DOI:
10.1039/D6RA01748G
(Paper)
RSC Adv., 2026,
16, 24794-24807
Spin-polarized DFT study of Pr2EuMO6 (M = Co, Fe) double perovskites for spintronic and energy applications
Received
28th February 2026
, Accepted 14th April 2026
First published on 12th May 2026
Abstract
Spin-polarized DFT-based computations have been performed using mBJ + U approximations to explore the structural, optoelectronic, magnetic, and thermoelectric characteristics of Pr2EuMO6 (M = Co, Fe) materials in the cubic (Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m) space group. The study has made the materials promising in spintronic applications due to their half-metallic nature. Magnetic analysis shows that the materials are ferromagnetic. The overall magnetic moments of Pr2EuCoO6 and Pr2EuFeO6 are 11 (µB) and 14 (µB), respectively. Optical analyses of the two materials are conducted in the 0 to 14 eV energy range. The optical parameter study indicates that the materials are excellent in terms of photovoltaic and high-energy absorbent applications. The Seebeck coefficient shows that Pr2EuCoO6 and Pr2EuFeO6 are n- and p-type semiconductors, respectively. The elevated values of ZT at room temperature suggest that both materials have good thermoelectric efficiency, and the rising values of PF with increasing temperature suggest that the two materials can be used in high-temperature thermoelectric applications.
m) space group. The study has made the materials promising in spintronic applications due to their half-metallic nature. Magnetic analysis shows that the materials are ferromagnetic. The overall magnetic moments of Pr2EuCoO6 and Pr2EuFeO6 are 11 (µB) and 14 (µB), respectively. Optical analyses of the two materials are conducted in the 0 to 14 eV energy range. The optical parameter study indicates that the materials are excellent in terms of photovoltaic and high-energy absorbent applications. The Seebeck coefficient shows that Pr2EuCoO6 and Pr2EuFeO6 are n- and p-type semiconductors, respectively. The elevated values of ZT at room temperature suggest that both materials have good thermoelectric efficiency, and the rising values of PF with increasing temperature suggest that the two materials can be used in high-temperature thermoelectric applications.
1 Introduction
The rising demand for innovative materials with enhanced features is propelling the technological development of the future. Consequently, the innovative design of new materials is changing considerably in the materials science field.1 Interest in the use of double perovskite materials has been both theoretical and empirical due to their numerous industrial and technical applications. The applications of these types of substances in spintronics, magneto-optics, half-metallic materials, thermoelectrics, and ferromagnetism are only a few of the areas that have found themselves in need of this flexibility. The materials also hold great potential in the production of semiconductors and magneto-dielectric materials; therefore, they rank among the top priorities of developing future electronics, energy conversion, and magnetic sensing technology.2–4 Rare earth (RE) elements and alkaline earth compounds have recently received significant attention because of their special properties and potential applications. The most important sub-type of double perovskites is the transition-metal perovskite.5,6 Considering that these substances can be tailored to work with the most recent technologies, such as electronics, magnetics, and energy-related uses, they are being carefully explored owing to their versatile array of possibilities.4,7,8
In the case of double perovskites, the general equation is A2BB′O6, which contains an oxygen atom O and the cations A, B, and B′. It is based on the fundamental perovskite chemical ABO3. Due to their ability to replace other components at the A, B and B′ positions, double perovskites have superior flexibility in the arrangement of atoms and thus are highly intriguing. These replacements can influence the optical, magnetic, and electrical properties of the material by enabling a very high level of control over the crystal structure. The tunability of double perovskites can be applied in many applications, including electrical devices, magnetism, spintronics, energy conversion, and storage.7,9,10 For decades, scientists have been studying these types of substances, focusing on their special qualities and uses, since the 1960s. Numerous investigations throughout the years have revealed their extraordinary flexibility and plasticity, igniting continued curiosity about theoretical and experimental studies.11,12 More than three hundred materials possessing a double perovskite structure have been effectively created. This illustrates the astounding adaptability of the double perovskite structure, which permits a wide range of structural and chemical changes. These substances are a major area of study in the field of materials science because they provide certain qualities for use in electronics, magnetism, and energy technologies.13–15
Due to their remarkable thermal properties, double perovskite (DPO) materials are also useful in thermal equipment, including sensors that monitor temperatures, coolers, motors, and thermocouples. Effective thermal control is essential in harsh settings, such as those encountered in airplanes or bases in space, where certain materials are especially well-suited for use. They are essential for space engineering and other cutting-edge thermoelectric systems because of their capacity to transform heat into electrical energy or to precisely regulate temperature.16–18 The addition of an electron's spin degree of motion has sparked an explosion in electronics and given rise to sophisticated spin-based technologies, or spintronics. The strong spin polarization of compounds is thought to be particularly beneficial for use in spintronic technologies. This novel method has several benefits over conventional electronic equipment, including higher processing rates, lower power usage, and permanent memory storage for information. Utilizing the inherent spin characteristics of electrons, spintronics holds promise for greatly enhancing device efficiency and opening the door for the future creation of electronic gadgets and devices. Further research on appropriate materials and procedures is essential to develop this innovative subject.19,20
A thorough literature review on double perovskites has been carried out to investigate previous studies on the materials. Towfiq et al.21 reported the half-metallic behavior and electronic structure of the Bi2FeMnO6 magnetic compound. Belhachi et al.6 also reported the half-metallic quality and electronic, magnetic and optical properties of the Ba2CoRhO6 compound. Mudasir et al.22 reported the spin-polarized properties of double perovskite K2GeMnX6 (X = Cl, Br, I). Bhuyan et al.23 reported the spintronic properties of Nd2CrFeO6, a double perovskite. Caid et al.24 explored the spin-dependent physical properties of Cs2B′GeCl6 (B′ = Zn, Cd), a double perovskite family. A Ali et al.25 recently investigated the magnetic, thermoelectric, and optoelectronic properties of Ba2GdXO6 (X = Nb and U) double perovskites using DFT analysis. These materials are ferromagnetic and semiconducting and exhibit excellent optical and thermoelectric characteristics. A Ali et al.26 reported double perovskite materials owing to their optoelectronic and magnetic properties using a spin-based first-principles investigation. These investigations suggest the antiferromagnetic and half-metallic nature of Sr2UFeO6 and the ferromagnetic and semiconducting nature of Sr2UNiO6. Zia et al.27 performed spin-based computations of Cs2XMoBr6 (X = Na, Li) double halide perovskites for investigations of the magnetic, thermoelectric, and optoelectronic characteristics of materials. Mazumdar et al.28 experimentally synthesized Pr2FeCrO6, a double perovskite material, and investigated its magnetic, structural, and magneto-caloric properties. Dhilip et al.29 computationally and experimentally analyzed the Pr2CoFeO6 double perovskite and investigated the structural, optoelectronic, and magnetic characteristics of the material. Saadi30 elaborated the half-metallic and magnetic nature of Sr2GdReO6 material using first principles investigations. The material is found to be half-metallic and ferromagnetic in nature. The ferromagnetic and half-metallic nature of X2MnUO6 (X = Sr or Ba) materials has been revealed by a first principles study using the FP-LAPW method.31 Saadi et al.32 reported the ferromagnetic and half-metallic nature of RBaMn2O6−δ (X = Nd, Pr, La and δ = 0, 1) compounds using a DFT study. A first principles investigation was performed to predict the thermoelectric and half-metallic characteristics of Sr2EuReO6, a double perovskite compound.33
The present research focuses on the first principles investigations of novel Pr2EuMO6 (M = Co, Fe) double oxide perovskites. This study aims to examine the structural, electronic, magnetic, and thermoelectric characteristics of double perovskites Pr2EuMO6 (M = Co, Fe) using first-principles calculations. The primary goals of this study are to investigate how the selection of transition metal (Co vs. Fe) affects the electronic band structure and magnetic ordering and to assess whether these materials display half-metallic ferromagnetism, positioning them as potential candidates for spintronic applications. This study also examines the thermoelectric performance of both compounds in relation to temperature, aiming to evaluate their potential for energy conversion applications. Variations in transition metals are believed to significantly influence their physical properties, resulting in unique magnetic moments and thermoelectric behaviors across various temperature ranges. The detailed research literature review predicts, both theoretically and experimentally, the novelty of the materials under study.
2 Computational method
The FP-LAPW technique, which has been implemented inside the DFT-based WIEN2K computational framework,34 is utilized to perform spin-polarized computational tasks to investigate the magnetic electronic and optical properties of materials in the cubic (Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m) space group.35,36 The WEIN2K code is selected to perform the structural, electronic, optical, and magnetic calculations of the materials with high accuracy, as it is based on the full potential method, which precisely incorporates the effects of core electrons, providing benefits over other DFT-based codes that utilize pseudopotentials. To account for both the precise band gap estimation and strong on-site Coulomb interactions in the localized d/f orbitals, electronic structure calculations were carried out using the modified Becke–Johnson (mBj) exchange potential in combination with the Hubbard U correction (mBj + U).37,38 The effective Hubbard U values are 6.0 eV for Pr-4f,39 6.5 eV for Eu-4f,40 3.5 eV for Co-3d,41 and 4.5 eV for Fe-3d orbitals.42 Although DFT + U, along with the TB-mBJ method, was used in the current study to consider the strong electron correlation in transition metal sites, it is acknowledged that the approach could still have a self-interaction error (SIE), which could negatively impact the accuracy of electronic property predictions. Other methods, including hybrid functionals, self-interaction-corrected (SIC) methods and other methods, have been demonstrated to provide better accuracy than standard DFT + U for systems containing transition metals.43,44 The wave function has been extended utilizing the sphere-shaped harmonic basis set as much as lmax = 10 across the not interconnected muffin-tin spheres. A plane-wave basis with a threshold value of RMT Kmax = 7 was implemented to extend the wave function throughout the interstitial region. The cut-off energy is chosen as −6.0 Ry to distinguish between the core and valence states. The incorporation of the Brillouin zone has been completed by employing a 10 × 10 × 10 k-mesh Monkhorst-Pack.45,46 In the present calculations, the energy and convergence criteria are considered whenever the charge difference is smaller than 0.001 e a.u.−3 per unit cell and the overall energy is steady within 0.0001 Ry. To avoid spurious oscillations and achieve higher quantitative accuracy, the optical characteristics were calculated using 15 × 15 × 15 k-points in the first Brillouin zone.47–50 To look into optical characteristics, the material's complex dielectric function is computed, as follows:51
m) space group.35,36 The WEIN2K code is selected to perform the structural, electronic, optical, and magnetic calculations of the materials with high accuracy, as it is based on the full potential method, which precisely incorporates the effects of core electrons, providing benefits over other DFT-based codes that utilize pseudopotentials. To account for both the precise band gap estimation and strong on-site Coulomb interactions in the localized d/f orbitals, electronic structure calculations were carried out using the modified Becke–Johnson (mBj) exchange potential in combination with the Hubbard U correction (mBj + U).37,38 The effective Hubbard U values are 6.0 eV for Pr-4f,39 6.5 eV for Eu-4f,40 3.5 eV for Co-3d,41 and 4.5 eV for Fe-3d orbitals.42 Although DFT + U, along with the TB-mBJ method, was used in the current study to consider the strong electron correlation in transition metal sites, it is acknowledged that the approach could still have a self-interaction error (SIE), which could negatively impact the accuracy of electronic property predictions. Other methods, including hybrid functionals, self-interaction-corrected (SIC) methods and other methods, have been demonstrated to provide better accuracy than standard DFT + U for systems containing transition metals.43,44 The wave function has been extended utilizing the sphere-shaped harmonic basis set as much as lmax = 10 across the not interconnected muffin-tin spheres. A plane-wave basis with a threshold value of RMT Kmax = 7 was implemented to extend the wave function throughout the interstitial region. The cut-off energy is chosen as −6.0 Ry to distinguish between the core and valence states. The incorporation of the Brillouin zone has been completed by employing a 10 × 10 × 10 k-mesh Monkhorst-Pack.45,46 In the present calculations, the energy and convergence criteria are considered whenever the charge difference is smaller than 0.001 e a.u.−3 per unit cell and the overall energy is steady within 0.0001 Ry. To avoid spurious oscillations and achieve higher quantitative accuracy, the optical characteristics were calculated using 15 × 15 × 15 k-points in the first Brillouin zone.47–50 To look into optical characteristics, the material's complex dielectric function is computed, as follows:51| | |
ε(ω) = ε1(ω) + ε2(ω).
| (1) |
The Kramers–Kronig relations were used to obtain the real component ε1 and the imaginary part ε2, which were determined by adding a suitably high number of unfilled states.52 Further, both ε1 and ε2 can be expressed mathematically using the following two equations:
| |
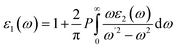 | (2) |
| |
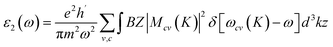 | (3) |
The real and imaginary parts of the dielectric function are utilized to determine the material's additional optical characteristics, employing the following derived formulas:
| |
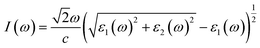 | (4) |
| |
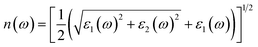 | (5) |
| |
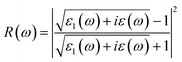 | (6) |
| |
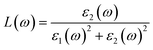 | (7) |
To compute the thermoelectric properties, the semi-classical Boltzmann transport theory was applied using the BoltzTraP code.53 The Seebeck coefficient, electrical conductivity and electronic thermal conductivity were determined using this code. The thermoelectric transport properties were calculated using the semi-classical Boltzmann transport theory within the constant relaxation time approximation (CRTA), as implemented in the BoltzTraP code. In this approach, only the electronic contribution to the transport coefficients (Seebeck coefficient, electrical conductivity, and electronic thermal conductivity) is evaluated based on the calculated electronic band structure. The lattice thermal conductivity (κl) is not included in this framework, as its calculation requires anharmonic phonon interactions and higher-order force constants, which are beyond the scope of the present study. In order to obtain precise values, a 1000 (10 × 10 × 10) k-mesh is employed to integrate the electronic energy levels of our mBJ + U calculation.
3 Results and discussions
3.1 Structural properties
Both Pr2EuCoO6 and Pr2EuFeO6 materials crystallize in the cubic Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group, as displayed in Fig. 1. The structures of these materials are optimized to obtain optimized bond lengths, lattice contents, and volumes. The energy vs. volume optimization curves are depicted in Fig. 7(a and b). The nature of the bonds among the material elements can be determined by computing the electronegativity difference between them. The electronegativity difference can be determined using Pauling values for each constituent element.54,55 For Pr2EuCoO6 and Pr2EuFeO6, the electronegativity difference for Pr–O, Eu–O, Co–O, and Fe–O bonds is 2.31, 2.24, 1.56, and 1.61, respectively, confirming that Pr–O and Eu–O have an ionic bonding nature while Co–O and Fe–O have a polar covalent bonding nature.56 The bonding nature can further be confirmed by studying the DOS plots (see Fig. 4), which show negligible hybridization between Pr–O and Eu–O, indicating ionic and strong hybridization near the Fermi level in the up-spin configurations between Co–O and Fe–O in Pr2EuCoO6 and Pr2EuFeO6, respectively, suggesting the covalent character of the materials. The tolerance factor provides insights into the crystal structure phase stability of the material. The concept of the tolerance factor for phase stability was first introduced by Megaw.57 Furthermore, Goldschmidt introduced the modified form of the tolerance factor, which is based on the ionic radii of the elements of the double perovskite materials. The modified form of the Goldschmidt tolerance factor (τ) formula for the materials under study is as follows:58
m space group, as displayed in Fig. 1. The structures of these materials are optimized to obtain optimized bond lengths, lattice contents, and volumes. The energy vs. volume optimization curves are depicted in Fig. 7(a and b). The nature of the bonds among the material elements can be determined by computing the electronegativity difference between them. The electronegativity difference can be determined using Pauling values for each constituent element.54,55 For Pr2EuCoO6 and Pr2EuFeO6, the electronegativity difference for Pr–O, Eu–O, Co–O, and Fe–O bonds is 2.31, 2.24, 1.56, and 1.61, respectively, confirming that Pr–O and Eu–O have an ionic bonding nature while Co–O and Fe–O have a polar covalent bonding nature.56 The bonding nature can further be confirmed by studying the DOS plots (see Fig. 4), which show negligible hybridization between Pr–O and Eu–O, indicating ionic and strong hybridization near the Fermi level in the up-spin configurations between Co–O and Fe–O in Pr2EuCoO6 and Pr2EuFeO6, respectively, suggesting the covalent character of the materials. The tolerance factor provides insights into the crystal structure phase stability of the material. The concept of the tolerance factor for phase stability was first introduced by Megaw.57 Furthermore, Goldschmidt introduced the modified form of the tolerance factor, which is based on the ionic radii of the elements of the double perovskite materials. The modified form of the Goldschmidt tolerance factor (τ) formula for the materials under study is as follows:58| |
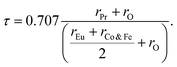 | (8) |
Here, the rPr, rEu, rCo, rFe, and rO are the ionic radii of the elements Pr, Eu, Co, Fe, and O, respectively. The values of these ionic radii are suggested by Shannon et al.59 The radii (in Å) of the material elements are rPr = 1.126, rEu = 1.120, rCo = 0.545, rFe = 0.55, and rO = 1.35, respectively. The computed tolerance factors of the Pr2EuCoO6 and Pr2EuFeO6 materials are 0.923 and 0.920, respectively. The value range of the tolerance factor for materials stable in the cubic (Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m) space group is 0.9–1, which suggests that the computed tolerance of the materials lying in the stable crystallographic phase range confirms the stable crystallographic phase of the materials.60 The prediction of formation energy helps in understanding the thermodynamic stability of materials.61,62 The formation energy ΔHF of the Pr2EuMO6 (M = Co, Fe) materials can be computed by employing the following relation:
m) space group is 0.9–1, which suggests that the computed tolerance of the materials lying in the stable crystallographic phase range confirms the stable crystallographic phase of the materials.60 The prediction of formation energy helps in understanding the thermodynamic stability of materials.61,62 The formation energy ΔHF of the Pr2EuMO6 (M = Co, Fe) materials can be computed by employing the following relation:| |
 | (9) |
 |
| | Fig. 1 Crystallographic structures of the Pr2EuMO6 (M = Co, Fe) perovskite materials. | |
The relation contains the total energy of the material, ET, and the energies of the constituent elements EPr, EEu, EM = Co & Fe and EO for Pr, Eu, (M = Co & Fe) and O elements, respectively. These energies are obtained by performing energy vs. volume optimizations of both materials in the most stable magnetic phase (FM). The computed formation energies per atom (in eV) of the materials Pr2EuCoO6 and Pr2EuFeO6 are −7.42 and −3.37, respectively. The negative formation enthalpy indicates the thermodynamic stability of the materials under study.25
3.1.1 Phonon stability. The phonon dispersion curves of Pr2EuCoO6 and Pr2EuFeO6, shown in Fig. 2(a) and (b), respectively, provide direct insight into their dynamical stability. In both compounds, all phonon branches remain positive throughout the high-symmetry Brillouin zone path (W–L–Γ–X–W–K), with no imaginary frequencies observed. This absence of soft modes indicates that both structures are dynamically stable in their optimized configurations. Notably, the acoustic branches smoothly approach zero frequency at the Γ-point, as expected from translational invariance, further confirming the numerical robustness of the calculations.
 |
| | Fig. 2 Phonon dispersion relations of (a) Pr2EuCoO6 and (b) Pr2EuFeO6 along the high-symmetry path, W–L–Γ–X–W–K, in the Brillouin zone. The absence of imaginary frequencies (indicated by the dashed horizontal line at 0 THz) confirms the dynamical stability of both compounds. The phonon branches are shown in black for Pr2EuCoO6 and in red for Pr2EuFeO6. | |
A comparison between the two systems reveals subtle differences in the optical phonon region. Pr2EuCoO6 exhibits a broader distribution of high-frequency modes extending beyond ∼10 THz, while Pr2EuFeO6 shows a relatively compressed spectrum with maximum frequencies below ∼8 THz. This shift can be attributed to differences in the mass and bonding characteristics of Co–O and Fe–O octahedra, where stronger bonding interactions in the Co-based system lead to higher vibrational frequencies.
3.2 Electronic properties
The cubic phase of the double perovskite oxide (DPO) material Pr2EuMO6 (M = Co and Fe) and its magneto-electronic characteristics have been studied. The spin-polarized band structures (BSs) of Pr2EuCoO6 and Pr2EuFeO6 are calculated within the energy spectrum of −4 to 4 eV on the Y-axis versus the high symmetric directions W, L, Γ, X, W, and K of the first Brillouin zone, as depicted in Fig. 3. According to the BS plots, both compounds have identical electronic band topologies, suggesting the same electronic activity within the studied energy spectrum. The plots suggest that both materials, Pr2EuCoO6 and Pr2EuFeO6, exhibit metallic characteristics in up-spin configurations, as the valence band (VB) energy levels cross the Fermi level, facilitating free electron motion. On the other hand, the down spin state of both materials demonstrates semiconducting behavior, with the conduction band minimum (CBM) and valence band maximum (VBM) prominently distinguished by forbidden energy gaps. Near the X symmetrical path, this shortest partition generates a straight band gap, highlighting the compound's versatility for spin-dependent purposes. The determined energy band gap values in spin down for Pr2EuCoO6 and Pr2EuFeO6 are 1.2 eV and 3.0 eV, respectively. The band gaps indicate their appropriateness for usage, whereby adjustable electrical characteristics are beneficial. These materials with a metallic nature in the spin-up and semiconducting in the spin-down channels are known to be half-metallic.63 The BS of both materials suggests that the overall nature of these materials is half-metallic. Half-metallic materials have attracted considerable interest in recent years because of their distinctive electronic structure, displaying metallic characteristics in one spin direction while exhibiting semiconducting properties in the other.64,65 Their ability to produce a fully spin-polarized current makes them perfect for use in spintronic applications. The distinct characteristics of half-metals have prompted studies into their magnetoresistance and possible uses in electronic devices.66 The contribution of material elements and electronic states to the conductivity and electronic nature of materials is explained by the density of states (DOS) analysis. The elemental contributions are given by the total density of states (TDOS), while the electronic state contribution is given by the partial density of states (PDOS), as displayed in Fig. 4(a and b). The DOS analysis of both materials provides more insight into the magnetic and conducting characteristics of the materials by highlighting their unique spin-polarized electronic structures. The plots suggest that the valence band in the up spin is formed from 0 to −5 eV, and the conduction band from 0.5 to 6 eV energy ranges. Additionally, in the down spin configurations, the valence band of Pr2EuCoO6 (see Fig. 4(a)) is from −0.5 to −5 eV, the conduction band is from 1.5 to 6 eV, the valence band of Pr2EuFeO6 (see Fig. 4(b)) is from 0.8 to 5 eV, and the conduction band ranges from 1 eV to 6 eV. The DOS plots follow the energy band gap explanations of materials by electronic structure analysis. The electronic structures in Pr2EuCoO6 and Pr2EuFeO6, which are mostly provided by the states of the Pr element, and the PDOS indicate that the Pr-f state overlaps the Fermi level in up spin configurations, which confirms the metallic nature of the materials. The valence band maximum and conduction band minimum of Pr2EuCoO6 and Pr2EuFeO6 in the up spin are made up of the Pr-f state. Similarly, in the down spin configuration, the valence band maximum and conduction band minimum of Pr2EuCoO6 material are formed by the Co-d state; the valence band maximum of Pr2EuFeO6 is developed by the O-p state, and the conduction band minimum is due to Fe-d states. The half-metallic nature of the materials is confirmed by the DOS plots. The DOS graphs additionally demonstrate significant hybridization involving the transition metals of Pr2EuCoO6, including the hybridization among Eu-f and Co-d (in up-spins) and Co-d and O-p states (in down-spins). The hybridizations also take place between the Eu-f and O-p states (in up-spins) of the Pr2EuFeO6 material. Both Pr2EuCoO6 and Pr2EuFeO6 exhibit half-metallic properties, attaining 100% spin polarization, resulting from the strong metallicity in the majority spin channel and the semiconducting function in the minority spin channel. This special characteristic raises the possibility that these materials might be used as efficient spin injectors in spintronic products, whereby information transportation and storage depend on the ability to manipulate the electron spin.
 |
| | Fig. 3 Spin-polarized electronic band structures of the (a) Pr2EuCoO6 and (b) Pr2EuFeO6 perovskites. | |
 |
| | Fig. 4 Spin-polarized total and partial density of states of the (a) Pr2EuCoO6 and (b) Pr2EuFeO6 perovskites. | |
3.3 Optical properties
A useful technique for figuring out a material's band structures and the response of the materials to the incident light is the analysis of the optical properties. The most significant aspect of the solid crystal's optical characteristics is its complex dielectric function, which is closely connected to the BS of crystals and consists of two parts: real and imaginary parts. The optical characteristics parameters are investigated in the photon energy range of 0–14 eV to study optical transitions. The computed real and imaginary parts of the materials in both spin configurations are displayed in Fig. 5(a–d).
 |
| | Fig. 5 Real ε1(ω) and imaginary ε2(ω) parts of the dielectric function ε(ω) of (a and b) Pr2EuCoO6 and (c and d) Pr2EuFeO6 in both spin configurations. | |
The real parts indicate the polarization of materials in response to incident electromagnetic radiation. This also indicates energy storage by materials in electric fields.67 The value of ε1(ω) at 0 eV, which is known as the static value ε1(0), is depicted in Fig. 5(a); for Pr2EuCoO6, the ε1(0) values are 45.0 (up-spin) and 4.5 (down-spin) states, while for Pr2EuFeO6, as illustrated in Fig. 5(c), these values are 137.5 (up-spin) and 12.0 (down-spin) states. The higher values in the up-spin configurations indicate and confirm the metallic nature, and the small values represent the semiconducting behavior of the materials, as depicted in the band structures (BSs) of the materials (see Fig. 3). This also confirms the spin-polarized electronic nature (half-metallic) of the materials. The ε1(0) higher value of Pr2EuFeO6 than Pr2EuCoO6 also indicates stronger hybridization of the Fe-d and O-p states than the Co-d and O-p states. The highest values of ε1(ω) for Pr2EuCoO6 are located in the visible region of the optical spectrum at energies of 0.42 eV (up spin) and 1.9 eV (down spin), with negligible values in the higher optical regions. The imaginary part ε2(ω) of the dielectric function, depicted in Fig. 5(b and d), expresses energy absorption or dissipation by the materials. The peaks in the imaginary parts express the intra-band electronic transitions. The plot suggests that the ε2(ω) values for Pr2EuCoO6, depicted in Fig. 5(b), reach their highest values at low energies in the up spin configurations and reach a threshold of 1.78 eV in the down spin configurations. The values of ε2(ω) for Pr2EuFeO6, shown in Fig. 5(d), reach their highest values at low energies in the up spin configurations and reach a threshold of 1.1 eV in the down spin configurations. The plots indicate certain absorptions at 24 eV for both materials and suggest negligible absorption at higher energies. The highest value of ε2(ω) at low energies indicates the metallic nature in the up spin, and threshold values in the down spin indicate the semiconducting nature of the materials, supporting the half-metallic electronic nature of the materials.68 The other computed optical parameters, including the absorption, energy loss, refractive index, and reflectivity of both materials, in an up-spin configuration, are displayed only in Fig. 6(a–d). The absorption coefficient (I(ω)) directly quantifies the optical power absorbed by the material. A high I(ω) at a specific photon energy signifies an intense interaction with light, along with effective energy dissipation or conversion within that spectral range. The absorption of energy by the materials can be understood by studying the absorption coefficient of the materials, as depicted in Fig. 6(a). The absorption coefficient I(ω) is a crucial metric for characterizing any optoelectronic compound.69,70 The absorption of the materials indicates that the absorption of incident radiation starts from the visible region, increases with an increase in incident radiation energy and reaches its maximum value at 21.8 eV for Pr2EuCoO6 and 22.5 eV for Pr2EuFeO6 material. All the absorptive peaks indicate intra-band electronic transitions. The maximum absorptive peak for both materials indicates the electronic transition from the Eu-f to Pr-f states. L(ω) represents the energy loss function, an important parameter that demonstrates the loss of energy from materials, as displayed in Fig. 6(b). The results depict that the energy loss by the materials is small in the visible energy range of the spectrum, increases with the increase in incident radiation energy and reaches the highest value at 22.7 eV energy. The refractive index n(ω) explains the dispersive nature of the materials and is plotted in Fig. 6(c). The static n(0) values are 13 for Pr2EuFeO6 and 7.5 for Pr2EuCoO6. The maximum dispersion of the incident radiation takes place in the visible region. The surface morphology can be explained by the study of the reflectivity coefficient of the materials, as displayed in Fig. 6(d). The static reflectivity R(0) has values of 0.65 (65%) and 0.78 (78%) for Pr2EuCoO6 and Pr2EuFeO6, respectively. The higher percentage of reflectivity under low static conditions in up spin confirms the metallic nature of the materials. The maximum reflectivity peaks indicate the plasmon resonance in the specified energy ranges of the spectrum. The optical parameters and electronic nature of the materials suggest that they are promising candidates for spintronic, photovoltaic, and solar cell applications.
 |
| | Fig. 6 (a) Absorption coefficient I(ω), (b) energy loss L(ω), (c) refractive index n(ω), and (d) reflectivity spectra R(ω) of the Pr2EuMO6 (M = Co and Fe) perovskites. | |
3.4 Magnetic nature
The magnetic properties of these double perovskites are crucial for magneto-optic applications, data storage systems, and spintronics because of their many magnetic properties, including ferromagnetism, ferrimagnetism, and anti-ferromagnetism.71 Materials with transitions and lanthanide constituent elements, having unpaired electrons in the d or f orbital, show magnetic behavior.72,73 The unpaired electrons in the outermost orbit of the material's element create magnetic moments that lead to the magnetic behavior of the material. To understand the magnetic strength of the Pr2EuMO6 (M = Co and Fe) materials, first-principles spin-based computations are performed. The computed magnetic moments (in µB) of the materials under study are displayed in Table 1. The table displays the magnetic moment due to interstitial sites (MMint), due to Pr (MMPr), due to Eu (MMEu), due to M = Co and Fe (MMM), and due to O (MMO), and the total magnetic moment of the material is (MMtotal). The total magnetic moments of the Pr2EuCoO6 and Pr2EuFeO6 are 11 (µB) and 14 (µB), respectively. The integral value of the total magnetic moments indicates the ferromagnetic nature of the materials.74 The larger value of the total magnetic moment of Pr2EuFeO6 indicates strong coupling between Eu and Fe compared to Eu and Co in the Pr2EuCoO6 material. The stronger coupling of Fe compared to Co suggests a stronger intrinsic magnetic moment of Fe than of Co. The table displays that the major contribution to the total magnetic moment of both materials is due to unpaired electrons of Pr, Eu, and Fe (only for Pr2EuFeO6), while other elements, including Co (for Pr2EuCoO6), have negligible contributions. The small contribution of the Co magnetic moment may be due to the lower spin state adopted by the unpaired electrons of the element. The negative magnetic moment of Co indicates antiferromagnetic alignment with the Pr and Eu spin configurations. Antiparallel alignment may arise from super-exchange interactions facilitated by oxygen atoms within the perovskite structure. The parallel alignment of Fe with Pr and Eu atom spins in Pr2EuFeO6 causes a higher value of the total magnetic moment than the Pr2EuCoO6 material's total magnetic moment. Furthermore, the magnetic nature of the materials can be confirmed using volume optimization analysis in different magnetic phases, including ferromagnetic (FM) and anti-ferromagnetic (AFM) phases. The computed optimization curves of both materials in the magnetic phases are plotted in Fig. 7(a and b). The optimization plots demonstrate that the ferromagnetic phase of both materials is the most stable magnetic phase among the other phases, which predicts the ferromagnetic nature of both materials. Table 2 illustrates the analytical analysis of the FM and AFM stability margin ΔE (in Ry and meV). The positive values of ΔE indicate and confirm the FM nature of the materials. Ferromagnetic materials have a large number of potential applications. Ferromagnetic materials offer a wide range of uses across different industries. They are utilized in magnetic data storage, transformer cores, electrical generators, and permanent magnets.75 In technologies such as data storage systems, refrigeration, and medical research, these materials are useful because they display special characteristics when subjected to external magnetic fields.76 New developments in ferromagnetic materials have expanded the potential uses of magneto-optical, magneto-resistive, magnetostrictive, and nanocrystalline soft magnetic materials.75
Table 1 Magnetic moments (µB) of the Pr2EuMO6 (M = Co and Fe) materials
| Materials |
MMint (µB) |
MMPr (µB) |
MMEu (µB) |
MMM (µB) |
MMO (µB) |
MMtotal (µB) |
| Pr2EuCoO6 |
0.30606 |
2.42867 |
6.24646 |
−0.04431 |
−0.06980 |
11 |
| Pr2EuFeO6 |
0.50719 |
2.26215 |
6.10179 |
4.05157 |
−0.20723 |
14 |
 |
| | Fig. 7 Optimization curves in the ferromagnetic (FM) and anti-ferromagnetic (AFM) magnetic phases of the (a) Pr2EuCoO6 and (b) Pr2EuFeO6 perovskites. | |
Table 2 Magnetic ground state verification: total energy and stability margin ΔE (in Ry and meV) for the FM and AFM phases of the Pr2EuMO6 (M = Co and Fe) materials
| Materials |
EFM (Ry) |
EAFM (Ry) |
ΔE (Ry) = EFM − EAFM |
ΔE (meV) = EFM − EAFM |
Magnetic phase |
| Pr2EuCoO6 |
−62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 366.804 366.804 |
−62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 366.752 366.752 |
0.052 |
+707.49 |
FM |
| Pr2EuFeO6 |
−62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 368.460 368.460 |
−62![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 368.170 368.170 |
0.290 |
+3945.65 |
FM |
3.5 Thermoelectric properties
The substantial rise in fossil fuel consumption due to recent global economic expansion has led to severe energy crises and considerable degradation of the environment. Excess heat can be transformed into electricity utilizing thermoelectric materials to mitigate energy crises and diminish hazardous emissions. The favorable thermoelectric properties of perovskite compounds, namely their elevated Seebeck coefficient and high carrier concentration, have garnered significant interest.77 Thermoelectric parameters (in the spin-down channel), including electrical conductivity, Seebeck coefficients, electronic thermal conductivity, specific heat capacity Cv, the power factor (PF), and ZT, are computed using the BoltzTraP computational code. The thermoelectric parameters are displayed in Fig. 8(a–f). The electrical conductivities of both materials are computed from 200 to 900 K, as displayed in Fig. 8(a), which shows the conductivity of electrons by the materials caused by the applied temperature gradient. The plots demonstrate the increasing trend of the electrical conductivity of the materials with temperature, which is a characteristic of semiconducting materials whose carrier mobility increases with temperature. Pr2EuFeO6 exhibits smaller electrical conductivity compared to Pr2EuCoO6 across the temperature range. The electrical conductivity of Pr2EuFeO6 increases slightly beyond 800 K, while Pr2EuCoO6 has an increasing electrical conductivity beyond 450 K of temperature. The electrical conductivity is strongly influenced by the energy band gaps of the materials; Pr2EuCoO6 has a smaller energy band gap (see Fig. 3(a and b)) compared to Pr2EuFeO6, resulting in higher electrical conductivity for Pr2EuCoO6 across the temperature range. The Seebeck coefficient is an important parameter that measures the voltage generated across the ends of materials by employing a temperature gradient across the ends. The computed Seebeck coefficient (S) for both materials is shown in Fig. 8(b). The plots display different behaviors for both materials with an increase in temperature. The Seebeck coefficient of Pr2EuFeO6 is −2.7 mV K−1 at 200 K and increases gradually to −0.8 mV K−1 at 800 K, while that of Pr2EuCoO6 is 1.7 mV K−1 at 200 K and decreases gradually to 0.20 mV K−1 at 800 K. In semiconducting materials, the Seebeck coefficient is an essential indicator of the type of carrier. The decreasing trend in the Seebeck coefficient of Pr2EuCoO6 indicates the n-type, and the increasing trend of Pr2EuFeO6 indicates the p-type nature of the materials.78 The thermal conductivity of the materials is determined by lattice vibrations and electronic movements. The present study focuses on electronic thermal conductivity, as the computation of lattice thermal conductivity requires more computational resources. The electronic thermoelectric conductivity of the materials is displayed in Fig. 8(c) and computed from the 200–900 K temperature range. Electronic thermal conductivity demonstrates the heat flow through materials via free electrons or holes. The thermal conductivity for both materials shows a similar trend of rise with temperature. The thermal conductivities of Pr2EuCoO6 and Pr2EuFeO6 start to increase beyond 450 K and 600 K, respectively. The thermal conductivity of Pr2EuCoO6 is significantly higher than that of Pr2EuFeO6 across the temperature range. The thermal conductivity of Pr2EuCoO6 is 0.21 × 1013 W (mKs)−1 at 450 K and rises to 2.75 × 1013 W (mKs)−1 at 800 K. Similarly, the thermal conductivity for Pr2EuCoO6 is 0.21 × 1013 W (mKs)−1 at 600 K and 0.3 × 1013 W (mKs)−1 at 800 K. The higher electronic thermal conductivity of Pr2EuCoO6 compared to that of Pr2EuFeO6 is due to the higher charge carrier concentration of Pr2EuCoO6. The specific heat capacity Cv at a constant volume for both materials, displayed in Fig. 8(d), has a similar nature of variation with temperature rise, which is the measure of heat required to increase the temperature of a material by 1 K. The specific heat capacity Cv, though thermodynamic, is included here due to its role in evaluating lattice thermal conductivity, which directly affects thermoelectric performance. The specific heat capacity Cv for Pr2EuCoO6 is very small up to 450 K and increases to 1.02 J mol−1 K−1 at 800 K, and that for Pr2EuFeO6 also has a small value up to 650 K and rises to 0.05 J mol−1 K−1 at 800 K. The maximum value of Cv for Pr2EuCoO6 is higher than that of Pr2EuFeO6. The elevated maximum Cv of Pr2EuCoO6 may be ascribed to its greater atomic mass, distinct bonding properties, or expanded vibrational spectrum. The performance of thermoelectric materials is commonly assessed by their power factor (PF), defined as the product of the square of the Seebeck coefficient (S) and electrical conductivity (σ).79 Fig. 8(e) displays the computed power factor (PF) of both materials. The plots indicate that the PF of Pr2EuCoO6 is higher than that of Pr2EuFeO6 across the temperature range. The PF of Pr2EuCoO6 starts increasing beyond 350 K and attains a value of 3.5 × 109 W mk−2 s−1 at 800 K, while the PF of Pr2EuFeO6 starts increasing beyond 600 K and reaches 0.8 × 109 W mk−2 s−1 at 800 K. The elevated power factor of Pr2EuCoO6 at higher temperatures suggests outstanding thermoelectric performance, which is attributed to the beneficial interplay between the Seebeck coefficient and electrical conductivity. The rise in the PF of both materials as the temperature increases is due to thermally activated carriers. The ZT value, also known as the figure of merit, serves as a dimensionless indicator of how effectively a thermoelectric material can convert heat into electricity. Fig. 8(f) displays the computed ZT value of both materials. The plots suggest higher values of ZT for both materials at room temperature. The ZT values of Pr2EuCoO6 and Pr2EuFeO6 at room temperature (300 K) are 1 and 0.6, respectively. The ZT values of both materials gradually decrease with an increase in temperature and reach 0.7 for Pr2EuCoO6 and 0.1 for Pr2EuFeO6 at 800 K. The temperature-dependent ZT value of the materials depends on the electrical conductivity (σ), the Seebeck coefficient (S), and the thermal conductivity (κ) of the materials. The different behaviors of Pr2EuCoO6 and Pr2EuFeO6 are due to their distinct electronic and thermal properties. The ZT suggests that both materials show excellent thermometric performance across the temperature range. These materials have a large number of thermoelectric applications, including temperature sensors,80 thermal energy harvesters,81 heat capacity-based thermometry,82 and thin-film infrared (IR) sensors.83
 |
| | Fig. 8 Thermoelectric parameters of Pr2EuMO6 (M = Co and Fe): (a) electrical conductivity (σ/τ), (b) Seebeck coefficient (S), (c) electronic thermal conductivity (κe), (d) specific heat capacity at constant volume Cv, (e) power factor (PF) and (f) ZT. | |
4 Conclusion
In summary, this study is carried out using the Wein2K package based on DFT and utilizing mBJ+U approximations to achieve ground state characteristics of Pr2EuMO6 (M = Co and Fe) perovskites. The structural, magnetic, optoelectronic, and thermoelectric properties of the materials are computed to explore their utilization in different technological applications. The electronic properties suggest that the materials have metallic, up-spin, and semiconducting natures in the down-spin configuration, predicting the half-metallic nature of both materials. Half-metallic materials have a large number of applications, specifically in spintronics. The magnetic nature of the materials is analyzed by performing spin-based computations. The computations predict the magnetic moment of the materials. The computed total magnetic moments of the materials Pr2EuCoO6 and Pr2EuFeO6 are 11 (µB) and 14 (µB), respectively. The integral value of the total magnetic moment indicates the ferromagnetic nature of both materials, which is verified by achieving the most stable volume optimization curve in FM compared to the AFM magnetic phase. The optical characteristics of the materials, including parts of the complex dielectric function, absorption, energy loss, refractive index, and reflectivity, are computed to understand their photonic nature for many potential applications. The study of optical parameters suggests that materials are good choices for photovoltaic and solar cell applications. Thermometric parameters of the materials are calculated (in spin-down) using the BoltzTraP code. Pr2EuCoO6 has higher electrical and electronic thermal conductivities compared to those of the Pr2EuFeO6 material at higher temperatures. The Seebeck coefficient of the materials suggests that Pr2EuCoO6 has an n-type semiconducting nature and Pr2EuFeO6 has a p-type semiconducting nature. The power factor (PF) of both materials is lower at room temperature and increases linearly with an increase in temperature. The PF of Pr2EuCoO6 is comparatively higher than that of Pr2EuFeO6 at higher temperatures. The ZT Pr2EuCoO6 is higher, with a value of 1.0 at room temperature (300 K) and decreases to 0.7 at 800 K, while Pr2EuFeO6 has a ZT value of 0.6 at room temperature and decreases to 0.1 at 800 K. The thermometric study suggests that Pr2EuCoO6 has a higher thermoelectric performance compared to Pr2EuFeO6 across the temperature range. The results highlight the future potential of Pr2EuCoO6 and Pr2EuFeO6 as viable options for spintronic and thermoelectric applications, attributed to their semiconducting characteristics and moderate thermoelectric performance.
Author contributions
Ahmad Ali: supervision, conceptualization, investigations, formal analysis, data curation, writing – original draft, and writing – review and editing. Gulzar Khan: software, investigation, data curation, formal analysis, writing – review & editing, and writing – original draft. Tania Gul: formal analysis, data curation, conceptualization, visualization, and writing – original draft. Fareha: investigations, formal analysis, data curation, conceptualization, validation, and writing – original draft. Sikander Azam: methodology, conceptualization, investigations, formal analysis, data curation, writing – original draft, and writing – review and editing. Osama Oqilat: conceptualization, investigations, methodology, writing – review and editing, and writing – original draft. Hijaz Ahmad: conceptualization, formal analysis, data curation, writing – review & editing, and writing – original draft.
Conflicts of interest
The authors declare that there are no conflicts of interest regarding the publication of this paper. All authors have contributed to this work according to the academic and research standards, and there are no competing interests, financial or otherwise, that could have influenced the outcomes of this study.
Data availability
The datasets generated and/or analyzed during this study are available from the corresponding author upon reasonable request.
Acknowledgements
This publication was supported by the project Quantum materials for applications in sustainable technologies (QM4ST), funded as project No. CZ.02.01.01/00/22_008/0004572 by the Programme Johannes Amos Comenius, call Excellent Research.
References
- S. Ahmad Dar, V. Srivastava, U. Kumar Sakalle, V. Parey and G. Pagare, Mater. Sci. Eng., B, 2018, 236–237, 217–224 CrossRef CAS.
- F. I. H. Alias, M. H. Ridzwan, M. K. Yaakob, C. W. Loy and Z. Mohamed, J. Mater. Res. Technol., 2022, 18, 1623–1630 CrossRef CAS.
- R. Sivasamy, P. Venugopal and R. Espinoza-González, Mater. Today Commun., 2020, 25, 101603 CrossRef CAS.
- M. Kumar, A. Raj, A. Kumar, S. Sharma, H. Bherwani, A. Gupta and A. Anshul, Optik, 2021, 242, 166764 CrossRef CAS.
- S. Haid, W. Benstaali, A. Abbad, B. Bouadjemi, S. Bentata and Z. Aziz, Mater. Sci. Eng., B, 2019, 245, 68–74 CrossRef CAS.
- S. Belhachi, S. Al-Qaisi, S. Samah, H. Rached, A. Zaman, T. A. Alrebdi, A. Boutramine, N. Erum, R. Ahmed and A. S. Verma, J. Inorg. Organomet. Polym. Mater., 2024, 35, 978–993 CrossRef.
- N. A. Azhar, I. S. Ismail, N. B. Mohamed, A. Hashim and Z. Mohamed, Crystals, 2020, 10, 521 CrossRef CAS.
- A. Khan, S. Chatterjee, T. K. Nath and A. Taraphder, Phys. Rev. B, 2021, 104, 035133 CrossRef.
- K. Yi, Q. Tang, Z. Wu and X. Zhu, Nanomaterials, 2022, 12, 979 CrossRef CAS PubMed.
- H. Khan, G. Murtaza, A. Ayyaz, M. B. Shakir, M. Touqir, K. Iqbal, A. Usman, S. Mumtaz and I. M. Moussa, Mater. Sci. Semicond. Process., 2024, 185, 108911 CrossRef.
- A. W. Sleight and R. Ward, J. Am. Chem. Soc., 1961, 83, 1088–1090 CrossRef CAS.
- F. S. Galasso, F. C. Douglas and R. J. Kasper, J. Chem. Phys., 1966, 44, 1672–1674 CrossRef CAS.
- M. Anderson, K. Greenwood, G. Taylor and K. Poeppelmeier, Prog. Solid State Chem., 1993, 22, 197–233 CrossRef CAS.
- M. Caid, D. Rached, Y. Rached and H. Rached, Phys. B, 2024, 677, 415742 CrossRef CAS.
- K. Assiouan, A. Marjaoui, J. El Khamkhami, M. Zanouni, H. Ziani, A. Bouchrit and A. Achahbar, J. Phys. Chem. Solids, 2024, 188, 111890 CrossRef CAS.
- Q. Mahmood, B. U. Haq, M. Yaseen, S. M. Ramay, M. G. B. Ashiq and A. Mahmood, Solid State Commun., 2019, 292, 17–23 CrossRef CAS.
- M. Waqas Iqbal, M. Manzoor, N. A. Noor, I. Rehman, N. Mushahid, S. Aftab, Y. M. Alanazi, H. Ullah and A. Muhammad Afzal, Sol. Energy, 2022, 239, 234–241 CrossRef CAS.
- S. Berri, Emerg. Mater., 2022, 5, 1849–1857 CrossRef CAS.
- A. Ali, M. Salman Khan, G. Khan, B. Gul, A. M Tawfeek, S. Azam, F. Abbas, S. Zulfiqar, S. A. Khattak, T. Khan and S. K. Shah, J. Solid State Chem., 2023, 326, 124211 CrossRef CAS.
- P. Kayser, S. Injac, B. J. Kennedy, T. Vogt, M. Avdeev, H. E. Maynard-Casely and Z. Zhang, Inorg. Chem., 2017, 56, 6565–6575 CrossRef CAS PubMed.
- T. Ahmed, A. Chen, D. A. Yarotski, S. A. Trugman, Q. Jia and J.-X. Zhu, APL Mater., 2016, 5, 035601 CrossRef.
- M. Y. Sofi, M. S. Khan and M. A. Khan, Mater. Adv., 2024, 5, 4913–4931 RSC.
- M. D. I. Bhuyan, R. Hossain, F. Ara and M. A. Basith, Phys. Chem. Chem. Phys., 2022, 24, 1569–1579 RSC.
- M. Caid, D. Rached, Y. Rached and H. Rached, Opt. Quant. Electron., 2024, 56, 980 CrossRef CAS.
- A. Ali, Q. Rafiq, G. Khan, S. S. Hayat, S. Azam, M. Aamer and M. Amina, Chem. Pap., 2024, 79, 1137–1153 CrossRef.
- A. Ali, B. Gul, M. S. Khan, G. Khan, S. Azam, A. A. Ifseisi, G. Wafa and F. Abbas, Chem. Phys. Lett., 2024, 841, 141168 CrossRef CAS.
- Z. Ullah, B. Gul, A. Salah Mohamed, A. Ali, G. Khan, M. Salman Khan and F. Abbas, Chem. Phys. Lett., 2024, 850, 141471 CrossRef CAS.
- D. Mazumdar and I. Das, Phys. Chem. Chem. Phys., 2021, 23, 5596–5606 RSC.
- M. Dhilip, S. Rameshkumar, R. K. Raji, T. Ramachandran, J. S. Punitha, F. R. M. Sundar Raj, K. S. Kumar, V. Anbarasu, N. Sekar, R. Chinnathambi and A. A. Ghfar, Mater. Today Commun., 2024, 38, 108120 CrossRef CAS.
- S. Berri, J. Magn. Magn. Mater., 2015, 385, 124–128 CrossRef CAS.
- S. Berri, Acta Phys. Pol., A, 2020, 138, 834–837 CrossRef CAS.
- S. Berri, N. Bouarissa and M. Attallah, J. Supercond. Novel Magn., 2020, 33, 17S Search PubMed.
- S. Berri, Comput. Condens. Matter, 2021, 28, e00586 CrossRef.
- P. Blaha, K. Schwarz, P. Sorantin and S. B. Trickey, Comput. Phys. Commun., 1990, 59, 399–415 CrossRef CAS.
- W. Kohn and L. J. Sham, Phys. Rev., 1965, 140, A1133–A1138 CrossRef.
- M. C. Payne, M. P. Teter, D. C. Allan, T. A. Arias and J. D. Joannopoulos, Rev. Mod. Phys., 1992, 64, 1045–1097 CrossRef CAS.
- F. Tran and P. Blaha, Phys. Rev. Lett., 2009, 102, 226401 CrossRef PubMed.
- S. L. Dudarev, G. A. Botton, S. Y. Savrasov, C. J. Humphreys and A. P. Sutton, Phys. Rev. B: Condens. Matter Mater. Phys., 1998, 57, 1505–1509 CrossRef CAS.
- M. Rezaiguia, W. Benstaali, A. Abbad, S. Bentata and B. Bouhafs, J. Supercond. Novel Magn., 2017, 30, 2581–2590 CrossRef CAS.
- J. Kunes, W. Ku and W. E Pickett, J. Phys. Soc. Jpn., 2004, 74, 1408–1411 CrossRef.
- A. Cadi-Essadek, A. Roldan, D. Santos-Carballal, P. E. Ngoepe, M. Claeys and N. H. De Leeuw, S. Afr. J. Chem., 2021, 74, 12–21 CrossRef.
- J. K. Shenton, D. R. Bowler and W. L. Cheah, J. Phys.: Condens. Matter, 2017, 29, 445501 CrossRef PubMed.
- R. Shinde, S. S. R. K. C. Yamijala and B. M. Wong, J. Phys.: Condens. Matter, 2020, 33, 115501 CrossRef PubMed.
- B. G. Janesko, Chem. Soc. Rev., 2021, 50, 8470–8495 RSC.
- H. J. Monkhorst and J. D. Pack, Phys. Rev. B: Condens. Matter Mater. Phys., 1976, 13, 5188–5192 CrossRef.
- A. D. Becke and M. R. Roussel, Phys. Rev. A, 1989, 39, 3761–3767 CrossRef CAS PubMed.
- Y. B. Idriss, M. Houari, B. Bouadjemi, M. Matougui, T. Lantri, S. Haid, A. Zitouni, M. Boudjelal, S. Bentata, Z. Aziz, S. Bin-Omran and R. Khenata, Opt. Quant. Electron., 2024, 57, 111 Search PubMed.
- A. Guesmia, M. C. Terkhi, I. Y. Bouchentouf, M. Houari, B. Bouadjemi, A. Zitouni, M. Matougui, S. Haid, T. Lantri, B. Achour, S. Mesbah, A. Khatar and S. Bentata, Indian J. Phys., 2024, 99, 2241–2255 CrossRef.
- M. H. Cherif, M. C. Terkhi, L. Beldi, M. Houari, B. Bouadjemi, S. Haid, M. Matougui, T. Lantri, S. Bentata, S. Mesbah and B. Bouhafs, Opt. Quant. Electron., 2024, 56, 1083 CrossRef.
- B. Bouadjemi, S. Bentata, A. Abbad, W. Benstaali and B. Bouhafs, Solid State Commun., 2013, 168, 6–10 CrossRef CAS.
- S. Ali, H. S. Ali, K. Ismail, A. R. Iftikhar, H. Ali and H. H. Raza, Opt. Quant. Electron., 2024, 56, 730 CrossRef.
- H. A. Kramers, Atti del Congresso Internazionale dei Fisici, 1927, 2, p. 10029437327 Search PubMed.
- P. Pichanusakorn and P. R. Bandaru, Appl. Phys. Lett., 2009, 94, 223108 CrossRef.
- A. L. Allred, J. Inorg. Nucl. Chem., 1961, 17, 215–221 CrossRef CAS.
- N. N. Greenwood and A. Earnshaw, Chemistry of the Elements, Butterworth-Heinemann, Oxford, 2nd edn, 1997 Search PubMed.
- D. I. Khomskii, Transition Metal Compounds, Cambridge University Press, Cambridge, 2014 Search PubMed.
- H. D. Megaw, Crystal Structures: A Working Approach, W. B. Saunders Co., Philadelphia, 1973 Search PubMed.
- V. M. Goldschmidt, Naturwissenschaften, 1926, 14, 477–485 CrossRef CAS.
- R. D. Shannon and C. T. Prewitt, Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem., 1970, 26, 1046–1048 CrossRef CAS.
- S. A. Khandy and D. C. Gupta, Int. J. Energy Res., 2021, 45, 13338–13354 CrossRef CAS.
- A. Agrawal, B. Meredig, C. Wolverton and A. Choudhary, A formation energy predictor for crystalline materials using ensemble data mining, 2016 IEEE 16th International Conference on Data Mining Workshops (ICDMW), 2016, pp. 1276–1279 Search PubMed.
- G. G. C. Peterson and J. Brgoch, J. Phys.: Energy, 2021, 3, 022002 CAS.
- L. Feng, E. K. Liu, W. X. Zhang, W. H. Wang and G. H. Wu, Stable Mn$_2$-based compensated ferrimagnetic Heusler compounds, Appl. Phys. Lett., 2014, 104, 012406 CrossRef.
- M. I. Katsnelson, V. Y. Irkhin, L. Chioncel, A. I. Lichtenstein and R. A. De Groot, Rev. Mod. Phys., 2008, 80, 315–378 CrossRef CAS.
- J. L. Wang, S. J. Campbell, J. M. Cadogan, A. J. Studer, R. Zeng and S. X. Dou, Presented in Part at the 34th Annual Condensed Matter and Materials Meeting, Waiheke Island, New Zealand, 2010 Search PubMed.
- X. Hu, Adv. Mater., 2011, 24, 294–298 CrossRef PubMed.
- R. R. Freeman, J. A. King and G. P. Lafyatis, Electromagnetic Radiation, Oxford University, 2019 Search PubMed.
- M. Dressel and G. Grüner, Choice Rev. Online, 2002, 40, 40–1598 Search PubMed.
- A. Bouhemadou, S. Al-Essa, D. Allali, M. A. Ghebouli and S. Bin-Omran, Solid State Sci., 2013, 20, 127–134 CrossRef CAS.
- O. Jani, C. Honsberg, A. Asghar, D. Nicol, I. Ferguson, S. Back, J. Parrish and S. Durbin, Proceedings IEEE Photovoltaic Specialists Conference, 2005, pp. 37–42 Search PubMed.
- S. Sidi Ahmed, M. Boujnah, L. Bahmad, A. Benyoussef and A. El Kenz, Chem. Phys. Lett., 2017, 685, 191–197 CrossRef CAS.
- B. R. Cooper, CRC Crit. Rev. Solid State Sci., 1972, 3, 83–129 CrossRef.
- S. Methfessel, IEEE Trans. Magn., 1965, 1, 144–155 CrossRef CAS.
- A. Ali, B. Gul, M. Salman Khan, G. Khan, A. Salah Mohamed and F. Abbas, J. Magn. Magn. Mater., 2024, 604, 172311 CrossRef CAS.
- H. J. Borg, Applications of Ferromagnetic and Optical Materials, Storage and Magnetoelectronics: Symposia Held April 16–20, 2001, San Francisco, California, U.S.A., Materials Research Society, 2001 Search PubMed.
- S. M. Abdelbasir and A. E. Shalan, Intriguing Properties and Applications of Functional Magnetic Materials, in Functional Materials, ed. D. Sahu, IntechOpen, London, 2019, ch. 4, pp. 63–80 Search PubMed.
- A. Ali, M. S. Khan, M. Irfan, G. Khan, B. Gul, S. Azam, H. Haider, A. Ullah, M. R. Karim, I. A. Alnaser and F. Abbas, Chem. Phys. Lett., 2023, 835, 141019 CrossRef.
- G. K. H. Madsen, J. Carrete and M. J. Verstraete, Comput. Phys. Commun., 2018, 231, 140–145 CrossRef CAS.
- A. Zakutayev, F. J. Luciano, V. P. Bollinger, A. K. Sigdel, P. F. Ndione, J. D. Perkins, J. J. Berry, P. A. Parilla and D. S. Ginley, Rev. Sci. Instrum., 2013, 84, 053905 CrossRef PubMed.
- D. M. Rowe, Thermoelectrics Handbook: Macro to Nano, CRC Press, 2005 Search PubMed.
- G. J. Snyder and E. S. Toberer, Nat. Mater., 2008, 7, 105–114 CrossRef CAS PubMed.
- Z. Gao, M. Reali, A. Yelon and C. Santato, Mater. Adv., 2022, 3, 7815–7823 RSC.
- H. J. Goldsmid, Basic Principles of Thermoelectrics, in Introduction to Thermoelectrics, Springer, Berlin, Heidelberg, 2nd edn, 2016, ch. 2, pp. 9–24 Search PubMed.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a,
Gulzar Khanb,
Tania Gulb,
Sikander Azam
*a,
Gulzar Khanb,
Tania Gulb,
Sikander Azam *cd,
Farehab,
Osama Oqilate and
Hijaz Ahmad*fgh
*cd,
Farehab,
Osama Oqilate and
Hijaz Ahmad*fgh
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m) space group. The study has made the materials promising in spintronic applications due to their half-metallic nature. Magnetic analysis shows that the materials are ferromagnetic. The overall magnetic moments of Pr2EuCoO6 and Pr2EuFeO6 are 11 (µB) and 14 (µB), respectively. Optical analyses of the two materials are conducted in the 0 to 14 eV energy range. The optical parameter study indicates that the materials are excellent in terms of photovoltaic and high-energy absorbent applications. The Seebeck coefficient shows that Pr2EuCoO6 and Pr2EuFeO6 are n- and p-type semiconductors, respectively. The elevated values of ZT at room temperature suggest that both materials have good thermoelectric efficiency, and the rising values of PF with increasing temperature suggest that the two materials can be used in high-temperature thermoelectric applications.
m) space group. The study has made the materials promising in spintronic applications due to their half-metallic nature. Magnetic analysis shows that the materials are ferromagnetic. The overall magnetic moments of Pr2EuCoO6 and Pr2EuFeO6 are 11 (µB) and 14 (µB), respectively. Optical analyses of the two materials are conducted in the 0 to 14 eV energy range. The optical parameter study indicates that the materials are excellent in terms of photovoltaic and high-energy absorbent applications. The Seebeck coefficient shows that Pr2EuCoO6 and Pr2EuFeO6 are n- and p-type semiconductors, respectively. The elevated values of ZT at room temperature suggest that both materials have good thermoelectric efficiency, and the rising values of PF with increasing temperature suggest that the two materials can be used in high-temperature thermoelectric applications.![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m) space group.35,36 The WEIN2K code is selected to perform the structural, electronic, optical, and magnetic calculations of the materials with high accuracy, as it is based on the full potential method, which precisely incorporates the effects of core electrons, providing benefits over other DFT-based codes that utilize pseudopotentials. To account for both the precise band gap estimation and strong on-site Coulomb interactions in the localized d/f orbitals, electronic structure calculations were carried out using the modified Becke–Johnson (mBj) exchange potential in combination with the Hubbard U correction (mBj + U).37,38 The effective Hubbard U values are 6.0 eV for Pr-4f,39 6.5 eV for Eu-4f,40 3.5 eV for Co-3d,41 and 4.5 eV for Fe-3d orbitals.42 Although DFT + U, along with the TB-mBJ method, was used in the current study to consider the strong electron correlation in transition metal sites, it is acknowledged that the approach could still have a self-interaction error (SIE), which could negatively impact the accuracy of electronic property predictions. Other methods, including hybrid functionals, self-interaction-corrected (SIC) methods and other methods, have been demonstrated to provide better accuracy than standard DFT + U for systems containing transition metals.43,44 The wave function has been extended utilizing the sphere-shaped harmonic basis set as much as lmax = 10 across the not interconnected muffin-tin spheres. A plane-wave basis with a threshold value of RMT Kmax = 7 was implemented to extend the wave function throughout the interstitial region. The cut-off energy is chosen as −6.0 Ry to distinguish between the core and valence states. The incorporation of the Brillouin zone has been completed by employing a 10 × 10 × 10 k-mesh Monkhorst-Pack.45,46 In the present calculations, the energy and convergence criteria are considered whenever the charge difference is smaller than 0.001 e a.u.−3 per unit cell and the overall energy is steady within 0.0001 Ry. To avoid spurious oscillations and achieve higher quantitative accuracy, the optical characteristics were calculated using 15 × 15 × 15 k-points in the first Brillouin zone.47–50 To look into optical characteristics, the material's complex dielectric function is computed, as follows:51
m) space group.35,36 The WEIN2K code is selected to perform the structural, electronic, optical, and magnetic calculations of the materials with high accuracy, as it is based on the full potential method, which precisely incorporates the effects of core electrons, providing benefits over other DFT-based codes that utilize pseudopotentials. To account for both the precise band gap estimation and strong on-site Coulomb interactions in the localized d/f orbitals, electronic structure calculations were carried out using the modified Becke–Johnson (mBj) exchange potential in combination with the Hubbard U correction (mBj + U).37,38 The effective Hubbard U values are 6.0 eV for Pr-4f,39 6.5 eV for Eu-4f,40 3.5 eV for Co-3d,41 and 4.5 eV for Fe-3d orbitals.42 Although DFT + U, along with the TB-mBJ method, was used in the current study to consider the strong electron correlation in transition metal sites, it is acknowledged that the approach could still have a self-interaction error (SIE), which could negatively impact the accuracy of electronic property predictions. Other methods, including hybrid functionals, self-interaction-corrected (SIC) methods and other methods, have been demonstrated to provide better accuracy than standard DFT + U for systems containing transition metals.43,44 The wave function has been extended utilizing the sphere-shaped harmonic basis set as much as lmax = 10 across the not interconnected muffin-tin spheres. A plane-wave basis with a threshold value of RMT Kmax = 7 was implemented to extend the wave function throughout the interstitial region. The cut-off energy is chosen as −6.0 Ry to distinguish between the core and valence states. The incorporation of the Brillouin zone has been completed by employing a 10 × 10 × 10 k-mesh Monkhorst-Pack.45,46 In the present calculations, the energy and convergence criteria are considered whenever the charge difference is smaller than 0.001 e a.u.−3 per unit cell and the overall energy is steady within 0.0001 Ry. To avoid spurious oscillations and achieve higher quantitative accuracy, the optical characteristics were calculated using 15 × 15 × 15 k-points in the first Brillouin zone.47–50 To look into optical characteristics, the material's complex dielectric function is computed, as follows:51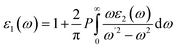
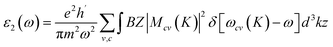
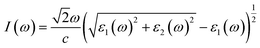
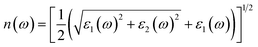
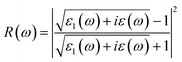
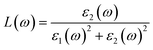
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m space group, as displayed in Fig. 1. The structures of these materials are optimized to obtain optimized bond lengths, lattice contents, and volumes. The energy vs. volume optimization curves are depicted in Fig. 7(a and b). The nature of the bonds among the material elements can be determined by computing the electronegativity difference between them. The electronegativity difference can be determined using Pauling values for each constituent element.54,55 For Pr2EuCoO6 and Pr2EuFeO6, the electronegativity difference for Pr–O, Eu–O, Co–O, and Fe–O bonds is 2.31, 2.24, 1.56, and 1.61, respectively, confirming that Pr–O and Eu–O have an ionic bonding nature while Co–O and Fe–O have a polar covalent bonding nature.56 The bonding nature can further be confirmed by studying the DOS plots (see Fig. 4), which show negligible hybridization between Pr–O and Eu–O, indicating ionic and strong hybridization near the Fermi level in the up-spin configurations between Co–O and Fe–O in Pr2EuCoO6 and Pr2EuFeO6, respectively, suggesting the covalent character of the materials. The tolerance factor provides insights into the crystal structure phase stability of the material. The concept of the tolerance factor for phase stability was first introduced by Megaw.57 Furthermore, Goldschmidt introduced the modified form of the tolerance factor, which is based on the ionic radii of the elements of the double perovskite materials. The modified form of the Goldschmidt tolerance factor (τ) formula for the materials under study is as follows:58
m space group, as displayed in Fig. 1. The structures of these materials are optimized to obtain optimized bond lengths, lattice contents, and volumes. The energy vs. volume optimization curves are depicted in Fig. 7(a and b). The nature of the bonds among the material elements can be determined by computing the electronegativity difference between them. The electronegativity difference can be determined using Pauling values for each constituent element.54,55 For Pr2EuCoO6 and Pr2EuFeO6, the electronegativity difference for Pr–O, Eu–O, Co–O, and Fe–O bonds is 2.31, 2.24, 1.56, and 1.61, respectively, confirming that Pr–O and Eu–O have an ionic bonding nature while Co–O and Fe–O have a polar covalent bonding nature.56 The bonding nature can further be confirmed by studying the DOS plots (see Fig. 4), which show negligible hybridization between Pr–O and Eu–O, indicating ionic and strong hybridization near the Fermi level in the up-spin configurations between Co–O and Fe–O in Pr2EuCoO6 and Pr2EuFeO6, respectively, suggesting the covalent character of the materials. The tolerance factor provides insights into the crystal structure phase stability of the material. The concept of the tolerance factor for phase stability was first introduced by Megaw.57 Furthermore, Goldschmidt introduced the modified form of the tolerance factor, which is based on the ionic radii of the elements of the double perovskite materials. The modified form of the Goldschmidt tolerance factor (τ) formula for the materials under study is as follows:58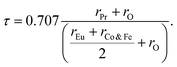
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m) space group is 0.9–1, which suggests that the computed tolerance of the materials lying in the stable crystallographic phase range confirms the stable crystallographic phase of the materials.60 The prediction of formation energy helps in understanding the thermodynamic stability of materials.61,62 The formation energy ΔHF of the Pr2EuMO6 (M = Co, Fe) materials can be computed by employing the following relation:
m) space group is 0.9–1, which suggests that the computed tolerance of the materials lying in the stable crystallographic phase range confirms the stable crystallographic phase of the materials.60 The prediction of formation energy helps in understanding the thermodynamic stability of materials.61,62 The formation energy ΔHF of the Pr2EuMO6 (M = Co, Fe) materials can be computed by employing the following relation:





![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 366.804
366.804![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 366.752
366.752![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 368.460
368.460![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 368.170
368.170


