 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Organosilica nanoparticle-reinforced resin-based dental composites: synthesis, characterisation, and evaluation of physicochemical properties
Mohammed A. Al-Khafaji *a,
Ali O. Imarahb,
Athmar A. Kadhimc,
Mudher K. Mohammedd,
Hayder O. Hashime,
Judith Mihályfg and
Zoltán Varga
*a,
Ali O. Imarahb,
Athmar A. Kadhimc,
Mudher K. Mohammedd,
Hayder O. Hashime,
Judith Mihályfg and
Zoltán Varga fh
fh
aDepartment of Basic Science, College of Dentistry, University of Babylon, Hilla, Iraq. E-mail: phar.mohammed.a.karam@uobabylon.edu.iq; moh_chem_84@yahoo.com
bDepartment of Chemical Engineering, College of Engineering, University of Babylon, Hilla, Iraq
cCollege of Kufa, Al-Furat Al-Awsat Technical University, Najaf, Iraq
dDepartment of Pharmacy, Al-Manara College of Medical Science, Amarah, Iraq
eDepartment of Clinical Laboratory Sciences, College of Pharmacy, University of Babylon, Hilla, Iraq
fInstitute of Materials and Environmental Chemistry, HUN-REN Research Centre for Natural Sciences, Budapest, Hungary
gDepartment of Chemistry, Eszterházy Károly Catholic University, Eger, Hungary
hDepartment of Physical Chemistry and Materials Science, Faculty of Chemical Technology and Biotechnology, Budapest University of Technology and Economics, Budapest, Hungary
First published on 14th April 2026
Abstract
Dental nanocomposites have recently gained significant attention in the scientific community due to their ability to improve the aesthetic outcomes of dental restorations, thereby improving appearance. Despite extensive research efforts, developing dental nanocomposites that meet both mechanical and aesthetic standards remains a challenging issue. Although nanoparticles are effective at enhancing dental nanocomposite materials, they also have drawbacks. The higher surface-to-volume ratio of silica particles, combined with their hydrophilic nature, increases water absorption and can cause the resin to dry, leading to nano- or micro-leakage. The study investigates the effect of a hydrophobic organo-silica shell framework on nanocomposite performance and the mechanical properties of a dental filler-based nanocomposite. Inorganic SiO2 nanoparticles were coated with 1,2-bis(triethoxysilyl)ethane (BTEE) to improve hydrophobicity and promote liquid-phase mixing. Core-shell organosilica nanoparticles (csOS NPs) and solid silica core nanoparticles (sSiO2 NPs) were conjugated with 3-(trimethoxysilyl)propyl methacrylate (γ-MPS), creating csOS-MA and sSiO2-MA nanoparticles with methyl methacrylate (MA) groups for better filler-resin binding. Characterization techniques included scanning electron microscopy (SEM), transmission electron microscopy (TEM), dynamic light scattering (DLS), and Fourier transform infrared spectroscopy (FTIR). Single-weight amounts of sSiO2-MA and csOS-MA nanoparticles were used as fillers in composites tested for compressive strength (CS), modulus, and flexural strength (FS), polymerisation shrinkage (PS), and degree of conversion (DC). The findings revealed that the organosilica shell has a significant impact on the mechanical characteristics and DC of the nanocomposite. The study examined the water sorption (WSP) and water solubility (WSL) of nanoparticles prior to and following surface modification. The findings indicated that the addition of an organosilica shell significantly reduced both WSP and WSL while also improving the overall physical properties of the nanoparticles.The findings show that incorporating an organosilica shell on the surface of an inorganic silica core significantly enhances the design of dental restorative materials for nanocomposite filling restorations.
1. Introduction
Dental nanocomposite materials are constructed of inorganic filler particles incorporated in a polymer resin matrix that polymerises upon application. Polymer composites are popular due to their advantageous properties such as nontoxicity, biocompatibility, aesthetic appeal, and antibacterial activity.1 Among numerous polymers, including urethane dimethacrylate (UDMA), bisphenol A-glycidyl methacrylate (Bis-GMA) is commonly regarded as the preferred dental polymer. Its high viscosity, low volatility, and limited polymerisation shrinkage make it ideal for both anterior and posterior dental restorations. In contrast, urethane methacrylate was chosen for its higher hardness and lower viscosity. Issues arise when Bis-GMA is used as a resin due to its high viscosity. To address this, diluents such as triethylene glycol dimethacrylate (TEGDMA) and 2-hydroxyethyl methacrylate (HEMA) are incorporated to reduce the viscosity in dental composites.2–4 Similarly, inorganic fillers can be utilised to enhance the physical and performance characteristics of composite resins.5,6 Numerous efforts have been dedicated to developing inorganic fillers that deliver superior performance and ensure long-term clinical durability. These advancements focus on incorporating antibacterial properties, minimising polymerisation shrinkage, enhancing wear resistance, and achieving optimal mechanical properties, including flexural strength, compressive strength, hardness, flexural modulus, diametral tensile strength, fracture toughness, and elastic modulus.1,7 A variety of functionalized fillers can be introduced to achieve this. Typically, powdered silica, hydroxyapatite, zirconium dioxide, and barium glass are used as inorganic fillers in composite resins.8–10 As a result, current research is frequently focused on creating and enhancing fillers, including filler type, size (which develops from macrohybrid to microfill and finally nanofill), and shape.7,8,11Including nanoparticles in dental composite materials improves mechanical, chemical, and optical properties, including better filler dispersion in the matrix.12,13 Among several compounds that are applied as inorganic fillers like apatite, zeolite and phosphate, silica nanoparticles are more promising due to their porous structure and bio-compatibility represented by low toxicity rates and low density.14,15 Karabela et al., for example, investigated how nanosilica particles with sizes ranging from 7 to 40 nm impact the physico-mechanical characteristics of composite resins. The effects of 3-methacryloxypropyltrimethoxysilane, a silane coupling agent, were also investigated. The results revealed that, independent of filler particle size, the MPS% on a silica surface increases with decreasing silica particle size. Composite conversion with sorbed water improves flexural strength and modulus.16
However, as noted, the nanoparticles added to the dental composite improve its physicochemical characteristics but still have limitations. The greater surface-to-volume ratio of the nanoparticles may cause increased water sorption, which could damage the resin–matrix interface.17 Another disadvantage of employing nanoparticles as fillers is that, when added to high-viscosity resin monomers, their enormous surface area leads to low filler loading and poor particle wetting. On the other hand, as the agglomerate expanded, its surface area decreased. The water sorption resulted in a more wetting composite. The surface charge of nanoparticles not only results in aggregated forms but also stops them from spreading throughout the matrix phase. Maintaining proper particle dispersion causes the composite to have many weak points, which eventually causes the dental restoration to fail.18
In addition to fillers, an adhesive agent (often silane) was utilised to bind the fillers to the resin matrix in the composite.19 In adhesive dentistry, the shift from classical salinisation to current intermolecular cross-linking methodology has been revolutionised. The addition of a silane coupling agent to the filler surface of composites is expected to increase cohesive strength via chemical interaction. This process is accomplished by the condensation of silanol functional groups with pre-hydrolysed silane and hydroxyl groups on the surface of silica nanoparticles.18,20 Water absorbed by the composites may break up the silane agent binding.21,22 Based on this, developing a resin composite that meets both the aesthetic and mechanical requirements for anterior and posterior restorations represented a challenge.23 In 2006 Matinlinna et al. were the first to apply a dipodal silane, namely 1,2-bis(triethoxysilyl)ethane (BTEE), as a solution to the hydrolytic instability commonly associated with traditional monopodal coupling agents.24 Afterwards, in their subsequent 2008 investigation, they also proved the influence of silane dilution; the maximum shearing bond strength and water resistance after heat treatment was observed at 1.0% BTEE formulation (1.0 vol% MPS + 1.0 vol% BTEE).25 This sort of research culminated in a novel experimental design by Matinlinna et al. in 2010, addressing silane applications in orthodontics, especially to increase the adhesion between dental resin and prepared titanium using the dipodal structure of BTEE. The focus is on developing a strong siloxane network on silicatized surfaces those functions as a coupling agent.26
The research assesses shear bond strength and the clinical behaviour of the BTEE-based system, measuring its influence on bond stability and failure rates resulting from its molecular design. It was found that the utilisation of specialised silane mixtures (e.g. 3-acryloxypropyltrimethoxysilane with BTEE) has the potential to maximise shear bond strength and durability and may lead to significantly enhanced bonding performance.27 It was also discovered that the modified silane layer containing BTEE may limit fungal colonisation, providing a dual benefit of enhanced structural bonding and increased biological resistance to peri-implant infections.28 To the best of our knowledge, the insertion of 1,2-bis(triethoxysilyl)ethane (BTEE) modified shell-modified SiO2 nanoparticles into dental composite systems has yet to be investigated.
The improvement of the filler phase in this work is one of the primary variables used to develop the resin-based dental nanocomposite material. This study investigated the impact of an organosilica shell framework on the surface of silica core nanoparticles, which created inorganic–organic silica core–shell nanoparticles (csOS NPs) reinforced with γ-MPS groups on some of the physicochemical properties of light-cured nanocomposites. The resin nanocomposite included a Bis-GMA/TEGDMA (49.5/49.5 wt/wt) resin matrix, a co-initiator of 0.8% EDMAB, and an initiator of 0.2% CQ. To accomplish this, silica core nanoparticles were created using hydrolysis and condensation. A similar method was used to coat the sSiO2 core nanoparticles with an organosilica shell. Finally, the post-grafting approach was employed to embed γ-MPS groups on the surface. All the prepared samples were examined utilising SEM, TEM, FTIR, and DLS techniques. The contact angle was measured both before and after surface coating in order to assess the hydrophilicity of the produced samples. To investigate the effects of the organosilica shell's hydrophobicity on the physicochemical characteristics of dental nanocomposites. Single weights of sSiO2-MA NPs and csOS-MA NPs were added to the nanocomposites. The degree of conversion (DC), polymerisation shrinkage (PS), flexural strength (FS), modulus, and compressive strength (CS) were assessed in connection with csOS-MA NPs. Additionally, water sorption (WSP) and polymerisation shrinkage (PS) were measured and compared to SiO2-MA nanocomposites with the same filler weight fractions. Due to its porous structure and adsorption properties. Nanoparticles of silica dioxide possess extremely high surface activity and adsorb various ions and molecules.
2. Materials and methods
2.1 Experimental materials
Tetraethyl orthosilicate (TEOS, 99% (GC)), triethylene glycol dimethacrylate (TEGDMA, 95%), 1,2-bis(triethoxysilyl)ethane (BTEE, 96%), 3-(trimethoxysilyl)propyl methacrylate (γ-MPS, 98%), 2,2-Bis[p-(2′-hydroxy-3′-methacryloxypropoxy)phenylene] propane (Bis-GMA, 99%) were all provided by SIGMA-ALDRICH CHEMIE GmbH (Steinheim, Germany). The co-initiator ethyl-4-dimethylaminobenzoate (4-EDMAB, 99%), and initiator camphorquinone (CQ, 98%) both were provided from J&K Scientific GmbH (Pforzheim, Germany). Ethyl alcohol (EtOH, 99.8%), ammonium hydroxide (NH4OH, 25%) (v/v), hydrochloric acid (HCl, 37%), and hexadecyltrimethylammonium bromide (CTAB, 98%) were purchased from Shanghai Chemical Reagent Co., Ltd (Shanghai, China). All chemicals were used as received from the supplier without any further purification. The main text of the article should appear here with headings as appropriate.2.2 Preparation of nanofiller
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water–ethanol mixture containing 1.1 mL of ammonia. After 30 minutes of stirring (500 rpm), the dispersed sSiO2 solution was gradually added dropwise while the CTAB solution continued stirring. After that, 10 µL of TEOS was quickly added to the previously mentioned mixture, followed by 50 µL of BTEE. The reaction was continued for 6 hours before the core–shell organosilica nanoparticles (csOS NPs) were collected by centrifugation (10 min at 5000×g at room temperature in Falcon™ 50 mL conical centrifuge tubes). The sample was then washed twice with absolute ethanol and twice with DDW before being vacuum-dried overnight.
1 water–ethanol mixture containing 1.1 mL of ammonia. After 30 minutes of stirring (500 rpm), the dispersed sSiO2 solution was gradually added dropwise while the CTAB solution continued stirring. After that, 10 µL of TEOS was quickly added to the previously mentioned mixture, followed by 50 µL of BTEE. The reaction was continued for 6 hours before the core–shell organosilica nanoparticles (csOS NPs) were collected by centrifugation (10 min at 5000×g at room temperature in Falcon™ 50 mL conical centrifuge tubes). The sample was then washed twice with absolute ethanol and twice with DDW before being vacuum-dried overnight.2.3 Preparation of dental nanocomposite
2.4 Measurements
 | (1) |
 | (2) |
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds (1638 cm−1) to an internal standard of aromatic C
C bonds (1638 cm−1) to an internal standard of aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bonds (1608 cm−1). Each nanocomposite sample was tested according to three separate tests. The DC (%) was determined using the formula below:
C bonds (1608 cm−1). Each nanocomposite sample was tested according to three separate tests. The DC (%) was determined using the formula below:
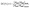 | (3) |
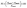 | (4) |
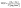 | (5) |
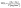 | (6) |
3. Results and discussion
3.1 Structure and morphology characterisation
Inorganic–organic silica core–shell nanoparticles (csOS NPs) were produced by creating a shell framework using 1,2-bis(triethoxysilyl)ethane (BTEE) as an organo-silica source on the surface of pre-synthesised solid silica cores. This was followed by grafting the surface of NPs with methacrylate (MA) groups using 3-(trimethoxysilyl)propyl methacrylate (γ-MPS). The preparation process is shown in Fig. 1. | ||
| Fig. 1 Representation of the preparation procedure of sSiO2 NPs, csOS and surface modification with MA groups. | ||
This is achieved by first creating monodisperse sSiO2 NPs using the Stöber technique, with a diameter of ∼210 nm and a spherical shape, as observed by SEM and TEM (Fig. 2a and d). The findings showed that the particle size of the silica (sSiO2) core was approximately consistent, as evidenced by the data obtained from SEM and TEM, as illustrated in Fig. 2(a and d). After successful formation of sSiO2 NPs, csOS NPs were created by hydrolysis and condensation of BTEE on the surface of sSiO2 NPs, with a percentage of CTAB surfactant as a pore formation agent, as shown in step 2 (Fig. 1). The particle size and surface shape of csOS NPs, as determined by SEM and TEM measurements, are also shown in Fig. 2. The finding suggested that the particle size of csOS NPs increased to about 256 nm in diameter after shell formation as observed by SEM (Fig. 2b). The surface modification of csOS NPs with MA groups has been carried out in an ethanol solution using γ-MPS to improve the adhesion force between the resin matrix and filler. The SEM examination of these particles revealed a slight increase in size, with an average size of around 259 nm (Fig. 2c). The particle size of synthesised csOS NPs after surface modification, denoted as (csOS-MA), was further confirmed by TEM examinations, which indicated a shell thickness of around 23 nm.The results demonstrated that the created shell formwork remained intact and homogeneous spherical shapes with no sign of particle aggregation. Furthermore, the low density of this shell is further proof of its formation, shown in the inset image of TEM measurement (Fig. 2e). To evaluate the size distribution of synthetic samples, at least 50 particles were analysed using ImageJ. Gaussian fitting was then used to calculate the average particle sizes; Fig. 2(g and h) display the size distribution of the particles measured by SEM and TEM, respectively.
The results were listed in Table 1. The hydrodynamic dimensions and PDIs of the synthesised NPs as determined by dynamic light scattering (DLS) are also displayed in Table 1. The results revealed that the hydrodynamic diameter of these NPs is slightly greater than that assessed by SEM and TEM for the same size. This is explained by the hydrodynamic diameter measured by DLS, which reflects the hydrated layer on the particle's outer surface. The PDI values of the measured particles were similarly low, indicating that the particles are monodisperse and show no evidence of aggregation, consistent with the results from SEM and TEM observations. Additionally, sSiO2 NPs were surface modified with MA groups using the same technique used to prepare csOS-MA NPs. These NPs were employed as a reference material to examine the water sorption and bonding strength of csOS-MA NPs when utilised as a filler in dental nanocomposites. The particle sizes of sSiO2-MA NPs, as measured by TEM and SEM, are given in Table 1 and are about 382 and 363 nm, respectively. In contrast to csOS NPs, it has been noted that sSiO2 particles exhibit a significant increase in particle size following silanation with MA groups. The hydrophilic characteristics of inorganic sSiO2 NPs may explain this, as they increase the particles' ability to hydrolyse silane groups, forming multiple layers on their surface and causing a significant shift in particle size. On the other hand, the hydrophobicity of the organic groups on the surface of csOS NPs reduces cocondensation of silane groups, resulting in a thinner, more uniform layer. This is indicated by a slight increase in particle size following surface modification.35,36 The successful fabrication of organosilica shells was further proved when TEM examinations of sSiO2-MA NPs (Fig. 2f) revealed a constant particle density, but TEM observations of csOS-MA NPs (Fig. 2e) revealed a low-density shell structure caused by organosilica shell creation. The SEM measurements revealed spherical particles, as shown in Fig. S1. According to the DLS data given in Table 1, the particle size was around 322 nm, with a polydispersity index (PDI) of 0.11.
| Sample | DLS | SEM | TEM | |||
|---|---|---|---|---|---|---|
| Dh (nm) | PDI | D (nm) | SD (nm) | D (nm) | SD (nm) | |
| sSiO2 NPs | 221.49 | 0.024 | 208.8 | 21.1 | 212.5 | 10.6 |
| csOS NPs | — | — | 256.1 | 23.3 | — | — |
| csOS-MA NPs | 292.23 | 0.083 | 259.7 | 17.4 | 254.4 | 15.3 |
| sSiO2-MA NPs | 322.16 | 0.117 | 363.3 | 14.3 | 382.5 | 18.4 |
3.2 Chemical structures
The chemical structures of the sSiO2 and csOS NPs before and after surface modifications with MA groups were examined using FT-IR, and the results are shown in Fig. 3. All synthesised samples exhibited absorption bands at 3733 cm−1, 3409 cm−1, 1108 cm−1, 943 cm−1, and 798 cm−1. The broad absorption bands at 3733 cm−1 and 3409 cm−1 correspond to the stretching vibrations of the O–H bond of Si–OH and water. The distinctive absorption band of the Si–O–Si bond is comprised of two peaks at about 1108 cm−1 and 798 cm−1, attributed to the symmetric and asymmetric vibrations, respectively. After the fabrication of the organosilica shell, it was found that the absorption band at 1108 cm−1, which was attributed to the asymmetric vibration of the regular tetrahedral structure of Si–O–Si in the sSiO2 spectrum, was shifted to a lower frequency and appeared at 1079 cm−1. This could be attributed to the deformation of the SiO4tetrahedron structure, which was briefly described in our previous study.29,35 It has also been demonstrated that surface modification of sSiO2 NPs and csOS NPs with MA further shifts the Si–O–Si absorption bands at 1108 cm−1 of the sSiO2 spectra to lower frequencies, reaching 1099 cm−1 and 1097 cm−1, respectively. The Si–OH bond vibration is represented by the absorption band at 943 cm−1. The shoulder, which was identified at about 1192 cm−1, was attributed to a split of longitudinal and transverse optical stretching motion.37,38 In comparison to the sSiO2 spectrum, when BTEE precursor and/or γ-MPA were fabricated on the surface of sSiO2, new peaks appeared at around 2964 cm−1, 2851 cm−1, and 1412 cm−1. The peaks at 2964 cm−1 and 2851 cm−1 represent the stretching vibration of the C–H group, while the band at 1412 cm−1 represented C–H deformation vibration, indicating successful surface modification.29,30 The water deformation band appears at around 1635 cm−1.39 As shown in Fig. 3 (red and blue lines), the hydrophobic effect of the organosilica shell, which prevents the accumulation of water molecules on the surface while also reducing the number of silanol hydroxyl groups on the surface, could explain the absence of this band in csOS spectra and its decreased intensity in csOS-MA spectra. The decreasing peak at 943 cm−1, ascribed to the Si–OH bond vibration, supports these predictions. Furthermore, when the csOS spectrum is compared to the SiO2-MA and csOS-MA spectra, as depicted in Fig. 3 (blue and brown lines), a new distinct peak at 1635 cm−1 is observed. This peak is attributed to the contraction vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C in γ-MPA and could be covered by the O–H vibration peak. This implies that γ-PMA was effectively grafted onto the csOS NPs and sSiO2 surfaces.40 These results demonstrate the effective synthesis of sSiO2 and csOS NPs modified with MA groups, which are consistent with TEM and SEM investigations.
C in γ-MPA and could be covered by the O–H vibration peak. This implies that γ-PMA was effectively grafted onto the csOS NPs and sSiO2 surfaces.40 These results demonstrate the effective synthesis of sSiO2 and csOS NPs modified with MA groups, which are consistent with TEM and SEM investigations.
The framework bridges reduce the density of hydrophilic silanol groups. The calculated p-value was p < 0.0001, which falls below the critical threshold of p < 0.05. This finding is consistent with that of letreture, which shows that the insertion of organosilica shell exhibits exceptional hydrolytic stability, being up to 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 times more resistant to water degradation than conventional silanes, resulting in consistent wettability in aqueous environments.41,42 This notion was also tested in our prior work, which found that, although ordinary silica is very hydrophilic and unstable in aqueous solution at high temperatures, the BTEE organosilica shell has moderate hydrophobicity and excellent water stability, even at high pH levels.29
000 times more resistant to water degradation than conventional silanes, resulting in consistent wettability in aqueous environments.41,42 This notion was also tested in our prior work, which found that, although ordinary silica is very hydrophilic and unstable in aqueous solution at high temperatures, the BTEE organosilica shell has moderate hydrophobicity and excellent water stability, even at high pH levels.29
3.3 Resin-based dental nanocomposites study
| Sample | Degree of conversion (%) | Flexural strength (MPa) | Flexural modulus (GPa) | Compressive strength (MPa) | Polymerisation shrinkage (%) | Water sorption (%) |
|---|---|---|---|---|---|---|
| a Note: statistical analysis was performed using the independent samples t-test. All obtained P-values were found to be (p < 0.05). | ||||||
| sSiO2-MA | 65.8 (2.2) | 126.5 (2.15) | 2.56 (0.04) | 132 (4.6) | 2.1 (0.07) | 1.2 (0.04) |
| csOS-MA | 76.2 (2.8) | 132.7 (2.22) | 2.72 (0.05) | 143 (5.2) | 0.45 (0.02) | 0.3 (0.01) |
| P-value | 0.012 | 0.041 | 0.033 | 0.003 | <0.001 | <0.001 |
The shell, characterised by its nanoporosity, creates internal surfaces that effectively trap photons. Additionally, the particles function as ‘nano-optical fibres’, channelling light both laterally and downward. This interaction facilitates efficient energy utilisation from the incident light throughout the entire functional thickness. As a result, appropriately controlled scattering by these hybrid fillers enhances both optical performance and polymerisation efficiency.
The flexural modulus of both under-investigation samples was also determined using the three-point bending test for nanocomposite samples, and the findings are presented in the table. The flexural modulus was increased by about 6.25% in the case of using csOS-MA as a filler instead of SiO2-MA. Additionally, the compression strength of dental restorations was examined and presented in Table 2. The csOS-MA NPs-based nanocomposite outperformed the SiO2-MA NPs-filled nanocomposite by about 8.3% in compressive strength. The compressive strength of the inorganic silica filler-based nanocomposite was around 132 MPa, whereas the organosilica filler-based nanocomposite was around 143 MPa. Employing csOS-MA NPs could enhance the compressive strength of dental restorations more effectively than SiO2-MA NPs, owing to their superior mechanical properties. In terms of modulus, flexural strength, and compression, the identical trend seen in Fig. 4b–d implies that the influence of the organosilica nanoparticle is similar in the changed specimens. Significant differences (p < 0.05) were identified across FS, FM, and CS groups, indicating significant diversity in performance. The improved qualities reported in the material, following surface fabrication with organosilica that contains ethylene bridage, are most likely due to the mechanical and structural phenomena of micro-flexibility. The Si–CH2–CH2–Si bridges of BTEE lend “organic flexibility” to the inorganic backbone, in contrast to the rigid and brittle character of inorganic silica.46 These bridges act as molecular-scale springs, giving the composite increased fracture toughness by allowing for minor structural deformation without breaking.47 The resin may produce an interpenetrating polymer network by penetrating the filler particles due to its high porosity. As a result, the resin solidifies inside the pores, improving bonding and creating “mechanical anchors”.48 This causes the failure mode to change from adhesive failure at the interface to cohesive strength. Additionally, the organosilica shell acts as a buffer zone between the flexible resin matrix and the rigid filler, significantly reducing interface stress and so successfully preventing macroscopic fractures in composites.49
4. Conclusions
In the current study, the impact of csOS-MA NPs on reinforced dental nanocomposite materials in comparison to sSiO2-MA NPs has been thoroughly investigated. NPs have been effectively synthesised using the sol–gel approach and silane-coated using the post-grafting technique. A single-weight NPs-to-matrix ratio was used to create the nanocomposite resins. The study's results showed that the overall mechanical characteristics and degree of conversion were improved by using csOS-MA NPs as a filler nanocomposite rather than sSiO2 NPs. The independent t-test results showed that samples with functionalized particles possessed a significant increase in flexural modulus, flexural strength, and compressive strength. The organic–inorganic framework and ethylene bridges contribute to these enhancements by improving the bonding with the polymer matrix. This alteration resulted in a change in contact angles, shifting from approximately 39.16° (hydrophilic) to around 112.76° (hydrophobic). The presence of a porous shell structure and effective surface modification was confirmed through FTIR and TEM analyses. These modifications have enhanced the performance of the nanocomposite and maintained its structural integrity. Combining these fillers with standard Bis-GMA/TEGDMA monomers increases hardness, strength, and conversion degree while decreasing polymerisation shrinkage and water sorption. Such advancements are especially important for restorative engineering in dentistry, where superior mechanical properties and conversion rates of reinforcing materials in nanocomposite resins can increase durability. A crucial finding of the study is that organic groups reduce water absorption, thereby minimising shrinkage and enhancing the longevity of dental nanocomposites. While this work focuses on the key physicochemical features of the produced nanocomposites. However, further study is needed to thoroughly evaluate the impacts of hybrid shell thickness and nanoporosity. It is also critical to assess long-term hydrolytic stability and biocompatibility to provide clinical dependability in complex oral environments.Author contributions
M. A. A., A. O. I. and A. A. K. contributed to the project conceptualisation; M. A. A., A. O. I., A. A. K., and H. O. H. conceived the methodology and designed the experiments; M. A. A., M. K. M., H. O. H., and Z. V. performed the experiments and analysed the data; A. O. I. and A. A. K. conducted the statistical analysis of the experimental data. M. A. A. and Z. V. carried out the writing-original draft preparation. All authors contributed to the writing-review of the paper. All authors have read and agreed to the published version of the manuscript.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data that support the findings of this study are available on reasonable request from the corresponding author.All the data produced and/or analyzed in the course of this study are presented within this manuscript and its accompanying supplementary information (SI) file. Supplementary information is available. See DOI: https://doi.org/10.1039/d6ra01974a.
Acknowledgements
This work was partially funded by the ADVANCED 150077 (to ZV) grant by the National Research, Development and Innovation Office, Hungary (NKFIH).References
- R. Yadav and M. Kumar, J. Oral Biosci., 2019, 61, 78–83 CrossRef PubMed.
- S. Shin, Y.-J. Kim, M. Toan, J.-G. Kim, T. Nguyen and J. Ku Cho, Eur. Polym. J., 2017, 92, 338–345 CrossRef CAS.
- M. Hosseinalipour, J. Javadpour, H. Rezaie, T. Dadras and A. N. Hayati, J. Prosthodontics, 2010, 19, 112–117 CrossRef PubMed.
- R. Srivastava, J. Wolska, J. Walkowiak-Kulikowska, H. Koroniak and Y. Sun, Eur. Polym. J., 2017, 90, 334–343 CrossRef CAS.
- J. L. Ferracane, Dent. Mater., 2011, 27, 29–38 CrossRef CAS PubMed.
- K. D. Son and Y.-J. Kim, Mater. Sci. Eng. C, 2013, 33, 499–506 CrossRef CAS PubMed.
- E. Habib, R. Wang, Y. Wang, M. Zhu and X. X. Zhu, ACS Biomater. Sci. Eng., 2016, 2, 1–11 CrossRef CAS PubMed.
- M. Ai, Z. Du, S. Zhu, H. Geng, X. Zhang, Q. Cai and X. Yang, Dent. Mater., 2017, 33, 12–22 CrossRef CAS PubMed.
- K. D. Jandt and B. W. Sigusch, Dent. Mater., 2009, 25, 1001–1006 CrossRef CAS PubMed.
- N. Ilie and R. Hickel, Clin. Oral Invest., 2009, 13, 485–487 CrossRef.
- H. A. Rodríguez, W. M. Kriven and H. Casanova, Mater. Sci. Eng. C, 2019, 101, 274–282 CrossRef PubMed.
- P. Saen, M. Atai, A. Nodehi and L. Solhi, Dent. Mater., 2016, 32, e185–e197 CrossRef CAS PubMed.
- D. Angerame and M. De Biasi, Operat. Dent., 2018, 43, E191–E209 CrossRef CAS PubMed.
- L. Mokeem, A. A. Balhaddad, I. M. Garcia, F. M. Collares and M. A. S. Melo, in Emerging Nanomaterials and Nano-Based Drug Delivery Approaches to Combat Antimicrobial Resistance, Elsevier, 2022, 661–700 Search PubMed.
- J. Y. Quek, E. Uroro, N. Goswami and K. Vasilev, Mater. Today Chem., 2022, 23, 100606 CrossRef CAS.
- M. M. Karabela and I. D. Sideridou, Dent. Mater., 2011, 27, 825–835 CrossRef CAS PubMed.
- A. R. Curtis, A. C. Shortall, P. M. Marquis and W. M. Palin, J. Dent., 2008, 36, 186–193 CrossRef CAS PubMed.
- J. M. Antonucci, S. H. Dickens, B. O. Fowler and H. H. K. Xu, J. Res. Natl. Inst. Stand. Technol., 2005, 110, 541 CrossRef PubMed.
- A. A. Zandinejad, M. Atai and A. Pahlevan, Dent. Mater., 2006, 22, 382–387 CrossRef CAS PubMed.
- M. Atai, A. Pahlavan and N. Moin, Dent. Mater., 2012, 28, 133–145 CrossRef CAS PubMed.
- R. K. Nayak, K. K. Mahato and B. C. Ray, Composites, Part A, 2016, 90, 736–747 CrossRef CAS.
- V. K. Balla, K. H. Kate, J. Satyavolu, P. Singh and J. G. D. Tadimeti, Composites, Part B, 2019, 174, 106956 CrossRef CAS.
- F. Ariyani and M. Anindhita, J. Med. Chem. Sci., 2022, 5, 335–340 CAS.
- J. P. Matinlinna, L. V. J. Lassila and P. K. Vallittu, J. Dent., 2006, 34, 436–443 CrossRef CAS PubMed.
- J. Matinlinna, M. Özcan, L. Lassila, W. Kalk and P. Vallittu, Acta Odontol. Scand., 2008, 66, 250–255 CrossRef CAS PubMed.
- J. P. Matinlinna and L. V. Lassila, Silicon, 2010, 2, 79–85 CrossRef CAS.
- A. A. Khan, A. A. Al Kheraif, J. Syed, D. D. Divakar and J. P. Matinlinna, Dent. Mater. J., 2017, 36, 111–116 CrossRef CAS PubMed.
- X. Liu, S. Chen, H. Ding and J. K. H. Tsoi, Coatings, 2024, 14, 1277 CrossRef CAS.
- M. A. Al-Khafaji, A. Gaál, B. Jezsó, J. Mihály, D. Bartczak, H. Goenaga-Infante and Z. Varga, Nanomaterials, 2022, 12, 1172 CrossRef CAS PubMed.
- M. A. Al-Khafaji, A. Gaál, B. Jezsó, J. Mihály and Z. Varga, Materials, 2022, 15, 2696 CrossRef CAS PubMed.
- F. Liu, R. Wang, Y. Cheng, X. Jiang, Q. Zhang and M. Zhu, Mater. Sci. Eng. C, 2013, 33, 4994–5000 CrossRef CAS PubMed.
- L. Calabrese, F. Fabiano, L. M. Bonaccorsi, V. Fabiano and C. Borsellino, Int. J. Biomater., 2015, 2015, 1–7 CrossRef PubMed.
- R. Wang, E. Habib and X. X. Zhu, Dent. Mater., 2018, 34, 1014–1023 CrossRef CAS PubMed.
- C. Santos, R. L. Clarke, M. Braden, F. Guitian and K. W. M. Davy, Biomaterials, 2002, 23, 1897–1904 CrossRef CAS PubMed.
- M. A. Al-Khafaji, Eötvös Loránd Tudományegyetem, 2023 Search PubMed.
- V. Gubala, G. Giovannini, F. Kunc, M. P. Monopoli and C. J. Moore, Cancer Nanotechnol., 2020, 11, 1 CrossRef CAS.
- S. Kanugala, S. Jinka, N. Puvvada, R. Banerjee and C. G. Kumar, Sci. Rep., 2019, 9, 6198 CrossRef PubMed.
- D.-M. Han, G.-Z. Fang and X.-P. Yan, J. Chromatogr. A, 2005, 1100, 131–136 CrossRef CAS PubMed.
- W. Yang, L. Zhu and Y. Chen, RSC Adv., 2015, 5, 58973–58979 RSC.
- L. Xie, X. Liang, H. Huang, L. Yang, F. Zhang, X. Li and Z. Luo, RSC Adv., 2019, 9, 1123–1133 RSC.
- J. D. Cyran, M. A. Donovan, D. Vollmer, F. Siro Brigiano, S. Pezzotti, D. R. Galimberti, M.-P. Gaigeot, M. Bonn and E. H. G. Backus, Proc. Natl. Acad. Sci. USA, 2019, 116, 1520–1525 CrossRef CAS PubMed.
- S. M. Mohd Ibrahim, K. Sawamura, K. Mishina, X. Yu, F. Salak, S. Miyata, N. Moriyama, H. Nagasawa, M. Kanezashi and T. Tsuru, Membranes, 2023, 14, 8 CrossRef PubMed.
- M. Santos, R. Fidalgo-Pereira, O. Torres, O. Carvalho, B. Henriques, M. Özcan and J. C. M. Souza, Clin. Oral Invest., 2024, 28, 454 CrossRef PubMed.
- F. Hoffmann, M. Cornelius, J. Morell and M. Fröba, Angew. Chem., Int. Ed., 2006, 45, 3216–3251 CrossRef CAS PubMed.
- C. H. Zanchi, F. A. Ogliari, R. Marques E Silva, R. G. Lund, H. H. Machado, C. Prati, N. L. V. Carreño and E. Piva, Appl. Adhes. Sci., 2015, 3, 27 CrossRef.
- T. Shimizu, K. Kanamori, A. Maeno, H. Kaji and K. Nakanishi, Langmuir, 2016, 32, 13427–13434 CrossRef CAS PubMed.
- T. Hamada, Y. Nakanishi, K. Okada and J. Ohshita, ACS Omega, 2021, 6, 8430–8437 CrossRef CAS PubMed.
- F. Liu, S. Yao, J. Li, K. Huang, D. Zhang, T. M. Wong, R. Tang, K. W. K. Yeung and J. Wu, Nat. Commun., 2025, 16, 4423 CrossRef CAS PubMed.
- H. L. Castricum, G. G. Paradis, M. C. Mittelmeijer-Hazeleger, R. Kreiter, J. F. Vente and J. E. Ten Elshof, Adv. Funct. Mater., 2011, 21, 2319–2329 CrossRef CAS.
- G. Dubois, W. Volksen, T. Magbitang, R. D. Miller, D. M. Gage and R. H. Dauskardt, Adv. Mater., 2007, 19, 3989–3994 CrossRef CAS.
- H. Chen, J. Luo, J. Yang, C. Zeng and X. Jiang, Biomolecules, 2023, 13, 1290 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |



