Open Access Article
Open Access ArticleCoaxial 3D printing of zeolite core–shell structured catalysts for integrated NOx adsorption and selective catalytic reduction in cold start application
Yingzhen Wei†
a,
Jingyi Feng†ab,
Dan Lia,
Youji Qia,
Mengyang Chen c,
Shuang Wangd,
Jinfeng Hanab and
Jihong Yu
c,
Shuang Wangd,
Jinfeng Hanab and
Jihong Yu *ab
*ab
aState Key Laboratory of Inorganic Synthesis and Preparative Chemistry, College of Chemistry, Jilin University, 2699 Qianjin Street, Changchun 130012, P. R. China. E-mail: jihong@jlu.edu.cn
bInternational Centre of Future Science, Jilin University, 2699 Qianjin Street, Changchun 130012, P. R. China
cSchool of Pharmaceutical and Chemical Engineering, Taizhou University, Taizhou 317000, P. R. China
dHenan Province Function-Oriented Porous Materials Key Laboratory, College of Chemistry and Chemical Engineering, Luoyang Normal University, Luoyang 471934, P. R. China
First published on 27th February 2026
Abstract
NH3 selective catalytic reduction (NH3-SCR) is the most effective technology to alleviate NOx emission from diesel vehicles but faces the cold start problem. Ideally, integrating passive NOx adsorption (PNA) and NH3-SCR could achieve a cost-effective and space-friendly tandem of these two units, but designing effective catalysts to achieve both high adsorption capacity and superior catalytic activity remains a challenge. Herein, we have successfully developed an integrated PNA-SCR catalyst system based on the Pd-SSZ-13@Cu-SSZ-13 core–shell structured zeolite composite via coaxial 3D printing, which affords ultra-high NOx removal efficiency (96%) over the entire PNA and NH3-SCR process. Over the core–shell structure with spatially confined effects, NOx can be effectively adsorbed in the Pd-SSZ-13 core at low temperature (<170 °C) with less H2O competition, which is subsequently released at 200–350 °C to react completely with NH3 over the Cu-SSZ-13 shell without excessive side reactions. Based on the optimal component of composite catalysts, Pd-SSZ-13@Cu-SSZ-13 displays high adsorption capacity (NOx/Pd = 0.54), high adsorption rate, optimized desorption temperature (∼250 °C), and excellent NH3-SCR activity, providing a potential solution to the cold start challenge in NOx elimination.
Introduction
The abatement of NOx emissions from diesel engine exhaust represents an imperative challenge, holding profound implications for environmental sustainability and public health. To this end, Cu-exchanged zeolites with small-pore structures have been rapidly developed for selective catalytic reduction of NOx with ammonia (NH3-SCR) and have made tremendous progress in the past decade.1–5 As the predominant catalyst, Cu-exchanged SSZ-13 zeolite (Cu-SSZ-13, CHA) has been successfully commercialized owing to its excellent hydrothermal stability and catalytic activity, which can achieve 100% NOx conversion at temperatures above 200 °C.6–9 However, the NOx emission during the cold start remains an essential challenge due to the insufficient low-temperature activity of NH3-SCR catalysts.10,11 An appealing strategy is introducing passive NOx adsorbers (PNAs) into exhaust-treatment systems to handle the cold start problem.12,13 Over PNA units, NOx can be trapped at low temperature (<170 °C) and released at the operating temperature of the NH3-SCR catalyst (200–350 °C).14–16 Currently, zeolite-supported Pd catalysts (e.g., Pd-SSZ-13) are the extensively studied PNA materials, which attract significant attention because of their superb NOx adsorption efficiency and capacity.17–20To avoid undesired oxidation of NH3 by precious metals, the PNA unit should be located ahead of the ammonia dosing point, upstream of the SCR catalysts.21,22 However, the introduction of an additional unit leads to a more complicated post-treatment system and higher cost. Selleri et al. proposed the “AdSCR” concept based on non-precious metal NOx adsorbers and SCR catalysts, which exhibited excellent NOx removal performance during the cold start.23–25 However, the “AdSCR” concept has been rarely investigated with the presence of H2O, which can seriously affect the adsorption behaviors of vehicle exhaust treatment catalysts. Creating tandem catalysts with Pd-based adsorbents and NH3-SCR catalysts can maintain adsorption and catalytic activity under humid conditions while avoiding the introduction of additional components. To this end, the Olsson group demonstrated a dual-layer monolith catalyst (Pd/SSZ-13 + Cu/SSZ-13) coated on cordierite for NOx abatement, which showed favorable coupled effects. However, this method still suffers from NOx storage losses due to the low loading of Pd-SSZ-13 and inhibited high-temperature catalytic conversion caused by the side reaction of non-selective ammonia oxidation occurred in precious metal Pd.26 Therefore, it is of great significance to develop facile strategies for effectively integrating PNA and SCR catalysts to overcome the above-mentioned issues, and thus improve the PNA-SCR technology.
As an emerging technology, 3D printing presents significant advantages in efficient and accurate construction of zeolite monolithic catalysts with high zeolite loading and desirable configurations, enabling accelerated heat and mass transfer.27–33 A series of zeolite monoliths such as ZSM-5, NaX, and SAPO-34 have been fabricated via 3D printing, which displayed outstanding adsorption, separation, and catalytic performance.34–37 Recently, our group developed a zeolite-based coaxial 3D printing technology that enables the general creation of core–shell structured catalysts with a variety of capabilities. Simultaneously, the thickness of the core/shell layers can be flexibly regulated to meet specific functional requirements.38 Employing coaxial 3D printing technology, PNA materials can be controllably integrated with NH3-SCR components to construct PNA-SCR core–shell monolithic catalysts for efficient coupling of low-temperature adsorption and high-temperature catalysis. The unique core–shell structure with spatially confined effects is expected to arrange adsorption and catalytic processes in a favorable order and suppress side reactions of ammonia oxidation. Meanwhile, the high zeolite loading and hierarchical structure are conducive to great adsorption capacity and rapid transfer rate. Therefore, we believe that coaxial 3D printing technology is a promising approach for flexibly constructing PNA-SCR coupled core–shell structured catalysts to maximize the effect of coupling while addressing the challenges of NOx storage losses and suppressed NH3-SCR activity.
In this work, an integrated PNA-SCR catalyst with Pd-SSZ-13 as the core and Cu-SSZ-13 as the shell has been successfully constructed via coaxial 3D printing (Fig. 1). The Pd-SSZ-13 in the core layer can capture NOx at <170 °C and desorb it as the temperature increases to 200–350 °C (Fig. 1a–c). The released NOx from the core layer diffuses to the Cu-SSZ-13 shell and launches an effective SCR reaction with NH3 at operating temperature (Fig. 1b and c). With the design of the core–shell structure, the Pd-SSZ-13@Cu-SSZ-13 catalyst exhibits high adsorption capacity (NOx/Pd = 0.54), high adsorption rate, optimized desorption temperature, as well as excellent NH3-SCR activity with suppressed ammonia oxidation, thus achieving an outstanding PNA-SCR coupled performance (Fig. 1c). This work provides a powerful strategy for the treatment of NOx emissions during the cold start of vehicles.
Results and discussion
Fabrication and characterization of integrated catalysts
Pd-SSZ-13@Cu-SSZ-13 core–shell structured catalysts were fabricated by coaxial 3D printing. Commercial Cu-SSZ-13 (Si/Al = 4.6, Fig. S1) was thoroughly mixed with an inorganic binder (halloysite nanotubes, HNTs, Fig. S2), an organic binder (hydroxypropyl methylcellulose, HPMC), and solvents (deionized water and ethanol) to obtain printing ink of the shell layer with suitable rheology (Fig. S3a and b). The printing ink of the core layer was prepared by mixing Pd(NO3)2 solution, NH4-SSZ-13 zeolites (Si/Al = 11, Fig. S4), and binders to generate suitable rheological behaviors (Fig. S3a–c). The Pd-SSZ-13@Cu-SSZ-13 monoliths were coaxially 3D printed using a customized coaxial nuzzle (20 G/16 G). Catalysts with different core–shell ratios were fabricated by regulating the extruding pressure. The pristine monoliths were dried at room temperature overnight and then calcined at 600 °C for 5 h to remove the organic binder. The prepared core–shell structured catalysts were donated as 3D-PdZ@CuZ. For comparison, Cu-SSZ-13 and Pd-SSZ-13 monoliths were also 3D printed, named 3D-CuZ and 3D-PdZ. According to inductively coupled plasma optical emission spectroscopy (ICP-OES) measurement (Table S1), the Pd and Cu loadings in 3D-PdZ and 3D-CuZ monoliths are 0.70 wt% and 4.10 wt%, respectively.By a coaxial 3D printing strategy, 3D-PdZ@CuZ zeolite monoliths with different sizes and geometric patterns were successfully fabricated (Fig. 2a). Honeycomb structured 3D-PdZ, 3D-CuZ, and 3D-PdZ@CuZ catalysts with inter-connected channels are illustrated in Fig. 2b. As observed from the top views of the scanning electron microscope (SEM) images (Fig. 2c), the crossing rods of the monolith possess a uniform size of about 800 µm. Fig. 2d–f illustrates cross-sectional SEM images of the representative catalyst at different magnifications, from which an integrated and uniform core–shell structure can be recognized. Zeolite particles in the core and shell layers are bonded by HNTs to form a monolith, as shown in Fig. 2f. SEM elemental mapping was performed to investigate the metal distribution in the cross-section of the catalyst (Fig. 2g). Pd and Cu elements are distributed in the core layer and shell layer, respectively, further confirming the successful construction of the Pd-SSZ-13@Cu-SSZ-13 monolithic core–shell structured catalyst. The denser distribution of Al in the shell also suggests that Cu-SSZ-13 zeolites with lower Si/Al are distributed in the shell layer.
N2 adsorption–desorption (Fig. S5 and Table S2) characterization displays similar isotherms and pore structure data over 3D-CuZ, 3D-PdZ, and 3D-PdZ@CuZ. As evidenced by SEM images (Fig. 2b–f) and pore size distributions (Fig. S5b), 3D printed core–shell catalysts feature hierarchical architectures, combining the inherent micropore structure of the zeolites, meso/macropores from the bonding/accumulation of zeolite crystals and inorganic binders, and the 3D-interconnected honeycomb structure channels. This hierarchical structure enables the free diffusion of NO (kinetic diameter 0.317 nm) and NH3 (kinetic diameter 0.260 nm) through the shell and reach the core under reaction conditions. The X-ray diffraction (XRD) patterns of the 3D-CuZ, 3D-PdZ, and 3D-PdZ@CuZ catalysts are presented in Fig. S6. The characteristic diffraction peaks of the CHA zeolite structure are observed on all catalysts, indicating that the zeolite framework structure is well preserved during the process of coaxial 3D printing. X-ray photoelectron spectroscopy (XPS, Fig. S7) and temperature-programmed reduction of hydrogen (H2-TPR, Fig. S8) patterns also show negligible changes of active Cu and Pd species between 3D-CuZ/3D-PdZ and 3D-PdZ@CuZ. These results demonstrate that the original physicochemical properties and the active species of the adsorbent and catalyst can be well maintained during the construction of core–shell structured catalysts by coaxial 3D printing.
Core–shell ratio control of integrated catalysts
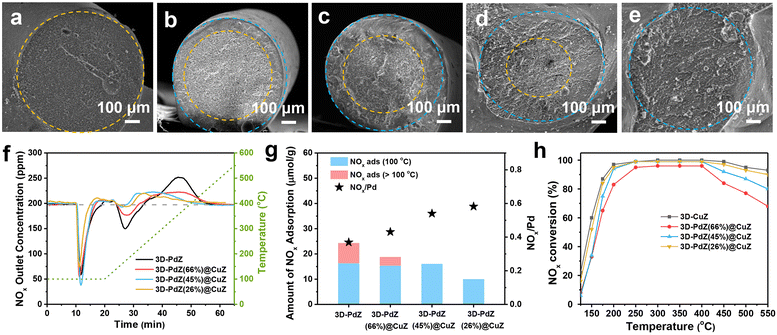
To investigate the optimal core–shell ratio in PNA-SCR performance, the monolithic catalysts with different core and shell thicknesses were constructed via coaxial 3D printing under different extruding pressures. Fig. 3a–e displays the cross-sectional SEM images of catalysts, 3D-PdZ, 3D-PdZ(66%)@CuZ (including 66% core layer and 34% shell layer), 3D-PdZ(45%)@CuZ (including 45% core layer and 55% shell layer), 3D-PdZ(26%)@CuZ (including 26% core layer and 74% shell layer), and 3D-CuZ, respectively. The percentages of the core and shell layers were calculated from ICP-OES results (Table S1).NOx storage and release curves were obtained to evaluate the PNA performance. As depicted in Fig. 3f, all samples capture NOx rapidly when the feed is switched to the catalysts, resulting in a dramatic decrease in NOx concentrations at 100 °C. After reaching the minimum value, the NOx concentrations gradually reapproach the initial values. Subsequently, as the temperature increases, a second decrease in NOx concentrations appears at ∼150 °C, which is mainly attributed to the desorption of H2O, releasing available Pd sites for NOx adsorption. Eventually, the NOx outlet concentrations exceed the initial concentration, indicating the desorption of NOx at high temperature. The amounts of NOx adsorption and NOx/Pd ratio are calculated and displayed in Fig. 3g. The adsorption capacities of the core–shell structured catalysts decrease with decreasing core thickness, which is associated with the reduced proportion of Pd-SSZ-13. It is worth noting that the NOx/Pd ratios of the core–shell structured catalysts are unexpectedly enhanced at lower Pd-SSZ-13 loadings, which demonstrates that more NOx can be adsorbed per Pd site on average, resulting in higher utilization of the precious metal Pd (Fig. S9).
The NH3-SCR tests were performed with the same weight of monolithic samples (WHSV = 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm3 g−1 h−1). As presented in Fig. 3h, increasing the proportion of the core layer leads to a gradual decline in catalytic conversion at low (<200 °C) and high (>400 °C) temperatures over catalysts, owing to the reduced loading of Cu-SSZ-13. As the percentage of the core increases to 66%, there is a considerable decline in NOx conversion across the temperature window. Among all samples, 3D-PdZ(45%)@CuZ exhibits a considerable NOx adsorption capacity (16.0 µmol g−1) and appropriate catalytic activity (maintaining ≥95% NOx conversion within the 200–400 °C range). The 3D-PdZ(45%)@CuZ is designed to adsorb NOx at low temperatures (<170 °C) and then release NOx at 200–350 °C to react completely with NH3 over the Cu-SSZ-13 shell as intended. Owing to this synergistic capacity, it is selected as a representative sample and renamed 3D-PdZ@CuZ for further discussion of its potential in PNA-SCR coupling systems.
000 cm3 g−1 h−1). As presented in Fig. 3h, increasing the proportion of the core layer leads to a gradual decline in catalytic conversion at low (<200 °C) and high (>400 °C) temperatures over catalysts, owing to the reduced loading of Cu-SSZ-13. As the percentage of the core increases to 66%, there is a considerable decline in NOx conversion across the temperature window. Among all samples, 3D-PdZ(45%)@CuZ exhibits a considerable NOx adsorption capacity (16.0 µmol g−1) and appropriate catalytic activity (maintaining ≥95% NOx conversion within the 200–400 °C range). The 3D-PdZ(45%)@CuZ is designed to adsorb NOx at low temperatures (<170 °C) and then release NOx at 200–350 °C to react completely with NH3 over the Cu-SSZ-13 shell as intended. Owing to this synergistic capacity, it is selected as a representative sample and renamed 3D-PdZ@CuZ for further discussion of its potential in PNA-SCR coupling systems.
PNA and NH3-SCR performance of integrated catalysts
To clarify the influence of the core–shell structure on catalyst performance, PNA and NH3-SCR tests were performed over fresh and aged 3D-CuZ, 3D-PdZ, and 3D-PdZ@CuZ in a fixed-bed reactor. The corresponding results are depicted in Fig. 4a–f. As shown in Fig. 4a, the NOx concentration profiles show a decline upon exposure to NOx, indicating the rapid adsorption of NOx on all samples at 100 °C. Immediately afterward, a NOx desorption peak appears for 3D-CuZ, illustrating that 3D-CuZ cannot trap NOx stably under the experimental conditions in the presence of H2O vapor. The NO/NO2 signal profiles of 3D-CuZ indicate that there is no significant NO/NO2 interconversion during the transient adsorption–desorption process (Fig. S10). Meanwhile, the 3D printed binder-only catalyst with the same structure exhibits negligible NOx adsorption, excluding weak physisorption and residence-time effects within the monolith (Fig. S11 and S12). Notably, 3D-CuZ exhibits considerable adsorption of NOx when evaluated under water-free conditions (Fig. S13). Based on the above results, it can be inferred that the adsorption sites of 3D-CuZ provide stronger interaction to H2O than NOx, leading to rapid desorption of NOx in the presence of H2O (Fig. S14a). In contrast, the Pd sites in Pd-SSZ-13 retain superior capacity of NOx adsorption in the presence of H2O vapor.39,40Over 3D-PdZ and 3D-PdZ@CuZ, the NOx concentration gradually approaches the starting concentrations as adsorption reaches saturation. 3D-PdZ@CuZ exhibits nearly identical NOx adsorption at 100 °C, despite having a smaller proportion of Pd-SSZ-13 compared to 3D-PdZ. A second NOx uptake occurs at around 150 °C for 3D-PdZ. As reported, adsorption of H2O at 100 °C weakens the binding of NOx to Pd sites.41,42 With the temperature elevating, the desorption of H2O frees up partial Pd sites leading to the additional NOx uptake (Fig. S14b). On 3D-PdZ@CuZ, there is little secondary adsorption of NOx. We suppose that the pre-adsorption of H2O on the Cu-SSZ-13 shell layer with a lower Si/Al ratio (4.6) effectively retards the diffusion of H2O molecules toward the Pd-SSZ-13 core, thereby extending the time window for preferential adsorption of NOx onto the active Pd sites prior to H2O breakthrough (Fig. S15 and S16). Therefore, the core–shell structured catalyst presents considerable NOx adsorption at 100 °C and the disappearance of re-adsorption peaks. Although there is only 45% loading of core materials, 3D-PdZ@CuZ (16.0 µmol g−1) exhibits 65% adsorption capacity of 3D-PdZ (24.3 µmol g−1) during the entire storage stage (Fig. 4b). The NOx/Pd ratio is also significantly improved in 3D-PdZ@CuZ (0.54) compared to 3D-PdZ (0.37), which indicates that the preparation of the core–shell structured material greatly enhances the utilization of the precious metal Pd and thus reduces the cost of PNA materials. The positive peaks above 200 ppm reflect the release of NOx when the temperatures exceed 200 °C. As shown in Fig. 4a, the desorption peaks of NOx appear at around 350 °C on 3D-PdZ and 250 °C on 3D-PdZ@CuZ, both within the appropriate temperature range of 200–350 °C for downstream NH3-SCR. Notably, compared to 3D-PdZ, the lower desorption temperature of 3D-PdZ@CuZ is conducive to offer available storage sites for the next run.
To further clarify the effect of H2O on the NOx adsorption and desorption process, PNA performance of 3D-PdZ was investigated with different H2O concentrations. As shown in Fig. S17, 3D-PdZ exhibits larger adsorption capacities, smaller re-adsorption peaks, and lower desorption temperatures at lower H2O concentrations. This further supports our supposition that the Cu-SSZ-13 shell creates an environment with lower H2O concentration for the Pd-SSZ-13 layer.
The NOx adsorption–desorption curves of the samples after hydrothermal aging (10% H2O, 750 °C for 16 h) are shown in Fig. 4d. The adsorption capacity of 3D-PdZ-A for NOx is greatly increased. This improvement can be ascribed to the redispersion of Pd species during hydrothermal aging, which provides more active Pd2+/[Pd(II)OH]+ sites for NOx adsorption.40 This inference is further corroborated by the CO-DRIFTS profiles and TEM images of 3D-PdZ@CuZ and 3D-PdZ@CuZ-A (Fig. S18 and S19). However, 3D-PdZ-A exhibits two NOx desorption peaks at ∼190 and ∼375 °C, which are outside the desirable temperature range. In contrast, the desorption temperatures of 3D-PdZ@CuZ-A, 225 °C and 285 °C, are still favorable for NH3-SCR, which can also be attributed to the weak damage on Pd-SSZ-13 from low-concentration H2O during hydrothermal aging. The adsorption capacity and NOx/Pd illustrate that Pd-SSZ-13 has comparable adsorption efficiency in 3D-PdZ-A and 3D-PdZ@CuZ-A (Fig. 4e). Notably, the core–shell structured catalysts exhibit fast adsorption rates before and after hydrothermal aging, thanks to the hierarchical structure of the 3D-printed monolithic catalysts and the rapid transfer of weakly adsorbed NOx in the Cu-SSZ-13 shell.
The NH3-SCR performances of fresh catalysts are depicted in Fig. 4c. 3D-PdZ exhibits poor NH3-SCR activity, with a maximal NOx conversion of 45% at 450 °C. With the same Cu-SSZ-13 loading (0.12 g Cu-SSZ-13, WHSVCu-SSZ-13 = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm3 g−1 h−1), 3D-PdZ@CuZ presents >95% NOx conversion at the temperature window from 200 to 400 °C, which is superior to that of 3D-CuZ.
000 cm3 g−1 h−1), 3D-PdZ@CuZ presents >95% NOx conversion at the temperature window from 200 to 400 °C, which is superior to that of 3D-CuZ.
This is associated with the fact that the Cu-SSZ-13 zeolites in 3D-PdZ@CuZ are mainly distributed in the ∼150 µm thick shell on the outer layer of the monolith, which can be sufficiently exposed to the reaction gas. As reported, the NH3-SCR reaction mainly occurs within 200 µm of the monolith surface,43 thus 3D-PdZ@CuZ presents a higher utilization efficiency of Cu-SSZ-13 than 3D-CuZ, where Cu-SSZ-13 zeolites are distributed throughout the monolith. This is also evidenced by the comparable NH3-SCR activity of 3D-CuZ and 3D-PdZ(26%)@CuZ with different loadings of Cu-SSZ-13 in Fig. 3h. It is well known that precious metals with high oxidizability usually lead to non-selective ammonia oxidation at high temperature,29 whereas these side reactions are hardly observed in 3D-PdZ@CuZ. The design of the core–shell structured catalysts with Pd-SSZ-13 as the core and the optimal core–shell ratio greatly addresses this problem (Fig. S20). Over 3D-PdZ@CuZ with spatially confined effects, the reactants are first reacted in the Cu-SSZ-13 shell layer and most of the reductant NH3 is consumed in the SCR reaction. The surplus NH3 diffuses into the Pd-SSZ-13 core layer and has a chance of being oxidized to NOx, which is in turn reduced to N2 by NH3 as it moves through the shell layer to leave the catalyst. By arranging the diffusion sequence of the gas flow over PNA and SCR catalysts, the problem of NOx escape caused by the non-selective oxidation in the PNA layer is overcome. As shown in Fig. 3h, the decrease in high-temperature activity due to non-selective ammonia oxidation is also observed on 3D-PdZ(66%)@CuZ with a thin shell layer, which further illustrates the importance of core–shell ratio controlling. As is known, hydrothermal aging generally results in a decrease in catalyst performance due to the de-alumination of zeolites and aggregation of CuOx. However, strikingly, 3D-PdZ@CuZ-A still presents about 8% higher conversion than 3D-CuZ-A at 200–550 °C because of the optimized distribution of Cu-SSZ-13 on the outer layer of the monolith (Fig. 4f).
N2O production, which is considered as an important aspect for SCR catalysts, is analyzed in Fig. S21. It is observed that fresh 3D-CuZ and 3D-PdZ@CuZ show minimal N2O production (<6 ppm) with excellent NH3-SCR selectivity, while fresh 3D-PdZ exhibits a much higher N2O yield (20–30 ppm) at 350–550 °C due to accelerated non-selective NH3 oxidation on Pd sites. After hydrothermal aging, 3D-CuZ-A and 3D-PdZ@CuZ-A maintain low N2O production (<6 ppm) below 400 °C but show increased yields (6–12 ppm) at 400–550 °C, which is attributed to the formation of additional CuOx species induced by aging that promotes non-selective NH3 oxidation. This explanation is evidenced by H2-TPR and XRD characterization studies of 3D-PdZ@CuZ and 3D-PdZ@CuZ-A (Fig. S22 and S23).
The influence of SO2 on catalyst performance has been also investigated. Upon the addition of 50 ppm SO2, the NOx adsorption amount of 3D-PdZ@CuZ increases to 20 µmol g−1 (Fig. S24a and b). The existence of SO2 seems to enhance the NOx storage in the PNA process via PdSO4 formation and intermediate-mediated synergistic adsorption.44 In the NH3-SCR reaction (Fig. S24c), with 50 ppm SO2 introduced, a significant decrease in NOx conversion is observed at temperatures below 300 °C, which is attributed to the pore blockage by (NH4)2SO4 (decomposing at 300 °C) or NH4HSO4 (decomposing at 350 °C).45–47 Notably, mid- and high-temperature conversions remain largely unchanged or slightly enhanced, which is attributed to sulfate species gradually decomposing and reopening the channel system of the zeolite.
To compare the catalysts with different assemblies, the layer-stacked 3D-printed catalyst with 3D-PdZ positioned in front of 3D-CuZ (3D-PdZ|CuZ) and the 3D-printed Pd-SSZ-13 and Cu-SSZ-13 mixed catalyst (3D-PdZ/CuZ) with the same PNA and SCR catalyst loading were also evaluated under the same reaction conditions (Fig. S25). The results are depicted in Fig. 4g–i. 3D-PdZ|CuZ exhibits a minimum adsorption at 100 °C and a maximum re-adsorption at 150 °C among the three samples, behaving with an adsorption–desorption profile similar to that of 3D-PdZ, as shown in Fig. 4g. Because 3D-CuZ is located downstream of 3D-PdZ in the layered 3D-PdZ|CuZ catalyst, it cannot influence the NOx adsorption and desorption process on 3D-PdZ. The dispersed Cu-SSZ-13 in 3D-PdZ/CuZ provides partial pre-adsorption of H2O and its adsorption–desorption curve is between those of 3D-PdZ@CuZ and 3D-PdZ/CuZ because the gas containing H2O diffuses randomly between Pd-SSZ-13 and Cu-SSZ-13 without a specific sequence. Notably, 3D-PdZ@CuZ provides the largest NOx adsorption capacity and the highest low-temperature adsorption percentage, indicating that the core–shell structure is superior to other combination forms in the PNA process (Fig. 4h). In the NH3-SCR experiments, the NOx conversions of 3D-PdZ|CuZ and 3D-PdZ/CuZ exhibit a drastic decrease at high temperature (Fig. 4i). On 3D-PdZ|CuZ, the oxidation of NH3 over Pd-SSZ-13 at high temperature leads to a shortage of reductant for the subsequent NH3-SCR reaction on Cu-SSZ-13, which causes an inferior high-temperature NH3-SCR performance (Fig. S26). For 3D-PdZ/CuZ, there is no spatial restriction between Pd-SSZ-13 and Cu-SSZ-13, which means NOx adsorption and desorption, ammonia oxidation, as well as NH3-SCR occur independently and synchronously (Fig. S27). Thus, the NOx raised by ammonia oxidation and the NOx desorbed from Pd-SSZ-13 might leave the catalyst directly without reacting further on Cu-SSZ-13, resulting in the suppressed NOx conversion. In contrast, 3D-PdZ@CuZ still achieves 80% conversion even at 550 °C owing to the controllable diffusion sequence of the gas flow as discussed above, which suggests that the core–shell structure also presents a significant advantage in the NH3-SCR reaction.
To further demonstrate the cost-effectiveness and practical viability of the core–shell catalyst, a double-coated sample (WC-PdZ@CuZ) with the same formulation was fabricated (Fig. S28). Under the PNA-SCR performance comparison, WC-PdZ@CuZ and 3D-PdZ@CuZ exhibit identical NOx adsorption capacities and NH3-SCR activity, indicating that the 3D-printed core–shell catalyst does not require higher loading and cost of precious metal or zeolite (Fig. S29). Compared to double-coated catalysts, coaxial 3D printed catalysts possess advantages of simplified fabrication of self-supporting core–shell catalysts and a tiny structure, which demonstrate a promising application prospect.
Mimicking test of integrated catalysts
To further demonstrate the potential of this core–shell structured catalysts, a complete mimicking test of cold start and operating conditions of 3D-CuZ, 3D-PdZ@CuZ, 3D-PdZ|CuZ, and 3D-PdZ/CuZ was carried out at an equivalent WHSV of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm3 g−1 h−1 (Fig. S30). In cold start transient experiments, 200 ppm of NOx and 5% H2O were fed in a stream of 10% O2 in N2 at 30 °C and the temperature was rapidly increased to 200 °C in about 2 min. The temperature was then ramped up to 350 °C at 20 °C min−1 and kept for 10 min to evaluate the behaviors of the coupled catalysts under operating conditions of vehicles. Finally, the temperature was increased to 550 °C to explore the performances of catalysts under high-temperature conditions. Note that, 500 ppm NH3 was injected at about 165 °C, which is consistent with the urea decomposition temperature.
000 cm3 g−1 h−1 (Fig. S30). In cold start transient experiments, 200 ppm of NOx and 5% H2O were fed in a stream of 10% O2 in N2 at 30 °C and the temperature was rapidly increased to 200 °C in about 2 min. The temperature was then ramped up to 350 °C at 20 °C min−1 and kept for 10 min to evaluate the behaviors of the coupled catalysts under operating conditions of vehicles. Finally, the temperature was increased to 550 °C to explore the performances of catalysts under high-temperature conditions. Note that, 500 ppm NH3 was injected at about 165 °C, which is consistent with the urea decomposition temperature.
For empty tubes without the catalyst, the corresponding signals can be detected immediately after NOx and NH3 are introduced, as shown in Fig. S31. Over 3D-CuZ, a large amount of NOx is released during the cold start stage (Fig. 5a). As the temperature increases, the NOx concentration gradually decreases after the addition of NH3 until it is completely reacted at 300 °C. 3D-CuZ exhibits great removal performance of NOx at medium and high temperatures, which is attributed to the NH3-SCR activity of Cu-SSZ-13.
As depicted in Fig. 5b, NOx concentration is remarkably reduced during the cold-start stage on 3D-PdZ@CuZ. NO and NO2 concentrations gradually increase up to 65 ppm and 18 ppm with Pd-SSZ-13 adsorption saturated. As the temperature increases to the active window for an effective NH3-SCR reaction, NOx is entirely removed throughout the medium and high temperature ranges. Over 3D-PdZ|CuZ (Fig. 5c) and 3D-PdZ/CuZ (Fig. 5d), the NOx removal efficiencies are also excellent in the mid-temperature stage. However, there is increased NOx overflow at low and high temperature stages because of the limited NOx adsorption capacity and the non-selective oxidation of NH3, particularly on 3D-PdZ|CuZ. For the entire mimicking process, 3D-PdZ@CuZ displays the best overall NOx removal efficiency up to 96%, compared to 3D-CuZ (90%), 3D-PdZ|CuZ (75%), and 3D-PdZ/CuZ (89%), demonstrating the significant advantages of coaxial 3D-printed core–shell composite structures in addressing cold-start NOx emissions. Besides, 3D-PdZ@CuZ-A also maintains an outstanding NOx removal efficiency (92%) after hydrothermal aging, which indicates the long-term stability of the core–shell integrated catalyst (Fig. S32).
A series of comparative tests were conducted to further clarify the contributions of Pd-SSZ-13 and Cu-SSZ-13 in the removal of NOx. NOx and NH3 were pre-stabilized in the bypass line to perform the mimicking test. After switching the gas mixture to the reaction line, the rapid decrease in NOx concentration can be clearly observed in Fig. S33 and S34, which directly demonstrates the adsorption process over Pd-SSZ-13 compared to Fig. 5b. The adsorption–desorption profile of NOx was also carried out in the presence of NH3 (Fig. S35). In addition to the adsorption peak of Pd-SSZ-13 at 100 °C, it is easily observed that the NOx concentration gradually tends to 0 as the temperature increases. This suggests that NOx reacts with NH3 on Cu-SSZ-13 and the NOx stored in Pd-SSZ-13 is consumed by the NH3-SCR reaction after desorption. The above discussion demonstrates the successful implementation of the adsorption and catalysis integrated core–shell structure, which not only exhibits excellent NOx removal activity but also avoids the introduction of costly additional components.
Conclusions
In summary, we have successfully fabricated 3D-PdZ@CuZ core–shell structured catalysts via coaxial 3D printing for integrated NOx capture and catalysis to efficiently remove NOx from the cold start stage. Over this integrated catalyst, NOx could be trapped in Pd-SSZ-13 at low temperature (<170 °C), and released from Pd-SSZ-13 to react with NH3 on Cu-SSZ-13 at 200–350 °C, achieving a boosted synergistic effect of PNA and SCR during the cold start. The outstanding behavior of the coupled catalyst is mainly attributed to the unique macroscopic core–shell structure with spatially confined effects. During the adsorption process, the Cu-SSZ-13 shell can pre-adsorb H2O, resulting in low H2O content in the Pd-SSZ-13 layer, hence 3D-PdZ@CuZ exhibits a higher adsorption capacity (NOx/Pd = 0.54), optimised desorption temperature (∼250 °C), and greater anti-aging properties compared to 3D-PdZ. On the other hand, the NH3-SCR occurred in the zeolite shell could effectively isolate the Pd-SSZ-13 core from NH3 and restrain the side reaction of ammonia oxidation, contributing to the excellent NH3-SCR activity of 3D-PdZ@CuZ. Additionally, the core/shell ratio plays an important role in balancing the adsorption and catalytic properties. By controlling the composition and distribution of the active components in the core–shell structure, 3D-PdZ@CuZ exhibits superior NOx removal efficiency (96%) in the mimicking test of the cold start and operating conditions, outperforming 3D-CuZ (90%), 3D-PdZ|CuZ (75%), and 3D-PdZ/Cu (89%). This work provides a facile strategy for coupling the PNA process into existing exhaust treatment systems to overcome the cold start challenge of NOx elimination from diesel vehicles by coaxial 3D printing of integrated PNA-SCR core–shell structured catalysts.Author contributions
Yingzhen Wei: conceptualization, formal analysis, and writing – original draft preparation. Jingyi Feng: investigation, formal analysis, and writing – original draft preparation. Dan Li: formal analysis. Youji Qi: data curation. Mengyang Chen: visualization. Shuang Wang: manuscript revision. Jinfeng Han: validation. Jihong Yu: conceptualization, project administration, writing – review & editing, and funding acquisition.Conflicts of interest
There is no conflict of interest to report.Data availability
All data associated with this article have been included in the main text and supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc08988c.Acknowledgements
The authors thank the National Natural Science Foundation of China (Grant No. 22288101, 22105093, and 22301209), the National Key Research and Development Program of China (Grant No. 2021YFA1501202), the ‘111 Center’ (B17020), and the Natural Science Foundation of Jilin Province (Grant No. SKL202502014JC) for supporting this work.Notes and references
- Y. Wu, T. Andana, Y. Wang, Y. Chen, E. D. Walter, M. H. Engelhard, K. G. Rappé, Y. Wang, F. Gao, U. Menon, R. Daya, D. Trandal, H. An, Y. Zha and K. Kamasamudram, Appl. Catal., B, 2022, 318, 121807 CrossRef CAS.
- J. Becher, D. F. Sanchez, D. E. Doronkin, D. Zengel, D. M. Meira, S. Pascarelli, J.-D. Grunwaldt and T. L. Sheppard, Nat. Catal., 2020, 4, 46–53 CrossRef.
- Y. Bai, D. Hao, Y. Wei, J. Han, D. Li, M. Chen and J. Yu, Mater. Chem. Front., 2024, 8, 2142–2148 RSC.
- T. Zhang, F. Qiu and J. Li, Appl. Catal., B, 2016, 195, 48–58 CrossRef CAS.
- Q. Lin, C. Lin, J. Liu, S. Liu, H. Xu, Y. Chen and Y. Dan, Chem. Res. Chin. Univ., 2020, 36, 1249–1254 CrossRef CAS.
- M. Chen, W. Zhao, Y. Wei, J. Han, J. Li, C. Sun, D. Mei and J. Yu, Nano Res., 2023, 16, 12126–12133 Search PubMed.
- Y. Wang, J. Han, M. Chen, W. Lv, P. Meng, W. Gao, X. Meng, W. Fan, J. Xu, W. Yan and J. Yu, Angew. Chem., Int. Ed., 2023, 62, e202306174 Search PubMed.
- M. Chen, J. Li, W. Xue, S. Wang, J. Han, Y. Wei, D. Mei, Y. Li and J. Yu, J. Am. Chem. Soc., 2022, 144, 12816–12824 Search PubMed.
- Y. Shan, G. He, J. Du, Y. Sun, Z. Liu, Y. Fu, F. Liu, X. Shi, Y. Yu and H. He, Nat. Commun., 2022, 13, 4606 Search PubMed.
- Y. Shan, J. Du, Y. Zhang, W. Shan, X. Shi, Y. Yu, R. Zhang, X. Meng, F. S. Xiao and H. He, Natl. Sci. Rev., 2021, 8, nwab010 CrossRef CAS PubMed.
- Y. Wei, M. Chen, X. Ren, Q. Wang, J. Han, W. Wu, X. Yang, S. Wang and J. Yu, CCS Chem., 2022, 4, 1708–1719 CrossRef CAS.
- E. Bello, V. J. Margarit, E. M. Gallego, F. Schuetze, C. Hengst, A. Corma and M. Moliner, Microporous Mesoporous Mater., 2020, 302, 110222 CrossRef CAS.
- H. Geerts-Claes, S. Smet, C. Hengst, F.-W. Schuetze, E. Verheyen, M. Minjauw, C. Detavernier, S. Pulinthanathu Sree and J. Martens, J. Phys. Chem. C, 2023, 127, 7119–7130 CrossRef CAS.
- K. Khivantsev, N. R. Jaegers, L. Kovarik, J. Z. Hu, F. Gao, Y. Wang and J. Szanyi, Emiss. Control Sci. Technol., 2019, 6, 126–138 CrossRef.
- D. Li, Q. Ding, D. Hao, J. Han, G. Yang, L. Pang, Y. Guo, J. Yu and T. Li, Environ. Sci. Technol., 2023, 57, 19956–19964 CrossRef CAS PubMed.
- M. Moliner and A. Corma, React. Chem. Eng., 2019, 4, 223–234 RSC.
- H. Zhao, X. Chen, A. Bhat, Y. Li and J. W. Schwank, Appl. Catal., B, 2021, 286, 119874 CrossRef CAS.
- K. Khivantsev, N. R. Jaegers, L. Kovarik, S. Prodinger, M. A. Derewinski, Y. Wang, F. Gao and J. Szanyi, Appl. Catal., A, 2019, 569, 141–148 CrossRef CAS.
- D. Li, Q. Ding, Y. Meng, Y. Guo, L. Pang and T. Li, Sep. Purif. Technol., 2023, 322, 124344 CrossRef CAS.
- K. Khivantsev, N. R. Jaegers, L. Kovarik, J. C. Hanson, F. Tao, Y. Tang, X. Zhang, I. Z. Koleva, H. A. Aleksandrov, G. N. Vayssilov, Y. Wang, F. Gao and J. Szanyi, Angew. Chem., Int. Ed., 2018, 57, 16672–16677 CrossRef CAS PubMed.
- D. Li, Y. Meng, D. Hao, Q. Ding, L. Pang, G. Yang, Y. Guo, J. Yu and T. Li, Chem. Eng. J., 2022, 446, 136779 CrossRef CAS.
- Y. Shan, Y. Sun, Y. Li, X. Shi, W. Shan, Y. Yu and H. He, Top. Catal., 2020, 63, 944–953 Search PubMed.
- M. E. Azzoni, F. S. Franchi, N. Usberti, N. D. Nasello, L. Castoldi, I. Nova and E. Tronconi, Appl. Catal., B, 2022, 315, 121544 CrossRef CAS.
- F. Gramigni, T. Selleri, I. Nova, E. Tronconi, S. Dieterich, M. Weibel and V. Schmeisser, Top. Catal., 2019, 62, 3–9 CrossRef CAS.
- T. Selleri, F. Gramigni, I. Nova, E. Tronconi, S. Dieterich, M. Weibel and V. Schmeisser, Catal. Sci. Technol., 2018, 8, 2467–2476 RSC.
- A. Wang, K. Xie, A. Kumar, K. Kamasamudram and L. Olsson, Catal. Today, 2021, 360, 356–366 CrossRef CAS.
- S. Lawson, X. Li, H. Thakkar, A. A. Rownaghi and F. Rezaei, Chem. Rev., 2021, 121, 6246–6291 CrossRef CAS PubMed.
- X. Wen, B. Zhang, W. Wang, F. Ye, S. Yue, H. Guo, G. Gao, Y. Zhao, Q. Fang, C. Nguyen, X. Zhang, J. Bao, J. T. Robinson, P. M. Ajayan and J. Lou, Nat. Mater., 2021, 20, 1506–1511 CrossRef CAS PubMed.
- L. Long, R. Pei, Y. Liu, X. Rao, Y. Wang, S.-f. Zhou and G. Zhan, J. Hazard. Mater., 2022, 423, 126983 CrossRef CAS PubMed.
- A. K. Mohammed, S. Usgaonkar, F. Kanheerampockil, S. Karak, A. Halder, M. Tharkar, M. Addicoat, T. G. Ajithkumar and R. Banerjee, J. Am. Chem. Soc., 2020, 142, 8252–8261 CrossRef CAS PubMed.
- C. Huo, S. Wu, R. Zhang, Z. Wu, X. Tian, L. Tan, K. Li, Y. Yang, B. Wang, Z. Hu, L. Wu and D. Li, Adv. Mater., 2025, 37, 2508078 CrossRef CAS PubMed.
- C. Y. Chaparro-Garnica, E. Bailón-García, D. Lozano-Castelló and A. Bueno-López, Catal. Sci. Technol., 2021, 11, 6490–6497 RSC.
- Y. Wang, S. Lin, M. Li, C. Zhu, H. Yang, P. Dong, M. Lu, W. Wang, J. Cao, Q. Liu, X. Feng, H. Hu, N. Tsubaki and M. Wu, Appl. Catal., B, 2024, 340, 123211 CrossRef CAS.
- Y. Wu, J. He, W. Huang, W. Chen, S. Zhou, X. She, W. Zhu, F. Huang, H. Li and H. Xu, Fuel, 2023, 332, 126021 CrossRef CAS.
- R. Wang, Y. Gong, P. Wang, W. He, Y. Song, M. Xin, Q. Jiang, Y. Sha, T. Cao, H. Song and W. Lin, J. Mater. Chem. A, 2023, 11, 13945–13955 RSC.
- S. Wang, P. Bai, M. Sun, W. Liu, D. Li, W. Wu, W. Yan, J. Shang and J. Yu, Adv. Sci., 2019, 6, 1901317 CrossRef PubMed.
- S. Wang, P. Bai, Y. Wei, W. Liu, X. Ren, J. Bai, Z. Lu, W. Yan and J. Yu, ACS Appl. Mater. Interfaces, 2019, 11, 38955–38963 CrossRef CAS PubMed.
- Y. Wei, S. Wang, M. Chen, J. Han, G. Yang, Q. Wang, J. Di, H. Li, W. Wu and J. Yu, Adv. Mater., 2024, 36, 2302912 CrossRef CAS PubMed.
- Y. Li, D. Chen, X. Xu, X. Wang, R. Kang, M. Fu, Y. Guo, P. Chen, Y. Li and D. Ye, Environ. Sci. Technol., 2023, 57, 3467–3485 CrossRef CAS PubMed.
- Y. Wang, X. Shi, Z. Liu, Y. Shan, W. Shi, Y. Yu and H. He, Appl. Catal., B, 2023, 324, 122254 CrossRef CAS.
- D. Li, G. Yang, M. Chen, L. Pang, Y. Guo, J. Yu and T. Li, Appl. Catal., B, 2022, 309, 121266 CrossRef CAS.
- A. Gupta, S. B. Kang and M. P. Harold, Catal. Today, 2021, 360, 411–425 CrossRef CAS.
- C. Zheng, L. Xiao, R. Qu, S. Liu, Q. Xin, P. Ji, H. Song, W. Wu and X. Gao, Chem. Eng. J., 2019, 361, 874–884 CrossRef CAS.
- D. Yao, P. H. Ho, J. C. Wurzenberger, T. Glatz, W. Di, R. F. Ilmasani, D. Creaser and L. Olsson, Chem. Eng. J., 2024, 487, 150406 CrossRef CAS.
- Y. Wang, X. Ren, K. Li, Y. Shao, Y. Zhou, M. Zhao, C. Liu, Y. Liu, X. Liu, A. Dong, H. Wu, M. Wang, L. Meng, W. Zhang, Z. Li and Q. Liu, Fuel, 2025, 392, 134818 CrossRef CAS.
- Y. Jangjou, Q. Do, Y. Gu, L.-G. Lim, H. Sun, D. Wang, A. Kumar, J. Li, L. C. Grabow and W. S. Epling, ACS Catal., 2018, 8, 1325–1337 CrossRef CAS.
- R. Yu, Z. Zhao, S. Huang and W. Zhang, Appl. Catal., B, 2020, 269, 118825 CrossRef CAS.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |