Open Access Article
Open Access ArticleSulfonic-acid functionalized hypercrosslinked porous organic polymer as a highly efficient heterogeneous catalyst for synthesis of 2H-chromene derivatives
Siriphong Somprasong ,
Thanchanok Ratvijitvech
,
Thanchanok Ratvijitvech ,
Thanakorn Tiyawarakul
,
Thanakorn Tiyawarakul ,
Chaiwat Rujirasereesakul,
Thanakorn Sitthasakul and
Torsak Luanphaisarnnont
,
Chaiwat Rujirasereesakul,
Thanakorn Sitthasakul and
Torsak Luanphaisarnnont *
*
Department of Chemistry and Center of Excellence for Innovation in Chemistry, Faculty of Science, Mahidol University, Bangkok 10400, Thailand. E-mail: torsak.lua@mahidol.ac.th
First published on 12th May 2026
Abstract
A sulfonic acid-functionalized hypercrosslinked porous organic polymer (HCP-H-SO3H) with high surface area (SBET = 604 m2 g−1) was successfully synthesized via a Friedel–Crafts alkylation of benzene and dimethoxymethane, followed by sulfonation of aromatic rings using sulfuric acid. This material was used as an efficient heterogeneous catalyst in a cyclization between various ortho-hydroxy aromatic aldehydes and acetylenic diesters, affording the corresponding 2H-chromenes in moderate to excellent yields. The material could be reused for at least five reaction cycles, suggesting the sustainable application potential of this sulfonic-acid functionalized materials as a heterogeneous acid catalyst. A mechanistic investigation using 13C NMR kinetic isotope effect at natural abundance experiments provided insights into a similar nature of the rate-limiting steps in the catalytic processes of the homogeneous catalyst (p-toluenesulfonic acid monohydrate; PTSA·H2O) and the heterogeneous catalyst (HCP-H-SO3H).
1 Introduction
2H-Chromeme and its derivatives are prevalent heterocyclic aromatic structural units in natural products and pharmaceutical targets,1 as well as in organic synthesis and material science.2 Over the past several decades, numerous homogeneous catalytic methods have been disclosed for the synthesis of 2H-chromenes. Transition metals3a–d and organo-catalysts3e–j,4 have been reported as efficient catalysts for the reaction. Recently, our research group reported a selective synthesis of 2H-chromenes via an intermolecular annulation reaction of ortho-hydroxy aromatic aldehydes with acetylenic diesters using a dual-organocatalytic system, a combination of p-toluenesulfonic acid monohydrate (PTSA·H2O) and pyrrolidine, under mild conditions giving the products in excellent yields.4Heterogeneous catalysis plays a key role in chemistry because of its eco-friendly conditions, reusability, and convenient separation from the reaction mixture.5 Recently, functionalized hypercrosslinked porous organic polymers (HCPs) have attracted significant attention owing to their high potential applications in gas storage and separations,6 small-molecules capture,7 drug delivery vehicle,8 antimicrobial materials,9 light-harvesting and light-emitting applications,10 sensors,11 energy storage and conversion,12 and catalysis of various important organic transformations.13 There are many advantages of HCPs over other porous materials such as high surface areas, good accessibilities to a broad range of monomers, convenient surface functionalization methods, low skeleton densities, and thermal and chemical stabilities.
The use of heterogeneous catalyst for the synthesis of 2H-chromenes from ortho-hydroxy aromatic aldehydes and acetylenic diesters such as silica gel,14 imidazole-functionalized silica nanoparticles,15 and ZnO nanoparticles have been reported.16 However, compared to homogeneous catalysts, these heterogeneous catalysts still have some limitations such as lower reactivity, poor selectivity of the conjugate addition products (2H-chromenes versus 4H-chromenes), and narrow substrate scopes.3c–e,4 Development of a new synthetic method using a heterogeneous catalyst to overcome this limitation is therefore important. We envision that the sulfonic-acid functionalized hypercrosslinked porous organic polymer may act as a highly efficient heterogeneous catalyst for the syntheses of 2H-chromenes. Previous reports have used chlorosulfuric acid as a sulfonating agent for various HCPs;17 however, the use of readily available sulfuric acid for sulfonation has not been thoroughly studied. Furthermore, although there have been reports on the use of heterogeneous acid catalysts for biomass valorization to biofuels and bioproducts18 and various organic reactions,19 the use of the sulfonic-acid functionalized material for mechanistically complex cyclization such as the synthesis of 2H-chromene derivatives has not been reported. Additionally, a mechanistic investigation to compare the reaction behaviors of a homogeneous sulfonic-acid catalyst and a heterogeneous sulfonic-acid catalyst has not been studied.
Herein, we reported a successful synthesis of derivatives of sulfonic-acid functionalized hypercrosslinked porous organic polymer (HCP-X-SO3H) using readily available concentrated sulfuric acid as a sulfonating agent. We also explored the application of the material as a heterogeneous catalyst in a complex cyclization to synthesize 2H-chromene derivatives. In addition, we used 13C NMR kinetic isotope effect experiments to study the rate-determining steps of the reaction catalyzed by homogeneous PTSA·H2O and that catalyzed by heterogeneous HCP-H-SO3H to compare the behaviors of the two catalysts.
2 Results and discussion
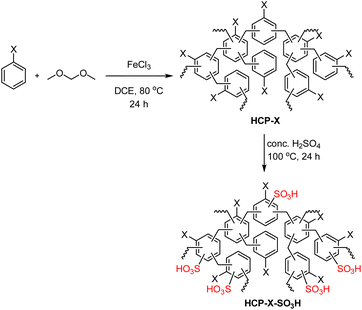
2.1 Synthesis of HCP-X-SO3H
Preparation of the HCP-X-SO3H involved a two-step procedure containing polymerization and sulfonation (a general reaction scheme is shown in Table 1). First, the non-functionalized hypercrosslinked porous organic polymers (HCP-X) were synthesized via Friedel–Crafts alkylation of benzene derivatives and dimethoxymethane using FeCl3 as a mediator. The polymers were obtained as red-brown solid in excellent yield (82–>99% yield).6f,20 For the sulfonation step, reaction optimization was performed using HCP-H as a model substrate. The reaction parameters under optimization included the concentration of H2SO4, the reaction temperature, and the ratio of HCP-H to H2SO4 (see SI). The optimized reaction conditions were conc. H2SO4 at 100 °C, leading to the formation of the HCP-H-SO3H as a black solid in 78% yield. The acid concentration was determined using a back acid–base titration method (see SI). The calculated acid concentration of HCP-H-SO3H was 4.16 mmol g−1. The scope of the sulfonation was investigated using substrates with various substituents. HCP-X with sterically different substituents (X = CH3, C2H5, n-C3H7, and i-C3H7) were sulfonated using the optimized reaction conditions to obtain HCP-X-SO3H in good to excellent yields (74–84% yield). The acid concentrations of these sulfonated polymers were determined to range from 3.81 to 4.11 mmol g−1. The similar yields and acid concentrations of sterically different substrates suggested that the steric property of the substituent on the HCP-X had minimal effects on the efficiency of the sulfonation method.| Entry | X = | Yield (%) | Acid concentration of HCP-X-SO3Hc (mmol g−1) | |
|---|---|---|---|---|
| HCP-Xa | HCP-X-SO3Hb | |||
| a Reaction conditions: benzene derivative (1.0 equiv.), dimethoxymethane (2.0 equiv.), and FeCl3 (2.0 equiv.) in DCE (120 mL) at 80 °C for 24 h.b Reaction conditions: HCP-X (1.0 equiv.) and conc. H2SO4 (50.0 equiv.) at 100 °C for 24 h.c The acid concentration of HCP-X-SO3H was calculated by the back acid-base titration method. | ||||
| 1 | H | >99 | 78 | 4.16 |
| 2 | CH3 | 96 | 74 | 3.81 |
| 3 | C2H5 | >99 | 82 | 4.11 |
| 4 | nC3H7 | 99 | 84 | 3.95 |
| 5 | iC3H7 | 82 | 81 | 4.04 |
2.2 Characterization of HCP-H-SO3H
After synthesis, the HCP-X derivatives were characterized, using Fourier transform infrared (FTIR) spectroscopy. The FTIR spectra of HCP-H and HCP-H-SO3H were shown in Fig. 1 and S3. The peaks located between 2900 and 3000 cm−1 corresponded to the C–H stretching vibration of methylene and benzene moieties. The peaks at approximately 1600 cm−1 corresponded to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations of benzene in the skeletons of the HCPs. The new peaks corresponding to the sulfonic acid groups were found in the FTIR spectrum of HCP-H-SO3H at approximately 1227 cm−1 and 1189 cm−1, which corresponded to the symmetric and asymmetric O
C stretching vibrations of benzene in the skeletons of the HCPs. The new peaks corresponding to the sulfonic acid groups were found in the FTIR spectrum of HCP-H-SO3H at approximately 1227 cm−1 and 1189 cm−1, which corresponded to the symmetric and asymmetric O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S
S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching of the sulfonic acid functional group.21 The peaks at approximately 1037 cm−1 and 625 cm−1 were responsible for the S–O stretching vibration21b,22 and the characteristic C–S stretching vibration,19d,23 respectively. The FTIR data suggested a successful installation of the sulfonic acid functional group onto the surface of the porous hypercrosslinked organic polymer. Similar trends were also observed in other FTIR spectra of HCP-X and HCP-X-SO3H (see SI).
O stretching of the sulfonic acid functional group.21 The peaks at approximately 1037 cm−1 and 625 cm−1 were responsible for the S–O stretching vibration21b,22 and the characteristic C–S stretching vibration,19d,23 respectively. The FTIR data suggested a successful installation of the sulfonic acid functional group onto the surface of the porous hypercrosslinked organic polymer. Similar trends were also observed in other FTIR spectra of HCP-X and HCP-X-SO3H (see SI).
To confirm the porous structure of HCP-H-SO3H, the porosity and the surface area of the material were further investigated using N2 adsorption–desorption analysis at 77 K (Fig. 2a). The HCP-H-SO3H gave a typical type IV isotherm with a significant hysteresis loop, indicating the presence of micropores and mesopores in the network.24 The adsorption isotherm displayed the steep nitrogen gas uptake at low relative pressure (P/P0 < 0.1), which reflected the abundant microporous structure. The rise at higher pressure indicated the presence of the larger pores (mesopores and macropores) in the material. The N2 sorption analysis exhibited a high Brunauer–Emmett–Teller (BET) surface area for HCP-H-SO3H of about 604 m2 g−1 with a total pore volume of approximately 0.41 cm3 g−1. The pore size distribution using the nonlocal density functional theory (NLDFT) for pillared clay model of the hypercrosslinked porous organic polymer after grafting of the sulfonic acid moiety exhibited a dominant pore width centered at ∼0.88–1.45 nm (Fig. 2b), suggesting the microporous structure in the material. In addition, the mesopores were also found in the material. The presence of large pore in material is also good for performing acid catalyzed-intermolecular cyclization of small organic molecules.25
 | ||
| Fig. 2 (a) N2 adsorption (closed symbols)–desorption (open symbols) isotherm and (b) pore size distribution calculated from NLDFT for pillared clay model of HCP-H-SO3H | ||
The thermal stabilities of the synthesized HCP-H and HCP-H-SO3H were studied by thermogravimetric analysis (TGA) at a heating rate of 10 °C min−1 under nitrogen atmosphere in the temperature range of 35 to 800 °C (Fig. 3). The mass loss at the initial heating stage before 100 °C were about ∼7% and ∼14% in HCP-H and HCP-H-SO3H, respectively owing to the evaporation of absorbed water molecules. The HCP-H was stable up to around 400 °C. A significant decrease in weight of about 25% was observed in the temperature range of 400 to 600 °C. On the other hand, the TGA plot of the HCP-H-SO3H showed the thermal stability of HCP-H-SO3H up to 200 °C before a sharp decrease in weight of about 27% was observed in the temperature range of 250 to 650 °C.26 The TGA results revealed that the materials were thermally stable at least up to 200 °C, which were in the compatibility range for the catalytic reaction of 2H-chromene synthesis.
The morphology of the HCP-H and HCP-H-SO3H were studied using field-emission scanning electron microscopy (FE-SEM) images. As showed in Fig. 4, both materials retained the irregular and agglomerated small particles in a range of ten to hundred nanometers. The FE-SEM images also confirmed that the morphology of the polymers did not change during the sulfonation process (Fig. 4c and d). Moreover, scanning electron microscopy with energy-dispersive X-ray spectroscopy (SEM-EDX) elemental mapping showed that sulfur and oxygen elements were evenly distributed along with carbon elements on the surface of HCP-H-SO3H (Fig. 5), revealing that sulfonic acid moiety were successfully introduced into the material. In addition, weight percentages of sulfur and oxygen increased to 8.13% and 32.65% in HCP-H-SO3H compared to 0.37% and 23.83% in HCP-H, respectively (see SI).
 | ||
| Fig. 5 (a) FE-SEM image and SEM-EDX elemental mappings of (b) carbon, (c) oxygen, and (d) sulfur for HCP-H-SO3H. | ||
2.3 Catalytic activity
The catalytic performance of the sulfonic-acid functionalized porous organic polymer HCP-H-SO3H was investigated in the synthesis of 2H-chromene derivatives. The reaction between salicylaldehyde (1a) and dimethyl acethylenedicarboxylate (2a) was chosen as the model reaction to optimize the reaction conditions. To understand the effect of both the acidic polymeric catalyst and the basic organocatalyst on this reaction, several other catalysts were also examined (Table 2). In the absence of the amine organocatalyst, only unreacted starting material was recovered (entries 1–3). The reaction without the acidic polymeric catalyst and the reaction with non-sulfonated HCP-H gave only a trace amount of the product 3aa (entries 4 and 5). The desired product 3aa could be obtained in 20% yield, using a combination of HCP-H-SO3H and pyrrolidine in a catalytic amount (entry 6). This result suggested that both acidic and basic catalysts were crucial in this reaction. The catalytic loading of the HCP-H-SO3H had a significant effect in the reaction. Increasing the acidic polymeric catalyst loading up to 10 mol% of HCP-H-SO3H led to a higher yield of the product (entries 6–9). However, a higher amount of the catalyst (20 mol%) led to a decrease in the yield (entry 9). Next, the type of base was investigated. Other amines such as piperidine, piperazine, and benzylamine gave the desired product 3aa in lower yields (entries 10–12). DABCO did not give any product (entry 13). Pyrrolidine was the optimal basic catalyst in the reaction, giving the product in the highest yield. Decreasing or increasing the pyrrolidine loading resulted in a reduction of the product yields (entries 14–17).| Entry | Catalyst (mol%) | Amine catalyst (mol%) | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: 1a (0.25 mmol, 1 equiv.), 2a (0.3 mmol, 1.2 equiv.), HCP-H-SO3H (10 mol%), and pyrrolidine (30 mol%) in EtOH (0.5 mL) at 75 °C for 8 h.b Determined by crude 1H NMR analysis. | |||
| 1 | — | — | 0 |
| 2 | HCP-H (10 mg) | — | 0 |
| 3 | HCP-H-SO3H (10) | — | 0 |
| 4 | — | Pyrrolidine (30) | Trace |
| 5 | HCP-H (10 mg) | Pyrrolidine (30) | Trace |
| 6 | HCP-H-SO3H (2.5) | Pyrrolidine (30) | 20 |
| 7 | HCP-H-SO3H (5) | Pyrrolidine (30) | 24 |
| 8 | HCP-H-SO3H (10) | Pyrrolidine (30) | 62 |
| 9 | HCP-H-SO3H (20) | Pyrrolidine (30) | 51 |
| 10 | HCP-H-SO3H (10) | Piperidine (30) | 38 |
| 11 | HCP-H-SO3H (10) | Piperazine (30) | 41 |
| 12 | HCP-H-SO3H (10) | Benzylamine (30) | 12 |
| 13 | HCP-H-SO3H (10) | DABCO (30) | 0 |
| 14 | HCP-H-SO3H (10) | Pyrrolidine (10) | 13 |
| 15 | HCP-H-SO3H (10) | Pyrrolidine (20) | 32 |
| 16 | HCP-H-SO3H (10) | Pyrrolidine (40) | 51 |
| 17 | HCP-H-SO3H (10) | Pyrrolidine (50) | 51 |
To further improve the yield of 2H-chromene 3aa, other parameters were investigated (Table 3). The solvent screening was performed. Alcoholic solvents such as EtOH, MeOH, tBuOH, and iPrOH afforded the desired product 3aa in 36–62% yields (entries 1–4). Water and aprotic solvents such as THF, MeCN, EtOAc, toluene, and CH2Cl2 were ineffective and gave 3aa in lower yields (entries 5–10). Subsequently, the effect of reaction temperature and time was investigated. Decreasing the temperature from 75 °C to room temperature and 60 °C did not increase the yield of the product (entries 11 and 12). Increasing the reaction time from 8 to 16 hours gave 3aa in 67% yield (entry 13); however, further prolonging the reaction time showed no improvement in the product yield (entry 14). Increasing the equivalent of 2a to 1.5 equivalent improved the yield of 3aa to 73% isolated yield (entry 15). The reaction also worked well at a larger scale, giving the product in 79% isolated yield (entry 16).
| Entry | Solvent | Temp (°C) | Time | Yieldb (%) |
|---|---|---|---|---|
| a Reaction conditions: 1a (0.25 mmol, 1 equiv.), 2a (0.3 mmol, 1.2 equiv.), HCP-H-SO3H (10 mol%), and pyrrolidine (30 mol%) in solvent (0.5 mL).b Determined by crude 1H NMR analysis. The value in parenthesis is an isolated yield.c 2a (0.375 mmol, 1.5 equiv.) was used for 16 h.d 1.0 mmol of 1a scale. | ||||
| 1 | EtOH | 75 | 8 | 62 |
| 2 | MeOH | 75 | 8 | 36 |
| 3 | tBuOH | 75 | 8 | 49 |
| 4 | iPrOH | 75 | 8 | 44 |
| 5 | H2O | 75 | 8 | 16 |
| 6 | THF | 75 | 8 | Trace |
| 7 | MeCN | 75 | 8 | Trace |
| 8 | EtOAc | 75 | 8 | Trace |
| 9 | PhMe | 75 | 8 | Trace |
| 10 | CH2Cl2 | 75 | 8 | 0 |
| 11 | EtOH | RT | 8 | 22 |
| 12 | EtOH | 60 | 8 | 26 |
| 13 | EtOH | 75 | 16 | 67 |
| 14 | EtOH | 75 | 24 | 59 |
| 15c | EtOH | 75 | 16 | 75 (73) |
| 16d | EtOH | 75 | 16 | 84 (79) |
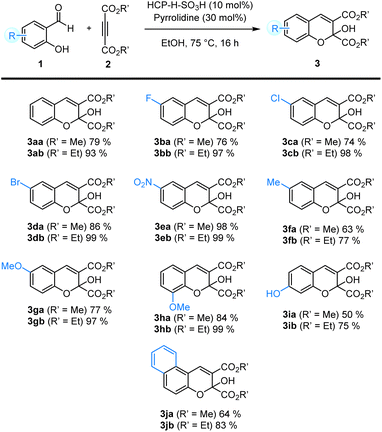
With the optimized reaction conditions in hand, we studied the substrate scope of the cyclization reaction using ortho-hydroxy aromatic aldehydes and dialkyl acethylene-dicarboxylates with various substituents (Table 4). Salicylaldehydes with different substituents worked well in the reaction, giving the 2H-chromene products in moderate to excellent yields. Substrates with electron-withdrawing substituents such as fluoro, chloro, bromo, and nitro groups provided the corresponding product 3ba–3ea in 74–98% yields. In contrast, salicylaldehydes with electron donating substituents such as methoxy and methyl groups gave the desired products 3fa–3ha in slightly lower yields (63–84%). The reaction of 2,5-dihydroxybenzaldehyde gave the desired product 3ia in only 50% yield. In addition, the reaction of 2-hydroxy-1-naphthaldehyde gave the corresponding product 3ja in 64% yield. When diethyl acethylenedicarboxylate 2b was used instead of dimethyl acethylenedicarboxylate 2a, the corresponding 2H chromene products 3ab–3jb were obtained in good to excellent yields (75–99% yields).
| a Reaction conditions: salicylaldehyde 1a–j (1.0 mmol, 1.0 equiv.), dialkyl acetylenedicarboxylate 2a–b (1.5 mmol, 1.5 equiv.), HCP-H-SO3H (10 mol%), and pyrrolidine (30 mol%) in EtOH (2.0 mL) at 75 °C for 16 h. |
|---|
 |
2.4 Reusability of the catalyst
The recyclability of the heterogeneous hypercrosslinked porous organic polymer was investigated using the reaction of o-vanillin (1h) and alkynoic diester 2b under the optimized reaction conditions as the model reaction (Table 5). After the completion of each run of the reaction, the HCP-H-SO3H catalyst was recovered by a simple filtration, washing, and drying processes. The catalyst can be used under the optimized reaction conditions for at least five consecutive runs with a minimal loss in the yields of the product. This result showed that the HCP-H-SO3H catalyst was highly stable in the optimized reaction conditions.| Run | Yield of 3hb (%) |
|---|---|
| a Reaction conditions: 1h (1.0 mmol, 1.0 equiv.) and 2b (1.5 mmol, 1.5 equiv,), HCP-H-SO3H (10 mol%), and pyrrolidine (30 mol%) in EtOH (2.0 mL) at 75 °C for 16 h. | |
| 1 | 99 |
| 2 | 99 |
| 3 | 99 |
| 4 | 98 |
| 5 | 96 |
2.5 Mechanistic investigation
Kinetic isotope effects (KIEs) is an important tool to study the change in bonding in the rate-determining step.27 To gain further insight into the nature of the rate-limiting step in the catalytic cyclization of salicylaldehyde and dialkyl acetylenedicarboxylate, competitive 13C KIEs were determined by natural abundant 12C/13C ratios using Singleton's 13C NMR methodology for product analysis.28 The reaction of 5-bromosalicylaldehyde (1d) and alkynoic diester 2a catalyzed by an acid catalyst was chosen for the determination of experimental 13C KIEs at natural abundance. The KIE measurements in the reaction were determined at low conversion to ensure that the faster reacting isotope of the product is enriched. Quantitative 13C measurements were carried out for the reactions in the presence of HCP-H-SO3H and PTSA·H2O as the heterogeneous catalyst and the corresponding homogeneous catalyst, respectively. To determine the relative proportion of 13C isotope compared to the original starting substrate, C4 in 1d and the methyl group (Cd) in 2a were used as an internal standard with the assumption that its isotopic composition does not change (see SI).The KIEs results were summarized in Table 6. The result from the reactions with PTSA·H2O and HCP-H-SO3H showed the same trend. The normal KIE was observed at C7 and Cb of the product 3da, indicating that both carbons were involved in the rate-determining step (RDS) in the catalytic process. Other carbon positions have near-unity KIEs. The results quantitatively validated our hypothesis that the carbon–carbon bond formation step may be important in the rate-determining step of the reaction and a plausible mechanism of 2H-chromene synthesis in the presence of HCP-H-SO3H as an acid catalyst is similar to that of the homogeneous catalytic reaction.
| Quantitative 13C KIEs | Acid catalyst | ||
|---|---|---|---|
| PTSA·H2Ob | HCP-H-SO3Hc | ||
| a Experimental 13C KIEs for product 3da.b Three experiments were carried to 15%, 16%, and 17% conversion to determine the KIEs for PTSA·H2O catalysis.c Three experiments were carried to 6%, 8%, and 10% conversion to determine the KIEs for HCP-H-SO3H catalysis. Each reaction was performed in five independent quantitative 13C NMR measurements. The numbers in parenthesis represent the standard deviation in the last digit. | |||
| KIEs determined from 1d | C1 | 1.001 (5) | 1.000 (1) |
| C2 | 1.000 (2) | 1.000 (1) | |
| C4 | 1.000 (assumed) | ||
| C5 | 1.003 (1) | 0.999 (1) | |
| C6 | 1.003 (1) | 1.003 (2) | |
| C7 | 1.039 (2) | 1.043 (1) | |
| KIEs determined from 2a | Ca | 0.998 (2) | 1.000 (1) |
| Cb | 1.022 (1) | 1.023 (1) | |
| Cc | 1.001 (4) | 1.000 (1) | |
| Cd | 1.000 (assumed) | ||
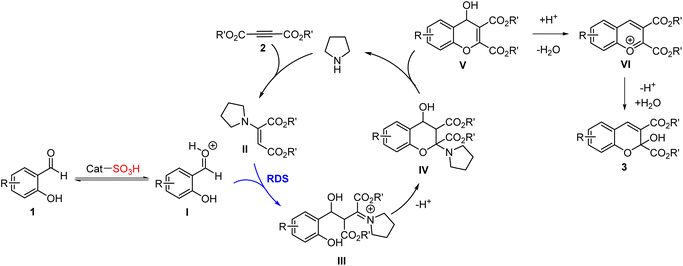
From the basis of the results and previous reports,4,29 a plausible mechanism for the formation of 2H-chromene products 3 in the presence of HCP-H-SO3H and pyrrolidine as the catalysts was outlined in Scheme 1. First, the carbonyl group of ortho-hydroxy aromatic aldehydes was activated by HCP-H-SO3H to form intermediate I. Meanwhile a conjugate addition reaction between acetylenic diester 2 and pyrrolidine gave the enamino ester intermediate II. Subsequently, the intermediate II reacted with the activated intermediate I to produce intermediate III. This step was proposed to be the rate-determining step. Next, an intramolecular nucleophilic attack by a hydroxyl group to the electron-deficient carbon atom of the iminium resulted in the six-membered ring intermediate IV. Subsequent elimination of pyrrolidine led to 4H-chromene intermediate V. Finally, transformation of the intermediate V to the desired 2H-chromene product 3 occurred via an isomerization process through a benzopyrylium intermediate VI.
3 Experimental section
3.1 General information
All reactions were performed under an ambient atmosphere in oven-dried glassware with magnetic stirrer. Reactions conducted above an ambient temperature were heated by an anodized aluminum block. Starting reagents and organic solvents were purchased from commercial sources (Sigma-Aldrich, TCI, Merck, and Alfa Aesar) and were used without further purification unless otherwise noted. Analytical thin layer chromatography (TLC) was performed on alumina sheets pre-coated with a Merck silica gel 60 F254 plate and compounds were visualized under UV light. Purification of reaction products was carried out by column chromatography, in which a Merck silica gel 60 (0.063–0.200 mm) was used as a stationary phase. Proton nuclear magnetic resonance (1H NMR) and proton-decoupled carbon nuclear magnetic resonance (13C NMR) spectra were recorded on a Bruker Avance 400 MHz and JEOL 400 MHz NMR spectrometer in deuterated chloroform (CDCl3) and deuterated acetone ((CD3)2CO). The chemical shifts were recorded in part per million (ppm) relative to the resonance of the residual protonated solvent (1H: CDCl3, δ = 7.24 ppm; acetone-d6, δ = 2.05 ppm and 13C: CDCl3, δ = 77.23 ppm; acetone-d6, δ = 29.84 ppm). Data are reported as following: (brs = broad, s = singlet, d = doublet, t = triplet, m = multiplet, dd = doublet of doublet, td = triplet of doublet; coupling constants, J, in Hz, integration). Melting points were determined in open glass capillaries using a Buchi melting point M-565 apparatus. FTIR spectra were recorded on a PerkinElmer Frontier FTIR spectrometer. The nitrogen adsorption–desorption isotherm (77 K) was obtained using Micromeritics 3Flex gas sorption analyser. Surface area was calculated in the range of 0.05–0.15 P/P0 using Brunauer–Emmett–Teller (BET) theory. TGA was performed under a flow of nitrogen by heating from room temperature to 800 °C with 10 °C min−1 on TA Instruments SDT 2960. SEM and EDX data were obtained from Field Emission Scanning Electron Microscopes (FE-SEM) (HITACHI SU-8010) mass spectrometric data were obtained with high resolution mass spectra (HRMS) on a Bruker micrOTOF spectrometer in the ESI mode.3.2 Synthesis of HCP-H
To a solution of benzene (4.68 g, 60 mmol, 1.0 equiv.) and dimethoxymethane (9.13 g, 120 mmol, 2.0 equiv.) in dichloroethane 120 mL was added FeCl3 (19.5 g, 120 mmol, 2.0 equiv.). The reaction was refluxed with stirring for 24 hours. The reaction was filtered. The brown solid was washed with methanol by a Soxhlet extractor until the yellow solution changed to colorless. The brown solid was dried in an oven for 24 hours. The reaction gave a brown solid as the product (3.60 g, quantitative yield).3.3 Synthesis of HCP-H-SO3H and acid concentration measurement
The reaction of HCP-H (2.01 g, 20 mmol, 1.0 equiv.) in conc. H2SO4 (56 mL, 50 equiv.) was stirred at 100 °C for 24 hours. The reaction was washed with H2O until the pH of the aqueous solution was neutral. The black solid was dried in an oven for 24 hours. The reaction gave a black solid as the product (2.80 g, 78%). The acid concentration was calculated by a back titration method. HCP-H-SO3H (20 mg, 0.2 mmol, 1.0 equiv.) was stirred with 0.0250 M NaOH (10 mL) for 1 hour. After that, the reaction was filtered and washed with water until the pH was neutral. The volume of the aqueous phase was adjusted to 100 mL, and 10 mL of the solution was pipetted to a flask containing 0.0250 M HCl solution (10 mL). The solution was titrated with 0.0125 M NaOH. The acid concentration in HCP-H-SO3H was determined to be 4.16 mmol g−1.3.4 General procedure for the synthesis of 2H-chromene derivatives
To a suspension of salicylaldehydes (1a–j, 1.0 mmol, 1.0 equiv.) and HCP-H-SO3H (0.10 mmol, 10 mol%) in ethanol (2.0 mL) was added dialkyl acetylenedicarboxylate (2a–b, 1.5 mmol, 1.5 equiv.) and pyrrolidine (24.6 µL, 0.30 mmol, 30 mol%). The reaction was stirred at 75 °C for 16 hours. The reaction mixture was filtered through a PTFE syringe filter, and the filter was thoroughly washed successively with water, methanol, and acetone. The filtrate was extracted with EtOAc, washed with brine, dried over anhydrous Na2SO4, and filtered. The solvent was removed in vacuo, and the crude product was purified by flash column chromatography (silica gel: EtOAc/hexane) to give the pure product 3.3.5 Recycling of the HCP-H-SO3H catalyst
After completion of the catalytic reaction, the acid catalyst HCP-H-SO3H was recovered through filtration for further use and thoroughly washed successively with water, methanol, and acetone. Then the recovered catalyst was dried overnight in an oven 120 °C.3.6 13C NMR kinetic isotope effects at natural abundance
The reaction between 5-bromosalicylaldehyde (1d) and dimethylacethylenedicarboxylate (2a) were chosen to determine the KIEs. The PTSA·H2O catalyzed cyclization reaction was repeated three times, giving the following yields: 15%, 16%, and 17%. The HCP-H-SO3H catalyzed cyclisation reaction was repeated three times, giving the following yields: 6%, 8%, and 10%. The percent yield of product was assumed as percent conversion. Two reference starting materials (1d and 2a) were used. The NMR samples were prepared in deuterated chloroform (CDCl3). The quantitative 13C NMR spectra were taken at 100 MHz on a Bruker Avance 400 MHz NMR spectrometer with inverse-gated 1H decoupling which used 30° pulses (zgig30 pulse program). Acquisition parameters were as follows: acquisition time 5.2 s; spectral width 240 ppm, size of fid 250k; recovery delays 75 s; size of real spectrum 64k points; transmitter frequency offset 110 ppm; number of dummy scans 8; pre-scan delay 50 µs; number of scans 512. 13C NMR measurements were carried out for the KIE values of the product 3da. 13C NMR data were processed using 1 Hz exponential multiplication. For the KIE determination, the integration of C4 was set to 100 when 1d was used as a reference, and the integration of Cd was set to 100 when 2a as a reference. The average integration values for the other carbons were used to calculate the KIE values using eqn (1).
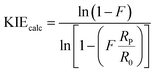 | (1) |
4 Conclusions
In summary, sulfonic-acid functionalized hypercrosslinked porous organic polymers (HCP-X-SO3H) were successfully prepared by a Friedel–Crafts alkylation and a sulfonation using readily available sulfuric acid. HCP-H-SO3H was used as an efficient heterogeneous catalyst for the synthesis of 2H-chromene derivatives from various ortho-hydroxy aromatic aldehydes and acetylenic diesters under mild reaction conditions. The catalyst could be recovered and reused for at least five times with minimal loss in the product yield. Mechanistic investigation of the cyclization reaction with heterogeneous HCP-H-SO3H catalyst and that with homogeneous PTSA·H2O catalyst using 13C KIEs suggested that the nature of their rate-determining steps was similar. Due to their high catalytic efficiency and stability, the sulfonic-acid functionalized hypercrosslinked porous organic polymers have great potential as an efficient heterogeneous acid catalyst for a wide range of mechanistically complex reactions. Application of the catalyst for other reactions is ongoing and will be reported in due course.Author contributions
Siriphong Somprasong designed the project, optimized the reaction conditions, investigated the substrate scope, analyzed the data, performed the mechanistic experiments, and wrote the draft of the manuscript. Thanchanok Ratvijitvech designed the project, synthesized and characterized hypercrosslinked porous organic polymers, analyzed the data, wrote the draft of the manuscript, and edited the manuscript. Thanakorn Tiyawarakul synthesized and characterized hypercrosslinked porous organic polymers. Chaiwat Rujirasereesakul performed mechanistic experiments. Thanakorn Sitthasakul performed mechanistic experiments. Torsak Luanphaisarnnont designed the project, analyzed the data, supervised the project, secured the funding, wrote the draft of the manuscript, and edited the manuscript. All the authors contributed to and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Experimental and analytical data supporting this article are available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra03062a.Acknowledgements
This research was supported by Mahidol University (Fundamental Fund: fiscal year 2025 by National Science Research and Innovation Fund (NSRF), FF-075/2568). Support from the Center of Excellence for Innovation in Chemistry (PERCH-CIC), Ministry of Higher Education, Science, Research and Innovation is gratefully acknowledged. We are also grateful for the support from the Department of Chemistry and the Central Instrumental Facility (CIF), Faculty of Science, Mahidol University for research facilities.Notes and references
-
(a) N. Majumdar, N. D. Paul, S. Mandal, B. de Bruin and W. D. Wulff, ACS Catal., 2015, 5, 2329–2366 CrossRef CAS
; (b) R. Pratap and V. J. Ram, Chem. Rev., 2014, 114, 10476–10526 CrossRef CAS PubMed
.
-
(a) A. D. Pugachev, M. B. Lukyanova, B. S. Lukyanov, I. V. Ozhogin, A. S. Kozlenko, I. A. Rostovtseva, N. I. Makarova, V. V. Tkachev and N. A. Aksenov, J. Mol. Struct., 2019, 1178, 590–598 CrossRef CAS
; (b) Y. Yue, C. Yin, F. Huo, J. Chao and Y. Zhang, Sens. Actuators, B, 2016, 223, 496–500 CrossRef CAS
; (c) S. N. Corns, S. M. Partington and A. D. Towns, Color. Technol., 2009, 125, 249–261 CrossRef CAS
.
- For selected examples of synthesis of 2H-chromenes, see:
(a) Q. J. Yang, Y. B. Wang, S. H. Luo and J. Wang, Angew. Chem., Int. Ed., 2019, 58, 5343–5347 CrossRef CAS PubMed
; (b) P. N. Moquist, T. Kodama and S. E. Schaus, Angew. Chem., Int. Ed., 2010, 49, 7096–7100 (Angew. Chem., 2010, 122, 7250–7254) CrossRef CAS PubMed
; (c) H. Hu, K.-Y. Ye, Q.-F. Wu, L.-X. Dai and S.-L. You, Adv. Synth. Catal., 2012, 354, 1084–1094 CrossRef
; (d) B.-S. Zeng, X.-Y. Yu, P. W. Siu and K. A. Scheidt, Chem. Sci., 2014, 5, 2277–2281 RSC
; (e) H. Li, J. Wang, T. E-Nunu, L. Zu, W. Jiang, S. Wei and W. Wang, Chem. Commun., 2007, 43, 507–509 RSC
; (f) D.-Q. Xu, Y.-F. Wang, S.-P. Luo, S. Zhang, A.-G. Zhong, H. Chen and Z.-Y. Xu, Adv. Synth. Catal., 2008, 350, 2610–2616 CrossRef CAS
; (g) H. Cai, L. Xia, Y. R. Lee, J.-J. Shim and S. H. Kim, Eur. J. Org Chem., 2015, 5212–5220 CrossRef CAS
; (h) G. Maiti, R. Karmakar, U. Kayal and R. N. Bhattacharya, Tetrahedron, 2012, 68, 8817–8822 CrossRef CAS
; (i) J. Yang, J.-N. Tan and Y. Gu, Green Chem., 2012, 14, 3304–3317 RSC
; (j) M. Rueping, U. Uria, M.-Y. Lin and I. Atodiresei, J. Am. Chem. Soc., 2011, 133, 3732–3735 CrossRef CAS PubMed
.
- S. Somprasong, W. Prasitwatcharakorn and T. Luanphaisarnnont, Tetrahedron Lett., 2020, 61, 152402 CrossRef CAS
.
-
(a) J. M. Thomas, R. Raja and D. W. Lewis, Angew. Chem., Int. Ed., 2005, 44, 6456–6482 CrossRef CAS PubMed
; (b) A. Álvarez, A. Bansode, A. Urakawa, A. V. Bavykina, T. A. Wezendonk, M. Makkee, J. Gascon and F. Kapteijn, Chem. Rev., 2017, 117, 9804–9838 CrossRef PubMed
; (c) L. Liu and A. Corma, Chem. Rev., 2018, 118, 4981–5079 CrossRef CAS PubMed
; (d) K. Huang, J.-Y. Zhang, F. Liu and S. Dai, ACS Catal., 2018, 8, 9079–9102 CrossRef CAS
; (e) S. Kim, E. E. Kwon, Y. T. Kim, S. Jung, H. J. Kim, G. W. Hubere and J. Lee, Green Chem., 2019, 21, 3715–3743 RSC
; (f) H. Liu, L. Wei, F. Liu, Z. Pei, J. Shi, Z.-J. Wang, D. He and Y. Chen, ACS Catal., 2019, 9, 5245–5267 CrossRef CAS
; (g) S. De, A. Dokania, A. Ramirez and J. Gascon, ACS Catal., 2020, 10, 14147–14185 CrossRef CAS
; (h) R. Nie, Y. Tao, Y. Nie, T. Lu, J. Wang, Y. Zhang, X. Lu and C. Xu, ACS Catal., 2021, 11, 1071–1095 CrossRef CAS
.
-
(a) Z. Chang, D.-S. Zhang, Q. Chen and X.-H. Bu, Phys. Chem. Chem. Phys., 2013, 15, 5430–5442 RSC
; (b) H. Furukawa and O. M. Yaghi, J. Am. Chem. Soc., 2009, 131, 8875–8883 CrossRef CAS PubMed
; (c) T. Ben, C. Pei, D. Zhang, J. Xu, F. Deng, X. Jing and S. Qiu, Energy Environ. Sci., 2011, 4, 3991–3999 RSC
; (d) R. Dawson, A. I. Cooper and D. J. Adams, Polym. Int., 2013, 62, 345–352 CrossRef CAS
; (e) P. Lindemann, M. Tsotsalas, S. Shishatskiy, V. Abetz, P. KrollaSidenstein, C. Azucena, L. Monnereau, A. Beyer, A. Gölzhäuser, V. Mugnaini, H. Gliemann, S. Bräse and C. Wöll, Chem. Mater., 2014, 26, 7189–7193 CrossRef CAS
; (f) R. T. Woodward, L. A. Stevens, R. Dawson, M. Vijayaraghavan, T. Hasell, I. P. Silverwood, A. V. Ewing, T. Ratvijitvech, J. D. Exley, S. Y. Chong, F. Blanc, D. J. Adams, S. G. Kazarian, C. E. Snape, T. C. Drage and A. I. Cooper, J. Am. Chem. Soc., 2014, 136(25), 9028–9035 CrossRef CAS PubMed
.
-
(a) K. V. Rao, S. Mohapatra, T. K. Maji and S. J. George, Chem.–Eur. J., 2012, 18, 4505–4509 CrossRef CAS PubMed
; (b) J. F. Van Humbeck, T. M. McDonald, X. Jing, B. M. Wiers, G. Zhu and J. R. Long, J. Am. Chem. Soc., 2014, 2432–2440 CrossRef CAS PubMed
; (c) B. Li, F. Su, H.-K. Luo, L. Liang and B. Tan, Microporous Mesoporous Mater., 2011, 138, 207–214 CrossRef CAS
; (d) T. Ratvijitvech, J. Polym. Environ., 2020, 28, 2211–2218 CrossRef CAS
.
- S. Bhunia, N. Chatterjee, S. Das, K. Das Saha and A. Bhaumik, ACS Appl. Mater. Interfaces, 2014, 6, 22569–22576 CrossRef CAS PubMed
.
-
(a) T. Ratvijitvecha and S. Na Pombejra, Mater. Today Commun., 2021, 28, 102617 CrossRef
; (b) D. Li, Y. Fang and X. Zhang, ACS Appl. Mater. Interfaces, 2020, 12(8), 8989–8999 CrossRef CAS PubMed
; (c) A. Mohan, M. H. Al-Sayah, A. Ahmed and O. M. El-Kadri, Sci. Rep., 2022, 12, 2638 CrossRef CAS PubMed
.
-
(a) Y. Xu, L. Chen, Z. Guo, A. Nagai and D. Jiang, J. Am. Chem. Soc., 2011, 133, 17622–17625 CrossRef CAS PubMed
; (b) J. X. Jiang, A. Trewin, D. J. Adams and A. I. Cooper, Chem. Sci., 2011, 2, 1777–1781 RSC
; (c) L. Chen, Y. Honsho, S. Seki and D. Jiang, J. Am. Chem. Soc., 2010, 132, 6742–6748 CrossRef CAS PubMed
.
- X. Liu, Y. Xu and D. Jiang, J. Am. Chem. Soc., 2012, 134, 8738–8741 CrossRef CAS PubMed
.
-
(a) F. Vilela, K. Zhang and M. Antonietti, Energy Environ. Sci., 2012, 5, 7819–7832 RSC
; (b) R. S. Sprick, J.-X. Jiang, B. Bonillo, S. Ren, T. Ratvijitvech, P. Guiglion, M. A. Zwijnenburg, D. J. Adams and A. I. Cooper, J. Am. Chem. Soc., 2015, 137, 3265–3270 CrossRef CAS PubMed
; (c) Y. Kou, Y. Xu, Z. Guo and D. Jiang, Angew. Chem., Int. Ed., 2011, 50, 8753–8757 CrossRef CAS PubMed
.
-
(a) P. Kaur, J. T. Hupp and S. T. Nguyen, ACS Catal., 2011, 1, 819–835 CrossRef CAS
; (b) Y. Zhang and S. N. Riduan, Chem. Soc. Rev., 2012, 41, 2083–2094 RSC
; (c) C. Thiamsiri, T. Ratvijitvech and T. Luanphaisarnnont, Mater. Adv., 2023, 4, 5184–5190 RSC
; (d) P. Schweng, D. Guerin-Faucheur, F. Kleitz and R. T. Woodward, Chem. Commun., 2024, 60, 14395–14398 RSC
.
- N. Noshiranzadeh and A. Ramazani, Synth. Commun., 2007, 37, 3181–3189 CrossRef CAS
.
- H. Valizadeh, L. Dinparast, S. Noorshargh and M. M. Heravi, C. R. Chem., 2016, 19, 395–402 CrossRef
.
- L. Dinparast and H. Valizadeh, Monatsh. Chem., 2015, 146, 313–319 CrossRef CAS
.
-
(a) R. M. N. Kalla, M.-R. Kim and I. Kim, Ind. Eng. Chem. Res., 2018, 57, 11583–11591 CrossRef CAS
; (b) S. S. Reddy, R. M. N. Kalla, A. Varyambath and I. Kim, Catal. Commun., 2019, 126, 15–20 CrossRef CAS
; (c) A. Blocher, F. Mayer, P. Schweng, T. M. Tikovits, N. Yousefi and R. T. Woodward, Mater. Adv., 2022, 3, 6335–6342 RSC
.
-
(a) W. Guan, C.-W. Tsang, C. S. K. Lin, C. Len, H. Hu and C. Lianga, Bioresour. Technol., 2020, 298, 122432 CrossRef CAS PubMed
; (b) S. M. Silva, A. F. Peixoto and C. Freire, Renewable Energy, 2020, 146, 2416–2429 CrossRef CAS
; (c) S. M. Silva, A. F. Peixoto and C. Freire, Appl. Catal., A, 2020, 568, 221–230 CrossRef
; (d) S. Mondal, J. Mondal and A. Bhaumik, ChemCatChem, 2015, 7, 3570–3578 CrossRef CAS
; (e) M. Du, A. M. Agrawal, S. Chakraborty, S. J. Garibay, R. Limvorapitux, B. Choi, S. T. Madrahimov and S. T. Nguyen, ACS Sustainable Chem. Eng., 2019, 7, 8126–8135 CrossRef CAS
; (f) B. Agarwal, K. Kailasam, R. S. Sangwan and S. Elumalai, Renewable Sustainable Energy Rev., 2018, 82, 2408–2425 CrossRef CAS
; (g) R. T. Woodward, M. Kessler, S. Lima and R. Rinaldi, Green Chem., 2018, 20, 2374–2381 RSC
.
-
(a) J. R. Pereira, M. C. Corvo, A. F. Peixoto, A. Aguiar-Ricardo and M. M. B. Marques, ChemCatChem, 2023, 15, e202201318 CrossRef CAS
; (b) A. Y. Sidorenko, N. S. Li-Zhulanov, P. Mäki-Arvela, T. Sandberg, A. V. Kravtsova, A. F. Peixoto, C. Freire, K. P. Volcho, N. F. Salakhutdinov, V. E. Agabekov and D. Yu Murzin, ChemCatChem, 2020, 12, 2605–2609 CrossRef CAS
; (c) L. Sekerová, P. Březinová, T. T. Do, E. Vyskočilová, J. Krupka, L. Červený, L. Havelková, B. Bashta and J. Sedláček, ChemCatChem, 2020, 12, 1075–1084 CrossRef
; (d) S. Mondal, B. C. Patra and A. Bhaumik, ChemCatChem, 2017, 9, 1469–1475 CrossRef CAS
; (e) B. Li, K. Leng, Y. Zhang, J. J. Dynes, J. Wang, Y. Hu, D. Ma, Z. Shi, L. Zhu, D. Zhang, Y. Sun, M. Chrzanowski and S. Ma, J. Am. Chem. Soc., 2015, 137, 4243–4248 CrossRef CAS PubMed
; (f) M. Hara, T. Yoshida, A. Takagaki, T. Takata, J. N. Kondo, S. Hayashi and K. Domen, Angew. Chem., Int. Ed., 2004, 43, 2955–2958 CrossRef CAS PubMed
.
- R. Dawson, T. Ratvijitvech, M. Corker, A. Laybourn, Y. Z. Khimyak, A. I. Cooper and D. J. Adams, Polym. Chem., 2012, 3, 2034–2038 RSC
.
-
(a) J. Juan-Alcañiz, R. Gielisse, A. B. Lago, E. V. Ramos-Fernandez, P. Serra-Crespo, T. Devic, N. Guillou, C. Serre, F. Kapteijna and J. Gasco, Catal. Sci. Technol., 2013, 3, 2311–2318 RSC
; (b) M. G. Goesten, À. Szécsényi, M. F. de Lange, A. V. Bavykina, K. B. Sai Sankar Gupta, F. Kapteijn and J. Gascon, ChemCatChem, 2016, 8, 961–967 CrossRef CAS
.
- S. Suganuma, K. Nakajima, M. Kitano, S. Hayashi and M. Hara, ChemSusChem, 2012, 5, 1841–1846 CrossRef CAS PubMed
.
- W.-Y. Lou, Q. Guo, W.-J. Chen, M.-H. Zong, H. Wu and T. J. Smith, ChemSusChem, 2012, 5, 1533–1541 CrossRef CAS PubMed
.
- R. Gomes and A. Bhaumik, J. Solid State Chem., 2015, 222, 4–11 CrossRef
.
- B. Coasne, New J. Chem., 2016, 40, 4078–4094 RSC
.
-
(a) M. Halder, P. Bhanja, M. M. Islam, S. Chatterjee, A. Khan, A. Bhaumik and S. ManirulIslam, Mol. Catal., 2020, 494, 111119 CAS
; (b) K. Dong, J. Zhang, W. Luo, L. Su and Z. Huang, Chem. Eng. Sci., 2018, 334, 1055–1064 CrossRef CAS
.
- For selected examples of isotope effects reveal the catalytic mechanism, see:
(a) D. T. Nowlan III, T. M. Gregg, H. M. L. Davies and D. A. Singleton, J. Am. Chem. Soc., 2003, 125, 15902–15911 CrossRef PubMed
; (b) L. K. Vo and D. A. Singleton, Org. Lett., 2004, 6, 2469–2472 CrossRef CAS PubMed
; (c) D. A. Singleton and Z. Wang, J. Am. Chem. Soc., 2005, 127, 6679–6685 CrossRef CAS PubMed
; (d) J. A. Izzo, P. H. Poulsen, J. A. Intrator, K. A. Jorgensen and M. J. Vetticatt, J. Am. Chem. Soc., 2018, 140, 8396–8400 CrossRef CAS PubMed
; (e) M. V. Joannou, J. M. Hoyt and P. J. Chirik, J. Am. Chem. Soc., 2020, 142, 5314–5330 CrossRef CAS PubMed
; (f) Y. Lin, W. J. Hirschi, A. Kunadia, A. Paul, I. Ghiviriga, K. A. Abboud, R. W. Karugu, M. J. Vetticatt, J. S. Hirschi and D. Seidel, J. Am. Chem. Soc., 2020, 142, 5627–5635 CrossRef CAS PubMed
; (g) J. N. Sanders, H. Jun, R. A. Yu, J. L. Gleason and K. N. Houk, J. Am. Chem. Soc., 2020, 142, 16877–16886 CrossRef CAS PubMed
; (h) K.-Y. Kuan and D. A. Singleton, J. Org. Chem., 2021, 86, 6305–6313 CrossRef CAS PubMed
; (i) V. Wambua, J. S. Hirschi and M. J. Vetticatt, ACS Catal., 2021, 11, 60–67 CrossRef CAS PubMed
; (j) C. Joshi, J. M. Macharia, J. A. Izzo, V. Wambua, S. Kim, J. S. Hirschi and M. J. Vetticatt, ACS Catal., 2022, 12, 2959–2966 CrossRef CAS PubMed
; (k) S. C. Mallojjala, V. O. Nyagilo, S. A. Corio, A. Adili, A. Dagar, K. A. Loyer, D. Seidel and J. S. Hirschi, J. Am. Chem. Soc., 2022, 144, 17692–17699 CrossRef CAS PubMed
; (l) S. Somprasong, M. Castiñeira Reis and S. R. Harutyunyan, Angew. Chem., Int. Ed., 2023, 62, e202217328 CrossRef CAS PubMed
; (m) S. Somprasong, M. Castiñeira Reis and S. R. Harutyunyan, ACS Catal., 2024, 14, 13030–13039 CrossRef CAS PubMed
; (n) S. Somprasong, B. Wan and S. R. Harutyunyan, Chem. Sci., 2025, 16, 802–808 RSC
.
-
(a) D. E. Frantz, D. A. Singleton and J. P. Snyder, J. Am. Chem. Soc., 1997, 119, 3383–3384 CrossRef CAS
; (b) D. E. Frantz and D. A. Singleton, J. Am. Chem. Soc., 2000, 122, 3288–3295 CrossRef CAS
; (c) M. J. Vetticatt and D. A. Singleton, Org. Lett., 2012, 14, 2370–2373 CrossRef CAS PubMed
; (d) S. Xiang and M. P. Mayer, J. Am. Chem. Soc., 2014, 136, 5832–5835 CrossRef CAS PubMed
; (e) D. E. Frantz and D. A. Singleton, J. Am. Chem. Soc., 2000, 122, 3288–3295 CrossRef CAS
.
-
(a) P. Saejong, S. Somprasong, C. Rujirasereesakul and T. Luanphaisarnnont, Synlett, 2022, 33, 1399–1404 CrossRef CAS
; (b) C. Kiattisewee, A. Kaidad, C. Jiarpinitnun and T. Luanphaisarnnont, Monatsh. Chem., 2018, 149, 1059 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2026 |