Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Neutral anion-detecting organic cages based on anion–π interactions
Yuyang
Lu†
a,
Ping
Zhou†
ab,
Hua
Tang†
a,
Yating
Wu
a,
Yueyan
Kuang
a,
Ze
Cao
a,
Jiyong
Liu
d,
Guangcheng
Wu
*e,
Hongliang
Chen
 *abc and
Hao
Li
*abc and
Hao
Li
 *ab
*ab
aStoddart Institute of Molecular, Science Department of Chemistry, Zhejiang University, Hangzhou 310058, China. E-mail: lihao2015@zju.edu.cn; hongliang.chen@zju.edu.cn
bZhejiang-Israel Joint Laboratory of Self-Assembling Functional Materials, ZJU-Hangzhou Global Scientific and Technological Innovation Center, Zhejiang University, Hangzhou 311215, China
cBeijing National Laboratory for Molecular Sciences, Beijing 100871, China
dDepartment of Chemistry, Zhejiang University, Hangzhou 310058, China
eDepartment of Chemistry, The University of Hong Kong, Hong Kong SAR 999077, China. E-mail: gcwu2023@hku.hk
First published on 23rd January 2026
Abstract
A series of neutral tetrahedral molecular cages were self-assembled in relatively high yields by condensing a triamino linker with triangular tris-aldehyde precursors. Each tris-aldehyde features a central triazine core, which imparts an electron-deficient cavity that facilitates anion encapsulation through fourfold anion–π interactions. The anion binding affinity is significantly influenced by substituents on the tris-aldehyde precursors: electron-donating groups (e.g., Ph) diminish binding by compromising the electron-deficient nature of the cage, whereas more electron-withdrawing substituents (e.g., Cl, Br, and CF3-Ph) enhance it. Interestingly, the strongly electron-withdrawing fluorine (F) substituents, in close proximity to the binding pocket, unexpectedly diminish binding affinity due to a repulsive field effect. Within each corner of the tetrahedral framework, intramolecular CH–π interactions occur between a phenyl proton ortho to the imine bond and an adjacent phenyl plane. The encapsulation of anionic guests within the cavity perturbed or reinforced these CH–π interactions to varying degrees, producing distinct NMR responses that serve as signatures for different anions.
Introduction
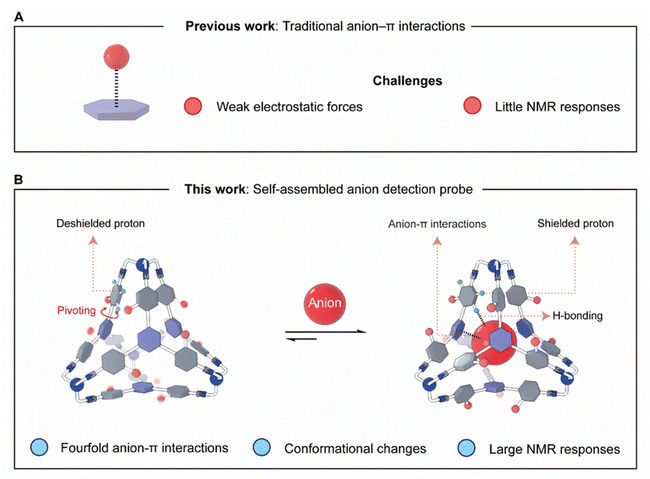
The recognition and detection of anions represent a major focus in supramolecular chemistry due to their critical roles in nature, such as metabolic regulation,1,2 aquatic eutrophication,3,4 and the maintenance of physiological homeostasis.5,6 A common approach of anion detection involves designing artificial anion acceptors that can bind with anionic targets by leveraging hydrogen bonding,7–16 halogen bonding,17–26 electrostatic forces,27 hydrophobic effects,28–32 and coordination to metal ions33–36 or boron atoms37 developed more recently. However, another type of weak supramolecular force—anion–π interaction38–41—was long overlooked in host–guest chemistry. This changed a few decades ago when some theoretical chemists proposed42–44 the feasibility of using electron-deficient π systems to bind negatively charged species (Fig. 1A). Typically, the so-called anion–π interactions are weaker in strength than commonly employed interactions including hydrogen bonds.45,46 As a consequence, anion–π interactions can hardly be used as the primary binding forces that solely drive guest recognition in the absence of other noncovalent forces. For example, many groups obtained47–50 various coordination cages bearing triazine walls, whose cavities can encapsulate anions. Here, electrostatic forces resulting from metal cations and anion–π interactions act as the primary and secondary driving forces, respectively, as inferred from the results that anion recognition can still occur even after the triazine units were replaced with electron rich phenyls.51,52 Developing neutral anion-detecting probes, in which anion–π interactions play the predominant roles, still needs to be exploited. Additionally, unlike hydrogen bonds, which exhibit partial covalent character and induce noticeable shifts in proton NMR spectra, anion–π interactions arise from electrostatic forces and usually result in minimal or no shifts53 in the corresponding 1H NMR spectra, unless hydrogen bonding is also involved.54 Other techniques for probing anion–π interactions also face limitations, including (i) crystallography,55 which only confirms binding in the solid state; (ii) mass spectrometry,56 which provides limited quantitative information; and (iii) UV-Vis absorption and fluorescence57 spectroscopies, which require dramatically different spectra for hosts and the corresponding host–anion complexes, a condition not always met. Therefore, it remains a key challenge to develop artificial hosts with high synthetic efficiency and the ability to produce distinct NMR responses for different anions as their signatures.In our group, a variety of complex molecules58 were synthesized through reversible organic reactions, including imine bond formation, which allows for error correction. For example, a series of tetrahedral molecules59–63 were self-assembled by condensing four equivalents of the trisamino linker tris(2-aminoethyl)amine (TREN) as the vertices with four equivalents of various trisformyl precursors as the faces. In some cases, the yields of these tetrahedral cages are nearly quantitative, partially due to the stabilizing intramolecular CH–π interactions at each tetrahedral corner between an ortho-phenyl proton (relative to an imine bond) and the adjacent phenyl moiety. The critical role that CH–π interactions play in driving the formation of tetrahedra was confirmed by a control experiment in which, when one of the ortho-phenyl protons was replaced by other nonacidic unit, including F, the tetrahedral cages cannot form.61 None of these tetrahedral cages exhibit the ability to accommodate anions, due to the electron rich nature of the phenyl building blocks. We propose that replacing the phenyl units on the faces with electron-deficient triazine moieties could generate a tetrahedron whose cavity is capable of anion recognition via fourfold anion–π interactions (Fig. 1B) in the absence of electrostatic attraction.
Results and discussion
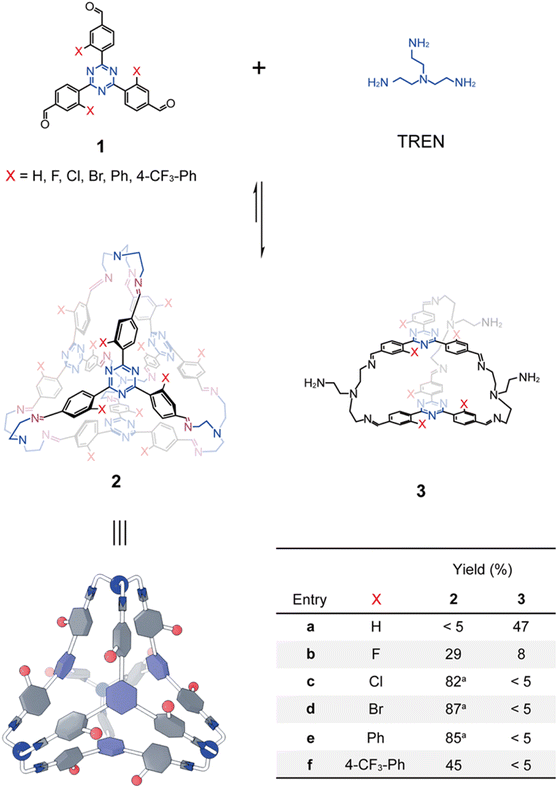
A series of trisformyl precursors each with a central triazine core 1a–1f (Fig. 2) were obtained either through commercial purchase or synthesis. The commercially available 1a was first combined with TREN in CDCl3 in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ratio. The solution was heated at 55 °C for 12 h to allow imine formation to reach equilibrium. However, the 1H NMR spectrum indicated that the expected tetrahedron 2a was not produced. Instead, a triangular prism-shaped molecule (3a) was self-assembled within a library of mixture, consistent with our previously reported results.64 The failure of tetrahedron formation stems from the fact that the tetrahedron formation requires the occurrence of CH–π interactions as the driving force, which necessitates that each benzaldehyde unit adopts an edge-in conformation. Given that the central triazine core orientates in a face-in manner within the tetrahedron framework, this in turn forces each benzaldehyde unit to twist into a specific dihedral angle relative to the central triazine unit. Unfortunately, 1a has a planar conformation, stabilized by intramolecular CH–N hydrogen bonds and phenyl-triazine conjugation. This conformational difference between the precursor 1a and the putative product 2a implies an energetic penalty that disfavors tetrahedron formation.
1 ratio. The solution was heated at 55 °C for 12 h to allow imine formation to reach equilibrium. However, the 1H NMR spectrum indicated that the expected tetrahedron 2a was not produced. Instead, a triangular prism-shaped molecule (3a) was self-assembled within a library of mixture, consistent with our previously reported results.64 The failure of tetrahedron formation stems from the fact that the tetrahedron formation requires the occurrence of CH–π interactions as the driving force, which necessitates that each benzaldehyde unit adopts an edge-in conformation. Given that the central triazine core orientates in a face-in manner within the tetrahedron framework, this in turn forces each benzaldehyde unit to twist into a specific dihedral angle relative to the central triazine unit. Unfortunately, 1a has a planar conformation, stabilized by intramolecular CH–N hydrogen bonds and phenyl-triazine conjugation. This conformational difference between the precursor 1a and the putative product 2a implies an energetic penalty that disfavors tetrahedron formation.
1b–1f are derivatives of 1a, each bearing a functional group at the meta-position relative to the formyl unit of the benzaldehyde function, including F (1b), Cl (1c), Br (1d), Ph (1e), and 4-CF3Ph (1f). These substituents introduce either steric hindrance or coulombic repulsion, thereby forcing each benzaldehyde unit to adopt a twisted dihedral angle relative to the triazine core. We thus proposed that the more twisted conformations of the trisformyl precursors 1b–1f relative to 1a might favor the formation of the corresponding tetrahedral products. The F-substituents in 1b, being the smallest non-hydrogen atom, endow this precursor with the smallest twisting dihedral angle. It is thus predictable that the formation of the tetrahedron 2b would be favored to the least extent. This hypothesis was confirmed, as a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 1b and TREN produced both tetrahedron 2b and prism 3b in a 2
1 mixture of 1b and TREN produced both tetrahedron 2b and prism 3b in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 manner, which was indicated by the 1H NMR spectrum (see Fig. S32 in the SI). By comparing the integrations of the corresponding resonances of the products and the formyl precursor relative to an internal standard, the cyclization yields of 2b and 3b were calculated to be around 29% and 8%, respectively.
1 manner, which was indicated by the 1H NMR spectrum (see Fig. S32 in the SI). By comparing the integrations of the corresponding resonances of the products and the formyl precursor relative to an internal standard, the cyclization yields of 2b and 3b were calculated to be around 29% and 8%, respectively.
As a comparison, combination of each of 1c–1f with TREN in CDCl3 produced only the corresponding tetrahedra 2c–2f as the sole observable products in their 1H NMR spectra (see the SI), further confirming that their preorganized twisted conformations are of importance in self-assembly, by favoring the occurrence of the CH–π interactions. 2c, 2d and 2e were isolated as solid-state compounds via precipitation by adding petroleum ether into the corresponding self-assembly solutions (see the detailed procedures in the SI). The isolated yields of 2c, 2d and 2e were determined to be 82%, 87% and 85%, respectively. These cages were re-dissolved in CDCl3 and their 1H NMR spectra were recorded, confirming their purity and indicating that 2c, 2d and 2e are rather stable or kinetically inert during precipitation, despite the dynamic nature of imine formation. Attempts to isolate 2f using the same procedure were unsuccessful. The 1H NMR spectrum recorded after precipitation showed a few resonances corresponding to impurities that were not detected in the in situ self-assembly solution, suggesting partial degradation of 2f during precipitation. Therefore, the in situ self-assembly solution of 2f was used for the following host–guest binding investigations. The cyclization yield of 2f was determined to be 45% by using an internal standard. The relatively low yield of 2f compared with other cage products was due to the production of some insoluble byproducts, which might be oligomers or polymers. Luckily, 2f is the only observable product in the 1H NMR spectrum, which can be used for host–guest recognition without further purification.
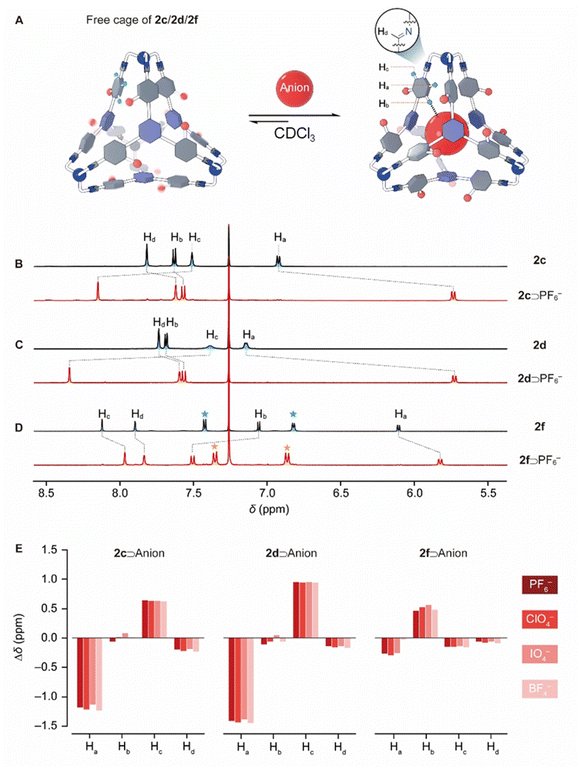
In the 1H NMR spectrum of 2c (Fig. 3B), the resonances corresponding to the protons a and c in the ortho positions relative to the imine bond were observed at around 6.93 and 7.82 ppm, which shifted upfield by 1.04 and 0.59 ppm, respectively, compared to the precursor 1c. These substantial upfield shifts for both protons confirm that each experiences a shielded magnetic environment due to intramolecular CH–π interactions with an adjacent phenyl moiety within the cage framework. The 1H NMR spectra of 2b and 2d exhibited similar patterns, except that in 2d, the resonances corresponding to protons a and c became relatively broad (Fig. 3C), indicating that rotation within 2d occurs at a slower rate compared to 2c. This is attributed to the bulkier Br atom in 2d, which introduces a higher energy barrier for rotation. In contrast, no rotation was observed in the framework of either 2e or 2f. In the 1H NMR spectrum of 2e (Fig. S47), the resonances corresponding to a and c appeared at 6.10 and 8.10 ppm, respectively, which are located in significantly upfield and downfield positions compared with their counterparts in 2c or 2d. This observation indicated that in 2e, proton a is oriented inward toward the cage cavity while proton c is oriented outward, away from the cavity at all times – a conformation driven by the large phenyl substituent, which cannot fit within the cage cavity. The 1H NMR spectrum of 2f (Fig. 3D) is similar to that of 2e, which is predictable given that its 4-CF3Ph substituent is even bulkier.
Given that each tetrahedral cage contains four triazine units, we hypothesized that they are capable of recognizing anionic guests via fourfold anion–π interactions. Various commercially available anions, all introduced as tetrabutylammonium (TBA+) salts, were added to CDCl3 solutions of 2b. Little or no changes were observed in the 1H NMR spectra (Fig. S69), indicating that 2b is incapable of recognizing any of these anions. In the case of 2c and 2d, addition of F−, Cl−, Br−, I−, H2PO4−, NO3−, AcO− and SiF62−, led to little or no changes in the corresponding 1H NMR spectra (Fig. S57 and S62) either. These observations indicated that the cage either 2c or 2d has little or no binding towards these anions, probably because of these less complementary sizes or weaker anion–π interactions. Addition of HSO4− led to degradation of 2c. To our delight, addition of PF6− to 2d led to a concomitant attenuation of the resonances corresponding to the “free” cage and an increase of a new set of resonances corresponding to the complex PF6−⊂2d. After approximately six hours, no further change in the composition of the reaction mixture was observed, indicating that complexation of PF6−⊂2d had reached equilibrium. In the case of the complex PF6−⊂2d, the resonances corresponding to protons a and c were observed at 5.74 and 8.34 ppm, which shifted upfield and downfield by 1.41 and 0.95 ppm compared with those of the “free” cage 2d, respectively (Fig. 3C, red trace). Apparently, these pronounced resonance shifts resulted from the conformational rearrangements upon guest encapsulation: the anionic guest forced the Br atoms and proton c to orient outward, while forcing proton a to reside inward at all times. The binding constant (Ka) for PF6−⊂2d was evaluated to be (5.6 ± 0.5) ×103 M−1 (Table 1) by integrating the resonances corresponding to the “free” cage and the complex in the 1H NMR spectrum of a mixture of PF6− and 2d. By performing similar experiments, a series of anionic complexes including ClO4−⊂2d, IO4−⊂2d and BF4−⊂2d were obtained. In their corresponding 1H NMR spectra (Fig. S63–S66), the resonances corresponding to the proton a were observed to undergo upfield shifts by 1.44, 1.38 and 1.45 ppm, respectively (Fig. 3E, middle). The binding constants of ClO4−⊂2d, IO4−⊂2d and BF4−⊂2d were determined to be (1.0 ± 0.1) ×103, (2.5 ± 0.1) ×102 and (1.6 ± 0.1) ×102 M−1, respectively (Table 1). All the above anionic guests were considered having little or no association with their counterion TBA+ in chlorinated solvents due to their lower basicity.65–67
| K a (M−1) | 2b | 2c | 2d | 2e | 2f |
|---|---|---|---|---|---|
| PF6− | N. D. | 8.2 (±0.3) × 102 | 5.6 (±0.5) × 103 | N. D. | 5.7 (±0.4) × 103 |
| ClO4− | N. D. | 2.2 (±0.1) × 102 | 1.0 (±0.1) × 103 | N. D. | 1.1 (±0.3) × 103 |
| IO4− | N. D. | 7.0 (±0.7) × 101 | 2.5 (±0.1) × 102 | N. D. | 1.2 (±0.2) × 103 |
| BF4− | N. D. | 3.0 (±0.4) × 101 | 1.6 (±0.1) × 102 | N. D. | 9.0 (±0.7) × 101 |
To investigate the influence of the solvophobic effect on binding constants, we also performed binding experiments in more polar solvents, namely CDCl3/CD3SOCD3 (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). The NMR titration results (Fig. S64) revealed that the binding constant of PF6−⊂2d was significantly reduced from 5.6 (±0.5) × 103 M−1 in pure CDCl3 to 1.5 × 102 M−1 in CDCl3/CD3SOCD3 (5
1, v/v). The NMR titration results (Fig. S64) revealed that the binding constant of PF6−⊂2d was significantly reduced from 5.6 (±0.5) × 103 M−1 in pure CDCl3 to 1.5 × 102 M−1 in CDCl3/CD3SOCD3 (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). This experiment indicated that more polar solvent disfavored host–guest binding by shielding its charge and thus suppressing anion–π interactions. We thus reasonably hypothesize that the solvophobic effect might play a less dominant role compared with anion–π interactions in anion recognition. The cage 2c bearing Cl atoms exhibited similar binding behaviors. In the 1H NMR spectra of the complexes PF6−⊂2c, ClO4−⊂2c, IO4−⊂2c and BF4−⊂2c (Fig. S58–S61), the resonances corresponding to proton a underwent upfield shifts by 1.18, 1.22, 1.14 and 1.23 ppm, respectively (Fig. 3E, left), relative to the “free” 2c. The corresponding binding constants for these complexes were determined to be (8.2 ± 0.3) ×102, (2.2 ± 0.1) ×102, (7.0 ± 0.7) ×101 and (3.0 ± 0.4) ×101 M−1, respectively (Table 1).
1, v/v). This experiment indicated that more polar solvent disfavored host–guest binding by shielding its charge and thus suppressing anion–π interactions. We thus reasonably hypothesize that the solvophobic effect might play a less dominant role compared with anion–π interactions in anion recognition. The cage 2c bearing Cl atoms exhibited similar binding behaviors. In the 1H NMR spectra of the complexes PF6−⊂2c, ClO4−⊂2c, IO4−⊂2c and BF4−⊂2c (Fig. S58–S61), the resonances corresponding to proton a underwent upfield shifts by 1.18, 1.22, 1.14 and 1.23 ppm, respectively (Fig. 3E, left), relative to the “free” 2c. The corresponding binding constants for these complexes were determined to be (8.2 ± 0.3) ×102, (2.2 ± 0.1) ×102, (7.0 ± 0.7) ×101 and (3.0 ± 0.4) ×101 M−1, respectively (Table 1).
The addition of these anions with TBA+ counterions to the CDCl3 solutions of 2e led to little or no shifts (Fig. S70). This outcome is somewhat surprising considering that the framework of 2e is identical to that of either 2c or 2d, aside from the different substituents. One possible explanation is that the phenyl substituents in the framework of 2e introduced steric hindrance to the anionic guests, increasing the energy barriers for host–guest association. This possibility was ruled out by the experiments that the combination of 1e and TREN in the presence of the putative anionic guests produced the “free” cage 2e exclusively (Fig. S71). Another explanation is that the phenyl substituents in 2e are relatively electron-rich, which jeopardized the electron deficient nature of the cage 2e. We thus synthesized 2f, an analogue of 2e with more electron-withdrawing 4-CF3Ph units, and tested its anion recognition ability. After adding these anions to the solution of 2f, we observed the formation of host–guest complexes, as evidenced by shifts in the 1H NMR spectra, although the complexation did not reach equilibrium until a few days. Therefore, the complexes PF6−⊂2f, ClO4−⊂2f, IO4−⊂2f and BF4−⊂2f were successfully obtained by self-assembling the cage 2f in the presence of these anionic guests as templates. In the 1H NMR spectra (Fig. 3D and S71–S74) of these complexes, the resonances corresponding to proton a underwent upfield shifts by 0.27, 0.29, 0.26 and 0.30 ppm (Fig. 3E, right), respectively, relative to the “free” 2f. These upfield shifts imply an enhanced shielded magnetic effect occurred to the protons a in the complexes compared with the “free” cage 2f. One possible explanation is that the accommodation of anionic guests leads to the expulsion of a solvent molecule (CDCl3) from the cage cavity, causing the cage framework to shrink slightly and enhanced CH–π interactions. It is noteworthy that the upfield shifts in 2f were less pronounced than those observed in 2c and 2d. This can be attributed to the fact that 2f undergoes fewer conformational changes during anion accommodation, i.e., the protons a are forced to reside inside the cage cavity by the 4-CF3Ph substituents regardless of whether anions are present. Ka values of PF6−⊂2f, ClO4−⊂2f, IO4−⊂2f and BF4−⊂2f were determined to be (5.7 ± 0.4) ×103, (1.1 ± 0.3) ×103, (1.2 ± 0.2) ×103 and (9.0 ± 0.7) ×101 M−1, respectively (Table 1), by integrating the corresponding resonances for both the complexes and the “free” cage.
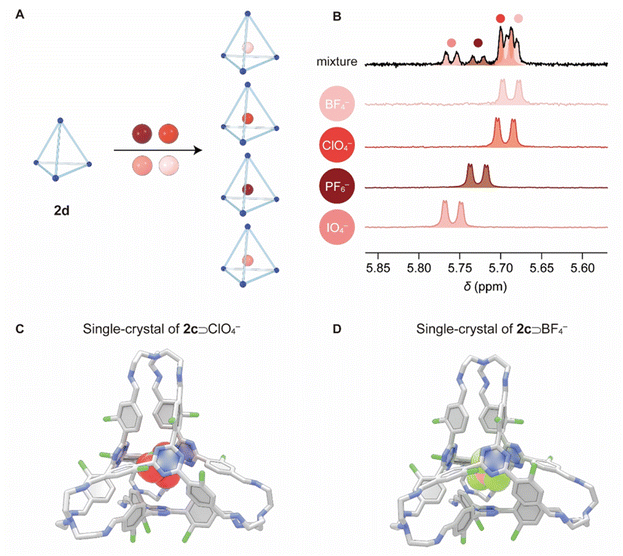
The ability of cages 2c, 2d, and 2f to produce distinct NMR responses for different anions encouraged us to test their utility as probes to detect multiple anions simultaneously when they are present as a mixture (Fig. 4A). A mixture of TBA+ salts of PF6−, BF4−, ClO4−, and IO4− (each at 3 mM) was added to a CDCl3 solution of 2d (0.5 mM). After the host–guest complexes reached equilibrium, the 1H NMR spectrum was recorded. The resonances corresponding to all four complexes namely PF6−⊂2d, ClO4−⊂2d, IO4−⊂2d and BF4−⊂2d were clearly observed (Fig. 4B). However, for some unknown reasons, the NMR integral ratios corresponding to different complexes do not match the binding constants. The probing ability is a key advantage resulting from the confined cavity of 2d, which led to slow exchange between the “free” cage and each of the different complexes on the timescale of 1H NMR spectroscopy. Such discrimination is typically unattainable in traditional systems with “open” structures, such as those reported by Wang,68,69 where complexes undergo fast association/dissociation exchange on the NMR timescale, leading to averaged signals for mixtures. The ability of our cage systems to detect multiple anions within a library of mixture simultaneously is reminiscent of the bambusuril rings reported by Sindelar et al.70
To gain a more quantitative understanding of the kinetic parameters controlling the formation and dissociation of the cage–anion complexes, we employed 1H NMR spectroscopy (Fig. S76–S108) to monitor the change in the composition of CDCl3 solutions initially containing the “free” cages (0.3 mM) after the addition of an excess of anionic guests (3–9 mM). The observed pseudo-first-order rate constants (kobs) were first calculated from plots of ln([A]0/[A]) versus time, where [A]0 and [A] represent the concentrations of the “free” cage at the start and at the given time after anion addition, respectively. Plots of kobsversus the initial anion concentrations were then constructed. The rates of association (kf) were defined by the slopes of these plots, while the rates of dissociation (kb) were calculated using the equation kb = kf/Ka. For PF6−⊂2d, ClO4−⊂2d, IO4−⊂2d and BF4−⊂2d, kf values were determined to be 30.8, 18.3, 2.3, and 11.3 M−1 h−1, and kb values were determined to be 5.5 × 10−3, 18.3 × 10−3, 9.2 × 10−3, and 70.6 × 10−3 h−1, respectively. In the case of PF6−⊂2f, ClO4−⊂2f, IO4−⊂2f and BF4−⊂2f, kf values were determined to be 1.06, 1.11, 0.33, and 1.28 M−1 h−1, and kb values were determined to be 0.19 × 10−3, 1.0 × 10−3, 0.03 × 10−3, and 14.2 × 10−3 h−1, respectively. For each anion, both kf and kb of 2d are approximately one order of magnitude larger than those of 2f, which is consistent with the fact that, compared with the Br units in 2d, the bulkier 4-CF3Ph substituents in 2f impose greater steric hindrance on the anionic guests.
Diffraction grade single crystals of ClO4−⊂2c·TBA+ (Fig. 4C) and BF4−⊂2c·TBA+ (Fig. 4D) were obtained by vapor diffusion of diisopropyl ether into their corresponding chloroform solutions, which unambiguously confirmed the formation of cage–anion complexes. The solid-state structures align with the 1H NMR spectroscopic results recorded in solution. As expected, all the Cl substituents in the framework of 2c are positioned outside the cage cavity, endowing the cage with a “free” cavity to accommodate the anionic guests. In the case of ClO4−⊂2c and BF4−⊂2c, close contacts were observed between the triazine units in the cages and the oxygen atoms (3.06 Å) in ClO4−, as well as the fluorine atoms (3.06 Å) of BF4−, indicating the occurrence of anion–π interactions. The distances between the protons in the meta positions relative to imine bonds and the oxygen atoms in ClO4− and fluorine atoms in BF4− were measured to be in the range of 2.55–2.83 Å, implying the formation of hydrogen bonds as secondary interactions.
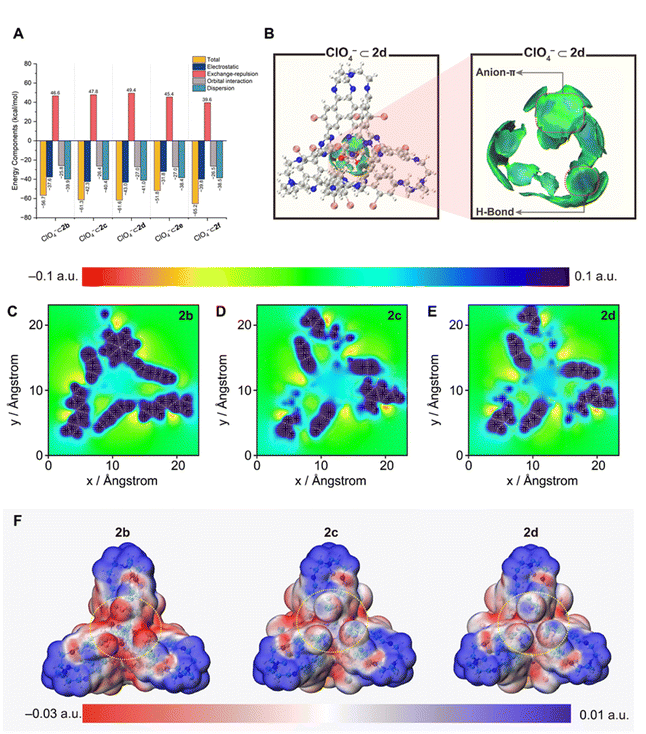
The unexpected trend in anion binding affinity prompted a comprehensive computational investigation to elucidate the physical nature of host–guest interactions. DFT calculations were initially employed to optimize the geometries of both the empty cages and their complexes with ClO4−. Subsequently, energy decomposition analysis (EDA) using the sobEDAw71 method was applied to partition the total interaction energy into electrostatic, exchange-repulsion, orbital, and dispersion terms (Fig. 5A and Table S2). The analysis reveals that electrostatic components are the predominant contributions, which govern the observed total interaction energy trend of |ΔEint(2b)| < |ΔEint(2c)| < |ΔEint(2d)| and |ΔEint(2e)| < |ΔEint(2f)| (Table S2). These computational trends align closely with the experimental binding affinity. Notably, although the molecular cages are charge-neutral, the dominant electrostatic attraction likely arises from their pronounced electron-deficient character. This finding motivated us to probe deeper into the origin of the observed affinity trend.
The independent gradient model based on Hirshfeld partition of molecular density (IGMH)72 analysis using Multiwfn 3.8(dev)73 was employed to visualize the interfragmental interactions within the complexes. The resulting isosurfaces (Fig. 5B and S112) display broad, mainly green regions between ClO4− and the triazine rings, indicative of attractive anion–π interactions with a substantial dispersion character. Additionally, stronger CH–O hydrogen bond interactions are also evident as the predominately blue isosurface regions between ClO4− and the inward-pointing protons at the meta-position of the substituent groups. To quantify the contributions of these interactions, we performed quantitative assessments based on calculated quadrupole moment (for anion–π interactions) and AIM topology analysis74 (for hydrogen bonds) (see SI, Section 6). The average quadrupole moments (Table S3) of the triazine rings for the empty cages do not correlate with the experimental affinity trend. In contrast, the cumulative hydrogen-bond energies derived from AIM analysis (Table S4) show better agreement, yet they account for only about 30% of the total interaction energy. These findings suggest that neither anion–π interactions nor hydrogen bonding serves as the primary determinant of the observed binding trend.
In an attempt to further probe the electrostatic character of the cage cavity, we analyzed the electrostatic potential (ESP) distribution75 of the empty cages. The ESP map (Fig. 5C–E and S109), visualized on a cross-section 3 Å above the triazine plane, reveals a pronounced electropositive region within the cage cavity. Intriguingly, the intensity of this electropositive potential exhibits a clear inverse correlation with the electronegativity of the substituent groups, with the most electronegative F substituent cage 2b having the weakest electropositive cavity. In order to clarify this counterintuitive observation, we derived restrained electrostatic potential (RESP) charges76 for each atom. The analysis (Table S6) reveals a slight increase in the net positive charge on the triazine fragments from 2d to 2b, consistent with the expected inductive effect applied by the electronegative halogen substituents. Concomitantly, the twelve halogen atoms exhibit substantial negative charges, with average values increasing in the order: 2d (−0.054 a.u.) < 2c (−0.083 a.u.) < 2b (−0.22 a.u.), as observed (Fig. 5F) on the ESP maps, where electron-rich regions exist over the surface of the halogen atoms.
The significant negative charge localized on the fluorine atoms in 2b motivated us to investigate their electrostatic influence by computing the electrostatic potential energy (Table S7) between cage components and an imaginary unit (−1) charge placed at the center of the cage. While the triazine units generated a notable attractive potential, it is effectively overshadowed by the repulsive potentials arising from the halogen atoms. Consequently, the net electrostatic repulsion follows the order: 2b > 2c > 2d, which aligns with the experimental anion affinity series and rationalizes the primacy of the repulsive field effect exerted by the adjacent halogen atoms in governing anion binding trends.
Finally, the affinity trends for cages 2e and 2f are explained by complementary mechanisms. The absence of observable anion affinity of 2e results from the electron-donating inductive effect of the Ph groups that significantly weaken the overall electrostatic term of attraction (Fig. 5A). In contrast, the strong binding affinity of 2f, where remote CF3 groups induce a negligible field effect, cannot be explained solely by an electron-withdrawing inductive effect. Instead, the enhanced affinity of 2f is also attributable to the marked reduction in Pauli repulsion (Table S2). This effect arises from the decreased overlap between the occupied orbitals of the anion and 2f, a consequence of orbital polarization induced by the electron-withdrawing CF3 groups.
Conclusions
To summarize, condensation of a trisamino linker with a series of trisformyl precursors yielded five tetrahedral cages in modest to high yields. Each cage features four electron-deficient faces each containing a triazine unit. Three of these cages utilize their cavities to accommodate anions of complementary sizes through fourfold anion–π interactions. The substituents—all grafted at the meta positions relative to the formyl groups—played crucial roles in both self-assembly and anion recognition. First, they preorganized each benzaldehyde unit into a twisted dihedral angle relative to the central triazine core in the precursors. This conformation favored intramolecular CH–π interactions, which served as the key driving force for tetrahedral cage formation. Second, the electronic nature of these substituents has significant impact on anion affinity. Generally, electron-donating groups (e.g., Ph) diminished binding affinity by reducing electrostatic attraction with the anionic guests. In contrast, these attractive forces were increased in cages with electron-withdrawing substituents (e.g., Br, Cl, and CF3-Ph), leading to enhanced anion binding. Notably, strongly electron-withdrawing substituents (e.g., F) in close proximity to the binding pocket also weaken the binding affinity through a repulsive field effect. The encapsulation of anionic guests induced conformational changes in the cage framework. This, in turn, perturbed or reinforced the intramolecular CH–π interactions at each corner to a varying extent for each anion. These perturbations produced distinct NMR responses that provided characteristic signatures for differentiating between anions.Author contributions
H. L. and Y. L. conceived the concept. Y. L. and H. T. performed the experiments and analyzed the data. P. Z. and G. W. carried out the computational studies. H. C. provided the image models. The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There is no conflict of interest to report.Data availability
CCDC 2483576 and 2483577 contain the supplementary crystallographic data for this paper.77a,bSupplementary information (SI): synthetic procedures, NMR spectra, ESI-MS spectra, computational details, and X-ray structure details. See DOI: https://doi.org/10.1039/d5sc08157b.
Acknowledgements
The research at Zhejiang University was supported by the Starry Night Science Fund of Zhejiang University Shanghai Institute for Advanced Study (No. SN-ZJU-SIAS-006). H. L. also want to thank the support from the Leading Innovation Team grant from Department of Science and Technology of Zhejiang Province (2022R01005), the Natural Science Foundation of Zhejiang Province (No. LZ24B020002) and the National Natural Science Foundation of China (Grant No. 22471240). We thank Prof. Qiaohong He, Dr Yaqin Liu, Dr Lina Gao and Mr Zeling Yu from the Chemistry Instrumentation Center Zhejiang University for the technical support.Notes and references
- X. Yin, J. Zhang, W. Zhao, Z. Liu and J. Wang, Combined Levo-Tetrahydropalmatine and Diphenyleneiodonium Chloride Enhances Antitumor Activity in Hepatocellular Carcinoma, Pharmacol. Res., 2022, 179, 106219 CrossRef CAS PubMed.
- J. O. Lundberg, M. Carlström and E. Weitzberg, Metabolic Effects of Dietary Nitrate in Health and Disease, Cell Metab., 2018, 28, 9–22 Search PubMed.
- H. Yan, Q. Chen, J. Liu, Y. Feng and K. Shih, Phosphorus Recovery through Adsorption by Layered Double Hydroxide Nano-Composites and Transfer into a Struvite-Like Fertilizer, Water Res., 2018, 145, 721–730 CrossRef CAS PubMed.
- M. Long, J. Zhu, X. Wang, S. Hu, J. Zhang, K. Cheng, T. Liu, W. Liu, J. R. Reinfelder, Y. Wu and F. Li, Hematite Enhances Microbial Autotrophic Nitrate Removal in Carbonate and Phosphate-Rich Environments by Increasing Fe(II) Activity, Sci. Total Environ., 2024, 949, 175002 Search PubMed.
- E. Pracucci, R. T. Graham, L. Alberio, G. Nardi, O. Cozzolino, V. Pillai, G. Pasquini, L. Saieva, D. Walsh, S. Landi, J. Zhang, A. J. Trevelyan and G.-M. Ratto, Daily Rhythm in Cortical Chloride Homeostasis Underpins Functional Changes in Visual Cortex Excitability, Nat. Commun., 2023, 14, 7108 CrossRef CAS.
- B. S. Yu, P. Chen, L. H. Nie and S. Z. Yao, Determination of Chloride in Human Body Fluids by Ion Chromatography with Piezoelectric Sensor as Detector, Anal. Lett., 1996, 29, 43–57 CrossRef CAS.
- Y. Liu, W. Zhao, C.-H. Chen and A. H. Flood, Chloride Capture Using a C–H Hydrogen-Bonding Cage, Science, 2019, 365, 159–161 CrossRef CAS PubMed.
- X. Wu, P. Wang, P. Turner, W. Lewis, O. Catal, D. S. Thomas and P. A. Gale, Tetraurea Macrocycles: Aggregation-Driven Binding of Chloride in Aqueous Solutions, Chem, 2019, 5, 1210–1222 CAS.
- M. A. Yawer, V. Havel and V. Sindelar, A Bambusuril Macrocycle That Binds Anions in Water with High Affinity and Selectivity, Angew Chem. Int. Ed. Engl., 2015, 54, 276–279 CrossRef CAS.
- X. Zhao, H. Wang, B. Li, W. Zhang, X. Li, W. Zhao, C. Janiak, A. W. Heard, X.-J. Yang and B. Wu, A Hydrogen-Bonded Ravel Assembled by Anion Coordination, Angew. Chem., Int. Ed., 2022, 61, e202115042 CrossRef CAS.
- H. Wang, S. Fang, G. Wu, Y. Lei, Q. Chen, H. Wang, Y. Wu, C. Lin, X. Hong, S. K. Kim, J. L. Sessler and H. Li, Constraining Homo- and Heteroanion Dimers in Ultraclose Proximity within a Self-Assembled Hexacationic Cage, J. Am. Chem. Soc., 2020, 142, 20182–20190 CrossRef CAS PubMed.
- L. Jing, E. Deplazes, J. K. Clegg and X. Wu, A Charge-Neutral Organic Cage Selectively Binds Strongly Hydrated Sulfate Anions in Water, Nat. Chem., 2024, 16, 335–342 CrossRef CAS PubMed.
- P. A. Gale, J. L. Sessler, V. Král and V. Lynch, Calix[4]Pyrroles: Old yet New Anion-Binding Agents, J. Am. Chem. Soc., 1996, 118, 5140–5141 CrossRef CAS.
- A. P. Bisson, V. M. Lynch, M.-K. C. Monahan and E. V. Anslyn, Recognition of Anions through NH–π Hydrogen Bonds in a Bicyclic Cyclophane—Selectivity for Nitrate, Angew Chem. Int. Ed. Engl., 1997, 36, 2340–2342 CrossRef CAS.
- M. Chvojka, D. Madea, H. Valkenier and V. Šindelář, Tuning Ch Hydrogen Bond-Based Receptors toward Picomolar Anion Affinity Via the Inductive Effect of Distant Substituents, Angew. Chem., Int. Ed., 2024, 63, e202318261 Search PubMed.
- R. Custelcean, A. Bock and B. A. Moyer, Selectivity Principles in Anion Separation by Crystallization of Hydrogen-Bonding Capsules, J. Am. Chem. Soc., 2010, 132, 7177–7185 CrossRef CAS PubMed.
- J. Pancholi and P. D. Beer, Halogen Bonding Motifs for Anion Recognition, Coord. Chem. Rev., 2020, 416, 213281 CrossRef CAS.
- J. T. Wilmore and P. D. Beer, Exploiting the Mechanical Bond Effect for Enhanced Molecular Recognition and Sensing, Adv. Mater., 2024, 36, 2309098 Search PubMed.
- M. J. Langton, S. W. Robinson, I. Marques, V. Félix and P. D. Beer, Halogen Bonding in Water Results in Enhanced Anion Recognition in Acyclic and Rotaxane Hosts, Nat. Chem., 2014, 6, 1039–1043 Search PubMed.
- A. M. S. Riel, D. A. Decato, J. Sun, C. J. Massena, M. J. Jessop and O. B. Berryman, Correction: The Intramolecular Hydrogen Bonded–Halogen Bond: A New Strategy for Preorganization and Enhanced Binding, Chem. Sci., 2018, 9, 6451 Search PubMed.
- M. G. Sarwar, B. Dragisic, L. J. Salsberg, C. Gouliaras and M. S. Taylor, Thermodynamics of Halogen Bonding in Solution: Substituent, Structural, and Solvent Effects, J. Am. Chem. Soc., 2010, 132, 1646–1653 Search PubMed.
- M. G. Sarwar, B. Dragisic, S. Sagoo and M. S. Taylor, A Tridentate Halogen-Bonding Receptor for Tight Binding of Halide Anions, Angew. Chem., Int. Ed., 2010, 49, 1674–1677 CrossRef CAS.
- N. L. Kilah, M. D. Wise, C. J. Serpell, A. L. Thompson, N. G. White, K. E. Christensen and P. D. Beer, Enhancement of Anion Recognition Exhibited by a Halogen-Bonding Rotaxane Host System, J. Am. Chem. Soc., 2010, 132, 11893–11895 CrossRef CAS PubMed.
- F. Zapata, A. Caballero, N. G. White, T. D. W. Claridge, P. J. Costa, V. t. Félix and P. D. Beer, Fluorescent Charge-Assisted Halogen-Bonding Macrocyclic Halo-Imidazolium Receptors for Anion Recognition and Sensing in Aqueous Media, J. Am. Chem. Soc., 2012, 134, 11533–11541 Search PubMed.
- A. Caballero, F. Zapata, N. G. White, P. J. Costa, V. Félix and P. D. Beer, A Halogen-Bonding Catenane for Anion Recognition and Sensing, Angew. Chem., Int. Ed., 2012, 51, 1876–1880 Search PubMed.
- M. Cametti, K. Raatikainen, P. Metrangolo, T. Pilati, G. Terraneo and G. Resnati, 2-Iodo-Imidazolium Receptor Binds Oxoanionsvia Charge-Assisted Halogen Bonding, Org. Biomol. Chem., 2012, 10, 1329–1333 Search PubMed.
- C. Xu, Q. G. Tran, D. Liu, C. Zhai, L. Wojtas and W. Liu, Charge-Assisted Hydrogen Bonding in a Bicyclic Amide Cage: An Effective Approach to Anion Recognition and Catalysis in Water, Chem. Sci., 2024, 15, 16040–16049 RSC.
- Y. Wu, C. Zhang, S. Fang, D. Zhu, Y. Chen, C. Ge, H. Tang and H. Li, A Self-Assembled Cage Binding Iodide Anions over Other Halide Ions in Water, Angew. Chem., Int. Ed., 2022, 61, e202209078 CrossRef CAS PubMed.
- E. G. Percástegui, J. Mosquera and J. R. Nitschke, Anion Exchange Renders Hydrophobic Capsules and Cargoes Water-Soluble, Angew. Chem., Int. Ed., 2017, 56, 9136–9140 CrossRef PubMed.
- W. Liu, L. O. Jones, H. Wu, C. L. Stern, R. A. Sponenburg, G. C. Schatz and J. F. Stoddart, Supramolecular Gold Stripping from Activated Carbon Using A-Cyclodextrin, J. Am. Chem. Soc., 2021, 143, 1984–1992 CrossRef CAS.
- T. Fiala, K. Sleziakova, K. Marsalek, K. Salvadori and V. Sindelar, Thermodynamics of Halide Binding to a Neutral Bambusuril in Water and Organic Solvents, J. Org. Chem., 2018, 83, 1903–1912 CrossRef CAS.
- M. Lisbjerg, B. E. Nielsen, B. O. Milhøj, S. P. A. Sauer and M. Pittelkow, Anion Binding by Biotin[6]Uril in Water, Org. Biomol. Chem., 2015, 13, 369–373 Search PubMed.
- Q.-Y. Hong, B. Huang, M.-X. Wu, J.-Y. Jiang, H.-B. Yang, X.-L. Zhao, G. H. Clever and X. Shi, Self-Assembly, Interlocking, Interconversion and Anion-Binding Catalysis in Phenoxazine-Based Pd2L4 and Pd4L8 Coordination Cages, Nat. Commun., 2025, 16, 2484 Search PubMed.
- S. Löffler, J. Lübben, L. Krause, D. Stalke, B. Dittrich and G. H. Clever, Triggered Exchange of Anionic for Neutral Guests inside a Cationic Coordination Cage, J. Am. Chem. Soc., 2015, 137, 1060–1063 Search PubMed.
- S. Sudan, D. W. Chen, C. Berton, F. Fadaei-Tirani and K. Severin, Synthetic Receptors with Micromolar Affinity for Chloride in Water, Angew. Chem., Int. Ed., 2023, 62, e202218072 CrossRef CAS PubMed.
- W.-L. Jiang, B. Huang, X.-L. Zhao, X. Shi and H.-B. Yang, Strong Halide Anion Binding within the Cavity of a Conformation-Adaptive Phenazine-Based Pd2L4 Cage, Chem, 2023, 9, 2655–2668 CAS.
- S. M. Butler, D. M. Beagan, W. Lewis, N. K. Szymczak and K. A. Jolliffe, Gem-Diboronic Acids: A Motif for Anion Recognition in Competitive Media, Angew. Chem., Int. Ed., 2025, 64, e202502582 Search PubMed.
- A. Frontera, P. Gamez, M. Mascal, T. J. Mooibroek and J. Reedijk, Putting Anion–π Interactions into Perspective, Angew. Chem., Int. Ed., 2011, 50, 9564–9583 Search PubMed.
- P. Gamez, The Anion–π Interaction: Naissance and Establishment of a Peculiar Supramolecular Bond, Inorg. Chem. Front., 2014, 1, 35–43 RSC.
- Y. Cotelle, V. Lebrun, N. Sakai, T. R. Ward and S. Matile, Anion-π Enzymes, ACS Cent. Sci., 2016, 2, 388–393 CrossRef CAS PubMed.
- D.-X. Wang and M.-X. Wang, Exploring Anion−π Interactions and Their Applications in Supramolecular Chemistry, Acc. Chem. Res., 2020, 53, 1364–1380 CrossRef CAS PubMed.
- D. Quiñonero, C. Garau, C. Rotger, A. Frontera, P. Ballester, A. Costa and P. M. Deyà, Angew. Chem., Int. Ed., 2002, 114, 3539–3542 CrossRef.
- M. Mascal, A. Armstrong and M. D. Bartberger, Anion−Aromatic Bonding: A Case for Anion Recognition by π-Acidic Rings, J. Am. Chem. Soc., 2002, 124, 6274–6276 CrossRef CAS.
- I. Alkorta, I. Rozas and J. Elguero, Interaction of Anions with Perfluoro Aromatic Compounds, J. Am. Chem. Soc., 2002, 124, 8593–8598 CrossRef CAS.
- P. Ballester, Experimental Quantification of Anion−π Interactions in Solution Using Neutral Host–Guest Model Systems, Acc. Chem. Res., 2013, 46, 874–884 CrossRef CAS PubMed.
- D.-X. Wang and M.-X. Wang, Anion−π Interactions: Generality, Binding Strength, and Structure, J. Am. Chem. Soc., 2013, 135, 892–897 Search PubMed.
- N. Bar Ziv, C. Chen, B. da Camara, R. R. Julian and R. J. Hooley, Selective Aqueous Anion Recognition in an Anionic Host, iScience, 2024, 27, 111348 Search PubMed.
- A. Ferguson, R. W. Staniland, C. M. Fitchett, M. A. Squire, B. E. Williamson and P. E. Kruger, Variation of Guest Selectivity within [Fe4L4]8+ Tetrahedral Cages through Subtle Modification of the Face-Capping Ligand, Dalton Trans., 2014, 43, 14550–14553 RSC.
- B. da Camara, N. B. Ziv, V. Carta, G. A. Mota Orozco, H.-T. Wu, R. R. Julian and R. J. Hooley, Gated, Selective Anion Exchange in Functionalized Self-Assembled Cage Complexes, Chem.–Eur. J., 2023, 29, e202203588 CrossRef CAS PubMed.
- A. B. Grommet and J. R. Nitschke, Directed Phase Transfer of an FeII4L4 Cage and Encapsulated Cargo, J. Am. Chem. Soc., 2017, 139, 2176–2179 CrossRef CAS PubMed.
- L. Xu, D. Zhang, T. K. Ronson and J. R. Nitschke, Improved Acid Resistance of a Metal–Organic Cage Enables Cargo Release and Exchange between Hosts, Angew. Chem., Int. Ed., 2020, 59, 7435–7438 CrossRef CAS PubMed.
- A. M. Castilla, T. K. Ronson and J. R. Nitschke, Sequence-Dependent Guest Release Triggered by Orthogonal Chemical Signals, J. Am. Chem. Soc., 2016, 138, 2342–2351 CrossRef CAS PubMed.
- D. X. Wang, Q. Y. Zheng, Q. Q. Wang and M. X. Wang, Halide Recognition by Tetraoxacalix[2]Arene[2]Triazine Receptors: Concurrent Noncovalent Halide–π and Lone-Pair–π Interactions in Host–Halide–Water Ternary Complexes, Angew. Chem., Int. Ed., 2008, 47, 7485–7488 CrossRef CAS PubMed.
- S.-Y. Zhuang, Y. Cheng, Q. Zhang, S. Tong and M.-X. Wang, Synthesis of I-Corona[6]Arenes for Selective Anion Binding: Interdependent and Synergistic Anion–π and Hydrogen-Bond Interactions, Angew. Chem., Int. Ed., 2020, 59, 23716–23723 CrossRef PubMed.
- X.-D. Wang, Q.-Q. Wang, Y.-F. Ao and D.-X. Wang, Conformational Control of Oxacalix[3]Arene[3]Triazine with Anion−π Interactions, Cryst. Growth Des., 2018, 18, 2707–2711 CrossRef CAS.
- J. Luo, J. Zhu, D.-H. Tuo, Q. Yuan, L. Wang, X.-B. Wang, Y.-F. Ao, Q.-Q. Wang and D.-X. Wang, Macrocycle-Directed Construction of Tetrahedral Anion–π Receptors for Nesting Anions with Complementary Geometry, Chem.–Eur. J., 2019, 25, 13275–13279 CrossRef CAS PubMed.
- D.-X. Wang, Q.-Y. Zheng, Q.-Q. Wang and M.-X. Wang, Halide Recognition by Tetraoxacalix[2]Arene[2]Triazine Receptors: Concurrent Noncovalent Halide–π and Lone-Pair–π Interactions in Host–Halide–Water Ternary Complexes, Angew. Chem., Int. Ed., 2008, 47, 7485–7488 CrossRef CAS.
- Y. Chen, H. Tang, H. Chen and H. Li, Self-Assembly Via Condensation of Imine or Its N-Substituted Derivatives, Acc. Chem. Res., 2023, 56, 2838–2850 CrossRef CAS PubMed.
- T. Jiao, L. Chen, D. Yang, X. Li, G. Wu, P. Zeng, A. Zhou, Q. Yin, Y. Pan, B. Wu, X. Hong, X. Kong, V. M. Lynch, J. L. Sessler and H. Li, Trapping White Phosphorus within a Purely Organic Molecular Container Produced by Imine Condensation, Angew. Chem., Int. Ed., 2017, 56, 14545–14550 Search PubMed.
- T. Jiao, H. Qu, L. Tong, X. Cao and H. Li, A Self-Assembled Homochiral Radical Cage with Paramagnetic Behaviors, Angew. Chem., Int. Ed., 2021, 60, 9852–9858 Search PubMed.
- Y. Chen, G. Wu, B. Chen, H. Qu, T. Jiao, Y. Li, C. Ge, C. Zhang, L. Liang, X. Zeng, X. Cao, Q. Wang and H. Li, Self-Assembly of a Purely Covalent Cage with Homochirality by Imine Formation in Water, Angew. Chem., Int. Ed., 2021, 60, 18815–18820 CrossRef CAS PubMed.
- Y. Chen, Z. Cao, T. Feng, X. Zhang, Z. Li, X. Dong, S. Huang, Y. Liu, X. Cao, A. C. H. Sue, C. Peng, X. Lin, L. Wang and H. Li, Enantioselective Self-Assembly of a Homochiral Tetrahedral Cage Comprising Only Achiral Precursors, Angew. Chem., Int. Ed., 2024, 63, e202400467 Search PubMed.
- H. Tang, Y. Lu, Y. Qian, C. Ge, J. Liu, H. Chen and H. Li, A Conformationally Adaptable Tetrahedral Cage with Different Guest Encapsulation Models, Chem. Sci., 2025, 16, 10867–10873 Search PubMed.
- T. Jiao, G. Wu, L. Chen, C.-Y. Wang and H. Li, Precursor Control over the Self-Assembly of Organic Cages Via Imine Condensation, J. Org. Chem., 2018, 83, 12404–12410 Search PubMed.
- S. Lee, C.-H. Chen and A. H. Flood, A Pentagonal Cyanostar Macrocycle with Cyanostilbene CH Donors Binds Anions and Forms Dialkylphosphate [3]Rotaxanes, Nat. Chem., 2013, 5, 704–710 Search PubMed.
- M. K. Deliomeroglu, V. M. Lynch and J. L. Sessler, Conformationally Switchable Non-Cyclic Tetrapyrrole Receptors: Synthesis of Tetrakis(1H-Pyrrole-2-Carbaldehyde) Derivatives and Their Anion Binding Properties, Chem. Commun., 2014, 50, 11863–11866 Search PubMed.
- M. K. Deliomeroglu, V. M. Lynch and J. L. Sessler, Non-Cyclic Formylated Dipyrromethanes as Phosphate Anion Receptors, Chem. Sci., 2016, 7, 3843–3850 Search PubMed.
- D. X. Wang, Q. Q. Wang, Y. Han, Y. Wang, Z. T. Huang and M. X. Wang, Versatile Anion–π Interactions between Halides and a Conformationally Rigid Bis(Tetraoxacalix[2]Arene[2]Triazine) Cage and Their Directing Effect on Molecular Assembly, Chem.–Eur. J., 2010, 16, 13053–13057 Search PubMed.
- X.-Y. Wang, J. Zhu, Q.-Q. Wang, Y.-F. Ao and D.-X. Wang, Anion−π-Directed Self-Assembly between Di- and Trisulfonates and a Rigid Molecular Cage with Three Electron-Deficient V-Clefts, Inorg. Chem., 2019, 58, 5980–5987 CrossRef CAS PubMed.
- V. Havel, M. A. Yawer and V. Sindelar, Real-Time Analysis of Multiple Anion Mixtures in Aqueous Media Using a Single Receptor, Chem. Commun., 2015, 51, 4666–4669 RSC.
- T. Lu and Q. Chen, Simple, Efficient, and Universal Energy Decomposition Analysis Method Based on Dispersion-Corrected Density Functional Theory, J. Phys. Chem. A, 2023, 127, 7023–7035 CrossRef CAS PubMed.
- T. Lu and Q. Chen, Independent Gradient Model Based on Hirshfeld Partition: A New Method for Visual Study of Interactions in Chemical Systems, J. Comput. Chem., 2022, 43, 539–555 CrossRef CAS PubMed.
- T. Lu and F. Chen, Multiwfn: A Multifunctional Wavefunction Analyzer, J. Comput. Chem., 2012, 33, 580–592 CrossRef CAS PubMed.
- E. Espinosa, E. Molins and C. Lecomte, Hydrogen Bond Strengths Revealed by Topological Analyses of Experimentally Observed Electron Densities, Chem. Phys. Lett., 1998, 285, 170–173 CrossRef CAS.
- J. Zhang and T. Lu, Efficient Evaluation of Electrostatic Potential with Computerized Optimized Code, Phys. Chem. Chem. Phys., 2021, 23, 20323–20328 RSC.
- C. I. Bayly, P. Cieplak, W. Cornell and P. A. Kollman, A Well-Behaved Electrostatic Potential Based Method Using Charge Restraints for Deriving Atomic Charges: The Resp Model, J. Phys. Chem., 1993, 97, 10269–10280 CrossRef CAS.
- (a) CCDC 2483576: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pccct.; (b) CCDC 2483577: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pccdv.
Footnote |
| † Y. L., P. Z., and H. T. contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |