Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Regioselective green synthesis of some novel indole-substituted 1H-benzo[f]chromenes via one-pot three-component reactions in water–ethanol media
Amirhossein Khanmohammadia,
Abolfazl Olyaei *a and
Mahdieh Sadeghpourb
*a and
Mahdieh Sadeghpourb
aDepartment of Chemistry, Faculty of Science, Imam Khomeini International University, Qazvin, Iran. E-mail: Olyaei_a@sci.ikiu.ac.ir
bDepartment of Chemistry, Qa.C, Islamic Azad University, Qazvin, Iran
First published on 2nd April 2026
Abstract
A regioselective, efficient and environmentally friendly method was developed for the synthesis of novel indole-substituted 1H-benzo[f]chromene derivatives through one-pot multicomponent condensation reaction of 3-cyanoacetyl indole, 2,7-dihydroxynaphthalene, and various aryl, heteroaryl and aliphatic aldehydes in EtOH/H2O under reflux conditions. The domino reaction proceeded via a piperidine-catalyzed Knoevenagel condensation between 3-cyanoacetyl indole and an aldehyde, followed by a Michael addition of 2,7-dihydroxynaphthalene and subsequent intramolecular heteroannulation. The attractive features of this method are operational simplicity, regioselectivity, green process, broad substrate scope, metal-free, shorter reaction time, easy workup, good to excellent yields and easy purification of products without utilization of any chromatography. The structures of the compounds were confirmed by FT-IR, 1H-NMR, and 13C-NMR spectroscopy and mass spectrometry.
Introduction
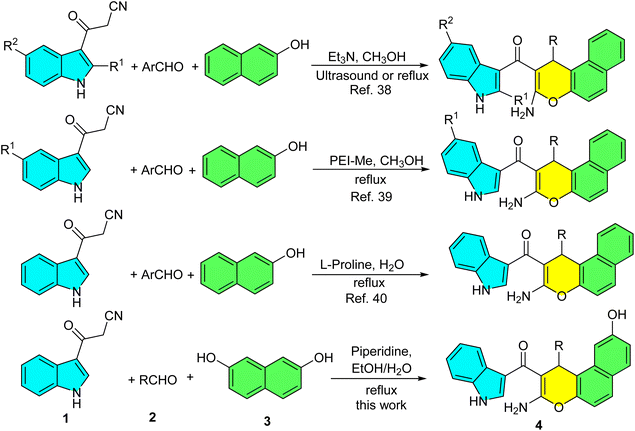
Chromene, benzochromene and their derivatives have been considered as an important class of oxygen-containing heterocycles. There has been increasing interest in the study of chromenes and benzochromenes due to their biological and pharmacological activities which include anti-cancer,1–4 anti-microbial,5–7 anti-influenza virus,8 anti-inflammatory effects,9 antiproliferative,10 antitubercular,11 antioxidant,12 anti-leishmanial,13 vascular disrupting14 and oestrogenic, anticoagulant and antispasmolytic15 activities. Among several synthesized chromene compounds, benzo[f]chromenes represent an important category of classical molecules and photochromic compounds.16 There has been considerable interest in chromenes and their benzo-derivatives because of their value for a variety of industrial, biological and chemical uses.17–19 Furthermore, 1H-benzo[f]chromene scaffolds have emerged as promising lead candidates for anticancer drug development due to their ability to target critical signaling pathways involved in cancer cell proliferation. These compounds have demonstrated diverse and potent anticancer mechanisms, including acting as c-Src kinase inhibitors and proapoptotic agents.20 They have also shown broad cytotoxic and apoptotic effects across a range of human cancer cell lines.21 Furthermore, others have been found to induce cell cycle arrest and apoptosis through the dual inhibition of topoisomerases and tubulin.22,23 Additionally, they have been utilized as powerful molecules that have apoptotic impacts with DNA binding attributes via an assortment of cell types.24Various synthetic routes have been developed for the construction of benzo[f]chromene derivatives. A prominent and widely used approach employs 2-naphthols as starting materials, which undergo a reaction with aldehydes and active methylene compounds under diverse catalytic or solvent conditions.25 Recent synthetic approaches for benzo[f]chromene derivatives has been reported. These include multi-component reactions such as: the condensation of 2-naphthol with α,β-unsaturated aldehydes and chiral 1-phenylethylamine;26 the reaction of 2-hydroxy-1-naphthaldehyde with indole derivatives and malononitrile catalyzed by Baker's yeast; 27 and the coupling of 2-naphthol with triphenylphosphine and an acetylenic ester in the presence of β-cyclodextrin.28 Additionally, methods using AuCl3/3AgOTf to catalyze the reaction of 2-naphthol with ketones have been developed.29 Additional synthetic strategies include the reaction of 2-naphthol with acetophenone derivatives and triethyl orthobenzoate using a bis[7-tert-butyl-2-anilinotropone]Ti complex;30 the ethylene diamine diacetate-catalyzed condensation of 2-naphthol with 3-methylbut-2-enal;31 and the CuCl2-mediated reaction of β-oxodithioesters and an S,S-diacetal with 2-hydroxy-1-naphthaldehyde.32 Other notable methods involve microwave-assisted reactions of 7-methoxy-2-naphthol with aromatic aldehydes and 2-cyanoethanethioamide;33 the reaction of 2-naphthol with aromatic aldehydes and 3,3,3-trifluoro-1-phenylpropan-1-one in 1,4-disulfo-1,4-diazoniabicyclo[2.2.2]octane chloride (DSDABCO),34 the use of In(OTf)3 to catalyze the coupling of β-naphthols with enals;35 the Kit-6-NH2@Schiff base complex promoted reaction of 2-naphthol with aldehyde and 4,4,4-trifluoro1-phenyl-1,3-butanedione,36 the NiFe2O4@Silicapropyl magnetic nanoparticles-catalyzed condensation of aromatic aldeydes with naphthalen-2-ol, and 4,4,4-trifluoro-1-phenylbutane1,3-dione.37 Moreover, indole-substituted chromene derivatives were obtained from the reaction of 2-naphthol with 3-cyanoacetylindoles and aryl aldehydes. This transformation employed Et3N38 and PEI-Me39 as catalysts in methanol, as well as L-proline in aqueous medium (Scheme 1).40 These compounds exhibit antibacterial, anti-inflammatory, and analgesic activities. Notably, they act as highly specific NorA efflux pump inhibitors, helping to mitigate drug-resistant strains of S. aureus. As shown in Scheme 1, these reactions were carried out either in methanol (a toxic solvent) with an expensive catalyst, or in water under reflux conditions with prolonged reaction times. Based on the established biological significance of chromene scaffolds and in continuation of our previous study on 3-cyanoacetyl indoles41 and multi-component synthetic methodologies,42 in the present work, we report a facial one-pot, three-component domino reaction of 3-cyanoacetyl indole, 2,7-dihydroxynaphthalene, various aryl, heteroaryl and aliphatic aldehydes using piperidine as an inexpensive catalyst in EtOH/H2O under heating conditions to afford the corresponding novel indole-substituted 1H-benzo[f]chromene derivatives in good to excellent yields. Furthermore, based on the biological properties of these compounds, the newly synthesized derivatives may also exhibit promising novel biological activities.
Results and discussion
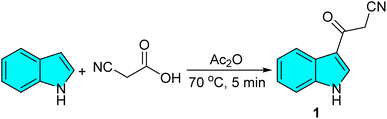
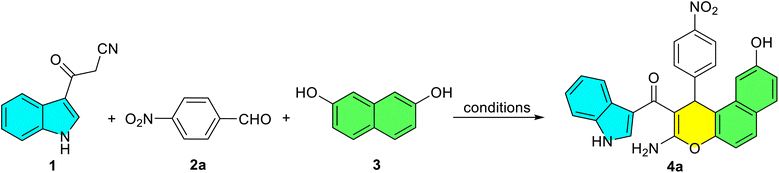
To initiate our study, 3-cyanoacetyl indole (1) was achieved via the reaction of indole and cyanoacetic acid in Ac2O at 70 °C for 5 min (Scheme 1). The identity of compound 1 was confirmed by comparison of its physical and spectral data with those reported in reference (Scheme 2).43 Then, the reaction of 3-cyanoacetyl indole (1) (1.0 mmol), 4-nitrobenzaldehyde 2a (1.0 mmol), and 2,7-dihydroxynaphthalene 3 (1.0 mmol) was chosen as a model (Table 1). First, carrying out this model reaction in the absence of a catalyst in ethanol at room temperature for 24 h yielded no product (Table 1, entry 1).| Entry | Solvent | Temperature (oC) | Catalyst loading (mol%) | Time (h) | Yieldb (%) |
|---|---|---|---|---|---|
| a All the reactions were performed with 1 (1 mmol), 2a (1 mmol), 3 (1 mmol), and solvent (10 mL).b Isolated yields. | |||||
| 1 | EtOH | r.t | — | 24 | — |
| 2 | EtOH | Reflux | — | 8 | 32 |
| 3 | EtOH | Reflux | PTSA (10%) | 8 | Trace |
| 4 | EtOH | Reflux | Et3N (10%) | 4 | 52 |
| 5 | EtOH | Reflux | Piperidine (10%) | 3 | 58 |
| 6 | MeOH | Reflux | Piperidine (10%) | 3 | 56 |
| 7 | HOAc | Reflux | — | 8 | Trace |
| 8 | CH3CN | Reflux | Piperidine (10%) | 6 | 51 |
| 9 | CH2Cl2 | Reflux | Piperidine (10%) | 8 | 43 |
| 10 | CHCl3 | Reflux | Piperidine (10%) | 8 | 47 |
| 11 | Toluene | Reflux | Piperidine (10%) | 1.5 | 50 |
| 12 | H2O | Reflux | Piperidine (10%) | 8 | 31 |
| 13 | EtOH + H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Reflux | Piperidine (10%) | 1.5 | 65 |
| 14 | EtOH + H2O (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Reflux | Piperidine (10%) | 1.5 | 52 |
| 15 | EtOH + H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
Reflux | Piperidine (10%) | 1.5 | 45 |
| 16 | EtOH + H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Reflux | Piperidine (5%) | 2 | 55 |
| 17 | EtOH + H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Reflux | Piperidine (15%) | 1 | 79 |
| 18 | EtOH + H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
Reflux | Piperidine (20%) | 1 | 80 |
| 19 | — | 80 | Piperidine (15%) | 1 | 52 |
| 20 | — | 100 | Piperidine (15%) | 1 | 54 |
When the reaction was stirred for 8 h in ethanol under reflux conditions without using any catalyst, the expected (3-amino-9-hydroxy-1-(4-nitrophenyl)-1H-benzo[f]chromen-2-yl)(1H-indol-3-yl)methanone (4a) was obtained in only 32% yield (Table 1, entry 2). We then investigated the reaction using p-TSA (10 mol%) in ethanol under reflux. Only trace amount of product 4a was formed (Table 1, entry 3). Next, we investigated base catalysts to optimize the yield and reaction time. The model reaction with Et3N (10 mol%) afforded product 4a in 52% yield, while piperidine (10 mol%) gave an improved yield of 58% in refluxing EtOH (Table 1, entries 4–5). Piperidine as a base catalyst seemed to be more efficient for the above transformation. Using piperidine (10 mol%) as the catalyst under reflux, the model reaction was evaluated in a series of protic and aprotic solvents (Table 1, entries 6–15). The protic solvents tested were MeOH, H2O HOAc, and EtOH/H2O mixtures (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 2
1, 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). The aprotic solvents were CH3CN, CH2Cl2, CHCl3, and toluene. The results revealed that using EtOH/H2O (1
2). The aprotic solvents were CH3CN, CH2Cl2, CHCl3, and toluene. The results revealed that using EtOH/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) provided the desired product in 65% yield after 1.5 h (Table 1, entry 13). To determine the optimal catalyst loading, reactions were performed in refluxing EtOH/H2O (1
1) provided the desired product in 65% yield after 1.5 h (Table 1, entry 13). To determine the optimal catalyst loading, reactions were performed in refluxing EtOH/H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) using 5, 15, and 20 mol% piperidine. A loading of 5 mol% afforded the product in 55% yield after 2 h. Surprisingly, increasing the catalyst to 15 mol% significantly improved the yield to 79% within 60 minutes (Table 1, entries 16–17). No significant difference in yield was observed when the catalyst loading was increased from 15 to 20 mol% (Table 1, entry 18). The reaction was also tested under solvent-free conditions at 80 °C and 100 °C, affording 54 and 52% yields after 1 h, respectively (Table 1, entries 19–20). On the basis of all of these experiments, the optimum reaction conditions were identified as 1
1) using 5, 15, and 20 mol% piperidine. A loading of 5 mol% afforded the product in 55% yield after 2 h. Surprisingly, increasing the catalyst to 15 mol% significantly improved the yield to 79% within 60 minutes (Table 1, entries 16–17). No significant difference in yield was observed when the catalyst loading was increased from 15 to 20 mol% (Table 1, entry 18). The reaction was also tested under solvent-free conditions at 80 °C and 100 °C, affording 54 and 52% yields after 1 h, respectively (Table 1, entries 19–20). On the basis of all of these experiments, the optimum reaction conditions were identified as 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) ethanol/water under reflux conditions catalyzed by piperidine (15 mol%).
1 (v/v) ethanol/water under reflux conditions catalyzed by piperidine (15 mol%).
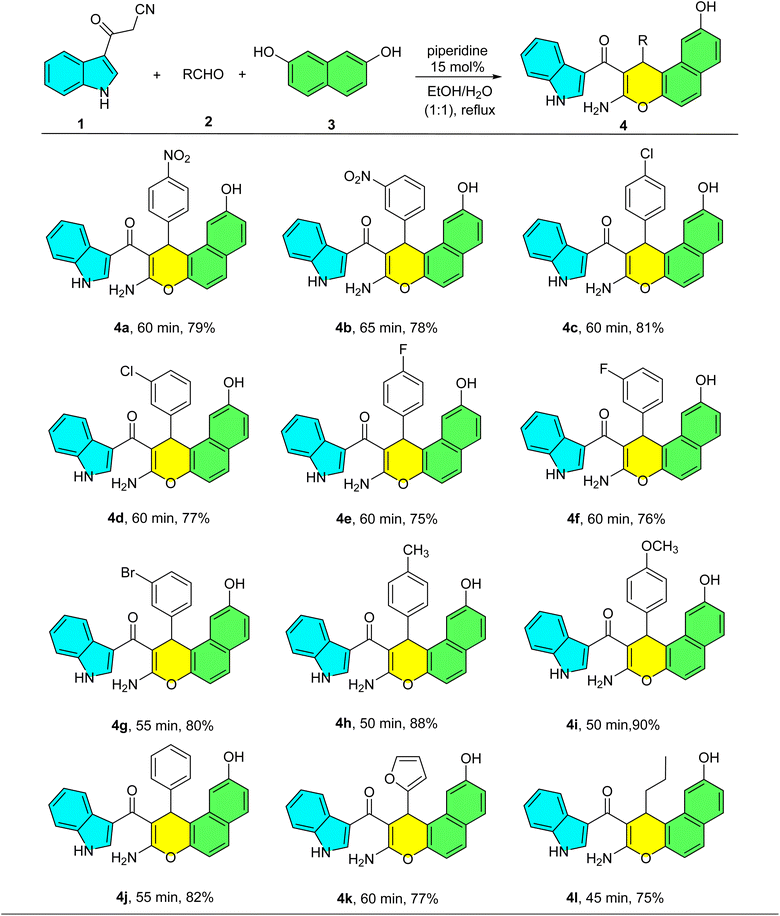
With the optimal conditions established, we evaluated the substrate scope using 12 diverse aldehydes 2. These included substituted aromatic aldehydes bearing electron-withdrawing groups (EWGs) such as 4-nitrobenzaldehyde, 3-nitrobenzaldehyde, 4-chlorobenzaldehyde, 3-chlorobenzaldehyde, 4-flourobenzaldehyde, 3-flourobenzaldehyde, 3-bromobenzaldehyde and electron-donating groups (EDGs) such as 4-methylbenzaldehyde, 4-methoxybenzaldehyde, a heteroaromatic aldehyde (furfural), and an aliphatic aldehyde (butyraldehyde). The results are summarized in Table 2. As shown in Table 2, under the optimized conditions, the reaction proceeded successfully with a range of aldehydes. Substrates bearing electron-withdrawing groups (EWGs) furnished products 4a–g in good yields (75–81%) over 55–65 min. In contrast, aldehydes with electron-donating groups (EDGs) reacted more rapidly, providing products 4h–i in excellent yields (88–90%) within a shorter time (50 min). Similarly, furfural, benzaldehyde, and butyraldehyde afforded products 4j–l in high yields (75–82%) with reaction times of 45–60 min. These results indicate that the reaction rate and yield are favorably influenced by EDGs. Notably, all products could be purified by simple trituration in boiling CHCl3, eliminating the need for chromatography.
a Reaction conditions: 3-cyanoacetyl indole (1 mmol), aldehyde (1 mmol), 2,7-dihydroxynaphthalene (1 mmol) and piperidine (15 mol%) in EtOH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1 H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) under reflux conditions. 1) under reflux conditions. |
|---|
 |
To the best of our knowledge, there is no report for the synthesis of indole-substituted 1H-benzo[f]chromene derivatives through the MCRs of 3-cyanoacetyl indole, 2,7-dihydroxynaphthalene, various aryl, heteroaryl and aliphatic aldehydes. Therefore, all the synthesized compounds were unknown, and were characterized by Fourier Transform Infrared (FT-IR), 1H and 13C-NMR, mass spectrometry analysis and melting points. The presence of the expected functional groups was confirmed by infrared spectroscopy. In the IR spectrum of compound 4a, stretching vibrations were observed for N–H and O–H groups (3501, 3451, 3128 cm−1), a carbonyl group (1637 cm−1), and a nitro group (1522 and 1340 cm−1). In the 1H NMR spectrum of 4a, the methine proton appeared as a singlet at δ 6.00 ppm. Aromatic protons resonated as a multiplet in the range δ 6.85–7.92 ppm. Three exchangeable proton singlets were observed at δ 8.70 (NH2), 9.79 (OH), and 11.66 ppm (NH). Upon addition of D2O to the NMR sample, the signals corresponding to the exchangeable protons were eliminated due to deuterium exchange. The 13C-NMR spectrum of 4a displayed signals corresponding to the methine carbon (δ 38.3 ppm), the carbonyl group (δ 188.0 ppm), and aromatic carbons (δ 88.8–162.0 ppm).
Mass spectral data for compounds 4a–k supported their structures, showing low-intensity molecular ions consistent with facile fragmentation. For compound 4a, the mass spectrum showed a molecular ion peak at m/z 477. This fragmented to yield key ions at m/z 355, 184, 144, 131, 116, and 103, with the base peak appearing at m/z 160.
Based on comprehensive experimental evidence, we propose a plausible reaction mechanism for the formation of glycine derivatives 4, as illustrated in Scheme 3. In the first step, a piperidine-mediated Knoevenagel condensation of 3-cyanoacetyl indole (1) and iminium ion intermediate 5 formed by condensation of piperidine with the aldehyde 2 gives Knoevenagel adduct 7. Deprotonation of 2,7-dihydroxynaphthalene (3) by piperidine forms the nucleophile 8, which adds to 7 in a Michael reaction to yield adduct 9. This is followed by an intramolecular O-cyclization, where the enolate moiety of 9 attacks the electrophilic nitrile carbon, forming intermediate 10. Tautomerization of 10 then provides the final product 4. The proposed mechanism was confirmed by studying the order of addition of reagents. To support the proposed mechanism, we adopted a two-component experimental approach. When 2,7-dihydroxynaphthalene (3) was reacted with aldehyde 2 under optimized conditions, no product was detected even after 2 hours. However, conducting the reaction between 1 and 2 under similar conditions led to the formation of intermediate 7 within 30 minutes. Subsequent addition of 3 to this mixture yielded the final product. These results clearly indicate that the reaction proceeds via in situ formation of the adduct 7.
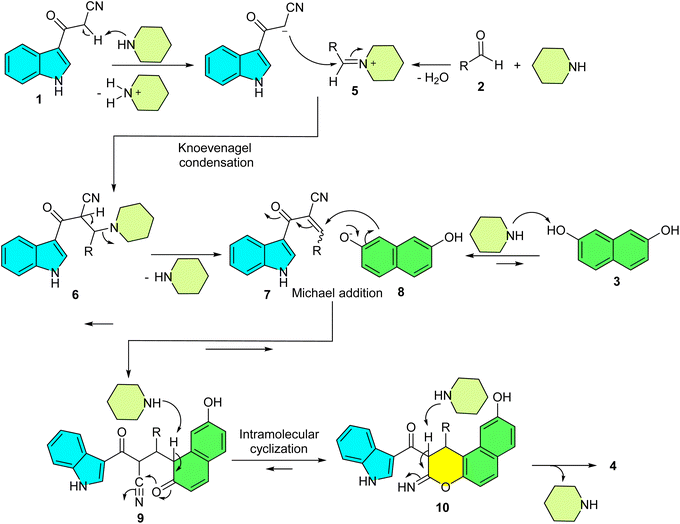 | ||
| Scheme 3 Proposed mechanism for the synthesis of indole-substituted 1H-benzo[f]chromene derivatives 4. | ||
Conclusions
In summary, we have described piperidine-catalyzed heteroannulation as an alternative method for the synthesis of a series of indole-substituted 1H-benzo[f]chromene derivatives 4 in a regiochemical manner by the reaction of 3-cyanoacetyl indole, aldehyde, and 2,7-dihydroxynaphthalene under conventional condition. This reaction involving Knoevenagel condensation, Michael addition and ring closure. The proposed protocol offers several advantages, including good to excellent yields, a broad substrate scope, the one-step conversion of simple, readily available starting materials into an interesting class of fused heterocyclic scaffolds of benzochromene derivatives. The procedure also features straightforward workup and product purification, which avoids the need for chromatographic methods. Studies to extend the reaction scope and explore further synthetic applications of this methodology are currently underway in our laboratory.Experimental methods and materials
General information
All commercially available chemicals and reagents were used without further purification. Melting points were determined with Gallenkamp MFB.595.010M apparatus. FT-IR spectra were recorded on a Bruker Tensor 27 spectrophotometer. The 1H-and 13C-NMR spectra were recorded in DMSO-d6 on Bruker DRX-300 Avance spectrometers. Chemical shifts (δ) are reported in parts per million and are referenced to the NMR solvent. Mass spectra of the products were obtained with a HP (Agilent technologies) 5973 Mass Selective Detector.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) H2O (1
H2O (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) (10 ml) was stirred at reflux for an appropriate time (Table 2), and the progress of the reaction was monitored by TLC (n-hexane/ethyl acetate: 1
1) (10 ml) was stirred at reflux for an appropriate time (Table 2), and the progress of the reaction was monitored by TLC (n-hexane/ethyl acetate: 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Upon completion of the reaction, the mixture was cooled to room temperature. The solid product was collected by filtration, washed with cold H2O/EtOH, and dried. For further purification, each precipitate was suspended in boiling CHCl3 and stirred for 5 minutes. After cooling, the solids were collected by filtration, washed with cold CHCl3, and dried to obtain pure products 4a–l in good to excellent yields. All compounds were fully characterized by FT-IR, 1H and 13C-NMR spectroscopy, and mass spectrometry.
1). Upon completion of the reaction, the mixture was cooled to room temperature. The solid product was collected by filtration, washed with cold H2O/EtOH, and dried. For further purification, each precipitate was suspended in boiling CHCl3 and stirred for 5 minutes. After cooling, the solids were collected by filtration, washed with cold CHCl3, and dried to obtain pure products 4a–l in good to excellent yields. All compounds were fully characterized by FT-IR, 1H and 13C-NMR spectroscopy, and mass spectrometry.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 477 [M]+, 355, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63, 51.
O); MS (EI, 70 ev) m/z (%): 477 [M]+, 355, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63, 51.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 477 [M]+, 184, 160 (100), 144, 131, 117, 116, 102, 103, 89, 77, 63, 51.
O); MS (EI, 70 ev) m/z (%): 477 [M]+, 184, 160 (100), 144, 131, 117, 116, 102, 103, 89, 77, 63, 51.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 468 [M + 2]+, 466 [M]+, 355, 308, 306, 279, 242, 184, 160, 144 (100), 131, 116, 89, 76, 63.
O); MS (EI, 70 ev) m/z (%): 468 [M + 2]+, 466 [M]+, 355, 308, 306, 279, 242, 184, 160, 144 (100), 131, 116, 89, 76, 63.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 468 [M + 2]+, 466 [M]+, 308, 306, 279, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63.
O); MS (EI, 70 ev) m/z (%): 468 [M + 2]+, 466 [M]+, 308, 306, 279, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 450 [M]+, 307, 290, 263, 224, 184, 160, 144 (100), 131, 116, 89, 63.
O); MS (EI, 70 ev) m/z (%): 450 [M]+, 307, 290, 263, 224, 184, 160, 144 (100), 131, 116, 89, 63.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 450 [M]+, 307, 290, 263, 224, 184, 160, 144 (100), 131, 116, 103, 89, 76, 63.
O); MS (EI, 70 ev) m/z (%): 450 [M]+, 307, 290, 263, 224, 184, 160, 144 (100), 131, 116, 103, 89, 76, 63.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 512 [M + 2]+, 510 [M]+, 352, 350, 325, 323, 271, 242, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63.
O); MS (EI, 70 ev) m/z (%): 512 [M + 2]+, 510 [M]+, 352, 350, 325, 323, 271, 242, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 446 [M]+, 286, 259, 242, 184, 160 (100), 144, 131, 116, 103, 89, 76, 63.
O); MS (EI, 70 ev) m/z (%): 446 [M]+, 286, 259, 242, 184, 160 (100), 144, 131, 116, 103, 89, 76, 63.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 462 [M]+, 302, 259, 242, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63.
O); MS (EI, 70 ev) m/z (%): 462 [M]+, 302, 259, 242, 184, 160 (100), 144, 131, 116, 103, 89, 77, 63.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 432 [M]+, 355, 306, 238, 209, 184, 160, 144 (100), 131, 117, 116, 103, 91, 89, 77, 69, 55.
O); MS (EI, 70 ev) m/z (%): 432 [M]+, 355, 306, 238, 209, 184, 160, 144 (100), 131, 117, 116, 103, 91, 89, 77, 69, 55.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 422 [M]+, 262, 238, 218, 197, 184, 160, 144 (100), 131, 116, 103, 89, 77, 63, 51.
O); MS (EI, 70 ev) m/z (%): 422 [M]+, 262, 238, 218, 197, 184, 160, 144 (100), 131, 116, 103, 89, 77, 63, 51.![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O); MS (EI, 70 ev) m/z (%): 398 [M]+, 355, 337, 382, 214, 184, 160, 144 (100), 131, 116, 103, 89, 77, 63.
O); MS (EI, 70 ev) m/z (%): 398 [M]+, 355, 337, 382, 214, 184, 160, 144 (100), 131, 116, 103, 89, 77, 63.Author contributions
Amirhossein Khanmohammadi conducted the synthesis and characterization; Abolfazl Olyaei supervision, project administration, scientific advice, writing – review & editing; Mahdieh Sadeghpour writing – original draft and editing. All authors reviewed and approved the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
Experimental data and characterization details for all compounds are provided in the article. 1H-NMR and 13C-NMR spectra for compounds 4a–l are available in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra02182d.Acknowledgements
All the authors are thankful to the Research Council of Imam Khomeini International University for financial support.References
- A. M. Fouda, R. M. Okasha, F. F. Alblewi, A. Mora, T. H. Afifi and A. M. El-Agrody, A proficient microwave synthesis with structure elucidation and the exploitation of the biological behavior of the newly halogenated 3-amino-1H-benzo[f]chromene molecules, targeting dual inhibition of topoisomerase II and microtubules, Bioorg. Chem., 2020, 95, 103549 CrossRef CAS PubMed.
- A. M. Fouda, M. A. Assiri, A. Mora, T. E. Ali, T. H. Afifi and A. M. El-Agrody, Microwave synthesis of novel halogenated β-enaminonitriles linked 9-bromo-1H-benzo[f]chromene moieties: induces cell cycle arrest and apoptosis in human cancer cells via dual inhibition of topoisomerase I and II, Bioorg. Chem., 2019, 93, 103289 CrossRef PubMed.
- F. F. Alblewi, R. M. Okasha, Z. M. Hritani, H. M. Mohamed, M. A. A. El-Nassag, A. H. Halawa, A. Mora, A. M. Fouda, M. A. Assirid, A. M. Al-Dies, T. H. Afifi and A. M. El-Agrody, Antiproliferative effect, cell cycle arrest and apoptosis generation of novel synthesized anticancer heterocyclic derivatives based 4H-benzo[h]chromene, Bioorg. Chem., 2019, 87, 560–571 CrossRef CAS PubMed.
- D. Hu, N. Miyagi, Y. Arai, H. Oguri, T. Miura, T. Nishinaka, T. Terada, H. Gouda, O. El-Kabbani, S. Xia, N. Toyooka, A. Hara, T. Matsunaga, A. Ikari and S. Endo, Synthesis of 8-hydroxy-2-iminochromene derivatives as selective and potent inhibitors of human carbonyl reductase 1, Org. Biomol. Chem., 2015, 13, 7487–7499 RSC.
- D. Ashok, K. R. Srinivas, G. Velagapuri and H. Rao, Synthesis of pyrazolylfuro[2,3-f]-chromenes and evaluation of their antimicrobial activity, Chem. Heterocycl. Compd., 2016, 52, 928–933 CrossRef CAS.
- H. K. Abd El-Mawgoud, H. A. M. Radwan, F. El-Mariah and A. M. El-Agrody, Synthesis characterization, biological activity of novel 1H-benzo[f]chromene and 12H-benzo[f]chromeno-[2,3-d]pyrimidine derivatives, Lett. Drug Des. Discov., 2018, 15, 857–865 CrossRef CAS.
- N. D. Vala, H. H. Jardosh and M. P. Patel, 5-PS-TBD triggered general protocol for the synthesis of 4H-chromenes, pyrano[4,3-b]pyran and pyrano[3,2-c]chromene derivatives of 1H-pyrazole and their biological activities, Chin. Chem. Lett., 2016, 27, 168–172 CrossRef CAS.
- A. M. Abdella, Y. Moatasim, M. A. Ali, A. H. M. Elwahy and I. A. Abdelhamid, Synthesis and anti-influenza virus activity of Novel bis(4H-chromene-3-carbonitrile) derivatives, J. Heterocycl. Chem., 2017, 54, 1854–1862 CrossRef CAS.
- I. J. Elenkov, B. Hrvacic, S. Markovic, M. Mesic, A. C. Klonkay, L. Lerman, A. F. Sucic, I. Vujasinovic, B. Bosnjak, K. Brajsa, D. Ziher, N. K. Hulita and I. Malnard, Synthesis and anti-inflammatory activity of novel furochromenes, Croat. Chem. Acta, 2013, 86, 253–264 CrossRef CAS.
- A. Parthiban, M. Kumaravel, J. Muthukumaran, R. Rukkumani, R. Krishna, H. Surya and P. Rao, Synthesis, in vitro and in silico anti-proliferative activity of 4-aryl-4H-chromene derivatives, Med. Chem. Res., 2016, 25, 1308–1315 CrossRef CAS.
- A. Termentzi, I. Khouri, T. Gaslonde, S. Prado, B. Saint-Joanis, F. Bardou, E. Amanatiadou, I. Vizirianakis, J. Kordulkova, M. Jackson, R. Brosch, Y. Janin, M. Daff, F. Tillequin and S. Michel, Synthesis, biological activity, and evaluation of the mode of action of novel antitubercular benzofurobenzopyrans substituted on A ring, 2010, Eur. J. Med. Chem., 2010, 45, 5833–5847 CrossRef CAS PubMed.
- W. Gregor, G. Grabner, C. Adelwöhrer, T. Rosenau and L. Gille, Antioxidant properties of natural and synthetic chromanol derivatives: study by fast kinetics and electron spin resonance spectroscopy, J. Org. Chem., 2005, 70, 3472–3483 CrossRef CAS PubMed.
- A. Foroumadi, S. Emami, M. Sorkhi, M. Nakhjiri, Z. Nazarian, S. Heydar, S. Ardestani, F. Poorrajab and A. Shafiee, Chromene-based synthetic chalcones as potent anti-leishmanial agents: synthesis and biological activity, Chem. Biol. Drug Des., 2010, 75, 590–596 CrossRef CAS PubMed.
- S. Kasibhatla, H. Gourdeau, K. Meerovitch, J. Drewe, S. Reddy, L. Qiu, H. Zhang, F. Bergeron, D. Bouffard, Q. Yang, J. Herich, S. Lamothe, S. X. Cai and B. Tseng, Discovery and mechanism of action of a novel series of apoptosis inducers with potential vascular targeting activity, Mol. Cancer Ther., 2004, 3, 1365–1374 CrossRef CAS PubMed.
- N. Jain, R. M. Kanojia, J. Xu, G. Jian-Zhong, E. Pacia, M.-T. Lai, F. Du, A. Musto, G. Allan, D. Hahn, S. Lundeen and Z. Sui, Novel chromene-derived selective estrogen receptor modulators useful for alleviating hot flushes and vaginal dryness, J. Med. Chem., 2006, 49, 3056–3059 CrossRef CAS PubMed.
- P. J. Coelho, L. M. Carvalho, S. Abrantes, M. M. Oliveira, M. F. Oliveira-Campos, A. Samat and R. Guglielmetti, Synthesis and spectrokinetic studies of spiro[thioxanthene-naphthopyrans], Tetrahedron, 2002, 58, 9505–9511 CrossRef CAS.
- C. D. Gabbutt, B. M. Heron, A. C. Instone, D. A. Thomas, S. M. Partington, M. B. Hursthouse and T. Gelbrich, Observations on the Synthesis of Photochromic Naphthopyrans, Eur. J. Org Chem., 2003, 1220–1230 CrossRef CAS.
- M. Kidwai, S. Saxena, M. K. R. Khan and S. S. Thukral, Aqua mediated synthesis of substituted 2-amino-4H-chromenes and in vitro study as antibacterial agents, Bioorg. Med. Chem. Lett., 2005, 15, 4295–4298 CrossRef CAS PubMed.
- A. H. Bedair, H. A. Emam, N. A. El-Hady, K. A. R. Ahmed and A. M. El-Agrody, Synthesis and antimicrobial activities of novel naphtho[2,1-b]pyran, pyrano[2,3-d]pyrimidine and pyrano[3,2-e][1,2,4]triazolo[2,3-c]-pyrimidine derivatives, Farmaco, 2001, 56, 965–973 CrossRef CAS PubMed.
- H. E. A. Ahmed, M. A. A. El-Nassag, A. H. Hassan, R. M. Okasha, S. Ihmaid, A. M. Fouda, T. H. Afif, A. Aljuhani and A. M. El-Agrody, Introducing Novel Potent Anticancer Agents of 1H-Benzo[f]chromene Scaffolds, Targeting C-Src Kinase Enzyme with MDA-MB-231 Cell Line Anti-invasion Effect, J. Enzyme Inhib. Med. Chem., 2018, 33, 1074–1088 CrossRef CAS PubMed.
- T. H. Afifi, R. M. Okasha, H. E. A. Ahmed, J. Ilas, T. Saleh and A. S. Abd-El-Aziz, Structure-activity Relationships and Molecular Docking Studies of Chromene and Chromene Based Azo Chromophores: A Novel Series of Potent Antimicrobial and Anticancer Agents, EXCLI J., 2017, 16, 868–902 Search PubMed.
- A. M. Fouda, M. A. Assiri, A. Mora, T. E. Ali, T. H. Afifi and A. M. El-Agrody, Microwave Synthesis of Novel Halogenated β-enaminonitriles Linked 9-Bromo-1h-Benzo[f]chromene Moieties: Induces Cell Cycle Arrest and Apoptosis in Human Cancer Cells via Dual Inhibition of Topoisomerase I and II, Bioorg. Chem., 2019, 93, 103289–103300 CrossRef PubMed.
- A. M. Fouda, R. M. Okasha, F. F. Alblewi, A. Mora, T. H. Afifi and A. M. El-Agrody, A Proficient Microwave Synthesis with Structure Elucidation and the Exploitation of the Biological Behavior of the Newly Halogenated 3-Amino-1h-Benzo[f]chromene Molecules, Targeting Dual Inhibition of Topoisomerase II and Microtubules, Bioorg. Chem., 2020, 95, 103549–103561 CrossRef CAS PubMed.
- Y. Dgachi, O. M. Bautista-Aguilera, M. Benchekroun, H. Martin, A. Bonet, D. Knez, J. Godyn, B. Malawska, S. Gobec, M. Chioua, J. Janockova, O. Soukup, F. Chabchoub, J. Marco-Contelles and L. Ismaili, Synthesis and biological evaluation of benzochromenopyrimidinones as cholinesterase inhibitors and potent antioxidant, non-hepatotoxic agents for Alzheimer's disease, Molecules, 2016, 21, 634 CrossRef PubMed.
- (a) R. Pratap and V. Ji Ram, Natural and Synthetic Chromenes, Fused Chromenes, and Versatility of Dihydrobenzo[h]chromenes in Organic Synthesis, Chem. Rev., 2014, 114, 10476–10526 CrossRef CAS PubMed; (b) M. Mamaghania, R. Hossein Nia, F. Tavakoli and P. Jahanshahi, Recent Advances in the MCRs Synthesis of Chromenes: A Review, Curr. Org. Chem., 2018, 22, 1–66 Search PubMed; (c) V. Patel, T. Bambharoliya, D. Shah, D. Patel, M. Patel, U. Shah, M. Patel, S. Patel, N. Solanki, A. Mahavar, A. Nagani, H. Patel, M. Rathod, B. Bhimani, V. Bhavsar, S. Padhiyar, S. Koradia, C. Chandarana, B. Patel, R. C. Dabhi and A. Patel, Eco-friendly Approaches to Chromene Derivatives: A Comprehensive Review of Green Synthesis Strategies, Curr. Top. Med. Chem., 2025, 25, 437–460 CrossRef CAS PubMed; (d) M. Chadha, A. Garg, A. Bhalla and S. Berry, Green methods mediated synthesis of chromene derivatives using magnetic nanoparticles as heterogeneous and reusable nanocatalyst: A review, Tetrahedron, 2024, 150, 133741 CrossRef CAS; (e) I. Yavari, M. Anary-Abbasinejad and Z. Hossaini, Reaction between naphthols and dimethyl acetylenedicarboxylate in the presence of phosphites. Synthesis of stable oxa-2λ5-phosphaphenanthrenes, and benzochromene derivatives, Org. Biomol. Chem., 2003, 1, 560–564 RSC.
- C. Cimarelli, D. Fratoni and G. Palmieri, Novel stereoselective synthesis of 2,3-dihydro-1H-benzo[f]chromen-3-amine derivatives through a one-pot three component reaction, Tetrahedron: Asymmetry, 2011, 22, 1542–1547 CrossRef CAS.
- N. G. Singh, R. Nongrum, C. Kathing, J. W. S. Rani and R. Nongkhlaw, Bakers' yeast: An environment benign catalyst for the one-pot synthesis of indolyl chromenes and bisindolyl alkanes, Green Chem. Lett. Rev., 2014, 7, 137–144 CrossRef.
- A. Ramazani, S. Ayoubi, Y. Ahmadi, H. Ahankar, H. Aghahosseini and S. W. Joo, Cyclodextrin nanoreactor” catalyzed synthesis of coumarin derivatives from in-situ generated stabilized phosphorus ylides in water, Phosphorus Sulfur Silicon Relat. Elem., 2015, 190, 2307–2314 CrossRef CAS.
- Y. Liu, J. Qian, Sh. Lou, J. Zhu and Zh. Xu, Gold (III)-catalyzed tandem reaction of ketones with phenols: Efficient and highly selective synthesis of functionalized 4H-chromenes, J. Org. Chem., 2010, 75, 1309–1312 CrossRef CAS PubMed.
- S. Damavandi, Heterogeneous titanium catalyst for the synthesis of novel 2-(4-aryl)-4-ethoxy-4-phenyl-4H-benzo[h] chromene derivatives, Synth. React. Inorg., Met.-Org., Nano-Met. Chem., 2012, 42, 1415–1417 CrossRef CAS.
- X. Wang, Y. Chen and Y. R. Lee, Concise Synthesis of (±)-Rhinacanthin A, Dehydro α-Lapachone, and β-Lapachone, and Pyranonaphthoquinone Derivatives, Bull. Korean Chem. Soc., 2011, 32, 153–156 CrossRef CAS.
- O. M. Singh, N. S. Devi, L. Ronibala, K. B. Lim, Y. J. Yoon and S.-G. Lee, Facile Synthesis of 3-Thioxo-3H-benzo[f]chromen-2-yl methanone and 3H-Benzo[f]chromene-3-one Under Solvent Free Condition, Bull. Korean Chem. Soc., 2011, 32, 175–178 CrossRef CAS.
- M. A. Fouda, A. Irfan, A. G. Al-Sehemi and A. M. El-Agrody, Synthesis, characterization, anti-proliferative activity and DFT study of 1H-benzo[f]chromene-2-carbothioamide derivatives, J. Mol. Struct., 2021, 1240, 130542 CrossRef.
- A. Masoumi Shahi, M. Nikpassand and L. Zare Fekri, An Efficient and Green Synthesis of New Benzo[f]chromenes Using 1,4-Disulfo-1,4-diazoniabicyclo[2.2.2]octane Chloride as a Novel Medium, Org. Prep. Proced. Int., 2019, 51, 521–529 CrossRef.
- R. Abdul Basit, S. Hassan and B. A. Bhat, Micelle-enabled synthesis of diverse 3-substituted-3H-benzo[f]chromene constructs in water, Tetrahedron Lett., 2026, 174, 155852 CrossRef.
- L. Zare Fekri, M. Nikpassand and S. Zeinali, Multicomponent Synthesis of Trifluoromethyl-1Hbenzo[f]chromenes Using Kit-6-NH2@Schiff Base Complex Nanoparticles, Org. Prep. Proced. Int., 2024, 56, 257–263 CrossRef CAS.
- L. Zare Fekri and M. Nikpassand, Synthesis of 3-Trifl uoromethyl-1H-benzo[f]chromenes Using Aminoglucose-Functionalized NiFe2O4@Silicapropyl as Green, Effi cient, and Magnetically Recoverable Heterogeneous Catalyst, Russ. J. Org. Chem., 2024, 60, 522–529 CrossRef.
- R. Hosseinnia, M. Mamaghani, K. Tabatabaeian, F. Shirini and M. Rassa, An expeditious regioselective synthesis of novel bioactive indole-substituted chromene derivatives via one-pot three-component reaction, Bioorg. Med. Chem. Lett., 2012, 22, 5956–5960 CrossRef CAS PubMed.
- A. Ganesan, L. R. Christena, H. M. V. Subbarao, U. Venkatasubramanian, R. Thiagarajan, V. Sivaramakrishnan, K. Kasilingam, N. Saisubramanian and S. S. Ganesan, Identification of benzochromene derivatives as a highly specific NorA efflux pump inhibitor to mitigate the drug resistant strains of S. aureus, RSC Adv., 2016, 6, 30258–30267 RSC.
- G. G. Reddy, Ch. Venkata Ramana Reddy and B. Srinivasa Reddy, Med. Chem., 2022, 18, 810–819 CrossRef CAS PubMed.
- A. Olyaei and M. Sadeghpour, Chemistry of 3-cyanoacetyl indoles: synthesis, reactions and applications: a recent update, RSC Adv., 2023, 13, 21710–21745 RSC.
- (a) F. Noruzian, A. Olyaei, R. Hajinasiri and M. Sadeghpour, Guanidine hydrochloride catalyzed efficient one-pot pseudo five-component synthesis of 4,4′-(arylmethylene)bis(1H-pyrazol-5-ols) in water, 2019, Synth. Commun., 2019, 49, 2717–2724 CrossRef CAS; (b) A. Olyaei, Z. Shafie and M. Sadeghpour, An efficient and one-pot green synthesis of novel 6-oxo-7-aryl-6, 7-dihydrochromeno pyrano [2, 3-b] pyridine derivatives, Tetrahedron Lett., 2018, 59, 3567–3570 CrossRef CAS; (c) R. Khoeiniha, A. Olyaei and M. Saraei, Catalyst-free synthesis of novel 4H-indeno[1,2-b]furan-4-ones and furo[2,3-d]pyrimidines in water, Synth. Commun., 2018, 48, 155–160 CrossRef CAS; (d) A. Olyaei, R. M. Ebrahimi, A. Adl and M. Sadeghpour, Green synthetic approach toward new chromeno[4,3-b]quinoline and chromeno[4,3-b]pyridine derivatives, Chem. Heterocycl. Compd., 2019, 55, 1104–1110 CrossRef CAS; (e) A. Olyaei, M. S. Shahsavari and M. Sadeghpour, Organocatalytic approach toward the green one-pot synthesis of novel benzo[f]chromenes and 12Hbenzo[5,6]chromeno[2,3-b]pyridines, Res. Chem. Intermed., 2018, 44, 943–956 CrossRef CAS; (f) S. B. Sajjadi, A. Olyaei and M. Shalbafan, Novel naphtho[2,3-b]furan-2,4,9(3H)-trione derivatives as potent ERα inhibitors: design, regioselective synthesis, HMBC-NMR characterization, in silico molecular Docking and ADME studies, BMC Chem., 2025, 19, 253 CrossRef CAS PubMed; (g) M. Kayyal, A. Olyaei, K. Pourshamsian and M. Sadeghpour, Acid-Mediated One-Pot Three-Component Tandem Cyclization: Access to Some New Series of Benzo [5, 6] Chromeno [2, 3-d] Pyrimidine Derivatives, ChemistrySelect, 2024, 9, e202304579 CrossRef CAS.
- J. Slatt, I. Romero and J. Bergman, Cyanoacetylation of Indoles, Pyrroles and Aromatic Amines with the Combination Cyanoacetic Acid and Acetic Anhydride, Synthesis, 2004, 2760–2765 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |