Open Access Article
Open Access ArticleTime-resolved tracking of hot carrier relaxation in two types of MBenes
Jie Zhaoab,
Qi Zhang*a,
Pan Xiongb,
Chunli Wangc,
Liangzhu Zhang *c,
Jiebo Li
*c,
Jiebo Li d,
Kun Zhao*a,
Ruifeng Lu
d,
Kun Zhao*a,
Ruifeng Lu a,
Kaijun Yuan
a,
Kaijun Yuan *bef and
Xueming Yang
*bef and
Xueming Yang beg
beg
aDepartment of Applied Physics, Nanjing University of Science and Technology, Nanjing 210094, China. E-mail: zhangqi24@njust.edu.cn; zhaokun@njust.edu.cn
bState Key Laboratory of Chemical Reaction Dynamics, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian 116023, China. E-mail: kjyuan@dicp.ac.cn
cSchool of Materials Science and Engineering, East China University of Science and Technology, Shanghai 200237, China. E-mail: zhangliangzhu@ecust.edu.cn
dInstitute of Medical Photonics, Beijing Advanced Innovation Center for Biomedical Engineering, School of Biological Science and Medical Engineering, Beihang University, Beijing 100191, China
eHefei National Laboratory, Hefei 230026, China
fUniversity of Chinese Academy of Sciences, Beijing 101408, China
gDepartment of Chemistry and Center for Advanced Light Source Research, College of Science, Southern University of Science and Technology, Shenzhen 518055, China
First published on 26th March 2026
Abstract
In metallic low-dimensional materials, the interaction between non-equilibrium electrons and phonons underlies various physical phenomena, such as the relaxation process of hot carriers. However, it is unclear how the surface, with or without termination, controls electron–phonon (e–p) interactions in two-dimensional (2D) transition metal borides (MBenes). Herein, ultrafast time-resolved spectroscopy was employed to measure the e–p interaction rate, revealing that MoB MBene without termination exhibits a lower rate than Mo4/3B2−xTz MBene with termination, indicating that its e–p interactions are weakened. Raman spectra and computational calculations indicate that the suppression is attributed to the fact that the excited electrons can only couple with a limited number of vibrational modes. This study paves the way for the tailored manipulation of hot carrier relaxation time, offering opportunities for advanced applications in electronics, optoelectronics, catalysis, and more.
Introduction
Novel two-dimensional (2D) transition metal borides (MBenes) have received significant attention recently as potentially useful materials for a wide range of applications.1–5 Unlike graphene and transition-metal dichalcogenides, MBenes feature numerous surface sites that can be chemically modified, making them analogous to 2D transition metal carbides (MXenes).6 The versatile chemical transformations of surface terminations (i.e., functional groups chemically attached to the surface atoms) in 2D materials further expand the design space for this broad class of functional materials, unlocking new possibilities for advanced applications. Experimental studies suggest that surface termination strongly affects the structure and properties of MXenes.7–10 However, research on MBenes is considerably less developed than that on MXenes.11–15 Research on MBenes is still at an early stage, and layered MBenes with surface terminations tend to generate defects during synthesis. Both terminations and defects not only compromise the stability and electronic structure of MBenes but also limit their performance in energy conversion and transport. Previous studies have shown that MBenes without surface termination exhibit outstanding electrical performance and are more stable than MXenes,16 thereby offering greater advantages in energy storage17 and catalysis.18 Molybdenum-based boride MBenes19 have been predicted to exhibit exceptional electronic conductivity, catalytic activity, high stability, thermoelectric performance, and superconductivity.17 Therefore, the exploration and development of MBenes without termination has become an emerging and critical research direction.The interactions between electrons and lattice vibrations (phonons), a phenomenon known as electron–phonon (e–p) interactions, are being increasingly studied because they are linked to electrical and thermal transport in molecular and condensed phase systems.20–23 The non-equilibrium e–p interaction plays a key role in hot carrier lifetimes,24 carrier mobility,25–27 and thermal conductivity22,28–30 of transition metal materials28 and MXenes.27,29,31 Surface chemistry, especially surface termination, has been shown to critically influence carrier dynamics and electron–phonon interactions in low-dimensional materials.7,18,21,32–34 So far, non-equilibrium e–p interactions have not been considered in the theoretical and experimental analyses on MBene because the investigation is a significant challenge due to complex influencing factors such as surface termination35–37 and ultrathin size confinement effects.38–40 According to traditional wisdom, the e–p interaction in 2D materials is enhanced due to the density of states (DOS) in atomically thin structures.41,42 However, the surface termination increases the thickness of 2D MBene, which may reduce size effects, leading to the suppression of e–p interactions. At present, the impact of surfaces, particularly the presence or absence of terminations, on non-equilibrium e–p interactions in MBenes remains unclear. Therefore, direct measurements of hot carrier relaxation dynamics of the two types of MBenes at ultrafast timescales are required.
Herein, we synthesized MoB and Mo4/3B2−xTz MBenes using an acid etching method. Pump-probe spectroscopy (Fig. 1) was used to observe the time-resolved dynamics of the novel MoB MBene sample without surface termination. Our experimental results show that the time constant for e–p interactions in MoB MBene without termination is ∼218 fs, which is unexpectedly longer than that in Mo4/3B2−xTz MBene (∼126 fs). This result indicates that surface terminations enhance e–p interactions, which differs from the findings reported for semiconductor nanocrystals.32 Furthermore, Raman spectra and density functional theory calculations show that phonon distributions are narrower in MoB MBene than in Mo4/3B2−xTz MBene. The narrow phonon distribution in MoB MBene indicates the presence of an electron that only interacts with a limited number of phonons (Fig. 1, left), which decreases the e–p interaction rate. In contrast, the surface-terminated Mo4/3B2−xTz MBene exhibits a broad phonon distribution, suggesting the presence of an electron that could interact with multiple phonons (Fig. 1, right). This study provides insights into the effect of surface termination on e–p interactions, which is crucial for understanding and advancing its prospects in electronics, optoelectronics, and photothermal applications.
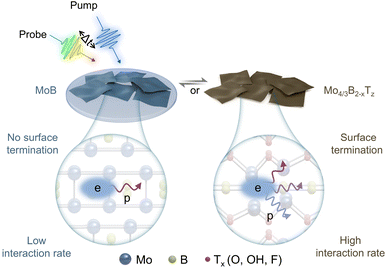 | ||
| Fig. 1 Schematic of the experimental method and the non-equilibrium electron–phonon (e–p) interaction in MoB and Mo4/3B2−xTz MBene. | ||
Results and discussion
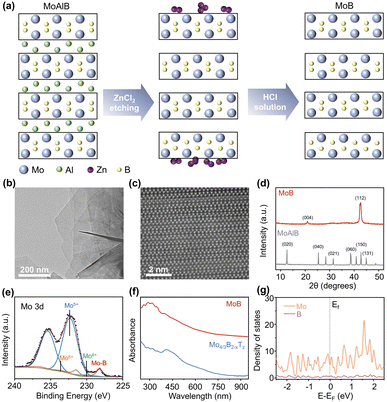
Synthesis and characterization of MoB
MBenes were synthesized by selectively etching Al atoms in the MAB phase43 because they have weak M–A bonds and strong M–B bonds, which facilitate etching and layering of 2D structures.1 M and B represent transition metal and boride atoms, respectively, and A denotes A-group element. Herein, we synthesized high-quality MoB MBene using an approach distinct from that reported in a previous study.35 A schematic of the synthesis process for MoB MBene is shown in Fig. 2a. MoB nanosheets were prepared by etching the Al layer of MoAlB in a Lewis acid molten salt, followed by removing Zn nanoparticles using a dilute hydrochloric acid solution (details in the SI). Transmission electron microscopy (TEM) and scanning electron microscopy images show the layered structure of MoB (Fig. 2b and S1). The scanning TEM (STEM) image of MoB is shown in Fig. 2c, in which Mo atoms appear bright but B atoms are not observed because of their lower mass than that of Mo atoms.35 The STEM image indicates that the MoB MBene exists as α-MoB with stacking faults (Fig. S2). The observed zigzag layers originate from the stacking fault structure, consistent with previous reports.35,43 The crystal structure of MoB was indicated using X-ray diffraction (XRD) patterns (Fig. 2d), which show stacking faults in α-MoB (Fig. S3).43 The high-resolution Mo 3d XPS spectrum of MoB is shown in Fig. 2e. Owing to spin–orbit coupling, the Mo 3d signal splits into the 3d5/2 and 3d3/2 components, with a typical spin–orbit energy separation of approximately 3.1 eV. Based on this constraint, peak deconvolution yields four pairs of characteristic peaks, with the 3d5/2 components located at 228.2, 230.0, 232.3, and 234.0 eV, corresponding to the Mo–B bonding state, Mo4+, Mo5+, and Mo6+ species, respectively. The Mo 3d spectrum of Mo4/3B2−xTz is shown in Fig. S4, where the Mo–B–Tz component appears at 229.6 eV, consistent with previous work.18 These results indicate that MoB contains only Mo–B bonds without surface terminations, whereas the presence of terminations is clearly observed in Mo4/3B2−xTz. Additionally, inductively coupled plasma optical emission spectrometry (ICP-OES) was used to characterize the chemical composition of MoB, which shows that MoB has a Mo/B atomic ratio of 49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 51, confirming the 1
51, confirming the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 stoichiometry of MoB. Meanwhile, the Mo/B atomic ratio in Mo4/3B2−xTz was determined to be 1
1 stoichiometry of MoB. Meanwhile, the Mo/B atomic ratio in Mo4/3B2−xTz was determined to be 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.31, corresponding to an x value of approximately 0.25. The absorption spectra of MoB and Mo4/3B2−xTz MBenes are shown in Fig. 2f. In Mo4/3B2−xTz MBene, Tz represents its surface termination. Both MBenes exhibit broad absorption spectra, with gradually increasing absorption from the visible to ultraviolet (UV) regions (<400 nm). The electronic DOS (Fig. 2g and S5) and band structure (Fig. S6 and S7), suggest that the Mo d orbitals in MoB and Mo4/3B2Tz MBene contribute the most to its electronic states near the Fermi level. The results indicate that MoB MBene exhibits metallic characteristics, similar to most MXenes.44,45
1.31, corresponding to an x value of approximately 0.25. The absorption spectra of MoB and Mo4/3B2−xTz MBenes are shown in Fig. 2f. In Mo4/3B2−xTz MBene, Tz represents its surface termination. Both MBenes exhibit broad absorption spectra, with gradually increasing absorption from the visible to ultraviolet (UV) regions (<400 nm). The electronic DOS (Fig. 2g and S5) and band structure (Fig. S6 and S7), suggest that the Mo d orbitals in MoB and Mo4/3B2Tz MBene contribute the most to its electronic states near the Fermi level. The results indicate that MoB MBene exhibits metallic characteristics, similar to most MXenes.44,45
Effect of the surface on e–p interactions
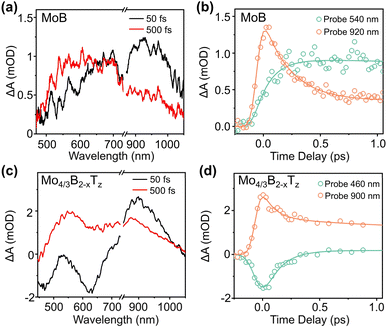
To investigate e–p interactions in MoB MBene, pump-probe transient absorption (TA) spectroscopy experiments were performed, in which a laser pulse excited electrons, and the optical response was measured as a function of time using a delayed probe pulse.46–48 Owing to the strong optical absorption of MoB MBene in the UV region (Fig. 2f, red line), a 360 nm photon was chosen as a pump pulse. Fig. 3a shows the TA spectrum of MoB MBene at 50 fs, which exhibits a broad photoinduced absorption feature ranging from 500 to 1000 nm, with gradually increasing absorption from the visible to near-infrared regions. In the TA spectrum of MoB MBene at 500 fs, the intensity of TA signals in the visible region (∼540 nm) increases, whereas that of signals in the near-infrared region (∼920 nm) distinctly decreases. The TA dynamics of MoB MBene at 920 nm (Fig. 3b) exhibit a fast decay of approximately 1 ps, whereas at 540 nm they show a significantly slower decay, with a timescale much greater than 1 ps. Based on the electronic density of states (Fig. S8), the long-lived feature at 540 nm may originate from the detection of low-energy carriers near the Fermi level, whereas the dynamics at 920 nm correspond to the relaxation of high-energy carriers further from the Fermi level. In non-bandgap, non-semiconducting 2D materials such as MXene and graphene, carriers primarily relax via e–p scattering within 1 ps, and during this time window, the dynamics are minimally affected by defects or structural disorder.Previous reports indicate that hot carrier relaxation induced by defects or structural disorder mainly occurs after 1 ps.49–52 Based on our calculations results (Fig. 2g and S6) and previous studies,18 MBene is classified as a metallic 2D material. Therefore, this study focuses on the carrier dynamics of MBene within 1 ps, primarily reflecting non-equilibrium e–p interactions. The mechanism of the decay and growth dynamic processes of MoB MBene may be as follows: after the metal absorbs photons, the electron temperature increases, and the lattice temperature remains near equilibrium. Then, with time delays, the high temperature of the electron decreases within hundreds of femtoseconds to several picoseconds as the high-temperature electron transfers energy to the low-temperature equilibrium phonon (decay dynamics), resulting in an increase in the temperature of the phonon (growth dynamics).53–55 Therefore, the TA dynamics of MoB MBene at 920 nm are consistent with the electron cooling feature, whereas its TA dynamics at 540 nm are consistent with the phonon heating process. The time required for dynamic decay and growth processes is the e–p interaction time (280 ± 20 fs) with global fitting (see SI).56,57
Next, we performed a control TA experiment on Mo4/3B2−xTz MBene with surface termination to determine the effect of surface termination on e–p scattering. The Mo4/3B2−xTz MBene sample was synthesized according to a procedure reported in a previous study.1 The high-resolution TEM image of Mo4/3B2−xTz MBene, shown in Fig. S4a, reveals a d-spacing of 0.31 nm, corresponding to the [100] plane of Mo4/3B2−xTz. The XRD pattern of Mo4/3B2−xTz MBene is shown in Fig. S4b. XPS (Fig. S4c and d) confirms the termination on the Mo4/3B2−xTz MBene surface. A wavelength of 390 nm was selected as a pump because of the strong UV absorption band of Mo4/3B2−xTz MBene (Fig. 2f, blue line). The TA spectra of Mo4/3B2−xTz (Fig. 3c) show a photoinduced bleaching feature in the 450–680 nm region and absorption signals in the 680–1000 nm range within the first 50 fs. Then, with time evolution at 500 fs, part of the photoinduced bleaching signal in the 450–680 nm range changes to a photoinduced absorption signal, whereas the photoinduced absorption signal intensity in the 680–1000 nm range decreases. The TA spectra of Mo4/3B2−xTz and MoB MBenes are different, suggesting that the surface termination can tune the optical and electrical properties of MBenes. Fig. 3d and S9 show the probe wavelength-dependent dynamics traces of Mo4/3B2−xTz. A global fitting was used to obtain an e–p interaction rate of 138 ± 6 fs for Mo4/3B2−xTz.
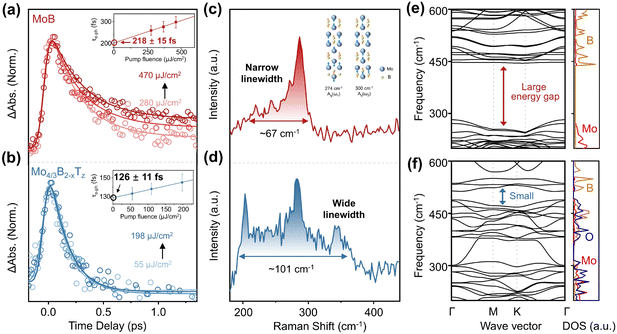
To obtain the intrinsic e–p scattering time, we conducted pump power–dependent TA experiments on MoB (Fig. 4a) and Mo4/3B2−xTz (Fig. 4b) MBenes. Fig. 4a and S10 show the pump fluence-dependent dynamics and TA spectrum of MoB, respectively. The dynamics lifetime probed at 920 nm is prolonged with the increasing pump fluence. This phenomenon is quite common in metals at high pump fluences.58,59 Global fitting of the dynamical traces (Fig. 4a, inset) reveals that the electron–phonon coupling time (τe–p) of MoB MBene increases with increasing pump fluence. The linear fit of pump power-dependent τe–p acquires the intercept (by extrapolating each curve to zero absorbed energy density) to exclude the influence of the thermal effect.60 The linear fit result displays an intercept τe–p of 218 ± 15 fs for MoB MBene. Fig. 4b shows the dynamics of Mo4/3B2−xTz MBene at 460 nm with different pump fluences. A wavelength of 460 nm was selected for analysis because of the small overlap between photon-induced bleaching and absorption signals. The obtained results suggest that the intercept τe–p of Mo4/3B2−xTz MBene is 126 ± 11 fs, significantly smaller than the intercept τe–p of MoB MBene, suggesting that the surface termination enhances e–p interaction. These results suggest that the size confinement effect, in which the surface termination increases the thickness of MBene and leads to weakened e–p interactions,38 is not a dominant mechanism in MBene. Additionally, the point that the surface termination enhances e–p interaction is supported by the measured Raman spectra of MBene. The Raman spectrum of MoB (Fig. 4d) shows the linewidth of the two phonon vibrations (Δν = 67 cm−1). The Raman spectral linewidth was analyzed via Gaussian fitting (Fig. S11). Two peaks are observed in the Raman spectrum of MoB MBene. According to the calculation results (Fig. S12), both peaks correspond to out-of-plane vibration modes. Meanwhile, the Raman spectrum of Mo4/3B2−xTz MBene with surface termination (Fig. 4e) shows four distinct peaks at 203, 240, 284 and 345 cm−1. The total linewidth (Δν = 101 cm−1) of the four peaks of Mo4/3B2−xTz (Fig. S13) is broader than the linewidth (Δν = 67 cm−1) of MoB MBene. According to the Raman spectra, the spectral linewidths of MoB are narrower than those of Mo4/3B2−xTz. Previous studies61 have shown that the linewidth of Raman peaks reflects the e–p interactions in metallic materials. Energy exchange between electrons near the Fermi level and phonons accelerates the relaxation of hot carriers and shortens the phonon lifetime, leading to a broadening of the Raman peaks.61–63 Raman spectra suggest that the wider linewidths of the phonon distribution are caused by surface termination, which leads to faster e–p interaction rates. TA experimental and Raman spectral results suggest that termination can increase the e–p interactions.
Further insights into the e–p interactions in MoB MBene were obtained by calculating its phonon dispersion bands and DOS, which are shown in Fig. 4g. The phonon dispersion bands of MoB MBene show a high-frequency vibration region (438–615 cm−1) attributed to B and a low-frequency vibration region (0–270 cm−1) corresponds to Mo. These results exhibit a large energy gap from 270 to 438 cm−1 in MoB MBene. The phonon dispersion bands and DOS of Mo4/3B2O2 MBene are shown in Fig. 4h. The phonon dispersion bands of Mo4/3B2O2 MBene show high (370–720 cm−1) and low (0–310 cm−1) frequency vibration regions attributed to B and Mo, respectively. Meanwhile, in Mo4/3B2O2 MBene, the vibration from O groups is observed in the vibrational regions of 0–315 cm−1 and 370–490 cm−1. These results reveal that the Mo4/3B2O2 MBene has more vibrational modes and a smaller energy gap than MoB MBene. Briefly, MBenes without surface termination exhibit a narrower phonon distribution. When terminating groups are introduced, the number of atoms in the unit cell increases, and the number of vibrational modes increases. This is consistent with the experimental Raman spectroscopy results, indicating that the surface terminations increase the number of phonon frequencies, thereby enhancing the e–p interactions.
Analysis of the symmetry and coupling strength of MoB MBene
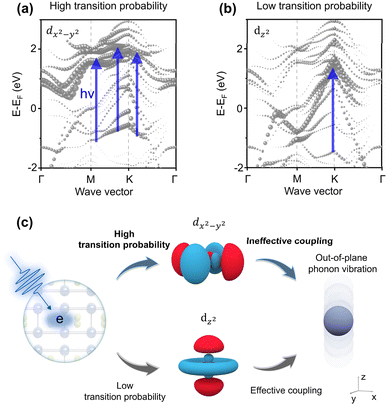
Finally, to understand the e–p interactions in MoB MBene after photon excitation, we examined whether the coupling strength of phonon modes is related to the band-to-band transition, as suggested by previous studies.24,64 The relatively weak e–p interaction strength in MoB MBene compared with MBenes with termination can be elucidated by considering the symmetry between the phonon vibration modes and electron orbitals. We calculated the DOS and electron structure of MoB MBene. The orbital-dependent DOS of MoB (Fig. S6) suggests that the energy band structure from −2 eV to 2 eV is contributed mainly by the 4d orbitals of Mo. The two orbital-dependent fat band structures dx2−y2 and dz2 are shown in Fig. 5a and b, respectively. Moreover, the size of each circle represents the DOS. By combining the fat-band (Fig. 5a and b) analysis with the excitation photon energy (360 nm), the probability of electronic transitions can be evaluated. Upon photon absorption, the dominant photoinduced band-to-band transitions are most likely to occur from approximately −1.0 to 2.0 eV in Fig. 5a and from −1.5 to 1.5 eV in Fig. 5b. The band structure of the dx2−y2 orbital (Fig. 5a) displays a higher DOS than the dz2 orbital. The fat band structures indicate that the band-to-band transitions are much more likely to occur in the dx2−y2 orbital than the dz2 orbital. The dxy electronic orbital band structure is shown in Fig. S14, which also displays a higher transition possibility. Therefore, the transition possibility of entering in-plane electronic orbitals, such as dx2−y2 and dxy, is high and the transition possibility of entering out-of-plane (dz2) electronic orbitals is low. Based on group theory arguments, the main in-plane excited electronic states, such as dx2−y2 and dxy, are generated after MoB absorbs a photon. The wave function of the excited electron is azimuthally symmetric, oriented along the in-plane direction, such as the x- or y-direction, which is effective in coupling in-plane phonon vibration modes. However, the Raman spectrum (Fig. 4c) shows that the phonons of MoB are primarily out-of-plane vibration modes. The mismatched symmetry between the dominated in-plane excited electron orbital and out-of-plane phonon vibration modes causes relatively weak e–p interactions.Our findings of MoB MBene suggest that the excited electrons primarily occupy in-plane orbitals (dx2−y2/dxy), which are symmetrically mismatched with the out-of-plane vibration mode (A1g). This is different from previous studies on MXenes with surface termination, in which the matched symmetry between the electron entering the out-of-plane orbital (dz2) and the out-of-plane phonon vibrational mode (A1g)24 is involved. This is also distinct from e–p interactions in graphite65 and graphene,66 where the electrons are excited to an in-plane orbital (px/py) strongly couple with in-plane phonon vibrational mode (Eg; Fig. S15). To further quantify the enhancement of e–p interactions by surface termination, we performed density functional perturbation theory calculations67 of the e–p coupling constants (see SI). The continuous density of states near the Fermi level (Fig. S16) shows that MoBO and MoBF are metallic. The results (Fig. S17) show that the e–p coupling constants for MoBO and MoBF are 0.51 and 0.445, respectively, both of which are higher than that (0.406) for MoB. This finding is in good agreement with previous studies.68 This further supports the conclusion that surface end groups in MBene enhance e–p interactions.
Discussion
The hot carrier dynamics of metallic MBene investigated in this work within a 1 ps timescale reflect non-equilibrium e–p interactions. In metals such as MBene, the high density of states near the Fermi level and the absence of a bandgap result in carrier relaxation being primarily dominated by electron–phonon scattering, typically occurring on a timescale of hundreds of femtoseconds to a few picoseconds, as observed in MXene, graphene, and noble metal nanoparticles.49–52,69,70With regard to the electronic structure, in semiconductor nanocrystals with surface terminations, the presence of a large bandgap reduces the thermal displacement of surface atoms and diminishes the spatial overlap between charge carriers and these large-amplitude atomic vibrations, leading to weaker electron–phonon coupling and slower carrier relaxation.32 Previous studies have shown that in metallic materials, where no bandgap exists, the relaxation rate of hot carriers is primarily influenced by the density of states near the Fermi level, high-frequency phonon modes, and the strength of electron–phonon coupling, with their relative importance decreasing in this order.30 For the density of states near the Fermi level, since the studied MoB and Mo4/3B2−xTz are both composed of the transition metal Mo,18 the electronic density of states near the Fermi level is primarily contributed by Mo.30 As a result, their densities of states near the Fermi level are similar, leading to only minor differences in the hot carrier-phonon scattering rates. However, their phonon modes differ significantly: in MoB without surface terminations, the number of high-frequency phonon modes is substantially reduced compared to Mo4/3B2−xTz with terminations, leading to a lower scattering rate between carriers and high-frequency phonons and consequently slower carrier relaxation. Unlike previous studies, which have mainly focused on surface-terminated Mo4/3B2−xTz MBenes,71,72 and relied on tuning the electronic structure by changing surface terminations to affect carrier dynamics, our work presents the first investigation of termination-free MoB MBene. We combine experimental and calculated results (Fig. 4 and S17) to demonstrate that, compared with MBenes with surface terminations, electron–phonon scattering is suppressed in the MBene without surface terminations, which can be attributed to its fewer phonon vibration modes. To further exclude the influence of other factors, a detailed discussion of the effects of stoichiometry and defects on the carrier dynamics in MoB and Mo4/3B2−xTz is provided in Section S6 of the SI.
Herein, we first observed the ultrafast dynamics of MoB and Mo4/3B2−xTz MBenes using TA spectroscopy. TA experimental results show that MoB MBene without surface termination exhibits slower relaxation dynamics compared with Mo4/3B2−xTz MBene, which suggests that surface termination enhances e–p interactions in 2D MBenes. The results differ from theoretical simulations of nanocrystals in that surface terminations suppress e–p interactions.32 In this work, the enhanced e–p interaction is caused by the introduction of low-mass elements as surface termination, which creates additional vibration modes, allowing the excited electron to interact with more phonons. In contrast, previous studies from computational simulations39 have investigated the influence of surface termination on e–p interaction by tuning the electronic structure. We provide experimental evidence from phonon structure and electron relaxation dynamics, analyzing the termination-dependent e–p interactions.
Although our study primarily focuses on Mo-based MBene systems, this mechanism may theoretically be applicable to other MBene systems as well. For metallic MBenes, surface terminations could enhance the e–p coupling constant. In addition, previous studies have demonstrated that the introduction of surface terminations can generate additional vibrational modes73 and increase the e–p coupling constant,74–76 thereby enhancing the e–p interactions. Detailed discussion of the role of surface terminations in electron–phonon scattering is provided in Section S7 of the SI. Nevertheless, the generality of surface termination enhancement of e–p interactions in other MBenes still requires further verification through additional studies. However, since their synthesis is still at an early stage and the available methods are limited. Future efforts could focus on developing diverse synthetic strategies to prepare MBenes with a variety of transition metals and surface terminations. This would enable systematic investigations of their dynamic behaviors.
These comprehensive findings collectively highlight that the control and manipulation of surface termination play a pivotal role in tuning e–p interactions in 2D metallic materials. In MBenes with surface termination, their multiphonon vibration modes tend to interact with electrons. The electron will rapidly cool and transfer its energy to heat the lattice, which can be utilized in the fields of thermoelectric conversion and nonlinear optics, such as saturable absorbers. By contrast, MBenes without surface termination are more suitable for photocatalysis and practical hot-electron optoelectronic devices due to their slower e–p interaction, which affords them prolonged electron lifetimes.
Author contributions
Q. Z., K. Y., L. Z., and K. Z. designed research; J. Z., Q. Z., and L. Z. performed research; Q. Z., J. Z., L. Z., and K. Y. analyzed data; J. L., P. X., R. L., K. Z., and X. Y. discussed the results; J. Z., Q. Z., K. Y., and L. Z. wrote the paper.Conflicts of interest
There are no conflicts to declare.Data availability
All study data are included in the article and/or supplementary information (SI). Supplementary information: SEM, STEM, XRD, XPS, Raman characterization, transient absorption spectra of MoB, calculations of electronic band structures, phonon dispersions, and electron–phonon coupling constants for MoB, MoBO, and MoBF. See DOI: https://doi.org/10.1039/d5sc06945a.Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 22303107, 22241304, 22225303, 22173082, 22209176, 22579053, 92365105, 12204237), the National Natural Science Foundation of China (NSFC Center for Chemical Dynamics (Grant No. 22288201)), the Innovation Program for Quantum Science and Technology (2021ZD0303304), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0970000 and XDB0970200), the Fundamental Research Funds for the Central Universities, (No. 30925010211), the Open Foundation of the State Key Laboratory of Molecular Reaction Dynamics in DICP (Grant No. SKLMRD-K202507), Shanghai Pujiang Talent plan project (23PJ1402000), Liaoning Natural Science Foundation (2023-BS-007), Beijing National Laboratory for Condensed Matter Physics (2024BNLCMPKF017), Jincheng Research institute of opto-mechatronics industry (2024SZKF08), the Natural Science Foundation of Jiangsu Province (Grant No. BK20220922).References
- J. Zhou, J. Palisaitis, J. Halim, M. Dahlqvist, Q. Tao, I. Persson, L. Hultman, P. O. Å. Persson and J. Rosen, Science, 2021, 373, 801–805 CrossRef CAS PubMed.
- L. Zhang, S. Xing, T. He, W. B. Wu, A. l. Zhang, Z. Guo, P. Das, S. Zheng, J. Y. Ge and X. Feng, Adv. Mater., 2025, 37, 2411765 CrossRef CAS PubMed.
- J. Si, J. Yu, H. Lan, L. Niu, J. Luo, Y. Yu, L. Li, Y. Ding, M. Zeng and L. Fu, J. Am. Chem. Soc., 2023, 145, 3994–4002 CrossRef CAS PubMed.
- Y. Wang, W. Xu, D. Yang, Y. Zhang, Y. Xu, Z. Cheng, X. Mi, Y. Wu, Y. Liu and Y. Hao, ACS Nano, 2023, 17, 24320–24328 CrossRef CAS PubMed.
- Z. Liu, W. Gao, L. Liu, Y. Gao, C. Zhang, L. Chen, F. Lv, J. Xi, T. Du and L. Luo, Nat. Commun., 2025, 16, 197 CrossRef CAS PubMed.
- M. Naguib, M. Kurtoglu, V. Presser, J. Lu, J. Niu, M. Heon, L. Hultman, Y. Gogotsi and M. W. Barsoum, MXene, 2023, 23, 4248–4253 Search PubMed.
- V. Kamysbayev, A. S. Filatov, H. Hu, X. Rui, F. Lagunas, D. Wang, R. F. Klie and D. V. Talapin, Science, 2020, 369, 979–983 CrossRef CAS PubMed.
- J. L. Hart, K. Hantanasirisakul, A. C. Lang, B. Anasori, D. Pinto, Y. Pivak, J. T. Van Omme, S. J. May, Y. Gogotsi and M. L. Taheri, Nat. Commun., 2019, 10, 522 CrossRef CAS PubMed.
- D. Li, W. Zheng, S. M. Gali, K. Sobczak, M. Horák, J. Polčák, N. Lopatik, Z. Li, J. Zhang and D. Sabaghi, Nat. Mater., 2024, 23, 1085–1092 CrossRef CAS PubMed.
- C. Zhou, Y.-H. Kim, B. Atterberry, V. Khokhar, A. S. Thind, F. Lagunas, R. Lin, D. Wang, W. Cho, Z. Zhou, M. E. Czaikowski, A. S. Filatov, J. S. Anderson, R. F. Klie, R. D. Schaller, D.-e. Jiang, A. J. Rossini and D. V. Talapin, J. Am. Chem. Soc., 2025, 147, 23743–23757 CrossRef CAS PubMed.
- J. Zhang, R. Muñoz-Mármol, S. Fu, X. Li, W. Zheng, A. Villa, G. M. Paternò, D. Pohl, A. Tahn and M. Hambsch, J. Am. Chem. Soc., 2025, 147, 10012–10022 CrossRef CAS PubMed.
- E. Colin-Ulloa, A. Fitzgerald, K. Montazeri, J. Mann, V. Natu, K. Ngo, J. Uzarski, M. W. Barsoum and L. V. Titova, Adv. Mater., 2023, 35, 2208659 CrossRef CAS PubMed.
- M. Volkov, E. Willinger, D. A. Kuznetsov, C. R. Müller, A. Fedorov and P. Baum, ACS Nano, 2021, 15, 14071–14079 CrossRef CAS PubMed.
- G. Li, N. Amer, H. A. Hafez, S. Huang, D. Turchinovich, V. N. Mochalin, F. A. Hegmann and L. V. Titova, Nano Lett., 2019, 20, 636–643 CrossRef PubMed.
- T. Parker, Y. Zhang, K. Shevchuk, T. Zhang, V. Khokhar, Y.-H. Kim, G. Kadagishvili, D. Bugallo, M. Tanwar, B. Davis, J. Kim, Z. Fakhraai, Y.-J. Hu, D.-e. Jiang, D. V. Talapin and Y. Gogotsi, ACS Nano, 2025, 19, 22228–22239 CrossRef CAS PubMed.
- X. Zhu, X. Zhou, Y. Jing and Y. Li, Nat. Commun., 2021, 12, 4080 CrossRef CAS PubMed.
- T. Xu, Y. Wang, Z. Xiong, Y. Wang, Y. Zhou and X. Li, Nano-Micro Lett., 2023, 15, 6 CrossRef CAS PubMed.
- P. Helmer, J. Halim, J. Zhou, R. Mohan, B. Wickman, J. Björk and J. Rosen, Adv. Funct. Mater., 2022, 32, 2109060 CrossRef CAS.
- J. Jin and U. Schwingenschlögl, npj 2D Mater. Appl., 2022, 6, 49 CrossRef CAS.
- U. De Giovannini, H. Hübener, S. A. Sato and A. Rubio, Phys. Rev. Lett., 2020, 125, 136401 CrossRef CAS PubMed.
- D. Bozyigit, N. Yazdani, M. Yarema, O. Yarema, W. M. M. Lin, S. Volk, K. Vuttivorakulchai, M. Luisier, F. Juranyi and V. Wood, Nature, 2016, 531, 618–622 CrossRef CAS PubMed.
- J. Zhou, H. D. Shin, K. Chen, B. Song, R. A. Duncan, Q. Xu, A. A. Maznev, K. A. Nelson and G. Chen, Nat. Commun., 2020, 11, 6040 CrossRef CAS PubMed.
- L. N. Quan, Y. Park, P. Guo, M. Gao, J. Jin, J. Huang, J. K. Copper, A. Schwartzberg, R. Schaller and D. T. Limmer, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2104425118 CrossRef CAS PubMed.
- Q. Zhang, J. Li, J. Wen, W. Li, X. Chen, Y. Zhang, J. Sun, X. Yan, M. Hu and G. Wu, Nat. Commun., 2022, 13, 7900 CrossRef CAS PubMed.
- A. D. Wright, C. Verdi, R. L. Milot, G. E. Eperon, M. A. Pérez-Osorio, H. J. Snaith, F. Giustino, M. B. Johnston and L. M. Herz, Nat. Commun., 2016, 7, 11755 CrossRef PubMed.
- A. Sood, J. B. Haber, J. Carlström, E. A. Peterson, E. Barre, J. D. Georgaras, A. H. Reid, X. Shen, M. E. Zajac and E. C. Regan, Nat. Nanotechnol., 2023, 18, 29–35 CrossRef CAS PubMed.
- W. Zheng, B. Sun, D. Li, S. M. Gali, H. Zhang, S. Fu, L. Di Virgilio, Z. Li, S. Yang and S. Zhou, Nat. Phys., 2022, 18, 544–550 Search PubMed.
- C. Li, N. K. Ravichandran, L. Lindsay and D. Broido, Phys. Rev. Lett., 2018, 121, 175901 CrossRef CAS PubMed.
- A. Wang, S. Li, X. Zhang and H. Bao, Phys. Rev. Mater., 2022, 6, 014009 CrossRef CAS.
- Y. Huang, J. Zhou, G. Wang and Z. Sun, J. Am. Chem. Soc., 2019, 141, 8503–8508 CrossRef CAS PubMed.
- B. Guzelturk, V. Kamysbayev, D. Wang, H. Hu, R. Li, S. B. King, A. H. Reid, M.-F. Lin, X. Wang and D. A. Walko, Nano Lett., 2023, 23, 2677–2686 CrossRef CAS PubMed.
- N. Yazdani, D. Bozyigit, K. Vuttivorakulchai, M. Luisier, I. Infante and V. Wood, Nano Lett., 2018, 18, 2233–2242 CrossRef CAS PubMed.
- G.-R. Siemann, P. A. E. Murgatroyd, T. Antonelli, E. A. Morales, S. Khim, H. Rosner, M. D. Watson, A. P. Mackenzie and P. D. C. King, Phys. Rev. B, 2025, 112, 085113 CrossRef CAS.
- X. Zhang, J. Zhou, F. Zheng, B. Yang, A. Jha and H.-G. Duan, Commun. Phys., 2025, 8, 71 CrossRef CAS.
- L. T. Alameda, P. Moradifar, Z. P. Metzger, N. Alem and R. E. Schaak, J. Am. Chem. Soc., 2018, 140, 8833–8840 CrossRef CAS PubMed.
- H. Zhang, H. Xiang, F.-z. Dai, Z. Zhang and Y. Zhou, J. Mater. Sci. Technol., 2018, 34, 2022–2026 CrossRef CAS.
- M. A. Purbayanto, M. Chandel, M. Birowska, A. Rosenkranz and A. M. Jastrzębska, Adv. Mater., 2023, 35, 2301850 CrossRef CAS PubMed.
- Z. Bai, D. He, S. Fu, Q. Miao, S. Liu, M. Huang, K. Zhao, Y. Wang and X. Zhang, Nano Sel., 2022, 3, 1112–1122 CrossRef CAS.
- K. G. Reeves, A. Schleife, A. A. Correa and Y. Kanai, Nano Lett., 2015, 15, 6429–6433 CrossRef CAS PubMed.
- N. Yazdani, S. Volk, O. Yarema, M. Yarema and V. Wood, ACS Photonics, 2020, 7, 1088–1095 CrossRef CAS.
- X. Li, J. T. Mullen, Z. Jin, K. M. Borysenko, M. Buongiorno Nardelli and K. W. Kim, Phys. Rev. B:Condens. Matter Mater. Phys., 2013, 87, 115418 CrossRef.
- S.-Y. Yue, R. Yang and B. Liao, Phys. Rev. B, 2019, 100, 115408 CrossRef CAS.
- K. J. Baumler, L. T. Alameda, R. R. Katzbaer, S. K. O'Boyle, R. W. Lord and R. E. Schaak, J. Am. Chem. Soc., 2023, 145, 1423–1432 CrossRef CAS PubMed.
- Y. Xie and P. Kent, Phys. Rev. B:Condens. Matter Mater. Phys., 2013, 87, 235441 CrossRef.
- M. Khazaei, A. Ranjbar, M. Ghorbani-Asl, M. Arai, T. Sasaki, Y. Liang and S. Yunoki, Phys. Rev. B, 2016, 93, 205125 CrossRef.
- C. Gadermaier, A. Alexandrov, V. Kabanov, P. Kusar, T. Mertelj, X. Yao, C. Manzoni, D. Brida, G. Cerullo and D. Mihailovic, Phys. Rev. Lett., 2010, 105, 257001 CrossRef CAS PubMed.
- J. Zhang, H. Sakai, K. Suzuki, T. Hasobe, N. V. Tkachenko, I. Y. Chang, K. Hyeon-Deuk, H. Kaji, T. Teranishi and M. Sakamoto, J. Am. Chem. Soc., 2021, 143, 17388–17394 CrossRef CAS PubMed.
- P. Kim, S. Roy, A. J. S. Valentine, X. Liu, S. Kromer, T. W. Kim, X. Li, F. N. Castellano and L. X. Chen, Chem. Sci., 2024, 15, 14766–14777 RSC.
- J. C. Johannsen, S. Ulstrup, F. Cilento, A. Crepaldi, M. Zacchigna, C. Cacho, I. C. E. Turcu, E. Springate, F. Fromm, C. Raidel, T. Seyller, F. Parmigiani, M. Grioni and P. Hofmann, Phys. Rev. Lett., 2013, 111, 027403 CrossRef PubMed.
- J. C. W. Song, M. Y. Reizer and L. S. Levitov, Phys. Rev. Lett., 2012, 109, 106602 CrossRef PubMed.
- Q. Zhang, Y. Chen, Y. Zhang, J. Sun, M. Hu, X. Yan, K. Yuan, X. Yang and J. Li, J. Phys. Chem. Lett., 2020, 11, 9521–9527 CrossRef CAS PubMed.
- J. Zhao, Q. Zhang, L. Sui, G. Niu, Y. Zhang, G. Wu, S. Yu, K. Yuan and X. Yang, ACS Nano, 2023, 17, 23714–23722 CrossRef CAS PubMed.
- L. Perfetti, P. Loukakos, M. Lisowski, U. Bovensiepen, H. Eisaki and M. Wolf, Phys. Rev. Lett., 2007, 99, 197001 CrossRef CAS PubMed.
- P. B. Allen, Phys. Rev. Lett., 1987, 59, 1460 CrossRef CAS PubMed.
- S. Brorson, A. Kazeroonian, J. Moodera, D. Face, T. Cheng, E. Ippen, M. Dresselhaus and G. Dresselhaus, Phys. Rev. Lett., 1990, 64, 2172 CrossRef CAS PubMed.
- S. Schott, L. Ress, J. Hrušák, P. Nuernberger and T. Brixner, Phys. Chem. Chem. Phys., 2016, 18, 33287–33302 RSC.
- I. H. M. van Stokkum, D. S. Larsen and R. van Grondelle, Bioenergetics, 2004, 1657, 82–104 CrossRef CAS PubMed.
- Z. Lu, A. Vallabhaneni, B. Cao and X. Ruan, Phys. Rev. B, 2018, 98, 134309 CrossRef CAS.
- A. M. Brown, R. Sundararaman, P. Narang, A. M. Schwartzberg, W. A. Goddard III and H. A. Atwater, Phys. Rev. Lett., 2017, 118, 087401 CrossRef PubMed.
- M. Zhou, C. Zeng, Y. Chen, S. Zhao, M. Y. Sfeir, M. Zhu and R. Jin, Nat. Commun., 2016, 7, 13240 CrossRef CAS PubMed.
- Z. Han, X. Yang, S. E. Sullivan, T. Feng, L. Shi, W. Li and X. Ruan, Phys. Rev. Lett., 2022, 128, 045901 CrossRef CAS PubMed.
- M. Lazzeri, S. Piscanec, F. Mauri, A. C. Ferrari and J. Robertson, Phys. Rev. B:Condens. Matter Mater. Phys., 2006, 73, 155426 CrossRef.
- J. Zhuang, X. Xu, Y. Du, K. Wu, L. Chen, W. Hao, J. Wang, W. K. Yeoh, X. Wang and S. X. Dou, Phys. Rev. B:Condens. Matter Mater. Phys., 2015, 91, 161409 CrossRef.
- B. R. Carvalho, L. M. Malard, J. M. Alves, C. Fantini and M. A. Pimenta, Phys. Rev. Lett., 2015, 114, 136403 CrossRef PubMed.
- T. Kampfrath, L. Perfetti, F. Schapper, C. Frischkorn and M. Wolf, Phys. Rev. Lett., 2005, 95, 187403 CrossRef PubMed.
- Y. Liu and R. F. Willis, Phys. Rev. B:Condens. Matter Mater. Phys., 2010, 81, 081406 CrossRef.
- T. Bo, P.-F. Liu, L. Yan and B.-T. Wang, Phys. Rev. Mater., 2020, 4, 114802 CrossRef CAS.
- X. Meng, Y. Shen, X. Yang, K. Cai, Q. Ai, Y. Shuai and Z. Zhou, Phys. Rev. B, 2025, 112, 104503 CrossRef CAS.
- J. Zhao, Y. Zhong, L. Zhang, L. Sui, G. Wu, J. Zhang, K. Han, Q. Zhang, K. Yuan and X. Yang, Nano Lett., 2024, 24, 15340–15347 CrossRef CAS PubMed.
- A. M. Brown, R. Sundararaman, P. Narang, A. M. Schwartzberg, W. A. Goddard and H. A. Atwater, Phys. Rev. Lett., 2017, 118, 087401 CrossRef PubMed.
- Y. Wang, Z. Nie and F. Wang, Light Sci. Appl., 2020, 9, 192 CrossRef CAS PubMed.
- J. Guo, Y. Hao, A. V. Kuklin, W. Bao, S. Wageh, O. A. Al-Hartomy, A. G. Al-sehemi, H. Ågren, L. Gao and H. Zhang, ACS Photonics, 2023, 10, 2353–2362 CrossRef CAS.
- J. Lei, A. Kutana and B. I. Yakobson, J. Mater. Chem. C, 2017, 5, 3438–3444 RSC.
- Z. Jing, J. Liu, N. Li, H. Wang, K. Wu, Y. Cheng and B. Xiao, J. Phys. D: Appl. Phys., 2021, 54, 015301 CrossRef CAS.
- J. Seeyangnok and U. Pinsook, Phys. Rev. B, 2026, 113, 054510 CrossRef.
- P. Jamwal, R. Ahuja and R. Kumar, Phys. Chem. Chem. Phys., 2025, 27, 11017–11024 RSC.
| This journal is © The Royal Society of Chemistry 2026 |