Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Shining light on the mechanism of photochemical alkene formation in vitamin B12
Alivia
Mukherjee
 a,
Summer Y.
Wu
a,
Summer Y.
Wu
 a,
David J.
Cooper
a,
David J.
Cooper
 ab,
Rachel
Hendrickson
ab,
Rachel
Hendrickson
 b,
Roseanne J.
Sension
b,
Roseanne J.
Sension
 ac,
Nicolai
Lehnert
ac,
Nicolai
Lehnert
 *ab and
James E.
Penner-Hahn
*ab and
James E.
Penner-Hahn
 *ab
*ab
aDepartment of Chemistry, University of Michigan, Ann Arbor, Michigan 48109-1055, USA. E-mail: lehnertn@umich.edu; jeph@umich.edu
bDepartment of Biophysics, University of Michigan, Ann Arbor, Michigan 48109-1055, USA
cDepartment of Physics, University of Michigan, Ann Arbor, Michigan 48109-1040, USA
First published on 23rd January 2026
Abstract
A wide range of proteins and enzymes depend on alkylcobalamins (alkylCbl or vitamin B12) for their activities, owing to the unique, biologically relevant Co–C bond. CarH, a regulatory protein in the bacterial carotenoid biosynthetic pathway, utilizes the photosensitivity of the Co–C bond in adenosylcobalamin (AdoCbl) for gene regulation. B12-dependent reductive dehalogenases rely on chemical Co–C bond cleavage to form a Co(III)–halide bond during catalysis. Ultrafast spectroscopy demonstrates that photolytic Co–C bond cleavage in cobalamins begins with the generation of a Co(II) species and an alkyl radical. Interestingly, both CarH and reductive dehalogenases are thought to generate a highly reactive Co(I) species as part of their reactivity. We have used time-resolved measurements of alkylCbls under single turnover conditions to better characterize this reactivity. We demonstrate that Co(I) can be generated in nearly quantitative yield during anaerobic photolysis of alkylCbls in aqueous solution. Remarkably, the addition of alkyl halide to this Co(I) species does not produce quantitative yield of Co(III)-alkylCbl species as would be expected given the “supernucleophilic” nature of the Co(I) center. Instead, we find a branching pathway which has significant implications in Cbl-dependent enzymes and vitamin B12 based organometallic photochemistry. Finally, we demonstrate that both the final oxidation state of the cobalamin product and the fate of the organic radical formed are solvent-dependent, an observation that has implications for CarH photochemistry.
1 Introduction
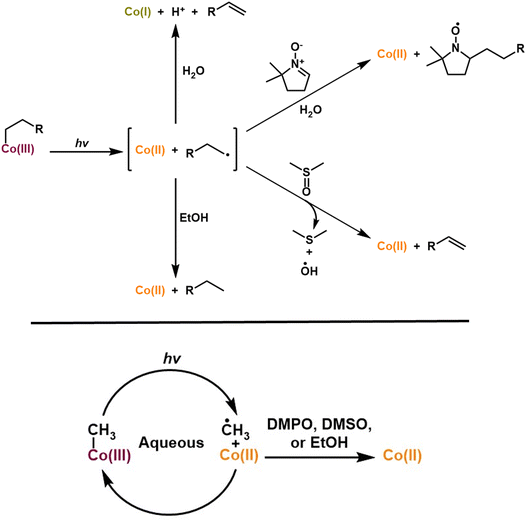
Cobalamin (vitamin B12) is essential in various biological processes, ranging from gene regulation to metabolic transformations.1 Known to be the most structurally intricate vitamin, it is involved in a myriad of different chemical reactions, including carbon skeleton isomerization, dehalogenation, halide coupling, hydrogenation, transmethylation, and cyclopropanation.2 Cobalamin derivatives, particularly alkylcobalamins (alkylCbls), have also garnered significant attention due to their pivotal roles as coenzymes in various biological processes including gene regulatory and dehalogenation pathways.3–7 Among their versatile functions, alkylCbls have been explored for their behavior upon exposure to light.8 Although the photosensitivity of alkyl derivatives of vitamin B12 has long been observed, it has recently been found to be biologically relevant in the regulation of the carotenoid biosynthetic pathway in certain bacteria.1 In the past decade, it has been shown that the photosensitivity of adenosylcobalamin (AdoCbl) plays a crucial role in gene regulation for certain non-photosynthetic bacteria via the transcription regulatory protein, CarH.7,9 The underlying mechanism behind this intriguing photochemistry is not fully understood, making it compelling for further investigation. Upon photolysis, the bond between the central cobalt atom and the adenosyl group cleaves, where the light induced cleavage generates Co(I) and 4′,5′-anhydroadenosine (alkene product). This outcome is contrary to that observed upon AdoCbl photolysis in solution, which typically produces 5′-deoxyadenosyl radicals that cyclize to form 5′,8-cycloadenosine (cyclic product) alongside a Co(II) species (Fig. 1).10–18 This divergence highlights fundamental gaps in our understanding of how protein environments and even experimental reaction conditions like solvents dictate cobalamin reactivity and product selectivity. The alkene product, 4′,5′-anhydroadenosine, has also been observed in adocobinamide (coenzyme B12 with the axial base removed) thermolysis in ethylene glycol. It was found that thermolysis of adocobinamide generates 5′-deoxyadenosyl radicals that are trapped within a solvent cage before undergoing β-H elimination.19,20 Base-mediated Co–C bond cleavage in 5′-deoxyadenosylcobaloxime,21 and AdoCbl bound to the enzymes ethanolamine deaminase and D,L-propanediol hydrolase22 were all reported to give the alkene as the observed product. Hoffman and co-workers have suggested an explanation for these differences. They trapped the 5′-deoxyadenosyl radical that was formed in a radical S-adenosyl-L-methionine enzyme and showed that it had the C5′–H antiperiplanar to the ribose-ring oxygen, potentially stabilizing the radical against elimination of the 4′−H; DFT calculations suggested that this might be an intrinsic feature of AdoCbl.23 In this model, the typical behavior of the 5′-deoxyadenosyl radical is intramolecular cyclization. While this explains the behavior of AdoCbl in solution, it makes the production of the alkene during photolysis of AdoCbl in CarH all the more surprising. Drennan and co-workers favor a homolytic pathway for Co–C bond cleavage, although this would require a different geometry for the 5′-deoxyadenosyl radical in order to explain the alkene product.10,19,24,25 Kutta and co-workers have suggested a heterolytic bond cleavage pathway for the Co–C bond in CarH, leading to the formation of ionic intermediates in the microsecond to millisecond timescales.26 A third possible pathway would be concerted β-elimination where a hydride ion directly forms hydridocobalamin and 4′,5′-anhydroadenosine, bypassing a radical pair intermediate. However, this was deemed unlikely as it would typically require a vacant coordination site on the metal and lacks prior examples in organocobalamins.27,28 The final piece of the puzzle involves the cobalt center. Sension and co-workers showed that with 550 nm excitation, the cobalamin in CarH forms an excited state with an unusually long lifetime (up to 3.8 ns), perhaps implicating the Co directly in the preference for alkene formation.29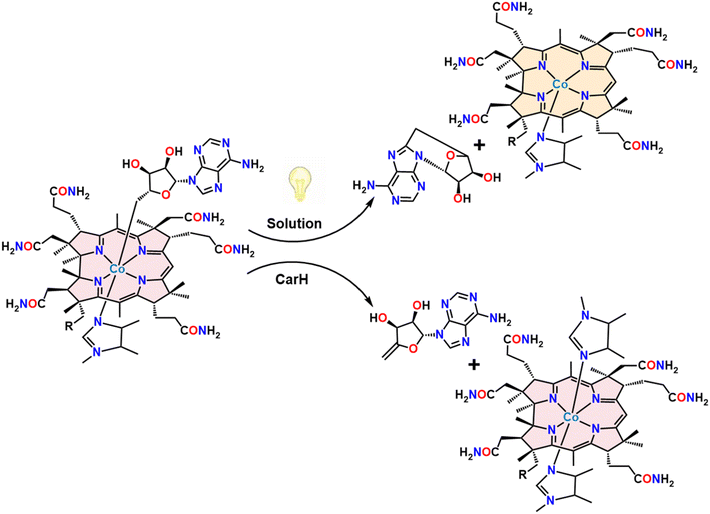 | ||
| Fig. 1 Products obtained by photolysis from free AdoCbl in solution and from AdoCbl bound in the CarH protein. The structures have been truncated for clarity. | ||
Although alkene formation following AdoCbl photolysis was suggested to be unique to CarH-type proteins, the generation of alkene products through the photolytic cleavage of simple alkylCbls is not a novel phenomenon. Over 50 years ago, alkenes were shown to be the product in the photolysis of alkylCbls such as n-ethylcobalamin.21,30–34 It has been proposed that alkene formation occurs via a Co(III)-hydride intermediate (which could be alternatively formulated as a protonated Co(I)); however, definitive evidence for such a species has yet to be reported.10,35–37 Recently, West and co-workers expanded on this proposal by demonstrating the photocatalytic synthesis of a range of primary alkenes from cobalamin and alkyl bromides in organic solvents, noting that the weak base HCO3− was necessary in order to favor alkene over alkane formation.38
In addition to uncertainty over the mechanism of alkene formation, there is also uncertainty over the oxidation state(s) of the Co center. As noted above, ultrafast photochemistry has shown that on the picosecond timescale, photolysis of alkylCbls results in the formation of a geminate pair of Co(II) + alkyl radical, followed by either recombination or cage-escape to form the Co(II) ground state, with no evidence for Co(I), at least not on the nanosecond time scale.39 In contrast, West and co-workers, in their work mentioned earlier, suggested a scheme similar to one of those proposed for CarH, with the Co cycling between Co(III)-alkyl and Co(I). In this mechanism, the reductant sodium borohydride was required, but only to initiate the cycle. This too echoes early observations of the photochemical formation of a Co(I) species alongside Co(II) in an aqueous solution of n-amylcobalamin under anaerobic conditions.8 This earlier work, substantiated by a single UV-Vis absorption spectrum, has not been widely appreciated within the scientific community. However, very recently CarH has been shown to form Co(I) on the millisecond time scale following photolysis.40 Furthermore, the reactivity of cobalamin extends to its crucial role in reductive dehalogenases (RDases), enzymes that play significant roles in enabling organohalide-respiration by catalyzing the removal of halogens from organic substrates. Understanding the mechanism involved in cobalamin-dependent reductive dehalogenation is essential, as this process underpins microbial strategies for detoxifying halogenated environmental pollutants. While several mechanistic pathways have been proposed, all center on Cob(I)alamin as the key reactive species. One proposed pathway involves Co(I) acting as a nucleophile, to form an organocobalt adduct; here, cobalt directly attacks the carbon backbone of the organohalide leading to alkylation of cobalt via the formation of a Co–C bond.6 This intermediate is then thought to be reduced further via electron transfer from an iron-sulfur cluster. Although organocobalt intermediates have been identified in solution studies, their existence during enzymatic catalysis has not been experimentally confirmed.41–43 Alternatively, studies have suggested a long range electron transfer, wherein Cob(I)alamin donates an electron to a substrate generating a radical intermediate, followed by the elimination of the halogen.44–48 A third mechanism proposes that Cob(I)alamin initiates nucleophilic attack on the halogenated substrates, abstracting a halide and forming a Co(III)-halide intermediate.49,50 This mechanism is also suggested by Leys and co-workers, whereby they postulate that carbon–halogen bond cleavage (either homolytically or heterolytically) and subsequent electron and proton transfers yield the dehalogenated product, with the pathway likely depending on the structure of the substrate.51 In all these cases, the active nucleophilic Cob(I)alamin form must be regenerated to sustain the catalytic cycle. Although structural analyses have helped to characterize these enzymes and suggest potential mechanisms, persistent gaps remain in our understanding of the dynamic steps in enzymatic dehalogenation.51 Critical uncertainties remain, such as the real-time sequence of bond-breaking and bond-forming events, and the precise nature of electron transfers involved, which can only be resolved through application of time-resolved spectroscopies or dynamic structural methodologies. Importantly, the presence of external electron donors in experimental setups often complicates the interpretation of Co(I) reactivity, as they can continuously regenerate the active form, masking potentially relevant intermediates.
To better understand the mechanism by which alkylCbl favors alkene formation following photolysis, and how this depends on experimental conditions, we have studied the millisecond to second reactivity of three alkylCbls: the well-studied propylcobalamin (PrCbl) along with two novel alkylCbl derivatives, phenylhexylcobalamin (PhHxCbl) and naphthylethylcobalamin (NapEtCbl). The latter two derivatives were selected for comparatively straightforward product identification since the aromatic groups in these compounds facilitate easy product analysis using gas chromatography coupled with mass spectrometry (GC-MS). Our study investigates the chemically reactive species that participate in the photochemistry of alkene formation from alkylCbls and aims to determine the extent to which the nature of the organic product correlates with the final Co oxidation state under anaerobic conditions. Our approach leverages the use of different solvent conditions to understand how experimental conditions affect the formation of the organic product(s). Our observations indicate that in water, a Co(I) species rapidly forms within seconds after the initiation of photolysis of alkylCbls, whereas Co(II) forms and remains unchanged in organic solvents including dimethyl sulfoxide (DMSO), ethanol (EtOH), and methanol (MeOH). We demonstrate that organic radical chemistry plays a crucial role in determining product selectivity under single turnover conditions. Finally, since we were able to generate a clean sample of cob(I)alamin by photolysis without the need for excess reductant, we investigated the reaction of this species with alkyl halides to obtain insight into the properties of Co(I) as a supernucleophile. Strikingly, we were able to observe a divergent reaction pathway rather than the typical SN2 reaction mechanism exhibited by the Co(I) nucleophile. Our mechanistic study has significant implications in not just cobalamin photochemistry but also its dehalogenase activity.
2 Experimental methods
2.1 Synthesis of alkylcobalamins
The synthesis and workup for these alkylCbls were performed in a dark room under red light. Synthesis and purification of phenylhexylcobalamin (PhHxCbl), naphthylethylcobalamin (NapEtCbl), and n-propylcobalamin (PrCbl) were carried out using a combination of different literature methods.33,34,52–552.2 Static and time-resolved spectral measurements
Static UV-Vis spectra were measured using a dual beam Shimadzu UV-Visible spectrophotometer (Shimadzu UV-2600 spectrometer) at room temperature. Time-resolved spectra were measured using an Avantes diode array (AvaSpec-2048-USB2-UA) coupled to the cuvette via a fiber optic cable. The ultraviolet and visual continua used for the photolysis measurements were generated with a Xenon arc lamp and a combination of various lenses and filters; the lamp was used both as our pump and probe. Using a beam splitter, 96% of the light was directed through a glass UV filter to photolyze the samples (∼1.2 mW at 532 nm), the remaining unfiltered 4% of the beam was used for absorption measurements. The 4% probe beam did not cause any measurable photolysis. Absorbance was calculated as Absorbance = −log10 [(I(t) − Idark − Iscatter)/(Ilamp − Idark)] for t > 0 and as Absorbance = −log10 [(I(t) − Idark)/(Ilamp − Idark)] for t < 0, where Idark is the dark current of the diode array and Ilamp is the signal in the absence of sample. Iscatter represents the scatter from the photolysis beam and was measured as the difference in I(t) averaged 1 s after and 1 s before t = 0.To show the spectral evolution as a function of time, time-resolved difference spectra were generated by calculating the initial absorbance spectrum of the sample prior to exposure to the photolysis beam. This was then subtracted from the absorbance spectra obtained during photolysis.
To investigate the mechanism of alkylCbl synthesis, we used photolytically generated Co(I) and conducted the reactions with 1-bromopropane as the alkyl halide, due to its relatively high solubility in water (∼20 mM). For these experiments, a PrCbl sample (typically between 0.03 to 0.06 mM) was photolyzed in water for 3 minutes to generate the Co(I) species.
2.3 GC-MS Analysis
Samples were prepared and photolyzed under anaerobic conditions as described above. After photolysis, a small-scale liquid–liquid extraction was performed to extract the organic photoproduct into hexanes. Samples photolyzed in EtOH or MeOH were either passed through a small silica column to collect the organic product or extracted into hexane. Gas chromatography-mass spectrometry (GC-MS) analyses were performed using a Shimadzu QP-2010 GC-MS equipped with a 30 meter-long DB-5 column with 0.25 mm ID (see the Supplemental Information, SI, for separation method). Fragmentation patterns were used to identify the peaks for 6-phenyl-1-hexene and phenylhexane. Chromatograms with total ion counts and extracted ion counts at specific m/z values are available in the SI.3 Results and analysis
3.1 Photolysis of alkylCbl in water under anaerobic conditions rapidly converts alkylCbl into a Co(I) species and the alkene product
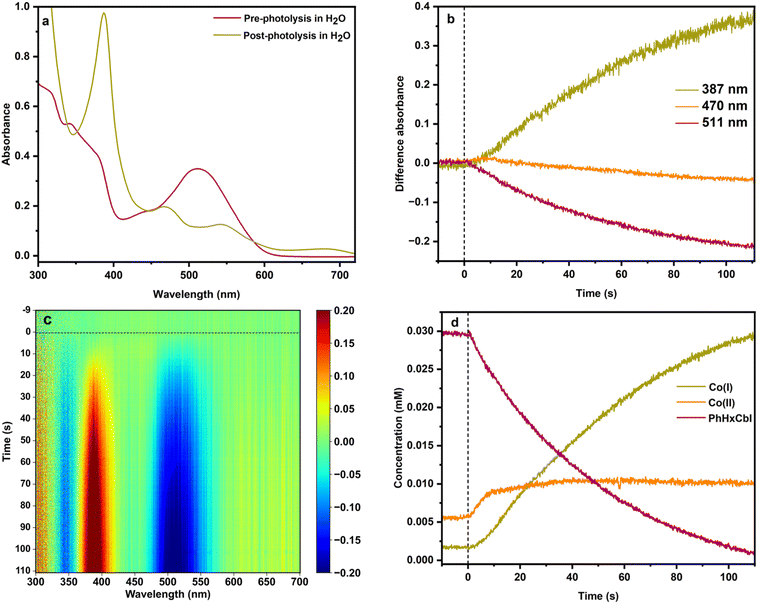
The different oxidation states of cobalamin have distinct UV-Vis spectra that can be used to distinguish between these forms. Representative spectra for Co(I), Co(II), PhHxCbl, PrCbl, cyanocobalamin (CNCbl), aquocobalamin (H2OCbl), and hydroxocobalamin (HOCbl) are shown in Fig. 2. We observed that alkylCbl converts to Co(I) within 2 minutes of photolysis in water under anaerobic conditions. For example, upon illuminating PhHxCbl in water at room temperature under strictly anaerobic conditions, we observed an almost immediate change of the sample color from red to very faint yellow. The UV-Vis absorption spectrum of the photolyzed alkylCbl has an intense peak at 387 nm after 2 minutes of illumination, consistent with Co(I) formation.56Fig. 3 shows the formation of the Co(I) species (indicated by the absorption maxima at 387 nm, 470 nm, 545 nm, and 680 nm) as the PhHxCbl sample is photolyzed in water. This highly reactive Co(I) species slowly oxidizes to a Co(II) species (see Fig. S9). Similarly, photolysis of NapEtCbl and PrCbl in water leads to the generation of cob(I)alamin (Fig. S13 and S15, respectively). In contrast to the rapid formation of Co(I), the oxidation of Co(I) to Co(II) is quite slow, with only ∼17% oxidation to Co(II) over 90 minutes, as obtained from fitting the data in Fig. S9 using references from Fig. 2.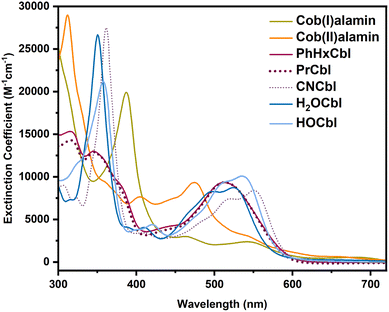 | ||
| Fig. 2 UV-Vis absorption reference spectra for cob(I)alamin, cob(II)alamin, PhHxCbl, PrCbl, CNCbl, H2OCbl, and HOCbl in water. | ||
To better understand the formation of cob(I)alamin, we explored the kinetics of the Co species. Differences in absorption at selected wavelengths that are characteristic of the different oxidation states are shown in Fig. 3b for PhHxCbl in water: Co(I) at 387 nm, Co(II) at 470 nm, and alkylcobalamin Co(III) at 511 nm. The time-resolved difference spectra of PhHxCbl photolyzed in water are shown in Fig. 3c.
The loss of the Co(III) PhHxCbl species starts instantaneously as illustrated by the prompt bleach at 511 nm and 350 nm. In contrast, the growth in Co(I), which gives rise to the peak at 387 nm, is delayed by ∼10 seconds. The same results are observed for the other two alkylCbls we studied (Fig. S24 and S28). There is, however, an increase in Co(II) concentration in the beginning which slows down over time as Co(I) starts forming (Fig. 3d). To quantitate these changes, the spectra were fitted using the Co(I), Co(II), and PhHxCbl reference spectra in Fig. 2 (refer to the Supplemental Information for details on the fitting procedure). As shown in Fig. 3d, these fits confirm that there is an initial increase then a plateau in the Co(II) concentration, and a lag in the formation of the Co(I) species which is the ultimate product of alkylCbl photolysis in water. The rate of Co(III) loss during the photolysis of alkylCbls in water is well described by first order kinetics, consistent with previous studies.57,58 If these experiments are performed in the presence of the radical trap 5,5-dimethyl-1-pyrroline N-oxide (DMPO), Co(I) is not formed and instead the final product is cob(II)alamin (Fig. S57 and Table S7), demonstrating that the alkyl radical is critical for the formation of Co(I). One possible mechanism for this would be proton-coupled electron transfer (PCET) involving the β-hydrogen of the radical. If this happened, a proton release should accompany Co(I) formation. To test for this, we carried out the photolysis in the presence of phenol-red and observed the expected decrease in pH (Fig. S58–S60), suggesting that a PCET mechanism is operable.59
3.2 Photolysis of alkylCbl in organic solvents under anaerobic conditions results in its predominant conversion to a Co(II) species
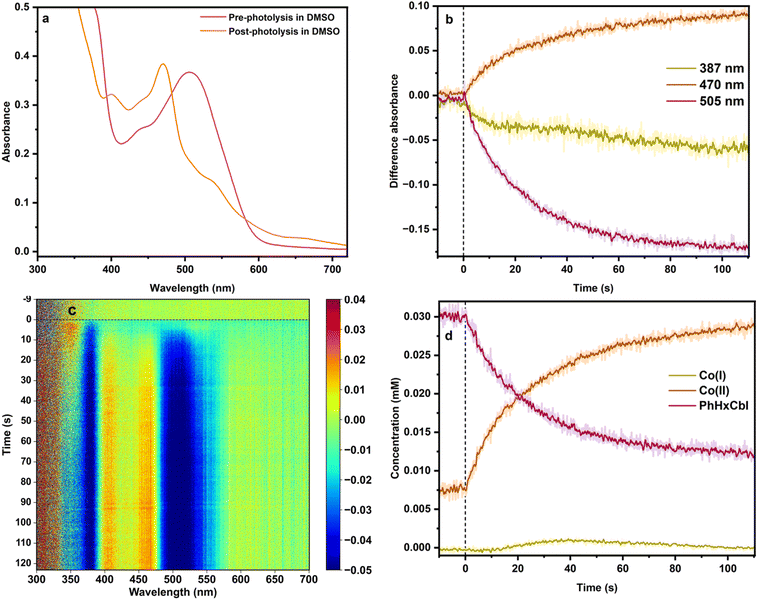
In contrast to the behavior in water, photolysis of alkylCbl in the aprotic solvent DMSO or the protic solvents MeOH and EtOH primarily generates cob(II)alamin (Fig. 4, S17–S21, S25–S27, S30 and S31). As before, there is a prompt loss in absorbance at 505 nm and 360 nm, characteristic of Co(III) spectra in DMSO (Fig. 4c). However, unlike in water, this spectral change is accompanied by an increase in absorbance at ∼400–470 nm, characteristic of cob(II)alamins.26,29,35,60 Analogous results were observed for the other alkylCbls. Quantitative analysis of these data confirms that they reflect primarily a conversion of Co(III) to Co(II), with negligible Co(I) formation.3.3 Solvent effects on cobalamin photochemistry
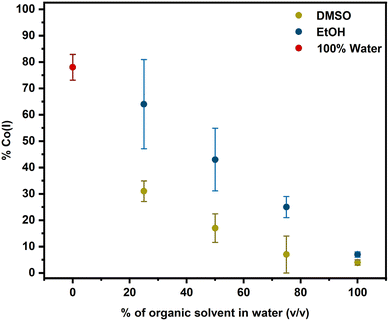
To investigate the solvent dependence further, we conducted photolysis experiments with DMSO/water mixtures. Since our data in Fig. 3 and also ultrafast spectroscopy have shown that Co(II) is the immediate first product of photolysis of alkylCbls, we wondered whether DMSO might be simply acting as a radical quencher—preventing formation of Co(I) from the initially formed [Co(II) + R˙]. To test this hypothesis, we investigated product formation after photolyzing alkylCbls in the presence of increasing amounts of DMSO. Photolysis in DMSO/water mixtures, or in the presence of known radical traps such as TEMPO and DMPO in water, causes a change from Co(I) to the Co(II) product (Fig. 5, S11, and S32–S33). The percentage of the Co(I) species decreases as the percentage of DMSO increases. We observe a similar trend with EtOH, although EtOH seems to be slightly less effective than DMSO at shifting the product towards Co(II) (Fig. S22 and S23). EPR data provides independent confirmation that Co(II) is formed when alkylCbls are photolyzed in DMSO and 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 ethanol/water and matches well with previously reported data (Fig. S50).61,62
50 ethanol/water and matches well with previously reported data (Fig. S50).61,62
3.4 Solvent choice influences the organic product, with water favoring alkene formation and ethanol favoring alkane formation
Aside from the reduced cobalamin, the other product of photolysis of alkylCbls is an organic fragment derived from the alkyl ligand. In addition to reducing Co(II), this radical could potentially dimerize or undergo disproportionation producing an alkene and an alkane.63,64 Alternatively, the radical could react with the solvent either donating an H atom, yielding alkene or accepting an H atom, generating an alkane. For photolysis at our low concentrations, bimolecular reactions of two radicals are comparatively less probable, and we have found no evidence for dimer formation. In order to explore how solvents affect the alkyl radical species, we investigated and compared the organic products that were formed during photolysis of alkylCbls in solvents including water, DMSO, EtOH, and MeOH.When PhHxCbl is photolyzed in 100% water, the predominant product is the alkene (∼98%) (see Table S4). When the photolysis is carried out in water/DMSO mixtures, there is a small decrease in the fraction of alkene, but it still remains the major product (Fig. 6 and Table S4). Ethanol shows a gradual decrease in the alkene fraction, and 100% EtOH gives majority conversion to the alkane (Fig. 6, and Table S5).
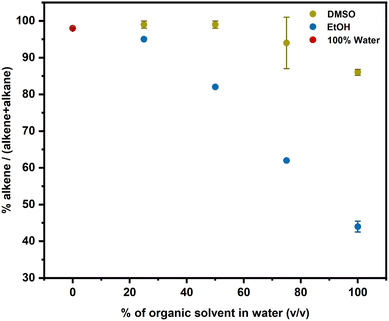 | ||
| Fig. 6 Ratio of alkene to total organic product for the photolysis of PhHxCbl in different solvent mixtures as determined by GC-MS. | ||
Across the spectrum of different solvents, the trend in alkene yield is as follows: water > DMSO > MeOH > EtOH (representative data shown in Fig. S54, and Tables S4, S5). Notably, both EtOH and DMSO prevent Co(I) formation, but increasing EtOH content favors alkane production while increasing DMSO content does not, indicating that the transition from Co(I) to Co(II) does not correlate with increased alkane formation. Both EtOH and MeOH are known to be strong hydrogen atom donors, so in principle they could quench the alkyl radical. Photolysis in ethanol-d1 showed no evidence for incorporation of deuterium into the alkane product. However, we do observe deuterium incorporation into the alkane product when the photolysis is carried out in either ethanol-d6 or methanol-d4. The fraction of the alkane product decreases in deuterated solvent (Table S8) perhaps due to a kinetic isotope effect.
It has been reported that the presence of sodium bicarbonate (NaHCO3) in acetonitrile is essential for the selective formation of alkene products rather than alkane products via photochemistry in a CNCbl based system.38 The insolubility of our alkylCbls and also CNCbl in acetonitrile prevented us from studying single turnover conditions for this reactivity in our system. However, in our tested solvents, we find that under single turnover conditions, NaHCO3 does not have any detectable effect on either the reaction rate, as measured by the loss of the Co(III) reactant, or product selectivity (Fig. S10, S14, S18–S19, S25, S27, and Table S6). It may be that in acetonitrile with excess NaBH4 present, the NaHCO3 is important not for deprotonating a putative H–Co(III) species as previously suggested,38 but rather for removing the β-H from the alkyl radical.
3.5 Mechanistic studies into the formation of alkylcobalamins
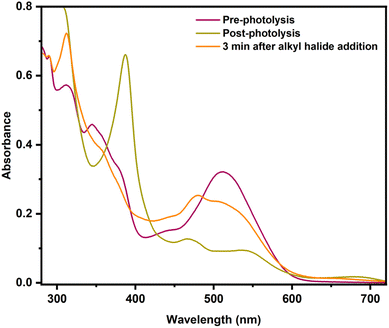
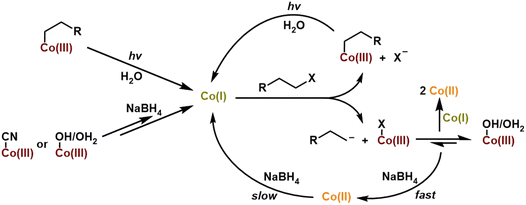
The common way to generate alkylCbls is by reaction of a cob(III)alamin, for example CNCbl or HOCbl, with an alkyl halide in the presence of a large excess of NaBH4 as the reductant.39,52,65 Here, it is believed that the reductant first generates the Co(I) form of cobalamin, which is considered a supernucleophile, and which is presumed to then react with the alkyl halide by oxidative addition to generate the alkylCbl. Since alkylCbls are not reducible by NaBH4, this leads to quantitative product formation. However, actual mechanistic information about this reaction is sparse, since the cob(I)alamin is typically formed by using a large excess of the chemical reductant38,66 or via electrochemistry.56 As shown above, we have been able to use single-turnover photolysis of alkylCbls in water to cleanly generate cob(I)alamin without the need for an external reductant. This provides a unique opportunity for us to investigate the reaction of cob(I)alamin with alkyl halides in single turnover experiments and to obtain mechanistic insight into this reaction. When PrCbl is photolyzed in a saturated stock solution of 1-bromopropane (PrBr) in water (∼20 mM) to generate Co(I) in situ, we obtained primarily a mixture of HOCbl and H2OCbl (Fig. S40, S41b, and Table S3). Photolysis of the longer alkyl chain PhHxCbl in the presence of excess alkyl halide yielded the same observations as seen for PrCbl (Fig. S44). These observations led us to further investigate the mechanism of the reaction between Co(I) and an alkyl halide by adding PrBr to pre-formed Co(I) rather than photolyzing in the presence of PrBr.Since 1-bromopropane has limited solubility in water, the addition of excess (ca. 100 µL) PrBr to water gives a saturated solution, with a PrBr concentration of ∼20 mM. When 100 µL of PrBr is added to photolytically pre-formed Co(I) in the dark, we obtain an ∼1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of Co(II) and the corresponding alkylCbl complex (PrCbl) (Fig. 7). A minor percentage of H2OCbl/HOCbl (∼5%) was also observed. The fact that the addition of RBr to the photogenerated Co(I) species gives a mixture of Co(II) and RCbl suggests a branching pathway.
1 mixture of Co(II) and the corresponding alkylCbl complex (PrCbl) (Fig. 7). A minor percentage of H2OCbl/HOCbl (∼5%) was also observed. The fact that the addition of RBr to the photogenerated Co(I) species gives a mixture of Co(II) and RCbl suggests a branching pathway.
To understand the mechanism behind formation of the Co(II) product upon adding the alkyl halide to the Co(I) species (generated by photolysis), we performed experiments using different concentrations of 1-bromopropane. One explanation for the formation of Co(II) is that Co(I) can react with RBr to form either RCbl + Br− or Co(II) + (R˙ + Br−).67 In this case, the ratio of RCbl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co(II) should be independent of the concentration of RCbl. Alternatively, if the branching was between formation of RCbl + Br− and formation of R− + BrCbl(III) (which would likely undergo ligand exchange to give H2OCbl(III)), the resulting Co(III) species would be expected to undergo rapid comproportionation with Co(I) to generate the observed Co(II) product.67,68 In this case, the ratio of PrCbl to Co(II) should increase as the concentration of RBr increases, since this will increase the likelihood that Co(I) reacts with RBr rather than with Co(III). As shown in Table 1, we added to a 0.03 mM PrCbl solution (a) 100 µL of degassed 1-bromopropane (making the solution saturated with alkyl halide, ∼20 mM) (Fig. 7 and S41d), (b) 100 µL of a 30 mM stock solution of bromopropane (final PrBr concentration: 1.5 mM; 50 equivalents) (Fig. S42), and (c) 50 µL of a 30 mM stock solution of bromopropane (final PrBr concentration: 0.75 mM; 25 equivalents) (Fig. S43). We found that the ratio PrCbl
Co(II) should be independent of the concentration of RCbl. Alternatively, if the branching was between formation of RCbl + Br− and formation of R− + BrCbl(III) (which would likely undergo ligand exchange to give H2OCbl(III)), the resulting Co(III) species would be expected to undergo rapid comproportionation with Co(I) to generate the observed Co(II) product.67,68 In this case, the ratio of PrCbl to Co(II) should increase as the concentration of RBr increases, since this will increase the likelihood that Co(I) reacts with RBr rather than with Co(III). As shown in Table 1, we added to a 0.03 mM PrCbl solution (a) 100 µL of degassed 1-bromopropane (making the solution saturated with alkyl halide, ∼20 mM) (Fig. 7 and S41d), (b) 100 µL of a 30 mM stock solution of bromopropane (final PrBr concentration: 1.5 mM; 50 equivalents) (Fig. S42), and (c) 50 µL of a 30 mM stock solution of bromopropane (final PrBr concentration: 0.75 mM; 25 equivalents) (Fig. S43). We found that the ratio PrCbl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Co(II) decreases at lower RBr concentrations, consistent with the Co(II) that we observe having been formed through comproportionation. We confirmed the feasibility of comproportionation by photolyzing PrCbl in the presence of an equivalent amount of H2OCbl; the only observed product was Co(II) with no indication of Co(I). This demonstrates that comproportionation occurs rapidly, as illustrated in Fig. S48.
Co(II) decreases at lower RBr concentrations, consistent with the Co(II) that we observe having been formed through comproportionation. We confirmed the feasibility of comproportionation by photolyzing PrCbl in the presence of an equivalent amount of H2OCbl; the only observed product was Co(II) with no indication of Co(I). This demonstrates that comproportionation occurs rapidly, as illustrated in Fig. S48.
| Sample | % Co(I) | % Co(II) | % PrCbl | % H2OCbl | % HOCbl | |ΔCo(I)| | ΔCo(II) | ΔPrCbl | |
|---|---|---|---|---|---|---|---|---|---|
| ∼20 mM | |||||||||
| Pre-photolysis | 4 | 7 | 89 | 0 | 0 | ||||
| Post-photolysis | 89 | 7 | 4 | 0 | 0 | ||||
| + RBr + 3 minutes | 1 | 48 | 46 | 0 | 5 | 88 | 41 | 42 | 49% |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||||
| 1.5 mM (50 eq.) | |||||||||
| Pre-photolysis | 5 | 8 | 87 | 0 | 0 | ||||
| Post-photolysis | 80 | 14 | 6 | 0 | 0 | ||||
| + RBr + 3 minutes | 5 | 65 | 29 | 0 | 0 | 75 | 51 | 23 | 33% |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||||||||
| 0.75 mM (25 eq.) | |||||||||
| Pre-photolysis | 3 | 5 | 92 | 0 | 0 | ||||
| Post-photolysis | 93 | 3 | 3 | 0 | 0 | ||||
| + RBr + 3 minutes | 40 | 44 | 16 | 0 | 0 | 53 | 41 | 13 | 26% |
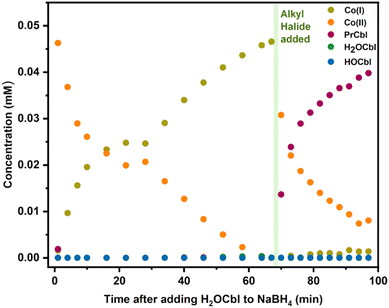
We also investigated the standard reaction conditions applied for the synthesis of alkylCbls, where cob(III)alamin is reduced with excess NaBH4 to generate Co(I), followed by the addition of alkyl halide. We observe that CNCbl initially reacts with NaBH4 to form Co(II), followed by gradual formation of Co(I) (Fig. S49). When excess 1-bromopropane is added, giving a saturated solution of alkyl halide, we initially observe Co(II) formation along with some alkylCbl, followed by gradual consumption of Co(II) and complete formation of alkylCbl (Fig. S49). We confirmed that Co(II) is inert towards 1-bromopropane by photolyzing PrCbl solution in water in the presence of both a radical scavenger, DMPO, and 1-bromopropane. We also photolyzed the alkylCbl independently with DMPO present followed by addition of alkyl halide afterwards. In all cases, only Co(II) was formed, confirming that alkyl halides do not react directly with Co(II) (Fig. S45–S47). For further clarification as to why, under synthetic conditions, cobalamin predominantly shows up as Co(II) immediately upon adding the alkyl halide, we then conducted an experiment adding NaBH4 to an aqueous solution of H2OCbl, which is the other plausible product of the reaction between Co(I) and alkyl halide alongside RCbl, due to the branching pathway. This led to the rapid formation of Co(II) (within the mixing time of the addition), followed by a slower reduction to Co(I) (Fig. 8). This indicates that NaBH4 initially leads to the rapid reduction of H2OCbl to Co(II) through a one-electron reduction process. This is followed by a slower one-electron reduction to Co(I), which in turn reacts rapidly with RBr. Therefore, in the presence of excess NaBH4, one would expect to observe significant cob(II)alamin which would gradually convert to the non-reducible alkylCbl (via Co(I)) as seen in Fig. 8. Despite the possibility of a two-electron reduction reaction, the fast first step and the slower second step explain the prevalence of Co(II) during alkylCbl synthesis.
4 Discussion
In this study, we have explored the photochemistry of alkylcobalamins on the milliseconds to seconds time scale, identifying both the cobalt species and the corresponding organic products that are formed in different solvents. Ultrafast UV-visible transient absorption spectroscopy has shown that on the picosecond time scale, photolysis of the Co(III)–R bond leads to the formation of cob(II)alamin and the organic radical, R˙, which may either recombine to reform the starting alkylcobalamin or undergo cage escape.11,13,14 Our work shows that when the photolysis is performed in water, the ultimate products are Co(I) and a primary alkene, consistent with loss of the β-hydrogen from the radical. This is reminiscent of work by Leys and co-workers showing the formation of Co(I) and an alkene product following photolysis of AdoCbl bound to the cobalamin binding domain of Thermus thermophilus CarH protein.40 However, our data demonstrate that the protein scaffold is not necessarily required for the generation of Co(I) and that, depending on the alkyl group, the protein is also not crucial for generating an alkene product. Rather, both seem to be fundamental properties of alkylCbls when photolyzed in water.We envision the mechanism of the process in water as follows: the initial photolysis generates Co(II) and an alkyl radical. If there is nothing available to quench the radical, it can readily lose the weak β-H to give the alkene product. In water, which is not a good H-atom acceptor, the Co(II) itself can act as a β-H acceptor, ultimately resulting in reduction of the cobalamin to Co(I). This could occur via a proton-coupled electron-transfer (PCET) mechanism, where the Co(II) center is reduced, and a proton is released into the solvent. Similar chemistry has been observed in Cu(II) complexes.69 Consistent with this model, methylcobalamin, which lacks the β-H, shows only limited photochemistry in water under anaerobic conditions, even after extensive photolysis (Fig. 9). This is similar behavior as observed for neopentylcobalamin, where the neopentyl radical, which also lacks a β-H, shows slow photochemistry in water under anaerobic conditions.70 Our finding that protons are released concomitant with Co(I) formation and that a radical trap prevents Co(I) formation are both consistent with a model in which a proton and an electron are both released during the formation of Co(I) following photolysis of alkylCbls in aqueous solutions.
 | ||
| Fig. 9 Photolysis of MeCbl (from left to right) in water, DMSO, and EtOH under anaerobic conditions. | ||
In contrast to the behavior in water, photolysis of alkylCbls in the organic solvents DMSO, EtOH and MeOH generate Co(II) rather than Co(I). It may be that the radical reacts with the solvent before it is able to donate the β-H to Co(II). In DMSO, the alkene remains the main organic product. This is consistent with a study by DiLabio and coworkers,71 finding that DMSO behaves as a hydrogen atom acceptor in reactions with the benzyloxyl radical (C6H5CH2O˙) in a process that involves proton-coupled electron transfer and ultimately generates DMS and a hydroxyl radical. Similar reactivity following the photolysis of alkylcobalamins would give alkene and a DMSO radical while leaving the cobalamin in the Co(II) oxidation state. It has been suggested previously that alcohols could serve as H-atom donors to the alkyl radical8,72 and our deuterium isotope experiments support this notion. Not only do we see deuterium incorporation into the alkane, but in addition we see a decrease in alkane formation, consistent with a kinetic isotope effect.
This proposal is consistent with the observation that MeCbl, which is stable to photolysis in water, can be made to form Co(II) either by adding a known radical trap (TEMPO) (Fig. S33) or by switching to DMSO or EtOH as the solvent (Fig. 9). Neopentylcobalamin similarly shows formation of Co(II) in the presence of 2-propanol,70 and early studies of MeCbl photolysis showed that longer-chain alcohols, which are more sensitive to radical mediated attack, promote the photolysis of MeCbl.8 In this model, if there is a pathway to quench the transient methyl radical Co(II) will form; only in water, where such quenching is not possible, is MeCbl stable to photolysis. This trend was further corroborated in subsequent studies involving ethylcobalamin and n-alkylcobalamin, where increasing the proportion of alcohol in aqueous solution similarly enhanced the photolysis rate and stabilized Co(II).72 The proposed mechanism for alkylCbl photolysis in various solvents is summarized in Fig. 10.
Work by West and coworkers previously found that photochemical alkene formation from alkylCbls was only seen in the presence of sodium bicarbonate.38 In contrast, we find that the organic product that is formed after photolyzing alkylCbls is an alkene as long as (a) there is a β-H present in the substrate/alkyl radical, and (b) there is nothing in solution that can intercept the radical or the β-H prior to cobalt reduction. In our homogeneous solutions, sodium bicarbonate has no effect on the organic product, the cobalt oxidation state, or the reaction rate. It may be that the requirement for bicarbonate in West's reaction is related to the solvent (acetonitrile) since alkylCbls are insoluble in acetonitrile, thus giving a heterogeneous mixture, unlike our homogenous system. It has been speculated that cobalamin reactivity involves a Co(III)-hydride species.10,38 We do not see any evidence for such a species and believe that our data are better explained by the loss of an H-atom (or proton coupled electron transfer) from the organic radical in water. Consistent with this, Co(I) formation is not required for alkene formation if there is a solvent such as DMSO that can act as the H-atom acceptor. As a final note, our results show that alkylCbl photolysis to give alkenes is a fast process in contrast with the slow, multi-hour timescale found in West's system. It may be that what ultimately limits the turnover frequency in the latter is the very low solubility of cobalamin in acetonitrile, which greatly hinders the reformation of the alkylCbl starting material via the chemical reductant after a turnover is finished. In this way, our results not only provide insight into the biologically relevant photochemistry of alkylCbls but also provide mechanistic clues for the development of improved catalytic reactions in organic chemistry that are based on alkylCbl photochemistry.
Finally, we investigated the mechanism of alkylCbl formation from the Co(I) form of cobalamin and an alkyl halide. In the literature, the Co(I) form is often referred to as a supernucleophile, implying that this reaction is a simple oxidative addition, leading to the clean formation of the alkylCbl. However, we find that, at least in water, the reaction Co(I) + RBr → Co(III)–R is not the only reaction. Instead, we observe that one of the major products is a Co(II) species together with some alkylCbl, with the fraction of alkylCbl increasing to half of the cobalamin product at the highest RBr concentrations. This is consistent with the reaction of Co(I) with alkyl halide having a branching point: one path forms alkylCbl (RCbl) while the other gives Br–Co(III) or H2OCbl, which undergoes rapid comproportionation in the presence of Co(I) (see Fig. 11). This quick reaction between excess Co(I) and Co(III) produces Co(II) and hinders complete formation of RCbl unless there is excess reductant present. The observation of an approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio of RCbl to Co(II) at the lowest RBr concentrations (0.75 mM) suggests a branching ratio of 40% RCbl + Br− and 60% BrCbl + R−, taking into consideration the stoichiometry involved (2 Co(I) + RBr → 2 Co(II) in the latter scenario). If Co(I) is generated in situ via photolysis, the steady-state concentration of Co(I) is too low to give significant comproportionation, and we instead observe a mixture of H2OCbl and HOCbl as the only cobalamin products (any RCbl that is formed will be rephotolyzed and thus does not accumulate). In the presence of NaBH4, HOCbl/H2OCbl is rapidly reduced to Co(II), followed by slower formation of Co(I). This Co(I) can then react with an additional alkyl halide, and over multiple such cycles, the alkylCbl product accumulates. Since NaBH4 cannot reduce the alkylCbl product, once it is formed it will accumulate. This reaction somewhat mirrors the previously reported dehalogenation reactions by Co(I) in vitamin B12 and similar complexes.38,45,50,51,56,66,73,74
3 ratio of RCbl to Co(II) at the lowest RBr concentrations (0.75 mM) suggests a branching ratio of 40% RCbl + Br− and 60% BrCbl + R−, taking into consideration the stoichiometry involved (2 Co(I) + RBr → 2 Co(II) in the latter scenario). If Co(I) is generated in situ via photolysis, the steady-state concentration of Co(I) is too low to give significant comproportionation, and we instead observe a mixture of H2OCbl and HOCbl as the only cobalamin products (any RCbl that is formed will be rephotolyzed and thus does not accumulate). In the presence of NaBH4, HOCbl/H2OCbl is rapidly reduced to Co(II), followed by slower formation of Co(I). This Co(I) can then react with an additional alkyl halide, and over multiple such cycles, the alkylCbl product accumulates. Since NaBH4 cannot reduce the alkylCbl product, once it is formed it will accumulate. This reaction somewhat mirrors the previously reported dehalogenation reactions by Co(I) in vitamin B12 and similar complexes.38,45,50,51,56,66,73,74
It is intriguing that HOCbl/H2OCbl is formed as the major product through illumination of alkylCbl in the presence of saturated concentrations of alkyl halide, while we observe almost complete formation of alkylCbl in the dark reaction using chemically reduced CNCbl or H2OCbl with NaBH4. These processes represent two different facets of an intersecting network of cycles. In the illuminated scenario, a mixture of H2OCbl and HOCbl serves as a terminal state because these species are not photoactive—once formed, it remains trapped; any alkylCbl produced can undergo further photolysis, continuing to cycle until complete conversion is achieved. If Co(I) is generated solely through photolysis and RBr is present at a relatively high concentration, the steady-state concentration of Co(I) will remain low, thereby preventing significant comproportionation of Co(III); hence primarily Co(III) accumulates. Conversely, in the dark reaction using NaBH4, alkylCbl becomes the terminal state as it is not reducible by the chemical reductant. With excess reductant present, any Co(III) other than the alkylCbl that is formed will be reduced back to Co(I), eventually leading to the accumulation of alkylCbl, since it cannot be further reduced. These observations align with the mechanism outlined in Fig. 11.
5 Conclusions
Our observations are important for understanding both the photochemical and enzymatic reactivity of cobalamins. Photolysis of alkylCbls in water rapidly generates a Co(I) species, accompanied by the transformation of the initially formed alkyl radical into the alkene. Although Co(II) is the initial photolytic product, the final product of the reaction in water is Co(I), presumably as a result of H-atom transfer. Notably, this occurs independent of a protein environment. Time-resolved spectroscopy and mass spectrometry reveal that both the final cobalt oxidation state and the organic product distribution are strongly influenced by the solvent environment. Although the nature of the organic product depends on the hydrogen atom donor or acceptor properties of the organic solvent, it is not directly linked to the final Co oxidation state and can be better rationalized by radical chemistry occurring upon photolysis. With a system for generating cob(I)alamin photochemically, without the need for chemical reductants, we were able to investigate its reactivity with alkyl bromides, directly observing two mechanistic pathways: formation of Co(III)-alkylcobalamin and a Co(II) product, likely through comproportionation between Co(I) and Co(III)-halide. This mechanistic branching, typically masked by excess reductant, not only broadens our understanding of cobalamin photochemistry but also has implications for the mechanisms of B12-dependent dehalogenases utilized in bacterial bioremediation. Overall, our results highlight the versatility of light-mediated cobalamin chemistry, expanding its relevance beyond bacterial physiology to inform new directions in gene regulation, biocatalysis, synthetic organic chemistry, and the remediation of halogenated environmental contaminants.Author contributions
The manuscript was written through contributions of all authors, who all have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sc07054f.Acknowledgements
This work was supported in part by NSF-CHE 2154157. R.H. was supported by a REU award (NSF-PHY 1757574). We thank Taylor (Eilidh) McClain for helping with photolysis setup and access to the Avantes instrument. We also thank Prof. Stephen Ragsdale and Kareem Aboulhosn for their assistance in the collection of EPR spectroscopic data.References
- M. C. Pérez-Marín, S. Padmanabhan, M. C. Polanco, F. J. Murillo and M. Elías-Arnanz, Vitamin B12 partners the CarH repressor to downregulate a photoinducible promoter in Myxococcus xanthus, Mol. Microbiol., 2008, 67, 804–819 CrossRef PubMed.
- M. Giedyk, K. Goliszewska and D. Gryko, Vitamin B12 catalysed reactions, Chem. Soc. Rev., 2015, 44, 3391–3404 RSC.
- J. Kim, L. Hannibal, C. Gherasim, D. W. Jacobsen and R. Banerjee, A Human Vitamin B12 Trafficking Protein Uses Glutathione Transferase Activity for Processing Alkylcobalamins, J. Biol. Chem., 2009, 284, 33418–33424 CrossRef CAS PubMed.
- L. Hannibal, J. Kim, N. E. Brasch, S. Wang, D. S. Rosenblatt, R. Banerjee and D. W. Jacobsen, Processing of alkylcobalamins in mammalian cells: A role for the MMACHC (cblC) gene product, Mol. Genet. Metab., 2009, 97, 260–266 CrossRef CAS PubMed.
- B. Kräutler, in Advances in Bioorganometallic Chemistry, Hirao, T., Moriuchi, T., e. Elsevier, 2019, p 399–430 Search PubMed.
- G. N. Schrauzer and E. Deutsch, Reactions of cobalt (I) supernucleophiles. The alkylation of vitamin B12s, cobaloximes (I), and related compounds, J. Am. Chem. Soc., 1969, 91, 3341–3350 CrossRef CAS PubMed.
- M. Jost, J. Fernández-Zapata, M. C. Polanco, J. M. Ortiz-Guerrero, P. Y.-T. Chen, G. Kang, S. Padmanabhan, M. Elías-Arnanz and C. L. Drennan, Structural basis for gene regulation by a B12-dependent photoreceptor, Nature, 2015, 526, 536–541 CrossRef CAS PubMed.
- R.-H. Yamada, S. Shimizu and S. Fukui, Factors affecting the anaerobic photolysis of the cobalt-carbon bond of cobalt-methylcobalamin, Biochim. Biophys. Acta (BBA) – General Subjects, 1966, 124, 195–197 CrossRef CAS PubMed.
- H. Takano, Y. Agari, K. Hagiwara, R. Watanabe, R. Yamazaki, T. Beppu, A. Shinkai and K. Ueda, LdrP, a cAMP receptor protein/FNR family transcriptional regulator, serves as a positive regulator for the light-inducible gene cluster in the megaplasmid of Thermus thermophilus, Microbiology, 2014, 160, 2650–2660 CrossRef CAS PubMed.
- M. Jost, J. H. Simpson and C. L. Drennan, The Transcription Factor CarH Safeguards Use of Adenosylcobalamin as a Light Sensor by Altering the Photolysis Products, Biochemistry, 2015, 54, 3231–3234 CrossRef CAS PubMed.
- R. J. Sension, D. A. Harris, A. Stickrath, A. G. Cole, C. C. Fox and E. N. G. Marsh, Time-Resolved Measurements of the Photolysis and Recombination of Adenosylcobalamin Bound to Glutamate Mutase, J. Phys. Chem. B, 2005, 109, 18146–18152 CrossRef CAS PubMed.
- R. J. Sension, A. G. Cole, A. D. Harris, C. C. Fox, N. W. Woodbury, S. Lin and E. N. G. Marsh, Photolysis and Recombination of Adenosylcobalamin Bound to Glutamate Mutase, J. Am. Chem. Soc., 2004, 126, 1598–1599 CrossRef CAS PubMed.
- L. M. Yoder, A. G. Cole, L. A. Walker and R. J. Sension, Time-Resolved Spectroscopic Studies of B12 Coenzymes: Influence of Solvent on the Photolysis of Adenosylcobalamin, J. Phys. Chem. B, 2001, 105, 12180–12188 CrossRef CAS.
- L. A. Walker, J. J. Shiang, N. A. Anderson, S. H. Pullen and R. J. Sension, Time-Resolved Spectroscopic Studies of B12 Coenzymes: The Photolysis and Geminate Recombination of Adenosylcobalamin, J. Am. Chem. Soc., 1998, 120, 7286–7292 CrossRef CAS.
- A. A. Mamun, M. J. Toda, P. Lodowski and P. M. Kozlowski, Photolytic Cleavage of Co–C Bond in Coenzyme B12-Dependent Glutamate Mutase, J. Phys. Chem. B, 2019, 123, 2585–2598 CrossRef CAS PubMed.
- J. F. Endicott and T. L. Netzel, Early events and transient chemistry in the photohomolysis of alkylcobalamins, J. Am. Chem. Soc., 1979, 101, 4000–4002 CrossRef CAS.
- E. Chen and M. R. Chance, Nanosecond transient absorption spectroscopy of coenzyme B12. Quantum yields and spectral dynamics, J. Biol. Chem., 1990, 265, 12987–12994 CrossRef CAS PubMed.
- H. P. C. Hogenkamp, A Cyclic Nucleoside Derived from Coenzyme B12, J. Biol. Chem., 1963, 238, 477–480 CrossRef CAS.
- C. D. Garr and R. G. Finke, Radical cage effects in adocobinamide (axial-base-off coenzyme B12): a simple method for trapping [Ado. .CoII] radical pairs. A new β-H elimination product from the radical pair and measurement of an unprecedentedly large cage-recombination efficiency factor, Fc ≥ 0.94, J. Am. Chem. Soc., 1992, 114, 10440–10445 Search PubMed.
- C. D. Garr, J. M. Sirovatka and R. G. A. Finke, the Base-off Analog of Coenzyme B12 (Adocobalamin). 2.1 Probing the “Base-on” Effect in Coenzyme B12 via Cobalt−Carbon Bond Thermolysis Product and Kinetic Studies as a Function of Exogenous Pyridine Bases, J. Am. Chem. Soc., 1996, 118, 11142–11154 CrossRef CAS.
- G. N. Schrauzer and J. W. Sibert, Coenzyme B12 and coenzyme B12 model compounds in the catalysis of the dehydration of glycols, J. Am. Chem. Soc., 1970, 92, 1022–1030 CrossRef CAS PubMed.
- R. N. Katz, T. M. Vickrey and G. N. Schrauzer, Detection of 4′,5′-Anhydroadenosine as the Cleavage Product of Coenzyme B12 in Functional Holoenzymes, Angew. Chem., Int. Ed. Engl., 1976, 15, 542–543 Search PubMed.
- H. M. Yang, S. Impano, A. S. Byer, R. J. Jodts, K. Yokoyama, W. E. Broderick, J. B. Broderick and B. M. Hoffman, The elusive 5′-deoxyadenosyl radical: Captured and characterized by electron paramagnetic resonance and electron nuclear double resonance spectroscopies, J. Am. Chem. Soc., 2019, 141, 12139–12146 Search PubMed.
- C. D. Garr and R. G. Finke, Adocobalamin (AdoCbl or coenzyme B12) cobalt-carbon bond homolysis radical-cage effects: product, kinetic, mechanistic, and cage efficiency factor (Fc) studies, plus the possibility that coenzyme B12-dependent enzymes function as "ultimate radical cages" and "ultimate radical traps, Inorg. Chem., 1993, 32, 4414–4421 CrossRef CAS.
- R. G. Fink, Vitamin B 12 and B 12 , Proteins, 1998, 383–402 Search PubMed.
- R. J. Kutta, S. J. O. Hardman, L. O. Johannissen, B. Bellina, H. L. Messiha, J. M. Ortiz-Guerrero, M. Elías-Arnanz, S. Padmanabhan, P. Barran, N. S. Scrutton and A. R. Jones, The photochemical mechanism of a B12-dependent photoreceptor protein, Nat. Commun., 2015, 6, 7907 CrossRef CAS PubMed.
- S. M. Chemaly, New light on vitamin B12: The adenosylcobalamin-dependent photoreceptor protein CarH, S. Afr. J. Sci., 2016, 112, 9 CrossRef PubMed.
- Organometallic Mechanisms and Catalysis, Kochi, J. K.,ed Academic Press, 1978, p 246–340 Search PubMed.
- N. A. Miller, A. K. Kaneshiro, A. Konar, R. Alonso-Mori, A. Britz, A. Deb, J. M. Glownia, J. D. Koralek, L. Mallik, J. H. Meadows, L. B. Michocki, T. B. van Driel, M. Koutmos, S. Padmanabhan, M. Elías-Arnanz, K. J. Kubarych, E. N. G. Marsh, J. E. Penner-Hahn and R. J. Sension, The Photoactive Excited State of the B12-Based Photoreceptor CarH, J. Phys. Chem. B, 2020, 124, 10732–10738 CrossRef CAS PubMed.
- D. Dolphin, A. W. Johnson and R. Rodrigo, Reactions of the alkylcobalamins, J. Chem. Soc. (Resumed), 1964, 3186–3193 RSC.
- D. Dolphin, R. L. N. Harris, J. L. Huppatz, A. W. Johnson, I. T. Kay and J. Leng, The synthesis of 5,5-bi-(dipyrromethenyls) and related compounds, J. Chem. Soc. C. Org, 1966, 98–106 RSC.
- G. N. Schrauzer, J. Sibert and R. J. Windgassen, Photochemical and thermal cobalt-carbon bond cleavage in alkylcobalamins and related organometallic compounds. Comparative study, J. Am. Chem. Soc., 1968, 90, 6681–6688 CrossRef CAS PubMed.
- G. N. Schrauzer, L.-P. Lee and J. W. Sibert, Alkylcobalamins and alkylcobaloximes. Electronic structure, spectra, and mechanism of photodealkylation, J. Am. Chem. Soc., 1970, 92, 2997–3005 CrossRef CAS PubMed.
- G. N. Schrauzer, in Coordination Chemistry–XIV, ed Lever, A. B. P., Butterworth-Heinemann, 1973, p 545–565 Search PubMed.
- S. M. Chemaly and J. M. Pratt, The chemistry of vitamin B12. Part 24. Evidence for hydride complexes of cobalt(III) corrinoids, J. Chem. Soc., Dalton trans., 1984, 595–599 RSC.
- M. J. Toda, P. Lodowski, A. A. Mamun and P. M. Kozlowski, Electronic and photolytic properties of hydridocobalamin, J. Photochem. Photobiol. B: Biol., 2021, 224, 112295 CrossRef CAS PubMed.
- D. C. Lacy, G. M. Roberts and J. C. Peters, The Cobalt Hydride that Never Was: Revisiting Schrauzer's “Hydridocobaloxime”, J. Am. Chem. Soc., 2015, 137, 4860–4864 CrossRef CAS PubMed.
- R. Bam, A. S. Pollatos, A. J. Moser and J. G. West, Mild olefin formation via bio-inspired vitamin B12 photocatalysis, Chem. Sci., 2021, 12, 1736–1744 RSC.
- A. G. Cole, L. M. Yoder, J. J. Shiang, N. A. Anderson, L. A. Walker, M. M. Banaszak Holl and R. J. Sension, Time-Resolved Spectroscopic Studies of B12 Coenzymes: A Comparison of the Primary Photolysis Mechanism in Methyl-, Ethyl-, n-Propyl-, and 5‘-Deoxyadenosylcobalamin, J. Am. Chem. Soc., 2002, 124, 434–441 CrossRef CAS PubMed.
- H. Poddar, R. Rios-Santacruz, D. J. Heyes, M. Shanmugam, A. Brookfield, L. O. Johannissen, C. W. Levy, L. N. Jeffreys, S. Zhang, M. Sakuma, J.-P. Colletier, S. Hay, G. Schirò, M. Weik, N. S. Scrutton and D. Leys, Redox driven B12-ligand switch drives CarH photoresponse, Nat. Commun., 2023, 14, 5082 CrossRef CAS PubMed.
- M. Fincker and A. M. Spormann, Biochemistry of Catabolic Reductive Dehalogenation, Annu. Rev. Biochem., 2017, 86, 357–386 CrossRef CAS PubMed.
- S. Lesage, S. Brown and K. Millar, A Different Mechanism for the Reductive Dechlorination of Chlorinated Ethenes: Kinetic and Spectroscopic Evidence, Environ. Sci. Technol., 1998, 32, 2264–2272 Search PubMed.
- K. M. McCauley, S. R. Wilson and W. A. van der Donk, Characterization of Chlorovinylcobalamin, A Putative Intermediate in Reductive Degradation of Chlorinated Ethylenes, J. Am. Chem. Soc., 2003, 125, 4410–4411 Search PubMed.
- R. P. H. Schmitz, J. Wolf, A. Habel, A. Neumann, K. Ploss, A. Svatos, W. Boland and G. Diekert, Evidence for a Radical Mechanism of the Dechlorination of Chlorinated Propenes Mediated by the Tetrachloroethene Reductive Dehalogenase of Sulfurospirillum multivorans, Environ. Sci. Technol., 2007, 41, 7370–7375 Search PubMed.
- J. Shey and W. A. van der Donk, Mechanistic Studies on the Vitamin B12-Catalyzed Dechlorination of Chlorinated Alkenes, J. Am. Chem. Soc., 2000, 122, 12403–12404 CrossRef CAS.
- W. Schumacher, C. Holliger, A. J. B. Zehnder and W. R. Hagen, Redox chemistry of cobalamin and iron-sulfur cofactors in the tetrachloroethene reductase of Dehalobacter restrictus, FEBS Lett., 1997, 409, 421–425 CrossRef CAS PubMed.
- B. Kräutler, W. Fieber, S. Ostermann, M. Fasching, K.-H. Ongania, K. Gruber, C. Kratky, C. Mikl, A. Siebert and G. Diekert, The Cofactor of Tetrachloroethene Reductive Dehalogenase of Dehalospirillum multivorans Is Norpseudo-B12, a New Type of a Natural Corrinoid, Helv. Chim. Acta, 2003, 86, 3698–3716 CrossRef.
- L. Ye, A. Schilhabel, S. Bartram, W. Boland and G. Diekert, Reductive dehalogenation of brominated ethenes by Sulfurospirillum multivorans and Desulfitobacterium hafniense PCE-S, Environ. Microbiol., 2010, 12, 501–509 CrossRef CAS PubMed.
- M. Bommer, C. Kunze, J. Fesseler, T. Schubert, G. Diekert and H. Dobbek, Structural basis for organohalide respiration, Science, 2014, 346, 455–458 CrossRef CAS PubMed.
- C. Kunze, M. Bommer, W. R. Hagen, M. Uksa, H. Dobbek, T. Schubert and G. Diekert, Cobamide-mediated enzymatic reductive dehalogenation via long-range electron transfer, Nat. Commun., 2017, 8, 15858 CrossRef CAS PubMed.
- K. A. P. Payne, C. P. Quezada, K. Fisher, M. S. Dunstan, F. A. Collins, H. Sjuts, C. Levy, S. Hay, S. E. J. Rigby and D. Leys, Reductive dehalogenase structure suggests a mechanism for B12-dependent dehalogenation, Nature, 2015, 517, 513–516 CrossRef CAS PubMed.
- Y. Takahata, A. Nishizawa, I. Kojima, M. Yamanishi and T. S. Toraya, Properties and Microbiological Activity of Hydrophobic Derivatives of Vitamin B12, J. Nutr. Sci. Vitaminol., 1995, 41, 515–526 CrossRef CAS PubMed.
- G. N. Schrauzer and M. Hashimoto, Studies on vitamin B12 and related compounds. 49. Direct synthesis of alkyl-and of ω-carboxyalkylcobalamins from vitamin B12 and aliphatic carboxylic acids under "oxidizing-reducing" conditions, J. Am. Chem. Soc., 1979, 101, 4593–4601 CrossRef CAS.
- G. Schrauzer and R. Windgassen, Cobalamin model compounds. Preparation and reactions of substituted alkyl-and alkenylcobaloximes and biochemical implications, J. Am. Chem. Soc., 1967, 89, 1999–2007 CrossRef CAS.
- G. Schrauzer and R. Holland, Hydridocobalamin and a new synthesis of organocobalt derivatives of vitamin B12, J. Am. Chem. Soc., 1971, 93, 4060–4062 CrossRef CAS PubMed.
- X. Yang, B. H. R. Gerroll, Y. Jiang, A. Kumar, Y. S. Zubi, L. A. Baker and J. C. Lewis, Controlling Non-Native Cobalamin Reactivity and Catalysis in the Transcription Factor CarH, ACS Catal., 2022, 12, 935–942 CrossRef CAS PubMed.
- J. M. Pratt, The chemistry of vitamin B12. Part II. Photochemical reactions, J. Chem. Soc., 1964, 5154–5160 RSC.
- D. H. Dolphin, A. W. Johnson, R. Rodrigo and N. Shaw, The vitamin B12 coenzyme, Pure Appl. Chem., 1963, 7, 539–550 CAS.
- J. T. Jarrett, C. Y. Choi and R. G. Matthews, Changes in Protonation Associated with Substrate Binding and Cob(I)alamin Formation in Cobalamin-Dependent Methionine Synthase, Biochem., 1997, 36, 15739–15748 CrossRef CAS PubMed.
- T. A. Stich, N. R. Buan and T. C. Brunold, Spectroscopic and Computational Studies of Co2+ Corrinoids: Spectral and Electronic Properties of the Biologically Relevant Base-On and Base-Off Forms of Co2+ Cobalamin, J. Am. Chem. Soc., 2004, 126, 9735–9749 CrossRef CAS PubMed.
- R. Padmakumar and R. Banerjee, Evidence from Electron Paramagnetic Resonance Spectroscopy of the Participation of Radical Intermediates in the Reaction Catalyzed by Methylmalonyl-coenzyme A Mutase, J. Biol. Chem., 1995, 270, 9295–9300 CrossRef CAS PubMed.
- S. O. Mansoorabadi, R. Padmakumar, N. Fazliddinova, M. Vlasie, R. Banerjee and G. H. Reed, Characterization of a Succinyl-CoA Radical−Cob(II)alamin Spin Triplet Intermediate in the Reaction Catalyzed by Adenosylcobalamin-Dependent Methylmalonyl-CoA Mutase, Biochem., 2005, 44, 3153–3158 CrossRef CAS PubMed.
- M. J. Gibian and R. C. Corley, Organic radical-radical reactions. Disproportionation vs. combination, Chem. Rev., 1973, 73, 441–464 CrossRef CAS.
- R. Chen, N. E. Intermaggio, J. Xie, J. A. Rossi-Ashton, C. A. Gould, R. T. Martin, J. Alcázar and D. W. C. MacMillan, Alcohol-alcohol cross-coupling enabled by SH2 radical sorting, Science, 2024, 383, 1350–1357 CrossRef CAS PubMed.
- S. M. Chemaly and J. M. Pratt, The chemistry of vitamin B12. Part 17. The effect of steric distortion of the cobalt–carbon bond on the pK values and spectra of organocobalamins, J. Chem. Soc., Dalton Trans., 1980, 2259–2266 RSC.
- A. Wincenciuk, P. Cmoch, M. Giedyk, M. P. Andersson and D. Gryko, Aqueous Micellar Environment Impacts the Co-Catalyzed Phototransformation: A Case Study, J. Am. Chem. Soc., 2024, 146, 19828–19838 CrossRef CAS PubMed.
- B. Heckel and M. Elsner, Exploring Mechanisms of Biotic Chlorinated Alkane Reduction: Evidence of Nucleophilic Substitution (SN2) with Vitamin B12, Environ. Sci. Technol., 2022, 56, 6325–6336 CrossRef CAS PubMed.
- D. Lexa and J. M. Saveant, The electrochemistry of vitamin B12, Acc. Chem. Res., 1983, 16, 235–243 CrossRef CAS.
- J. K. Kochi, A. Bemis and C. L. Jenkins, Mechanism of electron transfer oxidation of alkyl radicals by copper(II) complexes, J. Am. Chem. Soc., 1968, 90, 4616–4625 CrossRef CAS.
- G. N. Schrauzer and J. H. Grate, Sterically induced, spontaneous cobalt-carbon bond homolysis and beta-elimination reactions of primary and secondary organocobalamins, J. Am. Chem. Soc., 1981, 103, 541–546 CrossRef CAS.
- J. A. van Santen, S. Rahemtulla, M. Salamone, M. Bietti and G. A. DiLabio, A computational and experimental re-examination of the reaction of the benzyloxyl radical with DMSO, Comput. Theor. Chem., 2016, 1077, 74–79 CrossRef CAS.
- R.-H. Yamada, S. Shimizu and S. Fukui, Formation of vitamin B12s by anaerobic photolysis of cobalt-alkylcobalamins, Biochim. Biophys. Acta (BBA) - General Subjects, 1966, 124, 197–200 CrossRef CAS PubMed.
- L. O. Johannissen, D. Leys and S. Hay, A common mechanism for coenzyme cobalamin-dependent reductive dehalogenases, Phys. Chem. Chem. Phys., 2017, 19, 6090–6094 RSC.
- Z. Li, E. D. Greenhalgh, U. T. Twahir, A. Kallon, M. Ruetz, K. Warncke, T. C. Brunold and R. Banerjee, Chlorocob(II)alamin Formation Which Enhances the Thiol Oxidase Activity of the B12-Trafficking Protein CblC, Inorg. Chem., 2020, 59, 16065–16072 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |