Open Access Article
Open Access ArticleReductive desymmetric silylation of biaryl bis(triflates) enabled by a chiral nickel/picolinamide complex
Zhe
Chen
,
Junjie
Zhang
and
Chuan
Wang
 *
*
Department of Chemistry, University of Science and Technology of China, 96 Jinzhai Road, Hefei, Anhui 230026, P. R. China. E-mail: chuanw@ustc.edu.cn
First published on 5th February 2026
Abstract
Given the importance of organosilicons and atropisomerism, there is a fundamental need to develop new methods to synthesize axially chiral biaryl silanes. By use of an easily accessible chiral picolinamide as a ligand and inexpensive nickel as a catalyst, we realize a desymmetric silylation of prochiral biaryl bis(triflates) with silyl chlorides under net reductive conditions. This cross-electrophile reaction offers a new approach to preparing highly enantioenriched C1-symmetric axially chiral platform molecules, which incorporate both a silane and a triflate moiety as two distinct docking sites for various downstream derivatizations.
Introduction
Axially chiral biaryls are not only present as a core structure in numerous natural products1 and pharmaceutically relevant molecules2 but are also frequently used as privileged scaffolds for chiral ligands and organocatalysts in asymmetric catalysis.3 To date, catalytic methods have emerged as a powerful tool to synthesize axially chiral biaryls based on different strategies, including aryl–aryl cross-coupling, de novo arene ring formation, (dynamic) kinetic resolution of racemic biaryls, and desymmetrization of prochiral biaryls.4 In the desymmetrization variant, one of two distally positioned enantiotopic moieties is enantioselectively converted, and thus this unique reaction pattern circumvents direct construction of a sterically encumbered chiral axis while the unreacted functionality is available for further derivatizations.5 In the 1990s, Hayashi realized the first examples of desymmetric functionalization of prochiral biaryl bis(triflates) via palladium-catalyzed Kumada6 and Sonogashira coupling.7 Recently, this reaction mode has been further extended by utilizing other coupling partners including borohydride,8 aromatic amines,9 thiols,10 phosphines,11 alkyl bromides,12 aldehydes,12 and B2Pin2 (ref. 13) (Scheme 1A). However, desymmetric silylation of biaryl bis(halides) or bis(sulfonates) via enantioselective C–Si bond formation remains elusive.Organosilicons find broad applications in organic synthesis,14 material science,15 and medicinal chemistry.16 Consequently, organic chemists have endeavoured to develop new and efficient approaches to preparing such compounds.17 However, catalytic atroposelective synthesis of (hetero)biaryl silanes starting from achiral or racemic precursors is underdeveloped, and a handful of successful examples rely on either C–H silylation18 or cross-coupling between silicon nucleophiles and carbon electrophiles19 (Scheme 1B). On the other side, rapid progress has been witnessed in the transition metal-catalyzed cross-electrophile coupling (XEC) reactions between silyl halides and diverse carbon electrophiles, which provides new and efficient access to organosilanes.20 Driven by these elegant results and our continued interest in the development of asymmetric XEC reactions for the synthesis of atropisomers,21 we wondered whether reductive silylation of aryl (pseudo)halides with halo silanes could be applied as the enantiodetermining step of atroposelective desymmetrization. In this context, we realized a chiral nickel/picolinamide complex-catalyzed reductive desymmetric silylation of prochiral biaryl bis(triflates) with silyl chlorides towards the highly enantioselective synthesis of C1-symmetric axially chiral biaryls, which contain both a nucleophilic silane and an electrophilic triflate moiety as two distinct docking sites to introduce new functionalities (Scheme 1C).
Results and discussion
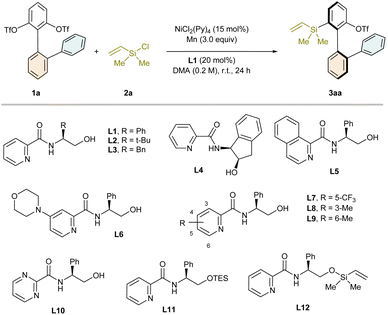
For optimization of the reaction conditions, the biaryl bis(triflate) 1a and chlorodimethyl(vinyl)silane (2a) were selected as the model substrates. While some privileged chiral ligands for asymmetric nickel catalysis, including BOX, BiOX, PyrOx, PyBOX, and PHOX, either could not promote the target reaction or only delivered a trace amount of the product as a racemic mixture, a ligand class of chiral picolinamides was surprisingly found to be effective for the intended desymmetric reductive silylation. To the best of our knowledge, enantioselective Ni-catalyzed cross-electrophile coupling reactions using chiral picolinamides as the ligands is unprecedented. Systematic screening of the reaction conditions revealed the optimized one as follows: NiCl2(Py)4 (15 mol%) as the precatalyst, the picolinamide L1 derived from L-phenylglycinol (20 mol%) as the ligand, and Mn (3 equiv.) as the reducing agent in DMA (0.2 M) as the solvent at room temperature for 24 h. In this case, the desired product 3aa was obtained in 80% yield and 91% ee (Table 1, entry 1). Notably, the formation of the bis(silylation) byproduct was not observed, revealing that this desymmetrization reaction is not followed by kinetic resolution. Next, we varied the reaction parameters from the optimized conditions to demonstrate their impact on the reaction outcome (Table 1, entries 2–17). Replacing Ph with t-Bu (L2) or Bn (L3) as the ligand arm resulted in decreased enantiocontrol (entries 2 and 3). The ligand L4 bearing a chiral indane scaffold was also competent but provided lower enantioselectivity (entry 4). In the case of the chiral 1-isoquinolinecarboxamide L5 as the ligand, both the reaction yield and the asymmetric induction were significantly reduced (entry 5). The introduction of an N-morpholinyl group to position 4 of the pyridine of the ligand did not improve the result (L6, entry 6), whereas the installation of CF3 on position 5 attenuated both the selectivity and the efficiency to some degree (L7, entry 7). Notably, substitution on positions 3 and 6 deteriorated the ligand performance significantly (L8 and L9, entries 8 and 9). Moreover, the reaction was almost shut down when using the ligand L10 based on pyrimidine-2-carboxylic acid (entry 10). When the hydroxyl group of the ligand was protected by TES, both the yield and the enantiomeric excess of the product diminished dramatically (L11, entry 11). However, a nearly identical result was achieved in the case of the ligand L12 with a pendant dimethylvinyl siloxy group, indicating that in situ silylation of the hydroxyl group of the ligand might occur (entry 12). When the nickel precatalysts lacking pyridine as a coligand were employed, the desired product was formed much less enantioselectively, suggesting that pyridine might remain coordinated to the catalyst species in the enantiodetermining step (entries 13 and 14). The use of 60 mol% pyridine as an additive did not lead to significantly improved enantiocontrol (entry 15). The desired reaction proceeded in other polar aprotic solvents tested but afforded only inferior results (entries 16 and 17). Only a trace amount of compound 3aa was generated when Zn was used as the reductant instead of Mn (entry 18).| Entry | Deviation from the optimized conditions | Yieldb (%) | eec (%) |
|---|---|---|---|
| a Unless otherwise specified, the reactions were performed on a 0.2 mmol scale of the biaryl bis(triflate) 1a using 3.0 equiv. of chlorodimethyl(vinyl)silane (2a), 15 mol% NiCl2(Py)4, 20 mol% ligand L1, and 3.0 equiv. of Mn in 1 mL DMA under N2 atmosphere for 24 h at room temperature. b Yield of the isolated product. c Determined by HPLC analysis on a chiral stationary phase. d The reaction was performed with 60 mol% pyridine as an additive. | |||
| 1 | None | 80 | 91 |
| 2 | L2 instead of L1 | 22 | 67 |
| 3 | L3 instead of L1 | 68 | 83 |
| 4 | L4 instead of L1 | 61 | 82 |
| 5 | L5 instead of L1 | 10 | 11 |
| 6 | L6 instead of L1 | 60 | 91 |
| 7 | L7 instead of L1 | 24 | 79 |
| 8 | L8 instead of L1 | 16 | 8 |
| 9 | L9 instead of L1 | 16 | 3 |
| 10 | L10 instead of L1 | Trace | n.d. |
| 11 | L11 instead of L1 | 19 | 21 |
| 12 | L12 instead of L1 | 78 | 91 |
| 13 | NiCl2 instead of NiCl2(Py)4 | 51 | 62 |
| 14 | Ni(COD)2 instead of NiCl2(Py)4 | 41 | 70 |
| 15d | Ni(COD)2 instead of NiCl2(Py)4 | 63 | 72 |
| 16 | DMF instead of DMA | 53 | 84 |
| 17 | NMP instead of DMA | 61 | 83 |
| 18 | Zn instead of Mn | Trace | n.d. |
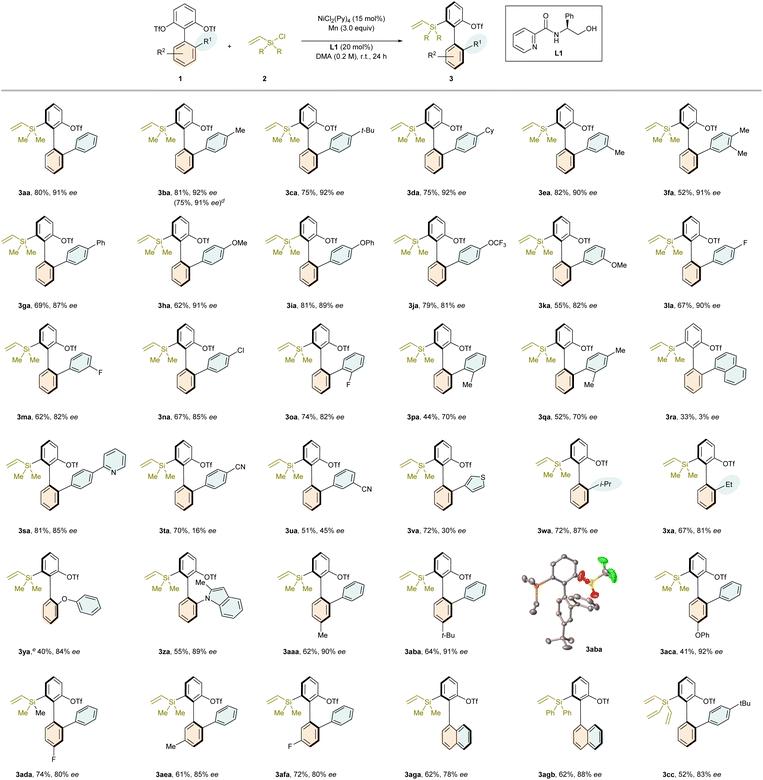
With the optimized reaction conditions in hand, we commenced to evaluate the substrate scope of this nickel-catalyzed desymmetrization reaction (Table 2). First, we focused on investigating the generality of the biaryl bis(triflates) 1. It turned out that suitable R1 substituents of compounds 1 include phenyl bearing alkyl (1b–1f), phenyl (1g), OMe (1h and 1k), OPh (1i), OCF3 (1j), and halide (1l–1n) on para or/and meta-position, and the corresponding products 3ba–na were obtained in 52–82% yields and 81–98% ee. The reaction also tolerated o-fluorine phenyl (1o), affording the product 3oa in 74% yield and 82% ee. In contrast, bulkier methyl on the ortho-position of the phenyl substituent (1j and 1k) had a significant detrimental effect on the asymmetric induction of the studied reaction, yielding the products 3pa and 3qa in much lower enantiomeric excesses. The sterically more encumbering α-naphthyl (1r) led to the formation of a nearly racemic product 3ra. Coordinative pyridine on the para-position of phenyl (1s) posed no problem, furnishing the product 3sa in 81% yield and 85% ee. Strong electron-withdrawing cyano group (1t and 1u) undermined the enantiocontrol of the studied reaction dramatically (3ta and 3ua). Replacing phenyl by 3-thienyl as the R1 substituent (1v) also resulted in a low enantiomeric excess (3va). In the case of isopropyl (1w) or ethyl (1x) as R1, the desired reactions proceeded smoothly, providing the products 3wa and 3xa in 87% ee and 81% ee, respectively. Notably, phenoxy (1y) and N-indolyl (1z) were also found to be pertinent R1 substituents, affording the products 3ya and 3za in 84% ee and 89% ee, respectively. Subsequently, permutation of the R2 substituent was performed. It turned out that alkyl (1aa, 1ab, and 1ae), PhO (1ac), and F (1ad and 1af) on either para or meta position were all compatible, delivering the corresponding products in 3aaa–3afa 41–74% yields and 80–92% ee. Moreover, moderate results in terms of both yield and enantiomeric excess were obtained in the case of α-naphthyl (3aga). Next, we turned our attention to assessing the scope of silyl chlorides. Chlorodiphenyl(vinyl)silane (2b) and chlorotrivinylsilane (2c) were both suitable precursors, providing the products 3agb and 3cc in 88% ee and 83% ee, respectively. The vinyl substituent of silyl chlorides was confirmed to be vital for this transformation since its absence shut down this target desymmetrization. The coordination-assisted oxidative addition of vinyl substituted silyl chlorides to low-valent nickel is likely. However, it could also be attributed to higher ability of vinyl substituted silyl chlorides regarding oxidative addition to low-valent nickel. Moreover, the reaction for the synthesis of compound 3ba on a 1 mmol scale afforded a similar result (75% yield, 91% ee). In addition, the absolute configuration of compound 3aba was unambiguously determined to be R through X-ray crystallography (CCDC number: 2452324).
| a Unless otherwise specified, the reactions were performed on a 0.2 mmol scale of the biaryl bis(triflate) 1 using 3.0 equiv. of the silyl chlorides 2, 15 mol% NiCl2(Py)4, 20 mol% ligand L1, and 3.0 equiv. of Mn in 1 mL DMA under N2 atmosphere for 24 h at room temperature. b Yields of the isolated products. c Determined by HPLC analysis on a chiral stationary phase. d The reaction was performed on a 1 mmol scale of 1b. e The reaction was performed using L5 as the ligand instead of L1. |
|---|
|
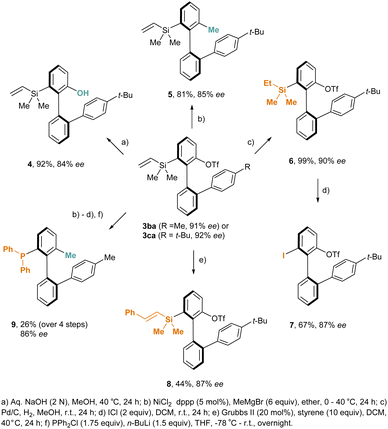
A variety of downstream derivatizations focusing on the transformations of the pendant triflate and/or silane moiety of the coupling products were carried out (Scheme 2). First, simple sodium hydroxide-mediated hydrolysis of the compound 3ca provided a chiral phenol 4 in 92% yield and 84% ee. Furthermore, the nickel-catalyzed Kumada methylation of the aryl triflate 3ca delivered a chiral silane 5 in 81% yield and 85% ee. Moreover, the vinyl group of 3ca was successfully hydrogenated using Pd/C in 99% yield and 90% ee. Upon treatment with ICl, the resultant saturated silane 6 underwent iodination, affording a chiral aryl iodide 7 in 67% yield and 87% ee. Besides, the compound 3ca was subjected to metathesis with styrene, furnishing a chiral silane 8 bearing an internal olefinic unit 8 in 44% yield and 86% ee. In addition, the sequential reactions consisting of Kumada methylation, hydrogenation, iodination, and n-BuLi-mediated phosphination with PPh2Cl converted the compound 3ba into a chiral phosphine 9 in 26% overall yield and 86% ee.
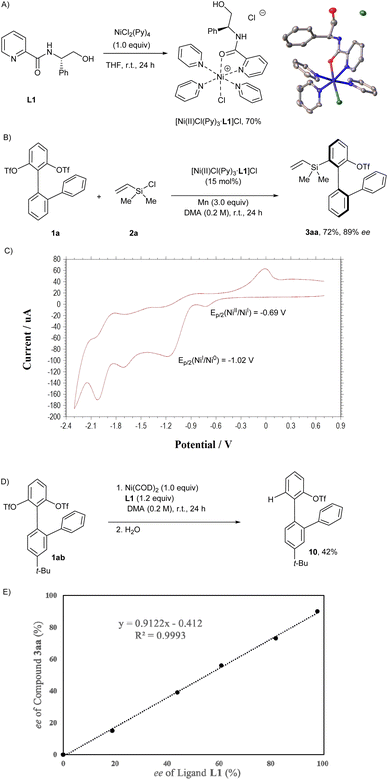
Next, a series of preliminary mechanistic studies were conducted, and the results were demonstrated in Scheme 3. First, it turned out that NiCl2(Py)4 could react with the picolinamide ligand L1 to afford a cationic Ni(II) complex [Ni(II)Cl(Py)3·L1]Cl, the structure of which was determined by X-ray crystallography (CCDC number: 2452325) (Scheme 3A). Furthermore, the direct use of [Ni(II)Cl(Py)3·L1]Cl as the precatalyst for the desymmetrization of the biaryl bis(triflate) 1a with chlorodimethyl(vinyl)silane (2a) provided a similar outcome to the one under the standard conditions (Scheme 3B). The cyclic voltammetry (CV) measurement of [Ni(II)Cl(Py)3·L1]Cl gave irreversible reduction waves at −0.89 V and at −1.02 V (vs. Ag/AgCl in DMA), which could be assigned to the reduction of Ni(II) to Ni(I) and Ni(I) to Ni(0), respectively (Scheme 3C). According to the seminal work of Stahl et al.,22 the thermodynamic potential of Mn2+/Mn is EO′ = −1.08 V (vs. Ag/AgCl in DMA), suggesting that Mn is capable of reducing the Ni(II) precatalyst into a Ni(0) species in situ. Moreover, the stoichiometric reaction between the biaryl bis(triflate) 1ab and Ni(COD)2 in the presence of the ligand L1 afforded the detriflated product 10 after quenching with water in 42% yield, indicating that the oxidative addition of biaryl bis(triflates) to Ni(0) is viable (Scheme 3D). The picolinamide ligand-enabled generation of Ni(0) via reduction might be the key for the success of this asymmetric silylation, since Mn is known to be unable to reduce Ni(II)PyrOx,23 Ni(II)BiOX,24 or Ni(II)BOX25 into their corresponding Ni(0) species. In addition, the dependence of the enantiomeric excess of the product 3aa on the enantiopurity of the ligand L1 was investigated, and a linear relationship was obtained, revealing that no high-order catalytically active on-cycle species containing multiple units of the chiral entity is present in this nickel-catalyzed reaction (Scheme 3E).
Based on the preliminary mechanistic studies and the previous report,20a a plausible catalytic cycle exemplified by the precursors 1a and 2a was proposed in Scheme 4. Initially, Ni(0) is generated under reductive conditions and undergoes two-electron oxidative addition with the biaryl bis(triflate) 1a in an enantioselective manner. The resultant axially chiral Ni(II) complex A is subsequently reduced by Mn into a Ni(I) species B, to which chlorodimethyl(vinyl)silane (2a) performs the second oxidative addition. Next, stereoretensive reductive elimination from the generated Ni(III) complex C provides the product 3aa and Ni(I)Cl, and the latter is reduced by Mn to Ni(0) for the next catalytic cycle.
Conclusions
In conclusion, we have developed a reductive desymmetric silylation of prochiral biaryl bis(triflates) with silyl chlorides. This desymmetrization reaction is promoted by a simple chiral nickel/picolinamide complex, allowing for the synthesis of C1-symmetric axially chiral biaryl silanes in a highly enantioselective fashion. The downstream derivatizations of the coupling products, focusing on the conversions of the pendant silane and triflate moieties, provide a variety of functionalized axially chiral biaryls. According to the preliminary mechanistic investigations, the picolinamide ligand enables the generation of a Ni(0) species via reduction, to which biaryl bis(triflates) perform enantiodiscriminating oxidative addition.Author contributions
C. W. and Z. C. conceived and designed the experiments. Z. C. and J. Z. performed experiments and prepared the SI. C. W. directed the project and wrote the paper. All authors discussed the results and commented on the manuscript.Conflicts of interest
There is no conflict to declare.Data availability
CCDC 2452324 (3aba) and 2452325 ([Ni(II)Cl(Py)3·L1]Cl) contain the supplementary crystallographic data for this paper.26a,bThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental procedure, spectra data, NMR-data, and HPLC-data. See DOI: https://doi.org/10.1039/d5sc09250g.
Acknowledgements
This work is supported National Natural Science Foundation of China (22471256, 22271267).Notes and references
- (a) M. H. McCormick, W. M. Stark, G. E. Pittenger, R. C. Pittenger and J. M. McGuire, Antibiot. Annu., 1955, 3, 606–611 Search PubMed; (b) S. M. Kupchacn, R. W. Britton, M. F. Ziegler, C. J. Gilmore, R. J. Restivo and R. F. Bryan, J. Am. Chem. Soc., 1973, 95, 1335–1336 CrossRef PubMed; (c) Y. F. Hallock, K. P. Manfredi, J. W. Blunt, J. H. Cardellina II, M. Schaeffer, K.-P. Gulden, G. Bringmann, A. Y. Lee, J. Clardy, G. François and M. R. Boy, J. Org. Chem., 1994, 59, 6349–6355 Search PubMed; (d) A. Zask, J. Murphy and G. A. Ellestad, Chirality, 2013, 25, 265–274 Search PubMed; (e) J. E. Smyth, N. M. Butler and P. A. Keller, Nat. Prod. Rep., 2015, 32, 1562–1583 Search PubMed; (f) C. A. Bewley, G. A. Sulikowski, Z. J. Yang, G. Bifulco, H.-M. Cho and C. R. Fullenkamp, Acc. Chem. Res., 2023, 56, 414–424 Search PubMed.
- (a) S. R. LaPlante, L. D. Fader, K. R. Fandrick, D. R. Fandrick, O. Hucke, R. Kemper, S. P. F. Miller and P. J. Edwards, J. Med. Chem., 2011, 54, 7005–7022 CrossRef CAS PubMed; (b) S. R. LaPlante, P. J. Edwards, L. D. Fader, A. Jakalian and O. Hucke, ChemMedChem, 2011, 6, 505–513 CrossRef CAS PubMed; (c) M. Basilaia, M. H. Chen, J. Secka and J. L. Gustafson, Acc. Chem. Res., 2022, 55, 2904–2919 Search PubMed; (d) P. W. Glunz, Bioorg. Med. Chem. Lett., 2018, 28, 53–60 Search PubMed; (e) S. Perreault, J. Chandrasekhar and L. Patel, Acc. Chem. Res., 2022, 55, 2581–2593 CrossRef CAS PubMed; (f) Z. Wang, L. Meng, X. Liu, L. Zhang, Z. Yu and G. Wu, Eur. J. Med. Chem., 2022, 243, 114700 CrossRef CAS PubMed.
- (a) R. Noyori and H. Takaya, Acc. Chem. Res., 1990, 23, 345–350 CrossRef CAS; (b) Y. Chen, S. Yekta and A. K. Yudin, Chem. Rev., 2003, 103, 3155–3212 Search PubMed; (c) W. Tang and X. Zhang, Chem. Rev., 2003, 103, 3029–3070 CrossRef CAS PubMed; (d) Y.-M. Li, F.-Y. Kwong, W.-Y. Yu and A. S. C. Chan, Coord. Chem. Rev., 2007, 251, 2119–2144 CrossRef CAS; (e) T. Akiyama, Chem. Rev., 2007, 107, 5744–5758 CrossRef CAS PubMed; (f) S. Shirakawa and K. Maruoka, Angew. Chem., Int. Ed., 2013, 52, 4312–4348 CrossRef CAS PubMed; (g) D. Parmar, E. Sugiono, S. Raja and M. Rueping, Chem. Rev., 2014, 114, 9047–9153 CrossRef CAS PubMed.
- For reviews on atroposelective synthesis of atropisomers, see: (a) G. Bringmann and D. Menche, Acc. Chem. Res., 2001, 34, 615–624 Search PubMed; (b) G. Bringmann, A. J. Price Mortimer, P. A. Keller, M. J. Gresser, J. Garner and M. Breuning, Angew. Chem., Int. Ed., 2005, 44, 5384–5427 Search PubMed; (c) M. C. Kozlowski, B. J. Morgan and E. C. Linton, Chem. Soc. Rev., 2009, 38, 3193–3207 Search PubMed; (d) G. Bringmann, T. Gulder, T. A. M. Gulder and M. Breuning, Chem. Rev., 2011, 111, 563–639 CrossRef CAS PubMed; (e) P. G. Cozzi, E. Emer and A. Gualandi, Angew. Chem., Int. Ed., 2011, 50, 3847–3849 CrossRef CAS PubMed; (f) G. Ma and M. P. Sibi, Chem.–Eur. J., 2015, 21, 11644–11657 CrossRef CAS PubMed; (g) J. Wencel-Delord, A. Panossian, F. R. Leroux and F. Colobert, Chem. Soc. Rev., 2015, 44, 3418–3430 Search PubMed; (h) Y.-N. Ma, S.-X. Li and S.-D. Yang, Acc. Chem. Res., 2017, 50, 1480–1492 Search PubMed; (i) D. Bonne and J. Rodriguez, Chem. Commun., 2017, 53, 12385–12393 RSC; (j) Y.-B. Wang and B. Tan, Acc. Chem. Res., 2018, 51, 534–547 CrossRef CAS PubMed; (k) A. Link and C. Sparr, Chem. Soc. Rev., 2018, 47, 3804–3815 Search PubMed; (l) B. Zilate, A. Castrogiovanni and C. Sparr, ACS Catal., 2018, 8, 2981–2988 CrossRef CAS; (m) A. J. Metrano and S. J. Miller, Acc. Chem. Res., 2019, 52, 199–215 Search PubMed; (n) Y.-C. Zhang, F. Jiang and F. Shi, Acc. Chem. Res., 2020, 53, 425–446 CrossRef CAS PubMed; (o) X. Bao, J. Rodriguez and D. Bonne, Angew. Chem., Int. Ed., 2020, 59, 12623–12634 CrossRef CAS PubMed; (p) J. K. Cheng, S.-H. Xiang, S. Li, L. Ye and B. Tan, Chem. Rev., 2021, 121, 4805–4902 CrossRef CAS PubMed; (q) T. A. Schmidt and C. Sparr, Acc. Chem. Res., 2021, 54, 2764–2774 CrossRef CAS PubMed; (r) R. Song, Y. Xie, Z. Jin and Y. R. Chi, Angew. Chem., Int. Ed., 2021, 60, 26026–26037 CrossRef CAS PubMed; (s) J. Wang, C. Zhao and J. Wang, ACS Catal., 2021, 11, 12520–12531 CrossRef CAS; (t) C.-X. Liu, W.-W. Zhang, S.-Y. Yin, Q. Gu and S.-L. You, J. Am. Chem. Soc., 2021, 143, 14025–14040 CrossRef CAS PubMed; (u) X. Zhang, K. Zhao and Z. Gu, Acc. Chem. Res., 2022, 55, 1620–1633 CrossRef CAS PubMed; (v) X.-F. Bai, Y.-M. Cui, J. Cao and L.-W. Xu, Acc. Chem. Res., 2022, 55, 2545–2561 CrossRef CAS PubMed; (w) W. Qin, Y. Liu and H. Yan, Acc. Chem. Res., 2022, 55, 2780–2795 CrossRef CAS PubMed; (x) S. Choppin and J. Wencel-Delord, Acc. Chem. Res., 2023, 56, 189–202 CrossRef CAS PubMed.
- (a) J. Moon, S. Kim, S. Lee, H.-A. Cho, A. Kim, M. K. Ham, S. H. Kim, J. Jung and Y. Kwon, ChemCatChem, 2024, 16, e202400690 CrossRef CAS; (b) X.-P. Zeng, Z.-Y. Cao, Y.-H. Wang, F. Zhou and J. Zhou, Chem. Rev., 2016, 116, 7330–7396 CrossRef CAS PubMed.
- T. Hayashi, S. Niizuma, T. Kamikawa, N. Suzuki and Y. Uozumi, J. Am. Chem. Soc., 1995, 117, 9101–9102 Search PubMed.
- T. Kamikawa, Y. Uozumi and T. Hayashi, Tetrahedron Lett., 1996, 37, 3161–3164 CrossRef CAS.
- X. Li, G.-W. Wang, L.-X. Liu, C.-B. Yu and Y.-G. Zhou, Angew. Chem., Int. Ed., 2023, 62, e202301337 CrossRef CAS PubMed.
- X. Wang, W.-G. Liu, L.-T. Liu, X.-D. Yang, S. Niu, C.-H. Tung, L.-Z. Wu and H. Cong, Org. Lett., 2021, 23, 5485–5490 CrossRef CAS PubMed.
- R. J. Armstrong and M. D. Smith, Angew. Chem., Int. Ed., 2014, 53, 12822–12826 Search PubMed.
- R. Cui, Y. Zhang, Z. Huang, L. Yuwen, Y. Xu and Q.-W. Zhang, Angew. Chem., Int. Ed., 2024, 63, e202412064 CrossRef CAS PubMed.
- Y.-D. Zhu, Z.-Y. Dai, M. Jiang and P.-S. Wang, ACS Catal., 2024, 14, 13860–13866 CrossRef CAS.
- Y. Xiao, A. L. Bartelt, E. Irran and M. Oestreich, Angew. Chem., Int. Ed., 2025, 64, e202515234 CrossRef CAS PubMed.
- (a) Y. Nakao and T. Hiyama, Chem. Soc. Rev., 2011, 40, 4893–4901 RSC; (b) P. L. Fuchs, in Handbook of Reagents for Organic Synthesis. Reagents for Silicon-Mediated Organic Synthesis, Wiley, New York, 2011 Search PubMed; (c) T. Komiyama, Y. Minami and T. Hiyama, ACS Catal., 2017, 7, 631–651 CrossRef CAS.
- (a) N. Auner and J. Weis, in Organosilicon Chemistry V: From Molecules to Materials, Wiley-VCH, Weinheim, 2003 CrossRef; (b) M. A. Brook, in Silicon in Organic, Organometallic, and Polymer Chemistry, Wiley, New York, 2000 Search PubMed; (c) H. Wang, X. Zhang, Y. Li and L.-W. Xu, Angew. Chem., Int. Ed., 2022, 61, e202210851 CrossRef CAS PubMed.
- (a) A. K. Franz and S. O. Wilson, J. Med. Chem., 2013, 56, 388–405 CrossRef CAS PubMed; (b) R. Ramesh and D. S. Reddy, J. Med. Chem., 2018, 61, 3779–3798 CrossRef CAS PubMed.
- (a) M. Oestreich, E. Hartmann and M. Mewald, Chem. Rev., 2013, 113, 402–441 CrossRef CAS PubMed; (b) G. K. Min, D. Hernández and T. Skrydstrup, Acc. Chem. Res., 2013, 46, 457–470 CrossRef CAS PubMed; (c) Z. Xu, W.-S. Huang, J. Zhang and L.-W. Xu, Synthesis, 2015, 47, 3645–3668 CrossRef CAS; (d) Y. Nakajima and S. Shimada, RSC Adv., 2015, 5, 20603–20616 RSC; (e) L. Li, Y. Zhang, L. Gao and Z. Song, Tetrahedron Lett., 2015, 56, 1466–1473 Search PubMed; (f) T. Hiyama, Y. Minami and A. Mori, in Organosilicon Chemistry: Novel Approaches and Reactions, ed. T. Hiyama and M. Oestreich, Wiley-VCH, Weinheim, 2019, pp. 271–332 CrossRef; (g) S. Bähr, W. Xue and M. Oestreich, ACS Catal., 2019, 9, 16–24 CrossRef; (h) X. Zhang, J. Fang, C. Cai and G. Lu, Chin. Chem. Lett., 2021, 32, 1280–1292 CrossRef CAS; (i) J.-J. Feng, W. Mao, L. Zhang and M. Oestreich, Chem. Soc. Rev., 2021, 50, 2010–2073 RSC; (j) Y. Wu, L. Zheng, Y. Wang and P. Wang, Chem, 2023, 9, 3461–3514 CrossRef CAS; (k) M. Liu, L. Qi and D. Zhao, Chin. J. Org. Chem., 2023, 43, 3508–3525 CrossRef CAS; (l) Y. Ge, J. Ke and C. He, Acc. Chem. Res., 2025, 58, 375–398 CrossRef CAS PubMed.
- (a) Y. Guo, M.-M. Liu, X. Zhu, L. Zhu and C. He, Angew. Chem., Int. Ed., 2021, 60, 13887–13891 CrossRef CAS; (b) B. Yang, J. Gao, X. Tan, Y. Ge and C. He, Angew. Chem., Int. Ed., 2023, 62, e202307812 CrossRef CAS PubMed.
- (a) X. Bi, J. Feng, X. Xue and Z. Gu, Org. Lett., 2021, 23, 3201–3206 CrossRef CAS PubMed; (b) Y. Xiao, A. L. Bartelt, E. Irran and M. Oestreich, ACS Catal., 2025, 15, 11307–11312 Search PubMed.
- (a) J. Duan, K. Wang, G.-L. Xu, S. Kang, L. Qi, X.-Y. Liu and X.-Z. Shu, Angew. Chem., Int. Ed., 2020, 59, 23083–23088 CrossRef CAS PubMed; (b) L. Zhang and M. Oestreich, Angew. Chem., Int. Ed., 2021, 60, 18587–18590 CrossRef CAS; (c) J. Duan, Y. Wang, L. Qi, P. Guo, X. Pang and X.-Z. Shu, Org. Lett., 2021, 23, 7855–7859 CrossRef CAS PubMed; (d) Z.-Z. Zhao, X. Pang, X.-X. Wei, X.-Y. Liu and X.-Z. Shu, Angew. Chem., Int. Ed., 2022, 61, e202200215 CrossRef CAS PubMed; (e) C.-H. Xu, Z.-Q. Xiong, J.-H. Qin, X.-H. Xu and J.-H. Li, Org. Lett., 2023, 25, 7263–7267 CrossRef CAS PubMed; (f) Q.-Q. Pan, L. Qi, X. Pang and X.-Z. Shu, Angew. Chem., Int. Ed., 2023, 62, e202215703 CrossRef CAS PubMed; (g) C. Li, S. Yang and X. Zeng, ACS Catal., 2023, 13, 12062–12073 CrossRef CAS; (h) X.-X. Wei, Z.-Z. Zhao, X. Pang and X.-Z. Shu, Org. Lett., 2024, 26, 6125–6129 CrossRef CAS PubMed; (i) L.-Y. Yang, Y. Qin, Z. Zhao and D. Zhao, Angew. Chem., Int. Ed., 2024, 63, e202407773 CrossRef CAS; (j) D. Xing, J. Liu, D. Cai, B. Huang, H. Jiang and L. Huang, Nat. Commun., 2024, 15, 4502 CrossRef CAS PubMed; (k) T. Yin, S. Sui, S. Li, J. Chang and D. Bai, Chem. Commun., 2024, 60, 14204–14207 RSC; (l) L. Qiu, Y. Liu, H. Chen, L. Song and W. Xie, Chem. Sci., 2025, 16, 9454–9461 RSC.
- (a) Z. Lin, W. Hu, L. Zhang and C. Wang, ACS Catal., 2023, 13, 6795–6803 CrossRef CAS; (b) H. Dong and C. Wang, J. Am. Chem. Soc., 2023, 145, 26747–26755 CrossRef CAS PubMed; (c) Y. Mao, W. Hu and C. Wang, ACS Catal., 2024, 14, 17039–17045 CrossRef CAS; (d) X. Ding and C. Wang, Org. Lett., 2025, 27, 2897–2901 CrossRef CAS PubMed.
- Z.-M. Su, R. Deng and S. S. Stahl, Nat. Chem., 2024, 16, 2036–2043 CrossRef CAS PubMed.
- C. L. Wagner, G. Herrera, Q. Lin, C. T. Hu and T. Diao, J. Am. Chem. Soc., 2021, 143, 5295–5300 CrossRef CAS PubMed.
- L. Ju, Q. Lin, L. N. J. Bretto, C. L. Wagner, C. T. Hu, J. T. Miller and T. Diao, J. Am. Chem. Soc., 2021, 143, 14458–14463 CrossRef CAS PubMed.
- R. F. Turro, J. L. H. Wahlman, Z. J. Tong, X. Chen, M. Yang, E. P. Chen, X. Hong, R. G. Hadt, K. N. Houk, Y.-F. Yang and S. Reisman, J. Am. Chem. Soc., 2023, 145, 14705–14715 CrossRef CAS PubMed.
- (a) CCDC 2452324: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n9v72; (b) CCDC 2452325: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n9v83.
| This journal is © The Royal Society of Chemistry 2026 |