 Open Access Article
Open Access ArticleOxidation of dibenzyl ether with nitric acid to benzaldehyde in continuous-flow microreactors
Xiang
Li
a,
Saier
Liu
a,
Minjing
Shang
*a and
Yuanhai
Su
 *ab
*ab
aState Key Laboratory of Polyolefins and Catalysis, School of Chemistry and Chemical Engineering, Shanghai Jiao Tong University, Shanghai 200240, PR China. E-mail: mshang@sjtu.edu.cn; y.su@sjtu.edu.cn; Tel: +86 21 54738710
bKey Laboratory of Thin Film and Microfabrication (Ministry of Education), Shanghai Jiao Tong University, Shanghai 200240, PR China
First published on 26th February 2026
Abstract
As an important chemical, benzaldehyde (BzH) can be synthesized by oxidizing dibenzyl ether (DBE) with nitric acid (HNO3). This is an environmentally friendly and chlorine-free production process, but it has problems such as intense in situ gas generation, low space–time yield and safety issues. This study developed a safe and efficient continuous-flow synthesis method using a packed-bed microreactor. Five critical parameters including reaction residence time, glass bead particle size, temperature, oxidant concentration, and initiator dosage were systematically investigated. Under optimal conditions (T = 85 °C, d = 104.5 μm, HNO3 eq. = 3 and NaNO2 eq. = 0.009), the conversion of DBE and the yield of BzH reached 80.11% and 96.35% after 17.92 min. The space–time yield was twice that of capillary microreactors and 40 times that of batch reactors. The reasons for in situ gas production in capillary microreactors and its negative impacts were explored. A kinetic model was developed, providing significant theoretical insights and practical value for better understanding the oxidation process and optimizing the process parameters.
1. Introduction
Benzaldehyde (BzH) is the simplest and most widely used aromatic aldehyde in industry and an important chemical raw material and intermediate product in many industrial processes, such as the synthesis of fiber auxiliaries, dyes, fragrances, medicines, seasonings, plastics, and coatings. In industry, there are several methods for producing BzH, mainly including the chlorination and hydrolysis of toluene, oxidation of benzyl alcohol, direct oxidation of toluene, direct formylation of benzene, and reduction of benzoic acid. In the reported literature, different raw materials, such as benzyl alcohol, styrene and toluene, were used to synthesize BzH through different reaction pathways with different catalysts, with a selectivity as high as 99%.1–6 However, all of these methods are highly dependent on catalytic systems, and the long reaction time up to several hours is typically needed to achieve the high conversion and selectivity.Some researchers have attempted to use low-value dibenzyl ether (DBE) as a raw material to synthesize BzH.7 DBE as a main by-product from the industrial production of benzyl alcohol has a large output but limited applications.8–14 Thus, dibenzyl ether becomes an intractable waste. As a result, the study of producing BzH from DBE has considerable research value. At present, there are relatively few studies on the reaction mechanism of generating BzH from the oxidation of DBE by nitric acid (HNO3). From the existing literature, the reaction mechanism can be inferred as shown in Fig. 1. First of all, HNO3 reacts with nitrous acid to produce nitrogen dioxide containing free radicals. Subsequently, nitrogen dioxide radicals attack the carbon near the ether bond in DBE, generating DBE radicals. Nitrogen dioxide radicals continue to attack this carbon until the ether bond on the other side breaks, generating BzH and benzyl nitrite. Benzyl nitrite, under the oxidation of HNO3, generates BzH and nitrite. Some of the accumulated nitrous acid will continue to react with HNO3 to form nitrogen dioxide radicals, while the other part will self-react to form HNO3 and nitric oxide. Nitric oxide can be oxidized by excess HNO3 to form nitrogen dioxide.15–18 When the amount of HNO3 is insufficient, benzyl nitrite will hydrolyze into benzyl alcohol and nitrous acid.17 In the case of excess HNO3, benzyl alcohol will react with the excess HNO3 to form BzH, which can be further oxidized to form benzoic acid by the excess HNO3.17
At present, research on the oxidation of DBE to BzH by HNO3 is mostly found in patents, which is often carried out in batch reactors. Since the reaction is relatively intense at high temperatures and generates a large amount of NOX gas, researchers can only adopt the dropwise feeding method to control the reaction conditions.19 Therefore, the reaction process is limited by its mass transfer efficiency, resulting in a long reaction time and low production efficiency.
A microreactor refers to a device with a certain microstructure manufactured through precision processing technology. As a core part of microchemical technology, it can take advantage of its characteristic dimension ranging from sub-microns to sub-millimeters to achieve the purposes of increasing specific surface area, improving mixing efficiency, enhancing mass and heat transfer rates and reducing safety risks. On the one hand, by taking advantage of the large specific surface area and low resistance to heat and mass transfer, chemical reaction systems controlled by mass and heat transfer can be enhanced in microreactors.20 On the other hand, the production processes with high safety hazards in traditional chemical industry, e.g., high-temperature and high-pressure reactions,21 peroxidation reactions,22,23 oxidation reactions,24 nitrification reactions,25 sulfonation reactions,26 hydrogenation reactions,27 polymerization reactions,28etc., can be improved by the use of microreactors.29 In addition, it is convenient to explore reaction kinetics and hydromechanics,30–32 develop various photochemical reaction paths,33 and transform some batch processes to continuous-flow processes in microreactors.34,35 Microreactors can be applied for multi-phase reactions, such as liquid–liquid two-phase,36,37 gas–liquid two-phase,38,39 and gas–liquid–liquid three-phase.40,41
The generation of in situ gas is a complex process for capillary microreactors. For instance, introducing inert gas can effectively enhance the liquid–liquid mass transfer rate,42,43 but sometimes it may actually reduce the reaction performance.44 However, using packed bed microreactors can effectively reduce the adverse impacts brought about by the generation of in situ gas.36,45
The research objective of this work is to regulate and optimize the rapid and highly exothermic reaction for producing BzH from DBE and HNO3 in microreactors and to establish a low-energy consumption, efficient and safe production method. Furthermore, it aims to deeply explore the reaction mechanism and hydrodynamic characteristics, which can guide the further expanding production of BzH.
2. Experiment
2.1 Materials
Nitric acid (AR), sodium nitrite (AR), methyl orange (AR), and sodium bicarbonate (AR) were purchased from Sinopharm Chemical Reagent Co., LTD. (Shanghai, China), and dibenzyl ether (≥99%, DBE as its abbreviation) was purchased from Adamas Beta (Shanghai) Chemical Reagent Co., LTD. (Shanghai, China). N-Decane (≥99%) and benzyl alcohol (AR) were purchased from Shanghai Aladdin Biochemical Technology Co., LTD. (Shanghai, China), and benzaldehyde (≥99%, BzH as its abbreviation) was purchased from Shanghai Mindray Biochemical Technology Co., LTD. (Shanghai, China). Benzoic acid (AR), n-hexane (AR), sodium chloride (AR), and anhydrous ethanol (AR) were purchased from Shanghai Titan Technology Co., LTD. (Shanghai, China). All reagents were used directly without further purification.2.2 Experimental setup and procedure
The schematic diagram of experimental setup for the oxidation of DBE by HNO3 to form BzH in a capillary microreactor or a packed-bed microreactor is shown in Fig. 2. The capillary microreactor system consisted of two micro-mixers and PFA tubes that were 1–10.5 meters long and had an inner diameter of 1 mm. The packed-bed microreactor system consisted of a micro-mixer and a 20 cm long glass tube with an inner diameter of 12 mm, in which inert glass beads (with a particle size of 104.5 μm) were filled. As a rapid reaction, there is no need to use solid catalysts instead of glass beads as the filler. Glass beads were used as filling materials to increase the surface area and promote the mixing and contact between reactants in the tube. HNO3, DBE and sodium nitrite solutions were introduced into the microreactor system by three syringe pumps. After premixing in the micromixers, the two-phase mixture was introduced into a capillary microreactor or a packed-bed microreactor in which the oxidation reaction took place. The reaction temperature (55–85 °C) was controlled by a constant temperature water bath circulator. The residual substances were collected from the exit of the microreactor and quenched in a small bottle containing ice water. The upper organic phase was separated from the aqueous phase through a separating funnel. Each experiment was repeated three times to control the experimental error below 5%. And the average measurement value was provided as the final result. | ||
| Fig. 2 Schematic representation of the capillary microreactor for the synthesis of BzH: (a) the capillary microreactor; (b) the packed-bed microreactor. | ||
2.3 Analysis
The concentration of HNO3 (CHNO3) in the aqueous phase was determined by titration. Because the cost of DBE is much higher than that of HNO3 and NaNO2, it was regarded as a key component. The organic product samples mainly contained BzH, DBE and benzyl nitrite, while the contents of other polar components could be ignored. After dilution with n-hexane, the organic product samples were analyzed by GC (Agilent 7890B, Japan) equipped with an HP-5 column (HP-5 column, 30 m × 0.32 mm × 0.25 μm) (for details, see the SI). The conversion of DBE (XDBE) is calculated by eqn (1): | (1) |
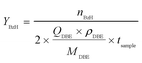 | (2) |
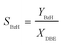 | (3) |
3. Results and discussion
3.1 Comparison of reaction performance in different reactors
The packed-bed microreactor can also solve the problem of reduced interfacial area due to the gas production. According to our previous work, the velocity of the generated gas in packed-bed microreactors is much higher than that of the liquid.36 It is beneficial for avoiding the effect of the in situ produced gas phase to the reactions to some certain extent. When the residence time was 24 min, the yield of BzH in the packed-bed microreactor was 75.47%, approximately twice that of the capillary microreactor. In contrast to the batch reactor, no by-product benzyl alcohol was produced in the packed-bed microreactor, and the reaction time was considerably reduced. In the packed-bed microreactor, the conversion of BzH could reach 60.24% in just 11.1 min, while in the batch reactor, it took more than 120 min to achieve the same level. Furthermore, the packed-bed microreactor has lower liquid holdup, and thus the oxidation process can be conducted at a higher reaction temperature with ensured safety.
3.2 Reaction condition dependence in packed-bed microreactors
| Glass bead particle size (μm) | Reactor volume (mL) |
|---|---|
| 104.5 | 8.06 |
| 167.9 | 8.29 |
| 380.2 | 8.49 |
3.3 Kinetic study on the synthesis of BzH from DBE and HNO3
Based on the obtained experimental results and the mechanism shown in Fig. 1, a kinetic model of the oxidation reaction between HNO3 and DBE was established. The reaction network is as shown in eqn (4) and (5). | (4) |
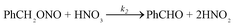 | (5) |
Some assumptions are made for the reaction kinetics establishment as follows: (a) the gas–liquid–liquid three-phase can be simplified to the liquid–liquid two-phase since the velocity of the gas phase produced in the packed-bed microreactor is much higher than that of the liquid phase. (b) Due to the fact that the yields of benzoic acid and benzyl alcohol in the packed-bed microreactor were less than 5%, the further oxidation of BzH is ignored.
Therefore, the simplified reaction kinetics of the formation of BzH from DBE oxidized by HNO3 can be expressed as eqn (6)–(10), which is fitted using the least squares fitting function in Python:
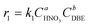 | (6) |
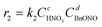 | (7) |
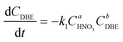 | (8) |
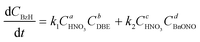 | (9) |
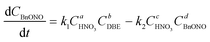 | (10) |
The rate constants and reaction orders of various reactants can be fitted based on the experimental data. After obtaining the reaction rate constants at different temperatures, the relationship between ln(k) and 1/T was correlated, as shown in Fig. 9(a) and (b). According to Arrhenius equation, the activation energy (Ea1) for the oxidation of DBE to BzH and benzyl nitrite could be determined to be 85.54 kJ mol−1, and the activation energy (Ea2) for the oxidation of benzyl nitrite to BzH could be determined to be 15.64 kJ mol−1. The results in Fig. 10 indicate that the calculated conversion of DBE fitted well with the experimental data. Finally, the complete reaction kinetics can be established as the eqn (11) and (12):
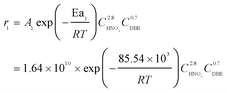 | (11) |
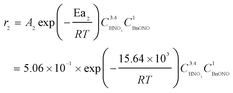 | (12) |
The Ha number was calculated using eqn (13) with the kinetic expressions established in the previous text (for details, see the SI) and is shown in Fig. 11, indicating that the mass transfer effect has been eliminated.47
 | (13) |
As shown in Fig. 12(a) and (b), the concentration of BzH and the DBE at different reaction temperatures and residence time for most of the cases involved in this work, especially at the intermediate, the predicted values of the reaction kinetics model fit well with the experimental results. However, when the reaction temperature rose to 328.15 K some of the calculated concentrations based on the kinetic model have certain errors compared with the experimental data. Especially when the residence time was 8.7 minutes, the predicted value did not fit well with the experimental value. This is because the model does not take into account the further oxidation reaction of BzH. When the temperature is high, HNO3 will further oxidize BzH to benzoic acid. In addition, the reactions among HNO3, HNO2 and NOX will also become more complex.
 | ||
| Fig. 12 Comparison between the experimental and predicted values of the concentrations of reactants and products at different temperatures by the reaction kinetic model: (a) BzH; (b) DBE. | ||
4. Conclusions
Here, we report the efficient and safe synthesis of BzH from DBE with HNO3 in microreactors for the first time. The reaction performance in the capillary microreactor, packed-bed microreactor and batch reactor was compared. The results showed that in the capillary microreactor, due to the in situ gas production, the interfacial area of liquid–liquid two phases decreased from the initial 3.54 × 103 m2 m−3 to 0.23 × 103 m2 m−3, resulting in limited reaction efficiency. In contrast, the packed-bed microreactor, by enhancing mass transfer and rapid gas discharge, achieved a BzH yield of 60.24% at a residence time of 11.1 min. This significantly reduced the reaction time compared to the batch reactor (which required more than 120 min), and no by-product (benzyl alcohol) was produced. According to the calculation, the space–time yield of the packed-bed microreactor was approximately twice that of the capillary microreactor and 40 times that of the batch reactor.By calculating the residence time distribution in the packed-bed microreactor, Bo number was obtained to be 170.38. The back mixing was relatively low and the flow in the packed-bed microreactor could be approximately regarded as a plug flow, which was suitable for consecutive reactions. Five parameters including the residence time, glass bead particle size, temperature, oxidant concentration and initiator dosage were investigated. It was confirmed that increasing the residence time, temperature and oxidant concentration was conducive to improving the conversion of DBE, up to 97% at most. And the yield of BzH first went up and then slowed down and the maximum could reach up to 80%. The increase in the temperature and oxidant concentration could effectively reduce the residence time. The glass bead particle size and initiator dosage had a relatively weak influence on the conversion of DBE and the yield of BzH. Under the optimal reaction conditions (T = 85 °C, d = 104.5 μm, HNO3 eq. = 3 and NaNO2 eq. = 0.009), the conversion rate of DBE and the yield of BzH reached 80.11% and 96.35% respectively at a residence time of 17.92 min.
A simplified reaction kinetics model was established. The first step reaction of HNO3 and DBE has a reaction order of 2.8 and 0.7, with an activation energy of 85.54 kJ mol−1; the second step reaction of HNO3 and benzenesulfonic anhydride has a reaction order of 3.4 and 1, with an activation energy of 15.64 kJ mol−1. The predicted concentration values of the model were in good agreement with the experimental data, indicating that this model can guide the optimization and scale-up study of the continuous-flow synthesis of BzH from DBE and HNO3.
Author contributions
Xiang Li: writing – original draft, investigation, writing –review & editing, and methodology; Saier Liu: writing –review & editing and investigation; Yuanhai Su: writing – review& editing, funding acquisition, supervision, and conceptualization; Minjing Shang: writing – review & editing and supervision.Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying this article will be shared on reasonable request to the corresponding author.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6re00004e.
Acknowledgements
We would like to acknowledge financial support from the National Natural Science Foundation of China (No. 22378258 and 22178215).References
- X. L. Bao, H. L. Li, Z. Y. Wang, F. X. Tong, M. Liu, Z. K. Zheng, P. Wang, H. F. Cheng, Y. Y. Liu, Y. Dai, Y. C. Fan, Z. Y. Li and B. B. Huang, Appl. Catal., B, 2021, 286, 119885 CrossRef CAS.
- C. Cheng, B. C. Zhu, B. Cheng, W. Macyk, L. X. Wang and J. G. Yu, ACS Catal., 2023, 13, 459–468 CrossRef CAS.
- X. H. Ren, Y. B. Wang, J. Li, K. F. Wang, M. Y. Shi, J. M. Li, J. D. Wang and C. Y. Guo, Appl. Surf. Sci., 2025, 705, 163434 CrossRef CAS.
- Y. X. Tan, Z. M. Chai, B. H. Wang, S. Tian, X. X. Deng, Z. J. Bai, L. Chen, S. Shen, J. K. Guo, M. Q. Cai, C. T. Au and S. F. Yin, ACS Catal., 2021, 11, 2492–2503 CrossRef CAS.
- M. Tayyab, Y. J. Liu, S. X. Min, R. M. Irfan, Q. H. Zhu, L. Zhou, J. Y. Lei and J. L. Zhang, Chin. J. Catal., 2022, 43, 1165–1175 CrossRef CAS.
- B. Xing, T. Wang, Z. Q. Zheng, S. J. Liu, J. J. Mao, C. Li and B. X. Li, Chem. Eng. J., 2023, 461, 141871 CrossRef CAS.
- V. P. Nekhoroshkov, G. L. Kamalov and V. I. Melnik, React. Kinet. Catal. Lett., 1984, 24, 103–105 CrossRef CAS.
- Y. S. Guan, W. Zhao, C. Zhao, D. K. Wang, L. Ao, K. S. Liu, H. M. Liu, S. L. Chen, X. Fan and X. Y. Wei, ChemistrySelect, 2018, 3, 11610–11615 CrossRef CAS.
- Q. S. Guo, Q. Miao, H. L. Wei, J. H. He, X. Y. Zhou, M. Liu, J. H. Xie and W. Jiang, Ind. Eng. Chem. Res., 2024, 63, 14018–14028 CrossRef CAS.
- Z. L. Jiang, S. B. Zhu, X. F. Tang, H. B. Tang, X. M. Zhu, L. Yu, L. T. Li, Y. D. Wang, H. L. Tang and X. L. Liu, Sustainable Energy Fuels, 2025, 9, 2421–2432 RSC.
- S. A. Krokhmal and T. N. Zueva, Probl. Atom. Sci. Technol., 2021, 111–114 Search PubMed.
- Z. Y. Li, L. Ju, R. P. Bao, X. B. Lin, T. Wang and X. H. Yu, Synlett, 2025, 36, 480–483 CrossRef CAS.
- M. X. Zhao, X. Y. Wei, M. Qu, J. Kong, Z. K. Li, J. Liu and Z. M. Zong, Fuel, 2016, 183, 531–536 CrossRef CAS.
- Y. N. Zheng, X. L. Song, Y. X. Fu and L. G. Gao, J. Saudi Chem. Soc., 2024, 28, 101825 CrossRef CAS.
- H. Feilchenfeld, S. Manor and J. A. Epstein, J. Chem. Soc., Dalton Trans., 1972, 2675–2680 RSC.
- Y. OGATA, in Oxidation in Organic Chemistry 5-C, ed. W. S. Trahanovsky, Elsevier Science & Technology, United States, 1978, vol. 5, pp. 295–342 Search PubMed.
- P. Strazzolini and A. Runcio, Eur. J. Org. Chem., 2003, 2003, 526–536 CrossRef.
- Z. F. Yan, G. Lee, J. Deng and G. S. Luo, Chem. Eng. Sci., 2025, 301, 120757 CrossRef CAS.
- S. R. Joshi, S. B. Sawant and J. B. Joshi, Org. Process Res. Dev., 2001, 5, 152–157 CrossRef CAS.
- K. Wang, Y. Lu, H. Shao and G. Luo, Ind. Eng. Chem. Res., 2008, 47, 9754–9758 CrossRef CAS.
- X. H. Zhang, Z. Chen, J. Chen and J. H. Xu, Chem. Eng. Sci., 2024, 288, 119777 CrossRef CAS.
- M. Qiu, L. Xiang, M. Shang and Y. Su, Adv. Polym. Technol., 2021, 2966912–2966920 Search PubMed.
- S. K. Xia, T. Yang, Z. Chen and J. H. Xu, Chem. Eng. Sci., 2024, 290, 119826 CrossRef CAS.
- G. Li, S. Liu, X. Dou, M. Shang, Z. Luo and Y. Su, Ind. Eng. Chem. Res., 2021, 60, 9389–9398 CrossRef CAS.
- Z. H. Wen, M. Yang, S. N. Zhao, F. Zhou and G. W. Chen, React. Chem. Eng., 2018, 3, 379–387 RSC.
- T. M. Xie, C. F. Zeng, C. Q. Wang and L. X. Zhang, Ind. Eng. Chem. Res., 2013, 52, 3714–3722 CrossRef CAS.
- N. Joshi and A. Lawal, Chem. Eng. Sci., 2012, 84, 761–771 CrossRef CAS.
- L. Xiang, Z. Zhong, S. Liu, M. Shang, Z. Luo and Y. Su, Ind. Eng. Chem. Res., 2022, 61, 15917–15932 CAS.
- K. Schubert, J. Brandner, M. Fichtner, G. Linder, U. Schygulla and A. Wenka, Microscale Thermophys. Eng., 2001, 5, 17–39 CrossRef CAS.
- S. Liu, G. Li, M. Shang, Z. H. Luo and Y. Su, AIChE J., 2021, 67, e17362 CrossRef CAS.
- M. Pasha, S. Liu, J. Zhang, M. Qiu and Y. Su, Ind. Eng. Chem. Res., 2022, 61, 12249–12268 CrossRef CAS.
- Z. F. Yan, J. X. Tian, C. C. Du, J. Deng and G. S. Luo, Chin. J. Chem. Eng., 2022, 41, 49–72 CrossRef CAS.
- Y. H. Su, N. Straathof, V. Hessel and T. Noël, Chem. - Eur. J., 2014, 20, 10562–10589 CrossRef CAS PubMed.
- L. Malet-Sanz and F. Susanne, J. Med. Chem., 2012, 55, 4062–4098 CrossRef CAS PubMed.
- J. Yoshida, Y. Takahashi and A. Nagaki, Chem. Commun., 2013, 49, 9896–9904 RSC.
- Y. S. Liu, M. J. Shang, S. E. Liu, X. Xue, Z. H. Zhong, Q. Niu and Y. H. Su, Chem. Eng. Sci., 2024, 299, 120499 CrossRef CAS.
- C. Shen, Q. B. Zheng, M. J. Shang, L. Zha and Y. H. Su, AIChE J., 2020, 66, e16260 CrossRef CAS.
- N. Straathof, Y. H. Su, V. Hessel and T. Noël, Nat. Protoc., 2016, 11, 10–21 CrossRef CAS PubMed.
- J. Zhang, K. Wang, X. Lin, Y. Lu and G. Luo, AIChE J., 2014, 60, 2724–2730 CrossRef CAS.
- Y. Liu, G. Chen and J. Yue, J. Flow Chem., 2020, 10, 103–121 CrossRef CAS.
- J. W. Zhang, Z. Chen and J. H. Xu, AIChE J., 2024, e18554 Search PubMed.
- T. F. Feng, J. Tan, W. S. Deng and Y. F. Su, Chem. Eng. Sci., 2018, 177, 270–283 CrossRef CAS.
- Y. H. Su, G. W. Chen, Y. C. Zhao and Q. Yuan, AIChE J., 2009, 55, 1948–1958 CrossRef CAS.
- A. Ufer, D. Sudhoff, A. Mescher and D. W. Agar, Chem. Eng. J., 2011, 167, 468–474 CrossRef CAS.
- Y. S. Liu, M. J. Shang, Q. Niu and Y. H. Su, Chem. Eng. Sci., 2025, 317, 121987 CrossRef CAS.
- N. M. Kashid, in Microstructured Devices for Chemical Processing, ed. A. Renken and L. Kiwi-Minsker, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2014, vol. 8, pp. 89–128 Search PubMed.
- T. Noël, Y. H. Su and V. Hessel, in Organometallic Flow Chemistry, ed. T. Noel, Springer Group, Berlin, 2016, vol. 57, pp. 1–41 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |










