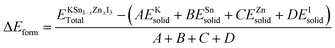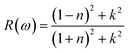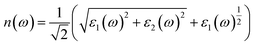Open Access Article
Open Access ArticleFirst-principles optimization of thermoelectric and optoelectronic performance of KSn1−xZnxI3 (x = 0, 0.25, 0.5, 0.75, 1) perovskites for sustainable energy harvesting
Usama Ahmeda,
Abu Sadat Md. Sayem Rahman *b,
Jesia Alam Jui
*b,
Jesia Alam Jui b,
Md. Mukter Hossain
b,
Md. Mukter Hossain a,
Md. Mohi Uddina and
Md. Ashraf Ali
a,
Md. Mohi Uddina and
Md. Ashraf Ali a
a
aAdvanced Computational Materials Research Laboratory, Department of Physics, Chittagong University of Engineering & Technology (CUET), Chattogram 4349, Bangladesh
bDepartment of Materials and Metallurgical Engineering (MME), Chittagong University of Engineering & Technology (CUET), Chittagong 4349, Bangladesh. E-mail: mdsayem@cuet.ac.bd; Tel: +880 01318503244
First published on 8th April 2026
Abstract
In the pursuit of non-toxic and high-efficiency perovskite solar cell materials, this study investigates the enhancement of thermoelectric and optoelectronic properties of Zn-doped KSn1−xZnxI3 (x = 0, 0.25, 0.5, 0.75, 1) perovskites. The study uses first-principles density functional theory (DFT) with the Vienna Ab initio Simulation Package (VASP). Structural analysis confirms a transition from orthorhombic (Pnma) to monoclinic (Pm) phases. All the compositions exhibit thermodynamic, mechanical, and dynamic stability. Electronic properties reveal a robust bandgap range of 1.47–1.96 eV (GGA-PBEsol) and 2.34–3.02 eV (HSE06), positioning these materials as promising candidates for the top cell in a tandem solar cell and UV-optoelectronics. An indirect-to-direct band structure transition occurs at 50% Zn doping, which primarily enhances the stiffness, Pugh's ratio (2.39–2.70), and Poisson's (0.316–0.335) ratio of the lattice for KSn1−xZnxI3. The elastic modulus (E), shear modulus (G), and bulk modulus (B) in KSn1−xZnxI3 also significantly increased upon addition of Zn in the compound. These behaviors indicate that although there is better lattice stiffness in the material, there is still very good ductility for making flexible devices. Near-perfect mechanical isotropy has been achieved in KZnI3 with a universal elastic anisotropy factor (AU) of only 0.15. This low level of anisotropic elastic behavior indicates that KZnI3 is unlikely to experience micro- fracture during or after manufacturing. Thermoelectric analysis shows that KSnI3 maintained a high Seebeck coefficient of 230 µV K−1 at low temperature, while KZnI3 showed a 225 µV K−1 Seebeck coefficient at elevated temperature. A high figure of merit (ZT) is achieved by both pristine compounds at high temperature, with values of 1.01 for KSnI3 and 1.27 for KZnI3. Furthermore, for optical properties, a high absorption coefficient of 7.32 × 105 cm−1 is observed by 25% Zn doping at UV-visible range. These findings make Zn-doped KSnI3 perovskite material suitable for efficient, non-toxic, low-cost optoelectronic and thermoelectric devices.
1. Introduction
Environmentalists have long been searching for new renewable energy sources. The growing demand for sustainable energy has driven researchers to develop new, eco-friendly novel materials. Scientists are looking for novel, eco-friendly materials that could convert sunlight to electricity by the photovoltaic effect.1 In parallel, researchers are also looking for novel materials that can generate electricity from waste heat by the thermoelectric effect.2 The ideal thermoelectric materials possess a large Seebeck coefficient, high electrical conductivity, and low thermal conductivity.3 Such a type of combination can be achieved in semiconducting materials that are engineered with alloying elements. The perovskites are newly emerged semiconducting materials with the formula ABX3, having good photovoltaic and thermoelectric properties.1,4 Desired properties can be easily tailored by changing the composition of the perovskite material by alloying with appropriate elements. The perovskite solar cells (PSCs) introduced in 2009 had an initial power conversion efficiency (PCE) of 3.8%.5 A significant advancement has been achieved in PCE value, which has increased to 26.7% over the past few years.6 Although this remarkable gain in efficiency has been achieved within a short period, a question remains whether it is the most suitable perovskite combination for environmental and public health due to the toxic nature of lead (Pb).7 To find solutions to this question, many researchers started to focus on much safer options than lead, such as Sn, Ge, Ti, Zn-based perovskite solar cells.8–10 These Pb-free perovskite components have generated a lot of interest because of their improved optoelectronic behavior than Pb-based perovskite material. Compared to other non-toxic perovskite materials, Sn-based perovskites stand out due to their lower toxicity, ideal band gap, good charge transport properties, and potential for environmentally friendly energy solutions.7,11 However, the easy oxidation process of Sn2+ to Sn4+ triggers p-type self-doping and generates Sn-vacancies. This phenomenon leads to Sn-based PSCs exhibiting inferior performance compared to Pb-based PSCs.12 However, alloying with Ge at the B-site makes the Sn-based perovskite much more stable with improved efficiency.13 At the same time, the development of non-toxic wide band gap perovskite is required for optoelectronic applications in the ultraviolet to the visible spectral range. The wide band gap perovskites have applications in the top cell of a tandem solar cell, UV-photodetectors, underwater photovoltaic devices, and light-emitting diodes (LEDs).14–16To increase the stability and fine-tune the compositions of perovskites, various elements are introduced as dopants at the B-site of perovskite structures. Among the various elements employed for B-site doping in perovskite structures, transition-metal ions are particularly common. Typical examples include manganese (Mn2+), zinc (Zn2+), lead (Pb2+), cadmium (Cd2+), aluminum (Al3+), tin (Sn2+), nickel (Ni2+), bismuth (Bi3+), and Cerium (Ce3+).17 Among these different elements, comprehensive studies on zinc (Zn2+) doping in perovskite compounds have established a pronounced effect on the photovoltaic and thermoelectric properties. The charge carrier collection efficiency and carrier lifetime in MAPbI3 perovskite are improved by Zn doping. Additionally, it also promotes the formation of larger grains with fewer pinholes. This significantly reduces non-radiative recombination and suppresses hysteresis.18,19 It is found in literature that the Sn-based perovskites are prone to intrinsic defects, undergo oxidation of Sn2+ to Sn4+, and experience local lattice strain. However, Zn doping effectively mitigates these intrinsic defects and lattice distortions. At the same time, it enhances the carrier diffusion length in Sn-based perovskite films.20 In general, higher oxidation resistance, low toxicity, and wide availability make zinc an excellent component for environmentally friendly solutions. Guo et al.21 proposed a method to fabricate a hybrid perovskite material, which is eco-friendly and operationally reliable. Lead content can be reduced up to 50% by co-doping CsPbBr3 with Zn and Mn without changing its optoelectronic properties. Kooijman et al.22 also reported that incorporating zinc halide into Pb-based perovskites significantly enhances the structural integrity, film morphology, and crystallinity. In addition, the zinc modified perovskites solar cell exhibited improved luminescence and achieved about a 33% increase in solar cell efficiency compared with their undoped materials. In a similar study, Ou et al.,23 where Zn2+ is added in Cs2AgBiBr6 instead of Ag, results in a high PCE value of 45.9% due to enhanced charge transport and more efficient interfacial charge extraction. Wei et al.24 investigate that zinc doping in halide perovskites weakens nonadiabatic coupling in the conduction band, which in turn slows down hot-carrier cooling. This behavior suggests a viable path for improving the lives of hot carriers in forthcoming solar technology. All things considered, these investigations highlight zinc doping as a successful method for developing stable, high-performing, and eco-friendly perovskite-based optoelectronic devices.
The thermoelectric potential of K and Sn-based perovskite compounds has become the focus of recent studies by many research groups. For example, the double perovskite compounds K2CuBiBr6 and K2AgBiBr6 were studied by Ali et al.25 These perovskite materials exhibit strong thermoelectric performance characterized by high Seebeck coefficients, good electrical conductivity, and ZT values of 0.84 for K2CuBiBr6 and 0.78 for K2AgBiBr6. Similarly, Abdullah and Gupta26 reported that the double perovskites A2GeSnF6 (A = K, Rb, Cs) are outstanding thermoelectric candidates with ZT values in the range of 0.94–0.97. The literature report indicates that Sn-based oxide perovskite materials, such as BaSnO3, exhibit excellent high temperature thermoelectric performance. In a study by Song et al.,27 BaSnO3 achieves a high p-type ZT of 1.52 and n-type ZT of 0.37 at 900 K due to its favorable electronic and thermal transport properties. The Zn-based perovskites, such as the inorganic cubic CsZnX3 (X = F, Cl, Br, I) studied by Aqili et al.28 also exhibit promising thermoelectric properties due to their favorable electronic and thermal transport characteristics. They observed that these compounds have ZT > 0.7 across a temperature range of 300–800 K, making them promising for thermoelectric applications. Similarly, Agouri et al.29 computationally investigated the thermoelectric properties of cubic (space group Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, no. 221) XZnI3 (X = Na, K, Rb, Cs) perovskites. They observed that these compounds are semiconductors with indirect band gaps in the range of 0.51–0.62 eV (TB-nmBJ) and ZT values reaching 0.55–0.66 at 1000 K. Specifically, the cubic KZnI3 phase exhibited a ZT value of approximately 0.55 at 1000 K and a band gap of 0.606 eV, respectively. Numerous beneficial effects of Zn doping on thermoelectric properties are also reported in the literature. For instance, Zn-doping in GeTe introduces resonance states and increases the Seebeck coefficient value by raising the heavy hole valence band on top of the light hole valence band. Zn-doping also reduces the lattice thermal conductivity in GeTe and hence improves the overall ZT value.30 The co-doping of Ge and Zn in SnTe is investigated by Shenoy et al.31 The Ge doping in SnTe reduces the thermal conductivity. The Zn doping in SnTe suppresses the bipolar effect at elevated temperature by widening the band gap, distorts the density of states (DOS) near the Fermi level by creating resonance states, which ultimately improves the power factor by co-doping strategy. When Zn is co-doped with Bi in SnTe, it drastically reduces the lattice thermal conductivity and results in a peak ZT value of ∼1.6 at elevated temperature.32 When Zn is co-doped with Ag in SnTe, it gives a high ZT value of ∼0.97 in the temperature range of 500 K to 840 K.33 Therefore, the doping of Zn, significantly improves the thermoelectric performance. The effect of doping on the thermoelectric properties can be accurately identified with the help of density functional theory (DFT) analysis. The DFT is a reliable scientific tool to accurately predict experimental results. For instance, DFT predicted the increment in band gap values due to Zn-doping from 0.079 eV to ∼0.2 eV in the Sn14Zn2Te16 compound. Experimentally, Zn-doped samples showed no “hump” in lattice thermal conductivity at high temperatures, unlike undoped SnTe. This confirms that the increased band gap due to Zn-doping prevented bipolar diffusion in SnTe.34 In the case of Pb–Zn co-doping in SnTe, the DFT confirmed the increment in the experimental Seebeck coefficient values. DFT predicted Pb–Zn co-doping introduces resonance states near the Fermi level and raises the heavy hole valence band approximately 0.1 eV above the light hole valence band at room temperature. This DFT prediction is confirmed by the experimental result,35 where because of the Pb–Zn co-doping in SnTe, significantly enhanced Seebeck coefficient value to 109 µV K−1 at 300 K and 229 µV K−1 at 840 K, respectively. In the case of Zn doping in Pb0.6Sn0.4Te,36 undoped Pb0.6Sn0.4Te has a zero band gap, which causes the Seebeck coefficient to drop after 570 K. DFT predicted that Zn opens a band gap (0.26 eV) in zero-gap Pb0.6Sn0.4Te. This is experimentally confirmed by the suppression of bipolar diffusion, evidenced by Seebeck values continuously increasing up to 840 K (221 µV K−1). It is also observed from Boltzmann transport calculations that Zn-doping in SnTe improves the ZT value. Experimentally, Sn0.96Zn0.08Te validated this by achieving a high ZT value of ∼0.28 at 300 K and a peak ZT of ∼1.49 at 840 K, respectively.34 Therefore, the DFT prediction has a good match with the experimental observation of thermoelectric properties.
m, no. 221) XZnI3 (X = Na, K, Rb, Cs) perovskites. They observed that these compounds are semiconductors with indirect band gaps in the range of 0.51–0.62 eV (TB-nmBJ) and ZT values reaching 0.55–0.66 at 1000 K. Specifically, the cubic KZnI3 phase exhibited a ZT value of approximately 0.55 at 1000 K and a band gap of 0.606 eV, respectively. Numerous beneficial effects of Zn doping on thermoelectric properties are also reported in the literature. For instance, Zn-doping in GeTe introduces resonance states and increases the Seebeck coefficient value by raising the heavy hole valence band on top of the light hole valence band. Zn-doping also reduces the lattice thermal conductivity in GeTe and hence improves the overall ZT value.30 The co-doping of Ge and Zn in SnTe is investigated by Shenoy et al.31 The Ge doping in SnTe reduces the thermal conductivity. The Zn doping in SnTe suppresses the bipolar effect at elevated temperature by widening the band gap, distorts the density of states (DOS) near the Fermi level by creating resonance states, which ultimately improves the power factor by co-doping strategy. When Zn is co-doped with Bi in SnTe, it drastically reduces the lattice thermal conductivity and results in a peak ZT value of ∼1.6 at elevated temperature.32 When Zn is co-doped with Ag in SnTe, it gives a high ZT value of ∼0.97 in the temperature range of 500 K to 840 K.33 Therefore, the doping of Zn, significantly improves the thermoelectric performance. The effect of doping on the thermoelectric properties can be accurately identified with the help of density functional theory (DFT) analysis. The DFT is a reliable scientific tool to accurately predict experimental results. For instance, DFT predicted the increment in band gap values due to Zn-doping from 0.079 eV to ∼0.2 eV in the Sn14Zn2Te16 compound. Experimentally, Zn-doped samples showed no “hump” in lattice thermal conductivity at high temperatures, unlike undoped SnTe. This confirms that the increased band gap due to Zn-doping prevented bipolar diffusion in SnTe.34 In the case of Pb–Zn co-doping in SnTe, the DFT confirmed the increment in the experimental Seebeck coefficient values. DFT predicted Pb–Zn co-doping introduces resonance states near the Fermi level and raises the heavy hole valence band approximately 0.1 eV above the light hole valence band at room temperature. This DFT prediction is confirmed by the experimental result,35 where because of the Pb–Zn co-doping in SnTe, significantly enhanced Seebeck coefficient value to 109 µV K−1 at 300 K and 229 µV K−1 at 840 K, respectively. In the case of Zn doping in Pb0.6Sn0.4Te,36 undoped Pb0.6Sn0.4Te has a zero band gap, which causes the Seebeck coefficient to drop after 570 K. DFT predicted that Zn opens a band gap (0.26 eV) in zero-gap Pb0.6Sn0.4Te. This is experimentally confirmed by the suppression of bipolar diffusion, evidenced by Seebeck values continuously increasing up to 840 K (221 µV K−1). It is also observed from Boltzmann transport calculations that Zn-doping in SnTe improves the ZT value. Experimentally, Sn0.96Zn0.08Te validated this by achieving a high ZT value of ∼0.28 at 300 K and a peak ZT of ∼1.49 at 840 K, respectively.34 Therefore, the DFT prediction has a good match with the experimental observation of thermoelectric properties.
Among different perovskite compositions, the non-toxic KSnI3 in its orthorhombic (Pnma) phase has been explored by different research groups for photovoltaic applications.37–40 The advantage of using potassium (K+) is that K+ is more cost-effective than its cesium (Cs+) and rubidium (Rb+) counterparts.8 The Zn is also non-toxic and earth abundant element. However, the use of Zn-doping at the B-site to stabilize the instability caused by Sn has remained largely unexplored for KSnI3 perovskite. Inspired by the success of Zn-doping at the B-site of different perovskites, we hypothesize that Zn-doping at the B-site of KSnI3 could synergistically enhance its multifunctional properties. Therefore, for the first time, this study employs density functional theory (DFT) to theoretically investigate the effect of Zn-doping on the structural, mechanical, electronic, optical, and thermoelectric properties of orthorhombic KSnI3 perovskite. Our goals are to: (i) identify the effect of Zn-doping on thermoelectric transport coefficients and ultimately to evaluate ZT, and (ii) elucidate doping-induced changes in the band structure and optical absorption for UV-visible light applications. This work investigates the effect of Zn-doping on the properties of KSnI3 perovskite for two distinct but equally important applications: (i) optoelectronics and (ii) thermoelectric. The optoelectronic properties are analyzed to determine the material's suitability to convert light to electricity. On the other hand, thermoelectric properties are studied independently to evaluate the potential of Zn-doped KSnI3 perovskites in waste heat recovery. The focus of this analysis is to produce a nontoxic Zn-doped KSnI3 perovskite material for advanced thermoelectric and optoelectronic applications.
2. Computational methodology
The density functional theory (DFT) calculations for the KSnxZn1−xI3 perovskite compositions are carried out by using the Vienna Ab initio Simulation Package (VASP).41 The Perdew–Burke–Ernzerhof for solids within the Generalized Gradient Approximation (GGA-PBEsol) functional is used to iteratively solve the Kohn–Sham equations in order to get the ground-state electronic structure.42 The electronic band structure, density of states (DOS), optical properties, and thermoelectric properties of the material were all studied in detail using the hybrid Heyd–Scuseria–Ernzerhof (HSE06) functional.43 The valence electron configurations included K (3s2 3p6 4s1), Sn (4d10 5s2 5p2), Zn (3d10 4s2), and I (4d10 5s2 5p5). For structural optimization, the energy and ionic convergence conditions were established at 1 × 10−6 eV and −2 × 10−2 eV Å−1, respectively. Monkhorst–Pack k-point meshes of 3 × 7 × 2 are utilized to sample the Brillouin zone, with a plane-wave kinetic energy cutoff of 450 eV. The Self-Consistent Field (SCF) iteration is performed until convergence is achieved by updating the charge density and computing an effective potential to solve for a new wave function.44 The calculation's post-processing analysis, including density of states (DOS), band structure, optical, and mechanical properties, is done using VASPKIT.45 Thermoelectric transport parameters are calculated using Boltzmann's theory with the Boltztrap2 tool,46 and the phonon dispersion is measured using the linear response method of the Cambridge Serial Total Energy Package (CASTEP) Code.47 The crystal structures of the KSnxZn1−xI3 perovskites are illustrated using the VESTA package.483. Results and discussion
3.1 Structural analysis
The perovskite structure (ABX3) in which the KSnI3 compound crystallizes, with K occupying the A-site, Sn at the B-site, and I at the X-site. The structure belongs to space group 62 and features an orthorhombic phase (Pnma).49 The phase changes from an orthorhombic to a monoclinic phase when Zn replaces Sn with space group 6 (Pm). Table 1 shows the atomic configuration of KSn1−xZnxI3 perovskite compositions. Fig. 1 shows the schematic of the crystal structure of KSn1−xZnxI3 perovskites. The lattice parameter and unit cell volume are optimized using the GGA-PBESol approximation. Table 2 provides a summary of the optimal lattice parameters for the orthorhombic and monoclinic phases.| Compounds | K | Sn | Zn | I |
|---|---|---|---|---|
| KSnI3 (orthorhombic) | (0.077304, 0.75, 0.672586) | (0.165021, 0.25, 0.941203) | — | (0.34052, 0.75, 0.011544), (0.017173, 0.25, 0.120918) and (0.30433, 0.25, 0.787371) |
| KSn0.75Zn0.25I3 (monoclinic) | (0.077304, 0.75, 0.672586) | (0.836324, 0.5, 0.952061), (0.331516, 0.5, 0.556637) and (0.663123, 0, 0.439629) | (0.1746, 0.0000, 0.5537) | (0.34052, 0.75, 0.011544), (0.017173, 0.25, 0.120918) and (0.30433, 0.25, 0.787371) |
| KSn0.5Zn0.5I3 (monoclinic) | (0.077304, 0.75, 0.672586) | (0.328786, 0.75, 0.549802) and (0.672324, 0.25, 0.452198) | (0.220023, 0.25, 0.02946) and (0.779967, 0.75, 0.97054) | (0.34052, 0.75, 0.011544), (0.017173, 0.25, 0.120918) and (0.30433, 0.25, 0.787371) |
| KSn0.25Zn0.75I3 (monoclinic) | (0.077304, 0.75, 0.672586) | (0.661255, 0, 0.441198) | (0.164057, 0, 0.046774), (0.833688, 0.5, 0.94975) and (0.324833, 0.5, 0.568874) | (0.34052, 0.75, 0.011544), (0.017173, 0.25, 0.120918) and (0.30433, 0.25, 0.787371) |
| KZnI3 (orthorhombic) | (0.077304, 0.75, 0.672586) | — | (0.165021, 0.25, 0.941203) | (0.34052, 0.75, 0.011544), (0.017173, 0.25, 0.120918) and (0.30433, 0.25, 0.787371) |
 | ||
| Fig. 1 The schematic representation of the unit cell structure of (a) KSnI3, (b) KSn0.75Zn0.25I3, (c) KSn0.5Zn0.5I3, (d) KSn0.25Zn0.75I3, and (e) KZnI3 perovskite compounds. | ||
| Compounds | a (Å) | b(Å) | c(Å) | V (Å3) | ΔEform (eV per atom) | TF | Ref. |
|---|---|---|---|---|---|---|---|
| KSnI3 | 10.20 | 4.611 | 16.79 | 790.13 | −1.333 | 0.93 | This work |
| 10.45 | 4.71 | 17.00 | — | — | — | 49 | |
| KSn0.75Zn0.25I3 | 10.16 | 4.52 | 16.55 | 759.33 | −1.324 | 0.93 | This work |
| KSn0.5Zn0.5I3 | 10.04 | 4.50 | 16.25 | 734.95 | −1.328 | 0.92 | This work |
| KSn0.25Zn0.75I3 | 10.04 | 4.30 | 16.26 | 703.74 | −1.304 | 0.92 | This work |
| KZnI3 | 9.84 | 4.23 | 16.03 | 669.41 | −1.304 | 0.92 | This work |
The Goldschmidt's tolerance factor (TF) has been calculated to verify the stability of KSn1−xZnxI3 perovskite compounds. The following is the equation for TF:50,51
The radii of K, B-site ions (Sn/Zn), and I are denoted as RA, RB, and RX, respectively. These values are taken from the Shannon ionic radii.52 As atomic size increases, the value of the tolerance factor (TF) decreases. The probability of a stable perovskite structure is indicated by the TF value, which ranges from 0.8 to 1.53 Table 2 contains the calculated TF values for KSn1−xZnxI3 perovskites.
By calculating its formation energy (ΔEform), the chemical endurance of the semiconductor KSn1−xZnxI3 perovskites has been further evaluated. The formula, as presented in ref. 54, is summarized here.
The number of K, Sn, Zn, and I atoms in the KSn1−xZnxI3 unit cell is represented by the variables A, B, C, and D in the provided equation. The total energy of the KSn1−xZnxI3 perovskite compound is denoted by the expressions 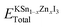 and the corresponding solid-state energies of K, Sn, Zn, and I atoms are denoted as; EKsolid, ESnsolid, EZnsolid, and EIsolid. The unit of the formation energy is eV per atom. The negative sign of the formation energy confirmed their stability. The computed formation energy values are shown in Table 2.
and the corresponding solid-state energies of K, Sn, Zn, and I atoms are denoted as; EKsolid, ESnsolid, EZnsolid, and EIsolid. The unit of the formation energy is eV per atom. The negative sign of the formation energy confirmed their stability. The computed formation energy values are shown in Table 2.
Fig. 2 illustrates the change of Goldschmidt tolerance factor (τ) and formation energy (ΔEform) with Zn content (x) in KSn1−xZnxI3 perovskites. For undoped KSnI3 and 50% Zn doping (x = 0.5), formation energy causes the thermodynamic stability to peak, providing stability to the lattice at these doping concentrations. This is because the lattice is properly accommodated when smaller Zn2+ ions partially replace larger Sn2+ ions, reducing lattice strain and distortion. According to the ideal perovskite stability criterion, the tolerance factor shows a minimum at x = 0.5. Beyond this, the little departure from perfect ionic size matching causes τ to increase as Zn content keeps rising, resulting in a subtle asymmetry in the structure. These trends validate that intermediate Zn doping levels, in particular 50%, present maximum structural stability and maximum geometric compatibility in the lattice.
 | ||
| Fig. 2 Effect of Zn doping on formation energy (ΔEform) and tolerance factor (τ) in KSn1−xZnxI3 perovskites. | ||
 | ||
| Fig. 3 Phonon dispersion curves of (a) KSn0.75Zn0.25I3, (b) KSn0.5Zn0.5I3, (c) KSn0.25Zn0.75I3, and (d) KZnI3 perovskite compounds. | ||
The frequency distribution of phonons extends from 0 to ∼6.5 THz in all cases. A denser phonon spectrum is observable in KZn0.5Sn0.5I3 (Fig. 3b) in the mid-frequency window (2–4 THz), implying stronger mode mixing and lattice complexities at the intermediate composition. Zn causes small blue shifts in some optical modes, mainly because Zn has a lower atomic mass than Sn, leading to the stiffening of Zn–I bonds. Acoustic modes show smooth and stable dispersion near the G point with an upward curvature, thus indicating that structural instabilities are absent. The Zn substitution creates many more such optical modes; consequently, coupling and splitting are more dominant in these crystals, especially for x = 0.5, suggesting enhanced phonon scatterings. At high-symmetry points Q and Z, phonon modes show flattening, which may affect the group velocities of vibrational modes and thus impinge upon thermal transport properties. The increased phonon mode density and dispersion complexity in KZn0.5Sn0.5I3 lead to a reduction of lattice thermal conductivity through enhanced phonon–phonon interactions and scatterings. Such behavior would be beneficial to thermoelectric applications, for which low thermal conductivity is a virtue. The systematic shift and redistribution of vibrational modes throughout the series further suggest a tunability of vibrational entropy, thus potentially affecting phase stability and defect formation energies in Zn-doped perovskites.
3.2 Electronic properties
The electronic properties of materials play a crucial role in governing carrier transport mechanisms and in distinguishing between metals, semiconductors, and insulators. In this study, the electronic properties have been evaluated using the GGA-PBEsol42 and HSE06 (ref. 43) functionals. A Gaussian smearing technique with a width of 0.05 eV was applied during the calculations.| Compound | XC-functionals | Band gap | References |
|---|---|---|---|
| KSnI3 | GGA-PBEsol | 1.8145 (Indirect) | This study |
| GGA-PBEsol | 1.84 (Indirect) | 38, 49, 55, and 56 | |
| HSE06 | 2.5218 (Indirect) | This study | |
| KSn0.75Zn0.25I3 | GGA-PBEsol | 1.6850 (Indirect) | This study |
| HSE06 | 2.4838 (Indirect) | This study | |
| KSn0.5Zn0.5I3 | GGA-PBEsol | 1.8510 (Direct) | This study |
| HSE06 | 2.6653 (Direct) | This study | |
| KSn0.25Zn0.75I3 | GGA-PBEsol | 1.4746 (Indirect) | This study |
| HSE06 | 2.3432 (Indirect) | This study | |
| KZnI3 | GGA-PBEsol | 1.9579 (Indirect) | This study |
| HSE06 | 3.0199 (Indirect) | This study |
 | ||
| Fig. 4 Electronic band structure of (a) KSnI3, (b) KSn0.75Zn0.25I3, (c) KSn0.5Zn0.5I3, (d) KSn0.25Zn0.75I3, and (e) KZnI3 perovskite compounds, calculated using the hybrid HSE06 functional. | ||
Pristine KSnI3 exhibits an indirect band gap of 2.52 eV. Although this value is too large for efficient single-junction photovoltaic absorption, it is well suited for transparent optoelectronic devices and window layers in photovoltaic devices. Such wide-band-gap materials are also relevant for tandem solar cell architectures, where selective absorption of high-energy photons is required. Beyond the band gap magnitude, the band-edge characteristics near the Fermi level provide qualitative insight into transport behavior. In particular, it has been seen that KSnI3 has relatively flatter band edges, which correlate to its larger Seebeck coefficient described in the thermoelectric section.
KSn0.75Zn0.25I3 has an indirect band gap of 2.48 eV at lower concentrations of Zn. This shows that we can achieve a modest amount of band gap tuning without significantly modifying the overall electronic structure of the material. Although it is still not the best choice for a primary photovoltaic absorber, it would be a promising candidate for a transparent optoelectronic device and the top cell in a tandem solar cell. The indirect-to-direct band gap transition is seen within KSn0.50Zn0.50I3, having a direct 2.67 eV band gap. The direct gap will increase the amount of radiative recombination and optical absorption, which is desirable for use in LEDs or high-speed photodetectors that are used in the visible to near ultraviolet spectral range. With further Zn incorporation, KSn0.25Zn0.75I3 shows an indirect band gap of 2.34 eV, while fully substituted KZnI3 exhibits the widest indirect band gap of 3.01 eV. These materials clearly fall within the wide-band-gap regime, supporting their potential use in UV optoelectronics, transparent electronics, and the top cell of a tandem solar cell. Moreover, the more dispersive band edges in KZnI3 are consistent with its enhanced carrier mobility and electrical conductivity. This is in agreement with the thermoelectric transport results discussed in the following section.
From the DOS plots, it is clear that Zn-doping in KZnxSn1−xI3 causes bandgap reduction, especially around x = 0.75, with Zn-4s and 4p states contribute significantly to the CBM. In pure KSnI3 (x = 0), the CBM mainly consists of Sn-5p orbitals, while the VBM is dominated by I-5p states, resulting in a relatively wider bandgap. When Zn replaces Sn, particularly at concentrations (x = 0.75), the Zn states hybridize significantly with I-5p states at the lower-energy CBM, thereby narrowing the bandgap. Additionally, the absence of a stereochemically active Sn2+ 5s2 lone pair in Zn2+, reduces local lattice distortions and promotes a better orbital alignment between VBM and CBM, producing a narrow bandgap. This behavior exemplifies how Zn substitution modifies the electronic structure and local symmetry, enabling cation engineering of tunable bandgap properties.
3.3 Optical properties
Light is a naturally abundant and renewable energy source that has high potential in optoelectronics. Currently, there is much research focused on the field of optoelectronics to develop new types of materials capable of converting light energy into electrical energy. This research focuses on parameters associated with the optical properties, such as light absorption, light reflection, refraction index, and optical conductivity of the KZnxSn1−xI3 perovskites. By choosing to alloy with a specific element, researchers can easily achieve wavelength-specific tunability of optical properties such as; refraction index (n), extinction coefficient (k), reflectivity (R), energy loss function (L), dielectric constant (ε), and absorption coefficient (α).| ε(ω) = ε1(ω) + iε2(ω) |
The real part of the dielectric function ε1(ω) describes the polarization response of materials to an applied electric field and the propagation of electromagnetic waves within the material. And, the imaginary component of the dielectric function describes how much energy is absorbed by that same material. In terms of perovskite solar cells, the imaginary part is particularly significant because it explains the mechanisms by which these types of solar cells convert electromagnetic wave energy into electrical energy.57
Fig. 6(a) illustrates how the value of dielectric function ε1(ω) varies for KZnxSn1−xI3 perovskites in the range of 0–10 eV. The static dielectric constant ε1(0), which is the value at 0 eV of the real part of the dielectric function.58 As shown in Fig. 6(a), KSnI3 (5.86) has the largest real value of the dielectric function at 0 eV as compared to other Zn-doped compounds and the KZnI3 (4.93) has the least value. Therefore, the addition of Zn in the KSn1−xZnxI3 material appears to cause a downward trend in the static dielectric constant and charge storage capacity. The decline of charge storage capacity is much less substantial than that of the original KSnI3 material, and so the doped materials are still appropriate for use in optoelectronic devices.
The imaginary component ε2(ω) of the dielectric function of a material is a measure of its capacity to absorb light. Electronic transitions between valence and conduction band states determine the value of ε2(ω). The peaks of the ε2(ω) curve correspond to these transitions and indicate the energy gaps associated with them.59 As illustrated in Fig. 6(b), an increase in ε2(ω) occurs at the point where the energy of the incoming photons exceeds the material's bandgap. For KZnxSn1−xI3, the value of ε2(ω) remains close to zero until the incident energy threshold is reached. Once this threshold has been crossed, ε2(ω) rises quickly and achieves maximum values of 9.63 KSnI3, 9.62 KZnI3, 7.97 KSn0.75Zn0.25I3, 6.61 KSn0.5Zn0.5I3, and 7.20 for KSn0.25Zn0.75I3 Perovskite compounds.
Fig. 6(c) shows the optical absorption coefficients of all the pristine and alloyed perovskite compositions: KSnI3, KSn0.25Zn0.75I3, KSn0.5Zn0.5I3, KSn0.75Zn0.25I3, and KZnI3. All compounds have strong UV and visible absorbance (200–600 nm) across the entire wavelength, which is an important characteristic for efficient optoelectronic operation.62 Except for the KSn0.75Zn0.25I3, the overall trend suggests that a slight increase in Zn content reduces optical absorption compared to pure KSnI3. The KSn0.75Zn0.25I3 exhibits relatively high absorption (8.15 × 105 cm−1), particularly at shorter wavelengths (UV-region), reflecting a strong light-matter interaction (Fig. 6(c)). This compound also exhibits a reduced band gap of 2.48 eV compared to 2.52 eV for pure KSnI3, suggesting promising potential for UV-photodetectors, blue-light LEDs, top cells in tandem solar cells, and photocatalytic systems that operate in the higher-energy spectrum. However, as Sn is gradually replaced by Zn (from KSn0.50Zn0.50I3 to KZnI3), there is a clear decline in absorption intensity. However, the pure KZnI3 shows the lowest absorption (3.15 × 105 cm−1) and highest bandgap (3.01 eV) among all, suggesting that only partial substitution of Zn (KSn0.25Zn0.75I3) plays an important role in boosting the light absorption capacity of these materials and broadens the range of usable light due to the band gap reduction.58
The best absorbing material for solar cells should have low reflectance in the visible and near infrared range (300–1100 nm or 1–4 eV) in order to ensure maximum photon absorption and conversion into electricity.62 Fig. 6(e) shows how the reflectance R(ω) of various perovskite compounds changes at different photon energy. The KSnI3 has the highest reflectance value of 37%, thus it reflects more sunlight than other types of perovskite compositions. In contrast, the KZnI3 exhibits a low reflectance value of 0.21, making it one of the least reflective perovskites, especially within the visible to near Infrared portion. Essentially, lowering the reflectance of a perovskite material by doping with Zn allows for a higher absorbance of incoming solar energy. Therefore, having lower reflectance in perovskite materials will save on energy while providing better quality products from a manufacturing standpoint.
At photon energies below 2 eV, all perovskites exhibit nearly zero optical conductivity. This occurs because photons with an energy lower than the bandgap energy do not provide enough energy to promote potentially conductive electrons from the valence band into the conduction band. After 2 eV, there is a sharp increase in conductivity because there is now enough energy in the incident photons to excite electrons into the conduction band. It is observed that above 6 eV, there is very strong optical conductivities, which gives KSn1−xZnxI3 perovskites huge potential for applications in UV-sensitive optoelectronics. In this case, KZnI3 has the highest conductivity due to its relatively high bandgap (3.01 eV), which suggests strong photoconductivity driven by the UV-range photon shining on the material.
As can be seen in Fig. 6(g), with the increase of Zn-doping in KSn1−xZnxI3 perovskite compounds, the static refractive index n(0) decreases and continues decreasing as the resulting amount of Zn-doping increases. The static refractive index of KSnI3, KSn0.75Zn0.25I3, KSn0.25Zn0.75I3, KSn0.5Zn0.5I3, and KZnI3 is 2.41, 2.33, 2.29, 2.29, and 2.20, respectively. Thus, Zn doping will cause perovskites to bend light less effectively, resulting in slightly reduced photon trapping in thin films. This can be considered an unfavorable characteristic for the design of layered tandem solar cells with KSn1−xZnxI3 perovskite compounds. However, this potential disadvantage can be minimized where the layered tandem solar cell layout uses a combination of anti-reflective coatings and light-trapping structures.
The extinction coefficient (k(ω)) of the KSn1−xZnxI3 compound is displayed in Fig. 6(h) over the energy range up to 10 eV. Initially, the extinction coefficient values remained almost zero up to 2 eV. Beyond this point, it increased gradually with increasing energy, corresponding to the onset of strong interband electronic transitions from the valence band to the conduction band. Pristine KSnI3 exhibits pronounced extinction features in the visible and near-ultraviolet regions, with a major peak around 4–5 eV, followed by additional high-energy peaks extending up to ∼8 eV. The fully substituted KZnI3 compound shows a comparatively weaker extinction coefficient in the lower-energy region but displays strong extinction in the higher-energy range (∼5–8 eV), reaching the largest peak intensity among all compositions. This behavior suggests enhanced optical transitions in the ultraviolet region for Zn-rich compositions. Fig. 6(h) also indicates that the gradual substitution of Sn with Zn in the perovskite structure leads to a decrease in extinction coefficients up to 5.44 eV photon energy, and after that, the trend becomes opposite. The impact of Zn doping on the evolution of k(ω) supports that alloying is an excellent means of modifying both the optical extinction coefficient and the spectral properties of KSn1−xZnxI3 perovskite compounds.
3.4 Thermo-mechanical properties
A complete comprehension of the internal structure of solid material is associated with elastic constants, which ensure the structural stability of the material. Elastic properties play a crucial role in material science, as they allow materials to spring back to their original shape after facing external forces.49 In this study, independent elastic constants are first evaluated using strain–stress calculations, which served as the basis for the investigation of mechanical properties.66 To find the mechanical behavior and structural stability of the KSn1−xZnxI3 perovskite series, key elastic parameters such as bulk modulus (B), Young's modulus (Y), shear modulus (G), and Poisson's ratio (ν) are calculated. The mechanical properties of Zn-doped KSn1−xZnxI3 perovskites with orthorhombic and monoclinic crystal structures are calculated with the GGA-PBEsol functional. The values of calculated elastic constants Cij for KSn1−xZnxI3 perovskites are provided in Table 4. Among different compositions of KSn1−xZnxI3; the KSnI3 and KZnI3 have an orthorhombic crystal structure, while the partially Zn-substituted compounds KSn0.75Zn0.25I3, KSn0.5Zn0.5I3, and KSn0.25Zn0.75I3 have a monoclinic structure. The conditions for mechanical stability depend on the type of crystal structure, where orthorhombic and monoclinic crystals each have their own specific elastic stability requirements. The mechanical stability criteria for the orthorhombic crystal structure are;67| C11 > 0, C44 > 0, C55 > 0, C66 > 0, (C11 × C12) > C122, |
| Compound | C11 | C12 | C13 | C15 | C22 | C23 | C25 | C33 | C35 | C44 | C46 | C55 | C66 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| KSnI3 (ref. 49) | 26.40 | 12.46 | 11.01 | — | 15.22 | 13.50 | — | 24.51 | — | 5.98 | — | 7.74 | 11.67 |
| KSnI3 | 18.52 | 16.02 | 15.39 | — | 30.12 | 13.10 | — | 33.31 | — | 8.29 | — | 5.93 | 14.02 |
| KSn0.75Zn0.25I3 | 16.87 | 9.73 | 10.98 | −0.18 | 21.49 | 10.30 | −1.06 | 21.27 | −0.42 | 3.66 | 0.26 | 3.88 | 11.90 |
| KSn0.50Zn0.50I3 | 24.38 | 13.92 | 16.10 | 3.16 | 25.45 | 14.88 | 2.55 | 35.15 | −0.33 | 10.30 | 3.08 | 5.37 | 8.241 |
| KSn0.25Zn0.75I3 | 30.72 | 15.56 | 16.08 | 4.18 | 37.69 | 16.51 | 3.43 | 29.45 | 2.34 | 10.78 | 1.57 | 6.74 | 10.89 |
| KZnI3 | 34.05 | 17.05 | 18.55 | — | 36.71 | 18.99 | — | 37.07 | — | 12.80 | — | 8.67 | 11.65 |
and
| (C11 × C22 × C33) + 2(C12 × C13 × C23) − (C11 × C232) − (C22 × C132) − (C33 × C122) > 0 |
The mechanical stability criteria for the monoclinic crystal system are;67
| C11 > 0, C22 > 0, C33 > 0, C44 > 0, C55 > 0, C66 > 0, |
| [C11 + C22 + C33 + 2(C12 + C13 + C23)] > 0, |
| [(C33 × C55) − C352 > 0, (C44 × C66) − C462 > 0, (C22 + C33 − 2C23)] > 0, |
| [C22(C33 × C55 − C352) + 2(C23 × C25 × C35) − (C232 × C55) − (C252 × C33)] > 0, |
and
| 2[C15 × C25(C33 × C12 − C13 × C23) + C15 × C35(C22 × C13 − C12 × C23) + C25 × C35(C11 ×C23 − C12 ×C13)] − [C15 × C15(C22 × C33 − C232) + C25 × C25(C11 × C33 − C132) + C35 × C35(C11 × C22 − C122)] + C55 × g > 0 |
It is observed that conditions mentioned in the above equations are satisfied for all KSn1−xZnxI3 perovskite compositions. Therefore, all the perovskite compositions are mechanically stable perovskite compounds.
By using the elastic constants provided in Table 4, the bulk and shear modulus are calculated with the Ruess (BR, GR) and Voigt approximation.68,69 The Hill approximation's bulk modulus (BH) and shear modulus (GH) are determined by taking the arithmetic mean of the values obtained from the Ruess and Voigt approximations.70 This approach provides a more accurate estimation of the overall mechanical properties by averaging the extremes of elastic responses. The values of the bulk and shear modulus of all the KSn1−xZnxI3 perovskite compositions are provided in Table 5. Table 5 and Fig. 7 indicate that the gradual substitution of Sn with Zn in the perovskite structure leads to a change in mechanical properties. While an initial reduction in bulk, shear, and Young's moduli is observed at low Zn content (x = 0.25), further increase is observed in these properties with increasing Zn concentration in the perovskite structure, which indicates enhanced mechanical stability and stiffness. The smaller ionic radius and stronger bonding characteristics of the Zn-ions compared to the Sn-ions contribute to a more compact and rigid crystal lattice.
| Compound | BV | GV | BR | GR | BH | GH | E | ν | B/G | AU |
|---|---|---|---|---|---|---|---|---|---|---|
| KSnI3 (ref. 49) | 15.57 | 7.02 | 14.54 | 5.21 | 15.05 | 6.11 | 16.16 | 0.321 | 2.46 | — |
| KSnI3 | 19.00 | 8.14 | 17.69 | 6.15 | 18.35 | 7.15 | 18.98 | 0.328 | 2.57 | 1.69 |
| KSn0.75Zn0.25I3 | 13.52 | 5.80 | 13.26 | 4.72 | 13.39 | 5.26 | 13.95 | 0.326 | 2.55 | 1.16 |
| KSn0.50Zn0.50I3 | 19.42 | 7.46 | 17.65 | 6.29 | 18.53 | 6.87 | 18.35 | 0.335 | 2.70 | 1.03 |
| KSn0.25Zn0.75I3 | 21.57 | 8.99 | 19.72 | 8.28 | 20.64 | 8.64 | 22.74 | 0.316 | 2.39 | 0.53 |
| KZnI3 | 24.11 | 10.17 | 24.03 | 9.89 | 24.07 | 10.03 | 26.43 | 0.317 | 2.40 | 0.15 |
The Pugh's ratio (B/G) measures whether a material will break easily (brittle) or deform before breaking, by comparing its stiffness to its resistance to shear.71 According to the B/G ratio criterion, materials with a B/G value greater than 1.75 are considered ductile, while those with a value less than 1.75 are classified as brittle. Similarly, another important elastic behavior, which also helps distinguish between brittle and ductile behavior, is Poisson's ratio (ν). According to Frantsevich's rule, materials with a Poisson's ratio greater than 0.26 (ν > 0.26) are considered ductile, while those with values below this threshold are classified as brittle.8 As evident from Table 5, all the investigated compounds of KSn1−xZnxI3 exhibit Poisson's ratio values ranging from 0.316 to 0.335 and B/G ratio ranging from 2.39 to 2.70 (Fig. 7(b), and Table 5). This indicates that all the studied compounds, KSnI3, KSn0.75Zn0.25I3, KSn0.5Zn0.5I3, KSn0.25Zn0.75I3, and KZnI3, are ductile and excellent for fabricating thin films on flexible substrates.
The universal anisotropy factor (AU) and its variation with different Zn-doping concentrations in KSn1−xZnxI3 perovskite compositions are presented in Table 5 and illustrated in Fig. 7(b). The completely elastic isotropic material has an AU value equal to zero. On the contrary, the AU value of an anisotropic material deviates from zero.58 All the compositions of Zn-doped KSn1−xZnxI3 perovskites exhibit an anisotropic nature, as the AU value differs from zero (Fig. 7(b), and Table 5). However, with increasing Zn concentration, the AU values progressively approach zero, indicating a gradual transition toward elastic isotropy induced by Zn incorporation. In particular, the perovskite compositions KSn0.25Zn0.75I3 and KZnI3 have AU values very close to zero, which indicates the nearly isotropic nature of these compositions. Therefore, the Zn-doped compositions are less prone to micro-crack formation after fabrication compared to pristine KSnI3 perovskite.
Table 6 summarizes the calculated density (ρ), longitudinal (νl), transverse (νt), and average sound velocities (νm), Debye temperature (ΘD), and melting temperature (Tm) of KSn1−xZnxI3 (x = 0, 0.25, 0.50, 0.75, and 1) perovskite compounds. These parameters play a crucial role in determining the lattice thermal conductivity and thermal stability, which are key factors for thermoelectric performance. The progressive substitution of Sn by Zn leads to a monotonic increase in density, indicating enhanced lattice rigidity and improved thermal stability with increasing Zn concentration. The formulas for the melting temperature of orthorhombic and monoclinic crystals are expressed as;64
| Tm = 412 + 8.2 [C11 + C121.25] |
| Compound | ρ (kg m−3) | νl (m s−1) | νt (m s−1) | νm (m s−1) | ΘD (K) | Tm (K) |
|---|---|---|---|---|---|---|
| KSnI3 | 4280 | 2482 | 1257 | 1409 | 123 | 835 |
| KSn0.75Zn0.25I3 | 4590 | 2210 | 1122 | 1257 | 108 | 845 |
| KSn0.5Zn0.5I3 | 4620 | 2447 | 1219 | 1367 | 123 | 915 |
| KSn0.25Zn0.75I3 | 4705 | 2614 | 1355 | 1516 | 138 | 950 |
| KZnI3 | 4814 | 2790 | 1444 | 1617 | 149 | 980 |
The increase in melting point with the addition of Zn from 835 K (KSnI3) to 980 K (KZnI3) suggests enhanced interatomic bonding from the incorporation of Zn. Thus, the Zn-doped compounds are suitable for high-temperature thermoelectric applications because of increased thermal stability. With Zn content, the average sound velocity and the Debye temperature vary non-monotonically. The KSn0.75Zn0.25I3 exhibits the lowest average sound velocity of 1257 m s−1 and the lowest Debye temperature of 108 K. This behavior suggests that the lattice thermal conductivity will be reduced due to the direct relation between lattice thermal conductivity, phonon group velocity, and Debye temperature. Low lattice thermal conductivities greatly improve the thermoelectric figure of merit, and this behavior makes them favorable to use in thermoelectric applications. As the concentration of Zn increases, both the value of νm and ΘD rise to their maximum values for KZnI3 composition. This is due to stronger bonding between the atoms and higher velocities of phonons within the structure, leading to improved overall thermal stability of the material. However, while compositions with Zn provide greater thermal stability, intermediate alloyed compositions should yield the best balance between reducing the thermal conductivity of the lattice and maintaining structural integrity.
Overall, the decrease in Debye temperature and sound velocities observed in the partially substituted zinc compositions provides evidence that the phonon transport is impeded due to alloy scattering arising from Sn/Zn disorder. Together, the softening mechanism of phonons and the sufficient range of melting temperatures indicate that KZnI3 is a potential candidate for future thermoelectric device applications. However, further investigation into the electrical transport properties will be necessary before establishing conclusively whether or not KZnI3 is suitable for use in thermoelectric devices.
Together, the computed melting temperatures, negative formation energies, and phonon stability show that KSn1−xZnxI3 compounds are suitable for high-temperature thermoelectric applications because they have good intrinsic structural robustness. Nevertheless, the current calculations do not specifically account for finite-temperature effects like thermal expansion or temperature-driven phase transitions because they are based on the optimized ground-state structures. At high temperatures, these effects could affect electronic transport, lattice thermal conductivity, and phonon scattering. Future research should focus on a more thorough evaluation of high-temperature structural evolution using ab initio molecular dynamics or quasi-harmonic calculations.
3.5 Thermoelectric properties
The graphs in Fig. 8 illustrate how the temperature affects each of the following variables: (a) Seebeck coefficient (S), (b) electrical conductivity (σ), (c) electronic conductivity (Ke), (d) lattice thermal conductivity (KL), (e) power factor (S2σ), and (f) thermoelectric figure of merit (ZT). The thermoelectric effect allows the direct conversion of a temperature difference into an electrical voltage and vice versa. The efficiency of a thermoelectric material is quantified by the dimensionless figure of merit, ZT,72 given by:Here, S is the Seebeck coefficient, σ is the electrical conductivity, K is the total thermal conductivity (the sum of electronic (Ke) and lattice (KL) contributions), and T is the absolute temperature. An optimal thermoelectric material requires a high-power factor (PF = S2σ) coupled with a low thermal conductivity (K).
In this study, key transport coefficients, including the Seebeck coefficient (S), electrical conductivity (σ), and electronic thermal conductivity (Ke) are calculated using the semi-classical Boltzmann transport theory within the constant relaxation time approximation (CRTA). Carrier concentration within the CRTA framework was utilized to compare intrinsic-temperature and composition-dependent trends across the KSn1−xZnxI3 series. This approximation assumes the electron relaxation time (τ) is energy-independent. This is a standard approach when precise τ calculations from first principles are computationally prohibitive. A constant τ value on the order of 10−14 s and a typical carrier concentration of 1 × 1020 cm−3 for semiconducting thermoelectric materials are employed. The assumption that is being made is consistent from current literature of DFT on similar materials. Calculations are completed using the BoltzTraP2 software,46 and lattice thermal conductivity (KL) is calculated using Slack's model.73
Here, the symbol γ denotes the Grüneisen parameter, a measure of the anharmonicity of a phonon. A reduction in phonon thermal conductivity at high γ values indicates the presence of significant anharmonic contributions to heat conduction. The A(γ) is a dimensionless function of the Gruneisen constant, Mav refers to the mean mass per molecule, θD is the Debye temperature, δ is the average distance between atoms, n is the number of atoms within one primitive unit cell, and T is the absolute temperature (in K). The impact of anharmonic phonon scattering and phonon group velocity on lattice heat transport is captured by the model. Thus, it is anticipated that compositions with lower average sound velocity and θD will have lower lattice thermal conductivity, which is in line with the trends found in this work. The dependence of the thermoelectric coefficient upon temperature is shown in Fig. 8. A complete evaluation of the thermoelectric power factor (S2σ), total thermal conductivity (K), and resulting figure of merit (ZT) will provide insight into the thermoelectric potential of KSn1−xZnxI3 (x = 0,0.25,0.50,0.75, and 1) perovskite compounds.
The Seebeck coefficient for KSnI3 is at its maximum value of 220–230 µV K1 at low temperature region (100 ≤ T ≤ 300 K). The reason that KSnI3 has such a high Seebeck coefficient is that it has a steep band curvature and the effective mass at the Fermi level is relatively large. As the temperature increases, KSnI3 shows a monotonic decrease in S value due to significant bipolar excitation typical for semiconductors. Conversely, KZnI3 shows a monotonic increase in S value as the temperature increases and surpasses all the other compositions at around 650 K with S value of 225 µV K1. The Seebeck coefficient of KZnI3 is maximized at the high temperature region with increased thermal excitation of the carriers, providing a more suitable band gap for thermoelectric operation at elevated temperatures. This makes KZnI3 a superior high-temperature thermoelectric material compared to compositions like KSnI3.
For a clearer assessment of performance, these results can be compared with the Seebeck coefficient (S) of well-known thermoelectric compounds found in the literature. For instance, polycrystalline tin selenide (SnSe) reaches the maximum Seebeck coefficient (S) value of 668 µV K−1 at 380 K.75 For the silicon-alloyed Bi0.4Sb1.6Te3 compounds, the Seebeck coefficient (S) value peaks at approximately 400 K, reaching values near 230 µV K−1.76 The n-type doped CsSnI3 and CsGeI3 perovskites exhibit Seebeck coefficient values of −300 and −280 µV K−1, respectively, at 700 K.77 In contrast, CsPbI3 perovskite exhibits a low value of the Seebeck coefficient of 150 µV K−1 at the same temperature.78 Among organic–inorganic mixed halide perovskites, the MAPbI3 single crystal displays a very high Seebeck coefficient of almost 1693 µV K−1 at 351 K.79 However, the low electrical conductivity of MAPbI3 makes the ZT value very small, approximately ∼0.1–0.9.80 Compared to these well-known thermoelectric compounds, the Seebeck coefficients of KSnI3 and KZnI3 fall within a competitive range along with the earth abundance of K compared to Se, Te, and Cs. This highlights their potential for further thermoelectric development.
The mixed alloys (x = 0.25, 0.5, 0.75) display noticeably lower Seebeck coefficients across all temperatures. This reduction occurs because alloying introduces structural changes, chemical disorder, and lattice scattering, which alter carrier concentration and reduce carrier mobility. These effects weaken the Seebeck coefficient compared to the well-ordered pure end compounds. Thus, KSnI3 dominates at low temperatures, while KZnI3 has the superior high-temperature Seebeck coefficient, making the two end-members the most promising thermoelectric compounds.
In contrast, the mixed alloys typically display lower electronic conductivity. For example, the electronic conductivity of KSn0.5Zn0.5I3 reduces to 0.39 × 1014 (m−1 k−1 s−1) at elevated temperatures due to alloy-induced disorder. This introduces potential variation and increases electron–phonon and point-defect scattering. The same trend is seen at 300 K as both KZnI3 and KSnI3 have conductivity greater than that of the respective alloy compositions, indicating that the Ke follows the same hierarchy as the σ in Fig. 8(b). This phenomenon demonstrates that structural ordering in the pure compounds results in significantly higher electronic conductivity than does disorder in the mixed alloys.
The mixed alloys exhibit reduced KL values, with KSn0.75Zn0.25I3 showing the lowest thermal conductivity among all compositions. This reduction originates from enhanced alloy scattering, where mass-fluctuation and strain-field disorder significantly shorten phonon mean free paths. Although lower KL is beneficial for increasing ZT, the alloys do not show improved thermoelectric performance because their PF values are drastically reduced. Thus, the thermal conductivity reduction alone is insufficient to compensate for the degradation in electronic transport.
For the intermediate alloys studied (x = 0.25, 0.5, 0.75), the measurements show a significantly reduced PF within the temperature range of 100 K to 1000 K. Disorder in the alloy structure leads to decreased mobility of carriers through alloy scattering and therefore leads to reduced S. Thus, the value of S2σ is significantly less than that of the pure compounds demonstrating the need for structural and electronic coherence across the pure compounds in order to achieve high performance transport. Finally, KZnI3 and KSnI3, as pure end compounds, retain superior pathways for carrier transport compared to disordered mixed alloys.
![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m, no. 221).83 In comparison, our work on the orthorhombic phase (Pnma, no. 62) reveals a substantially enhanced ZT of about 1.27 at 950 K. This exceptional value results from its high-power factor combined with favorable thermal transport characteristics, including relatively low lattice thermal conductivity with a strong Seebeck coefficient at elevated temperatures.
m, no. 221).83 In comparison, our work on the orthorhombic phase (Pnma, no. 62) reveals a substantially enhanced ZT of about 1.27 at 950 K. This exceptional value results from its high-power factor combined with favorable thermal transport characteristics, including relatively low lattice thermal conductivity with a strong Seebeck coefficient at elevated temperatures.To better evaluate the significance of this performance, a comparison with the well-established thermoelectric materials is essential. For example, tin selenide (SnSe) is one of the well-known thermoelectric compounds. The single crystal SnSe has one of the highest ZT values in the range of 2.2–2.6 at 913 K, and polycrystalline hole-doped SnSe provides ZT ∼ 3.1 at 783 K temperature.84 Another renowned thermoelectric material is Bi0.4Sb1.6Te3, used for low temperature thermoelectric applications, which has ZT ∼1.25 at about 100 °C temperature.84 However, selenium (Se) and tellurium (Te) are rare trace elements compared to K, Zn, and Sn present in KSn1−xZnxI3 perovskites. In case of inorganic perovskite materials, the peak value of ZT in CsSnI3 and CsGeI3 with the help of n-type doping can be reached to 0.95 and 1.05, respectively, at 700 K temperature.77 However, the ZT value of CsPbI3 is very low; ZT of 0.45 can be achieved with p-type doping at 700 K temperature.77 However, the K present in KSn1−xZnxI3 is much more earth-abundant compared to Cs, making these materials more promising from a sustainability perspective.
In the mixed alloys, ZT remains significantly lower throughout the temperature range, mainly due to structural shift. The pronounced disorder reduces carrier mobility and increases phonon scattering, thereby lowering PF and deteriorating ZT. Although increased phonon scattering can reduce KL, it is beneficial. The simultaneous suppression of σ and S2σ outweighs this advantage. As a result, alloying compromises the delicate balance required for high efficiency. Overall, the best thermoelectric performance is achieved in the pure compounds. The KSnI3 and KZnI3 are more promising for high-temperature thermoelectric applications. Whereas intermediate zinc concentrations reduce the overall thermoelectric efficiency. The tunability of thermoelectric behavior through Sn/Zn substitution highlights the potential of KSn1−xZnxI3 halide perovskites as next-generation, non-toxic, low-cost thermoelectric materials.
4. Conclusion
This study establishes KSn1−xZnxI3 (x = 0, 0.25, 0.5, 0.75, 1) perovskites as versatile, non-toxic, low-cost materials for thermoelectric energy harvesting and optoelectronic applications. The thermodynamic stability of all compositions is confirmed by their negative formation energies and phonon stability. It is successfully demonstrated that substituting Sn with Zn not only improves mechanical robustness (E, G, B) but also reduces elastic anisotropy (AU), which reduces micro-crack formation. These findings imply that there is potential to form thin films on a flexible substrate. The study of thermoelectric behavior indicates that the alloying of Zn to Sn results in the reduction of lattice thermal conductivity due to the scattering. This behavior results in a concurrent degradation of the electronic transport properties and a decrease in both the overall power factor and the overall figure of merit values. The thermoelectric performance is the greatest for two pure materials KSnI3 and KZnI3. The KSnI3 has the greatest performance at lower temperatures with a very high Seebeck coefficient of 220–230 µV K1, and the ZT value is 1.01. Meanwhile, KZnI3 archives optimized power factor ∼4.22 × 1011 W m−1 K−2 and ZT value of 1.27. The HSE06 functional is used to analyze the electronic characteristics Zn-doped KsnI3 materials. This analysis indicates that Zn can effectively engineer bandgaps of the perovskite compounds between 3.01 eV (KZnI3) and 2.34 eV (KSn0.25Zn0.75I3). As both compounds have wide bandgap, they can be used as suitable candidates for UV-visible optoelectronic devices or as the upper layer of tandem solar cells. Doping the perovskites with Zn allows for a systematic change to three important optical constants, such as optical absorption, reflectivity, and extinction coefficient, allowing for tailored light-matter interaction. In summary, Zn-doped KSnI3 perovskites represent a versatile, non-toxic platform. They are particularly promising for specialized optoelectronic applications in the UV-visible range and for high-temperature thermoelectric waste-heat recovery.Author contributions
U. Ahmed: conceptualization, data curation, methodology, formal analysis, investigation, visualization, validation, writing – original draft, writing – review & editing. A. S. M. S. Rahman: conceptualization, data curation, formal analysis, investigation, methodology, visualization, project administration, validation, supervision, writing – original draft, writing – review & editing. J. A. Jui: visualization, formal analysis, investigation, writing – review & editing. M. A. Ali: Software, validation, writing – review & editing. M. M. Hossain: validation, writing – review & editing. M. M. Uddin: validation, writing – review & editing.Conflicts of interest
There are no conflicts of interest to declare.Data availability
Data for this article including structural, electronic, mechanical, optical, and thermoelectric properties data, are available from the Zenodo repository (https://doi.org/10.5281/zenodo.18755200).Acknowledgements
This work was conducted at the ACMRL in the Department of Physics at CUET, which was established with the aid of a grant (grant number: 21-378 RG/PHYS/AS_G -FR3240319526) from UNESCO-TWAS and the Swedish International Development Co-operation Agency (SIDA). The views expressed herein do not necessarily represent those of UNESCO- TWAS, SIDA, or its Board of Governors.References
- A. S. A. Ahmed, F. S. M. Hashem, A. B. A. El-Adasy and T. A. Seaf El-Nasr, RSC Adv., 2025, 15, 23266–23301 RSC.
- R. Singh, S. Dogra, S. Dixit, N. I. Vatin, R. Bhardwaj, A. K. Sundramoorthy, H. C. S. Perera, S. P. Patole, R. K. Mishra and S. Arya, Hybrid Adv., 2024, 5, 100176 CrossRef.
- H. Ma, T. Lu, X. L. Shi, M. Li, S. Huo, P. Song, Z. G. Chen and M. Hong, Prog. Mater. Sci., 2026, 158, 101619 CrossRef CAS.
- C. Asker, C. Pipitone, F. Ursi, K. Chen, A. G. Ricciardulli, E. S. Suena Galindez, S. Luong, P. Samorì, M. Reece, A. Martorana, F. Giannici and O. Fenwick, J. Mater. Chem. A, 2025, 13, 26009–26018 RSC.
- A. Kojima, K. Teshima, Y. Shirai and T. Miyasaka, J. Am. Chem. Soc., 2009, 131, 6050–6051 CrossRef CAS PubMed.
- P. V. Kamat, ACS Energy Lett., 2025, 10, 896–897 CrossRef CAS.
- J. Li, H. L. Cao, W. Bin Jiao, Q. Wang, M. Wei, I. Cantone, J. Lü and A. Abate, Nat. Commun., 2020, 11, 310 CrossRef CAS PubMed.
- A. S. M. Sayem Rahman and K. M. Shorowordi, Mater. Sci. Semicond. Process., 2025, 186, 109114 CrossRef CAS.
- S. Mansouri, K. Zeghdar, L. Dehimi, F. Pezzimenti, R. Chakma, A. M. S. Alhuthali, A. Mohammad, A. A. Aly, N. Badi, M. H. H. Mahmoud, R. Haldhar and M. K. Hossain, J. Phys. Chem. Solids, 2026, 211, 113494 CrossRef CAS.
- K. Debnath, M. Debbarma, S. Das and S. Chattopadhyaya, Comput. Condens. Matter, 2026, 46, e01204 CrossRef.
- R. Kundara and S. Baghel, Mater. Sci. Eng., B, 2024, 307, 117536 CrossRef CAS.
- S. Wang, X. Zhang, W. Zhu, Z. Tang, J. Liu, H. Zhang, L. Ding and F. Hao, Appl. Surf. Sci., 2022, 602, 154393 CrossRef CAS.
- M. Liu, H. Pasanen, H. Ali-Löytty, A. Hiltunen, K. Lahtonen, S. Qudsia, J. H. Smått, M. Valden, N. V. Tkachenko and P. Vivo, Angew. Chem., Int. Ed., 2020, 59, 22117–22125 CrossRef CAS PubMed.
- C. Li, C. Chen, W. Gao, H. Dong, Y. Zhou, Z. Wu and C. Ran, ACS Nano, 2024, 18, 35130–35163 CrossRef CAS PubMed.
- C. Liu, H. Dong, Z. Zhang, W. Chai, L. Li, D. Chen, W. Zhu, H. Xi, J. Zhang, C. Zhang and Y. Hao, Sol. Energy, 2022, 233, 489–493 CrossRef CAS.
- J. Tong, Q. Jiang, F. Zhang, S. B. Kang, D. H. Kim and K. Zhu, ACS Energy Lett., 2021, 6, 232–248 CrossRef CAS PubMed.
- Y. Lu, F. Alam, J. Shamsi and M. Abdi-Jalebi, J. Phys. Chem. C, 2024, 128, 10084–10107 CrossRef CAS PubMed.
- W. Zhao, D. Yang, Z. Yang and S. (Frank) Liu, Mater. Today Energy, 2017, 5, 205–213 CrossRef.
- M. Badrooj, F. Jamali-Sheini and N. Torabi, Sol. Energy, 2022, 236, 63–74 CrossRef CAS.
- H. Jang, H. Y. Lim, C. B. Park, J. Seo, J. G. Son, T. Song, J. Lee, Y. S. Shin, J. Roe, S. K. Kwak, D. S. Kim and J. Y. Kim, J. Mater. Chem. A, 2023, 11, 10605–10611 RSC.
- Y. J. Guo, J. Su, L. Wang, Z. Lin, Y. Hao and J. Chang, J. Phys. Chem. Lett., 2021, 12, 3393–3400 CrossRef CAS PubMed.
- A. Kooijman, L. A. Muscarella and R. M. Williams, Appl. Sci., 2019, 9, 1678 CrossRef CAS.
- Y. Ou, Z. Lu, J. Lu, X. Zhong, P. Chen, L. Zhou and T. Chen, Opt. Mater., 2022, 129, 112452 CrossRef CAS.
- Q. Wei, J. Yin, O. M. Bakr, Z. Wang, C. Wang, O. F. Mohammed, M. Li and G. Xing, Angew. Chem., Int. Ed., 2021, 60, 10957–10963 CrossRef CAS PubMed.
- A. Ali, B. Anissa, D. Radouan, N. Al Bouzieh and N. Amrane, Mod. Phys. Lett. B, 2025, 39, 2550156 CrossRef CAS.
- D. Abdullah and D. C. Gupta, Sci. Rep., 2024, 14, 12644 CrossRef CAS PubMed.
- X. Song, G. Wang, L. Zhou, H. Yang, X. Li, H. Yang, Y. Shen, G. Xu, Y. Luo and N. Wang, ACS Appl. Energy Mater., 2023, 6, 9756–9763 CrossRef CAS.
- A. Aqili, A. Y. Al-Reyahi, S. M. Al Azar, S. Saad Essaoud, M. Elamin Ketfi, M. Maghrabi, N. Al Aqtash and A. Mufleh, Comput. Theor. Chem., 2024, 1238, 114721 CrossRef CAS.
- M. Agouri, H. Ouhenou, A. Waqdim, A. Zaghrane, E. Darkaoui, A. Abbassi, B. Manaut, S. Taj and M. Driouich, Epl, 2024, 146, 16005 CrossRef CAS.
- D. K. Bhat and U. S. Shenoy, New J. Chem., 2020, 44, 17664–17670 RSC.
- U. S. Shenoy, K. D. Goutham and D. K. Bhat, Mater. Adv., 2022, 3, 5941–5946 RSC.
- U. S. Shenoy and D. K. Bhat, J. Mater. Chem. C, 2020, 8, 2036–2042 RSC.
- U. S. Shenoy and D. K. Bhat, J. Alloys Compd., 2022, 892, 162221 CrossRef CAS.
- D. K. Bhat and U. S. Shenoy, Mater. Today Phys., 2019, 11, 100158 CrossRef.
- U. S. Shenoy and D. K. Bhat, J. Alloys Compd., 2021, 872, 159681 CrossRef CAS.
- D. K. Bhat and U. S. Shenoy, J. Alloys Compd., 2020, 834, 155181 CrossRef CAS.
- N. S. Alsaiari, I. Ahmed, S. Hanf, M. M. Rekha, M. Kundlas, M. Ouladsmane, K. Muhammad and J. Rehman, J. Inorg. Organomet. Polym. Mater., 2025, 35, 7458–7473 CrossRef CAS.
- M. Belarbi, Mater. Sci. Eng., B, 2025, 313, 117874 CrossRef CAS.
- P. Bhuvaneswari and P. Sriramalakshmi, J. Phys. Chem. Solids, 2026, 209, 113245 CrossRef CAS.
- A. Yousfi, O. Saidani, A. Benmakhlouf, T. Helaimia, A. S. Alsubaie, S. R. Al Ahmed and M. R. Islam, Sci. Rep., 2025, 15, 37022 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- L. A. Constantin, J. P. Perdew and J. M. Pitarke, Phys. Rev. B:Condens. Matter Mater. Phys., 2009, 79, 075126 CrossRef.
- J. Yang, L. Z. Tan and A. M. Rappe, Phys. Rev. B, 2018, 97, 085130 CrossRef CAS.
- H. Ehrenreich and M. H. Cohen, Phys. Rev., 1959, 115, 786–790 CrossRef.
- V. Wang, N. Xu, J. C. Liu, G. Tang and W. T. Geng, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- G. K. H. Madsen, J. Carrete and M. J. Verstraete, Comput. Phys. Commun., 2018, 231, 140–145 CrossRef CAS.
- S. J. Clark, M. D. Segall, C. J. Pickard, P. J. Hasnip, M. I. J. Probert, K. Refson and M. C. Payne, Z. Kristallogr. Cryst. Mater., 2005, 220, 567–570 CrossRef CAS.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2011, 44, 1272–1276 CrossRef CAS.
- G. Pindolia, S. M. Shinde and P. K. Jha, Mater. Chem. Phys., 2023, 297, 127426 CrossRef CAS.
- C. J. Bartel, C. Sutton, B. R. Goldsmith, R. Ouyang, C. B. Musgrave, L. M. Ghiringhelli and M. Scheffler, Sci. Adv., 2019, 5, eaav0693 CrossRef CAS PubMed.
- M. M. Rabbi, M. H. Mia, S. S. Saif, U. Ahmed, M. M. Hossain, M. M. Uddin and M. A. Ali, Comput. Condens. Matter, 2025, 44, e01093 CrossRef.
- R. D. Shannon, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef.
- A. E. Fedorovskiy, N. A. Drigo and M. K. Nazeeruddin, Small Methods, 2020, 4, 1900426 CrossRef CAS.
- M. W. Qureshi, X. Ma, G. Tang and R. Paudel, Sci. Rep., 2021, 11, 3260 CrossRef CAS PubMed.
- G. Pindolia and S. M. Shinde, Mater. Sci. Eng., B, 2023, 297, 116795 CrossRef CAS.
- I. Bin Abdul Ghani, M. Khalid, H. Yan, M. Kashif, B. Nawaz and J. Wang, J. Mater. Sci., 2024, 59, 14547–14572 CrossRef.
- M. Hilal, B. Rashid, S. H. Khan and A. Khan, Mater. Chem. Phys., 2016, 184, 41–48 CrossRef CAS.
- A. S. M. Sayem Rahman and K. M. Shorowordi, Heliyon, 2025, 11, e43425 CrossRef CAS.
- M. S. Alam, M. Saiduzzaman, A. Biswas, T. Ahmed, A. Sultana and K. M. Hossain, Sci. Rep., 2022, 12, 8663 CrossRef CAS PubMed.
- M. A. Rahman, R. Khatun, R. Ferdous, D. Chandra Roy, M. Z. Hasan, A. Irfan, M. A. Razzaque Sarker, M. M. Hossain, A. Hossain, S. C. Mouna and S. Sarker, J. Mater. Res. Technol., 2023, 26, 3840–3862 CrossRef CAS.
- R. Ahmad and N. Mehmood, J. Supercond. Novel Magn., 2018, 31, 1577–1586 CrossRef CAS.
- C. Yang, R. P. Xue, X. Li, X. Q. Zhang and Z. Y. Wu, Renewable Energy, 2020, 161, 836–845 CrossRef CAS.
- S. A. Dar, R. Sharma, V. Srivastava and U. K. Sakalle, RSC Adv., 2019, 9, 9522–9532 RSC.
- M. M. Hossain, M. A. Ali, M. M. Uddin, A. K. M. A. Islam and S. H. Naqib, J. Appl. Phys., 2021, 129, 175109 CrossRef CAS.
- M. M. Namisi, R. J. Musembi, W. M. Mulwa and B. O. Aduda, Comput. Condens. Matter, 2023, 34, e00772 CrossRef.
- M. H. Mia, U. Ahmed, S. K. Saha and M. A. Ali, J. Mater. Res. Technol., 2025, 36, 2468–2484 CrossRef CAS.
- F. Mouhat and F. X. Coudert, Phys. Rev. B:Condens. Matter Mater. Phys., 2014, 90, 224104 CrossRef.
- W. Voigt, Ann. Phys., 1889, 274, 573–587 CrossRef.
- A. Reuss, Z. angew. Math. Mech., 1929, 9, 49–58 CrossRef CAS.
- R. Hill, Proc. Phys. Soc., London, Sect. A, 1952, 65, 349–354 CrossRef.
- S. F. Pugh, London, Edinburgh Dublin Phil. Mag. J. Sci., 1954, 45, 823–843 CrossRef CAS.
- F. J. Disalvo, Science, 1999, 285, 703–706 CrossRef CAS PubMed.
- G. A. Slack, in Solid State Physics – Advances in Research and Applications, 1979, vol. 34, pp. 1–71 Search PubMed.
- D. Narducci, E. Selezneva, G. Cerofolini, S. Frabboni and G. Ottaviani, J. Solid State Chem., 2012, 193, 19–25 CrossRef CAS.
- F. Serrano-Sánchez, M. Gharsallah, N. M. Nemes, F. J. Mompean, J. L. Martínez and J. A. Alonso, Appl. Phys. Lett., 2015, 106, 083902 CrossRef.
- Y. Dou, X. Yan, Y. Du, J. Xu and D. Li, J. Mater. Sci.:Mater. Electron., 2020, 31, 4808–4814 CrossRef CAS.
- U. G. Jong, Y. S. Kim, C. H. Ri, Y. H. Kye and C. J. Yu, J. Phys. Chem. C, 2021, 125, 6013–6019 CrossRef CAS.
- U.-G. Jong, Y.-S. Kim, C.-H. Ri, Y.-H. Kye and C.-J. Yu, J. Phys. Chem. C, 2021, 125, 6013–6019 CrossRef CAS.
- T. Ye, X. Wang, X. Li, A. Q. Yan, S. Ramakrishna and J. Xu, J. Mater. Chem. C, 2017, 5, 1255–1260 RSC.
- M. Faizan, G. Zhao, T. Zhang, X. Wang, X. He and L. Zhang, Acta Phys.–Chim. Sin., 2024, 40, 2303004 CrossRef.
- J. P. Heremans, V. Jovovic, E. S. Toberer, A. Saramat, K. Kurosaki, A. Charoenphakdee, S. Yamanaka and G. J. Snyder, Science, 2008, 321, 554–557 CrossRef CAS PubMed.
- U. Ahmed, M. M. Hossian, M. M. Uddin, N. Jahan and M. A. Ali, Mater. Adv., 2026, 7, 403–424 RSC.
- M. Agouri, H. Ouhenou, A. Waqdim, A. Zaghrane, E. Darkaoui, A. Abbassi, B. Manaut, S. Taj and M. Driouich, A. Lett. J. Explor. Front. Phys., 2024, 146, 16005 CAS.
- C. Zhou, Y. K. Lee, Y. Yu, S. Byun, Z. Z. Luo, H. Lee, B. Ge, Y. L. Lee, X. Chen, J. Y. Lee, O. Cojocaru-Mirédin, H. Chang, J. Im, S. P. Cho, M. Wuttig, V. P. Dravid, M. G. Kanatzidis and I. Chung, Nat. Mater., 2021, 20, 1378–1384 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |