Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Translating across multiple representations of molecular structure: student reasoning, accuracy, and personas
Lyniesha Wright Ward a,
Fridah Rotich
a,
Fridah Rotich b,
Sara Arévalo
b,
Sara Arévalo b and
Maia Popova
b and
Maia Popova *b
*b
aIndiana University Indianapolis, Indianapolis, Indiana, USA
bUniversity of North Carolina at Greensboro, Greensboro, North Carolina, USA. E-mail: m_popova@uncg.edu
First published on 24th March 2026
Abstract
Translating between representations of molecular structure is a key component of representational competence. While studies have examined how students translate between two or three representations, no studies have comprehensively investigated how students translate between multiple representations of molecular structure. Herein, we use a semi-structured think-aloud protocol to investigate how 28 Organic Chemistry I students translate between seven representations of molecular structure (molecular formulae, condensed structures, Lewis structures, skeletal structures, wedge-dash diagrams, chair conformations, and Newman projections). We identified 18 strategies students use to translate, which we grouped into three primary categories: strategies associated with decoding, mapping, and drawing. Our data show that appropriately using the decoding and mapping strategies is associated with correct translations across all representations; however, the appropriate use of drawing strategies is associated with correctly translating representations that include implicit atoms and convey 3D information. Additionally, we identified four distinct personas that describe students’ approaches to translating: the Handyperson, the Apprentice, the Quick Fixer, and the Rule Follower. These personas can help instructors anticipate common challenges and tailor support accordingly.
Introduction and conceptual framework
Representations are integral tools in chemistry for visualizing imperceptible chemical particles. Chemists routinely leverage a variety of representations to communicate phenomena, often maneuvering between different representational formats (Kozma et al., 2000). As such, instruction in chemistry must support students in decoding the features of various representations, understanding how one representation connects to another, and using representations to solve problems and communicate information.The large number of representations introduced in chemistry courses is a challenge for students. Research has shown that students overly focus on the surface features of representations instead of the underlying phenomena embedded within them (Chi et al., 1981; Stains and Talanquer, 2008; Jaber and BouJaoude, 2012; Pande et al., 2015; Galloway et al., 2018; Popova and Bretz, 2018; Wright and Oliver-Hoyo, 2020; Atkinson et al., 2021; Rotich et al., 2024). Though instructors value using multiple representations, some acknowledge that the large number of representations they teach makes it challenging for students to decode underlying concepts (Patron et al., 2017; Popova and Jones, 2021).
These challenges become particularly apparent in the transition from General Chemistry to Organic Chemistry. General Chemistry curricula support students in communicating structure primarily through molecular formulae, condensed structures, and Lewis structures. This is expanded in the Organic Chemistry curricula to also include skeletal (bond-line) structures, wedge-dash diagrams, Newman projections, and chair conformations. The information embedded in these representations increases in complexity, with the General Chemistry representations addressing primarily the composition and connectivity of molecules and ions, and the Organic Chemistry representations also depicting various degrees of spatial information (Fig. 1). Organic Chemistry students are expected not only to learn each representation in isolation but also to recognize how they relate to one another in conveying different aspects of molecular structure.
 | ||
| Fig. 1 The increasing complexity of what is communicated through the diagrammatic features of molecular representations. | ||
Developing this proficiency is part of what scholars describe as representational competence—“a set of skills and practices that allow a person to reflectively use a variety of representations or visualizations, singly and together, to think about, communicate, and act on chemical phenomena in terms of underlying, aperceptual physical entities and processes’’ (Kozma and Russell, 2005, p. 131). This construct is composed of several highly interconnected skills (Nickel et al., 2025; Ward et al., 2025), including the ability to translate or make connections across different related representations by mapping features of one representation onto those of another. These skills are used by chemists to communicate structural information, predict reactivity and properties, and select appropriate representational forms for specific problems (Kozma et al., 2000). As students develop their representational competence, they begin to think more like practicing chemists.
In this study, we investigated how students reason when translating between seven commonly used representations in Organic Chemistry: molecular formulae, condensed structures, Lewis structures, skeletal structures, wedge-dash diagrams, Newman projections, and chair conformations. While all seven representations communicate chemical structure, they differ in surface features, such as the use of letters, numbers, dots, lines (thin, wedged, or dashed), and circles. Moreover, while all seven are symbolic representations (iconicity) and depict information on a similar level of abstraction (granularity), they differ based on their dimensionality and quantitativeness (Talanquer, 2022). Specifically, molecular formulae and condensed structures can be described as primarily compositional, while the remaining representations are more structural (dimensionality). Furthermore, Newman projections and chair conformations can convey mathematical information about the relative energy of chemical structure (quantitativeness). Finally, as described in Fig. 1, these representations vary in complexity, ranging from communicating composition only to a combination of composition, connectivity, and spatial information. Taken all together, differences in appearance, dimensions of variation, and complexity can impact student reasoning and representational competence. While prior work has highlighted the importance of the ability to translate for effective problem-solving and communication of ideas (Kozma and Russell, 2005; Ainsworth, 2006), little is known about how students navigate the distinct visual and conceptual features across this broad spectrum of representational forms.
Building on Talanquer's (2022) recent call for deeper insights into how the unique characteristics of chemical representations shape student reasoning, our study fills this gap by examining how students move across multiple representations of molecular structure. To our knowledge, this is the first study that comprehensively characterizes students’ ability to reason with and translate between such a large number of representations. In doing so, we provide a fuller characterization of students’ ability to translate, offering new insights and implications for both instruction and assessment.
Background and significance
Current instruction about translating between representations
Translating between representations is one of the most frequently cited representational competence skills that instructors aim to teach in organic chemistry (Popova and Jones, 2021). However, there is notable variation in which representations are emphasized and what instructional goals are associated with this skill (Jones et al., 2022). A key resource that supports students in making these connections is the textbook, but the extent and nature of that support differ depending on both the textbook and the specific representation (Gurung et al., 2022). Often, textbooks present translation tasks in a unidirectional manner, which can limit students' awareness of spatial features and promote a narrow, fixed interpretation of molecular structure (Kumi et al., 2013; Gurung et al., 2022). These limitations in textbook support likely contribute to instructors’ desire for professional development on how to teach representations more effectively (Popova and Jones, 2021). Additionally, many note that the content-heavy nature of organic chemistry limits the instructional time available to support student representational competence.A deeper understanding of how students actually translate between multiple representations can equip instructors to set more targeted goals and adopt more intentional strategies when teaching representations of molecular structure. Importantly, this does not mean instructors need to spend more time on these representations or teach more content—these representations are already part of their courses. What is needed are research-based insights into how students reason across multiple representations and what types of translation tasks or instructional sequences are most effective. Such insights can help instructors make more strategic, evidence-informed decisions about how they allocate their limited instructional time. In doing so, they can enhance students’ representational competence not by teaching more, but by teaching more intentionally.
Review of research on how students translate between different molecular representations
Several studies have investigated how students translate between representations of chemical structure in the context of two distinct yet related task types. In one common task, students are presented with multiple representations and asked to identify correspondences between them (e.g., identify a Newman projection among the four options below that corresponds to this wedge-dash diagram) (Koutalas et al., 2014; Stieff et al., 2014, 2018). In another, which is referred to by some researchers as a transformation task, students are asked to produce a new representation based on a given one (e.g., draw a Newman projection that corresponds to a given wedge-dash diagram) (Shane and Bodner, 2006; Cooper et al., 2010; Stieff, 2010; Tiettmeyer et al., 2017; Sandi-Urena et al., 2020). Regardless of the task type, studies have collectively focused on translations between specific representation pairs, such as between Lewis structures, molecular formulae, and/or condensed structures (Shane and Bodner, 2006; Cooper et al., 2010; Tiettmeyer et al., 2017; Sandi-Urena et al., 2020), as well as between Newman projections and wedge-dash diagrams (Stieff, 2010; Koutalas et al., 2014; Stieff et al., 2014, 2018). In contrast, far less is known about how students translate between other pairs, such as wedge-dash diagrams and chair conformations. Additionally, while previous studies have focused on students' ability to translate between two or three representations (Shane and Bodner, 2006; Cooper et al., 2010; Stieff, 2010; Koutalas et al., 2014; Stieff et al., 2014; Mohamed-Salah and Alain, 2016; Tiettmeyer et al., 2017; Sandi-Urena et al., 2020), no studies have comprehensively examined how students reason across the full range of representations of molecular structure used in organic chemistry. The paragraphs below summarize previous research on how students translate between some of these representations.Lewis structures have been widely studied across the chemistry curriculum to understand how learners shift from thinking about composition to reasoning about both composition and connectivity. Though there is an improvement with expertise, learners throughout the undergraduate curriculum struggle to translate molecular formulae and condensed structures into Lewis structures. This difficulty often stems from an over-reliance on memorized rules and surface features, rather than a deeper understanding of how those features map onto the actual chemical entities they represent (Shane and Bodner, 2006; Cooper et al., 2010; Tiettmeyer et al., 2017; Sandi-Urena et al., 2020).
Newman projections and wedge-dash diagrams have been widely studied together due to their shared emphasis on representing spatial information. Research on these representations has revealed several important findings. First, students tend to rely more heavily than experts on imagistic reasoning (Stieff, 2010)—the ability to generate and manipulate image-like mental representations for thinking and problem solving. They also often apply heuristics that treat the representations as two-dimensional diagrams rather than as depictions of submicroscopic entities (Stieff, 2010). Second, successful translation between these formats depends not only on spatial ability but also on representational competence (Stieff, 2010; Koutalas et al., 2014; Stieff et al., 2014, 2018). Third, manipulatives can support student understanding, especially when representations embed 3D spatial information, although their effectiveness varies by representation type (Mohamed-Salah and Alain, 2016). Finally, students’ performance on translation tasks is often influenced by the particular representational format used (Koutalas et al., 2014).
Collectively, these findings underscore the complexity of representational translation in organic chemistry and the multiple cognitive resources students must coordinate to be successful. Whether students are translating from compositional to structural representations (e.g., molecular formulae to Lewis structures) or between more spatial formats (e.g., wedge-dash diagrams to Newman projections), improving representational competence requires targeted instruction that improves reasoning with these representations.
Reasoning strategies used to translate between representations
Studies have investigated how experts and novices reason when translating between spatial representations such as wedge-dash diagrams, Newman projections, and Fischer projections. Three primary reasoning strategies have been identified: (a) algorithmic–diagrammatic, which involves non-spatial utterances, gestures, or inscriptions; (b) spatial-imagistic, which includes spatial utterances, gestures, or inscriptions; and (c) complex-mixed, which combines elements of both approaches (Stieff and Raje, 2010). Experts tend to employ a wider and more flexible repertoire of strategies, often selecting task-specific approaches that align with their personal preferences and goals (Stieff and Raje, 2010). Notably, experts rarely rely solely on spatial-imagistic reasoning (Stieff and Raje, 2010). Instead, they typically use it to evaluate or verify the accuracy of proposed structures. This ability to draw on multiple strategies allows experts to overcome any possible limitations in spatial ability (Stieff, 2007), which is less feasible for novices without knowledge of many strategies. Experts also appropriately apply heuristics and algorithmic strategies by generating additional structures or using imagistic reasoning to support and confirm their use of heuristics (Stieff and Raje, 2010).Novices often rely on spatial-imagistic strategies, such as mentally visualizing molecular models or 2D diagrams when translating between representations (Stieff, 2010); however, as their expertise develops, they incorporate a broader range of alternative strategies (Stieff et al., 2012). Students who use algorithmic–diagrammatic strategies take longer to solve the problem, but can do so with high accuracy (Stieff, 2010). A more fine-grained study on translations between wedge-dash diagrams and Newman projections has shown that students used strategies such as (a) counting atoms, (b) checking the connectivity of atoms, (c) sequential tracking to map the connectivity of atoms or functional groups, and (d) adopting an egocentric perspective-taking strategy (Ward et al., 2022).
Student strategies have also been captured when using manipulatives. For example, when working on an iPad, students used three strategies to translate between wedge-dash diagrams and ball-and-stick models (McCollum et al., 2016). In order of increasing sophistication, these strategies were (a) sequential tracking (i.e., focusing on the sequential order of connected atoms while simultaneously tracking corresponding atoms in both representations); (b) isolation and branching (i.e., fixating on a reference point and then branching out to other atoms); and (c) spatial orientation (i.e., considering the orientation and connectivity of atoms relative to a fixed point). Similarly, when translating between wedge-dash diagrams, Newman projections, and Fischer projections, students used five main strategies: (a) random assignment or guessing, (b) following rules or algorithms, (c) diagrammatic (i.e., drawing an intermediate diagram), (d) mental visualization, and (e) use of physical models (Padalkar and Hegarty, 2013).
These studies collectively show that reasoning when translating between spatial representations spans a continuum from less to more sophisticated strategies, with differences between novices and experts. Fine-grained analyses reveal a variety of approaches (i.e., from simple counting or guessing to more advanced strategies like spatial orientation) that differ in effectiveness and cognitive demand. The effectiveness of a given strategy depends on the specific task and representation, and strategies can be applied both appropriately and inappropriately (Ward et al., 2022). While no single strategy is universally most effective (Stieff, 2010), some are more productive in specific contexts (Padalkar and Hegarty, 2013; McCollum et al., 2016). Consequently, students would benefit most from instruction that not only develops a repertoire of multiple strategies but also emphasizes when and how each is best applied (Stieff et al., 2012).
The strategies described above were identified with studies that primarily investigate visuospatial skills, often in the context of representations that depict 3D spatial information (e.g., wedge-dash diagrams, Newman projections). While these studies provide important insights, representational competence extends beyond spatial reasoning alone (Harle and Towns, 2011; Carlisle et al., 2015). To holistically capture students’ ability to translate between representations of molecular structure, it is necessary to examine a broader range of representations, including those that do not explicitly convey spatial information. In this work, we extend prior research by examining how students translate between seven different representations of molecular structure and by exploring how the strategies they employ vary across these representations. Understanding which strategies students use and whether those strategies are productive is essential for providing targeted instructional guidance. Specifically, our research questions (RQ) are as follows:
• RQ1: What reasoning strategies do students use when translating between different representations of molecular structure?
• RQ2: How does students’ use of reasoning strategies vary across different representations?
• RQ3: How does students’ use of reasoning strategies impact their accuracy?
• RQ4: What patterns describe student proficiency when translating between representations of molecular structure?
Methods
Sample and data collection
The second author conducted semi-structured, think-aloud interviews (Bowen, 1994) with 28 students enrolled in their first semester of organic chemistry at a public southeastern university during Fall 2019 and Spring 2020. Four instructors taught these students across five course sections, all using the same curriculum and textbook (Bruice, 2016). We purposefully sampled participants (Patton, 2002) to represent a range of grades (from “A” to “C”) in General Chemistry II. Including students with varied grades in the previous chemistry course helped us capture reasoning that is more representative of what instructors encounter in real classrooms, as opposed to having only the “A” students volunteer to participate. Pseudonyms were assigned to protect student identities, and all procedures adhered to institutional review board (IRB) guidelines.The interview consisted of four phases addressing different representational competence skills and lasted approximately two hours. Sessions were audio- and video-recorded, then transcribed verbatim, including all student-generated diagrams and annotations. This manuscript focuses on the data from one phase of the interview in which students translated between seven representations of molecular structure: wedge-dash diagrams, chair conformations, Newman projections, condensed structures, skeletal structures, molecular formulas, and Lewis structures (see SI).
Students were randomly assigned to one of two protocol forms (Form 1: N = 15 and Form 2: N = 13), each containing 20 unique translation tasks spanning the seven representations (see SI). This design allowed us to include a wide range of representation pairs without requiring any participant to complete more than 20 translations, keeping the interviews manageable in both cognitive effort and time.
For each task, students judged whether the representations corresponded to each other and explained their reasoning. The tasks were informed by pilot interviews, which revealed common sources of confusion that were incorporated as distractors in the incorrect options. These distractors included issues with bond angles, molecular geometry, the number of atoms, connectivity, or selection based on surface-level similarity alone rather than structural features.
Data analyses
Our analysis followed a multi-stage process integrating qualitative coding, statistical testing, and cluster analysis to capture both the nature and patterns of students’ reasoning when translating between the representations. In Stage 1, we coded interview transcripts for reasoning strategies (see Table 1 in Results). We then categorized each response by the appropriateness of reasoning and accuracy of the answer, creating a four-category performance measure. In Stage 2, we used statistical tests to examine associations between student reasoning and correctness. Finally, in Stage 3, we conducted agglomerative cluster analysis to identify patterns across students’ performance across all representations. The sections below describe each stage of data analysis in more detail.Stage 1: qualitative analyses to characterize student reasoning and accuracy
The first and third authors analyzed interview transcripts to examine how students translate between representations of molecular structure, documenting both response accuracy and reasoning. Transcripts were uploaded into ATLAS.ti (version 9, https://atlasti.com/) and coded deductively using a codebook from pilot work (Ward et al., 2022). Given the broader range of representations in this study, inductive coding and constant comparative analysis (Glaser, 1965; Saldaña, 2013) were used to add new reasoning strategies (see Results for complete codebook) and refine categories. Credibility was supported through peer debriefing, code-recode strategies, and negative case analysis (Lincoln and Guba, 1985). The first and third authors independently coded all data for the 28 participants. The percent agreement for each code ranged between 77% and 100%, with only two codes having an agreement of less than 80% (Campbell et al., 2013). Every disagreement was discussed until a 100% inter-rater agreement was reached. Student performance was then classified into four categories reflecting reasoning appropriateness and answer accuracy: (a) correct answer supported with appropriate reasoning, (b) incorrect answer supported with appropriate reasoning, (c) correct answer supported with inappropriate reasoning, and (d) incorrect answer supported with inappropriate reasoning.Stage 2: statistical tests to examine associations between student reasoning and accuracy
After reaching a consensus in describing the accuracy and appropriateness of student reasoning, the counts of responses that fell into each category above were totaled. To investigate the relationship between student answers and reasoning, the two-sided Fisher's exact test or Pearson's χ2 test of independence was used. Fisher's exact test was used instead of Pearson's χ2 test when the number of counts within a frequency table was less than five. Statistical significance was set at p < 0.05 for all significance testing. All statistical analyses were completed using the R Stats Package in RStudio. Odds ratios were evaluated post hoc as a measure of effect size. The Haldane–Anscombe correction was used to determine odds ratios due to some frequencies equaling zero (Lawson, 2004). Small, moderate, and large effects corresponded to odds ratios equaling 1.68, 3.47, and 6.71, respectively (Chen et al., 2010).Stage 3: cluster analysis to identify patterns across students’ performance
We then used agglomerative cluster analysis to explore patterns among our participants (Clatworthy et al., 2005). The four categories reflecting the relationship between students’ reasoning and accuracy were scored: (a) correct answer supported with appropriate reasoning was scored as 3 points, (b) incorrect answer supported with appropriate reasoning was scored as 2 points, (c) correct answer supported with inappropriate reasoning was scored as 2 points, and (d) incorrect answer supported with inappropriate reasoning was scored as 1 point. We then averaged each student's responses by representation for an overall representation score. Aggregated scores for all participants can be found in the Appendix Table 5.The cluster analysis was conducted using R Version 4.3.0 (R Core Team) using Ward's method with the squared Euclidean distance similarity measure due to the elevation of scores (Clatworthy et al., 2005). We used the gap statistic and the dendrogram (Fig. 2) to determine that the optimal number of clusters was four (Tibshirani et al., 2001). To demonstrate the validity and stability of the clusters, we randomized the dataset ten times, and each time the clusters remained stable (Brandriet and Bretz, 2014; Harshman et al., 2017). Additional evidence of cluster validity, based on inferences about each group, is presented in the Results section (Clatworthy et al., 2005).
 | ||
| Fig. 2 A dendrogram showing the four-cluster solution from the agglomerative hierarchical cluster analysis. | ||
Results
RQ1: What reasoning strategies do students use when translating between different representations of molecular structure?
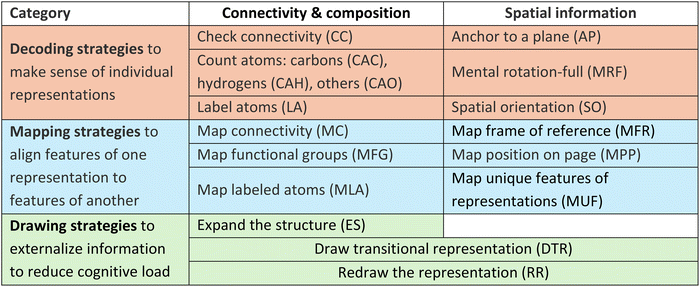
We identified 18 strategies that students use as they translate between the seven different representations of molecular structure (Table 1). Some matched reasoning strategies reported in the literature, which we coded deductively, while others were novel (to our knowledge) and developed through inductive coding. For the previously reported strategies, we intentionally retained the original terminology to maintain consistency with prior work (as opposed to creating new names). For example, we adopted ‘expanding the structure’ and ‘redrawing the representation’ from studies by Flynn and colleagues on student reasoning about reaction mechanisms (Bodé and Flynn, 2016; Flynn and Featherstone, 2017; Carle et al., 2020). Their work also described a ‘mapping strategy,’ which we observed as well; however, we distinguished between mapping different aspects of a representation (e.g., mapping connectivity, mapping functional groups, etc.). Similarly, McCollum and colleagues (2014) described ‘sequential tracking,’ in which students follow the order of connected atoms while aligning corresponding atoms across representations. We separated this strategy into ‘checking connectivity,’ where students described which atoms are connected within a single representation, and ‘mapping connectivity,’ where students compared the connectivity between multiple structures. We also adopted the terminology of previously reported spatial-imagistic strategies, such as ‘full mental rotation’ (Stieff, 2007; Stieff et al., 2018) and ‘spatial orientation’ (Lohman, 1979; Barnea, 2000; Harle and Towns, 2011; Oliver-Hoyo and Babilonia-Rosa, 2017).For novel strategies, we created new names that captured the essence of the approach. Examples include ‘anchor to a plane,’ where students determine whether atoms or bonds lie in or out of the plane of the page, and ‘mapping position on the page,’ where students focus on conserving the position of an atom or bond relative to the page (e.g., left, down) when translating between representations.
The 18 reasoning strategies represent the fine-grained processes students used to complete translation tasks. We grouped them into four categories: decoding, mapping, drawing, and general test-taking strategies (Tables 1 and 2). The decoding strategies, such as ‘counting atoms’ or ‘checking connectivity,’ enabled students to make sense of individual representations. The mapping strategies supported aligning features across representations. Examples include ‘mapping labeled atoms’ and ‘mapping functional groups,’ where students identified, compared, and mapped corresponding features between two representations. The drawing strategies include examples such as ‘redrawing a given representation’ and ‘expanding the structure’ by adding implicit atoms, which were used to support decoding of a single representation, as well as ‘drawing a transitional representation,’ which was used to support mapping of two representations. Students often drew to interpret features, make features explicit, or reduce the complexity of a representation. Drawing potentially enabled them to offload information onto the paper to create more working memory capacity to complete a task (Miller, 1956; Chandler and Sweller, 1991; Hegarty and Steinhoff, 1997; Johnstone, 2006; Fiorella and Zhang, 2018; Fan et al., 2023). Finally, we identified one general test-taking strategy, ‘unfamiliar elimination’ (Table 1), in which students dismissed a representation as non-corresponding because they thought that they had never seen it before.
We also organized the strategies by the type of representational features they helped make sense of (Table 2). Strategies such as ‘label atoms’ or ‘map connectivity’ were categorized as connectivity & composition because they helped students perceive the types of atoms in representations and how those atoms are connected. In contrast, strategies like ‘anchor to a plane’ or ‘map frame of reference’ were categorized as spatial information, as they supported reasoning about spatial relationships within representations. Two drawing strategies were considered translation-dependent because their focus, whether on connectivity & composition or on spatial features, varied depending on the representations being translated. For example, when students drew a transitional representation that is a condensed structure, they emphasized connectivity & composition. However, if the translational representation they drew was a wedge-dash diagram, they often discussed spatial information.
Each reasoning strategy could be used in isolation or in combination with others. On average, students used 2–4 strategies per task. Moreover, most strategies could be applied either appropriately or inappropriately. If participants reasoned appropriately, they applied the strategy normatively and made productive statements relevant to that specific context. For example, the ‘labeling atoms’ strategy was always appropriate and productive because atoms can be labeled in any manner to support sense-making; however, once labeled, students could ‘map the labeled atoms’ either appropriately or inappropriately. In contrast, ‘mapping position on the page’ is a non-normative and unproductive strategy. Rather than reasoning about the 3D spatial relationships of substituents in two representations, students simply compared the position of an atom, bond, or functional group relative to the page (e.g., left, down) to conclude that the representations depict the same molecule. ‘Unfamiliar elimination’ is another always inappropriate strategy, as students claimed they had never seen a certain representation despite having been taught all seven in class.
RQ2: How does students’ use of reasoning strategies vary across different representations?
Each translation required students to move between two different representations of molecular structure. While it is possible to analyze strategy use for each specific representation pair, we instead conducted our analyses at the level of individual representations. In other words, we counted the frequency of strategies whenever a given representation appeared in a translation task, regardless of whether it was the representation translated from or the representation translated to, and regardless of the other representation in the pair. This approach allowed us to identify patterns in strategy use that are more directly tied to the inherent features of each representation rather than patterns that might be specific to a particular pairing.On average, students used decoding strategies (75.2% of translations) about as often as mapping strategies (76.3% of translations), with a stronger emphasis on strategies that supported reasoning about connectivity & composition (78.2%) compared to those focused on spatial information (26.8%) (Table 3). This pattern is unsurprising, as all seven representations convey connectivity & composition, but only certain representations also convey spatial information (Fig. 1). Drawing strategies were used less frequently (17.6% of tasks).
The data reveal clear, representation-specific differences, with the most frequently used individual strategy varying from one representation to another. For condensed structures (CS), wedge-dash diagrams (WDD), chair conformation (CC), and Newman projections (NP), the most frequently used was ‘mapping functional groups.’ For Lewis structures (LS) and skeletal structures (SS), it was ‘checking connectivity,’ and for molecular formulae (MF), it was ‘counting atoms – carbon’ (Appendix Table 6). MF tasks elicited the fewest strategies overall, with a strong emphasis on decoding, reflecting the fact that this representation contains only compositional information and little else to map or interpret spatially. In contrast, LS and CS prompted both high decoding and high mapping use, suggesting that their explicit connectivity details require students to both interpret the representation and align it with others. Representations with embedded 3D information, such as WDD, CC, and NP, showed higher use of spatial strategies. Drawing strategies appeared most often with CS and NP, likely because CS often require expansion to make implicit atoms explicit, and transitional sketches are helpful with NP to clarify orientation.
RQ3: How does students’ use of reasoning strategies impact their accuracy?
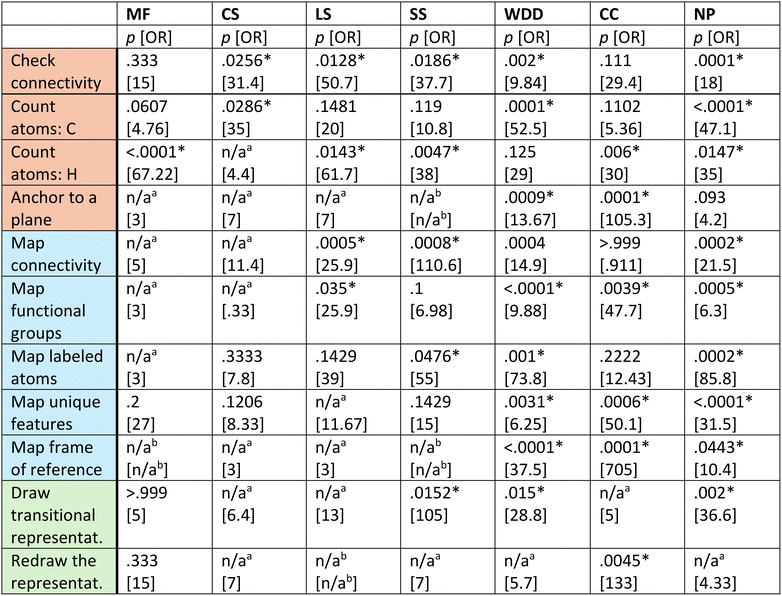
To investigate if there is a relationship between student answers and reasoning, the two-sided Fisher's exact test and Pearson's χ2 test of independence were used. Odds ratios (ORs) were evaluated post hoc as a measure of effect size. These statistical tests showed that there is a strong and consistent relationship between appropriate reasoning and answer accuracy across all representations, but with notable differences across strategy categories (Table 4). Across all strategies combined, p-values are <0.0001 for every representation, with high ORs. Therefore, for each representation, if students used strategies appropriately, they were far more likely to complete translation tasks correctly.| Legend: *Significant at p < 0.05.a n/a are contingency tables in which all frequencies in one row or column are zero. MF is molecular formulae, CS is condensed structures, LS is Lewis structures, SS is skeletal structures, WDD is wedge-dash diagrams, NP is Newman projections, and CC is chair conformations. |
|---|
 |
Breaking the data in Table 4 down by strategy category, the appropriate use of decoding and mapping strategies show statistically significant associations and high effect sizes with accuracy across all representations. The large mapping effect sizes highlight that accurate translation depends on connecting elements across representations, not just interpreting one in isolation. Likewise, the large decoding effect sizes emphasize that such translation is only possible when students can accurately interpret the features of each individual representation. Drawing strategies present a more mixed picture: their appropriate use is significantly associated with accuracy only for SS, WDD, CC, and NP, which are representations that are both novel to organic chemistry students, contain implicit features, and show a higher degree of 3D information. In these cases, drawing may help reduce representational complexity, highlight or visualize relevant features, and make implicit atoms explicit. Interestingly, students relied on different drawing strategies depending on the representation. When translating with SS, WDD, and NP, the ‘draw transitional representation strategy’ was associated with accurate answers (Appendix Table 7). The representations most frequently drawn were SS and MF. However, for CC, students used the ‘redraw the representation’ strategy, suggesting that redrawing the CC called their attention to the conventions of the representation to help them better translate. While students used the ‘expanding the structure’ drawing strategy to make the implicit explicit, it was not associated with better problem-solving. It should be noted that drawing was not beneficial in cases when essential conventions were overlooked. These findings underscore the importance of ensuring that students can accurately draw diagrammatic conventions of these representations, as this skill directly supports their ability to translate between forms, especially those showing a higher degree of 3D information.
When categorizing by representational features, connectivity & composition strategies are highly significant for every representation, with high ORs, especially for LS and CS. This suggests that correctly reasoning about connectivity and composition is universally important. Spatial strategies, in contrast, show significant associations only for translations that include WDD, CC, and NP, which are representations that encode 3D information. This suggests, perhaps unsurprisingly, that spatial reasoning is a necessary but representation-specific predictor of accuracy.
So far, we described the reasoning strategies students used when translating between different representations of molecular structure, and how students’ use of these strategies varied across different representations and impacted their accuracy. Besides these findings, we also noticed patterns across students’ performance, which are described in the section below.
RQ4: What patterns describe student proficiency when translating between representations of molecular structure?
The cluster analysis revealed four distinct groups of students based on their representation scores across all 20 translation tasks (Fig. 2). To validate the clustering solution, we compared these groups across additional external attributes not included in the clustering process (Clatworthy et al., 2005). Specifically, we examined the type and number of reasoning strategies used as well as the characteristics associated with students’ approaches to problem-solving. These criteria helped us develop a set of personas for each cluster, which is an approach adapted from ethnographic research, that captures the defining qualities of the problem solvers in each group (Pruitt and Aldin, 2006; Zagallo et al., 2019). We represent these personas through the metaphor of a contractor: just as contractors select tools from their toolbox to complete a task, students draw upon their repertoire of visualization tools and problem-solving strategies. We identified four distinct personas: The Handyperson, The Apprentice, The Quick Fixer, and The Rule Follower. Below, we describe the characteristics (Fig. 3) and reasoning strategies (Fig. 4) of each persona. | ||
| Fig. 3 Four personas identified through the cluster analysis. Circle colors represent average performance with each representation: green = correct on ≥2/3 of tasks, yellow = correct on 1/3 to 2/3 of tasks, and red = correct on <1/3 of tasks. The order and color for each persona align with its corresponding branch in the dendrogram in Fig. 2. | ||
 | ||
| Fig. 4 The reasoning strategies used by students within each persona. The strategies on the left are sorted by the four categories described in Tables 1 and 2: decoding (orange), mapping (blue), drawing (green), and general test-taking (purple). Bars are color-coded to demonstrate the accuracy of the answer (dark blue for correct and red for incorrect) and shaded to reflect the appropriateness of reasoning for a given context (solid for appropriate and striped for inappropriate). | ||
Will exemplifies the Handyperson persona. For example, when converting between a CS and a WDD (Task A, Fig. 5), Will used the ‘drawing a transitional representation’ strategy to draw a SS of the provided CS. He then compared his drawing to the WDD, reflecting aloud: “I don't know if that stereochemistry [in the wedge-dash diagram] is correct, yeah, it is. It's got to be correct. Boom! …this [CS] isn't supposed to display as much stereochemistry, whereas with the tertbutyl group [in WDD], we can presume that at the end, these all have to be on separate planes, right?” Will used a drawing strategy first to convert the CS into the SS and then compared it to the WDD. Additionally, Will acknowledged that the WDD affords the depiction of stereochemical information, but that spatial information is not represented in a CS.
Maggy exemplifies the Apprentice persona. While she performed some translations correctly and supported her answers with appropriate reasoning, other translations were a challenge. For example, when converting a WDD to a CC (Task B, Fig. 6), Maggy used the ‘redraw the representation’ strategy to redraw the cyclic WDD as a linear WDD. This mistake has been documented in our previous work in which over 15% of about 1500 students thought that cyclic structures can be opened up into linear structures (Ward et al., 2025). When asked about the purpose of the linear WDD, Maggy stated, “This is literally the skeletal structure since I didn't, um, do the molecular formula… This is just to check over it at the end, I guess, cause I didn't write the formula.” The interviewer followed up to clarify, asking Maggy how she would perform this translation in the opposite direction (from CC to WDD). Maggy doubled down on her approach: “…number it, starting at one of the ethyl [methyl] groups and then keep numbering it. Like you have to be consistent with the numbers. So, you can't do like one, two, like jump around like, you have to go continuously. And then, you know, each point represents like a carbon. So, then we start to draw, I would draw, draw something like this first [indicates the linear WDD she drew]. And then maybe if you told me to put it into a cyclohexane, I would put it there. But honestly, if you didn't tell me that it was a cyclohexane, then I wouldn't put it in that structure. I would just leave it like that [indicates the linear WDD she drew].” While Maggy correctly identified the two structures as corresponding due to the ‘mapping labeled atoms’ strategy, she also used the ‘redrawing the representation’ strategy inappropriately, equating a cyclic structure with a linear structure. This non-normative approach led to incorrect answers for other problems Maggy encountered. During the interview, similar to other students in this persona, Maggy acknowledged that she still needs support or guidance when translating.
Edith exemplifies the Quick Fixer persona. When translating between a cyclic WDD and a linear NP (Task C, Fig. 7), Edith described and drew the following: “So this wedge here [blue in WDD] shows a carbon and three hydrogens, which is shown here [blue in NP]. And then here and here [pink in WDD] show a carbon and hydrogen, which is shown here [pink in NP], they are hydrogens. And then this dash [red in WDD] is the reverse of this [blue in NP], which is shown here [red in NP] and it shows what's behind. And then, this shows a carbon with hydrogens [purple in WDD], and this shows a carbon with hydrogens [black in WDD], which is shown here with these hydrogens [purple and black in NP].” Edith overlooked the actual atomic composition and connectivity and, instead, applied a ‘mapping position on the page’ strategy, equating features simply because they appeared in similar positions across the two diagrams. By relying on surface similarity over structural meaning, she inappropriately treated a cyclic WDD as if it corresponded directly to a linear NP. All participants grouped as Quick Fixers used this inappropriate ‘mapping position on the page’ strategy.
Charlie and Vanessa are two participants characterized as Rule Followers. When translating between a CS and a WDD (Task A, Fig. 5), Charlie stated, “Uh, because the problem with [WDD] is that they, uh, indicate some sort of orientation or directionality of the substituent groups. Um, not to say that those are like wrong per se… there's not enough information from the orientation, from the representation to answer that.” Here, Charlie assumed that a CS could not correspond with a WDD because one affords spatial information and the other does not. This rigid reasoning prevented him from leveraging the complementary information across the two representations.
Vanessa would immediately state that structures they were less familiar with would not correspond. For example, she stated that she was not familiar with chair conformations (CC), which is why when completing Task B (Fig. 6), she stated “that one just doesn't look right [CC]… just because I'm unfamiliar with this one as well, I'm going to ‘X’ it out.” Their disregard for representations that they perceived as not familiar (even though they were taught them in class) led to Rule Followers struggling to complete many of the problems.
Limitations
There are several limitations that should be acknowledged. First, all interviews were conducted and recorded virtually, which limited our ability to fully capture student gestures. Gestures often play an important role in translation tasks, particularly those requiring spatial reasoning (McCollum et al., 2014; Flood et al., 2015; Stieff et al., 2016). Without a complete record of these nonverbal behaviors, our analysis does not fully capture the role gestures play in supporting or externalizing students’ reasoning processes.Second, the number of distinct representations varied across tasks. For example, there were more tasks involving translations using wedge–dash diagrams than molecular formulas. Because of this uneven distribution, we analyzed strategy use at the level of individual representations rather than representation pairs. This approach allowed us to identify patterns tied to the inherent features of each representation, but it comes with trade-offs. Certain strategies may emerge more or less frequently when students translate between specific representation pairs, and these nuances are not visible when strategies are aggregated at the representation level.
Third, the sample was from a single institution and reflects the characteristics of that student population. Other populations may use different strategies or bring different experiences to translation tasks. Additionally, our relatively small sample size limited our ability to test the statistical significance of strategy use within clusters or personas.
Finally, our interviews capture students’ translation strategies at a single moment in time. We do not account for how these strategies might develop or shift over the course of the Organic Chemistry course sequence.
Discussion and conclusions
We identified 18 distinct strategies that students used across seven representations of molecular structure, which we grouped into decoding, mapping, drawing, and general test-taking categories. In the context of our study, the test-taking strategy of ‘unfamiliar elimination,’ which has also been captured in other contexts (Sandi-Urena et al., 2020), was indicative of students who have not developed a thorough understanding of the course material. The strategies also differed in the kinds of representational features they helped students make sense of: connectivity & composition versus spatial relationships. Students typically used 2–4 strategies per translation task, and most strategies could be used either productively or inappropriately, underscoring the importance of not only knowing a strategy but also applying it effectively. Overall, decoding and mapping strategies were most common, used in about three-quarters of translation tasks, while drawing strategies appeared less often, in fewer than a quarter of tasks.Translating between representations is defined as the ability to “map features of one type of representation onto those of another” (Kozma and Russell, 2005, p. 132), and students’ mapping strategies (e.g., ‘mapping functional groups’) directly supported this process. Decoding strategies such as ‘checking connectivity’ or ‘counting atoms’ reflect interpretation skill, defined as “the ability to use words to identify and analyze features of a particular representation” (Kozma and Russell, 2005, p. 132). Students also used drawing strategies (e.g., ‘expanding the structure,’ ‘drawing transitional representations’), which represent generation skill (Kozma and Russell, 2005). Drawing is known to support students in offloading information from the working memory onto the paper (Miller, 1956; Chandler and Sweller, 1991; Hegarty and Steinhoff, 1997; Johnstone, 2006; Fiorella and Zhang, 2018; Fan et al., 2023), though some studies suggest that drawing increases cognitive load (Leutner et al., 2009). However, within this study, students were not required to draw but chose to, suggesting its utility in achieving their goal. These findings highlight how interconnected representational competence skills are: interpreting and generating directly support students’ ability to translate. This interconnectedness also extends to students’ meta-representational skills. For example, Rule Followers’ limited awareness of the affordances and limitations of representations hindered their translations. This issue is reflected in prior work showing that weak meta-representational reasoning constrains problem solving (Jaber and BouJaoude, 2012). Taken together, and similar to other studies (Nickel et al., 2025; Ward et al., 2025), these results emphasize the need to develop representational competence as an integrated whole, where interpretation, translation, generation, and meta-representational skills work in concert rather than as isolated skills.
Students’ strategy use varied across representations, which has been previously shown in other studies examining how students translate between representations (Koutalas et al., 2014). Molecular formulae elicited the fewest strategies, mostly focused on decoding by counting atoms. Lewis and condensed structures prompted both decoding and mapping, while 3D-rich representations such as wedge-dash diagrams, chair conformations, and Newman projections elicited more spatial reasoning. The most common strategies differed by representation: ‘counting atoms’ for molecular formulae, ‘checking connectivity’ for Lewis and skeletal structures, and ‘mapping functional groups’ for condensed, wedge-dash, chair, and Newman tasks. Drawing strategies appeared most often with condensed and Newman structures, where sketches helped make implicit and/or spatial features explicit. These findings highlight how representational features shape the reasoning strategies students use, pointing to the need for instruction tailored to the unique demands of each representation.
Across all representations, there was a strong and consistent relationship between appropriate strategy use and accuracy. Unsurprisingly, students are most proficient with translating between representations covered in general chemistry (molecular formulae, Lewis, and condensed structures) and are more challenged by the new representations introduced in organic chemistry, which also communicate more 3D information (skeletal structures, chair conformations, wedge-dash diagrams, and Newman projections). Similar to the findings of Olimpo and colleagues (2015), as the complexity of the representation increases, so does the chance that the students will respond incorrectly to a prompt. Decoding and mapping strategies were especially powerful predictors of correct translations, because accurate interpretation of individual representations and effective linking across them are both essential. Drawing strategies were associated with accuracy in tasks involving representations that use implicit features and depict spatial information (skeletal structures, chair conformations, wedge-dash diagrams, and Newman projections), where sketching reduced complexity or made implicit features explicit. Transitional drawings, especially with skeletal structures, wedge-dash diagrams, and Newman projections, supported accuracy by reducing complexity and making features explicit. Redrawing chair conformations also appeared to help students attend to representational conventions. The underuse but importance of drawing strategies, especially when complexity or 3D information is involved, highlights the importance of encouraging students to practice drawing representations (Ainsworth et al., 2011; Van Meter and Firetto, 2013). Connectivity & composition strategies were universally important, whereas spatial reasoning strategies predicted success only for 3D representations.
Finally, we identified four distinct personas that describe students’ approaches to translation tasks. The Handypersons demonstrated the most proficient and flexible use of strategies, applying them appropriately across all representations. For example, they drew on a wide repertoire of strategies and showed awareness of when to generate additional structures or when to rely on imagistic reasoning, which are practices often associated with expert problem solvers (Stieff and Raje, 2010). The Apprentices showed solid proficiency with more familiar representations (molecular formulae, Lewis and condensed structures) but struggled with more spatial representations that use implicit atoms (wedge-dash diagram, skeletal structures, chair conformations, and Newman projections), occasionally defaulting to rule-based reasoning. The Quick Fixers struggled across all representations because they relied on inappropriate surface-level comparisons. They also did not use any drawing strategies to support their reasoning. Finally, Rule Followers struggled with representations that communicate spatial information. They rigidly followed self-generated rules with limited awareness of representational affordances and limitations. These personas illustrate key differences in translation proficiency and provide a practical framework for instructors to recognize and respond to diverse student approaches.
Implications
This work provides insight into the strategies students use when translating between representations and responds to instructors’ reported need for guidance on how to effectively teach with them (Popova and Jones, 2021). Because instructors’ choices of representations are shaped by their perceptions of student knowledge (Patron et al., 2017), it is important to help students recognize the purpose of each. One way to make this explicit is through overview diagrams such as Fig. 1, which illustrate how representations vary in complexity and why different representations may be needed. Using such tools can strengthen students’ meta-representational awareness by helping them see not only how representations differ, but also what each affords and where each is limited. This is important as previous research in the context of resonance has shown that meta-representational awareness has a great impact on student conceptual understanding (Xue and Stains, 2020). To further develop this skill, instructors can design tasks that require students to choose and justify the most appropriate representation for a given context.This study underscores the need to treat representational competence as an integrated set of skills rather than as separate abilities. Instructors should recognize the complexity of representational tasks that may appear simple to them because of their years of experience in the discipline (Popova and Jones, 2021). They should scaffold and model their reasoning in contexts that provide integrated learning opportunities, where students can practice combining interpretation, translation, generation, and meta-representational awareness rather than treating these skills as isolated (Ward et al., 2025).
When translating between representations, students not only need to know a range of strategies but also to learn when and how to use them productively. For example, rather than letting students default to surface-level cues, instructors could design practice tasks that require students to justify their reasoning with structural features, such as connectivity or spatial orientation. The strong link between appropriate strategy use and accuracy suggests that instruction should focus on making strategies explicit and visible. Instructors can model their own reasoning aloud (e.g., “first I’ll count carbons to check connectivity, then I’ll map functional groups”), and then ask students to solve similar problems in think-pair-share or clicker formats. Reflection prompts or short written justifications can be built into homework and exams to reinforce metacognitive awareness of strategy choice. Drawing should be encouraged as a routine practice for representations that convey 3D information: students might be asked to sketch a wedge–dash diagram from a condensed structure, or draw a transitional representation before deciding whether two representations depict the same molecule.
Finally, the four personas identified in this study illustrate the diverse ways students approach translation tasks. These personas can help instructors anticipate common challenges and tailor support accordingly. For example, “Handypersons” can benefit from opportunities to explain their reasoning while translating. These students occasionally began with an incorrect answer, but through verbalizing their thought process, their reasoning often shifted toward the correct solution. “Apprentices” may need targeted practice with spatially rich representations. “Quick Fixers,” who rely on surface cues, may need assessments that probe beyond recognition to explanation. Similar to other studies (Stieff, 2010; Stieff and Raje, 2010; Stieff et al., 2012), we recommend that these students be modeled a broad repertoire of strategies, so that they have more problem-solving tools at their disposal. “Rule Followers” could benefit from assignments that explicitly ask students to compare the affordances and limitations of different representations and how multiple representations can be used in tandem to communicate different things. By recognizing these different approaches, instructors can better design scaffolds that move students from limited or rule-based reasoning toward more flexible, expert-like problem solving. While we provide suggestions tailored to each persona, their implementation would help all students.
Structured activities, such as clicker questions, think-pair-share, guided inquiry, or small-group discussions, can make reasoning not only visible but also actively develop it. Both “Apprentices” and “Quick Fixers” often relied on the unproductive ‘mapping position on the page’ strategy, focusing on the surface placement of substituents rather than reasoning about their spatial orientation. These students could benefit from explicit confrontation with such non-normative approaches. An example class activity, which creates opportunities for students to articulate their reasoning and recognize flaws in surface-level approaches, is shown in Fig. 8.
 | ||
| Fig. 8 An example think – write – pair – share activity to support students in translating between representations of molecular structure. | ||
Following the discussion, instructors could model how to redraw a wedge-dash diagram as a Newman projection for “Quick Fixers,” and use that moment to highlight the affordances and limitations of each representation for “Rule Followers.” Prompts should encourage students to consider the complementary functions of different visual representations rather than superficial similarities between them and to focus on conceptually relevant features (Rau, 2017). Feedback should go beyond right or wrong answers to highlight reasoning strategies and help students refine their approaches (Ward et al., 2022; Rotich et al., 2024).
Carefully designed assessment items can also reveal rule-based reasoning patterns, helping identify students who rely on them (see Fig. 3.3 in Ward et al., 2022). Physical model kits or virtual 3D models can further support students in translating between spatially rich representations. However, these tools function as representations themselves and, as studies have shown, students require assistance in using manipulatives and computer-generated models (Carlisle et al., 2015), because if students do not understand the affordances or limitations of manipulatives, they will not use them (Padalkar and Hegarty, 2013; McCollum et al., 2016).
Finally, to monitor the effectiveness of instruction for supporting students' ability to translate between different representations, instructors could use the Organic chemistry Representational Competence Assessment (ORCA; Ward et al., 2025). ORCA measures the learner's ability to interpret, translate, and use various commonly used representations of molecular structure in organic chemistry.
Ethical considerations
This study was conducted under the University of North Carolina at Greensboro IRB (20-0511).Conflicts of interest
There are no conflicts to declare.Data availability
The datasets generated and analyzed in this study are not publicly available due to the potential for participant identification and confidentiality agreements. However, de-identified excerpts of the data may be made available from the corresponding author upon reasonable request.The supplementary information (SI) contains the interview protocol is available. See DOI: https://doi.org/10.1039/d5rp00382b.
Appendix
Tables 5–7.| a Strategies characterized as spatial.b Strategies that emphasize connectivity and composition. The representations are abbreviated as molecular formulae (MF), condensed structures (CS), Lewis structures (LS), skeletal structures (SS), wedge-dash diagrams (WDD), Newman projections (NP), and chair conformations (CC). |
|---|
 |
Acknowledgements
We sincerely thank all the students who participated in this study. We also thank the National Science Foundation for funding this work through NSF DUE-2025216.References
- Ainsworth S., (2006), DeFT: a conceptual framework for considering learning with multiple representations, Learn. Instruct., 16(3), 183–198.
- Ainsworth S., Prain V. and Tytler R., (2011), Drawing to learn in science, Science, 333(6046), 1096–1097.
- Atkinson M. B., Croisant M. and Bretz S. L., (2021), Investigating first-year undergraduate chemistry students’ reasoning with reaction coordinate diagrams when choosing among particulate-level reaction mechanisms, Chem. Educ. Res. Pract., 22(1), 199–213.
- Barnea N., (2000), Teaching and Learning about Chemistry and Modelling with a Computer managed Modelling System, in Gilbert J. K. and Boulter C. J. (ed.), Developing Models in Science Education, Kluwer Academic Publishers, pp. 307–323.
- Bodé N. E. and Flynn A. B., (2016), Strategies of Successful Synthesis Solutions: Mapping, Mechanisms, and More, J. Chem. Educ., 93(4), 593–604.
- Bowen C. W., (1994), Think-aloud methods in chemistry education: understanding student thinking, J. Chem. Educ., 71(3), 184–190.
- Brandriet A. R. and Bretz S. L., (2014), Measuring meta-ignorance through the lens of confidence: examining students’ redox misconceptions about oxidation numbers, charge, and electron transfer, Chem. Educ. Res. Pract., 15(4), 729–746.
- Bruice P. Y., (2016), Organic Chemistry, 8th edn, Pearson.
- Campbell, J. L., Quincy, C., Osserman, J. and Pedersen, O. K., (2013), Coding In-depth Semistructured Interviews: Problems of Unitization and Intercoder Reliability and Agreement, Sociol. Methods Res., 42(3), 294–320.
- Carle M. S., Visser R. and Flynn A. B., (2020), Evaluating students’ learning gains, strategies, and errors using OrgChem101's module: Organic mechanisms – Mastering the arrows, Chem. Educ. Res. Pract., 21(2), 582–596.
- Carlisle D., Tyson J. and Nieswandt M., (2015), Fostering spatial skill acquisition by general chemistry students, Chem. Educ. Res. Pract., 16(3), 478–517.
- Chandler P. and Sweller J., (1991), Cognitive Load Theory and the Format of Instruction, Cogn. Instr., 8(4), 293–332.
- Chen H., Cohen P. and Chen S., (2010), How big is a big odds ratio? Interpreting the magnitudes of odds ratios in epidemiological studies, Commun. Stat.: Simul. Comput., 39(4), 860–864.
- Chi M. T. H., Feltovich P. J. and Glaser R., (1981), Categorization and representation of physics problems by experts and novices, Cogn. Sci., 5(2), 121–152.
- Clatworthy J., Buick D., Hankins M., Weinman J. and Horne R., (2005), The use and reporting of cluster analysis in health psychology: a review, Br. J. Health Psychol., 10(3), 329–358.
- Cooper M. M., Grove N., Underwood S. M. and Klymkowsky M. W., (2010), Lost in Lewis structures: an investigation of student difficulties in developing representational competence, J. Chem. Educ., 87(8), 869–874.
- Fan J. E., Bainbridge W. A., Chamberlain R. and Wammes J. D., (2023), Drawing as a versatile cognitive tool, Nat. Rev. Psychol., 2, 9, 556–568.
- Fiorella L. and Zhang Q., (2018), Drawing Boundary Conditions for Learning by Drawing, Educ. Psychol. Rev., 30(3), 1115–1137.
- Flood V. J., Amar F. G., Nemirovsky R., Harrer B. W., Bruce M. R. M. and Wittmann M. C., (2015), Paying attention to gesture when students talk chemistry: interactional resources for responsive teaching, J. Chem. Educ., 92(1), 11–22.
- Flynn A. B. and Featherstone R. B., (2017), Language of mechanisms: exam analysis reveals students’ strengths, strategies, and errors when using the electron-pushing formalism (curved arrows) in new reactions, Chem. Educ. Res. Pract., 18, 64–77.
- Galloway K. R., Leung M. W. and Flynn A. B., (2018), A Comparison of How Undergraduates, Graduate Students, and Professors Organize Organic Chemistry Reactions, J. Chem. Educ., 95(3), 355–365.
- Glaser B. G., (1965), The Constant Comparative Method of Qualitative Analysis, Soc. Problems, 12(4), 436–445.
- Gurung E., Jacob R., Bunch Z., Thompson B. and Popova M., (2022), Evaluating the Effectiveness of Organic Chemistry Textbooks for Promoting Representational Competence, J. Chem. Educ., 99, 2044–2054.
- Harle M. and Towns M., (2011), A review of spatial ability literature, its connection to chemistry, and implications for instruction, J. Chem. Educ., 88(3), 351–360.
- Harshman J., Yezierski E. and Nielsen S., (2017), Putting the R in CER: How the statistical program R transforms research capabilities. In Computer-Aided Data Analysis in Chemical Education Research (CADACER): Advances and Avenues, ACS Symposium Series, vol. 1260, pp. 65–90.
- Hegarty M. and Steinhoff K., (1997), Individual differences in use of diagrams as external memory in mechanical reasoning, Learn. Individual Differences, 9(1), 19–42.
- Jaber L. Z. and BouJaoude S., (2012), A Macro-Micro-Symbolic Teaching to Promote Relational Understanding of Chemical Reactions, Int. J. Sci. Educ., 34(7), 973–998.
- Johnstone A. H., (2006), Chemical education research in Glasgow in perspective, Chem. Educ. Res. Pract., 7(2), 49–63.
- Jones T., Romanov A., Pratt J. M. and Popova M., (2022), Multi-framework case study characterizing organic chemistry instructors’ approaches toward teaching about representations, Chem. Educ. Res. Pract., 23(4), 930–947.
- Koutalas V. G., Antonoglou L. D., Charistos N. D. and Sigalas M. P., (2014), Investigation of Students’ Ability to Transform and Translate 2D Molecular Diagrammatic Representations and its Relationship to Spatial Ability and Prior Chemistry Knowledge, Proc. Soc. Behav. Sci., 152, 698–703.
- Kozma R. and Russell J., (2005), Students Becoming Chemists: Developing Representational Competence, in Gilbert J. K. (ed.), Visualization in Science Education, Springer, pp. 121–145.
- Kozma R., Chin E., Russell J. and Marx N., (2000), The Roles of Representations and Tools in the Chemistry Laboratory and Their Implications for Chemistry Learning, J. Learn. Sci., 9(2), 105–143.
- Kumi B. C., Olimpo J. T., Bartlett F. and Dixon B. L., (2013), Evaluating the effectiveness of organic chemistry textbooks in promoting representational fluency and understanding of 2D-3D diagrammatic relationships, Chem. Educ. Res. Pract., 14(2), 177–187.
- Lawson R., (2004), Small sample confidence intervals for the odds ratio, Commun. Stat., Part B: Simul. Comput., 33(4), 1095–1113.
- Leutner D., Leopold C. and Sumfleth E., (2009), Cognitive load and science text comprehension: effects of drawing and mentally imagining text content. Comput. Human Behav., 25(2), 284–289.
- Lincoln Y. S. and Guba E. G., (1985), Naturalistic Inquiry, Sage Publications.
- Lohman D. F., (1979), Spatial ability: A review and reanalysis of the correlational literature (Tech. Rep. No. 8), Stanford, CA: Stanford University, School of Education, Aptitude Research Project.
- McCollum B. M., Regier L., Leong J., Simpson S. and Sterner S., (2014), The effects of using touch-screen devices on students’ molecular visualization and representational competence skills, J. Chem. Educ., 91(11), 1810–1817.
- McCollum B., Sepulveda A. and Moreno Y., (2016), Representational technologies and learner problem-solving strategies in chemistry, Teach. Learn. Inquiry, 4(2), 105–121.
- Miller G. A., (1956), The magical number seven, plus or minus two: some limits on our capacity for processing information, Psychol. Rev., 63(2), 81–97.
- Mohamed-Salah B. and Alain D., (2016), To what degree does handling concrete molecular models promote the ability to translate and coordinate between 2D and 3D molecular structure representations? A case study with Algerian students, Chem. Educ. Res. Pract., 17(4), 862–877.
- Nickel S., Brockmüller S., Boone W. J. and Habig S., (2025), Measuring representational competence–analyses of dimensionality and the relationship to general content knowledge, Int. J. Sci. Educ., 1–25.
- Olimpo J. T., Kumi B. C., Wroblewski R. and Dixon B. L., (2015), Examining the relationship between 2D diagrammatic conventions and students’ success on representational translation tasks in organic chemistry, Chem. Educ. Res. Pract., 16(1), 143–153.
- Oliver-Hoyo M. and Babilonia-Rosa M. A., (2017), Promotion of Spatial Skills in Chemistry and Biochemistry Education at the College Level, J. Chem. Educ., 94(8), 996–1006.
- Padalkar S. and Hegarty M., (2013), Undergraduate Students’ Understanding About Representations in Chemistry, Proc. EpiSTEME, 5, 288–294.
- Pande P., Shah P. and Chandrasekharan S., (2015), How Do Experts and Novices Navigate Chemistry Representations – an Eye-Tracking Investigation, in Proceedings of EPISTEME-6, Mumbai, India, pp 102–109.
- Patron E., Wikman S., Edfors I., Johansson-Cederblad B. and Linder C., (2017), Teachers’ reasoning: classroom visual representational practices in the context of introductory chemical bonding, Sci. Educ., 101(6), 887–906.
- Patton M. Q., (2002), Qualitative Research and Evaluation Methods, Sage Publications, 3rd edn.
- Popova M. and Bretz S. L., (2018), Organic chemistry students’ interpretations of the surface features of reaction coordinate diagrams, Chem. Educ. Res. Pract., 19(3), 919–931.
- Popova M. and Jones T., (2021), Chemistry instructors’ intentions toward developing, teaching, and assessing student representational competence skills, Chem. Educ. Res. Pract., 2(3), 733–748.
- Pruitt J. and Aldin T., (2006), The persona lifecycle: Keeping people in mind throughout product design, Amsterdam, Boston: Elsevier/Morgan Kaufmann Publishers.
- Rau M. A., (2017), How do Students Learn to See Concepts in Visualizations? Social Learning Mechanisms with Physical and Virtual Representations, J. Learn. Anal., 4(2), 240–263.
- Rotich F., Ward L., BeckC. and Popova M., (2024), Attention is currency: how surface features of Lewis structures influence organic chemistry student reasoning about stability, Chem. Educ. Res. Pract., 25, 1071–1089.
- Saldaña J., (2013), The Coding Manual for Qualitative Researchers, in Seaman J. (ed.), Thousand Oaks, CA: Sage Publications, 2nd edn.
- Sandi-Urena S., Loría Cambronero G. and Jinesta Chaves D., (2020), Conceptualisation of Lewis structures by chemistry majors, Chem. Teacher Int., 2(1), 791–849.
- Shane J. W. and Bodner G. M., (2006), General Chemistry Students’ Understanding of Structure-Function Relationships, Chem. Educ., 11, 1–8.
- Stains M. and Talanquer V., (2008), Classification of chemical reactions: stages of expertise, J. Res. Sci. Teach., 45(7), 771–793.
- Stieff M., (2007), Mental rotation and diagrammatic reasoning in science, Learn. Instruct., 17(2), 219–234.
- Stieff M., (2010), When Is a Molecule Three Dimensional? A Task-specific Role for Imagistic Reasoning in Advanced Chemistry, Sci. Educ., 95(2), 310–336.
- Stieff M. and Raje S., (2010), Expert algorithmic and imagistic problem solving strategies in advanced chemistry, Spatial Cogn. Comput., 10(1), 53–81.
- Stieff M., Ryu M., Dixon B. and Hegarty M., (2012), The Role of Spatial Ability and Strategy Preference for Spatial Problem Solving in Organic Chemistry, J. Chem. Educ., 89(7), 854–859.
- Stieff M., Lira M. and Desutter D., (2014), Representational Competence and Spatial Thinking in STEM, in ICLS Proceedings, pp. 987–991.
- Stieff M., Lira M. E. and Scopelitis S. A., (2016), Gesture Supports Spatial Thinking in STEM, Cogn. Instruct., 34(2), 80–99.
- Stieff M., Origenes A., DeSutter D., Lira M., Banevicius L., Tabang D. and Cabel G., (2018), Operational constraints on the mental rotation of STEM representations, J. Educ. Psychol., 110(8), 1160–1174.
- Talanquer V., (2022), The Complexity of Reasoning about and with Chemical Representations, JACS Au, 2(12), 2658–2669.
- Tibshirani R., Walther G. and Hastie T., (2001), Estimating the number of clusters in a data set via the gap statistics, J. R. Stat. Soc., Ser. B: Methodol., 63(2), 411–423.
- Tiettmeyer J. M., Coleman A. F., Balok R. S., Gampp T. W., Duffy P. L., Mazzarone K. M. and Grove N. P., (2017), Unraveling the Complexities: An Investigation of the Factors That Induce Load in Chemistry Students Constructing Lewis Structures, J. Chem. Educ., 94(3), 282–288.
- Van Meter P. and Firetto C. M., (2013), Cognitive Model of Drawing Construction, Learning Through Visual Displays, pp. 247–280.
- Ward L. W., Rotich F., Hoang J. and Popova M., (2022), Representational Competence Under the Magnifying Glass—The Interplay Between Student Reasoning Skills, Conceptual Understanding, and the Nature of Representations, in Graulich N. and Shultz G. (ed.), Student Reasoning in Organic Chemistry: Research Advances and Evidence-based Instructional Practices, Royal Society of Chemistry, pp. 36–55.
- Ward L. W., Rotich F., Raker J. R., Komperda R., Nedungadi S. and Popova M., (2025), Design, development, and evaluation of the organic chemistry representational competence assessment (ORCA), Chem. Educ. Res. Pract., 26, 244–258.
- Wright L. and Oliver-Hoyo M. T., (2020), Student assumptions and mental models encountered in IR spectroscopy instruction, Chem. Educ. Res. Pract., 21(1), 426–437.
- Xue D. and Stains M., (2020), Exploring Students’ Understanding of Resonance and Its Relationship to Instruction, J. Chem. Educ., 97(4), 894–902.
- Zagallo P., McCourt J., Idsardi R., Smith M. K., Urban-Lurain M., Andrews T. C., Haudek, K., Knight J. K., Merrill J., Nehm R., Prevost L. B. and Lemons P. P., (2019), Through the eyes of faculty: using personas as a tool for learner-centered professional development, CBE Life Sci. Educ., 18(4), 1–21.
| This journal is © The Royal Society of Chemistry 2026 |