Open Access Article
Open Access ArticleRhodium-catalyzed atroposelective C–H fluoroallylation of heteroarenes with gem-difluorocyclopropanes
Junwei Lia,
Fen Wang*a,
Xian Heb,
Genping Huang *bd,
Zi-Qiang Rong
*bd,
Zi-Qiang Rong c and
Xingwei Li
c and
Xingwei Li *a
*a
aSchool of Chemistry and Chemical Engineering, Shaanxi Normal University, Xi'an 710062, China
bDepartment of Chemistry, School of Science, Tianjin University, Tianjin, 300072, China
cFrontiers Science Center for Flexible Electronics (FSCFE), Shaanxi Institute of Flexible Electronics (SIFE), Northwestern Polytechnical University (NPU), Xi'an 710072, China
dState Key Laboratory of Chemistry and Utilization of Carbon-Based Energy Resources, College of Chemistry, Xinjiang University, Urumqi 830017, China
First published on 15th April 2026
Abstract
Atroposelective C–H bond allylation represents an efficient and sustainable strategy toward construction of axially chiral functionalized biaryls. Yet existing systems are limited in reaction patterns using olefins and allyl ethers/esters. The lack of synthetic methods is ascribed to limited mechanistic pathways underlying such transformations. Reported herein is Rh(I)-catalyzed stereoconvergent axially chiral C–H fluoroalkylation of non-electron-rich arenes using racemic gem-difluorocyclopropanes as bifunctional allyl precursors, which provides biaryls integrating an allyl moiety and a fluorine atom. Mechanistic studies reveal a Rh(I)/Rh(III) oxidative addition–reductive elimination cycle involving C–H activation, β–F elimination, and rapid epimerization of the chiral axis, enabling selective reductive elimination to afford the axially chiral biaryl with high regio- and enantioselectivity. A representative product exhibits circularly polarized luminescence (CPL), highlighting the potential for high-performance chiral optoelectronic applications.
Introduction
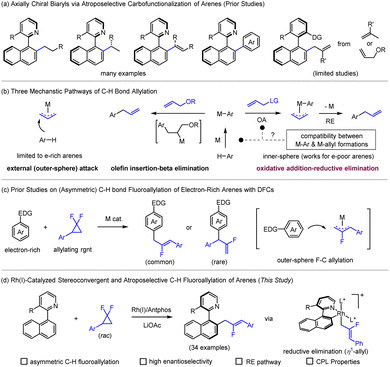
Axially chiral biaryls have emerged as privileged molecular scaffolds owing to their pivotal roles in asymmetric catalysis,1 drug discovery,2 and functional materials.3 Consequently, the efficient and precise construction of axial stereogenicity has long been a central objective in asymmetric synthesis.4 In recent years, atroposelective C–H functionalization of arenes has become a highly efficient and atom-economical strategy for building axially chiral architectures,5 offering a sustainable and more atom-economic alternative to classical cross-coupling methods that rely on prefunctionalized reagents. Among them, atroposelective C–H carbofunctionalization—including alkylation,6 alkenylation,7 and arylation8—has demonstrated remarkable advantages in enabling structural diversification with excellent stereocontrol (Scheme 1a). Despite these advances, atroposelective C–H allylation has been less explored and is largely limited to employment of olefins as allylating reagents.9 Although axially chiral allyl biaryls may serve as versatile synthetic intermediates enabling downstream diversifications, the inherent mechanistic complexity and the difficulty in balancing regio- and stereoselectivity make it challenging to develop a general and highly enantioselective strategy (Scheme 1a). Existing C–H bond allylation10 pathways typically proceed through (1) outer-sphere trapping of metal allyl by an arene or by a chelation-assisted acidic alkyl C–H group;11 (2) alkene insertion-β-elimination sequences;9,12 or (3) allyl oxidative addition–reductive elimination (OA-RE) processes.13 The last involves a high-valent M-allyl intermediate with frequent alterations of the coordination sphere, leading to poor compatibility with C–H allylation and making it difficult to achieve precise stereocontrol (Scheme 1b). Indeed, this OA-RE pathway has been less explored due to compatibility challenges between C–H activation and metal-allyl formation. Nevertheless, this pathway is theoretically applicable to electron-poor arenes and contingent on appropriate selection of a low-valent metal catalyst.Against this background, gem-difluorocyclopropanes14 (DFCs) have emerged as a unique allyl surrogate, providing an innovative and distinct approach to introduce a functionalized allyl fragment. Under transition-metal catalysis, their inherent ring strain drives C–C bond cleavage to generate a metal–allyl intermediate.15 Meanwhile, the strong electron-withdrawing nature of the fluorine substituents significantly modulates the electron density and leaving-group ability of this intermediate, enabling outer-sphere allylation with electron-rich arenes or olefins as in a Friedel–Crafts mechanism (Scheme 1b).16 The synergistic interplay between the strain-driven kinetic activation and the fluorine-induced electronic modulation provides an opportunity for the sequential realization of C–H activation, allylic intermediate generation, and C–C coupling in a stereochemically controlled fashion within a single catalytic cycle (Scheme 1c), thus offering an operationally simple and potentially general solution to atroposelective C–H allylation. Nevertheless, only very few reports on asymmetric C–H bond allylation using DFCs have been disclosed.17
Herein, we report a Rh(I)-catalyzed stereoconvergent atroposelective C–H fluoroalkylation using racemic gem-difluorocyclopropanes as bifunctional fluoroallyl precursors, providing an allyl framework with integrated fluorine atoms. This method efficiently constructs axially chiral biaryl frameworks with excellent regio- and enantioselectivity. Within the Rh(I)/Rh(III) redox cycle, the η1-allyl–Rh(III) intermediate undergoes enantio-determining reductive elimination, achieving the first enantioselective installation of a fluoroallyl unit via atropselective C–H functionalization (Fig. 1c) The resulting products exhibit pronounced circularly polarized luminescence (CPL), highlighting their potential in high-performance chiral optoelectronic applications.
Initial optimization studies
We began our investigation by screening chiral ligands using a Rh(I) catalyst in toluene in the presence of LiOAc (Fig. 1). Bidentate P–N and P–P ligands (L1–L5) exhibited negligible activity for the coupling of arene 1a with DFC 2a, whereas a chiral dppp-type ligand (L6) afforded the desired product 3 with only moderate enantioselectivity. Monodentate phosphoramidites L6 and L7 displayed both low reactivity and poor enantioselectivity. Encouragingly, Antphos L9 enabled the formation of 3 in excellent yield, albeit initially with modest reactivity. Notably, ortho substituents on the phenyl ring significantly influenced the reaction outcome, as exemplified by L10, which led to both poor reactivity and low enantioselectivity. Consequently, L9 was chosen for further optimization studies.Subsequent optimization revealed that an alkali acetate additive was essential to maintain catalytic activity (Table 1 and the SI). When NaOAc was used as an additive, the choice of solvent exerted a significant impact on both reactivity and enantioselectivity, with toluene providing the best outcomes (entries 2–7). The employment of LiOAc and AgBF4 (0.1 equiv) substantially enhanced the reaction efficiency (entry 8), while LiOAc still outperformed NaOAc in terms of both yield and selectivity (entry 9). Increasing the amount of AgBF4 led to diminished reaction efficiency, likely due to undesired single-electron oxidation of the phosphine ligand. By contrast, other additives such as lithium carbonate or magnesium acetate, or the omission of LiOAc, completely suppressed the reaction.
| Entry | Solvent | Base | Additive | ee (%)c | Yield (%)b |
|---|---|---|---|---|---|
| a Reactions were carried out using 1-(naphthalen-1-yl)isoquinoline 1a (0.1 mmol), 2a (2.0 equiv.), [Rh(cod)OMe]2 (3 mol%), L9 (6 mol%) and AgBF4 (10 mol%) in a solvent (2 mL) at 100 °C for 24 h under N2.b Isolated yield.c The ee was determined by HPLC analysis. | |||||
| 1 | DCE | NaOAc | — | — | 0 |
| 2 | PhMe | NaOAc | — | 90 | 21 |
| 3 | PhCl | NaOAc | — | 70 | 24 |
| 4 | PhCF3 | NaOAc | — | 50 | 28 |
| 5 | EA | NaOAc | — | 90 | 18 |
| 6 | THF | NaOAc | — | 76 | 15 |
| 7 | PhMe | LiOAc | — | 91 | 38 |
| 8 | PhMe | LiOAc | AgBF4 | 91 | 71 |
| 9 | PhMe | NaOAc | AgBF4 | 90 | 63 |
| 10 | PhMe | — | AgBF4 | — | 0 |
| 11 | PhMe | Li2CO3 | AgBF4 | — | <5 |
| 12 | PhMe | Mg(OAc)2 | AgBF4 | — | <5 |
Results and discussion
Substrate scope
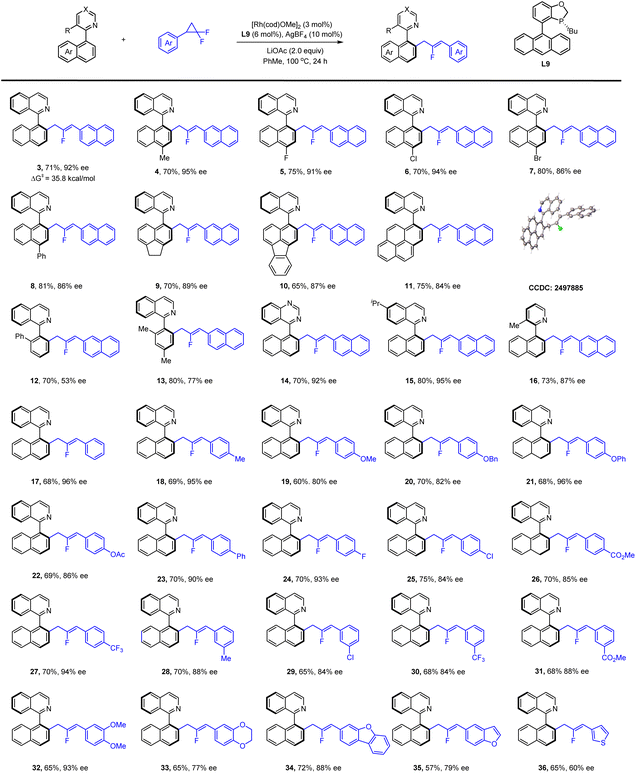
Having identified the optimal reaction conditions, we went on to investigate the generality of this catalytic system (Scheme 2). The scope of the heteroarene was investigated using 2a as the coupling reagent. It was found that introduction of alkyl, halogen, and aryl groups at the 4-positon of the naphthalene ring had marginal impact on the efficiency and enantioselectivity of this coupling system (products 4–8, 86–95% ee). The atropostability of the product has been measured, and the large racemization barrier of 35.8 kcal mol−1 indicated its high configurational stability. Fusing a carbocycle into different positions of the naphthalene was also tolerated, and the enantioselectivity was only slightly affected (9–11, 84–89% ee). The absolute configuration of product 11 has been determined by X-ray crystallography (CCDC 2497885). In contrast, replacing the naphthalene ring with a (2-phenyl)phenyl group resulted in lower enantioselectivity (12, 53% ee). Further improved enantioselectivity was found when a 2,4-xyl group was used (13, 77% yield). The heterocyclic directing group was next investigated. The simple isoquinoline ring was smoothly extended to an alkyl-substituted one (15), a quinazoline (14), and an alkyl-pyridine (16). The aryl group in the DFC reagent was then briefly investigated. Simple para alkyl-, alkoxy-, phenoxy-, acetoxy-, aryl-, ester-, CF3, and halogen-substituted phenyl groups were well tolerated (17–27, 80–96% ee). A series of electron-donating and -withdrawing meta groups (alkyl, sulfonyl, halogen, and ester) were also compatible (28–32, 84–96% ee). The aryl group was also switched to several heteroarene-fused aryls (33–35, 77–88% ee). In contrast, a 3-thienyl substituted DFC only reacted with moderate enantioselectivity (36). In all cases, the regioselectivity is excellent, and only the linear allylation product was observed.Synthetic applications
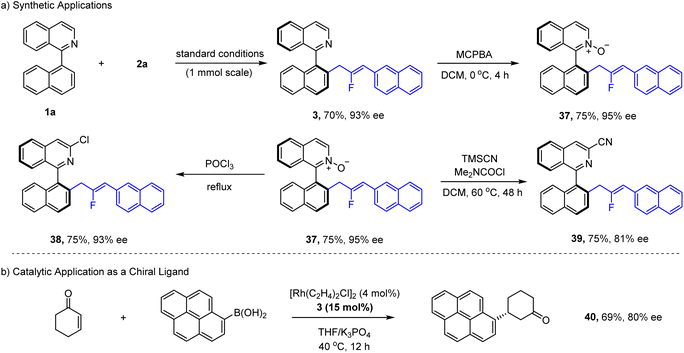
Synthetic transformations and catalytic applications were then investigated for a representative product (Scheme 3). The product 3 was prepared at a mmol scale, and no yield or enantioselectivity was compromised. The m-CPBA oxidative of 3 occurred chemoselectively at the nitrogen site (37). Treatment of 37 with POCl3 afforded a 3-chloroisioqunoline 38 in good yield. Deoxygenative cyanation proceeded smoothly to give 39 with good efficiency. In all cases, no erosion or only slight erosion of enantiopurity was observed. Moreover, product 37 also served as a chiral bidentate N-olefin ligand in Rh(I)-catalyzed Michael addition between an aryl boronic acid and cyclohexenone, affording chiral cyclohexanone 40 in good enantioselectivity.Besides the synthetic applications, the chiroptical properties of product 10 and its enantiomer were demonstrated (Fig. 2). Upon excitation (408 nm), strong fluorescence emission was observed with a maximum at 490 nm, and a quantum yield of 0.42 was measured (Fig. 2a). Strong circular dichroism with a mirror image was also observed for 10 and its enantiomer, indicating their chiral nature (Fig. 2b). To our delight, prominent CPL was also observed for these two enantiomers (Fig. 2c). Further analysis of the glum indicated that the luminescence dissymmetry factor has a magnitude of 10−3 (Fig. 2d). With these properties, this chiral product holds potential for applications as an organic semiconductor or organic optical material.
Experimental mechanistic studies
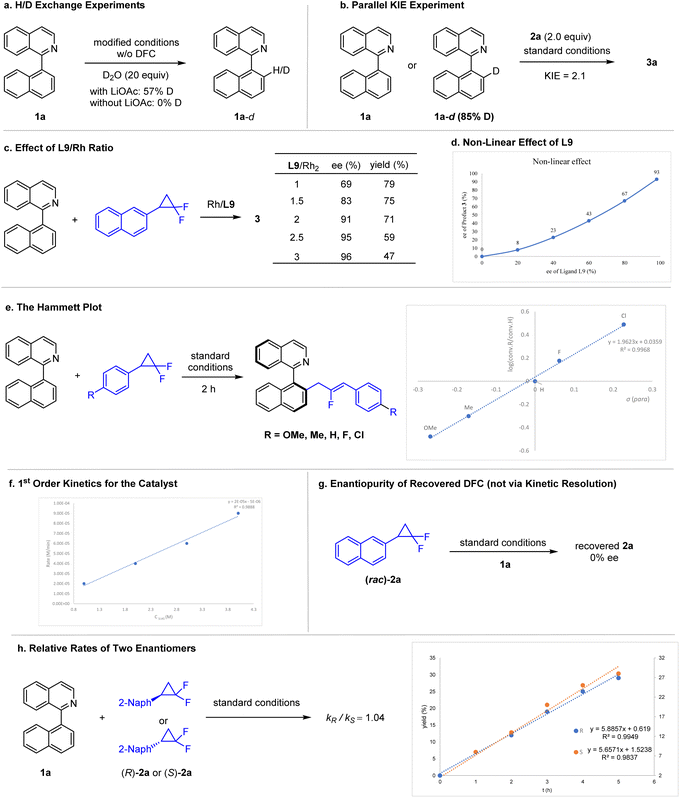
A series of experiments have been conducted to explore the mechanism of this catalytic system (Scheme 4). H/D exchange studies were carried out to probe the sequence of C–H bond activation versus C–C activation (Scheme 4a). Noticeable H/D exchange between 1-arylisoquinoline 1a and D2O was observed in the absence of a DFC reagent but in the presence of the Rh(I) catalysts and LiOAc (Scheme 4a). In contrast, no H/D exchange was detected when the LiOAc additive was omitted. In addition to the reversibility of C–H bond cleavage, these observations are consistent with the necessity of LiOAc in the catalytic reaction, and the reaction is likely initiated by C–H bond activation either via the CMD mechanism or via C–H bond oxidative addition-reductive elimination of AcOH. To further probe this C–H activation event, the KIE has been measured in parallel reactions (Scheme 4b). A KIE value of 2.1 suggests that the C–H cleavage probably occurs prior to the turnover-limiting step, and the barrier of C–H cleavage contributes to some extent to the overall barrier of the catalytic cycle. To probe the enantiocontrol event, the ratio of L9/Rh was evaluated. It was found that ee of product 3 increases as the L9/Rh ratio increases, and the highest value (95–96% ee) was reached when the L9/Rh ratio was ≥2.5 (Scheme 4c). This observation seems to indicate reversible ligation of L9 ligands, which also agrees with our non-linear effect study, where a negative NLE was observed (Scheme 4d). Our kinetic studies also revealed 1st order dependence of the Rh(I) catalyst (Scheme 4f). Therefore, a monomeric rhodium with two monodentate L9 ligands is likely operating during the EDS. To explore the electronic effect of the DFC reagent, linear free energy correlation has been investigated (Scheme 4e). The rather large positive slope of ρ = 2.0 from the Hammett plot indicates that C–C oxidative addition is likely involved either in the turnover-limiting step or prior to it. Furthermore, only racemic 2a was recovered from a catalytic reaction (Scheme 4g), suggesting that both enantiomers of 2a reacted indiscernibly during the C–C oxidative addition. In line with this observation, both enantiomers of 2a reacted with essentially the same rate in kinetic studies (Scheme 4h). Therefore, this stereoconvergent scenario stays contrast to the kinetic resolution pattern recently reported by Xia and coworkers.15d,17Computational mechanistic studies
Density functional theory (DFT) calculations were performed to gain deeper understanding of the reaction mechanism and the origin of the enantioselectivity (see the SI and Fig. 3). The reaction begins with C–H bond cleavage through the CMD process via transition state TS3, with an energy barrier of 21.3 kcal mol−1 relative to the reactant-coordinated intermediate IM2 (SI). Subsequently, with the incoming gem-difluorocyclopropane 2, the resulting intermediate IM4 undergoes C–C oxidative addition via TS4 to form the Rh(III) intermediate IM5. The subsequent β–F elimination proceeds through TS5, affording intermediate IM6. Anion exchange with the additive LiOAc and then with AgBF4 generates cationic intermediate (S)-IM0. It should be noted that formation of LiF is likely a strong driving force. The computations show that TS3 is lower in energy than TS4, indicating that the C–H bond cleavage step is reversible, which is consistent with the experimental observations (Scheme 4a). | ||
| Fig. 3 (a) DFT study on C–C reductive elimination (L* = L19). (b) Energies and bond distances are given in kcal mol−1 and Å, respectively. | ||
Accordingly, the energy profile of the full catalytic cycle can be divided into two regions: (i) prior to the C–C reductive elimination and (ii) the C–C reductive elimination step. Therefore, the energies associated with the C–C reductive elimination were calculated relative to intermediate (S)-IM0 and are presented in Fig. 3 (the key reductive elimination step is depicted in Fig. 3, with the results of the other steps included in the SI). Both bisligated and monoligated reaction pathways were considered. The computations indicate that the two pathways exhibit nearly identical activation barriers and enantioselectivities, implying that both pathways are likely accessible under the reaction conditions. It should be mentioned that the interconversion of the key rhodacyclic Rh(I) aryl intermediates (S)-IM0 and (R)-IM0 via transition state TS2 is kinetically feasible, confirming that the C–C reductive elimination is the enantioselectivity-determining step of the reaction. Furthermore, the C–C reductive elimination step has the highest energy barrier among all steps, which thus represents the rate-determining step of the reaction. Furthermore, the C–C reductive elimination step has the highest energy barrier among all steps, which thus represents the rate-determining step of the reaction.
The calculations indicate that in the bisligated reaction pathway, the transition state (S)-TS1 is 3.0 kcal mol−1 lower in energy than (R)-TS1, which corresponds to a predicted ratio of 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 at the reaction temperature, consistent with the experimentally observed enantioselectivity of 96% ee. Scrutiny of the optimized structures reveals that the enantioselectivity mainly arises from a stabilizing π–π interaction between the reacting arene 1a moiety and ligand L9 in (S)-TS1, which is absent in (R)-TS1, rendering the (S)-TS1 energetically more favorable. On the other hand, in the monoligated reaction pathway, L9 acts as a bidentate mode in both (S)-TS1′ and (R)-TS1′, with the aryl ring of L9 engaging in coordination interactions with the Pd center. The transition state (S)-TS1′ is 3.2 kcal mol−1 lower in energy than (R)-TS1′, primarily because the stabilizing interaction in (S)-TS1′ is stronger than that in (R)-TS1′.
2 at the reaction temperature, consistent with the experimentally observed enantioselectivity of 96% ee. Scrutiny of the optimized structures reveals that the enantioselectivity mainly arises from a stabilizing π–π interaction between the reacting arene 1a moiety and ligand L9 in (S)-TS1, which is absent in (R)-TS1, rendering the (S)-TS1 energetically more favorable. On the other hand, in the monoligated reaction pathway, L9 acts as a bidentate mode in both (S)-TS1′ and (R)-TS1′, with the aryl ring of L9 engaging in coordination interactions with the Pd center. The transition state (S)-TS1′ is 3.2 kcal mol−1 lower in energy than (R)-TS1′, primarily because the stabilizing interaction in (S)-TS1′ is stronger than that in (R)-TS1′.
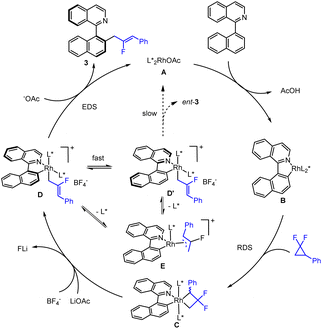
A plausible mechanism for this catalytic system is proposed (Scheme 5). Starting from a Rh(I) acetate bearing electron-rich chiral phosphine ligands (A), C–H bond oxidative addition and subsequent elimination of AcOH gives a rhodacyclic Rh(I) aryl intermediate (B). The alternative mechanism of acetate-assisted C–H activation cannot be ruled out at the current stage. Subsequently, indiscernible C–C bond oxidative addition of racemic DFC takes place to generate a four-membered Rh(III) intermediate (C), which then undergoes β–F elimination to give a cationic Rh(III) allyl species (D). It is proposed that fluoride abstraction by LiOAc plays an important role in ensuring the catalytic activity, driven by formation of insoluble LiF. The resulting Rh(III) allyl is proposed to undergo rapid epimerization of the chiral axis (D and D′),18 and the intermediate D undergoes more rapid C–C reductive elimination, producing product 3 together with Rh(I) for the next cycle. Based on our experimental mechanistic studies, while the Rh(III) allyl monophosphine intermediate (E) may also be reductively eliminated, the enantioselectivity of the product is lower from this manifold. In the catalytic system, the AgBF4 possibly participates in anion exchange with the acetate to give a cationic five-coordinate intermediate D, priming subsequent C–C reductive elimination.
Conclusions
In summary, we have developed a Rh(I)-catalyzed stereoconvergent C–H fluoroalkylation of non-electron-rich arenes in the presence of a chiral monodentate ligand for the efficient construction of axially chiral biaryls. Utilizing racemic gem-difluorocyclopropanes as bifunctional precursors, the reaction proceeds via a Rh(I)/Rh(III) oxidative addition–reductive elimination cycle, enabling C–H activation, β–F elimination, and rapid epimerization of the chiral axis in an intermediate to achieve excellent regio- and enantioselectivity. The LiOAc additive plays a key role as a fluoride scavenger as well as a base to facilitate C–H activation. DFT calculations elucidate the key factors governing stereocontrol, highlighting the precise synergy between C–H activation and allyl introduction. The resulting biaryls were accessed in high regio- and enantioselectivity and display pronounced circularly polarized luminescence (CPL), suggesting their promise as functional components in high-performance chiral optoelectronic materials.Author contributions
X. L. and F. W. conceived and directed the project. J. L. and F. W. performed the experimental studies. X. H. performed DFT studies under the guidance of G. H. F. W., Z.-Q. R., and X. L. wrote the manuscript.Conflicts of interest
The authors declare no competing financial interests.Data availability
CCDC 2497885 contains the supplementary crystallographic data for this paper.19Further details of the experimental procedure, 1H, 19F and 13C NMR spectra, and HPLC and X-ray crystallographic data are available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc08914j.
Acknowledgements
Financial support from the National Natural Science Foundation of China (No. 22371175 and 22471191) and a research fund from Shaanxi Normal University are gratefully acknowledged.Notes and references
- (a) J. Chang, J. Reiner and J. Xie, Chem. Rev., 2005, 105, 4581–4609 CrossRef CAS PubMed; (b) K. Ding, H. Guo, X. Li, Y. Yuan and Y. Wang, Top. Catal., 2005, 35, 105–116 CrossRef CAS; (c) Y.-M. Li, F.-Y. Kwong, W.-Y. Yu and A. S. C. Chan, Coord. Chem. Rev., 2007, 251, 2119–2144 CrossRef CAS; (d) M. P. Carroll and P. J. Guiry, Chem. Soc. Rev., 2014, 43, 819–833 RSC; (e) H. Zhang and F. Shi, Acc. Chem. Res., 2022, 55, 2562–2580 CrossRef CAS PubMed; (f) H. Zhang, T. Li, S. Liu and F. Shi, Angew. Chem., Int. Ed., 2024, 63, e202311053 CrossRef CAS PubMed; (g) A. Gaucherand, E. Yen-Pon, A. Domain, A. Bourhis, J. Rodriguez and D. Bonne, Chem. Soc. Rev., 2024, 53, 11165–11206 RSC.
- (a) J. Clayden, W. J. Moran, P. J. Edwards and S. R. LaPlante, Angew. Chem., Int. Ed., 2009, 48, 6398–6401 CrossRef CAS PubMed; (b) S. R. LaPlante, L. D. Fader, K. R. Fandrick, D. R. Fandrick, O. Hucke, R. Kemper, S. P. F Miller and P. J. Edwards, J. Med. Chem., 2011, 54, 7005–7022 CrossRef CAS PubMed; (c) A. Zask, J. Murphy and G. A. Ellestad, Chirality, 2013, 25, 265–274 CrossRef CAS PubMed; (d) S. T. Toenjes and J. L. Gustafson, Future Med. Chem., 2018, 10, 409–422 CrossRef CAS PubMed; (e) M. Basilaia, M. H. Chen, J. Secka and J. L. Gustafson, Acc. Chem. Res., 2022, 55, 2904–2919 CrossRef CAS PubMed; (f) S. Perreault, J. Chandrasekhar and L. Patel, Acc. Chem. Res., 2022, 55, 2581–2593 CrossRef CAS PubMed.
- (a) J. Chen and Y. Cao, Rapid Commun., 2007, 28, 1714–1742 CrossRef CAS; (b) Q. Li, L. Green, N. Venkataraman, I. Shiyanovskaya, A. Khan, A. Urbas and J. W. Doane, J. Am. Chem. Soc., 2007, 129, 12908–12909 CrossRef CAS PubMed; (c) L. Pu, Acc. Chem. Res., 2012, 45, 150–163 CrossRef CAS PubMed; (d) K. Takaishi, M. Yasui and T. Ema, J. Am. Chem. Soc., 2018, 140, 5334–5338 CrossRef CAS PubMed; (e) D. Zhang, M. Li and C. Chen, Chem. Soc. Rev., 2020, 49, 1331–1343 RSC.
- (a) E. Kumarasamy, R. Raghunathan, M. P. Sibi and J. Sivaguru, Chem. Rev., 2015, 115, 11239–11300 CrossRef CAS PubMed; (b) B. Zilate, A. Castrogiovanni and C. Sparr, ACS Catal., 2018, 8, 2981–2988 CrossRef CAS; (c) A. Link and C. Sparr, Stereoselective Arene Formation, Chem. Soc. Rev., 2018, 47, 3804–3815 RSC; (d) J. K. Cheng, S. Xiang, S. Li, L. Ye and B. Tan, Chem. Rev., 2021, 121, 4805–4902 CrossRef CAS PubMed; (e) G. Mei, W. L. Koay, C. Guan and Y. Lu, Chem, 2022, 8, 1855–1893 CrossRef CAS; (f) W. Qin, Y. Liu and H. Yan, Acc. Chem. Res., 2022, 55, 2780–2795 CrossRef CAS PubMed; (g) J. Feng, L. Xi, C. Lu and R.-R. Liu, Chem. Soc. Rev., 2024, 53, 9560–9581 RSC; (h) S. Xiang, W. Ding, Y. Wang and B. Tan, Nat. Catal., 2024, 7, 483–498 CrossRef CAS.
- For selected reviews, see: (a) G. Liao, T. Zhou, Q. Yao and B. F. Shi, Chem. Commun., 2019, 55, 8514–8523 RSC; (b) M. I. Lapuh, S. Mazeh and T. Besset, ACS Catal., 2020, 10, 12898–12919 CrossRef CAS; (c) C.-X. Liu, W. Zhang, S. Yin, Q. Gu and S.-L. You, J. Am. Chem. Soc., 2021, 143, 14025–14040 CrossRef CAS PubMed; (d) P. Qian, T. Zhou and B. F. Shi, Chem. Commun., 2023, 59, 12669–12684 RSC; (e) C.-X. Liu, S.-Y. Yin, F. Zhao, H. Yang, Z. Feng, Q. Gu and S.-L. You, Chem. Rev., 2023, 123, 10079–10134 CrossRef CAS PubMed; (f) S. Choppin and J. Wencel-Delor, Acc. Chem. Res., 2023, 56, 189–202 CrossRef CAS PubMed; (g) G. Liao and B.-F. Shi, Acc. Chem. Res., 2025, 58, 1562–1579 CrossRef CAS PubMed.
- (a) F. Kakiuchi, P. Le Gendre, A. Yamada, H. Ohtaki and S. Murai, Tetrahedron Asymmetry, 2000, 11, 2647–2651 CrossRef CAS; (b) M. Xiong, Z. Yan, S. Chen, J. Tang, Y. Fan and D. Xing, ACS Catal., 2024, 14, 7243–7255 CrossRef CAS; (c) F. Li, Y. Luo, J. Ren, Q. Yuan, D. Yan and W. B. Zhang, Org. Lett., 2024, 26, 6835–6840 CrossRef CAS PubMed.
- (a) J. Zheng and S.-L. You, Angew. Chem., Int. Ed., 2014, 53, 13244–13247 CrossRef CAS PubMed; (b) J. Zheng, W. Cui, C. Zheng and S.-L. You, J. Am. Chem. Soc., 2016, 138, 5242–5245 CrossRef CAS PubMed; (c) S. X. Li, Y. N. Ma and S. D. Yang, Org. Lett., 2017, 19, 1842–1845 CrossRef CAS PubMed; (d) J. Luo, T. Zhang, L. Wang, G. Liao, Q.-J. Yao, Y.-J. Wu, B. Zhan, Y. Lan, X. Lin and B.-F. Shi, Angew. Chem., Int. Ed., 2019, 58, 6708–6712 CrossRef CAS PubMed; (e) B. Zhan, L. Wang, J. Luo, X. Lin and B. F Shi, Angew. Chem., Int. Ed., 2020, 59, 3568–3572 CrossRef CAS PubMed; (f) P. Vázquez-Domínguez, A. Romero Arenas, R. Fernández, J. M. Lassaletta and A. Ros, ACS Catal., 2023, 13, 42–48 CrossRef; (g) Q. Zhou, S.-Y. Yin, D. Zheng, W. Zhang, S. Zhang, Q. Gu and S.-L. You, Synlett, 2023, 34(8), 1442–1446 CAS.
- (a) A. Ros, B. Estepa, P. Ramírez-López, E. Á lvarez, R. Fernández and J. M. Lassaletta, J. Am. Chem. Soc., 2013, 135, 15730–15733 CrossRef CAS PubMed; (b) Q. Wang, Z. Cai, C. Liu, Q. Gu and S.-L. You, J. Am. Chem. Soc., 2019, 141, 9504–9510 CrossRef CAS PubMed; (c) M. Tian, D. Bai, G. Zheng, J. Chang and X. Li, J. Am. Chem. Soc., 2019, 141, 9527–9532 CrossRef CAS PubMed; (d) Q. Wang, W. Zhang, H. Song, J. Wang, C. Zheng, Q. Gu and S.-L. You, J. Am. Chem. Soc., 2020, 142, 15678–15685 CrossRef CAS PubMed; (e) Q. Nguyen, S. Guo, T. Royal and O. Baudoin, J. Am. Chem. Soc., 2020, 142, 2161–2167 CrossRef CAS PubMed; (f) Q. Wang, W. Zhang, C. Zheng, Q. Gu and S.-L. You, J. Am. Chem. Soc., 2021, 143, 114–120 CrossRef CAS PubMed.
- (a) G. Liao, H. Chen, Q. Yao, Y. Xia, J. Luo and B.-F. Shi, Angew. Chem., Int. Ed., 2018, 57, 17151–17155 CrossRef CAS PubMed; (b) G. Liao, T. Zhang, L. Jin, B. Wang, C. Xu, Y. Lan, Y. Zhao and B.-F. Shi, Angew. Chem., Int. Ed., 2022, 61, e202115221 CrossRef CAS PubMed.
- Reviews on C–H bond allylation: N. K. Mishra, S. Sharma, J. Park, S. Han and I. S. Kim, Chem, 2021, 7, 555–605 Search PubMed.
- Asymmetric allylation of acidic C–H sites: (a) L. Fan, S. Luo, S. Chen, T. Wang, P. Wang and L. Gong, Angew. Chem., Int. Ed., 2019, 58, 16806–16810 CrossRef CAS PubMed; (b) T. Wang, L. Zhu, S. Luo, Z. Nong, P. Wang and L. Gong, J. Am. Chem. Soc., 2021, 143, 20454–20461 CrossRef CAS PubMed; (c) Z. Nong, L. Zhu, T. Wang, L. Fan, P. Wang and L. Gong, Nat. Synth., 2022, 1, 487–496 CrossRef CAS; (d) C.-X. Zhuo and S.-L. You, Acc. Chem. Res., 2014, 47, 2558–2573 CrossRef CAS PubMed; (e) B. M. Trost and M. U. Frederiksen, Angew. Chem., Int. Ed., 2005, 44, 308–310 CrossRef CAS PubMed.
- Allylation via olefin insertion and β–H elimination: (a) J. Kwak, Y. Ohk, Y. Jung and S. Chang, J. Am. Chem. Soc., 2012, 134, 17778–17788 CrossRef CAS PubMed; (b) S. Wang and N. Cramer, Angew. Chem., Int. Ed., 2019, 58, 2514–2518 CrossRef CAS PubMed; (c) R. Mi, G. Zheng, Z. Qi and X. Li, Angew. Chem., Int. Ed., 2019, 58, 17666–17670 CrossRef CAS PubMed; (d) X. Yang, G. Zheng and X. Li, Angew. Chem., Int. Ed., 2019, 58, 322–326 CrossRef CAS PubMed; (e) K. Ozols, S. Onodera, Ł. Wozniak and N. Cramer, Angew. Chem., Int. Ed., 2021, 60, 655–659 CrossRef CAS PubMed; (f) Y. Zheng, W. Zhang, Q. Gu, C. Zheng and S.-L. You, Nat. Commun., 2023, 14, 1094 CrossRef CAS PubMed.
- (a) S. Fan, F. Chen and X. Zhang, Angew. Chem., Int. Ed., 2011, 50, 5918–5923 CrossRef CAS PubMed; (b) S. Y. Lee and J. F. Hartwig, J. Am. Chem. Soc., 2016, 138, 15278–15284 CrossRef CAS PubMed; (c) W. Xie and S. Chang, Angew. Chem., Int. Ed., 2016, 55, 1876–1880 CrossRef CAS PubMed; (d) K. Liu, T. Li, D. Liu, W. Li, J. Han, C. Zhu and J. Xie, Sci. China Chem., 2021, 64, 1959–1963 CrossRef.
- (a) M. Fedorynski, Chem. Rev., 2003, 103, 1099–1132 CrossRef CAS PubMed; (b) F. Wang, T. Luo, J. Hu, Y. Wang, H. S. Krishnan, P. V. Jog, S. K. Ganesh, G. K. S. Prakash and G. A. Olah, Angew. Chem., Int. Ed., 2011, 50, 7153–7157 CrossRef CAS PubMed; (c) W. R. Dolbier and M. A. Battiste, Chem. Rev., 2003, 103, 1071–1098 CrossRef CAS PubMed; (d) Y. Zeng, Z.-T. Jiang and Y. Xia, Chem. Commun., 2024, 60, 3764–3773 RSC.
- (a) E.-A. M. Ahmed, A. M. Y. Suliman, T. Gong and Y. Fu, Org. Lett., 2019, 21, 5645–5649 CrossRef CAS PubMed; (b) Y. Zeng, H. Yang, J. Du, Q. Huang, G. Huang and Y. Xia, Chem. Sci., 2022, 13, 12419–12425 RSC; (c) Y. Ai, H. Yang, C. Duan, X. Li and S. Yu, Org. Lett., 2022, 24, 5051–5055 CrossRef CAS PubMed; (d) Z. Jiang, Z. Chen, Y. Zeng, J. Shi and Y. Xia, Org. Lett., 2022, 24, 6176–6181 CrossRef CAS PubMed; (e) J. Sun, H. Ye, F. Sun, Y. Pan, X. Zhu, X. Wu, Org. Lett., 2023, 25, 5220–5225 Search PubMed; (f) H. Qian, H. D. Nguyen, L. Lv, S. Chen and Z. Li, Angew. Chem., Int. Ed., 2023, 62, e202303271 CrossRef CAS PubMed.
- (a) Z. Jiang, J. Huang, Y. Zeng, F. Hu and Y. Xia, Angew. Chem., Int. Ed., 2021, 60, 10626–10631 CrossRef CAS PubMed; (b) Z. Fu, J. Zhu, S. Guo and A. Lin, Chem. Commun., 2021, 57, 1262–1265 RSC; (c) Z. Jiang, Y. Zeng and Y. Xia, Synlett, 2021, 32, 1675–1682 CrossRef CAS; (d) X. Wu, Y. Zeng, Z. Jiang, Y. Zhu, L. Xie and Y. Xia, Org. Lett., 2022, 24, 8429–8434 CrossRef CAS PubMed; (e) X. Wu, X. Song and Y. Xia, Adv. Sci., 2024, 11, 2401243 CrossRef CAS PubMed; (f) Y. Zeng, H. Gao, Z. Jiang, Y. Zhu, J. Chen, H. Zhang, G. Lu and Y. Xia, Nat. Commun., 2024, 15, 4317 CrossRef CAS PubMed.
- (a) H. Yang, Y. Zeng, X. Song, L. Che, Z. Jiang, G. Lu and Y. Xia, Angew. Chem., Int. Ed., 2024, 63, e202403602 CrossRef CAS PubMed; (b) Z. Su, B. Tan, H. He, K. Chen, S. Chen, H. Lei, T. Chen, S. Ni and Z. Li, Angew. Chem., Int. Ed., 2024, 63, e202402038 CrossRef CAS PubMed; (c) Z. Yang, Y. Gong, Q. Gu and S.-L. You, ACS Catal., 2025, 15, 4287–4293 CrossRef CAS.
- (a) J. A. Carmona, V. Hornillos, P. Ramírez-López, A. Ros, J. Iglesias-Sigüenza, E. Gómez-Bengoa, R. Fernández and J. M. Lassaletta, J. Am. Chem. Soc., 2018, 140, 11067–11075 CrossRef CAS PubMed; (b) A. Romero-Arenas, V. Hornillos, J. Iglesias-Sigüenza, R. Fernández, J. López-Serran, A. Ros and J. M. Lassaletta, J. Am. Chem. Soc., 2020, 142, 2628–2639 CrossRef CAS PubMed.
- CCDC 2497885: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pv7yr.
| This journal is © The Royal Society of Chemistry 2026 |