. Bai, Y. Li and K. M. Ok, Chem. Mater., 2025, 37, 1306–1313 CrossRef CAS.
- P. F. Li, C. L. Hu, B. Zhang, J. G. Mao and F. Kong, Chin. Chem. Lett., 2026, 37, 110588 CrossRef CAS.
- X. Y. Li, X. Cheng, C. L. Hu, B. X. Li, Z. Zhou, J. H. Zhang, S. Deng, J. G. Mao and F. Kong, Angew. Chem., Int. Ed., 2025, 64, e202501481 CrossRef CAS PubMed.
- X. Meng, X. Zhang, Q. Liu, Z. Zhou, X. Jiang, Y. Wang, Z. Lin and M. Xia, Angew. Chem., Int. Ed., 2022, 62, e202214848 Search PubMed.
- S. Choi and K. M. Ok, Chem.–Asian J., 2025, 20, e00622 CrossRef CAS PubMed.
- H. Wu, Z. Wei, Z. Hu, J. Wang, Y. Wu and H. Yu, Angew. Chem., Int. Ed., 2024, 63, e202406318 CrossRef CAS PubMed.
- (a) CCDC 2474438: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p1vl6; (b) CCDC 2474439: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2p1vm7.
| This journal is © The Royal Society of Chemistry 2026 |