 Open Access Article
Open Access ArticlePt–Y supported on magnesium–aluminium composite oxide catalysts for highly selective synthesis of 1,2-pentanediol from furfuryl alcohol under mild conditions
Xiaodong Zhouab,
Fengqin Guob,
Ziqiang Hanbc,
Jiajun Zhang bc,
Yan Caobc,
Huiquan Li
bc,
Yan Caobc,
Huiquan Li bc,
Guozhu Chen
bc,
Guozhu Chen *a and
Liguo Wang
*a and
Liguo Wang *bc
*bc
aSchool of Chemistry and Chemical Engineering, University of Jinan, Jinan, 250022, China. E-mail: chm_chengz@ujn.edu.cn
bNational Engineering Research Centre of Green Recycling for Strategic Metal Resources, Institute of Process Engineering, Chinese Academy of Science, Beijing, 100190, China. E-mail: lgwang@ipe.ac.cn
cSchool of Chemical Engineering, University of Chinese Academy of Sciences, Beijing, 100049, China
First published on 5th May 2026
Abstract
The selective hydrogenolysis of furfuryl alcohol (FFA) to 1,2-pentanediol (1,2-PeD) represents a promising route for biomass valorization, yet it remains challenging due to the competing over-hydrogenation of the furan ring and the recalcitrant C5–O bond cleavage. Herein, we report a highly efficient bimetallic Pt–Y catalyst supported on a MgO–AlO(OH) mixed oxide (MAO) that enables the selective hydrogenolysis of FFA to 1,2-PeD under remarkably mild conditions (140 °C, 0.8 MPa H2). Structural characterizations (AC-HAADF-STEM, XPS) confirm the formation of Pt–Y mixed clusters, which induce electron transfer from Pt to Y. This electronic modulation, combined with the tailored basicity of the MAO support, effectively suppresses the over-hydrogenation pathway while promoting the activation of the target C–O bond. In situ Fourier transform infrared spectroscopy shows that after the introduction of Y, FFA adsorbs on the catalyst surface in a vertical adsorption configuration through its O–H and C–O–C bonds, promoting the selective ring-opening. Consequently, the optimized 0.75Pt0.45Y/MAO catalyst achieves complete FFA conversion with an exceptional 1,2-PeD selectivity of 80.3% (110 mol 1,2-PeD·mol Pt per h), significantly outperforming its monometallic counterparts. Furthermore, the catalyst demonstrates outstanding stability in a continuous-flow reactor for over 200 hours without obvious deactivation. This work provides a strategy for designing synergistic bimetallic catalysts for the sustainable production of 1,2-PeD under mild conditions.
1 Introduction
The catalytic valorisation of biomass into high-value chemicals represents a pivotal and growing research frontier in sustainable chemistry.1–3 Furfuryl alcohol (FFA), as an important biomass platform molecule, can be efficiently produced through the acid-catalysed hydrolysis of agricultural and forestry waste (such as corn cobs, sugarcane bagasse, etc.) and subsequent reduction reactions.4,5 Furthermore, it can be converted into various downstream products through selective hydrogenation or hydrogenolysis, including 1,5-PeD, tetrahydrofurfuryl alcohol (THFOL), 1,2-PeD, 2-pentanol (2-PeOH), 1-pentanol (1-PeOH), and other high-value chemicals.6,7 Among these, the selective hydrogenation of furfuryl alcohol to produce 1,2-pentanediol (1,2-PeD) has attracted significant attention due to its broad application prospects. 1,2-Pentanediol is an important polyol compound widely used in pharmaceutical intermediates, high-performance polymer monomers, and environmentally friendly solvents. It is a key industrial raw material in the manufacturing processes of polyester fibres and surfactants.8,9 However, traditional production processes typically rely on multi-step oxidation or hydrolysis reactions of petroleum-based feedstocks, suffering from high energy consumption and low atom economy.10 Therefore, developing efficient catalyst based on biomass derivatives for the green synthesis of 1,2-pentanediol holds significant scientific importance and industrial value.11,12As early as the 1930s, the CuCr2O4 catalyst system was proven to exhibit catalytic activity for the direct hydrogenolysis of furfural/furfuryl alcohol (FA/FFA) compounds to produce pentanediols. However, the environmental pollution issue associated with chromium in this system significantly conflicts with contemporary green chemistry principles. To overcome this technical bottleneck, significant progress has been made in recent years in developing new catalytic systems: Zhang et al.13 reported a Ru/MnOx catalyst with basic sites as active sites for the hydrogenolysis of FFA to 1,2-PeD at 150 °C and 1.5 MPa H2 pressure, achieving 89.2% FFA conversion. However, the yield of 1,2-PeD was only 42.1%, and its catalytic stability and target product yield still need improvement. Kaneda et al.8 reported complete conversion of FA and 73% selectivity to 1,2-PeD catalysed by Pt/hydrotalcite (HT) at 150 °C and 3 MPa. Wang et al.14 studied the structure-performance relationship of platinum catalysts supported on CeO2. Under conditions of 165 °C and 2 MPa H2 pressure for 24 hours, Pt/CeO2-C exhibited the best catalytic performance: achieving complete conversion of furfural (FFA) and 77% selectivity towards the target product 1,2-pentanediol (1,2-PeD). This study revealed the important regulatory role of support crystal facet engineering on the hydrogenolysis performance of noble metal catalysts. Later, D.S. Pisal et al.15 reported complete FFA conversion and a high 1,2-PeD yield of 87% catalysed by a 1% Rh/OMS-2 catalyst at 160 °C and 3 MPa H2, representing the best result reported to date. In the development of non-noble metal systems, researchers have constructed alternative solution represented by copper-based catalysts, but their harsh reaction conditions and 1,2-pentanediol selectivity below 55% are difficult to meet industrial demands.16–19 Notably, current catalytic systems often rely on high-pressure hydrogen environments. This technical challenge has prompted the design and development of novel catalytic systems—specifically, the creation of catalysts that exhibit excellent catalytic performance under mild reaction conditions while maintaining a low loading of precious metals.
The selective hydrogenolysis of furfuryl alcohol (FFA) to 1,2-pentanediol (1,2-PeD) involves the cleavage of the C–O bond in the furan ring, which is typically facilitated by the presence of an unsaturated C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond adjacent to the oxygen atom. This unsaturation is crucial for the activation and ring-opening of the furan ring under mild conditions. However, a major competing reaction is the hydrogenation of the furan ring to form tetrahydrofurfuryl alcohol (THFA), which saturates the C
C bond adjacent to the oxygen atom. This unsaturation is crucial for the activation and ring-opening of the furan ring under mild conditions. However, a major competing reaction is the hydrogenation of the furan ring to form tetrahydrofurfuryl alcohol (THFA), which saturates the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds and results in a fully hydrogenated, stable cyclic ether structure. Once THFA is formed, the C–O bond within the saturated ring becomes significantly more resistant to cleavage, making subsequent ring-opening to 1,2-PeD virtually impossible under the same mild reaction conditions. Therefore, suppressing the formation of THFA is essential to preserve the integrity of the C
C bonds and results in a fully hydrogenated, stable cyclic ether structure. Once THFA is formed, the C–O bond within the saturated ring becomes significantly more resistant to cleavage, making subsequent ring-opening to 1,2-PeD virtually impossible under the same mild reaction conditions. Therefore, suppressing the formation of THFA is essential to preserve the integrity of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond, which serves as a key structural feature for facilitating the targeted C–O bond cleavage and ensuring high selectivity toward 1,2-PeD.20 Previous reports have indicated that the adsorption configuration of furfuryl alcohol on the catalyst is crucial for selectivity.5,21 Introducing a second metal as a promoter can modulate the electronic structure of the noble metal and create dual active sites.:22,23 the noble metal site is responsible for hydrogen activation, while the promoter site enhances C–O bond adsorption and activation capability. For instance, Liu et al.24 developed Pt/Ca co-modified nitrogen-doped carbon catalysts (xPtCa@CN) for the conversion of furfural (FA) to 1,2-pentanediol (1,2-PeD). Furthermore, previous reports have indicated that introducing transition metals benefit the adsorption of oxygen-containing groups and altering adsorption configurations due to the oxophilicity of transition metal cations.25–27 This demonstrates the effectiveness of the bimetallic strategy in promoting C–O hydrocracking of biomass-derived molecules and enhancing the effectiveness of selectivity through the regulation of adsorption configurations.
C bond, which serves as a key structural feature for facilitating the targeted C–O bond cleavage and ensuring high selectivity toward 1,2-PeD.20 Previous reports have indicated that the adsorption configuration of furfuryl alcohol on the catalyst is crucial for selectivity.5,21 Introducing a second metal as a promoter can modulate the electronic structure of the noble metal and create dual active sites.:22,23 the noble metal site is responsible for hydrogen activation, while the promoter site enhances C–O bond adsorption and activation capability. For instance, Liu et al.24 developed Pt/Ca co-modified nitrogen-doped carbon catalysts (xPtCa@CN) for the conversion of furfural (FA) to 1,2-pentanediol (1,2-PeD). Furthermore, previous reports have indicated that introducing transition metals benefit the adsorption of oxygen-containing groups and altering adsorption configurations due to the oxophilicity of transition metal cations.25–27 This demonstrates the effectiveness of the bimetallic strategy in promoting C–O hydrocracking of biomass-derived molecules and enhancing the effectiveness of selectivity through the regulation of adsorption configurations.
In this study, we attempted to introduce the transition metal Y into the catalyst to construct a new catalytic system Pt–Y/MAO (MAO refers to a mixture of magnesium oxide and basic aluminium oxide, which is used to provide a moderately strong alkaline environment). Yttrium (Y) was chosen as a promoter due to its strong oxophilicity, which enhances the adsorption of oxygen-containing groups in furfuryl alcohol (FFA) and facilitates C–O bond activation. The MgO–AlO(OH) (MAO) support, on the other hand, offers moderately basic sites that promote O–H bond activation and hydrogen transfer, while the presence of AlO(OH) modulates the surface basicity to avoid excessive side reactions. The high surface area of MAO also ensures good dispersion of Pt–Y clusters. Together, these features are anticipated to direct FFA adsorption into a vertical configuration, favouring ring-opening over ring hydrogenation. It can catalyse the FA/FFA hydrogenolysis reaction under mild conditions as low as 0.8 MPa hydrogen pressure and 140 °C, providing up to 80.3% selectivity for 1,2-PeD, and it is capable of achieving a production rate of up to 110 mol1,2-PeD·per molPt per h under highly selective conditions. The use of biomass-derived feedstock, green solvent (water), mild reaction conditions, and a recyclable solid catalyst make this process sustainable and environmentally friendly.
2 Experimental
2.1 Catalyst synthesis
Preparation of MAO support. MAO support was prepared using the ball milling method. Specifically, light magnesium oxide and basic aluminium oxide were first mixed at a Mg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al molar ratio of 3
Al molar ratio of 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and ball-milled at 800 rpm for 8 h. The resulting mixture was then calcined in a muffle furnace at 500 °C for 4 h. The MAO support was synthesized via a mechanochemical ball milling method, achieving thorough mixing of MgO and AlO(OH). This approach enables the generation of materials with high specific surface area and enhances metal–support interactions. The mechanical forces during ball milling induce lattice defects and surface activation, promoting the formation of heterojunction structures and facilitating the subsequent anchoring of Pt–Y clusters.28
1 and ball-milled at 800 rpm for 8 h. The resulting mixture was then calcined in a muffle furnace at 500 °C for 4 h. The MAO support was synthesized via a mechanochemical ball milling method, achieving thorough mixing of MgO and AlO(OH). This approach enables the generation of materials with high specific surface area and enhances metal–support interactions. The mechanical forces during ball milling induce lattice defects and surface activation, promoting the formation of heterojunction structures and facilitating the subsequent anchoring of Pt–Y clusters.28
Preparation of Pt–Y/MAO catalysts. The Pt–Y/MAO catalysts were prepared by the impregnation method. 0.01 g of chloroplatinic acid hexahydrate (H2PtCl6·6H2O, Aladdin) and 0.01 g of yttrium nitrate hexahydrate (Y(NO3)3·6H2O, Aladdin) were dissolved in 2 g of water. Then, 0.5 g of MAO was added, and the mixture was left to stand for 12 hours. The resulting catalyst precursor was dried at 80 °C for 12 h and reduced in H2 at 300 °C for 60 min. After cooling to room temperature in H2, the 0.75 wt% Pt-0.45 wt% Y/MAO catalyst was obtained, denoted as 0.75Pt0.45Y/MAO. Catalysts with 0.75 wt% Pt/MAO and 0.45 wt% Y/MAO were also prepared using the same method. The actual loads of Pt and Y are determined by ICP (Table S1).
2.2. Characterizations of catalyst
The crystal structure was characterized by wide-angle X-ray diffraction (XRD) using a PANalytical Empyrean multi-functional diffractometer equipped with a Cu Kα radiation source (40 kV, 40 mA). The XRD patterns were collected over a 2θ range of 5–90° with a scanning rate of 10° min−1.High-Angle Annular Dark-Field Scanning Transmission Electron Microscopy (HAADF-STEM) images were obtained on a Thermo Fisher Talos F200S G2 instrument. Before imaging, passivated samples were ultrasonically dispersed in ethanol and then dropped onto copper TEM grids. Spherical aberration-corrected transmission electron microscopy (AC-HAADF-STEM) images were acquired using a JEM-ARM300F instrument. Prior to imaging, the sample was ultrasonically dispersed in ethanol and then deposited onto a copper grid for observation.
CO2 temperature-programmed desorption (TPD) and H2 TPD were performed on the same chemisorption apparatus used for CO2 and H2 chemisorption. Prior to the measurement, 50 mg of the calcined sample was pretreated in pure H2 at 600 °C for 30 min, followed by a purge pretreatment under helium flow (50 mL min−1) to eliminate interference from moisture adsorption and the hydroxyl structure in AlO(OH) on the catalyst surface. After cooling to 50 °C, the sample was saturated with a 10% H2/Ar or CO2/He gas mixture (50 mL min−1) for 1 h. The gas flow was then switched to Ar or He (30–50 mL min−1) for 1 h to remove weakly physically adsorbed H2. Finally, desorption was carried out by heating to 700 °C at a ramp rate of 10 °C min−1 under Ar or He atmosphere, and the desorbed gas was detected by TCD.
Quasi-in situ X-ray Photoelectron Spectroscopy (XPS) was conducted on a Thermo Scientific K-Alpha spectrometer equipped with an Al Kα X-ray source. Before measurement, calcined samples were pre-reduced at 300 °C for 60 min and then transferred to the measurement chamber without exposure to air. The binding energies of Pt and Y were calibrated using the C 1s peak of adventitious carbon at 284.8 eV.
In situ Fourier Transform Infrared Spectroscopy (In situ FT-IR; Model: Mettler Toledo React IR 15) was used to characterize the interaction between the catalyst and reactants during the reaction and to monitor reaction intermediates and products. Infrared signals were acquired every 1 min using an N2 detector and a 9.5 mm AgX fiber probe. For the FFA adsorption experiments, 50 mg of the pre-reduced catalyst powder was placed in the cell. It was first pretreated at 300 °C for 1 hour under a nitrogen flow (30 mL min−1) to clean the surface. The gas flow was then switched to a continuous stream of H2 saturated with FFA (introduced via a temperature-controlled bubbler), while background and adsorption spectra were collected. To investigate the temperature-dependent evolution of the adsorption configuration, a temperature-programmed process (heating rate: 2 °C min−1) was initiated under maintained FFA/H2 flow, allowing in situ monitoring of the dynamic changes in the infrared spectra.
2.3 Activity testing
The catalyst test was conducted in a 20 mL high-pressure autoclave. Typically, 50 mg of catalyst, 0.1 g of furfuryl alcohol, and 4 mL of water were added to the autoclave. After sealing, the reactor was purged with H2 at least three times and then pressurized to the desired pressure. The reaction is carried out at the required temperature for the specified duration of hours with stirring at 500 rpm. After completion, the liquid product was separated by centrifugation and analysed using a gas chromatograph (Extend-C18) equipped with a flame ionization detector (FID) and an FFAP column, with biphenyl as the internal standard. The carbon balance for each reaction was calculated by comparing the total carbon in the products and residual furfuryl alcohol to the initial carbon in furfuryl alcohol. All liquid products were quantified by GC-FID using biphenyl as the internal standard. The carbon content of each compound was calculated based on its molecular formula.The stability of the 0.75Pt0.45Y/MAO catalyst was investigated in a continuous flow fixed-bed reactor under the conditions of 140 °C and 0.8 MPa H2 (Scheme 1). First, quartz wool was filled at the bottom of the stainless-steel tubular reactor (inner diameter 9 mm, length 370 mm), then 1 g of the catalyst was loaded into the middle of the stainless-steel tubular reactor, and finally quartz sand was filled at the top of the reactor. Subsequently, the catalyst was reduced at 300 °C under a hydrogen atmosphere of 30 mL min−1 for 60 minutes, and then cooled to 140 °C. A 2.5 wt% furfuryl alcohol aqueous solution was pumped into the reactor at a flow rate of 0.01 mL min−1 (LHSV = 0.15 h−1), while hydrogen was introduced at a flow rate of 8 mL min−1 (GHSV = 171 h−1). A portion of the liquid product was analysed using a gas chromatograph equipped with an FID detector and an FFAP column (Extend-C18), with biphenyl used as the internal standard.
The conversion of FFA, and the selectivity of 1,2-PeD are calculated as follows:
 | (1) |
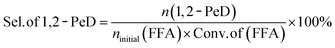 | (2) |
Among them, ninitial(FFA) represents the molar amount of furfuryl alcohol before the reaction; nresidual(FFA) denotes the molar amount of furfuryl alcohol after the reaction; and n(1,2-PeD) is the molar amount of 1,2-pentanediol.
3. Results and discussion
3.1. Structural investigation
The Pt–Y/MAO series catalysts were characterized to obtain structural information. XRD patterns showed the characteristic peaks of the MAO support composed of mixed phases of MgO and AlO(OH) (Fig. S1). The absence of XRD peaks corresponding to Pt or Y/Y2O3 phases, and the lack of new peaks in the XRD patterns of the metal-loaded catalysts compared to the MAO support without impregnation, indicate high dispersion of both components. Although a slight shift of the XRD peaks toward lower angles was observed after metal loading, incorporation of Y into the AlO(OH) lattice is unlikely, as such doping typically requires temperatures above 1100 °C, far exceeding our synthesis conditions.29,30 The reason may be that a small amount of Pt enters the near-surface lattice positions in the form of single atoms (Fig. S2), inducing local lattice expansion, which shifts the XRD diffraction peaks to lower angles. Regarding the decrease in peak width, we attribute this phenomenon to the grain growth of the support caused by heating during the reduction process of the catalyst. Subsequently, SEM characterization was conducted on the catalyst surface (Fig. S3). By comparing the morphologies of MAO and Pt–Y-MAO catalysts, it was found that they were basically the same, indicating that the impregnation method for loading metals does not significantly affect the surface morphology of the catalyst. The HAADF-STEM and HRTEM images of the 0.75 Pt/MAO and 0.75Pt0.45Y/MAO catalysts are shown in Fig. 1a and b. The results reveal that the MgO and AlO(OH) components are homogeneously mixed in both catalysts, with distinct lattice fringes from different phases clearly observable, exhibiting clear boundaries and close interconnection. The same phenomenon was also observed in the AC-HAADF-STEM image in Fig. 1c. The uniform distribution of Mg and Al elements also indicates that MgO and AlO(OH) are in close contact (Fig. 1f). These results provide sufficient evidence for the successful preparation of the heterojunction structure.31 The nanoparticles were evenly dispersed on the MAO support. The introduction of Y promoted nanoparticle growth (Fig. S4), suggesting a potential interaction between Pt and Y (Fig. 1d and e).Furthermore, aberration-corrected STEM characterization provided atomic-scale insight into the morphology of the metal species (Fig. 1h). For the 0.75Pt0.45Y/MAO catalyst, the loaded metals were predominantly present as sub-nanometric clusters. The co-localization of Pt and Y signals within the same clusters, as observed in elemental mapping, provides definitive evidence for their direct interaction.
3.2. The surface properties investigation
H2-TPD characterization was performed to evaluate the H2 activation capability of the MAO series catalysts (Fig. 2a). Corresponding to adsorbed hydrogen on the catalyst. As shown in Fig. 2a, the Pt-loaded sample exhibited the highest hydrogen adsorption capacity (431 µmol H2 gcat−1). After the introduction of the Y component, the hydrogen adsorption capacity decreased (196 µmol H2 gcat−1). Similarly, a comparable phenomenon was observed in CO2-TPD (Fig. 2b). The total base amount of the 0.75Pt0.45Y/MAO catalyst is 4.02 mmol g−1, which is significantly lower than that of the 0.75 Pt/MAO catalyst (6.56 mmol g−1). This can be explained by the significant weakening of H2 chemisorption due to the strong electronic interaction between Pt and Y. Similar phenomena have been observed previously in other systems.22,32,33 Fig. 2c presents the CO2-TPD profiles of the catalyst supports with different Mg/Al ratios, with a detailed discussion provided in the following section. As shown in Fig. S5, H2 uptake increased with Pt loading due to more surface Pt atoms. The desorption temperature rose slightly at higher loadings, indicating a weaker electronic effect from Y as the Y/Pt ratio decreased. The optimal 0.75Pt-0.45Y/MAO catalyst shows balanced H2 uptake and Pt–H strength, enabling selective C–O cleavage while suppressing ring hydrogenation. In contrast, lower Pt loading limits activity, while higher loading promotes Pt-like behaviour and increases ring hydrogenation risk.The electronic properties of Pt and Y in these Pt–Y/MAO catalysts were characterized by XPS. Although a portion of the Pt signal overlaps with the Al signal, a slight protrusion corresponding to the Pt4f7/2 component is observed.34 As shown in Fig. S6, for the freshly reduced 0.75 Pt/MAO catalyst, the Pt 4f7/2 binding energy was centred at 71.2 eV, indicating Pt is primarily in the metallic state (Pt0).35 In comparison, the Pt 4f7/2 peak binding energy for the 0.75Pt0.45Y/MAO catalyst was 71.6 eV, suggesting electron transfer from Pt to Y. On the other hand, Y 3d XPS showed that for the 0.8Pt0.4Y/MAO catalyst (Fig. 2e), the Y 3d3/2 binding energy was 156.2 eV, indicating a Y3+ valence state.36 While the Y 3d3/2 binding energy of the 0.45Y/MAO catalyst is centred at 156.6 eV, suggests electron transfer from Pt to Y, proving the bonding of Pt and Y, which is consistent with the TEM results. To further confirm, we measured the Pt 4d region (Fig. 2d), which is free from signal overlap. The Pt 4d binding energy of 0.75Pt0.45Y/MAO is 0.4 eV higher than that of 0.75 Pt/MAO, consistent with the Pt 4f results and confirming electron transfer from Pt to Y. The interaction between Pt and Y was further evaluated by CO pulse measurement and CO-TPD experiments. As shown in Fig. 2f and Table S2, the CO uptake decreased significantly upon Y addition, from 12.19 µmol g−1 for 0.75 Pt/MAO to 9.51 µmol g−1 for 0.75Pt-0.45Y/MAO. Concomitantly, the CO desorption temperature in the CO-TPD profiles shifted downward by 50 °C (Fig. 2f), providing direct evidence for the electronic modification of Pt by Y. Furthermore, previous reports have indicated that the presence of low-valent transition metal cations favours the adsorption of oxygen-containing groups due to their oxyphilicity.21–23 Therefore, it can be anticipated that the bonding of Y3+ with metallic Pt will facilitate the selective cleavage of the C–O bond on FFA.
3.3. Catalytic performance
The performance of materials for converting FFA to 1,2-PeD was evaluated (Fig. 3a). The conversion efficiency of FFA on MAO alone was very low. Similarly, Y/MAO also showed very low conversion efficiency. On the other hand, Pt/MAO catalyst exhibits high hydrogenation activity and achieves complete FFA conversion after six hours of reaction, simultaneously, the selectivity towards THFA was 31%, indicating that furan ring hydrogenation predominated, with a small portion undergoing cleavage reactions. To improve ring-opening selectivity, ring hydrogenation must be suppressed because once THFA is formed, this ring is difficult to cleave (Table S3). When a second component Y is present in the catalyst, even in very small amounts, THFA formation is greatly inhibited, and selectivity increases. Specifically, in order to compare the selectivity of 1,2-PeD after complete conversion, after the reaction lasted for six hours, the 0.75Pt0.45Y/MAO catalyst achieved 80.3% selectivity for 1,2-PeD, while hydrogenation byproducts including FFA and 1-butanol (1-BuOH) were marginal. This performance represents a significant improvement over the vast majority of those reported in the literature (Table 1). Among the Pt–Y/MAO catalysts mentioned above, the best-performing catalyst (0.75Pt0.45Y/MAO) had a Y/Pt atomic ratio of 1.67. We then investigated the influence of Pt content by fixing the Y/Pt ratio at 1.67, aiming to maximize Pt atomic efficiency. Pleasingly, when the Pt content was reduced from 0.75% to 0.4%, the 1,2-PeD selectivity only slightly decreased from 80.3% to 74%. When the Pt content was reduced to an extremely low value of 0.2%, the catalyst still maintained high selectivity towards ring-opening, achieving 82%, and although FFA conversion was only 12%, the 1,2-PeD selectivity hardly decreased, remaining above 65%. Previously, Shao et al. reported a selectivity of 63.9% for 1,2-PeD over a 2% Pt/Mg7AlO catalyst.18 Compared to that result, our Pt–Y/MAO clearly provides higher ring-opening selectivity and higher Pt metal efficiency. This demonstrates the significant promoting effect of Y. As revealed by XPS analysis, electron transfer from Pt to Y, thereby inhibiting ring hydrogenation on Pt sites. Therefore, the introduction of Y as a promoter should be the reason for the greatly improved ring-opening selectivity.| Feedstock | Catalyst | Conditions | Conversion (%) | Selectivity (%) | Ref. | |
|---|---|---|---|---|---|---|
| 1,2-PeD | 1,5-PeD | |||||
| FFA | 4Ru/MnOx | 150 °C 1.5 MPa 6 h | 89.2 | 41.4 | — | 13 |
| FFA | 10Ru/Al2O3 | 200 °C 10 MPa 1 h | 100 | 32.0 | — | 40 |
| FFA | 1Ru-5Sn/ZnO | 140 °C 3.5 MPa 6 h | 100 | 84.5 | 9.2 | 41 |
| FFA | 5Ru/MgO | 190 °C 3 MPa 1 h | 100 | 42.0 | 2.9 | 42 |
| FA | 20Ru/PVP | 125 °C 2 MPa 48 h | 100 | 36.0 | — | 43 |
| FA | 3Pd/MMT-K | 220 °C 3.5 MPa 5 h | >99.0 | 66.0 | — | 44 |
| FFA | 4.5Pt/CeO2 | 165 °C 2 MPa 24 h | 49.8 | 77.1 | 7.3 | 45 |
| FFA | 0.8Pt0.4Fe/MT | 140 °C 1 MPa 15 h | 100 | 75.0 | 20.0 | 23 |
| FFA | 0.75Pt0.45Y/MAO | 140 °C 0.8 MPa 6 h | 100 | 80.3 | 10.0 | This work |
Previous research has indicated that water is involved in hydrogen transfer during the reaction, which in turn facilitates the ring-opening pathway.37 We have proved through experiments that there can be rapid proton exchange between water and the substrate (Fig. S7). And MgO can also promote the activation of water in the reaction.38 To investigate the influence of different Mg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al molar ratios on reaction performance, the effect of Pt–Y/MAO catalysts on the selective hydrogenation of FFA to 1,2-PeD was studied. The results are shown in Fig. 3b. As the Mg
Al molar ratios on reaction performance, the effect of Pt–Y/MAO catalysts on the selective hydrogenation of FFA to 1,2-PeD was studied. The results are shown in Fig. 3b. As the Mg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al molar ratio decreased, the yield of 1,2-PeD initially increased and then decreased. The presence of acidic and basic sites has a significant impact on catalytic activity.39 When the support consisted only of AlO(OH), FFA conversion was almost negligible. This might be because AlO(OH), although possessing basic sites, has weak basic strength and may lack sufficient metal-support interaction, leading to insufficient electron density on Pt/AlO(OH) and rendering it unable to activate furfuryl alcohol or intermediates. Secondly, when the support was MgO, the reaction was incomplete and selectivity was low (50%). This is because MgO has strong basicity, which can promote FFA activation through dehydrogenation or hydrogenolysis involving the C
Al molar ratio decreased, the yield of 1,2-PeD initially increased and then decreased. The presence of acidic and basic sites has a significant impact on catalytic activity.39 When the support consisted only of AlO(OH), FFA conversion was almost negligible. This might be because AlO(OH), although possessing basic sites, has weak basic strength and may lack sufficient metal-support interaction, leading to insufficient electron density on Pt/AlO(OH) and rendering it unable to activate furfuryl alcohol or intermediates. Secondly, when the support was MgO, the reaction was incomplete and selectivity was low (50%). This is because MgO has strong basicity, which can promote FFA activation through dehydrogenation or hydrogenolysis involving the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond. However, its excessive basicity may lead to over-hydrogenation, generating the byproduct THFA. Additionally, pure MgO has a low specific surface area and poor dispersion of active sites, resulting in insufficient conversion.46 For mixed supports, complete FFA conversion was achieved across different ratios. This might be because when MgO is mixed with AlO(OH), the weak basicity of AlO(OH) neutralizes the strong basicity of MgO, forming moderate basic strength that can activate FFA while avoiding excessive reaction. Simultaneously, the acidic sites of AlO(OH) are suppressed by MgO, reducing side reactions, this can also be demonstrated by conducting CO2-TPD tests on catalyst supporter with different Mg
O bond. However, its excessive basicity may lead to over-hydrogenation, generating the byproduct THFA. Additionally, pure MgO has a low specific surface area and poor dispersion of active sites, resulting in insufficient conversion.46 For mixed supports, complete FFA conversion was achieved across different ratios. This might be because when MgO is mixed with AlO(OH), the weak basicity of AlO(OH) neutralizes the strong basicity of MgO, forming moderate basic strength that can activate FFA while avoiding excessive reaction. Simultaneously, the acidic sites of AlO(OH) are suppressed by MgO, reducing side reactions, this can also be demonstrated by conducting CO2-TPD tests on catalyst supporter with different Mg![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Al molar ratios (Fig. 2c). Moreover, the strong electron-donating effect of MgO can modulate the electron density of Pt, promoting H2 dissociation and hydrogen transfer; while the high specific surface area of AlO(OH) improves the dispersion of Pt–Y particles, increasing the number of active sites. The combination places Pt–Y in an optimal electronic state, favouring the selective formation of 1,2-PeD. The highest 1,2-PeD yield (80.3%) was obtained at the optimal Mg/Al ratio (3
Al molar ratios (Fig. 2c). Moreover, the strong electron-donating effect of MgO can modulate the electron density of Pt, promoting H2 dissociation and hydrogen transfer; while the high specific surface area of AlO(OH) improves the dispersion of Pt–Y particles, increasing the number of active sites. The combination places Pt–Y in an optimal electronic state, favouring the selective formation of 1,2-PeD. The highest 1,2-PeD yield (80.3%) was obtained at the optimal Mg/Al ratio (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). At this ratio, basic sites on the support surface (from MgO) dominate but are not excessive, ensuring initial FFA activation; a small amount of AlO(OH) (25%) provides sufficient dispersion and weak acidic sites, stabilizing reaction intermediates and reducing excessive side reactions.
1). At this ratio, basic sites on the support surface (from MgO) dominate but are not excessive, ensuring initial FFA activation; a small amount of AlO(OH) (25%) provides sufficient dispersion and weak acidic sites, stabilizing reaction intermediates and reducing excessive side reactions.
The selectivity of FFA to 1,2-PeD over the 0.75Pt0.45Y/MAO catalyst at different reaction temperatures was studied (Fig. 3c). Below 120 °C, although the main product was 1,2-PeD, the reactants were not completely converted, indicating that FFA hydrogenolysis and ring-opening proceed smoothly. As the reaction temperature increased from 120 °C to 160 °C, the yield of 1,2-PeD significantly increased. Correspondingly, more alcohols (1,5-PeD and 1-BuOH) were formed. The selectivity of 1,2-PeD increased from 68.3% at 100 °C to 80.3% at 140 °C, while further increasing to 160 °C resulted in a 1,2-PeD yield of 72.5%. The byproduct 1-BuOH was not detected at reaction temperatures below 100 °C. As the reaction temperature continued to increase, the 1-BuOH yield increased from 2% to 6%, indicating that higher temperatures promote 1-BuOH formation, possibly due to the partial conversion of 1,2-PeD route to 1-BuOH. The conversion of FFA to 1,2-PeD over the 0.75Pt0.45Y/MAO catalyst under different hydrogen pressures was further investigated (Fig. 3d). As shown, FFA was completely converted even at 0.8 MPa H2 pressure, proving H2 saturation. However, when pressure was further increased to 2 MPa, it was found that excessive pressure promotes the formation of 1,5-PeD. This is likely because the high-pressure environment facilitates the cleavage of the C1–O bond, leading to this phenomenon.
3.4. Reaction kinetics and mechanism
Since the adsorption configuration of furan ring compounds plays an important role in the hydrogenation pathway and product selectivity.47,48 FT-IR spectroscopy of FFA was conducted to investigate the adsorption configuration of FFA on the catalyst surface and elucidate the mechanism of FFA conversion to 1,2-PeD. First, the adsorption of FFA on the Pt–Y/MAO catalyst at different temperatures was compared (Fig. 4a). Characteristic peaks at 1149, 1038, and 1320 cm−1 were assigned to the C–O–C and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds of FFA adsorbed on the Pt–Y/MAO catalyst.49 It indicates that the O atom in the furan ring adsorb onto the active sites of Pt–Y/MAO. When the temperature was increased from 30 °C to 180 °C, it could be observed that at low temperatures, only the characteristic peak for the C–O–C bond was present, demonstrating that low temperatures are not favourable for the adsorption of the C
C bonds of FFA adsorbed on the Pt–Y/MAO catalyst.49 It indicates that the O atom in the furan ring adsorb onto the active sites of Pt–Y/MAO. When the temperature was increased from 30 °C to 180 °C, it could be observed that at low temperatures, only the characteristic peak for the C–O–C bond was present, demonstrating that low temperatures are not favourable for the adsorption of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond. However, as the temperature gradually increased, the characteristic peak for the C
C bond. However, as the temperature gradually increased, the characteristic peak for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond gradually appeared at 1320 cm−1. This indicates the emergence of a new adsorption configuration for the C
C bond gradually appeared at 1320 cm−1. This indicates the emergence of a new adsorption configuration for the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond on the catalyst, signifying that increased temperature leads to more ring hydrogenation reactions, and indeed, previous experiments have confirmed this.
C bond on the catalyst, signifying that increased temperature leads to more ring hydrogenation reactions, and indeed, previous experiments have confirmed this.
 | ||
| Fig. 4 Adsorption configurations represented by FT-IR: (a) FFA on 0.75Pt0.45Y/MAO at different temperatures; (b) FFA on different catalysts. | ||
Subsequently, the adsorption of FFA on different catalysts was compared (Fig. 4b). The characteristic peaks at 1590 and 1320 cm−1 were assigned to the adsorption of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of FFA on the Pt–Y/MAO catalyst, while the peaks at 1149 and 1038 cm−1 were attributed to the adsorption of the C–O–C bond. First, regarding C–O–C bond adsorption, near 1147 cm−1, almost no adsorption peak of the C–O–C bond was observed on Pt/MAO compared to Pt–Y/MAO and Y/MAO. This demonstrates that introducing the Y component significantly enhances the catalyst's adsorption toward the C–O–C bond in the furan ring of the substrate. Moreover, for the adsorption of the C
C bond of FFA on the Pt–Y/MAO catalyst, while the peaks at 1149 and 1038 cm−1 were attributed to the adsorption of the C–O–C bond. First, regarding C–O–C bond adsorption, near 1147 cm−1, almost no adsorption peak of the C–O–C bond was observed on Pt/MAO compared to Pt–Y/MAO and Y/MAO. This demonstrates that introducing the Y component significantly enhances the catalyst's adsorption toward the C–O–C bond in the furan ring of the substrate. Moreover, for the adsorption of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of FFA, distinct adsorption peaks corresponding to the C
C bond of FFA, distinct adsorption peaks corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond were observed at 1590 cm−1 on both Pt–Y/MAO and Y/MAO, whereas the adsorption peak on Pt–Y/MAO was notably weak. This indicates that the coexistence of Pt and Y strongly suppresses the tilted adsorption configuration of the C
C bond were observed at 1590 cm−1 on both Pt–Y/MAO and Y/MAO, whereas the adsorption peak on Pt–Y/MAO was notably weak. This indicates that the coexistence of Pt and Y strongly suppresses the tilted adsorption configuration of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond of FFA on the catalyst surface—a configuration generally favourable for furan ring hydrogenation. Taken together, in the Pt–Y bimetallic system, the introduction of Y enhances adsorption toward the substrate's C–O–C bond, while the synergistic effect of Pt and Y weakens adsorption toward the C
C bond of FFA on the catalyst surface—a configuration generally favourable for furan ring hydrogenation. Taken together, in the Pt–Y bimetallic system, the introduction of Y enhances adsorption toward the substrate's C–O–C bond, while the synergistic effect of Pt and Y weakens adsorption toward the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond in the furan ring. This results in FFA maintaining an upright orientation on the catalyst surface. Such an adsorption configuration on Pt–Y/MAO strongly promotes the ring-opening reaction of the furan ring, which also explains why more ring hydrogenation reactions occur on the Pt/MAO catalyst.
C bond in the furan ring. This results in FFA maintaining an upright orientation on the catalyst surface. Such an adsorption configuration on Pt–Y/MAO strongly promotes the ring-opening reaction of the furan ring, which also explains why more ring hydrogenation reactions occur on the Pt/MAO catalyst.
To systematically evaluate the impact of yttrium modification on the kinetic behaviour of platinum-based catalysts in the ring-opening reaction, this study employed furfuryl alcohol (FFA) as a probe molecule to comparatively investigate the reaction performance of 0.8 Pt/MAO and 0.8Pt0.4Y/MAO (Y/Pt atomic ratio = 1.67) catalysts (Fig. 5). The experiments were conducted within a hydrogen pressure range of 0.5–3 MPa, and intrinsic kinetic parameters were obtained by monitoring the FFA conversion process. The results show that both catalysts exhibit zero-order reactions in the hydrogen reaction rate. Suggesting that the surface of the active sites was nearly saturated with hydrogen under the given conditions, and hydrogen activation was not the rate-determining step.
Concurrently, the reaction orders with respect to FFA were 0.5 and 0.8 for the two catalysts, respectively, reflecting competitive adsorption behaviour of FFA on the catalyst surface and indicating that the adsorption and activation processes of FFA are partially involved in the rate-determining step of the reaction. Furthermore, the apparent activation energy measured for the 0.8 Pt/MAO catalyst was 54 kJ mol−1, while that for the 0.8Pt0.4Y/MAO catalyst decreased to 44 kJ mol−1. This difference further indicates that the incorporation of yttrium not only alters the energy barrier heights along the reaction pathway but may also induce a restructuring of the electronic structure or local coordination environment of the active sites. Consequently, it influences the adsorption modes of reactant molecules and the reaction mechanism, thereby promoting the reaction.
Before calculations, MgO(100)/AlOOH(010) lateral structure (MgO(100)/AlOOH(010)-ls) was constructed based on MgO conventional standard cell (JCPDS #04-001-7295) and AlOOH conventional standard cell (JCPDS #98-000-0120). Furthermore, in the previous experiments, we found that using only AlO(OH) to load Pt–Y clusters had no catalytic effect at all. Therefore, we built Pt4Y2O3 on MgO and constructed the MgO(100)/AlOOH(010)-ls-Pt4Y2O3 cluster model. In slab model, thickness along c direction was set at 30 Å to avoid weak interactions between image. Adsorption energy of molecule on slab model was determined by Eads. Eads = E(AB) − E(A) − E(B). E(AB) represents the total energy of slab model A with molecule B adsorbed on surface. E(A) represents the total energy of slab model A. E(B) represents the total energy of molecule B in its gas phase. In this case, configurations of furfuryl alcohol were optimized in a 20 × 20 × 20 Å−3 cell box, respectively.
As shown in Fig. 6, FFA exhibits the highest adsorption energy on Pt–Y/MAO, while the adsorption energy decreases on Pt/MAO. In this work, all calculations were carried out through the Vienna Ab initio Simulation Package (VASP) with the projector augmented wave (PAW) method. The Perdew–Burke–Ernzerhof (PBE) functional with the generalized gradient approximation (GGA) method was used to do with the exchange–correlation functional, in combination with the DFT-D3 correction. The cut-off energy of the plane-wave basis is set at 450 eV. For optimization of geometry of slab model, the Brillouin Zone integration was performed with a Monkhorst–Pack5 k-point mesh of 1 × 2 × 1. The self-consistent calculations apply a convergence energy threshold of 10−5 eV, and the equilibrium geometries are optimized with maximum stress on each atom within 0.02 eV Å−1. Spin polarization method was adopted to describe magnetism of slab models. For the adsorption reaction of molecules, the energy is calculated through the following equations:
| Ead = Esurface+absorbate − Esurface − Eabsorbate |
 | ||
| Fig. 6 (a and c) Model construction of Pt/MAO and Pt–Y/MAO; (b and d) Adsorption energy of FFA on Pt/MAO and Pt–Y/MAO. | ||
Based on the above experimental results and previous literature, a possible reaction mechanism for FFA conversion to 1,2-PeD on the Pt–Y/MAO catalyst is proposed (Fig. 7). First, the O–H bond of FFA and the C–O–C bond on the furan ring adsorb onto the catalyst support surface and the Y site in the supported metal, respectively. Simultaneously, H2 dissociates into two H atoms on the Pt surface. The dissociated H atoms are activated by basic sites. Then, H attacks the carbon atom at the C5–O bond, leading to furan ring opening. Subsequently, the dissociated hydrogen reduces the remaining C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond to generate 1,2-PeD.
C bond to generate 1,2-PeD.
3.5. Stability
Besides activity and selectivity, catalyst stability is another crucial performance indicator, particularly important for practical applications. In most previous studies, the reaction from FFA to pentanediol was conducted in batch reactors.44,50 Limiting the assessment of long-term catalyst stability. In this work, based on catalyst screening, we further evaluated the long-term operation of the 0.75Pt0.4Y/MAO catalyst in a continuous fixed-bed reactor. Moreover, we maintained the conversion rate at 90% by controlling the amount of the catalyst, thereby proving that the catalyst was not used in excess. (Table S5).51 The results are shown in Fig. 8. The catalyst performed quite stably over a 200-hour runtime. The XPS analysis of the used catalysts revealed that the oxidation states of Pt and Y remained unchanged after the reaction (Fig. S11). The HAADF-STEM images of the used catalysts (Fig. S12) showed that the Pt–Y nanoparticles were still highly dispersed and there was no obvious sintering, indicating that the nanostructure of the active sites was not disrupted during the long-term reaction.4. Conclusions
In summary, we have successfully developed a bimetallic Pt–Y/MAO catalyst for the highly selective hydrogenolysis of biomass-derived furfuryl alcohol to 1,2-pentanediol under mild reaction conditions. The key to the superior performance lies in the synergistic interplay between Pt and Y. Detailed characterization revealed that Y coexists with Pt in the form of intimately associated clusters, facilitating electron transfer from Pt to Y. This electronic engineering, coupled with the modulated surface basicity of the MgO–AlO(OH) support, plays a dual role: it effectively suppresses the undesired over-hydrogenation of the furan ring and concurrently enhances the adsorption and activation of the critical C5–O bond. In situ spectroscopic studies further elucidated that this synergy promotes a vertical adsorption mode of FFA, directing the reaction pathway toward selective ring-opening. The optimized catalyst achieves a remarkable 1,2-PeD selectivity of 80.3% at complete conversion, alongside demonstrating excellent long-term stability. This study not only presents a highly active and stable catalyst for biomass upgrading but also offers fundamental insights into the design principle of bimetallic catalysts, where promoter metals can tune both electronic properties and reactant adsorption configurations to achieve precise selectivity control in complex reaction networks. The strategy underscores the potential of utilizing non-precious promoters to enhance the efficiency and sustainability of noble metal-catalysed biomass conversion processes.Author contributions
Xiaodong Zhou: original draft, investigation, validation, data curation. Fengqin Guo: investigation, validation. Ziqiang Han: investigation, validation. Jiajun Zhang: resources, supervision. Yan Cao: resources, supervision. Huiquan Li: supervision, funding acquisition. Guozhu Chen: writing – review, supervision. Liguo Wang: conceptualization, writing – review & editing, supervision, funding acquisition.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its supplementary information (SI) files. Should any raw data files be needed in another format they are available from the corresponding author upon reasonable request. Source data are provided with this paper. Supplementary information: Fig. S1: XRD patterns of the series of MAO, Y/MAO, Pt/MAO, Pt–Y/MAO catalysts; Fig. S2: AC-HAADF-STEM images of 0.75Pt-0.45Y/MAO catalysts; Fig. S3: (a and c)SEM images of MAO and (b and d) 0.75Pt-0.45Y/MAO catalysts; Fig. S4: particle size statistics on 0.75 Pt/MAO (a) and 0.75Pt-0.45Y/MAO (b) catalysts; Fig. S5: The H2-TPD results of catalyst carriers with different Pt loading amounts; Fig. S6: TOF values under different reaction pressure over 0.75 Pt/MAO and 0.75Pt0.45Y/MAO catalysts. Reaction conditions: 50 mg of 0.75 Pt/MAO or 50 mg of 0.75Pt0.45Y/MAO catalyst, 140 °C. For the hydrogen pressure effect: 0.1 g of FFA in 4 g of water, PH2: 0.5, 1, 2, 3 MPa; Fig. S7: TOF values under different FFA concentration over 0.75 Pt/MAO and 0.75Pt0.45Y/MAO catalysts. Reaction conditions: 50 mg of 0.75 Pt/MAO or 50 mg of 0.75Pt0.45Y/MAO catalyst, 4 g of water, 1 MPa H2, CFFA: 1.5%, 2.5%, 3.5%, 4.5%; Fig. S8: TOF values under different temperature over 0.75 Pt/MAO and 0.75Pt0.45Y/MAO catalysts. Reaction conditions: 0.1 g of FFA in 4 g of water, 1 MPa H2, 50 mg of 0.75 Pt/MAO, temperature range of 120–150 °C or 50 mg of 0.75Pt0.45Y/MAO, temperature range of 120–150 °C; Table S1: the amount of Pt and Y in the impregnation solution; Table S2: basic physical-chemical properties of different catalysts; Table S3: catalytic performance of the 0.75Pt0.45Y/MAO catalyst for the THFA hydrogenolysis; Table S4: carbon balance for FFA hydrogenolysis over 0.75Pt0.45Y/MAO catalyst; Table S5: comparison of conversion rates between catalyst in excess and catalyst in insufficient amount in a fixed-bed reactor. See DOI: https://doi.org/10.1039/d6ra02263d.Acknowledgements
This work was financially supported by National Natural Science Foundation of China. (NSFC 22278409), The National Key R&D Program of China. (2022YFC3902200).Notes and references
- K. Lee, Y. Jing, Y. Wang and N. Yan, Nat. Rev. Chem., 2022, 6, 635–652 CrossRef PubMed.
- R. Mariscal, P. Maireles-Torres, M. Ojeda, I. Sadaba and M. Lopez Granados, Energy Environ. Sci., 2016, 9, 1144–1189 RSC.
- S. Wang, H. Li and M. Wu, J. Clean. Prod., 2021, 303, 126825 CrossRef CAS.
- Y. Liu, X. Huang, Z. Huang, F. Su and D. Song, Appl. Catal., A, 2026, 711, 120754 CrossRef CAS.
- S. Dou, L. Ma, Y. Dong, Q. Zhu and X. Kong, J. Colloid Interface Sci., 2024, 663, 345–357 CrossRef CAS PubMed.
- K. Huang, Z. J. Brentzel, K. J. Barnett, J. A. Dumesic, G. W. Huber and C. T. Maravelias, ACS Sustain. Chem. Eng., 2017, 5, 4699–4706 CrossRef CAS.
- Z. Yu, N. Ji, J. Xiong, Y. Han, X. Li, R. Zhang, Y. Qiao, M. Zhang and X. Lu, Small, 2022, 18, 2201361 CrossRef CAS PubMed.
- T. Mizugaki, T. Yamakawa, Y. Nagatsu, Z. Maeno, T. Mitsudome, K. Jitsukawa and K. Kaneda, ACS Sustain. Chem. Eng., 2014, 2, 2243–2247 CrossRef CAS.
- Y. Shi, Y. Zhu, Y. Yang, Y.-W. Li and H. Jiao, ACS Catal., 2015, 5, 4020–4032 CrossRef CAS.
- Y. Li, Y. Shen, J. Zhang, C. Chen, X. Zhang, B. Geng and G. Wang, Fuel, 2025, 387, 134398 CrossRef CAS.
- R. Yangcheng, J. Li, J. He, Y. Zheng, H. Yu, C. Chen and J. Wang, Small, 2024, 20, 2309821 CrossRef CAS PubMed.
- H. Yu, Y. Xu, K. Havener, M. Zhang, L. Zhang, W. Wu and K. Huang, Small, 2022, 18, 2106893 CrossRef CAS PubMed.
- B. Zhang, Y. Zhu, G. Ding, H. Zheng and Y. Li, Green Chem., 2012, 14, 3402–3409 RSC.
- T. Tong, X. Liu, Y. Guo, M. N. Banis, Y. Hu and Y. Wang, J. Catal., 2018, 365, 420–428 CrossRef CAS.
- D. S. Pisal and G. D. Yadav, ACS Omega, 2019, 4, 1201–1214 CrossRef CAS PubMed.
- H. Liu, Z. Huang, F. Zhao, F. Cui, X. Li, C. Xia and J. Chen, Catal. Sci. Technol., 2016, 6, 668–671 RSC.
- F. Gao, H. Liu, X. Hu, J. Chen, Z. Huang and C. Xia, Chin. J. Catal., 2018, 39, 1711–1723 CrossRef CAS.
- Y. Shao, J. Wang, H. Du, K. Sun, Z. Zhang, L. Zhang, Q. Li, S. Zhang, Q. Liu and X. Hu, ACS Sustain. Chem. Eng., 2020, 8, 5217–5228 CrossRef CAS.
- T. P. Sulmonetti, B. Hu, S. Lee, P. K. Agrawal and C. W. Jones, ACS Sustain. Chem. Eng., 2017, 5, 8959–8969 CrossRef CAS.
- Y. Nakagawa, M. Tamura and K. Tomishige, ACS Catal., 2013, 3, 2655–2668 CrossRef CAS.
- J. Wang, S. Zhu, Y. He, G. Fan, X. Li, X. Jia, M. Dong and W. Fan, Catal. Today, 2024, 433, 114647 CrossRef CAS.
- B. Hu, Z. Li, B. Wang, L. Chen, X. Wang, X. Hu, Z. Bai, Y. Li, G. Chen, X. Luo and S.-F. Yin, Appl. Catal. B Environ. Energy, 2025, 371, 125196 CrossRef CAS.
- C. Cao, W. Guan, Q. Liu, L. Li, Y. Su, F. Liu, A. Wang and T. Zhang, Green Chem., 2024, 26, 6511–6519 RSC.
- H. Liu, Y. Qiu, R. Shan, J. Zhang, H. Yuan and Y. Chen, J. Environ. Chem. Eng., 2025, 13, 118125 CrossRef CAS.
- Y. Ren, Y. Tang, L. Zhang, X. Liu, L. Li, S. Miao, D. S. Su, A. Wang, J. Li and T. Zhang, Nat. Commun., 2019, 10, 4500 CrossRef PubMed.
- Y. Zhang, L. Li, F. Liu, H. Qi, L. Zhang, W. Guan, Y. Liu, A. Wang and T. Zhang, ACS Catal., 2022, 12, 6302–6312 CrossRef CAS.
- Y. Zhang, F. Zhang, L. Li, H. Qi, Z. Yu, X. Liu, C. Cao, F. Liu, A. Wang and T. Zhang, J. Catal., 2023, 417, 301–313 CrossRef CAS.
- D. Leybo, K. L. Firestein, N. D. Evdokimenko, A. A. Ryzhova, V. S. Baidyshev, I. V. Chepkasov, Z. I. Popov, A. L. Kustov, A. S. Konopatsky, D. V. Golberg and D. V. Shtansky, ACS Appl. Nano Mater., 2022, 5, 16475–16488 CrossRef CAS.
- F. Nahif, S. Mráz, D. Music, P. Keuter and J. M. Schneider, Surf. Coat. Technol., 2014, 257, 333–337 CrossRef CAS.
- D. Xu, H. Jiang, M. Li, O. Hai and Y. Zhang, Ceram. Int., 2015, 41, 5355–5361 CrossRef CAS.
- X. Gong, Z. Fan, Y. Wang, X. Qiu, Y. Xie, Y. Ling and J. Zhao, Appl. Catal., A, 2023, 668, 119470 CrossRef CAS.
- N. Lei, X. Zhao, B. Hou, M. Yang, M. Zhou, F. Liu, A. Wang and T. Zhang, Chemcatchem, 2019, 11, 3903–3912 CrossRef CAS.
- N. Xi, Q. Li, Y. Chen, R. Bao, Q. Wang, Y. Lin, J. Yue, R. Wang, C. Yang, W. Yin and T. Qiu, Chem. Eng. J., 2025, 512, 162222 CrossRef CAS.
- J. Wang, S. Zhu, Z. Lv, X. Jia, X. Li, M. Dong and W. Fan, Catal. Today, 2025, 447, 115145 CrossRef CAS.
- X. Jin, R. Tsukimura, T. Aihara, H. Miura, T. Shishido and K. Nozaki, Nat. Catal., 2021, 4, 312–321 CrossRef CAS.
- S. Damyanova, I. Shtereva, B. Pawelec, L. Mihaylov and J. L. G. Fierro, Appl. Catal., B, 2020, 278 Search PubMed.
- R. Ma, X.-P. Wu, T. Tong, Z.-J. Shao, Y. Wang, X. Liu, Q. Xia and X.-Q. Gong, ACS Catal., 2017, 7, 333–337 CrossRef CAS.
- M. Feng, Z. Chen, H. Sun, S. Yao, Z. Liu, M. Lu, F. Li, H. Wang and L. Liu, J. Energy Chem., 2025, 107, 582–590 CrossRef CAS.
- M. Zhang, C.-B. Hong, Z. Ding, Y. Weng, H. Gong, H. Wang, Y. Zhang, Y. Liao and H. Liu, Chem. Eng. J., 2024, 502, 158100 CrossRef CAS.
- D. Goetz, M. Lucas and P. Claus, React. Chem. Eng., 2016, 1, 161–164 RSC.
- P. P. Upare, Y. Kim, K.-R. Oh, S. J. Han, S. K. Kim, D.-Y. Hong, M. Lee, P. Manjunathan, D. W. Hwang and Y. K. Hwang, ACS Sustain. Chem. Eng., 2021, 9, 17242–17253 CrossRef CAS.
- A. Yamaguchi, Y. Murakami, T. Imura and K. Wakita, Chemistryopen, 2021, 10, 731–736 CrossRef CAS PubMed.
- L. Bruna, M. Cardona-Farreny, V. Colliere, K. Philippot and M. R. Axet, Nanomaterials, 2022, 12, 328 CrossRef CAS PubMed.
- N. S. Date, R. C. Chikate, H.-S. Roh and C. V. Rode, Catal. Today, 2018, 309, 195–201 CrossRef CAS.
- T. Tong, Q. Xia, X. Liu and Y. Wang, Catal. Commun., 2017, 101, 129–133 CrossRef CAS.
- V. Hiremath, B. T. Shiferraw and J. G. Seo, J. CO2 Util., 2020, 42, 101294 CrossRef CAS.
- X. Chen, W. Liu, J. Luo, H. Niu, R. Li and C. Liang, Ind. Eng. Chem. Res., 2022, 61, 12953–12965 CrossRef CAS.
- Z. Tong, X. Li, J. Dong, R. Gao, Q. Deng, J. Wang, Z. Zeng, J.-J. Zou and S. Deng, ACS Catal., 2021, 11, 6406–6415 CrossRef CAS.
- Y. Zhu, W. Zhao, J. Zhang, Z. An, X. Ma, Z. Zhang, Y. Jiang, L. Zheng, X. Shu, H. Song, X. Xiang and J. He, ACS Catal., 2020, 10, 8032–8041 CrossRef CAS.
- P. P. Upare, Y. Kim, K.-R. Oh, S. J. Han, S. K. Kim, D.-Y. Hong, M. Lee, P. Manjunathan, D. W. Hwang and Y. K. Hwang, ACS Sustain. Chem. Eng., 2021, 9, 17242–17253 CrossRef CAS.
- S. L. Scott, ACS Catal., 2018, 8, 8597–8599 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |







