Open Access Article
Open Access ArticleSynthesis of condensed chiral pyranopyran derivatives with antiproliferative activity using domino Knoevenagel-IMHDA sequences
Sándor Balázs Király *a,
Balázs Buglyóab,
Otieno Michael Ookoab,
Attila Bényei
*a,
Balázs Buglyóab,
Otieno Michael Ookoab,
Attila Bényei c,
Erika Lisztesd,
Balázs István Tóthd,
Tamás Bíróe,
Kristóf Borozsnyaia,
Dávid Oláha,
Gábor Vasvárif,
Dániel Nemes
c,
Erika Lisztesd,
Balázs István Tóthd,
Tamás Bíróe,
Kristóf Borozsnyaia,
Dávid Oláha,
Gábor Vasvárif,
Dániel Nemes f,
Miklós Vecsernyésg,
Ferenc Fenyvesih and
Tibor Kurtán
f,
Miklós Vecsernyésg,
Ferenc Fenyvesih and
Tibor Kurtán *a
*a
aDepartment of Organic Chemistry, University of Debrecen, P. O. Box 400, 4002 Debrecen, Hungary. E-mail: kiraly.sandor.balazs@science.unideb.hu; kurtan.tibor@science.unideb.hu
bDoctoral School of Chemistry, University of Debrecen, Egyetem Square 1, 4032 Debrecen, Hungary
cDepartment of Physical Chemistry, University of Debrecen, Egyetem Square 1, 4032 Debrecen, Hungary
dDepartment of Physiology, University of Debrecen, Nagyerdei krt. 98., 4032 Debrecen, Hungary
eDepartment of Immunology, University of Debrecen, Egyetem Square 1, 4032 Debrecen, Hungary
fDepartment of Pharmaceutical Technology, University of Debrecen, Rex Ferenc Street 1, 4002 Debrecen, Hungary
gDepartment of Pharmaceutical Surveillance and Economics, University of Debrecen, Rex Ferenc Street 1, 4002 Debrecen, Hungary
hDepartment of Molecular and Nanopharmaceutics, University of Debrecen, Rex Ferenc Street 1, 4002 Debrecen, Hungary
First published on 6th May 2026
Abstract
We explored a domino Knoevenagel-intramolecular hetero-Diels-Alder (IMHDA) sequence, in which chiral pentaheterocyclic frameworks containing a pyrano[4,3-b]pyran unit condensed with a pyrone/pyridone and a tetrahydroquinoline/chroman subunit were synthesized. Substituted 2H-chromenes or 3,6-dihydro-2H-pyrans bearing an o-(formylaryl)-ether or -amine moiety were used as substrates, along with 4-hydroxypyrone, -pyridone, -coumarin and 2-quinolone derivatives as active methylene reagents, to assemble chiral condensed ring systems in a domino sequence. In the IMHDA cyclization step, two competing heterodiene subunits consisting of an α,β-unsaturated ketone or an ester/lactam carbonyl afforded regioisomeric condensed coumarin or chromone rings, respectively. Both heterodienes can undergo cyclization with an endo or exo transition state, resulting in two pairs of diastereomers. Up to four possible isomers were isolated in the cyclisation reactions and their structures elucidated using 2D NMR, chiroptical methods, and two single-crystal X-ray diffraction structures. The regio- and diastereoselectivity of the cyclisation was investigated in terms of temperature, solvent and the structures of the substrate and reagent. Simplified substrates were prepared from 5,6-dihydro-2H-pyran derivatives, the reactions of which took place with complete regio- and stereoselectivity, providing a single product with good yields. Some of the products exhibited antiproliferative activity against human cancer cell lines with IC50 values down to 5.7 µM.
Introduction
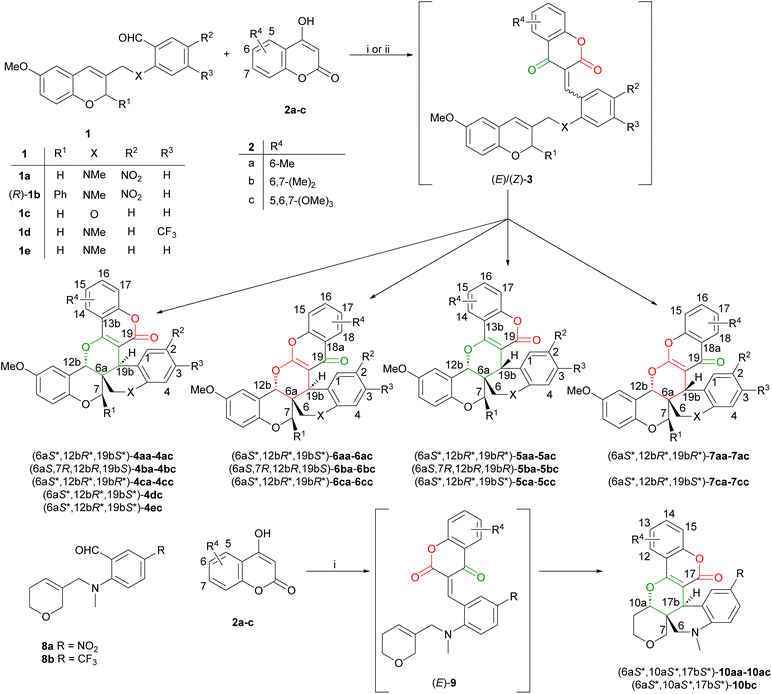
The pyranopyran motif is a common core structure in various natural secondary metabolites of pharmacological interest, and all of the six possible regioisomeric bicycles (Fig. 1a) are found in bioactive derivatives.1–4 The two most frequent ones are the pyrano[4,3-b]pyran, found in marine and fungal metabolites such as pyripyropene,5 territrems6 or cyclomorusin,7 (Fig. 1b) and the pyrano[3,2-b]pyran, which is a recurring motif in marine toxins like okadaic acid, ciguatoxins and brevetoxins.8 Pyrano[3,4-c]pyran and pyrano[2,3-b]pyran are also common building blocks, as the ring systems in the aglycone unit of many secoiridoid glycosides9,10 (Fig. 1c) and saponins,11–13 respectively. The pyrano[3,4-b]pyran and pyrano[4,3-c]pyran ring systems are much rarer, found mainly in rotenoids14 and ellagic acid. Tripyrano ring systems of a more complex nature are much less abundant, and synthetic availability is limited. It is evident that only a limited number of natural products15,16 or synthetic bioactive derivatives with antiviral,17 antibacterial,18 or antitumor19,20 activities, containing a tripyrano skeleton, are currently known.Hetero-Diels-Alder (HDA) reactions are a valuable tool in the synthesis of pyran heterocycles. They have been used in the total synthesis of various pyran natural products.21–27 The reaction can be conveniently combined with a condensation reaction in a tandem process, resulting in the domino Knoevenagel-intramolecular hetero-Diels-Alder (DKIMHDA) reaction.28–37 This is a versatile and robust tool in the preparation of fused pyran derivatives, with which complex scaffolds can be assembled in a single step, with excellent diastereoselectivities. If the dienophile or the diene already incorporates a heterocycle, an extended heterocyclic ring system is assembled during the cyclization reaction as also demonstrated by our recent work outlined in Fig. 1d. For the preparation of pyranopyran ring systems, 4-hydroxypyrone and -coumarin reagents were used, which are non-symmetric β-dicarbonyl reagents. In the Knoevenagel intermediate, there are two different heterodiene subunits, the α,β-unsaturated ketone or acyl moiety, capable of undergoing HDA cyclization (Fig. 1d). In many cases, the cyclization step occurs with excellent regioselectivity due to the different reactivity of the two heterodienes, with the α,β-unsaturated ketone moiety reacting exclusively.38–40 The α,β-unsaturated ester unit can also take part in the HDA reaction, especially if the cyclization is intramolecular,41–43 and the selectivity can be increased by the use of microwave irradiation44–46 or ionic liquids.47 While the regioselectivity varies based on the structure of the substrate, mainly on the substitution pattern of the dienophile, all reported reactions share the same cis diastereoselectivity. Other analogue reagents, such as 4-hydroxy-2H-piran-2-one48,49 and 4-hydroxyquinolin-2(1H)-one41,50 derivatives were rarely utilized in these reactions.
Our goal was to expand the scope of this reaction by using heterocyclic dienophiles (2H-chromene and 3,6-dihydro-2H-pyran) in the DKIMHDA reaction. In the reactions of 2H-chromenes 1 and active methylene reagents 2, the initial Knoevenagel intermediate 3 can undergo an IMHDA reaction through four different transition states (TSs), as demonstrated by Tietze and co-workers and our previous DFT calculations.51 In the case of these non-symmetric reagents, the four TSs lead to four isomers, yielding two diastereomeric pairs of the two regioisomers (Scheme 1a). The regioselectivity and diastereoselectivity of the IMHDA reaction were both found to be influenced by the substrate (1) and the reagent (2). In most cases, the regio- and diastereomers were separated by column chromatography. The regioisomers were identified by 13C-NMR and IR spectroscopy, the diastereomers by NOESY measurements, and in two cases, the planar structure and relative configuration were also confirmed by single-crystal X-ray diffraction analysis. The domino reactions of the simplified dihydropyran derivatives 8 demonstrated excellent regio- and diastereoselectivity, with each reaction yielding a single product, (6aS*, 10aS*, 17bS*)-10, and no other isomers being detected (Scheme 1b). The antiproliferative effect of the products was tested on three human cancer cell lines, and several derivatives showed promising activity with low micromolar IC50 values.
 | ||
| Scheme 1 Domino Knoevenagel-intramolecular hetero Diels–Alder reactions of (a) 2H-chromene derivatives 1 and (b) 3,6-dihydro-2H-pyrans 8 with cyclic non-symmetric β-dicarbonyl reagents 2. | ||
Results and discussion
Domino Knoevenagel-IMHDA reactions with 4-hydroxycoumarin reagents (2a–c)
Domino Knoevenagel-IMHDA reactions with hydroxycoumarins are usually carried out in the presence of a base and a wide variety of solvents can be utilized, including protic,49,50 polar aprotic,40,52 apolar41 and even ionic liquids47 or solvent-free conditions.38,39,44 Thus, we started our investigations with a solvent screening for the reaction of 1a and 2a using piperidine as a base (Table 1). The numbering employed for the cyclized products is as follows: the numbers 4–7 represent the regio- and stereochemistry as outlined in Table 1. The first letter refers to the structure of the substrate, and the second letter to that of the active methylene reagent.The yield, as well as the formation and ratio of the regio- (4aa and 5aa versus 6aa and 7aa) and diastereomers (4aa versus 5aa and 6aa versus 7aa), were found to be significantly dependent on the solvent utilized in the reaction. It was observed that, of the apolar solvents, toluene was the only suitable option for the reaction, as no reaction occurred in hexane (entry 1), due to the insolubility of substrate 1a. Toluene facilitated the formation of all four possible isomers, exhibiting low selectivity and favoring the formation of 7aa as the main product, which could not even be observed in most solvents. Furthermore, toluene was found to provide the best overall yield of 78% (entry 2). Among the protic solvents, ethanol exhibited surprisingly analogous results to toluene, producing all four isomers in comparable ratios (entry 8). Acetic acid was found to be incompatible with the reaction, resulting solely in decomposition of the substrate (entry 10). When using polar aprotic solvents, a clear correlation between the reaction's selectivity and the polarity of the solvent is absent. The formation of 7aa was not detected in these solvents, and 4aa was the major product, but the ratios did not follow an obvious pattern (entry 3–7, 9, 11–12). The ratio of isomers 4aa/6aa/5aa increased in the order ethanol < toluene < ethyl acetate < acetonitrile < 1,2-dichloroethane ≈ chloroform ≈ 1,4-dioxane < tetrahydrofuran < dimethylsulfoxide ≈ N,N-dimethylformamide from 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 to 4
3 to 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0. Since toluene showed the best overall yield, it was selected the solvent for the domino sequence. Although it showed the lowest selectivity, the preparation and isolation of multiple isomers allow a more comprehensive stereochemical study, and could assist in establishing a structure–activity relationship.
0. Since toluene showed the best overall yield, it was selected the solvent for the domino sequence. Although it showed the lowest selectivity, the preparation and isolation of multiple isomers allow a more comprehensive stereochemical study, and could assist in establishing a structure–activity relationship.
The formation of up to four isomeric products is most likely due to a lack of (E)/(Z) selectivity in the Knoevenagel condensation, since from the four possible TSs, only three are viable, according to Tietze et al.51 This is in accordance with our previous findings; in cyclization reactions of 2H-chromene derivatives 1a–c, exo-Z-syn and exo-E-anti TSs lead to epimeric products, according to our DFT calculations.34 Using a non-symmetric reagent for the domino reaction, the Knoevenagel condensation likely leads to a diastereomeric mixture of (E)/(Z)-3. Exo-Z-syn TSs from (Z)-3aa and (E)-3aa lead to products 6aa and 4aa, respectively, with trans orientation of methine protons 12b-H and 19b-H corresponding to (6aS*, 12bR*, 19bS*) relative configuration (RC) (Fig. 2). In the case of (Z)-3aa, the lactone carbonyl contributed to the heterodiene affording 6aa, while (E)-3aa leads to 4aa with the α,β-unsaturated ketone moiety acting as the heterodiene. Similarly, exo-E-anti TSs from (Z)- and (E)-3aa lead to products 5aa and 7aa, respectively, with cis orientation of the protons 12b-H and 19b-H implying (6aS*, 12bR*, 19bR*) RC. Through the exo-E-anti TS, (Z)-3aa produces 5aa with the ketone carbonyl contributing to the heterodiene, while (E)-3aa leads to 7aa with the participation of the lactone carbonyl (Fig. 2). Thus the selectivity of the domino reaction depends on two separate factors: (E)/(Z)-selectivity of the Knoevenagel condensation and the relative energies of the TSs leading to the given isomer in the subsequent cyclization.
The reactions of 1a–e with substituted 4-hydroxycoumarin reagents (2a–c) were carried out in refluxing toluene, using piperidine as the base. In the reactions of substrate 1c, possessing an ether linker, aza-Michael addition of piperidine to intermediate 3 was observed, impeding the cyclization. In these instances, triethylamine was utilized as a base (entries 5, 10, 16 and 17). For the simplified substrates 8a and 8b, containing a 3,6-dihydro-2H-pyran moiety, all reactions took place with complete regio- and diastereoselectivity affording solely the product (6aS*, 10aS*, 17bS*)-10 with up to 77% yield, irrespective of the substitution pattern of the reagent (Table 2, entries 6, 11, 20 and 21).
| Entry | Solvent | Ratio of isomersb (%) | Yielda (%) | |||
|---|---|---|---|---|---|---|
| 4aa | 5aa | 6aa | 7aa | |||
| a The products were isolated as a mixture of isomers by crystallization, yield given is isolated yield.b Ratio of isomers was determined by HPLC separation from the crystallized mixture.c No reaction.d Full conversion, but no product observed. | ||||||
| 1 | Hexane | — | — | — | — | 0%c |
| 2 | Toluene | 18 | 24 | 24 | 34 | 78% |
| 3 | 1,2-Dichloroethane | 52 | 22 | 26 | — | 51% |
| 4 | Tetrahydrofuran | 60 | 24 | 16 | — | 55% |
| 5 | Chloroform | 50 | 27 | 23 | — | 50% |
| 6 | Ethyl acetate | 39 | 30 | 31 | — | 50% |
| 7 | Dioxane | 52 | 30 | 18 | — | 58% |
| 8 | Ethanol | 16 | 19 | 32 | 33 | 46% |
| 9 | Acetonitrile | 42 | 27 | 31 | — | 67% |
| 10 | Acetic acid | — | — | — | — | 0%d |
| 11 | N,N-dimethylformamide | 81 | — | 19 | — | 35% |
| 12 | Dimethylsulfoxide | 77 | 3 | 20 | — | 33% |
In order to ascertain the effect of the temperature on the reaction, the reaction of 1a and 2a was carried out at three different conditions; at reflux temperature (entry 1), at room temperature (entry 2), and at 150 °C with microwave activation (entry 3). An increased diastereoselectivity was observed at room temperature, favouring derivatives 4aa and 7aa (entry 2). This is most likely caused by the different selectivity of the initial Knoevenagel condensation, as the formation of these two isomers is possible from the same (E)-3aa intermediate through exo-Z-syn and exo-E-anti transition states for 4aa and 7aa, respectively (Fig. 2). Microwave irradiation (150 °C) was found to provide 4aa exclusively, but in a low yield (22%), no other isomers were detected in the reaction. The low yield is attributed to decomposition; as multiple side-products were observed in the reaction mixture (entry 3). Substrate-controlled stereoselective domino reaction was tested with the optically active substrate (R)-1b (entries 4), which promoted the formation of isomer 5ba as the major product (31%) but diastereomeric 4ba was also isolated with 28% yield. The inherent chirality of the substrate affected mostly the regioselectivity, favouring the enone subunit as the heterodiene, while the diastereselectivity remained quite low as also demonstrated by entries 14 and 15. The dimethyl derivative 2b showed superior selectivity in the domino reactions. The formation of isomer 7 was not detected with either substrate (entry 7–11) and full diastereoselectivity was observed with 8a and 1a (entries 7, 8 and 11). The effect of temperature was also assessed in the reaction of 1a and 2b (entries 7 and 8). The reaction afforded regioisomers 4ab and 6ab with full diastereoselectivity at both temperatures, and the regioselectivity was again higher at room temperature, favouring 4ab with 54% yield. The trimethoxy-substituted reagent 2c afforded three isomers with high regioselectivity and moderate to low diastereoselectivity in the domino reaction with all three substates (entry 12–17). Carrying out the reaction at room temperature induced no significant change in either the regio- or diastereoselectivity (entries 13, 15 and 17). Since these products showed the best antiproliferative activities, two additional derivatives were prepared to study the effect of the substitution of the benzaldehyde moiety (R2 and R3). In these reactions, only the major products were isolated, but the formation of the other two minor isomers was detected (entry 18–19).
The planar structures of the regioisomers were determined on the basis of the 13C-chemical shift of the C-19 carbonyl carbon: the lactone moiety of the coumarin derivatives 4 and 5 exhibited a characteristic peak in the 165–166 ppm range, while the ketone of the chromone derivatives 6 and 7 in the range of 180–181 ppm. The planar structure is supported by the νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibrations in the IR spectra of the products: the isomers 4/5 and 6/7 showed the characteristic IR absorption bands for α,β-unsaturated esters (1697–1711 cm−1) and ketones (1667–1669 cm−1), respectively. The relative configuration of each diastereomeric product was assigned using characteristic NOE correlations. 12b-H and C-6 are cis in all isomers, owing to the diastereospecificity of the Diels–Alder reaction (Z configuration of the dienophile double bond in substrates 1a–e). The trans configuration of the methine protons 12b-H and 19b-H in products 4 and 5 was determined using key correlations 12b-Heq/6-Heq, 19b-Hax/6-Hax and 19b-Hax/7-Heq (Fig. S3 and S9). In 6 and 7, 12b-H and 19b-H are cis, which was determined from the characteristic 12b-Hax/6-Hax, 19b-Hax/12b-Hax and 19b-Hax/6-Hax correlations (Figure S6).
O stretching vibrations in the IR spectra of the products: the isomers 4/5 and 6/7 showed the characteristic IR absorption bands for α,β-unsaturated esters (1697–1711 cm−1) and ketones (1667–1669 cm−1), respectively. The relative configuration of each diastereomeric product was assigned using characteristic NOE correlations. 12b-H and C-6 are cis in all isomers, owing to the diastereospecificity of the Diels–Alder reaction (Z configuration of the dienophile double bond in substrates 1a–e). The trans configuration of the methine protons 12b-H and 19b-H in products 4 and 5 was determined using key correlations 12b-Heq/6-Heq, 19b-Hax/6-Hax and 19b-Hax/7-Heq (Fig. S3 and S9). In 6 and 7, 12b-H and 19b-H are cis, which was determined from the characteristic 12b-Hax/6-Hax, 19b-Hax/12b-Hax and 19b-Hax/6-Hax correlations (Figure S6).
In the case of optically active products 4ba–4bc, 6ba–6bc and 5ba–5bc, since the absolute configuration (AC) of the C-2 chirality center in (R)-1b (C-7 in the products) is known,53 the ACs (6aS, 7R, 12bR, 19bS)-4ba–bc, (6aS, 7R, 12bR, 19bS)-6ba–bc and (6aS, 7R, 12bR, 19bR)-5ba–bc were assigned. The enantiomers of racemates 4aa–4ac, 5aa–5ac and 6aa–6ac were separated by chiral HPLC, and the on-line HPLC-ECD spectra of the enantiomers were recorded (Fig. S181–S205). By comparing these spectra to the ECD spectra of the corresponding C-7 phenyl derivatives with known absolute configuration, the ACs of the separated enantiomers were assigned (Fig. S182, S184 and S186). Surprisingly, the HPLC-ECD spectra of 4aa and 6aa did not show sufficient similarity with the ECD spectra of 4ba and 6ba, and hence the absolute configurations were not assigned in these cases.
The structures of racemic 4cc and 6cc were determined independently by single crystal X-ray diffraction analysis (CCDC no. 2408634 and 2408635, respectively, Fig. 3), which supported both the connectivity and the stereochemistry of the assigned structures (Tables S1–S6).
| Entry | 1 | 2 | Product (yield) | Regioselectivity (4 + 5)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (6 + 7) (6 + 7) |
Diastereoselectivity (4 + 6)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (5 + 7) (5 + 7) |
|---|---|---|---|---|---|
| a Isolated yield.b Isolated as a mixture, yield calculated from NMR integrals.c Reaction carried out at room temperature.d Reaction carried out with microwave irradiation.e No reaction at r.t., reaction carried out at 55 °C.f Only one isomer isolated, ratios not determined. | |||||
| 1 | 1a | 2a | 4aa (14%)a | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 58 58 |
42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 58 58 |
| 6aa (19%)b | |||||
| 5aa (19%)b | |||||
| 7aa (27%)a | |||||
| 2c | 1a | 2a | 4aa (27%)a | 52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 48 48 |
61![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 39 39 |
| 6aa (12%)b | |||||
| 5aa (6%)b | |||||
| 7aa (19%)a | |||||
| 3d | 1a | 2a | 4aa (22%)a | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 4 | (R)-1b | 2a | 4ba (28%)b | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45 45 |
| 6ba (14%)b | |||||
| 5ba (34%)a | |||||
| 5 | 1c | 2a | 4ca (34%)a | 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33 33 |
70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 30 |
| 6ca (19%)a | |||||
| 5ca (17%)a | |||||
| 7ca (6%)a | |||||
| 6 | 8a | 2a | 10aa (44%) | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 7 | 1a | 2b | 4ab (40%)a | 47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 53 53 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 6ab (45%)a | |||||
| 8c | 1a | 2b | 4ab (54%)a | 74![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26 26 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 6ab (19%)a | |||||
| 9 | (R)-1b | 2b | 4bb (31%)a | 74![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26 26 |
77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
| 6bb (16%)a | |||||
| 5bb (14%)a | |||||
| 10 | 1c | 2b | 4cb (35%)a | 75![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 25 |
72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 28 |
| 6cb (19%)a | |||||
| 5cb (21%)a | |||||
| 11 | 8a | 2b | 10ab (57%) | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 12 | 1a | 2c | 4ac (49%)a | 82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18 18 |
72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 28 |
| 6ac (16%)a | |||||
| 5ac (25%)a | |||||
| 13c | 1a | 2c | 4ac (21%)a | 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22 22 |
69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21 21 |
| 6ac (10%)a | |||||
| 5ac (14%)a | |||||
| 14 | (R)-1b | 2c | 4bc (42%)a | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
| 6bc (10%)a | |||||
| 5bc (44%)a | |||||
| 15c | (R)-1b | 2c | 4bc (32%)a | 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9 |
43![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 57 57 |
| 6bc (8%)a | |||||
| 5bc (53%)a | |||||
| 16 | 1c | 2c | 4cc (34%)a | 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
| 6cc (12%)a | |||||
| 5cc (14%)a | |||||
| 17e | 1c | 2c | 4cc (29%)a | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
74![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26 26 |
| 6cc (8%)a | |||||
| 5cc (13%)a | |||||
| 18 | 1d | 2c | 4dc (42%)a | n.d.f | n.d.f |
| 19 | 1e | 2c | 4ec (32%)a | n.d.f | n.d.f |
| 20 | 8a | 2c | 10ac (77%) | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 21 | 8b | 2c | 10bc (52%) | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
 | ||
| Fig. 3 ORTEP view at 50% probability level of (a) rac-(6aS*, 12bR*, 19bR*)-4cc (CCDC no. 2408634) and (b) rac-(6aS*, 12bR*, 19bR*)-6cc (CCDC no. 2408635). | ||
Domino Knoevenagel-IMHDA reactions with 4-hydroxy-2-quinolone (2d)
In order to test the effect of the substitution in the carbonyl moiety on the regioselectivity, the reaction was also carried out with the lactam derivative 4-hydroxy-2-quinolone (2d) as a reagent (Scheme 2). Due to its poor solubility in toluene, the reactions had to be conducted in ethanol. The selectivity was governed primarily by the substitution pattern of the 4H-chromene substrate, and this reagent produced the largest differences among substrates. The 2H-chromene substrate 1a afforded three isomers, with 4ad being the major product (48%), and the lactam carbonyl also reacted as part of the heterodiene to form 6ad with 12% yield. The 2-phenyl derivative rac-1b gave a mixture of two diastereomers with full regioselectivity, favouring the formation of 4bd (60%). In the reaction of the arylether substrate 1c, only one isomer was isolated with 24% yield (Scheme 2). The regioselectivity was higher than those with the 4-hydroxycoumarins 2a-c, as the lactam carbonyl moiety of 2d is even less reactive than the ester group of 2a-c. The domino sequence with the simplified analogue 8a was completely regio- and diastereoselective, affording 10ad as the only product with 42% yield.Domino Knoevenagel-IMHDA reaction with 4-hydroxypyrone (2e) and -pyridone reagents (2f)
Since the presence or absence of the condensed benzene ring in the substrates (1a–e or 8a, 8b) had a decisive effect on both the regio- and diastereoselectivity of the domino sequence, reagents lacking the condensed benzene ring, 4-hydroxy-6-methyl-2H-pyran-2-one (2e) and its lactam derivative 4-hydroxy-1,6-dimethylpyridin-2(1H)-one (2f), were also tested. In the domino reaction, substrates containing an amine linker (1a and 1b) gave three or four isomers, while the substrate 1c containing an arylether linker afforded the two regioisomers with complete diastereoselectivity. To test the effect of the reaction temperature on the selectivity of the IMHDA reaction, the domino reaction sequences were carried out both at reflux and at room temperature as well (entries 1–6). In the reaction of (R)-1b, the temperature had a significant effect on the diastereoselectivity: the main product was 4be at reflux, and 5be at room temperature. The regioselectivity however, was only slightly affected by the temperature, the ratio of 6be increases at lower temperature (entry 3–4). In the reaction of 1a, isomer 5ae was the major product at both temperatures. Lowering the temperature had the same effects as for 1b: formation of 5ae is preferred at lower temperature, but the 4ae/6ae ratio changes only slightly (entry 3–4). Substrate 1c did not react at room temperature, so the reaction was carried out at 55 °C (entry 6). The temperature had no effect on the diastereoselectivity, only 4ce and 6ce were formed at both temperatures, the regioselectivity however, was governed by the temperature: the major product was 6ce at reflux, and 4ce at lower temperature (entry 5–6). In the reactions of amide derivative 2f, the two competing heterodiene moieties are an α,β-unsaturated lactam and a -ketone subunit. Surprisingly, there was just a minor change in the regioselectivity compared to reactions of the pyran derivative 2e. This suggests that the decisive factor in the selectivity is the favorable or unfavorable intramolecular interactions in the transition states (steric effects), and not the HOMO–LUMO orbital energies (electronic effects). In the reaction of 1c with 2f only one isomer was isolated, which was due to the observed decomposition of the other isomers, and not the selectivity of the cyclization (entry 11) (Table 3).| Entry | 1 | 2 | Product (yield)a | Regioselectivity (4 + 5)/(6 + 7) | Diastereoselectivity (4 + 6)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) (5 + 7) (5 + 7) |
|---|---|---|---|---|---|
| a Isolated yield.b Reaction carried out at room temperature.c No reaction at r.t., reaction carried out at 55 °C. | |||||
| 1 | 1a | 2e | 4ae (16%) | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 58 58 |
72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 28 |
| 6ae (35%) | |||||
| 5ae (14%) | |||||
| 7ae (6%) | |||||
| 2b | 1a | 2e | 4ae (14%) | 53![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 47 47 |
65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35 35 |
| 6ae (37%) | |||||
| 5ae (27%) | |||||
| 3 | (R)-1b | 2e | 4be (32%) | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
74![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26 26 |
| 6be (8%) | |||||
| 5be (14%) | |||||
| 4b | (R)-1b | 2e | 4be (13%) | 76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24 24 |
47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 53 53 |
| 6be (14%) | |||||
| 5be (31%) | |||||
| 5 | 1c | 2e | 4ce (10%) | 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 72 72 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 6ce (26%) | |||||
| 6c | 1c | 2e | 4ce (16%) | 72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 28 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 6ce (6%) | |||||
| 7 | 8a | 2e | 10ae (42%) | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 8 | 8b | 2e | 10be (32%) | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 0 |
| 9 | 1a | 2f | 4af (31%) | 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 46 46 |
76![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 24 24 |
| 6af (31%) | |||||
| 5af (13%) | |||||
| 7af (7%) | |||||
| 10 | rac-1b | 2f | 4bf (15%) | 49![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 51 51 |
77![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 23 23 |
| 6bf (29%) | |||||
| 5bf (13%) | |||||
| 11 | 1c | 2f | 4cf (11%) | n.d. | n.d. |
Antiproliferative activity
The antiproliferative activity of the condensed heterocyclic products were tested on three human cancer cell lines, and results are shown in Table 4. Derivatives with no significant inhibition at 50 µM concentration were considered inactive, IC50 values were determined where significant (>50%) antiproliferative effect was observed. All the active derivatives have (S*, R*, S*) relative configuration and all except for one belong to the series of products 4, in which the α,β-unsaturated ketone moiety acts as a heterodiene in the IMHDA cyclization step. The trimethoxy derivative 4ac showed the best activity with an IC50 value of 5.7 µM on the CaCo-2 cell line (entry 1), while it was inactive (>50 µM) against A2780 and WM35 cells. Doxorubicin has a reported IC50 value of 12.5 µM on the Caco-2 cell line.54,55 The structural analogues 4ec and 4aa, lacking the C-2 nitro substituent of the condensed benzene ring or two methoxy groups of the coumarin subunit, respectively, had lower activity against the CaCo-2 cell line (entry 2 and 3). In 4dc, the replacement of the C-2 nitro group of 4ac with a trifluoromethyl one resulted in the complete elimination of activity (IC50 > 50 µM, entry 4). The additional C-7 phenyl group and the resultant chirality center of rac-4ba and (6aS, 7R, 12bR, 19bS)-4ba decreased the activity compared to that of 4ac (entry 5 and 6). Against the A2780 cell line, moderate activities with IC50 values of 25.1 and 17.7 µM were identified for 4ce and 4cd, containing an oxygen linker instead of the NMe in position 5 and condensed 2-quinolone or 2-pyrone subunits, respectively. From the other diastereomeric (5) and regioisomeric products (6 and 7), only the regioisomeric 6ae exhibited activity against the CaCo-2 cell line with an IC50 value of 11.5 µM.| Entry | Compound | Cell lines/IC50 (µM) | ||
|---|---|---|---|---|
| CaCo-2 | A2780 | WM35 | ||
| a nd: not determined. | ||||
| 1 | 4ac | 5.7 | >50 | >50 |
| 2 | 4ec | 24.1 | >50 | >50 |
| 3 | 4aa | 13.4 | >50 | >50 |
| 4 | 4dc | >50 | >50 | >50 |
| 5 | rac-4ba | 13.3 | >50 | >50 |
| 6 | (6aS, 7R, 12bR, 19bS)-4ba | 26.3 | >50 | >50 |
| 7 | 4ce | nd | 25.1 | >50 |
| 8 | 4cd | nd | 17.7 | >50 |
| 9 | 6ae | 11.5 | >50 | >50 |
Experimental
Materials and methods
Chemicals were purchased puriss p.a. from commercial suppliers, and solvents were purified by distillation before use. For thin-layer chromatography (TLC), silica gel plates Merck 60 F254 were used and compounds were visualized by irradiation with UV light. Column chromatography was performed using silica gel Merck 60 (particle size 0.063–0.200 mm) (Merck, Darmstadt, Germany). Melting points were determined on a Kofler hot-stage apparatus and are uncorrected. The NMR spectra were recorded on Bruker Avance II 400 (1H: 400 MHz. 13C: 100 MHz) and Bruker Avance II 500 MHz (1H: 500 MHz. 13C: 125 MHz) spectrometers using TMS as internal standard (Bruker, Billerica, MA, USA). Chemical shifts were reported as δ in ppm and 3JH,H coupling constants in Hz. IR spectra were recorded on a JASCO (Tokyo, Japan) FT/IR-4100 spectrometer and absorption bands are presented as wavenumber in cm−1. The optical rotations of the compounds were measured by a PerkinElmer (Norwalk, CT) 341 polarimeter. ECD spectra were recorded on a JASCO (Tokyo, Japan) J-810 spectropolarimeter. Electrospay Quadrupole Time-of-Flight HRMS measurements were performed with a MicroTOF-Q type QqTOF MS instrument equipped with an ESI source from Bruker (Bruker Daltoniks, Bremen, Germany).Chiral HPLC separation and ECD measurement
Separation of 4aa–af, 5aa–af and 6aa–af were performed on a JASCO (Tokyo, Japan) HPLC system with Chiralpak-IC column (5 µm, 250 × 4.6 mm, hexane/dichloromethane/methanol eluent, 1 mL min−1 flow rate) and HPLC-ECD spectra were recorded in stopped-flow mode on a JASCO (Tokyo, Japan) J-810 spectropolarimeter equipped with a 10 mm HPLC flow cell. ECD ellipticity (ε) values were not corrected for concentration. For an HPLC-ECD spectrum, three consecutive scans were recorded and averaged with 2 nm bandwidth, 1 s response, and standard sensitivity. The HPLC-ECD spectrum of the eluent recorded in the same way was used as background. The concentration of the injected sample was set so that the HT value did not exceed 500 V in the HT channel down to 230 nm.Microwave irradiation experiments
Reactions were performed using single-mode CEM Discover Systems (CEM Corporation, Charlotte, NC, USA) in dynamic mode. Reaction times refer to the hold time at the desired set temperature and not to the total irradiation time. Stirring speed was set to “High” (ca. 700 rpm). Standard 10 ml volume cylindrical Pyrex® reaction vessels (inner diameter 12 mm, obtained from CEM Corporation, USA) were used, equipped with small cylindrical magnetic stirring bars. The vessels were sealed with PEEK snap caps and standard PTFE-coated silicone septa. The external IR thermometer of the equipment was used to monitor the reaction temperatures. Pressure sensing is achieved by a hydraulic sensor. Reaction cooling is performed by compressed air automatically after the heating period has elapsed.X-ray crystallographic study
X-ray quality crystals could be grown from appropriate solvents by slow evaporation. A chosen crystal was then fixed under a microscope onto a Mitegen loop using high-density oil. Diffraction Intensity data was collected at ambient temperature on a Bruker-D8 Venture diffractometer (Bruker AXS GmbH, Karlsruhe, Germany) equipped with INCOATEC IµS 3.0 (Incoatec GmbH, Geesthacht, Germany) dual (Cu and Mo) sealed tube micro sources and a Photon II Charge-Integrating Pixel Array detector (Bruker AXS GmbH, Karlsruhe, Germany) using Mo Kα (λ = 0.71073 Å) radiation. High-multiplicity data collection and integration were performed using APEX5 (version 2017.3-0, Bruker AXS Inc., 2017, Madison, WI, USA) software. Data reduction and multiscan absorption correction were performed using SAINT (version 8.38A, Bruker AXS Inc., 2017, Madison, WI, USA). The structure was solved using direct methods and refined on F2 using the SHELXL program56 incorporated into the APEX5 suite. Refinement was performed anisotropically for all non-hydrogen atoms. Hydrogen atoms were placed in idealized positions on parent atoms. The CIF file was manually edited using publCIF software,57 while graphics were designed using the Mercury program.58 The results for the X-ray diffraction structure determinations followed the Checkcif functionality of PLATON software (Utrecht University, Utrecht, the Netherlands).59Molecular structure analysis
The single crystal X-ray diffraction study had proved the relative configuration of the stereogenic carbon centers. In structure 4cc there are two molecules in the asymmetric unit with slightly different conformation (Fig. S218). Solving the structure in centrosymmetric space group No. 14 (P21/n) gives disordered structure so the space group of Pn was accepted as correct space group. The lattice symmetry generates the enantiomeric pair of the compound. In case of 6cc the crystals were very thin needles resulting in high Rint value. Nevertheless, in both cases we consider the structures as reliable and correct.MTT assay60,61
A2780 and WM35 cells were purchased from Sigma (St. Louis, MO, USA) and ATCC (Manassas, VA, USA), respectively. Human White colon adenocarcinoma (Caco-2) cells (European Collection of Cell Cultures (ECACC, UK) were isolated from a primary colonic tumor in a 72-year-old White male using the explant culture technique. Caco-2 human epithelial colorectal adenocarcinoma cells were grown in plastic cell culture flasks in Dulbecco's Modified Eagle's Medium, supplemented with 3.7 g L−1 NaHCO3, 10% (v/v) heat-inactivated fetal bovine serum (FBS), 1% (v/v) non-essential amino acids solution, 1% (v/v) L-glutamine, 100 IU ml−1 penicillin, and 100 µg ml−1 streptomycin at 37 °C in an atmosphere of 5% CO2. The cells were routinely maintained by regular passaging. The number of viable cells was indirectly determined by measuring the conversion of the tetrazolium salt MTT [3-(4,5-dimethilthiasol-2-il)-2,5-diphenyltetrasolium bromide, Sigma-Aldrich] to formazan by mitochondrial dehydrogenases. Cells were plated in 96-well multi-titer plates (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cells per well density) in quadruplicates and were cultured for 3 days and treated by the compounds daily. Negative control group was treated with equal amount of vehicle solvent (DMSO) and the positive control group was treated with 1 µg ml−1 doxorubicin (for A2780 and WM35). Cells were then incubated with 5 mg ml−1 MTT for 3 hours, precipitated formazan crystals were desolved in acidic isopropanol (10% 1 M HCl in isopropanol supplemented with 10% Triton X 100) and concentration of formazan was assessed colorimetrically by measuring absorbance at 565 nm. Procedure is based on the one outlined in the cell proliferation kit (Sigma, St. Louis, MO, USA).
000 cells per well density) in quadruplicates and were cultured for 3 days and treated by the compounds daily. Negative control group was treated with equal amount of vehicle solvent (DMSO) and the positive control group was treated with 1 µg ml−1 doxorubicin (for A2780 and WM35). Cells were then incubated with 5 mg ml−1 MTT for 3 hours, precipitated formazan crystals were desolved in acidic isopropanol (10% 1 M HCl in isopropanol supplemented with 10% Triton X 100) and concentration of formazan was assessed colorimetrically by measuring absorbance at 565 nm. Procedure is based on the one outlined in the cell proliferation kit (Sigma, St. Louis, MO, USA).
Determination of IC50
Logistic dose–response curves were fitted using the equation y = A2 + (A1 − A2)/(1 + (x/x0)p) where the parameters are the following: A1: initial value (ymin), A2: final value (ymax), x0: center (EC/IC50) and p is the calculated power. Fittings were carried out and parameters were calculated using Origin 8.6 (OriginLab Corporation, Northampton, MA, USA).General methods for the domino Knoevenagel-hetero Diels–Alder reactions
Domino Knoevenagel-IMHDA reactions of 2H-chromene derivatives 1a–e with 4-hydroxy-coumarin derivatives 2a–c
rac-(6aS*, 12bR*, 19bS*)- and (6aS*, 12bR*, 19bR*)-11-methoxy-5,15,-dimethyl-2-nitro-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,4′:5, 6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [rac-(6 aS*,12bR*,19bS*)-4aa and rac-(6aS*, 12bR*, 19bR*)-5aa] and rac-(6aS*, 12bR*, 19bS*)- and (6aS*, 12bR*, 19bR*)-11-methoxy-5,17-dimethyl-2-nitro-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,2′:5,6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [rac-(6 aS*,12bR*,19bS*)-6aa and rac-(6aS*, 12bR*, 19bR*)-7aa]The reaction of 1a and 2a was carried out according to method A. Cooling the reaction to room temperature, a precipitate formed which was filtered and washed with cold ether, affording the mixture of rac-(6aS*, 12bR*, 19bS*)-4aa, rac-(6aS*, 12bR*, 19bS*)-6aa, rac-(6aS*, 12bR*, 19bR*)-5aa and rac-(6aS*, 12bR*, 19bR*)-7aa as yellow powder. The mixture was separated by column chromatography (hexane/chloroform/acetone 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 19bS*)-4aa as yellow powder (14%), the 1
1), affording rac-(6aS*, 12bR*, 19bS*)-4aa as yellow powder (14%), the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of rac-(6aS*, 12bR*, 19bS*)-6aa and rac-(6aS*, 12bR*, 19bR*)-5aa as yellow powder (37%) and rac-(6aS*, 12bR*, 19bR*)-7aa as yellow powder (27%).
1 mixture of rac-(6aS*, 12bR*, 19bS*)-6aa and rac-(6aS*, 12bR*, 19bR*)-5aa as yellow powder (37%) and rac-(6aS*, 12bR*, 19bR*)-7aa as yellow powder (27%).
Alternatively, the reaction was carried out at room temperature, and after workup, afforded rac-(6aS*, 12bR*, 19bS*)-4aa as yellow powder (27%), the 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of rac-(6aS*, 12bR*, 19bS*)-6aa and rac-(6aS*, 12bR*, 19bR*)-5aa as yellow powder (17%) and rac-(6aS*, 12bR*, 19bR*)-7aa as yellow powder (19%).
1 mixture of rac-(6aS*, 12bR*, 19bS*)-6aa and rac-(6aS*, 12bR*, 19bR*)-5aa as yellow powder (17%) and rac-(6aS*, 12bR*, 19bR*)-7aa as yellow powder (19%).
The reaction was also repeated using microwave irradiation. The sealed vessel was heated to 150 °C for 30 minutes, it was then cooled down, and the solvent was removed in vacuo. After workup, the reaction afforded rac-(6aS*, 12bR*, 19bS*)-4aa as yellow powder (22%) and rac-(6aS*, 12bR*, 19bR*)-7aa as yellow powder (19%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.35 (s, 3H, 3′-H), 2.97 (s, 3H, 2′-H), 3.29 (d, J = 13.2 Hz, 1H, 6-Ha), 3.68 (d, J = 13.2 Hz, 1H, 6-Hb), 3.86 (s, 3H, 1′-H), 3.91 (d, J = 11.4 Hz, 1H, 7-Ha), 3.98 (dd, J = 11.4 and 1.8 Hz, 1H, 7-Hb), 4.24 (s, 1H, 19b-H), 5.05 (d, J = 1.8 Hz, 1H, 12b-H), 6.57 (d, J = 9.2 Hz, 1H, 4-H), 6.90 (d, J = 10.0 Hz, 1H, 9-H), 6.95–7.03 (m, 2H, 10-H and 12-H), 7.27 (d, J = 8.6 Hz, 1H, 17-H), 7.36 (dd, J = 8.6 and 2.2 Hz, 1H, 16-H), 7.43 (bs, 1H, 14-H), 7.99 (dd, J = 9.2 and 2.7 Hz, 1H, 3-H), 8.09 (d, J = 2.7 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.9 (C-3′), 30.7 (C-6a), 33.1 (C-19b), 39.7 (C-2′), 54.6 (C-6), 55.9 (C-1′), 67.5 (C-7), 72.2 (C-12b), 100.6 (C-19a), 110.3 (C-4), 114.4 (C-13b), 115.8 (C-9), 116.7 (C-14), 117.9 (C-10), 118.1 (C-12), 118.2 (C-12a), 121.3 (C-19c), 122.7 (C-17), 124.8 (C-3), 126.1 (C-1), 133.6, (C-16) 133.9 (C-15), 138.5 (C-2), 146.7 (C-8a), 148.9 (C-4a), 151.1 (C-17a), 154.1 (C-11), 157.2 (C-13a), 163.9 (C-19). IR (KBr) ν: 1047, 1217, 1276, 1298, 1318, 1499, 1702, 2853, 2925 cm−1; HRMS: calcd for C29H24N2O7Na [M + Na]+ 535.1476, found 535.1478.
1). 1H NMR (400 MHz, CDCl3) δ 2.35 (s, 3H, 3′-H), 2.97 (s, 3H, 2′-H), 3.29 (d, J = 13.2 Hz, 1H, 6-Ha), 3.68 (d, J = 13.2 Hz, 1H, 6-Hb), 3.86 (s, 3H, 1′-H), 3.91 (d, J = 11.4 Hz, 1H, 7-Ha), 3.98 (dd, J = 11.4 and 1.8 Hz, 1H, 7-Hb), 4.24 (s, 1H, 19b-H), 5.05 (d, J = 1.8 Hz, 1H, 12b-H), 6.57 (d, J = 9.2 Hz, 1H, 4-H), 6.90 (d, J = 10.0 Hz, 1H, 9-H), 6.95–7.03 (m, 2H, 10-H and 12-H), 7.27 (d, J = 8.6 Hz, 1H, 17-H), 7.36 (dd, J = 8.6 and 2.2 Hz, 1H, 16-H), 7.43 (bs, 1H, 14-H), 7.99 (dd, J = 9.2 and 2.7 Hz, 1H, 3-H), 8.09 (d, J = 2.7 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.9 (C-3′), 30.7 (C-6a), 33.1 (C-19b), 39.7 (C-2′), 54.6 (C-6), 55.9 (C-1′), 67.5 (C-7), 72.2 (C-12b), 100.6 (C-19a), 110.3 (C-4), 114.4 (C-13b), 115.8 (C-9), 116.7 (C-14), 117.9 (C-10), 118.1 (C-12), 118.2 (C-12a), 121.3 (C-19c), 122.7 (C-17), 124.8 (C-3), 126.1 (C-1), 133.6, (C-16) 133.9 (C-15), 138.5 (C-2), 146.7 (C-8a), 148.9 (C-4a), 151.1 (C-17a), 154.1 (C-11), 157.2 (C-13a), 163.9 (C-19). IR (KBr) ν: 1047, 1217, 1276, 1298, 1318, 1499, 1702, 2853, 2925 cm−1; HRMS: calcd for C29H24N2O7Na [M + Na]+ 535.1476, found 535.1478.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), and the on line HPLC-ECD spectra were recorded.
5), and the on line HPLC-ECD spectra were recorded.
(6 aR,12bS,19bR)-4aa: tR = 9.08 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 387.5 (2.30), 331.5sh (−4.01), 316 (−10.38), 304.5sh (−4.80), 289 (3.42), 277 (2.91), 254.5 (−5.33), 229 (−51.26).
5]: 387.5 (2.30), 331.5sh (−4.01), 316 (−10.38), 304.5sh (−4.80), 289 (3.42), 277 (2.91), 254.5 (−5.33), 229 (−51.26).
(6 aS,12bR,19bS)-4aa: tR = 11.94 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 383.5 (−1.83), 331.5sh (2.57), 317 (6.54), 304.5sh (3.01), 289.5 (−2.62), 277.5 (−2.74), 255 (3.38), 228.5 (33.91).
5]: 383.5 (−1.83), 331.5sh (2.57), 317 (6.54), 304.5sh (3.01), 289.5 (−2.62), 277.5 (−2.74), 255 (3.38), 228.5 (33.91).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.38 and 2.48 (s, 3H, 3′-H), 2.96 and 3.05 (s, 3H, 2′-H), 3.19–3.32 (m, 3H, 6-H and 6-Ha), 3.35 (dd, J = 11.7 and 2.1 Hz, 1H, 7-Ha), 3.68 (d, J = 13.2 Hz, 1H, 6-Hb), 3.80 (s, 3H, 1′-H), 3.82–3.89 (m, 4H, 1′-H and 7-Hb), 3.96 (s, 2H, 7-H), 4.18 and 4.47 (s, 1H, 19b-H), 4.89 (d, J = 1.1 Hz, 1H, 12b-H), 5.20 (s, 1H, 12b-H), 6.57 (d, J = 9.2 Hz, 1H, 4-H), 6.66 (d, J = 9.1 Hz, 1H, 4-H), 6.83–6.91 (m, 3H, 2 × 9-H and 12-H), 6.94 (d, J = 3.0 Hz, 1H, 12-H), 6.95–7.02 (m, 2H, 2 × 10-H), 7.24 (d, J = 8.5 Hz, 1H, 15-H), 7.29 (d, J = 8.6 Hz, 1H, 17-H), 7.39 (dd, J = 8.5 and 2.3 Hz, 1H, 16-H), 7.44 (dd, J = 8.6 and 2.3 Hz, 1H, 16-H), 7.52 (s, 1H, 14-H), 7.78 (d, J = 2.3 Hz, 1H, 1-H), 7.98 (dd, J = 9.2 and 2.3 Hz, 1H, 3-H), 8.04 (bs, 1H, 1-H), 8.09 (bs, 1H, 18-H), 8.11 (dd, J = 9.1 and 2.4 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 21.0 and 21.1 (C-3′), 31.2 (C-6a), 32.6 (C-19b), 35.5 (C-6a), 37.2 (C-19b), 38.3 and 39.8 (C-2′), 54.5 and 54.6 (C-6), 55.9 and 56.0 (C-1′), 63.2 and 67.7 (C-7), 75.0 and 76.3 (C-12b), 97.3 and 99.0 (C-19a), 109.9 and 110.4 (C-4), 114.7 (C-13b), 115.2 and 115.5 (C-10), 116.7 (C-17), 117.1 (C-15), 117.4 (C-12a), 117.8 and 118.4 (C-12), 118.5 and 119.0 (C-9), 119.6 and 121.8 (C-19c), 122.3 (C-18a), 122.3 (C-1), 122.7 (C-14), 124.8 and 125.1 (C-3), 125.9 (C-18), 126.6 (C-1), 133.8 (C-16), 134.1 (C-15), 134.7 (C-16), 135.7 (C-17), 138.1 and 138.8 (C-2), 146.9 and 148.6 (C-8a), 149.0 (C-4a), 151.2 (C-17a), 151.6 (C-14a), 151.8 (C-4a), 153.9 and 154.4 (C-11), 160.3 (C-19), 160.5 and 161.5 (C-13a), 178.3 (C-19). IR (KBr) ν: 1215, 1289, 1318, 1498, 1606, 1630, 1732, 2926 cm−1; HRMS: calcd for C29H24N2O7Na [M + Na]+ 535.1476, found 535.1478.
1). 1H NMR (400 MHz, CDCl3) δ 2.38 and 2.48 (s, 3H, 3′-H), 2.96 and 3.05 (s, 3H, 2′-H), 3.19–3.32 (m, 3H, 6-H and 6-Ha), 3.35 (dd, J = 11.7 and 2.1 Hz, 1H, 7-Ha), 3.68 (d, J = 13.2 Hz, 1H, 6-Hb), 3.80 (s, 3H, 1′-H), 3.82–3.89 (m, 4H, 1′-H and 7-Hb), 3.96 (s, 2H, 7-H), 4.18 and 4.47 (s, 1H, 19b-H), 4.89 (d, J = 1.1 Hz, 1H, 12b-H), 5.20 (s, 1H, 12b-H), 6.57 (d, J = 9.2 Hz, 1H, 4-H), 6.66 (d, J = 9.1 Hz, 1H, 4-H), 6.83–6.91 (m, 3H, 2 × 9-H and 12-H), 6.94 (d, J = 3.0 Hz, 1H, 12-H), 6.95–7.02 (m, 2H, 2 × 10-H), 7.24 (d, J = 8.5 Hz, 1H, 15-H), 7.29 (d, J = 8.6 Hz, 1H, 17-H), 7.39 (dd, J = 8.5 and 2.3 Hz, 1H, 16-H), 7.44 (dd, J = 8.6 and 2.3 Hz, 1H, 16-H), 7.52 (s, 1H, 14-H), 7.78 (d, J = 2.3 Hz, 1H, 1-H), 7.98 (dd, J = 9.2 and 2.3 Hz, 1H, 3-H), 8.04 (bs, 1H, 1-H), 8.09 (bs, 1H, 18-H), 8.11 (dd, J = 9.1 and 2.4 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 21.0 and 21.1 (C-3′), 31.2 (C-6a), 32.6 (C-19b), 35.5 (C-6a), 37.2 (C-19b), 38.3 and 39.8 (C-2′), 54.5 and 54.6 (C-6), 55.9 and 56.0 (C-1′), 63.2 and 67.7 (C-7), 75.0 and 76.3 (C-12b), 97.3 and 99.0 (C-19a), 109.9 and 110.4 (C-4), 114.7 (C-13b), 115.2 and 115.5 (C-10), 116.7 (C-17), 117.1 (C-15), 117.4 (C-12a), 117.8 and 118.4 (C-12), 118.5 and 119.0 (C-9), 119.6 and 121.8 (C-19c), 122.3 (C-18a), 122.3 (C-1), 122.7 (C-14), 124.8 and 125.1 (C-3), 125.9 (C-18), 126.6 (C-1), 133.8 (C-16), 134.1 (C-15), 134.7 (C-16), 135.7 (C-17), 138.1 and 138.8 (C-2), 146.9 and 148.6 (C-8a), 149.0 (C-4a), 151.2 (C-17a), 151.6 (C-14a), 151.8 (C-4a), 153.9 and 154.4 (C-11), 160.3 (C-19), 160.5 and 161.5 (C-13a), 178.3 (C-19). IR (KBr) ν: 1215, 1289, 1318, 1498, 1606, 1630, 1732, 2926 cm−1; HRMS: calcd for C29H24N2O7Na [M + Na]+ 535.1476, found 535.1478.The enantiomers of rac-(6aS*, 12bR*, 19bR*)-5aa were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and the on-line HPLC-ECD spectra were recorded.
7), and the on-line HPLC-ECD spectra were recorded.
1st eluting enantiomer: tR = 4.57 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 360.5 (−0.81), 313.5 (0.71), 293.5 (−2.03), 269 (−2.88), 248 (−2.85).
7]: 360.5 (−0.81), 313.5 (0.71), 293.5 (−2.03), 269 (−2.88), 248 (−2.85).
2nd eluting enantiomer: tR = 7.41 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 352 (0.74), 313 (−0.34), 293.5 (1.66), 269 (2.84), 247.5 (3.06).
7]: 352 (0.74), 313 (−0.34), 293.5 (1.66), 269 (2.84), 247.5 (3.06).
The enantiomers of rac-(6 aS*,12bR*,19bS*)-6aa were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and the on-line HPLC-ECD spectra were recorded.
7), and the on-line HPLC-ECD spectra were recorded.
1st eluting enantiomer: tR = 6.51 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 387 (−14.58), 314 (−18.50), 294.5sh (−7.83), 283sh (5.83), 275.5 (8.52), 241.5sh (−12.47).
7]: 387 (−14.58), 314 (−18.50), 294.5sh (−7.83), 283sh (5.83), 275.5 (8.52), 241.5sh (−12.47).
2nd eluting enantiomer: tR = 12.30 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 389 (9.21), 313.5 (11.62), 294.5sh (4.76), 284sh (−3.39), 275 (−5.29), 241.5sh (7.54).
7]: 389 (9.21), 313.5 (11.62), 294.5sh (4.76), 284sh (−3.39), 275 (−5.29), 241.5sh (7.54).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.48 (s, 3H, 3′-H), 3.06 (s, 3H, 2′-H), 3.26 (s, 2H, 6-H), 3.36 (dd, J = 11.7 and 2.2 Hz, 1H, 7-Ha), 3.80 (s, 3H, 1′-H), 3.87 (d, J = 11.7 Hz, 1H, 7-Hb), 4.15 (s, 1H, 19b-H), 4.90 (d, J = 2.2 Hz, 1H, 12b-H), 6.69 (d, J = 9.2 Hz, 1H, 4-H), 6.81 (d, J = 8.9 Hz, 1H, 9-H), 6.92 (d, J = 2.9 Hz, 1H, 12-H), 6.95 (dd, J = 8.9, 2.9 Hz, 1H, 10-H), 7.27 (d, J = 8.9 Hz, 1H, 15-H), 7.52 (dd, J = 8.7 and 2.4 Hz, 1H, 16-H), 7.74 (dd, J = 2.7 and 1.2 Hz, 1H, 1-H), 7.96 (bs, 1H, 18-H), 8.15 (dd, J = 9.2 and 2.7 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 20.9 (C-3′), 34.9 (C-6a), 36.8 (C-19b), 38.2 (C-2′), 54.3 (C-6), 55.9 (C-1′), 62.8 (C-7), 77.0 (C-12b), 98.6 (C-19a), 109.9 (C-4), 112.1 (C-18a), 115.3 (C-12), 116.5 (C-12a), 116.8 (C-15), 117.7 (C-9), 118.7 (C-19c), 118.9 (C-10), 121.8 (C-1), 123.8 (C-18), 125.1 (C-3), 135.2 (C-17), 136.3 (C-16), 137.9 (C-2), 148.4 (C-8a), 151.6 (C-4a), 153.9 (C-14a), 154.7 (C-11), 158.9 (C-19), 162.3 (C-13a). IR (KBr) ν: 1216, 1287, 1315, 1498, 1607, 1631, 1736, 2852, 2925 cm−1; HRMS: calcd for C29H24N2O7Na [M + Na]+ 535.1476, found 535.1478.
1). 1H NMR (400 MHz, CDCl3) δ 2.48 (s, 3H, 3′-H), 3.06 (s, 3H, 2′-H), 3.26 (s, 2H, 6-H), 3.36 (dd, J = 11.7 and 2.2 Hz, 1H, 7-Ha), 3.80 (s, 3H, 1′-H), 3.87 (d, J = 11.7 Hz, 1H, 7-Hb), 4.15 (s, 1H, 19b-H), 4.90 (d, J = 2.2 Hz, 1H, 12b-H), 6.69 (d, J = 9.2 Hz, 1H, 4-H), 6.81 (d, J = 8.9 Hz, 1H, 9-H), 6.92 (d, J = 2.9 Hz, 1H, 12-H), 6.95 (dd, J = 8.9, 2.9 Hz, 1H, 10-H), 7.27 (d, J = 8.9 Hz, 1H, 15-H), 7.52 (dd, J = 8.7 and 2.4 Hz, 1H, 16-H), 7.74 (dd, J = 2.7 and 1.2 Hz, 1H, 1-H), 7.96 (bs, 1H, 18-H), 8.15 (dd, J = 9.2 and 2.7 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 20.9 (C-3′), 34.9 (C-6a), 36.8 (C-19b), 38.2 (C-2′), 54.3 (C-6), 55.9 (C-1′), 62.8 (C-7), 77.0 (C-12b), 98.6 (C-19a), 109.9 (C-4), 112.1 (C-18a), 115.3 (C-12), 116.5 (C-12a), 116.8 (C-15), 117.7 (C-9), 118.7 (C-19c), 118.9 (C-10), 121.8 (C-1), 123.8 (C-18), 125.1 (C-3), 135.2 (C-17), 136.3 (C-16), 137.9 (C-2), 148.4 (C-8a), 151.6 (C-4a), 153.9 (C-14a), 154.7 (C-11), 158.9 (C-19), 162.3 (C-13a). IR (KBr) ν: 1216, 1287, 1315, 1498, 1607, 1631, 1736, 2852, 2925 cm−1; HRMS: calcd for C29H24N2O7Na [M + Na]+ 535.1476, found 535.1478.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and the on-line HPLC-ECD spectra were recorded.
7), and the on-line HPLC-ECD spectra were recorded.
(6 aS,12bR,19bR)-7aa: tR = 6.11 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 386 (−21.92), 312.5 (−16.39), 275.5 (−1.22), 258 (−20.20), 241 (−13.70).
7]: 386 (−21.92), 312.5 (−16.39), 275.5 (−1.22), 258 (−20.20), 241 (−13.70).
(6 aR,12bS,19bS)-7aa: tR = 13.64 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 387 (13.61), 312 (10.40), 254.5 (12.97), 242sh (7.80).
7]: 387 (13.61), 312 (10.40), 254.5 (12.97), 242sh (7.80).
(6aS, 7R, 12bR, 19bS)– and (6aS, 7R, 12bR, 19bR)-11-methoxy-5,15-dimethyl-2-nitro-7-phenyl-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,4':5,6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [(6aS, 7R, 12bR, 19bS)-4ba and (6aS, 7R, 12bR, 19bR)-5ba] and (6aS, 7R, 12bR, 19bS)-11-methoxy-5,17-dimethyl-2-nitro-7-phenyl-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,2′:5,6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [(6aS, 7R, 12bR, 19bS)-6ba]
The reaction of (R)-1b and 2a was carried out according to method A. Cooling the reaction to room temperature, a precipitate formed which was filtered and washed with cold ether, affording (6aS, 7R, 12bR, 19bS)-4ba as yellow powder (10%). The filtrate was concentrated in vacuo, and it was purified by column chromatography (hexane/ethyl acetate 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording the mixture of (6aS, 7R, 12bR, 19bS)-4ba and (6aS, 7R, 12bR, 19bS)-6ba as yellow powder (32%) and (6aS, 7R, 12bR, 19bR)-5ba as yellow powder (34%).
1), affording the mixture of (6aS, 7R, 12bR, 19bS)-4ba and (6aS, 7R, 12bR, 19bS)-6ba as yellow powder (32%) and (6aS, 7R, 12bR, 19bR)-5ba as yellow powder (34%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = +35 (c = 0.80 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (231 µg/4 ml): 354sh (7.71), 323 (7.85), 296sh (−0.93), 267 (−7.81), 245.5 (9.22), 210.5 (−31.69), 197.5 (66.34).1H NMR (400 MHz, CD2Cl2) δ 2.50 (s, 3H, 3″-H), 2.93 (s, 3H, 2″-H), 3.30 (d, J = 13.1 Hz, 1H, 6-Ha), 3.55 (d, J = 13.1 Hz, 1H, 6-Hb), 3.84 (s, 3H, 1″-H), 3.97 (s, 1H, 19b-H), 5.07 (s, 1H, 7-H), 5.23 (s, 1H, 12b-H), 6.59 (d, J = 9.3 Hz, 1H, 4-H), 7.03 (m, 2H, 9-H and 10-H), 7.07 (s, 1H, 12-H), 7.25 (d, J = 8.5 Hz, 1H, 17-H), 7.29–7.38 (m, 2H, 2′-H and 6′-H), 7.44 (m, 3H, 3′-H, 4′-H and 5′-H), 7.54 (dd, J = 8.5 and 2.3 Hz, 1H, 16-H), 7.97 (dd, J = 9.3 and 2.7 Hz, 1H, 3-H), 7.99–8.06 (m, 2H, 1-H and 14-H).13C NMR (100 MHz, CDCl3) δ 21.0 (C-3″), 32.7 (C-19b), 33.1 (C-6a), 40.0 (C-2″), 50.4 (C-6), 56.0 (C-1″), 73.3 (C-12b), 77.1 (C-7), 101.2 (C-19a), 110.4 (C-4), 114.4 (C-13b), 115.8 (C-17), 116.9 (C-9), 118.0 (C-10), 118.3 (C-12), 118.4 (C-12a), 122.4 (C-19c), 122.9 (C-3), 124.6 (C-1), 126.2 (C-14), 127.5 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.3 (C-4′), 133.9 (C-16), 134.0 (C-1′), 134.1 (C-17), 138.5 (C-2), 147.9 (C-14a), 148.5 (C-8a), 151.5 (C-4a), 154.4 (C-11), 158.1 (C-13a), 163.6 (C-19). IR (KBr) ν: 1117, 1228, 1262, 1315, 1497, 1556, 1726, 2915 cm−1. HRMS: calcd for C35H28N2O7Na [M + Na]+ 611.1789, found 611.1790.
1). [α]D = +35 (c = 0.80 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (231 µg/4 ml): 354sh (7.71), 323 (7.85), 296sh (−0.93), 267 (−7.81), 245.5 (9.22), 210.5 (−31.69), 197.5 (66.34).1H NMR (400 MHz, CD2Cl2) δ 2.50 (s, 3H, 3″-H), 2.93 (s, 3H, 2″-H), 3.30 (d, J = 13.1 Hz, 1H, 6-Ha), 3.55 (d, J = 13.1 Hz, 1H, 6-Hb), 3.84 (s, 3H, 1″-H), 3.97 (s, 1H, 19b-H), 5.07 (s, 1H, 7-H), 5.23 (s, 1H, 12b-H), 6.59 (d, J = 9.3 Hz, 1H, 4-H), 7.03 (m, 2H, 9-H and 10-H), 7.07 (s, 1H, 12-H), 7.25 (d, J = 8.5 Hz, 1H, 17-H), 7.29–7.38 (m, 2H, 2′-H and 6′-H), 7.44 (m, 3H, 3′-H, 4′-H and 5′-H), 7.54 (dd, J = 8.5 and 2.3 Hz, 1H, 16-H), 7.97 (dd, J = 9.3 and 2.7 Hz, 1H, 3-H), 7.99–8.06 (m, 2H, 1-H and 14-H).13C NMR (100 MHz, CDCl3) δ 21.0 (C-3″), 32.7 (C-19b), 33.1 (C-6a), 40.0 (C-2″), 50.4 (C-6), 56.0 (C-1″), 73.3 (C-12b), 77.1 (C-7), 101.2 (C-19a), 110.4 (C-4), 114.4 (C-13b), 115.8 (C-17), 116.9 (C-9), 118.0 (C-10), 118.3 (C-12), 118.4 (C-12a), 122.4 (C-19c), 122.9 (C-3), 124.6 (C-1), 126.2 (C-14), 127.5 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.3 (C-4′), 133.9 (C-16), 134.0 (C-1′), 134.1 (C-17), 138.5 (C-2), 147.9 (C-14a), 148.5 (C-8a), 151.5 (C-4a), 154.4 (C-11), 158.1 (C-13a), 163.6 (C-19). IR (KBr) ν: 1117, 1228, 1262, 1315, 1497, 1556, 1726, 2915 cm−1. HRMS: calcd for C35H28N2O7Na [M + Na]+ 611.1789, found 611.1790.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H-NMR (400 MHz, CDCl3): 2.37 and 2.53 (s, 3H, 3″-H), 2.88 and 2.92 (s, 3H, 2″-H), 3.26 (d, J = 13.2 Hz, 1H, 6-Hb), 3.54 (d, J = 12.8 Hz, 1H, 6-Ha), 3.85 and 3.88 (s, 3H, 1″-H), 4.05 and 4.32 (s, 1H, 19b-H), 5.05 and 5.13 (s, 1H, 7-H), 5.15 and 5.28 (s, 1H, 12b-H), 6.52 (d, J = 8.8 Hz, 1H, 4-H), 7.03 (m, 3H, 9-H, 10-H and 12-H), 7.38 (m, 8H, 16-H, 17-H, 15-H and Ph), 7.89 (d, J = 7.4 Hz, 1H, 3-H), 8.06 and 8.21 (bs, 1H, 1-H), 8.14 (s, 1H, 18-H). 13C-NMR (100 MHz, CDCl3): 20.9 and 21.0 (C-3″), 31.9 and 32.6 (C-19b), 32.9 and 33.2 (C-6a), 39.8 and 39.9 (C-2″), 50.1 and 50.3 (C-6), 55.8 and 55.9 (C-1″), 73.2 and 75.7 (C-12b), 77.0 (C-7), 97.5 and 101.1 (C-19a), 110.2 and 110.3 (C-4), 114.3 (C-13b), 114.8 and 115.6 (C-9), 116.6 and 117.0 (C-10), 117.3 and 118.3 (C-12a), 118.0 (C-15), 118.1 and 118.4 (C-12), 118.9 (C-17), 122.2 and 122.3 (C-19c), 122.6 (C-18a), 122.8 (C-18), 124.4 and 124.5 (C-3), 126.0 and 126.1 (C-1), 126.5 (C-15), 127.5 (C-3′ and C-5′), 128.5 (C-2′ and C-6′), 129.1 and 129.2 (C-4′), 133.7 (C-16), 133.9 and 134.0 (C-1′), 134.1 (C-17), 134.7 (C-14), 135.5 (C-16), 138.3 and 138.4 (C-2), 147.8 (C-14a), 148.3 and 148.4 (C-18a), 151.3 and 151.5 (C-4a), 154.3 and 154.4 (C-11), 158.0 (C-13a), 160.9 (C-19), 163.5 (C-13a), 177.6 (C-19). IR (KBr) ν: 1231, 1313, 1498, 1582, 1614, 1628, 1707 cm−1; HRMS: calcd for C35H28N2O7Na [M + Na]+ 611.1789, found 611.1790.
1). 1H-NMR (400 MHz, CDCl3): 2.37 and 2.53 (s, 3H, 3″-H), 2.88 and 2.92 (s, 3H, 2″-H), 3.26 (d, J = 13.2 Hz, 1H, 6-Hb), 3.54 (d, J = 12.8 Hz, 1H, 6-Ha), 3.85 and 3.88 (s, 3H, 1″-H), 4.05 and 4.32 (s, 1H, 19b-H), 5.05 and 5.13 (s, 1H, 7-H), 5.15 and 5.28 (s, 1H, 12b-H), 6.52 (d, J = 8.8 Hz, 1H, 4-H), 7.03 (m, 3H, 9-H, 10-H and 12-H), 7.38 (m, 8H, 16-H, 17-H, 15-H and Ph), 7.89 (d, J = 7.4 Hz, 1H, 3-H), 8.06 and 8.21 (bs, 1H, 1-H), 8.14 (s, 1H, 18-H). 13C-NMR (100 MHz, CDCl3): 20.9 and 21.0 (C-3″), 31.9 and 32.6 (C-19b), 32.9 and 33.2 (C-6a), 39.8 and 39.9 (C-2″), 50.1 and 50.3 (C-6), 55.8 and 55.9 (C-1″), 73.2 and 75.7 (C-12b), 77.0 (C-7), 97.5 and 101.1 (C-19a), 110.2 and 110.3 (C-4), 114.3 (C-13b), 114.8 and 115.6 (C-9), 116.6 and 117.0 (C-10), 117.3 and 118.3 (C-12a), 118.0 (C-15), 118.1 and 118.4 (C-12), 118.9 (C-17), 122.2 and 122.3 (C-19c), 122.6 (C-18a), 122.8 (C-18), 124.4 and 124.5 (C-3), 126.0 and 126.1 (C-1), 126.5 (C-15), 127.5 (C-3′ and C-5′), 128.5 (C-2′ and C-6′), 129.1 and 129.2 (C-4′), 133.7 (C-16), 133.9 and 134.0 (C-1′), 134.1 (C-17), 134.7 (C-14), 135.5 (C-16), 138.3 and 138.4 (C-2), 147.8 (C-14a), 148.3 and 148.4 (C-18a), 151.3 and 151.5 (C-4a), 154.3 and 154.4 (C-11), 158.0 (C-13a), 160.9 (C-19), 163.5 (C-13a), 177.6 (C-19). IR (KBr) ν: 1231, 1313, 1498, 1582, 1614, 1628, 1707 cm−1; HRMS: calcd for C35H28N2O7Na [M + Na]+ 611.1789, found 611.1790.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = −566 (c = 0.83 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (232 µg/4 ml): 395 (−4.96), 345 (−0.41), 312.5 (−7.45), 280.5 (5.64), 251 (−7.04), 235 (3.59), 208.5 (−113.53), 197.5 (109.55). 1H NMR (400 MHz, CDCl3) δ 2.43 (s, 3H,3″-H), 2.91 (s, 3H, 2″-H), 3.26 (d, J = 12.4 Hz, 1H, 6-Ha), 3.76 (d, J = 12.8 Hz, 1H, 6-Hb), 3.89 (s, 3H, 1″-H), 4.31 (s, 1H, 19b-H), 4.93 (s, 1H, 12b-H), 5.35 (s, 1H, 7-H), 5.99 (d, J = 9.2 Hz, 1H, 4-H), 6.89 (m, 4H, 2′-H, 3′-H, 5′-H and 6′-H), 6.94–7.03 (m, 3H, 9-H, 12-H and 4′-H), 7.06 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.34 (d, J = 8.5 Hz, 1H, 17-H), 7.43 (d, J = 1.8 Hz, 1H, 16-H), 7.47 (dd, J = 8.6 and 1.6 Hz, 1H, 1-H), 7.57 (dd, J = 9.2 and 2.4 Hz, 1H, 3-H), 7.61 (s, 1H, 14-H). 13C NMR (100 MHz, CDCl3) δ 20.9 C-3″), 29.7 (C-2″), 38.2 (C-6a), 38.6 (C-19b), 53.4 (C-6), 55.9 (C-1″), 77.3 (C-7), 79.3 (C-12b), 99.5 (C-19a), 109.5 (C-4), 114.6 (C-13b), 115.0 (C-12), 116.5 (C-17), 117.1 (C-12a), 117.6 (C-11), 118.2 (C-19c), 118.6 (C-10), 122.6 (C-14), 124.2 and 124.3 (C-1 and C-3), 128.3 (Ph), 133.8 (C-16), 134.0 (C-15), 136.8 (C-1′), 137.3 (C-2), 148.8 (C-8a), 151.1 (C-17a), 152.2 (C-4a), 153.9 (C-11), 160.7 (C-13a), 161.1 (C-19). IR (KBr) ν: 1040, 1230, 1304, 1497, 1579, 1609, 1715 cm−1; HRMS: calcd for C35H28N2O7Na [M + Na]+ 611.1789, found 611.1790.
1). [α]D = −566 (c = 0.83 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (232 µg/4 ml): 395 (−4.96), 345 (−0.41), 312.5 (−7.45), 280.5 (5.64), 251 (−7.04), 235 (3.59), 208.5 (−113.53), 197.5 (109.55). 1H NMR (400 MHz, CDCl3) δ 2.43 (s, 3H,3″-H), 2.91 (s, 3H, 2″-H), 3.26 (d, J = 12.4 Hz, 1H, 6-Ha), 3.76 (d, J = 12.8 Hz, 1H, 6-Hb), 3.89 (s, 3H, 1″-H), 4.31 (s, 1H, 19b-H), 4.93 (s, 1H, 12b-H), 5.35 (s, 1H, 7-H), 5.99 (d, J = 9.2 Hz, 1H, 4-H), 6.89 (m, 4H, 2′-H, 3′-H, 5′-H and 6′-H), 6.94–7.03 (m, 3H, 9-H, 12-H and 4′-H), 7.06 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.34 (d, J = 8.5 Hz, 1H, 17-H), 7.43 (d, J = 1.8 Hz, 1H, 16-H), 7.47 (dd, J = 8.6 and 1.6 Hz, 1H, 1-H), 7.57 (dd, J = 9.2 and 2.4 Hz, 1H, 3-H), 7.61 (s, 1H, 14-H). 13C NMR (100 MHz, CDCl3) δ 20.9 C-3″), 29.7 (C-2″), 38.2 (C-6a), 38.6 (C-19b), 53.4 (C-6), 55.9 (C-1″), 77.3 (C-7), 79.3 (C-12b), 99.5 (C-19a), 109.5 (C-4), 114.6 (C-13b), 115.0 (C-12), 116.5 (C-17), 117.1 (C-12a), 117.6 (C-11), 118.2 (C-19c), 118.6 (C-10), 122.6 (C-14), 124.2 and 124.3 (C-1 and C-3), 128.3 (Ph), 133.8 (C-16), 134.0 (C-15), 136.8 (C-1′), 137.3 (C-2), 148.8 (C-8a), 151.1 (C-17a), 152.2 (C-4a), 153.9 (C-11), 160.7 (C-13a), 161.1 (C-19). IR (KBr) ν: 1040, 1230, 1304, 1497, 1579, 1609, 1715 cm−1; HRMS: calcd for C35H28N2O7Na [M + Na]+ 611.1789, found 611.1790.rac-(6aS*, 12bR*, 19bR*)- and rac-(6 aS*,12bR*,19bS*)-11-methoxy-15-methyl-12bH,19H,19bH-pyrano[3,2-c:3,4-c':5,6-c'']trichromen-19-one [rac-(6aS*, 12bR*, 19bR*)-4ca and rac-(6aS*, 12bR*, 19bR*)-5ca] and rac-(6aS*, 12bR*, 19bR*)– and rac-(6 aS*,12bR*,19bS*)-11-methoxy-17-methyl-12bH,19H,19bH-pyrano[2,3-b:5,4-c':5,6-c'']trichromen-19-one [rac-(6aS*, 12bR*, 19bR*)-6ca and rac-(6aS*, 12bR*, 19bR*)-7ca]
The reaction of 1c and 2a was carried out according to method B, and the crude product was purified by column chromatography (hexane/ethyl acetate 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 19bR*)-4ca as white crystals (34%), rac-(6 aS*,12bR*,19bS*)-5ca as pale yellow amorphous solid (17%), rac-(6aS*, 12bR*, 19bR*)-6ca as pale yellow crystals (19%) and rac-(6 aS*,12bR*,19bS*)-7ca as white crystals (6%).
1), affording rac-(6aS*, 12bR*, 19bR*)-4ca as white crystals (34%), rac-(6 aS*,12bR*,19bS*)-5ca as pale yellow amorphous solid (17%), rac-(6aS*, 12bR*, 19bR*)-6ca as pale yellow crystals (19%) and rac-(6 aS*,12bR*,19bS*)-7ca as white crystals (6%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.36 (s, 3H, 2′-H), 3.88 (s, 3H, 1′-H), 3.95 (d, J = 11.7 Hz, 1H, 7-Ha), 3.99 (d, J = 11.6 Hz, 1H, 7-Hb), 4.14 (d, J = 11.8 Hz, 1H, 6-Ha), 4.28 (s, 1H, 19b-H), 4.34 (d, J = 11.8 Hz, 1H, 6-Hb), 5.31 (m, 1H, 12b-H), 6.86 (d, J = 8.2 Hz, 1H, 4-H), 6.89–6.97 (m, 2H, 2-H and 9-H), 7.01 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.06 (d, J = 2.9 Hz, 1H, 12-H), 7.17 (t, J = 7.7 Hz, 1H, 3-H), 7.25 (d, J = 8.5 Hz, 1H, 17-H), 7.35 (dd, J = 8.5 and 1.6 Hz, 1H, 16-H), 7.47 (s, 1H, 14-H), 7.53 (d, J = 7.9 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.8 (C-2′), 30.5 (C-6a), 32.0 (C-19b), 55.9 (C-1′), 66.1 (C-7), 68.4 (C-6), 71.1 (C-12b), 101.1 (C-19a), 114.5 (C-13b), 116.0 (C-12), 116.4 (C-17), 117.0,(C-4), 118.0 (C-9 and C-10), 118.2 (C-12a), 122.1 (C-2), 122.3 (C-19c), 122.8 (C-14), 128.4 (C-3), 129.9 (C-1), 133.4 (C-16), 133.9 (C-15), 146.7 (C-8a), 150.9 (C-17a), 152.3 (C-4a), 154.1 (C-11), 157.1 (C-13a), 164.3 (C-19). IR (KBr) ν: 1048, 1219, 1498, 1583, 1631, 1698, 2928 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1308.
1). 1H NMR (400 MHz, CDCl3) δ 2.36 (s, 3H, 2′-H), 3.88 (s, 3H, 1′-H), 3.95 (d, J = 11.7 Hz, 1H, 7-Ha), 3.99 (d, J = 11.6 Hz, 1H, 7-Hb), 4.14 (d, J = 11.8 Hz, 1H, 6-Ha), 4.28 (s, 1H, 19b-H), 4.34 (d, J = 11.8 Hz, 1H, 6-Hb), 5.31 (m, 1H, 12b-H), 6.86 (d, J = 8.2 Hz, 1H, 4-H), 6.89–6.97 (m, 2H, 2-H and 9-H), 7.01 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.06 (d, J = 2.9 Hz, 1H, 12-H), 7.17 (t, J = 7.7 Hz, 1H, 3-H), 7.25 (d, J = 8.5 Hz, 1H, 17-H), 7.35 (dd, J = 8.5 and 1.6 Hz, 1H, 16-H), 7.47 (s, 1H, 14-H), 7.53 (d, J = 7.9 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.8 (C-2′), 30.5 (C-6a), 32.0 (C-19b), 55.9 (C-1′), 66.1 (C-7), 68.4 (C-6), 71.1 (C-12b), 101.1 (C-19a), 114.5 (C-13b), 116.0 (C-12), 116.4 (C-17), 117.0,(C-4), 118.0 (C-9 and C-10), 118.2 (C-12a), 122.1 (C-2), 122.3 (C-19c), 122.8 (C-14), 128.4 (C-3), 129.9 (C-1), 133.4 (C-16), 133.9 (C-15), 146.7 (C-8a), 150.9 (C-17a), 152.3 (C-4a), 154.1 (C-11), 157.1 (C-13a), 164.3 (C-19). IR (KBr) ν: 1048, 1219, 1498, 1583, 1631, 1698, 2928 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1308.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.37 (s, 3H, 2′-H), 3.43 (dd, J = 11.6 and 2.0 Hz, 1H, 7-Ha), 3.81–3.89 (m, 4H, 7-Hb and 1′-H), 3.96 (dd, J = 10.4 and 1.8 Hz, 1H, 6-Ha), 4.24 (dd, J = 10.4 and 1.0 Hz, 1H, 6-Hb), 4.39 (s, 1H, 19b-H), 4.89 (d, J = 2.0 Hz, 1H, 12b-H), 6.85 (d, J = 9.0 Hz, 1H, 9-H), 6.88–6.98 (m, 3H, 12-H, 2-H and 4-H), 6.98 (dd, J = 9.0 and 3.1 Hz, 1H, 10-H), 7.06 (d, J = 7.5 Hz, 1H, 1-H), 7.16–7.25 (m, 1H, 3-H), 7.27 (d, J = 8.6 Hz, 1H, 17-H), 7.37 (dd, J = 8.5 and 1.8 Hz, 1H, 16-H), 7.53 (bs, 1H, 14-H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-2′), 35.6 (C-19b), 36.4 (C-6a), 56.0 (C-1′), 63.4 (C-7), 69.5 (C-6), 75.6 (C-12b), 99.3 (C-19a), 114.8 (C-13b), 115.4 (C-12), 116.5 (C-17), 116.8 (C-4), 117.4 (C-12a), 117.8 (C-9), 118.5 (C-10), 121.3 (C-2), 122.8 (C-14), 122.9 (C-19c), 126.3 (C-1), 128.4 (C-3), 133.5 (C-16), 134.0 (C-15), 148.9 (C-8a), 151.2 (C-17a), 153.7 (C-11), 154.8 (C-4a), 159.7 (C-13a), 161.7 (C-19). IR (KBr) ν: 1043, 1221, 1498, 1578, 1625, 1714, 2834, 2931 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1308.
1). 1H NMR (400 MHz, CDCl3) δ 2.37 (s, 3H, 2′-H), 3.43 (dd, J = 11.6 and 2.0 Hz, 1H, 7-Ha), 3.81–3.89 (m, 4H, 7-Hb and 1′-H), 3.96 (dd, J = 10.4 and 1.8 Hz, 1H, 6-Ha), 4.24 (dd, J = 10.4 and 1.0 Hz, 1H, 6-Hb), 4.39 (s, 1H, 19b-H), 4.89 (d, J = 2.0 Hz, 1H, 12b-H), 6.85 (d, J = 9.0 Hz, 1H, 9-H), 6.88–6.98 (m, 3H, 12-H, 2-H and 4-H), 6.98 (dd, J = 9.0 and 3.1 Hz, 1H, 10-H), 7.06 (d, J = 7.5 Hz, 1H, 1-H), 7.16–7.25 (m, 1H, 3-H), 7.27 (d, J = 8.6 Hz, 1H, 17-H), 7.37 (dd, J = 8.5 and 1.8 Hz, 1H, 16-H), 7.53 (bs, 1H, 14-H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-2′), 35.6 (C-19b), 36.4 (C-6a), 56.0 (C-1′), 63.4 (C-7), 69.5 (C-6), 75.6 (C-12b), 99.3 (C-19a), 114.8 (C-13b), 115.4 (C-12), 116.5 (C-17), 116.8 (C-4), 117.4 (C-12a), 117.8 (C-9), 118.5 (C-10), 121.3 (C-2), 122.8 (C-14), 122.9 (C-19c), 126.3 (C-1), 128.4 (C-3), 133.5 (C-16), 134.0 (C-15), 148.9 (C-8a), 151.2 (C-17a), 153.7 (C-11), 154.8 (C-4a), 159.7 (C-13a), 161.7 (C-19). IR (KBr) ν: 1043, 1221, 1498, 1578, 1625, 1714, 2834, 2931 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1308.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.45 (s, 3H, 2′-H), 3.82 (s, 3H, 1′-H), 3.94 (s, 2H, 7-H), 4.10 (d, J = 11.9 Hz, 1H, 6-Ha), 4.20 (s, 1H, 19b-H), 4.33 (d, J = 11.8 Hz, 1H, 6-Hb), 5.40 (s, 1H, 12b-H), 6.84 (m, 2H, 4-H and 9-H), 6.92 (m, 2H, 2-H and 10-H), 7.10 (d, J = 2.9 Hz, 1H, 12-H), 7.15 (d, J = 8.0 Hz, 1H, 3-H), 7.21 (d, J = 8.5 Hz, 1H, 15-H), 7.36 (d, J = 7.8 Hz, 1H, 1-H), 7.46 (dd, J = 8.5 and 1.6 Hz, 1H, 16-H), 7.91 (s, 1H, 18-H). 13C NMR (100 MHz, CDCl3) δ 20.9 (C-2′), 30.8 (C-6a), 31.7 (C-19b), 55.9 (C-1′), 66.3 (C-7), 68.2 (C-6), 71.8 (C-12b), 100.9 (C-19a), 112.3 (C-17a), 115.2 (C-12), 116.8 (C-15), 117.2 (C-9), 117.6 (C-19c), 117.9 (C-4), 119.1 (C-10), 121.5 (C-12a), 122.1 (C-2), 123.5 (C-18), 128.7 (C-3), 129.4 (C-1), 135.0 (C-17), 136.0 (C-16), 146.5 (C-8a), 151.7 (C-14a), 152.5 (C-4a), 154.4 (C-11), 159.7 (C-19), 161.8 (C-13a). IR (KBr) ν: 1042, 1218, 1267, 1453, 1498, 1574, 1624 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1310.
1). 1H NMR (400 MHz, CDCl3) δ 2.45 (s, 3H, 2′-H), 3.82 (s, 3H, 1′-H), 3.94 (s, 2H, 7-H), 4.10 (d, J = 11.9 Hz, 1H, 6-Ha), 4.20 (s, 1H, 19b-H), 4.33 (d, J = 11.8 Hz, 1H, 6-Hb), 5.40 (s, 1H, 12b-H), 6.84 (m, 2H, 4-H and 9-H), 6.92 (m, 2H, 2-H and 10-H), 7.10 (d, J = 2.9 Hz, 1H, 12-H), 7.15 (d, J = 8.0 Hz, 1H, 3-H), 7.21 (d, J = 8.5 Hz, 1H, 15-H), 7.36 (d, J = 7.8 Hz, 1H, 1-H), 7.46 (dd, J = 8.5 and 1.6 Hz, 1H, 16-H), 7.91 (s, 1H, 18-H). 13C NMR (100 MHz, CDCl3) δ 20.9 (C-2′), 30.8 (C-6a), 31.7 (C-19b), 55.9 (C-1′), 66.3 (C-7), 68.2 (C-6), 71.8 (C-12b), 100.9 (C-19a), 112.3 (C-17a), 115.2 (C-12), 116.8 (C-15), 117.2 (C-9), 117.6 (C-19c), 117.9 (C-4), 119.1 (C-10), 121.5 (C-12a), 122.1 (C-2), 123.5 (C-18), 128.7 (C-3), 129.4 (C-1), 135.0 (C-17), 136.0 (C-16), 146.5 (C-8a), 151.7 (C-14a), 152.5 (C-4a), 154.4 (C-11), 159.7 (C-19), 161.8 (C-13a). IR (KBr) ν: 1042, 1218, 1267, 1453, 1498, 1574, 1624 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1310.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.47 (s, 3H, 2′-H), 3.46 (dd, J = 11.5 and 2.1 Hz, 1H, 7-Ha), 3.81 (s, 3H, 1′-H), 3.86 (d, J = 11.5 Hz, 1H, 7-Hb), 3.95 (d, J = 10.4 Hz, 1H, 6-Ha), 4.26 (d, J = 10.4 Hz, 1H, 6-Hb), 4.37 (s, 1H, 19b-H), 4.93 (d, J = 1.3 Hz, 1H, 12b-H), 6.81 (d, J = 8.7 Hz, 1H, 9-H), 6.90–6.98 (m, 4H, 2-H, 4-H, 10-H and 12-H), 7.01 (d, J = 7.3 Hz, 1H, 3-H), 7.22 (m, 1H, 1-H), 7.26 (d, J = 8.6 Hz, 1H, 15-H), 7.50 (dd, J = 8.6 and 2.3 Hz, 1H, 16-H), 7.93 (bs, 1H, 18-H). 13C NMR (90 MHz, CDCl3) δ 21.0 (C-2′), 35.3 (C-19b), 36.1 (C-6a), 55.9 (C-1′), 63.0 (C-7), 69.4 (C-6), 76.5 (C-12b), 99.1 (C-19a), 112.3 (C-14a), 115.1 (C-4), 116.7 (C-12a), 116.8 (C-15), 117.0 (C-12), 117.8 (C-9), 119.0 (C-10), 121.2 (C-2), 122.0 (C-19c), 123.6 (C-18), 126.0 (C-3), 128.6 (C-1), 135.1 (C-17), 136.1 (C-16), 148.7 (C-8a), 151.8 (C-17a), 153.9 (C-4a), 154.7 (C-11), 159.0 (C-19), 162.2 (C-13a). IR (KBr) ν: 1041, 1113, 1218, 1276, 1498, 1552, 1629, 1733, 2929 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1310.
1). 1H NMR (400 MHz, CDCl3) δ 2.47 (s, 3H, 2′-H), 3.46 (dd, J = 11.5 and 2.1 Hz, 1H, 7-Ha), 3.81 (s, 3H, 1′-H), 3.86 (d, J = 11.5 Hz, 1H, 7-Hb), 3.95 (d, J = 10.4 Hz, 1H, 6-Ha), 4.26 (d, J = 10.4 Hz, 1H, 6-Hb), 4.37 (s, 1H, 19b-H), 4.93 (d, J = 1.3 Hz, 1H, 12b-H), 6.81 (d, J = 8.7 Hz, 1H, 9-H), 6.90–6.98 (m, 4H, 2-H, 4-H, 10-H and 12-H), 7.01 (d, J = 7.3 Hz, 1H, 3-H), 7.22 (m, 1H, 1-H), 7.26 (d, J = 8.6 Hz, 1H, 15-H), 7.50 (dd, J = 8.6 and 2.3 Hz, 1H, 16-H), 7.93 (bs, 1H, 18-H). 13C NMR (90 MHz, CDCl3) δ 21.0 (C-2′), 35.3 (C-19b), 36.1 (C-6a), 55.9 (C-1′), 63.0 (C-7), 69.4 (C-6), 76.5 (C-12b), 99.1 (C-19a), 112.3 (C-14a), 115.1 (C-4), 116.7 (C-12a), 116.8 (C-15), 117.0 (C-12), 117.8 (C-9), 119.0 (C-10), 121.2 (C-2), 122.0 (C-19c), 123.6 (C-18), 126.0 (C-3), 128.6 (C-1), 135.1 (C-17), 136.1 (C-16), 148.7 (C-8a), 151.8 (C-17a), 153.9 (C-4a), 154.7 (C-11), 159.0 (C-19), 162.2 (C-13a). IR (KBr) ν: 1041, 1113, 1218, 1276, 1498, 1552, 1629, 1733, 2929 cm−1; HRMS: calcd for C28H22O6Na [M + Na]+ 477.1309, found 477.1310.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 19bS*)-4ab as yellow crystals (40%) and rac-(6aS*, 12bR*, 19bS*)-6ab as yellow powder (45%).
1), affording rac-(6aS*, 12bR*, 19bS*)-4ab as yellow crystals (40%) and rac-(6aS*, 12bR*, 19bS*)-6ab as yellow powder (45%).Alternatively, the reaction was carried out at room temperature, affording rac-(6aS*, 12bR*, 19bS*)-4ab as yellow crystals (54%) and rac-(6aS*, 12bR*, 19bS*)-6ab as yellow powder (19%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.23 (s, 3H, 3′-H), 2.33 (s, 3H, 4′-H), 2.95 (s, 3H, 2′-H), 3.27 (d, J = 13.2 Hz, 1H, 6-Ha), 3.68 (d, J = 13.2 Hz, 1H, 6-Hb), 3.85 (s, 3H, 1′-H), 3.90 (d, J = 11.5 Hz, 1H, 7-Ha), 3.99 (d, J = 11.5 Hz, 1H, 7-Hb), 4.24 (s, 1H, 19b-H), 5.02 (s, 1H, 12b-H), 6.53 (d, J = 9.2 Hz, 1H, 4-H), 6.89 (d, J = 9.8 Hz, 1H, 9-H), 6.95–7.01 (m, 2H, 10-H and 12-H), 7.14 (s, 1H, 17-H), 7.36 (s, 1H, 14-H), 7.95 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.07 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.3 (C-3′), 20.4 (C-4′), 30.7 (C-6a), 33.2 (C-19b), 39.8 (C-2), 54.7 (C-6), 56.0 (C-1′), 67.6 (C-7), 72.2 (C-12b), 100.0 (C-19a), 110.3 (C-4), 112.4 (C-13b), 116.0 (C-12), 117.5 (C-17), 117.9 (C-10), 118.1 (C-9), 118.5 (C-12a), 121.6 (C-19c), 123.1 (C-14), 124.8 (C-3), 126.2 (C-1), 133.1 (C-15), 138.5 (C-2), 142.8 (C-16), 146.9 (C-8a), 149.1 (C-4a), 151.6 (C-17a), 154.2 (C-11), 157.5 (C-13a), 164.2 (C-19). IR (KBr) ν: 1037, 1208, 1281, 1322, 1498, 1630, 1705, 2926, cm−1; HRMS: calcd for C30H26N2O7Na [M + Na]+ 549.1632, found 549.1631.
1). 1H NMR (400 MHz, CDCl3) δ 2.23 (s, 3H, 3′-H), 2.33 (s, 3H, 4′-H), 2.95 (s, 3H, 2′-H), 3.27 (d, J = 13.2 Hz, 1H, 6-Ha), 3.68 (d, J = 13.2 Hz, 1H, 6-Hb), 3.85 (s, 3H, 1′-H), 3.90 (d, J = 11.5 Hz, 1H, 7-Ha), 3.99 (d, J = 11.5 Hz, 1H, 7-Hb), 4.24 (s, 1H, 19b-H), 5.02 (s, 1H, 12b-H), 6.53 (d, J = 9.2 Hz, 1H, 4-H), 6.89 (d, J = 9.8 Hz, 1H, 9-H), 6.95–7.01 (m, 2H, 10-H and 12-H), 7.14 (s, 1H, 17-H), 7.36 (s, 1H, 14-H), 7.95 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.07 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.3 (C-3′), 20.4 (C-4′), 30.7 (C-6a), 33.2 (C-19b), 39.8 (C-2), 54.7 (C-6), 56.0 (C-1′), 67.6 (C-7), 72.2 (C-12b), 100.0 (C-19a), 110.3 (C-4), 112.4 (C-13b), 116.0 (C-12), 117.5 (C-17), 117.9 (C-10), 118.1 (C-9), 118.5 (C-12a), 121.6 (C-19c), 123.1 (C-14), 124.8 (C-3), 126.2 (C-1), 133.1 (C-15), 138.5 (C-2), 142.8 (C-16), 146.9 (C-8a), 149.1 (C-4a), 151.6 (C-17a), 154.2 (C-11), 157.5 (C-13a), 164.2 (C-19). IR (KBr) ν: 1037, 1208, 1281, 1322, 1498, 1630, 1705, 2926, cm−1; HRMS: calcd for C30H26N2O7Na [M + Na]+ 549.1632, found 549.1631.The enantiomers of rac-(6aS*, 12bR*, 19bS*)-4ab were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), and the on-line HPLC-ECD spectra were recorded.
5), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS, 19bR)-4ab: tR = 10.14 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 390 (3.30), 330.5sh (−6.59), 316 (−16.13), 302.5sh (−7.42), 288sh (3.94), 277.5 (4.82), 255.5 (−4.44), 230 (−58.44).
5]: 390 (3.30), 330.5sh (−6.59), 316 (−16.13), 302.5sh (−7.42), 288sh (3.94), 277.5 (4.82), 255.5 (−4.44), 230 (−58.44).
(6aS, 12bR, 19bS)-4ab: tR = 13.11 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 390 (−2.51), 330.5sh (5.53), 316 (13.82), 302.5sh (5.89), 288sh (−3.85), 277.5 (−4.16), 255.5 (3.87), 230 (48.37).
5]: 390 (−2.51), 330.5sh (5.53), 316 (13.82), 302.5sh (5.89), 288sh (−3.85), 277.5 (−4.16), 255.5 (3.87), 230 (48.37).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (500 MHz, CDCl3) δ 2.37 (s, 3H, 3′-H), 2.38 (s, 3H, 4′-H), 2.96 (s, 3H, 2′-H), 3.26 (d, J = 13.2 Hz, 1H, 6-Ha), 3.66 (d, J = 13.2 Hz, 1H, 6-Hb), 3.81 (s, 3H, 1′-H), 3.95 (q, J = 11.8 Hz, 2H, 7-H), 4.45 (s, 1H, 19b-H), 5.18 (s, 1H, 12b-H), 6.58 (d, J = 9.2 Hz, 1H, 4-H), 6.86–6.92 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 9.0 and 2.9 Hz, 1H, 10-H), 7.11 (s, 1H, 15-H), 8.01 (dd, J = 9.0 and 2.0 Hz, 1H, 3-H), 8.04 (s, 1H, 18-H), 8.05 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.4 (C-4′), 20.5 (C-3′), 31.1 (C-6a), 32.5 (C-19b), 39.8 (C-2′), 54.6 (C-6), 55.9 (C-1′), 67.7 (C-7), 74.8 (C-12b), 97.1 (C-19a), 110.3 (C-4), 115.2 (C-12), 117.5 (C-15), 118.4 (C-9), 118.9 (C-10), 120.4 (C-18a), 122.0 (C-19c), 124.8 (C-3), 126.2 (C-18), 126.6 (C-1), 134.8 (C-17), 138.7 (C-2), 143.9 (C-16), 146.9 (C-8a), 149.0 (C-4a), 151.8 (C-14a), 154.3 (C-11), 160.2 (C-13a), 178.2 (C-19). IR (KBr) ν: 1042, 1213, 1267, 1291, 1320, 1499, 1619, 1706, 2924 cm−1; HRMS: calcd for C30H26N2O7Na [M + Na]+ 549.1632, found 549.1631.
1). 1H NMR (500 MHz, CDCl3) δ 2.37 (s, 3H, 3′-H), 2.38 (s, 3H, 4′-H), 2.96 (s, 3H, 2′-H), 3.26 (d, J = 13.2 Hz, 1H, 6-Ha), 3.66 (d, J = 13.2 Hz, 1H, 6-Hb), 3.81 (s, 3H, 1′-H), 3.95 (q, J = 11.8 Hz, 2H, 7-H), 4.45 (s, 1H, 19b-H), 5.18 (s, 1H, 12b-H), 6.58 (d, J = 9.2 Hz, 1H, 4-H), 6.86–6.92 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 9.0 and 2.9 Hz, 1H, 10-H), 7.11 (s, 1H, 15-H), 8.01 (dd, J = 9.0 and 2.0 Hz, 1H, 3-H), 8.04 (s, 1H, 18-H), 8.05 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.4 (C-4′), 20.5 (C-3′), 31.1 (C-6a), 32.5 (C-19b), 39.8 (C-2′), 54.6 (C-6), 55.9 (C-1′), 67.7 (C-7), 74.8 (C-12b), 97.1 (C-19a), 110.3 (C-4), 115.2 (C-12), 117.5 (C-15), 118.4 (C-9), 118.9 (C-10), 120.4 (C-18a), 122.0 (C-19c), 124.8 (C-3), 126.2 (C-18), 126.6 (C-1), 134.8 (C-17), 138.7 (C-2), 143.9 (C-16), 146.9 (C-8a), 149.0 (C-4a), 151.8 (C-14a), 154.3 (C-11), 160.2 (C-13a), 178.2 (C-19). IR (KBr) ν: 1042, 1213, 1267, 1291, 1320, 1499, 1619, 1706, 2924 cm−1; HRMS: calcd for C30H26N2O7Na [M + Na]+ 549.1632, found 549.1631.The enantiomers of rac-(6aS*, 12bR*, 19bS*)-6ab were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), and the on-line HPLC-ECD spectra were recorded.
5), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS, 19bR)-6ab: tR = 8.12 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 355 (−4.02), 301.5sh (−9.02), 294 (−11.85), 264.5sh (−16.06), 246.5 (−31.16).
5]: 355 (−4.02), 301.5sh (−9.02), 294 (−11.85), 264.5sh (−16.06), 246.5 (−31.16).
(6aS, 12bR, 19bS)-6ab: tR = 15.72 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 355 (1.33), 301.5sh (3.11), 294 (3.90), 264.5sh (5.68), 246.5 (10.64).
5]: 355 (1.33), 301.5sh (3.11), 294 (3.90), 264.5sh (5.68), 246.5 (10.64).
(6aS, 7R, 12bR, 19bS) and (6aS, 7R, 12bR, 19bR)-11-methoxy-5,15,16-trimethyl-2-nitro-7-phenyl-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,4':5,6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [(6aS, 7R, 12bR, 19bS)-4bb and (6aS, 7R, 12bR, 19bR)-5bb] and (6aS, 7R, 12bR, 19bS)-11-methoxy-5,16,17-trimethyl-2-nitro-7-phenyl-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,2′:5,6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [(6aS, 7R, 12bR, 19bS)-6bb]
The reaction of (R)-1b and 2b was carried out according to method A, and the crude product was purified by column chromatography (hexane/chloroform/ethyl acetate 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording (6aS, 7R, 12bR, 19bS)-4bb as yellow powder (26%), (6aS, 7R, 12bR, 19bS)-6bb as yellow powder (16%) and (6aS, 7R, 12bR, 19bR)-5bb as yellow powder (44%).
1), affording (6aS, 7R, 12bR, 19bS)-4bb as yellow powder (26%), (6aS, 7R, 12bR, 19bS)-6bb as yellow powder (16%) and (6aS, 7R, 12bR, 19bR)-5bb as yellow powder (44%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = −7 (c = 1.00 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (288 µg/4 ml): 331sh (1.67), 317 (5.73), 307sh (2.84), 288.5 (−4.26), 277 (−3.93), 252.5 (0.28), 230 (24.87), 212 (−67.01), 198.5 (113.93). 1H NMR (400 MHz, CDCl3) δ 2.27 (s, 3H, 3″-H), 2.36 (s, 3H, 4″-H), 2.92 (s, 3H, 2″-H), 3.24 (d, J = 12.9 Hz, 1H, 6-Ha), 3.53 (d, J = 12.9 Hz, 1H, 6-Hb), 3.88 (s, 3H, 1″-H), 4.03 (s, 1H, 19b-H), 5.05 (s, 1H, 7-H), 5.12 (s, 1H, 12b-H), 6.52 (d, J = 9.2 Hz, 1H, 4-H), 7.03 (m, 3H, 9-H, 10-H and 12-H), 7.20 (s, 1H, 17-H), 7.29 (d, J = 6.8 Hz, 2H, 2′-H and 6′-H), 7.38 (m, 3H, 3′-H, 4′-H and 5′-H), 7.44 (s, 1H, 14-H), 7.93 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.12 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.3(C-3″), 20.5 (C-4″), 32.7 (C-19b), 33.1 (C-6a), 39.9 (C-2″), 50.5 (C-6), 56.0 (C-1″), 73.2 (C-12b), 77.1 (C-7), 100.3 (C-19a), 110.3 (C-4), 112.3 (C-13b), 115.8 (C-12), 117.6 (C-17), 118.0 (C-10), 118.2 (C-9), 118.5 (C-12a), 122.6 (C-19c), 123.1 (C-14), 124.6 (C-3), 126.2 (C-1), 127.5 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.3 (C-4′), 133.2 (C-16), 134.1 (C-1′), 138.5 (C-2), 143.0 (C-15), 148.0 (C-8a), 148.6 (C-4a), 151.8 (C-17a), 154.4 (C-11), 158.3 (C-13a), 163.8 (C-19). IR (KBr) ν: 1036, 1233, 1314, 1498, 1630, 1705, 2921 cm−1; HRMS: calcd for C36H30N2O7Na [M + Na]+ 625.1945, found 625.1948.
1). [α]D = −7 (c = 1.00 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (288 µg/4 ml): 331sh (1.67), 317 (5.73), 307sh (2.84), 288.5 (−4.26), 277 (−3.93), 252.5 (0.28), 230 (24.87), 212 (−67.01), 198.5 (113.93). 1H NMR (400 MHz, CDCl3) δ 2.27 (s, 3H, 3″-H), 2.36 (s, 3H, 4″-H), 2.92 (s, 3H, 2″-H), 3.24 (d, J = 12.9 Hz, 1H, 6-Ha), 3.53 (d, J = 12.9 Hz, 1H, 6-Hb), 3.88 (s, 3H, 1″-H), 4.03 (s, 1H, 19b-H), 5.05 (s, 1H, 7-H), 5.12 (s, 1H, 12b-H), 6.52 (d, J = 9.2 Hz, 1H, 4-H), 7.03 (m, 3H, 9-H, 10-H and 12-H), 7.20 (s, 1H, 17-H), 7.29 (d, J = 6.8 Hz, 2H, 2′-H and 6′-H), 7.38 (m, 3H, 3′-H, 4′-H and 5′-H), 7.44 (s, 1H, 14-H), 7.93 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.12 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.3(C-3″), 20.5 (C-4″), 32.7 (C-19b), 33.1 (C-6a), 39.9 (C-2″), 50.5 (C-6), 56.0 (C-1″), 73.2 (C-12b), 77.1 (C-7), 100.3 (C-19a), 110.3 (C-4), 112.3 (C-13b), 115.8 (C-12), 117.6 (C-17), 118.0 (C-10), 118.2 (C-9), 118.5 (C-12a), 122.6 (C-19c), 123.1 (C-14), 124.6 (C-3), 126.2 (C-1), 127.5 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.3 (C-4′), 133.2 (C-16), 134.1 (C-1′), 138.5 (C-2), 143.0 (C-15), 148.0 (C-8a), 148.6 (C-4a), 151.8 (C-17a), 154.4 (C-11), 158.3 (C-13a), 163.8 (C-19). IR (KBr) ν: 1036, 1233, 1314, 1498, 1630, 1705, 2921 cm−1; HRMS: calcd for C36H30N2O7Na [M + Na]+ 625.1945, found 625.1948.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = 115 (c = 1.01 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (299 µg/4 ml): 390 (2.15), 310.5 (−1.85), 300.5sh (1.50), 293.5 (2.27), 266sh (4.66), 246.5 (7.15), 211.5 (−33.58), 199 (35.49), 191.5 (−94.02). 1H NMR (400 MHz, CDCl3) δ 2.42 (s, 3H, 3″-H), 2.44 (s, 3H, 4″-H), 2.93 (s, 3H, 2″-H), 3.22 (d, J = 12.9 Hz, 1H, 6-Ha), 3.55 (d, J = 12.9 Hz, 1H, 6-Hb), 3.85 (s, 3H, 1″-H), 4.34 (s, 1H, 19b-H), 5.15 (s, 1H, 7-H), 5.27 (s, 1H, 12b-H), 6.53 (d, J = 9.2 Hz, 1H, 4-H), 6.97 (s, 1H, 12-H), 7.03 (m, 2H, 9-H and 10-H), 7.19 (s, 1H, 15-H), 7.26–7.33 (m, 2H, 2′-H and 6′-H), 7.40 (m, 3H, 3′-H, 4′-H and 5′-H), 7.96 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H), 8.07 (bs, 1H, 1-H), 8.16 (s, 1H, 18-H). 13C NMR (100 MHz, CDCl3) δ 19.4 (C-4″), 20.5 (C-3″), 32.0 (C-19b), 33.4 (C-6a), 39.9 (C-2″), 50.3 (C-6), 56.0 (C-1″), 75.7 (C-12b), 77.0 (C-7), 97.4 (C-19a), 110.3 (C-4), 115.0 (C-12), 117.5 (C-12a), 117.6 (C-15), 118.5 (C-10), 119.0 (C-9), 120.5 (C-18a), 122.9 (C-19c), 124.5 (C-3), 126.5 (C-18), 126.7 (C-1), 127.6 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.2 (C-4′), 134.0 (C-1′), 134.8 (C-17), 138.7 (C-2), 144.0 (C-16), 148.0 (C-8a), 148.4 (C-4a), 151.9 (C-14a), 154.5 (C-11), 160.8 (C-13a), 177.7 (C-19). IR (KBr) ν: 1261, 1314, 1432, 1463, 1498, 1621, 2922 cm−1; HRMS: calcd for C36H30N2O7Na [M + Na]+ 625.1945, found 625.1948.
1). [α]D = 115 (c = 1.01 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (299 µg/4 ml): 390 (2.15), 310.5 (−1.85), 300.5sh (1.50), 293.5 (2.27), 266sh (4.66), 246.5 (7.15), 211.5 (−33.58), 199 (35.49), 191.5 (−94.02). 1H NMR (400 MHz, CDCl3) δ 2.42 (s, 3H, 3″-H), 2.44 (s, 3H, 4″-H), 2.93 (s, 3H, 2″-H), 3.22 (d, J = 12.9 Hz, 1H, 6-Ha), 3.55 (d, J = 12.9 Hz, 1H, 6-Hb), 3.85 (s, 3H, 1″-H), 4.34 (s, 1H, 19b-H), 5.15 (s, 1H, 7-H), 5.27 (s, 1H, 12b-H), 6.53 (d, J = 9.2 Hz, 1H, 4-H), 6.97 (s, 1H, 12-H), 7.03 (m, 2H, 9-H and 10-H), 7.19 (s, 1H, 15-H), 7.26–7.33 (m, 2H, 2′-H and 6′-H), 7.40 (m, 3H, 3′-H, 4′-H and 5′-H), 7.96 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H), 8.07 (bs, 1H, 1-H), 8.16 (s, 1H, 18-H). 13C NMR (100 MHz, CDCl3) δ 19.4 (C-4″), 20.5 (C-3″), 32.0 (C-19b), 33.4 (C-6a), 39.9 (C-2″), 50.3 (C-6), 56.0 (C-1″), 75.7 (C-12b), 77.0 (C-7), 97.4 (C-19a), 110.3 (C-4), 115.0 (C-12), 117.5 (C-12a), 117.6 (C-15), 118.5 (C-10), 119.0 (C-9), 120.5 (C-18a), 122.9 (C-19c), 124.5 (C-3), 126.5 (C-18), 126.7 (C-1), 127.6 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.2 (C-4′), 134.0 (C-1′), 134.8 (C-17), 138.7 (C-2), 144.0 (C-16), 148.0 (C-8a), 148.4 (C-4a), 151.9 (C-14a), 154.5 (C-11), 160.8 (C-13a), 177.7 (C-19). IR (KBr) ν: 1261, 1314, 1432, 1463, 1498, 1621, 2922 cm−1; HRMS: calcd for C36H30N2O7Na [M + Na]+ 625.1945, found 625.1948.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 5
5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = 453 (c = 1.10 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (199 µg/4 ml): 399.5 (−6.64), 313 (−8.66), 282.5 (7.96), 251.5 (−9.34), 237 (2.85), 208.5 (−143.57), 198.5 (129.65). 1H NMR (400 MHz, CDCl3) δ 2.31 (s, 3H, 4″-H), 2.41 (s, 3H, 3″-H), 2.91 (s, 3H, 2″-H), 3.23 (d, J = 12.4 Hz, 1H, 6-Ha), 3.75 (d, J = 12.4 Hz, 1H, 6-Hb), 3.87 (s, 3H, 1″-H), 4.30 (s, 1H, 19b-H), 4.90 (s, 1H, 12b-H), 5.34 (s, 1H, 7-H), 6.00 (d, J = 9.1 Hz, 1H, 4-H), 6.90 (brs, 4H, 2′-H, 3′-H, 5′-H and 6′-H), 6.93–7.00 (m, 3H, 9-H, 12-H and 4′-H), 7.05 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.23 (s, 1H, 17-H), 7.47 (d, J = 2.4 Hz, 1H, 1-H), 7.55 (s, 1H, 14-H), 7.58 (dd, J = 9.1 and 2.4 Hz, 1H, 3-H). 13C NMR (90 MHz, CDCl3) δ 19.4 (C-4″), 20.6 (C-3″), 37.9 (C-2″), 38.5 (C-6a), 38.9 (C-19b), 53.7 (C-7), 56.1 (C-1″), 77.4 (C-7), 79.5 (C-12b), 98.8 (C-19c), 109.6 (C-4), 112.6 (C-13b), 115.2 (C-9), 117.4 (C-17), 117.8 (C-12), 118.6 (C-19a and C-12a), 118.7 (C-10), 123.0 (C-14), 124.4 (C-1 and C-3), 127.7–128.5 (Ph), 133.2 (C-15), 137.0 (C-16), 137.7 (C-1′), 143.1 (C-2), 149.1 (C-8a), 151.6 (C-17a), 152.3 (C-4a), 154.1 (C-11), 160.9 (C-13a), 161.4 (C-19). IR (KBr) ν: 1037, 1228, 1302, 1319, 1498, 1613, 1717, 2920 cm−1; HRMS: calcd for C36H30N2O7Na [M + Na]+ 625.1945, found 625.1948.
1). [α]D = 453 (c = 1.10 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (199 µg/4 ml): 399.5 (−6.64), 313 (−8.66), 282.5 (7.96), 251.5 (−9.34), 237 (2.85), 208.5 (−143.57), 198.5 (129.65). 1H NMR (400 MHz, CDCl3) δ 2.31 (s, 3H, 4″-H), 2.41 (s, 3H, 3″-H), 2.91 (s, 3H, 2″-H), 3.23 (d, J = 12.4 Hz, 1H, 6-Ha), 3.75 (d, J = 12.4 Hz, 1H, 6-Hb), 3.87 (s, 3H, 1″-H), 4.30 (s, 1H, 19b-H), 4.90 (s, 1H, 12b-H), 5.34 (s, 1H, 7-H), 6.00 (d, J = 9.1 Hz, 1H, 4-H), 6.90 (brs, 4H, 2′-H, 3′-H, 5′-H and 6′-H), 6.93–7.00 (m, 3H, 9-H, 12-H and 4′-H), 7.05 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.23 (s, 1H, 17-H), 7.47 (d, J = 2.4 Hz, 1H, 1-H), 7.55 (s, 1H, 14-H), 7.58 (dd, J = 9.1 and 2.4 Hz, 1H, 3-H). 13C NMR (90 MHz, CDCl3) δ 19.4 (C-4″), 20.6 (C-3″), 37.9 (C-2″), 38.5 (C-6a), 38.9 (C-19b), 53.7 (C-7), 56.1 (C-1″), 77.4 (C-7), 79.5 (C-12b), 98.8 (C-19c), 109.6 (C-4), 112.6 (C-13b), 115.2 (C-9), 117.4 (C-17), 117.8 (C-12), 118.6 (C-19a and C-12a), 118.7 (C-10), 123.0 (C-14), 124.4 (C-1 and C-3), 127.7–128.5 (Ph), 133.2 (C-15), 137.0 (C-16), 137.7 (C-1′), 143.1 (C-2), 149.1 (C-8a), 151.6 (C-17a), 152.3 (C-4a), 154.1 (C-11), 160.9 (C-13a), 161.4 (C-19). IR (KBr) ν: 1037, 1228, 1302, 1319, 1498, 1613, 1717, 2920 cm−1; HRMS: calcd for C36H30N2O7Na [M + Na]+ 625.1945, found 625.1948.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 19bR*)-4cb as white crystals (35%), rac-(6aS*, 12bR*, 19bR*)-6cb as pale yellow amorphous solid (19%) and rac-(6aS*, 12bR*, 19bS*)-5cb as yellow amorphous solid (21%).
1), affording rac-(6aS*, 12bR*, 19bR*)-4cb as white crystals (35%), rac-(6aS*, 12bR*, 19bR*)-6cb as pale yellow amorphous solid (19%) and rac-(6aS*, 12bR*, 19bS*)-5cb as yellow amorphous solid (21%).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.26 (s, 3H, 3′-H), 2.35 (s, 3H, 2′-H), 3.88 (s, 3H, 1′-H), 3.95 (dd, J = 11.7, 1.4 Hz, 1H, 7-Ha), 3.99 (d, J = 11.6 Hz, 1H, 7-Hb), 4.14 (d, J = 11.8 Hz, 1H, 6-Ha), 4.26 (s, 1H, 19b-H), 4.33 (d, J = 11.8 Hz, 1H, 6-Hb), 5.31 (s, 1H, 12b-H), 6.85 (d, J = 8.2 Hz, 1H, 4-H), 6.89–6.96 (m, 2H, 2-H and 9-H), 7.00 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.05 (d, J = 2.9 Hz, 1H, 12-H), 7.12–7.19 (m, 2H, 3-H and 17-H), 7.40 (s, 1H, 14-H), 7.53 (d, J = 7.9 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.2 (C-3′), 20.3 (C-2′), 30.5 (C-6a), 32.0 (C-19b), 55.9 (C-1′), 66.1 (C-7), 68.4 (C-6), 71.0 (C-12b), 100.2 (C-19a), 112.4 (C-13b), 116.0 (C-12), 116.9 (C-4), 117.1 (C-17), 117.9 (C-2), 118.0 (C-10), 118.3 (C-12a), 122.1 (C-9), 122.5 (C-19c), 123.0 (C-14), 128.3 (C-3), 129.9 (C-1), 132.9 (C-15), 142.4 (C-16), 146.7 (C-8a), 151.2 (C-17a), 152.3 (C-4a), 154.1 (C-11), 157.3 (C-13a), 164.6 (C-19). IR (KBr) ν: 1039, 1217, 1231, 1499, 1573, 1630, 1699, 2930 cm−1; HRMS: calcd for C29H24O6Na [M + Na]+ 491.1465, found 491.1465.
1). 1H NMR (400 MHz, CDCl3) δ 2.26 (s, 3H, 3′-H), 2.35 (s, 3H, 2′-H), 3.88 (s, 3H, 1′-H), 3.95 (dd, J = 11.7, 1.4 Hz, 1H, 7-Ha), 3.99 (d, J = 11.6 Hz, 1H, 7-Hb), 4.14 (d, J = 11.8 Hz, 1H, 6-Ha), 4.26 (s, 1H, 19b-H), 4.33 (d, J = 11.8 Hz, 1H, 6-Hb), 5.31 (s, 1H, 12b-H), 6.85 (d, J = 8.2 Hz, 1H, 4-H), 6.89–6.96 (m, 2H, 2-H and 9-H), 7.00 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.05 (d, J = 2.9 Hz, 1H, 12-H), 7.12–7.19 (m, 2H, 3-H and 17-H), 7.40 (s, 1H, 14-H), 7.53 (d, J = 7.9 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.2 (C-3′), 20.3 (C-2′), 30.5 (C-6a), 32.0 (C-19b), 55.9 (C-1′), 66.1 (C-7), 68.4 (C-6), 71.0 (C-12b), 100.2 (C-19a), 112.4 (C-13b), 116.0 (C-12), 116.9 (C-4), 117.1 (C-17), 117.9 (C-2), 118.0 (C-10), 118.3 (C-12a), 122.1 (C-9), 122.5 (C-19c), 123.0 (C-14), 128.3 (C-3), 129.9 (C-1), 132.9 (C-15), 142.4 (C-16), 146.7 (C-8a), 151.2 (C-17a), 152.3 (C-4a), 154.1 (C-11), 157.3 (C-13a), 164.6 (C-19). IR (KBr) ν: 1039, 1217, 1231, 1499, 1573, 1630, 1699, 2930 cm−1; HRMS: calcd for C29H24O6Na [M + Na]+ 491.1465, found 491.1465.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.35 (s, 3H, 3′-H), 2.37 (s, 3H, 2′-H), 3.81 (s, 3H, 1′-H), 3.93 (dd, J = 11.8 and 1.5 Hz, 1H, 7-Ha), 4.01 (d, J = 11.8 Hz, 1H, 7-Hb), 4.13 (d, J = 11.8 Hz, 1H, 6-Ha), 4.28 (d, J = 11.7 Hz, 1H, 6-Hb), 4.48 (s, 1H, 19b-H), 5.43 (s, 1H, 12b-H), 6.81 (dd, J = 8.1 and 1.0 Hz, 1H, 4-H), 6.85–6.91 (m, 2H, 2-H and 9-H), 6.91–7.01 (m, 2H, 10-H and 12-H), 7.07–7.17 (m, 2H, 3-H and 15-H), 7.41 (d, J = 7.9 Hz, 1H, 1-H), 8.00 (s, 1H, 18-H). 13C NMR (100 MHz, CDCl3) δ 19.4 (C-3′), 20.5 (C-2′), 31.0 (C-19b), 31.4 (C-6a), 55.9 (C-1′), 66.2 (C-7), 68.3 (C-6), 73.7 (C-12b), 97.4 (C-19a), 115.3 (C-10), 116.8 (C-4), 117.3 (C-18a), 117.5 (C-15), 118.3 (C-9), 119.1 (C-12), 120.5 (C-19c), 122.2 (C-2), 123.0 (C-12a), 125.8 (C-18), 128.4 (C-3), 130.5 (C-1), 134.6 (C-17), 143.8 (C-16), 146.9 (C-8a), 151.7 (C-14a), 152.3 (C-4a), 154.3 (C-11), 160.4 (C-13a), 178.7 (C-19). IR (KBr) ν: 1217, 1267, 1464, 1498, 1563, 1620, 2925 cm−1; HRMS: calcd for C29H24O6Na [M + Na]+ 491.1465, found 491.1466.
1). 1H NMR (400 MHz, CDCl3) δ 2.35 (s, 3H, 3′-H), 2.37 (s, 3H, 2′-H), 3.81 (s, 3H, 1′-H), 3.93 (dd, J = 11.8 and 1.5 Hz, 1H, 7-Ha), 4.01 (d, J = 11.8 Hz, 1H, 7-Hb), 4.13 (d, J = 11.8 Hz, 1H, 6-Ha), 4.28 (d, J = 11.7 Hz, 1H, 6-Hb), 4.48 (s, 1H, 19b-H), 5.43 (s, 1H, 12b-H), 6.81 (dd, J = 8.1 and 1.0 Hz, 1H, 4-H), 6.85–6.91 (m, 2H, 2-H and 9-H), 6.91–7.01 (m, 2H, 10-H and 12-H), 7.07–7.17 (m, 2H, 3-H and 15-H), 7.41 (d, J = 7.9 Hz, 1H, 1-H), 8.00 (s, 1H, 18-H). 13C NMR (100 MHz, CDCl3) δ 19.4 (C-3′), 20.5 (C-2′), 31.0 (C-19b), 31.4 (C-6a), 55.9 (C-1′), 66.2 (C-7), 68.3 (C-6), 73.7 (C-12b), 97.4 (C-19a), 115.3 (C-10), 116.8 (C-4), 117.3 (C-18a), 117.5 (C-15), 118.3 (C-9), 119.1 (C-12), 120.5 (C-19c), 122.2 (C-2), 123.0 (C-12a), 125.8 (C-18), 128.4 (C-3), 130.5 (C-1), 134.6 (C-17), 143.8 (C-16), 146.9 (C-8a), 151.7 (C-14a), 152.3 (C-4a), 154.3 (C-11), 160.4 (C-13a), 178.7 (C-19). IR (KBr) ν: 1217, 1267, 1464, 1498, 1563, 1620, 2925 cm−1; HRMS: calcd for C29H24O6Na [M + Na]+ 491.1465, found 491.1466.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.26 (s, 3H, 2′-H), 2.36 (s, 3H, 3′-H), 3.43 (dd, J = 11.6 and 1.9 Hz, 1H, 7-Ha), 3.84 (s, 3H, 1′-H), 3.84 (dd, J = 11.6 and 1.5 Hz, 1H, 7-Hb), 3.96 (dd, J = 10.4 and 1.8 Hz, 1H, 6-Ha), 4.23 (d, J = 10.4 Hz, 1H, 6-Hb), 4.38 (s, 1H, 19b-H), 4.87 (d, J = 1.8 Hz, 1H, 12b-H), 6.85 (d, J = 8.9 Hz, 1H, 9-H), 6.89–6.96 (m, 3H, 2-H, 4-H and 12-H), 6.98 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.06 (d, J = 7.6 Hz, 1H, 1-H), 7.15 (s, 1H, 17-H), 7.20 (t, J = 7.7 Hz, 1H, 3-H), 7.46 (s, 1H, 14-H). 13C NMR (100 MHz, CDCl3) δ 19.3 (C-2′), 20.5 (C-3′), 35.5 (C-6a), 36.4 (C-19b), 56.0 (C-1′), 63.4 (C-7), 69.5 (C-6), 75.5 (C-12b), 98.4 (C-19a), 112.7 (C-13b), 115.4 (C-12), 116.7 (C-4), 117.2 (C-17), 117.5 (C-12a), 117.8 (C-9), 118.5 (C-10), 121.2 (C-2), 123.0 (C-14), 123.0 (C-19c), 126.4 (C-1), 128.4 (C-3), 133.0 (C-15), 142.6 (C-16), 148.9 (C-8a), 151.4 (C-17a), 153.7 (C-11), 154.8 (C-4a), 159.9 (C-13a), 162.0 (C-19). IR (KBr) ν: 1044, 1220, 1268, 1294, 1391, 1455, 1499, 1625, 1712, 2942 cm−1; HRMS: calcd for C29H24O6Na [M + Na]+ 491.1465, found 491.1465.
1). 1H NMR (400 MHz, CDCl3) δ 2.26 (s, 3H, 2′-H), 2.36 (s, 3H, 3′-H), 3.43 (dd, J = 11.6 and 1.9 Hz, 1H, 7-Ha), 3.84 (s, 3H, 1′-H), 3.84 (dd, J = 11.6 and 1.5 Hz, 1H, 7-Hb), 3.96 (dd, J = 10.4 and 1.8 Hz, 1H, 6-Ha), 4.23 (d, J = 10.4 Hz, 1H, 6-Hb), 4.38 (s, 1H, 19b-H), 4.87 (d, J = 1.8 Hz, 1H, 12b-H), 6.85 (d, J = 8.9 Hz, 1H, 9-H), 6.89–6.96 (m, 3H, 2-H, 4-H and 12-H), 6.98 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.06 (d, J = 7.6 Hz, 1H, 1-H), 7.15 (s, 1H, 17-H), 7.20 (t, J = 7.7 Hz, 1H, 3-H), 7.46 (s, 1H, 14-H). 13C NMR (100 MHz, CDCl3) δ 19.3 (C-2′), 20.5 (C-3′), 35.5 (C-6a), 36.4 (C-19b), 56.0 (C-1′), 63.4 (C-7), 69.5 (C-6), 75.5 (C-12b), 98.4 (C-19a), 112.7 (C-13b), 115.4 (C-12), 116.7 (C-4), 117.2 (C-17), 117.5 (C-12a), 117.8 (C-9), 118.5 (C-10), 121.2 (C-2), 123.0 (C-14), 123.0 (C-19c), 126.4 (C-1), 128.4 (C-3), 133.0 (C-15), 142.6 (C-16), 148.9 (C-8a), 151.4 (C-17a), 153.7 (C-11), 154.8 (C-4a), 159.9 (C-13a), 162.0 (C-19). IR (KBr) ν: 1044, 1220, 1268, 1294, 1391, 1455, 1499, 1625, 1712, 2942 cm−1; HRMS: calcd for C29H24O6Na [M + Na]+ 491.1465, found 491.1465.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 19bS*)-4ac as yellow powder (49%), rac-(6aS*, 12bR*, 19bS*)-6ac as yellow amorphous solid (16%) and rac-(6aS*, 12bR*, 19bR*)-5ac as yellow powder (25%).
1), affording rac-(6aS*, 12bR*, 19bS*)-4ac as yellow powder (49%), rac-(6aS*, 12bR*, 19bS*)-6ac as yellow amorphous solid (16%) and rac-(6aS*, 12bR*, 19bR*)-5ac as yellow powder (25%).Alternatively, the reaction was carried out at room temperature, affording rac-(6aS*, 12bR*, 19bS*)-4ac as yellow powder (21%), rac-(6aS*, 12bR*, 19bS*)-6ac as yellow amorphous solid (10%) and rac-(6aS*, 12bR*, 19bR*)-5ac as yellow powder (14%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.96 (s, 3H, 5′-H), 3.29 (d, J = 13.6 Hz, 1H, 6-Ha), 3.50 (s, 3H, 2′-H), 3.69 (d, J = 13.2 Hz, 1H, 6-Hb), 3.78 (s, 3H, 3′-H), 3.80 (s, 3H, 1′-H), 3.92 (m, 4H, 4′-H and 7-Ha), 4.01 (d, J = 11.2 Hz, 1H, 7-Hb), 4.23 (s, 1H, 19b-H), 5.05 (s, 1H, 12b-H), 6.53 (d, J = 8.8 Hz, 1H, 4-H), 6.70 (s, 1H, 17-H), 6.86 (d, J = 8.8 Hz, 1H, 9-H), 6.94 (d, J = 9.2 Hz, 1H, 10-H), 6.99 (s, 1H, 12-H), 7.97–7.93 (m, 2H, 1-H and 3-H). 13C NMR (100 MHz, CDCl3) δ 30.1 (C-6a), 32.8 (C-19b), 39.6 (C-5′), 54.4 (C-6), 54.4 (C-4′), 56.3 (C-1′), 61.2 (C-3′), 62.1 (C-2′), 67.6 (C-7), 72.0 (C-12b), 96.6 (C-17), 98.1 (C-19a), 103.2 (C-13b), 110.0 (C-4), 115.2 (C-12), 118.1 (C-10), 118.2 (C-12a), 118.3 (C-9), 121.2 (C-19c), 138.1 (C-2), 140.3 (C-15), 146.6 (C-8a), 148.8 (C-4a), 150.6 (C-14), 150.8 (C-17a), 154.0 (C-11), 157.0 (C-16), 158.8 (C-13a), 163.6 (C-19). IR (KBr) ν: 1042, 1100, 1285, 1299, 1317, 1391, 1498, 1607, 1703 cm−1; HRMS: calcd for C31H28N2O10Na [M + Na]+ 611.1636, found 611.1637.
1). 1H NMR (400 MHz, CDCl3) δ 2.96 (s, 3H, 5′-H), 3.29 (d, J = 13.6 Hz, 1H, 6-Ha), 3.50 (s, 3H, 2′-H), 3.69 (d, J = 13.2 Hz, 1H, 6-Hb), 3.78 (s, 3H, 3′-H), 3.80 (s, 3H, 1′-H), 3.92 (m, 4H, 4′-H and 7-Ha), 4.01 (d, J = 11.2 Hz, 1H, 7-Hb), 4.23 (s, 1H, 19b-H), 5.05 (s, 1H, 12b-H), 6.53 (d, J = 8.8 Hz, 1H, 4-H), 6.70 (s, 1H, 17-H), 6.86 (d, J = 8.8 Hz, 1H, 9-H), 6.94 (d, J = 9.2 Hz, 1H, 10-H), 6.99 (s, 1H, 12-H), 7.97–7.93 (m, 2H, 1-H and 3-H). 13C NMR (100 MHz, CDCl3) δ 30.1 (C-6a), 32.8 (C-19b), 39.6 (C-5′), 54.4 (C-6), 54.4 (C-4′), 56.3 (C-1′), 61.2 (C-3′), 62.1 (C-2′), 67.6 (C-7), 72.0 (C-12b), 96.6 (C-17), 98.1 (C-19a), 103.2 (C-13b), 110.0 (C-4), 115.2 (C-12), 118.1 (C-10), 118.2 (C-12a), 118.3 (C-9), 121.2 (C-19c), 138.1 (C-2), 140.3 (C-15), 146.6 (C-8a), 148.8 (C-4a), 150.6 (C-14), 150.8 (C-17a), 154.0 (C-11), 157.0 (C-16), 158.8 (C-13a), 163.6 (C-19). IR (KBr) ν: 1042, 1100, 1285, 1299, 1317, 1391, 1498, 1607, 1703 cm−1; HRMS: calcd for C31H28N2O10Na [M + Na]+ 611.1636, found 611.1637.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), and the on-line HPLC-ECD spectra were recorded.
5), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS, 19bR)-4ac: tR = 8.00 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 389 (2.05), 335sh (−6.63), 318 (−14.22), 298sh (3.92), 289.5 (4.10), 236 (−26.08).
5]: 389 (2.05), 335sh (−6.63), 318 (−14.22), 298sh (3.92), 289.5 (4.10), 236 (−26.08).
(6aS, 12bR, 19bS)-4ac: tR = 11.35 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 388.5 (−1.82), 335sh (4.38), 318 (9.87), 297sh (−3.65), 285.5 (−3.73), 236 (18.61).
5]: 388.5 (−1.82), 335sh (4.38), 318 (9.87), 297sh (−3.65), 285.5 (−3.73), 236 (18.61).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.95 (s, 3H, 5′-H), 3.24 (d, J = 13.2 Hz, 1H, 6-Ha), 3.71 (d, J = 13.2 Hz, 1H, 6-Hb), 3.80 (s, 3H, 1′-H), 3.89 (s, 3H, 2′-H), 3.90–3.98 (m, 4H, 3′-H and 7-Ha), 4.01 (dd, J = 11.7 and 2.0 Hz, 1H, 7-Hb), 4.08 (s, 3H, 4′-H), 4.49 (s, 1H, 19b-H), 5.14 (d, J = 2.0 Hz, 1H, 12b-H), 6.55 (d, J = 9.3 Hz, 1H, 4-H), 6.61 (s, 1H, 15-H), 6.85–6.93 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 9.1 and 3.0 Hz, 1H, 10-H), 7.98 (dd, J = 9.3 and 2.7 Hz, 1H, 3-H), 8.08 (d, J = 2.7 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 31.0 (C-6a), 32.0 (C-19b), 39.7 (C-5′), 54.5 (C-6), 55.8 (C-1′), 56.3 (C-2′), 61.6 (C-3′), 62.5 (C-4′), 67.5 (C-7), 74.6 (C-12b), 95.9 (C-15), 97.1 (C-19a), 110.1 (C-4), 111.1 (C-18a), 115.0 (C-12), 117.4 (C-12a), 118.3 (C-9), 118.8 (C-10), 121.9 (C-19c), 124.6 (C-1), 126.5 (C-3), 138.5 (C-2), 140.8 (C-17), 146.8 (C-8a), 148.8 (C-4a), 151.3 (C-14a), 153.0 (C-18), 154.2 (C-11), 157.5 (C-16), 158.8 (C-13a), 177.0 (C-19). IR (KBr) ν: 1217, 1265, 1421, 1498, 1604, 2852, 2925 cm−1. HRMS: calcd for C31H28N2O10Na [M + Na]+ 611.1636, found 611.1639.
1). 1H NMR (400 MHz, CDCl3) δ 2.95 (s, 3H, 5′-H), 3.24 (d, J = 13.2 Hz, 1H, 6-Ha), 3.71 (d, J = 13.2 Hz, 1H, 6-Hb), 3.80 (s, 3H, 1′-H), 3.89 (s, 3H, 2′-H), 3.90–3.98 (m, 4H, 3′-H and 7-Ha), 4.01 (dd, J = 11.7 and 2.0 Hz, 1H, 7-Hb), 4.08 (s, 3H, 4′-H), 4.49 (s, 1H, 19b-H), 5.14 (d, J = 2.0 Hz, 1H, 12b-H), 6.55 (d, J = 9.3 Hz, 1H, 4-H), 6.61 (s, 1H, 15-H), 6.85–6.93 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 9.1 and 3.0 Hz, 1H, 10-H), 7.98 (dd, J = 9.3 and 2.7 Hz, 1H, 3-H), 8.08 (d, J = 2.7 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 31.0 (C-6a), 32.0 (C-19b), 39.7 (C-5′), 54.5 (C-6), 55.8 (C-1′), 56.3 (C-2′), 61.6 (C-3′), 62.5 (C-4′), 67.5 (C-7), 74.6 (C-12b), 95.9 (C-15), 97.1 (C-19a), 110.1 (C-4), 111.1 (C-18a), 115.0 (C-12), 117.4 (C-12a), 118.3 (C-9), 118.8 (C-10), 121.9 (C-19c), 124.6 (C-1), 126.5 (C-3), 138.5 (C-2), 140.8 (C-17), 146.8 (C-8a), 148.8 (C-4a), 151.3 (C-14a), 153.0 (C-18), 154.2 (C-11), 157.5 (C-16), 158.8 (C-13a), 177.0 (C-19). IR (KBr) ν: 1217, 1265, 1421, 1498, 1604, 2852, 2925 cm−1. HRMS: calcd for C31H28N2O10Na [M + Na]+ 611.1636, found 611.1639.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), and the on-line HPLC-ECD spectra were recorded.
5), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS, 19bR)-6ac: tR = 8.44 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 365.5 (−1.80), 322.5 (0.93), 299.5 (−10.31), 281 (2.24), 251.5 (−20.78), 232.5 (−2.41).
5]: 365.5 (−1.80), 322.5 (0.93), 299.5 (−10.31), 281 (2.24), 251.5 (−20.78), 232.5 (−2.41).
(6aS, 12bR, 19bS)-6ac: tR = 15.56 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 365.5 (1.03), 321.5 (−0.36), 300 (4.28), 281.5 (−1.79), 249.5 (9.07).
5]: 365.5 (1.03), 321.5 (−0.36), 300 (4.28), 281.5 (−1.79), 249.5 (9.07).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 3.05 (s, 3H, 5′-H), 3.26 (m, 2H, 6-H), 3.35 (dd, J = 11.8 and 2.1 Hz, 1H, 7-Ha), 3.63 (s, 3H, 2′-H), 3.81 (s, 3H, 1′-H), 3.83 (s, 3H, 3′-H), 3.89 (d, J = 11.8 Hz, 1H, 7-Hb), 3.94 (s, 3H, 4′-H), 4.13 (s, 1H, 19b-H), 4.86 (d, J = 2.1 Hz, 1H, 12b-H), 6.66 (d, J = 9.2 Hz, 1H, 4-H), 6.73 (s, 1H, 17-H), 6.84 (d, J = 8.9 Hz, 1H, 9-H), 6.91 (d, J = 3.0 Hz, 1H, 12-H), 6.97 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.73 (d, J = 2.6 Hz, 1H, 1-H), 8.12 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 34.9 (C-6a), 36.9 (C-19b), 38.2 (C-5′), 54.4 (C-6), 55.9 (C-1′), 56.4 (C-4′), 61.4 (C-3′), 62.2 (C-2′), 63.1 (C-7), 76.0 (C-12b), 96.4 (C-19a), 96.5 (C-17), 103.4 (C-13b), 109.7 (C-4), 115.0 (C-12), 117.4 (C-12a), 117.7 (C-9), 118.3 (C-10), 119.6 (C-19c), 122.1 (C-1), 124.9 (C-3), 138.0 (C-2), 140.4 (C-15), 148.4 (C-8a), 150.6 (C-14), 150.8 (C-17a), 151.7 (C-4a), 153.7 (C-11), 157.1 (C-16), 161.2 (C-19), 161.7 (C-13a). IR (KBr) ν: 1290, 1313, 1498, 1607, 1707, 2853, 2926 cm−1. HRMS: calcd for C31H28N2O10Na [M + Na]+ 611.1636, found 611.1639.
1). 1H NMR (400 MHz, CDCl3) δ 3.05 (s, 3H, 5′-H), 3.26 (m, 2H, 6-H), 3.35 (dd, J = 11.8 and 2.1 Hz, 1H, 7-Ha), 3.63 (s, 3H, 2′-H), 3.81 (s, 3H, 1′-H), 3.83 (s, 3H, 3′-H), 3.89 (d, J = 11.8 Hz, 1H, 7-Hb), 3.94 (s, 3H, 4′-H), 4.13 (s, 1H, 19b-H), 4.86 (d, J = 2.1 Hz, 1H, 12b-H), 6.66 (d, J = 9.2 Hz, 1H, 4-H), 6.73 (s, 1H, 17-H), 6.84 (d, J = 8.9 Hz, 1H, 9-H), 6.91 (d, J = 3.0 Hz, 1H, 12-H), 6.97 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.73 (d, J = 2.6 Hz, 1H, 1-H), 8.12 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 34.9 (C-6a), 36.9 (C-19b), 38.2 (C-5′), 54.4 (C-6), 55.9 (C-1′), 56.4 (C-4′), 61.4 (C-3′), 62.2 (C-2′), 63.1 (C-7), 76.0 (C-12b), 96.4 (C-19a), 96.5 (C-17), 103.4 (C-13b), 109.7 (C-4), 115.0 (C-12), 117.4 (C-12a), 117.7 (C-9), 118.3 (C-10), 119.6 (C-19c), 122.1 (C-1), 124.9 (C-3), 138.0 (C-2), 140.4 (C-15), 148.4 (C-8a), 150.6 (C-14), 150.8 (C-17a), 151.7 (C-4a), 153.7 (C-11), 157.1 (C-16), 161.2 (C-19), 161.7 (C-13a). IR (KBr) ν: 1290, 1313, 1498, 1607, 1707, 2853, 2926 cm−1. HRMS: calcd for C31H28N2O10Na [M + Na]+ 611.1636, found 611.1639.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and the on-line HPLC-ECD spectra were recorded.
7), and the on-line HPLC-ECD spectra were recorded.
(6aS, 12bR, 19bR)-5ac: tR = 5.99 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 387 (−13.22), 314 (−18.67), 288.5 (6.43), 264.5sh (3.90).
7]: 387 (−13.22), 314 (−18.67), 288.5 (6.43), 264.5sh (3.90).
(6aR, 12bS, 19bS)-5ac: tR = 14.05 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 387.5 (6.79), 315 (9.23), 287.5 (−3.37), 265sh (−2.31).
7]: 387.5 (6.79), 315 (9.23), 287.5 (−3.37), 265sh (−2.31).
(6aS, 7R, 12bR, 19bS)- and (6aS, 7R, 12bR, 19bR)-11,14,15,16-tetramethoxy-5-methyl-2-nitro-7-phenyl-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,4':5,6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [(6aS, 7R, 12bR, 19bS)-4bc and (6aS, 7R, 12bR, 19bR)-5bc] and (6aS, 7R, 12bR, 19bS)-11,16,17,18-tetramethoxy-5-methyl-2-nitro-7-phenyl-5,19b-dihydro-6H,12bH,19H-dichromeno[3′,2′:5,6; 4″,3″:2,3]pyrano[3,4-c]quinolin-19-one [(6aS, 7R, 12bR, 19bS)-6bc]
The reaction of (R)-1b and 2c was carried out according to method A, and the crude product was purified by column chromatography (hexane/chloroform/ethyl acetate 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording (6aS, 7R, 12bR, 19bS)-4bc as yellow powder (42%), (6aS, 7R, 12bR, 19bS)-6bc as yellow amorphous solid (10%) and (6aS, 7R, 12bR, 19bR6aS, 7R, 12bR, 19bR)-5bc as yellow crystals (44%).
1), affording (6aS, 7R, 12bR, 19bS)-4bc as yellow powder (42%), (6aS, 7R, 12bR, 19bS)-6bc as yellow amorphous solid (10%) and (6aS, 7R, 12bR, 19bR6aS, 7R, 12bR, 19bR)-5bc as yellow crystals (44%).
Alternatively, the reaction was carried out at room temperature, affording (6aS, 7R, 12bR, 19bS)-4bc as yellow powder (32%), (6aS, 7R, 12bR, 19bS)-6bc as yellow amorphous solid (8%) and (6aS, 7R, 12bR, 19bR)-5bc as yellow crystals (53%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = 0.2 (c = 0.88 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (218 µg/4 ml): 319 (9.42), 296.5 (−5.63), 235 (19.47), 219 (−40.95), 199.5 (92.52). 1H NMR (400 MHz, CDCl3) δ 2.93 (s, 3H, 5″-H), 3.25 (d, J = 13.0 Hz, 1H, 6-Ha), 3.54 (m, 4H, 3″-H and 6-Hb), 3.81 (s, 3H, 2″-H), 3.84 (s, 3H, 1″-H), 3.95 (s, 3H, 4″-H), 4.04 (s, 1H, 19b-H), 5.07 (s, 1H, 7-H), 5.15 (s, 1H, 12b-H), 6.52 (d, J = 9.2 Hz, 1H, 4-H), 6.77 (s, 1H, 17-H), 6.99 (s, 2H, 9-H and 10-H), 7.05 (s, 1H, 12-H), 7.31 (d, J = 6.9 Hz, 2H, 2′-H and 6′-H), 7.35–7.46 (m, 3H, 3′-H, 4′-H and 5′-H), 7.96 (dd, J = 9.2 and 2.7 Hz, 1H, 3-H), 8.05 (s, 1H, 1-H). 13C-NMR (100 MHz, CDCl3): 32.4 (C-19b), 32.7 (C-6a), 39.8 (C-5″), 50.2 (C-6), 55.9 (C-1″), 56.4 (C-3″), 61.3 (C-2″), 62.2 (C-3″), 73.0 (C-12b), 77.2 (C-7), 96.8 (C-17), 98.5 (C-13b), 103.2 (C-19a), 110.0 (C-19c), 110.1 (C-4), 115.1 (C-12), 118.2 (C-9), 118.3 (C-10), 118.4 (C-12a), 122.4 (C-1′), 124.5 (C-3), 126.1 (C-1), 127.5 (C-3′ and C-5′), 128.5 (C-2′ and C-6′), 129.2 (C-4′), 133.9 (C-15), 138.4 (C-2), 147.7 (C-14), 148.4 (C-17a), 150.8 (C-8a), 151.2 (C-4a), 154.3 (C-11), 157.2 (C-16), 159.5 (C-13a), 163.1 (C-19). IR (KBr) ν: 1036, 1098, 1233, 1300, 1314, 1393, 1460, 1498, 1607, 1705 cm−1; HRMS: calcd for C37H32N2O10Na [M + Na]+ 687.1949, found 687.1952.
1). [α]D = 0.2 (c = 0.88 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (218 µg/4 ml): 319 (9.42), 296.5 (−5.63), 235 (19.47), 219 (−40.95), 199.5 (92.52). 1H NMR (400 MHz, CDCl3) δ 2.93 (s, 3H, 5″-H), 3.25 (d, J = 13.0 Hz, 1H, 6-Ha), 3.54 (m, 4H, 3″-H and 6-Hb), 3.81 (s, 3H, 2″-H), 3.84 (s, 3H, 1″-H), 3.95 (s, 3H, 4″-H), 4.04 (s, 1H, 19b-H), 5.07 (s, 1H, 7-H), 5.15 (s, 1H, 12b-H), 6.52 (d, J = 9.2 Hz, 1H, 4-H), 6.77 (s, 1H, 17-H), 6.99 (s, 2H, 9-H and 10-H), 7.05 (s, 1H, 12-H), 7.31 (d, J = 6.9 Hz, 2H, 2′-H and 6′-H), 7.35–7.46 (m, 3H, 3′-H, 4′-H and 5′-H), 7.96 (dd, J = 9.2 and 2.7 Hz, 1H, 3-H), 8.05 (s, 1H, 1-H). 13C-NMR (100 MHz, CDCl3): 32.4 (C-19b), 32.7 (C-6a), 39.8 (C-5″), 50.2 (C-6), 55.9 (C-1″), 56.4 (C-3″), 61.3 (C-2″), 62.2 (C-3″), 73.0 (C-12b), 77.2 (C-7), 96.8 (C-17), 98.5 (C-13b), 103.2 (C-19a), 110.0 (C-19c), 110.1 (C-4), 115.1 (C-12), 118.2 (C-9), 118.3 (C-10), 118.4 (C-12a), 122.4 (C-1′), 124.5 (C-3), 126.1 (C-1), 127.5 (C-3′ and C-5′), 128.5 (C-2′ and C-6′), 129.2 (C-4′), 133.9 (C-15), 138.4 (C-2), 147.7 (C-14), 148.4 (C-17a), 150.8 (C-8a), 151.2 (C-4a), 154.3 (C-11), 157.2 (C-16), 159.5 (C-13a), 163.1 (C-19). IR (KBr) ν: 1036, 1098, 1233, 1300, 1314, 1393, 1460, 1498, 1607, 1705 cm−1; HRMS: calcd for C37H32N2O10Na [M + Na]+ 687.1949, found 687.1952.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = 45 (c = 1.00 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (197 µg/4 ml): 389.5 (3.53), 319.5 (−0.94), 298.5 (8.06), 281 (−0.32), 248 (15.29), 235sh (8.35), 212 (−20.71), 201.5 (18.73), 191.5 (−123.48). 1H NMR (400 MHz, CDCl3) δ 2.90 (s, 3H, 5″-H), 3.20 (d, J = 12.9 Hz, 1H, 6-Ha), 3.50 (d, J = 12.9 Hz, 1H, 6-Hb), 3.83 (s, 3H, 1″-H), 3.92 (s, 3H, 2″-H), 3.98 (s, 3H, 3″-H), 4.12 (s, 3H, 4″-H), 4.37 (s, 1H, 19b-H), 5.14 (s, 1H, 7-H), 5.21 (s, 1H, 12b-H), 6.51 (d, J = 9.2 Hz, 1H, 4-H), 6.66 (s, 1H, 15-H), 6.93 (m, 1H, 12-H), 7.02 (m, 2H, 9-H and 10-H), 7.30 (m, 2H, 2′-H and 6′-H), 7.37 (m, 1H, 4′-H), 7.42 (m, 2H, 3′-H and 5′-H), 7.94 (dd, J = 9.2 and 2.6 Hz, 1H, 3-H), 8.16 (dd, J = 2.6 and 1.3 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 31.5 (C-19b), 33.1 (C-6a), 39.8 (C-5″), 50.2 (C-6), 55.8 (C-1″), 56.3 (C-2″), 61.8 (C-3″), 62.7 (C-4″), 75.4 (C-12b), 76.8 (C-7), 96.0 (C-15), 97.6 (C-19c), 110.2 (C-4), 111.3 (C-18a), 114.9 (C-12), 117.4 (C-12a), 118.3 (C-9), 118.8 (C-10), 122.9 (C-19c), 124.2 (C-1), 126.7 (C-3), 127.5 (C-2′ and C-6′), 128.5 (C-3′ and C-5′), 129.1 (C-4′), 133.8 (C-1′), 138.5 (C-2), 140.9 (C-17), 147.8 (C-8a), 148.2 (C-4a), 151.4 (C-14a), 153.1 (C-18), 154.3 (C-11), 157.5 (C-16), 159.3 (C-13a), 176.5 (C-19). IR (KBr) ν: 1234, 1264, 1313, 1421, 1498, 1604, 1628, 2926 cm−1; HRMS: calcd for C37H32N2O10Na [M + Na]+ 687.1949, found 687.1951.
1). [α]D = 45 (c = 1.00 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (197 µg/4 ml): 389.5 (3.53), 319.5 (−0.94), 298.5 (8.06), 281 (−0.32), 248 (15.29), 235sh (8.35), 212 (−20.71), 201.5 (18.73), 191.5 (−123.48). 1H NMR (400 MHz, CDCl3) δ 2.90 (s, 3H, 5″-H), 3.20 (d, J = 12.9 Hz, 1H, 6-Ha), 3.50 (d, J = 12.9 Hz, 1H, 6-Hb), 3.83 (s, 3H, 1″-H), 3.92 (s, 3H, 2″-H), 3.98 (s, 3H, 3″-H), 4.12 (s, 3H, 4″-H), 4.37 (s, 1H, 19b-H), 5.14 (s, 1H, 7-H), 5.21 (s, 1H, 12b-H), 6.51 (d, J = 9.2 Hz, 1H, 4-H), 6.66 (s, 1H, 15-H), 6.93 (m, 1H, 12-H), 7.02 (m, 2H, 9-H and 10-H), 7.30 (m, 2H, 2′-H and 6′-H), 7.37 (m, 1H, 4′-H), 7.42 (m, 2H, 3′-H and 5′-H), 7.94 (dd, J = 9.2 and 2.6 Hz, 1H, 3-H), 8.16 (dd, J = 2.6 and 1.3 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 31.5 (C-19b), 33.1 (C-6a), 39.8 (C-5″), 50.2 (C-6), 55.8 (C-1″), 56.3 (C-2″), 61.8 (C-3″), 62.7 (C-4″), 75.4 (C-12b), 76.8 (C-7), 96.0 (C-15), 97.6 (C-19c), 110.2 (C-4), 111.3 (C-18a), 114.9 (C-12), 117.4 (C-12a), 118.3 (C-9), 118.8 (C-10), 122.9 (C-19c), 124.2 (C-1), 126.7 (C-3), 127.5 (C-2′ and C-6′), 128.5 (C-3′ and C-5′), 129.1 (C-4′), 133.8 (C-1′), 138.5 (C-2), 140.9 (C-17), 147.8 (C-8a), 148.2 (C-4a), 151.4 (C-14a), 153.1 (C-18), 154.3 (C-11), 157.5 (C-16), 159.3 (C-13a), 176.5 (C-19). IR (KBr) ν: 1234, 1264, 1313, 1421, 1498, 1604, 1628, 2926 cm−1; HRMS: calcd for C37H32N2O10Na [M + Na]+ 687.1949, found 687.1951.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = −424 (c = 0.82 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (212 µg/4 ml): 397.5 (−8.02), 315.5 (−10.95), 283.5 (10.44), 248.5 (−14.47), 210.5 (−121.64), 199.5 (70.97). 1H NMR (400 MHz, CDCl3) δ 2.91 (s, 3H, 5″-H), 3.28 (d, J = 12.4 Hz, 1H, 6-Ha), 3.70 (s, 3H, 2″-H), 3.78 (d, J = 12.5 Hz, 1H, 6-Hb), 3.84 (s, 3H, 4″-H), 3.88 (s, 3H, 3″-H), 3.98 (s, 3H, 1″-H), 4.29 (s, 1H, 19b-H), 4.91 (s, 1H, 12b-H), 5.41 (s, 1H, 7-H), 5.96 (d, J = 9.2 Hz, 1H, 4-H), 6.77 (s, 1H, 17-H), 6.81–6.99 (m, 7H, Ph, 9-H and 12-H), 7.03 (dd, J = 8.9 and 2.9 Hz, 1H, 10-H), 7.46 (d, J = 2.3 Hz, 1H, 1-H), 7.58 (dd, J = 9.1 and 2.3 Hz, 1H, 3-H). 13C-NMR (100 MHz, CDCl3): 37.7 (C-6a), 37.8 (C-5″), 38.7 (C-19b), 53.5 (C-6), 55.9 (C-4″), 56.4 (C-1″), 61.3 (C-3″), 62.2 (C-2″), 77.3 (C-7), 79.3 (C-12b), 96.5 (C-17), 97.2 (C-19a), 103.4 (C-13b), 109.4 (C-4), 114.6 (C-12), 117.3 (C-12a) 117.7 (C-9), 118.3 (C-10), 118.5 (C-19c), 124.0 (C-1), 124.3 (C-3), 128.3 (C-2′, C-3′, C-4′, C-5′ and C-6′), 136.9 (C-2), 137.3 (C-15), 140.4 (C-1′), 148.8 (C-8a), 150.5 (C-4a), 150.7 (C-14), 152.2 (C-17a), 153.8 (C-11), 157.2 (C-16), 160.8 (C-13a), 162.2 (C-19). IR (KBr) ν: 1231, 1302, 1389, 1497, 1608, 1712 cm−1; HRMS: calcd for C37H32N2O10Na [M + Na]+ 687.1949, found 687.1949.
1). [α]D = −424 (c = 0.82 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (212 µg/4 ml): 397.5 (−8.02), 315.5 (−10.95), 283.5 (10.44), 248.5 (−14.47), 210.5 (−121.64), 199.5 (70.97). 1H NMR (400 MHz, CDCl3) δ 2.91 (s, 3H, 5″-H), 3.28 (d, J = 12.4 Hz, 1H, 6-Ha), 3.70 (s, 3H, 2″-H), 3.78 (d, J = 12.5 Hz, 1H, 6-Hb), 3.84 (s, 3H, 4″-H), 3.88 (s, 3H, 3″-H), 3.98 (s, 3H, 1″-H), 4.29 (s, 1H, 19b-H), 4.91 (s, 1H, 12b-H), 5.41 (s, 1H, 7-H), 5.96 (d, J = 9.2 Hz, 1H, 4-H), 6.77 (s, 1H, 17-H), 6.81–6.99 (m, 7H, Ph, 9-H and 12-H), 7.03 (dd, J = 8.9 and 2.9 Hz, 1H, 10-H), 7.46 (d, J = 2.3 Hz, 1H, 1-H), 7.58 (dd, J = 9.1 and 2.3 Hz, 1H, 3-H). 13C-NMR (100 MHz, CDCl3): 37.7 (C-6a), 37.8 (C-5″), 38.7 (C-19b), 53.5 (C-6), 55.9 (C-4″), 56.4 (C-1″), 61.3 (C-3″), 62.2 (C-2″), 77.3 (C-7), 79.3 (C-12b), 96.5 (C-17), 97.2 (C-19a), 103.4 (C-13b), 109.4 (C-4), 114.6 (C-12), 117.3 (C-12a) 117.7 (C-9), 118.3 (C-10), 118.5 (C-19c), 124.0 (C-1), 124.3 (C-3), 128.3 (C-2′, C-3′, C-4′, C-5′ and C-6′), 136.9 (C-2), 137.3 (C-15), 140.4 (C-1′), 148.8 (C-8a), 150.5 (C-4a), 150.7 (C-14), 152.2 (C-17a), 153.8 (C-11), 157.2 (C-16), 160.8 (C-13a), 162.2 (C-19). IR (KBr) ν: 1231, 1302, 1389, 1497, 1608, 1712 cm−1; HRMS: calcd for C37H32N2O10Na [M + Na]+ 687.1949, found 687.1949.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 19bR*)-4cc as pale yellow powder (34%), rac-(6aS*, 12bR*, 19bR*)-6cc as pale yellow powder (12%) and rac-(6aS*, 12bR*, 19bS*)-5cc as pale yellow powder (14%).
1), affording rac-(6aS*, 12bR*, 19bR*)-4cc as pale yellow powder (34%), rac-(6aS*, 12bR*, 19bR*)-6cc as pale yellow powder (12%) and rac-(6aS*, 12bR*, 19bS*)-5cc as pale yellow powder (14%).Alternatively, the reaction was carried out at 55 °C (there is no reaction at room temperature), affording rac-(6aS*, 12bR*, 19bR*)-4cc as pale yellow powder (29%), rac-(6aS*, 12bR*, 19bR*)-6cc as pale yellow powder (8%) and rac-(6 aS*,12bR*,19bS*)-5cc as pale yellow powder (13%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 3.53 (s, 3H, 2′-H), 3.77 (s, 3H, 3′-H), 3.80 (s, 3H, 1′-H), 3.89 (s, 3H, 4′-H), 3.95 (m, 2H, 7-H), 4.11 (d, J = 11.8 Hz, 1H, 6-Ha), 4.22 (s, 1H, 19b-H), 4.31 (d, J = 11.8 Hz, 1H, 6-Hb), 5.30 (s, 1H, 12b-H), 6.67 (s, 1H, 17-H), 6.82 (m, 1H, 4-H), 6.88 (m, 2H, 2-H and 9-H), 6.93 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.03 (d, J = 2.9 Hz, 1H, 12-H), 7.12 (t, J = 7.8 Hz, 1H, 3-H), 7.39 (d, J = 8.0 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 30.0 (C-6a), 31.8 (C-19b), 55.8 (C-1′), 56.3 (C-4′), 61.2 (C-3′), 62.3 (C-2′), 66.2 (C-7), 68.3 (C-6), 70.9 (C-12b), 96.4 (C-17), 98.6 (C-13b), 103.3 (C-19a), 115.3 (C-12), 116.9 (C-4), 118.1 (C-9), 118.1 (C-12a), 118.5 (C-10), 122.0 (C-2), 122.4 (C-19c), 128.3 (C-3), 129.8 (C-1), 140.3 (C-15), 146.6 (C-8a), 150.7 (C-17a), 150.7 (C-14), 152.3 (C-4a), 154.0 (C-11), 156.9 (C-16), 158.7 (C-13a), 164.1 (C-19). IR (KBr) ν: 1040, 1102, 1215, 1282, 1398, 1463, 1498, 1605, 1697 cm−1; HRMS: calcd for C30H26O9Na [M + Na]+ 533.1469, found 533.1468.
1). 1H NMR (400 MHz, CDCl3) δ 3.53 (s, 3H, 2′-H), 3.77 (s, 3H, 3′-H), 3.80 (s, 3H, 1′-H), 3.89 (s, 3H, 4′-H), 3.95 (m, 2H, 7-H), 4.11 (d, J = 11.8 Hz, 1H, 6-Ha), 4.22 (s, 1H, 19b-H), 4.31 (d, J = 11.8 Hz, 1H, 6-Hb), 5.30 (s, 1H, 12b-H), 6.67 (s, 1H, 17-H), 6.82 (m, 1H, 4-H), 6.88 (m, 2H, 2-H and 9-H), 6.93 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.03 (d, J = 2.9 Hz, 1H, 12-H), 7.12 (t, J = 7.8 Hz, 1H, 3-H), 7.39 (d, J = 8.0 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 30.0 (C-6a), 31.8 (C-19b), 55.8 (C-1′), 56.3 (C-4′), 61.2 (C-3′), 62.3 (C-2′), 66.2 (C-7), 68.3 (C-6), 70.9 (C-12b), 96.4 (C-17), 98.6 (C-13b), 103.3 (C-19a), 115.3 (C-12), 116.9 (C-4), 118.1 (C-9), 118.1 (C-12a), 118.5 (C-10), 122.0 (C-2), 122.4 (C-19c), 128.3 (C-3), 129.8 (C-1), 140.3 (C-15), 146.6 (C-8a), 150.7 (C-17a), 150.7 (C-14), 152.3 (C-4a), 154.0 (C-11), 156.9 (C-16), 158.7 (C-13a), 164.1 (C-19). IR (KBr) ν: 1040, 1102, 1215, 1282, 1398, 1463, 1498, 1605, 1697 cm−1; HRMS: calcd for C30H26O9Na [M + Na]+ 533.1469, found 533.1468.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 3.82 (s, 3H, 1′-H), 3.91 (s, 3H, 2′-H), 3.92–3.96 (m, 4H, 3′-H and 7-Ha), 4.01 (d, J = 11.7 Hz, 1H, 7-Hb), 4.07 (s, 3H, 4′-H), 4.15 (d, J = 11.8 Hz, 1H, 6-Ha), 4.29 (d, J = 11.8 Hz, 1H, 6-Hb), 4.48 (s, 1H, 19b-H), 5.42 (d, J = 1.2 Hz, 1H, 12-H), 6.62 (s, 1H, 15-H), 6.83 (dd, J = 8.2 and 1.1 Hz, 1H, 4-H), 6.87–6.93 (m, 2H, 2-H and 9-H), 6.94 (d, J = 2.9 Hz, 1H, 12-H), 6.98 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.15 (td, J = 7.4 and 1.2 Hz, 1H, 3-H), 7.46 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 31.0 (C-6a), 31.0 (C-19b), 55.8 (C-1′), 56.2 (C-2′), 61.5 (C-3′), 62.3 (C-4′), 66.1 (C-7), 68.1 (C-6), 73.5 (C-12b), 95.9 (C-15), 97.3 (C-19a), 111.2 (C-12), 115.1 (C-4), 116.7 (C-12a), 117.3 (C-9), 118.2 (C-10), 122.1 (C-2), 122.8 (C-19c), 128.2 (C-3), 130.5 (C-1), 140.6 (C-17), 146.7 (C-8a), 151.2 (C-13a), 152.2 (C-4a), 152.8 (C-18), 154.2 (C-11), 157.3 (C-16), 159.1 (C-12a), 177.4 (C-19). IR (KBr) ν: 1218, 1274, 1420, 1499, 1616, 2938 cm−1; HRMS: calcd for C30H26O9Na [M + Na]+ 533.1469, found 533.1469.
1). 1H NMR (400 MHz, CDCl3) δ 3.82 (s, 3H, 1′-H), 3.91 (s, 3H, 2′-H), 3.92–3.96 (m, 4H, 3′-H and 7-Ha), 4.01 (d, J = 11.7 Hz, 1H, 7-Hb), 4.07 (s, 3H, 4′-H), 4.15 (d, J = 11.8 Hz, 1H, 6-Ha), 4.29 (d, J = 11.8 Hz, 1H, 6-Hb), 4.48 (s, 1H, 19b-H), 5.42 (d, J = 1.2 Hz, 1H, 12-H), 6.62 (s, 1H, 15-H), 6.83 (dd, J = 8.2 and 1.1 Hz, 1H, 4-H), 6.87–6.93 (m, 2H, 2-H and 9-H), 6.94 (d, J = 2.9 Hz, 1H, 12-H), 6.98 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.15 (td, J = 7.4 and 1.2 Hz, 1H, 3-H), 7.46 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 31.0 (C-6a), 31.0 (C-19b), 55.8 (C-1′), 56.2 (C-2′), 61.5 (C-3′), 62.3 (C-4′), 66.1 (C-7), 68.1 (C-6), 73.5 (C-12b), 95.9 (C-15), 97.3 (C-19a), 111.2 (C-12), 115.1 (C-4), 116.7 (C-12a), 117.3 (C-9), 118.2 (C-10), 122.1 (C-2), 122.8 (C-19c), 128.2 (C-3), 130.5 (C-1), 140.6 (C-17), 146.7 (C-8a), 151.2 (C-13a), 152.2 (C-4a), 152.8 (C-18), 154.2 (C-11), 157.3 (C-16), 159.1 (C-12a), 177.4 (C-19). IR (KBr) ν: 1218, 1274, 1420, 1499, 1616, 2938 cm−1; HRMS: calcd for C30H26O9Na [M + Na]+ 533.1469, found 533.1469.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 3.45 (d, J = 11.6 Hz, 1H, 7-Ha), 3.61 (s, 3H, 4′-H), 3.80 (s, 3H, 1′-H), 3.82 (s, 3H, 3′-H), 3.89 (d, J = 12.1 Hz, 1H, 7-Hb), 3.93 (s, 3H, 2′-H), 3.97 (d, J = 10.5 Hz, 1H, 6-Ha), 4.27 (d, J = 10.3 Hz, 1H, 6-Hb), 4.36 (s, 1H, 19b-H), 4.88 (s, 1H, 12b-H), 6.70 (s, 1H, 17-H), 6.83 (d, J = 8.9 Hz, 1H, 9-H), 6.93 (m, 4H, 2-H, 4-H, 10-H and 12-H), 7.01 (d, J = 7.4 Hz, 1H, 1-H), 7.20 (t, J = 7.6 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 35.3 (C-19b), 35.9 (C-6a), 55.9 (C-1′), 56.3 (C-2′), 61.3 (C-3′), 62.2 (C-4′), 63.3 (C-7), 69.5 (C-6), 75.4 C-12b), 96.3 (C-17), 96.7 (C-19a), 103.5 (C-13b), 114.9 (C-9), 116.6C(-4), 117.4 (C-12a), 117.7 (C-10), 118.4 (C-2), 121.0 (C-12), 122.8 (C-19c), 126.1 (C-1), 128.2 (C-3), 140.3 (C-15), 148.7 (C-8a), 150.7 (C-14), 152.2 (C-17a), 153.6 (C-11), 154.7 (C-4a), 156.9 (C-16), 161.1 (C-13a), 161.3 (C-19). IR (KBr) ν: 1226, 1390, 1497, 1609, 1704 cm−1; HRMS: calcd for C30H26O9Na [M + Na]+ 533.1469, found 533.1469.
1). 1H NMR (400 MHz, CDCl3) δ 3.45 (d, J = 11.6 Hz, 1H, 7-Ha), 3.61 (s, 3H, 4′-H), 3.80 (s, 3H, 1′-H), 3.82 (s, 3H, 3′-H), 3.89 (d, J = 12.1 Hz, 1H, 7-Hb), 3.93 (s, 3H, 2′-H), 3.97 (d, J = 10.5 Hz, 1H, 6-Ha), 4.27 (d, J = 10.3 Hz, 1H, 6-Hb), 4.36 (s, 1H, 19b-H), 4.88 (s, 1H, 12b-H), 6.70 (s, 1H, 17-H), 6.83 (d, J = 8.9 Hz, 1H, 9-H), 6.93 (m, 4H, 2-H, 4-H, 10-H and 12-H), 7.01 (d, J = 7.4 Hz, 1H, 1-H), 7.20 (t, J = 7.6 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 35.3 (C-19b), 35.9 (C-6a), 55.9 (C-1′), 56.3 (C-2′), 61.3 (C-3′), 62.2 (C-4′), 63.3 (C-7), 69.5 (C-6), 75.4 C-12b), 96.3 (C-17), 96.7 (C-19a), 103.5 (C-13b), 114.9 (C-9), 116.6C(-4), 117.4 (C-12a), 117.7 (C-10), 118.4 (C-2), 121.0 (C-12), 122.8 (C-19c), 126.1 (C-1), 128.2 (C-3), 140.3 (C-15), 148.7 (C-8a), 150.7 (C-14), 152.2 (C-17a), 153.6 (C-11), 154.7 (C-4a), 156.9 (C-16), 161.1 (C-13a), 161.3 (C-19). IR (KBr) ν: 1226, 1390, 1497, 1609, 1704 cm−1; HRMS: calcd for C30H26O9Na [M + Na]+ 533.1469, found 533.1469.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6 aS*,12bR*,19bS*)-4dc as pale yellow powder (32%). Two other isomers were detected, but were not isolated.
1), affording rac-(6 aS*,12bR*,19bS*)-4dc as pale yellow powder (32%). Two other isomers were detected, but were not isolated.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.87 (s, 1H, 5′-H), 3.16 (d, J = 12.4 Hz, 1H, 6-Ha), 3.46 (d, J = 12.8 Hz, 1H, 6-Hb), 3.52 (s, 3H, 2′-H), 3.79 (s, 3H, 3′-H), 3.81 (s, 3H, 1′-H), 3.91 (s, 3H, 4′-H), 3.93 (s, 2H, 7-H), 4.16 (s, 1H, 17b-H), 5.16 (s, 1H, 12b-H), 6.68 (s, 1H, 17-H), 6.83 (s, 1H, 4-H), 6.86 (d, J = 9.0 Hz, 1H, 9-H), 6.88–6.96 (m, 2H, 2-H and 10-H), 7.00 (d, J = 2.8 Hz, 1H, 12-H), 7.31 (d, J = 8.0 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 30.9 (C-6a), 33.4 (C-19b), 39.4 (C-5′), 54.9 (C-6), 55.8 (C-3′), 56.2 (C-4′), 61.8 (C-1′), 62.2 (C-2′), 67.5 (C-7), 72.4 (C-12b), 96.4 (C-17), 98.5 (C-19a), 103.3 (C-13b), 108.0 (C-4), 114.4 (C-2), 115.3 (C-12), 118.0 (C-9), 118.2 (C-10), 118.5 (C-12a), 126.1 (C-19c), 129.8 (C-1), 130.1 (d, J = 31.9 Hz, C-3), 140.3 (C-15), 144.9 (C-4a), 146.7 (C-8a), 150.8 (C-4a and C-17a), 153.9 (C-11), 156.9 (C-16), 158.7 (C-13a), 163.8 (C-19). IR (KBr) ν: 1038, 1100, 1120, 1151, 1172, 1214, 1339, 1399, 1498, 1608, 1703 cm−1; HRMS: calcd for C32H28F3NO8Na [M + Na]+ 634.1659, found 634.1662.
1). 1H NMR (400 MHz, CDCl3) δ 2.87 (s, 1H, 5′-H), 3.16 (d, J = 12.4 Hz, 1H, 6-Ha), 3.46 (d, J = 12.8 Hz, 1H, 6-Hb), 3.52 (s, 3H, 2′-H), 3.79 (s, 3H, 3′-H), 3.81 (s, 3H, 1′-H), 3.91 (s, 3H, 4′-H), 3.93 (s, 2H, 7-H), 4.16 (s, 1H, 17b-H), 5.16 (s, 1H, 12b-H), 6.68 (s, 1H, 17-H), 6.83 (s, 1H, 4-H), 6.86 (d, J = 9.0 Hz, 1H, 9-H), 6.88–6.96 (m, 2H, 2-H and 10-H), 7.00 (d, J = 2.8 Hz, 1H, 12-H), 7.31 (d, J = 8.0 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 30.9 (C-6a), 33.4 (C-19b), 39.4 (C-5′), 54.9 (C-6), 55.8 (C-3′), 56.2 (C-4′), 61.8 (C-1′), 62.2 (C-2′), 67.5 (C-7), 72.4 (C-12b), 96.4 (C-17), 98.5 (C-19a), 103.3 (C-13b), 108.0 (C-4), 114.4 (C-2), 115.3 (C-12), 118.0 (C-9), 118.2 (C-10), 118.5 (C-12a), 126.1 (C-19c), 129.8 (C-1), 130.1 (d, J = 31.9 Hz, C-3), 140.3 (C-15), 144.9 (C-4a), 146.7 (C-8a), 150.8 (C-4a and C-17a), 153.9 (C-11), 156.9 (C-16), 158.7 (C-13a), 163.8 (C-19). IR (KBr) ν: 1038, 1100, 1120, 1151, 1172, 1214, 1339, 1399, 1498, 1608, 1703 cm−1; HRMS: calcd for C32H28F3NO8Na [M + Na]+ 634.1659, found 634.1662.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27
27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and the on-line HPLC-ECD spectra were recorded.
3), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS, 19bR)-4dc: tR = 6.43 min (Chiralpak IC, hexane/dichloromethane/methanol 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27
27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). ECD [nm (Δε), hexane/dichloromethane/methanol 70
3). ECD [nm (Δε), hexane/dichloromethane/methanol 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27
27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3]: 324.5 (−34.27), 299 (7.18), 273.5 (−7.78), 262.5 (4.18), 255 (−3.16), 250 (5.51), 236.5 (−37.73).
3]: 324.5 (−34.27), 299 (7.18), 273.5 (−7.78), 262.5 (4.18), 255 (−3.16), 250 (5.51), 236.5 (−37.73).
(6aS, 12bR, 19bS)-4dc: tR = 6.80 min (Chiralpak IC, hexane/dichloromethane/methanol 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27
27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). ECD [nm (Δε), hexane/dichloromethane/methanol 70
3). ECD [nm (Δε), hexane/dichloromethane/methanol 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27
27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3]: 326.5 (47.92), 298.5 (−10.57), 275 (11.34), 263 (−5.37), 255.5 (4.57), 250 (−7.34), 237 (53.12).
3]: 326.5 (47.92), 298.5 (−10.57), 275 (11.34), 263 (−5.37), 255.5 (4.57), 250 (−7.34), 237 (53.12).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6 aS*,12bR*,19bS*)-4ec as pale yellow amorphous solid (32%). Two other isomers were detected, but were not isolated.
1), affording rac-(6 aS*,12bR*,19bS*)-4ec as pale yellow amorphous solid (32%). Two other isomers were detected, but were not isolated.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.82 (s, 1H, 5′-H), 3.09 (d, J = 12.4 Hz, 1H, 6a-H), 3.41 (d, J = 12.4 Hz, 1H, 6b-H), 3.52 (s, 3H, 2′-H), 3.79 (s, 3H, 3′-H), 3.81 (s, 3H, 1′-H), 3.93 (s, 3H, 4′-H), 3.93 (s, 2H, 7-H), 4.15 (s, 1H, 17b-H), 5.22 (s, 1H, 12b-H), 6.64–6.71 (m, 3H, 2-H, 4-H and 17-H), 6.85 (d, J = 9.2 Hz, 1H, 9-H), 6.93 (dd, J = 9.2 and 2.8 Hz, 1H, 10-H), 7.00 (d, J = 2.8 Hz, 1H, 12-H), 7.13 (t, J = 7.6 Hz, 1H, 3-H), 7.20 (d, J = 8.0 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 33.3 (C-5′), 36.6 (C-6a), 39.5 (C-19b), 55.1 (C-6), 55.8 (C-1′), 56.2 (C-4′), 61.2 (C-3′), 62.2 (C-2′), 67.7 (C-7), 72.5 (C-12b), 96.4 (C-17), 99.2 (−19a), 103.5 (13b), 111.7 (C-4), 115.4 (C-2), 117.9 (C-10), 118.0 (C-12), 118.2 (C-9), 120.6 (C-12a), 122.7 (C-19c), 127.8 (C-3), 129.3 (C-1), 141.2 (C-15), 144.8 (C-4a), 146.9 (C-14), 150.7 (C-17a), 153.8 (C-11), 156.6 (C-16), 158.5 (C-13a), 163.9 (C-19). IR (KBr) ν: 1217, 1228, 1365, 1497, 1608, 1713, 1738 cm−1; HRMS: calcd for C31H29NO8Na [M + Na]+ 566.1785, found 566.1781.
1). 1H NMR (400 MHz, CDCl3) δ 2.82 (s, 1H, 5′-H), 3.09 (d, J = 12.4 Hz, 1H, 6a-H), 3.41 (d, J = 12.4 Hz, 1H, 6b-H), 3.52 (s, 3H, 2′-H), 3.79 (s, 3H, 3′-H), 3.81 (s, 3H, 1′-H), 3.93 (s, 3H, 4′-H), 3.93 (s, 2H, 7-H), 4.15 (s, 1H, 17b-H), 5.22 (s, 1H, 12b-H), 6.64–6.71 (m, 3H, 2-H, 4-H and 17-H), 6.85 (d, J = 9.2 Hz, 1H, 9-H), 6.93 (dd, J = 9.2 and 2.8 Hz, 1H, 10-H), 7.00 (d, J = 2.8 Hz, 1H, 12-H), 7.13 (t, J = 7.6 Hz, 1H, 3-H), 7.20 (d, J = 8.0 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 33.3 (C-5′), 36.6 (C-6a), 39.5 (C-19b), 55.1 (C-6), 55.8 (C-1′), 56.2 (C-4′), 61.2 (C-3′), 62.2 (C-2′), 67.7 (C-7), 72.5 (C-12b), 96.4 (C-17), 99.2 (−19a), 103.5 (13b), 111.7 (C-4), 115.4 (C-2), 117.9 (C-10), 118.0 (C-12), 118.2 (C-9), 120.6 (C-12a), 122.7 (C-19c), 127.8 (C-3), 129.3 (C-1), 141.2 (C-15), 144.8 (C-4a), 146.9 (C-14), 150.7 (C-17a), 153.8 (C-11), 156.6 (C-16), 158.5 (C-13a), 163.9 (C-19). IR (KBr) ν: 1217, 1228, 1365, 1497, 1608, 1713, 1738 cm−1; HRMS: calcd for C31H29NO8Na [M + Na]+ 566.1785, found 566.1781.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and the on-line HPLC-ECD spectra were recorded.
2), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS, 19bR)-4ec: tR = 20.57 min (Chiralpak IC, hexane/dichloromethane/methanol 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). ECD [nm (Δε), hexane/dichloromethane/methanol 80
2). ECD [nm (Δε), hexane/dichloromethane/methanol 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2]: 318 (−3.13), 275 (−1.04), 260 (0.89), 248.5 (1.65), 230.5 (−7.35).
2]: 318 (−3.13), 275 (−1.04), 260 (0.89), 248.5 (1.65), 230.5 (−7.35).
(6 aS,12bR,19bS)-4ec: tR = 21.95 min (Chiralpak IC, hexane/dichloromethane/methanol 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). ECD [nm (Δε), hexane/dichloromethane/methanol 80
2). ECD [nm (Δε), hexane/dichloromethane/methanol 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 18
18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2]: 319 (3.41), 282 (1.51), 261.5 (−0.44), 246 (−1.19), 231 (8.39).
2]: 319 (3.41), 282 (1.51), 261.5 (−0.44), 246 (−1.19), 231 (8.39).
Domino Knoevenagel-IMHDA reactions of 2H-chromene derivatives 1a-c with 2,4-dihydroxyquinoline 2d
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).1H NMR (400 MHz, DMSO-d6) δ 2.94 and 2.95 (s, 3H, 2′-H), 3.04 (d, J = 8.3 Hz, 4H, 2′-H and 6-Ha), 3.12–3.24 (m, 2H, 6-Ha), 3.49 (d, J = 11.7 Hz, 1H, 6-Hb), 3.71 (d, J = 11.2 Hz, 4H, 1′-H and 7-Ha), 3.76 and 3.77 (s, 3H, 1′-H), 3.84 (d, J = 13.4 Hz, 2H, 6-Hb and 7-Hb), 4.09 (d, J = 11.3 Hz, 1H, 7-Hb), 4.23, 4.32 and 4.43 (s, 1H, 19-Hb), 5.07 and 5.18 (s, 1H, 12b-H), 6.74 (d, J = 9.4 Hz, 1H, 4-H), 6.81 (d, J = 9.3 Hz, 1H, 4-H), 6.84 (d, J = 9.0 Hz, 1H, 9-H), 6.87 (d, J = 8.9 Hz, 1H, 9-H), 6.95–7.03 (m, 1H, 10-H), 7.07 (d, J = 3.0 Hz, 1H, 12-H), 7.11 (d, J = 8.0 Hz, 1H, 15-H), 7.16 (d, J = 7.9 Hz, 1H, 15-H), 7.19 and 7.23 (d, J = 3.0 Hz, 1H, 12-H), 7.34 (d, J = 8.3 Hz, 1H, 17-H), 7.37–7.42 (m, 1H, 17-H), 7.50 (t, J = 8.3 Hz, 1H, 16-H), 7.55 (d, J = 8.5 Hz, 1H, 16-H), 7.59 (d, J = 8.0 Hz, 1H, 14-H), 7.67 (d, J = 8.7 Hz, 1H, 14-H), 7.93 (dd, J = 9.3, 2.5 Hz, 1H, 3-H), 7.96 and 8.03 (m, 1H, 1-H), 8.06 and 8.18 (m, 1H, 3-H), 11.85 (s, 1H, N–H). 13C NMR (100 MHz, DMSO-d6) δ 30.3 and 30.6 (C-6a), 32.1 and 32.3 (C-19b), 34.6 (C-6a), 37.2 (C-19b), 38.5 and 39.8 (C-2′), 54.2 and 55.3 (C-6), 56.1 and 56.2 (C-1′), 63.3 (C-6), 67.5 and 67.6 (C-7), 71.0, 72.4 and 74.4 (C-12b), 99.2, 105.0 and 107.4 (C-19a), 110.0, 110.9 and 111.1 (C-4), 114.12 and 114.6 (C-13b), 115.6 and 115.8 (C-17), 116.2, 116.5 and 116.6 (C-12), 117.4 and 117.9 (C-9), 118.0 (C-10), 118.0 (C-9), 118.4 and 118.5 (C-10), 118.9, 119.1, and 119.8 (C-12a), 120.2 (C-19c), 122.0 and 122.1 (C-15), 122.2 (C-19c), 122.7 (C-14), 122.9 (C-19c), 123.1 (C-14), 123.5 (C-18a), 124.6 and 125.6 (C-3), 126.2 and 126.5 (C-1), 131.3 and 131.9 (C-16), 136.6 and 137.2 (C-2), 137.3 (C-17a), 138.5 (C-17a and C-14a), 146.8, 147.0 and 148.5 (C-8a), 149.8, 150.0 and 152.6 (C-4a), 153.4, 153.7 and 153.9 (C-13a), 154.0 and 154.1 (C-11), 157.3, 162.4 and 163.7 (C-19). IR (KBr) ν: 1215, 1297, 1319, 1498, 1606, 1649 cm−1; HRMS: calcd for C28H23N3O6Na [M + Na]+ 520.1479, found 520.1479.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 7R*, 12bR*, 19bS*)-4bd as yellow powder (60%) and rac-(6aS*, 7R*, 12bR*,1 9bR*)-5bd as yellow powder (17%).
1), affording rac-(6aS*, 7R*, 12bR*, 19bS*)-4bd as yellow powder (60%) and rac-(6aS*, 7R*, 12bR*,1 9bR*)-5bd as yellow powder (17%).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, DMSO-d6) δ 2.92 (s, 3H, 2″-H), 3.28 (d, J = 13.5 Hz, 1H, 6-Ha), 3.53 (d, J = 13.5 Hz, 1H, 6-Hb), 3.79 (s, 3H, 1″-H), 3.87 (s, 1H, 19b-H), 5.01 (s, 1H, 7-H), 5.17 (s, 1H, 12b-H), 6.72 (d, J = 9.4 Hz, 1H, 4-H), 7.00 (d, J = 9.0 Hz, 1H, 9-H), 7.04 (dd, J = 9.0 and 2.8 Hz, 1H, 10-H), 7.15 (t, J = 7.5 Hz, 1H, 15-H), 7.24–7.30 (m, 2H, 2′-H and 6′-H), 7.31 (d, J = 2.8 Hz, 1H, 12-H), 7.34–7.45 (m, 4H, 3′-H, 4′-H, 5′-H and 17-H), 7.54 (td, J = 7.7 and 1.1 Hz, 1H, 16-H), 7.67 (d, J = 7.9 Hz, 1H, 14-H), 7.89 (dd, J = 9.2 and 2.6 Hz, 1H, 3-H), 8.01 (m, 1H, 1-H), 11.92 (s, 1H, N–H). 13C NMR (100 MHz, DMSO-d6) δ 31.6 (C-19b), 32.1 (C-6a), 49.8 (C-6), 55.5 (C-1″), 71.3 (C-12b), 76.7 (C-7), 106.8 (C-19a), 110.9 (C-4), 113.6 (C-13b), 115.4 (C-17), 115.9 (C-12), 117.5 (C-9 and C-10), 119.3 (C-12a), 121.8 (C-15), 122.3 (C-14), 122.4 (C-19c), 124.0 (C-3), 125.6 (C-1), 127.8 (C-2′ and C-6′), 128.0 (C-3′ and C-5′), 128.7 (C-4′), 131.1 (C-16), 134.4 (C-1′), 136.6 (C-2), 138.2 (C-17a), 147.3 (C-8a), 148.9 (C-4a), 153.8 (C-11), 154.2 (C-13a), 163.0 (C-19). IR (KBr) ν: 1261, 1299, 1312, 1499, 1605, 1646, 2854, 2925 cm−1; HRMS: calcd for C34H27N3O6Na [M + Na]+ 596.1792, found 596.1792.
1). 1H NMR (400 MHz, DMSO-d6) δ 2.92 (s, 3H, 2″-H), 3.28 (d, J = 13.5 Hz, 1H, 6-Ha), 3.53 (d, J = 13.5 Hz, 1H, 6-Hb), 3.79 (s, 3H, 1″-H), 3.87 (s, 1H, 19b-H), 5.01 (s, 1H, 7-H), 5.17 (s, 1H, 12b-H), 6.72 (d, J = 9.4 Hz, 1H, 4-H), 7.00 (d, J = 9.0 Hz, 1H, 9-H), 7.04 (dd, J = 9.0 and 2.8 Hz, 1H, 10-H), 7.15 (t, J = 7.5 Hz, 1H, 15-H), 7.24–7.30 (m, 2H, 2′-H and 6′-H), 7.31 (d, J = 2.8 Hz, 1H, 12-H), 7.34–7.45 (m, 4H, 3′-H, 4′-H, 5′-H and 17-H), 7.54 (td, J = 7.7 and 1.1 Hz, 1H, 16-H), 7.67 (d, J = 7.9 Hz, 1H, 14-H), 7.89 (dd, J = 9.2 and 2.6 Hz, 1H, 3-H), 8.01 (m, 1H, 1-H), 11.92 (s, 1H, N–H). 13C NMR (100 MHz, DMSO-d6) δ 31.6 (C-19b), 32.1 (C-6a), 49.8 (C-6), 55.5 (C-1″), 71.3 (C-12b), 76.7 (C-7), 106.8 (C-19a), 110.9 (C-4), 113.6 (C-13b), 115.4 (C-17), 115.9 (C-12), 117.5 (C-9 and C-10), 119.3 (C-12a), 121.8 (C-15), 122.3 (C-14), 122.4 (C-19c), 124.0 (C-3), 125.6 (C-1), 127.8 (C-2′ and C-6′), 128.0 (C-3′ and C-5′), 128.7 (C-4′), 131.1 (C-16), 134.4 (C-1′), 136.6 (C-2), 138.2 (C-17a), 147.3 (C-8a), 148.9 (C-4a), 153.8 (C-11), 154.2 (C-13a), 163.0 (C-19). IR (KBr) ν: 1261, 1299, 1312, 1499, 1605, 1646, 2854, 2925 cm−1; HRMS: calcd for C34H27N3O6Na [M + Na]+ 596.1792, found 596.1792.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, DMSO-d6) δ 2.90 (s, 3H, 2″-H), 3.44 (d, J = 12.8 Hz, 1H, 6-Ha), 3.61 (d, J = 12.7 Hz, 1H, 6-Hb), 3.79 (s, 3H, 1″-H), 4.36 (s, 1H, 19b-H), 5.27 (s, 1H, 12b-H), 5.34 (s, 1H, 7-H), 6.02 (d, J = 9.2 Hz, 1H, 4-H), 6.82 (m, 2H, 2′-H and 6′-H), 6.91 (d, J = 8.9 Hz, 1H, 9-H), 7.05 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.10 (d, J = 3.0 Hz, 1H, 12-H), 7.19 (t, J = 7.6 Hz, 1H, 15-H), 7.39 (d, J = 8.2 Hz, 1H, 17-H), 7.41 (m, 1H, 1-H), 7.45 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H), 7.58 (td, J = 7.7 and 1.2 Hz, 1H, 16-H), 7.74 (d, J = 7.9 Hz, 1H, 14-H), 11.82 (s, 1H, N–H). 13C NMR (100 MHz, DMSO-d6) δ 36.7 (C-6a), 37.4 (C-2″), 38.2 (C-19b), 52.6 (C-6), 55.6 (C-1″), 76.8 (C-12b), 76.9 (C-7), 105.1 (C-19a), 108.9 (C-4), 114.2 (C-13b), 115.0 (C-12 and C-17), 116.8 (C-9), 118.0 (C-10), 118.3 (C-19c), 118.3 (C-12a), 121.5 (C-15), 122.3 (C-14), 123.4 (C-3), 124.6 (C-1), 127.5 (C-2′ and C-6′), 130.9 (C-16), 135.3 (C-2), 137.6 (C-1′), 137.9 (C-17a), 148.3 (C-8a), 152.6 (C-4a), 153.2 (C-11), 157.4 (C-13a), 161.9 (C-19). IR (KBr) ν: 1230, 1289, 1497, 1607, 1644, 2853, 2924 cm−1; HRMS: calcd for C34H27N3O6Na [M + Na]+ 596.1792, found 596.1792.
1). 1H NMR (400 MHz, DMSO-d6) δ 2.90 (s, 3H, 2″-H), 3.44 (d, J = 12.8 Hz, 1H, 6-Ha), 3.61 (d, J = 12.7 Hz, 1H, 6-Hb), 3.79 (s, 3H, 1″-H), 4.36 (s, 1H, 19b-H), 5.27 (s, 1H, 12b-H), 5.34 (s, 1H, 7-H), 6.02 (d, J = 9.2 Hz, 1H, 4-H), 6.82 (m, 2H, 2′-H and 6′-H), 6.91 (d, J = 8.9 Hz, 1H, 9-H), 7.05 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.10 (d, J = 3.0 Hz, 1H, 12-H), 7.19 (t, J = 7.6 Hz, 1H, 15-H), 7.39 (d, J = 8.2 Hz, 1H, 17-H), 7.41 (m, 1H, 1-H), 7.45 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H), 7.58 (td, J = 7.7 and 1.2 Hz, 1H, 16-H), 7.74 (d, J = 7.9 Hz, 1H, 14-H), 11.82 (s, 1H, N–H). 13C NMR (100 MHz, DMSO-d6) δ 36.7 (C-6a), 37.4 (C-2″), 38.2 (C-19b), 52.6 (C-6), 55.6 (C-1″), 76.8 (C-12b), 76.9 (C-7), 105.1 (C-19a), 108.9 (C-4), 114.2 (C-13b), 115.0 (C-12 and C-17), 116.8 (C-9), 118.0 (C-10), 118.3 (C-19c), 118.3 (C-12a), 121.5 (C-15), 122.3 (C-14), 123.4 (C-3), 124.6 (C-1), 127.5 (C-2′ and C-6′), 130.9 (C-16), 135.3 (C-2), 137.6 (C-1′), 137.9 (C-17a), 148.3 (C-8a), 152.6 (C-4a), 153.2 (C-11), 157.4 (C-13a), 161.9 (C-19). IR (KBr) ν: 1230, 1289, 1497, 1607, 1644, 2853, 2924 cm−1; HRMS: calcd for C34H27N3O6Na [M + Na]+ 596.1792, found 596.1792.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 19bR*)-4cd as white powder (24%), mp > 330 °C. Rf = 0.14 (hexane/ethyl acetate 2
1), affording rac-(6aS*, 12bR*, 19bR*)-4cd as white powder (24%), mp > 330 °C. Rf = 0.14 (hexane/ethyl acetate 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).1H NMR (400 MHz, CDCl3) δ 3.85 (s, 3H, 1′-H), 3.94 (dd, J = 11.7 and 1.5 Hz, 1H, 7-Ha), 4.00 (d, J = 11.6 Hz, 1H, 7-Hb), 4.20 (d, J = 11.8 Hz, 1H, 6-Ha), 4.36 (d, J = 11.8 Hz, 1H, 6-Hb), 4.48 (s, 1H, 19b-H), 5.31 (s, 1H, 12b-H), 6.82–6.91 (m, 3H, 9-H, 2-H and 4-H), 6.96 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.06 (d, J = 2.9 Hz, 1H, 12-H), 7.14 (t, J = 7.6 Hz, 2H, 3-H and 15-H), 7.38 (d, J = 8.1 Hz, 1H, 17-H), 7.47 (td, J = 7.7 and 1.1 Hz, 2H, 16-H), 7.63 (d, J = 7.8 Hz, 1H, 1-H), 7.78 (d, J = 8.1 Hz, 1H, 14-H), 11.89 (s, 1H, N–H). 13C NMR (100 MHz, CDCl3) δ 30.5 (C-6a), 31.8 (C-19b), 56.0 (C-1′), 66.5 (C-7), 68.7 (C-6), 70.4 (C-12b), 106.6 (C-19a), 114.8 (C-12a), 115.9 (C-12), 115.9 (C-17), 117.0 (C-9), 118.0 (C-10), 118.0 (C-4), 119.1 (C-13b), 122.0 (C-2), 122.4 (C-15), 123.1 (C-19c), 123.2 (C-14), 128.3 (C-3), 130.6 (C-1), 131.2 (C-16), 137.7 (C-17a), 146.8 (C-8a), 152.6 (C-4a), 154.2 (C-11), 155.4 (C-13a), 165.8 (C-19). IR (KBr) ν: 1043, 1215, 1259, 1401, 1498, 1605, 1646, 2862, 2947, 2993 cm−1; HRMS: calcd for C27H21NO5Na [M + Na]+ 462.1312, found 462.1311.
Domino Knoevenagel-IMHDA reactions of 2H-chromene derivatives 1a-c with 4-hydroxy-6-methyl-2H-pyran-2-one 2e and 4-hydroxy-1,6-dimethyl-pyridine-2(1H)-one 2f
rac-(6aS*,12bR*,17bS*)- (6aS*,12bR*,17bS*)-11-methoxy-5,15-dimethyl-2-nitro-5,17b-dihydro-6H,12bH,17H-chromeno[4′,3′:2,3]pyrano[3′,4':5,6]pyrano[3,4-c]quinolin-17-one [rac-(6aS*,12bR*,17bS*)-4ae and rac-(6 aS*,12bR*,17bS*)-5ae] and rac-(6aS*,12bR*,17bS*)- and rac-(6aS*,12bR*,17bR*)-11-methoxy-5,15-dimethyl-2-nitro-5,17b-dihydro-6H,12bH,17H-chromeno[4′,3′:2,3]pyrano[3′,2′:5,6]pyrano[3,4-c]quinolin-17-one [rac-(6aS*,12bR*,17bS*)-6ae and rac-(6aS*,12bR*,17bR*)-7ae]The reaction of 1a and 2e was carried out according to method A, and the crude product was purified by column chromatography (hexane/ethyl acetate 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 17bS*)-4ae as yellow powder (16%), rac-(6 aS*, 12bR*,17bS*)-6ae as yellow oil (35%), rac-(6 aS*, 12bR*, 17bS*)-5ae as orange powder (14%) and rac-(6aS*, 12bR*, 17bR*)-7ae as yellow powder (6%).
1), affording rac-(6aS*, 12bR*, 17bS*)-4ae as yellow powder (16%), rac-(6 aS*, 12bR*,17bS*)-6ae as yellow oil (35%), rac-(6 aS*, 12bR*, 17bS*)-5ae as orange powder (14%) and rac-(6aS*, 12bR*, 17bR*)-7ae as yellow powder (6%).
Alternatively, the reaction was carried out at room temperature, affording rac-(6aS*, 12bR*, 17bS*)-4ae as yellow powder (14%), rac-(6aS*, 12bR*, 17bS*)-6ae as yellow oil (37%) and rac-(6aS*, 12bR*, 17bS*)-5ae as orange powder (27%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.26 (s, 3H, 3′-H), 2.94 (s, 3H, 2′-H), 3.24 (d, J = 13.2 Hz, 1H, 6-Ha), 3.64 (d, J = 13.2 Hz, 1H, 6-Hb), 3.81 (s, 3H, 1′-H), 3.87 (d, J = 11.5 Hz, 1H, 7-Ha), 3.96 (dd, J = 11.4 and 1.1 Hz, 1H, 7-Hb), 4.09 (s, 1H, 17b-H), 4.89 (s, 1H, 12b-H), 5.76 (s, 1H, 14-H), 6.55 (d, J = 9.2 Hz, 1H, 4-H), 6.85 (d, J = 2.9 Hz, 1H, 12-H), 6.89 (d, J = 9.0 Hz, 1H, 9-H), 6.97 (dd, J = 9.0 and 2.9 Hz, 1H, 10-H), 7.99 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.11 (m, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.1 (C-3′), 30.7 (C-6a), 32.6 (C-17b), 39.8 (C-2′), 54.7 (C-6), 55.9 (C-7), 67.5 (C-12b), 72.0 (C-17a), 98.5 (C-14), 100.1 (C-14), 110.3 (C-4), 115.4 (C-12), 118.2 (C-10), 118.3 (C-9 and C-12a), 121.6 (C-17c), 124.9 (C-3), 126.2 (C-1), 138.5 (C-2), 146.7 (C-8a), 149.0 (C-4a), 154.2 (C-11), 162.1 (C-13a), 162.5 (C-15), 165.6 (C-17). IR (KBr) ν: 1211, 1266, 1326, 1498, 1579, 1697 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1319.
1). 1H NMR (400 MHz, CDCl3) δ 2.26 (s, 3H, 3′-H), 2.94 (s, 3H, 2′-H), 3.24 (d, J = 13.2 Hz, 1H, 6-Ha), 3.64 (d, J = 13.2 Hz, 1H, 6-Hb), 3.81 (s, 3H, 1′-H), 3.87 (d, J = 11.5 Hz, 1H, 7-Ha), 3.96 (dd, J = 11.4 and 1.1 Hz, 1H, 7-Hb), 4.09 (s, 1H, 17b-H), 4.89 (s, 1H, 12b-H), 5.76 (s, 1H, 14-H), 6.55 (d, J = 9.2 Hz, 1H, 4-H), 6.85 (d, J = 2.9 Hz, 1H, 12-H), 6.89 (d, J = 9.0 Hz, 1H, 9-H), 6.97 (dd, J = 9.0 and 2.9 Hz, 1H, 10-H), 7.99 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.11 (m, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.1 (C-3′), 30.7 (C-6a), 32.6 (C-17b), 39.8 (C-2′), 54.7 (C-6), 55.9 (C-7), 67.5 (C-12b), 72.0 (C-17a), 98.5 (C-14), 100.1 (C-14), 110.3 (C-4), 115.4 (C-12), 118.2 (C-10), 118.3 (C-9 and C-12a), 121.6 (C-17c), 124.9 (C-3), 126.2 (C-1), 138.5 (C-2), 146.7 (C-8a), 149.0 (C-4a), 154.2 (C-11), 162.1 (C-13a), 162.5 (C-15), 165.6 (C-17). IR (KBr) ν: 1211, 1266, 1326, 1498, 1579, 1697 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1319.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), and the on-line HPLC-ECD spectra were recorded.
5), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS, 17bR)-4ae: tR = 9.31 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 387 (1.25), 335 (−0.83), 316.5 (−1.13), 310 (0.66), 291 (−7.98), 248.5 (−3.10), 226.5 (−25.04).
5]: 387 (1.25), 335 (−0.83), 316.5 (−1.13), 310 (0.66), 291 (−7.98), 248.5 (−3.10), 226.5 (−25.04).
(6aS, 12bR, 17bS)-4ae: tR = 12.02 min (Chiralpak IC, hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5). ECD [nm (Δε), hexane/dichloromethane/methanol 50
5). ECD [nm (Δε), hexane/dichloromethane/methanol 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 45
45![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5]: 392.5 (−0.87), 334.5 (1.06), 316.5 (1.49), 309 (−0.24), 291 (7.85), 250 (3.05), 226 (23.20).
5]: 392.5 (−0.87), 334.5 (1.06), 316.5 (1.49), 309 (−0.24), 291 (7.85), 250 (3.05), 226 (23.20).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.30 (s, 3H, 3′-H), 3.02 (s, 3H, 2′-H), 3.18 (s, 2H, 6-H), 3.32 (d, J = 11.7 Hz, 1H, 7-Ha), 3.79 (m, 4H, 1′-H and 7-Hb), 4.02 (s, 1H, 17b-H), 4.73 (s, 1H, 12b-H), 5.86 (s, 1H, 14-H), 6.65 (d, J = 9.1 Hz, 1H, 4-H), 6.82 (m, 2H, 9-H and 12-H), 6.95 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.79 (d, J = 2.7 Hz, 1H, 1-H), 8.12 (dd, J = 9.1 and 2.7 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 20.1 (C-3′), 35.2 (C-6a), 36.4 (C-17b), 38.3 (C-2′), 54.4 (C-6), 56.0 (C-1′), 63.1 (C-7), 75.9 (C-12b), 96.4 (C-17a), 100.1 (C-14), 109.9 (C-4), 115.0 (C-12), 117.2 (C-12a), 117.8 (C-9), 118.7 (C-10), 119.4 (C-17c), 122.5 (C-1), 125.1 (C-3), 138.2 (C-2), 148.4 (C-8a), 151.8 (C-4a), 153.9 (C-11), 162.7 (C-15), 163.1 (C-17), 164.9 (C-13a). IR (KBr) ν: 1040, 1217, 1289, 1313, 1498, 1575, 1604, 1710, 2925 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1319.
1). 1H NMR (400 MHz, CDCl3) δ 2.30 (s, 3H, 3′-H), 3.02 (s, 3H, 2′-H), 3.18 (s, 2H, 6-H), 3.32 (d, J = 11.7 Hz, 1H, 7-Ha), 3.79 (m, 4H, 1′-H and 7-Hb), 4.02 (s, 1H, 17b-H), 4.73 (s, 1H, 12b-H), 5.86 (s, 1H, 14-H), 6.65 (d, J = 9.1 Hz, 1H, 4-H), 6.82 (m, 2H, 9-H and 12-H), 6.95 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.79 (d, J = 2.7 Hz, 1H, 1-H), 8.12 (dd, J = 9.1 and 2.7 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 20.1 (C-3′), 35.2 (C-6a), 36.4 (C-17b), 38.3 (C-2′), 54.4 (C-6), 56.0 (C-1′), 63.1 (C-7), 75.9 (C-12b), 96.4 (C-17a), 100.1 (C-14), 109.9 (C-4), 115.0 (C-12), 117.2 (C-12a), 117.8 (C-9), 118.7 (C-10), 119.4 (C-17c), 122.5 (C-1), 125.1 (C-3), 138.2 (C-2), 148.4 (C-8a), 151.8 (C-4a), 153.9 (C-11), 162.7 (C-15), 163.1 (C-17), 164.9 (C-13a). IR (KBr) ν: 1040, 1217, 1289, 1313, 1498, 1575, 1604, 1710, 2925 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1319.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and the on-line HPLC-ECD spectra were recorded.
7), and the on-line HPLC-ECD spectra were recorded.
(6aS, 12bR, 17bR)-5ae: tR = 6.03 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 387 (−19.42), 312 (−24.98), 264.5 (5.93), 236.5 (−13.46).
7]: 387 (−19.42), 312 (−24.98), 264.5 (5.93), 236.5 (−13.46).
(6aR, 12bS, 17bS)-5ae: tR = 13.46 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 388.5 (10.03), 312 (12.61), 263 (−3.35), 238 (6.60).
7]: 388.5 (10.03), 312 (12.61), 263 (−3.35), 238 (6.60).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.19 (s, 3H, 3′-H), 2.89 (s, 3H, 2′-H), 3.18 (d, J = 13.3 Hz, 1H, 6-Ha), 3.67 (d, J = 13.2 Hz, 1H, 6-Hb), 3.76 (s, 3H, 1′-H), 3.87 (d, J = 11.6 Hz, 1H, 7-Ha), 4.01 (dd, J = 11.6, 1.2 Hz, 1H, 7-Hb), 4.33 (s, 1H, 17b-H), 5.07 (s, 1H, 12b-H), 6.13 (s, 1H, 16-H), 6.47 (d, J = 9.3 Hz, 1H, 4-H), 6.83 (d, J = 2.9 Hz, 1H, 12-H), 6.86 (d, J = 9.0 Hz, 1H, 9-H), 6.93 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.88 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 7.96 (d, J = 2.5, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.2 (C-3′), 30.7 (C-6a), 31.8 (C-17b), 39.6 (C-2′), 54.2 (C-6), 55.8 (C-1′), 67.2 (C-7), 74.6 (C-12b), 99.8 (C-17a), 110.1 (C-4), 112.6 (C-16), 115.0 (C-9), 117.3 (C-17c), 118.2 (C-10), 118.7 (C-12), 121.5 (C-12a), 124.6 (C-3), 126.4 (C-1), 138.2 (C-2), 146.7 (C-8a), 148.8 (C-4a), 154.1 (C-11), 160.4 (C-15), 161.4 (C-13a), 180.1 (C-17). IR (KBr) ν: 1265, 1300, 1323, 1498, 1585, 1603, 1668 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1319.
1). 1H NMR (400 MHz, CDCl3) δ 2.19 (s, 3H, 3′-H), 2.89 (s, 3H, 2′-H), 3.18 (d, J = 13.3 Hz, 1H, 6-Ha), 3.67 (d, J = 13.2 Hz, 1H, 6-Hb), 3.76 (s, 3H, 1′-H), 3.87 (d, J = 11.6 Hz, 1H, 7-Ha), 4.01 (dd, J = 11.6, 1.2 Hz, 1H, 7-Hb), 4.33 (s, 1H, 17b-H), 5.07 (s, 1H, 12b-H), 6.13 (s, 1H, 16-H), 6.47 (d, J = 9.3 Hz, 1H, 4-H), 6.83 (d, J = 2.9 Hz, 1H, 12-H), 6.86 (d, J = 9.0 Hz, 1H, 9-H), 6.93 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.88 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 7.96 (d, J = 2.5, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.2 (C-3′), 30.7 (C-6a), 31.8 (C-17b), 39.6 (C-2′), 54.2 (C-6), 55.8 (C-1′), 67.2 (C-7), 74.6 (C-12b), 99.8 (C-17a), 110.1 (C-4), 112.6 (C-16), 115.0 (C-9), 117.3 (C-17c), 118.2 (C-10), 118.7 (C-12), 121.5 (C-12a), 124.6 (C-3), 126.4 (C-1), 138.2 (C-2), 146.7 (C-8a), 148.8 (C-4a), 154.1 (C-11), 160.4 (C-15), 161.4 (C-13a), 180.1 (C-17). IR (KBr) ν: 1265, 1300, 1323, 1498, 1585, 1603, 1668 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1319.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and the on-line HPLC-ECD spectra were recorded.
7), and the on-line HPLC-ECD spectra were recorded.
(6aR, 12bS,1 7bR)-6ae: tR = 5.81 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 402.5 (0.73), 319 (−3.48), 296 (5.98), 243.5 (−58.93).
7]: 402.5 (0.73), 319 (−3.48), 296 (5.98), 243.5 (−58.93).
(6aS, 12bR, 17bS)-6ae: tR = 11.71 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 406.5 (−0.51), 318.5 (3.23), 297.5 (−5.76), 242 (54.73).
7]: 406.5 (−0.51), 318.5 (3.23), 297.5 (−5.76), 242 (54.73).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.29 (s, 3H, 3′-H), 3.03 (s, 3H, 2′-H), 3.20 (s, 2H, 6-H), 3.34 (dd, J = 11.7 and 1.6 Hz, 1H, 7-Ha), 3.80 (s, 3H, 1′-H), 3.88 (d, J = 11.7 Hz, 1H, 7-Hb), 4.14 (s, 1H, 17b-H), 4.94 (d, J = 1.4 Hz, 1H, 12b-H), 6.19 (s, 1H, 16-H), 6.64 (d, J = 9.1 Hz, 1H, 4-H), 6.83 (m, 2H, 9-H and 12-H), 6.96 (dd, J = 8.9 and 3.1 Hz, 1H, 10-H), 7.66 (d, J = 2.5 Hz, 1H, 1-H), 8.11 (dd, J = 9.1 and 2.5 Hz, 1H, 3-H). 13C NMR (90 MHz, CDCl3) δ 19.2 (C-3′), 35.6 (C-6a), 36.9 (C-17b), 38.3 (C-2′), 54.2 (C-69, 56.0 (C-1′), 63.1 (C-7), 78.1 (C-12b), 97.4 (C-17a), 109.7 (C-4), 113.5 (C-16), 114.9 (C-12), 116.2 (C-12a), 118.0 (C-9), 119.3 (C-10), 119.3 (C-17c), 123.0 (C-1), 125.1 (C-3), 138.2 (C-2), 148.6 (C-8a), 151.6 (C-4a), 153.9 (C-11), 161.1 (C-15), 162.4 (C-13a), 179.4 (C-17). IR (KBr) ν: 1216, 1364, 1498, 1604, 1715, 2925, 2969, 3005 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1321.
1). 1H NMR (400 MHz, CDCl3) δ 2.29 (s, 3H, 3′-H), 3.03 (s, 3H, 2′-H), 3.20 (s, 2H, 6-H), 3.34 (dd, J = 11.7 and 1.6 Hz, 1H, 7-Ha), 3.80 (s, 3H, 1′-H), 3.88 (d, J = 11.7 Hz, 1H, 7-Hb), 4.14 (s, 1H, 17b-H), 4.94 (d, J = 1.4 Hz, 1H, 12b-H), 6.19 (s, 1H, 16-H), 6.64 (d, J = 9.1 Hz, 1H, 4-H), 6.83 (m, 2H, 9-H and 12-H), 6.96 (dd, J = 8.9 and 3.1 Hz, 1H, 10-H), 7.66 (d, J = 2.5 Hz, 1H, 1-H), 8.11 (dd, J = 9.1 and 2.5 Hz, 1H, 3-H). 13C NMR (90 MHz, CDCl3) δ 19.2 (C-3′), 35.6 (C-6a), 36.9 (C-17b), 38.3 (C-2′), 54.2 (C-69, 56.0 (C-1′), 63.1 (C-7), 78.1 (C-12b), 97.4 (C-17a), 109.7 (C-4), 113.5 (C-16), 114.9 (C-12), 116.2 (C-12a), 118.0 (C-9), 119.3 (C-10), 119.3 (C-17c), 123.0 (C-1), 125.1 (C-3), 138.2 (C-2), 148.6 (C-8a), 151.6 (C-4a), 153.9 (C-11), 161.1 (C-15), 162.4 (C-13a), 179.4 (C-17). IR (KBr) ν: 1216, 1364, 1498, 1604, 1715, 2925, 2969, 3005 cm−1; HRMS: calcd for C25H22N2O7Na [M + Na]+ 485.1319, found 485.1321.The enantiomers were separated on a Chiralpak IC column (hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7), and the on-line HPLC-ECD spectra were recorded.
7), and the on-line HPLC-ECD spectra were recorded.
1st eluting enantiomer: tR = 7.29 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 389.5 (−18.95), 325.5 (0.30), 313.5 (−10.49), 275 (4.80), 230 (−64.54).
7]: 389.5 (−18.95), 325.5 (0.30), 313.5 (−10.49), 275 (4.80), 230 (−64.54).
2nd eluting enantiomer: tR = 21.45 min (Chiralpak IC, hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7). ECD [nm (Δε), hexane/dichloromethane/methanol 30
7). ECD [nm (Δε), hexane/dichloromethane/methanol 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 63
63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7]: 387.5 (5.05), 326 (−0.56), 313.5 (2.47), 276 (−2.02), 227.5 (17.86).
7]: 387.5 (5.05), 326 (−0.56), 313.5 (2.47), 276 (−2.02), 227.5 (17.86).
(6aS, 7R, 12bR, 17bS)- and (6aS, 7R, 12bR, 17bR)-11-methoxy-5,15-dimethyl-2-nitro-7-phenyl-5,17b-dihydro-6H,12bH,17H-chromeno[4′,3′:2,3]pyrano[3′,4':5,6]pyrano[3,4-c]quinolin-17-one [(6aS, 7R, 12bR, 17bS)-4be and (6aS, 7R, 12bR, 17bR)-5be] and (6aS, 7R, 12bR, 17bS)-11-methoxy-5,15-dimethyl-2-nitro-7-phenyl-5,17b-dihydro-6H,12bH,17H-chromeno[4′,3′:2,3]pyrano[3′,2′:5,6]pyrano[3,4-c]quinolin-17-one [(6aS, 7R, 12bR, 17bS)-6be]
The reaction of (R)-1b and 2e was carried out according to method A, and the crude product was purified by column chromatography (hexane/chloroform/ethyl acetate 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8
8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording (6aS, 7R, 12bR, 17bS)-4be as yellow powder (32%), (6aS, 7R, 12bR, 17bS)-6be as orange powder (8%) and (6aS, 7R, 12bR, 17bR)-5be as orange powder (14%).
1), affording (6aS, 7R, 12bR, 17bS)-4be as yellow powder (32%), (6aS, 7R, 12bR, 17bS)-6be as orange powder (8%) and (6aS, 7R, 12bR, 17bR)-5be as orange powder (14%).
Alternatively, the reaction was carried out at room temperature, affording (6aS, 7R, 12bR, 17bS)-4be as yellow powder (13%), (6aS, 7R, 12bR, 17bS)-6be as orange powder (14%) and (6aS, 7R, 12bR, 17bR)-5be as orange powder (31%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = +41 (c = 0.81 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (211 µg/4 ml): 372 (1.41), 323.5 (−0.56), 309.5 (−1.66), 293 (3.46), 269.5 (−2.09), 223 (17.38), 202.5 (−69.51), 195.5 (−32.93), 191.5 (−45.85). 1H-NMR (400 MHz, CDCl3): 2.23 (s, 3H, 3″-H), 2.81 (s, 3H, 2″-H), 3.11 (d, J = 12.8 Hz, 1H, 6-Ha), 3.39 (d, J = 12.8 Hz, 1H, 6-Hb), 3.74 (s, 3H, 1″-H), 3.81 (s, 1H, 17b-H), 4.89 (s, 1H, 12b-H), 4.93 (s, 1H, 7-H), 5.76 (s, 1H, 14-H), 6.42 (d, J = 9.2 Hz, 1H, 4-H), 6.81 (s, 1H, 12-H), 6.92 (s, 2H, 9-H and 10-H), 7.31 (d, J = 6.9 Hz, 1H, 2′-H and 6′-H), 7.31 (m, 3H, 3′-H, 4′-H and 5′-H), 7.86 (d, J = 9.2 Hz, 1H, 3-H), 8.05 (s, 1H, 1-H). 13C-NMR (100 MHz, CDCl3): 20.0 (C-3″), 32.0 (C-17b), 32.9 (C-6a), 39.8 (C-2″), 50.3 (C-6), 55.8 (C-1″), 72.8 (C-12b), 76.9 (C-7), 98.6 (C-17a), 100.0 (C-14), 110.2 (C-4), 115.1 (C-12), 118.1 (C-9 and C-10), 118.3 (C-12a), 122.4 (C-17c), 124.5 (C-3), 126.1 (C-1), 127.4 (C-2′ and C-6′), 128.5 (C-3′ and C-5′), 129.2 (C-4′), 134.0 (C-1′), 138.4 (C-2), 147.7 (C-8a), 148.4 (C-4a), 154.2 (C-11), 162.8 (C-13a and C-15), 165.1 (C-17). IR (KBr) ν: 1038, 1232, 1261, 1313, 1497, 1574, 1698 cm−1; HRMS: calcd for C31H26N2O7Na [M + Na]+ 561.1632, found 561.1636.
1). [α]D = +41 (c = 0.81 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (211 µg/4 ml): 372 (1.41), 323.5 (−0.56), 309.5 (−1.66), 293 (3.46), 269.5 (−2.09), 223 (17.38), 202.5 (−69.51), 195.5 (−32.93), 191.5 (−45.85). 1H-NMR (400 MHz, CDCl3): 2.23 (s, 3H, 3″-H), 2.81 (s, 3H, 2″-H), 3.11 (d, J = 12.8 Hz, 1H, 6-Ha), 3.39 (d, J = 12.8 Hz, 1H, 6-Hb), 3.74 (s, 3H, 1″-H), 3.81 (s, 1H, 17b-H), 4.89 (s, 1H, 12b-H), 4.93 (s, 1H, 7-H), 5.76 (s, 1H, 14-H), 6.42 (d, J = 9.2 Hz, 1H, 4-H), 6.81 (s, 1H, 12-H), 6.92 (s, 2H, 9-H and 10-H), 7.31 (d, J = 6.9 Hz, 1H, 2′-H and 6′-H), 7.31 (m, 3H, 3′-H, 4′-H and 5′-H), 7.86 (d, J = 9.2 Hz, 1H, 3-H), 8.05 (s, 1H, 1-H). 13C-NMR (100 MHz, CDCl3): 20.0 (C-3″), 32.0 (C-17b), 32.9 (C-6a), 39.8 (C-2″), 50.3 (C-6), 55.8 (C-1″), 72.8 (C-12b), 76.9 (C-7), 98.6 (C-17a), 100.0 (C-14), 110.2 (C-4), 115.1 (C-12), 118.1 (C-9 and C-10), 118.3 (C-12a), 122.4 (C-17c), 124.5 (C-3), 126.1 (C-1), 127.4 (C-2′ and C-6′), 128.5 (C-3′ and C-5′), 129.2 (C-4′), 134.0 (C-1′), 138.4 (C-2), 147.7 (C-8a), 148.4 (C-4a), 154.2 (C-11), 162.8 (C-13a and C-15), 165.1 (C-17). IR (KBr) ν: 1038, 1232, 1261, 1313, 1497, 1574, 1698 cm−1; HRMS: calcd for C31H26N2O7Na [M + Na]+ 561.1632, found 561.1636.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = +13 (c = 0.80 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (204 µg/4 ml): 386.5 (1.71), 299 (−4.03), 237.5 (20.15), 208 (−36.62), 193.5 (−107.66). 1H NMR (400 MHz, CDCl3) δ 2.30 (s, 3H, 3″-H), 2.90 (s, 3H, 2″-H), 3.18 (d, J = 12.9 Hz, 1H, 6-Ha), 3.49 (d, J = 12.9 Hz, 1H, 6-Hb), 3.82 (s, 3H, 1″-H), 4.13 (s, 1H, 17b-H), 5.07 (s, 1H, 7-H), 5.18 (s, 1H, 12b-H), 6.30 (d, J = 1.4 Hz, 1H, 16-H), 6.51 (d, J = 9.2 Hz, 1H, 4-H), 6.88 (m, 1H, 12-H), 7.01 (m, 2H, 9-H and 10-H), 7.20–7.31 (m, 2H, 2′-H and 6′-H), 7.31–7.46 (m, 3H, 3′-H, 4′-H and 5′-H), 7.95 (dd, J = 9.2 and 2.6 Hz, 1H, 3-H), 8.03 (dd, J = 2.6 and 1.4 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.5 (C-3″), 31.6 (C-17b), 33.3 (C-6a), 39.9 (C-2″), 50.1 (C-6), 55.9 (C-1″), 75.7 (C-12b), 76.9 (C-7), 100.1 (C-17a), 110.3 (C-4), 113.1 (C-16), 114.9 (C-12), 117.4 (C-12a), 118.5 (C-10), 118.9 (C-9), 122.5 (C-17c), 124.6 (C-3), 126.6 (C-1), 127.5 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.3 (C-4′), 133.8 (C-1′), 138.7 (C-2), 147.9 (C-8a), 148.3 (C-4a), 154.5 (C-11), 161.1 (C-13a), 161.8 (C-15), 179.7 (C-17). IR (KBr) ν: 1242, 1312, 1428, 1498, 1583, 1604, 1668, 2925 cm−1; HRMS: calcd for C31H26N2O7Na [M + Na]+ 561.1632, found 561.1636.
1). [α]D = +13 (c = 0.80 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (204 µg/4 ml): 386.5 (1.71), 299 (−4.03), 237.5 (20.15), 208 (−36.62), 193.5 (−107.66). 1H NMR (400 MHz, CDCl3) δ 2.30 (s, 3H, 3″-H), 2.90 (s, 3H, 2″-H), 3.18 (d, J = 12.9 Hz, 1H, 6-Ha), 3.49 (d, J = 12.9 Hz, 1H, 6-Hb), 3.82 (s, 3H, 1″-H), 4.13 (s, 1H, 17b-H), 5.07 (s, 1H, 7-H), 5.18 (s, 1H, 12b-H), 6.30 (d, J = 1.4 Hz, 1H, 16-H), 6.51 (d, J = 9.2 Hz, 1H, 4-H), 6.88 (m, 1H, 12-H), 7.01 (m, 2H, 9-H and 10-H), 7.20–7.31 (m, 2H, 2′-H and 6′-H), 7.31–7.46 (m, 3H, 3′-H, 4′-H and 5′-H), 7.95 (dd, J = 9.2 and 2.6 Hz, 1H, 3-H), 8.03 (dd, J = 2.6 and 1.4 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.5 (C-3″), 31.6 (C-17b), 33.3 (C-6a), 39.9 (C-2″), 50.1 (C-6), 55.9 (C-1″), 75.7 (C-12b), 76.9 (C-7), 100.1 (C-17a), 110.3 (C-4), 113.1 (C-16), 114.9 (C-12), 117.4 (C-12a), 118.5 (C-10), 118.9 (C-9), 122.5 (C-17c), 124.6 (C-3), 126.6 (C-1), 127.5 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.3 (C-4′), 133.8 (C-1′), 138.7 (C-2), 147.9 (C-8a), 148.3 (C-4a), 154.5 (C-11), 161.1 (C-13a), 161.8 (C-15), 179.7 (C-17). IR (KBr) ν: 1242, 1312, 1428, 1498, 1583, 1604, 1668, 2925 cm−1; HRMS: calcd for C31H26N2O7Na [M + Na]+ 561.1632, found 561.1636.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 4
4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). [α]D = −556 (c = 0.84 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (211 µg/4 ml): 401.5 (−5.77), 336.5 (−0.30), 309 (−9.31), 280 (5.38), 251 (−5.26), 232.5 (8.35), 207.5 (−105.70). 1H-NMR (400 MHz, CDCl3): 2.34 (s, 3H, 3″-H), 2.87 (s, 3H, 2″-H), 3.16 (d, J = 12.4 Hz, 1H, 6-Ha), 3.70 (d, J = 12.5 Hz, 1H, 6-Hb), 3.83 (s, 3H, 1″-H), 4.14 (s, 1H, 17b-H), 4.76 (s, 1H, 12b-H), 5.27 (s, 1H, 7-H), 5.94 (m, 1H, 14-H), 5.97 (d, J = 9.2 Hz, 1H, 4-H), 6.85–6.95 (m, 6H, 12-H and Ph), 6.98 (d, J = 3.0 Hz, 1H, 9-H), 7.01 (m, 1H, 10-H), 7.47 (dd, J = 2.5 and 1.2 Hz, 1H, 1-H), 7.57 (dd, J = 9.1 Hz and 2.5 Hz, 1H, 3-H). 13C-NMR (100 MHz, CDCl3): 19.9 (C-3″), 37.8 (C-2″), 37.9 (C-17b), 38.0 (C-6a), 53.4 (C-6), 55.9 (C-1″), 77.2 (C-7), 79.0 (C-12b), 96.9 (C-17a), 99.8 (C-14), 109.4 (C-4), 114.6 (C-12), 117.1 (C-17c), 117.7 (C-10), 118.7 (C-9), 122.4 (C-12a), 124.3 (C-3), 124.3 (C-1), 127.4 (C-2′ and C-6′), 129.2 (C-3′, C-4′ and C-5′), 136.8 (C-1′), 137.3 (C-2), 148.7 (C-8a), 152.2 (C-11), 153.8 (C-4a), 162.6 (C-15), 162.7 (C-13a), 165.3 (C-17). IR (KBr) ν: 1002, 1034, 1166, 1231, 1288, 1301, 1317, 1496, 1576, 1606, 1711, 2838, 2915 cm−1; HRMS: calcd for C31H26N2O7Na [M + Na]+ 561.1632, found 561.1635.
1). [α]D = −556 (c = 0.84 g/100 ml in CHCl3). ECD [nm (Δε), MeCN] (211 µg/4 ml): 401.5 (−5.77), 336.5 (−0.30), 309 (−9.31), 280 (5.38), 251 (−5.26), 232.5 (8.35), 207.5 (−105.70). 1H-NMR (400 MHz, CDCl3): 2.34 (s, 3H, 3″-H), 2.87 (s, 3H, 2″-H), 3.16 (d, J = 12.4 Hz, 1H, 6-Ha), 3.70 (d, J = 12.5 Hz, 1H, 6-Hb), 3.83 (s, 3H, 1″-H), 4.14 (s, 1H, 17b-H), 4.76 (s, 1H, 12b-H), 5.27 (s, 1H, 7-H), 5.94 (m, 1H, 14-H), 5.97 (d, J = 9.2 Hz, 1H, 4-H), 6.85–6.95 (m, 6H, 12-H and Ph), 6.98 (d, J = 3.0 Hz, 1H, 9-H), 7.01 (m, 1H, 10-H), 7.47 (dd, J = 2.5 and 1.2 Hz, 1H, 1-H), 7.57 (dd, J = 9.1 Hz and 2.5 Hz, 1H, 3-H). 13C-NMR (100 MHz, CDCl3): 19.9 (C-3″), 37.8 (C-2″), 37.9 (C-17b), 38.0 (C-6a), 53.4 (C-6), 55.9 (C-1″), 77.2 (C-7), 79.0 (C-12b), 96.9 (C-17a), 99.8 (C-14), 109.4 (C-4), 114.6 (C-12), 117.1 (C-17c), 117.7 (C-10), 118.7 (C-9), 122.4 (C-12a), 124.3 (C-3), 124.3 (C-1), 127.4 (C-2′ and C-6′), 129.2 (C-3′, C-4′ and C-5′), 136.8 (C-1′), 137.3 (C-2), 148.7 (C-8a), 152.2 (C-11), 153.8 (C-4a), 162.6 (C-15), 162.7 (C-13a), 165.3 (C-17). IR (KBr) ν: 1002, 1034, 1166, 1231, 1288, 1301, 1317, 1496, 1576, 1606, 1711, 2838, 2915 cm−1; HRMS: calcd for C31H26N2O7Na [M + Na]+ 561.1632, found 561.1635.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 17bR*)-4ce as pale yellow amorphous solid (10%) and rac-(6aS*, 12bR*, 17bR*)-6ce as yellow powder (26%).
1), affording rac-(6aS*, 12bR*, 17bR*)-4ce as pale yellow amorphous solid (10%) and rac-(6aS*, 12bR*, 17bR*)-6ce as yellow powder (26%).Alternatively, the reaction was carried out at 55 °C (there is no reaction at room temperature), affording rac-(6aS*, 12bR*, 17bR*)-4ce as pale yellow amorphous solid (16%) and rac-(6aS*, 12bR*, 17bR*)-6ce as yellow powder (6%).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.21 (s, 3H, 2′-H), 3.79 (s, 3H, 1′-H), 3.87 (d, J = 11.7 Hz, 1H, 7-Ha), 3.91 (d, J = 11.6 Hz, 1H, 7-Hb), 4.05 (d, J = 11.8 Hz, 1H, 6-Ha), 4.08 (s, 1H, 17b-H), 4.25 (d, J = 11.7 Hz, 1H, 6-Hb), 5.12 (s, 1H, 12b-H), 5.73 (s, 1H, 14-H), 6.81 (d, J = 8.1 Hz, 1H, 4-H), 6.86 (m, 2H, 9-H and 12-H), 6.88–7.00 (m, 2H, 10-H and 2-H), 7.15 (t, J = 7.6 Hz, 1H, 3-H), 7.52 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.0 (C-2′), 30.6 (C-6a), 31.6 (C-17b), 55.9 (C-1′), 66.2 (C-7), 68.5 (C-6), 70.9 (C-12b), 98.9 (C-17a), 100.2 (C-14), 115.5 (C-12), 117.0 (C-4), 118.1 (C-9), 118.2 (C-12a), 118.5 (C-10), 122.2 (C-2), 122.7 (C-17c), 128.5 (C-3), 130.1 (C-1), 146.7 (C-8a), 152.4 (C-4a), 154.2 (C-11), 161.9 (C-15), 162.0 (C-13a), 166.1 (C-17). IR (KBr) ν: 1046, 1215, 1269, 1489, 1501, 1583, 1697, 2833, 2875, 2925, 2953 cm−1; HRMS: calcd for C24H20O6Na [M + Na]+ 427.1152, found 427.1152.
1). 1H NMR (400 MHz, CDCl3) δ 2.21 (s, 3H, 2′-H), 3.79 (s, 3H, 1′-H), 3.87 (d, J = 11.7 Hz, 1H, 7-Ha), 3.91 (d, J = 11.6 Hz, 1H, 7-Hb), 4.05 (d, J = 11.8 Hz, 1H, 6-Ha), 4.08 (s, 1H, 17b-H), 4.25 (d, J = 11.7 Hz, 1H, 6-Hb), 5.12 (s, 1H, 12b-H), 5.73 (s, 1H, 14-H), 6.81 (d, J = 8.1 Hz, 1H, 4-H), 6.86 (m, 2H, 9-H and 12-H), 6.88–7.00 (m, 2H, 10-H and 2-H), 7.15 (t, J = 7.6 Hz, 1H, 3-H), 7.52 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.0 (C-2′), 30.6 (C-6a), 31.6 (C-17b), 55.9 (C-1′), 66.2 (C-7), 68.5 (C-6), 70.9 (C-12b), 98.9 (C-17a), 100.2 (C-14), 115.5 (C-12), 117.0 (C-4), 118.1 (C-9), 118.2 (C-12a), 118.5 (C-10), 122.2 (C-2), 122.7 (C-17c), 128.5 (C-3), 130.1 (C-1), 146.7 (C-8a), 152.4 (C-4a), 154.2 (C-11), 161.9 (C-15), 162.0 (C-13a), 166.1 (C-17). IR (KBr) ν: 1046, 1215, 1269, 1489, 1501, 1583, 1697, 2833, 2875, 2925, 2953 cm−1; HRMS: calcd for C24H20O6Na [M + Na]+ 427.1152, found 427.1152.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.20 (s, 3H, 2′-H), 3.79 (s, 3H, 1′-H), 3.92 (m, 2H, 7-H), 4.07 (d, J = 11.8 Hz, 1H, 6-Ha), 4.24 (d, J = 11.8 Hz, 1H, 6-Hb), 4.29 (s, 1H, 17b-H), 5.34 (s, 1H, 12b-H), 6.13 (s, 1H, 16-H), 6.80 (d, J = 8.1 Hz, 1H, 4-H), 6.86 (m, 2H, 9-H and 12-H), 6.91 (d, J = 7.5 Hz, 1H, 2-H), 6.95 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.14 (t, J = 7.6 Hz, 1H, 3-H), 7.41 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.3 (C-2′), 31.0 (C-17b), 31.0 (C-6a), 55.9 (C-1′), 66.1 (C-7), 68.2 (C-6), 73.7 (C-12b), 100.1 (C-17a), 112.7 (C-16), 115.2 (C-12), 116.9 (C-4), 117.3 (C-12a), 118.3 (C-9), 119.1 (C-10), 122.3 (C-2), 122.7 (C-17c), 128.4 (C-3), 130.4 (C-1), 146.8 (C-8a), 152.3 (C-4a), 154.3 (C-11), 160.7 (C-13a), 161.2 (C-15), 180.8 (C-17). IR (KBr) ν: 1019, 1047, 1217, 1245, 1262, 1422, 1498, 1587, 1667 cm−1; HRMS: calcd for C24H20O6Na [M + Na]+ 427.1152, found 427.1154.
1). 1H NMR (400 MHz, CDCl3) δ 2.20 (s, 3H, 2′-H), 3.79 (s, 3H, 1′-H), 3.92 (m, 2H, 7-H), 4.07 (d, J = 11.8 Hz, 1H, 6-Ha), 4.24 (d, J = 11.8 Hz, 1H, 6-Hb), 4.29 (s, 1H, 17b-H), 5.34 (s, 1H, 12b-H), 6.13 (s, 1H, 16-H), 6.80 (d, J = 8.1 Hz, 1H, 4-H), 6.86 (m, 2H, 9-H and 12-H), 6.91 (d, J = 7.5 Hz, 1H, 2-H), 6.95 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.14 (t, J = 7.6 Hz, 1H, 3-H), 7.41 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.3 (C-2′), 31.0 (C-17b), 31.0 (C-6a), 55.9 (C-1′), 66.1 (C-7), 68.2 (C-6), 73.7 (C-12b), 100.1 (C-17a), 112.7 (C-16), 115.2 (C-12), 116.9 (C-4), 117.3 (C-12a), 118.3 (C-9), 119.1 (C-10), 122.3 (C-2), 122.7 (C-17c), 128.4 (C-3), 130.4 (C-1), 146.8 (C-8a), 152.3 (C-4a), 154.3 (C-11), 160.7 (C-13a), 161.2 (C-15), 180.8 (C-17). IR (KBr) ν: 1019, 1047, 1217, 1245, 1262, 1422, 1498, 1587, 1667 cm−1; HRMS: calcd for C24H20O6Na [M + Na]+ 427.1152, found 427.1154.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9
9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 17bS*)-4af as yellow powder (31%), rac-(6aS*, 12bR*, 17bS*)-6af as yellow powder (31%), rac-(6aS*, 12bR*, 17bS*)-5af as yellow powder (13%) and rac-(6aS*, 12bR*, 17bS*)-7af as yellow powder (7%).
1), affording rac-(6aS*, 12bR*, 17bS*)-4af as yellow powder (31%), rac-(6aS*, 12bR*, 17bS*)-6af as yellow powder (31%), rac-(6aS*, 12bR*, 17bS*)-5af as yellow powder (13%) and rac-(6aS*, 12bR*, 17bS*)-7af as yellow powder (7%).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.33 (s, 3H, 4′-H), 2.95 (s, 3H, 2′-H), 3.23 (d, J = 13.1 Hz, 1H, 6-Ha), 3.62 (s, 3H, 3′-H), 3.65 (d, J = 13.1 Hz, 1H, 6-Hb), 3.80 (s, 3H, 1′-H), 3.87 (s, 2H, 7-H), 4.25 (s, 1H, 17b-H), 4.82 (s, 1H, 12b-H), 5.74 (s, 1H, 14-H), 6.55 (d, J = 9.2 Hz, 1H, 4-H), 6.81–6.90 (m, 2H, 9-H and 12-H), 6.94 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 8.00 (dd, J = 9.1 and 2.7 Hz, 1H, 3-H), 8.07 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 21.1 (C-4′), 30.6 (C-6a), 31.4 (C-3′), 33.3 (C-17b), 39.7 (C-2′), 55.0 (C-6), 55.8 (C-1′), 68.0 (C-7), 70.6 (C-12b), 100.1 (C-14), 104.5 (C-17a), 110.0 (C-4), 115.5 (C-12), 117.9 (C-10), 117.9 (C-9), 119.4 (C-12a), 122.4 (C-17c), 124.6 (C-3), 126.7 (C-1), 138.5 (C-2), 146.3 (C-15), 146.7 (C-8a), 149.2 (C-4a), 154.0 (C-11), 158.0 (C-13a), 164.8 (C-17). IR (KBr) ν: 1215, 1229, 1495, 1545, 1644, 1717, 1738, 2923, 2969 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1636.
1). 1H NMR (400 MHz, CDCl3) δ 2.33 (s, 3H, 4′-H), 2.95 (s, 3H, 2′-H), 3.23 (d, J = 13.1 Hz, 1H, 6-Ha), 3.62 (s, 3H, 3′-H), 3.65 (d, J = 13.1 Hz, 1H, 6-Hb), 3.80 (s, 3H, 1′-H), 3.87 (s, 2H, 7-H), 4.25 (s, 1H, 17b-H), 4.82 (s, 1H, 12b-H), 5.74 (s, 1H, 14-H), 6.55 (d, J = 9.2 Hz, 1H, 4-H), 6.81–6.90 (m, 2H, 9-H and 12-H), 6.94 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 8.00 (dd, J = 9.1 and 2.7 Hz, 1H, 3-H), 8.07 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 21.1 (C-4′), 30.6 (C-6a), 31.4 (C-3′), 33.3 (C-17b), 39.7 (C-2′), 55.0 (C-6), 55.8 (C-1′), 68.0 (C-7), 70.6 (C-12b), 100.1 (C-14), 104.5 (C-17a), 110.0 (C-4), 115.5 (C-12), 117.9 (C-10), 117.9 (C-9), 119.4 (C-12a), 122.4 (C-17c), 124.6 (C-3), 126.7 (C-1), 138.5 (C-2), 146.3 (C-15), 146.7 (C-8a), 149.2 (C-4a), 154.0 (C-11), 158.0 (C-13a), 164.8 (C-17). IR (KBr) ν: 1215, 1229, 1495, 1545, 1644, 1717, 1738, 2923, 2969 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1636.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.36 (s, 3H, 4′-H), 3.03 (s, 3H, 2′-H), 3.15 (d, J = 11.7 Hz, 1H, 6-Ha), 3.19 (d, J = 11.7 Hz, 1H, 6-Hb), 3.29 (dd, J = 11.6 and 1.5 Hz, 1H, 7-Ha), 3.56 (s, 3H, 3′-H), 3.80 (s, 3H, 1′-H), 3.86 (d, J = 11.5 Hz, 1H, 7-Hb), 4.07 (s, 1H, 17b-H), 4.60 (d, J = 1.2 Hz, 1H, 12b-H), 5.85 (s, 1H, 14-H), 6.63 (d, J = 9.1 Hz, 1H, 4-H), 6.76–6.84 (m, 2H, 9-H and 12-H), 6.92 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.69 (d, J = 2.5 Hz, 1H, 1-H), 8.09 (dd, J = 9.1 and 2.5 Hz, 1H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-4′), 31.3 (C-3′), 35.3 (C-6a), 37.6 (C-17b), 38.2 (C-2′), 54.8 (C-6), 56.0 (C-1′), 63.5 (C-7), 75.0 (C-12b), 100.2 (C-14), 102.1 (C-17a), 109.6 (C-4), 115.0 (C-9), 117.6 (C-12), 118.1 (C-12a), 118.4 (C-10), 120.1 (C-17c), 123.1 (C-1), 124.7 (C-3), 137.9 (C-2), 146.3 (C-15), 148.5 (C-8a), 152.1 (C-4a), 153.7 (C-11), 160.8 (C-13a), 163.4 (C-17). IR (KBr) ν: 1216, 1227, 1365, 1498, 1571, 1636, 1717, 1738, 2926, 2970, 3003 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1636.
1). 1H NMR (400 MHz, CDCl3) δ 2.36 (s, 3H, 4′-H), 3.03 (s, 3H, 2′-H), 3.15 (d, J = 11.7 Hz, 1H, 6-Ha), 3.19 (d, J = 11.7 Hz, 1H, 6-Hb), 3.29 (dd, J = 11.6 and 1.5 Hz, 1H, 7-Ha), 3.56 (s, 3H, 3′-H), 3.80 (s, 3H, 1′-H), 3.86 (d, J = 11.5 Hz, 1H, 7-Hb), 4.07 (s, 1H, 17b-H), 4.60 (d, J = 1.2 Hz, 1H, 12b-H), 5.85 (s, 1H, 14-H), 6.63 (d, J = 9.1 Hz, 1H, 4-H), 6.76–6.84 (m, 2H, 9-H and 12-H), 6.92 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 7.69 (d, J = 2.5 Hz, 1H, 1-H), 8.09 (dd, J = 9.1 and 2.5 Hz, 1H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-4′), 31.3 (C-3′), 35.3 (C-6a), 37.6 (C-17b), 38.2 (C-2′), 54.8 (C-6), 56.0 (C-1′), 63.5 (C-7), 75.0 (C-12b), 100.2 (C-14), 102.1 (C-17a), 109.6 (C-4), 115.0 (C-9), 117.6 (C-12), 118.1 (C-12a), 118.4 (C-10), 120.1 (C-17c), 123.1 (C-1), 124.7 (C-3), 137.9 (C-2), 146.3 (C-15), 148.5 (C-8a), 152.1 (C-4a), 153.7 (C-11), 160.8 (C-13a), 163.4 (C-17). IR (KBr) ν: 1216, 1227, 1365, 1498, 1571, 1636, 1717, 1738, 2926, 2970, 3003 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1636.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.31 (s, 3H, 4′-H), 2.97 (s, 3H, 2′-H), 3.24 (d, J = 13.2 Hz, 1H, 6-Ha), 3.35 (s, 3H, 3′-H), 3.66 (d, J = 13.2 Hz, 1H, 6-Hb), 3.83 (m, 4H, 1′-H and 7-Ha), 3.91 (dd, J = 11.5 and 1.0 Hz, 1H, 7-Hb), 4.42 (s, 1H, 17b-H), 5.05 (s, 1H, 12b-H), 6.30 (s, 1H, 16-H), 6.56 (d, J = 9.1 Hz, 1H, 4-H), 6.85–6.93 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 8.00 (dd, J = 9.1 and 2.4 Hz, 1H, 3-H), 8.03 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.4 (C-4′), 30.8 (C-6a), 31.9 (C-3′), 32.4 (C-17b), 39.7 (C-2′), 54.7 (C-6), 55.9 (C-1′), 68.0 (C-7), 73.3 (C-12b), 105.0 (C-17a), 110.0 (C-4), 115.0 (C-16), 115.5 (C-12), 117.9 (C-10), 118.3 (C-9), 118.6 (C-12a), 121.9 (C-17c), 124.7 (C-3), 126.8 (C-1), 138.6 (C-2), 145.7 (C-15), 146.9 (C-8a), 149.1 (C-4a), 151.4 (C-13a), 154.2 (C-11), 178.1 (C-17). IR (KBr) ν: 1234, 1297, 1316, 1494, 1638 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1638.
1). 1H NMR (400 MHz, CDCl3) δ 2.31 (s, 3H, 4′-H), 2.97 (s, 3H, 2′-H), 3.24 (d, J = 13.2 Hz, 1H, 6-Ha), 3.35 (s, 3H, 3′-H), 3.66 (d, J = 13.2 Hz, 1H, 6-Hb), 3.83 (m, 4H, 1′-H and 7-Ha), 3.91 (dd, J = 11.5 and 1.0 Hz, 1H, 7-Hb), 4.42 (s, 1H, 17b-H), 5.05 (s, 1H, 12b-H), 6.30 (s, 1H, 16-H), 6.56 (d, J = 9.1 Hz, 1H, 4-H), 6.85–6.93 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 8.9 and 3.0 Hz, 1H, 10-H), 8.00 (dd, J = 9.1 and 2.4 Hz, 1H, 3-H), 8.03 (bs, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.4 (C-4′), 30.8 (C-6a), 31.9 (C-3′), 32.4 (C-17b), 39.7 (C-2′), 54.7 (C-6), 55.9 (C-1′), 68.0 (C-7), 73.3 (C-12b), 105.0 (C-17a), 110.0 (C-4), 115.0 (C-16), 115.5 (C-12), 117.9 (C-10), 118.3 (C-9), 118.6 (C-12a), 121.9 (C-17c), 124.7 (C-3), 126.8 (C-1), 138.6 (C-2), 145.7 (C-15), 146.9 (C-8a), 149.1 (C-4a), 151.4 (C-13a), 154.2 (C-11), 178.1 (C-17). IR (KBr) ν: 1234, 1297, 1316, 1494, 1638 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1638.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.34 (s, 3H, 4′-H), 3.02 (s, 3H, 2′-H), 3.16 (d, J = 11.2 Hz, 1H, 6-Ha), 3.22 (d, J = 11.2 Hz, 1H, 6-Hb), 3.32 (d, J = 11.9 Hz, 1H. 7-Ha), 3.45 (s, 3H, 3′-H), 3.81 (s, 3H, 1′-H), 3.85 (d, J = 11.9 Hz, 1H, 7-Hb), 4.20 (s, 1H, 17b-H), 4.79 (s, 1H, 12b-H), 6.27 (s, 1H, 16-H), 6.61 (d, J = 9.1 Hz, 1H, 4-H), 6.79–6.87 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.68 (d, J = 2.5 Hz, 1H, 1-H), 8.08 (dd, J = 9.1 and 2.5 Hz, 1H, 3-H). 13C NMR (90 MHz, CDCl3) δ 20.2 (C-4′), 32.0 (C-3′), 35.2 (C-6a), 37.7 (C-17b), 38.3 (C-2′), 54.5 (C-6), 56.0 (C-1′), 63.5 (C-7), 76.9 (C-12b) 102.2 (C-17a), 109.6 (C-4), 115.3 (C-12), 115.8 (C-16), 117.2 (C-12a), 117.8 (C-9), 118.4 (C-10), 119.9 (C-17c), 123.2 (C-1), 124.8 (C-3), 138.2 (C-2), 145.3 (C-15), 148.8 (C-8a), 151.9 (C-4a), 153.7 (C-11), 177.9 (C-17). IR (KBr) ν: 1217, 1364, 1603, 1639, 1716, 1738, 2925, 2970 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1639.
1). 1H NMR (400 MHz, CDCl3) δ 2.34 (s, 3H, 4′-H), 3.02 (s, 3H, 2′-H), 3.16 (d, J = 11.2 Hz, 1H, 6-Ha), 3.22 (d, J = 11.2 Hz, 1H, 6-Hb), 3.32 (d, J = 11.9 Hz, 1H. 7-Ha), 3.45 (s, 3H, 3′-H), 3.81 (s, 3H, 1′-H), 3.85 (d, J = 11.9 Hz, 1H, 7-Hb), 4.20 (s, 1H, 17b-H), 4.79 (s, 1H, 12b-H), 6.27 (s, 1H, 16-H), 6.61 (d, J = 9.1 Hz, 1H, 4-H), 6.79–6.87 (m, 2H, 9-H and 12-H), 6.97 (dd, J = 9.0 and 3.0 Hz, 1H, 10-H), 7.68 (d, J = 2.5 Hz, 1H, 1-H), 8.08 (dd, J = 9.1 and 2.5 Hz, 1H, 3-H). 13C NMR (90 MHz, CDCl3) δ 20.2 (C-4′), 32.0 (C-3′), 35.2 (C-6a), 37.7 (C-17b), 38.3 (C-2′), 54.5 (C-6), 56.0 (C-1′), 63.5 (C-7), 76.9 (C-12b) 102.2 (C-17a), 109.6 (C-4), 115.3 (C-12), 115.8 (C-16), 117.2 (C-12a), 117.8 (C-9), 118.4 (C-10), 119.9 (C-17c), 123.2 (C-1), 124.8 (C-3), 138.2 (C-2), 145.3 (C-15), 148.8 (C-8a), 151.9 (C-4a), 153.7 (C-11), 177.9 (C-17). IR (KBr) ν: 1217, 1364, 1603, 1639, 1716, 1738, 2925, 2970 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 498.1636, found 498.1639.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 7R*, 12bR*, 17bS*)-4bf as yellow powder (29%), rac-(6aS*, 7R*, 12bR*, 17bR*)-5bf as yellow powder (29%) and rac-(6aS*, 7R*, 12bR*, 17bS*)-6bf as yellow powder (13%).
1), affording rac-(6aS*, 7R*, 12bR*, 17bS*)-4bf as yellow powder (29%), rac-(6aS*, 7R*, 12bR*, 17bR*)-5bf as yellow powder (29%) and rac-(6aS*, 7R*, 12bR*, 17bS*)-6bf as yellow powder (13%).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.37 (s, 3H, 4″-H), 2.87 (s, 3H, 2″-H), 3.14 (d, J = 12.8 Hz, 1H, 6-Ha), 3.45 (d, J = 12.8 Hz, 1H, 6-Hb), 3.69 (s, 3H, 3″-H), 3.81 (s, 3H, 1″-H), 4.10 (s, 1H, 17b-H), 4.87 (s, 1H, 12b-H), 5.05 (s, 1H, 7-H), 5.77 (s, 1H, 14-H), 6.47 (d, J = 9.1 Hz, 1H, 4-H), 6.89 (s, 1H, 12-H), 6.98 (s, 2H, 9-H and 10-H), 7.26 (m, 2H, 2′-H and 6′-H), 7.30–7.46 (m, 3H, 3′-H, 4′-H and 5′-H), 7.93 (d, J = 9.1 Hz, 1H, 3-H), 8.09 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 21.2 (C-4″), 31.6 (C-3″), 32.7 (C-17b), 32.9 (C-6a), 39.8 (C-2″), 50.6 (C-6), 55.9 (C-1″), 71.5 (C-12b), 77.0 (C-7), 99.8 (C-14), 104.8 (C-17a), 110.0 (C-4), 115.2 (C-12), 118.0 (C-10), 118.0 (C-9), 119.4 (C-12a), 123.4 (C-17c), 124.3 (C-3), 126.9 (C-1), 127.5 (C-2′ and C-6′), 128.5 (C-3′ and C-5′), 128.9 (C-4′), 134.6 (C-1′), 138.5 (C-2), 146.6 (C-15), 147.8 (C-8a), 148.7 (C-4a), 154.2 (C-11), 158.5 (C-13a), 164.2 (C-17). IR (KBr) ν: 1226, 1364, 1499, 1573, 1643, 1715, 2970, 3005 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 574.1949, found 574.1954.
1). 1H NMR (400 MHz, CDCl3) δ 2.37 (s, 3H, 4″-H), 2.87 (s, 3H, 2″-H), 3.14 (d, J = 12.8 Hz, 1H, 6-Ha), 3.45 (d, J = 12.8 Hz, 1H, 6-Hb), 3.69 (s, 3H, 3″-H), 3.81 (s, 3H, 1″-H), 4.10 (s, 1H, 17b-H), 4.87 (s, 1H, 12b-H), 5.05 (s, 1H, 7-H), 5.77 (s, 1H, 14-H), 6.47 (d, J = 9.1 Hz, 1H, 4-H), 6.89 (s, 1H, 12-H), 6.98 (s, 2H, 9-H and 10-H), 7.26 (m, 2H, 2′-H and 6′-H), 7.30–7.46 (m, 3H, 3′-H, 4′-H and 5′-H), 7.93 (d, J = 9.1 Hz, 1H, 3-H), 8.09 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 21.2 (C-4″), 31.6 (C-3″), 32.7 (C-17b), 32.9 (C-6a), 39.8 (C-2″), 50.6 (C-6), 55.9 (C-1″), 71.5 (C-12b), 77.0 (C-7), 99.8 (C-14), 104.8 (C-17a), 110.0 (C-4), 115.2 (C-12), 118.0 (C-10), 118.0 (C-9), 119.4 (C-12a), 123.4 (C-17c), 124.3 (C-3), 126.9 (C-1), 127.5 (C-2′ and C-6′), 128.5 (C-3′ and C-5′), 128.9 (C-4′), 134.6 (C-1′), 138.5 (C-2), 146.6 (C-15), 147.8 (C-8a), 148.7 (C-4a), 154.2 (C-11), 158.5 (C-13a), 164.2 (C-17). IR (KBr) ν: 1226, 1364, 1499, 1573, 1643, 1715, 2970, 3005 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 574.1949, found 574.1954.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.40 (s, 3H, 4″-H), 2.87 (s, 3H, 2″-H), 3.17 (d, J = 12.3 Hz, 1H, 6-Ha), 3.56 (s, 3H, 3″-H), 3.67 (dd, J = 12.3 and 1.2 Hz, 1H, 6-Hb), 3.82 (s, 3H, 1″-H), 4.20 (s, 1H, 17b-H), 4.62 (s, 1H, 12b-H), 5.37 (s, 1H, 7-H), 5.91 (m, 2H, 4-H and 14-H), 6.47–7.00 (m, 8H, 9-H, 10-H, 12-H and Ph), 7.42 (dd, J = 2.6 and 1.2 Hz, 1H, 1-H), 7.54 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-4″), 31.3 (C-3″), 37.8 (C-2″), 38.0 (C-6a), 39.3 (C-17b), 53.9 (C-6), 56.0 (C-1″), 77.6 (C-7), 78.2 (C-12b), 100.0 (C-14), 102.7 (C-17a), 109.3 (C-4), 114.6 (C-12), 117.6 (C-9), 118.0 (C-12a), 118.5 (C-10), 118.7 (C-17c), 124.2 (C-3), 125.2 (C-1), 128.2 (C-3′ and C-5′), 137.3 (C-2), 137.5 (C-1′), 146.4 (C-15), 148.8 (C-8a), 152.6 (C-4a), 153.8 (C-11), 161.4 (C-13a), 163.3 (C-17). IR (KBr) ν: 1224, 1284, 1365, 1494, 1644, 1715, 2927, 2969, 3003 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 574.1949, found 574.1956.
1). 1H NMR (400 MHz, CDCl3) δ 2.40 (s, 3H, 4″-H), 2.87 (s, 3H, 2″-H), 3.17 (d, J = 12.3 Hz, 1H, 6-Ha), 3.56 (s, 3H, 3″-H), 3.67 (dd, J = 12.3 and 1.2 Hz, 1H, 6-Hb), 3.82 (s, 3H, 1″-H), 4.20 (s, 1H, 17b-H), 4.62 (s, 1H, 12b-H), 5.37 (s, 1H, 7-H), 5.91 (m, 2H, 4-H and 14-H), 6.47–7.00 (m, 8H, 9-H, 10-H, 12-H and Ph), 7.42 (dd, J = 2.6 and 1.2 Hz, 1H, 1-H), 7.54 (dd, J = 9.1 and 2.6 Hz, 1H, 3-H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-4″), 31.3 (C-3″), 37.8 (C-2″), 38.0 (C-6a), 39.3 (C-17b), 53.9 (C-6), 56.0 (C-1″), 77.6 (C-7), 78.2 (C-12b), 100.0 (C-14), 102.7 (C-17a), 109.3 (C-4), 114.6 (C-12), 117.6 (C-9), 118.0 (C-12a), 118.5 (C-10), 118.7 (C-17c), 124.2 (C-3), 125.2 (C-1), 128.2 (C-3′ and C-5′), 137.3 (C-2), 137.5 (C-1′), 146.4 (C-15), 148.8 (C-8a), 152.6 (C-4a), 153.8 (C-11), 161.4 (C-13a), 163.3 (C-17). IR (KBr) ν: 1224, 1284, 1365, 1494, 1644, 1715, 2927, 2969, 3003 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 574.1949, found 574.1956.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.34 (s, 3H, 4″-H), 2.88 (s, 3H, 2″-H), 3.13 (d, J = 12.9 Hz, 1H, 6-Ha), 3.39 (s, 3H, 3″-H), 3.48 (d, J = 12.9 Hz, 1H, 6-Hb), 3.83 (s, 3H, 1″-H), 4.27 (s, 1H, 17b-H), 5.03 (s, 1H, 7-H), 5.08 (s, 1H, 12b-H), 6.38 (s, 1H, 16-H), 6.47 (d, J = 9.2 Hz, 1H, 4-H), 6.92 (s, 1H, 12-H), 7.00 (m, 2H, 9-H and 10-H), 7.29 (d, J = 7.2 Hz, 2H, 2′-H and 6′-H), 7.35 (d, J = 7.2 Hz, 1H, 4′-H), 7.40 (m, 2H, 3′-H and 5′-H), 7.93 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.05 (dd, J = 2.5 and 1.2 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.5 (C-4″), 31.9 (C-17b and C-3″), 33.0 (C-6a), 39.8 (C-2″), 50.3 (C-6), 56.0 (C-1″), 74.2 (C-12b), 77.0 (C-7), 105.2 (C-17a), 109.9 (C-4), 115.4 (C-12), 115.5 (C-16), 118.0 (C-10), 118.3 (C-9), 118.6 (C-12a), 123.0 (C-17c), 124.4 (C-3), 127.0 (C-1), 127.6 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.1 (C-4′), 134.0 (C-1′), 138.7 (C-2), 145.7 (C-15), 148.0 (C-8a), 148.5 (C-4a), 151.9 (C-13a), 154.3 (C-11), 177.6 (C-17). IR (KBr) ν: 1224, 1364, 1494, 1644, 1714, 2924, 2969, 3005 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 574.1949, found 574.1954.
1). 1H NMR (400 MHz, CDCl3) δ 2.34 (s, 3H, 4″-H), 2.88 (s, 3H, 2″-H), 3.13 (d, J = 12.9 Hz, 1H, 6-Ha), 3.39 (s, 3H, 3″-H), 3.48 (d, J = 12.9 Hz, 1H, 6-Hb), 3.83 (s, 3H, 1″-H), 4.27 (s, 1H, 17b-H), 5.03 (s, 1H, 7-H), 5.08 (s, 1H, 12b-H), 6.38 (s, 1H, 16-H), 6.47 (d, J = 9.2 Hz, 1H, 4-H), 6.92 (s, 1H, 12-H), 7.00 (m, 2H, 9-H and 10-H), 7.29 (d, J = 7.2 Hz, 2H, 2′-H and 6′-H), 7.35 (d, J = 7.2 Hz, 1H, 4′-H), 7.40 (m, 2H, 3′-H and 5′-H), 7.93 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.05 (dd, J = 2.5 and 1.2 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.5 (C-4″), 31.9 (C-17b and C-3″), 33.0 (C-6a), 39.8 (C-2″), 50.3 (C-6), 56.0 (C-1″), 74.2 (C-12b), 77.0 (C-7), 105.2 (C-17a), 109.9 (C-4), 115.4 (C-12), 115.5 (C-16), 118.0 (C-10), 118.3 (C-9), 118.6 (C-12a), 123.0 (C-17c), 124.4 (C-3), 127.0 (C-1), 127.6 (C-2′ and C-6′), 128.6 (C-3′ and C-5′), 129.1 (C-4′), 134.0 (C-1′), 138.7 (C-2), 145.7 (C-15), 148.0 (C-8a), 148.5 (C-4a), 151.9 (C-13a), 154.3 (C-11), 177.6 (C-17). IR (KBr) ν: 1224, 1364, 1494, 1644, 1714, 2924, 2969, 3005 cm−1; HRMS: calcd for C32H29N3O6Na [M + Na]+ 574.1949, found 574.1954.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 12bR*, 17bR*)-4cf as white powder (11%).
1), affording rac-(6aS*, 12bR*, 17bR*)-4cf as white powder (11%).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1). 1H NMR (400 MHz, CDCl3) δ 2.28 (s, 3H, 3′-H), 3.58 (s, 3H, 2′-H), 3.79 (s, 3H, 1′-H), 3.83 (dd, J = 11.6 and 1.5 Hz, 1H, 7-Ha), 3.90 (d, J = 11.6 Hz, 1H, 7-Hb), 4.09 (d, J = 11.7 Hz, 1H, 6-Ha), 4.20–4.31 (m, 2H, 6-Hb and 17b-H), 5.06 (d, J = 1.5 Hz, 1H, 12b-H), 5.71 (s, 1H, 14-H), 6.79 (dd, J = 7.7 and 1.0 Hz, 1H, 4-H), 6.83 (d, J = 8.8 Hz, 1H, 9-H), 6.85–6.96 (m, 3H, 2-H, 10-H and 12-H), 7.11 (t, J = 7.7 Hz, 1H, 3-H), 7.48 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-3′), 30.4 (C-6a), 31.3 (C-2′), 32.2 (C-17b), 55.8 (C-1′), 66.4 (C-7), 68.7 (C-6), 69.6 (C-12b), 100.3 (C-14), 105.1 (C-17a), 115.5 (C-2), 116.7 (C-4), 117.9 (C-9), 118.1 (C-10), 119.2 (C-12a), 121.9 (C-12), 123.5 (C-17c), 128.0 (C-3), 130.5 (C-1), 145.5 (C-15), 146.6 (C-8a), 152.4 (C-4a), 154.0 (C-11), 158.0 (C-13a), 165.2 (C-17). IR (KBr) ν: 1216, 1227, 1276, 1365, 1497, 1577, 1643, 1715, 2938, 2970 cm−1; HRMS: calcd for C25H23NO5Na [M + Na]+ 440.1468, found 440.1468.
1). 1H NMR (400 MHz, CDCl3) δ 2.28 (s, 3H, 3′-H), 3.58 (s, 3H, 2′-H), 3.79 (s, 3H, 1′-H), 3.83 (dd, J = 11.6 and 1.5 Hz, 1H, 7-Ha), 3.90 (d, J = 11.6 Hz, 1H, 7-Hb), 4.09 (d, J = 11.7 Hz, 1H, 6-Ha), 4.20–4.31 (m, 2H, 6-Hb and 17b-H), 5.06 (d, J = 1.5 Hz, 1H, 12b-H), 5.71 (s, 1H, 14-H), 6.79 (dd, J = 7.7 and 1.0 Hz, 1H, 4-H), 6.83 (d, J = 8.8 Hz, 1H, 9-H), 6.85–6.96 (m, 3H, 2-H, 10-H and 12-H), 7.11 (t, J = 7.7 Hz, 1H, 3-H), 7.48 (d, J = 7.8 Hz, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 21.0 (C-3′), 30.4 (C-6a), 31.3 (C-2′), 32.2 (C-17b), 55.8 (C-1′), 66.4 (C-7), 68.7 (C-6), 69.6 (C-12b), 100.3 (C-14), 105.1 (C-17a), 115.5 (C-2), 116.7 (C-4), 117.9 (C-9), 118.1 (C-10), 119.2 (C-12a), 121.9 (C-12), 123.5 (C-17c), 128.0 (C-3), 130.5 (C-1), 145.5 (C-15), 146.6 (C-8a), 152.4 (C-4a), 154.0 (C-11), 158.0 (C-13a), 165.2 (C-17). IR (KBr) ν: 1216, 1227, 1276, 1365, 1497, 1577, 1643, 1715, 2938, 2970 cm−1; HRMS: calcd for C25H23NO5Na [M + Na]+ 440.1468, found 440.1468.Domino Knoevenagel-IMHDA reactions of 5,6-dihydro-2H-pyran derivatives 8a–b
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), affording rac-(6aS*, 10aS*, 17bS*)-10aa as yellow amorphous solid (44%). Rf = 0.17 (hexane/ethyl acetate 1
2), affording rac-(6aS*, 10aS*, 17bS*)-10aa as yellow amorphous solid (44%). Rf = 0.17 (hexane/ethyl acetate 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).1H NMR (400 MHz, CDCl3) δ 2.09 (d, J = 14.6 Hz, 1H, 10-Ha), 2.27 (m, 1H, 10-Hb), 2.41 (s, 3H, 2′-H), 3.13 (s, 3H, 1′-H), 3.42 (d, J = 12.0 Hz, 1H, 7-Ha), 3.50 (d, J = 11.7 Hz, 1H, 7-Hb), 3.55 (d, J = 13.1 Hz, 1H, 6-Ha), 3.68 (t, J = 11.4 Hz, 1H, 9-Ha), 3.86 (d, J = 13.2 Hz, 1H, 6-Hb), 3.88–3.98 (m, 2H, 17b-H and 9-Hb), 4.52 (s, 1H, 10a-H), 6.55 (d, J = 9.2 Hz, 1H, 4-H), 7.28 (d, J = 8.4 Hz, 1H, 15-H), 7.38 (d, J = 8.4 Hz, 1H, 14-H), 7.57 (s, 1H, 12-H), 7.93 (dd, J = 9.1 and 2.4 Hz, 1H, 3-H), 8.10 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 20.9 (C-2′), 27.7 (C-10), 31.6 (C-6a), 34.1 (C-17b), 39.8 (C-1′), 55.9 (C-6), 62.8 (C-9), 69.4 (C-7), 72.1 (C-10a), 101.3 (C-17a), 110.3 (C-4), 114.4 (C-11b), 116.7 (C-15), 121.8 (C-17c), 122.3 (C-12), 124.5 (C-3), 126.2 (C-1), 133.5 (C-14), 134.0 (C-13), 138.3 (C-2), 149.1 (C-15a), 151.0 (C-4a), 159.0 (C-17), 164.0 (C-11a). IR (KBr) ν: 1305, 1319, 1497, 1581, 1630, 1702, 2859, 2925 cm−1; HRMS: calcd for C24H22N2O6Na [M + Na]+ 457.1370, found 457.1370.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2).
2).1H NMR (400 MHz, CDCl3) δ 2.09 (d, J = 14.7 Hz, 1H, 10-Ha), 2.21–2.30 (m, 1H, 10-Hb), 2.32 (s, 3H, 3′), 2.37 (s, 3H, 2′-H), 3.13 (s, 3H, 1′-H), 3.42 (d, J = 12.1 Hz, 1H, 7-Ha), 3.48 (d, J = 12.1 Hz, 1H, 7-Hb), 3.54 (d, J = 13.1 Hz, 1H, 6-Ha), 3.69 (t, J = 11.4 Hz, 1H, 9-Ha), 3.85 (d, J = 13.2 Hz, 1H, 6-Hb), 3.90–4.00 (m, 2H, 17b-H and 9-Hb), 4.52 (s, 1H, 10a-H), 6.56 (d, J = 9.2 Hz, 1H, 4-H), 7.17 (s, 1H, 15-H), 7.51 (s, 1H, 12-H), 7.95 (dd, J = 9.2 and 2.5 Hz, 1H, 3-H), 8.12 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.3 (C-3′), 20.3 (C-2′), 27.7 (C-10), 31.7 (C-6a), 34.1 (C-17b), 39.8 (C-1′), 56.0 (C-6), 62.8 (C-9), 69.4 (C-7), 71.9 (C-10a), 100.4 (C-17a), 110.3 (C-4), 112.3 (C-11b), 117.4 (C-15), 122.0 (C-17c), 122.6 (C-12), 124.5 (C-3), 126.5 (C-1), 133.1 (C-13), 138.4 (C-2), 142.6 (C-14), 149.1 (C-15a), 151.3 (C-4a), 159.2 (C-17), 164.2 (C-11a). IR (KBr) ν: 1289, 1305, 1629, 1697, 2869, 2927 cm−1; HRMS: calcd for C25H24N2O6Na [M + Na]+ 471.1527, found 471.1526.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), affording rac-(6aS*, 10aS*, 17bS*)-10ac as yellow amorphous solid (77%). Rf = 0.23 (hexane/ethyl acetate 1
2), affording rac-(6aS*, 10aS*, 17bS*)-10ac as yellow amorphous solid (77%). Rf = 0.23 (hexane/ethyl acetate 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2).
2).1H NMR (400 MHz, CDCl3) δ 2.00 (d, J = 14.1 Hz, 1H, 10-Ha), 2.23 (s, 1H, 10-Hb), 3.09 (s, 3H, 4′-H), 3.38–3.59 (m, 3H, 7-H and 6-Ha), 3.59–3.74 (m, 1H, 9-Ha), 3.74–3.86 (m, 7H, 1′-H, 2′-H and 6-Hb), 3.91 (s, 5H, 3′-H, 17b-H and 9-Hb), 4.49 (s, 1H, 10a-H), 6.52 (d, J = 8.9 Hz, 1H, 4-H), 6.71 (s, 1H, 15-H), 7.90 (d, J = 8.9 Hz, 1H, 3-H),8.00 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 27.8 (C-10), 31.1 (C-6a), 34.0 (C-17b), 39.8 (C-4′), 55.9 (C-6), 56.4 (C-1′), 61.3 (C-2′), 62.1 (C-3′), 62.9 (C-9), 69.4 (C-7), 71.9 (C-10a), 96.7 (C-15), 98.9 (C-17a), 103.0 (C-11b), 110.3 (C-4), 122.1 C-17c), 124.6 (C-3), 126.5 (C-1), 138.3 (C-2), 140.4 (C-13), 149.1 (C-4a), 150.8 (C-12), 157.0 (C-14), 160.8 (C-11a), 163.8 (C-17). IR (KBr) ν: 1094, 1291, 1607, 1700, 2856, 2925 cm−1; HRMS: calcd for C26H26N2O9Na [M + Na]+ 533.1531 found 533.1530.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 10aS*, 17bS*)-10bc as white powder (52%), mp 232–235 °C. Rf = 0.28 (hexane/ethyl acetate 1
1), affording rac-(6aS*, 10aS*, 17bS*)-10bc as white powder (52%), mp 232–235 °C. Rf = 0.28 (hexane/ethyl acetate 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2).
2).1H NMR (400 MHz, CDCl3) δ 2.01 (d, J = 14.7 Hz, 1H, 10-Ha), 2.23 (m, 1H, 10-Hb), 3.02 (s, 3H, 4′-H), 3.39 (d, J = 12.5 Hz, 1H, 6-Ha), 3.46 (s, 2H, 7-H), 3.68 (d, J = 12.5 Hz, 1H, 6-Hb), 3.73 (m, 1H, 9-Ha), 3.85 (s, 3H, 2′-H), 3.87 (s, 4H, 1′-H, 17b-H), 3.91 (d, J = 5.2 Hz, 1H, 9-Hb), 3.94 (s, 3H, 3′-H), 4.58 (s, 1H, 10a-H), 6.67 (d, J = 8.6 Hz, 1H, 4-H), 6.73 (s, 1H, 15-H), 7.33 (d, J = 8.5 Hz, 1H, 3-H), 7.44 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 27.7 (C-10), 31.7 (C-6a), 34.3 (C-17b), 39.6 (C-4′), 56.3 (C-3′), 56.3 (C-6), 61.2 (C-2′), 62.0 (C-1′), 62.8 (C-9), 69.4 (C-7), 72.1 (C-10a), 96.6 (C-15), 99.1 (C-17a), 103.1 (C-11b), 110.0 (CF3), 111.2 (C-4), 119.7 (q, J = 32.3 Hz, C-2), 123.0 (C-17c), 123.4 (CF3), 124.93 (d, J = 3.5 Hz, C-3), 126.1 (CF3), 126.83 (d, J = 3.2 Hz, C-1), 140.3 (C-13), 147.2 (C-4a), 150.7 (C-15a), 150.8 (C-12), 156.8 (C-14), 160.8 (C-11a), 163.9 (C-17). IR (KBr) ν: 1099, 1330, 1399, 1606, 1700, 2940 cm−1; HRMS: calcd for C27H26F3NO7Na [M + Na]+ 556.1554 found 556.1554.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), affording rac-(6aS*, 10aS*, 17bS*)-10ad as yellow powder (42%), mp 244–246 °C. Rf = 0.29 (hexane/ethyl acetate 1
2), affording rac-(6aS*, 10aS*, 17bS*)-10ad as yellow powder (42%), mp 244–246 °C. Rf = 0.29 (hexane/ethyl acetate 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2).
2).1H NMR (400 MHz, DMSO-d6) δ 1.84 (s, 2H, 10-H), 2.39 (s, 3H, 1′-H), 3.18 (s, 2H, 6-H), 3.40 (m, 2H, 7-H), 3.46 (s, 2H, 9-H), 5.40 (s, 1H, 10a-H), 6.49 (s, 1H, 17b-H), 7.23 (d, J = 9.0 Hz, 1H, 4-H), 7.27 (t, J = 7.6 Hz, 1H, 13-H), 7.43 (d, J = 8.2 Hz, 1H, 15-H), 7.57 (t, J = 7.7 Hz, 1H, 14-H), 7.94 (d, J = 7.5 Hz, 1H, 12-H), 8.00 (d, J = 2.0 Hz, 1H, 1-H), 8.06 (dd, J = 8.9 and 2.6 Hz, 1H, 3-H), 12.22 (s, 1H, N–H). 13C NMR (100 MHz, DMSO-d6) δ 25.0 (C-10), 34.7 (C-17b), 58.1 (C-6), 63.5 (C-7), 66.2 (C-9), 116.3 (C-15), 116.8 (C-17a), 121.5 (C-4), 123.0 (C-13), 123.0 (C-3), 123.3 (C-10a), 123.6 (C-12), 125.4 (C-1), 130.6 (C-13b), 131.6 (C-14), 134.0 (C-17c), 137.2 (C-2), 141.8 (C-15a), 158.3 (C-4a), 161.6 (C-13a), 166.1 (C-17). IR (KBr) ν: 1333, 1393, 1492, 1604, 1637 cm−1; HRMS: calcd for C23H21N3O5Na [M + Na]+ 442.1373 found 442.1373.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), affording rac-(6 aS*, 10aS*, 15bS*)-10ae as yellow powder (42%), mp 279–282 °C. Rf = 0.29 (hexane/ethyl acetate 1
2), affording rac-(6 aS*, 10aS*, 15bS*)-10ae as yellow powder (42%), mp 279–282 °C. Rf = 0.29 (hexane/ethyl acetate 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2).
2).1H NMR (400 MHz, CDCl3) δ 1.87 (d, J = 14.6 Hz, 1H, 10-Ha), 2.16 (t, J = 11.7 Hz, 1H, 10-Hb), 2.27 (s, 3H, 2′-H), 3.08 (s, 3H, 1′-H), 3.32–3.55 (m, 3H, 7-H and 6-Ha), 3.62 (t, J = 11.6 Hz, 1H, 9-Ha), 3.78 (m, 2H, 15b-H and 6-Hb), 3.87 (d, J = 7.0 Hz, 1H, 9-Hb), 4.33 (s, 1H, 10a-H), 5.83 (s, 1H, 12-H), 6.52 (d, J = 9.2 Hz, 1H, 4-H), 7.93 (d, J = 9.0 Hz, 1H, 3-H), 8.10 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.9 (C-2′), 27.5 (C-10), 31.7 (C-6a), 33.5 (C-15b), 39.7 (C-1′), 55.9 (C-6), 62.6 (C-9), 69.2 (C-7), 71.6 (C-10a), 98.9 (C-15a), 99.8 (C-12), 110.2 (C-4), 122.0 (C-15c), 124.5 (C-3), 126.4 (C-1), 138.3 (C-2), 149.1 (C-4a), 162.1 (C-13), 163.9 (C-11a), 165.6 (C-15). IR (KBr) ν: 1243, 1301, 1587, 1704, 2861, 2925 cm−1; HRMS: calcd for C20H20N2O6Na [M + Na]+ 407.1214, found 407.1213.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), affording rac-(6aS*, 10aS*, 15bS*)-10be as white amorphous solid (32%). Rf = 0.18 (hexane/ethyl acetate 2
1), affording rac-(6aS*, 10aS*, 15bS*)-10be as white amorphous solid (32%). Rf = 0.18 (hexane/ethyl acetate 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).1H NMR (400 MHz, CDCl3) δ 1.84 (d, J = 14.6 Hz, 1H, 10-Ha), 2.13 (m, 1H, 10-Hb), 2.26 (s, 3H, 2′-H), 2.98 (s, 3H, 1′-H), 3.31 (d, J = 12.5 Hz, 1H, 6-Ha), 3.37 (s, 2H, 7-H), 3.63 (m, 2H, 6-Hb and 9-Ha), 3.70 (s, 1H, 15b-H), 3.86 (dd, J = 11.8 and 5.4 Hz, 1H, 9-Hb), 4.40 (s, 1H, 10a-H), 5.82 (s, 1H, 12-H), 6.64 (d, J = 8.6 Hz, 1H, 4-H), 7.32 (d, J = 8.6 Hz, 1H, 3-H), 7.52 (s, 1H, 1-H). 13C NMR (100 MHz, CDCl3) δ 19.9 (C-2′), 27.5 (C-10), 32.4 (C-6a), 33.8 (C-15b), 39.6 (C-1′), 56.3 (C-6), 62.6 (C-9), 69.2 (C-7), 99.1 (C-15a), 99.8 (C-12), 111.2 (C-4), 119.6 and 119.9 (C-2), 123.0 (C-15c), 124.9 (C-3), 126.1 (C-1), 147.2 (C-4a), 161.7 (C-13), 163.9 (C-11a), 165.8 (C-15). IR (KBr) ν: 1106, 1328, 1524, 1583, 1617, 1655, 1702, 2861, 2937 cm−1; HRMS: calcd for C21H20F3NO4Na [M + Na]+ 430.1237, found 430.1235.
Conclusion
Pentaheterocyclic chiral frameworks of novel skeletons containing a pyrano[4,3-b]pyran unit condensed with a pyrone or pyridone and a tetrahydroquinoline or chroman subunit were prepared in a domino Knoevenagel-intramolecular hetero-Diels-Alder reaction, utilizing heterocyclic dienophiles and heterodienes. Solvent-dependence was explored and toluene was selected as the solvent of the reaction, affording the best overall yields and access to four isomers, which could be readily separated by column chromatography. The reaction of 2H-chromene derivatives 1a–e with 4-hydroxycoumarine reagents 2a–c produced isomer 4 as the main product in most cases with the α,β-unsaturated ketone subunit acting as the heterodiene in the IMHDA step. Simplified 3,6-dihydro-2H-pyran substrates 8a–b, containing a dihydropyran moiety as the dienophile showed a remarkable improvement in both regio- and diastereoselectivity, affording a single isomer irrespective of the substitution pattern of the reagent or the substrate. 4-Hydroxy-2-pyrone and -2-pyridone reagents 2e and 2f, lacking the condensed benzene ring, were also tested in the domino reaction, in which substrates 1a-c afforded a mixture of 2–4 regio- and diastereomers, while substrate 8a gave a single isomer in each case. The planar structure and relative configuration of the products 4–7 were determined on the basis of 13C chemical shifts, NOE correlations and IR carbonyl stretching vibrations supported by two single crystal X-ray diffraction analysis. The antiproliferative activities of the condensed heterocycles were tested on three human cancer cell lines, which revealed that all the active derivatives have the same (S*, R*, S*) relative configuration and all except for one belong to the series of products 4, in which the α,β-unsaturated ketone moiety acts as a heterodiene in the IMHDA cyclization step. Compound 4ac, containing a condensed trimethoxycoumarin residue, exhibited promising antiproliferative activity with an IC50 value of 5.7 µM.Author contributions
Conceptualization, S. B. K. and T. K.; methodology, S. B. K., B. I. T. and G. V.; software, A. B., B. I. T. and G. V.; validation, B. I. T., G. V., E. L., D. N. and A. B.; formal analysis, A. B.; investigation, S. B. K., B. B., O. M. O., D. O., K. B., A. B., E. L., G. V., B. I. T. and D. N.; resources, T. K., F. F., M. V. and T. B.; data curation, S. B. K., B. I. T. and T. K.; writing – original draft preparation, S. B. K.; writing – review and editing, S. B. K., T. K., F. F., T. B. and M. V.; visualization, S. B. K., A. B., B. I. T. and D. N.; supervision, S. B. K., T. K., T. B., F. F. and M. V; project administration, T. K.; funding acquisition, T. K, T. B., E. L., F. F. and M. V. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2408634 (4cc) and 2408635 (6cc) contain the supplementary crystallographic data for this paper.62a,bAll experimental (recipies for the preparation of substrates 1a–e and 8a–b), spectral (1H, 13C and 2D-NMR, ECD and on-line HPLC-ECD spectra of products) and bioactivity (inhibitory effect at 50 µM concentration and concentration-dependent effect) data is available in the supporting information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra02756c.
Acknowledgements
This research was funded by the National Research Development and Innovation Office (Grant numbers: Advanced-153095 T. K.; PD 134791 E. L). The authors thank the University of Debrecen for the Scientific Research Bridging Fund (DETKA). E. L. was supported by the Hungarian Academy of Science's János Bolyai grant and by the EKÖP-24-4-II-DE-19 University Research Scholarship Program of the Ministry for Culture and Innovation from the Source of the National Research, Development and Innovation Fund.References
- A. Evidente, L. Maddau, E. Spanu, A. Franceschini, S. Lazzaroni and A. Motta, J. Nat. Prod., 2003, 66, 313–315 CrossRef CAS PubMed.
- L. C. Meade-Tollin, E. M. K. Wijeratne, D. Cooper, M. Guild, E. Jon, A. Fritz, G.-X. Zhou, L. Whitesell, J.-y. Liang and A. A. L. Gunatilaka, J. Nat. Prod., 2004, 67, 2–4 CrossRef CAS PubMed.
- M. C. Edler, R. M. Buey, R. Gussio, A. I. Marcus, C. D. Vanderwal, E. J. Sorensen, J. F. Díaz, P. Giannakakou and E. Hamel, Biochemistry, 2005, 44, 11525–11538 CrossRef PubMed.
- M. J. Towle, K. A. Salvato, J. Budrow, B. F. Wels, G. Kuznetsov, K. K. Aalfs, S. Welsh, W. Zheng, B. M. Seletsky, M. H. Palme, G. J. Habgood, L. A. Singer, L. V. DiPietro, Y. Wang, J. J. Chen, D. A. Quincy, A. Davis, K. Yoshimatsu, Y. Kishi, M. J. Yu and B. A. Littlefield, Cancer Res., 2001, 61, 1013–1021 CAS.
- S. Omura, H. Tomoda, Y. K. Kim and H. Nishida, J. Antibiot., 1993, 46, 1168–1169 CrossRef CAS PubMed.
- K. H. Ling, C. K. Yang and F. T. Peng, Appl. Environ. Microbiol., 1979, 37, 355–357 CrossRef CAS PubMed.
- J. Du, Z.-D. He, R.-W. Jiang, W.-C. Ye, H.-X. Xu and P. P.-H. But, Phytochemistry, 2003, 62, 1235–1238 CrossRef CAS PubMed.
- T. Yasumoto and M. Murata, Chem. Rev., 1993, 93, 1897–1909 CrossRef CAS.
- S. Ray, H. K. Majumder, A. K. Chakravarty, S. Mukhopadhyay, R. R. Gil and G. A. Cordell, J. Nat. Prod., 1996, 59, 27–29 CrossRef CAS PubMed.
- I. Souzu and H. Mitsuhashi, Tetrahedron Lett., 1969, 10, 2725–2728 CrossRef PubMed.
- H. Otsuka, T. Akiyama, K.-I. Kawai, S. Shibata, O. Inoue and Y. Ogihara, Phytochemistry, 1978, 17, 1349–1352 CrossRef CAS.
- A. K. Chakravarty, T. Sarkar, K. Masuda, K. Shiojima, T. Nakane and N. Kawahara, Phytochemistry, 2001, 58, 553–556 CrossRef CAS PubMed.
- E. Fujita, M. Taoka, M. Shibuya, T. Fujita and T. Shingu, J. Chem. Soc., Perkin Trans. 1, 1973, 2277–2281 RSC.
- L. Li, H.-K. Wang, J.-J. Chang, A. T. McPhail, D. R. McPhail, H. Terada, T. Konoshima, M. Kokumai, M. Kozuka, J. R. Estes and K.-H. Lee, J. Nat. Prod., 1993, 56, 690–698 CrossRef CAS PubMed.
- T. Tanaka, T. Ito, M. Iinuma, Y. Takahashi and H. Naganawa, Phytochemistry, 1998, 48, 1423–1427 CrossRef CAS.
- J. Li, Y.-F. Tan, S. Liu, X.-Q. Wu, J. Wang, K.-P. Xu, G.-S. Tan, Z.-X. Zou and W.-X. Wang, Phytochemistry, 2022, 200, 113205 CrossRef CAS PubMed.
- N. Damuka, K. Kammari, A. M. Potshangbam, A. K. Kondapi and V. Vindal, ChemistrySelect, 2021, 6, 5941–5948 CrossRef CAS.
- N. C. Dige, P. G. Mahajan and D. M. Pore, Med. Chem. Res., 2018, 27, 224–233 CrossRef CAS.
- R. Sagar, J. Park, M. Koh and S. B. Park, J. Org. Chem., 2009, 74, 2171–2174 CrossRef CAS PubMed.
- P. Kumari, S. Gupta, C. Narayana, S. Ahmad, N. Vishnoi, S. Singh and R. Sagar, New J. Chem., 2018, 42, 13985–13997 RSC.
- L. Zeng, S. Liu, Y. Lan and L. Gao, Nat. Commun., 2023, 14, 3511 CrossRef CAS PubMed.
- M. Jin, C. Tang, Y. Li, S. Yang, Y.-T. Yang, L. Peng, X.-N. Li, W. Zhang, Z. Zuo, F. Gagosz and L.-L. Wang, Nat. Commun., 2021, 12, 7188 CrossRef CAS PubMed.
- Y. Zheng, S. Qian, P. Xu, T. Ma and S. Huang, Adv. Synth. Catal., 2022, 364, 3800–3804 CrossRef CAS.
- V. Arredondo, D. E. Roa, S. Yan, F. Liu-Smith and D. L. Van Vranken, Org. Lett., 2019, 21, 1755–1759 CrossRef CAS PubMed.
- X. R. Nan Zhao, J. Ren, H. Lu, S. Ma, R. Gao, Y. Li, S. Xu, Li Li and S. Yu, Org. Lett., 2015, 17, 3118–3121 CrossRef PubMed.
- L. N. Aldrich, E. S. Dawson and C. W. Lindsley, Org. Lett., 2010, 12, 1048–1051 CrossRef CAS PubMed.
- B. B. Snider and Q. Lu, J. Org. Chem., 1996, 61, 2839–2844 CrossRef CAS PubMed.
- S. Bhuyan, D. Das, A. Chakraborty, S. Mandal, K. Dhanabal and B. G. Roy, Chem.–Asian J., 2021, 16, 4108–4121 CrossRef CAS PubMed.
- K. A. Krasnov, P. V. Dorovatovskii, Y. V. Zubavichus, T. V. Timofeeva and V. N. Khrustalev, Tetrahedron, 2017, 73, 542–549 CrossRef CAS.
- P. Padmaja, B. V. Subba Reddy, N. Jain, S. R. Mutheneni, P. Bollepelli, S. Polepalli, G. Rambabu and P. N. Reddy, New J. Chem., 2016, 40, 8305–8315 RSC.
- A. Mishra, N. Rastogi and S. Batra, Tetrahedron, 2012, 68, 2146–2154 CrossRef CAS.
- S. Majumder and P. J. Bhuyan, Tetrahedron Lett., 2012, 53, 137–140 CrossRef CAS.
- S. B. Király, A. Bényei, E. Lisztes, T. Bíró, B. I. Tóth and T. Kurtán, Eur. J. Org Chem., 2021, 2021, 6161–6170 CrossRef.
- S. Balázs Király, L. Tóth, T. Kovács, A. Bényei, E. Lisztes, B. István Tóth, T. Bíró, A. Kiss-Szikszai, K. E. Kövér, A. Mándi and T. Kurtán, Adv. Synth. Catal., 2023, 365, 3301–3319 CrossRef.
- L.-F. Tietze, S. Brand and T. Pfeiffer, Angew. Chem., Int. Ed., 1985, 24, 784–786 CrossRef.
- B. Baruah and P. J. Bhuyan, Tetrahedron, 2009, 65, 7099–7104 CrossRef CAS.
- L. F. Tietze and P. Saling, Synlett, 1992, 1992, 281–282 CrossRef.
- M. Bakthadoss and D. Kannan, RSC Adv., 2014, 4, 11723–11731 RSC.
- M. Bakthadoss and G. Sivakumar, Tetrahedron Lett., 2014, 55, 1765–1770 CrossRef CAS.
- S. Kathiravan and R. Raghunathan, Synlett, 2010, 2010, 1927–1930 CrossRef.
- Y. R. Lee and T. V. Hung, Tetrahedron, 2008, 64, 7338–7346 CrossRef CAS.
- N. J. Parmar, S. B. Teraiya, H. A. Barad, D. Sharma and V. K. Gupta, Synth. Commun., 2013, 43, 1577–1586 CrossRef CAS.
- S. Maiti, S. K. Panja and C. Bandyopadhyay, Tetrahedron, 2010, 66, 7625–7632 CrossRef CAS.
- E. Ramesh and R. Raghunathan, Tetrahedron Lett., 2008, 49, 1812–1817 CrossRef CAS.
- M. Shanmugasundaram, S. Manikandan and R. Raghunathan, Tetrahedron, 2002, 58, 997–1003 CrossRef CAS.
- R. D. R. S. Manian, J. Jayashankaran and R. Raghunathan, Tetrahedron Lett., 2007, 48, 1385–1389 CrossRef CAS.
- J. S. Yadav, B. V. S. Reddy, V. Naveenkumar, R. S. Rao and K. Nagaiah, Synthesis, 2004, 1783–1788 CrossRef CAS.
- M. Ghandi, S. Feizi, M. T. Nazeri and B. Notash, J. Iran. Chem. Soc., 2017, 14, 177–187 CrossRef CAS.
- Y. Dommaraju, S. Borthakur and D. Prajapati, ChemistrySelect, 2016, 1, 6768–6771 CrossRef CAS.
- S. Manikandan, M. Shanmugasundaram and R. Raghunathan, Tetrahedron, 2002, 58, 8957–8962 CrossRef CAS.
- L. F. Tietze, H. Geissler, J. Fennen, T. Brumby, S. Brand and G. Schulz, J. Org. Chem., 1994, 59, 182–191 CrossRef CAS.
- G. Cravotto, G. M. Nano, S. Tagliapietra, G. Palmisano and T. Pilati, J. Heterocycl. Chem., 2001, 38, 965–971 CrossRef CAS.
- H. Sunden, I. Ibrahem, G. L. Zhao, L. Eriksson and A. Cordova, Chem. Eur. J., 2007, 13, 574–581 CrossRef CAS PubMed.
- R. R. Raslan, Y. A. Ammar, S. A. Fouad, S. A. Hessein, N. A. M. Shmiess and A. Ragab, RSC Adv., 2023, 13, 10440–10458 RSC.
- A. Ragab, R. R. Raslan, M. S. Abusaif, H. K. Thabet, Y. A. Ammar and N. A. Gohar, Eur. J. Med. Chem., 2025, 294, 117751 CrossRef CAS PubMed.
- G. Sheldrick, Acta Crystallogr., Sect. A, 2008, 64, 112–122 CrossRef CAS PubMed.
- S. Westrip, J. Appl. Crystallogr., 2010, 43, 920–925 CrossRef CAS.
- C. F. Macrae, I. Sovago, S. J. Cottrell, P. T. A. Galek, P. McCabe, E. Pidcock, M. Platings, G. P. Shields, J. S. Stevens, M. Towler and P. A. Wood, J. Appl. Crystallogr., 2020, 53, 226–235 CrossRef CAS PubMed.
- A. Spek, J. Appl. Crystallogr., 2003, 36, 7–13 CrossRef CAS.
- A. Szappanos, A. Mandi, K. Gulacsi, E. Lisztes, B. I. Toth, T. Biro, S. Antus and T. Kurtan, Org. Biomol. Chem., 2020, 18, 2148–2162 RSC.
- Á. Szappanos, A. Mándi, K. Gulácsi, E. Lisztes, B. I. Tóth, T. Bíró, A. Kónya-Ábrahám, A. Kiss-Szikszai, A. Bényei, S. Antus and T. Kurtán, Biomolecules, 2020, 10, 1462 CrossRef PubMed.
- (a) CCDC 2408634: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2lvcwq; (b) CCDC 2408635: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2lvcxr.
| This journal is © The Royal Society of Chemistry 2026 |