 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Dinitrogen complexes N2L2 (L = N2, CO, CS, NO+, CN−)
Yahui
Li
a,
Chengxiang
Ding
a,
Lianbin
Xie
a,
Sudip
Pan
 *a and
Gernot
Frenking
*a and
Gernot
Frenking
 *bcd
*bcd
aInstitute of Atomic and Molecular Physics, Jilin University, Changchun 130023, China. E-mail: sudip@jlu.edu.cn
bInstitute of Advanced Synthesis, School of Chemistry and Molecular Engineering, Nanjing Tech University, Nanjing 211816, China. E-mail: frenking@chemie.uni-marburg.de
cFachbereich Chemie, Philipps-Universität Marburg, Hans-Meerwein-Strasse 4, D-35043 Marburg, Germany
dDonostia International Physics Center (DIPC), M. de Lardizabal Pasealekua 3, 20018 Donostia, Euskadi, Spain
First published on 9th February 2026
Abstract
Quantum chemical calculations using ab initio methods and density functional theory have been carried out on the equilibrium structures and the vibrational spectra of the (valence) isoelectronic compounds N2L2 (L = N2, CO, CS, NO+, CN−). The molecules have a trans-periplanar arrangement of the L2 ligands at the N2 unit. The complexes with L = N2, CO, NO+, CN− are predicted as thermodynamically unstable for dissociation into N2 + 2L with ΔG298 value lying in between −257 kcal mol−1 (L = NO+) and −73 kcal mol−1 (L = CO), but the adduct N2(CS)2 is calculated as slightly stable with ΔG298 = 4 kcal mol−1. The homolytic dissociation reaction into two fragments N2L2 → 2 NL is energetically less favorable than the heterolytic fragmentation N2L2 → N2 + 2 L, which proceeds synchronously but asymmetrically. The activation barriers for the fragmentation reaction N2L2 → N2 + 2L have values between ΔG≠(298 K) = 17 kcal mol−1 for L = N2 and ΔG≠(298 K) = 84 kcal mol−1 for L = CS. The calculated vibrational frequencies suggest that the molecules N2L2 can be identified by the IR active antisymmetric stretching mode νas of the ligands L, which is blue shifted for L = CO (Δ = 55 cm−1) and L = NO+ (Δ = 118 cm−1) but it is red shifted for L = CS (Δ = −242 cm−1) and L = CN− (Δ = −133 cm−1) relative to the νas mode of L = N2. The analysis of the bonding situation reveals that there is a total charge donation L→(1Γ-N2)←L in all complexes, ranging between 1.38 e (L = CN−) and 0.56 e (L = N2), except in the dication with L = NO+, where a small backdonation in reverse direction L←(1Γ-N2)→L with 0.10 e is calculated. EDA-NOCV calculations of N6 show that the best description of the bonding situation is given in terms of dative interactions N2→(1Γ-N2)←N2 between central N2 in the excited (1)1Γg singlet state and the terminal N2 fragments in the 1Σg+ electronic ground state. In contrast, the best description of the complexes with L = CO, CS, NO+ is calculated for the interactions between the central N2 in the 5Σu+ quintet state and the terminal ligands in the symmetry-adapted (L)2 quintet state. For N2L2 with L = CN−, it is found that the bonding is best described for the interaction between N2− in the electronic quartet (4Σu+) state and the terminal (L)2− ligand as symmetry-adapted quartet. In contrast to the common bonding model for N6 using Lewis structures N−![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+
N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–N
N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+=N−, the donor–acceptor model N2→(N2)←N2 explains that the lowest activation barrier is found for the concerted cleavage of the two formal double bonds, leading to the experimentally observed dissociation into 3 N2.
N+=N−, the donor–acceptor model N2→(N2)←N2 explains that the lowest activation barrier is found for the concerted cleavage of the two formal double bonds, leading to the experimentally observed dissociation into 3 N2.
Introduction
In 1964, Appel and Schöllhorn reported that triphenylphosphinazine, which was sketched with the formula Ph3P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–N
N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) PPh3, is a thermally stable diamagnetic species that has a melting point of 184°.1 However, neither precise structural information about the compound was given in the work, nor in their further study.2 Later, quantum chemical calculations showed that N2(PPh3)2 is thermodynamically unstable for the release of N2 by ∼90 kcal mol−1,3 which raised doubts about the structure of the isolated compound. A subsequent X-ray structure analysis confirmed that the species is indeed triphenylphosphinazine, which has an antiperiplanar arrangement of the phosphine groups in Ph3P-(N2)-PPh3 with bending angles P–N–N of 107o, and a long N–N bond of 1.497 Å.4 The theoretical analysis of the bonding situation suggested that the N2 moiety binds through its highly excited (1)1Γg state, where the out-of-plane π and π* orbitals are doubly occupied and the in-plane π MO is vacant, which leads to strong dative interactions Ph3P→(N2)←PPh3 with N2 as a double Lewis acid. The same type of dative interactions was suggested for the related compound with NHC (N-Heterocyclic Carbene) ligand, NHC→(N2)←NHC, which was reported to have an antiperiplanar arrangement of the NHC groups and a long N–N bond of 1.415 Å.5
PPh3, is a thermally stable diamagnetic species that has a melting point of 184°.1 However, neither precise structural information about the compound was given in the work, nor in their further study.2 Later, quantum chemical calculations showed that N2(PPh3)2 is thermodynamically unstable for the release of N2 by ∼90 kcal mol−1,3 which raised doubts about the structure of the isolated compound. A subsequent X-ray structure analysis confirmed that the species is indeed triphenylphosphinazine, which has an antiperiplanar arrangement of the phosphine groups in Ph3P-(N2)-PPh3 with bending angles P–N–N of 107o, and a long N–N bond of 1.497 Å.4 The theoretical analysis of the bonding situation suggested that the N2 moiety binds through its highly excited (1)1Γg state, where the out-of-plane π and π* orbitals are doubly occupied and the in-plane π MO is vacant, which leads to strong dative interactions Ph3P→(N2)←PPh3 with N2 as a double Lewis acid. The same type of dative interactions was suggested for the related compound with NHC (N-Heterocyclic Carbene) ligand, NHC→(N2)←NHC, which was reported to have an antiperiplanar arrangement of the NHC groups and a long N–N bond of 1.415 Å.5
Very recently, Qian, Mardyukov and Schreiner (QMS) reported the synthesis, via gas-phase reaction, of the new nitrogen allotrope N6, which was trapped in low-temperature argon matrices at 10 K and as a film at liquid nitrogen temperature of 77 K.6 The molecule was identified by IR and UV-vis spectroscopy, and by ab initio calculations, which predict a structure where two N3 fragments are bonded in a trans-arrangement through a long (1.460 Å) N–N bond. The viewpoint of N6 as the dimer of N3 is reasonable, because hexanitrogen was synthesized by treating AgN3 with Cl2, which yields ClN3 that reacts with AgN3 and leads to the formation of the new nitrogen allotrope N6. Hexanitrogen was heralded as a molecule of the year 2025 wth a long N–N bond connecting two N3 fragments.7 But the structural similarity to the N2(PPh3)2 and N2(NHC)2 species led us suspect that the N6 species is another example of the compound class N2L2 where the ligands L are bonded through dative interactions L→(1Γ-N2)←L. Dinitrogen N2 is generally known as weakly bonded ligand, but the highly excited (1)1Γg state of the central N2 is a strong σ acceptor and strong π donor, which is capable to bind two N2 ligands. The (1)1Γg state of N2 is 294.3 kcal mol−1 above the X1Σg+ ground state,8 which, however, does not occur in N2L2 as a free species, but as a reference state that is strongly stabilised by orbital interactions.
Another acceptor, which binds even eight N2 ligands in the octa-coordinated complexes M(N2)8 (M = Ca, Sr, Ba), is the alkaline-earth atom M in an excited triplet state with (n−1)d2 electron configuration via strong M→(N2)8 π backdonation.9
The new findings prompted us to investigate the electronic structure of N6 in terms of dative bonding N2→(1Γ-N2)←N2 and to compare the homolytic and heterolytic bond dissociation with quantum chemical methods. We also calculated the (valence) isoelectronic compounds N2L2 (L = N2, CO, CS, NO+, CN−). Here, we report about the equilibrium geometries, bond dissociation energies (BDEs), and the vibrational spectra of the molecules. We also present a thorough analysis of the nature of the chemical bonds using a variety of methods. The results may be useful as a guideline for future experimental studies.
Methods
The geometrical optimizations, followed by the frequency calculations of N2L2 (L = N2, CO, CS, NO+, CN−) compounds, were first carried out at the M06-2X/cc-pVTZ level10 in their respective singlet and triplet spin states. Taking the most stable spin state (singlet), further reoptimizations and frequency calculations were performed at the CCSD(T)-Full/cc-pVTZ level.11 These calculations were done using the Gaussian 16 program.12 Natural atomic charges and Mayer bond order13 were calculated using NBO7 (ref. 14) and Multiwfn programs,15 respectively.Energy decomposition analysis (EDA)16 in conjunction with the natural orbital for chemical valence theory (NOCV)17 was carried out at the M06-2X/TZ2P-ZORA18//CCSD(T)/cc-pVTZ level using the ADF 2020 package.19 The ZORA method considers relativistic effects, which are unimportant for this work, but further work by us on heavier analogues requires a uniform level of theory. In the EDA-NOCV20 analysis, the intrinsic interaction energy (ΔEint) between two fragments is dissected into three distinct energy components, as follows:
| ΔEint = ΔEelstat + ΔEPauli + ΔEorb | (1) |
The electrostatic ΔEelstat term represents the quasiclassical electrostatic interaction between the unperturbed charge distributions of the prepared fragments. The Pauli repulsion, ΔEPauli accounts for the energy change during the transformation from the superposition of unperturbed electron densities of the individual fragments to a wavefunction that explicitly adheres to the Pauli principle, achieved through the necessary antisymmetrization and wavefunction renormalization. The orbital term ΔEorb comes from the mixing of orbitals, charge transfer, and polarization between the isolated fragments.
The EDA-NOCV enables the partition of the total ΔEorb into pairwise contributions of the orbital interactions that are very important to get a complete picture of the bonding. The charge deformation Δρk(r), resulting from the mixing of the orbital pairs ψk(r) and ψ−k(r) of the interacting fragments presents the amount and the shape of the charge flow due to the orbital interactions (eqn (2)), and the associated energy term ΔEorb provides with the size of stabilizing orbital energy originated from such interaction (eqn (3)).
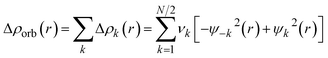 | (2) |
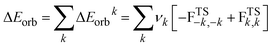 | (3) |
Several papers extensively discussed details of the EDA-NOCV method and its application, offering different perspectives and viewpoints.21
Results and discussion
Fig. 1 shows the calculated geometries, atomic partial charges (q), and bond orders (P) of the compounds N2L2 (L = N2, CO, CS, NO+, CN−) at the CCSD(T)/cc-pVTZ level. All molecules exhibit a trans-periplanar arrangement of the L2 ligands at the N2 unit with a C2h point group and 1Ag electronic state. The singlet-triplet gap computed at the M06-2X/cc-pVTZ level is very high (39.7–65.7 kcal mol; see Table S1 in SI). The central N–N bond lengths in N2(N2)2 (1.450 Å) and [N2(CN)2]2− (1.488 Å) compare favorably with the bond length in H2N–NH2 (1.445 Å),6 whereas the N–N bonds in N2(CO)2 (1.386 Å), N2(CS)2 (1.355 Å) and [N2(NO)2]2+ (1.382 Å) are significantly shorter. The small differences between our values for N6 and the data reported by QMS are due to the fact that the CCSD(T)/cc-pVTZ calculations were carried out by us with the full core option, whereas the previous study used the frozen core approximation. Geometry optimisations of a syn isomer did not result in another conformational isomer, but rather in the trans form as the only acyclic energy minimum structure. An extensive search of the potential energy surface using various starting geometries gave planar four-membered cyclic structures N2(CX)2 (X = O, S) as energy minima, which are > 25 kcal mol−1 less stable than the acyclic N2L2 forms. We also located nonplanar four-membered cyclic isomers for all species except for N6 where the central N–N bond is broken, which are > 40 kcal mol−1 higher in energy than the acyclic N2L2 forms. They are shown in Fig. S1 of the SI. Since the present work focuses on the dinitrogen complexes N2L2, we do not discuss the cyclic isomers in this work.Table 1 gives the calculated BDEs for the fragmentation N2L2 → N2 + 2L at the CCSD(T)/cc-pVTZ level, which show a remarkably high variation. The purely electronic values suggest that [N2(NO)2]2+ is even less stable (De = −233.3 kcal mol−1) than N2(N)2 (De = −179.1 kcal mol−1). The exoenergetic values are much lower for [N2(CN)2]2− (De = −116.7 kcal mol−1) and N2(CO)2 (De = −49.5 kcal mol−1), and N2(CS)2 is even predicted to be energetically stable (De = 27.7 kcal mol−1). The corrections by vibrational frequencies and entropic and thermal contributions lead to ΔG298 values where the N2L2 compounds are seen as thermodynamically unstable in the order L = NO+ > N2 > CN− > CO, but the molecule N2(CS)2 is calculated as thermodynamically stable at room temperature with ΔG298 = 3.6 kcal mol−1.
| Complex | D e | D 0 | ΔH | ΔG |
|---|---|---|---|---|
| (a) N 2 L 2 → N 2 + 2L | ||||
| N2(N2)2 | −179.1 | −184.1 | −182.0 | −200.7 |
| N2(CO)2 | −49.5 | −55.8 | −53.7 | −73.4 |
| N2(CS)2 | 27.7 | 21.8 | 23.6 | 3.6 |
| [N2(CN)2]2− | −116.7 | −122.2 | −120.1 | −139.7 |
| [N2(NO)2]2+ | −233.3 | −238.7 | −236.5 | −256.8 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||||
| (b) N 2 L 2 → 2NL | ||||
| N2(N2)2 | 34.4 | 29.6 | 30.5 | 22.2 |
| N2(CO)2 | 62.1 | 58.5 | 59.1 | 48.9 |
| N2(CS)2 | 27.8 | 24.1 | 25.0 | 13.4 |
| [N2(CN)2]2− | −51.8 | −56.8 | −55.8 | −60.8 |
| [N2(NO)2]2+ | −70.2 | −74.5 | −73.7 | −84.6 |
Table 1 also gives the BDEs for breaking the central N–N bond in the fragmentation N2L2 → 2 NL at the CCSD(T)/cc-pVTZ level. It becomes obvious that the homolytic bond rupture of the N–N bond is thermodynamically strongly disfavoured compared with the heterolytic cleavage of the L–N2–L bonds, except for L = CS. The energies of the two fragmentation reactions of the latter species are very similar, but the free energy of the heterolytic process (ΔG = 3.6 kcal mol−1) makes it clearly more favourable than the homolytic fragmentation (ΔG = 13.4 kcal mol−1). The calculated BDEs at the M06-2X/cc-pVTZ level are very similar (Table S2 of SI) to the CCSD(T)/cc-pVTZ values, which indicates that the DFT values are quite reliable.
Fig. 2 shows the reaction profile for the heterolytic dissociation reaction N2L2 → N2 + 2 L along with the calculated activation free energy barriers ΔG≠(298 K), which vary between 16.9 kcal mol−1 (L = N2) and 84.2 kcal mol−1 (L = CS). The trend of the ΔG≠ follows, in general, the reaction energies, except for the very exergonic reactions where L = N2, NO+. The calculated barrier for the fragmentation of N2 is in good agreement with the value reported by QMS (14.8 kcal mol−1).6 The computed value of the T1 diagnostics suggests that the multi-reference character of the CCSD(T) calculations of the N2L2 complexes and the transition states is very low (Table S3, SI), which indicates that the single-reference approach is sufficient. The activation barrier for the homolytic reaction N6 → 2 N3 was reported by QMS to be significantly higher (ΔG≠(298 K) = 26.1 kcal mol−1) than the heterolytic process.6 We carried out energy calculations with stretched N–N distances of the other L2L2 species, which suggest that the barriers are also higher than for the heterolytic fragmentation. Since the NL fragments are also less stable than N2 + 2 L (Table 1), it is unlikely that the homolytic reaction course plays a role in the fragmentation reaction of the N2L2 species.
The higher barrier for breaking the bond in the central N2 moiety of N6 compared to breaking the (N2)–(N2)2 bonds is surprising, given the calculated bond orders, which are much higher and have shorter distances in the latter bonds than the former one. It turns out that the energy required to break a bond depends not only on the strength of the bond, but also on the reorganisation of the electronic structure of the fragments during the cleavage reaction. The N3–N3 bond in N6 is weaker than the (N2)–(N2)2 bonds, but the electronic charge migration during rupture of the latter heterolytic process is energetically more favourable than the homolytic reaction. The lower barrier for breaking the (N2)–(N2)2 bonds than the N3–N3 bond supports the use of our bonding model for N6 in terms of dative interactions N2→(N2)←N2 rather than the more conventional model using Lewis structures N−![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+
N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–N
N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+
N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N−, since it accounts for the experimental finding that N6 directly dissociated into 3 N2. The same applies for N2(CO)2, which photolytically dissociates into N2 + 2 CO whereas cleavage into NCO radical was observed only to a small extent.22a
N−, since it accounts for the experimental finding that N6 directly dissociated into 3 N2. The same applies for N2(CO)2, which photolytically dissociates into N2 + 2 CO whereas cleavage into NCO radical was observed only to a small extent.22a
Examination of the transition state structures for the dissociation reaction reveals surprising features (Fig. 3). The geometries of [N2L2]≠ possess a non-planar staggered geometry with a syn conformation of the ligands except for [N2(CS)2]≠, which exhibits a nearly linear NNCS moiety with one CS ligand, where the bond to the second CS ligand is significantly stretched. This implies that the fragmentation reaction may possibly be a two-step process in which an intermediate product, NN-L is formed along the dissociation reaction. We calculated the intrinsic reaction coordinate starting from the transition states and found that it smoothly connects to the reaction products N2 + 2L. The fragmentation reaction is thus predicted as a concerted but highly asynchronous process where the two N2–L bonds break one after the other. Note that the central N–N bond of the transition state [N2L2]≠ is clearly shorter than in the equilibrium structures.
Hexanitrogen was clearly identified by spectroscopic signals, and the authors presented a careful analysis of the vibrational spectrum of the molecule.6 The IR spectrum of N6 exhibits an intense vibrational band at 2076.6 cm−1, which comes from an asymmetric stretching mode of the terminal N2 ligands. Fig. 4 shows the calculated IR spectra of the five molecules N2L2. They show a similar pattern where the asymmetric stretching mode νas of the terminal L2 ligands, which has the second largest wavenumber of all 12 fundamentals, exhibits the strongest signal. The symmetric stretching mode νs with a slightly higher wavenumber is IR inactive. The calculated wavenumber of 2263.7 cm−1 for the asymmetric mode of N6 is blue shifted for L = CO (Δ = 55 cm−1) and L = NO+ (Δ = 118 cm−1), but it is red shifted for L = CS (Δ = −242 cm−1) and L = CN− (Δ = −133 cm−1). The calculated frequency shift of the intense asymmetric stretching mode νs is very helpful information for identifying the molecules.
 | ||
| Fig. 4 Calculated IR spectra of N2L2 complexes (L = N2, CO, CS, CN−, NO+) at the M06-2X/cc-pVTZ level. | ||
In addition to N6 [N2(N2)2], the complexes N2(CO)2 and N2(CS)2 have been synthesised and identified spectroscopically and they were also the subject of theoretical works. The dicarbonyl adduct was introduced as diisocynate O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–N
N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O following the conventional descriptions using Lewis structures.22 Photolysis of the matrix-isolated N2(CO)2 gives N2 + 2 CO as products, which agrees with our calculated reaction profile (Fig. 2). The IR spectrum shows one very intensive band at 2200.6 cm−1 for the asymmetric NCO stretching mode besides several signals with lower intensity.22a The comparison with the IR spectrum of N6 gives a red shift of 124 cm−1, which agrees with the direction but is higher than our computed value of 54 cm−1. The diisothiocyanate complex N2(CS)2 was identified as product of the photolysis of the energetically lower lying isomer S2(CN)2 with a characteristic vibrational mode of the IR spectrum at 1910 cm−1.23 The experimental red-shift compared to the asymmetric stretching mode of N6 is also given by our calculations, but the observed value of −166 cm−1 is smaller than our computed value of −242 cm−1. The computed wavenumber for [N2(NO)2]2+ (2382 cm−1) agrees quite well with the experimental value of NO+ (2340 cm−1) whereas the calculated value for [N2(CN)2]2− (2131 cm−1) is red-shifted compared with an approximately interaction-free anion CN− (2244 cm−1).24 The complete set of the calculated vibrational spectra of the five molecules N2L2 is given in Table S4 of the SI.
O following the conventional descriptions using Lewis structures.22 Photolysis of the matrix-isolated N2(CO)2 gives N2 + 2 CO as products, which agrees with our calculated reaction profile (Fig. 2). The IR spectrum shows one very intensive band at 2200.6 cm−1 for the asymmetric NCO stretching mode besides several signals with lower intensity.22a The comparison with the IR spectrum of N6 gives a red shift of 124 cm−1, which agrees with the direction but is higher than our computed value of 54 cm−1. The diisothiocyanate complex N2(CS)2 was identified as product of the photolysis of the energetically lower lying isomer S2(CN)2 with a characteristic vibrational mode of the IR spectrum at 1910 cm−1.23 The experimental red-shift compared to the asymmetric stretching mode of N6 is also given by our calculations, but the observed value of −166 cm−1 is smaller than our computed value of −242 cm−1. The computed wavenumber for [N2(NO)2]2+ (2382 cm−1) agrees quite well with the experimental value of NO+ (2340 cm−1) whereas the calculated value for [N2(CN)2]2− (2131 cm−1) is red-shifted compared with an approximately interaction-free anion CN− (2244 cm−1).24 The complete set of the calculated vibrational spectra of the five molecules N2L2 is given in Table S4 of the SI.
We analyzed the bonding situation in N2L2 with a variety of methods. Fig. 1 shows that the Mayer bond order (MBO) of the central N–N bond is between 0.83 (L = CN−) and 0.96 (L = CS), which is a rather small variation considering the differences in the N–N bond length between 1.488 Å (L = CN−) and 1.355 Å (L = CS). The atomic partial charges suggest that there is a total charge donation in the neutral complexes L→(N2)←L with 0.98 e (L = CO), 0.78 e (L = CS), and 0.56 e (L = N2). There is an even stronger charge donation of 1.38 e in the dianion (L = CN−) and a small backdonation in reverse direction L←(N2)→L with 0.10 e in the dication (L = NO+).
We proposed in our earlier study that the diatomic N2 species in N2(PPh3)2 binds through its highly excited (1)1Γg state where the out-of-plane π and π* orbitals are doubly occupied.4Fig. 5 shows schematically the valence MOs of N2 in the (1)1Γg state. The donation of the L2 ligands L→(N2)←L may take place into the vacant 1πu′ (bonding) and 1πg′ (antibonding) orbitals of N2. Note that the assignment of π symmetry refers to free N2, which has two mirror planes that contain the atoms. The complexes L-N2-L have only one mirror plane and the donation is correctly assigned as in-plane σ(+,−) and σ(+,+) orbital interaction.
There are two occupied out-of-plane π MOs, 1πu (bonding) and 1πg (antibonding), in the (1)1Γg state of N2. If this holds true also for the N2L2 complexes in the present study, the number of occupied valence π MOs should be four (π and π* from N2 and one π from each ligand L) and not three. Examination of the shape of the Kohn–Sham MOs shows that this is indeed the case. Fig. 6 displays the four occupied valence π MOs of N6. The number of nodes follows the common symmetry rules with zero (HOMO-8), one (HOMO-7), two (HOMO-2), and three (HOMO). The same number of occupied valence π is found for the other N2L2 complexes. The complete set of occupied valence MOs of all N2L2 complexes is presented in Fig. S2–S6. Fig. 6 shows also the HOMO of the N2L2 complexes, which can be identified with the out-of-plane π* orbital of N2 mixing with the antibonding π* orbitals of the ligands L.
 | ||
| Fig. 6 Top row: plot of the four occupied valence π MOs of N6. Bottom row: plot of the HOMO of N2L2 with L = CO, CS, NO+, CN−. | ||
A more detailed insight into the nature of the N2–L2 bonds is provided by the EDA-NOCV analysis of the interactions between the fragments. In our previous study of N2(PPh3)2, we considered only neutral N2 in the excited (1)1Γg state and 2 PPh3 ligands in the electronic ground state.4 We analyzed N2L2 using various electronic states and charges of the fragments N2 and 2L. Numerous studies have shown that the strength of the orbital term ΔEorb, which considers the change in the wave function during bond formation, is a reliable indicator of the optimal fragments for describing the bond between them.7,25 The fragments with the lowest absolute values of ΔEorb are the best choice for explaining the bonding interactions. Table 2 shows the numerical results for L = N2.
| Energies | Orbital interaction | N2 (singlet) + 2L (singlet) | N2 (triplet) + 2L (triplet) | N2 (quintet) + 2L (quintet) | N2−(doublet) + 2L+(doublet) | N2−(quartet) + 2L+(quartet) |
|---|---|---|---|---|---|---|
| a The values in parentheses give the percentage contribution to the total attractive interactions ΔEelstat + ΔEorb. b The values in parentheses give the percentage contribution to the total orbital interactions ΔEorb. | ||||||
| ΔEint | −165.0 | −368.7 | −401.3 | −387.3 | −519.0 | |
| ΔEPauli | 1036.0 | 1022.8 | 1028.3 | 1042.8 | 1090.1 | |
| ΔEelstata | −364.9 (30.4%) | −388.8 (27.9%) | −438.1 (30.6%) | −538.1 (37.6%) | −615.1 (38.6%) | |
| ΔEorba | −836.1 (69.6%) | −1002.7 (72.1%) | −991.5 (69.4%) | −894.5 (62.4%) | −980.0 (61.4%) | |
| ΔEorb(1)b | L–NN–L σ-bond (+,−) | −328.7 (39.3%) | −317.3 (31.6%) | −328.7 (33.2%) | −329.8 (36.9%) | −350.4 (35.8%) |
| ΔEorb(2)b | L–NN–L σ-bond (+,+) | −296.4 (35.5%) | −296.9 (29.6%) | −314.1 (31.7%) | −261.4 (29.2%) | −337.7 (34.5%) |
| ΔEorb(3)b | L–NN–L π-bond (+,−) | −98.6 (11.8%) | −174.9 (17.4%) | −151.4 (15.3%) | −123.1 (13.8%) | −90.9 (9.3%) |
| ΔEorb(4)b | L–NN–L π-bond (+,+) | −44.7 (5.3%) | −86.5 (8.6%) | −94.8 (9.6%) | −54.0 (6.0%) | −56.2 (5.7%) |
| ΔEorb(rest)b | −67.7 (8.1%) | −127.1 (12.7%) | −102.2 (10.3%) | −126.2 (14.1%) | −144.8 (14.8%) | |
It becomes obvious that the chemical bonds in N6 between the central N2 moiety and the terminal N2 species are indeed best described in terms of dative interactions N2→(N2)←N2 between central N2 in the excited (1)1Γg singlet state and two terminal N2 fragments in the 1Σg+ electronic ground state. The interactions between central N2 in the 3Σu+ triplet state and the terminal ligands in the symmetry-adapted (N2)2 triplet state, which exhibit a mixture of electron-sharing and dative bonds, give a bigger ΔEorb value. The same holds for the formation of electron-sharing double bonds between central N2 in the 5Σu+ quintet state and the terminal ligands in the symmetry-adapted (N2)2 quintet state. EDA-NOCV calculations using central N2− as an anion in the electronic doublet (2Σg+) or quartet (4Σu+) state and the terminal (N2)2+ ligand as a cation in the symmetry-adapted doublet or quartet state also result in bigger ΔEorb values.
Further examination of the dative interactions between central N2 and the terminal N2 ligands reveals that they come mainly from N2→(N2) N2σ donation through out-of-phase (ΔEorb(1)) and in-phase (ΔEorb(2)) orbital pairs, which provide 75% of the total orbital (covalent) bonding. The π backdonation N2←(N2)→N2via out-of-phase (ΔEorb(3)) and in-phase (ΔEorb(4)) orbital interactions deliver only 17% of ΔEorb. The nature of the individual orbital interactions ΔEorb(1)–ΔEorb(4) becomes clear when considering the corresponding deformation densities and the associated orbitals, which are shown in Fig. 7. The dominant orbital interactions through (+,−) and (+,+) σ donation N2→(N2)←N2 nicely explain the bond shortening of the central N2 moiety compared to free N2 in the 1Γ state, which has a calculated bond length of 1.608 Å and is 294.3 kcal mol−1 above the X1Σg+ ground state.8 The donation takes place into the vacant in-plane and out-of-plane π orbitals of N2, which are bonding orbitals (see Fig. 5). Note that the (1)1Γg state of N2 is a reference state which is strongly stabilized by the orbital interaction. It is not formed as a free species during the reaction process.
The numerical EDA-NOCV results of the other N2L2 compounds are given in Tables S5–S8 of SI. In contrast to the results for N6, the lowest ΔEorb values with L = CO, CS, NO+ are calculated for the interactions between the central N2 in the 5Σu+ quintet state and the terminal ligands in the symmetry-adapted (L)2 quintet state, which indicate electron-sharing double bonds between the fragments. The central N–N bond in the latter species is much shorter (between 1.355–1.386 Å) than in N6 (1.450 Å). The lowest ΔEorb value of N2L2 with L = CN− is found for the interactions between N2− in the electronic quartet (4Σu+) state and the terminal (CN)2− ligands as symmetry-adapted quartets. The individual orbital interactions are identified by the corresponding deformation densities and the associated orbitals, which are shown in Fig. S7–S14. The latter electron-sharing interactions also lead to four occupied π valence MOs in all five N2L2 molecules.
In order to compare the N2L2 compounds with each other, we have chosen the same fragments with the central N2 in the excited (1)1Γg singlet state and two terminal N2 fragments in the 1Σg+ electronic ground state as model for the bonding interactions. Table 3 gives the numerical results. The calculated interaction energies ΔEint show the same order CS > CO > CN− > NO+ > N2 as the calculated activation barriers ΔE≠ (Fig. 2), which indicates that the choice of the singlet fragments is a valid model for the trend of the chemical bonds. The orbital (covalent) interactions always make the largest percentage contribution to the chemical bonds, which is particularly high for L = NO+ (75%). The breakdown of ΔEorb into the pairwise orbital interactions reveals that the σ donations through out-of-phase (ΔEorb(1)) and in-phase (ΔEorb(2)) orbital pairs are always the largest components of the total orbital bonding. The percentage contribution of ΔEorb(1) and ΔEorb(2) is higher for L = CN− and lower for L = NO+, which is due to the charges of the ligands. A surprising result concerns the low contribution of the π backdonation ΔEorb(3) and ΔEorb(4) for L = CO, which is smaller than for L = N2. CO is known to be a better π acceptor than N2 in transition metal complexes.26 The peculiar results for the orbital interactions in the neutral N2L2 compounds can be explained with the dominance of the σ donation L→N2←L over π backdonation L←N2→L, which comes to the fore by the calculated partial charges (Fig. 1). The energy of the σ lone-pair HOMO suggests that the donor strength of the ligands has the order CS (ε = −10.1 eV) > CO (ε = −12.2 eV) > N2 (ε = −13.9 eV). But the overall strength of the dative interactions cannot simply be derived from the orbital interactions. The orbital interaction ΔEorb of the negatively charged CN− is significantly weaker (−972.1 kcal mol−1) than that of NO+ (−1088.0 kcal mol−1), but the total interaction energy ΔEint of the former ligand is clearly higher (−301.4 kcal mol−1) than the latter (−202.5 kcal mol−1). The electrostatic interaction ΔEelstat but also the Pauli repulsion ΔEPauli, which makes the largest contribution to ΔEint, are equally relevant for the trend of the interatomic interactions. It has been shown that the Pauli repulsion is the crucial factor for the equilibrium geometry of molecules.27
| Energies | Orbital interaction | L = N2 | L = CO | L = CS | L = NO+ | L = CN− |
|---|---|---|---|---|---|---|
| a The values in parentheses give the percentage contribution to the total attractive interactions ΔEelstat + ΔEorb. b The values in parentheses give the percentage contribution to the total orbital interactions ΔEorb. | ||||||
| ΔEint | −165.0 | −306.0 | −387.0 | −202.5 | −301.4 | |
| ΔEPauli | 1036.0 | 1400.4 | 1542.2 | 1242.3 | 1155.6 | |
| ΔEelstata | −364.9 (30.4%) | −530.8 (31.1%) | −595.4 (30.9%) | −356.9 (24.7%) | −484.9 (33.3%) | |
| ΔEorba | −836.1 (69.6%) | −1175.7 (68.9%) | −1333.8 (69.1%) | −1088.0 (75.3%) | −972.1 (66.7%) | |
| ΔEorb(1)b | L–NN–L in-plane(σ)-bond (+,−) | −328.7 (39.3%) | −483.0 (41.1%) | −536.4 (40.2%) | −373.2 (34.3%) | −405.8 (41.7%) |
| ΔEorb(2)b | L–NN–L in-plane(σ)-bond (+,+) | −296.4 (35.5%) | −434.3 (37.4%) | −493.4 (37.0%) | −322.3 (29.6%) | −400.0 (41.1%) |
| ΔEorb(3)b | L–NN–L out-of-plane(π)-bond (+,−) | −98.6 (11.8%) | −76.1 (6.5%) | −134.6 (10.1%) | −182.1 (16.7%) | −70.4 (7.2%) |
| ΔEorb(4)b | L–NN–L out-of-plane(π)-bond (+,+) | −44.7 (5.3%) | −45.2 (3.8%) | −48.0 (3.6%) | −75.5 (6.9%) | −34.6 (3.6%) |
| ΔEorb(rest)b | −67.7 (8.1%) | −137.1 (11.7%) | −121.4 (9.1%) | −134.9 (12.4%) | −61.3 (6.3%) | |
We also want to comment on the ΔEorb(rest) term, which is comparatively large in the systems with L = CO, CS, NO+. It stems from the relaxation of the fragment orbitals with respect to the isolated species. The central N2 moieties in the latter complexes have significantly shorter N–N distances than in the 1Γg singlet state (1.606 Å at the M06-2X/cc-pVTZ level), and the electronic relaxation is, therefore, larger than in the complexes with L = N2, CN− which have longer central N–N bonds (Fig. 1). The impact of the geometry relaxation on the BDE and the stability of molecules has been pointed out by Bickelhaupt in his activation strain model.28 The strong influence of the fragment relaxation and the geometry of the interacting species becomes obvious when the trend of the interaction energy ΔEint between the frozen fragments (L = CS > CO > CN− > NO+ > N2) is compared with the BDEs that are calculated using the fragments at their equilibrium geometries and electronic ground state (L = CS > CO > CN− > N2 > NO+). Finally, we want to mention that the chemistry of N2(PPh3)2 was recently studied in joint experimental and theoretical works by the groups of Stephan and Grimme, which showed a surprising reactivity of the member of the N2L2 compound class.29
Summary and conclusion
Geometry optimizations of the compounds N2L2 (L = N2, CO, CS, NO+, CN−) at the CCSD(T)/cc-pVTZ level give the energy minimum structures with a trans-periplanar arrangement of the L2 ligands at the N2 unit. Attempts to localize a syn isomer failed, and the calculations gave the trans isomer as the only conformational minimum. Energy calculations suggest that all compounds N2L2 (L = N2, CO, CS, NO+, CN−) may be synthesized and should be observable under appropriate conditions. The complexes with L = N2, CO, NO+, CN− are predicted as thermodynamically unstable for dissociation into N2 + 2 L with ΔG298 value lying in between −257 kcal mol−1 (L = NO+) and −73 kcal mol−1 (L = CO), but the adduct N2(CS)2 is calculated as slightly stable with ΔG298 = 4 kcal mol−1. The homolytic dissociation reaction into two fragments N2L2 → 2 NL is energetically less favorable than the heterolytic fragmentation N2L2 → N2 + 2 L, which proceeds synchronously but asymmetrically. The activation barriers for the fragmentation reaction N2L2 → N2 + 2 L have values between ΔG≠(298 K) = 17 kcal mol−1 for L = N2 and ΔG≠(298 K) = 84 kcal mol−1 for L = CS. The calculated vibrational frequencies show that all molecules N2L2 have a characteristic vibrational mode with the second highest wavenumber νas that comes from the antisymmetric stretch of the ligands L, which is blue shifted for L = CO (Δ = 55 cm−1) and L = NO+ (Δ = 118 cm−1) but it is red shifted for L = CS (Δ = −54 cm−1) and L = CN− (Δ = −133 cm−1) relative to the νas mode of L = N2.The analysis of the bonding situation using the charge distribution reveals that there is a total charge donation L→(N2)←L in all complexes ranging between 1.38 e (L = CN−) and 0.56 e (L = N2), except in the dication with L = NO+, where a small backdonation in reverse direction L→(N2)←L with 0.10 e is calculated. EDA-NOCV calculations of N6 using the central N2 moiety and the terminal N2 ligands as interacting fragments in various electronic states and with different partial charges show that the best description of the bonding situation is given in terms of dative interactions N2→(N2)←N2 between central N2 in the excited (1)1Γg singlet state and the terminal N2 fragments in the 1Σg+ electronic ground state. In contrast to the results for N6, the best description of the complexes with L = CO, CS, NO+ is calculated for the interactions between the central N2 in the 5Σu+ quintet state and the terminal ligands in the symmetry-adapted (L)2 quintet state, which indicates electron-sharing double bonds between the fragments. For N2L2 with L = CN−, it is found that the bonding is best described for the interaction between N2− in the electronic quartet (4Σu+) state and the terminal (L)2− ligand as symmetry-adapted quartet. A comparative analysis of the five N2L2 compounds using the same fragments with central N2 in the excited (1)1Γg singlet state and the terminal L2 fragments in the 1Σg+ electronic ground state reveals that the σ donation L→(N2)←L makes the largest contribution to the stabilizing interactions and that the π backdonation L←(N2)←L is much weaker. In contrast to the common bonding model for N6 using Lewis structures N−![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+
N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N–N
N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N+
N+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N−, the donor–acceptor model N2→(N2)←N2 explains that the lowest activation barrier is found for the concerted cleavage of the two formal double bonds, leading to the experimentally observed dissociation into 3 N2.
N−, the donor–acceptor model N2→(N2)←N2 explains that the lowest activation barrier is found for the concerted cleavage of the two formal double bonds, leading to the experimentally observed dissociation into 3 N2.
Author contributions
G. F. and S. P. conceived the project, wrote the draft and finalized it, Y. L., C. D. and L. X. performed the calculations, Y. L. analyzed the data. All authors took part in the discussions and approved the final version.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc08399k.Acknowledgements
GF acknowledges financial support by Nanjing Tech University. SP is grateful for financial assistance by Jilin University.References
- R. Appel and R. Schöllhorn, Angew. Chem., Int. Ed., 1964, 3, 805 Search PubMed.
- R. Appel, B. Blaser and G. Siegemund, Z. Anorg. Allg. Chem., 1968, 363, 176–182 Search PubMed.
- D. J. Wilson, S. A. Couchman and J. L. Dutton, Inorg. Chem., 2012, 51, 7657–7668 Search PubMed.
- N. Holzmann, D. Dange, C. Jones and G. Frenking, Angew. Chem., Int. Ed., 2013, 52, 3004–3008 CrossRef CAS PubMed.
- M. Reinmuth, C. Neuhäuser, P. Walter, M. Enders, E. Kaifer and H.-J. Himmel, Eur. J. Inorg. Chem., 2011, 2011, 83–90 CrossRef.
- W. Qian, A. Mardyukov and P. R. Schreiner, Nature, 2025, 642, 356–360 Search PubMed.
- C&EN, Am. Chem. Soc., 2025, vol. 103, 23 Search PubMed.
- M. Hochlaf, H. Ndome, D. Hammoutène and M. Vervloet, J. Phys. B: At. Mol. Opt. Phys., 2010, 43, 245101 Search PubMed.
- Q. Wang, S. Pan, S. Lei, J. Jin, G. Deng, G. Wang, L. Zhao, M. Zhou and G. Frenking, Nat. Commun., 2019, 10, 3375 Search PubMed.
- (a) Y. Zhao and D. G. Truhlar, Theor. Chem. Acc., 2008, 120, 215–241 Search PubMed; (b) T. H. Dunning, Jr, J. Chem. Phys., 1989, 90, 1007–1023 Search PubMed.
- G. D. Purvis III and R. J. Bartlett, J. Chem. Phys., 1982, 76, 1910–1918 CrossRef.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, Gaussian 16 Rev. B.01, Wallingford, CT, 2016 Search PubMed.
- I. Mayer, Chem. Phys. Lett., 1983, 97, 270–274 CrossRef CAS.
- E. D. Glendening, C. R. Landis and F. Weinhold, J. Comput. Chem., 2019, 40, 2234–2241 CrossRef CAS PubMed.
- T. Lu and F. Chen, J. Comput. Chem., 2012, 33, 580–592 Search PubMed.
- T. Ziegler and A. Rauk, Theor. Chim. Acta, 1977, 46, 1–10 CrossRef CAS.
- (a) M. Mitoraj and A. Michalak, Organometallics, 2007, 26, 6576–6580 CrossRef CAS; (b) M. Mitoraj and A. Michalak, J. Mol. Model., 2008, 14, 681–687 CrossRef CAS PubMed.
- (a) E. Van Lenthe and E. J. Baerends, J. Comput. Chem., 2003, 24, 1142–1156 CrossRef CAS PubMed; (b) E. van Lenthe, J. G. Snijders and E. J. Baerends, J. Chem. Phys., 1996, 105, 6505–6516 Search PubMed.
- (a) G. te Velde, F. M. Bickelhaupt, E. J. Baerends, C. Fonseca Guerra, S. J. A. van Gisbergen, J. G. Snijders and T. Ziegler, J. Comput. Chem., 2001, 22, 931–967 CrossRef CAS; (b) ADF 2020, ADF SCM, Theoretical Chemistry, Vrije Universiteit, Amsterdam, The Netherlands, https://www.scm.com/ Search PubMed.
- (a) A. Michalak, M. Mitoraj and T. Ziegler, J. Phys. Chem. A, 2008, 112, 1933–1939 CrossRef CAS PubMed; (b) M. P. Mitoraj, A. Michalak and T. Ziegler, J. Chem. Theory Comput., 2009, 5, 962–975 CrossRef CAS PubMed.
- (a) L. Zhao, S. Pan and G. Frenking, Energy Decomposition Analysis of the Chemical Bond: Scope and Limitation in Comprehensive Computational Chemistry, ed. M. Yáñez and R. J. Boyd, Elsevier, Oxford, 2024, vol. 2, pp. 322–361 Search PubMed; (b) F. M. Bickelhaupt, C. Fonseca Guerra, M. Mitoraj, F. Sagan, A. Michalak, S. Pan and G. Frenking, Phys. Chem. Chem. Phys., 2022, 24, 15726–15735 Search PubMed; (c) L. Zhao, M. Hermann, W. H. E. Schwarz and G. Frenking, Nat. Rev. Chem., 2019, 3, 48–63 CrossRef CAS; (d) L. Zhao, S. Pan, N. Holzmann, P. Schwerdtfeger and G. Frenking, Chem. Rev., 2019, 119, 8781–8845 CrossRef CAS PubMed.
- (a) G. Maier, M. Naumann, H. P. Reisenauer and J. Eckwert, Angew Chem. Int. Ed. Engl., 1996, 35, 1696–1697 CrossRef CAS; (b) T. Pasinszki, Phys. Chem. Chem. Phys., 2008, 10, 1411–1418 RSC.
- T. Vörös, G. Bazsó, G. Tarzay and T. Pasinszki, J. Mol. Struct., 2012, 1025, 117–123 CrossRef.
- T. A. Engesser, C. Friedmann, A. Martens, D. Kratzert, P. J. Malinowski and I. Krossing, Chem. Eur. J., 2016, 22, 15085–15094 CrossRef CAS PubMed.
- (a) D. M. Andrada, J. L. Casals-Sainz, Á. Martín Pendás and G. Frenking, Chem. Eur. J., 2018, 24, 9083–9089 CrossRef CAS PubMed; (b) P. Jerabek, P. Schwerdtfeger and G. Frenking, J. Comput. Chem., 2019, 40, 247–264 CrossRef CAS PubMed; (c) R. Saha, S. Pan, G. Merino and P. K. Chattaraj, Angew. Chem., Int. Ed., 2019, 58, 8372–8377 Search PubMed; (d) W. Su, Y. Ma, L. Xiang, J. Wang, S. Wang, L. Zhao, G. Frenking and Q. Ye, Chem. Eur. J., 2021, 27, 10006–10011 CrossRef CAS PubMed; (e) C. Ding, L. Yao, L. Zhao and G. Frenking, Phys. Chem. Chem. Phys., 2022, 24, 16732–16745 RSC; (f) R. Liu, L. Qin, Z. Zhang, L. Zhao, F. Sagan, M. Mitoraj and G. Frenking, Chem. Sci., 2023, 14, 4872–4887 RSC; (g) L. Qin, R. Liu, F. Sagan, Z. Zhang, L. Zhao, M. Mitoraj and G. Frenking, Phys. Chem. Chem. Phys., 2024, 26, 24294–24313 RSC; (h) L. Qin, Y.-q. Liu, R. Liu, X. Yang, Z.-h. Cui, L. Zhao, S. Pan, S. Fau and G. Frenking, Chem. Eur. J., 2024, 30, e202304136 Search PubMed; (i) W.-X. Chen, W.-J. Tian, Z.-S. Li, J.-J. Wang, A. Muñoz-Castro, G. Frenking and Z.-M. Sun, Nat. Synth., 2025, 4, 471–478 Search PubMed; (j) Y.-H. Xu, X. Yang, Y.-N. Yang, L. Zhao, G. Frenking and Z.-M. Sun, Nat. Chem., 2025, 17, 556–563 CrossRef CAS PubMed.
- G. Frenking and N. Fröhlich, Chem. Rev., 2000, 100, 717–774 CrossRef CAS PubMed.
- A. Krapp, F. M. Bickelhaupt and G. Frenking, Chem. Eur. J., 2006, 12, 9196–9216 Search PubMed.
- I. Fernandez and F. M. Bickelhaupt, Chem. Soc. Rev., 2014, 43, 4953–4967 RSC.
- (a) L. Miao, J. Yeung, A. Yeganeh-Salman, Z.-w. Qu, S. Grimme and D. W. Stephan, J. Am. Chem. Soc., 2025, 147, 4720–4725 CrossRef CAS PubMed; (b) V. Bedi, A. N. L. Ocampochua, Z.-W. Qu, S. Grimme and D. W. Stephan, Angew. Chem., Int. Ed., 2025, 64, e202503331 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





