 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Selective NH3-to-N2H4 conversion electrocatalysed by ruthenium(II)-cymene complexes
Xi
Zhang
a,
Shan
Zhao
a,
Chen
Zhou
a,
Guo
Chen
 b,
Liru
Cao
c,
Jian
Lin
b,
Liru
Cao
c,
Jian
Lin
 c,
Chen
Tang
a,
Zhi-Yan
Liu
a,
Piao
He
c,
Chen
Tang
a,
Zhi-Yan
Liu
a,
Piao
He
 a and
Xiao-Yi
Yi
a and
Xiao-Yi
Yi
 *a
*a
aCollege of Chemistry and Chemical Engineering, Central South University, Changsha, Hunan 410083, P. R. China. E-mail: xyyi@csu.edu.cn
bSchool of Chemistry and Chemical Engineering, Southwest University, Chongqing, 400715, P. R. China
cCAS Key Laboratory of Science and Technology on Applied Catalysis, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian, 116023, P. R. China
First published on 13th January 2026
Abstract
A series of ruthenium(II)-cymene complexes [(η6-p-cymene)Ru(pp)Cl] (1–4) and corresponding NH3-ligated complexes [(η6-p-cymene)Ru(pp)(NH3)]PF6 ([1-NH3]PF6 to [4-NH3]PF6), where cymene = 4-isopropyltoluene and pp− = pyridylpyrrole ligand, have been designed and synthesized. Structural modifications of pp− ligands are accomplished through the use of an increasing number of electron-donating methyl groups on the pyrrole unit. Solid-state structural analysis shows that these complexes have a typical piano-stool structure. Electrochemical studies of these complexes illustrate that the introduction of a methyl group to the pp− ligand can greatly decrease the oxidation potential of RuIII/II from 0.49 V vs. Cp2Fe+/0 for [1-NH3]PF6 to 0.16 V vs. Cp2Fe+/0 for [4-NH3]PF6. Controlled potential coulometry experiments show that these complexes exhibit selective catalysis for the oxidation of NH3 to N2H4 with a turnover number of up to 453.2 at Eapp 0.8 V vs. Cp2Fe+/0 for the [4-NH3]PF6 complex. Kinetic and theoretical thermodynamic studies show that the pathway of bimolecular coupling of RuII-aminyl species and the pathway of ammonia nucleophilic attack of RuIV-imide (generated from the disproportionation of RuIII-amide) are involved in N–N formation.
To meet the challenges of a large-scale energy crisis and environmental pollution, energy-rich H2 of a green and sustainable nature has attracted much interest as an alternative energy source, although storage and distribution of liquid H2 still suffer from harsh conditions along with a lack of infrastructure. Ammonia (NH3) is a good candidate as a hydrogen energy carrier, and offers approximately 1.7 times the energy density of liquid H2.1–4 The NH3 industry has not only seen applications worldwide but also has widespread facilities for storage, transport and handing. The classical catalytic cracking reaction for NH3-to-H2 conversion (eqn (1)) requires a precious metal catalyst and high temperature, which are of relatively high cost. Moreover, the generated N2 product is directly discharged into the air, resulting in low atomic utilization and relatively poor economy in NH3-to-H2 conversion.
| 2NH3(g) → N2(g) + 3H2(g) ΔGθ = 7.9 kcal mol−1 | (1) |
| 2NH3(g) → N2H4(l) + H2(g) ΔGθ = 43.6 kcal mol−1 | (2) |
| N2H4(l) → N2(g) + 2H2(g) ΔGθ = −35.7 kcal mol−1 | (3) |
Selective electrocatalytic conversion of NH3 into N2H4 and H2 (NH3-to-N2H4 conversion, eqn (2)) seems more appealing than NH3-to-H2 conversion due to the advantages of not only generating H2, but also simultaneously producing high value-added N2H4 (the price of anhydrous hydrazine is about 58![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 USD t−1). However, this route is a thermodynamically demanding process (ΔGθ = 43.6 kcal mol−1), and needs to overcome the competitive reaction of spontaneous dehydrogenation of N2H4 to N2 (eqn (3), ΔGθ = −35.7 kcal mol−1). Hence, highly efficient and selective NH3-to-N2H4 conversion is appealing, but remains a huge scientific challenge.5
000 USD t−1). However, this route is a thermodynamically demanding process (ΔGθ = 43.6 kcal mol−1), and needs to overcome the competitive reaction of spontaneous dehydrogenation of N2H4 to N2 (eqn (3), ΔGθ = −35.7 kcal mol−1). Hence, highly efficient and selective NH3-to-N2H4 conversion is appealing, but remains a huge scientific challenge.5
Molecular catalysts can offer several advantages over their heterogeneous counterparts, such as controllable structure, convenient characterization, and well-defined active site nature, which allow for mechanistic studies to elucidate the factors controlling the catalytic activity and selectivity. Since the seminal work by Hamann and Smith III on the electro-oxidation of NH3 to N2 catalyzed by [(trpy)(bpyNMe2)Ru(NH3)](PF6)2 (trpy = 2,2′:6′,2″-terpyridine, bpyNMe2 = 4,4′-bis(dimethylamino)-2,2′-bipyridine),6 significant progress has been made in the development of molecular catalysts for ammonia oxidation.7–22 Nevertheless, the reported catalytic systems are mostly concerned with the oxidation of NH3 to N2, and there are few reports on the selective catalytic conversion of NH3 into N2H4. In 2023, we demonstrated that [Ru(κ2-N,N′-dpp)(bpy)(dmso)(NH3)]PF6 (Hdpp = 2,5-di(pyridin-2-yl)-1H-pyrrole)15a can catalyze the electrocatalytic conversion of NH3 into N2H4 with unprecedentedly high selectivity (over 97.9%) and turnover frequency (238.9 h−1). Although a similar ligated-N2H4 intermediate and a similar N–N formation pathway—such as either bimolecular coupling of a metal-imide15,18,19,20a or ammonia nucleophilic attack of a metal-imide6,10,17,22—are involved, this catalyst is in sharp contrast with conventional ones that usually generate N2 as the major N–N coupling product with relatively low turnover frequencies. N2H4/N2 selectivity is usually represented by a branch from a ligated-N2H4 intermediate,14,15a which could directly release N2H4 or continue to be over-oxidized to release N2. The release of N2H4 from a ligated-N2H4 intermediate to restart the catalytic cycle is one of the key issues for the selective oxidation of NH3 to N2H4. Thus, rational design of the ancillary ligand backbone is still a desirable strategy for developing highly efficient and selective catalysts for NH3-to-N2H4 conversion.
We have long been interested in the study of metal complexes based on the pyridylpyrrole (pp−) ligand, which is structurally analogous to bipyridine.23 Notwithstanding the similar structure and coordination properties, the π-donation from the pyrrolyl group in the pp− ligand increases the energy of the metal-based LUMO in the M–N2H4 intermediate, thus weakening the M–N2H4 bond and facilitating the release of N2H4.24 In addition, the negative charge of the pyrrolyl unit not only lowers the overpotential of the metal complex but also reduces the overall positive charge of the reactive intermediate, which is a major cause of instability in the intermediates during the AO catalytic cycle.23 Herein, we design an ancillary pp− ligand using an increasing number of electron-donating methyl groups on the pyrrole unit to regulate the electronic structure of the metal complex. Corresponding ruthenium(II)-cymene complexes [(η6-p-cymene)Ru(pp)Cl] (where pp− is HL1 = 2-(1H-pyrrol-2-yl)pyridine (1), HL2 = 2-(4-methyl-1H-pyrrol-2-yl)pyridine (2), HL3 = 2-(3,5-dimethyl-1H-pyrrol-2-yl)pyridine (3), and HL4 = 2-(3,4,5-trimethyl-1H-pyrrol-2-yl)pyridine (4)) and corresponding NH3-ligated complexes [(η6-p-cymene)Ru(pp)(NH3)]PF6 ([1-NH3]PF6 to [4-NH3]PF6) are reported. Their selective catalysis for NH3-to-N2H4 conversion and the catalytic mechanism are also presented.
As shown in Fig. 1a, complexes 1–4 are synthesized by treatment of dimeric precursor of [(η6-p-cymene)RuCl2]2 and a deprotonated pyridylpyrrole ligand in CH2Cl2 at 0 °C in moderate yield (∼50%). The corresponding NH3-ligated complexes [(η6-p-cymene)Ru(pp)(NH3)]PF6 ([1-NH3]PF6 to [4-NH3]PF6) are synthesized in over 60% yield by treatment of 1–4 with one equiv. of AgPF6 in CH3CN followed by bubbling NH3 gas. These complexes are stable in common organic solvents, and are fully characterized by ESI-MS, NMR and IR spectroscopy (Fig. S6–S29). Compared to 1–4, the 1H NMR spectra of [1-NH3]PF6 to [4-NH3]PF6 show a newly added broad single peak at ∼2.0 ppm due to the ligated-NH3. Similarly, the IR spectra of [1-NH3]PF6 to [4-NH3]PF6 show an additional band at ∼3340–3350 cm−1 due to N–H stretching, also indicating that NH3 binds to the Ru center. The solid-state structures of 1–4 and [2-NH3]PF6 are shown in Fig. 1b. The crystallographic data and selected bond distances and angles are listed in Tables S1–S6. These complexes exhibit a typical piano-stool structure with one pp−, one cymene and one Cl− (or NH3) ligand coordinating to the ruthenium center. The bond distance of Ru–Npyrrole (2.042(10)–2.074(3) Å) is slightly shorter than that of Ru–Npyridine (2.101(2)–2.113(3) Å), mainly due to the electrostatic interaction between the Ru atom and the anionic pyrrolide N atom. In [2-NH3]PF6, NH3 coordinates to the Ru center with a Ru–N bond distance of 2.138(12) Å, which is similar to that of other NH3-ligated Ru(II) complexes.
The electrochemical behavior of the title complexes in CH3CN is investigated by cyclic voltammetry (CV) and differential pulse voltammetry (DPV) using a AgCl/Ag electrode in a saturated KCl solution as a reference electrode. Unless otherwise specified, all potentials are converted into E1/2vs. Cp2Fe+/0 in CH3CN by adding −0.43 V to the measured potential.
The electrochemical behavior of 1–4 is shown in Fig. 2a. The first oxidation wave is assigned to ruthenium center oxidation (RuII → RuIII). The electro-donating nature of the pp− ligand significantly influences the redox potential of these complexes, leading to a decreasing RuIII/II reduction potential from 0.37 V for 1 to 0.04 V for 4 with an increase in the number of electron-donating methyl groups on the pyrrole unit. The redox potentials of the second oxidation wave at ∼1.2 V and third oxidation wave at ∼1.6 V (see the DPV curves in Fig. 2a) are independent of the methyl substituted pp− ligand. These could clearly be assigned to Cl− and cymene ligand oxidation events, respectively.25,26Fig. 2b shows that [1-NH3]PF6 to [4-NH3]PF6 exhibit two oxidation waves. The first one (0.49 V, 0.36 V, 0.23 V and 0.16 V for [1-NH3]PF6 to [4-NH3]PF6, respectively) is attributed to their metal center oxidation from RuII to RuIII. Compared to 1–4, the RuIII/II redox potentials of [1-NH3]PF6 to [3-NH3]PF6 are anodically shifted by ∼0.12 V, mainly due to the π-donating capability of the Cl− ligand in the former. Upon expanding the voltage window, unlike the second oxidation peak corresponding to RuIV/III reported in our previous literature,15 these complexes exhibited no additional metal-centered oxidation waves except for the second oxidation peak assigned to cymene ligand oxidation at ∼1.6 V.26 However, a weak new wave marked by a rhombus appears at 0.75–0.88 V when scanning in the cathodic direction (Fig. 2c). This suggests that a disproportionation of RuIII species possibly occurs to give RuII and RuIV species, and then RuIV is reduced to RuIII on the reverse scan. Taking [1-NH3]PF6 as an example, its RuIII intermediate formed by 1e− oxidation is proposed to undergo rapid disproportionation to RuII [1-NH3]+ and a RuIV imido intermediate and simultaneously reach equilibrium. Thus, the wave at 0.88 V on the reverse scan can be assigned to the reduction of RuIV species to RuIII species of [1-NH3]+. To further confirm the 1e− oxidation to form a RuIII intermediate, CV was employed, with ferrocene added as the internal reference at an equimolar concentration to [1-NH3]+. As shown in Fig. S30, the diffusion coefficient of complex [1-NH3]+ (1.58 × 10−5 cm2 s−1) is very close to that of ferrocene (1.37 × 10−5 cm2 s−1), and the peak areas of [1-NH3]+ and ferrocene are almost the same in their respective CV plots, preliminarily indicating that the first oxidation wave of [1-NH3]+ involved only one electron transfer. The more critical evidence in the reversible charge transfer process is the slope analysis of E vs. lg[(Il − I)/I] (where E is the potential and Il is the limiting current).27 As shown in Fig. S31, a linear relationship was observed between E and lg[(Ip − I)/I] (where Ip is the peak current, used in place of Il due to the irreversible oxidation wave of [1-NH3]+), with a slope m = 2.3RT/nF (where R is the ideal gas constant, T = 298.15 K, and F = 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485C mol−1). The fitted slope from the experimental data was 0.84, yielding an electron transfer number n ≈ 0.84. These results collectively confirm that only a single electron transfer occurs at the electrode surface, followed by a redox disproportionation chemical step.
485C mol−1). The fitted slope from the experimental data was 0.84, yielding an electron transfer number n ≈ 0.84. These results collectively confirm that only a single electron transfer occurs at the electrode surface, followed by a redox disproportionation chemical step.
The electrochemical behavior of [1-NH3]PF6 to [4-NH3]PF6 in the presence of 0.05 M NH3 is shown in Fig. 3. Complexes [1-NH3]PF6 and [2-NH3]PF6 display a slightly increased catalytic current (icat) over RuIII species with low onset potentials of 0.15 V and 0.10 V, respectively (inset figures of Fig. 3). Although RuIV species of the title complexes are not clearly observed in the CV studies, the catalytic current increases sharply at potentials over 0.6 V. For complexes [3-NH3]PF6 and [4-NH3]PF6, a catalytic current only appears at a high potential, and there is no catalytic current over RuIII species. Anodic currents (ip) of the RuIII/II redox couple and the catalytic current of [1-NH3]PF6 to [4-NH3]PF6 in the presence of 0.05 M NH3 increase linearly with the square root of the scan rate (Fig. S33), indicating diffusion-controlled behavior of these ruthenium catalysts under test conditions. The rate constant (kcat) and maximum turnover frequency (TOFmax) of [1-NH3]PF6 to [4-NH3]PF6 for ammonia oxidation are estimated. The diffusion coefficients (DRu) and rate constants (kcat) of [1-NH3]PF6 to [4-NH3]PF6 are 1.58 × 10−5 cm2 s−1, 1.71 × 10−5 cm2 s−1, 1.82 × 10−5 cm2 s−1, and 1.94 × 10−5 cm2 s−1; and 4.5 s−1, 4.9 s−1, 5.2 s−1, and 5.4 s−1, respectively, which are determined from the dependence of ip and ν1/2 based on the Randles–Sevcik relation (eqn (4)) and linear fitting of icat/ip with ν−1/2 based on eqn (5), respectively. The TOFmax values at a scan rate of 0.1 V s−1 based on eqn (6) are estimated to be 4.92 × 10−2 s−1, 5.48 × 10−2 s−1, 5.81 × 10−2 s−1, and 6.29 × 10−2 s−1.
 | (4) |
 | (5) |
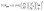 | (6) |
To confirm the electrocatalysis of [1-NH3]PF6 to [4-NH3]PF6 for NH3-to-N2H4 conversion, controlled potential coulometry (CPC) experiments are conducted in a conventional sealed three-electrode cell with 2.0 M NH3, 0.01 mM catalyst and 0.1 M [n-Bu4N]PF6 electrolyte in dried CH3CN under an Ar atmosphere. Carbon cloth (1 cm2), a Pt plate (1 cm2) and Ag/AgCl in saturated KCl aqueous solution are used as the working electrode, counter electrode and reference electrode, respectively. Possible gas products (such as N2, H2) are determined by the GC method (Fig. S1). Possible products in the electrolyte (such as N2H4, NO2−, NO3−) are quantitatively analyzed via chemical methods (Fig. S2–S5).28–30
To obtain general information for the CPC experiments, first, control experiments are employed. As shown in Table S7, only negligible N2H4 and N2 are generated at low applied potential (such as Eapp 0.39 V) for 24 h. However, 21.6 µmol of N2H4 and 0.5 µmol of N2 are produced at Eapp 0.8 V for 2 h, indicating that the bare electrode causes slow ammonia oxidation at relatively high potential. Subsequently, the complex [2-NH3]PF6 is chosen to investigate the relationship between catalytic performance over time and applied potential (Table 1 and Fig. S38, S39, Tables S8, S9). The amounts of N2H4 as an absolutely dominant anodic product and H2 as a cathodic product increase with an increase of the applied potential from 0.2 V to 1.0 V (Fig. S38). Other anode products of NO2− and NO3− are not determined due to the absence of oxygen sources in the catalytic system. Holding the applied potential at 0.8 V, the generation of N2H4 and H2 keeps increasing over time, however, the turnover frequency (TOF) and Faraday efficiency (FE) of N2H4 formation continuously decrease (Fig. S39 and Table S9). The loss of TOF and FE for long-term electrolysis might be caused by over-oxidation of the produced H2 in the sealed CPC cell. A decrease in icat under a H2 atmosphere indicating the current consumption of H2 oxidation cannot be ignored (Fig. S40). The icat remains almost unchanged after 100 consecutive CV cycles or electrolysis for 2 h, indicating that these Ru catalysts under catalytic conditions have satisfactory stability (Fig. S41 and S42). A thoroughly rinsed electrode after catalysis shows no ruthenium deposition and no catalytic activity, indicating solution-based electrocatalysis (Fig. S43–4S5 and Table S7). Notably, N2H4 is prone to undergo either catalytic oxidation or disproportionation decomposition to yield N2. In contrast, complexes [1-NH3]PF6–[4-NH3]PF6 exhibit good selectivity toward N2H4 formation. CV measurements of [1-NH3]PF6 in MeCN containing N2H4 confirm that they are inactive toward N2H4 oxidation (Fig. S54).
| Entry | Cat | [NH3] (M) | E app | Time (h) | TONH2/nH2(µmol) | TOFH2 (h−1) | TONN2H4/nN2H4(µmol) | TOFN2H4 (h−1) | TONN2/nN2(µmol) | TOFN2 (h−1) | FEN2H4b (%) | S N2H4 (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| a Conditions: carbon cloth (1 cm2) as the working electrode, a platinum plate (1 cm2) as the counter electrode, AgCl/Ag in saturated KCl aqueous solution as the reference electrode, [cat] = 0.01 mM. The background is subtracted to obtain the number of moles of the products H2, N2 and N2H4. b FEN2H4 = 2 nN2H4F/it × 100%. c S N2H4 = nN2H4/(nN2 + nN2H4) × 100%. | ||||||||||||
| 1 | [1-NH3]PF6 | 2.0 | 0.39 | 24 | 17.2 | 0.7 | 16.5 | 0.7 | Trace | — | 86.3 | 100 |
| 13.7 | 13.2 | |||||||||||
| 2 | [2-NH3]PF6 | 2.0 | 0.26 | 24 | 11.5 | 0.5 | 11.0 | 0.5 | Trace | — | 89.7 | 100 |
| 9.2 | 8.8 | |||||||||||
| 3 | [3-NH3]PF6 | 2.0 | 0.13 | 24 | Trace | — | Trace | — | Trace | — | — | — |
| 4 | [4-NH3]PF6 | 2.0 | 0.06 | 24 | Trace | — | Trace | — | Trace | — | — | — |
| 5 | [1-NH3]PF6 | 2.0 | 0.8 | 2 | 377.3 | 188.7 | 356.2 | 178.1 | 5.3 | 2.7 | 88.2 | 98.6 |
| 301.9 | 284.9 | 4.2 | ||||||||||
| 6 | [2-NH3]PF6 | 2.0 | 0.8 | 2 | 403.5 | 201.8 | 393.9 | 197.0 | 2.2 | 1.1 | 90.8 | 99.4 |
| 322.8 | 315.2 | 1.8 | ||||||||||
| 7 | [3-NH3]PF6 | 2.0 | 0.8 | 2 | 430.1 | 215.1 | 418.2 | 209.1 | 4.3 | 2.2 | 93.5 | 99.0 |
| 344.1 | 334.5 | 3.4 | ||||||||||
| 8 | [4-NH3]PF6 | 2.0 | 0.8 | 2 | 461.6 | 230.8 | 453.2 | 230.8 | 2.4 | 1.2 | 91.9 | 99.5 |
| 369.3 | 362.5 | 1.9 | ||||||||||
| 9 | [4-NH3]PF6 | 0.05 | 0.8 | 2 | 156.6 | 78.3 | 151.0 | 75.5 | 1.5 | 0.8 | 87.6 | 99.0 |
| 125.3 | 120.8 | 1.2 | ||||||||||
| 10 | 5 (ref. 15a) | 2.0 | 1.0 | 24 | 5870 | 244.6 | 5735 | 238.9 | 43.9 | 1.8 | 36.6 | 97.8 |
| 2348 | 2293.8 | 17.56 | ||||||||||
| 11 | 6 (ref. 15b) | 2.0 | 1.0 | 2 | 801.5 | 400.8 | 721.5 | 360.8 | 13.7 | 6.9 | 99.2 | 98.1 |
| 561.1 | 505.1 | 9.6 | ||||||||||
| 12 | 7 (ref. 15c) | 2.0 | 1.0 | 2 | 844.2 | 422.1 | 786.8 | 393.4 | 19.8 | 9.9 | 99.3 | 93.1 |
| 616.2 | 566.4 | 14.3 | ||||||||||
To clarify the catalytic performance of the catalysts at the initial stage of RuIII species formation, we selected a potential 0.1 V lower than the RuIII/II potential of the catalysts (0.39 V for [1-NH3]PF6, 0.26 V for [2-NH3]PF6, 0.13 V for [3-NH3]PF6, and 0.06 V for [4-NH3]PF6) as the applied potential in the CPC experiment. N2H4 and almost equiv. molar of H2 are produced in the [1-NH3]PF6 and [2-NH3]PF6 catalyst systems, and the generation of N2 could be negligible (entries 1 and 2). From comparison to the result of the control CPC experiments at Eapp 0.39 V, this indicates that the catalytic activity originates from the catalyst but not the bare electrode. The catalytic conversion of NH3 to N2H4 achieves ∼100% selectivity. This behavior also aligns with our earlier ruthenium catalysts containing pyridylpyrrole operated at low applied potentials via bimolecular N–N coupling of RuIII-amide to form N2H4.15,18,19,20a As expected in the CV studies, no ammonia oxidation products (N2H4 or N2) are generated in the [3-NH3]PF6 and [4-NH3]PF6 catalyst systems, only over RuIII species at low potential (entries 3 and 4). Next, we carried out CPC experiments at higher applied potential Eapp of 0.80 V. Correspondingly, the catalytic efficiency of [1-NH3]PF6 to [4-NH3]PF6 significantly increased. As shown in Table 1, entries 5–8, N2H4 is still determined as the dominant anodic product with a yield range from 284.9 µmol to 362.5 µmol. The turnover frequency (TOF) of N2H4 formation reaches 178.1 h−1 for [1-NH3]PF6, 197.0 h−1 for [2-NH3]PF6, 209.1 h−1 for [3-NH3]PF6 and 226.6 h−1 for [1-NH3]PF6. The selectivity and Faraday efficiency based on N2H4 formation are over 98.6% and 88.2%, respectively. In this work, [4-NH3]PF6 also shows excellent selectivity and high catalytic activity for the oxidation of low-concentration NH3 to generate N2H4. We carried out CPC experiments in a low-concentration NH3 solution (0.05 M) at the same potential. After 2 h, catalytic amounts of H2 (125.3 µmol), N2H4 (120.8 µmol) and N2 (1.2 µmol) were generated. This result breaks through the limitation of conventional catalytic systems that rely on high ammonia concentrations to achieve high selectivity (entry 9).
The CV and CPC experiments illustrate that complexes [1-NH3]PF6 and [2-NH3]PF6 for RuIII species and [1-NH3]PF6 to [4-NH3]PF6 for RuIV species readily undergo ammonia oxidation to generate N2H4. To explore the detailed mechanism of these complexes, we carried out theoretical calculations on the activation energy of each step in the catalytic procedure for the [1-NH3]PF6 and [4-NH3]PF6 catalysts. A summary of the proposed mechanism and alternative pathway is shown in Fig. 4.
As shown in Fig. 4a, complexes [1-NH3]+ and [4-NH3]+ are first oxidized to RuIII-amide 2[1-I]+ and 2[4-I]+ (the left superscript shows the spin state) via a 1e−/H+ transfer process with ΔG of 8.0 and 6.3 kcal mol−1, respectively. Spin density distribution calculations (Fig. S55) reveal that the spin densities of 2[1-I]+ and 2[4-I]+ are primarily located on the Ru center (0.50 and 0.47 e−) and N atom of NH2 (0.50 and 0.50 e−), indicating that the RuIII-amide species has RuII-aminyl character via inter-conversion of RuIII–NH2− ![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif) RuII–NH2͘͘˙. The RuII-aminyl species is an active intermediate and can readily generate N2H4via a bimolecular RuII–NH2− coupling reaction.15,18,19,20a For the [1-NH3]PF6 catalyst, theoretical calculations elucidate the coupling process between two 2[1-I]+ molecules, which proceeds through transition states 1[1-III]2+ and 1[1-IV]2+ to ultimately form the thermodynamically stable hydrazine-bridged bimetallic complex 1[1-V]2+. The catalytic cycle is completed by a mildly endothermic hydrazine dissociation process, with an overall energy barrier of 12.3 kcal mol−1.
RuII–NH2͘͘˙. The RuII-aminyl species is an active intermediate and can readily generate N2H4via a bimolecular RuII–NH2− coupling reaction.15,18,19,20a For the [1-NH3]PF6 catalyst, theoretical calculations elucidate the coupling process between two 2[1-I]+ molecules, which proceeds through transition states 1[1-III]2+ and 1[1-IV]2+ to ultimately form the thermodynamically stable hydrazine-bridged bimetallic complex 1[1-V]2+. The catalytic cycle is completed by a mildly endothermic hydrazine dissociation process, with an overall energy barrier of 12.3 kcal mol−1.
This bimolecular coupling pathway is supported by the CPC experiment of [1-NH3]PF6 at low applied potential (0.39 V), where only N2H4 as an ammonia oxidation product is observed. In addition, its catalytic current over RuIII species is independent of the increase of [NH3] (10–50 mM), also indicating that ammonia seems not to be involved in N2H4 formation (Fig. S35) only over RuIII species. A similar bimolecular coupling pathway to form N2H4 over RuIII species is observed in our reported ruthenium catalysts,15 and has recently been confirmed in the [(trpy)(bpyNMe2)Ru(NH3)](PF6)2 catalyst system.14 Interestingly, in the bimolecular coupling pathway, the energy barrier for the conversion of 1[4-III]2+ to 1[4-V]2+ (17.3 kcal mol−1) is only 5 kcal mol−1 higher than that for 1[1-III]2+ to 1[1-V]2+. However, CV and CPC studies on the structurally analogous complex [4-NH3]PF6 demonstrate that its RuIII species (2[4-I]+) cannot trigger ammonia oxidation. This suggests that the difference in energy barrier does not play a decisive role in determining whether N–N bond formation occurs. From a kinetic perspective, an increased number of methyl groups (three methyl groups in [4-NH3]PF6) enhances steric hindrance, which disfavors the bimolecular coupling pathway and appears to be the dominant controlling factor.
Following the 1e−/H+ transfer process, the subsequent second 1e−/H+ oxidation to form RuIV-imide species 1[1-II]+ and 1[4-II]+ is less endergonic with ΔG of 5.3 and 1.5 kcal mol−1, respectively. Compared to the first oxidation step, the significantly reduced ΔG values indicate thermodynamically more favorable formation of RuIV-imide species. Analysis of the combined free energy changes reveals that the redox disproportionation of RuIII-amide to generate RuII-ammine and RuIV-imide exhibits ΔG values of −2.7 and −4.8 kcal mol−1, respectively, demonstrating a highly spontaneous thermodynamic process. Therefore, the redox disproportionation pathway to form RuIV-imide species is thermodynamically preferred over direct oxidation of RuIII-amide, explaining the absence of an observable RuIV/III redox couple under CV test conditions. Notably, the redox disproportionation of 2[4-I]+ seems to be more favorable than that of 2[1-I]+, which is less exergonic. Subsequently, the RuIV-imide species 1[1-II]+ and 1[4-II]+ initiate ammonia oxidation via nucleophilic attack pathways. First, 1[1-II]+ and 1[4-II]+ readily interact with NH3 to generate 1[1-VI]+ and 1[4-VI]+ intermediates (ΔG = −4.5 and −4.3 kcal mol−1, respectively) due to the formation of a hydrogen bond between the H atom of the imide and the N atom of the approaching NH3. Subsequently, terminal N2H4-ligated 1[1-IX]+ and 1[4-IX]+ are formed through transition states 1[1-VII]+ and 1[4-VII]+ (ΔG‡ = 5.4 and 6.0 kcal mol−1, respectively) and intermediates 1[1-Viii]+ and 1[4-Viii]+. The formation of 1[1-IX]+ and 1[4-IX]+ N2H4-ligated intermediates through the reaction of ammonia and RuIV-imides 1[1-II]+ and 1[4-II]+ is highly exergonic by 46.4 and 44.1 kcal mol−1, respectively. Finally, the catalytic cycle is restarted by endergonic evolution of N2H4 through N2H4-by-NH3 substitution of 1[1-IX]+ and 1[4-IX]+ (ΔG = 10.2 and 9.8 kcal mol−1, respectively). Unlike other ruthenium molecular catalysts only generating N2 as the N–N coupling product, the π-donor capability of the pyrrolyl group of the ancillary pp− ligand in the title complexes helps to release N2H4 through N2H4-by-NH3 substitution of the N2H4-ligated intermediate, thus hindering N2H4 overoxidation to generate N2.24
The kinetic studies of the title complexes also support an ammonia nucleophilic attack route over RuIV-imide species. As shown in Fig. S35 and S36, the catalytic current icat (at E = 1.2 V) linearly increases with the increase of [NH3] (0.010–0.050 M) and [cat] (0.2–1.0 mM), clearly indicating that there is a single-site molecular catalytic pathway. Notably, unlike icat at 0.05 M NH3 showing a linear relationship with ν1/2, the icat at 1.0 M NH3 almost does not change with increasing scan rate, indicating that the icat is no longer determined by the bulk diffusion of catalyst or NH3 but by the rate of regeneration of active RuIV-imide species at the electrode.11 This seems to confirm that when ammonia is present in high concentrations, redox disproportionation to generate RuIV-imide could be the slow step, and the nucleophilic coupling pathway could prevail, which is consistent with the recent results of mechanism studies of the [(trpy)(bpyNMe2)Ru(NH3)](PF6)2 catalyst system.14
In summary, a series of ruthenium(II)-cymene NH3-ligated complexes are synthesized and fully characterized. By regulating the electronic structure of the ancillary ligand, the oxidation potential of the ruthenium center is gradually reduced from 0.49 V for [1-NH3]PF6 to 0.16 V for [4-NH3]PF6. Unlike the structurally analogous half-sandwich ferric catalyst [Cp*Fe(1,2-Ph2PC6H4NH)(NH3)]+ containing a phosphinoamido ligand to only generate N2H4 stoichiometrically,17[1-NH3]PF6 to [4-NH3]PF6 exhibit good performance for the selective electrocatalytic conversion of NH3 to N2H4 with at least 98.6% selectivity and 86.3% Faraday efficiency. The mechanism studies illustrate that the RuIII-amide intermediate has radical RuII-aminyl character via the inter-conversion RuIII–NH2 ![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif) RuII–NH2͘͘˙. Bimolecular coupling of the RuII-aminyl species readily generates a N2H4-bridged biruthenium intermediate in [1-NH3]PF6 and [2-NH3]PF6 catalyst systems, but not in [3-NH3]PF6 and [4-NH3]PF6 catalyst systems. Another pathway of N2H4 formation in [1-NH3]PF6 and [4-NH3]PF6, the nucleophilic attack of RuIV-imide species by ammonia, is more feasible due to lower energy barriers of 5.4 kcal mol−1 and 6.0 kcal mol−1, compared to the bimolecular coupling pathway with energy barriers of 12.3 and 17.3 kcal mol−1, respectively. This single-site molecular catalytic pathway is supported by a linear relationship between the catalytic current and concentration of catalyst and ammonia.
RuII–NH2͘͘˙. Bimolecular coupling of the RuII-aminyl species readily generates a N2H4-bridged biruthenium intermediate in [1-NH3]PF6 and [2-NH3]PF6 catalyst systems, but not in [3-NH3]PF6 and [4-NH3]PF6 catalyst systems. Another pathway of N2H4 formation in [1-NH3]PF6 and [4-NH3]PF6, the nucleophilic attack of RuIV-imide species by ammonia, is more feasible due to lower energy barriers of 5.4 kcal mol−1 and 6.0 kcal mol−1, compared to the bimolecular coupling pathway with energy barriers of 12.3 and 17.3 kcal mol−1, respectively. This single-site molecular catalytic pathway is supported by a linear relationship between the catalytic current and concentration of catalyst and ammonia.
Author contributions
Xiao-Yi Yi as the corresponding author contributed to project design and paper revision. Xi Zhang mainly contributed to synthesis and electrocatalysis studies of the titled complexes, writing the preliminary draft. Shan Zhao and Guo Chen contributed to the assist with synthesis. Contribution of Liru Cao and Jian Lin lied in assisting with project design. Zhi-Yan Liu contributed to supplement data during the paper revision process. Piao He contributed to the DFT calculations.Conflicts of interest
The authors declare no competing financial interest.Data availability
The raw data including synthesis, characterization, and photophysical and catalytic properties of these complexes are available from the corresponding author, upon reasonable request.CCDC 1 (2320736), 2 (2324232), 3 (2321021), 4 (2321506) and [2-NH3]PF6 (2427138) contain the supplementary crystallographic data for this paper.
The authors confirm that the data supporting the findings of this study are available within the article and/or its supplementary information (SI). Supplementary information: general methods for synthesis and characterization, crystallographic refinement, cyclic voltammetry experiments and electrolysis experiments, and DFT calculations. See DOI: https://doi.org/10.1039/d5sc08826g.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22471291) and the Foundation of Central South University Research Programme of Advanced Interdisciplinary (2023QYJC019) and Key Program of Hunan Provincial Natural Science Foundation of China (2025JJ30006).References
- (a) H. Tan, Z. Sang, Y. Tian, W. Peng, X. Liu and J. Liang, Ammonia as a Green Carbon-Free Fuel: A Pathway to the Sustainable Energy Economy, ACS Energy Lett., 2024, 9, 5120–5136 CrossRef CAS; (b) D. T. Tran, T. H. Nguyen, H. Jeong, P. K. L. Tran, D. Malhotra, K. U. Jeong, N. H. Kim and J. H. Lee, Recent Engineering Advances in Nanocatalysts for NH3-to-H2 Conversion Technologies, Nano Energy, 2022, 94, 106929 CrossRef CAS.
- S. A. Lee, M. G. Lee and H. W. Jang, Catalysts for Electrochemical Ammonia Oxidation: Trend, Challenge, and Promise, Sci. China Mater., 2022, 65, 3334–3352 CrossRef.
- L. Jiang and X. Fu, An Ammonia-Hydrogen Energy Roadmap for Carbon Neutrality: Opportunity and Challenges in China, Engineering, 2021, 7, 1688–1691 CrossRef.
- N. M. Adli, H. Zhang, S. Mukherjee and G. Wu, Review—Ammonia Oxidation Electrocatalysis for Hydrogen Generation and Fuel Cells, J. Electrochem. Soc., 2018, 165, J3130–J3147 CrossRef CAS.
- (a) J. Li, F. Zhang, H. Xiong, Y. Cai and B. Zhang, Molecular Catalysts for Electrocatalytic Ammonia Oxidation, Sci. China Chem., 2024, 67, 3976–3993 CrossRef CAS; (b) D. N. Stephens and M. T. Mock, Molecular Complexes for Catalytic Ammonia Oxidation to Dinitrogen and the Cleavage of N-H Bonds, Eur. J. Inorg. Chem., 2024, e202400039 CrossRef CAS; (c) H. Y. Liu, H. M. C. Lant, C. C. Cody, J. Jelusic, R. H. Crabtree and G. W. Brudvig, Electrochemical Ammonia Oxidation with Molecular Catalysts, ACS Catal., 2023, 13, 4675–4682 CrossRef CAS.
- F. Habibzadeh, S. L. Miller, T. W. Hamann and M. R. Smith, Homogeneous Electrocatalytic Oxidation of Ammonia to N2 under Mild Conditions, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 2849–2853 CrossRef CAS PubMed.
- K. Nakajima, H. Toda, K. Sakata and Y. Nishibayashi, Ruthenium-catalysed oxidative conversion of ammonia into dinitrogen, Nat. Chem., 2019, 11, 702–709 CrossRef CAS PubMed.
- P. L. Dunn, S. I. Johnson, W. Kaminsky and R. M. Bullock, Diversion of Catalytic C-N Bond Formation to Catalytic Oxidation of NH3 through Modification of the Hydrogen Atom Abstractor, J. Am. Chem. Soc., 2020, 142, 3361–3365 CrossRef CAS PubMed.
- J. Holub, N. Vereshchuk, F. J. Sanchez-Baygual, M. Gil-Sepulcre, J. Benet-Buchholz and A. Llobet, Synthesis, Structure, and Ammonia Oxidation Catalytic Activity of Ru-NH3 Complexes Containing Multidentate Polypyridyl Ligands, Inorg. Chem., 2021, 60, 13929–13940 CrossRef CAS PubMed.
- M. J. Trenerry, C. M. Wallen, T. R. Brown, S. V. Park and J. F. Berry, Spontaneous N2 formation by a diruthenium complex enables electrocatalytic and aerobic oxidation of ammonia, Nat. Chem., 2021, 13, 1221–1227 CrossRef CAS PubMed.
- S. I. Jacob, A. Chakraborty, A. Chamas, R. Bock, L. Sepunaru and G. Ménard, Rapid Aqueous Ammonia Oxidation to N2 Using a Molecular Ru Electrocatalyst, ACS Energy Lett., 2023, 8, 3760–3766 CrossRef CAS.
- A. M. Beiler, A. Denisiuk, J. Holub, F. J. Sánchez-Baygua, M. Gil-Sepulcre, M. Z. Ertem, D. Moonshiram, A. Piccioni and A. Llobet, Heterogeneous Electrochemical Ammonia Oxidation with a Ru-bda Oligomer Anchored on Graphitic Electrodes via CH-π Interactions, ACS Energy Lett., 2023, 8, 172–178 CrossRef CAS PubMed.
- S. Feng, J. Chen, R. Wang, H. Li, J. Xie, Z. Guo, T.-C. Lau and Y. Liu, Dual Pathways in Catalytic Ammonia Oxidation by a Ruthenium Complex Bearing a Tetradentate Bipyridine–Bipyrazole Ligand: Isolation of a Diruthenium Intermediate with a µ-Hexazene Derivative, J. Am. Chem. Soc., 2024, 146, 21490–21495 CrossRef PubMed.
- B. Yuan, G. L. Tripodi, M. T. G. M. Derks, Y. Pereverzew, S. Zhou and J. Roithová, Mapping the Catalytic Cycle of Ru-Catalyzed Ammonia Oxidation, Angew. Chem., Int. Ed., 2025, 64, e202501617 CrossRef CAS PubMed.
- (a) G. Chen, P. He, C. Liu, X.-F. Mo, J.-J. Wei, Z.-W. Chen, T. Cheng, L.-Z. Fu and X.-Y. Yi, Direct synthesis of hydrazine by efficient electrochemical ruthenium-catalysed ammonia oxidation, Nat. Catal., 2023, 6, 949–958 CrossRef CAS; (b) C. Zhou, X. Zhang, S. Zhao, S.-D. Zhong, X.-L. Ding, S.-P. Yang, F. Pan, P. He and X.-Y. Yi, Catalytic Selective Conversion of Ammonia into Hydrazine by a RuII(trpy) Complex Bearing a Pyridylpyrrole Ligand, ACS Catal., 2025, 15, 3535–3545 CrossRef CAS; (c) S. Zhao, X. Zhang, G. Chen, T. Cheng, X.-L. Ding, S.-D. Zhong, S.-P. Yang, P. He and X.-Y. Yi, Selective Electrocatalytic Oxidation of Ammonia by Ru-dpp Complexes Containing Aromatic Nitrogen Donor as Axial Ligand, Inorg. Chem., 2024, 63, 23150–23157 CrossRef CAS PubMed.
- (a) M. D. Zott, P. Garrido-Barros and J. C. Peters, Electrocatalytic Ammonia Oxidation Mediated by a Polypyridyl Iron Catalyst, ACS Catal., 2019, 9, 10101–10108 CrossRef CAS; (b) M. D. Zott and J. C. Peters, Enhanced Ammonia Oxidation Catalysis by a Low-Spin Iron Complex Featuring cis Coordination Sites, J. Am. Chem. Soc., 2021, 143, 7612–7616 CrossRef CAS PubMed; (c) M. D. Zott and J. C. Peters, Improving Molecular Iron Ammonia Oxidation Electrocatalysts via Substituent Effects that Modulate Standard Potential and Stability, ACS Catal., 2023, 13, 14052–14057 CrossRef CAS PubMed.
- Y. Li, J.-Y. Chen, Q. Miao, X. Yu, L. Feng, R.-Z. Liao, S. Ye, C.-H. Tung and W. Wang, A Parent Iron Amido Complex in Catalysis of Ammonia Oxidation, J. Am. Chem. Soc., 2022, 144, 4365–4275 CrossRef CAS PubMed.
- L. Liu, S. I. Johnson, A. M. Appel and R. M. Bullock, Oxidation of Ammonia Catalyzed by a Molecular Iron Complex: Translating Chemical Catalysis to Mediated Electrocatalysis, Angew. Chem., Int. Ed., 2024, 63, e202402635 CrossRef CAS PubMed.
- M. E. Ahmed, R. J. Staples, T. R. Cundari and T. H. Warren, Electrocatalytic Ammonia Oxidation by Pyridyl-Substituted Ferrocenes, J. Am. Chem. Soc., 2025, 147, 6514–6522 CrossRef CAS PubMed.
- (a) M. E. Ahmed, M. Raghibi Boroujeni, P. Ghosh, C. Greene, S. Kundu, J. A. Bertke and T. H. Warren, Electrocatalytic Ammonia Oxidation by a Low-Coordinate Copper Complex, J. Am. Chem. Soc., 2022, 144, 21136–21145 CrossRef CAS PubMed; (b) H. Y. Liu, H. M. C. Lant, J. L. Troiano, G. Hu, B. Q. Mercado, R. H. Crabtree and G. W. Brudvig, Electrocatalytic, Homogeneous Ammonia Oxidation in Water to Nitrate and Nitrite with a Copper Complex, J. Am. Chem. Soc., 2022, 144, 8449–8453 CrossRef CAS PubMed.
- (a) D. N. Stephens, R. K. Szilagyi, P. N. Roehling, N. Arulsamy and M. T. Mock, Catalytic Ammonia Oxidation to Dinitrogen by a Nickel Complex, Angew. Chem., Int. Ed., 2023, 62, e202213462 CrossRef CAS PubMed; (b) N. X. Gu, P. H. Oyala and J. C. Peters, Hydrazine Formation via Coupling of a Nickel(III)-NH2 radical, Angew. Chem., Int. Ed., 2021, 60, 4009–4013 CrossRef CAS PubMed.
- H. Toda, K. Kuroki, R. Kanega, S. Kuriyama, K. Nakajima, Y. Himeda, K. Sakata and Y. Nishibayashi, Manganese-Catalyzed Ammonia Oxidation into Dinitrogen under Chemical or Electrochemical Conditions, ChemPlusChem, 2021, 86, 1511–1516 CrossRef CAS PubMed.
- J. N. McPherson, B. Das and S. B. Colbran, Tridentate pyridine–pyrrolide chelate ligands: An under-appreciated ligand set with an immensely promising coordination chemistry, Coord. Chem. Rev., 2018, 375, 285–332 Search PubMed.
- G. Chen, X. L. Ding, P. He, T. Cheng, Y. Chen, J. Lin, X. Zhang, S. Zhao, N. Qiao and X.-Y. Yi, Understanding the factors governing the ammonia oxidation reaction by mononuclear ruthenium complex, Chem. Sci., 2025, 16, 7573–7578 Search PubMed.
- P. Štěpnička, J. Ludvík, J. Canivet and G. Süss-Fink, Relating catalytic activity and electrochemical properties: The Case of Arene-ruthenium Phenanthroline Complexes Catalytically Active in Transfer Hydrogenation, Inorg. Chim. Acta, 2006, 359, 2369–2374 CrossRef.
- J. P. da Silva, O. M. Fuganti, G. Kramer, G. Facchin, L. E. N. Aquino, J. Ellena, D. F. Back, A. C. S. Gondim, E. H. S. Sousa, L. G. F. Lopes, S. Machado, I. D. L. Guimarães, K. Wohnrath and M. P. de Araujo, Electrochemical, Mechanistic, and DFT Studies of Amine Derived Diphosphines Containing Ru(ii)-cymene Complexes with Potent in vitro Cytotoxic Activity Against HeLa and Triple-negative Breast Cancer Cells MDA-MB-231, Dalton Trans., 2020, 49, 16498–16514 RSC.
- A. J. Bard, L. R. Faulkner and H. S. White, Electrochemical Methods: Fundamentals and Applications, 2022, p. 248 Search PubMed.
- G. W. Watt and J. D. Chrisp, Spectrophotometric Method for Determination of Hydrazine, Anal. Chem., 1952, 24, 2006–2008 CrossRef CAS.
- H. Y. Liu, H. M. C. Lant, J. L. Troiano, G. Hu, B. Q. Mercado, R. H. Crabtree and G. W. Brudvig, Electrocatalytic, Homogeneous Ammonia Oxidation in Water to Nitrate and Nitrite with a Copper Complex, J. Am. Soc. Chem., 2022, 144, 8449–8453 CrossRef CAS PubMed.
- Q. Chen, J. Liang, L. Yue, Y. Luo, Q. Liu, N. Li, A. A. Alshehri, T. Li, H. Guo and X. Sun, CoO Nanoparticle Decorated N-doped Carbon Nanotubes: a High-efficiency Catalyst for Nitrate Reduction to Ammonia, Chem. Commun., 2022, 58, 5901–5904 RSC.
- (a) CCDC 2320736: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2hwxgs; (b) CCDC 2324232: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2j0k7c; (c) CCDC 2321021: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2hx6n9; (d) CCDC 2321506: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2hxq9g; (e) CCDC 2427138: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2mgmsj.
| This journal is © The Royal Society of Chemistry 2026 |




