 Open Access Article
Open Access ArticleEfficient removal of methylene blue dye using magnetized titanate nanotubes
Rania N. Zohny,
Ayman H. Zaki *,
A. A. Farghali and
Mohamed Taha
*,
A. A. Farghali and
Mohamed Taha
Material Science and Nanotechnology Department, Faculty of Postgraduate Studies for Advanced Sciences, Beni-Suef University, Beni-Suef, Egypt. E-mail: ayman.zaki@psas.bsu.edu.eg
First published on 27th April 2026
Abstract
This study investigates the removal of methylene blue (MB) dye, a model organic pollutant, using sodium titanate nanotubes (Na/TNTs) and magnetized titanate nanotubes (Mag/TNTs) as efficient nano-adsorbent materials. Mag/TNTs consist of cobalt ferrite (CoFe2O4) nanoparticles supported on Na/TNTs. Both adsorbents were synthesized via a simple hydrothermal method and characterized using SEM, HRTEM, XRD, BET, zeta potential analysis, and FT-IR techniques. Multiwalled open-ended nanotubes were formed, as demonstrated by TEM images. Their typical lengths ranged from 100 to 150 nm, and their average outer diameters were 12.46 and 12.88 nm for Na/TNTs and Mag/TNTs, respectively. Batch adsorption experiments were conducted to evaluate the effects of pH, adsorbent dose, initial dye concentration, temperature, contact time, and adsorbent regeneration on the removal efficiency of MB dye. Among five isotherm models (Langmuir, Freundlich, Langmuir–Freundlich, Sips, Dubinin–Radushkevich), Langmuir–Freundlich and Sips provided the best fits, indicating heterogeneous surface adsorption. Kinetic data were best described by pseudo-second-order and mixed 1,2-order models among the four tested models (pseudo-first-order, pseudo-second-order, mixed 1,2-order, Avrami), suggesting combined physisorption and chemisorption. The maximum adsorption capacities for Na/TNTs and Mag/TNTs increased with temperature, reaching 178.98 mg g−1 and 180.50 mg g−1 at 328 K, respectively. Thermodynamic parameters confirmed that the process is spontaneous and exothermic. Monte Carlo simulations revealed that while CoFe2O4 exhibits stronger individual interactions with MB, the higher surface area of Na/TNTs leads to greater overall removal. The study demonstrates that Mag/TNTs are efficient, separable (using an external magnet), and reusable adsorbents for cationic dye removal from wastewater.
1. Introduction
Clean water resources and their sustainability have been considered one of the most important global issues over the centuries. Freshwater quality enhancement has received great attention from researchers worldwide.1 Urbanization, overpopulation, and human activities such as industrialization and agriculture have side effects on clean water resources.2 Water pollution arises from the discharge of industrial, agricultural, urban, and domestic effluents into water.3 The existence of these pollutants causes several diseases such as dysentery, diarrhea, hepatitis A, and cholera. It is speculated that more than 842![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 people die from diarrhea around the world.4
000 people die from diarrhea around the world.4
Organic dyes are serious pollutants in wastewater, and cause a threat to the aquatic environment through the prevention of light transfer to the aquatic life in water, and consequently hinder the photosynthetic process.5 Every year, thousands of tons of dyes are produced, with global production estimated at approximately 7 × 105 tons.6,7 Methylene blue (MB) is one of the most common cationic dyes, widely used for dyeing leathers, food, pharmaceuticals, cosmetics,8 papers, and fabrics, and is discharged as effluents into surface and groundwater.9 Several risks to human health come from MB presence in water, such as tissue necrosis, vomiting, cyanosis, jaundice, Heinz body formation, increased heart beat rate, shock, etc.10 Several methods have been proposed for removing the organic contaminants from water. These methods include adsorption, ion exchange, advanced oxidation processes, photochemical treatments, and biological treatment methods.7 One of the primary and advantageous methods for dye removal is the adsorption process, as it has a simple design, low cost, high efficiency, and avoids the generation of toxic materials.2,11,12 Numerous adsorbent materials that have been investigated, such as carbon nanotubes,13 hydroxyapatite nanoparticles,14 zinc oxide nanoparticles,15 titanium dioxide nanoparticles,16 cobalt zinc ferrite nanoparticles,17 and titanate nanotubes (TNTs). These adsorbents are characterized by several properties such as high surface area, ion-exchange ability, high stability, large pore volume, and surface functionalized with hydroxyl (OH) groups.2,18,19 The ion-exchange capability of titanate nanotubes enables efficient removal of cationic dyes from wastewater.2 However, the separation of TNTs after water treatment remains a great challenge. Modifying TNTs with magnetic materials will overcome this challenge, as they can be easily separated from wastewater using an external magnet and could be easily reused for many treatment cycles.
CoFe2O4 is a promising magnetic adsorbent for dye removal from wastewater due to its good chemical and thermal stability, a simple chemical composition, and high magnetic properties at the nanoscale.20,21 The spinel structure of CoFe2O4 provides numerous surface-active sites that enhance the adsorption of water pollutants.22 Moreover, the magnetic properties of CoFe2O4 enable rapid separation using an external magnetic field.23
This work aims to combine the advantages of titanate nanotubes as an excellent adsorbent for dye removal with the good magnetic properties of CoFe2O4.24,25 In this regard, we have fabricated Na/TNTs and Mag/TNTs and tested their ability for efficient MB removal from water under various conditions. Furthermore, the adsorption mechanism was investigated using Monte Carlo (MC) simulation to provide molecular-level insights into the interactions between MB molecules and the adsorbent surfaces.
2. Materials and methods
2.1. Materials
All chemicals were of analytical grade and used as received without any further purification. Sodium hydroxide (NaOH) was purchased from PioChem laboratory reagents (Kenya, packed in Egypt), and anatase TiO2 powder was obtained from Loba Chemie laboratory reagents (India). Cobalt sulfate (CoSO4), ferric nitrate (Fe(NO3)3), and MB powder were purchased from Oxford Laboratory Reagents (India) and Central Drug House Ltd (India), respectively.2.2. Synthesis of Na/TNTs and Mag/TNTs
2.3. Batch adsorption of MB
The adsorption performance of Na/TNTs and Mag/TNTs for MB was investigated through different adsorption parameters. 40 mL of MB dye solution was shaken and tested under different parameters such as (i) pH (3 to 12), (ii) initial dye concentration (5 ppm to 320 ppm), (iii) adsorbent dose (20 mg to 160 mg), temperature (298 K to 328 K), and (iv) time (10 min to 210 min). For kinetic studies, samples were withdrawn from the solution at regular intervals and centrifuged at 4000 rpm for 3 min to separate the adsorbent. For adsorption isotherm studies, different initial concentrations of MB were contacted with a fixed amount of adsorbent and stirred for a certain time. After equilibrium was reached, the samples were centrifuged at 4000 rpm for 3 min, and the supernatant was collected for concentration analysis. A UV-visible spectrophotometer (Alpha-1900S) was used to measure the concentration of MB dye in the supernatant at a wavelength of 664 nm. It is worth mentioning that every adsorption experiment was carried out in triplets. The equilibrium adsorption capacity (qe, mg g−1) (eqn (1)) and removal efficiency (% R) (eqn (2)) of MB by Na/TNTs and Mag/TNTs were calculated as:
 | (1) |
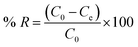 | (2) |
2.4. MC simulation
In this study, the BIOVIA Materials Studio 2020 package [https://www.3ds.com] was used to investigate the adsorption mechanism of MB on Na2Ti3O7 and CoFe2O4 surfaces via MC simulation. The crystal structures of Na2Ti3O7 and CoFe2O4 were obtained from the open-access Materials Project database.29 The Bravais lattice of Na2Ti3O7 is triclinic, with lattice parameters a = 8.546 Å, b = 3.792 Å, and c = 9.137 Å, and lattice angles α = 90°, β = 101.88°, and γ = 90°. CoFe2O4 exhibits a cubic structure, with all angles equal to 90° and a lattice constant of a = 8.446 Å. Using the Forcite module in Materials Studio, geometry optimization of the MB molecule, Na2Ti3O7, and CoFe2O4 was carried out before performing the MC simulations. Compass III force30 with its charges is used in all calculations. An atom-based summation method was employed to calculate both electrostatic and van der Waals interactions during the dye geometry optimization. A cubic-spline truncation scheme was applied, using a cutoff distance of 18.5 Å, a spline width of 1 Å, and a buffer width of 2 Å. For Na2Ti3O7, and CoFe2O4 optimizations, the electrostatic interactions were treated using the Ewald summation, whereas the van der Waals interactions were computed using an atom-based method. In this atom-based scheme, the same cubic-spline truncation parameters used for MB optimization were applied. For the electrostatic summation, an Ewald accuracy of 1.0 × 10−5 kcal mol−1 and a 2 Å buffer width were used. The geometry optimization was considered converged when the energy change, maximum force, and atomic displacement decreased to less than 1.0 × 10−4 kcal mol−1, 0.005 kcal mol−1·Å−1, and 5.0 × 10−5 Å, respectively. In the case of unit-cell optimizations, these convergence criteria were applied together with a stress tolerance of 0.001 GPa and an external pressure of 0 GPa. The smart optimization algorithm is employed in all optimization processes. The optimized lattice parameters of Na2Ti3O7 were a = 8.269 Å (3.2% deviation), b = 3.744 Å (1.3% deviation), and c = 8.865 Å (3.0% deviation), with lattice angles α = 90°, β = 103.05° (1.1% deviation), and γ = 90°. The optimized lattice parameters of CoFe2O4 retained a cubic structure, with all angles equal to 90° (0% deviation) and a lattice constant of a = 9.418 Å (11.5% deviation). The small deviations between the optimized and experimental lattice parameters demonstrate the reliability of the COMPASS forcefield for Na2Ti3O7 and CoFe2O4. This validation supports its use in subsequent MC simulations to explore the adsorption mechanism, despite the slightly higher deviation in CoFe2O4. This forcefield was chosen for its broad parameterization, enabling accurate modeling of hybrid inorganic–organic systems. Mainly, it provides reliable representations of metal oxides and aromatic organic molecules. COMPASS III force field effectively described the associated metal–π interactions through its optimized van der Waals and electrostatic potential terms.28,31 Forcefield atom types assigned to the constituent atoms of Na2Ti3O7, CoFe2O4, and MB dye are mentioned in Table S1 (SI). Supercells of Na2Ti3O7 (45.499 × 45.68 × 76.167 Å) and CoFe2O4 (41.378 × 41.807 × 59.714 Å) were generated. The Na2Ti3O7 (1 0 0) and CoFe2O4 (3 1 1) surfaces were constructed with 50 Å vacuum layers to prevent interlayer interactions. In all MC simulations, non-bonded interactions were treated using the same summation methods applied during the optimization of Na2Ti3O7 and CoFe2O4. The Adsorption Locator module was employed to perform simulated annealing within the canonical ensemble using the Metropolis MC method. The parameters for the simulated annealing process were as follows: five heating cycles of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps each. Simulated annealing MC simulation was conducted with a maximum temperature of 1.0 × 105 K to enable broad configurational sampling, followed by cooling to 100 K (final temperature) to achieve the lowest-energy adsorption configuration.2,28,32,33 This approach helps identify the lowest-energy configuration of dye molecules on Na2Ti3O7 and CoFe2O4 surfaces, from which the corresponding adsorption energies (ΔEads) were calculated (eqn (3)).34 During the simulations, the Na2Ti3O7 and CoFe2O4 surfaces were kept fixed, and their energies were defined as zero reference points. The ΔEads was calculated as the total energy change associated with the adsorption of the relaxed dye on the Na2Ti3O7 and CoFe2O4 surfaces. It is defined as the sum of the rigid adsorption energy (ΔErigid), which corresponds to the energy change when the unrelaxed adsorbate initially adsorbs on the substrate, and the deformation energy (ΔEdeformation), which is the energy released (or required) as the adsorbate relaxes on the surface.
000 steps each. Simulated annealing MC simulation was conducted with a maximum temperature of 1.0 × 105 K to enable broad configurational sampling, followed by cooling to 100 K (final temperature) to achieve the lowest-energy adsorption configuration.2,28,32,33 This approach helps identify the lowest-energy configuration of dye molecules on Na2Ti3O7 and CoFe2O4 surfaces, from which the corresponding adsorption energies (ΔEads) were calculated (eqn (3)).34 During the simulations, the Na2Ti3O7 and CoFe2O4 surfaces were kept fixed, and their energies were defined as zero reference points. The ΔEads was calculated as the total energy change associated with the adsorption of the relaxed dye on the Na2Ti3O7 and CoFe2O4 surfaces. It is defined as the sum of the rigid adsorption energy (ΔErigid), which corresponds to the energy change when the unrelaxed adsorbate initially adsorbs on the substrate, and the deformation energy (ΔEdeformation), which is the energy released (or required) as the adsorbate relaxes on the surface.| ΔEads = ΔErigid + ΔEdeformation, | (3) |
This definition clarifies the contributions of both initial adsorption and subsequent structural relaxation of the adsorbate. The Cartesian coordinates of the optimized dye-surface configurations are provided in the supporting information, including the lowest-energy geometries. Different configurational move types were assigned to each species during the MC simulation. A conformer (0.32 probability), translation (0.32 probability), rotation (0.32 probability), and regrowth (0.03 probability) move ratio of 1 was applied to the MB dye molecules to fully sample their positional changes.35,36 In contrast, chloride ions were restricted to translational moves only, consistent with their spherical geometry. After five annealing cycles (5 × 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 steps), both the total energy and adsorption configuration stop changing and reach a stable state, confirming convergence. Increasing the number of simulation cycles produced no significant change in the lowest-energy configuration or adsorption energy.
000 steps), both the total energy and adsorption configuration stop changing and reach a stable state, confirming convergence. Increasing the number of simulation cycles produced no significant change in the lowest-energy configuration or adsorption energy.
3. Results and characterization
3.1. Characterization of Na/TNTs and Mag/TNTs
Fig. 1A–D shows high-resolution SEM (Zeiss Sigma 500 VP Analytical FESEM, Carl Zeiss, Germany) and TEM (JEM-1230 (600Kx)) of Na/TNTs and Mag/TNTs, both with a uniform tubular structure. These images proved the successful formation of nanotubes. TEM images showed multiwalled open-ended nanotubes with an average length between 100 nm and 150 nm for both, and average outer diameters of 12.46 nm and 12.88 nm for Na/TNTs and Mag/TNTs, respectively. The spherical particles that spread over the nanotubes indicate the presence of CoFe2O4 in the prepared material.24,37 EDX analysis investigated the elemental composition of the prepared Na/TNTs and Mag/TNTs. The analysis detected the presence of Ti, O, and Na, which are the components of Na/TNTs (Fig. 1E) with weight percentages (35.83, 51.02, and 23.15) wt%, respectively, and the presence of Ti, O, Na, Co, and Fe which are the main components of Mag/TNTs (Fig. 1F) with weight percentages (11.67, 68.85, 18.17, 0.52, and 0.79) wt%, respectively.The crystalline structure of Na/TNTs and Mag/TNTs was examined using XRD (Malvern PANalytical Empyrean 3 diffractometer). Fig. 2 shows the XRD patterns of Na/TNTs and Mag/TNTs. The four reflections at 2θ values of 9.27°, 24.41°, 28.17°, and 48.29° confirm the successful preparation of Na/TNTs.38 The formation of Na/TNTs is verified according to the ICDD (International Center for Diffraction Data) card no. 00-059-0666.39 XRD peak at 2θ = 9.27° refers to the interlayer spacing of the layered Na/TNTs structure. After magnetization, the XRD pattern of Mag/TNTs retains the characteristic titanate peak and exhibits additional diffraction peaks attributed to the crystalline CoFe2O4 phase. The new peaks appear at (35.6°, 57.2°, 62.8°), corresponding to magnetic CoFe2O4, matching the ICDD card no. 22-1086.24
The surface functional groups of Na/TNTs and Mag/TNTs were identified by Fourier transform infrared (FTIR) spectroscopy (Bruker Vertex 70 spectrometer). The broad peak at 3388.01 cm−1 represents the O–H stretching vibration of surface hydroxyl groups and adsorbed water molecules. Also, the H–O–H bending vibration appears with strong absorption at 1625.81 cm−1, confirming the existence of water molecules in the titanate nanotubes. The spectrum of Na/TNTs (Fig. 3a) shows a characteristic peak of Ti–O stretching vibration at 915.62 cm−1,40 as well as an absorption peak appearing at 463.89 cm−1, referring to the Ti–O–Ti crystal phonons.2 As shown in Fig. 3b, the band observed at 451 cm−1 refers to the presence of Co–Fe vibration.24
The BET results revealed that the surface area reached 138.1 m2 g−1 and 96.253 m2 g−1, with total pore volume of 0.2238 cm3 g−1 and 0.2762 cm3 g−1 at an adsorption temperature of 77 K for both Na/TNTs and Mag/TNTs, respectively. In the adsorption isotherm (Fig. 4a), both Na/TNTs and Mag/TNTs showed a gradual increase in nitrogen quantity at P/P0 near zero, with a rapid increase at high relative pressure (P/P0 > 0.8). The results indicated that Na/TNTs exhibited a higher surface area and a small pore volume, whereas Mag/TNTs exhibited a lower surface area and a large pore volume. Pore size distribution plays a role in determining how much the nitrogen adsorption uptake contributes to surface area and pore volume. In the case of Na/TNTs, there are many narrow mesopores, which lead to a large internal surface area of 138.1 m2 g−1 with an overall small pore volume of 0.2238 cm3 g−1. Resulting in a high quantity of nitrogen adsorbed close to P0 for Na/TNTs. On the contrary, Mag/TNTs have wider mesopores that provide a larger pore volume (0.2762 cm3 g−1) with a small surface area (96.253 m2 g−1). According to the IUPAC classification, the adsorption isotherm is an IV-type isotherm, and H3 hysteresis loop which is characteristic of mesoporous materials with cylindrical pores.2,24,41 The BJH pore size distribution (Fig. 4b) revealed that both materials have mesoporous properties. Also, the figure pointed out that Na/TNTs have a narrow pore size distribution compared to Mag/TNTs with a uniform pore structure ranging from 2.5 nm to 25 nm for Na/TNTs and 3.47 nm to 25 nm for Mag/TNTs, respectively.
3.2. Investigation of the MB adsorption
| Kinetic models | Adjustable parameters | Values | R2 | ||
|---|---|---|---|---|---|
| Na/TNTs | Mag/TNTs | Na/TNTs | Mag/TNTs | ||
| PFO | k1 | 0.139 | 0.192 | 0.992 | 0.990 |
| qe | 133.9 | 129.5 | |||
| PSO | k2 | 0.002 | 0.0036 | 0.999 | 0.998 |
| qe | 140.5 | 133.99 | |||
| MO | k1,2 | 0.0017 | 0.0018 | 0.999 | 0.998 |
| qe | 140.1 | 133.7 | |||
| f2 | 0.99 | 0.99 | |||
| Avrami | kav | 0.38 | 0.45 | 0.992 | 0.990 |
| nav | 1.35 | 0.42 | |||
| qe | 133.9 | 129.5 | |||
| Isotherm model | Equation | Kinetics model | Equation |
|---|---|---|---|
| a qe: equilibrium adsorption capacity of adsorbent (mg g−1). Ce: equilibrium concentration of the adsorbate (mg L−1). qmax: maximum adsorption capacity of adsorbent (mg g−1). KL: Langmuir constant(L mg−1). Kf: Freundlich constant (L g−1). 1/nf: Freundlich adsorption intensity. KLF: equilibrium constant for heterogeneous solid. nLF: heterogeneous parameter. KS: Sips isotherm model constant (L mg−1). 1/ns: Sips isotherm exponent. Kad: Dubinin–Radushkevich constant related to the mean free energy of adsorption (mol2 J−2). ε: Polyani potential that is related to equilibrium concentration (J mol−1). qm: Dubinin–Radushkevich monolayer saturation capacity. qt: adsorption capacity of adsorbent at time t (mg g−1). k1: rate constant of pseudo first-order model (min−1). k2: rate constant of pseudo second-order model (g mg−1 min−1). k1,2: rate constant of mixed 1,2-order model (min−1). t: time (min). kav: Avrami rate constant (min−1). nav: Avrami component (dimensionless). | |||
| Langmuir | 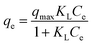 |
Pseudo-first order | qt = qe(1 − e−k1t) |
| Freundlich | 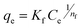 |
Pseudo-second order | 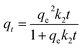 |
| Sips |  |
Mixed 1,2-order | 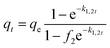 |
| Langmuir–Freundlich |  |
Avrami |  |
| Dubinin–Radushkevich | qe = (qm)exp(−Kadε2) | ||
Adsorption capacities of 126.8 mg g−1 and 124.4 mg g−1 were obtained for Na/TNTs and Mag/TNTs, respectively, within the first 30 min, then increased till reaching equilibrium at 100–210 min. The final adsorption capacity (qt) reached 137.4 mg g−1 and 133.3 mg g−1 for Na/TNTs and Mag/TNTs at 210 min, respectively. The PSO and MO models provided the best fits to the experimental data, both models yielding correlation coefficients (R2) of 0.999 for Na/TNTs and 0.998 for Mag/TNTs (Table 1). The excellent fitting of these two models to the experimental data (Fig. 7) suggests that the adsorption proceeds through mixed mechanisms involving both physisorption and chemisorption.50
Fig. 9 presents the fitting of the experimental adsorption data of MB onto Na/TNTs (a–d) and Mag/TNTs (e–h) at temperatures of 298, 308, 318, and 328 K, respectively, using different isotherm models. The qe values increase sharply at low Ce values and then reach a plateau. Even though the Langmuir model explains the behavior of monolayer adsorption, the L–F and Sips models fit the data better, indicating adsorption on energetically heterogeneous active sites. The heterogeneous adsorption surface may be due to the presence of magnetic nanoparticles and a nanotubular structure. Conversely, the Freundlich model exhibited a low R2 (0.60), indicating poor agreement with experimental data and suggesting that a purely empirical multilayer model cannot well explain the adsorption process. The D–R model supports that adsorption includes diffusion into micropore filling in addition to surface coverage. In general, the agreement of these models suggests that MB adsorption onto both Na/TNTs and Mag/TNTs occurs via a mechanism that includes pore filling, heterogeneous multi-energy interactions, and monolayer adsorption on active sites. In addition, the Langmuir–Freundlich and Sips models show consistently strong agreement with the experimental data at all other investigated temperatures, as supported by the R2 values presented in Table 3 for both Na/TNTs and Mag/TNTs. The strong alignment of experimental data with the fitted models demonstrates the reliability and validity of the isothermal analysis. The qmax values Na/TNTs increased from (91.23 to 178.98) mg g−1 as the temperature increased from (298to 328) K, whereas Mag/TNTs showed a similar but more pronounced enhancement from (62.51 to 180.50) mg g−1, as obtained from the L–F model. The sharper increase observed for Mag/TNTs indicates that the incorporation of magnetic components enhances the thermal activation of adsorption sites and strengthens the adsorbate–adsorbent interactions at elevated temperatures.64,65
 | ||
| Fig. 9 Fitting experimental qe data with isothermal models for MB adsorption onto Na/TNTs (a, b, c, d) and Mag/TNTs (e, f, g, h) at different temperatures (298, 308, 318, 328) K. | ||
3.3. Adsorption thermodynamics
A thermodynamic analysis was conducted to further evaluate the feasibility of MB adsorption onto Na/TNTs and Mag/TNTs, as well as the associated energetic changes. Several thermodynamic parameters were determined to provide a better understanding of the adsorption mechanism, including the thermodynamic equilibrium constant (Keq) and the standard Gibbs free energy of adsorption (ΔG°) at each temperature, along with the standard enthalpy (ΔH°) and entropy (ΔS°) of adsorption. These parameters were estimated using eqn (4)–(7).52,66
 | (4) |
ΔG° = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Keq Keq
| (5) |
| ΔG° = ΔH° − TΔS° | (6) |
 | (7) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Keq versus 1/T for both Na/TNTs and Mag/TNTs (Fig. 10). The linear regression yielded R2 values of 0.82 for Na/TNTs and 0.81 for Mag/TNTs, indicating that the adsorption process does not fully conform to ideal Van't Hoff behavior. Such deviations typically arise from heterogeneous adsorption sites or temperature-dependent adsorption pathways.67 Consequently, the derived thermodynamic parameters should be interpreted with caution, and the uncertainties (standard errors) are provided to reflect the limited precision of the linear fit. Table 4 reports the thermodynamic parameters along with their associated standard errors. The uncertainties in ΔG° were propagated from the errors in ΔH° and ΔS°. From the slope and intercept of the line, the ΔH° and ΔS° were calculated, respectively. The relatively large standard errors for ΔH° and ΔS° reflect the limited precision of the linear fit.
Keq versus 1/T for both Na/TNTs and Mag/TNTs (Fig. 10). The linear regression yielded R2 values of 0.82 for Na/TNTs and 0.81 for Mag/TNTs, indicating that the adsorption process does not fully conform to ideal Van't Hoff behavior. Such deviations typically arise from heterogeneous adsorption sites or temperature-dependent adsorption pathways.67 Consequently, the derived thermodynamic parameters should be interpreted with caution, and the uncertainties (standard errors) are provided to reflect the limited precision of the linear fit. Table 4 reports the thermodynamic parameters along with their associated standard errors. The uncertainties in ΔG° were propagated from the errors in ΔH° and ΔS°. From the slope and intercept of the line, the ΔH° and ΔS° were calculated, respectively. The relatively large standard errors for ΔH° and ΔS° reflect the limited precision of the linear fit.
| T (K) | Na/TNTs | Mag/TNTs | ||||||
|---|---|---|---|---|---|---|---|---|
| ΔG° (kJ mol−1) | ΔH° (kJ mol−1) | ΔS° (J mol−1 K−1) | R2 | ΔG° (kJ mol−1) | ΔH° (kJ mol−1) | ΔS° (J mol−1 K−1) | R2 | |
| 298 | −34.3 ± 1.7 | −42.9 ± 13.8 | −28.8 ± 44.3 | 0.82 | −31.8 ± 1.6 | −37.9 ± 12.9 | −20.4 ± 41.4 | 0.81 |
| 308 | −34.0 ± 1.7 | — | — | — | −31.6 ± 1.6 | — | — | — |
| 318 | −33.7 ± 1.7 | — | — | — | −31.4 ± 1.6 | — | — | — |
| 328 | −33.4 ± 1.7 | — | — | — | −31.2 ± 1.6 | — | — | — |
The nature of the adsorption process, whether exothermic or endothermic, is determined by the positive or negative value of ΔH°. The sign and magnitude of ΔS° indicate the degree of randomness at the solid–solution interface.68 Moreover, the ΔG° provides insights into the spontaneity of the adsorption process at various temperatures.69 The ΔG° values for both adsorbents were negative at all studied temperatures (Table 4), indicating that the adsorption process is spontaneous. The negative ΔH° values confirm that the adsorption process is exothermic for both materials. Additionally, the negative ΔS° values suggest a decrease in randomness at the solid–solution interface during adsorption. Although the negative ΔH° and ΔS° values indicate an exothermic process with reduced interfacial randomness, the adsorption capacity increased with temperature. This suggests that although the process is thermodynamically exothermic, the overall adsorption performance is governed primarily by kinetic and diffusion effects,28,42,51,70 which is consistent with the trends observed in our concentration and temperature studies.
3.4. Adsorbent reusability
The adsorption capacity of Na/TNTs and Mag/TNTs showed a slight decrease after five successive regeneration cycles during the removal of MB, as explained in Fig. 11, with values of (78.7 and 78.6, 71.5 and 69.5, 71.06) mg g−1 and (68.2, 70.7, and 67.3, and 65.2 and 61.8) mg g−1, respectively. The results confirmed their good reusability and potential as efficient adsorbents for wastewater treatment applications.This study presents a comparative analysis of sodium titanate nanotubes with other related materials of the same chemical structure for the removal of MB dye. In our research, TNTs showed higher adsorption capacity than other adsorbents listed in Table 5, and for the first time, Mag/TNTs were used for dye removal, showing significant adsorption efficiency.
| Adsorbent | Adsorption capacity (mg g−1) | Reference |
|---|---|---|
| Graphene oxide/ZnTiO3/TiO2 | 78 | 71 |
| Activated carbon fiber supported titanate nanotubes (ACF@TNTs) | 32.7 | 72 |
| H-TNT and H-TNS (H2Ti3O7) | 7.25 and 6.67 | 73 |
| Na–BaLa4Ti4O15 | 37.6 | 74 |
| Cerium titanate cellulose fiber nanocomposite (Ce–Ti/Cf) | 54.29 | 75 |
| Titanate nanotube (TNTs) | 151.51 | 76 |
| Potassium titanate (K2TiO3) | 21.92 | 77 |
| Carbonaceous mesoporous titanate-based materials (Na2Ti3O7/Na3Ti6O13) | 27.5 | 78 |
| Activated charcoal-supported TNTs (TNTs@PAC) | 173.30 | 79 |
| Na/TNTs | 178.98 (at 328 K) | Our study |
| Mag/TNTs | 180.5 (at 328 K) | Our study |
3.5. Mechanism of MB adsorption
MC simulations were employed to estimate the adsorption sites and energies of a single MB molecule, a monolayer, and multilayers of MB dye on the Na2Ti3O7 and CoFe2O4 surfaces. The corresponding adsorption energies were −109.37 kcal mol−1 for Na2Ti3O7 and −446.33 kcal mol−1 for CoFe2O4, respectively. These values are significantly higher than typical experimental adsorption enthalpies for dye removal, which is explained by several factors. First, the calculated energy represents the sum of multiple simultaneous interactions per MB molecule. As shown in Fig. 12, when a single MB molecule attaches to the surface, its hydrogen atoms (from methyl or aromatic groups) form favorable electrostatic interactions with surface oxygen atoms. These interactions occur at distances of 2.58–2.97 Å for Na2Ti3O7 and 2.3–2.8 Å for CoFe2O4. Additionally, metal–π interactions were observed, wherein the aromatic rings of MB orient parallel to surface metal atoms. Specifically, interactions were identified between the Ti atoms in Na2Ti3O7 (Fig. 12A) and the Fe atoms in CoFe2O4 (Fig. 12B) with the aromatic ring of the dye. The distances between the centroid of the benzenoid moiety of MB and the Ti and Fe were approximately 4.3 Å and 3.5 Å, respectively. These interaction distances are consistent with those reported for similar systems in the Cation–Aromatic Database (CAD), where cation–π distances typically range from 3.5 to 4.5 Å.80 Second, the simulation uses clean surfaces with a high density of unsaturated-coordinated metal and oxygen atoms, which are more reactive than fully coordinated surface sites found under experimental conditions. Consequently, each dye molecule simultaneously forms multiple contacts with the surface, and the calculated adsorption energy represents the cumulative sum of all these individual interactions. Third, the calculations are performed in vacuum, completely omitting the dielectric screening effect of water molecules and the competition from solvent molecules for adsorption sites. In aqueous solution, competition from water molecules and charge screening will significantly reduce the net adsorption energy. Consequently, these values should not be directly compared to experimental free energies of adsorption; instead, they are useful for comparing relative binding affinities and identifying interaction geometries. Although Na2Ti3O7 exhibits weaker interactions with MB, as indicated by its lower adsorption energies compared to CoFe2O4, its experimentally confirmed higher surface area facilitates the adsorption of a greater number of MB molecules. This accounts for the higher removal efficiency of Na2Ti3O7 observed experimentally. | ||
| Fig. 12 The lowest-energy structure of the adsorption of a single MB dye molecule on the surface of Na2Ti3O7 (A) and CoFe2O4 (B). | ||
Fig. 13 illustrates the lowest-energy configuration of a monolayer composed of 14 MB dye molecules adsorbed on the surfaces of Na2Ti3O7 and CoFe2O4. This configuration was selected to elucidate both dye–surface and dye–dye interactions. In the side view, close contacts are observed between the methyl or aromatic hydrogen atoms of MB with the surface oxygen atoms, with interaction distances ranging from 2.48 Å to 2.95 Å for Na2Ti3O7, and from 2.29 Å to 2.81 Å for CoFe2O4. Furthermore, metal–π interactions are evident, where the aromatic rings of MB align parallel to surface metal atoms, specifically Ti in Na2Ti3O7 and Fe/Co in CoFe2O4. In the top view, intermolecular attractions between adjacent dye molecules are apparent. These include van der Waals interactions between methyl hydrogen atoms and either other methyl hydrogens or the aromatic ring hydrogens/carbons of neighboring molecules. The distances associated with these interactions range from 2.46 Å to 2.95 Å for Na2Ti3O7 and from 2.34 Å to 2.55 Å for CoFe2O4.
 | ||
| Fig. 13 The lowest-energy structure of the adsorption of 14 MB dye molecules (monolayer) on the surface of Na2Ti3O7 (A) and CoFe2O4 (B). | ||
Fig. 14 presents the lowest-energy configuration of 28 MB dye molecules adsorbed as a multilayer on the surfaces of Na2Ti3O7 and CoFe2O4. This configuration primarily illustrates the intermolecular interactions (MB–MB) within the multilayer structure formed on the adsorbent surfaces. In the side view, interactions are observed between the methyl and aromatic hydrogen atoms of the dye molecules and the surface oxygen atoms, with distances ranging from 2.66 Å to 2.98 Å for Na2Ti3O7 and from 2.31 Å to 2.85 Å for CoFe2O4. The top view reveals multiple modes of MB–MB interaction. These include van der Waals forces between methyl hydrogen atoms and either other methyl hydrogens or aromatic ring hydrogens/carbons. The corresponding interaction distances range from 2.20 Å to 2.97 Å for Na2Ti3O7 and from 2.07 Å to 2.37 Å for CoFe2O4.
 | ||
| Fig. 14 The lowest-energy structure of the adsorption of 28 MB dye molecules (multilayer) on the surface of Na2Ti3O7 (A) and CoFe2O4 (B). | ||
4. Conclusion
In this study, Na/TNTs and Mag/TNTs were successfully prepared via hydrothermal method. The incorporation of cobalt ferrite into titanate nanotubes enhanced the rate of adsorption of MB dye and allowed easy separation. The synthesized nanotubes were characterized by SEM, HR-TEM, FTIR, BET, XRD, and zeta potential analysis. Na/TNTs and Mag/TNTs exhibited effective performance in the adsorption of MB dye, with adsorption capacities of (90.5, 146.1, 171.9, and 182.4) mg g−1 for Na/TNTs and (67, 120.8, 130.5, and 180) mg g−1 for Mag/TNTs at pH 5 and different temperatures (298, 308, 318, 328) K. Various parameters were studied to optimize the MB dye adsorption, such as pH, adsorbent dose, initial MB concentration, temperature, time, and adsorbent reusability. Isotherm models were used to fit the adsorption data obtained at different concentrations. In general, the best-fitting models for Na/TNTs and Mag/TNTs were Langmuir–Freundlich, Sips, and Dubinin–Radushkevich at the investigated temperature, indicating that the adsorbent materials possess heterogeneous surfaces and exhibit saturated monolayer adsorption behavior. Additionally, the pseudo-first-order, pseudo-second order, mixed 1,2-order, and Avrami models were employed to fit the experimental kinetic data. Among these models, the pseudo-second order and mixed 1,2-order models provided the best fit to the experimental results, implying that the adsorption proceeds through mixed mechanisms involving both physisorption and chemisorption. MC simulations revealed that although CoFe2O4 exhibits stronger individual interactions with MB dye, Na2Ti3O7 demonstrates higher overall dye removal efficiency, attributed to its larger surface area. The adsorption geometries confirmed favorable electrostatic and metal–π–π interactions, particularly between the hydrogen/aromatic rings of MB and the surface oxygen or metal atoms. In both monolayer and multilayer configurations, MB–MB interactions, primarily van der Waals forces, play a crucial role in stabilizing the adsorbed structures. These findings highlight the combined influence of surface characteristics and intermolecular interactions in governing the adsorption behavior of MB dye. Although Na/TNTs exhibited higher removal efficiency than Mag/TNTs, this work paves the way for the design of efficient magnetic titanate composites for wastewater treatment.Author contributions
Rania N. Zohny: writing–original draft, visualization, software, investigation, formal analysis, data curation. Ayman H. Zaki: conceptualization, methodology, investigation, supervision, writing review & editing. A. A. Farghali: supervision. Mohamed Taha: writing– review & editing, visualization, supervision, software, methodology, investigation, conceptualization.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
All data generated or analysed during this study are included in the manuscript.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d6ra02268e.
Acknowledgements
All gratitude to the Academy of Scientific Research and Technology (ASRT) in Egypt for their valuable financial support for this work. Scientists of the next generation (SNG) scholarship, Cycle 8.References
- J. A. Aznar-Sánchez, L. J. Belmonte-Ureña, J. F. Velasco-Muñoz and F. Manzano-Agugliaro, J. Cleaner Prod., 2018, 198, 1120–1132 CrossRef.
- R. Saleh, A. H. Zaki, F. I. A. El-Ela, A. A. Farghali, M. Taha and R. Mahmoud, J. Environ. Chem. Eng., 2021, 9, 104726 CrossRef CAS.
- N. Zolkefli, S. S. Sharuddin, M. Z. M. Yusoff, M. A. Hassan, T. Maeda and N. Ramli, Water, 2020, 12, 3417 CrossRef CAS.
- M. K. Hasan, A. Shahriar and K. U. Jim, Heliyon, 2019, 5, e02145 CrossRef PubMed.
- V. K. Garg, M. Amita, R. Kumar and R. Gupta, Dyes Pigm., 2004, 63, 243–250 CrossRef CAS.
- F. D. Chequer, G. R. De Oliveira, E. A. Ferraz, J. C. Cardoso, M. B. Zanoni and D. P. De Oliveira, Eco-Friendly Text. Dyeing Finish., 2013, 6, 151–176 Search PubMed.
- V. Katheresan, J. Kansedo and S. Y. Lau, J. Environ. Chem. Eng., 2018, 6, 4676–4697 CrossRef CAS.
- S. Dardouri and J. Sghaier, Korean J. Chem. Eng., 2017, 34, 1037–1043 CrossRef CAS.
- P. O. Oladoye, T. O. Ajiboye, E. O. Omotola and O. J. Oyewola, Results Eng., 2022, 100678 CrossRef CAS.
- R. Ahmad and R. Kumar, J. Environ. Manage., 2010, 91, 1032–1038 CrossRef CAS PubMed.
- B. Mu and A. Wang, J. Environ. Chem. Eng., 2016, 4, 1274–1294 CrossRef CAS.
- A. S. Abdulhameed, A. H. Jawad and A.-T. Mohammad, Bioresour. Technol., 2019, 293, 122071 CrossRef CAS PubMed.
- L. Ceroni, S. Benazzato, S. Pressi, L. Calvillo, E. Marotta and E. Menna, Nanomaterials, 2024, 14, 522 CrossRef CAS PubMed.
- M. Aaddouz, K. Azzaoui, N. Akartasse, E. Mejdoubi, B. Hammouti, M. Taleb, R. Sabbahi and S. Alshahateet, J. Mol. Struct., 2023, 1288, 135807 CrossRef CAS.
- M. H. Kahsay, Appl. Water Sci., 2021, 11, 45 CrossRef CAS.
- A. K. Inamdar, N. R. Hulsure, A. S. Kadam, R. S. Rajenimbalkar, R. Karpoormath, S. B. Shelke and S. N. Inamdar, Results Chem., 2023, 5, 100854 CrossRef CAS.
- T. Tatarchuk, N. Paliychuk, R. B. Bitra, A. Shyichuk, M. Naushad, I. Mironyuk and D. Ziółkowska, Desalin. Water Treat., 2019, 150, 374–385 CrossRef CAS.
- M. Hinojosa-Reyes, R. Camposeco-Solis and F. Ruiz, Microporous Mesoporous Mater., 2019, 276, 183–191 CrossRef CAS.
- X. Lei, X. Li, Z. Ruan, T. Zhang, F. Pan, Q. Li, D. Xia and J. Fu, J. Mol. Liq., 2018, 266, 122–131 CrossRef CAS.
- T. Tuutijärvi, J. Lu, M. Sillanpää and G. Chen, J. Hazard. Mater., 2009, 166, 1415–1420 CrossRef PubMed.
- H. Jing, C. G. Hua and I. Lo, Water Res., 2005, 39, 4528–4536 CrossRef PubMed.
- S. J. Salih and Z. S. Tahseen, Next Sustain., 2026, 7, 100240 CrossRef.
- P. A. Vinosha, A. Manikandan, A. C. Preetha, A. Dinesh, Y. Slimani, M. A. Almessiere, A. Baykal, B. Xavier and G. F. Nirmala, J. Supercond. Novel Magn., 2021, 34, 995–1018 CrossRef CAS.
- S. Kumar, F. Ahmed, N. M. Shaalan, R. Kumar, A. Alshoaibi, N. Arshi, S. Dalela, F. Sayeed, S. Dwivedi and K. Kumari, Materials, 2022, 15, 7955 CrossRef CAS PubMed.
- A. Zaki, M. A. Hafiez, W. M. El Rouby, S. El-Dek and A. Farghali, J. Magn. Magn. Mater., 2019, 476, 207–212 CrossRef CAS.
- J. Zhu, Q. Liu, Z. Li, J. Liu, H. Zhang, R. Li and J. Wang, J. Hazard. Mater., 2018, 353, 9–17 CrossRef CAS PubMed.
- Y. Hu, C. Zou, T. Xiong and H. Wang, J. Ind. Eng. Chem., 2023, 128, 294–305 CrossRef CAS.
- A. Abdelkarim, A. H. Zaki, S. I. El-Dek and M. Taha, J. Mol. Liq., 2024, 415, 126414 CrossRef CAS.
- A. Jain, S. P. Ong, G. Hautier, W. Chen, W. D. Richards, S. Dacek, S. Cholia, D. Gunter, D. Skinner and G. Ceder, APL Mater., 2013, 1, 011002 CrossRef.
- H. Sun, Z. Jin, C. Yang, R. L. Akkermans, S. H. Robertson, N. A. Spenley, S. Miller and S. M. Todd, J. Mol. Model., 2016, 22, 1–10 Search PubMed.
- M. Makowska-Janusik, K. Filipecka-Szymczyk, D. Pelczarski, W. Stampor and M. Zalas, Molecules, 2025, 30, 1312 CrossRef CAS PubMed.
- D. Delahaye, S. Chaimatanan and M. Mongeau, in Handbook of Metaheuristics, Springer, 2018, pp. 1–35 Search PubMed.
- E. S. Elbanna, A. A. Farghali, M. H. Khedr and M. Taha, J. Mol. Liq., 2024, 409, 125538 CrossRef.
- W. Gao, Y. Chen, B. Li, S.-P. Liu, X. Liu and Q. Jiang, Nat. Commun., 2020, 11, 1196 CrossRef CAS PubMed.
- B. B. Majumdar, V. Prytkova, E. K. Wong, J. A. Freites, D. J. Tobias and M. Heyden, J. Chem. Theory Comput., 2019, 15, 1399–1408 CrossRef CAS PubMed.
- B. Verberck, in Applications of Monte Carlo Method in Science and Engineering, IntechOpen, 2011 Search PubMed.
- N. M. Refat, M. Y. Nassar and S. A. Sadeek, RSC Adv., 2022, 12, 25081–25095 RSC.
- A. H. Zaki and M.-J. Lee, ACS Omega, 2019, 4, 19623–19634 CrossRef CAS PubMed.
- M. Morsy, A. I. Abdel-Salam, D. A. Rayan, I. Gomaa and A. Elzwawy, J. Nanopart. Res., 2022, 24, 226 CrossRef CAS.
- A. H. Zaki, A. A. Naeim and S. I. El-Dek, Environ. Sci. Pollut. Res., 2019, 26, 36388–36400 CrossRef CAS PubMed.
- Y. Zhang, D. Shao, J. Yan, X. Jia, Y. Li, P. Yu and T. Zhang, J. Nat. Gas Geosci., 2016, 1, 213–220 CrossRef.
- B. S. Yadav and S. Dasgupta, Inorg. Chem. Commun., 2022, 137, 109203 CrossRef CAS.
- H. Ighnih, R. Haounati, H. Ouachtak, A. Regti, B. El Ibrahimi, N. Hafid, A. Jada, M. L. Taha and A. A. Addi, Inorg. Chem. Commun., 2023, 153, 110886 CrossRef CAS.
- H. Ouachtak, R. El Haouti, A. El Guerdaoui, R. Haounati, E. Amaterz, A. A. Addi, F. Akbal and M. L. Taha, J. Mol. Liq., 2020, 309, 113142 CrossRef CAS.
- Z. Iqbal, M. S. Tanweer and M. Alam, ACS Omega, 2023, 8, 6376–6390 CrossRef CAS PubMed.
- J. Wang and X. Guo, J. Hazard. Mater., 2020, 390, 122156 CrossRef CAS PubMed.
- K. Tan and B. Hameed, J. Taiwan Inst. Chem. Eng., 2017, 74, 25–48 CrossRef CAS.
- J. Wang and X. Guo, Chemosphere, 2022, 309, 136732 CrossRef CAS PubMed.
- X. Guo, Y. Liu and J. Wang, Mar. Pollut. Bull., 2019, 145, 547–554 CrossRef CAS PubMed.
- E. P. Komarala, S. Doshi, S. Thiyagarajan, M. Aslam and D. Bahadur, New J. Chem., 2018, 42, 129–136 RSC.
- W. Jia, L. Sun, H. Li, T. Lv, Y. Tang and Y. Pi, Front. Mar. Sci., 2025, 12, 1542584 CrossRef.
- B. M. Thamer, F. A. Al-Aizari and H. S. Abdo, Molecules, 2023, 28, 7712 CrossRef CAS PubMed.
- R. Gopinathan, A. Bhowal and C. Garlapati, J. Chem. Thermodyn., 2017, 107, 182–188 CrossRef CAS.
- G. K. Rajahmundry, C. Garlapati, P. S. Kumar, R. S. Alwi and D.-V. N. Vo, Chemosphere, 2021, 276, 130176 CrossRef CAS PubMed.
- I. Narin, Y. Surme, M. Soylak and M. Dogan, J. Hazard. Mater., 2006, 136, 579–584 CrossRef CAS PubMed.
- S. N. Shankar, D. R. Dinakaran, D. K. Chandran, G. Mantha, B. Srinivasan and U. P. N. Kannaian, Energy Nexus, 2023, 10, 100197 CrossRef.
- A. B. D. Nandiyanto, G. C. S. Girsang, R. Maryanti, R. Ragadhita, S. Anggraeni, F. M. Fauzi, P. Sakinah, A. P. Astuti, D. Usdiyana and M. Fiandini, Commun. Sci. Technol., 2020, 5, 31–39 CrossRef.
- I. Langmuir, J. Am. Chem. Soc., 1916, 38, 2221–2295 CrossRef CAS.
- F. Okeola and E. Odebunmi, Adv. Nat. Appl. Sci., 2010, 281–289 CAS.
- R. A. Koble and T. E. Corrigan, Ind. Eng. Chem., 1952, 44, 383–387 CrossRef CAS.
- J. Wang and X. Guo, Chemosphere, 2020, 258, 127279 CrossRef CAS PubMed.
- H. Freundlich, Kapillarchemie, eine Darstellung der Chemie der Kolloide und verwandter Gebiete, Akademische Verlagsgesellschaft, 1930 Search PubMed.
- R. Sips, J. Chem. Phys., 1948, 16, 490–495 CrossRef CAS.
- X.-L. Wu, P. Xiao, S. Zhong, K. Fang, H. Lin and J. Chen, RSC Adv., 2017, 7, 28145–28151 RSC.
- J. Wang, M. Guan, Z. Qin, S. Zhang, J. Cheng and B. Xin, Water, 2025, 17, 1630 CrossRef CAS.
- E. C. Lima, A. Hosseini-Bandegharaei, J. C. Moreno-Piraján and I. Anastopoulos, J. Mol. Liq., 2019, 273, 425–434 CrossRef CAS.
- F. Gritti and G. Guiochon, Anal. Chem., 2006, 78, 4642–4653 CrossRef CAS PubMed.
- E. M. A. Rahman, G. M. El-Subruiti, E. R. Atta and A. H. Moustafa, ACS Omega, 2025, 10, 32680–32690 CrossRef PubMed.
- K. M. Doke and E. M. Khan, Rev. Environ. Sci. Bio/Technol., 2013, 12, 25–44 CrossRef CAS.
- A. N. Ebelegi, N. Ayawei and D. Wankasi, Open J. Phys. Chem., 2020, 10, 166–182 CrossRef CAS.
- X. Jaramillo-Fierro and G. Cuenca, Int. J. Mol. Sci., 2024, 25, 4367 CrossRef CAS PubMed.
- Q. Wang, X. Lei, F. Pan, D. Xia, Y. Shang, W. Sun and W. Liu, Colloids Surf., A, 2018, 555, 605–614 CrossRef CAS.
- A. Zaki, S. Rashad, M.-J. Lee and N. Shehata, Mater. Adv., 2022, 3, 8977–8988 RSC.
- A.-L. Chang, B.-S. Nguyen, V.-H. Nguyen and C. Hu, Mater. Chem. Phys., 2022, 276, 125363 CrossRef CAS.
- Y. H. Kotp, Environ. Sci. Pollut. Res., 2022, 29, 81583–81608 CrossRef CAS PubMed.
- S. Mohanty and S. K. Maji, Water Sci. Technol., 2020, 82, 2562–2575 CrossRef CAS PubMed.
- S. Pysarenko, O. Kaminskyi, O. Chyhyrynets, R. Denysiuk and V. Chernenko, Mater. Today: Proc., 2022, 62, 7754–7758 CAS.
- N. L. M. Tri, D. T. Thoa, N. H. Tri, N. M. Viet, T. T. V. Ha and N. C. Khoa, Vietnam J. Chem., 2022, 60, 708–717 CrossRef.
- Y. Lin, J. Ma, W. Liu, Z. Li and K. He, Environ. Sci. Pollut. Res., 2019, 26, 10263–10273 CrossRef CAS PubMed.
- A. S. Mahadevi and G. N. Sastry, Chem. Rev., 2013, 113, 2100–2138 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |












