 Open Access Article
Open Access ArticleArtificial recombinant high-density protein nanocarriers for precision drug delivery
Wenpan Li a,
Nan Jia
a,
Nan Jia a,
Mengwen Li
a,
Mengwen Li a,
Yanhao Jiang
a,
Yanhao Jiang a and
Jianqin Lu
a and
Jianqin Lu *abcd
*abcd
aSkaggs Pharmaceutical Sciences Center, Department of Pharmacology & Toxicology, R. Ken Coit College of Pharmacy, The University of Arizona, Tucson, Arizona 85721, USA. E-mail: lu6@arizona.edu; Fax: +520-626-2466; Tel: +520-626-1786
bClinical and Translational Oncology Program, The University of Arizona Cancer Center, Tucson, AZ 85721, USA
cBIO5 Institute, The University of Arizona, Tucson, Arizona 85721, USA
dSouthwest Environmental Health Sciences Center, The University of Arizona, Tucson, 85721, USA
First published on 11th May 2026
Abstract
High-density lipoprotein (HDL) is a naturally occurring nanoparticle characterized by excellent biocompatibility and intrinsic receptor-mediated targeting capabilities. Consequently, recombinant high-density lipoprotein (rHDL) has emerged as a highly promising platform for precision drug delivery. This review focuses on the latest advancements in rHDL design, highlighting how key parameters-such as the stoichiometry of lipids to proteins, the engineering of apolipoproteins or mimetic peptides, and interfacial functionalization-govern the nanoparticle's structure, payload integration, and biological performance. We specifically demonstrate how these design principles enable the precise and tunable control of particle size, stability, and targeting efficiency across diverse therapeutic applications, including oncology, atherosclerosis, and neurodegenerative diseases. Compared to conventional nanocarriers, rHDL exhibits distinct advantages by virtue of its biomimetic properties and receptor-specific delivery pathways. In summary, these collective advancements establish rHDL as a “chemically programmable” nanoplatform, underscoring its immense potential to drive the development of targeted and multifunctional therapeutics.
1 Introduction
High-density lipoprotein (HDL) is a natural nanoparticle composed of various biomacromolecules. Multiple physicochemical features such as particle size, morphology, and surface chemistry play critical roles in determining its biological functions.1,2 Compared with other lipoproteins (e.g., low density lipoprotein and very low-density lipoprotein), HDL has a higher density and a smaller diameter of approximately 8–12 nm.3,4 Known as “good cholesterol,” HDL primarily removes excess cholesterol from peripheral tissues and transports it to the liver for metabolism or excretion through the reverse cholesterol transport (RCT) pathway.5,6 In addition, HDL particles possess anti-inflammatory, antioxidant, anti-apoptotic, and anti-infective properties, making HDL an essential protective factor in cardiovascular health and a key component in reducing the risk of coronary artery disease.7–9Recently, the exploitation of HDL particles as therapeutic delivery vehicles has garnered substantial interest. HDL offers several inherent advantages: it is fully biodegradable, exhibits excellent biocompatibility, and, as an endogenous nanostructure, is less susceptible to rapid clearance by the mononuclear phagocyte system (MPS) and does not provoke undesirable immune activation. Emerging evidence indicates that native HDL transports lipids, proteins, and microRNAs between donor and recipient cells,10 underscoring its intrinsic targeting capabilities and making it an attractive scaffold for drug delivery. Despite the attractive biological properties of native HDL, its practical application as a drug delivery vehicle is limited by scarce availability from plasma sources, complex and costly purification procedures, batch-to-batch heterogeneity, and difficulties in customizing cargo loading for diverse therapeutics. Consequently, increasing attention has shifted toward the fabrication of reconstituted HDL (rHDL) nanoparticles engineered from phospholipids and apolipoproteins or apolipoprotein mimetic peptides as versatile and scalable platforms for therapeutic cargo delivery.
The composition of rHDL including lipid species, apolipoprotein selection, and lipid to protein ratios can be precisely tuned, enabling fine control over key physicochemical properties such as particle homogeneity, zeta potential, and the loading capacities of both the core and surface domains. Hydrophobic therapeutics can be sequestered within the hydrophobic core of rHDL; amphiphilic agents can be stably integrated into the lipid bilayer; and hydrophilic cargos may be incorporated by conjugation with hydrophobic moieties for surface anchoring or by facilitated encapsulation within the core.11,12 Collectively, these modular design features endow rHDL with substantial versatility and position it as a highly promising platform for drug delivery applications. Table 1 summarizes the major differences between native HDL and rHDL systems, highlighting why rHDL has become the predominant and more translatable platform for precision drug delivery across a wide range of biomedical applications.
| Aspect | Natural HDL | Artificial recombinant HDL (rHDL) |
|---|---|---|
| Source & scalability | Endogenous (plasma-derived); limited supply | Synthetic (recombinant components); highly scalable; facilitates large-scale manufacturing and clinical translation |
| Composition | Heterogeneous (multiple apolipoproteins and proteins) | Homogeneous; precisely tunable lipid: protein ratio; enhances reproducibility and control over properties |
| Size & morphology | 8–12 nm; natural discoidal/spherical subclasses | 8–50 nm; controllable (discoidal or spherical); optimization for circulation time or tissue penetration |
| Targeting specificity | Natural selectivity via SR-B1, ABCG1etc. | Retains SR-B1 targeting; easily' functionalized with additional ligands (e.g., folate, EGFR); improves tumor/lesion specificity and reduced off-target effects |
| Cargo loading versatility | Primarily lipophilic drugs; limited customization | High efficiency for hydrophobic, amphiphilic, and hydrophilic cargos; greater adaptability for chemotherapeutics, nucleic acids, imaging agentsetc. |
Importantly, the rationale for a timely new review is not merely the expansion of disease applications, but the emergence of new chemical design logic in the rHDL field over the past three to five years. Earlier generations of HDL-mimetic systems largely emphasized biomimicry of native HDL structure and receptor engagement. By contrast, recent studies increasingly treat rHDL as a programmable chemical nanoplatform, in which lipid–protein stoichiometry, apolipoprotein or peptide architecture, lipid/surface chemistry, and assembly method can be deliberately tuned to control particle morphology, cargo integration, receptor interactions, and ultimately biological fate. Recent progress in rational apoA-I mimetic design, ligand- and cholesterol-based surface engineering, and increasingly quantitative composition-property analyses has therefore shifted the field from simple HDL reconstruction toward modular and chemically engineered lipoprotein nanocarriers. This conceptual transition provides the main motivation for the present review.13
2 Structure and physiological functions of high-density lipoprotein
HDL is predominantly synthesized in the liver and small intestine, and its principal protein constituent is apolipoprotein A-I (apoA-I), which represents roughly 70% of the total protein content associated with HDL particles. ApoA-I is a 28 kDa protein composed of eight amphipathic α-helical repeats, each containing 22 amino acids, and these structural motifs play a central role in dictating HDL's architecture and particle size. The conformational dynamics and lipid binding interactions of apoA-I ultimately govern the morphology and biophysical characteristics of mature HDL particles.14Natural HDL particles exist in several forms, including discoidal pre-β-HDL and the two major spherical subclasses, HDL2 and HDL3, both of which contain a neutral lipid core composed of cholesteryl esters and triglycerides. HDL2 and HDL3 constitute the predominant species in human plasma. Nascent HDL produced in the liver is primarily composed of phospholipids and apoA-I and initially adopts a discoidal configuration. Through the catalytic activity of lecithin cholesterol acyltransferase (LCAT), free cholesterol is esterified and sequestered into the particle core, driving the structural transition from discoidal HDL to mature spherical HDL3. Subsequent remodeling mediated by lipoprotein lipase (LPL) yields HDL2. HDL plays a central role in reverse cholesterol transport (RCT), mobilizing cholesterol from peripheral tissues to the liver for excretion. This process maintains intracellular cholesterol equilibrium and mitigates atherogenesis.15 By converting free cholesterol into cholesteryl esters, LCAT not only supports HDL maturation but also reduces plasma free cholesterol levels, establishes a favorable gradient for cholesterol efflux, and prevents pathological cholesterol accumulation in peripheral tissues.16
Emerging evidence indicates that HDL particles possess a broad spectrum of bioactive functions, including antioxidant,17 anti-inflammatory,18 anti-apoptotic,19 and antithrombotic effects,20 as well as immunomodulatory activities.21 HDL has also been reported to support pancreatic β-cell survival and enhance insulin secretion, thereby contributing to glucose homeostasis.22,23 Proteomic analyses have identified over 85 HDL associated proteins, which participate in interconnected biological processes such as the acute phase response, inflammation, hemostasis, and immune regulation. A comprehensive understanding of HDL's structural complexity and functional diversity may facilitate the rational design of more effective and precisely targeted rHDL based drug delivery platforms.
3 Endogenous receptors of high-density lipoprotein
A detailed understanding of the ligands present on HDL particles and their corresponding cellular receptors is critical for elucidating both the physiological roles of endogenous HDL and its potential as a targeted drug delivery platform. HDL function is closely linked to its apolipoprotein composition, which includes apoA-I, apoA-II, apoA-IV, apoA-V, and apoC-M.24 Among these, apoA-I is the predominant component, comprising approximately 70% of HDL's total protein content. ApoA-I containing HDL interacts with a diverse array of cellular receptors, including scavenger receptor class B type 1 (SR-B1), ectopic F-ATPase, ABCG1, and cubilin, facilitating receptor mediated uptake and signaling.Cubilin, an endocytic receptor highly expressed in proximal renal tubules, mediates the uptake of apoA-I/HDL, thereby contributing to the recycling of HDL through endocytosis. Both ABCG1 and SR-B1 facilitate cholesterol efflux from peripheral cells to HDL; however, SR-B1 is particularly notable for its role in the selective uptake of cholesteryl esters, enabling efficient lipid influx. Consequently, SR-B1 is regarded as the principal receptor mediating rHDL based cargo delivery to target cells.25,26 Beyond lipid transport, SR-B1 also functions as an endocytic receptor, mediating the internalization of lipopolysaccharides and facilitating hepatitis C virus entry.27,28 Furthermore, Tall et al. demonstrated that SR-B1 mediates HDL endocytosis in polarized hepatocytes.29
SR-B1 is highly expressed in the liver, adrenal gland, ovary, and placenta,30,31 with the highest levels observed in the liver, where it functions as the principal receptor mediating hepatic uptake of HDL delivered cholesteryl esters during reverse transport.32 Leveraging these receptor mediated interactions, HDL represents a highly efficient and specific platform for targeted drug delivery with considerable therapeutic potential. However, the use of endogenous HDL is constrained by limited availability, complex purification procedures, and the potential for immunogenicity in allogeneic applications. These limitations underscore the need for scalable strategies to engineer HDL mimetic nanoparticles for clinical translation.
4 Advantages of rHDL nanocarriers compared to other systems
While liposomes and polymeric nanoparticles remain dominant in clinical nanomedicine, recombinant high-density lipoprotein (rHDL) nanocarriers offer distinct advantages rooted in their biomimetic nature, particularly for receptor-mediated precision targeting in cancer, atherosclerosis, and neurodegenerative disease.Liposomes are well-established delivery vehicles that excel in biocompatibility and the encapsulation of both hydrophilic and hydrophobic cargos, with several FDA-approved formulations in clinical use (e.g., Doxil®).33–35 However, they are susceptible to rapid clearance by the mononuclear phagocyte system (MPS), limited tumor penetration due to their relatively large size (50–200 nm), and payload leakage during circulation.36 In direct comparisons using tumor spheroid models, rHDL nanoparticles (∼8–20 nm) demonstrated significantly greater penetration depth and intracellular accumulation than conventional and PEGylated liposomes, attributable to their smaller size, favorable surface chemistry, and SR-B1-mediated uptake that promotes cytosolic delivery while reducing endosomal entrapment.2,37
Polymeric nanoparticles, such as PLGA-based systems, offer controlled drug release, mechanical robustness, and tunable degradation, making them well suited for long-term therapeutic applications.38,39 Hybrid PLGA–rHDL architectures further combine these properties with the targeting and stealth capabilities of the rHDL corona.40,41 Nevertheless, polymeric carriers can exhibit higher immunogenicity, potential cytotoxicity from acidic degradation products, and comparatively slower cellular internalization relative to lipid-based systems.42,43 In these respects, rHDL holds clear advantages: negligible immunogenicity, intrinsic anti-inflammatory and antioxidant activity conferred by apoA-I, and high receptor-mediated selectivity via SR-B1 overexpression in tumors, macrophages, and blood–brain barrier endothelium-enabling efficient targeted delivery without extensive surface functionalization.
Taken together, rHDL addresses key limitations of both liposomes and polymeric nanoparticles by exploiting endogenous transport pathways to achieve superior tissue penetration, reduced off-target effects, and inherent multifunctionality, including theranostic applications through co-loading of imaging agents. Although recombinant apoA-I production costs and manufacturing scalability remain practical challenges, rHDL's established translational safety profile and demonstrated preclinical superiority in receptor-targeted applications position it as a compelling next-generation platform for precision nanomedicine.
5 Preparation of recombinant HDL (rHDL)
The preparation of recombinant HDL (rHDL) nanoparticles is a critical step that determines their physicochemical properties, structural integrity, and biological performance. A variety of reconstitution strategies have been developed, each differing in complexity, scalability, and control over lipid–protein interactions. In this section, we summarize the most commonly used fabrication approaches and highlight their respective advantages and limitations in the context of drug delivery applications.5.1 Bile salt dialysis method
This method is currently the most widely used technique for preparing rHDL particles.44 The process begins with dissolving all lipid constituents in an organic solvent, which is then evaporated under a gentle stream of nitrogen to produce a uniform thin lipid film. This step ensures homogeneous lipid distribution and facilitates subsequent particle assembly. The lipid film is hydrated with an aqueous buffer containing bile salt derivatives, most commonly sodium cholate, which acts as a solubilizing agent to stabilize lipid aggregates during particle formation and can be readily removed afterward. ApoA-I or apolipoprotein mimetic peptides are then incubated with the solubilized lipid mixture at 4 °C for approximately 12 h, allowing the apolipoproteins to integrate with the lipid bilayer and adopt their native α-helical conformation, which is critical for structural stability and receptor recognition. The final step involves the removal of sodium cholate through extensive dialysis against buffer, resulting in the self-assembly of discrete, stable rHDL nanoparticles with defined size, morphology, and surface properties.45 This method allows precise control over particle composition, lipid to protein ratio, and the incorporation of therapeutic cargos, making it highly versatile for targeted drug delivery applications.5.2 Urea-assisted method
To enhance the efficiency and practicality of rHDL fabrication, Fukuda et al. developed a rapid reconstruction approach known as the urea-assisted method.46 This technique is simple, time efficient, and does not require specialized equipment, making it particularly attractive for scalable production. In this method, DMPC is first dissolved in ethanol, while apoA-I is solubilized in PBS containing urea, which serves as a protein denaturant to promote partial unfolding and facilitate lipid protein interactions. The two solutions are then combined and incubated at the gel liquid crystalline phase transition temperature of DMPC for 6 h, allowing self-assembly into rHDL nanoparticles. The yield obtained using the urea-assisted method is comparable to that achieved with the conventional bile salt dialysis technique.Characterization of the resulting nanoparticles indicates that they exhibit diameters and heights of approximately 19 ± 3 nm and 3.1 ± 0.3 nm, respectively, closely matching those produced via bile salt dialysis (18 ± 4 nm and 4.0 ± 0.5 nm). Importantly, comparative drug loading studies demonstrated that the urea-assisted method achieves similar encapsulation efficiencies for hydrophilic, hydrophobic, and amphiphilic agents, including doxorubicin (DOX), all-trans retinoic acid (ATRA), and amphotericin B. This indicates that the structural integrity and functional versatility of rHDL particles are preserved, regardless of the preparation technique.
The urea-assisted approach also offers additional advantages beyond efficiency. By promoting controlled partial denaturation of apoA-I, it may enhance the exposure of amphipathic α-helices, facilitating more uniform lipid protein interactions and potentially improving nanoparticle stability and drug loading capacity. Moreover, this method could enable easier incorporation of sensitive or labile therapeutic cargos, as it avoids the extensive dialysis and prolonged handling associated with bile salt methods. Collectively, these attributes highlight the urea-assisted technique as a robust, scalable alternative for rHDL fabrication, supporting its application in both preclinical studies and potential clinical translation.
5.3 Ultrasonication method
In this approach, phospholipids and cholesteryl oleate are first dissolved in chloroform and dried under a stream of nitrogen, followed by dehydration in a vacuum oven for 1 h to generate a uniform lipid film. The film is then hydrated with PBS buffer and vortexed for 5 minutes to produce a lipid suspension. To achieve particle size reduction and homogeneity, the suspension is ultrasonicated for 60 minutes at 48 °C under a nitrogen atmosphere. ApoA-I or apolipoprotein mimetic peptides dissolved in PBS are subsequently added, and the resulting mixture is filtered through a 0.2 µm membrane and further purified by gel filtration chromatography.Ultrasonication plays a critical role in controlling particle size and uniformity. The alternating compression and rarefaction waves generate microbubbles that undergo cavitation upon reaching a critical size, leading to localized high energy forces that reduce lipid aggregates into discrete nanoparticles. This method typically produces spherical rHDL particles with well-defined size distribution.47 Beyond size control, ultrasonication may enhance lipid protein interactions, promoting efficient incorporation of therapeutic cargos and stable particle assembly. The combination of ultrasonication and gel filtration purification ensures both structural integrity and reproducibility, making this technique a reliable alternative for the preparation of rHDL nanoparticles for drug delivery applications.
5.4 Microfluidic reconstitution method
Microfluidic devices are engineered to manipulate fluids within nanoscale channels, offering precise control over processing parameters. In conventional preparation techniques, fluid behavior is dominated by inertial and viscous forces,48 often leading to nonlinear flow phenomena, including turbulence.49 In contrast, microfluidic systems operate under laminar flow conditions with negligible inertial forces, enabling highly controlled mixing.Nanoparticle formation typically involves a change in solvent environment, such as transitioning from an organic solvent to an aqueous phase. Optimal mixing achieved through rapid and homogeneous blending of solvent and antisolvent is critical for controlling particle size. If mixing occurs more slowly than the nucleation and polymer chain self-assembly processes, nanoparticles form under heterogeneous solvent conditions, resulting in broad size distributions, structural instability, and aggregation. Conversely, rapid mixing that outpaces nucleation ensures self-assembly under uniform conditions, allowing hydrophilic polymer segments to stabilize the nanoparticles and yield a narrow, monodisperse size distribution.50
Microfluidics exploits this principle by enabling ultrafast, homogeneous mixing, thereby facilitating the formation of uniform nanoparticle populations. Compared with conventional approaches such as bile salt dialysis and ultrasonication,2 microfluidic techniques offer superior scalability, continuous operation, and high reproducibility, supporting continuous flow, high-throughput nanoparticle production while maintaining stringent control over particle quality. Several microfluidic strategies including hydrodynamic flow focusing, micromixing, droplet based systems, and turbulent flow reactors have been applied to nanoparticle fabrication.49 Microfluidic platforms (e.g., NanoAssemblr™) are already widely applied for the fabrication of liposomes,51 polymeric nanoparticles,52 and lipid–polymer hybrid nanoparticles.53 This technology has also been extended to the large-scale synthesis of bioactive HDL like particles.
In a typical three-inlet microfluidic setup, rHDL nanoparticles are generated via micromixing driven self-assembly. An ethanolic phospholipid solution flows through the central channel, flanked by aqueous streams containing apoA-I. This configuration enables precise control of lipid to apolipoprotein ratios, yielding nanoparticles that closely mimic the size, morphology, and functional properties of native HDL.54 Microfluidic platforms also allow the incorporation of imaging agents or therapeutic cargos, expanding their versatility for both diagnostic and therapeutic applications.
Moreover, this approach has been extended to the development of radiotherapeutic rHDL systems with enhanced targeting and therapeutic efficacy. For instance, microfluidic synthesis has been employed to fabricate 225Ac-loaded rHDL nanoparticles by incorporating a lipophilic 225Ac-chelate into the lipid phase, enabling controlled assembly and high reproducibility. In this system, rHDL facilitates selective recognition of scavenger receptor class B type I (SR-BI) and direct cytoplasmic delivery, resulting in efficient deposition of high radiation doses within tumor cells. The resulting 225Ac-rHDL nanoparticles exhibit high radiochemical purity and serum stability, along with significant tumor retention and potent anticancer effects in vitro and in vivo (Fig. 1).55
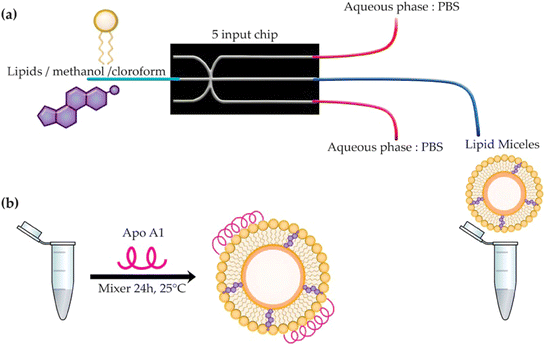 | ||
| Fig. 1 (a) Synthesis of lipid micelles. Preparation of micelles via a microfluidic system with hydrodynamic flow focusing, using a 5-way glass 3D chip that allows the diffusion of lipids in water, as well as water in alcohol, until its concentration decreases below the limit of lipid solubility, triggering the formation of lipid micelles. (b) rHDL synthesis. In the second step, the incorporation of apolipoprotein Apo AI was performed for the formation of rigid rHDL.55 | ||
While a variety of methods have been developed for rHDL fabrication, including bile salt dialysis, urea-assisted assembly, ultrasonication, and microfluidic reconstitution, these techniques exhibit distinct trade-offs in terms of chemical reproducibility, scalability, and preservation of biomolecular integrity. Bile salt dialysis remains the most established and widely adopted approach, offering excellent control over lipid–protein stoichiometry and particle composition. However, the multistep nature of detergent removal and prolonged processing time may introduce batch-to-batch variability and limit scalability. In contrast, the urea-assisted method provides a simplified and time-efficient alternative, enabling rapid assembly with comparable structural and loading characteristics, although partial protein denaturation may influence apolipoprotein conformation. Ultrasonication-based approaches facilitate particle size reduction and uniformity through cavitation-driven energy input, but the associated mechanical stress and localized heating may affect sensitive biomolecules and compromise structural integrity under certain conditions. Microfluidic reconstitution represents a significant advancement toward controlled and scalable manufacturing. By enabling rapid and homogeneous mixing under laminar flow conditions, microfluidic platforms provide improved control over nanoparticle size, dispersity, and composition, resulting in enhanced reproducibility. Moreover, continuous-flow operation and precise tuning of mixing parameters make microfluidics particularly attractive for large-scale production and translational applications. Importantly, these fabrication methods not only influence particle size and morphology, but also impact protein structure, lipid organization, and ultimately biological performance. As such, the selection of an appropriate reconstitution strategy should consider the balance between compositional precision, process scalability, and preservation of biomolecular functionality, depending on the intended application.
6 Chemical design principles and therapeutic applications of recombinant HDL nanocarriers
Recent advances in rHDL research have made increasingly clear that biological performance is governed not by cargo identity alone, but by a constellation of underlying chemical design variables that collectively determine nanoparticle structure and function. What distinguishes modern rHDL platforms from earlier HDL-inspired systems is precisely the degree to which these variables are now treated as deliberate engineering parameters rather than passive formulation choices. Lipid-to-protein stoichiometry has emerged as a quantitative determinant of particle size, morphology, and remodeling behavior; apolipoproteins and mimetic peptides are rationally engineered to control receptor recognition and structural stability; and lipid and interfacial chemistry has been expanded through ligand conjugation, cholesterol-derived anchors, and multifunctional surface modifications. Taken together, these developments signal a transition from empirical formulation toward a more principled, composition-driven design logic. While fully predictive models linking particle composition to in vivo fate remain at an early stage, recent work has begun to establish a quantitative formulation-to-function framework-one that positions rHDL nanocarriers as a chemically tunable and increasingly rational class of nanomedicine.6.1 Control of lipid-to-protein stoichiometry and its impact on physicochemical properties and targeting
A major recent advance has been the recognition that lipid-to-protein stoichiometry is not merely a formulation parameter, but a programmable chemical variable that influences particle remodeling, receptor interactions, and in vivo transport behavior. Importantly, recent studies have further demonstrated that rHDL composition directly governs particle remodeling through dynamic interactions with endogenous lipoproteins, establishing a clear link between compositional design and biological fate.56 One of the most powerful chemical design levers in rHDL nanocarrier engineering is the precise control of lipid-to-protein stoichiometry (i.e., the molar ratio of phospholipid molecules to apolipoprotein A-I or its mimetic peptides). This ratio, typically tunable between 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 150
1 and 150![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, serves as a fundamental parameter that dictates nearly all key physicochemical properties of the resulting nanoparticles. At an optimal ratio of approximately 100
1, serves as a fundamental parameter that dictates nearly all key physicochemical properties of the resulting nanoparticles. At an optimal ratio of approximately 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, rHDL particles consistently form highly uniform nanostructures with diameters of 8–12 nm.56,57
1, rHDL particles consistently form highly uniform nanostructures with diameters of 8–12 nm.56,57
The ability to fine-tune stoichiometry is readily achieved through established preparation methods. For instance, the bile salt dialysis method allows exquisite control by varying the initial lipid-to-apoA-I feeding ratio, while microfluidic reconstitution enables continuous and highly reproducible adjustment of this parameter in a single step.54,58 Recent advances in controlled production and characterization further suggest that formulation parameters can be quantitatively linked to biodistribution profiles, reinforcing stoichiometry as a central determinant of in vivo performance rather than a purely synthetic variable.59 Higher lipid![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) protein ratios not only expand the hydrophobic core volume, thereby increasing loading capacity for poorly water-soluble drugs, but also promote the formation of hybrid architectures such as PLGA–rHDL nanoparticles. In these systems, an enlarged lipid corona surrounding a polymeric core confers sustained drug release while preserving SR-B1-mediated macrophage targeting, as demonstrated in atherosclerotic plaque models.58 This structural control also governs the encapsulation of hydrophobic therapeutics. For example, peptide-based HDL (pHDL) nanodiscs with defined hydrophobic cores enable efficient loading of lipophilic compounds such as curcumin and tanshinone IIA, while preserving nanoparticle integrity and enhancing bioavailability.60 Conversely, lower ratios yield smaller, more discoidal particles with enhanced tissue penetration and faster cellular internalization.
protein ratios not only expand the hydrophobic core volume, thereby increasing loading capacity for poorly water-soluble drugs, but also promote the formation of hybrid architectures such as PLGA–rHDL nanoparticles. In these systems, an enlarged lipid corona surrounding a polymeric core confers sustained drug release while preserving SR-B1-mediated macrophage targeting, as demonstrated in atherosclerotic plaque models.58 This structural control also governs the encapsulation of hydrophobic therapeutics. For example, peptide-based HDL (pHDL) nanodiscs with defined hydrophobic cores enable efficient loading of lipophilic compounds such as curcumin and tanshinone IIA, while preserving nanoparticle integrity and enhancing bioavailability.60 Conversely, lower ratios yield smaller, more discoidal particles with enhanced tissue penetration and faster cellular internalization.
This stoichiometric tunability translates into broad therapeutic versatility across disease contexts. In solid tumors, ∼10 nm rHDL particles optimized at ∼100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratios exhibit superior extravasation and deep penetration into tumor spheroids compared with larger counterparts, significantly improving delivery of chemotherapeutics and cytolytic peptides (Fig. 2).61,62 More importantly, emerging evidence demonstrates that compositional variations-particularly lipid species and surface charge-can dominate lymphatic transport efficiency and lymph node retention, even when particle size and morphology are comparable, underscoring the decisive role of chemical composition in governing in vivo biological disposition.63 Thus, rational manipulation of lipid-to-protein stoichiometry represents a foundational chemical strategy that unifies rHDL design, enabling tailored size, stability, cargo capacity, and receptor-mediated targeting for diverse precision medicine applications.
1 ratios exhibit superior extravasation and deep penetration into tumor spheroids compared with larger counterparts, significantly improving delivery of chemotherapeutics and cytolytic peptides (Fig. 2).61,62 More importantly, emerging evidence demonstrates that compositional variations-particularly lipid species and surface charge-can dominate lymphatic transport efficiency and lymph node retention, even when particle size and morphology are comparable, underscoring the decisive role of chemical composition in governing in vivo biological disposition.63 Thus, rational manipulation of lipid-to-protein stoichiometry represents a foundational chemical strategy that unifies rHDL design, enabling tailored size, stability, cargo capacity, and receptor-mediated targeting for diverse precision medicine applications.
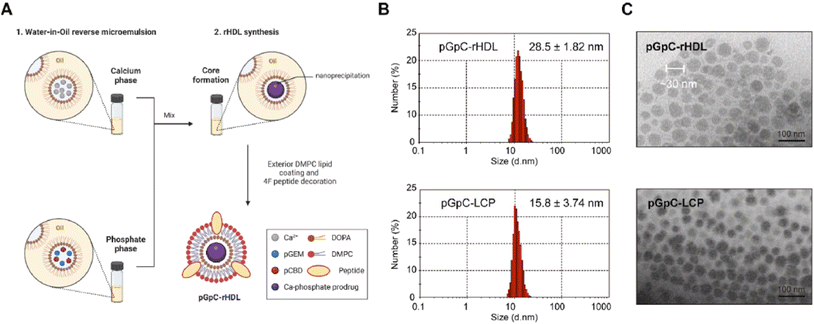 | ||
| Fig. 2 Preparation and characterization of pGpC-rHDL. (A) pGpC-rHDL was prepared mainly through reverse microemulsion and thin film hydration. (B) The mean size of pGpC-rHDL and pGpC-LCP characterized by dynamic light scattering. (C) Morphology of pGpC-rHDL and pGpC-LCP observed by TEM. Scale bars, 100 nm.62 | ||
6.2 Apolipoprotein selection and mimetic peptides: structure–function relationships in receptor recognition and targeting
Another important shift has been the move from simple apoA-I reconstitution toward rationally engineered mimetic peptides and modular protein architectures with tunable structure–function properties. The protein component of rHDL nanocarriers-primarily apolipoprotein A-I (apoA-I) or its engineered mimetic peptides-serves as the critical structural scaffold that not only stabilizes the phospholipid monolayer but also governs receptor-mediated targeting and biological functionality.14 The structure–function relationships of apoA-I are rooted in its unique amphipathic α-helical repeats (eight 22-mer helices in the full-length 243-amino-acid protein), which enable lipid binding on one face while presenting polar/charged residues on the opposite face for receptor interactions. These helices adopt flexible, belt-like conformations around the particle perimeter, facilitating high-affinity binding to scavenger receptor class B type 1 (SR-B1), the principal receptor mediating selective uptake of rHDL cargos in hepatocytes, steroidogenic cells, macrophages, tumor cells, and brain endothelial cells.25,26,30Full-length recombinant apoA-I preserves the native conformational dynamics essential for optimal SR-B1 recognition, cholesterol efflux promotion, and anti-inflammatory signaling, making it the preferred choice when maximal biomimicry is required. However, its production cost, purification complexity, and potential for batch variability have driven the development of shorter apoA-I mimetic peptides (typically 18–22 amino acids per helix, often arranged as single or bihelical constructs).64 These peptides retain the core amphipathic α-helical motif, lipid-binding capability, and SR-B1 affinity while offering advantages in scalability, chemical modification, and reduced immunogenicity. In addition, emerging work highlights the importance of peptide valency and spatial display, showing that multivalent presentation of apoA-I-derived peptides can significantly enhance lipid binding, structural integrity, and receptor engagement, underscoring the role of higher-order architecture as a key chemical design parameter.65
These structure–function principles are further reflected in modular peptide engineering strategies. Sequence-level modifications of apoA-I mimetic peptides further illustrate the importance of structure–function relationships. For example, engineered variants of the 22A peptide, including 22A-F and 22A-P-18A, have been shown to enhance cholesterol efflux, improve plasma stability, and increase LCAT activation without disrupting HDL remodeling behavior. These findings highlight that subtle sequence variations can modulate multiple functional pathways, reinforcing peptide engineering as a powerful strategy for tuning rHDL performance (Fig. 3).66 In neurodegenerative diseases, substitution with ApoE-derived peptides or full ApoE-rHDL exploits SR-B1/transcytosis pathways for blood–brain barrier penetration and targeted Aβ clearance in Alzheimer's models, or macropinocytosis in Ras-hyperactivated glioblastoma cells.67–69 In atherosclerosis, apoA-I mimetics enhance macrophage uptake and cholesterol efflux without triggering immune activation.70,71
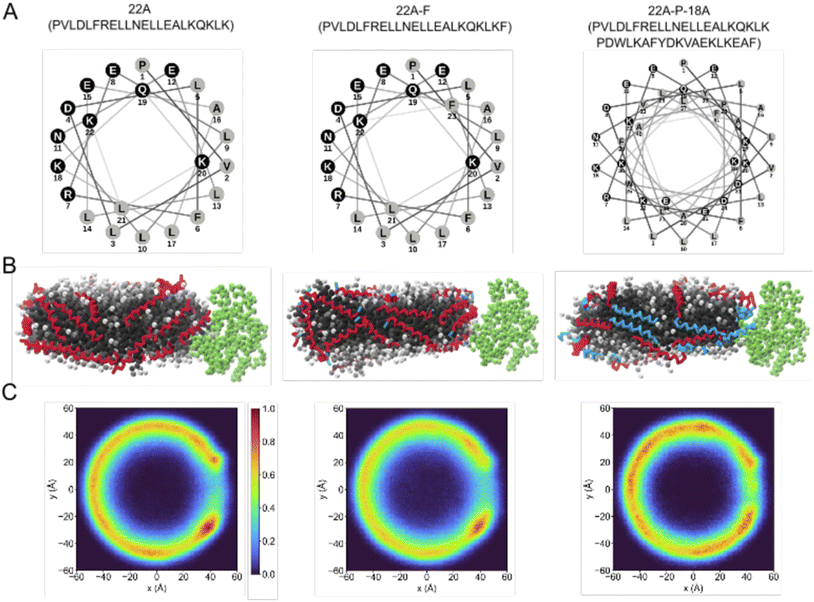 | ||
| Fig. 3 Design of amphipathic peptides via modification of the 22A apoA-I mimetic peptide sequence. (A) Helical wheel plots of the peptides and their sequences used in this study, generated using the online server NetWheels. Hydrophobic amino acids are color-coded in gray. (B) A snapshot from molecular dynamics simulations of an sHDL particle, illustrating its molecular arrangement. (C) A heatmap (top view) depicting the spatial distribution of peptide chains within the sHDL particle.66 | ||
Recent work further illustrates peptide-apoA-I synergy: co-assembly of full-length apoA-I with CIGB-258 (a 3 kDa HSP60-derived altered peptide ligand) at apoA-I![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CIGB-258 ratios of 1
CIGB-258 ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 to 1
0 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 produced dose-dependent size increases (∼60 nm to 83 nm) and markedly enhanced resistance to urea denaturation, antioxidant activity against LDL oxidation, and inhibition of CML-induced HDL glycation (up to 34% at 1
1 produced dose-dependent size increases (∼60 nm to 83 nm) and markedly enhanced resistance to urea denaturation, antioxidant activity against LDL oxidation, and inhibition of CML-induced HDL glycation (up to 34% at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). In zebrafish models of CML toxicity, higher CIGB-258 content improved survivability, reduced inflammation/apoptosis, and lowered serum markers (triglycerides, AST/ALT, IL-6). These results show CIGB-258 reinforces apoA-I helical stability and anti-inflammatory function by binding phospholipids/cholesterol, exemplifying how HSP60-derived peptides can act as multifunctional structural adjuvants.72 More broadly, these developments indicate a transition from biomimetic reconstruction toward programmable protein–lipid nanostructures, in which apolipoproteins and peptides are no longer passive structural components but active chemical design elements that govern nanoparticle assembly, stability, and biological performance.
1). In zebrafish models of CML toxicity, higher CIGB-258 content improved survivability, reduced inflammation/apoptosis, and lowered serum markers (triglycerides, AST/ALT, IL-6). These results show CIGB-258 reinforces apoA-I helical stability and anti-inflammatory function by binding phospholipids/cholesterol, exemplifying how HSP60-derived peptides can act as multifunctional structural adjuvants.72 More broadly, these developments indicate a transition from biomimetic reconstruction toward programmable protein–lipid nanostructures, in which apolipoproteins and peptides are no longer passive structural components but active chemical design elements that govern nanoparticle assembly, stability, and biological performance.
Collectively, the ability to modulate apolipoprotein structure-from full-length apoA-I for native-like function to rationally engineered mimetic peptides for scalable and customizable systems-represents a central chemical design strategy in rHDL engineering. This flexibility not only preserves intrinsic receptor recognition but also enables modular enhancements in targeting fidelity, cargo integration, and disease-specific adaptation, underscoring the platform's versatility for precision nanomedicine.
6.3 Surface functionalization and emerging multifunctional enhancements
Surface functionalization represents a critical dimension in the chemical engineering of rHDL nanocarriers, enabling the integration of targeting ligands, imaging agents, and therapeutic functionalities. A defining recent advance in rHDL design is the diversification of lipid and interfacial chemistry, shifting these systems from native HDL mimics toward chemically engineered interfaces with programmable biological functions. In this context, rHDL nanoparticles are increasingly understood not as passive biomimetic carriers, but as modular platforms whose biological behavior is governed by interfacial chemical design.73The phospholipid monolayer and exposed apolipoprotein helices provide versatile sites for covalent and non-covalent modification, allowing incorporation of ligands, stimuli-responsive moieties, imaging agents, and bioactive components without compromising particle stability or receptor recognition. Importantly, such modifications should be regarded as interfacial chemical strategies that actively regulate nanoparticle–cell interactions, receptor engagement, and downstream biological responses, rather than simple functional add-ons.74
Ligand modification expands targeting specificity beyond endogenous SR-B1-mediated recognition. Functionalization with EGFR-targeting ligands enhances selective accumulation in receptor-overexpressing tumors,75 while GM1-modified ApoE-rHDL improves Aβ recognition and promotes microglia-mediated clearance in neurodegenerative models.69 ROS-responsive ANG-modified PEGylated lipids further enable blood–brain barrier penetration and concurrent mitigation of oxidative stress.76 Recent advances further demonstrate that ligand chemistry can be used to modulate immune cell specificity. For example, mannose-functionalized rHDL nanoparticles selectively target tumor-associated macrophages (TAMs), illustrating that interfacial ligand design can actively reprogram nanoparticle–immune interactions rather than merely enhance receptor binding (Fig. 4).74
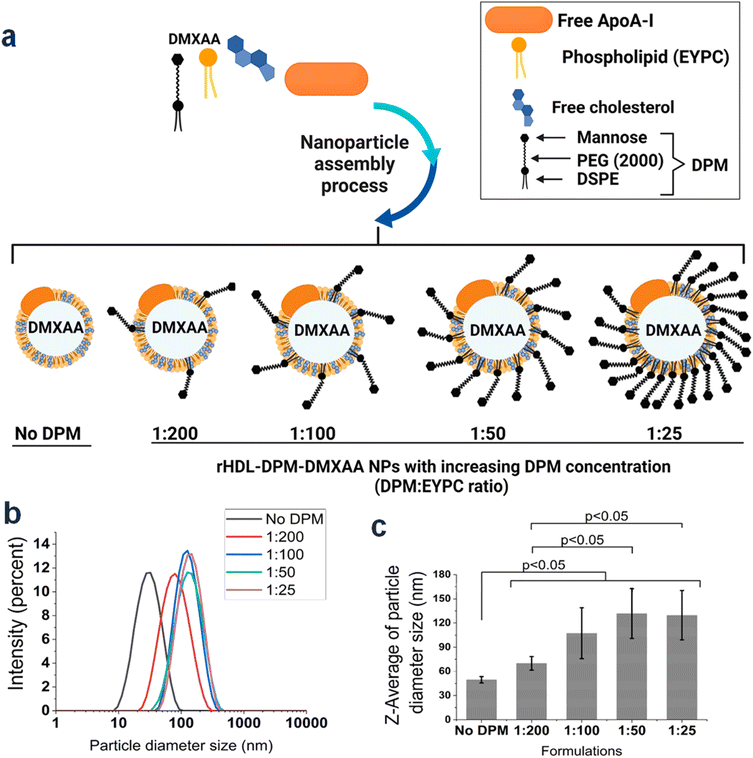 | ||
| Fig. 4 (a) Optimization of rHDL-DPM-DMXAA nanoparticles with increasing DPM ratios, while EYPC, cholesterol, and ApoA-I were held constant; particle size, PDI, zeta potential, and loading efficiencies were evaluated. (b) Intensity-weighted particle size distribution with or without DPM (no DPM). DPM ratios denote molar ratios to total phospholipids. (c) Z-Average particle size.74 | ||
Beyond ligand conjugation, chemically engineered anchoring strategies introduce an additional level of structural and functional control. In particular, cholesterol-derived chemical anchoring strategies enable stable incorporation of imaging agents, radionuclides, and therapeutic cargos into rHDL without disrupting particle architecture, thereby supporting modular and multifunctional theranostic design. Such approaches exploit the intrinsic affinity of cholesterol for lipoprotein assemblies, allowing functional components to be integrated while preserving nanoparticle stability and pharmacokinetic behavior (Fig. 5).77
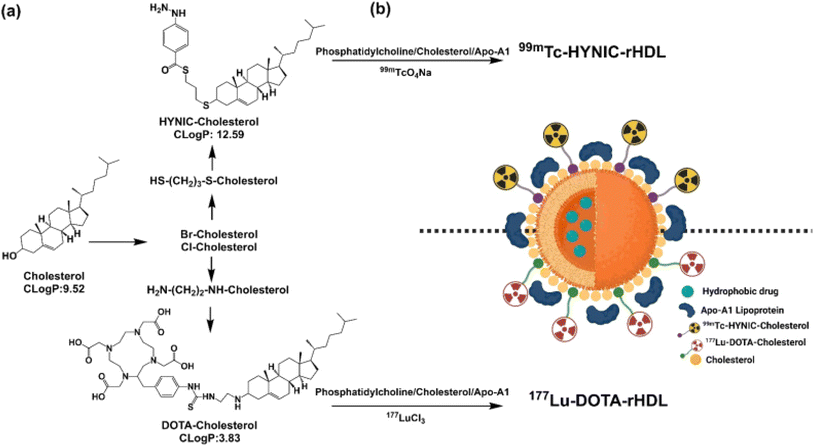 | ||
| Fig. 5 Schematic methodology for the preparation of a suitable platform for theranostic applications. (a) Synthesis of cholesterol derivatives; (b) HYNIC–rHDL or DOTA–rHDL nanoparticles radiolabeled with 99mTc and 177Lu.77 | ||
In parallel, rHDL design has expanded beyond native lipid composition through the deliberate incorporation of non-natural lipid analogs. Synthetic lipid components, such as porphyrin-conjugated lipids, introduce intrinsic optical, photodynamic, and diagnostic functionalities, representing a transition from biomimetic systems toward chemically reprogrammed lipoprotein nanostructures. This strategy enables direct encoding of new physicochemical properties into the lipid framework, substantially broadening the functional landscape of rHDL platforms.78–80 Beyond structural modification, rHDL nanoparticles exhibit intrinsically dynamic behavior in biological environments. During systemic circulation, rHDL can interact with endogenous lipoproteins and plasma components, leading to compositional remodeling through lipid and cargo exchange processes. These interactions are mediated by circulating lipoproteins, lipid transfer proteins, and enzymes, and may influence both structural integrity and drug retention.
Importantly, the extent of this remodeling is strongly dependent on particle composition, lipid–protein stoichiometry, and the chemical nature of the incorporated payload. Labile hydrophobic cargos or lipid-associated probes may partially dissociate during circulation, whereas more stable formulations-such as those incorporating conjugated lipids or optimized lipid compositions-can reduce exchange dynamics and improve retention. In parallel, emerging evidence indicates that surface and compositional chemistry governs protein corona formation and, consequently, biological fate.81 Studies in nanomedicine have shown that nanoparticle composition dictates protein corona signatures, which in turn regulate cellular uptake pathways, biodistribution, and therapeutic performance. Together, these findings highlight an emerging paradigm in which rHDL delivery efficiency arises from a balance between structural stability and dynamic remodeling, rather than from static particle integrity alone, and that composition-protein interaction-ate relationships are becoming central design principles for predictive nanocarrier engineering.82
Building on these chemical strategies, multifunctional rHDL systems have been developed through the integration of targeting, imaging, and therapeutic components within a single nanoparticle. For example, bioactive lipid incorporation-such as GM3-functionalized rHDL for enhanced anti-atherosclerotic efficacy-demonstrates how lipid composition can synergize with apoA-I-mediated targeting to improve therapeutic outcomes.83 Similarly, theranostic platforms combining imaging agents and drug payloads enable simultaneous disease detection and treatment,69,84 while co-delivery of drugs in HDL-mimicking nanodiscs has been shown to dramatically amplify therapeutic efficacy (Fig. 6).85 Although stimuli-responsive features (e.g., pH- or enzyme-triggered release) remain underexplored, the modular phospholipid/apoA-I architecture provides a versatile framework for future integration of responsive elements, enabling on-demand drug release in pathological microenvironments.86,87
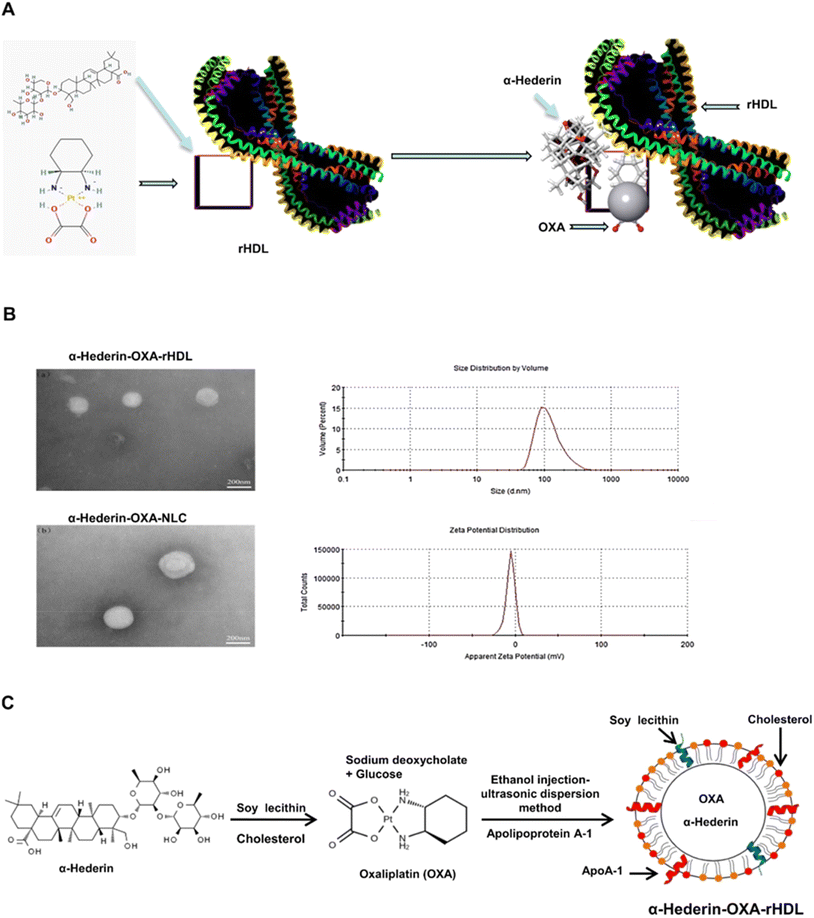 | ||
| Fig. 6 Characterization of α-hederin/oxaliplatin dual-loaded rHDL modified liposomes. (A) Chemical and structure of α-hederin/oxaliplatin dual-loaded rHDL modified liposomes (α-hederin-OXA-rHDL). (B) The appearance (a) and TEM image (b) of α-hederin-OXA-rHDL. The particle size distribution (a) and surface zeta potential (b) of α-hederin-OXA-rHDL with an average particle size from 42 to 120 nm, with an average size of 124.5 ± 3.5 nm. (C) Preparation scheme for dual-loaded liposomes with α-hederin and oxaliplatin conjugated soybean lecithin and cholesterol with apolipoprotein A-I.85 | ||
Collectively, these advances demonstrate that surface functionalization in rHDL has evolved from simple ligand decoration into a sophisticated chemical design space encompassing ligand engineering, anchoring strategies, synthetic lipid components, and interface-mediated biological regulation. This progression toward chemically programmable and multifunctional lipoprotein nanocarriers establishes a unified framework linking interfacial chemistry to biological function and therapeutic performance, and highlights the potential of rHDL systems as next-generation platforms for precision nanomedicine (Table 2).
| Design principle | rHDL system | Design parameters | Key properties (quantitative) | Applications |
|---|---|---|---|---|
| Control of lipid-to-protein stoichiometry | PLGA–rHDL hybrid nanoparticles | Lipid corona + polymer core | Size and PDI varied with the ratio of PLGA/lipids | Sustained release of hydrophobic agent (Nile Red); targeting atherosclerosis lesions of ApoE−/− mice |
| PDI: N/A | ||||
| pHDL nanodiscs | Peptide HDL; core loading | Size: 7.62–15.5 nm | Improves the bioavailability and delivery of curcumin and tanshinone IIA in atherosclerotic lesions of ApoE−/− mice | |
| Zeta potential: N/A | ||||
| PDI: N/A | ||||
| pGpC–rHDL nanodiscs | Stoichiometry-controlled nanodisc assembly | Size: ∼28.5 nm | Enhances tumor penetration of cannabidiol and gemcitabinein in pancreatic ductal adenocarcinoma | |
| Zeta potential: −1.38 mV | ||||
| PDI: ∼0.2–0.4 | ||||
| Apolipoprotein selection and mimetic peptides | 22A, 18A, and 5A peptide rHDL | Mimetic peptide design | Size: ∼8.54–11.26 nm | Enhances the efficiency of reverse cholesterol transport in cells |
| Zeta potential: N/A | ||||
| PDI: N/A | ||||
| 22A-F/22A-P-18A | Sequence engineering | Size: ∼8.9–10.1 nm | Improve cholesterol efflux from macrophages cell and structural stability of sHDL particles in human plasma | |
| Zeta potential: N/A | ||||
| PDI: N/A | ||||
| ApoE–rHDL (ANC-α-M) | Full length recombinant ApoE3 | Size: ∼35.95 nm | Delivery of α-garcinone D in SAMP8 mice reduces amyloid-β deposition, attenuates microglial proliferation, and improves memory function | |
| Zeta potential: −19.96 mV | ||||
| PDI: 0.23 | ||||
| ApoE–rHDL (siRNA-CaP-rHDL) | Apolipoprotein E3 | Size: ∼20–40 nm | Delivery of ATF5 siRNA to C6 glioblastoma model | |
| Zeta potential: −15–25 mV | ||||
| PDI: 0.2–0.4 | ||||
| CIGB-258 + apoA-I rHDL | Peptide–protein hybrid | Size: ∼60–83 nm | Enhances antioxidant and anti-glycation activities in vitro, while augmenting anti-inflammatory effects in zebrafish via CIGB-258-loaded rHDL | |
| Zeta potential: N/A | ||||
| PDI: N/A | ||||
| Surface functionalization and emerging multifunctional enhancements | EGFR–rHDL | EGFR ligand | Size: ∼10–30 nm | Enhances the targeting capability of rHDL in the KB tumor model |
| Zeta potential: ∼0.087 mV | ||||
| PDI: N/A | ||||
| αNAP-GM1-rHDL | GM1 modification | Size: ∼25.42 nm | Reduce Aβ deposition, ameliorate neurological dysfunction, and reverse memory loss in AD model | |
| Zeta potential: −15.7 mV | ||||
| PDI: 0.218 | ||||
| Mannose–rHDL | Mannose ligand | Size and zeta potential varied with the amount of DSPE-PEG-mannose | Delivery of the immunomodulatory drug DMXAA to macrophages cells | |
| PDI less than 0.3 | ||||
| ANG-PEG–rHDL | ANG ligand + ROS sensitive | Size: ∼94.3 nm | Delivery of RLA to AD mice improves cognitive function through Aβ clearance | |
| Zeta potential: −20.38 mV | ||||
| PDI: N/A | ||||
| GM3-functionalized rHDL | GM3-functionalized | Size: ∼142.3 nm | Enhances the macrophage-targeting capability of lovastatin within atherosclerotic plaques in ApoE−/− mice | |
| Zeta potential: −34.7 mV | ||||
| PDI: 0.46 | ||||
| Cholesterol-anchored rHDL | Cholesterol-radionuclide | Size: ∼60–75 nm | Delivery of various radionuclides for SPECT/PET imaging or targeted radiotherapy | |
| Zeta potential: above 18 mV | ||||
| PDI: 0.85–0.97 | ||||
| Porphyrin-lipid rHDL | Porphyrin–lipid | Size: ∼10–30 nm | Enable high-dose photosensitizer delivery for photodynamic therapy in CHO cells | |
| Zeta potential: N/A | ||||
| PDI: N/A | ||||
| α-Hederin/oxaliplatin rHDL | Dual-drug loading | Size: 125.6 nm | Delivery of alpha-hederin and oxaliplatin in a colon adenocarcinoma model | |
| Zeta potential: −15.82 mV | ||||
| PDI: 0.217 |
7 Immunogenicity, safety, and clinical translation of rHDL
Recombinant high-density lipoprotein (rHDL) nanocarriers exhibit excellent immunogenicity and safety, primarily due to their biomimetic composition: biocompatible phospholipids and recombinant apolipoprotein A-I (or a peptide mimicking lipoprotein). These components are highly similar to endogenous high-density lipoprotein and can be recognized as “self” by the immune system. Preclinical animal studies have shown that even at high doses, complement activation, cytokine release, or antibody responses are minimal, and no hemolysis or allergic reactions have been observed.88 This low immunogenicity supports repeated dosing without significant clearance by the mononuclear phagocyte system (MPS), a significant advantage over many synthetic nanoparticles. Extensive experience with rHDL-like formulations in humans, particularly in cardiovascular indications, strongly supports the clinical translation of rHDL and provides direct insights for the development of drug delivery platforms. CSL-112 (plasma-derived apolipoprotein A-I bound to phospholipids) demonstrated excellent safety in both Phase II and Phase III trials. Weekly infusion therapy for four consecutive weeks did not reveal major organ toxicity, immunogenicity signals, or hypersensitivity reactions other than mild, transient events.89 Although the AEGIS-II trial failed to meet its primary endpoint of reducing the incidence of major adverse cardiovascular events (MACE) over 90 days and no regulatory applications have been recently submitted, safety was confirmed in all subgroups, with no increase in adverse event incidence compared to the placebo group. Exploratory analyses suggested potential benefit in high-risk subgroups (e.g., elevated baseline LDL-C or inflammatory markers), but did not meet the primary efficacy.90 Earlier recombinant formulations, such as CER-001 (apolipoprotein A-I with sphingomyelin), were well-tolerated in clinical trials without immunogenicity issues.91,92 These data demonstrate the potential of rHDL for precision drug delivery, enabling high-dose administration with minimal carrier-related toxicity and facilitating the delivery of targeted payloads (e.g., chemotherapeutic drugs, siRNA) via SR-B1 while minimizing off-target effects.93Challenges include the high production cost of recombinant apolipoprotein A-I (apoA-I), the scalability of preparation methods (e.g., microfluidics), potential interactions with endogenous lipoproteins at high doses, and the need for disease-specific efficacy trials in oncology or neurodegenerative diseases (where preclinical applications of rHDL have shown promise, but large-scale human data are lacking).94,95 Nevertheless, rHDL has demonstrated low immunogenicity, good safety profiles in thousands of patients, and inherent targeting capabilities, making it a highly attractive and clinically viable platform that promises to expand the application of precision medicine beyond cardiovascular disease.96
8 Outlook and perspectives
The field of recombinant high-density lipoprotein (rHDL) nanocarriers has advanced significantly as a versatile platform for precision drug delivery, leveraging endogenous pathways for enhanced targeting fidelity, biocompatibility, and multifunctionality. While preclinical successes in cancer (e.g., synergistic multi-drug delivery and neoantigen vaccination), atherosclerosis (e.g., macrophage-targeted anti-inflammatory and imaging agents), and neurodegenerative diseases (e.g., BBB-penetrating Aβ clearance and gene silencing) underscore its promise, several key directions will shape its future development and clinical impact.First, multifunctional and stimuli-responsive designs hold strong potential. Integrating rHDL with responsive elements (e.g., pH-, ROS-, or enzyme-triggered release) could enable on-demand payload delivery in diseased microenvironments, such as acidic tumor cores or inflamed plaques.86,97,98 Hybrid systems (e.g., PLGA–rHDL or peptide-mimetic variants) may further combine sustained release with intrinsic SR-B1 targeting, improving pharmacokinetics and therapeutic indices.99,100 Advances in microfluidic fabrication and scalable recombinant apoA-I production will be critical to overcoming current cost and batch-to-batch variability barriers.101,102
Second, personalized and precision medicine integration emerges as a high-impact frontier. Patient-specific factors (e.g., SR-B1 expression levels in tumors or plaques, genetic variations in lipid metabolism) could guide rHDL formulation optimization.103,104 Combining rHDL with omics data (genomics/proteomics) may enable tailored cargos, such as neoantigen vaccines or siRNA for Ras-driven glioblastomas, advancing toward individualized therapies in oncology and neurodegeneration.105,106 Third, expanded clinical translation requires addressing remaining hurdles: long-term safety in non-cardiovascular indications, interactions with endogenous lipoproteins at therapeutic doses, and rigorous efficacy demonstration in human trials beyond CVD (where related HDL formulations like CSL-112 showed excellent safety but mixed efficacy).107,108 Focused Phase I/II studies in high-need areas (e.g., glioblastoma or pancreatic cancer) could accelerate progress, building on the validated low immunogenicity and high tolerability from thousands of patients in cardiovascular trials.
Finally, interdisciplinary efforts-spanning nanotechnology, lipid biology, immunology, and AI-driven design-will drive innovation, such as AI-optimized particle compositions or theranostic rHDL (co-loaded imaging + therapy).109–111 With these advancements, rHDL nanocarriers are poised to transition from promising biomimetic tools to transformative precision therapeutics, offering safer, more effective interventions for lipid/inflammatory-driven pathologies and broadening their biomedical footprint in the coming decade.
9 Conclusion
Biomimetic nanoparticle systems have enabled a shift toward exploiting endogenous transport pathways for precise therapeutic delivery. Recombinant high-density lipoprotein (rHDL) exemplifies this approach, combining defined composition, intrinsic biocompatibility, and favorable pharmacokinetics to support efficient and selective cargo transport. Beyond biomimicry, advances in rHDL engineering now enable programmable control over nanoparticle structure, targeting, and functional integration. Preclinical and emerging clinical studies further support its safety and translational potential across diverse disease settings. Nevertheless, challenges in scalable production, in vivo remodeling, and consistent therapeutic performance remain. Continued refinement of composition-driven design principles is expected to advance rHDL toward clinically effective precision nanomedicines.Author contributions
Wenpan Li: conceptualization, investigation, writing – original draft. Nan Jia, Mengwen Li and Yanhao Jiang: writing – original draft. Jianqin Lu: conceptualization, supervision, writing – original draft, funding acquisition.Conflicts of interest
There are no conflicts of interest to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
This work was supported in part by Faculty Retention Funds from the R. Ken Coit College of Pharmacy at the University of Arizona (UArizona) and the National Institutes of Health (NIH) grants (R35GM147002 and R01CA272487).References
- A. von Eckardstein, B. G. Nordestgaard, A. T. Remaley and A. L. Catapano, High-density lipoprotein revisited: biological functions and clinical relevance, Eur. Heart J., 2023, 44, 1394–1407 CrossRef PubMed.
- R. Kuai, D. Li, Y. E. Chen, J. J. Moon and A. Schwendeman, High-density lipoproteins: nature's multifunctional nanoparticles, ACS Nano, 2016, 10, 3015–3041 CrossRef CAS PubMed.
- B. He and Q. Yang, Recent development of LDL-based nanoparticles for cancer therapy, Pharmaceuticals, 2022, 16, 18 CrossRef PubMed.
- M. G. Damiano, R. K. Mutharasan, S. Tripathy, K. M. McMahon and C. S. Thaxton, Templated high density lipoprotein nanoparticles as potential therapies and for molecular delivery, Adv. Drug Delivery Rev., 2013, 65, 649–662 CrossRef CAS PubMed.
- K. T. Trajkovska and S. Topuzovska, High-density lipoprotein metabolism and reverse cholesterol transport: strategies for raising HDL cholesterol, Anatolian J. Cardiol., 2017, 18, 149 CAS.
- H. J. Pownall, C. Rosales, B. K. Gillard and A. M. Gotto Jr, High-density lipoproteins, reverse cholesterol transport and atherogenesis, Nat. Rev. Cardiol., 2021, 18, 712–723 CrossRef CAS PubMed.
- M. Casula, O. Colpani, S. Xie, A. L. Catapano and A. Baragetti, HDL in atherosclerotic cardiovascular disease: in search of a role, Cells, 2021, 10, 1869 CrossRef CAS PubMed.
- X. Su, G. Zhang, Y. Cheng and B. Wang, New insights into the emerging effects of inflammatory response on HDL particles structure and function, Mol. Biol. Rep., 2021, 48, 5723–5733 CrossRef CAS PubMed.
- A. Giammanco, et al., Hyperalphalipoproteinemia and beyond: the role of HDL in cardiovascular diseases, Life, 2021, 11, 581 CrossRef CAS PubMed.
- K. C. Vickers, B. T. Palmisano, B. M. Shoucri, R. D. Shamburek and A. T. Remaley, MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins, Nat. Cell Biol., 2011, 13, 423–433 CrossRef CAS PubMed.
- H. Huang, W. Cruz, J. Chen and G. Zheng, Learning from biology: synthetic lipoproteins for drug delivery, Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol., 2015, 7, 298–314 CrossRef CAS PubMed.
- S. Busatto, et al., Lipoprotein-based drug delivery, Adv. Drug Delivery Rev., 2020, 159, 377–390 CrossRef CAS PubMed.
- Z. Xie, et al., A natural nano-platform: advances in drug delivery system with recombinant high-density lipoprotein, Chin. Chem. Lett., 2024, 35, 109584 CrossRef CAS.
- Z.-C. Mo, K. Ren, X. Liu, Z.-L. Tang and G.-H. Yi, A high-density lipoprotein-mediated drug delivery system, Adv. Drug Delivery Rev., 2016, 106, 132–147 CrossRef CAS PubMed.
- C. L. Millar, Q. Duclos and C. N. Blesso, Effects of dietary flavonoids on reverse cholesterol transport, HDL metabolism, and HDL function, Adv. Nutr., 2017, 8, 226–239 CrossRef CAS PubMed.
- A. Ossoli, S. Simonelli, C. Vitali, G. Franceschini and L. Calabresi, Role of LCAT in atherosclerosis, J. Atheroscler. Thromb., 2016, 23, 119–127 CrossRef CAS PubMed.
- A. Kontush, et al., Preferential sphingosine-1-phosphate enrichment and sphingomyelin depletion are key features of small dense HDL3 particles: relevance to antiapoptotic and antioxidative activities, Arterioscler., Thromb., Vasc. Biol., 2007, 27, 1843–1849 CrossRef CAS PubMed.
- S. Patel, et al., Reconstituted high-density lipoprotein increases plasma high-density lipoprotein anti-inflammatory properties and cholesterol efflux capacity in patients with type 2 diabetes, J. Am. Coll. Cardiol., 2009, 53, 962–971 CrossRef CAS PubMed.
- J. A. De Souza, et al., Small, dense HDL 3 particles attenuate apoptosis in endothelial cells: pivotal role of apolipoprotein A-I, J. Cell. Mol. Med., 2010, 14, 608–620 CrossRef CAS PubMed.
- J. R. Nofer, M. F. Brodde and B. E. Kehrel, High-density lipoproteins, platelets and the pathogenesis of atherosclerosis, Clin. Exp. Pharmacol. Physiol., 2010, 37, 726–735 CrossRef CAS PubMed.
- K. T. Creasy, J. P. Kane and M. J. Malloy, Emerging roles of HDL in immune function, Curr. Opin. Lipidol., 2018, 29, 486–487 CrossRef CAS PubMed.
- A. von Eckardstein and C. Widmann, High-density lipoprotein, beta cells, and diabetes, Cardiovasc. Res., 2014, 103, 384–394 CrossRef CAS PubMed.
- B. G. Drew, K.-A. Rye, S. J. Duffy, P. Barter and B. A. Kingwell, The emerging role of HDL in glucose metabolism, Nat. Rev. Endocrinol., 2012, 8, 237–245 CrossRef CAS PubMed.
- W. Annema and A. von Eckardstein, High-Density Lipoproteins–Multifunctional but Vulnerable Protections from Atherosclerosis, Circ. J., 2013, 77, 2432–2448 CrossRef CAS PubMed.
- R. Johnson, N. Sabnis, X. Sun, R. Ahluwalia and A. G. Lacko, SR-B1-targeted nanodelivery of anti-cancer agents: a promising new approach to treat triple-negative breast cancer, Breast Cancer: Targets Ther., 2017, 383–392 CAS.
- S. Acton, et al., Identification of scavenger receptor SR-BI as a high density lipoprotein receptor, Science, 1996, 271, 518–520 CrossRef CAS PubMed.
- C. Voisset, et al., High density lipoproteins facilitate hepatitis C virus entry through the scavenger receptor class B type I, J. Biol. Chem., 2005, 280, 7793–7799 CrossRef CAS PubMed.
- T. G. Vishnyakova, et al., Binding and internalization of lipopolysaccharide by Cla-1, a human orthologue of rodent scavenger receptor B1, J. Biol. Chem., 2003, 278, 22771–22780 CrossRef CAS PubMed.
- D. L. Silver, N. Wang, X. Xiao and A. R. Tall, High density lipoprotein (HDL) particle uptake mediated by scavenger receptor class B type 1 results in selective sorting of HDL cholesterol from protein and polarized cholesterol secretion, J. Biol. Chem., 2001, 276, 25287–25293 CrossRef CAS PubMed.
- W.-J. Shen, S. Asthana, F. B. Kraemer and S. Azhar, Scavenger receptor B type 1: expression, molecular regulation, and cholesterol transport function, J. Lipid Res., 2018, 59, 1114 CrossRef CAS PubMed.
- G. Cao, et al., Structure and localization of the human gene encoding SR-BI/CLA-1: evidence for transcriptional control by steroidogenic factor 1, J. Biol. Chem., 1997, 272, 33068–33076 CrossRef CAS PubMed.
- M. Krieger, Charting the fate of the “good cholesterol”: identification and characterization of the high-density lipoprotein receptor SR-BI, Annu. Rev. Biochem., 1999, 68, 523–558 CrossRef CAS PubMed.
- R. Eugster and P. Luciani, Liposomes: Bridging the gap from lab to pharmaceuticals, Curr. Opin. Colloid Interface Sci., 2025, 75, 101875 CrossRef CAS.
- Y. C. Barenholz, Doxil®—The first FDA-approved nano-drug: Lessons learned, J. Controlled Release, 2012, 160, 117–134 CrossRef CAS PubMed.
- Y. Jiang, W. Li, Z. Wang and J. Lu, Lipid-based nanotechnology: liposome, Pharmaceutics, 2023, 16, 34 CrossRef PubMed.
- C. Stavnsbjerg, et al., Accelerated blood clearance and hypersensitivity by PEGylated liposomes containing TLR agonists, J. Controlled Release, 2022, 342, 337–344 CrossRef CAS PubMed.
- K. K. Ng, J. F. Lovell and G. Zheng, Lipoprotein-inspired nanoparticles for cancer theranostics, Acc. Chem. Res., 2011, 44, 1105–1113 CrossRef CAS PubMed.
- M. Alvi, A. Yaqoob, K. Rehman, S. M. Shoaib and M. S. H. Akash, PLGA-based nanoparticles for the treatment of cancer: current strategies and perspectives, AAPS Open, 2022, 8, 12 CrossRef.
- A. K. Shakya, et al., Review on PLGA polymer based nanoparticles with antimicrobial properties and their application in various medical conditions or infections, Polymers, 2023, 15, 3597 CrossRef CAS PubMed.
- J. Chen, et al., High density lipoprotein mimicking nanoparticles for atherosclerosis, Nano Convergence, 2020, 7, 6 CrossRef CAS PubMed.
- Z. Song, et al., Biologically responsive nanosystems targeting cardiovascular diseases, Curr. Drug Delivery, 2021, 18, 892–913 CrossRef CAS PubMed.
- A. Narmani, et al., Biomedical applications of PLGA nanoparticles in nanomedicine: advances in drug delivery systems and cancer therapy, Expert Opin. Drug Delivery, 2023, 20, 937–954 CrossRef CAS PubMed.
- J. Yang, et al., Recent applications of PLGA in drug delivery systems, Polymers, 2024, 16, 2606 CrossRef CAS PubMed.
- A. N. A. Jonas in Methods in Enzymology, Elsevier, 1986, vol. 128, pp. 553–582 Search PubMed.
- W. J. McConathy, M. P. Nair, S. Paranjape, L. Mooberry and A. G. Lacko, Evaluation of synthetic/reconstituted high-density lipoproteins as delivery vehicles for paclitaxel, Anti-Cancer Drugs, 2008, 19, 183–188 CrossRef CAS PubMed.
- R. Fukuda, et al., Urea-Assisted Reconstitution of Discoidal High-Density Lipoprotein, Biochemistry, 2020, 59, 1455–1464 CrossRef CAS PubMed.
- X. Zhang and B. Chen, Recombinant high density lipoprotein reconstituted with apolipoprotein AI cysteine mutants as delivery vehicles for 10-hydroxycamptothecin, Cancer Lett., 2010, 298, 26–33 CrossRef CAS PubMed.
- T. Chen, A.-P. Hynninen, R. K. Prud’homme, I. G. Kevrekidis and A. Z. Panagiotopoulos, Coarse-grained simulations of rapid assembly kinetics for polystyrene-b-poly (ethylene oxide) copolymers in aqueous solutions, J. Phys. Chem. B, 2008, 112, 16357–16366 CrossRef CAS PubMed.
- J. Ahn, et al., Microfluidics in nanoparticle drug delivery; From synthesis to pre-clinical screening, Adv. Drug Delivery Rev., 2018, 128, 29–53 CrossRef CAS PubMed.
- P. M. Valencia, O. C. Farokhzad, R. Karnik and R. Langer, Microfluidic technologies for accelerating the clinical translation of nanoparticles, Nano-Enabled Med. Appl., 2020, 93–112 Search PubMed.
- J. S. Hong, et al., Microfluidic directed self-assembly of liposome− hydrogel hybrid nanoparticles, Langmuir, 2010, 26, 11581–11588 CrossRef CAS PubMed.
- R. Karnik, et al., Microfluidic platform for controlled synthesis of polymeric nanoparticles, Nano Lett., 2008, 8, 2906–2912 CrossRef CAS PubMed.
- A. J. Mieszawska, et al., Synthesis of polymer–lipid nanoparticles for image-guided delivery of dual modality therapy, Bioconjugate Chem., 2013, 24, 1429–1434 CrossRef CAS PubMed.
- Y. Kim, et al., Single step reconstitution of multifunctional high-density lipoprotein-derived nanomaterials using microfluidics, ACS Nano, 2013, 7, 9975–9983 CrossRef CAS PubMed.
- T. Hernández-Jiménez, et al., 225Ac-rHDL nanoparticles: a potential agent for targeted alpha-particle therapy of tumors overexpressing SR-BI proteins, Molecules, 2022, 27, 2156 CrossRef PubMed.
- D. Pedersbæk, et al., The composition of reconstituted high-density lipoproteins (rHDL) dictates the degree of rHDL cargo-and size-remodeling via direct interactions with endogenous lipoproteins, Bioconjugate Chem., 2019, 30, 2634–2646 CrossRef PubMed.
- J. B. Simonsen, Evaluation of reconstituted high-density lipoprotein (rHDL) as a drug delivery platform–a detailed survey of rHDL particles ranging from biophysical properties to clinical implications, Nanomed. Nanotechnol. Biol. Med., 2016, 12, 2161–2179 CrossRef CAS PubMed.
- B. L. Sanchez-Gaytan, et al., HDL-mimetic PLGA nanoparticle to target atherosclerosis plaque macrophages, Bioconjugate Chem., 2015, 26, 443–451 CrossRef CAS PubMed.
- S. Rosanaly, et al., Production, characterization and biodistribution of therapeutic high-density lipoprotein-like nanoparticles reconstituted with or without histidine-tagged recombinant ApoA1, Biochim. Biophys. Acta, Mol. Cell Biol. Lipids, 2025, 1870, 159606 CrossRef CAS PubMed.
- J. Gao, et al., Peptide-based HDL as an effective delivery system for lipophilic drugs to restrain atherosclerosis development, Int. J. Nanomed., 2022, 3877–3892 CrossRef CAS PubMed.
- C. Huang, et al., Hybrid melittin cytolytic peptide-driven ultrasmall lipid nanoparticles block melanoma growth in vivo, ACS Nano, 2013, 7, 5791–5800 CrossRef CAS PubMed.
- Y. Huang, et al., High-density lipoprotein-based nanoplatform reprograms tumor microenvironment and enhances chemotherapy against pancreatic ductal adenocarcinoma, Biomaterials, 2025, 318, 123147 CrossRef CAS PubMed.
- G. Gracia, et al., High-density lipoprotein composition influences lymphatic transport after subcutaneous administration, Mol. Pharm., 2020, 17, 2938–2951 CrossRef CAS PubMed.
- W. Yuan, et al., Systematic evaluation of the effect of different apolipoprotein AI mimetic peptides on the performance of synthetic high-density lipoproteins in vitro and in vivo, Nanomed. Nanotechnol. Biol. Med., 2023, 48, 102646 CrossRef CAS PubMed.
- M. D. Shin, O. A. Ortega-Rivera and N. F. Steinmetz, Multivalent display of ApoAI peptides on the surface of tobacco mosaic virus nanotubes improves cholesterol efflux, Bioconjugate Chem., 2022, 33, 1922–1933 CrossRef CAS PubMed.
- S. Nouri et al., Modifications of the 22A apoA-I mimetic peptide sequence improve the anti-atherosclerotic properties of synthetic HDL, bioRxiv, 2025, preprint, 2025-2008, DOI:10.1101/2025.08.13.670037.
- J.-L. Huang, et al., Lipoprotein-biomimetic nanostructure enables efficient targeting delivery of siRNA to Ras-activated glioblastoma cells via macropinocytosis, Nat. Commun., 2017, 8, 15144 CrossRef PubMed.
- Q. Song, et al., Biomimetic ApoE-reconstituted high density lipoprotein nanocarrier for blood–brain barrier penetration and amyloid beta-targeting drug delivery, Mol. Pharm., 2016, 13, 3976–3987 CrossRef CAS PubMed.
- M. Huang, et al., GM1-modified lipoprotein-like nanoparticle: multifunctional nanoplatform for the combination therapy of Alzheimer's disease, ACS Nano, 2015, 9, 10801–10816 CrossRef CAS PubMed.
- A. B. Sigalov, Nature-inspired nanoformulations for contrast-enhanced in vivo MR imaging of macrophages, Contrast Media Mol. Imaging, 2014, 9, 372–382 CrossRef CAS PubMed.
- R. Duivenvoorden, et al., A statin-loaded reconstituted high-density lipoprotein nanoparticle inhibits atherosclerotic plaque inflammation, Nat. Commun., 2014, 5, 3065 CrossRef PubMed.
- K.-H. Cho, J.-E. Kim, D.-J. Kang, M. d. C. Dominguez-Horta and G. Martinez-Donato, Synergistic anti-inflammatory activity of apolipoprotein AI and CIGB-258 in reconstituted high-density lipoproteins (rHDL) against acute toxicity of carboxymethyllysine in zebrafish and its embryo, Pharmaceuticals, 2024, 17, 165 CrossRef CAS PubMed.
- J. Han, et al., Development of recombinant high-density lipoprotein platform with innate adipose tissue-targeting abilities for regional fat reduction, ACS Nano, 2024, 18, 13635–13651 CrossRef CAS PubMed.
- A. S. Dossou, et al., Mannose-coated reconstituted lipoprotein nanoparticles for the targeting of tumor-associated macrophages: optimization, characterization, and in vitro evaluation of effectiveness, Pharmaceutics, 2023, 15, 1685 CrossRef CAS PubMed.
- Z. Zhang, et al., HDL-Mimicking peptide–lipid nanoparticles with improved tumor targeting, Small, 2010, 6, 430–437 CrossRef CAS PubMed.
- H. Wang, et al., Versatile lipoprotein-inspired nanocomposites rescue Alzheimer's cognitive dysfunction by promoting Aβ degradation and lessening oxidative stress, Nanoscale, 2023, 15, 15717–15729 RSC.
- L. Aranda-Lara, et al., Engineered rHDL nanoparticles as a suitable platform for theranostic applications, Molecules, 2022, 27, 7046 CrossRef CAS PubMed.
- E. Huynh and G. Zheng, Porphysome nanotechnology: a paradigm shift in lipid-based supramolecular structures, Nano Today, 2014, 9, 212–222 CrossRef CAS.
- R. Chen, et al., Detection of atherosclerotic plaques with HDL-like porphyrin nanoparticles using an intravascular dual-modality optical coherence tomography and fluorescence system, Sci. Rep., 2024, 14, 12359 CrossRef CAS PubMed.
- K. K. Ng, J. F. Lovell, A. Vedadi, T. Hajian and G. Zheng, Self-assembled porphyrin nanodiscs with structure-dependent activation for phototherapy and photodiagnostic applications, ACS Nano, 2013, 7, 3484–3490 CrossRef CAS PubMed.
- M. Morbidelli, E. Papini and R. Tavano, Essential protocols for decoding the composition and the functional effects of the nanoparticle protein corona, Front. Nanotechnol., 2024, 6, 1500567 CrossRef.
- K. Liu, et al., Multiomics analysis of naturally efficacious lipid nanoparticle coronas reveals high-density lipoprotein is necessary for their function, Nat. Commun., 2023, 14, 4007 CrossRef CAS PubMed.
- B. Wei, et al., Ganglioside GM3-functionalized reconstituted high-density lipoprotein (GM3-rHDL) as a novel nanocarrier enhances antiatherosclerotic efficacy of statins in apoE−/− C57BL/6 mice, Pharmaceutics, 2022, 14, 2534 CrossRef CAS PubMed.
- S. Marrache and S. Dhar, Biodegradable synthetic high-density lipoprotein nanoparticles for atherosclerosis, Proc. Natl. Acad. Sci. U. S. A., 2013, 110, 9445–9450 CrossRef CAS PubMed.
- G. Wang, X.-N. Xu, Z. Zhi-Min, K. Wang and F. Li, Prediction and verification of targets for α-hederin/oxaliplatin dual-loaded rHDL modified liposomes: Reversing effector T-cells dysfunction and improving anti-COAD efficiency in vitro and in vivo, Int. J. Pharm., 2024, 662, 124512 CrossRef CAS PubMed.
- M. Rui, et al., Tumor microenvironment-responsive conformational activation of apoA-I mimetic peptides for targeted cancer therapy, Drug Delivery, 2026, 33, 2604086 CrossRef PubMed.
- Z. Taheri, et al., Integrin-specific stimuli-responsive nanomaterials for cancer theranostics, Pharmaceutics, 2024, 16, 1441 CrossRef CAS PubMed.
- S. Raut, et al., Reconstituted HDL: drug delivery platform for overcoming biological barriers to cancer therapy, Front. Pharmacol., 2018, 9, 1154 CrossRef CAS PubMed.
- P. Tricoci, et al., Infusion of Reconstituted High-Density Lipoprotein, CSL 112, in Patients With Atherosclerosis: Safety and Pharmacokinetic Results From a Phase 2a Randomized Clinical Trial, J. Am. Heart Assoc., 2015, 4, e002171 CrossRef PubMed.
- C. M. Gibson, et al., Rationale and design of ApoA-I Event Reducing in Ischemic Syndromes II (AEGIS-II): A phase 3, multicenter, double-blind, randomized, placebo-controlled, parallel-group study to investigate the efficacy and safety of CSL112 in subjects after acute myocardial infarction, Am. Heart J., 2021, 231, 121–127 CrossRef CAS PubMed.
- C. H. Keyserling et al., CER-001, a synthetic HDL-mimetic, safely mobilizes cholesterol in healthy dyslipidemic volunteers, Lippincott Williams & Wilkins, 2011 Search PubMed.
- K. H. Zheng, et al., HDL mimetic CER-001 targets atherosclerotic plaques in patients, Atherosclerosis, 2016, 251, 381–388 CrossRef CAS PubMed.
- A. G. Lacko, N. A. Sabnis, B. Nagarajan and W. J. McConathy, HDL as a drug and nucleic acid delivery vehicle, Front. Pharmacol., 2015, 6, 247 Search PubMed.
- Y. Mei, et al., Reconstituted high density lipoprotein (rHDL), a versatile drug delivery nanoplatform for tumor targeted therapy, J. Mater. Chem. B, 2021, 9, 612–633 RSC.
- D. Sviridov and A. T. Remaley, High-density lipoprotein mimetics: promises and challenges, Biochem. J., 2015, 472, 249–259 CrossRef CAS PubMed.
- X. Ma, Q. Song and X. Gao, Reconstituted high-density lipoproteins: novel biomimetic nanocarriers for drug delivery, Acta Pharm. Sin. B, 2018, 8, 51–63 CrossRef PubMed.
- Y. Song, H. Jing, L. B. Vong, J. Wang and N. Li, Recent advances in targeted stimuli-responsive nano-based drug delivery systems combating atherosclerosis, Chin. Chem. Lett., 2022, 33, 1705–1717 CrossRef CAS.
- C. Guo, et al., Dual stimulus-responsive renewable nanoadsorbent for selective adsorption of low-density lipoprotein in serum, Regener. Biomater., 2024, 11, rbae045 CrossRef CAS PubMed.
- K. R. Gajbhiye, et al., Lipid polymer hybrid nanoparticles: a custom-tailored next-generation approach for cancer therapeutics, Mol. Cancer, 2023, 22, 160 CrossRef PubMed.
- S. Parveen, P. Gupta, S. Kumar and M. Banerjee, Lipid polymer hybrid nanoparticles as potent vehicles for drug delivery in cancer therapeutics, Med. Drug Discovery, 2023, 20, 100165 CrossRef CAS.
- F. Jia, Y. Gao and H. Wang, Recent advances in drug delivery system fabricated by microfluidics for disease therapy, Bioengineering, 2022, 9, 625 CrossRef CAS PubMed.
- S. Siavashy, et al., A comprehensive review of one decade of microfluidic platforms applications in synthesis of enhanced carriers utilized in controlled drug delivery, Adv. Mater. Technol., 2022, 7, 2101615 CrossRef.
- M. Ullah, et al., Engineered metal nanoparticles for precision drug delivery: Pioneering the future of medicine: Mini review, J. Chin. Chem. Soc., 2024, 71, 1358–1367 CrossRef CAS.
- K. Hristova-Panusheva, C. Xenodochidis, M. Georgieva and N. Krasteva, Nanoparticle-mediated drug delivery systems for precision targeting in oncology, Pharmaceuticals, 2024, 17, 677 CrossRef CAS PubMed.
- Y. Wang, et al., Multi-omics approaches to decipher the interactions of nanoparticles and biological systems, Nat. Rev. Bioeng., 2025, 3, 333–348 CrossRef CAS.
- L. Qi, Z. Li, J. Liu and X. Chen, Omics-enhanced nanomedicine for cancer therapy, Adv. Mater., 2024, 36, 2409102 CrossRef CAS PubMed.
- G. Olatunji, et al., Can CSL-112 revolutionize atherosclerosis treatment? A critical look at the evidence, Curr. Probl. Cardiol., 2024, 49, 102680 CrossRef PubMed.
- C. R. Sirtori, G. Cincotto, S. Castiglione and C. Pavanello, HDL-replacement therapy: from traditional to emerging clinical applications, Atheroscler. Plus, 2025, 59, 68–79 CrossRef PubMed.
- H. Mazumdar, K. R. Khondakar, S. Das, A. Halder and A. Kaushik, Artificial intelligence for personalized nanomedicine; from material selection to patient outcomes, Expert Opin. Drug Delivery, 2025, 22, 85–108 CrossRef PubMed.
- S. A.-D. H. Hassan, et al., Development of nanotechnology by artificial intelligence: a comprehensive review, J. Nanostruct., 2023, 13, 915–932 Search PubMed.
- W. C. Chou, A. Canchola, F. Zhang and Z. Lin, Machine learning and artificial intelligence in nanomedicine, Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol., 2025, 17, e70027 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |
